The Periconceptional Environment and Cardiovascular Disease: Does In Vitro Embryo Culture and Transfer Influence Cardiovascular Development and Health?
Abstract
:1. Introduction
- (1)
- When is the periconceptional period?
- (2)
- Why is the periconceptional period a critical window of embryonic development?
- (3)
- What are the different procedures involved in ARTs and how do these impact the periconceptional environment?
- (4)
- What is the evidence that the periconceptional environment influences cardiovascular health in fetal life and in adulthood?
- (5)
- What is the evidence that ARTs influence cardiovascular health before and after birth?
- (6)
- What are the most likely mechanisms linking ARTs and risk of cardiovascular disease in fetal and adult life?
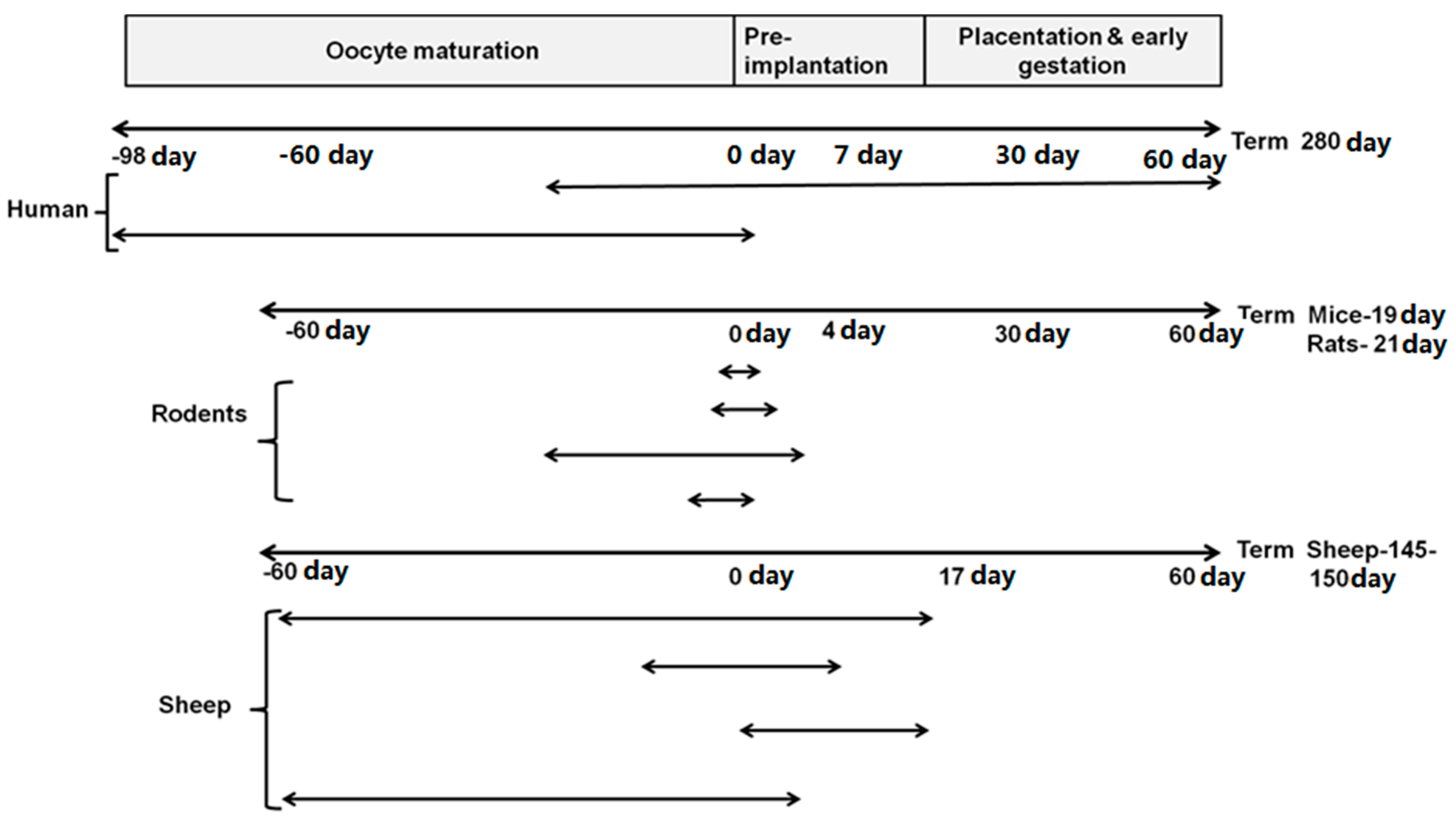
2. When Is the Periconceptional Period?
3. Why Is Periconceptional Period a Critical Window of Development?
3.1. Oogenesis Occurs Early in Life
3.2. Key Events in Embryonic Development during the Periconceptional Period in Different Species
3.2.1. Humans
| Timing of events during the periconceptional period | Human | Mouse | Rat | Sheep |
|---|---|---|---|---|
| Time between ovulations | 28 days | 4–5 days | 4–5 days | 17 days |
| Time to 2 cell stage (first cleavage) | 24 h | 21–23 | 20.6 h | 24–26 h |
| Time to 4 cell stage | 40 h | 38–50 | 72 h | 30–36 h |
| Time to 8 cell stage | 50 h | 50–60 | 78 h | 42–45 h |
| Time to 16–32 cells (morula stage) | 72 h | 60–70 | 84 h | 63–86 h |
| Formation of blastocyst | 5 days | 3–4 days | 4–5 days | 5–6 days |
| Zona hatching | 5–7 days | 3.5 | 6 days | 8 days |
| Implantation | 7–9 days | 4–5 days | 6–7 days | 16 days |
| Zygotic gene activation | 40–50 h | 24 h | 24 h | 30–45 h |
3.2.2. Rats
3.2.3. Sheep
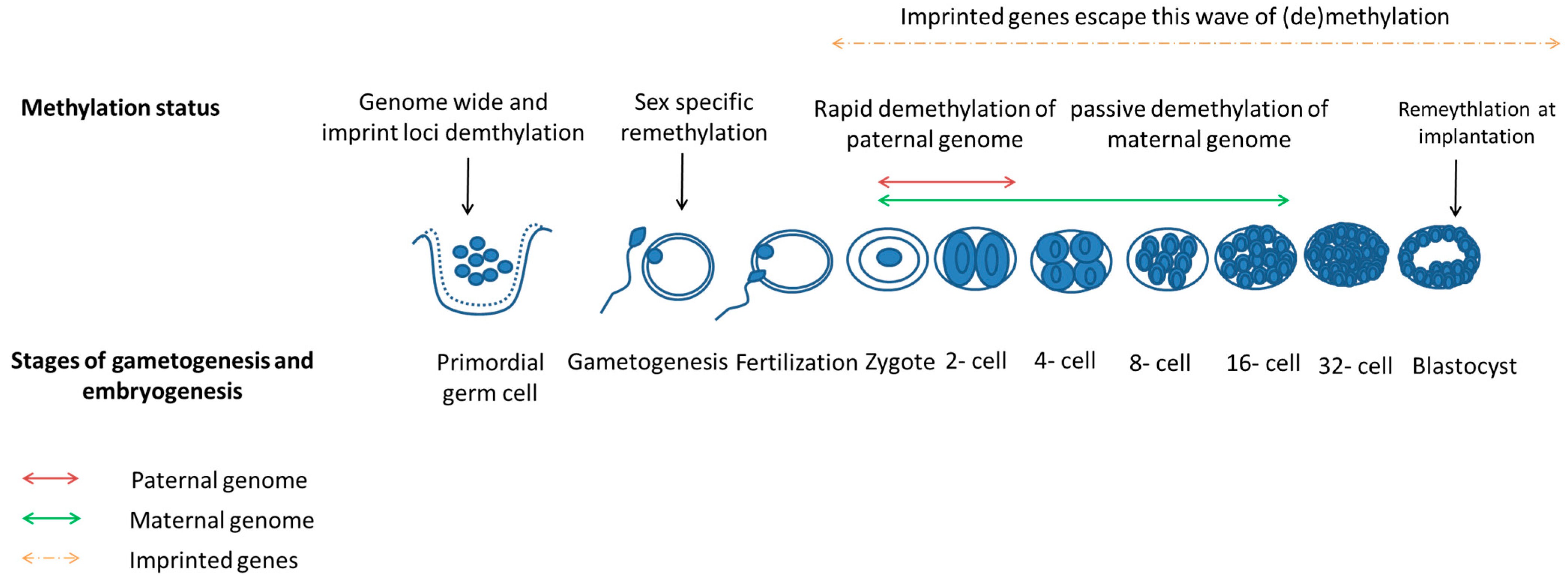
3.3. Zygotic Gene Activation Occurs during Early Embryogenesis
| ART Treatment | Infertility Treated | Procedures Involved and Manipulation of Oocyte/Embryo Development |
|---|---|---|
| IVF | Blocked Fallopian tubes, endometriosis, unexplained infertility, ovarian failure, ovulatory disorders and male infertility | Controlled ovarian hyperstimulation—Oocyte and follicular development |
| Oocyte retrieval and transfer—Oocyte and follicular development | ||
| Sperm retrieval and preparation—No direct effect on embryo | ||
| In vitro fertilization—zygote | ||
| In vitro embryo culture—zygote, cleavage, morula, blastocyst | ||
| Embryo transfer—blastocyst | ||
| GIFT | Sperm dysfunction, endometriosis or unknown fertility | Controlled ovarian hyperstimulation—Oocyte and follicular development |
| Oocyte retrieval—Oocyte and follicular development | ||
| Sperm retrieval and preparation—No direct effect on embryo | ||
| ZIFT | Severe male infertility, immunologic infertility or unexplained infertility | Controlled ovarian hyperstimulation—Oocyte and follicular development |
| Oocyte retrieval—Oocyte and follicular development | ||
| Sperm retrieval and preparation—No direct effect on embryo | ||
| In vitro fertilization—zygote | ||
| In vitro embryo culture—zygote, cleavage | ||
| AI and IUI | Male infertility | No direct effect on oocyte development |
| ICSI | Male infertility | Controlled ovarian hyperstimulation—Oocyte and follicular development |
| Oocyte retrieval—Oocyte and follicular development | ||
| Sperm retrieval and preparation—No direct effect on embryo | ||
| In vitro fertilization—Zygote | ||
| In vitro embryo culture—Zygote, cleavage, morula, blastocyst | ||
| Embryo transfer—Blastocyst |
3.4. Epigenetics Reprogramming Is an Important Event in Both Gametogenesis and Embryogenesis
4. What Are the Different Procedures Involved in ARTs and How Do These Impact the Periconceptional Environment?
4.1. History, Procedures and Current Status of ARTs
4.2. Periconceptional Manipulations Associated with ARTs
4.3. In Vitro Embryo Culture: A Periconceptional Manipulation of the Nutritional Environment of the Gametes and/or Zygote Associated with ARTs
5. What Is the Evidence that the Periconceptional Environment Influences Cardiovascular Health in Fetal Life and Adulthood?
Periconceptional Manipulations and Cardiovascular Disorders: Insights from Human and Animal Studies
| Species | Periconceptional Manipulation | Time | Blood Pressure | Baroreflex Sensitivity | Congenital Heart Defects | Risks for Heart Diseases | Heart Weight | Vaso-Constriction | Vasodilation |
|---|---|---|---|---|---|---|---|---|---|
| Human | Undernutrition | Early gestation | ↑ [196] | n/a | n/a | ↑ [195] | n/a | n/a | n/a |
| Alcohol consumption | −30–+90 days | n/a | n/a | ↑ [199] | n/a | n/a | n/a | n/a | |
| Low maternal dietary nicotinamide and exposure to a range of medicines | −30–+60 days | n/a | n/a | ↑ [200] | n/a | n/a | n/a | n/a | |
| Rodent | Protein restriction | 0–4.25 days | ↑ [201] | n/a | n/a | n/a | n/a | n/a | n/a |
| Low protein diet | −3.5–0 days | ↑ [202] | n/a | n/a | n/a | n/a | n/a | ↓ | |
| 0–3.5 days | ↑ [203] | n/a | n/a | n/a | ↓ [203] | n/a | n/a | ||
| Sheep | Maternal undernutrition | −60–7 days | ↑ [204] | n/a | n/a | n/a | n/a | n/a | n/a |
| 1–30days | ↔ [205] | ↓[205] | n/a | n/a | n/a | n/a | n/a | ||
| 0–95 days | ↑ [206] | ↓ [206] | n/a | n/a | n/a | n/a | n/a | ||
| −30–0 days | n/a | n/a | n/a | n/a | n/a | n/a | ↓ [17] | ||
| −15–15 days | n/a | n/a | n/a | n/a | n/a | ↑ [17] | ↓ [17] | ||
| 1–31days | ↑ [207] | n/a | n/a | n/a | ↑ [207] | ↑ [207] | n/a | ||
| −61–30 days | n/a | n/a | n/a | n/a | ↑ [11] | n/a | n/a | ||
| −61–0 days | n/a | n/a | n/a | n/a | ↓ [208] | n/a | n/a | ||
| −61–30 days | n/a | n/a | n/a | n/a | ↓ [208] | n/a | n/a | ||
| −2–30 days | n/a | n/a | n/a | n/a | ↓ [208] | n/a | n/a |
6. What Is the Evidence that ARTs Influence Cardiovascular Health Before and After Birth?
6.1. Congenital Heart Defects in Human Population
6.2. Evidence of Risk Factors for Long-Term Cardiovascular Outcomes in Humans
| Study Type; Population; Year | Sample Size (N) | Congenital Heart Defects | Reference | |
|---|---|---|---|---|
| Registry of IVF and GIFT pregnancies in Australia and New Zealand | IVF-1694 | 4-cases of transposition of great arteries (p = 0.0034). | [217] | |
| Population based study; Children conceived by IVF or IUI at the University of Iowa; 1989–2002 | IVF-1462 IUI-343 Controls-8422 |
| [218] | |
| Population-wide cohort study; South Australian Perinatal Statistics Collection; January 1986–December 2002 | ART-6163 Controls-302,811 |
| [219] | |
| Population based study; Reproductive Technology Register; 1993–1997 | IVF-837 ICSI-301 Controls-4000 | Increased prevalence of cardiovascular defects in children conceived with IVF, but not those conceived with ICSI compared to controls (p < 0.001). | [220] | |
| Population based study; National professional Perinatal and Neonatal Registers; 1995–1996 | IVF-4224 Controls-314,605 |
| [221] | |
| Population based study; Medical Birth Register, Finland; 1996–1998 | IVF-4559 Other ARTs-4467 Controls-27,078 |
| ||
| Case-control; California Patient Discharge Linked Birth Cohort Database Dataset; 2006 to 2007 | ART-4795 Controls-46,025 |
| [50] | |
| Retrospective cohort study: Ottawa; Fertility Centre; 1996–2005 | ART-1044 Controls-1910 |
| [223] | |
| Cross-sectional descriptive study; Royan Institute, Tehran; | ART-400 | 8 cases (2%) of defects in cardiovascular system. | [224] | |
| Population based cohort study; IVF Outpatient Clinic, University of Oulu and Infertility Clinic of the Family Federation of Finland and Oulu Controls- Finnish Medical Birth Register; 1990–1996 | IVF-304 Controls-569 | 4 fold increase in atrial septal and ventricular septal defects (OR 4.0, 95% CI 1.4–11.7). | [225] | |
| Historical cohort study; Child Health and Development Research Centre; January 2008–December 2010 | ART-326 Controls-652 | Increased risks of cardiovascular malformations in ART children (1.07% vs. 1.53%). | [226] | |
| Population based, multicenter case-control study, National Birth Defects Prevention Study; October 1997–December 2003 | Cases-9584 (with birth defects) Controls-4792 (no birth defects) |
| [227] | |
| Population based study; Swedish Medical Birth Register, Swedish Registry of Congenital Malformations, and Swedish Hospital Discharge Register; 1982–March 31, 2001 | ART-16,280 Controls-2,039,943 |
| [228] | |
| Population based study; Swedish Medical Birth Register, Swedish Registry of Congenital Malformations, and Swedish Hospital Discharge Register; 31 March 2001–1 January 2007 | IVF-15,570 Controls-5,689,157 |
| [229] | |
| Case control study; Paris Registry of Congenital Malformations; 1987–2006 | Cases-5493 (with congenital heart defects) Malformed controls-3487 (malformations not previously associated with ART) |
| [230] | |
| Case-control; Paris Registry of Congenital Malformations; 1987–2009. Prospective cohort study, congenital heart disease in children (EPICARD); 2005–2008 | Case-1583 (with congenital heart defects) Malformed controls-4104 (malformations not previously associated with ART) |
| [231] | |
| Study Type; Population; Year | Sample Size (N); Age(s) | Cardiovascular Risk Factors | Reference |
|---|---|---|---|
| Prospective cohort study; Maternal-Fetal Medicine Unit, Spain. | ART-100 Controls-100 28–30 weeks gestation, 1 month, 6 months |
| [232] |
| Retrospective cohort study; OMEGA study, VU university Medical centre, Netherland; 1980–1995 | IVF-225 Controls-225 8–18 year (mean age 12 year) | Increased SBP and DBP pressure in children conceived through IVF compared to control population at a mean age of 12.3 (109 ± 11 vs. 105 ± 10 mmHg, p < 0.001; and 61 ± 7 vs. 59 ±7 mmHg, p < 0.001), respectively. | [233] |
| Cross- sectional, case-control study; IVF cases-IVF section, Department of Obstetrics and Gynaecology; University of Athens Controls-Aghai Sophia Children’s hospital; 1990–1996 | IVF-106 Controls-68 4–14 year | Increased SBP and DBP standard deviation score (0.3 vs. −0.3, p < 0.001; 0.7 vs. 0.2 p < 0.001) in children conceived through IVF. | [237] |
| Clinical Trial; Swiss children IVF and Control siblings of IVF children; October 2007–April 2010 | ART-65 Controls-57 Mean-11 year | Smaller flow mediated dilation of the brachial artery (6.7 ± 1.6 vs. 8.6 ± 1.7 %; p < 0.0001), faster carotid-femoral pulse wave velocity (7.8 ± 2.4 vs. 6.5 ± 1.3 m/s; p < 0.001), increase in carotid intima-media thickness (410 ± 30 vs. 370 ± 20 μm; p < 0.0001), higher systolic pulmonary artery pressure (39 ± 11 vs. 30 ± 9 mmHg; p < 0.0001) in ART children compared to control children. An inverse relationship existed between pulmonary artery pressure and flow mediated dilation (r= −0.30, p = 0.001). | [238] |
6.3. Evidence of Cardiovascular Defects from Animal Models of ARTs
| Species | Age(s) | Cardiovascular Risk Factors | Reference |
|---|---|---|---|
| Sheep | 61 day gestation 125 day gestation | Increased allometric growth coefficients of heart at 61and 125 days of gestation. | [163] |
| 125 day gestation | Increased absolute (25.8 ± 8.9 vs. 19.0 ± 4.1, p = 0.004) and relative heart weight (6.5 ± 1.05 vs. 5.7 ± 0.76, p = 0.022) in in vitro embryo culture group when serum was supplemented during precompaction period. | [246] | |
| 125 day gestation | Inverse relation between heart weight and IGFR2 gene expression (r = −0.675, p < 0.001) and loss of methylation of IGF2R in embryo culture group when compared to control group. | [58] | |
| 125 day gestation | A strong inverse relation between heart weight and IGFR2 gene expression in the in vitro embryo culture groups(r = −0.73, p < 0.001). | [247] | |
| Cows | 222 day gestation (7 months) | Increased heart girth (56.5 ± 1.2 vs. 52.4 ± 0.9, p = 0.01) and heart weight (139.7 ± 8.3 vs. 116.2 ± 5.8, p = 0.02) in embryo transfer and culture groups compared to controls. | [248] |
| At birth | Increased intra-ventricular septum in in vitro produced groups compared to embryo transfer group (SOF: 11.8 ± 0.3, Co-culture: 12.0 ± 0.3, embryo transfer 10 ± 0.3, p < 0.05) Thicker left ventricular wall during diastole in in vitro produced group (co-culture) when compared to embryo transfer group (14.5 ± 0.5 vs. 11.8 ± 0.6, p < 0.05). | [249] | |
| 1 year | Increased relative heart weight in embryo culture group (4.01 ± 0.08 vs. 3.56 ± 0.12, p < 0.02). | [250] | |
| Rodents | 21 days after birth | Raised SBP in males. | [62] |
| 12–14 weeks after birth |
| [63,251] | |
| 2 years after birth | Increased heart weight in embryo culture with serum compared to without serum group (0.29 ± 0.02 vs. 0.20 ± 0.01, p < 0.05). | [54] |
6.3.1. Evidence from Ruminants
6.3.2. Evidence from Rodents
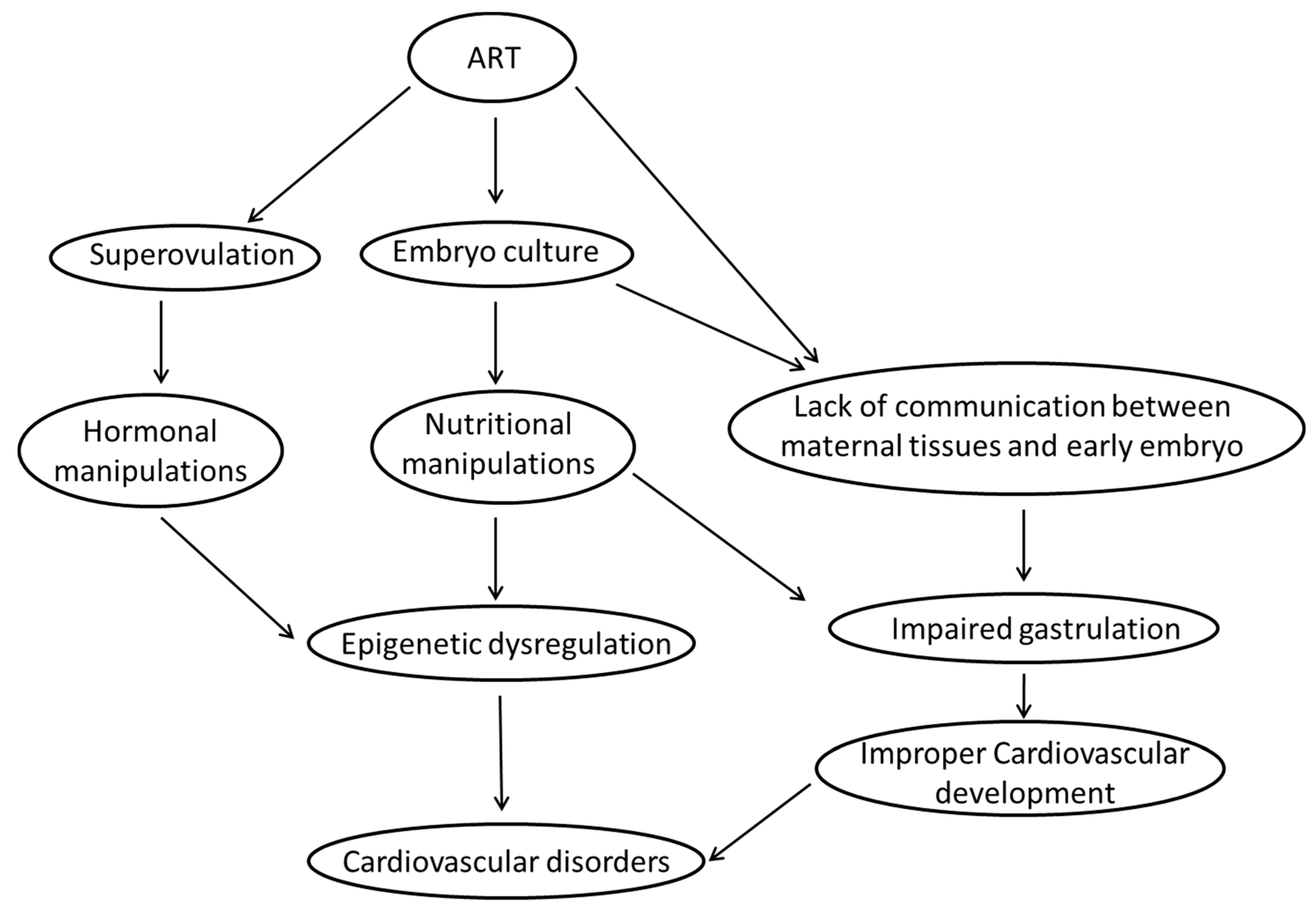
7. What Are the Most Likely Mechanisms Linking ART and Risk of Cardiovascular Disease in Fetal and Adult Life?
7.1. Epigenetic Dysregulation
7.1.1. Ovarian Hyperstimulation/Superovulation
7.1.2. In Vitro Embryo Culture
7.2. Impaired Gastrulation
8. Conclusions
Acknowledgments
Conflicts of Interest
References
- Jurema, M.W.; Nogueira, D. In vitro maturation of human oocytes for assisted reproduction. Fertil. Steril. 2006, 86, 1277–1291. [Google Scholar] [CrossRef] [PubMed]
- Fleming, T.P.; Kwong, W.Y.; Porter, R.; Ursell, E.; Fesenko, I.; Wilkins, A.; Miller, D.J.; Watkins, A.J.; Eckert, J.J. The embryo and its future. Biol. Reprod. 2004, 71, 1046–1054. [Google Scholar] [CrossRef] [PubMed]
- Louis, G.M.B.; Cooney, M.A.; Lynch, C.D.; Handal, A. Periconception window: Advising the pregnancy-planning couple. Fertil. Steril. 2008, 89, e119–e121. [Google Scholar] [CrossRef] [PubMed]
- Czeizel, A.E. Prevention of congenital abnormalities by periconceptional multivitamin supplementation. BMJ 1993, 306, 1645–1648. [Google Scholar] [CrossRef] [PubMed]
- Van Uitert, E.M.; van der Elst-Otte, N.; Wilbers, J.J.; Exalto, N.; Willemsen, S.P.; Eilers, P.H.C.; Koning, A.H.J.; Steegers, E.A.P.; Steegers-Theunissen, R.P.M. Periconception maternal characteristics and embryonic growth trajectories: The Rotterdam Predict Study. Hum. Reprod. 2013, 28, 3188–3196. [Google Scholar]
- Schelbach, C.J.; Robker, R.L.; Bennett, B.D.; Gauld, A.D.; Thompson, J.G.; Kind, K.L. Altered pregnancy outcomes in mice following treatment with the hyperglycaemia mimetic, glucosamine, during the periconception period. Reprod. Fertil. Dev. 2013, 25, 405–416. [Google Scholar] [CrossRef] [PubMed]
- Watkins, A.J.; Lucas, E.S.; Wilkins, A.; Cagampang, F.R.A.; Fleming, T.P. Maternal periconceptional and gestational low protein diet affects mouse offspring growth, cardiovascular and adipose phenotype at 1 year of age. PLoS One 2011, 6, e28745. [Google Scholar] [CrossRef] [PubMed]
- Maloney, C.A.; Hay, S.M.; Reid, M.D.; Duncan, G.; Nicol, F.; Sinclair, K.D.; Rees, W.D. A methyl-deficient diet fed to rats during the pre- and peri-conception periods of development modifies the hepatic proteome in the adult offspring. Genes Nutr. 2013, 8, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Kwong, W.Y.; Miller, D.J.; Ursell, E.; Wild, A.E.; Wilkins, A.P.; Osmond, C.; Anthony, F.W.; Fleming, T.P. Imprinted gene expression in the rat embryo-fetal axis is altered in response to periconceptional maternal low protein diet. Reproduction 2006, 132, 265–277. [Google Scholar] [CrossRef] [PubMed]
- Bloomfield, F.H. Periconceptional undernutrition in sheep accelerates maturation of the fetal hypothalamic-pituitary-adrenal axis in late gestation. Endocrinology 2004, 145, 4278–4278. [Google Scholar] [CrossRef] [PubMed]
- Oliver, M.H.; Hawkins, P.; Harding, J.E. Periconceptional undernutrition alters growth trajectory and metabolic and endocrine responses to fasting in late-gestation fetal sheep. Pediatr. Res. 2005, 57, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Spencer, T.E.; Johnson, G.A.; Bazer, F.W.; Burghardt, R.C. Implantation mechanisms: Insights from the sheep. Reproduction 2004, 128, 657–668. [Google Scholar] [CrossRef] [PubMed]
- Gardner, D.S.; van Bon, B.W.M.; Dandrea, J.; Goddard, P.J.; May, S.F.; Wilson, V.; Stephenson, T.; Symonds, M.E. Effect of periconceptional undernutrition and gender on hypothalamic-pituitary-adrenal axis function in young adult sheep. J. Endocrinol. 2006, 190, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Bazer, F.W.; Wu, G.; Johnson, G.A.; Kim, J.; Song, G. Uterine histotroph and conceptus development: select nutrients and secreted phosphoprotein 1 affect mechanistic target of rapamycin cell signaling in ewes. Biol. Reprod. 2011, 85, 1094–1107. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Rattanatray, L.; Morrison, J.L.; Nicholas, L.M.; Lie, S.; McMillen, I.C. Maternal obesity and the early origins of childhood obesity: Weighing up the benefits and costs of maternal weight loss in the periconceptional period for the offspring. Exp. Diabetes Res. 2011, 2011, 10. [Google Scholar] [CrossRef]
- Edwards, L.J.; McFarlane, J.R.; Kauter, K.G.; McMillen, I.C. Impact of periconceptional nutrition on maternal and fetal leptin and fetal adiposity in singleton and twin pregnancies. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2005, 288, R39–45. [Google Scholar] [CrossRef] [PubMed]
- Torrens, C.; Snelling, T.H.; Chau, R.; Shanmuganathan, M.; Cleal, J.K.; Poore, K.R.; Noakes, D.E.; Poston, L.; Hanson, M.A.; Green, L.R. Effects of pre- and periconceptional undernutrition on arterial function in adult female sheep are vascular bed dependent. Exp. Physiol. 2009, 94, 1024–1033. [Google Scholar] [CrossRef] [PubMed]
- Edwards, L.J.; McMillen, I.C. Impact of maternal undernutrition during the periconceptional period, fetal number, and fetal sex on the development of the hypothalamo-pituitary adrenal axis in sheep during late gestation. Biol. Reprod. 2002, 66, 1562–1569. [Google Scholar] [CrossRef] [PubMed]
- MacLaughlin, S.M.; Mühlhäusler, B.S.; Gentili, S.; McMillen, I.C. When in gestation do nutritional alterations exert their effects? A focus on the early origins of adult disease. Curr. Opin. Endocrinol. Diabetes Obes. 2006, 13, 516–522. [Google Scholar] [CrossRef]
- McMillen, I.C.; MacLaughlin, S.M.; Muhlhausler, B.S.; Gentili, S.; Duffield, J.L.; Morrison, J.L. Developmental origins of adult health and disease: The role of periconceptional and foetal nutrition. Basic Clin. Pharmacol. Toxicol. 2008, 102, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Bachvarova, R. Gene expression during oogenesis and oocyte development in mammals. In Oogenesis; Browder, L., Ed.; Springer US: New York, NY, USA, 1985; Volume 1, pp. 453–524. [Google Scholar]
- Eddy, E.; Clark, J.M.; Gong, D.; Fenderson, B.A. Origin and migration of primordial germ cells in mammals. Gamete Res. 1981, 4, 333–362. [Google Scholar] [CrossRef]
- Schoenwolf, G.C.; Bleyl, S.; Brauer, P.R.; Francis-West, P.H. Larsen’s Human Embryology; Churchill Livingstone/Elsevier Philadelphia: Philadelphia, PA, USA, 2009. [Google Scholar]
- Carlson, B.M. Human Embryology and Developmental Biology; Elsevier Health Sciences: Philadelphia, PA, USA, 2013. [Google Scholar]
- Wilcox, A.J. Fertility and Pregnancy: An Epidemiologic Perspective: An Epidemiologic Perspective; Oxford University Press: USA: New York, NY, USA, 2010. [Google Scholar]
- Gougeon, A. Regulation of ovarian follicular development in primates: Facts and hypotheses. Endocr. Rev. 1996, 17, 121–155. [Google Scholar] [CrossRef] [PubMed]
- Bernal, A.B.; Vickers, M.H.; Hampton, M.B.; Poynton, R.A.; Sloboda, D.M. Maternal undernutrition significantly impacts ovarian follicle number and increases ovarian oxidative stress in adult rat offspring. PLoS One 2010, 5, e15558. [Google Scholar] [CrossRef] [PubMed]
- Connor, K.L.; Vickers, M.H.; Beltrand, J.; Meaney, M.J.; Sloboda, D.M. Nature, nurture or nutrition? Impact of maternal nutrition on maternal care, offspring development and reproductive function. J. Physiol. 2012, 590, 2167–2180. [Google Scholar] [CrossRef] [PubMed]
- Sloboda, D.M.; Hickey, M.; Hart, R. Reproduction in females: The role of the early life environment. Hum. Reprod. Update 2011, 17, 210–227. [Google Scholar] [CrossRef] [PubMed]
- Guzmán, C.; Cabrera, R.; Cárdenas, M.; Larrea, F.; Nathanielsz, P.W.; Zambrano, E. Protein restriction during fetal and neonatal development in the rat alters reproductive function and accelerates reproductive ageing in female progeny. J. Physiol. 2006, 572, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Intapad, S.; Alexander, B.T. The double hit of growth restriction: Its origins and outcome on this generation and the next. J. Physiol. 2012, 590, 1019–1019. [Google Scholar] [CrossRef] [PubMed]
- Gallo, L.A.; Tran, M.; Moritz, K.M.; Jefferies, A.J.; Wlodek, M.E. Pregnancy in aged rats that were born small: Cardiorenal and metabolic adaptations and second-generation fetal growth. FASEB J. 2012, 26, 4337–4347. [Google Scholar] [CrossRef] [PubMed]
- Torrens, C.; Poston, L.; Hanson, M.A. Transmission of raised blood pressure and endothelial dysfunction to the F2 generation induced by maternal protein restriction in the F0, in the absence of dietary challenge in the F1 generation. Br. J. Nutr. 2008, 100, 760–766. [Google Scholar] [CrossRef] [PubMed]
- Benyshek, D.C.; Johnston, C.S.; Martin, J.F.; Ross, W.D. Insulin sensitivity is normalized in the third generation (F3) offspring of developmentally programmed insulin resistant (F2) rats fed an energy-restricted diet. Nutr. Metab. (Lond.) 2008, 5, 26. [Google Scholar] [CrossRef]
- Kott, R. Montana farm flock sheep production handbook. In Animal & Range Sciences, Extension Service; Montana State University: Bozeman, MT, USA, 2006. [Google Scholar]
- Wilcox, A.J.; Dunson, D.; Baird, D.D. The timing of the “fertile window” in the menstrual cycle: Day specific estimates from a prospective study. BMJ 2000, 321, 1259–1262. [Google Scholar] [CrossRef] [PubMed]
- Long, J.A.; Evans, H.M. The Oestrous Cycle in the Rat and Its Associated Phenomena; University of California Press: Berkeley, CA, USA, 1922; Volume 6. [Google Scholar]
- Allen, E. The oestrous cycle in the mouse. Am. J. Anat. 1922, 30, 297–371. [Google Scholar] [CrossRef]
- Fortune, J.E. Ovarian follicular growth and development in mammals. Biol. Reprod. 1994, 50, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Hirshfield, A.N. Development of follicles in the mammalian ovary. Int. Rev. Cytol. 1991, 124, 43–101. [Google Scholar] [PubMed]
- McGee, E.A.; Hsueh, A.J. Initial and cyclic recruitment of ovarian follicles. Endocr. Rev. 2000, 21, 200–214. [Google Scholar] [PubMed]
- Wassarman, P.M. Zona Pellucida Glycoproteins. Annu. Rev. Biochem. 1988, 57, 415–442. [Google Scholar] [CrossRef] [PubMed]
- Hennet, M.L.; Combelles, C.M. The antral follicle: A microenvironment for oocyte differentiation. Int. J. Dev. Biol. 2012, 56, 819–831. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, R.J.; Irving-Rodgers, H.F. Formation of the ovarian follicular antrum and follicular fluid. Biol. Reprod. 2010, 82, 1021–1029. [Google Scholar] [CrossRef] [PubMed]
- Hill, M.A. Embryology Fertilization. Available online: https://php.med.unsw.edu.au/embryology/index.php?title=Fertilization (accessed on 18 November 2014).
- Senger, P.L. Pathways to Pregnancy and Parturition, 1st ed.; Current Conceptions, Inc.: Pullman, WA, USA, 1999; pp. 206–246. [Google Scholar]
- Krisher, R.L. The effect of oocyte quality on development. J. Anim. Sci. 2004, 82, E14–E23. [Google Scholar] [PubMed]
- Ertzeid, G.; Storeng, R. The impact of ovarian stimulation on implantation and fetal development in mice. Hum. Reprod. 2001, 16, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Van der Auwera, I.; D’Hooghe, T. Superovulation of female mice delays embryonic and fetal development. Hum. Reprod. 2001, 16, 1237–1243. [Google Scholar] [CrossRef] [PubMed]
- Kelley-Quon, L.I.; Tseng, C.-H.; Janzen, C.; Shew, S.B. Congenital malformations associated with assisted reproductive technology: A California statewide analysis. J. Pediatr. Surg. 2013, 48, 1218–1224. [Google Scholar] [CrossRef] [PubMed]
- Ceelen, M.; van Weissenbruch, M.M.; Vermeiden, J.P.W.; van Leeuwen, F.E.; Delemarre-van de Waal, H.A. Growth and development of children born after in vitro fertilization. Fertil. Steril. 2008, 90, 1662–1673. [Google Scholar] [CrossRef] [PubMed]
- Sutcliffe, A.G.; Ludwig, M. Outcome of assisted reproduction. Lancet 2007, 370, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Stouder, C.; Deutsch, S.; Paoloni-Giacobino, A. Superovulation in mice alters the methylation pattern of imprinted genes in the sperm of the offspring. Reprod. Toxicol. 2009, 28, 536–541. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Gonzalez, R.; Moreira, P.; Bilbao, A.; Jiménez, A.; Pérez-Crespo, M.; Ramírez, M.A.; de Fonseca, F.R.; Pintado, B.; Gutiérrez-Adán, A. Long-term effect of in vitro culture of mouse embryos with serum on mRNA expression of imprinting genes, development, and behavior. Proc. Natl. Acad. Sci. USA 2004, 101, 5880–5885. [Google Scholar] [CrossRef] [PubMed]
- Mainigi, M.A.; Olalere, D.; Burd, I.; Sapienza, C.; Bartolomei, M.; Coutifaris, C. Peri-implantation hormonal milieu: Elucidating mechanisms of abnormal placentation and fetal growth. Biol. Reprod. 2014, 90, 26. [Google Scholar] [CrossRef] [PubMed]
- Fauque, P.; Mondon, F.; Letourneur, F.; Ripoche, M.-A.; Journot, L.; Barbaux, S.; Dandolo, L.; Patrat, C.; Wolf, J.-P.; Jouannet, P.; et al. In vitro fertilization and embryo culture strongly impact the placental transcriptome in the mouse model. PLoS One 2010, 5, e9218. [Google Scholar] [CrossRef] [PubMed]
- Alikani, M.; Calderon, G.; Tomkin, G.; Garrisi, J.; Kokot, M.; Cohen, J. Cleavage anomalies in early human embryos and survival after prolonged culture in vitro. Hum. Reprod. 2000, 15, 2634–2643. [Google Scholar] [CrossRef] [PubMed]
- Young, L.E.; Fernandes, K.; McEvoy, T.G.; Butterwith, S.C.; Gutierrez, C.G.; Carolan, C.; Broadbent, P.J.; Robinson, J.J.; Wilmut, I.; Sinclair, K.D. Epigenetic change in IGF2R is associated with fetal overgrowth after sheep embryo culture. Nat. Genet. 2001, 27, 153–154. [Google Scholar] [CrossRef] [PubMed]
- Watkins, A.J.; Fleming, T.P. Blastocyst environment and its influence on offspring cardiovascular health: The heart of the matter. J. Anat. 2009, 215, 52–59. [Google Scholar] [CrossRef] [PubMed]
- McEvoy, T.G.; Sinclair, K.D.; Young, L.E.; Wilmut, I.; Robinson, J.J. Large offspring syndrome and other consequences of ruminant embryo culture in vitro: Relevance to blastocyst culture in human ART. Hum. Fertil. 2000, 3, 238–246. [Google Scholar] [CrossRef]
- Khosla, S.; Dean, W.; Brown, D.; Reik, W.; Feil, R. Culture of preimplantation mouse embryos affects fetal development and the expression of imprinted genes. Biol. Reprod. 2001, 64, 918–926. [Google Scholar] [CrossRef] [PubMed]
- Watkins, A.J.; Platt, D.; Papenbrock, T.; Wilkins, A.; Eckert, J.J.; Kwong, W.Y.; Osmond, C.; Hanson, M.; Fleming, T.P. Mouse embryo culture induces changes in postnatal phenotype including raised systolic blood pressure. Proc. Natl. Acad. Sci. USA 2007, 104, 5449–5454. [Google Scholar] [CrossRef] [PubMed]
- Rimoldi, S.F.; Sartori, C.; Rexhaj, E.; Cerny, D.; Von Arx, R.; Soria, R.; Germond, M.; Allemann, Y.; Scherrer, U. Vascular dysfunction in children conceived by assisted reproductive technologies: Underlying mechanisms and future implications. Swiss Med. Wkly. 2014, 144, w13973. [Google Scholar] [PubMed]
- Valbuena, D.; Jasper, M.; Remohí, J.; Pellicer, A.; Simón, C. Ovarian stimulation and endometrial receptivity. Hum. Reprod. 1999, 14, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Alonso, M.; Blesa, D.; Simón, C. The genomics of the human endometrium. Biochim. Biophys. Acta—Mol. Basis Dis. 2012, 1822, 1931–1942. [Google Scholar] [CrossRef]
- Rahman, A.; Abdullah, R.; Wan-Khadijah, W. Gametogenesis, fertilization and early embryogenesis in mammals with special reference to goat: A review. J. Biol. Sci. 2008, 8, 1115–1128. [Google Scholar] [CrossRef]
- Ecochard, R.; Gougeon, A. Side of ovulation and cycle characteristics in normally fertile women. Hum. Reprod. 2000, 15, 752–755. [Google Scholar] [CrossRef] [PubMed]
- Minami, N.; Suzuki, T.; Tsukamoto, S. Zygotic Gene Activation and Maternal Factors in Mammals. J. Reprod. Dev. 2007, 53, 707–715. [Google Scholar] [CrossRef] [PubMed]
- Zernicka-Goetz, M. Activation of embryonic genes during preimplantation rat development. Mol. Reprod. Dev. 1994, 38, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Young, W.C.; Boling, J.L.; Blandau, R.J. The vaginal smear picture, sexual receptivity and time of ovulation in the albino rat. Anat. Rec. 1941, 80, 37–45. [Google Scholar] [CrossRef]
- Green, E.L. Biology of the laboratory mouse; Dover Publications Inc.: New York, NY, USA, 1966. [Google Scholar]
- Hill, M.A. Embryology Rat Development. Available online: https://php.med.unsw.edu.au/embryology/index.php?title=Rat_Development (accessed on 20 November 2014).
- Hill, M.A. Embryology Mouse Development. Available online: https://php.med.unsw.edu.au/embryology/index.php?title=Mouse_Development (accessed on 20 November 2014).
- Spencer, T.E.; Johnson, G.A.; Bazer, F.W.; Burghardt, R.C.; Palmarini, M. Pregnancy recognition and conceptus implantation in domestic ruminants: Roles of progesterone, interferons and endogenous retroviruses. Reprod. Fertil. Dev. 2006, 19, 65–78. [Google Scholar] [CrossRef]
- Schultz, R.M. The molecular foundations of the maternal to zygotic transition in the preimplantation embryo. Hum. Reprod. Update 2002, 8, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Schultz, R.M. Regulation of zygotic gene activation in the mouse. Bioessays 1993, 15, 531–538. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Lu, X.; Dean, J. The Maternal to Zygotic Transition in Mammals. Mol. Asp. Med. 2013, 34, 919–938. [Google Scholar] [CrossRef]
- Kafri, T.; Ariel, M.; Brandeis, M.; Shemer, R.; Urven, L.; McCarrey, J.; Cedar, H.; Razin, A. Developmental pattern of gene-specific DNA methylation in the mouse embryo and germ line. Genes Dev. 1992, 6, 705–714. [Google Scholar] [CrossRef] [PubMed]
- Stojanov, T.; O’Neill, C. Ontogeny of expression of a receptor for platelet-activating factor in mouse preimplantation embryos and the effects of fertilization and culture in vitro on its expression. Biol. Reprod. 1999, 60, 674–682. [Google Scholar] [CrossRef] [PubMed]
- Stojanov, T.; O’Neill, C. In vitro fertilization causes epigenetic modifications to the onset of gene expression from the zygotic genome in mice. Biol. Reprod. 2001, 64, 696–705. [Google Scholar] [CrossRef] [PubMed]
- Van Voorhis, B.J. In vitro fertilization. N. Engl. J. Med. 2007, 356, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Abyholm, T.; Tanbo, T. GIFT, ZIFT, and related techniques. Curr. Opin. Obstet. Gynecol. 1993, 5, 615–622. [Google Scholar] [PubMed]
- Sutherland, J.E.; Costa, M.A.X. Epigenetics and the environment. Ann. N. Y. Acad. Sci. 2003, 983, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Egger, G.; Liang, G.; Aparicio, A.; Jones, P.A. Epigenetics in human disease and prospects for epigenetic therapy. Nature 2004, 429, 457–463. [Google Scholar] [CrossRef] [PubMed]
- Tchurikov, N.A. Molecular mechanisms of epigenetics. Biochemistry (Moscow) 2005, 70, 406–423. [Google Scholar] [CrossRef]
- Gu, L.; Wang, Q.; Sun, Q.Y. Histone modifications during mammalian oocyte maturation: Dynamics, regulation and functions. Cell Cycle 2010, 9, 1942–1950. [Google Scholar] [CrossRef] [PubMed]
- Reik, W.; Dean, W.; Walter, J. Epigenetic reprogramming in mammalian development. Science 2001, 293, 1089–1093. [Google Scholar] [CrossRef] [PubMed]
- Morgan, H.D.; Santos, F.; Green, K.; Dean, W.; Reik, W. Epigenetic reprogramming in mammals. Hum. Mol. Genet. 2005, 14, R47–R58. [Google Scholar] [CrossRef] [PubMed]
- Santos, F.; Dean, W. Epigenetic reprogramming during early development in mammals. Reproduction 2004, 127, 643–651. [Google Scholar] [CrossRef] [PubMed]
- Mayer, W.; Niveleau, A.; Walter, J.; Fundele, R.; Haaf, T. Embryogenesis: Demethylation of the zygotic paternal genome. Nature 2000, 403, 501–502. [Google Scholar] [CrossRef] [PubMed]
- Allegrucci, C.; Thurston, A.; Lucas, E.; Young, L. Epigenetics and the germline. Reproduction 2005, 129, 137–149. [Google Scholar] [CrossRef] [PubMed]
- Li, E.; Beard, C.; Jaenisch, R. Role for DNA methylation in genomic imprinting. Nature 1993, 366, 362–365. [Google Scholar] [CrossRef] [PubMed]
- Reik, W.; Walter, J. Genomic imprinting: Parental influence on the genome. Nat. Rev. Genet. 2001, 2, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Osorio, N.; Dogan, S.; Memili, E. Epigenetics of mammalian gamete and embryo development. In Livestock Epigenetics, 1st ed.; Hassan, K., Ed.; Wiley-Blackwell: Oxford, UK, 2011; pp. 3–25. [Google Scholar]
- De Rycke, M.; Liebaers, I.; van Steirteghem, A. Epigenetic risks related to assisted reproductive technologies: Risk analysis and epigenetic inheritance. Hum. Reprod. 2002, 17, 2487–2494. [Google Scholar] [CrossRef] [PubMed]
- Reik, W.; Constância, M.; Fowden, A.; Anderson, N.; Dean, W.; Ferguson-Smith, A.; Tycko, B.; Sibley, C. Regulation of supply and demand for maternal nutrients in mammals by imprinted genes. J. Physiol. 2003, 547, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Lambertini, L.; Marsit, C.J.; Sharma, P.; Maccani, M.; Ma, Y.; Hu, J.; Chen, J. Imprinted gene expression in fetal growth and development. Placenta 2012, 33, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Abu-Amero, S.; Monk, D.; Apostolidou, S.; Stanier, P.; Moore, G. Imprinted genes and their role in human fetal growth. Cytogenet. Genome Res. 2006, 113, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.G. Genomic imprinting: Review and relevance to human diseases. Am. J. Hum. Genet. 1990, 46, 857–873. [Google Scholar] [PubMed]
- Amor, D.J.; Halliday, J. A review of known imprinting syndromes and their association with assisted reproduction technologies. Hum. Reprod. 2008, 23, 2826–2834. [Google Scholar] [CrossRef] [PubMed]
- Dominguez-Salas, P.; Moore, S.E.; Baker, M.S.; Bergen, A.W.; Cox, S.E.; Dyer, R.A.; Fulford, A.J.; Guan, Y.; Laritsky, E.; Silver, M.J.; et al. Maternal nutrition at conception modulates DNA methylation of human metastable epialleles. Nat. Commun. 2014, 5. [Google Scholar] [CrossRef]
- Niemitz, E.L.; Feinberg, A.P. Epigenetics and assisted reproductive technology: A call for investigation. Am. J. Hum. Genet. 2004, 74, 599–609. [Google Scholar] [CrossRef] [PubMed]
- Schieve, L.A.; Meikle, S.F.; Ferre, C.; Peterson, H.B.; Jeng, G.; Wilcox, L.S. Low and very low birth weight in infants conceived with use of assisted reproductive technology. N. Engl. J. Med. 2002, 346, 731–737. [Google Scholar] [CrossRef] [PubMed]
- Zegers-Hochschild, F.; Adamson, G.D.; de Mouzon, J.; Ishihara, O.; Mansour, R.; Nygren, K.; Sullivan, E.; van der Poel, S. The International Committee for Monitoring Assisted Reproductive Technology (ICMART) and the World Health Organization (WHO) revised glossary on ART terminology, 2009. Hum. Reprod. 2009, 24, 2683–2687. [Google Scholar] [CrossRef] [PubMed]
- Heape, W. Preliminary note on the transplantation and grwoth of mammalian ova within a uterine foster mother. Proc. R. Soc. 1890, 48, 457–458. [Google Scholar] [CrossRef]
- Betteridge, K.J. Reflections on the golden anniversary of the first embryo transfer to produce a calf. Theriogenology 2000, 53, 3–10. [Google Scholar] [CrossRef] [PubMed]
- De Kretzer, D.; Dennis, P.; Hudson, B.; Leeton, J.; Lopata, A.; Outch, K.; Talbot, J.; Wood, C. Transfer of a human zygote. Lancet 1973, 302, 728–729. [Google Scholar] [CrossRef]
- Steptoe, P.C.; Edwards, R.G. Birth after the reimplantation of a human embryo. Lancet 1978, 312, 366–366. [Google Scholar] [CrossRef]
- Kamel, R.M. Assisted reproductive technology after the birth of louise brown. J. Reprod. Infertil. 2013, 14, 96–109. [Google Scholar] [PubMed]
- ART Fact Sheet. Available online: http://www.eshre.eu/sitecore/content/Home/Guidelines%20and%20Legal/ART%20fact%20sheet (accessed on 16 November 2014).
- Balasch, J. Investigation of the infertile couple: Investigation of the infertile couple in the era of assisted reproductive technology: A time for reappraisal. Hum. Reprod. 2000, 15, 2251–2257. [Google Scholar] [CrossRef] [PubMed]
- Wyndham, N.; Marin Figueira, P.G.; Patrizio, P. A persistent misperception: Assisted reproductive technology can reverse the “aged biological clock”. Fertil. Steril. 2012, 97, 1044–1047. [Google Scholar] [CrossRef] [PubMed]
- Hilder, L.; Zhichao, Z.; Parker, M.; Jahan, S.; Chambers, G.M. Australia's mothers and babies 2012; Perinatal statistics series no. 30. Cat. no. PER 69; Australian Institute of Health and Welfare and University: Canberra, Australia, 2014. [Google Scholar]
- Nash, M. Making ‘Postmodern’ Mothers: Pregnant Embodiment, Baby Bumps and Body Image; Palgrave Macmillan: England, UK, 2012. [Google Scholar]
- Australian Institute of Family Studies. Available online: http://www.aifs.gov.au/institute/info/charts/births/#age (accessed on 16 November 2014).
- Martin, J.A.; Hamilton, B.E.; Osterman, M.J.K.; Curtin, S.C.; Mathews, T.J. Births: Final Data for 2012; National Vital Statistics Reports, Vol 62 No 9; National Center for Health Statistics: Hyattsville, MD, USA, 30 December 2013.
- Millan, A. Fertility: Overview, 2008; Report on the Demographic Situation in Canada, Catalogue no. 91-209-X.; Authority of the Minister responsible for Statistics Canada: Ottawa, Canada, 2011 August.
- Dunson, D.B.; Colombo, B.; Baird, D.D. Changes with age in the level and duration of fertility in the menstrual cycle. Hum. Reprod. 2002, 17, 1399–1403. [Google Scholar] [CrossRef] [PubMed]
- Van Noord-Zaadstra, B.M.; Looman, C.W.; Alsbach, H.; Habbema, J.D.; te Velde, E.R.; Karbaat, J. Delaying childbearing: Effect of age on fecundity and outcome of pregnancy. BMJ 1991, 302, 1361. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.A.; Killick, S.R. Effect of male age on fertility: Evidence for the decline in male fertility with increasing age. Fertil. Steril. 2003, 79, 1520–1527. [Google Scholar] [CrossRef] [PubMed]
- Leridon, H. Can assisted reproduction technology compensate for the natural decline in fertility with age? A model assessment. Hum. Reprod. 2004, 19, 1548–1553. [Google Scholar] [CrossRef] [PubMed]
- Macaldowie, A.; Wang, Y.A.; Chambers, G.M.; Sullivan, E.A. Assisted Reproductive Technology in Australia and New Zealand 2012; AIHW: Canberra, Australia, 2012. [Google Scholar]
- Moore, K.; Thatcher, W.W. Major advances associated with reproduction in dairy cattle. J. Dairy Sci. 2006, 89, 1254–1266. [Google Scholar] [CrossRef] [PubMed]
- Sejian, V.; Meenambigai, T.V.; Chandirasegaran, M.; Naqvi, S.M.K. reproductive technology in farm animals: New facets and findings: A review. J. Biol. Sci. 2010, 10, 686–700. [Google Scholar] [CrossRef]
- Chakravarthi, P.; Balaji, N. Use of assisted reproductive technologies for livestock development. Vet. World 2010, 3, 238–240. [Google Scholar]
- Amiridis, G.S.; Cseh, S. Assisted reproductive technologies in the reproductive management of small ruminants. Anim. Reprod. Sci. 2012, 130, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Bertolini, M.; Bertolini, L.R. Advances in reproduc tive technologies in cattle: From artificial insemination to cloning. Rev. Med. Vet. Zoot. 2009, 56, 184–194. [Google Scholar]
- Solti, L.; Crichton, E.G.; Loskutoff, N.M.; Cseh, S. Economical and ecological importance of indigenous livestock and the application of assisted reroduction to their preservation. Theriogenology 2000, 53, 149–162. [Google Scholar] [CrossRef] [PubMed]
- Barbieri, E.; kashyap, S.; Chung, P. Infertility and in vitro fertilization. In Priniciples of Gender Specific Medicine, 2nd ed.; Legato, M.J., Ed.; Academic Press: San Diego, CA, USA, 2010; pp. 381–398. [Google Scholar]
- Tanbo, T.; Omland, A.; Dale, P.O.; Åbyholm, T. In vitro fertilization/embryo transfer in unexplained infertility and minimal peritoneal endometriosis. Acta Obstet. Gynecol. Scand. 1995, 74, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Chandler, C.J.; Harris, C.P. Gamete intrafallopian transfer. Br. J. Hosp. Med. 1996, 55, 468–471. [Google Scholar] [PubMed]
- Carnevale, E.M. Gamete intrafallopian transfer. Vet. Clin. N. Am. Equine Pract. 1996, 12, 47–60. [Google Scholar]
- Jansen, R.P.S. Gamete intra-fallopian transfer. In Clinical in Vitro Fertilization; Springer London: London, UK, 1989; pp. 63–80. [Google Scholar]
- Molloy, D.; Speirs, A.; Du Plessis, Y.; Gellert, S.; Bourne, H.; Johnston, W.I. Gamete intra-fallopian transfer. Med. J. Aust. 1985, 143, 428. [Google Scholar] [PubMed]
- Perone, N. Gamete intrafallopian transfer (GIFT): Historic perspective. J. Vitr. Fertil. Embryo Transf. 1991, 8, 1–4. [Google Scholar] [CrossRef]
- Asch, R.H.; Balmaceda, J.P.; Cittadini, E.; Casas, P.F.; Gomel, V.; Hohl, M.K.; Johnston, I.; Leeton, J.; Escudero, F.J.R.; Noss, U.; et al. Gamete intrafallopian transfer. Ann. N. Y. Acad. Sci. 1988, 541, 722–727. [Google Scholar] [CrossRef] [PubMed]
- Asch, R.H.; Balmaceda, J.P.; Ellsworth, L.R.; Wong, P.C. Gamete intra-fallopian transfer (GIFT): A new treatment for infertility. Int. J. Fertil. 1985, 30, 41–45. [Google Scholar] [PubMed]
- Hummel, W.P.; Kettel, L.M. Assisted reproductive technology: The state of the ART. Ann. Med. 1997, 29, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Devroey, P.; Staessen, C.; Camus, M.; de Grauwe, E.; Wisanto, A.; van Steirteghem, A.C. Zygote intrafallopian transfer as a successful treatment for unexplained infertility. Fertil. Steril. 1989, 52, 246–249. [Google Scholar] [PubMed]
- Hamori, M.; Stuckensen, J.; Rumpf, D.; Kniewald, T.; Kniewald, A.; Marquez, M. Zygote intrafallopian transfer (ZIFT): Evaluation of 42 cases. Fertil. Steril. 1988, 50, 519–521. [Google Scholar] [PubMed]
- Palermo, G.; Devroey, P.; Camus, M.; de Grauwe, E.; Khan, I.; Staessen, C.; Wisanto, A.; van Steirteghem, A.C. Zygote intra-Fallopian transfer as an alternative treatment for male infertility. Hum. Reprod. 1989, 4, 412–415. [Google Scholar] [PubMed]
- Simpson, R. Assisted Reproductive Technology; NSW Parliamentary Library Research Service: Sydney, Australia, 1998; pp. 1–65.
- Allen, V.M.; Wilson, R.D.; Cheung, A. Pregnancy outcomes after assisted reproductive technology. J. Obstet. Gynaecol. Can. 2006, 28, 220–250. [Google Scholar] [PubMed]
- Wang, Y.A.C.G.; Sullivan, E.A. Assisted Reproductive Technology in Australia and New Zealand 2008; AIHW: Canberra, Australia, 2010. [Google Scholar]
- Gianaroli, L.; Plachot, M.; van Kooij, R.; Al-Hasani, S.; Dawson, K.; DeVos, A.; Magli, M.C.; Mandelbaum, J.; Selva, J.; van Inzen, W. ESHRE guidelines for good practice in IVF laboratories. Committee of the special interest group on embryology of the european society of human reproduction and embryology. Hum. Reprod. 2000, 15, 2241–2246. [Google Scholar] [CrossRef] [PubMed]
- Engmann, L.; Maconochie, N.; Tan, S.L.; Bekir, J. Trends in the incidence of births and multiple births and the factors that determine the probability of multiple birth after IVF treatment. Hum. Reprod. 2001, 16, 2598–2605. [Google Scholar] [CrossRef] [PubMed]
- Pinborg, A. IVF/ICSI twin pregnancies: Risks and prevention. Hum. Reprod. Update 2005, 11, 575–593. [Google Scholar] [CrossRef] [PubMed]
- El-Toukhy, T.; Khalaf, Y.; Braude, P. IVF results: Optimize not maximize. Am. J. Obstet. Gynecol. 2006, 194, 322–331. [Google Scholar] [CrossRef] [PubMed]
- De Sutter, P.; van der Elst, J.; Coetsier, T.; Dhont, M. Single embryo transfer and multiple pregnancy rate reduction in IVF/ICSI: A 5-year appraisal. Reprod. BioMed. Online 2003, 6, 464–469. [Google Scholar] [CrossRef] [PubMed]
- Khalaf, Y.; El-Toukhy, T.; Coomarasamy, A.; Kamal, A.; Bolton, V.; Braude, P. Selective single blastocyst transfer reduces the multiple pregnancy rate and increases pregnancy rates: A pre- and postintervention study. BJOG 2008, 115, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Tiitinen, A.; Unkila-Kallio, L.; Halttunen, M.; Hyden-Granskog, C. Impact of elective single embryo transfer on the twin pregnancy rate. Hum. Reprod. 2003, 18, 1449–1453. [Google Scholar] [CrossRef] [PubMed]
- Menezes, J. Embryo culture systems. In Insights into Infertility Management, 2nd ed.; Jaypee Brothers Medical Publishers: New Delhi, India, 2012; pp. 188–198. [Google Scholar]
- Gardner, D.K.; Lane, M. Embryo culture systems. In In Vitro Fertilization: A Practical Approach; Gardner, D.K., Ed.; Informa Healthcare USA, Inc.: New York, NY, USA, 2000; pp. 221–264. [Google Scholar]
- Zander-Fox, D.; Lane, M. The Future of Human Embryo Culture Media–Or Have We Reached the Ceiling? In The Human Embryo; Yamada, S., Takakuwa, T., Eds.; InTech: Rijeka, Croatia, 2012; pp. 73–98. [Google Scholar]
- Quinn, P. Review of media used in art laboratories. J. Androl. 2000, 21, 610–615. [Google Scholar] [PubMed]
- Lane, M.; Gardner, D.K. Embryo culture medium: Which is the best? Best Pract. Res. Clin. Obstet. Gynaecol. 2007, 21, 83–100. [Google Scholar] [CrossRef] [PubMed]
- Quinn, P.; Kerin, J.F.; Warnes, G.M. Improved pregnancy rate in human in vitro fertilization with the use of a medium based on the composition of human tubal fluid. Fertil. Steril. 1985, 44, 493–498. [Google Scholar] [PubMed]
- Gardner, D.K.; Lane, M. Culture and selection of viable blastocysts: A feasible proposition for human IVF? Hum. Reprod. Update 1997, 3, 367–382. [Google Scholar] [CrossRef] [PubMed]
- Gardner, D.K. Mammalian embryo culture in the absence of serum or somatic cell support. Cell Biol. Int. 1994, 18, 1163–1180. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S. Media in ART. In Manual of Assisted Reproductive Technologies and Clinical Embryology, 2nd ed.; Jaypee Brothers Medical Publishers: New Delhi, India, 2012; pp. 44–57. [Google Scholar]
- Grace, K.S.; Sinclair, K.D. Assisted reproductive technology, epigenetics, and long-term health: A developmental time bomb still ticking. Semin. Reprod. Med. 2009, 27, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, K.D.; McEvoy, T.G.; Carolan, C.; Maxfield, E.K.; Maltin, C.A.; Young, L.E.; Wilmut, I.; Robinson, J.J.; Broadbent, P.J. Conceptus growth and development following in vitro culture of ovine embryos in media supplemented with bovine sera. Theriogenology 1998, 49, 218–218. [Google Scholar] [CrossRef]
- Sinclair, K.D.; McEvoy, T.G.; Maxfield, E.K.; Maltin, C.A.; Young, L.E.; Wilmut, I.; Broadbent, P.J.; Robinson, J.J. Aberrant fetal growth and development after in vitro culture of sheep zygotes. J. Reprod. Fertil. 1999, 116, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Gonzalez, R.; Ramirez, M.A.; Bilbao, A.; de Fonseca, F.R.; Gutiérrez-Adán, A. Suboptimal in vitro culture conditions: An epigenetic origin of long-term health effects. Mol. Reprod. Dev. 2007, 74, 1149–1156. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.G.; Gardner, D.K.; Pugh, P.A.; McMillan, W.H.; Tervit, H.R. Lamb birth weight is affected by culture system utilized during in vitro pre-elongation development of ovine embryos. Biol. Reprod. 1995, 53, 1385–1391. [Google Scholar] [CrossRef] [PubMed]
- Gardner, D.K.; Lane, M. Culture of viable human blastocysts in defined sequential serum-free media. Hum. Reprod. 1998, 13, 148–159; discussion 160. [Google Scholar] [CrossRef] [PubMed]
- Holst, N.; Bertheussen, K.; Forsdahl, F.; Hakonsen, M.B.; Hansen, L.J.; Nielsen, H.I. Optimization and simplification of culture conditions in human in vitro fertilization (IVF) and preembryo replacement by serum-free media. J. Vitr. Fertil. Embryo Transf. 1990, 7, 47–53. [Google Scholar] [CrossRef]
- Alwan, A. Global Status Report on Noncommunicable Diseases 2010; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- World Health Organization. Global Health Observatory (GHO) Data. Available online: http://www.who.int/gho/ncd/mortality_morbidity/cvd/en/ (accessed on 16 November 2014).
- Stampfer, M.J.; Hu, F.B.; Manson, J.E.; Rimm, E.B.; Willett, W.C. Primary Prevention of Coronary Heart Disease in Women through Diet and Lifestyle. N. Engl. J. Med. 2000, 343, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Mensah, G.A.; Mendis, S.; Greenland, K.; MacKay, J. The Atlas of Heart Disease and Stroke; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- McMillen, I.C.; Robinson, J.S. Developmental origins of the metabolic syndrome: prediction, plasticity, and programming. Physiol. Rev. 2005, 85, 571–633. [Google Scholar] [CrossRef] [PubMed]
- Barker, D.J. The developmental origins of adult disease. Eur. J. Epidemiol. 2003, 18, 733–736. [Google Scholar] [CrossRef] [PubMed]
- De Boo, H.A.; Harding, J.E. The developmental origins of adult disease (Barker) hypothesis. Aust. N. Z. J. Obstet. Gynaecol. 2006, 46, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Botting, K.J.; Wang, K.C.; Padhee, M.; McMillen, I.C.; Summers-Pearce, B.; Rattanatray, L.; Cutri, N.; Posterino, G.S.; Brooks, D.A.; Morrison, J.L. Early origins of heart disease: Low birth weight and determinants of cardiomyocyte endowment. Clin. Exp. Pharmacol. Physiol. 2012, 39, 814–823. [Google Scholar] [CrossRef] [PubMed]
- Gluckman, P.D.; Hanson, M.A.; Buklijas, T.; Low, F.M.; Beedle, A.S. Epigenetic mechanisms that underpin metabolic and cardiovascular diseases. Nat. Rev. Endocrinol. 2009, 5, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-P.; Bruneau, B.G. Epigenetics and cardiovascular development. Annu. Rev. Physiol. 2012, 74, 41–68. [Google Scholar] [CrossRef] [PubMed]
- Vallaster, M.; Vallaster, C.D.; Wu, S.M. Epigenetic mechanisms in cardiac development and disease. Acta Biochim. Biophys. Sin. 2012, 44, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Ordovás, J.M.; Smith, C.E. Epigenetics and cardiovascular disease. Nat. Rev. Cardiol. 2010, 7, 510–519. [Google Scholar] [CrossRef] [PubMed]
- Zak, R. Growth of the Heart in Health and Disease; Raven Press: New York, NY, USA, 1984. [Google Scholar]
- Nag, A.C. Study of non-muscle cells of the adult mammalian heart: a fine structural analysis and distribution. Cytobios 1980, 28, 41–61. [Google Scholar] [PubMed]
- Soonpaa, M.H.; Kim, K.K.; Pajak, L.; Franklin, M.; Field, L.J. Cardiomyocyte DNA synthesis and binucleation during murine development. Am. J. Physiol. 1996, 271, H2183–H2189. [Google Scholar] [PubMed]
- Oparil, S.; Bishop, S.P.; Clubb, F.J. Myocardial cell hypertrophy or hyperplasia. Hypertension 1984, 6, III38–III43. [Google Scholar] [CrossRef] [PubMed]
- Schmid, G.; Pfitzer, P. Mitoses and binucleated cells in perinatal human hearts. Virchows Arch. B 1985, 48, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Burrell, J.H.; Boyn, A.M.; Kumarasamy, V.; Hsieh, A.; Head, S.I.; Lumbers, E.R. Growth and maturation of cardiac myocytes in fetal sheep in the second half of gestation. Anat. Rec. A Discov. Mol. Cell. Evol. Biol. 2003, 274, 952–961. [Google Scholar] [CrossRef] [PubMed]
- Ahuja, P.; Sdek, P.; MacLellan, W.R. Cardiac Myocyte Cell Cycle Control in Development, Disease, and Regeneration. Physiol. Rev. 2007, 87, 521–544. [Google Scholar] [CrossRef] [PubMed]
- Jonker, S.S.; Zhang, L.; Louey, S.; Giraud, G.D.; Thornburg, K.L.; Faber, J.J. Myocyte enlargement, differentiation, and proliferation kinetics in the fetal sheep heart. J. Appl. Physiol. 2007, 102, 1130–1142. [Google Scholar] [CrossRef] [PubMed]
- Woodcock, E.A.; Matkovich, S.J. Cardiomyocytes structure, function and associated pathologies. Int. J. Biochem. Cell Biol. 2005, 37, 1746–1751. [Google Scholar] [CrossRef] [PubMed]
- Walker, S.K.; Hartwich, K.; Robinson, J.S. Long-term effects on offspring of exposure of oocytes and embryos to chemical and physical agents. Hum. Reprod. Update 2000, 6, 564–577. [Google Scholar] [CrossRef] [PubMed]
- Mollova, M.; Bersell, K.; Walsh, S.; Savla, J.; Das, L.T.; Park, S.-Y.; Silberstein, L.E.; dos Remedios, C.G.; Graham, D.; Colan, S.; et al. Cardiomyocyte proliferation contributes to heart growth in young humans. Proc. Natl. Acad. Sci. USA 2013, 110, 1446–1451. [Google Scholar] [CrossRef] [PubMed]
- Clubb, F.J., Jr.; Bishop, S.P. Formation of binucleated myocardial cells in the neonatal rat. An index for growth hypertrophy. Lab. Investig. 1984, 50, 571–577. [Google Scholar] [PubMed]
- Porrello, E.R.; Mahmoud, A.I.; Simpson, E.; Hill, J.A.; Richardson, J.A.; Olson, E.N.; Sadek, H.A. Transient regenerative potential of the neonatal mouse heart. Science 2011, 331, 1078–1080. [Google Scholar] [CrossRef] [PubMed]
- Walsh, S.; Pontén, A.; Fleischmann, B.K.; Jovinge, S. Cardiomyocyte cell cycle control and growth estimation in vivo—An analysis based on cardiomyocyte nuclei. Cardiovasc. Res. 2010, 86, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Wang, X.; Capasso, J.M.; Gerdes, A.M. Rapid transition of cardiac myocytes from hyperplasia to hypertrophy during postnatal development. J. Mol. Cell. Cardiol. 1996, 28, 1737–1746. [Google Scholar] [CrossRef] [PubMed]
- Roseboom, T.J.; van der Meulen, J.H.P.; Osmond, C.; Barker, D.J.P.; Ravelli, A.C.J.; Schroeder-Tanka, J.M.; van Montfrans, G.A.; Michels, R.P.J.; Bleker, O.P. Coronary heart disease after prenatal exposure to the Dutch famine, 1944–1945. Heart 2000, 84, 595–598. [Google Scholar] [CrossRef] [PubMed]
- Painter, R.C.; de Rooij, S.R.; Bossuyt, P.M.; Phillips, D.I.; Osmond, C.; Barker, D.J.; Bleker, O.P.; Roseboom, T.J. Blood pressure response to psychological stressors in adults after prenatal exposure to the Dutch famine. J. Hypertens. 2006, 24, 1771–1778. [Google Scholar] [CrossRef] [PubMed]
- Malik, S.; Cleves, M.A.; Honein, M.A.; Romitti, P.A.; Botto, L.D.; Yang, S.; Hobbs, C.A.; National Birth Defects Prevention, S. Maternal Smoking and Congenital Heart Defects. Pediatrics 2008, 121, e810–e816. [Google Scholar] [CrossRef] [PubMed]
- Karatza, A.A.; Giannakopoulos, I.; Dassios, T.G.; Belavgenis, G.; Mantagos, S.P.; Varvarigou, A.A. Periconceptional tobacco smoking and Xisolated congenital heart defects in the neonatal period. Int. J. Cardiol. 2011, 148, 295–299. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, S.L.; Shaw, G.M.; Yang, W.; Lammer, E.J. Maternal periconceptional alcohol consumption and risk for conotruncal heart defects. Birth Defects Res. Part A Clin. Mol. Teratol. 2003, 67, 875–878. [Google Scholar] [CrossRef] [PubMed]
- Van Driel, L.M.J.W.; Smedts, H.P.M.; Helbing, W.A.; Isaacs, A.; Lindemans, J.; Uitterlinden, A.G.; van Duijn, C.M.; de Vries, J.H.M.; Steegers, E.A.P.; Steegers-Theunissen, R.P.M. Eight-fold increased risk for congenital heart defects in children carrying the nicotinamide N-methyltransferase polymorphism and exposed to medicines and low nicotinamide. Eur. Heart J. 2008, 29, 1424–1431. [Google Scholar] [CrossRef] [PubMed]
- Kwong, W.Y.; Wild, A.E.; Roberts, P.; Willis, A.C.; Fleming, T.P. Maternal undernutrition during the preimplantation period of rat development causes blastocyst abnormalities and programming of postnatal hypertension. Development 2000, 127, 4195–4202. [Google Scholar] [PubMed]
- Watkins, A.J.; Wilkins, A.; Cunningham, C.; Perry, V.H.; Seet, M.J.; Osmond, C.; Eckert, J.J.; Torrens, C.; Cagampang, F.R.A.; Cleal, J.; et al. Low protein diet fed exclusively during mouse oocyte maturation leads to behavioural and cardiovascular abnormalities in offspring. J. Physiol. 2008, 586, 2231–2244. [Google Scholar] [CrossRef] [PubMed]
- Watkins, A.J.; Ursell, E.; Panton, R.; Papenbrock, T.; Hollis, L.; Cunningham, C.; Wilkins, A.; Perry, V.H.; Sheth, B.; Kwong, W.Y.; et al. Adaptive responses by mouse early embryos to maternal diet protect fetal growth but predispose to adult onset disease. Biol. Reprod. 2008, 78, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Edwards, L.J.; McMillen, I.C. Periconceptional nutrition programs development of the cardiovascular system in the fetal sheep. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2002, 283, R669–R679. [Google Scholar] [PubMed]
- Gardner, D.S.; Pearce, S.; Dandrea, J.; Walker, R.; Ramsay, M.M.; Stephenson, T.; Symonds, M.E. Peri-Implantation undernutrition programs blunted angiotensin ii evoked baroreflex responses in young adult sheep. Hypertension 2004, 43, 1290–1296. [Google Scholar] [CrossRef] [PubMed]
- Gopalakrishnan, G.S.; Gardner, D.S.; Rhind, S.M.; Rae, M.T.; Kyle, C.E.; Brooks, A.N.; Walker, R.M.; Ramsay, M.M.; Keisler, D.H.; Stephenson, T.; et al. Programming of adult cardiovascular function after early maternal undernutrition in sheep. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2004, 287, R12–R20. [Google Scholar] [CrossRef] [PubMed]
- Cleal, J.K.; Poore, K.R.; Boullin, J.P.; Khan, O.; Chau, R.; Hambidge, O.; Torrens, C.; Newman, J.P.; Poston, L.; Noakes, D.E.; et al. Mismatched pre- and postnatal nutrition leads to cardiovascular dysfunction and altered renal function in adulthood. Proc. Natl. Acad. Sci. USA 2007, 104, 9529–9533. [Google Scholar] [CrossRef] [PubMed]
- Jaquiery, A.L.; Oliver, M.H.; Honeyfield-Ross, M.; Harding, J.E.; Bloomfield, F.H. Periconceptional undernutrition in sheep affects adult phenotype only in males. J. Nutr. Metab. 2012, 2012, 7. [Google Scholar] [CrossRef]
- Chen, M.; Norman, R.J.; Heilbronn, L.K. Does in vitro fertilisation increase type 2 diabetes and cardiovascular risk? Curr. Diabetes Rev. 2011, 7, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, E. Long-term outcomes in children born after assisted conception. Semin. Reprod. Med. 2012, 30, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Hart, R.; Norman, R.J. The longer-term health outcomes for children born as a result of IVF treatment: Part I—General health outcomes. Hum. Reprod. Update 2013, 19, 232–243. [Google Scholar] [CrossRef] [PubMed]
- Hart, R.; Norman, R.J. The longer-term health outcomes for children born as a result of IVF treatment. Part II—Mental health and development outcomes. Hum. Reprod. Update 2013, 19, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, K.D.; Young, L.E.; Wilmut, I.; McEvoy, T.G. In-utero overgrowth in ruminants following embryo culture: Lessons from mice and a warning to men. Hum. Reprod. 2000, 15, 68–86. [Google Scholar] [CrossRef] [PubMed]
- Rebecca, C.P.; Susanne, R.D.R.; Patrick, M.B.; Timothy, A.S.; Clive, O.; David, J.B.; Otto, P.B.; Tessa, J.R. Early onset of coronary artery disease after prenatal exposure to the Dutch famine. Am. J. Clin. Nutr. 2006, 84, 322–327. [Google Scholar] [PubMed]
- Watkins, A.J.; Lucas, E.S.; Fleming, T.P. Impact of the periconceptional environment on the programming of adult disease. J. Dev. Orig. Health Dis. 2010, 1, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Taitson, P.F.; Kwong, D.D.; Lima, G.C.D.A.; Coelho, L.S.; Bruce, W.D.; Bernardes, N.D.O. Incidence and anatomy of cardiac malformations in children conceived by assisted reproduction techniques—A review. JBRA Assist. Reprod. 2014, 18, 52–54. [Google Scholar]
- Lancaster, P.A. Congenital malformations after in vitro fertilisation. Lancet 1987, 2, 1392–1393. [Google Scholar] [CrossRef] [PubMed]
- Olson, C.K.; Keppler-Noreuil, K.M.; Romitti, P.A.; Budelier, W.T.; Ryan, G.; Sparks, A.E.; van Voorhis, B.J. In vitro fertilization is associated with an increase in major birth defects. Fertil. Steril. 2005, 84, 1308–1315. [Google Scholar] [CrossRef] [PubMed]
- Davies, M.J.; Moore, V.M.; Willson, K.J.; van Essen, P.; Priest, K.; Scott, H.; Haan, E.A.; Chan, A. Reproductive technologies and the risk of birth defects. N. Engl. J. Med. 2012, 366, 1803–1813. [Google Scholar] [CrossRef] [PubMed]
- Hansen, M.; Kurinczuk, J.J.; Bower, C.; Webb, S. The risk of major birth defects after intracytoplasmic sperm injection and in vitro fertilization. N. Engl. J. Med. 2002, 346, 725–730. [Google Scholar] [CrossRef] [PubMed]
- Anthony, S.; Buitendijk, S.E.; Dorrepaal, C.A.; Lindner, K.; Braat, D.D.; den Ouden, A.L. Congenital malformations in 4224 children conceived after IVF. Hum. Reprod. 2002, 17, 2089–2095. [Google Scholar] [CrossRef] [PubMed]
- Klemetti, R.; Gissler, M.; Sevon, T.; Koivurova, S.; Ritvanen, A.; Hemminki, E. Children born after assisted fertilization have an increased rate of major congenital anomalies. Fertil. Steril. 2005, 84, 1300–1307. [Google Scholar] [CrossRef] [PubMed]
- Wen, S.W.; Leader, A.; White, R.R.; Leveille, M.-C.; Wilkie, V.; Zhou, J.; Walker, M.C. A comprehensive assessment of outcomes in pregnancies conceived by in vitro fertilization/intracytoplasmic sperm injection. Eur. J. Obstet. Gynecol. Reprod. Biol. 2010, 150, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Mozafari Kermani, R.; Nedaeifard, L.; Nateghi, M.R.; Shahzadeh Fazeli, A.; Ahmadi, E.; Osia, M.A.; Jafarzadehpour, E.; Nouri, S. Congenital anomalies in infants conceived by assisted reproductive techniques. Arch. Iran. Med. 2012, 15, 228–231. [Google Scholar] [PubMed]
- Koivurova, S.; Hartikainen, A.L.; Gissler, M.; Hemminki, E.; Sovio, U.; Jarvelin, M.R. Neonatal outcome and congenital malformations in children born after in vitro fertilization. Hum. Reprod. 2002, 17, 1391–1398. [Google Scholar] [CrossRef] [PubMed]
- Farhangniya, M.; Dortaj Rabori, E.; Mozafari Kermani, R.; Haghdoost, A.A.; Bahrampour, A.; Bagheri, P.; Lancestar, P.A.L.; Ashrafi, M.; Vosough Taqi Dizaj, A.; Gourabi, H.; et al. Comparison of congenital abnormalities of infants conceived by assisted reproductive techniques versus infants with natural conception in Tehran. Int. J. Fertil. Steril. 2013, 7, 217–224. [Google Scholar] [PubMed]
- Reefhuis, J.; Honein, M.A.; Schieve, L.A.; Correa, A.; Hobbs, C.A.; Rasmussen, S.A. Assisted reproductive technology and major structural birth defects in the United States. Hum. Reprod. 2009, 24, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Källén, B.; Finnström, O.; Nygren, K.G.; Olausson, P.O. In vitro fertilization (IVF) in Sweden: Risk for congenital malformations after different IVF methods. Birth Defects Res. Part A: Clin. Mol. Teratol. 2005, 73, 162–169. [Google Scholar] [CrossRef]
- Källén, B.; Finnström, O.; Lindam, A.; Nilsson, E.; Nygren, K.-G.; Otterblad, P.O. Congenital malformations in infants born after in vitro fertilization in Sweden. Birth Defects Res. Part A: Clin. Mol. Teratol. 2010, 88, 137–143. [Google Scholar]
- Tararbit, K.; Houyel, L.; Bonnet, D.; de Vigan, C.; Lelong, N.; Goffinet, F.; Khoshnood, B. Risk of congenital heart defects associated with assisted reproductive technologies: A population-based evaluation. Eur. Heart J. 2011, 32, 500–508. [Google Scholar] [CrossRef] [PubMed]
- Tararbit, K.; Lelong, N.; Thieulin, A.-C.; Houyel, L.; Bonnet, D.; Goffinet, F.; Khoshnood, B.; EPICARD Study Group. The risk for four specific congenital heart defects associated with assisted reproductive techniques: A population-based evaluation. Hum. Reprod. 2013, 28, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela-Alcaraz, B.; Crispi, F.; Bijnens, B.; Cruz-Lemini, M.; Creus, M.; Sitges, M.; Bartrons, J.; Civico, S.; Balasch, J.; Gratacos, E. Assisted reproductive technologies are associated with cardiovascular remodeling in utero that persists postnatally. Circulation 2013, 128, 1442–1450. [Google Scholar] [CrossRef] [PubMed]
- Ceelen, M.; van Weissenbruch, M.M.; Vermeiden, J.P.W.; van Leeuwen, F.E.; Delemarre-van de Waal, H.A. Cardiometabolic differences in children born after in vitro fertilization: follow-up study. J. Clin. Endocrinol. Metab. 2008, 93, 1682–1688. [Google Scholar] [CrossRef] [PubMed]
- Ceelen, M.; van Weissenbruch, M.M.; Prein, J.; Smit, J.J.; Vermeiden, J.P.; Spreeuwenberg, M.; van Leeuwen, F.E.; Delemarre-van de Waal, H.A. Growth during infancy and early childhood in relation to blood pressure and body fat measures at age 8–18 years of IVF children and spontaneously conceived controls born to subfertile parents. Hum. Reprod. 2009, 24, 2788–2795. [Google Scholar] [CrossRef] [PubMed]
- Sinaiko, A.R.; Donahue, R.P.; Jacobs, D.R.; Prineas, R.J. Relation of weight and rate of increase in weight during childhood and adolescence to body size, blood pressure, fasting insulin, and lipids in young adults: the minneapolis children’s blood pressure study. Circulation 1999, 99, 1471–1476. [Google Scholar] [CrossRef] [PubMed]
- Law, C.M.; Shiell, A.W.; Newsome, C.A.; Syddall, H.E.; Shinebourne, E.A.; Fayers, P.M.; Martyn, C.N.; de Swiet, M. Fetal, infant, and childhood growth and adult blood pressure: a longitudinal study from birth to 22 years of age. Circulation 2002, 105, 1088–1092. [Google Scholar] [CrossRef] [PubMed]
- Sakka, S.D.; Loutradis, D.; Kanaka-Gantenbein, C.; Margeli, A.; Papastamataki, M.; Papassotiriou, I.; Chrousos, G.P. Absence of insulin resistance and low-grade inflammation despite early metabolic syndrome manifestations in children born after in vitro fertilization. Fertil. Steril. 2010, 94, 1693–1699. [Google Scholar] [CrossRef] [PubMed]
- Scherrer, U.; Rimoldi, S.F.; Rexhaj, E.; Stuber, T.; Duplain, H.; Garcin, S.; de Marchi, S.F.; Nicod, P.; Germond, M.; Allemann, Y.; et al. Systemic and pulmonary vascular dysfunction in children conceived by assisted reproductive technologies. Circulation 2012, 125, 1890–1896. [Google Scholar] [CrossRef] [PubMed]
- Charakida, M.; Deanfield, J.E.; Halcox, J.P. Childhood origins of arterial disease. Curr. Opin. Pediatr. 2007, 19, 538–545. [Google Scholar] [CrossRef] [PubMed]
- Urbina, E.M.; Williams, R.V.; Alpert, B.S.; Collins, R.T.; Daniels, S.R.; Hayman, L.; Jacobson, M.; Mahoney, L.; Mietus-Snyder, M.; Rocchini, A.; et al. Noninvasive assessment of subclinical atherosclerosis in children and adolescents: recommendations for standard assessment for clinical research: A scientific statement from the american heart association. Hypertension 2009, 54, 919–950. [Google Scholar] [CrossRef] [PubMed]
- Bonetti, P.O.; Lerman, L.O.; Lerman, A. Endothelial dysfunction a marker of atherosclerotic risk. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Sitia, S.; Tomasoni, L.; Atzeni, F.; Ambrosio, G.; Cordiano, C.; Catapano, A.; Tramontana, S.; Perticone, F.; Naccarato, P.; Camici, P. From endothelial dysfunction to atherosclerosis. Autoimmun. Rev. 2010, 9, 830–834. [Google Scholar] [CrossRef] [PubMed]
- Van Popele, N.M.; Grobbee, D.E.; Bots, M.L.; Asmar, R.; Topouchian, J.; Reneman, R.S.; Hoeks, A.P.; van der Kuip, D.A.; Hofman, A.; Witteman, J.C. Association between arterial stiffness and atherosclerosis The Rotterdam Study. Stroke 2001, 32, 454–460. [Google Scholar] [CrossRef] [PubMed]
- Bots, M.L.; Dijk, J.M.; Oren, A.; Grobbee, D.E. Carotid intima-media thickness, arterial stiffness and risk of cardiovascular disease: Current evidence. J. Hypertens. 2002, 20, 2317–2325. [Google Scholar] [CrossRef] [PubMed]
- Simons, P.C.; Algra, A.; Bots, M.L.; Grobbee, D.E.; van der Graaf, Y. Common carotid intima-media thickness and arterial stiffness: Indicators of cardiovascular risk in high-risk patients. The SMART Study (Second Manifestations of ARTerial disease). Circulation 1999, 100, 951–957. [Google Scholar] [CrossRef] [PubMed]
- Rooke, J.A.; McEvoy, T.G.; Ashworth, C.J.; Robinson, J.J.; Wilmut, I.; Young, L.E.; Sinclair, K.D. Ovine fetal development is more sensitive to perturbation by the presence of serum in embryo culture before rather than after compaction. Theriogenology 2007, 67, 639–647. [Google Scholar] [CrossRef] [PubMed]
- Powell, K.; Rooke, J.A.; McEvoy, T.G.; Ashworth, C.J.; Robinson, J.J.; Wilmut, I.; Young, L.E.; Sinclair, K.D. Zygote donor nitrogen metabolism and in vitro embryo culture perturbs in utero development and IGF2R expression in ovine fetal tissues. Theriogenology 2006, 66, 1901–1912. [Google Scholar] [CrossRef] [PubMed]
- Farin, P.W.; Farin, C.E. Transfer of bovine embryos produced in vivo or in vitro: Survival and fetal development. Biol. Reprod. 1995, 52, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Van Wagtendonk-de Leeuw, A.M.; Mullaart, E.; de Roos, A.P.W.; Merton, J.S.; den Daas, J.H.G.; Kemp, B.; de Ruigh, L. Effects of different reproduction techniques: AI, moet or IVP, on health and welfare of bovine offspring. Theriogenology 2000, 53, 575–597. [Google Scholar] [CrossRef] [PubMed]
- McEvoy, T.G.; Sinclair, K.D.; Broadbent, P.J.; Goodhand, K.L.; Robinson, J.J. Post-natal growth and development of Simmental calves derived from in vivo or in vitro embryos. Reprod. Fertil. Dev. 1998, 10, 459–464. [Google Scholar] [CrossRef] [PubMed]
- Rexhaj, E.; Paoloni-Giacobino, A.; Rimoldi, S.F.; Fuster, D.G.; Anderegg, M.; Somm, E.; Bouillet, E.; Allemann, Y.; Sartori, C.; Scherrer, U. Mice generated by in vitro fertilization exhibit vascular dysfunction and shortened life span. J. Clin. Investig. 2013, 123, 5052–5060. [Google Scholar] [CrossRef] [PubMed]
- Young, L.E.; Sinclair, K.D.; Wilmut, I. Large offspring syndrome in cattle and sheep. Rev. Reprod. 1998, 3, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Farin, P.W.; Piedrahita, J.A.; Farin, C.E. Errors in development of fetuses and placentas from in vitro-produced bovine embryos. Theriogenology 2006, 65, 178–191. [Google Scholar] [CrossRef] [PubMed]
- McEvoy, T.G.; Robinson, J.J.; Sinclair, K.D. Developmental consequences of embryo and cell manipulation in mice and farm animals. Reproduction 2001, 122, 507–518. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.C.W.; Botting, K.J.; Padhee, M.; Zhang, S.; McMillen, I.C.; Suter, C.M.; Brooks, D.A.; Morrison, J.L. Early origins of heart disease: Low birth weight and the role of the insulin-like growth factor system in cardiac hypertrophy. Clin. Exp. Pharmacol. Physiol. 2012, 39, 958–964. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.C.; Zhang, L.; McMillen, I.C.; Botting, K.J.; Duffield, J.A.; Zhang, S.; Suter, C.M.; Brooks, D.A.; Morrison, J.L. Fetal growth restriction and the programming of heart growth and cardiac insulin-like growth factor 2 expression in the lamb. J. Physiol. 2011, 589, 4709–4722. [Google Scholar] [CrossRef] [PubMed]
- Vinci, M.; Polvani, G.; Pesce, M. Epigenetic programming and risk: The birthplace of cardiovascular disease? Stem Cell Rev. Rep. 2013, 9, 241–253. [Google Scholar] [CrossRef]
- Heijmans, B.T.; Tobi, E.W.; Stein, A.D.; Putter, H.; Blauw, G.J.; Susser, E.S.; Slagboom, P.E.; Lumey, L.H. Persistent epigenetic differences associated with prenatal exposure to famine in humans. Proc. Natl. Acad. Sci. USA 2008, 105, 17046–17049. [Google Scholar] [CrossRef] [PubMed]
- Tobi, E.W.; Lumey, L.H.; Talens, R.P.; Kremer, D.; Putter, H.; Stein, A.D.; Slagboom, P.E.; Heijmans, B.T. DNA methylation differences after exposure to prenatal famine are common and timing- and sex-specific. Hum. Mol. Genet. 2009, 18, 4046–4053. [Google Scholar] [CrossRef] [PubMed]
- Roseboom, T.; de Rooij, S.; Painter, R. The Dutch famine and its long-term consequences for adult health. Early Hum. Dev. 2006, 82, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, K.D.; Allegrucci, C.; Singh, R.; Gardner, D.S.; Sebastian, S.; Bispham, J.; Thurston, A.; Huntley, J.F.; Rees, W.D.; Maloney, C.A.; et al. DNA methylation, insulin resistance, and blood pressure in offspring determined by maternal periconceptional B vitamin and methionine status. Proc. Natl. Acad. Sci. USA 2007, 104, 19351–19356. [Google Scholar] [CrossRef] [PubMed]
- Celermajer, D.S. Manipulating nature: Might there be a cardiovascular price to pay for the miracle of assisted conception? Circulation 2012, 125, 1832–1834. [Google Scholar] [CrossRef] [PubMed]
- Horsthemke, B.; Ludwig, M. Assisted reproduction: The epigenetic perspective. Hum. Reprod. Update 2005, 11, 473–482. [Google Scholar] [CrossRef] [PubMed]
- DeBaun, M.R.; Niemitz, E.L.; Feinberg, A.P. Association of in vitro fertilization with Beckwith-Wiedemann syndrome and epigenetic alterations of LIT1 and H19. Am. J. Hum. Genet. 2003, 72, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Gicquel, C.; Gaston, V.; Mandelbaum, J.; Siffroi, J.P.; Flahault, A.; le Bouc, Y. In vitro fertilization may increase the risk of Beckwith-Wiedemann syndrome related to the abnormal imprinting of the KCN1OT gene. Am. J. Hum. Genet. 2003, 72, 1338–1341. [Google Scholar] [CrossRef] [PubMed]
- Maher, E.R.; Brueton, L.A.; Bowdin, S.C.; Luharia, A.; Cooper, W.; Cole, T.R.; Macdonald, F.; Sampson, J.R.; Barratt, C.L.; Reik, W.; Hawkins, M.M. Beckwith-Wiedemann syndrome and assisted reproduction technology (ART). J. Med. Genet. 2003, 40, 62–64. [Google Scholar] [CrossRef] [PubMed]
- Halliday, J.; Oke, K.; Breheny, S.; Algar, E.; David, J.A. Beckwith-Wiedemann syndrome and IVF: A case-control study. Am. J. Hum. Genet. 2004, 75, 526–528. [Google Scholar] [CrossRef] [PubMed]
- Lim, D.; Bowdin, S.C.; Tee, L.; Kirby, G.A.; Blair, E.; Fryer, A.; Lam, W.; Oley, C.; Cole, T.; Brueton, L.A.; et al. Clinical and molecular genetic features of Beckwith-Wiedemann syndrome associated with assisted reproductive technologies. Hum. Reprod. 2009, 24, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Rossignol, S.; Steunou, V.; Chalas, C.; Kerjean, A.; Rigolet, M.; Viegas-Pequignot, E.; Jouannet, P.; le Bouc, Y.; Gicquel, C. The epigenetic imprinting defect of patients with Beckwith-Wiedemann syndrome born after assisted reproductive technology is not restricted to the 11p15 region. J. Med. Genet. 2006, 43, 902–907. [Google Scholar] [CrossRef] [PubMed]
- Moll, A.C.; Imhof, S.M.; Cruysberg, J.R.; Schouten-van Meeteren, A.Y.; Boers, M.; van Leeuwen, F.E. Incidence of retinoblastoma in children born after in vitro fertilisation. Lancet 2003, 361, 309–310. [Google Scholar] [CrossRef] [PubMed]
- Mantikou, E.; Youssef, M.A.F.M.; van Wely, M.; van der Veen, F.; Al-Inany, H.G.; Repping, S.; Mastenbroek, S. Embryo culture media and IVF/ICSI success rates: A systematic review. Hum. Reprod. Update 2013, 19, 210–220. [Google Scholar] [CrossRef] [PubMed]
- Bromer, J.G.; Sakkas, D.; Seli, E. Metabolomic profiling of embryo culture media to predict IVF outcome. Expert Rev. Obstet. Gynecol. 2008, 3, 441–447. [Google Scholar] [CrossRef]
- Katari, S.; Turan, N.; Bibikova, M.; Erinle, O.; Chalian, R.; Foster, M.; Gaughan, J.P.; Coutifaris, C.; Sapienza, C. DNA methylation and gene expression differences in children conceived in vitro or in vivo. Hum. Mol. Genet. 2009, 18, 3769–3778. [Google Scholar] [CrossRef] [PubMed]
- Lui, J.C.; Finkielstain, G.P.; Barnes, K.M.; Baron, J. An imprinted gene network that controls mammalian somatic growth is down-regulated during postnatal growth deceleration in multiple organs. Am. J. Physiol.Regul. Integr. Comp. Physiol. 2008, 295, R189–R196. [Google Scholar] [CrossRef] [PubMed]
- Olofsson, L.E.; Orho-Melander, M.; William-Olsson, L.; Sjoholm, K.; Sjostrom, L.; Groop, L.; Carlsson, B.; Carlsson, L.M.; Olsson, B. CCAAT/enhancer binding protein alpha (C/EBPalpha) in adipose tissue regulates genes in lipid and glucose metabolism and a genetic variation in C/EBPalpha is associated with serum levels of triglycerides. J. Clin. Endocrinol. Metab. 2008, 93, 4880–4886. [Google Scholar] [CrossRef] [PubMed]
- Mzhavia, N.; Yu, S.; Ikeda, S.; Chu, T.T.; Goldberg, I.; Dansky, H.M. Neuronatin: A new inflammation gene expressed on the aortic endothelium of diabetic mice. Diabetes 2008, 57, 2774–2783. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Smit, E.; Brouwers, M.C.G.J.; Goossens, G.H.; van der Kallen, C.J.H.; van Greevenbroek, M.M.J.; Mariman, E.C.M. Plasma pigment epithelium-derived factor is positively associated with obesity in Caucasian subjects, in particular with the visceral fat depot. Eur. J. Endocrinol. 2008, 159, 713–718. [Google Scholar] [CrossRef] [PubMed]
- Linke, M.; May, A.; Reifenberg, K.; Haaf, T.; Zechner, U. The impact of ovarian stimulation on the expression of candidate reprogramming genes in mouse preimplantation embryos. Cytogenet. Genome Res. 2013, 139, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Fauque, P. Ovulation induction and epigenetic anomalies. Fertil. Steril. 2013, 99, 616–623. [Google Scholar] [CrossRef] [PubMed]
- Geuns, E.; Hilven, P.; van Steirteghem, A.; Liebaers, I.; de Rycke, M. Methylation analysis of KvDMR1 in human oocytes. J. Med. Genet. 2007, 44, 144–147. [Google Scholar] [CrossRef] [PubMed]
- Sato, C.; Shimada, M.; Mori, T.; Kumasako, Y.; Otsu, E.; Watanabe, H.; Utsunomiya, T. Assessment of human oocyte developmental competence by cumulus cell morphology and circulating hormone profile. Reprod. BioMed. Online 2007, 14, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Haaf, T. Aberrant methylation patterns at the two-cell stage as an indicator of early developmental failure. Mol. Reprod. Dev. 2002, 63, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Market-Velker, B.A.; Zhang, L.; Magri, L.S.; Bonvissuto, A.C.; Mann, M.R.W. Dual effects of superovulation: Loss of maternal and paternal imprinted methylation in a dose-dependent manner. Hum. Mol. Genet. 2010, 19, 36–51. [Google Scholar] [CrossRef] [PubMed]
- Laprise, S.L. Implications of epigenetics and genomic imprinting in assisted reproductive technologies. Mol. Reprod. Dev. 2009, 76, 1006–1018. [Google Scholar] [CrossRef] [PubMed]
- Paoloni-Giacobino, A.; Chaillet, J.R. Genomic imprinting and assisted reproduction. Reprod. Health 2004, 1, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Vu, T.H.; Ulaner, G.A.; Littman, E.; Ling, J.-Q.; Chen, H.-L.; Hu, J.-F.; Behr, B.; Giudice, L.; Hoffman, A.R. IVF results in de novo DNA methylation and histone methylation at an Igf2-H19 imprinting epigenetic switch. Mol. Hum. Reprod. 2005, 11, 631–640. [Google Scholar] [CrossRef] [PubMed]
- Doherty, A.S.; Mann, M.R.; Tremblay, K.D.; Bartolomei, M.S.; Schultz, R.M. Differential effects of culture on imprinted H19 expression in the preimplantation mouse embryo. Biol. Reprod. 2000, 62, 1526–1535. [Google Scholar] [CrossRef] [PubMed]
- Mann, M.R.; Lee, S.S.; Doherty, A.S.; Verona, R.I.; Nolen, L.D.; Schultz, R.M.; Bartolomei, M.S. Selective loss of imprinting in the placenta following preimplantation development in culture. Development 2004, 131, 3727–3735. [Google Scholar] [CrossRef] [PubMed]
- Wolff, G.L.; Kodell, R.L.; Moore, S.R.; Cooney, C.A. Maternal epigenetics and methyl supplements affect agouti gene expression in Avy/a mice. FASEB J. 1998, 12, 949–957. [Google Scholar] [PubMed]
- Armant, D.R. Blastocysts don’t go it alone. Extrinsic signals fine-tune the intrinsic developmental program of trophoblast cells. Dev. Biol. 2005, 280, 260–280. [Google Scholar] [CrossRef] [PubMed]
- Saito, S. Cytokine cross-talk between mother and the embryo/placenta. J. Reprod. Immunol. 2001, 52, 15–33. [Google Scholar] [CrossRef] [PubMed]
- Burke, A.C.; Nelson, C.E.; Morgan, B.A.; Tabin, C. Hox genes and the evolution of vertebrate axial morphology. Development 1995, 121, 333–346. [Google Scholar] [PubMed]
- Boerjan, M.L.; den Daas, J.H.G.; Dieleman, S.J. Embryonic origins of health: Long term effects of IVF in human and livestock. Theriogenology 2000, 53, 537–547. [Google Scholar] [CrossRef] [PubMed]
- Van Soom, A.; Boerjan, M.L.; Bols, P.E.; Vanroose, G.; Lein, A.; Coryn, M.; de Kruif, A. Timing of compaction and inner cell allocation in bovine embryos produced in vivo after superovulation. Biol. Reprod. 1997, 57, 1041–1049. [Google Scholar] [CrossRef] [PubMed]
- Van Soom, A.; Boerjan, M.; Ysebaert, M.-T.; de Kruif, A. Cell allocation to the inner cell mass and the trophectoderm in bovine embryos cultured in two different media. Mol. Reprod. Dev. 1996, 45, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Olson, E.N.; Schneider, M.D. Sizing up the heart: Development redux in disease. Genes Dev. 2003, 17, 1937–1956. [Google Scholar] [CrossRef] [PubMed]
- Van Montfoort, A.P.; Hanssen, L.L.; de Sutter, P.; Viville, S.; Geraedts, J.P.; de Boer, P. Assisted reproduction treatment and epigenetic inheritance. Hum. Reprod. Update 2012, 18, 171–197. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Padhee, M.; Zhang, S.; Lie, S.; Wang, K.C.; Botting, K.J.; McMillen, I.C.; MacLaughlin, S.M.; Morrison, J.L. The Periconceptional Environment and Cardiovascular Disease: Does In Vitro Embryo Culture and Transfer Influence Cardiovascular Development and Health? Nutrients 2015, 7, 1378-1425. https://doi.org/10.3390/nu7031378
Padhee M, Zhang S, Lie S, Wang KC, Botting KJ, McMillen IC, MacLaughlin SM, Morrison JL. The Periconceptional Environment and Cardiovascular Disease: Does In Vitro Embryo Culture and Transfer Influence Cardiovascular Development and Health? Nutrients. 2015; 7(3):1378-1425. https://doi.org/10.3390/nu7031378
Chicago/Turabian StylePadhee, Monalisa, Song Zhang, Shervi Lie, Kimberley C. Wang, Kimberley J. Botting, I. Caroline McMillen, Severence M. MacLaughlin, and Janna L. Morrison. 2015. "The Periconceptional Environment and Cardiovascular Disease: Does In Vitro Embryo Culture and Transfer Influence Cardiovascular Development and Health?" Nutrients 7, no. 3: 1378-1425. https://doi.org/10.3390/nu7031378
APA StylePadhee, M., Zhang, S., Lie, S., Wang, K. C., Botting, K. J., McMillen, I. C., MacLaughlin, S. M., & Morrison, J. L. (2015). The Periconceptional Environment and Cardiovascular Disease: Does In Vitro Embryo Culture and Transfer Influence Cardiovascular Development and Health? Nutrients, 7(3), 1378-1425. https://doi.org/10.3390/nu7031378