Lactobacillus plantarum TWK10 Supplementation Improves Exercise Performance and Increases Muscle Mass in Mice
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials, Animals, and Experiment Design
2.2. Forelimb Grip Strength Test
2.3. Swimming Exercise Performance Test
2.4. Determination of Fatigue-Associated Biochemical Variables
2.5. Clinical Biochemical Profiles
2.6. Histology of Tissues
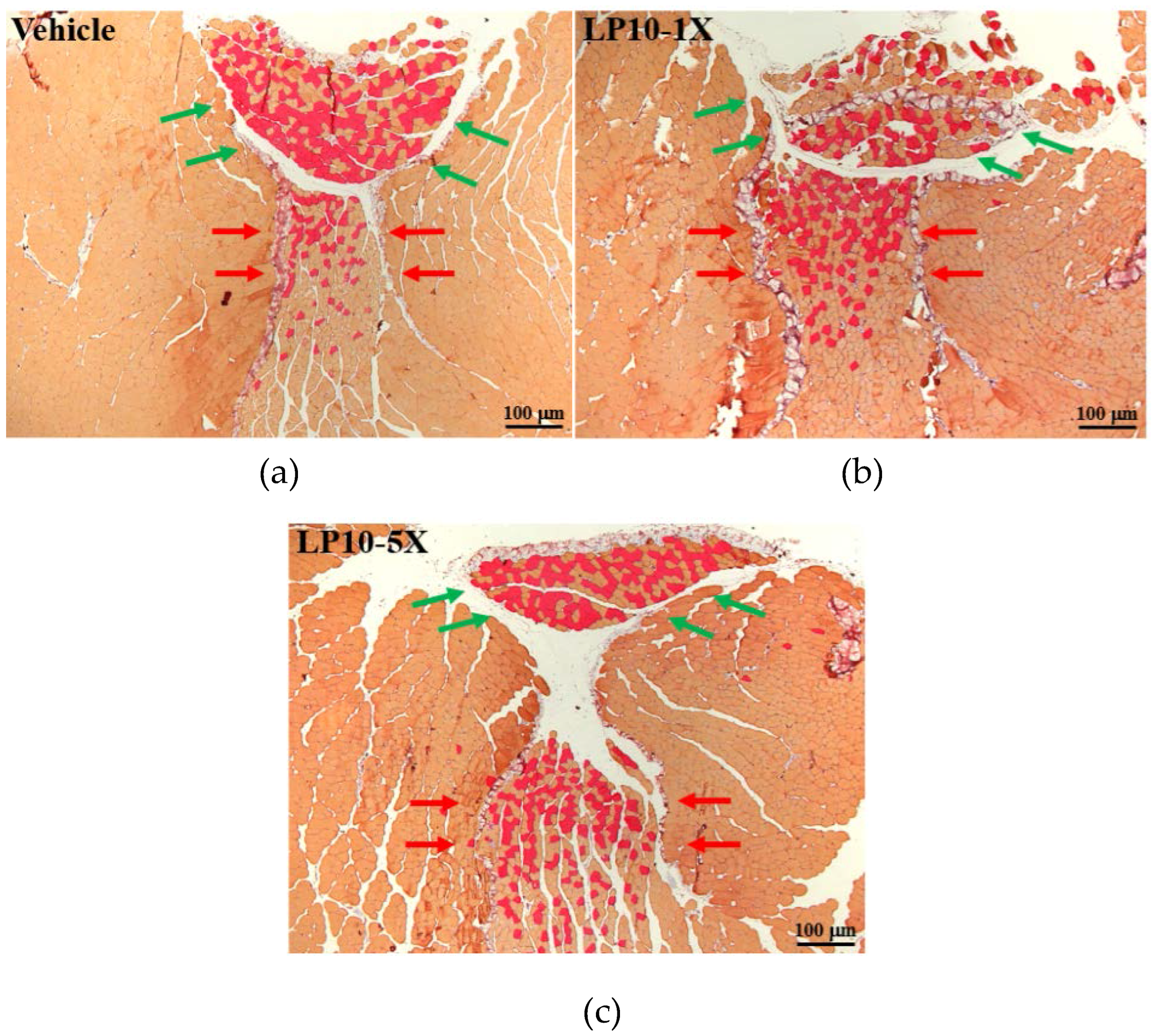
2.7. Immunohistochemical Staining of Gastrocnemius Muscles
2.8. Statistical Analysis
3. Results and Discussion
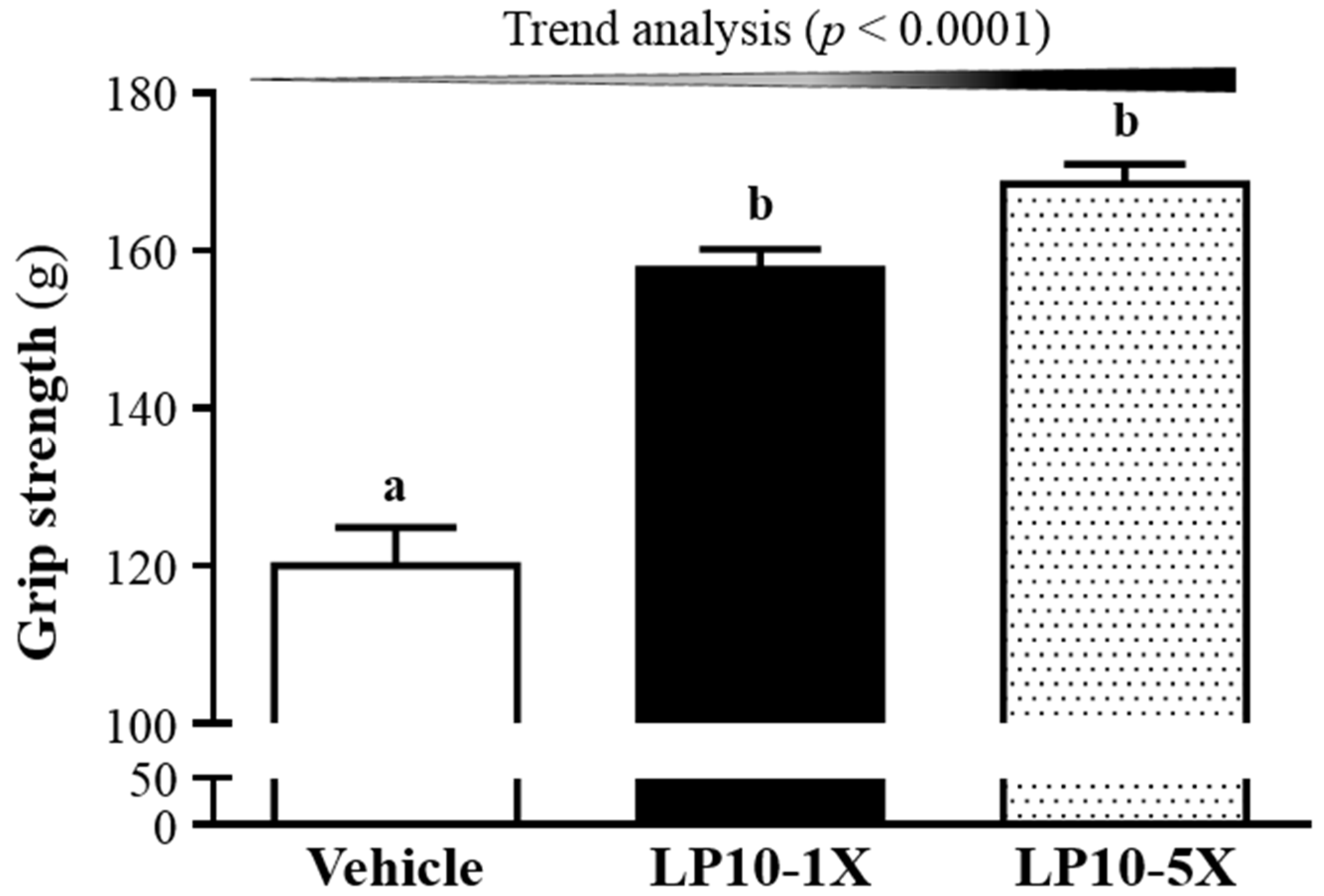
3.1. Effects of LP10 on Forelimb Grip Strength
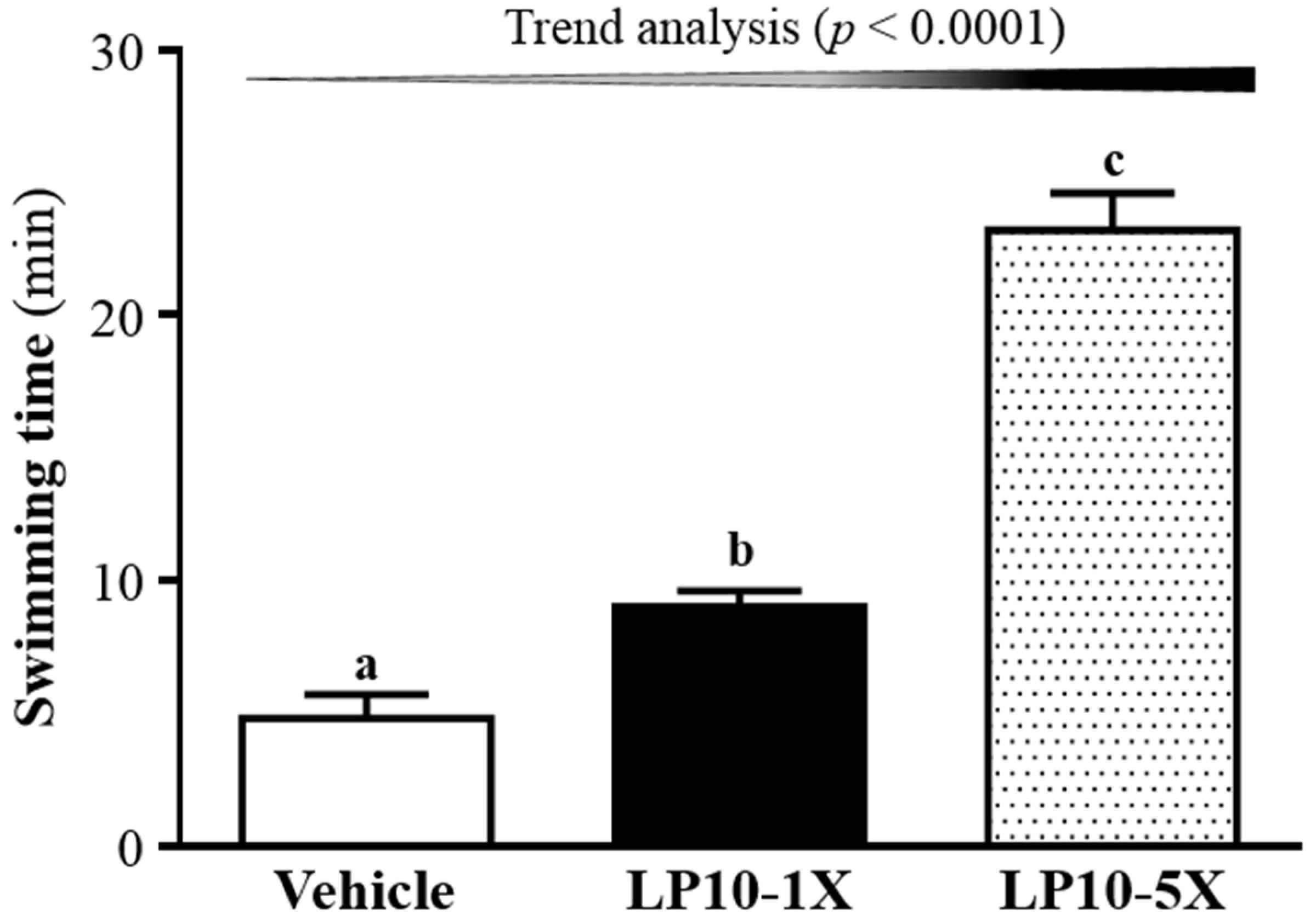
3.2. Effect of LP10 on Exercise Performance in a Weight-Loaded Swimming Test
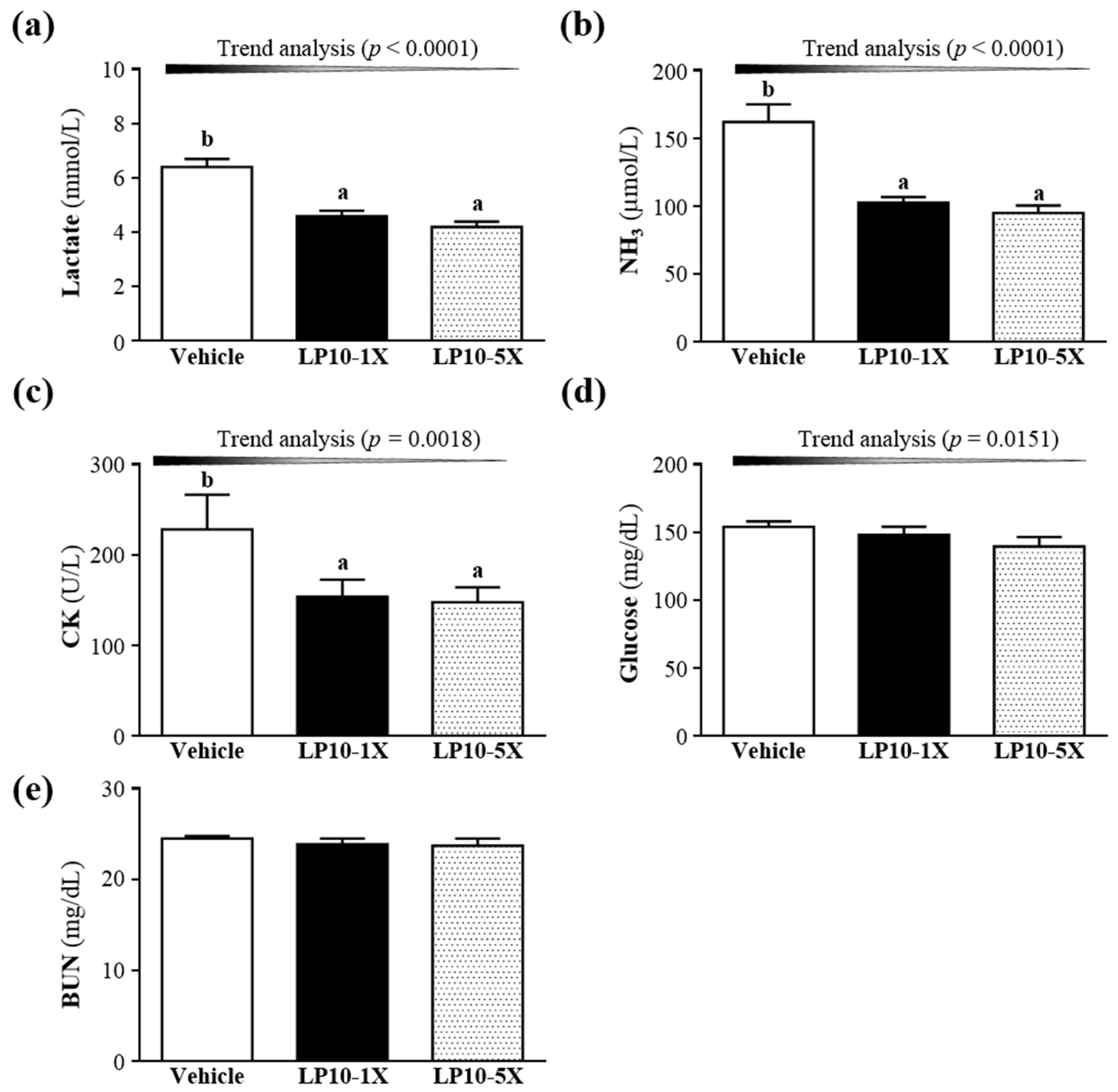
3.3. Effect of LP10 Supplementation on Serum Lactate, Ammonia, Glucose, CK and BUN Levels after Acute Exercise Challenge
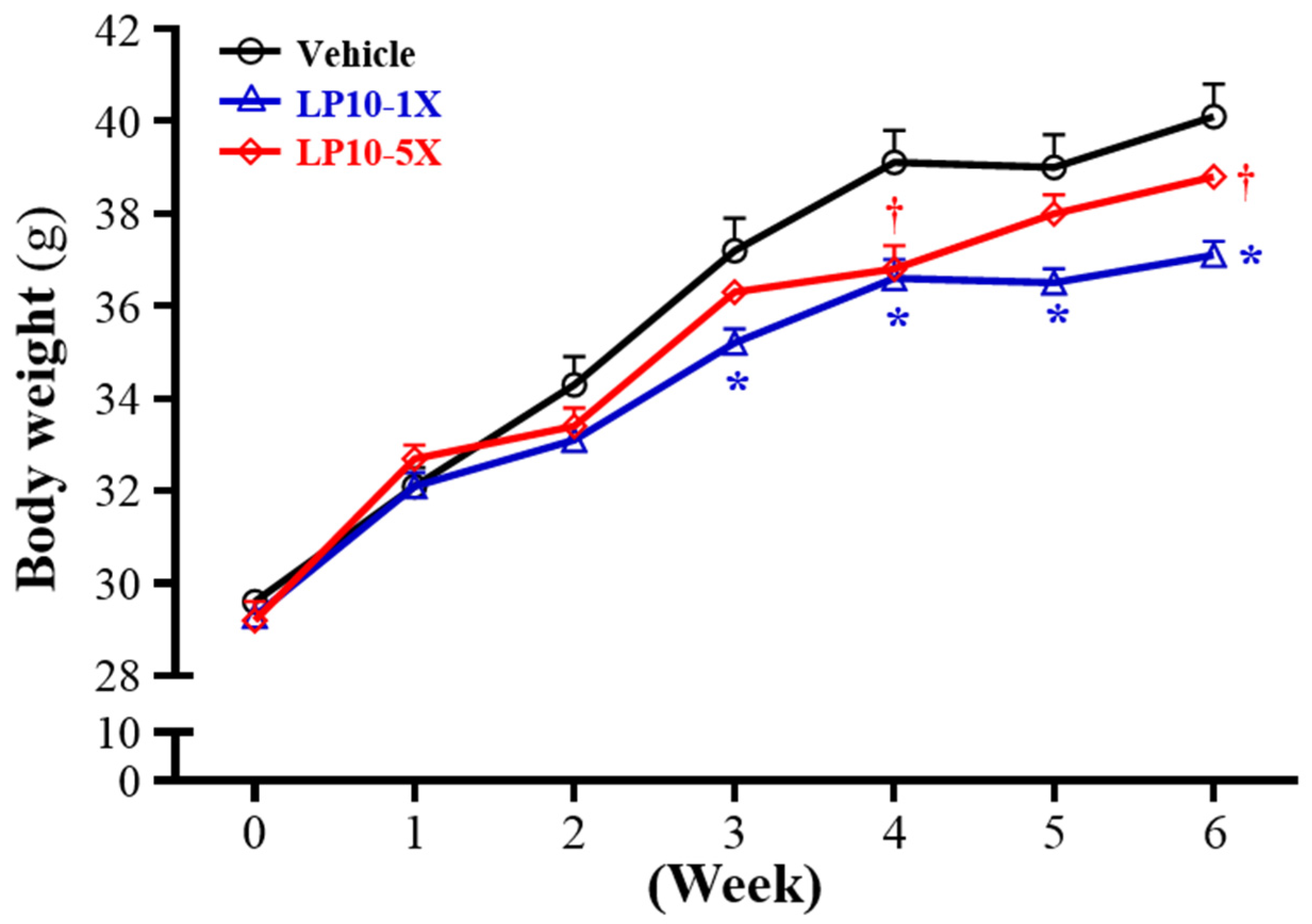
3.4. General Characteristics of Mice with LP10 Supplementation for Six Weeks
3.5. Effect of LP10 Supplementation on Biochemical Variables at the End of the Experiment
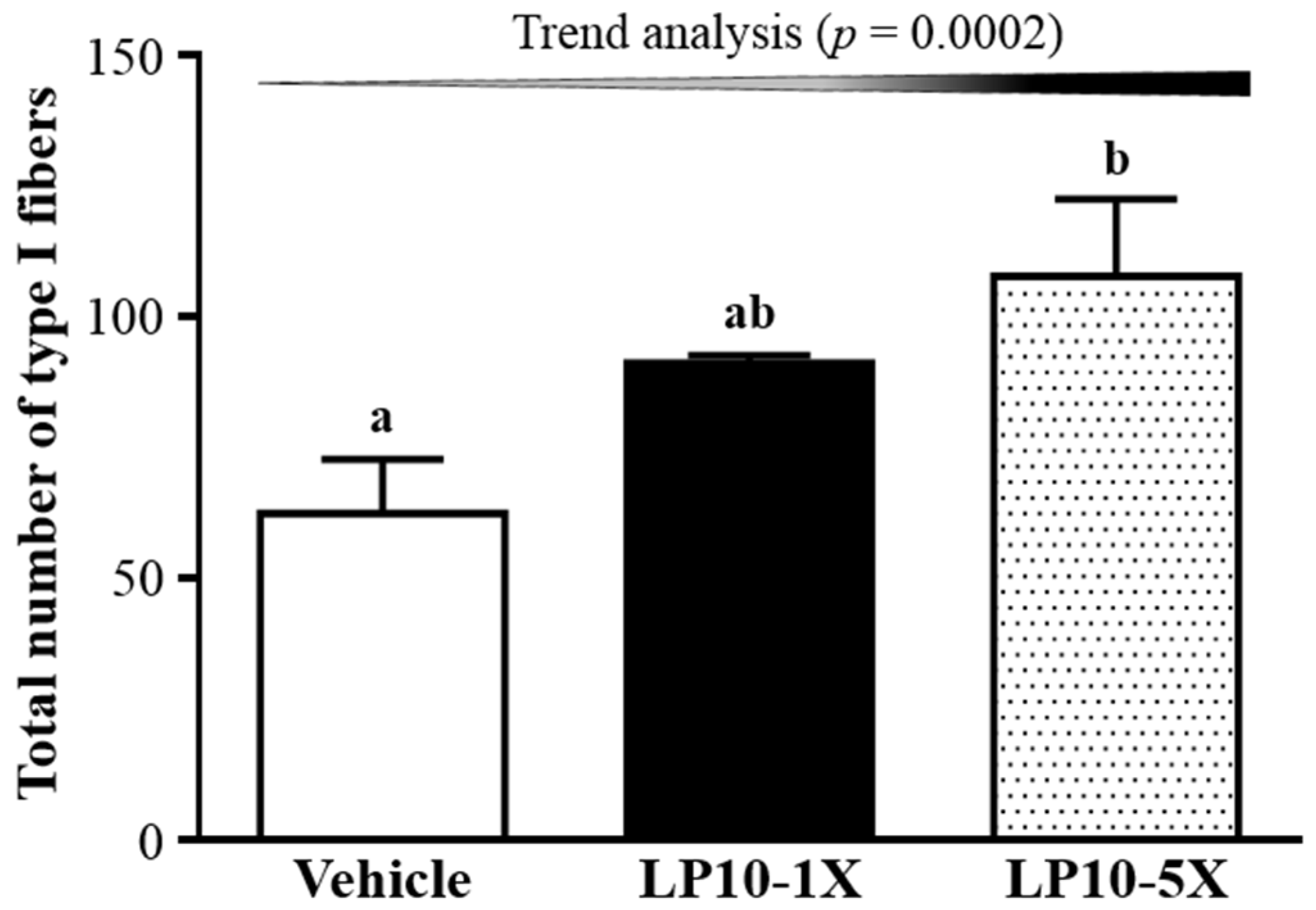
3.6. IHC of Gastrocnemius Muscles for Type I and Type II Muscle Fibers
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Andreasen, A.S.; Larsen, N.; Pedersen-Skovsgaard, T.; Berg, R.M.; Møller, K.; Svendsen, K.D.; Pedersen, B.K. Effects of Lactobacillus acidophilus NCFM on insulin sensitivity and the systemic inflammatory response in human subjects. Br. J. Nutr. 2010, 104, 1831–1838. [Google Scholar] [CrossRef] [PubMed]
- Iyer, C.; Kosters, A.; Sethi, G.; Kunnumakkara, A.B.; Aggarwal, B.B.; Versalovic, J. Probiotic Lactobacillus reuteri promotes TNF-induced apoptosis in human myeloid leukemiaderived cells by modulation of NF-kappaB and MAPK signalling. Cell Microbiol. 2008, 10, 1442–1452. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.Y.; Zeng, S.Y.; Leu, Y.L.; Tsai, T.Y. Antihypertensive Effect of a Combination of Uracil and Glycerol Derived from Lactobacillus plantarum Strain TWK10-Fermented Soy Milk. J. Agric. Food Chem. 2015, 63, 7333–7342. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.C.; Chen, Y.M.; Kan, N.W.; Ho, C.S.; Wei, L.; Chan, C.H.; Huang, C.C. Hypolipidemic Effects and Safety of Lactobacillus Reuteri 263 in a Hamster Model of Hyperlipidemia. Nutrients 2015, 7, 3767–3782. [Google Scholar] [CrossRef] [PubMed]
- Mack, D.R.; Ahrne, S.; Hyde, L.; Wei, S.; Hollingsworth, M.A. Extracellular MUC3 mucin secretion follows adherence of Lactobacillus strains to intestinal epithelial cells in vitro. Gut 2003, 52, 827–833. [Google Scholar] [CrossRef] [PubMed]
- Reis, J.A.; Paula, A.T.; Casarotti, S.N.; Penna, A.L.B. Lactic acid bacteria antimicrobial compounds: Characteristics and applications. Food Eng. Rev. 2012, 4, 124–140. [Google Scholar] [CrossRef]
- Amaretti, A.; Di Nunzio, M.; Pompei, A.; Raimondi, S.; Rossi, M.; Bordoni, A. Antioxidant properties of potentially probiotic bacteria: In vitro and in vivo activities. Appl. Microbiol. Biotechnol. 2013, 97, 809–817. [Google Scholar] [CrossRef] [PubMed]
- Gauchard, G.C.; Gangloff, P.; Vouriot, A.; Mallie, J.P.; Perrin, P.P. Effects of exercise-induced fatigue with and without hydration on static postural control in adult human subjects. Int. J. Neurosci. 2002, 112, 1191–1206. [Google Scholar] [CrossRef]
- Cheng, A.J.; Rice, C.L. Fatigue-induced reductions of torque and shortening velocity are muscle dependent. Med. Sci. Sports Exerc. 2010, 42, 1651–1659. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, J.H.; Blomqvist, G. Maximal oxygen uptake. N. Engl. J. Med. 1971, 284, 1018–1022. [Google Scholar] [CrossRef] [PubMed]
- Kreider, R.B.; Ferreira, M.; Wilson, M.; Grindstaff, P.; Plisk, S.; Reinardy, J.; Almada, A.L. Effects of creatine supplementation on body composition, strength, and sprint performance. Med. Sci. Sports Exerc. 1998, 30, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Conterno, L.; Fava, F.; Viola, R.; Tuohy, K.M. Obesity and the gut microbiota: Does up-regulating colonic fermentation protect against obesity and metabolic disease? Genes Nutr. 2011, 6, 241–260. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, J.K.; Holmes, E.; Kinross, J.; Burcelin, R.; Gibson, G.; Jia, W.; Pettersson, S. Host-gut microbiota metabolic interactions. Science 2012, 336, 1262–1267. [Google Scholar] [CrossRef] [PubMed]
- Duncan, S.H.; Louis, P.; Flint, H.J. Lactate-utilizing bacteria, isolated from human feces, that produce butyrate as a major fermentation product. Appl. Environ. Microbiol. 2004, 70, 5810–5817. [Google Scholar] [CrossRef] [PubMed]
- Bindels, L.B.; Delzenne, N.M. Muscle wasting: The gut microbiota as a new therapeutic target? Int. J. Biochem. Cell Biol. 2013, 45, 2186–2190. [Google Scholar] [CrossRef] [PubMed]
- Su, K.Y.; Yu, C.Y.; Chen, Y.W.; Huang, Y.T.; Chen, C.T.; Wu, H.F.; Chen, Y.L. Rutin, a flavonoid and principal component of Saussurea involucrata, attenuates physical fatigue in a forced swimming mouse model. Int. J. Med. Sci. 2014, 11, 528–537. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.E.; Huang, W.C.; Liao, C.C.; Chang, Y.K.; Kan, N.W.; Huang, C.C. Resveratrol protects against physical fatigue and improves exercise performance in mice. Molecules 2013, 18, 4689–4702. [Google Scholar] [CrossRef] [PubMed]
- Finkel, T. Signal transduction by reactive oxygen species. J. Cell Biol. 2011, 194, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Zhao, Y.; Zhang, L.; Zhang, X.; Huang, L.; Li, D.; Wang, Q. Antioxidant activity of Lactobacillus plantarum strains isolated from traditional Chinese fermented foods. Food chem. 2012, 135, 1914–1919. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, J.K.; Kumar, A.; Duary, R.K.; Mohanty, A.K.; Grover, S.; Batish, V.K. Functional and probiotic attributes of an indigenous isolate of Lactobacillus plantarum. PLoS ONE 2009, 4, e8099. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.I.; Huang, W.C.; Chen, W.C.; Kan, N.W.; Wei, L.; Chiu, Y.S.; Huang, C.C. Effect of whole-body vibration training on body composition, exercise performance and biochemical responses in middle-aged mice. Metabolism 2015, 64, 1146–1156. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.M.; Shih, T.W.; Chiu, C.P.; Pan, T.M.; Tsai, T.Y. Effects of lactic acid bacteria-fermented soy milk on melanogenesis in B16F0 melanocytes. J. Funct. Foods 2013, 5, 395–405. [Google Scholar] [CrossRef]
- Van Baarlen, P.; Troost, F.J.; van Hemert, S.; van der Meer, C.; de Vos, W.M.; de Groot, P.J.; Hooiveld, G.J.; Brummer, R.J.; Kleerebezem, M. Differential NF-kappaB pathways induction by Lactobacillus plantarum in the duodenum of healthy humans correlating with immune tolerance. Proc. Natl. Acad. Sci. USA 2009, 106, 2371–2376. [Google Scholar] [CrossRef] [PubMed]
- Million, M.; Angelakis, E.; Paul, M.; Armougom, F.; Leibovici, L.; Raoult, D. Comparative meta-analysis of the effect of Lactobacillus species on weight gain in humans and animals. Microb. Pathog. 2012, 53, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.C.; Chiu, W.C.; Chuang, H.L.; Tang, D.W.; Lee, Z.M.; Wei, L.; Huang, C.C. Effect of curcumin supplementation on physiological fatigue and physical performance in mice. Nutrients 2015, 7, 905–921. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.C.; Tseng, T.L.; Huang, W.C.; Chung, Y.H.; Chuang, H.L.; Wu, J.H. Whole-body vibration training effect on physical performance and obesity in mice. Int. J. Med. Sci. 2014, 11, 1218–1227. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.C.; Huang, W.C.; Chiu, C.C.; Chang, Y.K.; Huang, C.C. Whey protein improves exercise performance and biochemical profiles in trained mice. Med. Sci. Sports Exerc. 2014, 46, 1517–1524. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.M.; Tsai, Y.H.; Tsai, T.Y.; Chiu, Y.S.; Wei, L.; Chen, W.C.; Huang, C.C. Fucoidan Supplementation Improves Exercise Performance and Exhibits Anti-Fatigue Action in Mice. Nutrients 2014, 7, 239–252. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.C.; Chen, Y.M.; Kan, N.W.; Chao, H.L.; Ho, C.S.; Hsu, M.C. Cornu cervi pantotrichum supplementation improves exercise performance and protects against physical fatigue in mice. Molecules 2014, 19, 4669–4680. [Google Scholar] [CrossRef] [PubMed]
- Bindels, L.B.; Beck, R.; Schakman, O.; Martin, J.C.; De Backer, F.; Sohet, F.M.; Verrax, J. Restoring specific lactobacilli levels decreases inflammation and muscle atrophy markers in an acute leukemia mouse model. PLoS ONE 2012, 7, e37971. [Google Scholar]
- White, J.P.; Baynes, J.W.; Welle, S.L.; Kostek, M.C.; Matesic, L.E.; Sato, S.; Carson, J.A. The regulation of skeletal muscle protein turnover during the progression of cancer cachexia in the Apc(Min/+) mouse. PLoS ONE 2011, 6, e24650. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.H.; Pomare, E.W.; Branch, W.J.; Naylor, C.P.; Macfarlane, G.T. Short chain fatty acids in human large intestine, portal, hepatic and venous blood. Gut 1987, 28, 1221–1227. [Google Scholar] [CrossRef] [PubMed]
- Murase, M.; Kimura, Y.; Nagata, Y. Determination of portal short-chain fatty acids in rats fed various dietary fibers by capillary gas chromatography. J. Chromatogr. B Biomed. Appl. 1995, 664, 415–420. [Google Scholar] [CrossRef]
- Den Besten, G.; van Eunen, K.; Groen, A.K.; Venema, K.; Reijngoud, D.J.; Bakker, B.M. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J. Lipid Res. 2013, 54, 2325–2340. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.C.; Lin, C.I.; Chiu, C.C.; Lin, Y.T.; Huang, W.K.; Huang, H.Y.; Huang, C.C. Chicken essence improves exercise performance and ameliorates physical fatigue. Nutrients 2014, 6, 2681–2696. [Google Scholar] [CrossRef] [PubMed]
- Shing, C.M.; Peake, J.M.; Lim, C.L.; Briskey, D.; Walsh, N.P.; Fortes, M.B.; Vitetta, L. Effects of probiotics supplementation on gastrointestinal permeability, inflammation and exercise performance in the heat. Eur. J. Appl. Physiol. 2014, 114, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Maathuis, A.; Keller, D.; Farmer, S. Survival and metabolic activity of the GanedenBC30 strain of Bacillus coagulans in a dynamic in vitro model of the stomach and small intestine. Benef. Microbes 2009, 1, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Strojnik, V.; Komi, P.V. Fatigue after submaximal intensive stretch-shortening cycle exercise. Med. Sci. Sports Exerc. 2000, 32, 1314–1319. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo, M.; González-Izal, M.; Navarro-Amezqueta, I.; Calbet, J.A.; Ibañez, J.; Malanda, A.; Mallor, F.; Häkkinen, K.; Kraemer, W.J.; Gorostiaga, E.M. Effects of strength training on muscle fatigue mapping from surface EMG and blood metabolites. Med. Sci. Sports Exerc. 2011, 43, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Green, H.; Halestrap, A.; Mockett, C.; O’Toole, D.; Grant, S.; Ouyang, J. Increases in muscle MCT are associated with reductions in muscle lactate after a single exercise session in humans. Am. J. Physiol. Endocrinol. Metab. 2002, 282, E154–E160. [Google Scholar] [PubMed]
- Hobson, R.M.; Saunders, B.; Ball, G.; Harris, R.C.; Sale, C. Effects of β-alanine supplementation on exercise performance: A meta-analysis. Amino Acids 2012, 43, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Brooks, S.P.; Storey, K.B. A quantitative evaluation of the effect of enzyme complexes on the glycolytic rate in vivo: Mathematical modeling of the glycolytic complex. J. Theor. Biol. 1991, 149, 361–375. [Google Scholar] [CrossRef]
- Poutahidis, T.; Springer, A.; Levkovich, T.; Qi, P.; Varian, B.J.; Lakritz, J.R.; Erdman, S.E. Probiotic microbes sustain youthful serum testosterone levels and testicular size in aging mice. PLoS ONE 2014, 9, e84877. [Google Scholar]
- Chorell, E.; Karlsson Videhult, F.; Hernell, O.; Antti, H.; West, C.E. Impact of probiotic feeding during weaning on the serum lipid profile and plasma metabolome in infants. Br. J. Nutr. 2013, 110, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Finsterer, J. Biomarkers of peripheral muscle fatigue during exercise. BMC Musculoskelet. Disord. 2012, 13, 218. [Google Scholar] [CrossRef] [PubMed]
- Bercik, P.; Verdu, E.F.; Foster, J.A.; Macri, J.; Potter, M.; Huang, X.; Collins, S.M. Chronic gastrointestinal inflammation induces anxiety-like behavior and alters central nervous system biochemistry in mice. Gastroenterology 2010, 6, 2102–2112. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.C.; Hsu, M.C.; Huang, W.C.; Yang, H.R.; Hou, C.C. Triterpenoid-rich extract from Antrodia camphorata improves physical fatigue and exercise performance in mice. Evid. Based Complement Alternat. Med. 2012, 2012, 364741. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.J.; Chiu, C.C.; Li, Y.P.; Huang, W.C.; Huang, Y.T.; Huang, C.C.; Chuang, H.L. Effect of intestinal microbiota on exercise performance in mice. J. Strength Cond. Res. 2015, 29, 552–558. [Google Scholar] [CrossRef] [PubMed]
- Coyle, E.F.; Hagberg, J.M.; Hurley, B.F.; Martin, W.H.; Ehsani, A.A.; Holloszy, J.O. Carbohydrate feeding during prolonged strenuous exercise can delay fatigue. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 1983, 55, 230–235. [Google Scholar] [PubMed]
- Zou, D.; Chen, K.; Liu, P.; Chang, H.; Zhu, J.; Mi, M. Dihydromyricetin improves physical performance under simulated high altitude. Med. Sci. Sports Exerc. 2014, 46, 2077–2084. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Y.; Huang, W.C.; Liu, C.C.; Wang, M.F.; Ho, C.S.; Huang, W.P.; Huang, C.C. Pumpkin (Cucurbita moschata) fruit extract improves physical fatigue and exercise performance in mice. Molecules 2012, 17, 11864–11876. [Google Scholar] [CrossRef] [PubMed]
- Schwiertz, A.; Taras, D.; Schäfer, K.; Beijer, S.; Bos, N.A.; Donus, C.; Hardt, P.D. Microbiota and SCFA in lean and overweight healthy subjects. Obesity 2010, 18, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Bull-Otterson, L.; Feng, W.; Kirpich, I.; Wang, Y.; Qin, X.; Liu, Y.; Barve, S. Metagenomic analyses of alcohol induced pathogenic alterations in the intestinal microbiome and the effect of Lactobacillus rhamnosus GG treatment. PLoS ONE 2013, 8, e53028. [Google Scholar]
- Ge, H.; Li, X.; Weiszmann, J.; Wang, P.; Baribault, H.; Chen, J.L.; Tian, H.; Li, Y. Activation of G protein-coupled receptor 43 in adipocytes leads to inhibition of lipolysis and suppression of plasma free fatty acids. Endocrinology 2008, 149, 4519–4526. [Google Scholar] [CrossRef] [PubMed]
- Tisdale, M.J. Mechanisms of cancer cachexia. Physiol. Rev. 2009, 89, 381–410. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Vehicle | LP10-1X | LP10-5X | Trend Analysis |
|---|---|---|---|---|
| Initial BW (g) | 29.6 ± 0.2 | 29.3 ± 0.3 | 29.2±1.2 | 0.5370 |
| Final BW (g) | 40.1 ± 0.7 c | 37.1 ± 0.3 a | 38.8 ± 0.1 a,b | 0.6493 |
| Food intake (g/day) | 6.3 ± 0.1 a | 6.2 ± 0.0 a | 7.5 ± 0.1 b | <0.0001 (↑) |
| Water intake (mL/day) | 6.9 ± 0.1 a | 6.8 ± 0.2 a | 7.6 ± 0.0 b | <0.0001 (↑) |
| Weight (g) | ||||
| Liver (g) | 2.13 ± 0.05 | 2.10 ± 0.05 | 2.10 ± 0.03 | 0.9075 |
| Kidney (g) | 0.68 ± 0.02 | 0.67 ± 0.04 | 0.72 ± 0.04 | 0.1272 |
| EFP (g) | 0.85 ± 0.07 b | 0.55 ± 0.03 a | 0.42 ± 0.05 a | <0.0001 (↓) |
| Heart (g) | 0.20 ± 0.01 | 0.20 ± 0.01 | 0.20 ± 0.00 | 0.3908 |
| Lung (g) | 0.21 ± 0.01 | 0.22 ± 0.01 | 0.21 ± 0.00 | 0.9353 |
| Muscle (g) | 0.36 ± 0.01 | 0.37 ± 0.01 | 0.37 ± 0.01 | 0.4790 |
| BAT (g) | 0.13 ± 0.01 | 0.12 ± 0.00 | 0.13 ± 0.01 | 0.9473 |
| Relative weight (%) | ||||
| Liver | 5.29 ± 0.03 a | 5.65 ± 0.09 b | 5.43 ± 0.06 a | 0.1073 |
| Kidney | 1.70 ± 0.02 a | 1.81 ± 0.02 b | 1.86 ± 0.03 b | <0.0001 (↑) |
| EFP | 2.09 ± 0.16 c | 1.48 ± 0.09 b | 1.08 ± 0.15 a | <0.0001 (↓) |
| Heart | 0.49 ± 0.10 a | 0.54 ± 0.07 b | 0.53 ± 0.07 b | 0.0018 (↑) |
| Lung | 0.53 ± 0.03 a | 0.58 ± 0.03 b | 0.55 ± 0.01 a,b | 0.2009 |
| Muscle | 0.90 ± 0.02 a | 0.99 ± 0.01 b | 0.96 ± 0.02 b | 0.0326 (↑) |
| BAT | 0.31 ± 0.01 | 0.31 ± 0.01 | 0.33 ± 0.02 | 0.6881 |
| Variable | Vehicle | LP10-1X | LP-5X | Trend Analysis |
|---|---|---|---|---|
| CK (U/L) | 193 ± 36 | 169 ± 22 | 181 ± 25 | 0.8469 |
| TP (g/dL) | 4.8 ± 0.1 | 4.9 ± 0.1 | 4.9 ± 0.1 | 0.9571 |
| Albumin (g/dL) | 3.6 ± 0.0 b | 3.6 ± 0.0 b | 3.3 ± 0.1 a | 0.012(↓) |
| BUN (mg/dL) | 26.5 ± 0.5 b | 22.4 ± 0.6 a | 23.0 ± 0.9 a | 0.0017(↓) |
| Creatinine (mg/dL) | 0.27 ± 0.01 | 0.27 ± 0.01 | 0.29 ± 0.01 | 0.4627 |
| UA (mg/dL) | 0.91 ± 0.03 | 1.01 ± 0.10 | 1.03 ± 0.10 | 0.5858 |
| TC (mg/dL) | 143 ± 6 | 144 ± 6 | 130 ± 3 | 0.1804 |
| TG (mg/dL) | 205 ± 12 b | 159 ± 11 a | 151 ± 6 a | 0.0005(↓) |
| Glucose (mg/dL) | 166 ± 4 | 161 ± 5 | 157 ± 5 | 0.1336 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.-M.; Wei, L.; Chiu, Y.-S.; Hsu, Y.-J.; Tsai, T.-Y.; Wang, M.-F.; Huang, C.-C. Lactobacillus plantarum TWK10 Supplementation Improves Exercise Performance and Increases Muscle Mass in Mice. Nutrients 2016, 8, 205. https://doi.org/10.3390/nu8040205
Chen Y-M, Wei L, Chiu Y-S, Hsu Y-J, Tsai T-Y, Wang M-F, Huang C-C. Lactobacillus plantarum TWK10 Supplementation Improves Exercise Performance and Increases Muscle Mass in Mice. Nutrients. 2016; 8(4):205. https://doi.org/10.3390/nu8040205
Chicago/Turabian StyleChen, Yi-Ming, Li Wei, Yen-Shuo Chiu, Yi-Ju Hsu, Tsung-Yu Tsai, Ming-Fu Wang, and Chi-Chang Huang. 2016. "Lactobacillus plantarum TWK10 Supplementation Improves Exercise Performance and Increases Muscle Mass in Mice" Nutrients 8, no. 4: 205. https://doi.org/10.3390/nu8040205
APA StyleChen, Y.-M., Wei, L., Chiu, Y.-S., Hsu, Y.-J., Tsai, T.-Y., Wang, M.-F., & Huang, C.-C. (2016). Lactobacillus plantarum TWK10 Supplementation Improves Exercise Performance and Increases Muscle Mass in Mice. Nutrients, 8(4), 205. https://doi.org/10.3390/nu8040205





