Ginseng Protects Against Respiratory Syncytial Virus by Modulating Multiple Immune Cells and Inhibiting Viral Replication
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cells, Virus, Reagents
2.2. Preparation of RSV Stock
2.3. RSV Immunoplaque Assay
2.4. Cell Viability Assay
2.5. Cytopathogenic Effect (CPE) Reduction Assay
2.6. Assays for Cytokines
2.7. Treatment of Mice with RGE and RSV A2 Virus
2.8. Lung Virus Titer and Cytokine Assays
2.9. Preparation of Bronchoalveolar Lavage (BAL) and Flow Cytometric Analysis
2.10. Statistical Analysis
3. Results
3.1. Influence of RGE on RSV Replication in Human Epithelial Cells
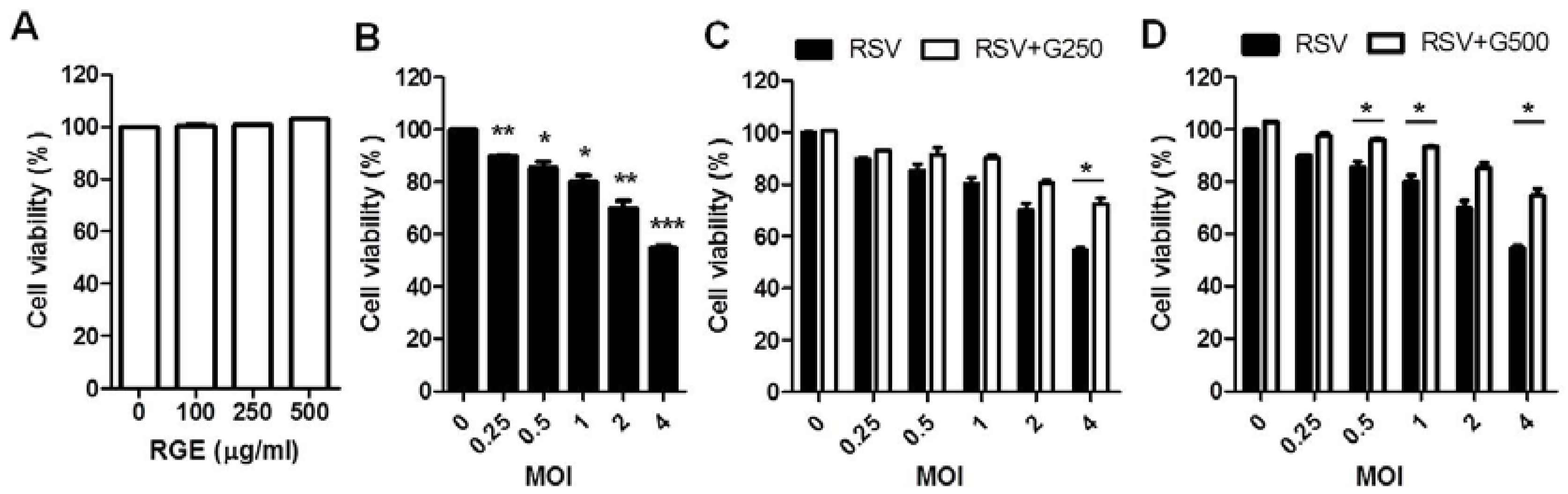
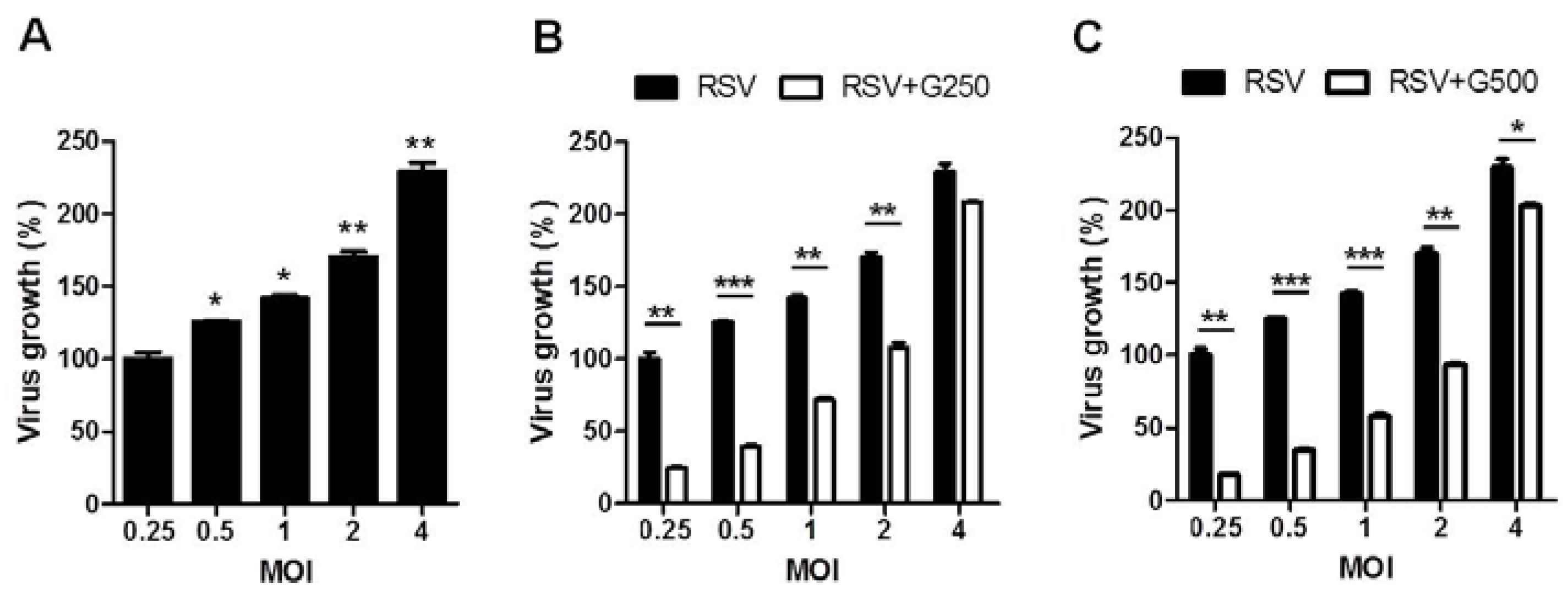
3.2. Effects of RGE on RSV-Induced Cytokine Production in Dendritic and Macrophage Cells
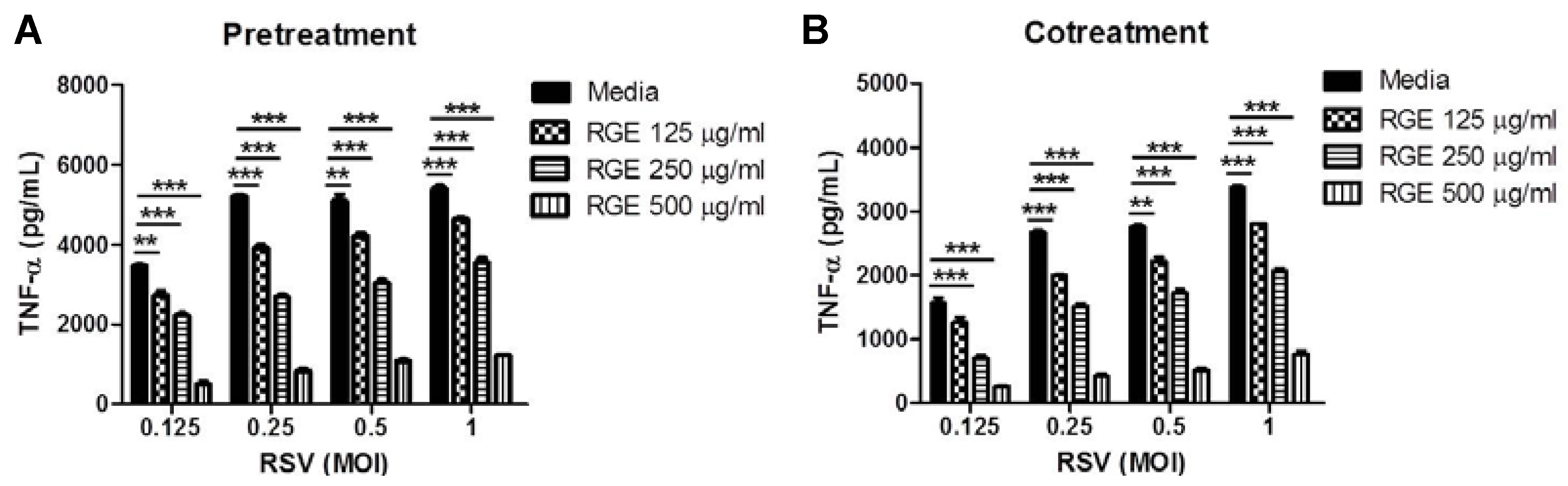
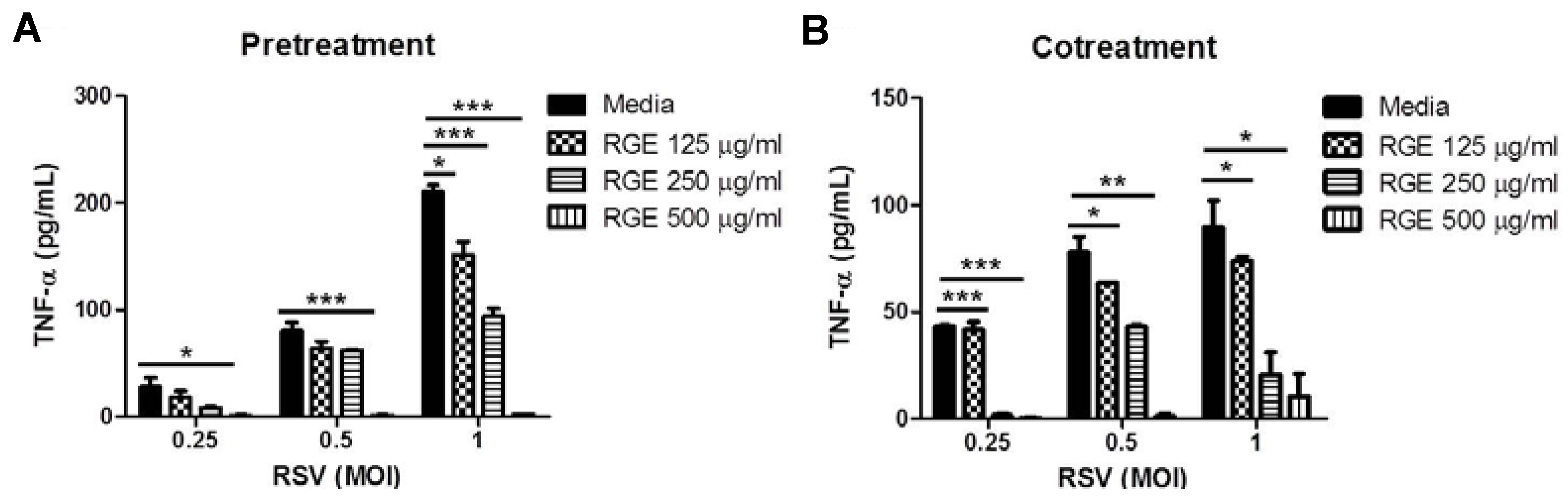
3.3. RGE Treatment Improves Clinical Outcomes upon RSV Infection of Mice
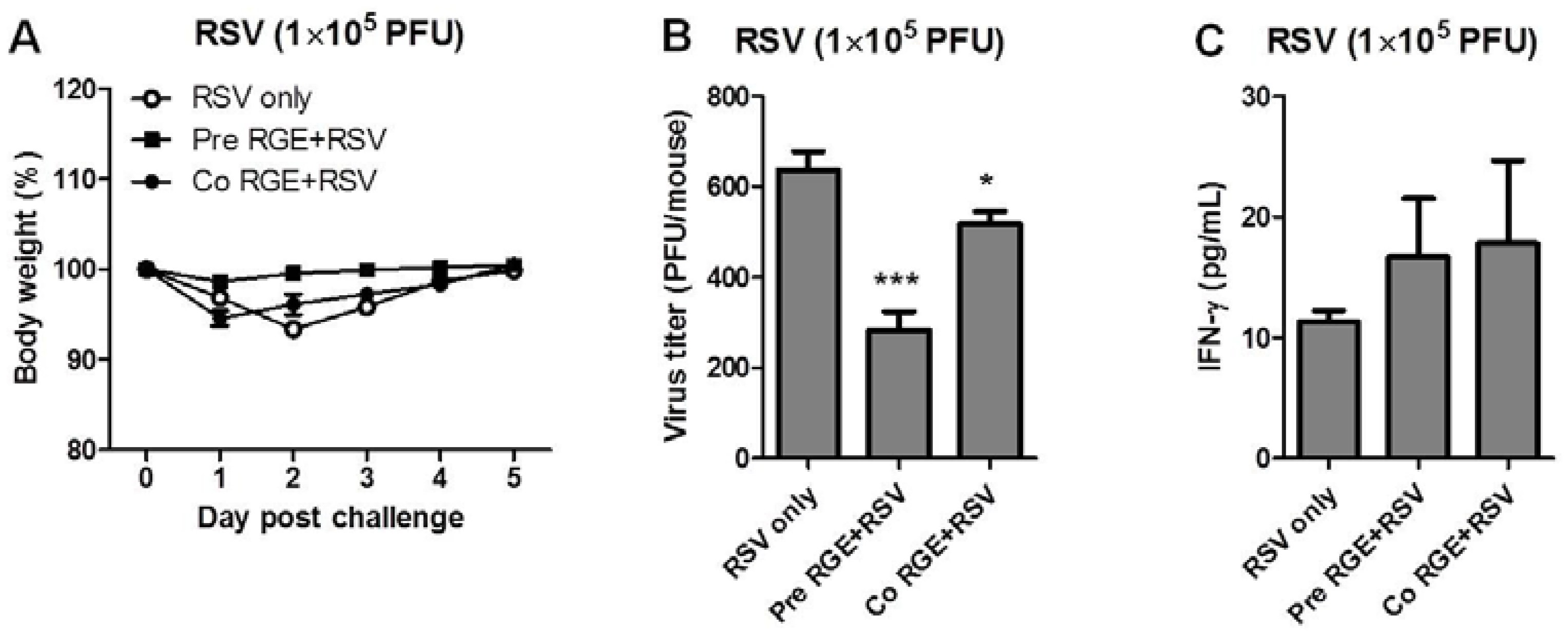
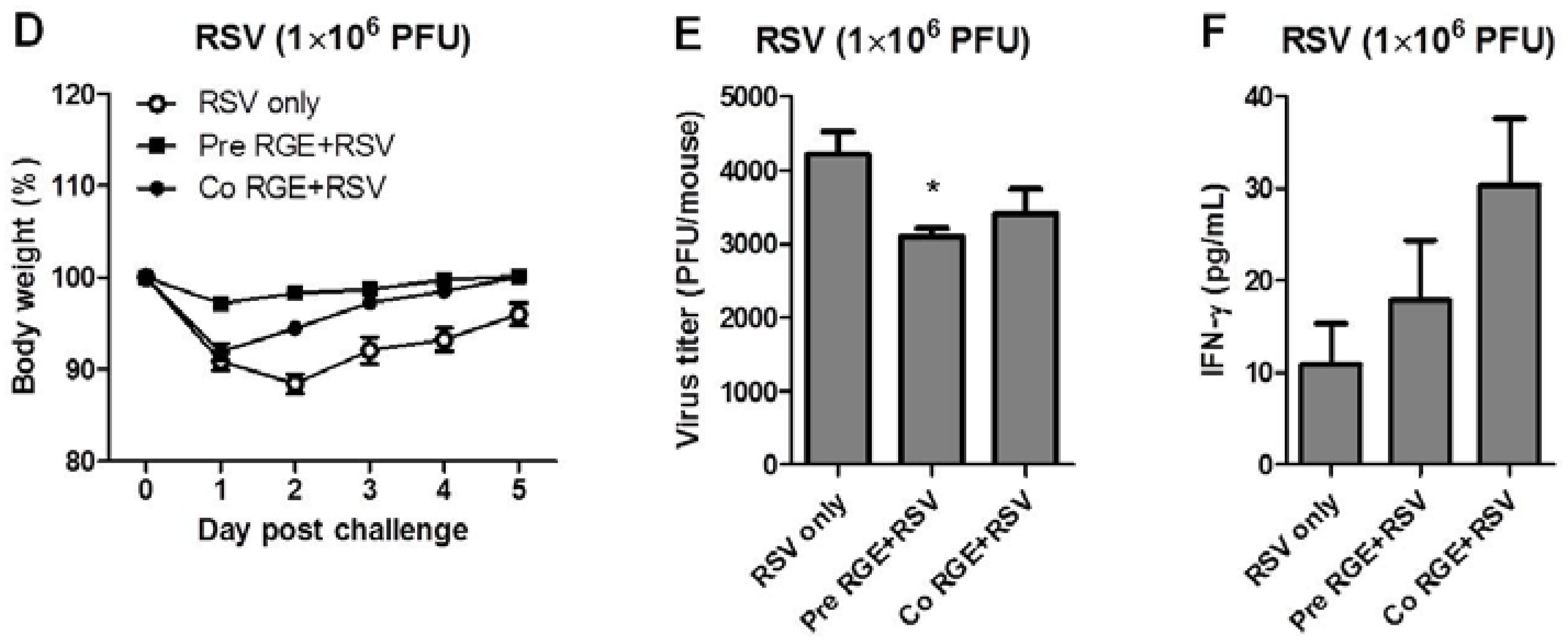
3.4. RGE Modulates Bronchoalveolar Immune Cells upon RSV Infection of Mice

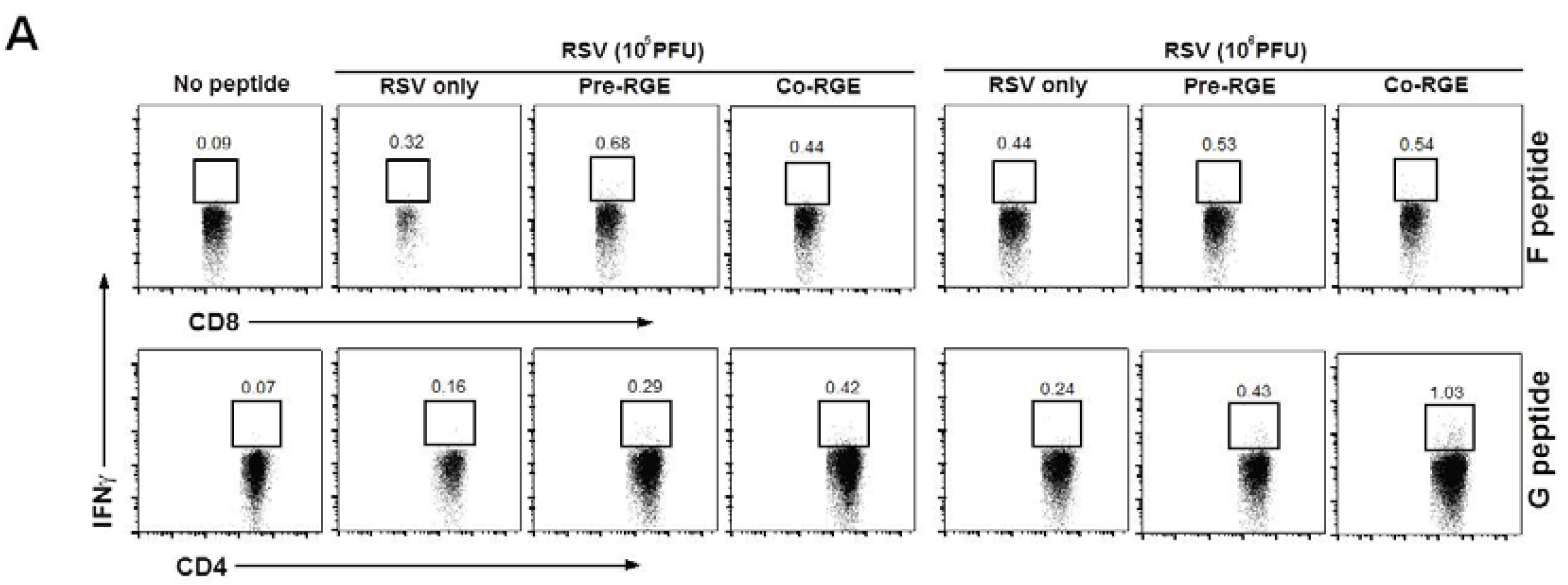
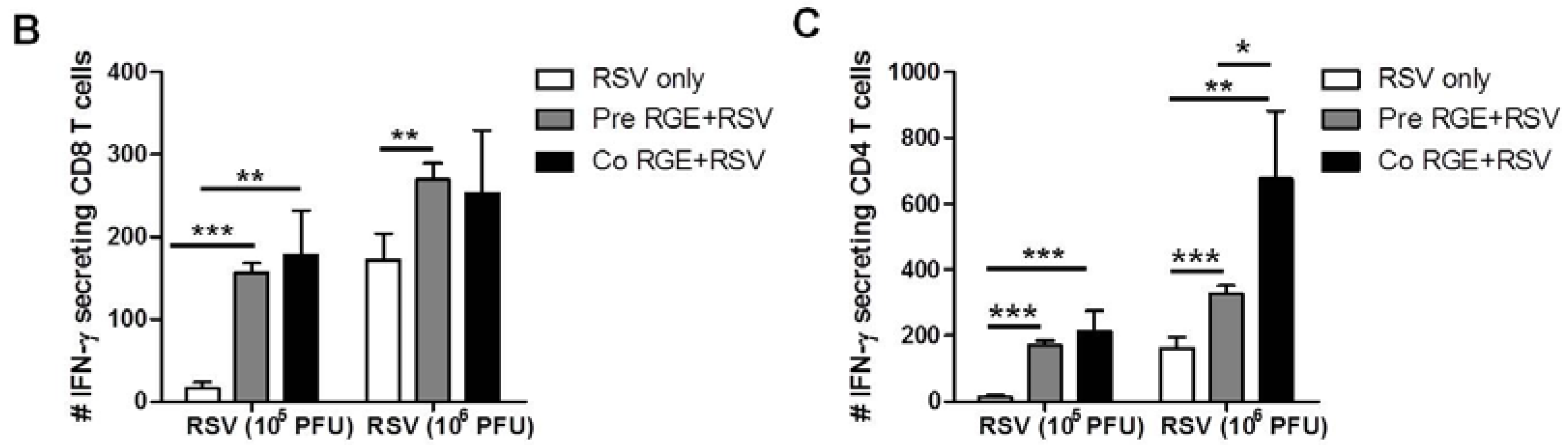
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Graham, B.S. Biological challenges and technological opportunities for respiratory syncytial virus vaccine development. Immunol. Rev. 2011, 239, 149–166. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.W.; Canchola, J.G.; Brandt, C.D.; Pyles, G.; Chanock, R.M.; Jensen, K.; Parrott, R.H. Respiratory syncytial virus disease in infants despite prior administration of antigenic inactivated vaccine. Am. J. Epidemiol. 1969, 89, 422–434. [Google Scholar] [PubMed]
- Legg, J.P.; Hussain, I.R.; Warner, J.A.; Johnston, S.L.; Warner, J.O. Type 1 and type 2 cytokine imbalance in acute respiratory syncytial virus bronchiolitis. Am. J. Respir. Crit. Care Med. 2003, 168, 633–639. [Google Scholar] [CrossRef] [PubMed]
- Waris, M.E.; Tsou, C.; Erdman, D.D.; Day, D.B.; Anderson, L.J. Priming with live respiratory syncytial virus (RSV) prevents the enhanced pulmonary inflammatory response seen after RSV challenge in BALB/c mice immunized with formalin-inactivated RSV. J. Virol. 1997, 71, 6935–6939. [Google Scholar] [PubMed]
- Waris, M.E.; Tsou, C.; Erdman, D.D.; Zaki, S.R.; Anderson, L.J. Respiratory synctial virus infection in BALB/c mice previously immunized with formalin-inactivated virus induces enhanced pulmonary inflammatory response with a predominant Th2-like cytokine pattern. J. Virol. 1996, 70, 2852–2860. [Google Scholar] [PubMed]
- Tripp, R.A.; Moore, D.; Jones, L.; Sullender, W.; Winter, J.; Anderson, L.J. Respiratory syncytial virus G and/or SH protein alters Th1 cytokines, natural killer cells, and neutrophils responding to pulmonary infection in BALB/c mice. J. Virol. 1999, 73, 7099–7107. [Google Scholar] [PubMed]
- Zhang, Y.; Luxon, B.A.; Casola, A.; Garofalo, R.P.; Jamaluddin, M.; Brasier, A.R. Expression of respiratory syncytial virus-induced chemokine gene networks in lower airway epithelial cells revealed by cDNA microarrays. J. Virol. 2001, 75, 9044–9058. [Google Scholar] [CrossRef] [PubMed]
- Blanco, J.C.; Richardson, J.Y.; Darnell, M.E.; Rowzee, A.; Pletneva, L.; Porter, D.D.; Prince, G.A. Cytokine and chemokine gene expression after primary and secondary respiratory syncytial virus infection in cotton rats. J. Infect. Dis. 2002, 185, 1780–1785. [Google Scholar] [CrossRef] [PubMed]
- Graham, B.S. Pathogenesis of respiratory syncytial virus vaccine-augmented pathology. Am. J. Respir. Crit. Care Med. 1995, 152, 63–66. [Google Scholar] [CrossRef]
- Schwarze, J. Lung dendritic cells in respiratory syncytial virus bronchiolitis. Pediatr. Infect. Dis. J. 2008, 27, 89–91. [Google Scholar] [CrossRef] [PubMed]
- Pulendran, B.; Ahmed, R. Translating innate immunity into immunological memory: Implications for vaccine development. Cell 2006, 124, 849–863. [Google Scholar] [CrossRef] [PubMed]
- Jia, L.; Zhao, Y.; Liang, X.J. Current evaluation of the millennium phytomedicine—Ginseng (II): Collected chemical entities, modern pharmacology, and clinical applications emanated from traditional Chinese medicine. Curr. Med. Chem. 2009, 16, 2924–2942. [Google Scholar] [CrossRef] [PubMed]
- Attele, A.S.; Wu, J.A.; Yuan, C.S. Ginseng pharmacology: Multiple constituents and multiple actions. Biochem. Pharmacol. 1999, 58, 1685–1693. [Google Scholar] [CrossRef] [PubMed]
- Scaglione, F.; Ferrara, F.; Dugnani, S.; Falchi, M.; Santoro, G.; Fraschini, F. Immunomodulatory effects of two extracts of Panax ginseng C.A. Meyer. Drugs Exp. Clin. Res. 1990, 16, 537–542. [Google Scholar] [PubMed]
- Larsen, M.W.; Moser, C.; Hoiby, N.; Song, Z.; Kharazmi, A. Ginseng modulates the immune response by induction of interleukin-12 production. APMIS 2004, 112, 369–373. [Google Scholar] [CrossRef] [PubMed]
- Lemmon, H.R.; Sham, J.; Chau, L.A.; Madrenas, J. High molecular weight polysaccharides are key immunomodulators in North American ginseng extracts: Characterization of the ginseng genetic signature in primary human immune cells. J. Ethnopharmacol. 2012, 142, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.Y.; Choi, I.S.; Shim, J.Y.; Yun, E.K.; Yun, Y.S.; Jeong, G.; Song, J.Y. The immunomodulator ginsan induces resistance to experimental sepsis by inhibiting Toll-like receptor-mediated inflammatory signals. Eur. J. Immunol. 2006, 36, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.Y.; Song, J.Y.; Yun, Y.S.; Jeong, G.; Choi, I.S. Protection of Staphylococcus aureus-infected septic mice by suppression of early acute inflammation and enhanced antimicrobial activity by ginsan. FEMS Immunol. Med. Microbiol. 2006, 46, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Y.Z.; Shang, H.C.; Gao, X.M.; Zhang, B.L. A comparison of the ancient use of ginseng in traditional Chinese medicine with modern pharmacological experiments and clinical trials. Phytother. Res. 2008, 22, 851–858. [Google Scholar] [CrossRef] [PubMed]
- Moore, M.L.; Chi, M.H.; Luongo, C.; Lukacs, N.W.; Polosukhin, V.V.; Huckabee, M.M.; Newcomb, D.C.; Buchholz, U.J.; Crowe, J.E., Jr.; Goleniewska, K.; et al. A chimeric A2 strain of respiratory syncytial virus (RSV) with the fusion protein of RSV strain line 19 exhibits enhanced viral load, mucus, and airway dysfunction. J. Virol. 2009, 83, 4185–4194. [Google Scholar] [CrossRef] [PubMed]
- Quan, F.S.; Kim, Y.; Lee, S.; Yi, H.; Kang, S.M.; Bozja, J.; Moore, M.L.; Compans, R.W. Viruslike particle vaccine induces protection against respiratory syncytial virus infection in mice. J. Infect. Dis. 2011, 204, 987–995. [Google Scholar] [CrossRef] [PubMed]
- Hargadon, K.M.; Forrest, O.A.; Reddy, P.R. Suppression of the maturation and activation of the dendritic cell line DC2.4 by melanoma-derived factors. Cell. Immunol. 2012, 272, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Fu, R.H.; Hran, H.J.; Chu, C.L.; Huang, C.M.; Liu, S.P.; Wang, Y.C.; Lin, Y.H.; Shyu, W.C.; Lin, S.Z. Lipopolysaccharide-stimulated activation of murine DC2.4 cells is attenuated by N-butylidenephthalide through suppression of the NF-κB pathway. Biotechnol. Lett. 2011, 33, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Hong, E.K. Immunostimulating activity of the polysaccharides isolated from Cordyceps militaris. Int. Immunopharmacol. 2011, 11, 1226–1233. [Google Scholar] [CrossRef] [PubMed]
- Geiler, J.; Michaelis, M.; Naczk, P.; Leutz, A.; Langer, K.; Doerr, H.W.; Cinatl, J., Jr. N-acetyl-l-cysteine (NAC) inhibits virus replication and expression of pro-inflammatory molecules in A549 cells infected with highly pathogenic H5N1 influenza A virus. Biochem. Pharmacol. 2010, 79, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Quan, F.S.; Kim, Y.C.; Yoo, D.G.; Compans, R.W.; Prausnitz, M.R.; Kang, S.M. Stabilization of influenza vaccine enhances protection by microneedle delivery in the mouse skin. PLoS One 2009, 4, e7152. [Google Scholar] [CrossRef] [PubMed]
- Song, J.M.; Hossain, J.; Yoo, D.G.; Lipatov, A.S.; Davis, C.T.; Quan, F.S.; Chen, L.M.; Hogan, R.J.; Donis, R.O.; Compans, R.W.; et al. Protective immunity against H5N1 influenza virus by a single dose vaccination with virus-like particles. Virology 2010, 405, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Villenave, R.; Shields, M.D.; Power, U.F. Respiratory syncytial virus interaction with human airway epithelium. Trends Microbiol. 2013, 21, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Panuska, J.R.; Midulla, F.; Cirino, N.M.; Villani, A.; Gilbert, I.A.; McFadden, E.R., Jr.; Huang, Y.T. Virus-induced alterations in macrophage production of tumor necrosis factor and prostaglandin E2. Am. J. Physiol. 1990, 259, 396–402. [Google Scholar]
- Hammad, H.; Lambrecht, B.N. Dendritic cells and epithelial cells: Linking innate and adaptive immunity in asthma. Nat. Rev. Immunol. 2008, 8, 193–204. [Google Scholar] [CrossRef] [PubMed]
- Culley, F.J.; Pennycook, A.M.; Tregoning, J.S.; Hussell, T.; Openshaw, P.J. Differential chemokine expression following respiratory virus infection reflects Th1- or Th2-biased immunopathology. J. Virol. 2006, 80, 4521–4527. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Kou, J.P.; Yu, B.Y. Ginsenoside Rg1 protects against hydrogen peroxide-induced cell death in PC12 cells via inhibiting NF-κB activation. Neurochem. Int. 2011, 58, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.E.; Lyu, S.Y. Anti-inflammatory and anti-oxidative effects of Korean red ginseng extract in human keratinocytes. Immune Netw. 2011, 11, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Mata, M.; Morcillo, E.; Gimeno, C.; Cortijo, J. N-acetyl-l-cysteine (NAC) inhibit mucin synthesis and pro-inflammatory mediators in alveolar type II epithelial cells infected with influenza virus A and B and with respiratory syncytial virus (RSV). Biochem. Pharmacol. 2011, 82, 548–555. [Google Scholar] [CrossRef] [PubMed]
- Michaelis, M.; Geiler, J.; Naczk, P.; Sithisarn, P.; Leutz, A.; Doerr, H.W.; Cinatl, J., Jr. Glycyrrhizin exerts antioxidative effects in H5N1 influenza A virus-infected cells and inhibits virus replication and pro-inflammatory gene expression. PLoS One 2011, 6, e19705. [Google Scholar] [CrossRef] [PubMed]
- DeVincenzo, J.P.; El Saleeby, C.M.; Bush, A.J. Respiratory syncytial virus load predicts disease severity in previously healthy infants. J. Infect. Dis. 2005, 191, 1861–1868. [Google Scholar] [CrossRef] [PubMed]
- Karron, R.A.; Wright, P.F.; Crowe, J.E., Jr.; Clements-Mann, M.L.; Thompson, J.; Makhene, M.; Casey, R.; Murphy, B.R. Evaluation of two live, cold-passaged, temperature-sensitive respiratory syncytial virus vaccines in chimpanzees and in human adults, infants, and children. J. Infect. Dis. 1997, 176, 1428–1436. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Kunimoto, M.; Patel, J.A. Autocrine regulation and experimental modulation of interleukin-6 expression by human pulmonary epithelial cells infected with respiratory syncytial virus. J. Virol. 1998, 72, 2496–2499. [Google Scholar] [PubMed]
- Panuska, J.R.; Merolla, R.; Rebert, N.A.; Hoffmann, S.P.; Tsivitse, P.; Cirino, N.M.; Silverman, R.H.; Rankin, J.A. Respiratory syncytial virus induces interleukin-10 by human alveolar macrophages. Suppression of early cytokine production and implications for incomplete immunity. J. Clin. Invest. 1995, 96, 2445–2453. [Google Scholar] [CrossRef] [PubMed]
- Tsutsumi, H.; Matsuda, K.; Sone, S.; Takeuchi, R.; Chiba, S. Respiratory syncytial virus-induced cytokine production by neonatal macrophages. Clin. Exp. Immunol. 1996, 106, 442–446. [Google Scholar] [CrossRef] [PubMed]
- Schwarze, J.; Mackenzie, K.J. Novel insights into immune and inflammatory responses to respiratory viruses. Thorax 2013, 68, 108–110. [Google Scholar] [CrossRef] [PubMed]
- Johnson, T.R.; Hong, S.; Van Kaer, L.; Koezuka, Y.; Graham, B.S. NK T cells contribute to expansion of CD8(+) T cells and amplification of antiviral immune responses to respiratory syncytial virus. J. Virol. 2002, 76, 4294–4303. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, Y.; Gilmore, X.; Xu, K.; Wyde, P.R.; Mbawuike, I.N. An aged mouse model for RSV infection and diminished CD8(+) CTL responses. Exp. Biol. Med. 2002, 227, 133–140. [Google Scholar]
- Castilow, E.M.; Olson, M.R.; Meyerholz, D.K.; Varga, S.M. Differential role of gamma interferon in inhibiting pulmonary eosinophilia and exacerbating systemic disease in fusion protein-immunized mice undergoing challenge infection with respiratory syncytial virus. J. Virol. 2008, 82, 2196–2207. [Google Scholar] [CrossRef] [PubMed]
- Jung, I.D.; Kim, H.Y.; Park, J.W.; Lee, C.M.; Noh, K.T.; Kang, H.K.; Heo, D.R.; Lee, S.J.; Son, K.H.; Park, H.J.; et al. RG-II from Panax ginseng C.A. Meyer suppresses asthmatic reaction. BMB Rep. 2012, 45, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.; Shin, H.S.; Kang, S.C. Inhibition of inflammations and macrophage activation by ginsenoside-Re isolated from Korean ginseng (Panax ginseng C.A. Meyer). Food Chem. Toxicol. 2012, 50, 1354–1361. [Google Scholar] [CrossRef] [PubMed]
- Yoo, D.G.; Kim, M.C.; Park, M.K.; Park, K.M.; Quan, F.S.; Song, J.M.; Wee, J.J.; Wang, B.Z.; Cho, Y.K.; Compans, R.W.; et al. Protective effect of ginseng polysaccharides on influenza viral infection. PLoS One 2012, 7, e33678. [Google Scholar] [CrossRef] [PubMed]
- Banchereau, J.; Steinman, R.M. Dendritic cells and the control of immunity. Nature 1998, 392, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Guilliams, M.; Lambrecht, B.N.; Hammad, H. Division of labor between lung dendritic cells and macrophages in the defense against pulmonary infections. Mucosal Immunol. 2013, 6, 464–473. [Google Scholar] [CrossRef] [PubMed]
- Lambrecht, B.N.; Hammad, H. Lung dendritic cells in respiratory viral infection and asthma: From protection to immunopathology. Annu. Rev. Immunol. 2012, 30, 243–270. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.S.; Lee, Y.-N.; Lee, Y.-T.; Hwang, H.S.; Kim, K.-H.; Ko, E.-J.; Kim, M.-C.; Kang, S.-M. Ginseng Protects Against Respiratory Syncytial Virus by Modulating Multiple Immune Cells and Inhibiting Viral Replication. Nutrients 2015, 7, 1021-1036. https://doi.org/10.3390/nu7021021
Lee JS, Lee Y-N, Lee Y-T, Hwang HS, Kim K-H, Ko E-J, Kim M-C, Kang S-M. Ginseng Protects Against Respiratory Syncytial Virus by Modulating Multiple Immune Cells and Inhibiting Viral Replication. Nutrients. 2015; 7(2):1021-1036. https://doi.org/10.3390/nu7021021
Chicago/Turabian StyleLee, Jong Seok, Yu-Na Lee, Young-Tae Lee, Hye Suk Hwang, Ki-Hye Kim, Eun-Ju Ko, Min-Chul Kim, and Sang-Moo Kang. 2015. "Ginseng Protects Against Respiratory Syncytial Virus by Modulating Multiple Immune Cells and Inhibiting Viral Replication" Nutrients 7, no. 2: 1021-1036. https://doi.org/10.3390/nu7021021
APA StyleLee, J. S., Lee, Y.-N., Lee, Y.-T., Hwang, H. S., Kim, K.-H., Ko, E.-J., Kim, M.-C., & Kang, S.-M. (2015). Ginseng Protects Against Respiratory Syncytial Virus by Modulating Multiple Immune Cells and Inhibiting Viral Replication. Nutrients, 7(2), 1021-1036. https://doi.org/10.3390/nu7021021



