The Long Term Impact of Micronutrient Supplementation during Infancy on Cognition and Executive Function Performance in Pre-School Children
Abstract
:1. Introduction
2. Methods
2.1. Study Design
2.2. Participant Recruitment
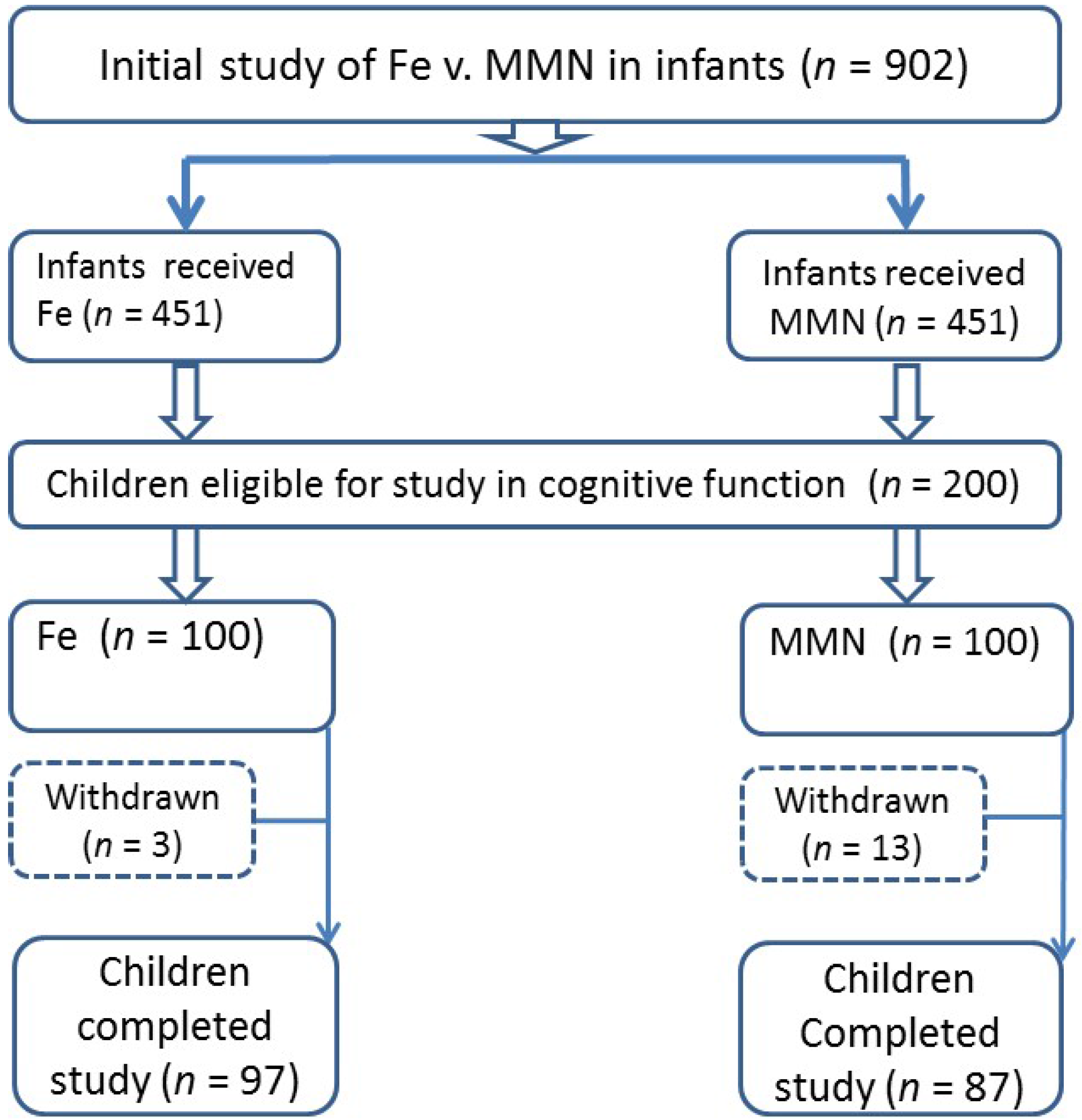
2.3. Study Methodology
2.4. Test Procedure for Executive Function Cognitive Outcome
2.4.1. Day-Night Stroop Test
2.4.2. Nine Boxes
2.4.3. Theory of Mind
2.4.4. BITSEA
2.5. Test Procedure for Intelligence Testing
WPPSI
2.6. Statistical Analysis Plan
3. Results
3.1. Cognitive Tests
3.1.1. Day-Night Stroop Test
| Variable | Fe (n = 97) | MMN (n = 87) | p value | ||||
|---|---|---|---|---|---|---|---|
| ± SD | Min. | Max. | ± SD | Min. | Max. | ||
| Age (months) | |||||||
| All children | 41.55 ± 3.16 | 36.07 | 47.87 | 41.40 ± 3.23 | 36.03 | 47.93 | 0.22 |
| Boys | 41.65 ± 3.13 | 36.07 | 47.87 | 40.99 ± 3.04 | 36.03 | 47.38 | |
| Girls | 41.42 ± 3.23 | 36.95 | 47.15 | 41.92 ± 3.42 | 36.36 | 47.93 | |
| Weight (kg) | |||||||
| All children | 15.49 ± 2.14 | 11.5 | 22.5 | 15.41 ± 2.01 | 12 | 21.6 | 0.60 |
| Boys | 15.59 ± 2.02 | 12.4 | 21.7 | 15.71 ± 1.98 | 12 | 21.6 | |
| Girls | 15.37 ± 2.29 | 11.5 | 22.5 | 15.02 ±2.02 | 12.4 | 19.7 | |
| Height (cm) | |||||||
| All children | 97.43 ± 3.64 | 90.8 | 107.5 | 97.16 ± 3.94 | 89 | 105.2 | 0.66 |
| Boys | 97.9 ± 3.45 | 91.3 | 105.2 | 97.49 ± 3.83 | 91 | 105.2 | |
| Girls | 96.85 ± 3.81 | 90.8 | 107.5 | 96.74 ± 4.09 | 89 | 104.2 | |
| Fe | MMN | p value | Eta squared η2 | Cohen’s d | Effect size r | |
|---|---|---|---|---|---|---|
| ± SD | ± SD | |||||
| Longest correct run for Day-Night test † | ||||||
| All children | 7.75 ± 4.79 | 7.98 ± 4.96 | 0.73 | 0.0001 | −0.02 | −0.009 |
| Boys | 6.76 ± 4.35 * | 6.81 ± 4.39 * | 0.01 * | 1.04 | ||
| Girls | 9 ± 5.12 * | 9.63 ± 5.37 * | ||||
| Longest correct run for nine boxes test | ||||||
| All children | 4.46 ± 2.05 | 4.77 ± 2.20 | 0.26 | 0.007 | −0.15 | −0.07 |
| Boys | 4.64 ± 2.11 | 4.57 ± 2.38 | 0.92 | 0.00006 | ||
| Girls | 4.25 ± 1.89 | 5.03 ± 1.95 | ||||
| Aggregate score for the Theory of Mind (false belief task) | ||||||
| All children | 0.20 ± 0.49 | 0.25 ± 0.55 | 0.38 | 0.004 | −0.12 | −0.06 |
| Boys | 0.21 ± 0.49 | 0.18 ± 0.49 | 0.39 | 0.004 | ||
| Girls | 0.18 ± 0.50 | 0.34 ± 0.63 | ||||
3.1.2. Nine Boxes
3.1.3. Theory of Mind (False Belief Task)
3.2. Social Emotional Functioning
| BITSEA | Fe (n = 97 ) | MMN(n = 87) | p value | Eta squared η2 | Cohen’s d | Effect size r | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| ± SD | Min. | Max. | ± SD | Min.Max. | ||||||
| Problem | ||||||||||
| All children | 14.13 ± 5.99 | 3 | 33 | 15.17 ± 6.31 | 4 | 33 | 0.21 | 0.008 | −0.26 | −0.12 |
| Boys | 15.70 ± 5.85 * | 5 | 33 | 15.69 ± 6.78 * | 4 | 33 | 0.01 | 0.035 | ||
| Girls | 12.25 ± 5.65 | 3 | 29 | 14.50 ± 5.67 | 4 | 28 | ||||
| Competence | ||||||||||
| All children | 16.37 ± 3.22 | 7 | 22 | 16.45 ± 2.54 | 9 | 22 | 0.80 | 0.0004 | −0.03 | −0.014 |
| Boys | 15.79 ± 2.99 | 7 | 21 | 15.82 ± 2.63 | 9 | 22 | 0.05 | |||
| Girls | 17.07 ± 3.37 * | 8 | 22 | 17.26 ± 2.19 * | 13 | 22 | <0.01 | |||
WPPSI
| Fe(n = 93) | MMN(n = 81) | p value | Maximum points (Number of questions) | |||
|---|---|---|---|---|---|---|
| ± SD | ± SD | |||||
| Boys (n = 52) | Girls (n = 41) | Boys (n = 44) | Girls (n = 37) | |||
| Executive subtests | ||||||
| Object assembly | 8.10 ± 3.27 | 7.78 ± 3.30 | 8.09 ± 3.24 | 8.49 ± 2.99 | 0.47 | 32 (6) |
| Picture Completion | 9.06 ± 2.82 | 8.90 ± 2.62 | 9.25 ± 2.54 | 9.14 ± 2.57 | 0.96 | 28 (28) |
| Block design | 10.06± 2.49 | 9.88 ± 2.26 | 9.98 ± 2.67 | 10.54 ± 2.82 | 0.34 | 42 (14) |
| Animal Peg | 9.17 ± 3.20 | 9.66 ± 3.34 | 9.41 ± 3.24 | 9.84 ± 3.43 | 0.96 | 70 (28 pegs) |
| Verbal subtests | ||||||
| Comprehension | 6.78 ± 1.90 | 6.80 ± 1.52 | 6.95 ± 1.68 | 6.75 ± 1.92 | 0.69 | 30 (15) |
| Vocabulary | 5.88 ± 1.86 | 5.78 ± 1.59 ** | 5.68 ± 1.85 | 6.83 ± 2.05 ** | 0.03 ** | 47 (25) |
| Similarities | 7.41 ± 2.69 | 7.72 ± 2.43 | 7.43 ± 2.50 | 7.86 ± 2.95 | 0.89 | 28 (12) |
| Arithmetic | 8.57 ± 2.27 | 8.08 ± 2.60 | 7.75 ± 2.63 | 7.97 ± 2.70 | 0.36 | 23 (23) |
| Sentences | 6.82 ± 2.67 | 7.95 ± 2.95 * | 7.20 ± 2.48 | 7.83 ± 2.87 * | 0.04 * | 37 (12) |
| Fe (n = 93) | MMN (n = 81) | Mean difference | p value | |
|---|---|---|---|---|
| ± SD | ± SD | |||
| Executive IQ | ||||
| All children | 93.95 ± 14.80 | 94.48 ± 16.75 | 0.53 | 0.87 |
| Boys | 93.67 ± 14.65 | 95.48 ±13.78 | 1.81 | |
| Girls | 94.29 ±15.16 | 93.30 ± 19.86 | 0.99 | |
| Verbal IQ | ||||
| All children | 83.31 ± 9.86 | 83.38 ± 11.09 | 0.07 | 0.92 |
| Boys | 83.12 ± 10.30 | 82.23 ± 10.31 | 0.89 | |
| Girls | 83.56 ± 9.40 | 84.76 ± 11.94 | 1.2 | |
| Total IQ score | ||||
| All children | 86.61 ± 12.23 | 87.90 ± 12.91 | 1.29 | 0.48 |
| Boys | 86.37 ± 12.02 | 86.73 ± 12.15 | 0.36 | |
| Girls | 86.93 ± 12.64 | 89.30 ± 13.79 | 2.37 | |
4. Discussion
5 Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| BITSEA | Brief Infant Toddler Social Emotional Assessment |
| BITSEA/P | BITSEA Problem total score |
| BITSEA/C | BITSEA Competence total score |
| Fe | Iron supplemented group |
| IQ | Intelligence Quotient |
| IIN | Instituto de Investigación Nutricional |
| MMN | Multiple micronutrient (including zinc) supplemented group |
| RCT | Randomized control trial |
| RDA | Recommended Daily Allowance |
| SD | Standard Deviation |
| UCLAN | University of Central Lancashire |
| UNICEF | United Nations Children’s Fund |
| WPPSI | Wechsler Preschool and Primary Scale of Intelligence |
| WPPSI -R | Wechsler Preschool and Primary Scale of Intelligence Revised |
References
- Morgane, P.J.; Mokler, D.J.; Galler, J.R. Effects of prenatal protein malnutrition on the hippocampal formation. Neurosci. Biobehav. Rev. 2002, 26, 471–483. [Google Scholar] [CrossRef]
- Pollitt, E.; Gorman, K.S.; Engle, P.L.; Martorell, R.; Rivera, J. Early supplementary feeding and cognition: Effects over two decades. Monogr. Soc. Res. Child. Dev. 1993, 58, 1–99. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.L.; Pollitt, E. Malnutrition, poverty and intellectual development. Sci. Am. 1996, 274, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Black, M.M. Micronutrient deficiencies and cognitive functioning. J. Nutr. 2003, 133, 3927S–3931S. [Google Scholar] [PubMed]
- Olson, C.R.; Mello, C.V. Significance of vitamin a to brain function, behavior and learning. Mol. Nutr. Food Res. 2010, 54, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Cocco, S.; Diaz, G.; Stancampiano, R.; Diana, A.; Carta, M.; Curreli, R.; Sarais, L.; Fadd, F. Vitamin a deficiency produces spatial learning and memory impairment in rats. Neuroscience 2002, 115, 475–482. [Google Scholar] [CrossRef]
- McGarel, C.; Pentieva, K.; Strain, J.J.; McNulty, H. Emerging roles for folate and related B-vitamins in brain health across the lifecycle. Proc. Nutr. Soc. 2015, 74, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Harrison, F.E.; May, J.M. Vitamin C function in the brain: Vital role of the ascorbate transporter SVCT2. Free Radic. Biol. Med. 2009, 46, 719–730. [Google Scholar] [CrossRef] [PubMed]
- Rosales, F.J.; Zeisel, S.H. Perspectives from the symposium: The role of nutrition in infant and toddler brain and behavioral development. Nutr. Neurosci. 2008, 11, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Bourre, J.M. Effects of nutrients (in food) on the structure and function of the nervous system: Update on dietary requirements for brain. Part 1: Micronutrients. J. Nutr. Health Aging 2006, 10, 377–385. [Google Scholar] [PubMed]
- Gómez-Pinilla, F. Brain foods: The effects of nutrients on brain function. Nat. Rev. Neurosci. 2008, 9, 568–578. [Google Scholar] [CrossRef] [PubMed]
- Muthayya, S.; Eilander, A.; Transler, C.; Thomas, T.; van der Knaap, H.C.; Srinivasan, K.; van Klinken, B.J.W.; Osendarp, S.J.; Kurpad, A.V. Effect of fortification with multiple micronutrients and n−3 fatty acids on growth and cognitive performance in indian schoolchildren: The CHAMPION (Children’s Health and Mental Performance Influenced by Optimal Nutrition) Study. Am. J. Clin. Nutr. 2009, 89, 1766–1775. [Google Scholar] [CrossRef] [PubMed]
- Eilander, A.; Gera, T.; Sachdev, H.S.; Transler, C.; van der Knaap, H.C.M.; Kok, F.J.; Osendarp, S.J.M. Multiple micronutrient supplementation for improving cognitive performance in children: Systematic review of randomized controlled trials. Am. J. Clin. Nutr. 2010, 91, 115–130. [Google Scholar] [CrossRef] [PubMed]
- Fischer Walker, C.; Kordas, K.; Stoltzfus, R.J.; Black, R.E. Interactive effects of iron and zinc on biochemical and functional outcomes in supplementation trials. Am. J. Clin. Nutr. 2005, 82, 5–12. [Google Scholar] [PubMed]
- Grantham-McGregor, S.M.; Fernald, L.C.; Sethuraman, K. Effects of health and nutrition on cognitive and behavioural development in children in the first three years of life. Part 1: Low birthweight, breastfeeding, and protein-energy malnutrition. Food Nutr. Bull. 1999, 20, 53–75. [Google Scholar]
- Levitsky, D.A.; Strupp, B.J. Malnutrition and the brain: Changing concepts, changing concerns. J. Nutr. 1995, 125, 2212S–2220S. [Google Scholar] [PubMed]
- Birberg-Thornberg, U.; Karlsson, T.; Gustafsson, P.A.; Duchen, K. Nutrition and theory of mind—The role of polyunsaturated fatty acids (PUFA) in the development of theory of mind. Prostaglandins Leukot. Essent. Fatty Acids 2006, 75, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Gogia, S.; Sachdev, H.S. Zinc supplementation for mental and motor development in children. Cochrane Database Syst. Rev. 2012, 12, CD007991. [Google Scholar] [PubMed]
- Grantham-McGregor, S.M.; Ani, C.C. The role of micronutrients in psychomotor and cognitive development. Br. Med. Bull. 1999, 55, 511–527. [Google Scholar] [CrossRef] [PubMed]
- Stoch, M.B.; Smythe, P.M. Does undernutrition during infancy inhibit brain growth and subsequent intellectual development? Arch. Dis. Child. 1963, 38, 546–552. [Google Scholar] [CrossRef] [PubMed]
- Rosales, F.J.; Reznick, J.S.; Zeisel, S.H. Understanding the role of nutrition in the brain and behavioral development of toddlers and preschool children: Identifying and addressing methodological barriers. Nutr. Neurosci. 2009, 12, 190–202. [Google Scholar] [CrossRef] [PubMed]
- Wachs, T.D. Relation of mild-to-moderate malnutrition to human development: Correlational studies. J. Nutr. 1995, 125, 2245S–2254S. [Google Scholar] [PubMed]
- Pollitt, E.; Saco-Pollitt, C.; Jahari, A.; Husaini, M.A.; Huang, J. Effects of an energy and micronutrient supplement on mental development and behavior under natural conditions in undernourished children in indonesia. Eur. J. Clin. Nutr. 2000, 54, S80–S90. [Google Scholar] [CrossRef] [PubMed]
- Grantham-McGregor, S.; Baker-Henningham, H. Review of the evidence linking protein and energy to mental development. Public Health Nutr. 2005, 8, 1191–1201. [Google Scholar] [CrossRef] [PubMed]
- Morgane, P.J.; Austin-LaFrance, R.; Bronzino, J.; Tonkiss, J.; Diaz-Cintra, S.; Cintra, L.; Kemper, T.; Galler, J.R. Prenatal malnutrition and development of the brain. Neurosci. Biobehav. Rev. 1993, 17, 91–128. [Google Scholar] [CrossRef]
- International zinc nutrition consultative group (IZiNCG). Quantifying the Risk of Zinc Deficiency: Recommended Indicators. Available online: http://www.Izincg.Org/files/english-brief1.Pdf (accessed on 3 August 2015).
- Instituto nacional de estadística e informatica (INEI). Series Nacionales. Available online: http://series.Inei.Gob.Pe:8080/sirtod-series/ (accessed on 3 August 2015).
- Sánchez-Abanto, J. Evolution of chronic malnutrition in children under five in Peru. Rev. Peru Med. Exp. Salud Publica 2012, 29, 402–405. [Google Scholar] [CrossRef] [PubMed]
- Munayco, C.V.; Ulloa-Rea, M.E.; Medina-Osis, J.; Lozano-Revollar, C.R.; Tejada, V.; Castro-Salazar, C.; Munarriz-Villafuerte, J.; de Bustos, C.; Arias, L. Evaluation of the impact of multiple micronutrient powders on children anemia in three andean regiones in Peru. Rev. Peru Med. Exp. Salud Publica 2013, 30, 229–234. [Google Scholar] [PubMed]
- Huamán-Espino, L.; Aparco, J.P.; Nuñez-Robles, E.; Gonzáles, E.; Pillaca, J.; Mayta-Tristán, P. Consumption of chispitas® multimicronutrient supplements and anemia in 6–35-month-old children: Cross-cut study in the context of a populational health intervention in apurimac, Peru. Rev. Peru Med. Exp. Salud Publica 2012, 29, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Zavaleta, N.; Loza, D.; Egoavil, P.; Sanchez, J.; Mosqueira, R.; Neufeld, L. Efficacy of different schemes of supplementation with micronutrient powders on anemia and micronutrient status in infants. FASEB J. 2013, 27, 36–38. [Google Scholar]
- Schauer, C.; Zlotkin, S. Home fortification with micronutrient sprinkles—A new approach for the prevention and treatment of nutritional anemias. Paediatr. Child Health 2003, 8, 87–90. [Google Scholar] [PubMed]
- Sharieff, W.; Bhutta, Z.; Schauer, C.; Tomlinson, G.; Zlotkin, S. Micronutrients (including zinc) reduce diarrhoea in children: The pakistan sprinkles diarrhoea study. Arch. Dis. Child. 2006, 91, 573–579. [Google Scholar] [CrossRef] [PubMed]
- ISRCTN. International standard randomised controlled trial number register (ISRCTN). Available online: http://www.Controlled-trials.Com/isrctn39244429 (accessed on 3 August 2015).
- Zavaleta, N.; Huasasquiche, C.; Callalli, J.; Garcia, T.; Caulfield, L. Changes in iron nutritional status during the first 6 months of life in infants born to iron supplemented mothers. Pediatr. Res. 2005, 57, 920. [Google Scholar] [CrossRef]
- López de Romaña, G.; Cusirramos, S.; López de Romaña, D.; Gross, R. Efficacy of multiple micronutrient supplementation for improving anemia, micronutrient status, growth, and morbidity of peruvian infants. J. Nutr. 2005, 135, 646S–652S. [Google Scholar] [PubMed]
- Iannotti, L.L.; Zavaleta, N.; Leon, Z.; Caulfield, L.E. Growth and body composition of peruvian infants in a periurban setting. Food Nutr. Bull. 2009, 30, 245–253. [Google Scholar] [PubMed]
- Caulfield, L.E.; Putnick, D.L.; Zavaleta, N.; Lazarte, F.; Albornoz, C.; Chen, P.; DiPietro, J.A.; Bornstein, M.H. Maternal gestational zinc supplementation does not influence multiple aspects of child development at 54 mo of age in Peru. Am. J. Clin. Nutr. 2010, 92, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Yang, H.; Li, L.; Chen, T.; Lynn, R. An increase of intelligence measured by the wppsi in China, 1984–2006. Intelligence 2012, 40, 139–144. [Google Scholar] [CrossRef]
- Kaufman, A.S. Essentials of WISC-III and WPPSI-R Assessment; Lichtenberger, E.O., Ed.; Wiley: New York, NY, USA, 2000. [Google Scholar]
- Caulfield, L.E.; Zavaleta, N.; Chen, P.; Lazarte, F.; Albornoz, C.; Putnick, D.L.; Bornstein, M.H.; DiPietro, J.A. Maternal zinc supplementation during pregnancy affects autonomic function of peruvian children assessed at 54 months of age. J. Nutr. 2011, 141, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Colombo, J.; Zavaleta, N.; Kannass, K.N.; Lazarte, F.; Albornoz, C.; Kapa, L.L.; Caulfield, L.E. Zinc supplementation sustained normative neurodevelopment in a randomized, controlled trial of peruvian infants aged 6–18 months. J. Nutr. 2014, 144, 1298–1305. [Google Scholar] [CrossRef] [PubMed]
- Gerstadt, C.L.; Hong, Y.J.; Diamond, A. The relationship between cognition and action: Performance of children 3½–7 years old on a stroop- like day-night test. Cognition 1994, 53, 129–153. [Google Scholar] [CrossRef]
- Carlson, S.M.; Moses, L.J. Individual differences in inhibitory control and children’s theory of mind. Child Dev. 2001, 72, 1032–1053. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.E.; Blake, P.R.; Harris, P.L. I should but I won’t: Why young children endorse norms of fair sharing but do not follow them. PLoS ONE 2013, 8, e59510. [Google Scholar] [CrossRef] [PubMed]
- Nigg, J.T. On inhibition/disinhibition in developmental psychopathology: Views from cognitive and personality psychology and a working inhibition taxonomy. Psychol. Bull. 2000, 126, 220–246. [Google Scholar] [CrossRef] [PubMed]
- Dowsett, S.M.; Livesey, D.J. The development of inhibitory control in preschool children: Effects of “executive skills” training. Dev. Psychobiol. 2000, 36, 161–174. [Google Scholar] [CrossRef]
- Diamond, A.; Prevor, M.B.; Callender, G.; Druin, D.P. Prefrontal cortex cognitive deficits in children treated early and continuously for PKU. Monogr. Soc. Res. Child Dev. 1997, 62, 1–205. [Google Scholar] [CrossRef]
- Fernald, L.C.; Kariger, P.; Engle, P.; Raikes, A. Examining Early Child Development in Low-income Countries; The World Bank: Washington, DC, USA, 2009. [Google Scholar]
- Kane, M.J.; Engle, R.W. The role of prefrontal cortex in working-memory capacity, executive attention, and general fluid intelligence: An individual-differences perspective. Psychon. Bull. Rev. 2002, 9, 637–671. [Google Scholar] [CrossRef] [PubMed]
- Seamon, J.G.; kenrick, D.T. Psychology; Prentice Hall: Englewood Cliffs, NJ, USA, 1994. [Google Scholar]
- Engle, R.W.; Tuholski, S.W.; Laughlin, J.E.; Conway, A.R. Working memory, short-term memory, and general fluid intelligence: A latent-variable approach. J. Exp. Psychol. Gen. 1999, 128, 309–331. [Google Scholar] [CrossRef] [PubMed]
- Gathercole, S.E.; Alloway, T.P. Working memory and classroom learning. Dyslexia Rev. 2004, 15, 4–9. [Google Scholar]
- Perner, J.; Lang, B. Development of theory of mind and executive control. Trends Cogn. Sci. 1999, 3, 337–344. [Google Scholar] [CrossRef]
- Wimmer, H.; Perner, J. Beliefs about beliefs—Representation and constraining function of wrong beliefs in young childrens understanding of deception. Cognition 1983, 13, 103–128. [Google Scholar] [CrossRef]
- Halford, G.S.; Andrews, G.; Bowden, D. Relational Complexity and Theory-of-mind. Available online: http://eric.ed.gov/?id=ED421673 (accessed on 3 August 2015).
- Flavell, J.H.; Flavell, E.R.; Green, F.L.; Moses, L.J. Young children’s understanding of fact beliefs versus value beliefs. Child Dev. 1990, 61, 915–928. [Google Scholar] [CrossRef] [PubMed]
- Briggs-Gowan, M.J.; Carter, A.S. Bitsea: Brief Infant-toddler Social and Emotional Assessment. Examiner’s Manual; Harcourt Assessment: San Diego, CA, USA, 2006. [Google Scholar]
- Briggs-Gowan, M.J.; Carter, A.S.; McCarthy, K.; Augustyn, M.; Caronna, E.; Clark, R. Clinical validity of a brief measure of early childhood social-emotional/behavioral problems. J. Pediatr. Psychol. 2013, 38, 577–587. [Google Scholar] [CrossRef] [PubMed]
- Briggs-Gowan, M.J.; Carter, A.S.; Skuban, E.M.; Horwitz, S.M. Prevalence of social-emotional and behavioral problems in a community sample of 1- and 2-year-old children. J. Am. Acad. Child Adolesc. Psychiatry 2001, 40, 811–819. [Google Scholar] [CrossRef] [PubMed]
- Carlson, S.M. Developmentally sensitive measures of executive function in preschool children. Dev. Neuropsychol. 2005, 28, 595–616. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, J.M.; Astington, J.W. Cognitive factors and family structure associated with theory of mind development in young children. Dev. Psychol. 1996, 32, 70–78. [Google Scholar] [CrossRef]
- Astington, J.W.; Jenkins, J.M. A longitudinal study of the relation between language and theory-of-mind development. Dev. Psychol. 1999, 35, 1311–1320. [Google Scholar] [CrossRef] [PubMed]
- Keceli Kaysili, B.; Acarlar, F. The development of theory of mind according to false belief performance of children ages 3 to 5. Educ. Sci. Theory Practice 2011, 11, 1821–1826. [Google Scholar]
- Bland, J.M.; Altman, D.G. Multiple significance tests: The bonferroni method. BMJ 1995, 310, 170. [Google Scholar] [CrossRef] [PubMed]
- Wellman, H.M.; Cross, D.; Watson, J. Meta-analysis of theory-of-mind development: The truth about false belief. Child Dev. 2001, 72, 655–684. [Google Scholar] [CrossRef] [PubMed]
- Briggs-Gowan, M.J.; Carter, A.S.; Irwin, J.R.; Wachtel, K.; Cicchetti, D.V. The brief infant-toddler social and emotional assessment: Screening for social-emotional problems and delays in competence. J. Pediatr. Psychol. 2004, 29, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Prettell, E.A.; Higa, A.M. Peru Celebrates 25 Years of Sustained Elimination of IDD. Available online: http://www.Ign.Org/newsletter/idd_nl_feb09_peru.Pdf (accessed on 3 August 2015).
- United Nations Children’s Fund (Unicef) and International Zinc Association (IZA), Unicef-IZA Partnership. Zinc Saves Kids (ZSK). Available online: http://zincsaveskids.Org/documents/zsk%20progress%20report%20peru%202014.Pdf (accessed on 3 August 2015).
- Ministry of health (MoH). Proporción de Niños de 6 Meses a 36 Meses Con Anemia—Año. 2014. Available online: http://www.App.Minsa.Gob.Pe/bsc/detalle_indbsc.Asp?Lcind=8&lcobj=2&lcper=1&lcfreg=10/4/2015 (accessed on 2 July 2015).
- Zavaleta, N. Instituto de Investigación Nutricional (IIN), lima, perú. Unpublished work. 2012. [Google Scholar]
- Taneja, S.; Bhandari, N.; Bahl, R.; Bhan, M.K. Impact of zinc supplementation on mental and psychomotor scores of children aged 12 to 18 months: A randomized, double-blind trial. J. Pediatr. 2005, 146, 506–511. [Google Scholar] [CrossRef] [PubMed]
- Murray-Kolb, L.E.; Khatry, S.K.; Katz, J.; Schaefer, B.A.; Cole, P.M.; Le Clerq, S.C.; Morgan, M.E.; Tielsch, J.M.; Christian, P. Preschool micronutrient supplementation effects on intellectual and motor function in school-aged nepalese children. Arch. Pediatr. Adolesc. Med. 2012, 166, 404–410. [Google Scholar] [PubMed]
- Christian, P.; Murray-Kolb, L.E.; Khatry, S.K.; Katz, J.; Schaefer, B.A.; Cole, P.M.; LeClerq, S.C.; Tielsch, J.M. Prenatal micronutrient supplementation and intellectual and motor function in early school-aged children in Nepal. JAMA 2010, 304, 2716–2723. [Google Scholar] [CrossRef] [PubMed]
- Recommended Dietary Allowances. Recommended Dietary Allowances; National Research Council-National Academy Press: Washington, DC, USA, 1989. [Google Scholar]
- Penland, J.G.; Sandstead, H.H.; Alcock, N.W.; Dayal, H.H.; Chen, X.C.; Li, J.S.; Zhao, F.J.; Yang, J.J. A preliminary report: Effects of zinc and micronutrient repletion on growth and neuropsychological function of urban chinese children. J. Am. Coll. Nutr. 1997, 16, 268–272. [Google Scholar] [CrossRef] [PubMed]
- Sandstead, H.H.; Penland, J.G.; Alcock, N.W.; Dayal, H.H.; Chen, X.C.; Li, J.S.; Zhao, F.; Yang, J.J. Effects of repletion with zinc and other micronutrients on neuropsychologic performance and growth of chinese children. Am. J. Clin. Nutr. 1998, 68, 470S–475S. [Google Scholar] [PubMed]
- Palejwala, M.H.; Fine, J.G. Gender differences in latent cognitive abilities in children aged 2 to 7. Intelligence 2015, 48, 96–108. [Google Scholar] [CrossRef]
- Quereshi, M.; Seitz, R. Gender differences on the WPPSI, the WISC-R, and the WPPSI-R. Curr. Psychol. 1994, 13, 117–123. [Google Scholar] [CrossRef]
- Liu, J.; Lynn, R. Factor structure and sex differences on the wechsler preschool and primary scale of intelligence in China, Japan and United States. Pers. Individ. Dif. 2011, 50, 1222–1226. [Google Scholar] [CrossRef] [PubMed]
- Carlson, S.M.; Moses, L.J.; Breton, C. How specific is the relation between executive function and theory of mind? Contributions of inhibitory control and working memory. Infant Child Dev. 2002, 11, 73–92. [Google Scholar] [CrossRef]
- Gale, C.R.; O’Callaghan, F.J.; Godfrey, K.M.; Law, C.M.; Martyn, C.N. Critical periods of brain growth and cognitive function in children. Brain 2004, 127, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Coulson, S.; Vitetta, L. Nutrients and the brain. J. Complement. Med. 2009, 8, 24–29. [Google Scholar]
- Warthon-Medina, M.; Moran, V.H.; Stammers, A.L.; Dillon, S.; Qualter, P.; Nissensohn, M.; Serra-Majem, L.; Lowe, N.M. Zinc intake, status and indices of cognitive function in adults and children: A systematic review and meta-analysis. Eur. J. Clin. Nutr. 2015, 69, 649–661. [Google Scholar] [CrossRef] [PubMed]
- Thatcher, R.W. Maturation of the human frontal lobes: Physiological evidence for staging. Dev. Neuropsychol. 1991, 7, 397–419. [Google Scholar] [CrossRef]
- Bryan, J.; Osendarp, S.; Hughes, D.; Calvaresi, E.; Baghurst, K.; Van Klinken, J.W. Nutrients for cognitive development in school-aged children. Nutr. Rev. 2004, 62, 295–306. [Google Scholar] [CrossRef] [PubMed]
- Georgieff, M.K. Nutrition and the developing brain: Nutrient priorities and measurement. Am. J. Clin. Nutr. 2007, 85, 614S–620S. [Google Scholar] [PubMed]
- Lenroot, R.K.; Giedd, J.N. Brain development in children and adolescents: Insights from anatomical magnetic resonance imaging. Neurosci. Biobehav. Rev. 2006, 30, 718–729. [Google Scholar] [CrossRef] [PubMed]
- Dekaban, A.S.; Sadowsky, D. Changes in brain weights during the span of human life: Relation of brain weights to body heights and body weights. Ann. Neurol. 1978, 4, 345–356. [Google Scholar] [CrossRef] [PubMed]
- Bailey, R.L.; West, K.P., Jr.; Black, R.E. The epidemiology of global micronutrient deficiencies. Ann. Nutr. Metab. 2015, 66, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Leung, B.M.Y.; Wiens, K.P.; Kaplan, B.J. Does prenatal micronutrient supplementation improve children’s mental development? A systematic review. BMC Pregnancy Childbirth 2011, 11. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Yan, H.; Zeng, L.; Cheng, Y.; Liang, W.; Dang, S.; Wang, Q.; Tsuji, I. Effects of maternal multimicronutrient supplementation on the mental development of infants in rural western china: Follow-up evaluation of a double-blind, randomized, controlled trial. Pediatrics 2009, 123, E685–E692. [Google Scholar] [CrossRef] [PubMed]
- Prado, E.L.; Alcock, K.J.; Muadz, H.; Ullman, M.T.; Shankar, A.H.; SUMMIT Study Group. Maternal multiple micronutrient supplements and child cognition: A randomized trial in Indonesia. Pediatrics 2012, 130, e536–e546. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, F.R. Minority and Cross-cultural Aspects of Neuropsychological Assessment; CRC Press: Boca Raton, FL, USA, 2002. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Warthon-Medina, M.; Qualter, P.; Zavaleta, N.; Dillon, S.; Lazarte, F.; Lowe, N.M. The Long Term Impact of Micronutrient Supplementation during Infancy on Cognition and Executive Function Performance in Pre-School Children. Nutrients 2015, 7, 6606-6627. https://doi.org/10.3390/nu7085302
Warthon-Medina M, Qualter P, Zavaleta N, Dillon S, Lazarte F, Lowe NM. The Long Term Impact of Micronutrient Supplementation during Infancy on Cognition and Executive Function Performance in Pre-School Children. Nutrients. 2015; 7(8):6606-6627. https://doi.org/10.3390/nu7085302
Chicago/Turabian StyleWarthon-Medina, Marisol, Pamela Qualter, Nelly Zavaleta, Stephanie Dillon, Fabiola Lazarte, and Nicola M. Lowe. 2015. "The Long Term Impact of Micronutrient Supplementation during Infancy on Cognition and Executive Function Performance in Pre-School Children" Nutrients 7, no. 8: 6606-6627. https://doi.org/10.3390/nu7085302
APA StyleWarthon-Medina, M., Qualter, P., Zavaleta, N., Dillon, S., Lazarte, F., & Lowe, N. M. (2015). The Long Term Impact of Micronutrient Supplementation during Infancy on Cognition and Executive Function Performance in Pre-School Children. Nutrients, 7(8), 6606-6627. https://doi.org/10.3390/nu7085302






