Intravenous Administration of Lycopene, a Tomato Extract, Protects against Myocardial Ischemia-Reperfusion Injury
Abstract
:1. Introduction
2. Methods and Materials
2.1. Animals, Cell Lines and Chemicals
2.2. In Vitro Hypoxia Model and Trypan Blue Exclusion Assay
2.3. In Vivo Myocardial I/R Model
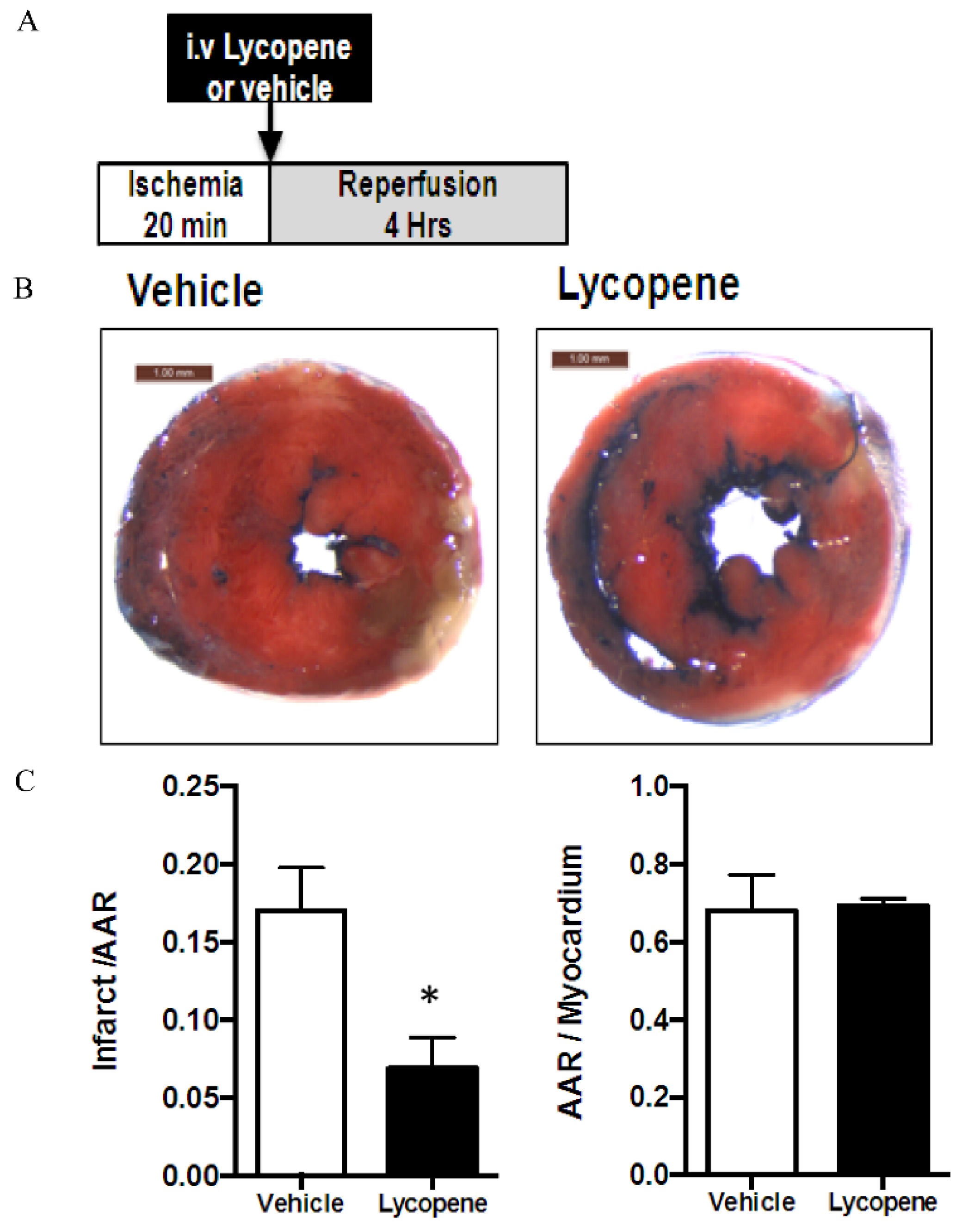
2.4. Measurement of Myocardial Infarct
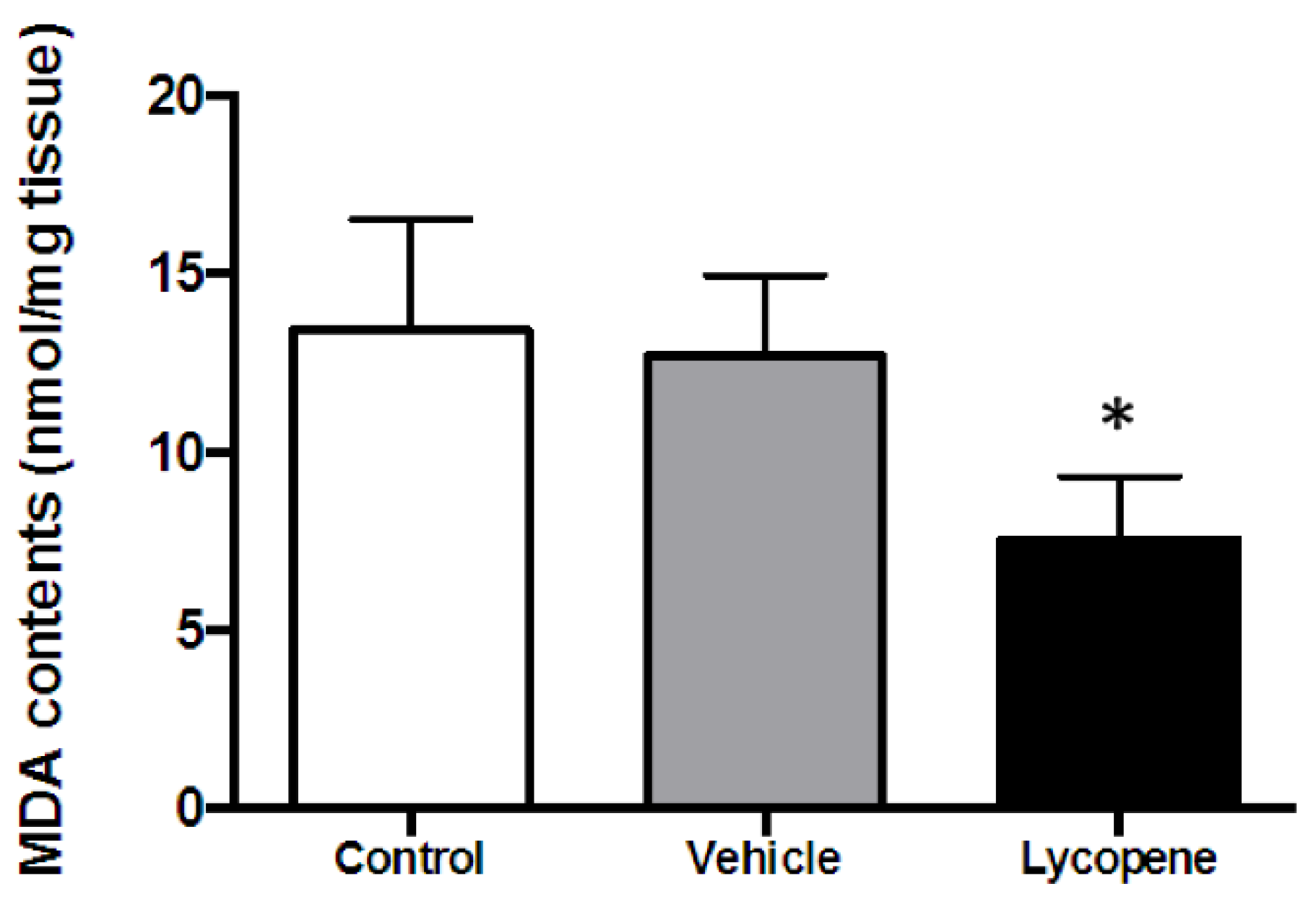
2.5. MDA Assay
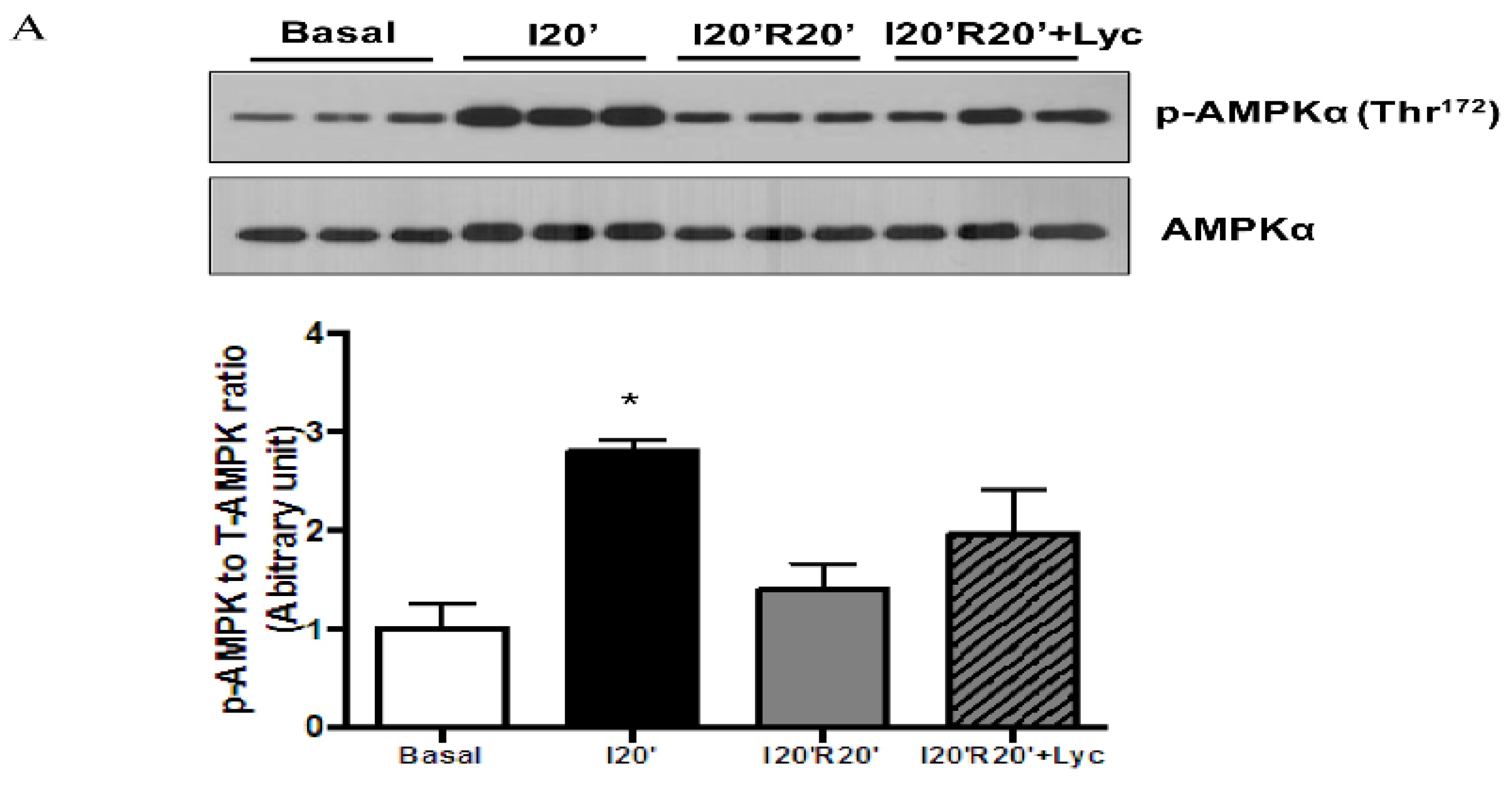
2.6. Immunoblotting Analysis
2.7. Statistical Analysis
3. Results
3.1. Lycopene Protects against I/R Induced Cardiomyocyte Death
3.2. Effects of Intravenous Administration of Lycopene on I/R Induced Myocardial Infarct
3.3. Effect of Lycopene on ROS Generation during I/R
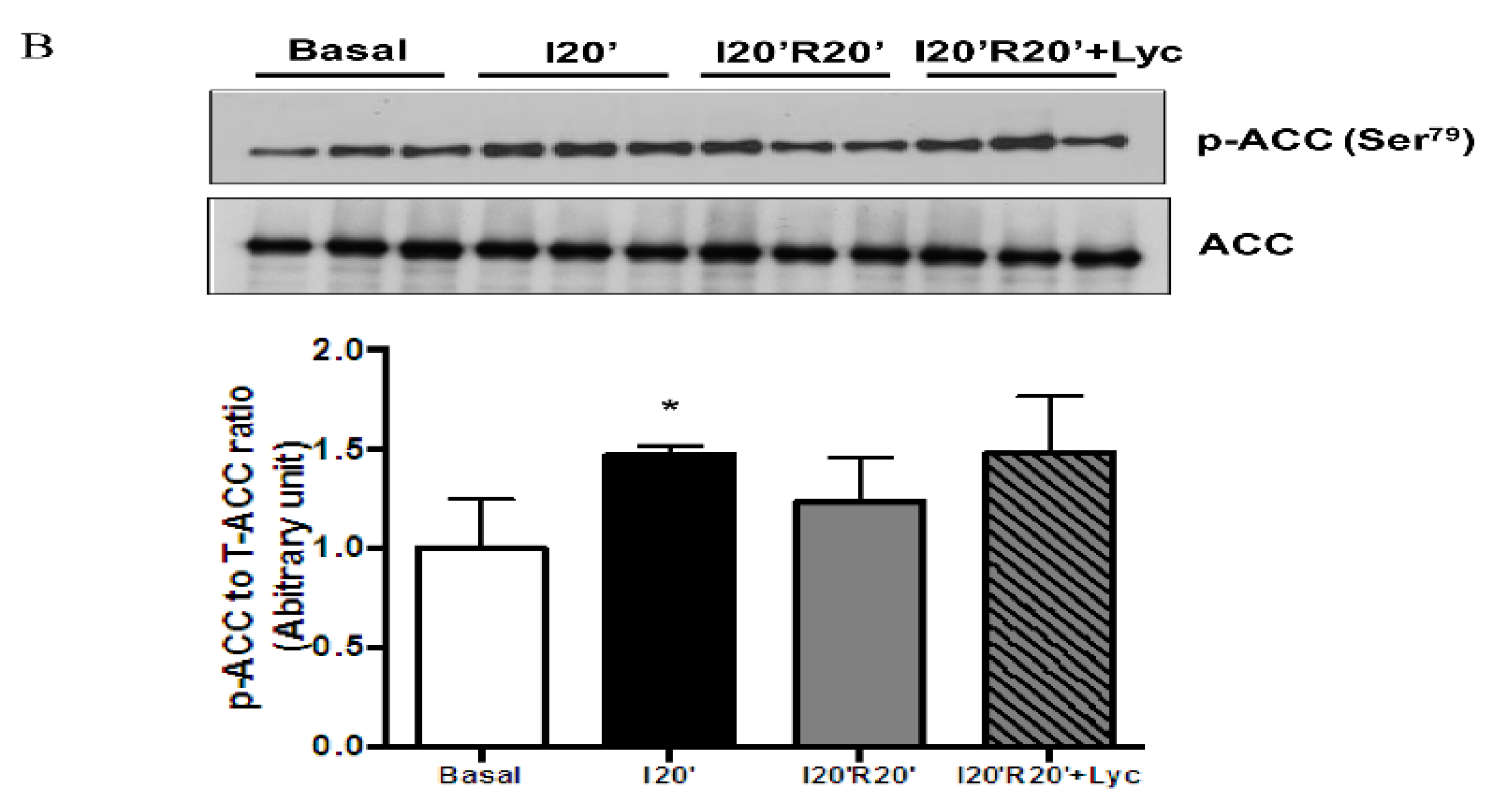
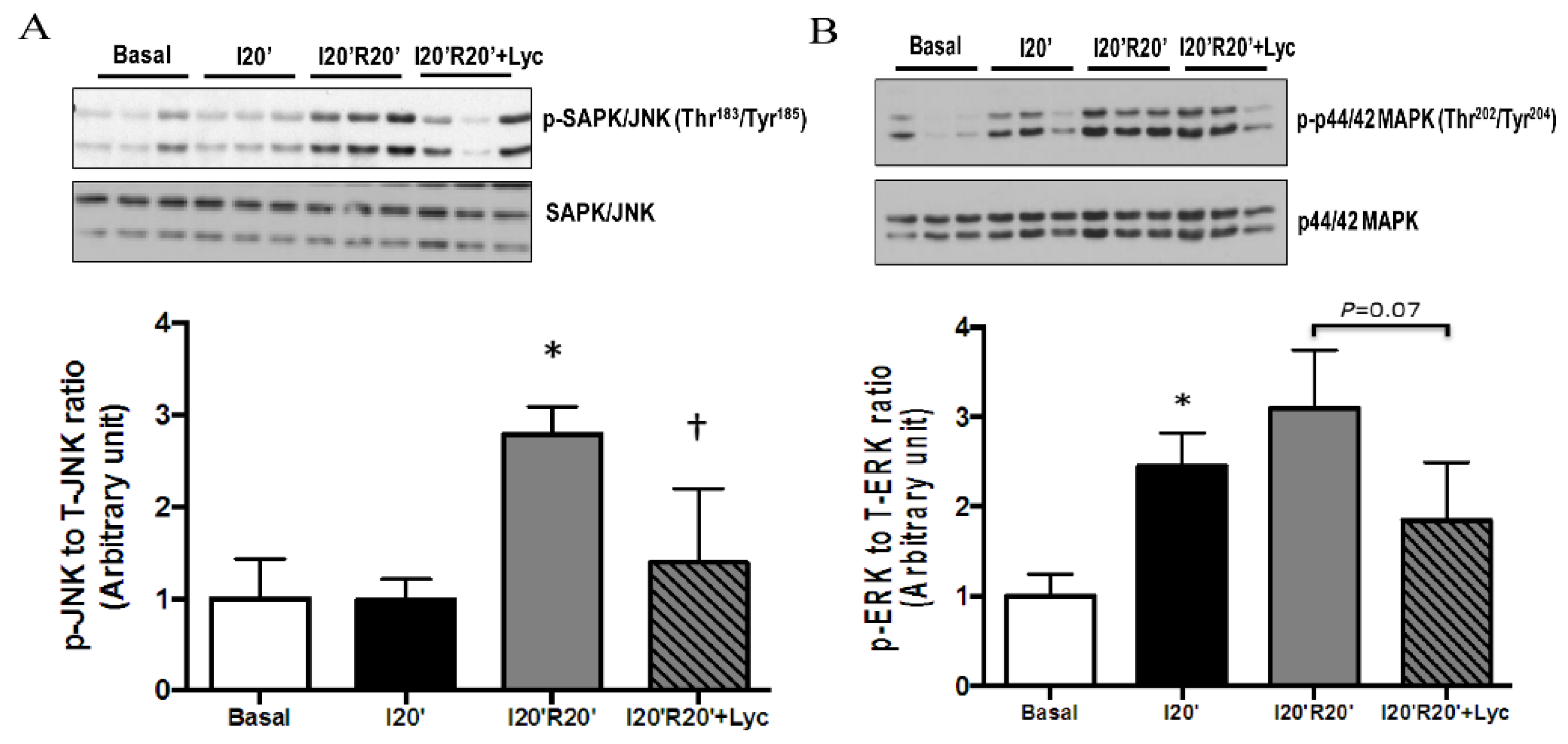
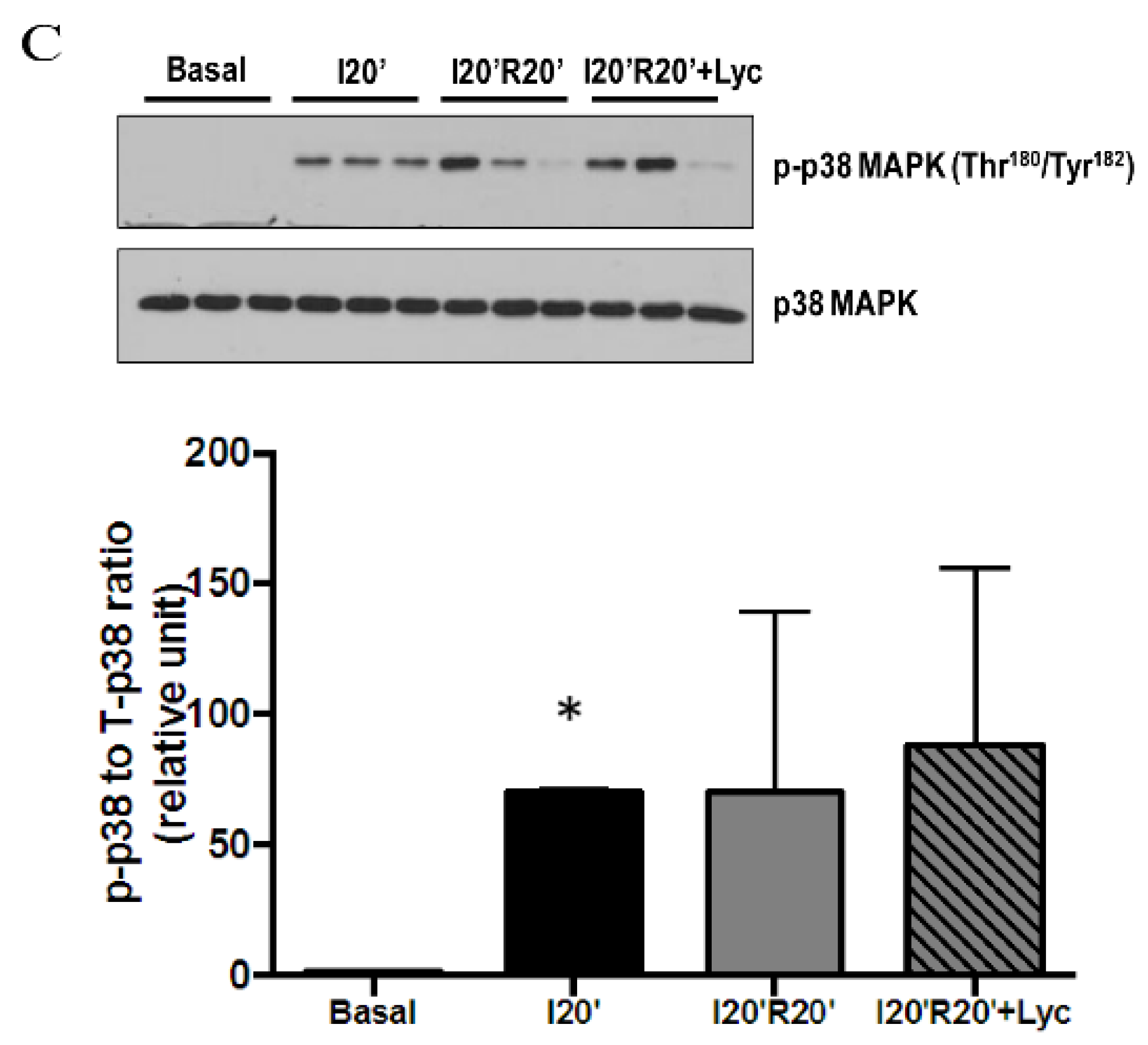
3.4. Signaling Pathways Regulated by Acute Lycopene Treatment during I/R
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Liu, Y.H.; Yang, X.P.; Sharov, V.G.; Sigmon, D.H.; Sabbah, H.N.; Carretero, O.A. Paracrine systems in the cardioprotective effect of angiotensin-converting enzyme inhibitors on myocardial ischemia reperfusion injury in rats. Hypertension 1996, 27, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Moghaddas, S.; Hoppel, C.L.; Lesnefsky, E.J. Ischemic defects in the electron transport chain increase the production of reactive oxygen species from isolated rat heart mitochondria. Am. J. Physiol. Cell Physiol. 2008, 294, C460–C466. [Google Scholar] [CrossRef] [PubMed]
- Jolly, S.R.; Kane, W.J.; Bailie, M.B.; Abrams, G.D.; Lucchesi, B.R. Canine myocardial reperfusion injury—Its reduction by the combined administration of superoxide-dismutase and catalase. Circ. Res. 1984, 54, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Piao, C.S.; Gao, S.; Lee, G.H.; Kim, D.S.; Park, B.H.; Chae, S.W.; Chae, H.J.; Kim, S.H. Sulforaphane protects ischemic injury of hearts through antioxidant pathway and mitochondrial k-atp channels. Pharmacol. Res. 2010, 61, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Di Mascio, P.; Kaiser, S.; Sies, H. Lycopene as the most efficient biological carotenoid singlet oxygen quencher. Arch. Biochem. Biophys. 1989, 274, 532–538. [Google Scholar] [CrossRef]
- Khachik, F.; Carvalho, L.; Bernstein, P.S.; Muir, G.J.; Zhao, D.Y.; Katz, N.B. Chemistry, distribution, and metabolism of tomato carotenoids and their impact on human health. Exp. Biol. Med. 2002, 227, 845–851. [Google Scholar]
- Schwarz, S.; Obermuller-Jevic, U.C.; Hellmis, E.; Koch, W.; Jacobi, G.; Biesalski, H.K. Lycopene inhibits disease progression in patients with benign prostate hyperplasia. J. Nutr. 2008, 138, 49–53. [Google Scholar] [PubMed]
- Qu, M.; Li, L.; Chen, C.; Li, M.; Pei, L.; Chu, F.; Yang, J.; Yu, Z.; Wang, D.; Zhou, Z. Protective effects of lycopene against amyloid beta-induced neurotoxicity in cultured rat cortical neurons. Neurosci. Lett. 2011, 505, 286–290. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, M.; Fechner, M.; Kalkowski, J.; Frohlich, K.; Trautmann, A.; Bohm, V.; Liebisch, G.; Lehneis, S.; Schmitz, G.; Ludwig, A.; et al. Effects of lycopene on the initial state of atherosclerosis in New Zealand white (nzw) rabbits. PLoS ONE 2012, 7, e30808. [Google Scholar] [CrossRef] [PubMed]
- Ghavipour, M.; Saedisomeolia, A.; Djalali, M.; Sotoudeh, G.; Eshraghyan, M.R.; Moghadam, A.M.; Wood, L.G. Tomato juice consumption reduces systemic inflammation in overweight and obese females. Br. J. Nutr. 2013, 109, 2031–2035. [Google Scholar] [CrossRef] [PubMed]
- Giovannucci, E. Tomatoes, tomato-based products, lycopene, and cancer: Review of the epidemiologic literature. J. Natl. Cancer Inst. 1999, 91, 317–331. [Google Scholar] [CrossRef] [PubMed]
- Quilliot, D.; Forbes, A.; Dubois, F.; Gueant, J.L.; Ziegler, O. Carotenoid deficiency in chronic pancreatitis: The effect of an increase in tomato consumption. Eur. J. Clin. Nutr. 2011, 65, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Sanderson, M.J.; White, K.L.; Drake, I.M.; Schorah, C.J. Vitamin e and carotenoids in gastric biopsies: The relation to plasma concentrations in patients with and without helicobacter pylori gastritis. Am. J. Clin. Nutr. 1997, 65, 101–106. [Google Scholar] [PubMed]
- Rissanen, T.H.; Voutilainen, S.; Nyyssonen, K.; Salonen, R.; Kaplan, G.A.; Salonen, J.T. Serum lycopene concentrations and carotid atherosclerosis: The kuopio ischaemic heart disease risk factor study. Am. J. Clin. Nutr. 2003, 77, 133–138. [Google Scholar] [PubMed]
- Sesso, H.D.; Buring, J.E.; Norkus, E.P.; Gaziano, J.M. Plasma lycopene, other carotenoids, and retinol and the risk of cardiovascular disease in women. Am. J. Clin. Nutr. 2004, 79, 47–53. [Google Scholar] [PubMed]
- Kohlmeier, L.; Kark, J.D.; Gomez-Gracia, E.; Martin, B.C.; Steck, S.E.; Kardinaal, A.F.; Ringstad, J.; Thamm, M.; Masaev, V.; Riemersma, R.; et al. Lycopene and myocardial infarction risk in the euramic study. Am. J. Epidemiol. 1997, 146, 618–626. [Google Scholar] [CrossRef] [PubMed]
- Bansal, P.; Gupta, S.K.; Ojha, S.K.; Nandave, M.; Mittal, R.; Kumari, S.; Arya, D.S. Cardioprotective effect of lycopene in the experimental model of myocardial ischemia-reperfusion injury. Mol. Cell. Biochem. 2006, 289, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Aman, U.; Vaibhav, P.; Balaraman, R. Tomato lycopene attenuates myocardial infarction induced by isoproterenol: Electrocardiographic, biochemical and anti-apoptotic study. Asian Pac. J. Trop. Biomed. 2012, 2, 345–351. [Google Scholar] [PubMed]
- Ross, A.B.; Vuong le, T.; Ruckle, J.; Synal, H.A.; Schulze-Konig, T.; Wertz, K.; Rumbeli, R.; Liberman, R.G.; Skipper, P.L.; Tannenbaum, S.R.; et al. Lycopene bioavailability and metabolism in humans: An accelerator mass spectrometry study. Am. J. Clin. Nutr. 2011, 93, 1263–1273. [Google Scholar] [CrossRef] [PubMed]
- Moran, N.E.; Clinton, S.K.; Erdman, J.W., Jr. Differential bioavailability, clearance, and tissue distribution of the acyclic tomato carotenoids lycopene and phytoene in mongolian gerbils. J. Nutr. 2013, 143, 1920–1926. [Google Scholar] [CrossRef] [PubMed]
- Collins, J.K.; Arjmandi, B.H.; Claypool, P.L.; Perkins-Veazie, P.; Baker, R.A.; Clevidence, B.A. Lycopene from two food sources does not affect antioxidant or cholesterol status of middle-aged adults. Nutr. J. 2004, 3. [Google Scholar] [CrossRef] [PubMed]
- Claycomb, W.C.; Lanson, N.A., Jr.; Stallworth, B.S.; Egeland, D.B.; Delcarpio, J.B.; Bahinski, A.; Izzo, N.J., Jr. Hl-1 cells: A cardiac muscle cell line that contracts and retains phenotypic characteristics of the adult cardiomyocyte. Proc. Natl. Acad. Sci. USA 1998, 95, 2979–2984. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.J.; Li, J.; Leng, L.; McDonald, C.; Atsumi, T.; Bucala, R.; Young, L.H. Macrophage migration inhibitory factor stimulates AMP-activated protein kinase in the ischaemic heart. Nature 2008, 451, U578–U579. [Google Scholar] [CrossRef] [PubMed]
- Tong, C.; Morrison, A.; Mattison, S.; Qian, S.; Bryniarski, M.; Rankin, B.; Wang, J.; Thomas, D.P.; Li, J. Impaired sirt1 nucleocytoplasmic shuttling in the senescent heart during ischemic stress. FASEB J. 2013, 27, 4332–4342. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Wang, J.; Thomas, D.P.; Tong, C.; Leng, L.; Wang, W.; Merk, M.; Zierow, S.; Bernhagen, J.; Ren, J.; et al. Impaired macrophage migration inhibitory factor-AMP-activated protein kinase activation and ischemic recovery in the senescent heart. Circulation 2010, 122, 282–292. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Ma, H.; Tong, C.; Zhang, H.; Lawlis, G.B.; Li, Y.; Zang, M.; Ren, J.; Nijland, M.J.; Ford, S.P.; et al. Overnutrition and maternal obesity in sheep pregnancy alter the JNK-IRS-1 signaling cascades and cardiac function in the fetal heart. FASEB J. 2010, 24, 2066–2076. [Google Scholar] [CrossRef] [PubMed]
- Hladunewich, M.; Karumanchi, S.A.; Lafayette, R. Pathophysiology of the clinical manifestations of preeclampsia. Clin. J. Am. Soc. Nephrol. CJASN 2007, 2, 543–549. [Google Scholar] [CrossRef] [PubMed]
- Schierle, J.; Bretzel, W.; Buhler, I.; Faccin, N.; Hess, D.; Steiner, K.; Schuep, W. Content and isomeric ratio of lycopene in food and human blood plasma. Food Chem. 1997, 59, 459–465. [Google Scholar] [CrossRef]
- Kyriakis, J.M.; Avruch, J. Mammalian mapk signal transduction pathways activated by stress and inflammation: A 10-year update. Physiol. Rev. 2012, 92, 689–737. [Google Scholar] [CrossRef] [PubMed]
- Fujita, K.; Yoshimoto, N.; Kato, T.; Imada, H.; Matsumoto, G.; Inakuma, T.; Nagata, Y.; Miyachi, E. Lycopene inhibits ischemia/reperfusion-induced neuronal apoptosis in gerbil hippocampal tissue. Neurochem. Res. 2013, 38, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Hekimoglu, A.; Kurcer, Z.; Aral, F.; Baba, F.; Sahna, E.; Atessahin, A. Lycopene, an antioxidant carotenoid, attenuates testicular injury caused by ischemia/reperfusion in rats. Tohoku J. Exp. Med. 2009, 218, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Boileau, T.W.; Clinton, S.K.; Erdman, J.W., Jr. Tissue lycopene concentrations and isomer patterns are affected by androgen status and dietary lycopene concentration in male f344 rats. J. Nutr. 2000, 130, 1613–1618. [Google Scholar] [PubMed]
- Li, H.; Deng, Z.; Liu, R.; Loewen, S.; Tsao, R. Carotenoid compositions of coloured tomato cultivars and contribution to antioxidant activities and protection against H2O2-induced cell death in H9C2. Food Chem. 2013, 136, 878–888. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Jia, P.; Shu, W.; Jia, D. The protective effect of lycopene on hypoxia/reoxygenation-induced endoplasmic reticulum stress in H9C2 cardiomyocytes. Eur. J. Pharmacol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Yue, R.; Hu, H.; Yiu, K.H.; Luo, T.; Zhou, Z.; Xu, L.; Zhang, S.; Li, K.; Yu, Z. Lycopene protects against hypoxia/reoxygenation-induced apoptosis by preventing mitochondrial dysfunction in primary neonatal mouse cardiomyocytes. PLoS ONE 2012, 7, e50778. [Google Scholar]
- Rosano, G.M.C.; Fini, M.; Caminiti, G.; Barbaro, G. Cardiac metabolism in myocardial ischemia. Curr. Pharm. Des. 2008, 14, 2551–2562. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.F.; Salem, J.E.; Saidel, G.M.; Stanley, W.C.; Cabrera, M.E. Mechanistic model of cardiac energy metabolism predicts localization of glycolysis to cytosolic subdomain during ischemia. Am. J. Physiol. Heart C 2005, 288, H2400–H2411. [Google Scholar] [CrossRef] [PubMed]
- Paradies, G.; Petrosillo, G.; Pistolese, M.; Di Venosa, N.; Federici, A.; Ruggiero, F.M. Decrease in mitochondrial complex I activity in ischemic/reperfused rat heart—Involvement of reactive oxygen species and cardiolipin. Circ. Res. 2004, 94, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Ye, G.; Metreveli, N.S.; Donthi, R.V.; Xia, S.; Xu, M.; Carlson, E.C.; Epstein, P.N. Catalase protects cardiomyocyte function in models of type 1 and type 2 diabetes. Diabetes 2004, 53, 1336–1343. [Google Scholar] [CrossRef] [PubMed]
- Krishnamoorthy, G.; Selvakumar, K.; Venkataraman, P.; Elumalai, P.; Arunakaran, J. Lycopene supplementation prevents reactive oxygen species mediated apoptosis in sertoli cells of adult albino rats exposed to polychlorinated biphenyls. Interdiscip. Toxicol. 2013, 6, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Ben-Dor, A.; Steiner, M.; Gheber, L.; Danilenko, M.; Dubi, N.; Linnewiel, K.; Zick, A.; Sharoni, Y.; Levy, J. Carotenoids activate the antioxidant response element transcription system. Mol. Cancer Ther. 2005, 4, 177–186. [Google Scholar] [PubMed]
- Goo, Y.A.; Li, Z.; Pajkovic, N.; Shaffer, S.; Taylor, G.; Chen, J.; Campbell, D.; Arnstein, L.; Goodlett, D.R.; van Breemen, R.B. Systematic investigation of lycopene effects in lncap cells by use of novel large-scale proteomic analysis software. Proteom. Clin. Appl. 2007, 1, 513–523. [Google Scholar] [CrossRef] [PubMed]
- Russell, R.R., 3rd; Li, J.; Coven, D.L.; Pypaert, M.; Zechner, C.; Palmeri, M.; Giordano, F.J.; Mu, J.; Birnbaum, M.J.; Young, L.H. AMP-activated protein kinase mediates ischemic glucose uptake and prevents postischemic cardiac dysfunction, apoptosis, and injury. J. Clin. Investig. 2004, 114, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Santulli, G.; Xie, W.; Reiken, S.R.; Marks, A.R. Mitochondrial calcium overload is a key determinant in heart failure. Proc. Natl. Acad. Sci. USA 2015, 112, 11389–11394. [Google Scholar] [CrossRef] [PubMed]
- Hawley, S.A.; Pan, D.A.; Mustard, K.J.; Ross, L.; Bain, J.; Edelman, A.M.; Frenguelli, B.G.; Hardie, D.G. Calmodulin-dependent protein kinase kinase-beta is an alternative upstream kinase for AMP-activated protein kinase. Cell Metab. 2005, 2, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, S.C. Protein kinase activation and myocardial ischemia/reperfusion injury. Cardiovasc. Res. 2004, 61, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Yue, T.L.; Wang, C.; Gu, J.L.; Ma, X.L.; Kumar, S.; Lee, J.C.; Feuerstein, G.Z.; Thomas, H.; Maleeff, B.; Ohlstein, E.H. Inhibition of extracellular signal-regulated kinase enhances ischemia/reoxygenation-induced apoptosis in cultured cardiac myocytes and exaggerates reperfusion injury in isolated perfused heart. Circ. Res. 2000, 86, 692–699. [Google Scholar] [CrossRef] [PubMed]
- Ferrandi, C.; Ballerio, R.; Gaillard, P.; Giachetti, C.; Carboni, S.; Vitte, P.A.; Gotteland, J.P.; Cirillo, R. Inhibition of c-Jun N-terminal kinase decreases cardiomyocyte apoptosis and infarct size after myocardial ischemia and reperfusion in anaesthetized rats. Br. J. Pharmacol. 2004, 142, 953–960. [Google Scholar] [CrossRef] [PubMed]
- Qi, D.; Hu, X.; Wu, X.; Merk, M.; Leng, L.; Bucala, R.; Young, L.H. Cardiac macrophage migration inhibitory factor inhibits jnk pathway activation and injury during ischemia/reperfusion. J. Clin. Investig. 2009, 119, 3807–3816. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Lee, J.S.; Han, Y.S.; Lee, J.H.; Bae, I.; Yoon, Y.M.; Kwon, S.M.; Lee, S.H. Pretreatment with lycopene attenuates oxidative stress-induced apoptosis in human mesenchymal stem cells. Biomol. Ther. 2015, 23, 517–524. [Google Scholar] [CrossRef] [PubMed]
- Strohm, C.; Barancik, T.; Bruhl, M.L.; Kilian, S.A.; Schaper, W. Inhibition of the ER-kinase cascade by PD98059 and UO126 counteracts ischemic preconditioning in pig myocardium. J. Cardiovasc. Pharmacol. 2000, 36, 218–229. [Google Scholar] [CrossRef] [PubMed]
- Barancik, M.; Htun, P.; Strohm, C.; Kilian, S.; Schaper, W. Inhibition of the cardiac p38-MAPK pathway by SB203580 delays ischemic cell death. J. Cardiovasc. Pharmacol. 2000, 35, 474–483. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Miller, E.J.; Ninomiya-Tsuji, J.; Russell, R.R., 3rd; Young, L.H. AMP-activated protein kinase activates p38 mitogen-activated protein kinase by increasing recruitment of p38 MAPK to tab1 in the ischemic heart. Circ. Res. 2005, 97, 872–879. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Sun, Z.W.; Ye, L.F.; Fu, G.S.; Mou, Y.; Hu, S.J. Lycopene protects against apoptosis in hypoxia/reoxygenationinduced H9C2 myocardioblast cells through increased autophagy. Mol. Med. Rep. 2015, 11, 1358–1365. [Google Scholar] [PubMed]
- Xu, J.; Hu, H.; Chen, B.; Yue, R.; Zhou, Z.; Liu, Y.; Zhang, S.; Xu, L.; Wang, H.; Yu, Z. Lycopene protects against hypoxia/reoxygenation injury by alleviating er stress induced apoptosis in neonatal mouse cardiomyocytes. PLoS ONE 2015, 10, e0136443. [Google Scholar] [CrossRef] [PubMed]

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tong, C.; Peng, C.; Wang, L.; Zhang, L.; Yang, X.; Xu, P.; Li, J.; Delplancke, T.; Zhang, H.; Qi, H. Intravenous Administration of Lycopene, a Tomato Extract, Protects against Myocardial Ischemia-Reperfusion Injury. Nutrients 2016, 8, 138. https://doi.org/10.3390/nu8030138
Tong C, Peng C, Wang L, Zhang L, Yang X, Xu P, Li J, Delplancke T, Zhang H, Qi H. Intravenous Administration of Lycopene, a Tomato Extract, Protects against Myocardial Ischemia-Reperfusion Injury. Nutrients. 2016; 8(3):138. https://doi.org/10.3390/nu8030138
Chicago/Turabian StyleTong, Chao, Chuan Peng, Lianlian Wang, Li Zhang, Xiaotao Yang, Ping Xu, Jinjin Li, Thibaut Delplancke, Hua Zhang, and Hongbo Qi. 2016. "Intravenous Administration of Lycopene, a Tomato Extract, Protects against Myocardial Ischemia-Reperfusion Injury" Nutrients 8, no. 3: 138. https://doi.org/10.3390/nu8030138
APA StyleTong, C., Peng, C., Wang, L., Zhang, L., Yang, X., Xu, P., Li, J., Delplancke, T., Zhang, H., & Qi, H. (2016). Intravenous Administration of Lycopene, a Tomato Extract, Protects against Myocardial Ischemia-Reperfusion Injury. Nutrients, 8(3), 138. https://doi.org/10.3390/nu8030138




