Dietary Intake of Trans Fatty Acids in Children Aged 4–5 in Spain: The INMA Cohort Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Dietary Assessment
2.3. Covariates
2.4. Statistical Analysis
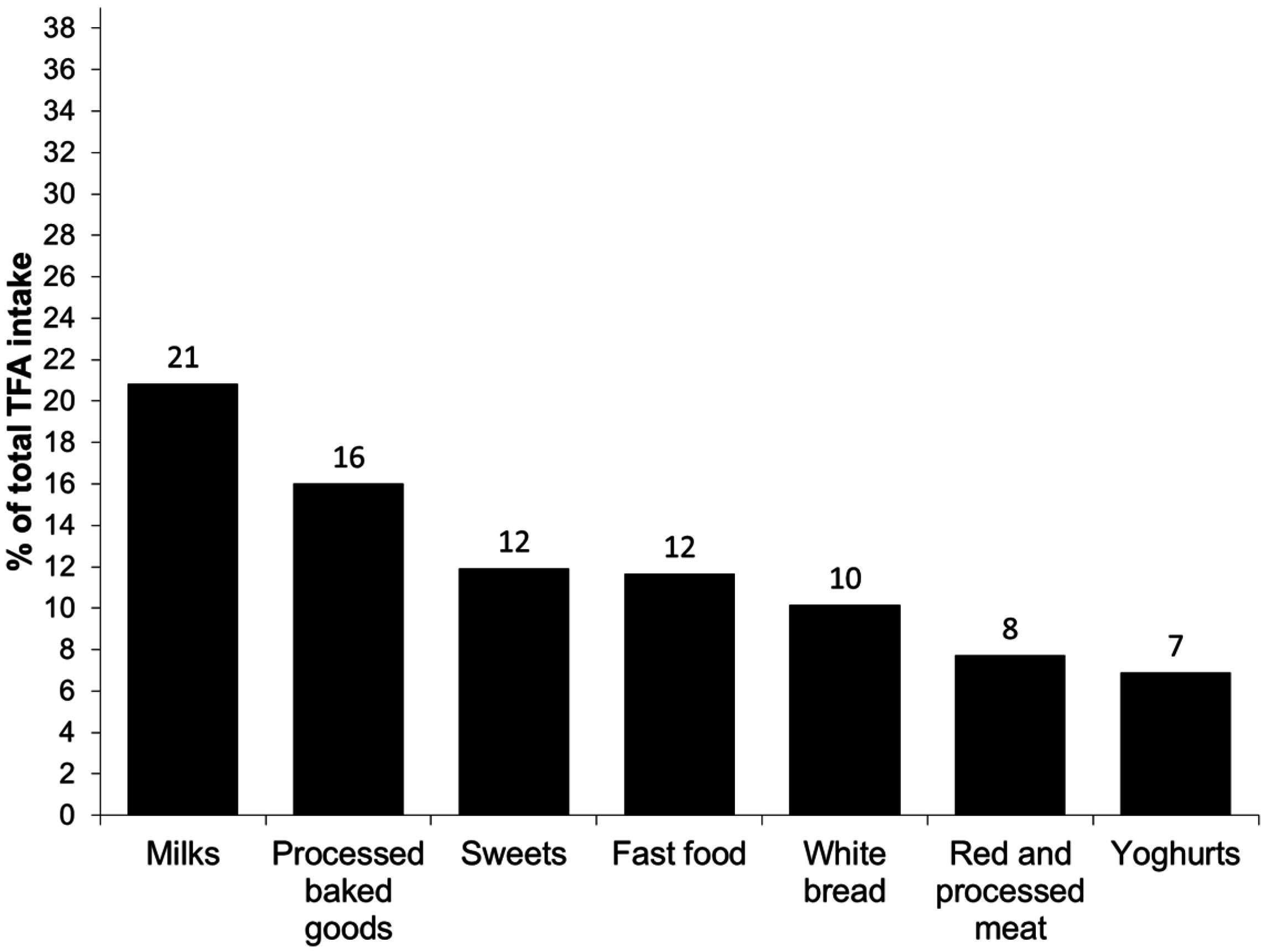
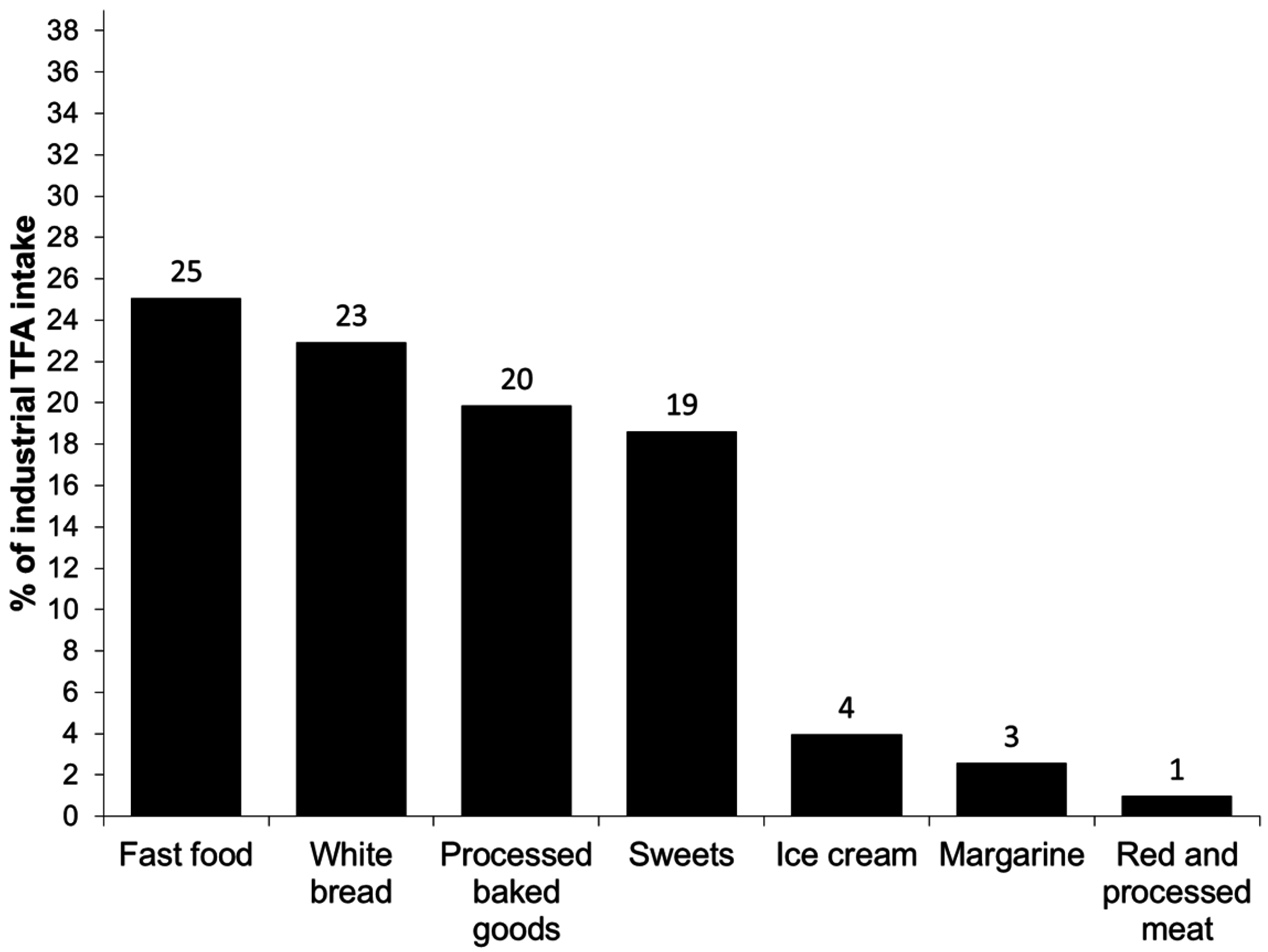
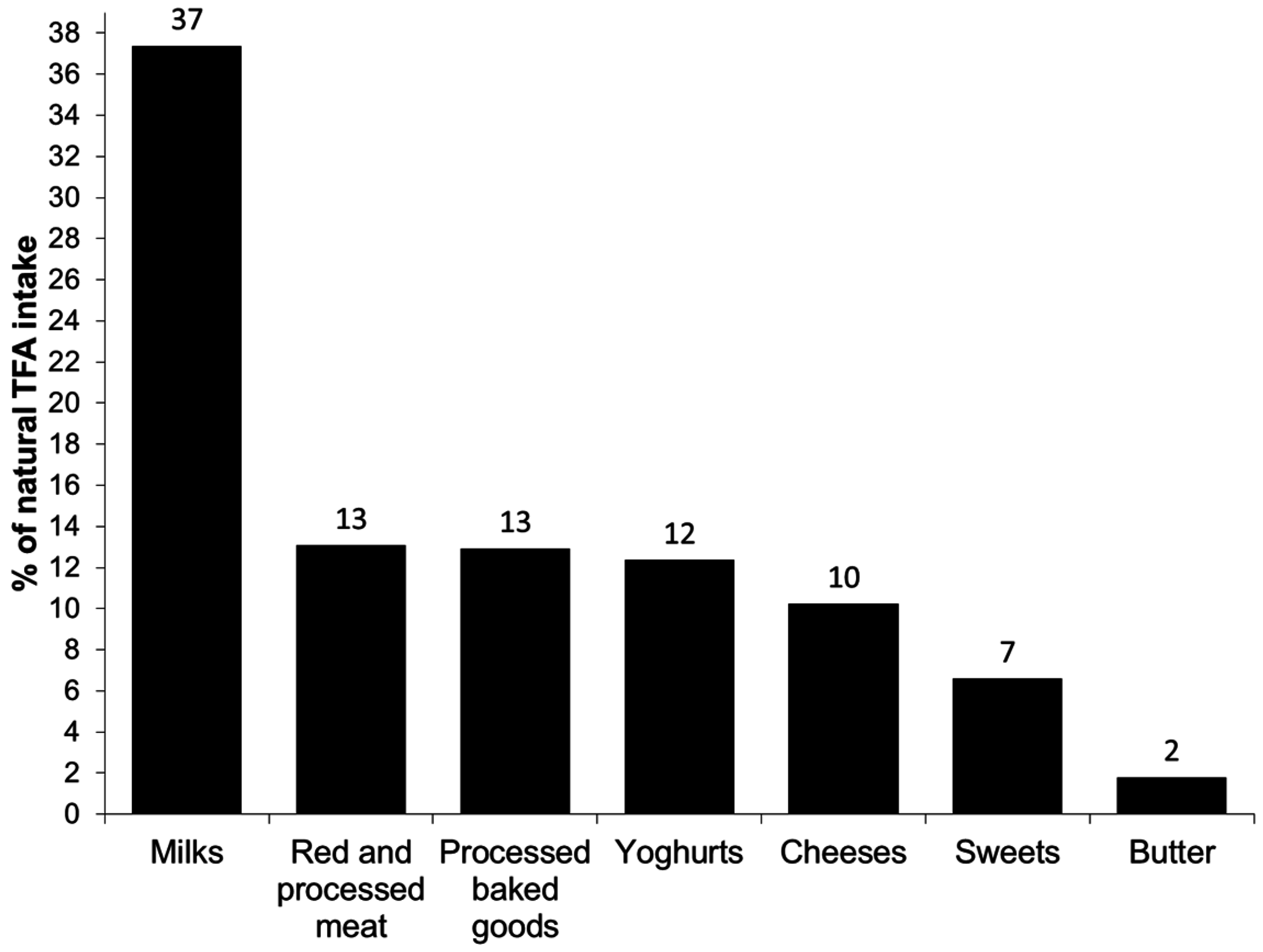
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nishida, C.; Uauy, R. WHO Scientific Update on health consequences of trans fatty acids: Introduction. Eur. J. Clin. Nutr. 2009, 63, S1–S4. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, S.; Passi, S.J.; Misra, A. Overview of trans fatty acids: Biochemistry and health effects. Diabetes Metab. Syndr. 2011, 5, 161–164. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Katan, M.B.; Ascherio, A.; Stampfer, M.J.; Willett, W.C. Trans Fatty Acids and Cardiovascular Disease. N. Engl. J. Med. 2006, 354, 1601–1613. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Aro, A.; Willett, W.C. Health effects of trans-fatty acids: Experimental and observational evidence. Eur. J. Clin. Nutr. 2009, 63 (Suppl. S2), S5–S21. [Google Scholar] [CrossRef] [PubMed]
- Thompson, A.K.; Minihane, A.-M.; Williams, C.M. Trans fatty acids and weight gain. Int. J. Obes. 2011, 35, 315–324. [Google Scholar] [CrossRef] [PubMed]
- De Souza, R.J.; Mente, A.; Maroleanu, A.; Cozma, A.I.; Ha, V.; Kishibe, T.; Uleryk, E.; Budylowski, P.; Schünemann, H.; Beyene, J.; et al. Intake of saturated and trans unsaturated fatty acids and risk of all cause mortality, cardiovascular disease, and type 2 diabetes: Systematic review and meta-analysis of observational studies. BMJ 2015, 351, h3978. [Google Scholar] [CrossRef] [PubMed]
- Kleber, M.E.; Delgado, G.E.; Lorkowski, S.; März, W.; von Schacky, C. Trans fatty acids and mortality in patients referred for coronary angiography: The Ludwigshafen Risk and Cardiovascular Health Study. Eur. Heart J. 2016, 37, 1072–1078. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, I.A.; Wanders, A.J.; Katan, M.B. Trans fatty acids and cardiovascular health: Research completed? Eur. J. Clin. Nutr. 2013, 67, 541–547. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Fats and Fatty Acids in Human Nutrition. Available online: http://www.who.int/nutrition/publications/nutrientrequirements/fatsandfattyacids_humannutrition/en/ (accessed on 16 March 2016).
- Mulder, K.A.; Ferdinands, A.R.; Richardson, K.J.; Innis, S.M. Sources of trans and saturated fatty acids in the diets of Vancouver children. Can. J. Diet. Pract. Res. 2013, 74, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Innis, S.M. Trans fatty intakes during pregnancy, infancy and early childhood. Atheroscler. Suppl. 2006, 7, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Bauer, L.R.; Waldrop, J. Trans fat intake in children: Risks and recommendations. Pediatr. Nurs. 2009, 35, 346–351. [Google Scholar] [PubMed]
- Guxens, M.; Ballester, F.; Espada, M.; Fernández, M.F.; Grimalt, J.O.; Ibarluzea, J.; Olea, N.; Rebagliato, M.; Tardón, A.; Torrent, M.; et al. INMA Project Cohort Profile: The INMA—INfancia y Medio Ambiente—(Environment and Childhood) Project. Int. J. Epidemiol. 2012, 41, 930–940. [Google Scholar] [CrossRef] [PubMed]
- Vioque, J.; Navarrete-Muñoz, E.-M.; Gimenez-Monzó, D.; García-de-la-Hera, M.; Granado, F.; Young, I.S.; Ramón, R.; Ballester, F.; Murcia, M.; Rebagliato, M.; et al. INMA-Valencia Cohort Study Reproducibility and validity of a food frequency questionnaire among pregnant women in a Mediterranean area. Nutr. J. 2013, 12, 26. [Google Scholar] [CrossRef] [PubMed]
- Vioque, J.; Gimenez-Monzó, D.; Navarrete-Muñoz, E.M.; García-de-la-Hera, M.; Gonzalez-Palacios, S.; Rebagliato, M.; Ballester, F.; Murcia, M.; Iñiguez, C.; Granado, F. Reproducibility and validity of a Food Frequency Questionnaire designed to assess diet in children aged 4–5 years. PLoS ONE 2016, in press. [Google Scholar]
- Ortiz-Andrellucchi, A.; Henríquez-Sánchez, P.; Sánchez-Villegas, A.; Peña-Quintana, L.; Mendez, M.; Serra-Majem, L. Dietary assessment methods for micronutrient intake in infants, children and adolescents: A systematic review. Br. J. Nutr. 2009, 102 (Suppl. S1), S87–S117. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Agriculture: Agricultural Research Service. USDA National Nutrient Database for Standard Reference, Release 27; U.S. Department of Agriculture: Washington, DC, USA, 2015.
- Leth, T.; Ovesen, L.; Hansen, K. Fatty acid composition of meat from ruminants, with special emphasis on trans fatty acids. J. Am. Oil Chem. Soc. 1998, 75, 1001–1005. [Google Scholar]
- Larqué, E.; Garaulet, M.; Pérez-Llamas, F.; Zamora, S.; Tebar, F.J. Fatty acid composition and nutritional relevance of most widely consumed margarines in Spain. Grasas Aceites 2003, 54, 65–70. [Google Scholar]
- Vicario, I.M.; Griguol, V.; León-Camacho, M. Multivariate characterization of the fatty acid profile of Spanish cookies and bakery products. J. Agric. Food Chem. 2003, 51, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Fernández-San Juan, P.M. Trans fatty acids (TFA): Sources and intake levels, biological effects and content in commercial Spanish food. Nutr. Hosp. 2009, 24, 515–520. [Google Scholar] [PubMed]
- Kuhnt, K.; Baehr, M.; Rohrer, C.; Jahreis, G. Trans fatty acid isomers and the trans-9/trans-11 index in fat containing foods. Eur. J. Lipid Sci. Technol. 2011, 113, 1281–1292. [Google Scholar] [CrossRef] [PubMed]
- Willett, W. Nutritional Epidemiology; Oxford University Press: New York, NY, USA, 2013. [Google Scholar]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.D.; Li, J.W.; Liu, Z.P.; Zhou, P.P.; Mao, W.F.; Li, N.; Zhang, L. Trans Fatty Acid Levels in Foods and Intakes among Population Aged 3 Years and above in Beijing and Guangzhou Cities, China. Biomed. Environ. Sci. 2015, 28, 477–485. [Google Scholar] [PubMed]
- Matheson, D.M.; Killen, J.D.; Wang, Y.; Varady, A.; Robinson, T.N. Children’s food consumption during television viewing. Am. J. Clin. Nutr. 2004, 79, 1088–1094. [Google Scholar] [PubMed]
- Miller, S.A.; Taveras, E.M.; Rifas-Shiman, S.L.; Gillman, M.W. Association between television viewing and poor diet quality in young children. Int. J. Pediatr. Obes. 2008, 3, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Utter, J.; Scragg, R.; Schaaf, D. Associations between television viewing and consumption of commonly advertised foods among New Zealand children and young adolescents. Public Health Nutr. 2006, 9, 606–612. [Google Scholar] [CrossRef] [PubMed]
- Agudo, A.; Pera, G. Vegetable and fruit consumption associated with anthropometric, dietary and lifestyle factors in Spain. EPIC Group of Spain. European Prospective Investigation into Cancer. Public Health Nutr. 1999, 2, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Dennison, B.A.; Rockwell, H.L.; Baker, S.L. Fruit and vegetable intake in young children. J. Am. Coll. Nutr. 1998, 17, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Keller, K.L.; Kirzner, J.; Pietrobelli, A.; St-Onge, M.-P.; Faith, M.S. Increased sweetened beverage intake is associated with reduced milk and calcium intake in 3- to 7-year-old children at multi-item laboratory lunches. J. Am. Diet. Assoc. 2009, 109, 497–501. [Google Scholar] [CrossRef] [PubMed]
- Chisaguano, A.M.; Montes, R.; Castellote, A.I.; Morales, E.; Júlvez, J.; Vioque, J.; Sunyer, J.; López-Sabater, M.C. Elaidic, vaccenic, and rumenic acid status during pregnancy: Association with maternal plasmatic LC-PUFAs and atopic manifestations in infants. Pediatr. Res. 2014, 76, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Garland, M.; Sacks, F.M.; Colditz, G.A.; Rimm, E.B.; Sampson, L.A.; Willett, W.C.; Hunter, D.J. The relation between dietary intake and adipose tissue composition of selected fatty acids in US women. Am. J. Clin. Nutr. 1998, 67, 25–30. [Google Scholar] [PubMed]
- Willett, W.; Mozaffarian, D. Ruminant or industrial sources of trans fatty acids: Public health issue or food label skirmish? Am. J. Clin. Nutr. 2008, 87, 515–516. [Google Scholar] [PubMed]
| All Cohorts (n = 1793) | Asturias (n = 387) | Guipuzcoa (n = 395) | Sabadell (n = 429) | Valencia (n = 582) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| g/day | E% 1 | g/day | E% 1 | g/day | E% 1 | g/day | E% 1 | g/day | E% 1 | |
| Total TFA intake | ||||||||||
| Mean 2 | 1.36 | 0.77 | 1.40 | 0.76 | 1.22 | 0.74 | 1.35 | 0.74 | 1.43 | 0.81 |
| S.D. 2 | 0.45 | 0.17 | 0.47 | 0.16 | 0.38 | 0.17 | 0.41 | 0.17 | 0.50 | 0.18 |
| Percentile 3 | ||||||||||
| P10 | 0.85 | 0.56 | 0.89 | 0.56 | 0.77 | 0.54 | 0.89 | 0.56 | 0.86 | 0.59 |
| P25 | 1.03 | 0.65 | 1.08 | 0.65 | 0.93 | 0.62 | 1.07 | 0.63 | 1.06 | 0.68 |
| P50 | 1.30 | 0.75 | 1.32 | 0.74 | 1.17 | 0.74 | 1.29 | 0.73 | 1.38 | 0.80 |
| P75 | 1.60 | 0.87 | 1.59 | 0.86 | 1.46 | 0.85 | 1.60 | 0.83 | 1.73 | 0.92 |
| P90 | 1.95 | 1.00 | 1.97 | 0.99 | 1.67 | 0.98 | 1.90 | 0.94 | 2.11 | 1.05 |
| Industrial TFA intake | ||||||||||
| Mean 2 | 0.60 | 0.34 | 0.54 | 0.30 | 0.54 | 0.33 | 0.63 | 0.35 | 0.65 | 0.37 |
| S.D. 2 | 0.27 | 0.13 | 0.26 | 0.11 | 0.21 | 0.11 | 0.27 | 0.13 | 0.30 | 0.13 |
| Percentile 3 | ||||||||||
| P10 | 0.31 | 0.20 | 0.28 | 0.17 | 0.31 | 0.21 | 0.35 | 0.21 | 0.33 | 0.21 |
| P25 | 0.42 | 0.26 | 0.38 | 0.22 | 0.39 | 0.25 | 0.45 | 0.26 | 0.45 | 0.28 |
| P50 | 0.55 | 0.32 | 0.49 | 0.29 | 0.50 | 0.32 | 0.57 | 0.33 | 0.59 | 0.35 |
| P75 | 0.73 | 0.40 | 0.64 | 0.35 | 0.66 | 0.38 | 0.76 | 0.41 | 0.81 | 0.44 |
| P90 | 0.94 | 0.50 | 0.85 | 0.43 | 0.81 | 0.46 | 0.98 | 0.52 | 1.06 | 0.55 |
| Natural TFA intake | ||||||||||
| Mean 2 | 0.71 | 0.40 | 0.82 | 0.45 | 0.64 | 0.39 | 0.65 | 0.36 | 0.72 | 0.41 |
| S.D. 2 | 0.28 | 0.12 | 0.31 | 0.13 | 0.26 | 0.12 | 0.23 | 0.10 | 0.28 | 0.11 |
| Percentile 3 | ||||||||||
| P10 | 0.40 | 0.26 | 0.48 | 0.29 | 0.36 | 0.23 | 0.40 | 0.23 | 0.41 | 0.27 |
| P25 | 0.51 | 0.32 | 0.59 | 0.35 | 0.46 | 0.30 | 0.49 | 0.29 | 0.52 | 0.34 |
| P50 | 0.67 | 0.39 | 0.80 | 0.43 | 0.60 | 0.38 | 0.62 | 0.35 | 0.70 | 0.40 |
| P75 | 0.86 | 0.47 | 0.97 | 0.53 | 0.76 | 0.46 | 0.79 | 0.43 | 0.88 | 0.48 |
| P90 | 1.10 | 0.56 | 1.21 | 0.62 | 0.97 | 0.55 | 0.95 | 0.49 | 1.11 | 0.56 |
| Total TFA (mg/Day) | Industrial TFA (mg/Day) | Natural TFA (mg/Day) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| β 1 | 95% CI | I2, % 2 | β 1 | 95% CI | I2, % 2 | β 1 | 95% CI | I2, % 2 | |
| Parental characteristics | |||||||||
| Maternal age, years | −1.6 | −11.8; 8.6 | 81.3 3 | 0.7 | −4.4; 5.8 | 57.8 | −2.2 | −7.4; 2.9 | 64.7 3 |
| Paternal age, years | −0.6 | −4.2; 3.1 | 59.6 | −0.5 | −3.2; 2.3 | 0.0 | −0.3 | −6.1; 5.5 | 81.0 3 |
| Country of origin other than Spain | 45.5 | 1.3; 89.6 | 0.0 | −13.6 | −47.8; 20.5 | 0.0 | 65.7 | 37.4; 94.0 | 0.0 |
| Mother’s educational level | |||||||||
| Primary or no education | ref. | ref. | ref. | ref. | ref. | ref. | |||
| Secondary | −11.0 | −48.1; 26.0 | 25.6 | −1.5 | −42.9; 40.0 | 53.0 | −17.6 | −42.1; 6.9 | 25.9 |
| University | −8.0 | −82.6; 66.6 | 61.3 | 15.3 | −49.7; 19.2 | 35.2 | 13.9 | −16.8; 44.6 | 44.0 |
| Mother’s social class | |||||||||
| Low | ref | ref | ref | ref | ref | ref | |||
| Middle | 11.1 | −28.2; 50.3 | 0.0 | 0.4 | −28.1; 29.6 | 26.3 | 5.0 | −21.9; 31.9 | 0.0 |
| High | −1.1 | −43.0; 40.8 | 31.2 | −20.4 | −51.5; 10.7 | 20.4 | 16.0 | −12.7; 44.7 | 0.0 |
| Child’s characteristics | |||||||||
| Physical activity (parentally reported) | |||||||||
| Sedentary | ref. | ref. | ref. | ref. | ref. | ref. | |||
| Active | −1.0 | −28.7; 26.6 | 0.0 | −16.4 | −37.0; 4.2 | 0.0 | 12.2 | −6.5; 30.9 | 0.0 |
| TV viewing, h/day | 16.0 | −0.5; 32.6 | 39.9 | 18.3 | 5.9; 30.7 | 0.0 | −2.9 | −13.9; 8.1 | 22.4 |
| Fruit and vegetable intake, 100 g/day | −85.5 | −104.8; −66.1 | 63.6 3 | −46.6 | −61.9; −31.3 | 67.7 3 | −36.9 | −44.9; −28.9 | 40.3 |
| Sweetened beverage intake, 100 g/day | −27.6 | −41.1; −14.0 | 0.0 | 8.5 | −1.7; 18.7 | 0.0 | −34.8 | −43.6; −26.0 | 0.0 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scholz, A.; Gimenez-Monzo, D.; Navarrete-Muñoz, E.M.; Garcia-de-la-Hera, M.; Fernandez-Somoano, A.; Tardon, A.; Santa Marina, L.; Irazabal, A.; Romaguera, D.; Guxens, M.; et al. Dietary Intake of Trans Fatty Acids in Children Aged 4–5 in Spain: The INMA Cohort Study. Nutrients 2016, 8, 625. https://doi.org/10.3390/nu8100625
Scholz A, Gimenez-Monzo D, Navarrete-Muñoz EM, Garcia-de-la-Hera M, Fernandez-Somoano A, Tardon A, Santa Marina L, Irazabal A, Romaguera D, Guxens M, et al. Dietary Intake of Trans Fatty Acids in Children Aged 4–5 in Spain: The INMA Cohort Study. Nutrients. 2016; 8(10):625. https://doi.org/10.3390/nu8100625
Chicago/Turabian StyleScholz, Alexander, Daniel Gimenez-Monzo, Eva Maria Navarrete-Muñoz, Manuela Garcia-de-la-Hera, Ana Fernandez-Somoano, Adonina Tardon, Loreto Santa Marina, Amaia Irazabal, Dora Romaguera, Mònica Guxens, and et al. 2016. "Dietary Intake of Trans Fatty Acids in Children Aged 4–5 in Spain: The INMA Cohort Study" Nutrients 8, no. 10: 625. https://doi.org/10.3390/nu8100625
APA StyleScholz, A., Gimenez-Monzo, D., Navarrete-Muñoz, E. M., Garcia-de-la-Hera, M., Fernandez-Somoano, A., Tardon, A., Santa Marina, L., Irazabal, A., Romaguera, D., Guxens, M., Julvez, J., Llop, S., Lopez-Espinosa, M.-J., & Vioque, J. (2016). Dietary Intake of Trans Fatty Acids in Children Aged 4–5 in Spain: The INMA Cohort Study. Nutrients, 8(10), 625. https://doi.org/10.3390/nu8100625








