Prevalence of Nutrition and Health-Related Claims on Pre-Packaged Foods: A Five-Country Study in Europe
Abstract
:1. Introduction
1.1. Background
1.2. Research Questions
- What proportion of pre-packaged foods and drinks available in-store in the five countries carry NHC?
- What proportion of these claims is symbolic?
- What types of NHC can be found on pre-packaged foods and drinks?
- To which nutrients or other food components do NHC refer?
- To which health-relationships do health claims refer?
- Which types of foods and drinks carry NHC?
2. Methods and Materials
2.1. Data Collection
2.2. Data Extraction
2.3. Analysis
- A nutrition claim was defined as “any claim which states, suggests or implies that a food has particular beneficial nutritional properties due to (a) the energy (calorific value) it (i) provides; (ii) provides at a reduced or increased rate; or (iii) does not provide; and/or (b) the nutrients or other substances it (i) contains; (ii) contains in reduced or increased proportions; or (iii) does not contain” [1].
- Nutrition claims were classified as either a nutrient content claim when it “describes the level of a nutrient contained in a food (or its energy value)” (e.g., “high in fibre” and “low fat”) or a nutrient comparative claim “when it compares the composition of the food in question with the composition of other foods” (e.g., “higher in fibre” and “reduced sugar”) [1].
- A health-related ingredient claim was defined as a claim communicating the presence of an ingredient(s) which is not a nutrient or other substance as defined in the EU Regulation [1] but which implies health benefits. In most cases, these claims related to the content of ingredients that are considered as a healthy (e.g., “Contains one of your five a day”) or at least a healthier alternative (e.g., “Sweetened only with brown sugar”).
- A health claim was defined as “any claim that states, suggests or implies that a relationship exists between a food category, a food or one of its constituents and health” [1].
- A general health claim (covered by Article 10(3) of the EU Regulation [1]) was defined as a claim referring to benefits for general health or well-being. A typical example of such a claim is “Good for your health” or “Healthier choice (within this product group)”.
- A nutrient and other function claim (as covered by Article 13 of the EU Regulation [1]) was defined as a health claim that describes or refers to one of the following: (a) the role of a nutrient or other substance in growth, development and the functions of the body; (b) psychological and behavioral functions; or (c) slimming or weight-control or a reduction in the sense of hunger or an increase in the sense of satiety or a reduction of the available energy from the diet. Typical examples of such a claim are “Calcium builds strong teeth” and “Fibre helps maintain a healthy digestive system”.
- A reduction of disease risk claim (covered by Article 14.1(a) of the EU Regulation [1]) was defined as a claim communicating that the consumption of a food category, a food or one of its constituents significantly reduces a risk factor in the development of a human disease. Typical examples of such a claim are “Plant sterols reduce blood cholesterol. High cholesterol is a risk factor in the development of coronary heart disease” and “Reduces the risk factor for development of dental caries”.
- A children’s development and health claim (covered by Article 14.1(b) of the EU Regulation [1]) was defined as a health claim where children’s development and/or health was specifically mentioned. Typical examples of such a claim are “Calcium is needed for normal growth and development of bones in children” and “For your baby’s safe and balanced diet”. Claims where children were not specifically mentioned, even if these claims were found on foods intended solely for use by children, were not considered children’s development and health claims.
3. Results
3.1. What Proportion of Pre-Packaged Foods and Drinks Available In-Store in the Five Countries Carry NHC?
3.2. What Proportion of These Claims is Symbolic?
3.3. What Types of NHC Can Be Found on Pre-Packaged Food and Drinks?
3.4. To Which Nutrients or Other Food Components Do NHC Refer?
3.5. To Which Health-Relationships Do Health Claims Refer?
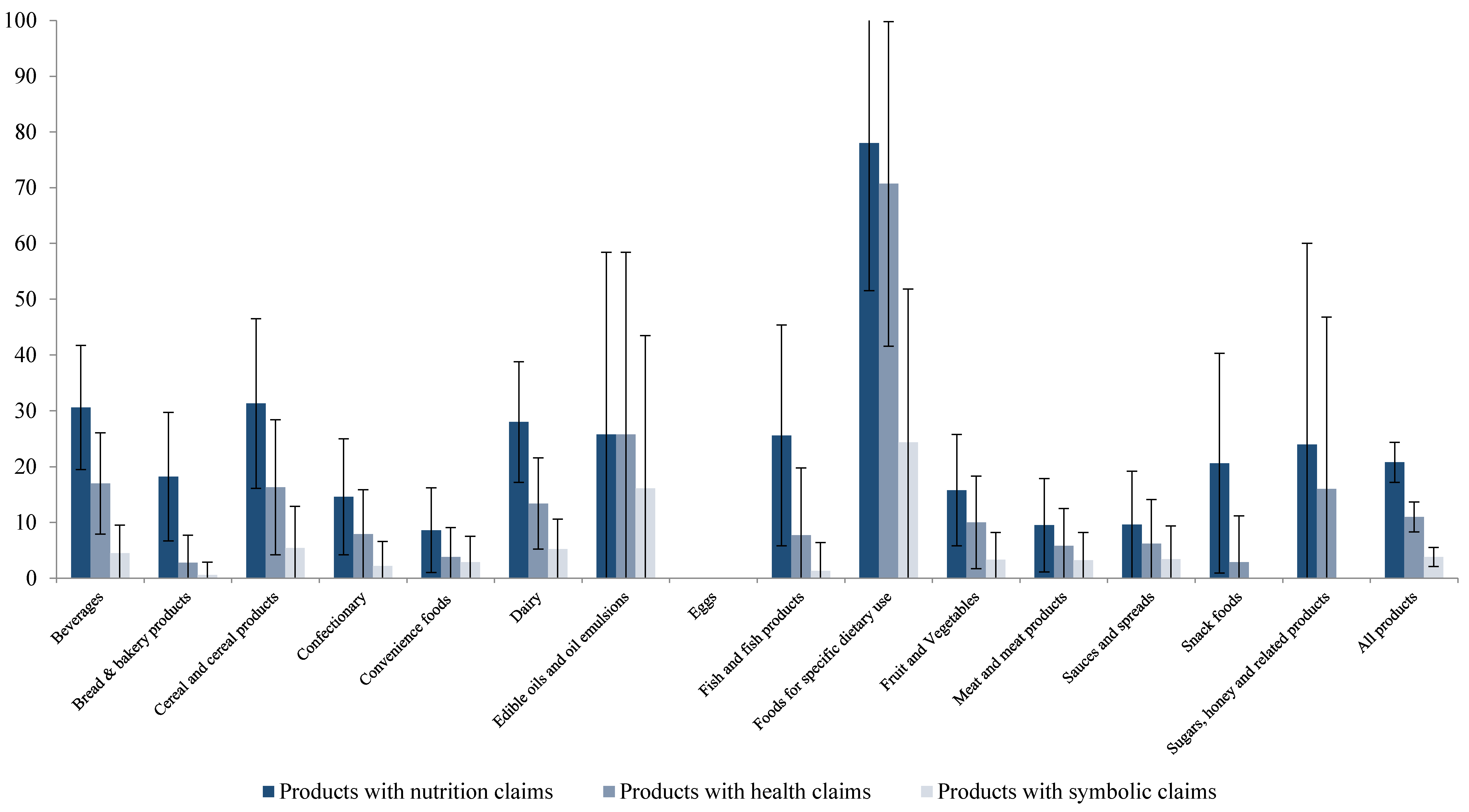
3.6. Which Types of Foods Carry NHC?
4. Discussion
4.1. Strengths
4.2. Limitations
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Commission Regulation (EC) No 1924/2006 of the European Parliament and of the Council on Nutrition and Health Claims Made on Foods. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:02006R1924-20141213 (accessed on 1 March 2016).
- Pravst, I. Functional Foods in Europe: A Focus on Health Claims. In Scientific, Health and Social Aspects of the Food Industry; Valdez, B., Ed.; In Tech: Rijeka, Croatia, 2012; pp. 165–208. [Google Scholar]
- Commission Regulation (EU) No 432/2012 Establishing a List of Permitted Health Claims Made on Foods, Other than Those Referring to the Reduction of Disease Risk and to Children’s Development and Health. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:02012R0432-20160101 (accessed on 1 March 2016).
- Kušar, A.; Pravst, I. Quality and safety of botanical food products and their labelling. Agro Food Ind. Hi Tech 2014, 25, 33–35. [Google Scholar]
- Hieke, S.; Kuljanic, N.; Wills, J.M.; Pravst, I.; Kaur, A.; Raats, M.M.; van Trijp, H.C.M.; Verbeke, W.; Grunert, K.G. The role of health-related claims and health-related symbols in consumer behaviour. Nutr. Bull. 2015, 40, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Brecher, S.J.; Bender, M.M.; Wilkening, V.L.; McCabe, N.M.; Anderson, E.M. Status of nutrition labeling, health claims, and nutrient content claims for processed foods: 1997 Food Label and Package Survey. J. Am. Diet. Assoc. 2000, 100, 1057–1062. [Google Scholar] [CrossRef]
- Colby, S.E.; Johnson, L.; Scheett, A.; Hoverson, B. Nutrition marketing on food labels. J. Nutr. Educ. Behav. 2013, 42, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Sacco, J.E.; Sumanac, D.; Tarasuk, V. Front-of-package references to fiber on foods in Canadian supermarkets highlight the need for increased nutrition knowledge among consumers. J. Nutr. Educ. Behav. 2013, 45, 518–524. [Google Scholar] [CrossRef] [PubMed]
- Schermel, A.; Emrich, T.E.; Arcand, J.; Wong, C.L.; L’Abbé, M.R. Nutrition marketing on processed food packages in Canada: 2010 Food Label Information Program. Appl. Physiol. Nutr. Metab. 2013, 38, 666–672. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.; Yeatman, H.; Zakrzewski, S.; Aboozaid, B.; Henshaw, S. Nutrition and related claims used on packaged Australian foods - implications for regulation. Asia Pac. J. Clin. Nutr. 2003, 12, 138–150. [Google Scholar] [PubMed]
- Williams, P.; Yeatman, H.; Ridges, L.; Houston, A.; Rafferty, J.; Roesler, A.; Sobierajski, M.; Spratt, B. Nutrition function, health and related claims on packaged Australian food products—prevalence and compliance with regulations. Asia Pac. J. Clin. Nutr. 2006, 15, 10–20. [Google Scholar] [PubMed]
- Walker, K.Z.; Woods, J.L.; Rickard, C.A.; Wong, C.K. Product variety in Australian snacks and drinks: How can the consumer make a healthy choice? Public Health Nutr. 2008, 11, 1046–1053. [Google Scholar] [CrossRef] [PubMed]
- Hughes, C.; Wellard, L.; Lin, J.; Suen, K.L.; Chapman, K. Regulating health claims on food labels using nutrient profiling: What will the proposed standard mean in the Australian supermarket? Public Health Nutr. 2013, 16, 2154–2161. [Google Scholar] [CrossRef] [PubMed]
- Devi, A.; Eyles, H.; Rayner, M.; Mhurchu, C.N.; Swinburn, B.; Lonsdale-Cooper, E.; Vandevijvere, S. Nutritional quality, labelling and promotion of breakfast cereals on the New Zealand market. Appetite 2014, 81, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Genannt Bonsmann, S.S.; Celemin, L.F.; Grunert, K.G. Food labelling to advance better education for life. Eur. J. Clin. Nutr. 2010, 64, S14–S19. [Google Scholar] [CrossRef] [PubMed]
- Genannt Bonsmann, S.S.; Celemin, L.F.; Larranaga, A.; Egger, S.; Wills, J.M.; Hodgkins, C.; Raats, M.M. Penetration of nutrition information on food labels across the EU-27 plus Turkey. Eur. J. Clin. Nutr. 2010, 64, 1379–1385. [Google Scholar] [CrossRef] [PubMed]
- Kaur, A.; Scarborough, P.; Matthews, A.; Payne, S.; Mizdrak, A.; Rayner, M. How many foods in the UK carry health and nutrition claims, and are they healthier than those that do not? Public Health Nutr. 2015. [Google Scholar] [CrossRef] [PubMed]
- Lalor, F.; Kennedy, J.; Flynn, M.A.T.; Wall, P.G. A study of nutrition and health claims—A snapshot of what’s on the Irish market. Public Health Nutr. 2010, 13, 704–711. [Google Scholar] [CrossRef] [PubMed]
- Pravst, I.; Kušar, A. Consumers’ exposure to nutrition and health claims on pre-packed foods: Use of sales weighting for assessing the food supply in Slovenia. Nutrients 2015, 7, 9353–9368. [Google Scholar] [CrossRef] [PubMed]
- Menard, C.; Dumas, C.; Gillot, N.; Laurent, L.; Labarbe, B.; Ireland, J.; Volatier, J.-L. The French OQALI survey on dairy products: Comparison of nutrient contents and other nutrition information on labels among types of brands. J. Hum. Nutr. Diet. 2012, 25, 323–333. [Google Scholar] [CrossRef] [PubMed]
- Debeljak, K.; Pravst, I; Košmelj, K.; Kač, M. “Healthier” and “less healthy” classifications according to three nutrient profiling systems relative to nutrition and health claims on food labels. Acta Aliment. 2015, 44, 561–565. [Google Scholar]
- Davidovic, D.B.; Tomic, D.V.; Paunovic, K.Ž.; Vasiljevic, N.D.; Jorga, J.B. Nutrition labelling of pre-packaged foods in Belgrade, Serbia: Current situation. Public Health Nutr. 2015, 18, 1969–1978. [Google Scholar] [CrossRef] [PubMed]
- Dunford, E.; Webster, J.; Metzler, A.B.; Czernichow, S.; Mhurchu, C.N.; Snowdon, W.; L’Abbe, M.; Li, N.; Maulik, P.K.; Barquera, S.; et al. International collaborative project to compare and monitor the nutritional composition of processed foods. Eur. J. Prev. Cardiol. 2012, 19, 1326–1332. [Google Scholar] [CrossRef] [PubMed]
- Dunford, E.; Webster, J.; Metzler, A.; Blanco, S.; Ni Mhurchu, C.; Wolmarans, P.; Snowdon, W.; L’Abbe, M. International collaborative project to compare and track the nutritional composition of fast foods. BMC Public Health 2012, 12, 1–7. [Google Scholar]
- Regulation (EU) No 1169/2011 on the Provision of Food Information to Consumers. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:02011R1169-20140219 (accessed on 29 February 2016).
- Roodenburg, A.J.C.; Popkin, B.M.; Seidell, J.C. Development of international criteria for a front of package food labelling system: The International Choices Programme. Eur. J. Clin. Nutr. 2011, 65, 1190–1200. [Google Scholar] [CrossRef] [PubMed]
- Cinar, A.B.; Murtomaa, H. A holistic food labelling strategy for preventing obesity and dental caries. Obes. Rev. 2009, 10, 357–361. [Google Scholar] [CrossRef] [PubMed]
- Rayner, M.; Wood, A.; Lawrence, M.; Mhurchu, C.N.; Albert, J.; Barquera, S.; Friel, S.; Hawkes, C.; Kelly, B.; Kumanyika, S.; et al. Monitoring the health-related labelling of foods and non-alcoholic beverages in retail settings. Obes. Rev. 2013, 14, 70–81. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation. International Classification of Functioning, Disability and Health (ICF). Available online: http://www.who.int/classifications/icf/en/ (accessed on 29 February 2016).
- Raats, M.M.; Malcolm, R.N.; Lähteenmaki, L.; Pravst, I.; Gage, H.; Cleary, A.; Klopčič, M. Understanding the impact of European Regulation on the substantiation and use of claims on food and drinks: Design of the REDICLAIM project and initial results. Nutr. Bull. 2015, 40, 340–348. [Google Scholar] [CrossRef]

| UK | Netherlands | Germany | Slovenia | Spain | Total | |
|---|---|---|---|---|---|---|
| Large supermarket/ national retailer | floor plan, n = 248 | store list, n = 252 | store list, n = 248 | store list, n = 260 | store list, n = 251 | 1259 (62%) |
| Discounter | floor plan, n = 75 | store list, n = 81 | store list, n = 76 | store list, n = 78 | store list, n = 78 | 388 (19%) |
| Neighborhood store | floor plan, n = 75 | store list, n = 83 | floor plan, n = 75 | store list, n = 78 | store list, n = 76 | 387 (19%) |
| Total no of foods | 398 (20%) | 416 (20%) | 399 (20%) | 416 (20%) | 405 (20%) | 2034 (100%) |
| Country | Claim Type | No. of Claims | … of Which Are Symbolic | No. of Foods with a Claim | % of Foods with Claim (95% CIs) |
|---|---|---|---|---|---|
| All Countries N = 2034 Foods | Nutrition claim | 865 | 1 | 423 | 20.8% (19.0%–22.5%) |
| Nutrient content claim | 797 | 1 | 399 | 19.6% (17.8%–21.3%) | |
| Nutrient comparative claims | 68 | 0 | 49 | 2.4% (1.7%–3.1%) | |
| Health-related ingredient claim | 105 | 6 | 72 | 3.5% (2.7%–4.3%) | |
| Health claim | 392 | 74 | 222 | 10.9% (9.6%–12.3%) | |
| General health claim | 153 | 64 | 137 | 6.7% (5.6%–7.8%) | |
| Nutrient and other function claim | 185 | 9 | 106 | 5.2% (4.2%–6.2%) | |
| Reduction of disease risk claim | 21 | 1 | 12 | 0.6% (0.2%–0.09%) | |
| Children’s development & health claims | 33 | 0 | 15 | 0.7% (0.4%–1.1%) | |
| Any type of claim (NHC) | 1362 | 81 | 528 | 26.0% (24.0%–27.9%) | |
| UK N = 398 foods | Nutrition claim | 247 | 0 | 118 | 29.6 (25.1%–34.1%) |
| Health-related ingredient claim | 65 | 3 | 40 | 10.1% (7.1%–13.0%) | |
| Health claim | 85 | 2 | 44 | 11.1% (8.0%–14.1%) | |
| General health claim | 30 | 0 | 23 | 5.8% (3.5%–8.1%) | |
| Nutrient and other function claim | 38 | 2 | 26 | 6.5% (4.1%–9.0%) | |
| Reduction of disease risk claim | 10 | 0 | 4 | 1.0% (0.0%–0.2%) | |
| Children’s development & health claims | 7 | 0 | 4 | 1.0% (0.0%–2.0%) | |
| Any type of claim (NHC) | 397 | 5 | 140 | 35.2% (30.4%–40.0%) | |
| Nether-lands N = 416 foods | Nutrition claim | 154 | 0 | 70 | 16.8% (13.2%–20.4%) |
| Health-related ingredient claim | 12 | 0 | 12 | 2.9% (1.3%–4.5%) | |
| Health claim | 73 | 50 | 60 | 14.4% (8.9%–15.2%) | |
| General health claim | 52 | 49 | 50 | 12.0% (8.9%–15.2%) | |
| Nutrient and other function claim | 19 | 1 | 12 | 2.9% (1.3%–4.5%) | |
| Reduction of disease risk claim | 2 | 0 | 1 | 0.2% (0.0%–0.1%) | |
| Children’s development & health claims | 0 | 0 | 0 | 0.0% | |
| Any type of claim (NHC) | 239 | 50 | 103 | 24.8% (20.6%–29.0%) | |
| Germany N = 399 foods | Nutrition claim | 123 | 0 | 64 | 16.0% (12.4%–19.7%) |
| Health-related ingredient claim | 19 | 1 | 13 | 3.3% (1.5%–5.0%) | |
| Health claim | 82 | 0 | 37 | 9.3% (6.4%–12.1%) | |
| General health claim | 29 | 0 | 23 | 5.8% (3.5%–8.1%) | |
| Nutrient and other function claim | 45 | 0 | 20 | 5.0% (2.9%–3.2%) | |
| Reduction of disease risk claim | 1 | 0 | 1 | 0.3% (–0.2%–0.7%) | |
| Children’s development & health claims | 7 | 0 | 5 | 1.3% (0.1%–0.2%) | |
| Any type of claim (NHC) | 224 | 1 | 82 | 20.6% (16.6%–24.5%) | |
| Slovenia N = 416 foods | Nutrition claim | 144 | 0 | 78 | 18.8% (15.0%–22.5%) |
| Health-related ingredient claim | 3 | 1 | 2 | 0.5% (–0.2%–1.1%) | |
| Health claim | 88 | 7 | 52 | 12.5% (0.9%–15.7%) | |
| General health claim | 24 | 1 | 23 | 5.5% (3.3%–7.7%) | |
| Nutrient and other function claim | 58 | 6 | 36 | 8.7% (5.9%–11.4%) | |
| Reduction of disease risk claim | 4 | 0 | 3 | 0.7% (–0.1%–1.5%) | |
| Children’s development & health claims | 2 | 0 | 1 | 0.2% (–0.2%–0.7%) | |
| Any type of claim (NHC) | 235 | 8 | 103 | 24.8% (20.6%–28.9%) | |
| Spain N = 405 | Nutrition claim | 196 | 1 | 93 | 23.0% (18.8%–27.1%) |
| Health-related ingredient claim | 6 | 1 | 5 | 1.2% (0.2%–2.3%) | |
| Health claim | 64 | 15 | 29 | 7.2% (4.6%–9.7%) | |
| General health claim | 18 | 14 | 18 | 4.4% (2.4%–6.5%) | |
| Nutrient and other function claim | 25 | 0 | 12 | 3.0% (1.3%–4.6%) | |
| Reduction of disease risk claim | 4 | 1 | 3 | 0.7% (–0.1%–1.6%) | |
| Children’s development & health claims | 17 | 0 | 5 | 1.2% (0.2%–2.3%) | |
| Any type of claim (NHC) | 266 | 17 | 100 | 24.7% (20.5%–28.9%) |
| Mean Number of Nutrition Claims 1 | Highest Number of Nutrition Claims on a Single Product | Mean Number of Health Claims 1 | Highest Number of Health Claims on a Single Product | Mean Number of Any Claim 1 | Highest Number of Any Claims on a Single Product | |
|---|---|---|---|---|---|---|
| All countries | 2.0 | 13 | 1.9 | 15 | 2.6 | 17 |
| UK | 2.1 | 11 | 1.9 | 5 | 2.8 | 15 |
| Netherlands | 2.2 | 8 | 1.2 | 6 | 2.3 | 9 |
| Germany | 1.9 | 13 | 2.2 | 15 | 2.7 | 17 |
| Slovenia | 1.8 | 8 | 1.7 | 5 | 2.3 | 11 |
| Spain | 2.1 | 12 | 2.2 | 11 | 2.7 | 17 |
| p value | 0.94 | <0.01 | 0.52 |
| Nutrient | Nutrition Claim (No.)s | % of All Nutrition Claims | Health Claim (No.)s | % of All Health Claims |
|---|---|---|---|---|
| Energy | 40 | 5% | 1 | <1% |
| Protein | 35 | 4% | 8 | 2% |
| Carbohydrates | 109 | 13% | 14 | 4% |
| Of which sugars | 100 | 12% | 2 | 1% |
| Fat | 206 | 24% | 31 | 8% |
| Total fat | 127 | 15% | 5 | 1% |
| Saturated fat | 7 | 1% | 3 | 1% |
| Unsaturated fat | 50 | 6% | 23 | 6% |
| Omega-3 fatty acids | 33 | 4% | 15 | 4% |
| Fiber | 74 | 9% | 14 | 4% |
| Sodium/Salt | 35 | 4% | 0 | 0% |
| Vitamins and/or minerals | 305 | 35% | 64 | 16% |
| Vitamins and Minerals | 2 | <1% | 3 | 1% |
| Vitamins (any) | 187 | 22% | 38 | 10% |
| Vitamin C | 47 | 5% | 8 | 2% |
| Vitamin D | 15 | 2% | 9 | 2% |
| Vitamin E | 19 | 2% | 5 | 1% |
| Other specified vitamins | 55 | 6% | 11 | 3% |
| Unspecified vitamins | 51 | 6% | 5 | 1% |
| Minerals (any) | 116 | 13% | 23 | 6% |
| Calcium | 55 | 6% | 13 | 3% |
| Iron | 21 | 2% | 3 | 1% |
| Other specified minerals | 26 | 3% | 6 | 2% |
| Unspecified minerals | 14 | 2% | 1 | <1% |
| Probiotics | 23 | 3% | 2 | 1% |
| Phytosterols/stanols | 6 | 1% | 5 | 1% |
| Whole products | 0 | 0% | 84 | 21% |
| Unspecified nutrient | 4 | <1% | 141 | 36% |
| Other nutrients | 27 | 3% | 9 | 2% |
| Ingredients that are not nutrients | 1 | <1% | 19 | 5% |
| Herbs | 0 | 0% | 12 | 3% |
| Seeds | 0 | 0% | 3 | 1% |
| Whole grain/Whole wheat / Whole foods/Whole meal | 1 | <1% | 2 | 1% |
| TOTAL | 865 | 100% | 392 | 100% |
| ICF Chapter | No. of Claims | % of Claims (95% CIs) | |
|---|---|---|---|
| Mental Functions | Global psychosocial functions | 4 | 2.2% (0.0%–4.3%) |
| Energy and drive functions | 8 | 4.4% (1.4%–7.4%) | |
| Sleep functions | 2 | 1.1% (–0.4%–0.26%) | |
| Specific mental functions | 1 | 0.5% (–0.5%–1.6%) | |
| Higher-level cognitive functions | 1 | 0.5% (–0.5%–1.6%) | |
| Total | 20 | 11.0% (6.4%–15.6%) | |
| Sensory Functions and Pain | Seeing and related functions | 1 | 0.5% (–0.5%–1.6%) |
| Total | 1 | 0.5% (–0.5%–1.6%) | |
| Voice and Speech Functions | Voice functions | 4 | 2.2% (0.0%–4.3%) |
| Total | 4 | 2.2% (0.0%–4.3%) | |
| Functions of the cardiovascular, hematological, immunological and respiratory systems | Heart functions | 9 | 4.9% (1.8%–8.1%) |
| Blood vessel functions | 1 | 0.5% (–0.5%–1.6%) | |
| Immunological system functions | 16 | 8.8% (4.6%–12.9%) | |
| Functions of the respiratory system | 1 | 0.5% (–0.5%–1.6%) | |
| Total | 27 | 14.8% (9.6%–20.0%) | |
| Functions of the digestive, metabolic and endocrine systems | Digestive functions | 19 | 10.4% (6.0%–14.9%) |
| Defecation functions | 5 | 2.7% (0.3%–5.1%) | |
| Weight maintenance functions | 14 | 7.7% (3.8%–11.6%) | |
| General metabolic functions | 27 | 14.8% (9.6%–20.0%) | |
| Water, mineral and electrolyte balance functions | 6 | 3.3% (0.7%–5.9%) | |
| Endocrine gland functions | 2 | 1.1% (–0.4%–2.6%) | |
| Total | 73 | 40.1% (32.9%–47.3%) | |
| Genitourinary and reproductive functions | Urinary excretory functions | 1 | 0.5% (–0.5%–1.6%) |
| Sexual functions | 2 | 1.1% (–0.4%–2.6%) | |
| Total | 3 | 1.6% (–0.2%–3.5%) | |
| Neuro-musculoskeletal and movement related functions | Functions of the joints and bones | 15 | 8.2% (4.2%–12.3%) |
| Muscle endurance functions | 4 | 2.2% (0.0%–4.3%) | |
| Total | 19 | 10.4% (6.0%–14.9%) | |
| Functions of the skin | Functions of the skin | 2 | 1.1% (–0.4%–2.6%) |
| Functions of the hair and nails | 1 | 0.5% (–0.5%–1.6%) | |
| Total | 3 | 1.6% (–0.2%–3.5%) | |
| Others | Functions related to the digestive system: Teeth | 15 | 8.2% (4.2%–12.3%) |
| Functions of the hematological and immunological systems: Anti-oxidants | 8 | 4.4% (1.4%–7.4%) | |
| Growth | 9 | 4.9% (1.8%–8.1%) | |
| Total | 32 | 18.1% (12.5%–23.8%) | |
| TOTAL | 185 | 100.0% 5.2% (4.2%–6.2%) | |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hieke, S.; Kuljanic, N.; Pravst, I.; Miklavec, K.; Kaur, A.; Brown, K.A.; Egan, B.M.; Pfeifer, K.; Gracia, A.; Rayner, M. Prevalence of Nutrition and Health-Related Claims on Pre-Packaged Foods: A Five-Country Study in Europe. Nutrients 2016, 8, 137. https://doi.org/10.3390/nu8030137
Hieke S, Kuljanic N, Pravst I, Miklavec K, Kaur A, Brown KA, Egan BM, Pfeifer K, Gracia A, Rayner M. Prevalence of Nutrition and Health-Related Claims on Pre-Packaged Foods: A Five-Country Study in Europe. Nutrients. 2016; 8(3):137. https://doi.org/10.3390/nu8030137
Chicago/Turabian StyleHieke, Sophie, Nera Kuljanic, Igor Pravst, Krista Miklavec, Asha Kaur, Kerry A. Brown, Bernadette M. Egan, Katja Pfeifer, Azucena Gracia, and Mike Rayner. 2016. "Prevalence of Nutrition and Health-Related Claims on Pre-Packaged Foods: A Five-Country Study in Europe" Nutrients 8, no. 3: 137. https://doi.org/10.3390/nu8030137
APA StyleHieke, S., Kuljanic, N., Pravst, I., Miklavec, K., Kaur, A., Brown, K. A., Egan, B. M., Pfeifer, K., Gracia, A., & Rayner, M. (2016). Prevalence of Nutrition and Health-Related Claims on Pre-Packaged Foods: A Five-Country Study in Europe. Nutrients, 8(3), 137. https://doi.org/10.3390/nu8030137








