Sesamin Ameliorates Advanced Glycation End Products-Induced Pancreatic β-Cell Dysfunction and Apoptosis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Drugs and Reagents
2.2. Preparation and Validation of AGEs
2.3. Laboratory Rodent Studies
2.4. Intraperitoneal Glucose Tolerance Test (GTT), Insulin Releasing Test (IRT) and Intraperitoneal Insulin Tolerance Test (ITT)
2.5. Immunofluorescent Staining for Insulin in Mice Pancreatic Islets
2.6. Assessment of Apoptosis in Mice Pancreatic Islets
2.7. In Situ Detection of ROS Production in Mice Pancreatic Islets
2.8. Cell Culture and Treatment
2.9. Measurement of Insulin Secretion in MIN6 Cells
2.10. Western Blot Analysis
2.11. Detection of Cell Death
2.12. Evaluation of ROS Production in MIN6 Cells
2.13. Measurement of NADPH Oxidase Activity in MIN6 Cells
2.14. Statistical Analysis
3. Results
3.1. Characterization of the AGEs
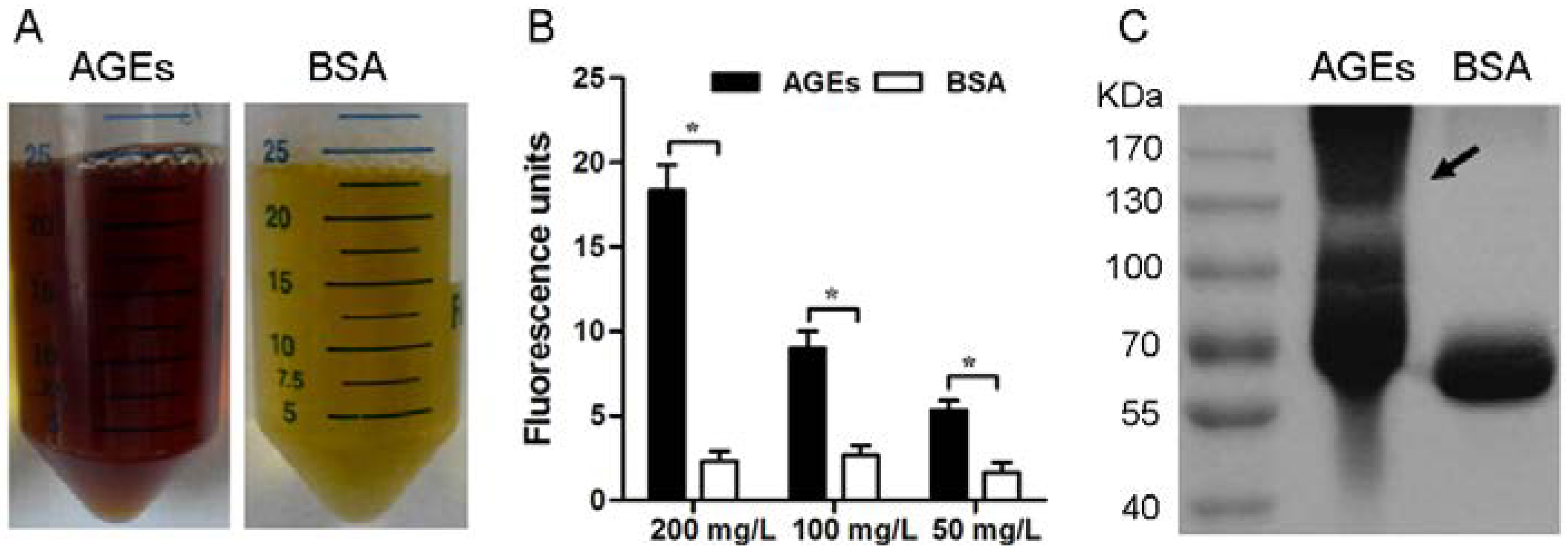
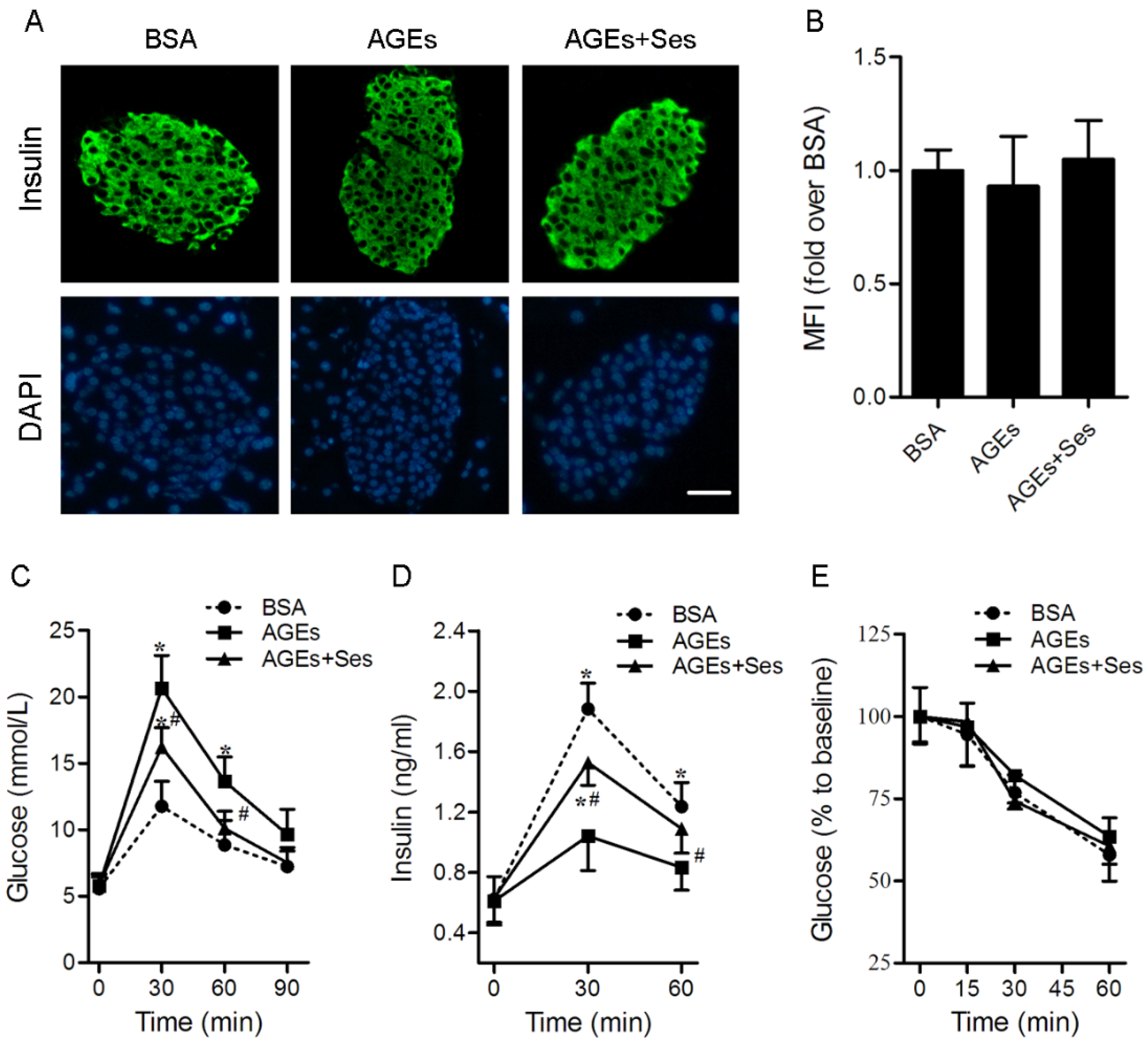
3.2. Insulin Content, GTT, IRT, ITT, BW and Food Intake in Mice
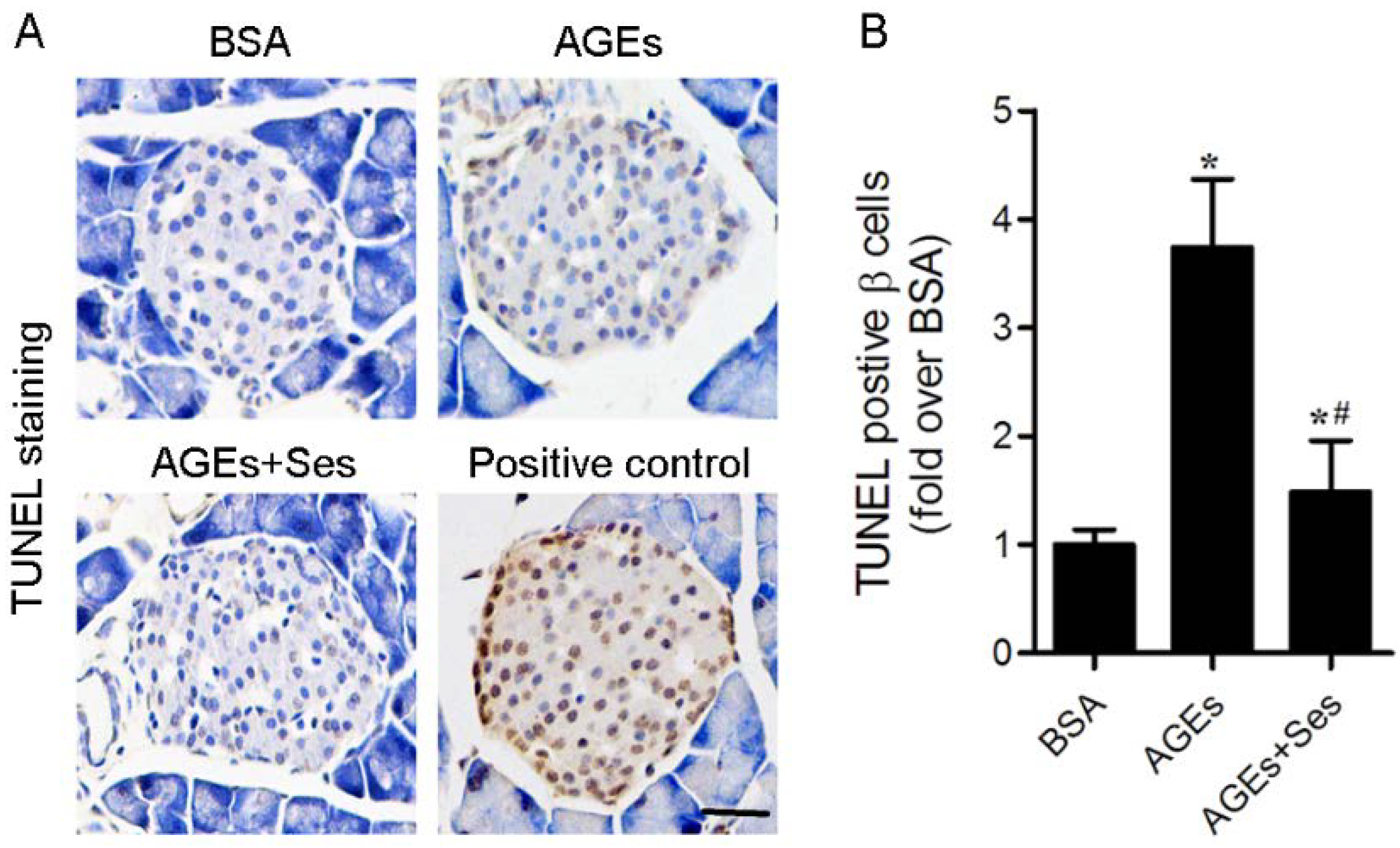
3.3. Apoptosis in Mice Pancreatic Islets
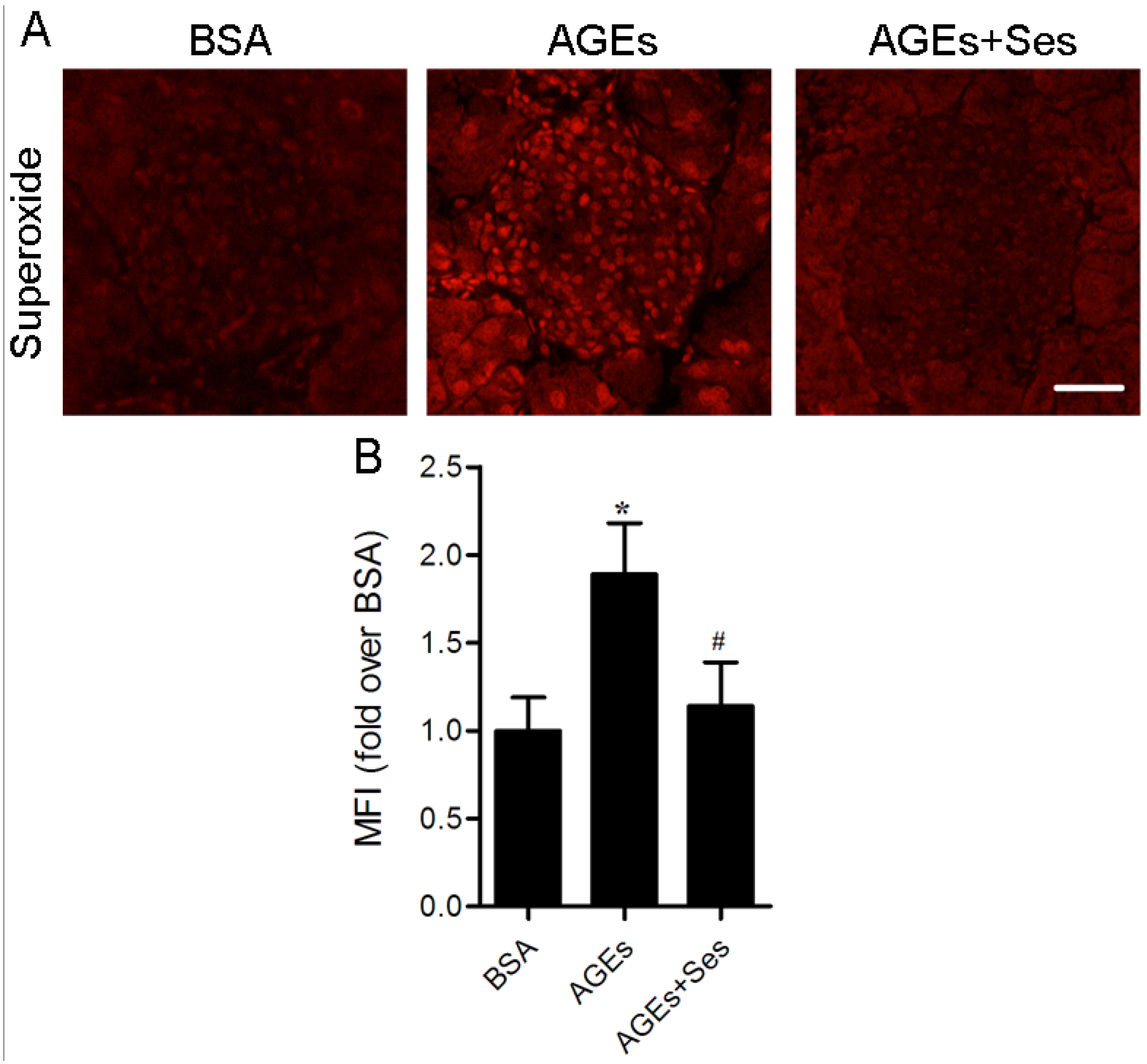
3.4. ROS Production in Mice Pancreatic Islets
3.5. Insulin Secretion in MIN6 Cells

3.6. Apoptosis in MIN6 Cells
3.7. ROS Production in MIN6 Cells
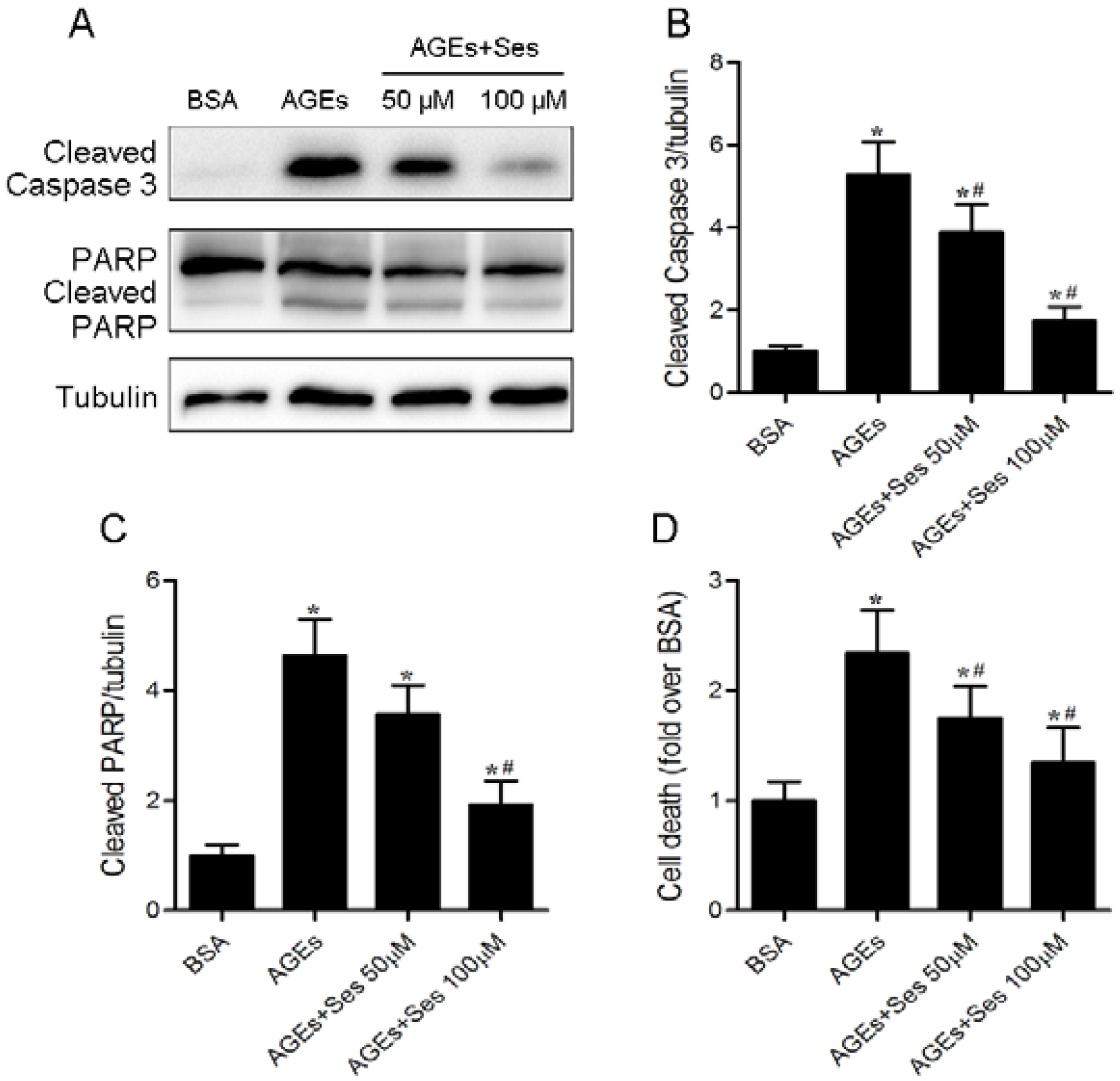
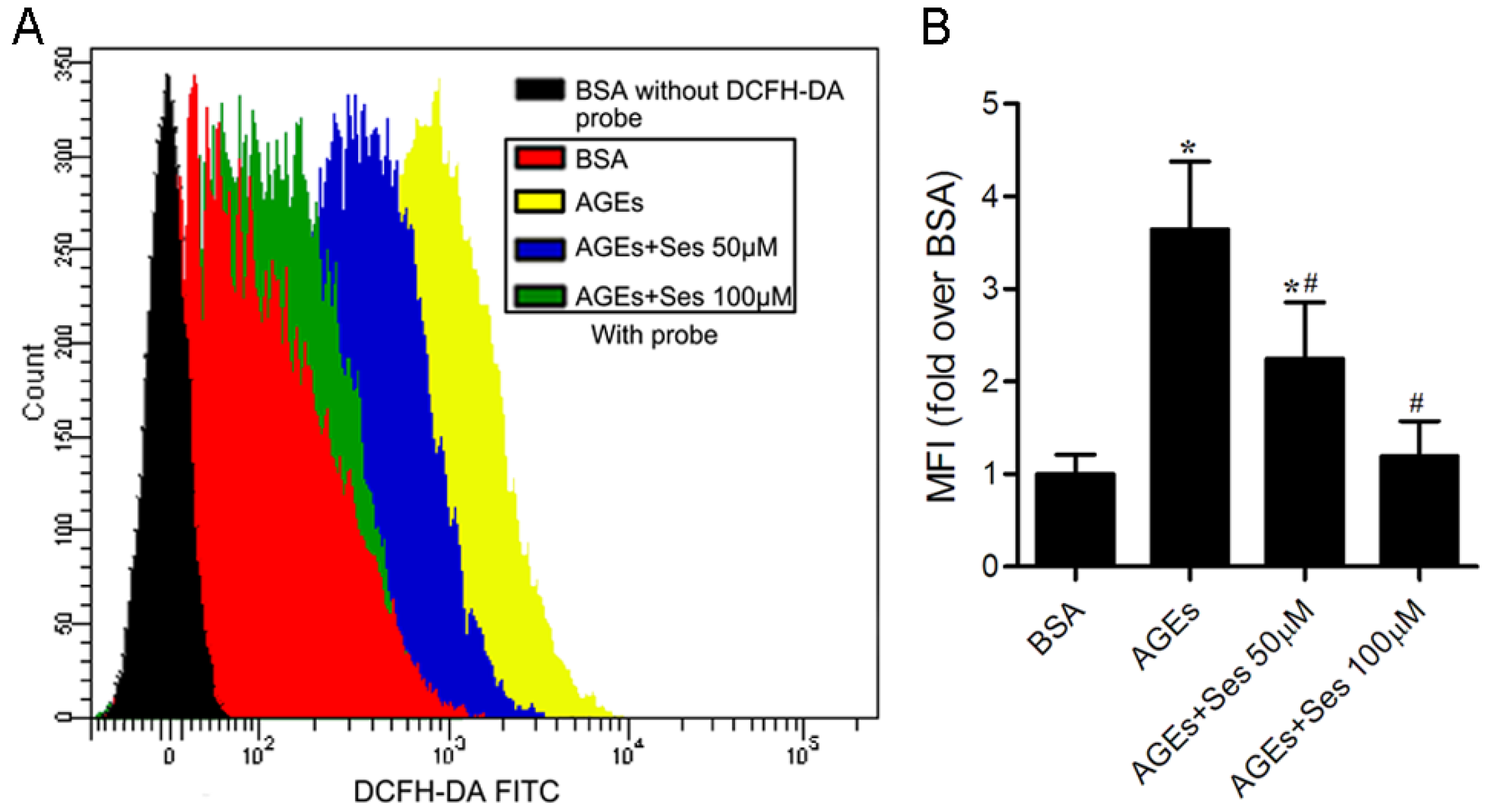
3.8. Expression and Activity of NADPH Oxidase in MIN6 Cells
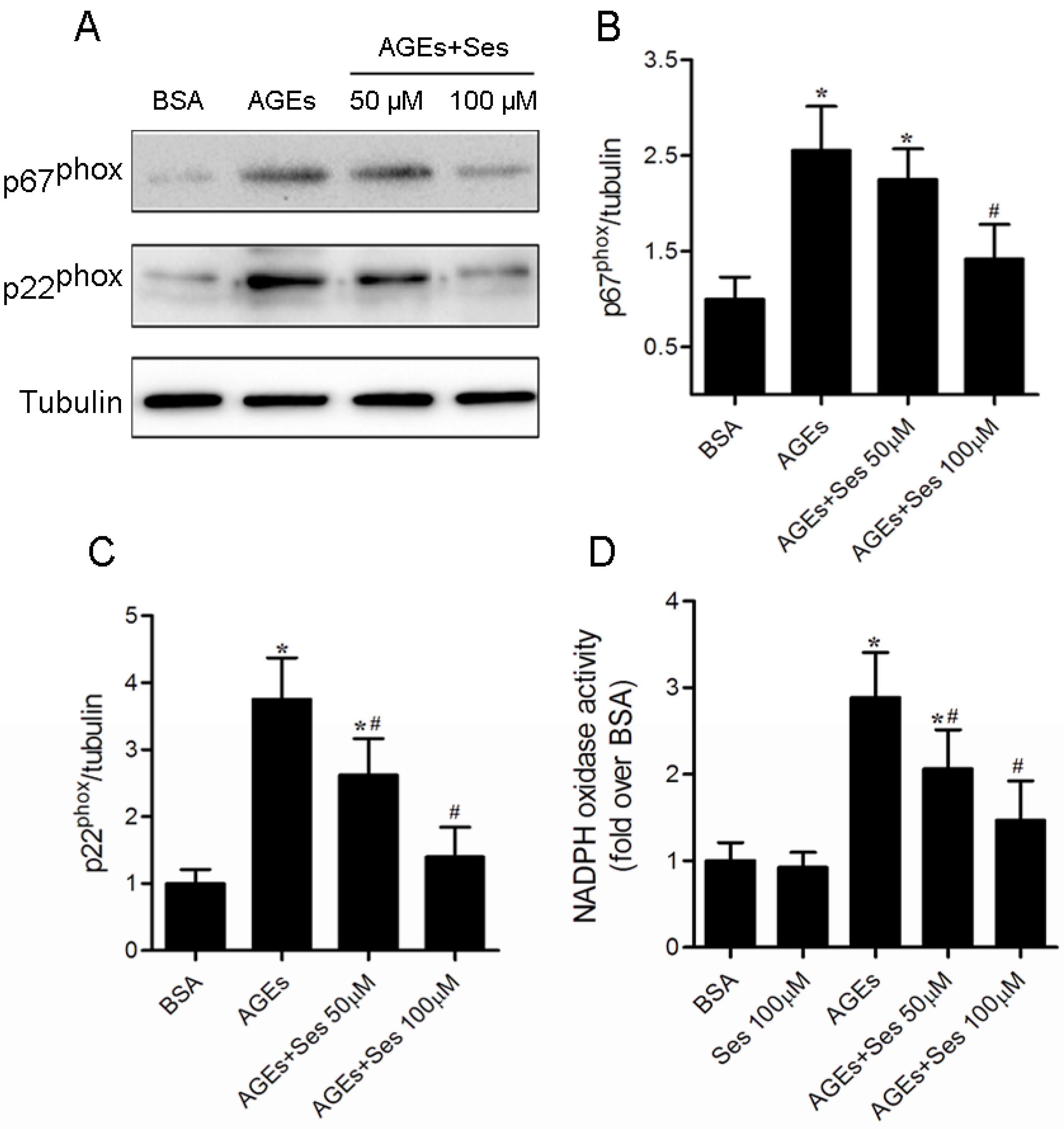
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Peyroux, J.; Sternberg, M. Advanced glycation endproducts (AGEs): Pharmacological inhibition in diabetes. Pathol. Biol. 2006, 54, 405–419. [Google Scholar] [PubMed]
- Vlassara, H.; Palace, M.R. Diabetes and advanced glycation endproducts. J. Intern. Med. 2002, 251, 87–101. [Google Scholar] [CrossRef] [PubMed]
- Kong, X.; Ma, M.Z.; Huang, K.; Qin, L.; Zhang, H.M.; Yang, Z.; Li, X.Y.; Su, Q. Increased plasma levels of the methylglyoxal in patients with newly diagnosed type 2 diabetes 2. J. Diabetes 2014, 6, 535–540. [Google Scholar] [CrossRef] [PubMed]
- Wendt, T.M.; Tanji, N.; Guo, J.; Kislinger, T.R.; Qu, W.; Lu, Y.; Bucciarelli, L.G.; Rong, L.L.; Moser, B.; Markowitz, G.S.; et al. RAGE drives the development of glomerulosclerosis and implicates podocyte activation in the pathogenesis of diabetic nephropathy. Am. J. Pathol. 2003, 162, 1123–1137. [Google Scholar] [CrossRef]
- Ahmed, N.; Thornalley, P.J. Advanced glycation endproducts: What is their relevance to diabetic complications? Diabetes Obes. Metab. 2007, 9, 233–245. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Zhao, C.; Zhang, X.H.; Zheng, F.; Cai, W.; Vlassara, H.; Ma, Z.A. Advanced glycation end products inhibit glucose-stimulated insulin secretion through nitric oxide-dependent inhibition of cytochrome c oxidase and adenosine triphosphate synthesis. Endocrinology 2009, 150, 2569–2576. [Google Scholar] [CrossRef] [PubMed]
- Coughlan, M.T.; Yap, F.Y.; Tong, D.C.; Andrikopoulos, S.; Gasser, A.; Thallas-Bonke, V.; Webster, D.E.; Miyazaki, J.; Kay, T.W.; Slattery, R.M.; et al. Advanced glycation end products are direct modulators of beta-cell function. Diabetes 2011, 60, 2523–2532. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.S.; Cheng, Y.H.; Lee, C.Y.; Chung, C.Y.; Chang, W.C. Resveratrol protects against methylglyoxal-induced hyperglycemia and pancreatic damage in vivo. Nutrients 2015, 7, 2850–2865. [Google Scholar] [CrossRef] [PubMed]
- Ge, Q.M.; Dong, Y.; Su, Q. Effects of glucose and advanced glycation end products on oxidative stress in MIN6 cells. Cell. Mol. Biol. 2010, 56, L1231–L1238. [Google Scholar]
- Lin, N.; Zhang, H.; Su, Q. Advanced glycation end-products induce injury to pancreatic beta cells through oxidative stress. Diabetes Metab. 2012, 38, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Costal, F.; Oliveira, E.; Raposo, A.; Machado-Lima, A.; Peixoto, E.; Roma, L.; Santos, L.; Lopes, F.J.; Carpinelli, A.R.; Giannella-Neto, D.; et al. Dual effect of advanced glycation end products in pancreatic islet apoptosis. Diabetes Metab. Res. Rev. 2013, 29, 296–307. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Wan, X.; Ye, T.; Fang, F.; Chen, X.; Chen, Y.; Dong, Y. Matrix metalloproteinase 2 contributes to pancreatic Beta cell injury induced by oxidative stress. PLoS ONE 2014, 9, e110227. [Google Scholar] [CrossRef] [PubMed]
- Kong, X.; Yang, J.R.; Guo, L.Q.; Xiong, Y.; Wu, X.Q.; Huang, K.; Zhou, Y. Sesamin improves endothelial dysfunction in renovascular hypertensive rats fed with a high-fat, high-sucrose diet. Eur. J. Pharmacol. 2009, 620, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Kong, X.; Zhang, D.Y.; Wu, H.B.; Li, F.X. Losartan and pioglitazone ameliorate nephropathy in experimental metabolic syndrome rats. Biol. Pharm. Bull. 2011, 34, 693–699. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.J.; Ou, H.C.; Wu, C.M.; Lee, I.T.; Lin, S.Y.; Lin, L.Y.; Tsai, K.L.; Lee, S.D.; Sheu, W.H. Sesamin mitigates inflammation and oxidative stress in endothelial cells exposed to oxidized low-density lipoprotein. J. Agric. Food Chem. 2009, 57, 11406–11417. [Google Scholar] [CrossRef] [PubMed]
- Kiso, Y. 15 Antioxidative roles of sesamin, a functional lignan in sesame seed, and its effect on lipid- and alcohol-metabolism in the liver: A DNA microarray study. Biofactors 2004, 21, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.X.; Yang, J.R.; Chen, G.X.; Tang, L.J.; Li, W.X.; Yang, H.; Kong, X. Sesamin ameliorates arterial dysfunction in spontaneously hypertensive rats via downregulation of NADPH oxidase subunits and upregulation of eNOS expression. Acta Pharmacol. Sin. 2013, 34, 912–920. [Google Scholar] [CrossRef] [PubMed]
- Miyawaki, T.; Aono, H.; Toyoda-Ono, Y.; Maeda, H.; Kiso, Y.; Moriyama, K. Antihypertensive effects of sesamin in humans. J. Nutr. Sci. Vitaminol. 2009, 55, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Hong, L.; Yi, W.; Liangliang, C.; Juncheng, H.; Qin, W.; Xiaoxiang, Z. Hypoglycaemic and hypolipidaemic activities of sesamin from sesame meal and its ability to ameliorate insulin resistance in KK-Ay mice. J. Sci. Food Agric. 2013, 93, 1833–1838. [Google Scholar] [CrossRef] [PubMed]
- Makita, Z.; Vlassara, H.; Cerami, A.; Bucala, R. Immunochemical detection of advanced glycosylation end products in vivo. J Biol Chem. 1992, 267, 5133–5138. [Google Scholar] [PubMed]
- Kong, X.; Zhang, Y.; Wu, H.B.; Li, F.X.; Zhang, D.Y.; Su, Q. Combination therapy with losartan and pioglitazone additively reduces renal oxidative and nitrative stress induced by chronic high fat, sucrose, and sodium intake. Oxid. Med. Cell. Longev. 2012, 2012, 856085. [Google Scholar] [CrossRef] [PubMed]
- Kong, X.; Ma, M.Z.; Zhang, Y.; Weng, M.Z.; Gong, W.; Guo, L.Q.; Zhang, J.X.; Wang, G.D.; Su, Q.; Quan, Z.W.; et al. Differentiation therapy: Sesamin as an effective agent in targeting cancer stem-like side population cells of human gallbladder carcinoma. BMC Complement. Altern. Med. 2014, 14, 254. [Google Scholar] [CrossRef] [PubMed]
- Mizutari, K.; Ono, T.; Ikeda, K.; Kayashima, K.; Horiuchi, S. Photo-enhanced modification of human skin elastin in actinic elastosis by N(epsilon)-(carboxymethyl)lysine, one of the glycoxidation products of the Maillard reaction. J. Investig. Dermatol. 1997, 108, 797–802. [Google Scholar] [CrossRef] [PubMed]
- Robertson, R.; Zhou, H.; Zhang, T.; Harmon, J.S. Chronic oxidative stress as a mechanism for glucose toxicity of the beta cell in type 2 diabetes. Cell Biochem. Biophys. 2007, 48, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Lupi, R.; Del, P.S. Beta-cell apoptosis in type 2 diabetes: Quantitative and functional consequences. Diabetes Metab. 2008, 34 (Suppl. S2), S56–S64. [Google Scholar] [CrossRef]
- Lenzen, S.; Drinkgern, J.; Tiedge, M. Low antioxidant enzyme gene expression in pancreatic islets compared with various other mouse tissues. Free Radic. Biol. Med. 1996, 20, 463–466. [Google Scholar] [CrossRef]
- Lenzen, S. Oxidative stress: The vulnerable beta-cell. Biochem. Soc. Trans. 2008, 36, 343–347. [Google Scholar] [CrossRef] [PubMed]
- Turrens, J.F. Mitochondrial formation of reactive oxygen species. J. Physiol. 2003, 552, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Guichard, C.; Moreau, R.; Pessayre, D.; Epperson, T.K.; Krause, K.H. NOX family NADPH oxidases in liver and in pancreatic islets: A role in the metabolic syndrome and diabetes? Biochem. Soc. Trans. 2008, 36, 920–929. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Li, Q.; Liu, Y.; Xiong, C.; Li, J.; Zhang, R.; Niu, Y.; Zhao, L.; Wang, Y.; Guo, H. Sesamin ameliorates doxorubicin-induced cardiotoxicity: Involvement of Sirt1 and Mn-SOD pathway. Toxicol. Lett. 2014, 224, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Lahaie-Collins, V.; Bournival, J.; Plouffe, M.; Carange, J.; Martinoli, M.G. Sesamin modulates tyrosine hydroxylase, superoxide dismutase, catalase, inducible NO synthase andinterleukin-6 expression in dopaminergic cells under MPP+-induced oxidative stress. Oxid. Med. Cell. Longev. 2008, 1, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Ghafoorunissa, S.; Hemalatha, S.; Rao, M.V. Sesame lignans enhance antioxidant activity of vitamin E in lipid peroxidation systems. Mol. Cell. Biochem. 2004, 262, 195–202. [Google Scholar] [PubMed]
- Lei, H.; Han, J.; Wang, Q.; Guo, S.; Sun, H.; Zhang, X. Effects of sesamin on streptozotocin (STZ)-induced NIT-1 pancreatic β-cell damage. Int. J. Mol. Sci. 2012, 13, 16961–16970. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, P.F.; Hou, C.W.; Yao, P.W.; Wu, S.P.; Peng, Y.F.; Shen, M.L.; Lin, C.H.; Chao, Y.Y.; Chang, M.H.; Jeng, K.C. Sesamin ameliorates oxidative stress and mortality in kainic acid-induced status epilepticus by inhibition of MAPK and COX-2 activation. J. Neuroinflammation 2011, 8, 57. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.H.; Kang, Y.P.; Wang, N.H.; Jou, H.J.; Wang, T.A. Sesame ingestion affects sex hormones, antioxidant status, and blood lipids in postmenopausal women. J. Nutr. 2006, 136, 1270–1275. [Google Scholar] [PubMed]
- Palipoch, S.; Punsawad, C.; Koomhin, P.; Suwannalert, P. Hepatoprotective effect of curcumin and alpha-tocopherol against cisplatin-induced oxidative stress. BMC Complement. Altern. Med. 2014, 14, 111. [Google Scholar] [CrossRef] [PubMed]
- Thakurta, I.G.; Chattopadhyay, M.; Ghosh, A.; Chakrabarti, S. Dietary supplementation with N-acetyl cysteine, α-tocopherol and α-lipoic acid reduces the extent of oxidative stress and proinflammatory state in aged rat brain. Biogerontology 2012, 13, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Pazdro, R.; Burgess, J.R. Differential effects of α-tocopherol and N-acetyl-cysteine on advanced glycation end product-induced oxidative damage and neurite degeneration in SH-SY5Y cells. Biochim. Biophys. Acta 2012, 1822, 550–556. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kong, X.; Wang, G.-D.; Ma, M.-Z.; Deng, R.-Y.; Guo, L.-Q.; Zhang, J.-X.; Yang, J.-R.; Su, Q. Sesamin Ameliorates Advanced Glycation End Products-Induced Pancreatic β-Cell Dysfunction and Apoptosis. Nutrients 2015, 7, 4689-4704. https://doi.org/10.3390/nu7064689
Kong X, Wang G-D, Ma M-Z, Deng R-Y, Guo L-Q, Zhang J-X, Yang J-R, Su Q. Sesamin Ameliorates Advanced Glycation End Products-Induced Pancreatic β-Cell Dysfunction and Apoptosis. Nutrients. 2015; 7(6):4689-4704. https://doi.org/10.3390/nu7064689
Chicago/Turabian StyleKong, Xiang, Guo-Dong Wang, Ming-Zhe Ma, Ru-Yuan Deng, Li-Qun Guo, Jun-Xiu Zhang, Jie-Ren Yang, and Qing Su. 2015. "Sesamin Ameliorates Advanced Glycation End Products-Induced Pancreatic β-Cell Dysfunction and Apoptosis" Nutrients 7, no. 6: 4689-4704. https://doi.org/10.3390/nu7064689
APA StyleKong, X., Wang, G.-D., Ma, M.-Z., Deng, R.-Y., Guo, L.-Q., Zhang, J.-X., Yang, J.-R., & Su, Q. (2015). Sesamin Ameliorates Advanced Glycation End Products-Induced Pancreatic β-Cell Dysfunction and Apoptosis. Nutrients, 7(6), 4689-4704. https://doi.org/10.3390/nu7064689



