In Vivo Protective Effects of Diosgenin against Doxorubicin-Induced Cardiotoxicity
Abstract
:1. Introduction
2. Experimental Section
2.1. Chemicals
2.2. Animals and Experimental Protocol
2.3. Heart Rate, Blood Pressure Monitoring, and Blood Analysis
2.4. Measurement of Thiobarbituric Acid Relative Substances (TBARS), ROS and Antioxidant Status in Heart
2.5. Enzyme Immunoassay
2.6. Real-Time Polymerase Chain Reaction (Real-Time PCR) for mRNA Expression
| Gene | Primer | Sequence |
|---|---|---|
| PDE5A | Forward | 5′-AAATGGTGGGACCTTCACTG-3′ |
| Reverse | 5′-GTGGCCGCTATCTTCTTCAG-3′ | |
| PDE3A | Forward | 5′-AATGTGGCCGTATTCTGAGC-3′ |
| Reverse | 5′-GAATCGGCTGTGTTGTGAGA-3′ | |
| NF-κB | Forward | 5′-CAGACCGCAGTATCCATAGC-3′ |
| Reverse | 5′-CGTGAAAGGGGTTATTGTTGG-3′ | |
| TGF-β | Forward | 5′-TGACGTCACTGGAGTTGTACGG-3′ |
| Reverse | 5′-GGTTCATGTCATGGATGGTGC-3′ | |
| GADPH | Forward | 5′-TGTGTCCGTCGTGGATCTGA-3′ |
| Reverse | 5′-TTGCTGTTGAAGTCGCAGGAG-3′ |
2.7. Protein Preparation and Western Blot Analysis
2.8. Isolation of Mitochondria and Cytochrome C Assay
2.9. Statistical Analysis
3. Results
3.1. Diosgenin Diminished DOX-Induced Losses of Heart Weight, the Ratio of Heart Weight to Body Weight, and Heart Rate, and Attenuated DOX-Increased Blood Pressure, and Serum Levels of Cardiotoxicity Markers
| Normal | DOX | DOX + Diosgenin | |
|---|---|---|---|
| Body weight (g) | 28.00 ± 0.00 b | 20.77 ± 3.64 a | 21.73 ± 2.84 a |
| Heart weight (mg) | 166.43 ± 15.82 b | 131.78 ± 21.74 a | 148.05 ± 15.51 ab |
| Heart weight/body weight ratio | 5.94 ± 0.57 b | 4.66 ± 1.05 a | 5.83 ± 0.75 b |
| Heart rate (bpm) # | 611.40 ± 15.65 b | 566.31 ± 27.36 a | 595.59 ± 32.83 ab |
| Blood pressure (mmHg) | 98.08 ± 11.40 a | 119.93 ± 26.25 b | 98.88 ± 15.49 a |
| Serum LDH (U/L) | 173.02 ± 8.70 a | 263.91 ± 54.35 b | 152.13 ± 29.25 a |
| Serum CPK (U/L) | 106.31 ± 17.06 a | 241.22 ± 18.77 b | 165.32 ± 16.02 ab |
| Serum CK-MB (U/L) | 86.90 ± 6.72 a | 154.28 ± 15.03 b | 107.15 ± 10.33 ab |
3.2. Diosgenin Restored DOX-Induced Alterations in Oxidative Status
| Normal | DOX | DOX + Diosgenin | |
|---|---|---|---|
| TBARS (nmol/mg protein) | 0.16 ± 0.02 a | 0.27 ± 0.07 b | 0.17 ± 0.04 a |
| ROS (RFU/mg protein) | 1.02 ± 0.16 a | 1.51 ± 0.50 b | 1.05 ± 0.16 a |
| GSH (nmol/mg protein) | 5.97 ± 0.28 b | 2.36 ± 0.99 a | 4.43 ± 1.37 ab |
| GPx (U/mg protein) | 43.79 ± 0.73 b | 27.52 ± 3.42 a | 40.19 ± 4.75 b |
| SOD (U/mg protein) | 4.29 ± 0.64 b | 2.90 ± 0.92 a | 3.94 ± 0.51 ab |
3.3. Diosgenin Improved DOX-Regulated Levels of cAMP and cGMP, and mRNA Expression of PDE5A

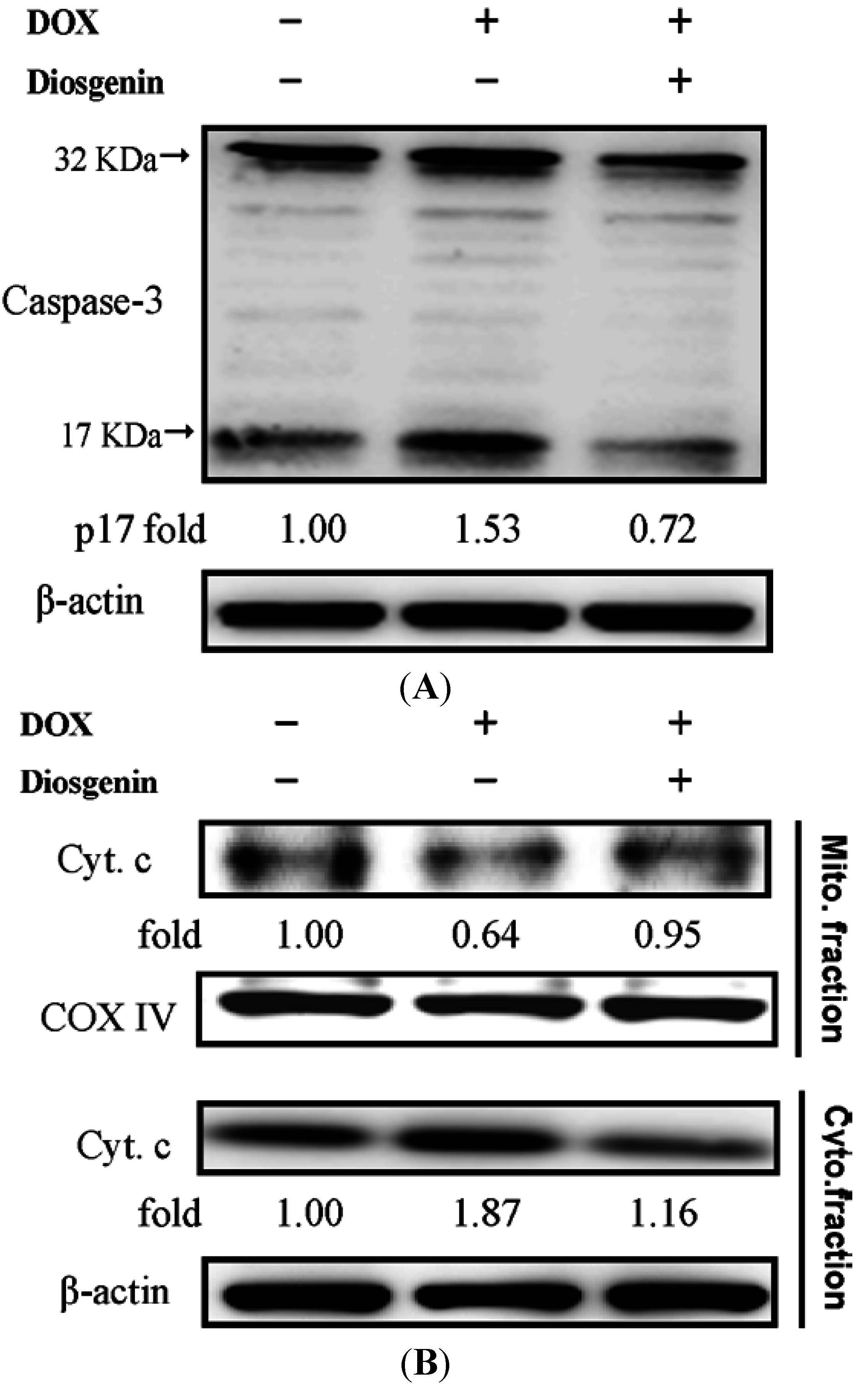
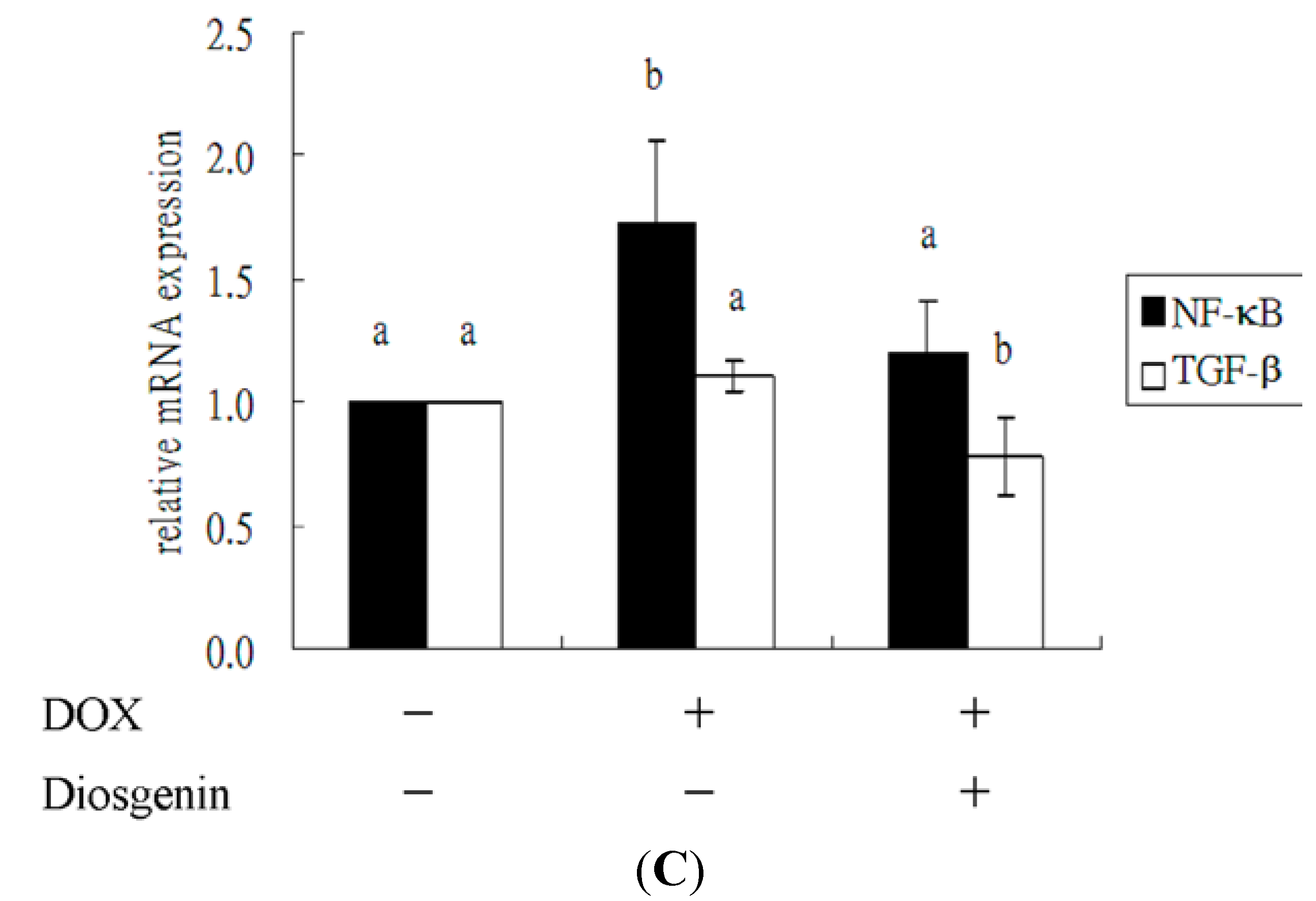
3.4. Diosgenin Weakened DOX-Induced Caspase-3 Activation and NF-κB Expression
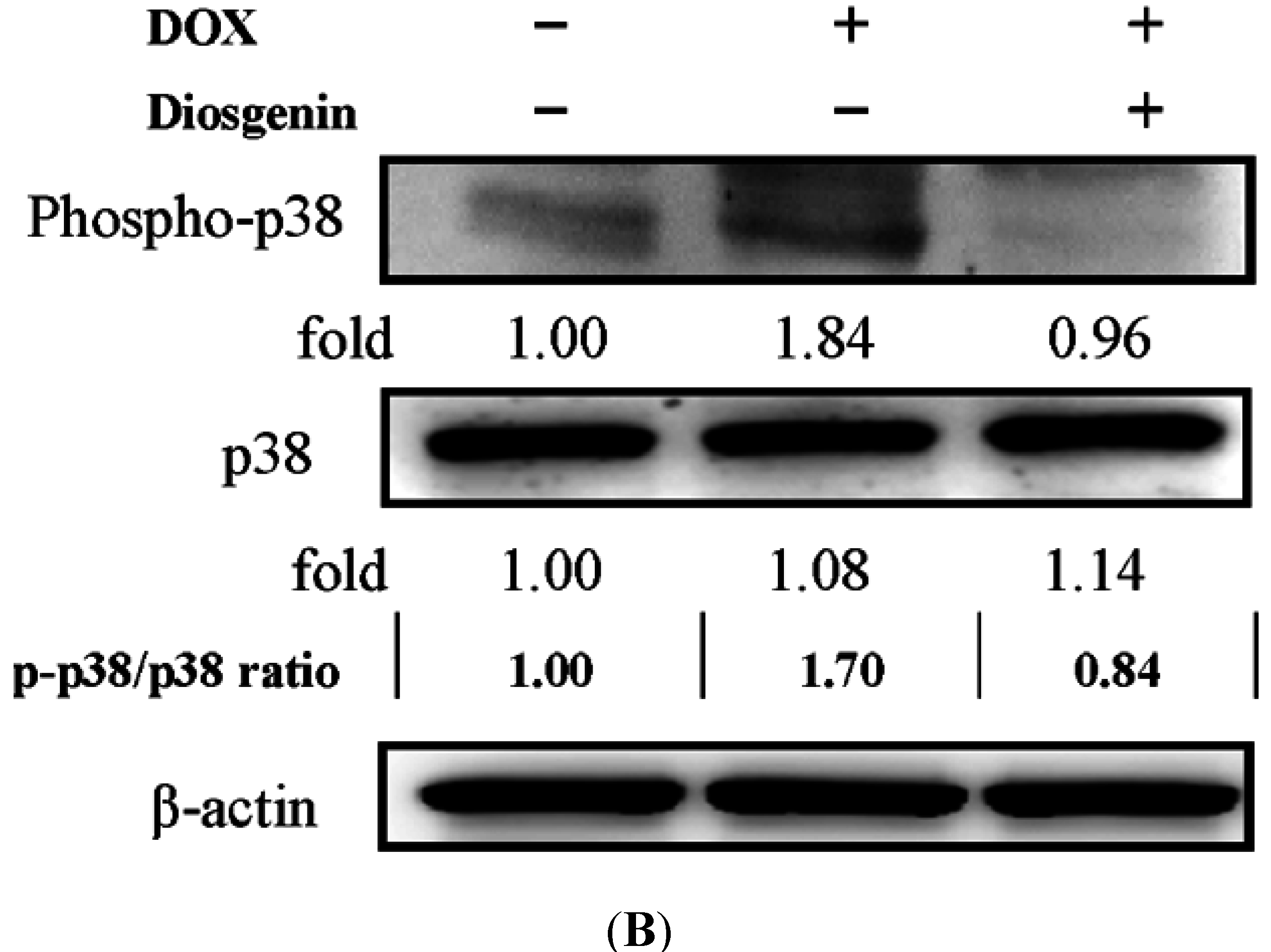
3.5. Diosgenin Restored DOX-Regulated PKA and p38 Activation

4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Weiss, R.B. The anthracyclines: Will we ever find a better doxorubicin? Semin. Oncol. 1992, 19, 670–686. [Google Scholar] [PubMed]
- Jain, D. Cardiotoxicity of doxorubicin and other anthracycline derivatives. J. Nucl. Cardiol. 2000, 7, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.X.; Waagstein, F.; Hjalmarson, A. A new insight into adriamycin-induced cardiotoxicity. Int. J. Cardiol. 1990, 29, 15–20. [Google Scholar] [CrossRef]
- Singal, P.K.; Iliskovic, N. Adriamycin cardiomyopathy. N. Engl. J. Med. 1998, 339, 900–905. [Google Scholar] [CrossRef] [PubMed]
- Kalyanaraman, B.; Joseph, J.; Kalivendi, S.; Wang, S.; Konorev, U.; Kotamraju, S. Doxorubicin-induced apoptosis: Implications in cardiotoxicity. Mol. Cell. Biochem. 2002, 234–235, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Tong, J.; Ganguly, P.K.; Singal, P.K. Myocardial adrenergic changes at two stages of heart failure due to adriamycin treatment in rats. Am. J. Physiol. 1991, 260, H909–H916. [Google Scholar] [PubMed]
- Kumar, D.; Kirshenbaum, L.A.; Li, T.; Danelisen, I.; Singal, P.K. Apoptosis in Adriamycin cardiomyopathy and its modulation by probucol. Antioxid. Redox Signal. 2001, 3, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Hadi, N.; Yousif, N.G.; Al-amran, F.G.; Huntei, N.K.; Mohammad, B.I.; Ali, S.J. Vitamin E and telmisartan attenuates doxorubicin induced cardiac injury in rat through down regulation of inflammatory response. BMC Cardiovasc. Disord. 2012, 12, 63. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.C.; Yin, M.C. Preventive effects of ellagic acid against doxorubicin-induced cardio-toxicity in mice. Cardiovasc. Toxicol. 2013, 13, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Nitobe, J.; Yamaguchi, S.; Okuyama, M.; Nozaki, N.; Sata, M.; Miyamoto, T.; Takeishi, Y.; Kubota, I.; Tomoike, H. Reactive oxygen species regulate FLICE inhibitory protein (FLIP) and susceptibility to Fas mediated apoptosis in cardiac myocytes. Cardiovasc. Res. 2003, 57, 119–128. [Google Scholar] [CrossRef]
- Ueno, M.; Kakinuma, Y.; Yuhki, K.; Murakoshi, N.; Iemitsu, M.; Miyauchi, T.; Yamaguchi, I. Doxorubicin induces apoptosis by activation of caspase-3 in cultured cardiomyocytes in vitro and rat cardiac ventricles in vivo. J. Pharmacol. Sci. 2006, 101, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Salloum, F.N.; Abbate, A.; Das, A.; Houser, J.E.; Mudrick, C.A.; Qureshi, I.; Hoke, N.N.; Roy, S.K.; Brown, W.R.; Prabhakar, S.; et al. Sildenafil (viagra) attenuates ischemic cardiomyopathy and improves left ventricular function in mice. Am. J. Physiol. Heart Circ. Physiol. 2008, 294, H1398–H1406. [Google Scholar] [CrossRef] [PubMed]
- Chau, V.Q.; Salloum, F.N.; Hoke, N.N.; Abbate, A.; Kukreja, R.C. Mitigation of the progression of heart failure with sildenafil involves inhibition of RhoA/Rho-kinase pathway. Am. J. Physiol. Heart Circ. Physiol. 2011, 300, H2272–H2279. [Google Scholar] [CrossRef] [PubMed]
- Das, A.; Xi, L.; Kukreja, R.C. Protein kinase G-dependent cardioprotective mechanism of phosphodiesterase-5 inhibition involves phosphorylation of ERK and GSK3. Biol. Chem. 2008, 283, 29572–29585. [Google Scholar] [CrossRef] [PubMed]
- Tsai, E.J.; Kass, D.A. Cyclic GMP signaling in cardiovascular pathophysiology and therapeutics. Pharmacol. Ther. 2009, 122, 216–238. [Google Scholar] [CrossRef] [PubMed]
- Marker, R.E.; Turner, D.L.; Ulshafer, P.R. Sterols. CIV. Diosgenin from certain American plants. J. Am. Chem. Soc. 1940, 62, 2542–2543. [Google Scholar] [CrossRef]
- Yamada, T.; Hoshino, M.; Hayakawa, T.; Ohhara, H.; Yamada, H.; Nakazawa, T.; Inagaki, T.; Iida, M.; Ogasawara, T.; Uchida, A.; et al. Dietary diosgenin attenuates subacute intestinal inflammation associated with in domethacin in rats. Am. J. Physiol. 1997, 273, G355–G364. [Google Scholar] [PubMed]
- McAnuff, M.A.; Omoruyi, F.O.; Morrison, E.Y.; Asemota, H.N. Plasma and liver lipid distributions in streptozotocin-induced diabetic rats fed sapogenin extract of the Jamaican bitter yam (Dioscorea polygonoides). Nutr. Res. 2002, 22, 1427–1434. [Google Scholar] [CrossRef]
- McAnuff, M.A.; Harding, W.W.; Omoruyi, F.O.; Jacobs, H.; Morrison, E.Y.; Asemota, H.N. Hypoglycemic effects of steroidal sapogenins isolated from Jamaican bitter yam, Dioscorea polygonoides. Food Chem. Toxicol. 2005, 43, 1667–1672. [Google Scholar] [CrossRef] [PubMed]
- Son, I.S.; Kim, J.H.; Sohn, H.Y.; Son, K.H.; Kim, J.S.; Kwon, C.S. Antioxidative and hypolipidemic effects of diosgenin, a steroidal saponin of yam (Dioscorea spp.) om high-cholesterol fed rats. Biosci. Biotechnol. Biochem. 2007, 71, 3063–3071. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.C.; Huang, Y.C.; Yin, M.C.; Lin, S.J. Effect of yam (Dioscorea alata vs. Dioscorea japonica) on gastrointestinal function and antioxidant activity in mice. J. Food Sci. 2006, 71, 513–516. [Google Scholar] [CrossRef]
- Vasanthi, H.R.; Mukherjee, S.; Ray, D.; Pandian Jayachandran, K.S.; Lekli, I.; Das, D.K. Protective role of air potato (Dioscorea bulbifera) of yam family in myocardial ischemic reperfusion injury. Food Funct. 2010, 1, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Jayachandran, K.S.; Vasanthi, H.R.; Rajamanickam, G.V. Antilipoperoxidative and membrane stabilizing effect of diosgenin, in experimentally induced myocardial infarction. Mol. Cell. Biochem. 2009, 327, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.W.; Park, H.J.; Jung, D.H.; Kim, T.W.; Park, Y.M.; Kim, B.O.; Sohn, E.H.; Moon, E.Y.; Um, S.H.; Rhee, D.K.; et al. Inhibition of TNF-α-induced adhesion molecule expression by diosgenin in mouse vascular smooth muscle cells via downregulation of the MAPK, Akt and NF-κB signaling pathways. Vasc. Pharmacol. 2010, 53, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Pari, L.; Monisha, P.; Mohamed Jalaludeen, A. Beneficial role of diosgenin on oxidative stress in aorta of streptozotocin induced diabetic rats. Eur. J. Pharmacol. 2012, 691, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Patil, L.; Balaramanb, R. Effect of green tea extract on doxorubicin induced cardiovascular abnormalities: Antioxidant action. Iran. J. Pharm. Res. 2011, 10, 89–96. [Google Scholar] [PubMed]
- Chen, J.H.; Tsai, C.W.; Wang, C.P.; Lin, H.H. Anti-atherosclerotic potential of gossypetin via inhibiting LDL oxidation and foam cell formation. Toxicol. Appl. Pharmacol. 2013, 272, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.; Jung, S.H.; Ryu, J.K.; Kim, B.; Kim, H.S.; Yang, S.K. Isolation and characterization of smooth muscle cells from rat corpus cavernosum tissue for the study of erectile dysfunction. Korean J. Urol. 2012, 53, 556–563. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Feng, Y.; Qu, S.; Wei, X.; Zhu, H.; Luo, Q.; Liu, M.; Chen, G.; Xiao, X. Resveratrol attenuates doxorubicin-induced cardiomyocyte apoptosis in mice through SIRT1-mediated deacetylation of p53. Cardiovasc. Res. 2011, 90, 538–545. [Google Scholar] [CrossRef] [PubMed]
- Elberry, A.A.; Abdel-Naim, A.B.; Abdel-Sattar, E.A.; Nagy, A.A.; Mosli, H.A.; Mohamadin, A.M.; Ashour, O.M. Cranberry (Vaccinium macrocarpon) protects against doxorubicin-induced cardiotoxicity in rats. Food Chem. Toxicol. 2010, 48, 1178–1184. [Google Scholar] [CrossRef] [PubMed]
- Susin, S.A.; Zamzami, N.; Castedo, M.; Daugas, E.; Wang, H.G.; Geley, S.; Fassy, F.; Reed, J.C.; Kroemer, G. The central executioner of apoptosis: Multiple connections between protease activation and mitochondria in Fas/APO-1/CD95- and ceramide-induced apoptosis. J. Exp. Med. 1997, 186, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Green, P.S.; Leeuwenburgh, C. Mitochondrial dysfunction is an early indicator of doxorubicin-induced apoptosis. Biochim. Biophys. Acta 2002, 1588, 94–101. [Google Scholar] [CrossRef]
- Maurice, D.H.; Palmer, D.; Tilley, D.G.; Dunkerley, H.A.; Netherton, S.J.; Raymond, D.R.; Elbatarny, H.S.; Jimmo, S.L. Cyclic nucleotide phosphodiesterase activity, expression, and targeting in cells of the cardiovascular system. Mol. Pharmacol. 2003, 64, 533–546. [Google Scholar] [CrossRef] [PubMed]
- Dhalla, N.S.; Mueller, A.L. Protein kinases as drug development targets for heart disease therapy. Pharmaceuticals 2010, 3, 2111–2145. [Google Scholar] [CrossRef]
- Naidu, M.U.R.; Vijay Kumar, K.; Krishna Mohan, I.; Sundaram, C.; Singh, S. Protective effect of Gingko biloba extract against doxorubicin-induced cardiotoxicity in mice. Indian J. Exp. Biol. 2002, 40, 894–900. [Google Scholar] [PubMed]
- Hardina, R.; Gersl, V.; Klimtova, I.; Simunek, T.; Machackova, J.; Adamcova, M. Anthracycline induced cardiotoxicity. Acta Medica. 2000, 43, 75–82. [Google Scholar]
- Ladas, E.J.; Jacobson, J.S.; Kennedy, D.D.; Teel, K.; Fleischauer, A.; Kelly, K.M. Antioxidants and cancer therapy: A systematic review. J. Clin. Oncol. 2004, 22, 517–528. [Google Scholar] [CrossRef] [PubMed]
- Liagre, B.; Vergne-Salle, P.; Corbiere, C.; Charissoux, J.L.; Beneytout, J.L. Diosgenin, a plant steroid, induces apoptosis in human rheumatoid arthritis synoviocytes with cyclooxygenase-2 overexpression. Arthritis Res. Ther. 2004, 6, R373–R383. [Google Scholar] [CrossRef] [PubMed]
- Trouillas, P.; Corbière, C.; Liagre, B.; Duroux, J.L.; Beneytout, J.L. Structurefunction relationship for saponin effects on cell cycle arrest and apoptosis in the human osteosarcoma cells: A molecular modelling approach of natural molecules structurally close to Diosgenin. Bioorg. Med. Chem. 2005, 13, 1141–1149. [Google Scholar] [CrossRef] [PubMed]
- Manivannan, J.; Shanthakumar, J.; Arunagiri, P.; Raja, B.; Balamurugan, E. Diosgenin interferes coronary vasoconstriction and inhibits osteochondrogenic transdifferentiation of aortic VSMC in CRF rats. Biochimie 2014, 102, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.L.; Cai, B.; Cui, C.B.; Liu, H.W.; Wu, C.F.; Yao, X.S. Diosgenin-3-Oalpha- L-rhamnopyranosyl-(14)-beta-D-glucopyranoside obtained as a new anticancer agent from Dioscorea futschauensis induces apoptosis on human colon carcinoma HCT-15 cells via mitochondria-controlled apoptotic pathway. J. Asian Nat. Prod. Res. 2004, 6, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.J.; Wang, Z.; Ju, Y.; Wong, R.N.; Wu, Q.Y. Diosgenin induces cell cycle arrest and apoptosis in human leukemia K562 cells with the disruption of Ca2+ homeostasis. Cancer Chemother. Pharmacol. 2005, 55, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, S.; Koduru, S.; Kumar, R.; Venguswamy, G.; Kyprianou, N.; Damodaran, C. Diosgenin targets Akt-mediated prosurvival signaling in human breast cancer cells. Int. J. Cancer 2009, 125, 961–967. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Fernandez, P.P.; Rajendran, P.; Hui, K.M.; Sethi, G. Diosgenin, a steroidal saponin, inhibits STAT3 signaling pathway leading to suppression of proliferation and chemosensitization of human hepatocellular carcinoma cells. Cancer Lett. 2010, 292, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.T.; Zheng, L.H.; Bao, Y.L.; Yu, C.L.; Wu, Y.; Meng, X.Y.; Li, Y.X. Reversal effect of Dioscin on multidrug resistance in human hepatoma HepG2/adriamycin cells. Eur. J. Pharmacol. 2011, 654, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Menna, P.; Salvatorelli, E.; Cairo, G.; Gianni, L. Anthracyclines: Molecular advances and pharmacologic developments in antitumor activity and cardiotoxicity. Pharmacol. Rev. 2004, 56, 185–229. [Google Scholar]
- Zhang, Y.W.; Shi, J.; Li, Y.J.; Wei, L. Cardiomyocyte death in doxorubicin-induced cardiotoxicity. Arch. Immunol. Ther. Exp. (Warsz) 2009, 57, 435–445. [Google Scholar] [CrossRef] [PubMed]
- Das, A.; Smolenski, A.; Lohmann, S.M.; Kukreja, R.C. Cyclic GMP-dependent protein kinase I alpha attenuates necrosis and apoptosis following ischemia/reoxygenation in adult cardiomyocyte. J. Biol. Chem. 2006, 281, 38644–38652. [Google Scholar] [CrossRef] [PubMed]
- Tomita, H.; Nazmy, M.; Kajimoto, K.; Yehia, G.; Molina, C.A.; Sadoshima, J. Inducible cAMP early repressor (ICER) is a negative-feedback regulator of cardiac hypertrophy and an important mediator of cardiac myocyte apoptosis in response to beta-adrenergic receptor stimulation. Circ. Res. 2003, 93, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Booz, G.W. Putting the brakes on cardiac hypertrophy: Exploiting the NO-cGMP counter-regulatory system. Hypertension 2005, 45, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, S.K.; Patil, C.S. Phosphodiesterase 5 enzyme and its inhibitors: Update on pharmacological and therapeutical aspects. Methods Find Exp. Clin. Pharmacol. 2004, 26, 789–799. [Google Scholar] [CrossRef] [PubMed]
- Takimoto, E.; Champion, H.C.; Li, M.; Belardi, D.; Ren, S.; Rodriguez, E.R.; Bedja, D.; Gabrielson, K.L.; Wang, Y.; Kass, D.A. Chronic inhibition of cyclic GMP phosphodiesterase 5A prevents and reverses cardiac hypertrophy. Nat. Med. 2005, 11, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Westermann, D.; Becher, P.M.; Lindner, D.; Savvatis, K.; Xia, Y.; Fröhlich, M.; Hoffmann, S.; Schultheiss, H.P.; Tschöpe, C. Selective PDE5A inhibition with sildenafil rescues left ventricular dysfunction, inflammatory immune response and cardiac remodeling in angiotensin II-induced heart failure in vivo. Basic Res. Cardiol. 2012, 107, 308. [Google Scholar] [CrossRef] [PubMed]
- Zeidan, A.; Siam, A.; Al Kaabba, A.; Mohammad, M.; Khatib, S. The ability of phosphodiesterase-5 inhibitors sildenafil and ordonafil to reverse L-NAME induced cardiac hypertrophy in the rabbit: Possible role of calcineurin and p38. Can. J. Physiol. Pharmacol. 2012, 90, 1247–1255. [Google Scholar] [CrossRef] [PubMed]
- Henaff, M.; Hatem, S.N.; Mercadier, J.J. Low catecholamine concentrations protect adult rat ventricular myocytes against apoptosis through cAMP dependent extracellular signal-regulated kinase activation. Mol. Pharmacol. 2000, 58, 1546–1553. [Google Scholar] [PubMed]
- Liao, P.; Georgakopoulos, D.; Kovacs, A.; Zheng, M.; Lerner, D.; Pu, H.; Saffitz, J.; Chien, K.; Xiao, R.P.; Kass, D.A.; et al. The in vivo role of p38 MAP kinases in cardiac remodeling and restrictive cardiomyopathy. Proc. Natl. Acad. Sci. USA 2001, 98, 12283–12288. [Google Scholar] [CrossRef] [PubMed]
- Trivedi, P.P.; Kushwaha, S.; Tripathi, D.N.; Jena, G.B. Cardioprotective effects of hesperetin against doxorubicin-induced oxidative stress and DNA damage in rat. Cardiovasc. Toxicol. 2011, 11, 215–225. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.L.; Loh, S.H.; Chen, J.J.; Tsai, C.S. Urotensin II prevents cardiomyocyte apoptosis induced by doxorubicin via Akt and ERK. Eur. J. Pharmacol. 2012, 680, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Yue, T.L.; Wang, C.; Gu, J.L.; Ma, X.L.; Kumar, S.; Lee, J.C.; Feuerstein, G.Z.; Thomas, H.; Maleeff, B.; Ohlstein, E.H. Inhibition of extracellular signal-regulated kinase enhances ischemia/reoxygenation-induced apoptosis in cultured cardiac myocytes and exaggerates reperfusion injury in isolated perfused heart. Circ. Res. 2000, 86, 692–699. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, C.-T.; Wang, Z.-H.; Hsu, C.-C.; Lin, H.-H.; Chen, J.-H. In Vivo Protective Effects of Diosgenin against Doxorubicin-Induced Cardiotoxicity. Nutrients 2015, 7, 4938-4954. https://doi.org/10.3390/nu7064938
Chen C-T, Wang Z-H, Hsu C-C, Lin H-H, Chen J-H. In Vivo Protective Effects of Diosgenin against Doxorubicin-Induced Cardiotoxicity. Nutrients. 2015; 7(6):4938-4954. https://doi.org/10.3390/nu7064938
Chicago/Turabian StyleChen, Chih-Tai, Zhi-Hong Wang, Cheng-Chin Hsu, Hui-Hsuan Lin, and Jing-Hsien Chen. 2015. "In Vivo Protective Effects of Diosgenin against Doxorubicin-Induced Cardiotoxicity" Nutrients 7, no. 6: 4938-4954. https://doi.org/10.3390/nu7064938
APA StyleChen, C.-T., Wang, Z.-H., Hsu, C.-C., Lin, H.-H., & Chen, J.-H. (2015). In Vivo Protective Effects of Diosgenin against Doxorubicin-Induced Cardiotoxicity. Nutrients, 7(6), 4938-4954. https://doi.org/10.3390/nu7064938





