Does Motivation for Exercise Influence Post-Exercise Snacking Behavior?
Abstract
:1. Introduction
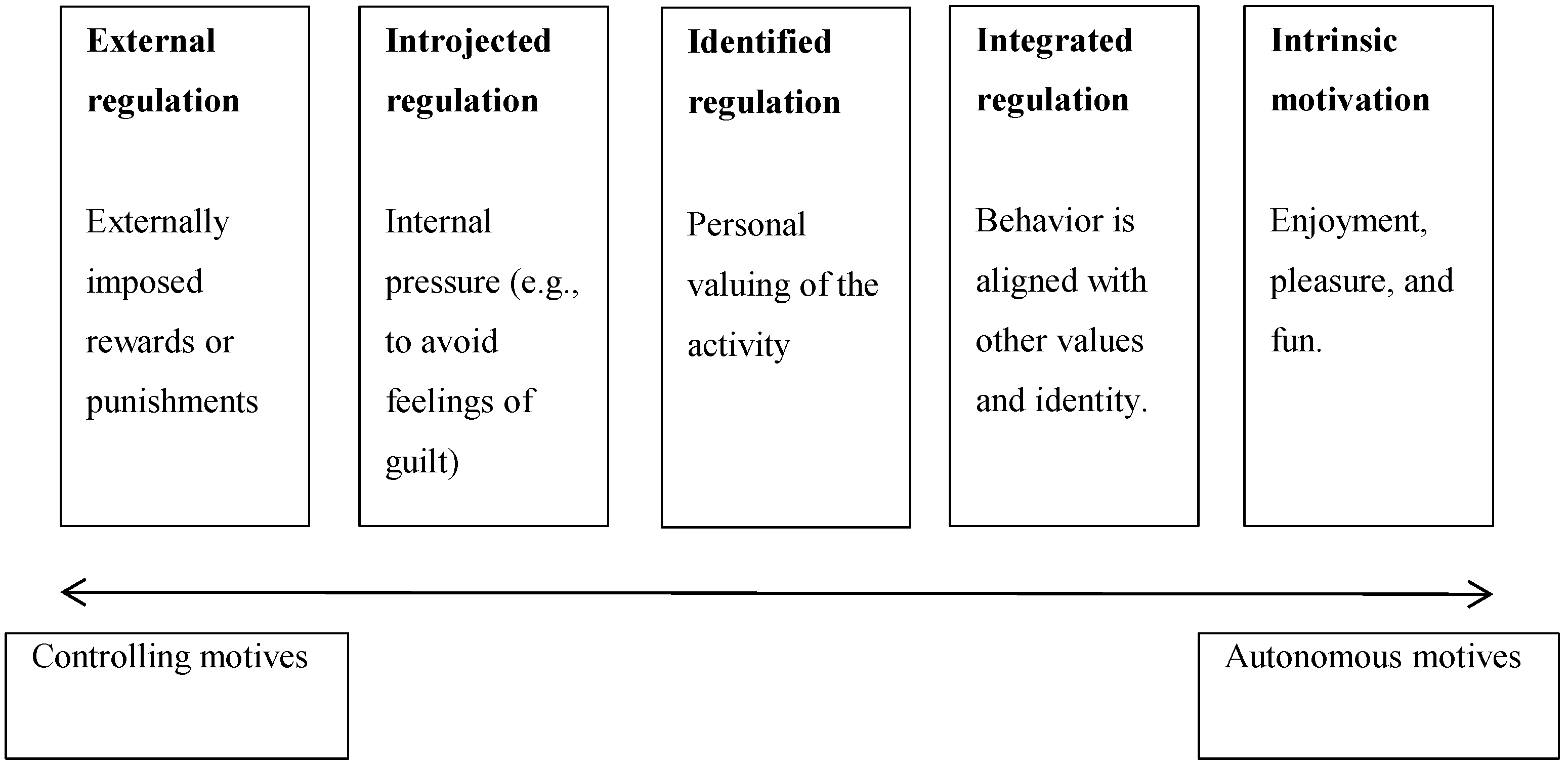
Exercise Motivation from the Lens of Self-Determination Theory
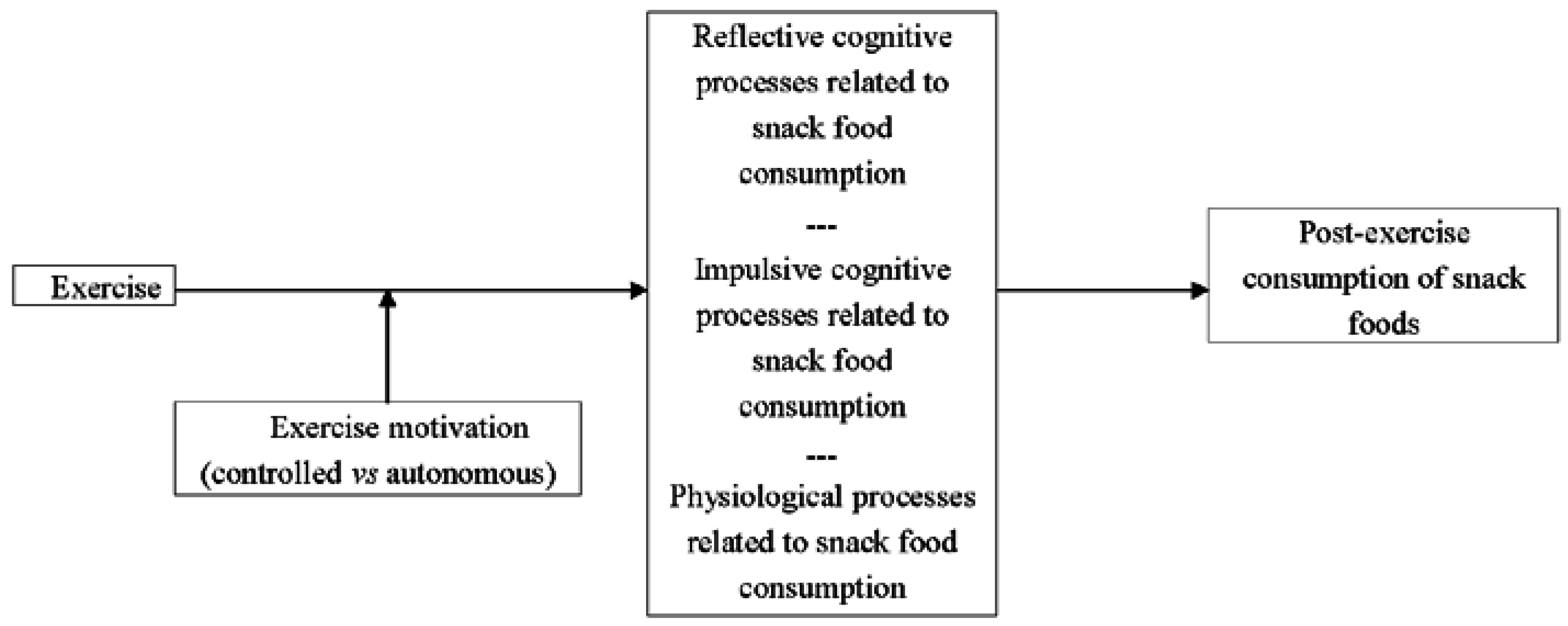
2. Mechanisms for the Influence of Exercise Motivation on Post-Exercise Snacking
2.1. Exercise Motivation and Reflective Justifications for Snacking
2.2. Exercise Motivation and Impulsive Post-Exercise Snack Consumption
2.3. Exercise Motivation and Physiological Regulation of Post-Exercise Snack Behavior
3. Conclusions
Author Contributions
Conflicts of Interest
References
- Blundell, J.E.; Gibbons, C.; Caudwell, P.; Finlayson, G.; Hopkins, M. Appetite control and energy balance: Impact of exercise. Obes. Rev. 2015, 16, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Schubert, M.M.; Desbrow, B.; Sabapathy, S.; Leveritt, M. Acute exercise and subsequent energy intake. A meta-analysis. Appetite 2013, 63, 92–104. [Google Scholar] [CrossRef] [PubMed]
- Hall, K.D.; Heymsfield, S.B.; Kemnitz, J.W.; Klein, S.; Schoeller, D.A.; Speakman, J.R. Energy balance and its components. Implications for body weight regulation. Am. J. Clin. Nutr. 2012, 95, 989–994. [Google Scholar] [CrossRef] [PubMed]
- Werle, C.O.C.; Wansink, B.; Payne, C.R. Just thinking about exercise makes me serve more food: Physical activity and calorie compensation. Appetite 2011, 56, 332–335. [Google Scholar] [CrossRef] [PubMed]
- Deci, E.L.; Ryan, R.M. Intrinsic Motivation and Self-Determination in Human Behavior; Springer US: New York, NY, USA, 1985. [Google Scholar]
- Warburton, D.E.R.; Nicol, C.W.; Bredin, S.S.D. Health benefits of physical activity: The evidence. Can. Med. Assoc. J. 2006, 174, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Thøgersen-Ntoumani, C.; Ntoumanis, N. The role of self-determined motivation in the understanding of exercise-related behaviours, cognitions, and physical self-evaluations. J. Sport. Sci. 2006, 24, 393–404. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, P.J.; Carraça, E.V.; Markland, D.; Silva, M.N.; Ryan, R.M. Exercise, physical activity, and self-determination theory: A systematic review. Int. J. Behav. Nutr. Phys. 2012, 9, 78. [Google Scholar] [CrossRef] [PubMed]
- Maltby, J.; Day, L. The relationship between exercise motives and psychological well-being. J. Psychcol. 2001, 135, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Motl, R.W.; Berger, B.G.; Leuschen, P.S. The role of enjoyment in the exercise-mood relationship. Int. J. Sport. Psychol. 2000, 31, 347–363. [Google Scholar]
- Raedeke, T.D. The relationship between enjoyment and affective responses to exercise. J. Appl. Sport. Psychol. 2007, 19, 105–115. [Google Scholar] [CrossRef]
- Fenzl, N.; Bartsch, K.; Koenigstorfer, J. Labeling exercise fat-burning increases post-exercise food consumption in self-imposed exercisers. Appetite 2014, 81, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ajzen, I. The theory of planned behavior. Organ. Behav. Hum. Dec. 1991, 50, 179–211. [Google Scholar] [CrossRef]
- Bandura, A. Health promotion from the perspective of social cognitive theory. Psychol. Health 1998, 13, 623–649. [Google Scholar] [CrossRef]
- Webb, T.L.; Sheeran, P. Does changing behavioral intentions engender behavior change? A meta-analysis of the experimental evidence. Psychol. Bull. 2006, 132, 249–268. [Google Scholar] [CrossRef] [PubMed]
- Perugini, M. Predictive models of implicit and explicit attitudes. Brit. J. Soc. Psychol. 2005, 44, 29–45. [Google Scholar] [CrossRef] [PubMed]
- Connor, M.T.; Perugini, M.; O’Gorman, R.; Ayres, K.; Prestwich, A. Relations between implicit and explicit measures of attitudes and measures of behavior: Evidence of moderation by individual difference variables. Pers. Soc. Psychol. B 2007, 33, 1727–1740. [Google Scholar] [CrossRef] [PubMed]
- Friese, M.; Hofmann, W.; Wänke, M. When impulses take over: Moderated predictive validity of explicit and implicit attitude measures in predicting food choice and consumption behavior. Br. J. Soc. Psychol. 2008, 47, 397–419. [Google Scholar] [CrossRef] [PubMed]
- Muraven, M.; Baumeister, R.F. Self-regulation and depletion of limited resources. Does self-control resemble a muscle? Psychol. Bull. 2000, 126, 247–259. [Google Scholar] [CrossRef] [PubMed]
- Ramanathan, S.; Menon, G. Time-varying effects of chronic hedonic goals on impulsive behavior. J. Mark. Res. 2006, 43, 628–641. [Google Scholar] [CrossRef]
- Knäuper, B.; Rabiau, M.; Cohen, O.; Patriciu, N. Compensatory health beliefs. Scale development and psychometric properties. Psychol. Health 2004, 19, 607–624. [Google Scholar] [CrossRef]
- Festinger, L. A Theory of Cognitive Dissonance; Stanford University Press: Stanford, CA, USA, 1957. [Google Scholar]
- Rabiau, M.; Knäuper, B.; Miquelon, P. The eternal quest for optimal balance between maximizing pleasure and minimizing harm. The compensatory health beliefs model. Br. J. Health. Psychol. 2006, 11, 139–153. [Google Scholar] [CrossRef] [PubMed]
- Nix, G.A.; Ryan, R.M.; Manly, J.B.; Deci, E.L. Revitalization through self-regulation: The effects of autonomous and controlled motivation on happiness and vitality. J. Exp. Soc. Psychol. 1999, 35, 266–284. [Google Scholar] [CrossRef]
- Miquelon, P.; Knäuper, B.; Vallerand, R.J. Motivation and goal attainment: The role of compensatory beliefs. Appetite 2012, 58, 608–615. [Google Scholar] [CrossRef] [PubMed]
- Fishbach, A.; Dhar, R. Goals as excuses or guides: The liberating effect of perceived goal progress on choice. J. Consum. Res. 2005, 32, 370–377. [Google Scholar] [CrossRef]
- Fishbach, A.; Shah, J.Y. Self-control in action. Implicit dispositions toward goals and away from temptations. J. Pers. Soc. Psychol. 2006, 90, 820–832. [Google Scholar] [CrossRef] [PubMed]
- Ryan, R.M.; Deci, E.L. Self-determination theory and the facilitation of intrinsic motivation, social development, and well-being. Am. Psychol. 2000, 55, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Kiberstis, P. Food as reward. Sci. Signal. 2013, 6, 193. [Google Scholar] [CrossRef]
- Volkow, N.D.; Wang, G.J.; Baler, R.D. Reward, dopamine and the control of food intake: Implications for obesity. Trends Cogn. Sci. 2011, 15, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Moshier, S.J.; Landau, A.J.; Hearon, B.A.; Stein, A.T.; Greathouse, L.; Smits, J.A.J.; Otto, M.W. The development of a novel measure to assess motives for compensatory eating in response to exercise: The CEMQ. Behav. Med. 2014, 22, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Strack, F.; Deutsch, R. Reflective and impulsive determinants of social behavior. Pers. Soc. Psychol. Rev. 2004, 8, 220–247. [Google Scholar] [CrossRef] [PubMed]
- Gawronski, B.; Bodenhausen, G.V. Associative and propositional processes in evaluation: An integrative review of implicit and explicit attitude change. Psychol. Bull. 2006, 132, 692–731. [Google Scholar] [CrossRef] [PubMed]
- Baumeister, R.F.; Vohs, K.D.; Tice, D.M. The strength model of self-control. Curr. Dir. Psychol. Sci. 2007, 16, 351–355. [Google Scholar] [CrossRef]
- Hagger, M.S.; Wood, C.; Stiff, C.; Chatzisarantis, N.L.D. Ego depletion and the strength model of self-control. A meta-analysis. Psychol. Bull. 2010, 136, 495–525. [Google Scholar] [CrossRef] [PubMed]
- Dewitte, S.; Bruyneel, S.; Geyskens, K. Self-regulating enhances self-regulation in subsequent consumer decisions involving similar response conflicts. J. Consum. Res. 2009, 36, 394–405. [Google Scholar] [CrossRef]
- Geyskens, K.; Dewitte, S.; Pandelaere, M.; Warlop, L. Tempt me just a little bit more: The effect of prior food temptation actionability on goal activation and consumption. J. Consum. Res. 2008, 35, 600–610. [Google Scholar]
- Ryan, R.M.; Patrick, H.; Deci, E.L.; Williams, G.C. Facilitating health behavior change and its maintenance: Interventions based on Self-Determination Theory. Euro. Health. Psychol. 2008, 10, 2–5. [Google Scholar]
- Magaraggia, C.; Dimmock, J.A.; Jackson, B. The effect of learning climate on snack consumption and ego depletion among undergraduate students. Appetite 2013, 69, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, W.; Rauch, W.; Gawronski, B. And deplete us not into temptation: Automatic attitudes, dietary restraint, and self-regulatory resources as determinants of eating behavior. J. Exp. Soc. Psychol. 2007, 43, 497–504. [Google Scholar] [CrossRef]
- Gailliot, M.T.; Baumeister, R.F.; DeWall, C.N.; Maner, J.K.; Plant, E.A.; Tice, D.M.; Brewer, L.E.; Schmeichel, B.J. Self-control relies on glucose as a limited energy source. Willpower is more than a metaphor. J. Pers. Soc. Psychol. 2007, 92, 325–336. [Google Scholar] [CrossRef] [PubMed]
- Mayer, J. Regulation of energy intake and the body weight: The glucostatic theory and the lipostatic hypothesis. Ann. N. Y. Acad. Sci. 1955, 63, 15–43. [Google Scholar] [CrossRef] [PubMed]
- DeWall, C.N.; Baumeister, R.F.; Gailliot, M.T.; Maner, J.K. Depletion makes the heart grow less helpful: Helping as a function of self-regulatory energy and genetic relatedness. Pers. Soc. Psychol. B 2008, 34, 1653–1662. [Google Scholar] [CrossRef] [PubMed]
- Crum, A.J.; Corbin, W.R.; Brownell, K.D.; Salovey, P. Mind over milkshakes: Mindsets, not just nutrients, determine ghrelin response. Health. Psychol. 2011, 30, 424–429. [Google Scholar] [CrossRef] [PubMed]
- Knee, C.R.; Neighbors, C.; Vietor, N. Self-determination theory as a framework for understanding road rage. J. Appl. Soc. Psychol. 2001, 31, 889–904. [Google Scholar] [CrossRef]
- Weinstein, N.; Ryan, R.M. A self-determination theory approach to understanding stress incursion and responses. Stress Health 2011, 27, 4–17. [Google Scholar] [CrossRef]
- Crum, A.J.; Salovey, P.; Achor, S. Rethinking stress: The role of mindsets in determining the stress response. J. Pers. Soc. Psychol. 2013, 104, 716–733. [Google Scholar] [CrossRef] [PubMed]
- Kunz-Ebrecht, S.R.; Mohamed-Ali, V.; Feldman, P.J.; Kirschbaum, C.; Steptoe, A. Cortisol responses to mild psychological stress are inversely associated with proinflammatory cytokines. Brain. Behav. Immun. 2003, 17, 373–383. [Google Scholar] [CrossRef]
- McEwen, B. Stressed or stressed out: What is the difference? J. Psychiatry Neurosci. 2005, 30, 315–318. [Google Scholar] [PubMed]
- McEwen, B. Central effects of stress hormones in health and disease: Understanding the protective and damaging effects of stress and stress mediators. Eur. J. Pharmacol. 2008, 583, 174–185. [Google Scholar] [CrossRef] [PubMed]
- Yau, Y.H.; Potenza, M.N. Stress and eating behaivours. Minerva Endocrinol. 2013, 38, 255–267. [Google Scholar] [PubMed]
- Dallman, M.F. Stress-induced obesity and the emotional nervous system. Trends Endocrinol. Metab. 2010, 21, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Reeve, J.; Tseng, C.M. Cortisol reactivity to a teacher’s motivating style: The biology of being controlled versus supporting autonomy. Motiv. Emotion. 2011, 35, 63–74. [Google Scholar] [CrossRef]
- Di Bartolo, P.M.; Lin, L.; Montoya, S.; Neal, H.; Shaffer, C. Are there “healthy” and “unhealthy” reasons for exercise? Examining individual differences in exercise motivations using the function of exercise scale. J. Clin. Sport. Psychol. 2007, 1, 93–120. [Google Scholar]
- Quested, E.; Bosch, J.A.; Burns, V.E.; Cumming, J.; Ntoumanis, N.; Duda, J.L. Basic psychological need satisfaction, stress-related appraisals, and dancers’ cortisol and anxiety responses. J. Sport. Exerc. Psychol. 2011, 33, 828–846. [Google Scholar] [PubMed]
- Hagger, M.; Chatzisarantis, N.L.D. Intrinsic Motivation and Self-Determination in Exercise and Sport; Human Kinetics: Champaign, IL, USA, 2007. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dimmock, J.A.; Guelfi, K.J.; West, J.S.; Masih, T.; Jackson, B. Does Motivation for Exercise Influence Post-Exercise Snacking Behavior? Nutrients 2015, 7, 4804-4816. https://doi.org/10.3390/nu7064804
Dimmock JA, Guelfi KJ, West JS, Masih T, Jackson B. Does Motivation for Exercise Influence Post-Exercise Snacking Behavior? Nutrients. 2015; 7(6):4804-4816. https://doi.org/10.3390/nu7064804
Chicago/Turabian StyleDimmock, James A., Kym J. Guelfi, Jessica S. West, Tasmiah Masih, and Ben Jackson. 2015. "Does Motivation for Exercise Influence Post-Exercise Snacking Behavior?" Nutrients 7, no. 6: 4804-4816. https://doi.org/10.3390/nu7064804
APA StyleDimmock, J. A., Guelfi, K. J., West, J. S., Masih, T., & Jackson, B. (2015). Does Motivation for Exercise Influence Post-Exercise Snacking Behavior? Nutrients, 7(6), 4804-4816. https://doi.org/10.3390/nu7064804



