Immune-Relevant and Antioxidant Activities of Vitellogenin and Yolk Proteins in Fish
Abstract
:1. Introduction

2. Immune-Relevant Activities of Vtgs and Yolk Proteins
2.1. Immune Roles of Vtgs
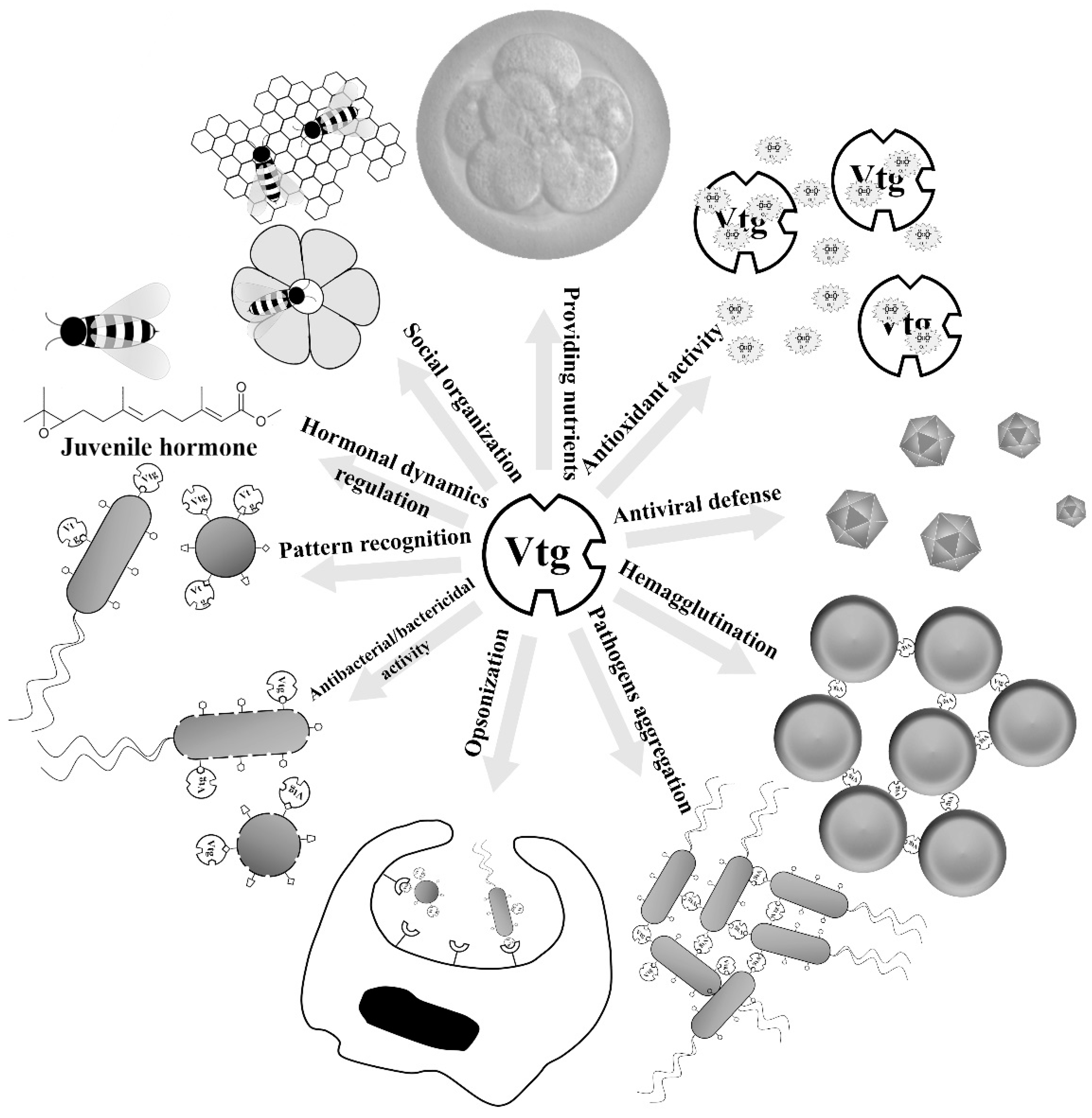
2.2. Immune Roles of Yolk Proteins
3. Antioxidant Activities of Vtgs and Yolk Proteins
4. Potential Applications in Human Health
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jalabert, B. Particularities of reproduction and oogenesis in teleost fish compared to mammals. Reprod. Nutr. Dev. 2005, 45, 261–279. [Google Scholar] [CrossRef] [PubMed]
- Lubzens, E.; Young, G.; Bobe, J.; Cerda, J. Oogenesis in teleosts: How eggs are formed. Gen. Comp. Endocrinol. 2010, 165, 367–389. [Google Scholar] [CrossRef]
- Patiño, R.; Sullivan, C. Ovarian follicle growth, maturation, and ovulation in teleost fish. Fish Physiol. Biochem. 2002, 26, 57–70. [Google Scholar] [CrossRef]
- Finn, R.N. Vertebrate yolk complexes and the functional implications of phosvitins and other subdomains in vitellogenins. Biol. Reprod. 2007, 76, 926–935. [Google Scholar] [CrossRef] [PubMed]
- Smolenaars, M.M.; Madsen, O.; Rodenburg, K.W.; van der Horst, D.J. Molecular diversity and evolution of the large lipid transfer protein superfamily. J. Lipid Res. 2007, 48, 489–502. [Google Scholar] [CrossRef] [PubMed]
- Avarre, J.C.; Lubzens, E.; Babin, P.J. Apolipocrustacein, formerly vitellogenin, is the major egg yolk precursor protein in decapod crustaceans and is homologous to insect apolipophorin II/I and vertebrate apolipoprotein B. BMC Evol. Biol. 2007, 7. [Google Scholar] [CrossRef] [PubMed]
- Tufail, M.; Takeda, M. Molecular characteristics of insect vitellogenins. J. Insect Physiol. 2008, 54, 1447–1458. [Google Scholar] [PubMed]
- Matozzo, V.; Gagne, F.; Marin, M.G.; Ricciardi, F.; Blaise, C. Vitellogenin as a biomarker of exposure to estrogenic compounds in aquatic invertebrates: A review. Environ. Int. 2008, 34, 531–545. [Google Scholar] [PubMed]
- Wu, L.T.; Hui, J.H.; Chu, K.H. Origin and evolution of yolk proteins: Expansion and functional diversification of large lipid transfer protein superfamily. Biol. Reprod. 2013, 88. [Google Scholar] [CrossRef] [PubMed]
- Finn, R.N.; Kolarevic, J.; Kongshaug, H.; Nilsen, F. Evolution and differential expression of a vertebrate vitellogenin gene cluster. BMC Evol. Biol. 2009, 9. [Google Scholar] [CrossRef] [PubMed]
- Silva, R.; Fischer, A.H.; Burch, J.B. The major and minor chicken vitellogenin genes are each adjacent to partially deleted pseudogene copies of the other. Mol. Cell. Biol. 1989, 9, 3557–3562. [Google Scholar] [CrossRef] [PubMed]
- Van het Schip, F.D.; Samallo, J.; Broos, J.; Ophuis, J.; Mojet, M.; Gruber, M.; Ab, G. Nucleotide sequence of a chicken vitellogenin gene and derived amino acid sequence of the encoded yolk precursor protein. J. Mol. Biol. 1987, 196, 245–260. [Google Scholar] [CrossRef]
- Germond, J.E.; Walker, P.; ten Heggeler, B.; Brown-Luedi, M.; de Bony, E.; Wahli, W. Evolution of vitellogenin genes: Comparative analysis of the nucleotide sequences downstream of the transcription initiation site of four Xenopus laevis and one chicken gene. Nucleic Acids Res. 1984, 12, 8595–8609. [Google Scholar] [CrossRef] [PubMed]
- Wahli, W.; Dawid, I.B.; Wyler, T.; Jaggi, R.B.; Weber, R.; Ryffel, G.U. Vitellogenin in Xenopus laevis is encoded in a small family of genes. Cell 1979, 16, 535–549. [Google Scholar] [CrossRef]
- Blumenthal, T.; Squire, M.; Kirtland, S.; Cane, J.; Donegan, M.; Spieth, J.; Sharrock, W. Cloning of a yolk protein gene family from Caenorhabditis elegans. J. Mol. Biol. 1984, 174, 1–18. [Google Scholar] [CrossRef]
- Wang, H.; Yan, T.; Tan, J.T.; Gong, Z. A zebrafish vitellogenin gene (vg3) encodes a novel vitellogenin without a phosvitin domain and may represent a primitive vertebrate vitellogenin gene. Gene 2000, 256, 303–310. [Google Scholar] [CrossRef]
- Wang, H.; Tan, J.T.; Emelyanov, A.; Korzh, V.; Gong, Z. Hepatic and extrahepatic expression of vitellogenin genes in the zebrafish, Danio rerio. Gene 2005, 356, 91–100. [Google Scholar] [PubMed]
- Kang, B.J.; Jung, J.H.; Lee, J.M.; Lim, S.G.; Saito, H.; Kim, M.H.; Kim, Y.J.; Saigusa, M.; Han, C.H. Structural and expression analyses of two vitellogenin genes in the carp, Cyprinus carpio. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2007, 148, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Williams, V.N.; Reading, B.J.; Hiramatsu, N.; Amano, H.; Glassbrook, N.; Hara, A.; Sullivan, C.V. Multiple vitellogenins and product yolk proteins in striped bass, Morone saxatilis: Molecular characterization and processing during oocyte growth and maturation. Fish Physiol. Biochem. 2014, 40, 395–415. [Google Scholar] [CrossRef] [PubMed]
- Reading, B.J.; Hiramatsu, N.; Sawaguchi, S.; Matsubara, T.; Hara, A.; Lively, M.O.; Sullivan, C.V. Conserved and variant molecular and functional features of multiple egg yolk precursor proteins (vitellogenins) in white perch (Morone americana) and other teleosts. Mar. Biotechnol. 2009, 11, 169–187. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Hu, L.; Liu, S.; Gao, Z.; Zhang, S. Functional analysis of domain of unknown function (DUF) 1943, DUF1944 and von Willebrand factor type D domain (VWD) in vitellogenin 2 in zebrafish. Dev. Comp. Immunol. 2013, 41, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Hayward, A.; Takahashi, T.; Bendena, W.G.; Tobe, S.S.; Hui, J.H. Comparative genomic and phylogenetic analysis of vitellogenin and other large lipid transfer proteins in metazoans. FEBS Lett. 2010, 584, 1273–1278. [Google Scholar] [CrossRef] [PubMed]
- Finn, R.N.; Kristoffersen, B.A. Vertebrate vitellogenin gene duplication in relation to the “3R hypothesis”: Correlation to the pelagic egg and the oceanic radiation of teleosts. PLoS ONE 2007, 2, e169. [Google Scholar] [CrossRef] [PubMed]
- Meusy, J.J. Vitellogenin, the extraovarian precursor of the protein yolk in Crustacea: A review. Reprod. Nutr. Dev. 1980, 20, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Girish, B.P.; Swetha, C.; Reddy, P.S. Hepatopancreas but not ovary is the site of vitellogenin synthesis in female fresh water crab, Oziothelphusa senex senex. Biochem. Biophys. Res. Commun. 2014, 447, 323–327. [Google Scholar] [CrossRef] [PubMed]
- Mak, A.S.; Choi, C.L.; Tiu, S.H.; Hui, J.H.; He, J.G.; Tobe, S.S.; Chan, S.M. Vitellogenesis in the red crab Charybdis feriatus: Hepatopancreas-specific expression and farnesoic acid stimulation of vitellogenin gene expression. Mol. Reprod. Dev. 2005, 70, 288–300. [Google Scholar] [CrossRef] [PubMed]
- Kolarevic, J.; Nerland, A.; Nilsen, F.; Finn, R.N. Goldsinny wrasse (Ctenolabrus rupestris) is an extreme vtgAa-type pelagophil teleost. Mol. Reprod. Dev. 2008, 75, 1011–1020. [Google Scholar] [CrossRef] [PubMed]
- Sawaguchi, S.; Ohkubo, N.; Koya, Y.; Matsubara, T. Incorporation and utilization of multiple forms of vitellogenin and their derivative yolk proteins during vitellogenesis and embryonic development in the mosquitofish, Gambusia affinis. Zool. Sci. 2005, 22, 701–710. [Google Scholar] [CrossRef] [PubMed]
- Amano, H.; Fujita, T.; Hiramatsu, N.; Kagawa, H.; Matsubara, T.; Sullivan, C.V.; Hara, A. Multiple vitellogenin-derived yolk proteins in gray mullet (Mugil cephalus): Disparate proteolytic patterns associated with ovarian follicle maturation. Mol. Reprod. Deve. 2008, 75, 1307–1317. [Google Scholar] [CrossRef] [PubMed]
- Wallace, R.A.; Selman, K. Ultrastructural aspects of oogenesis and oocyte growth in fish and amphibians. J. Electron Microsc. Tech. 1990, 16, 175–201. [Google Scholar] [PubMed]
- Conner, S.D.; Schmid, S.L. Regulated portals of entry into the cell. Nature 2003, 422, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Hiramatsu, N.; Todo, T.; Sullivan, C.V.; Schilling, J.; Reading, B.J.; Matsubara, T.; Ryu, Y.W.; Mizuta, H.; Luo, W.; Nishimiya, O.; et al. Ovarian yolk formation in fishes: Molecular mechanisms underlying formation of lipid droplets and vitellogenin-derived yolk proteins. Gen. Comp. Endocrinol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Reading, B.J.; Hiramatsu, N.; Schilling, J.; Molloy, K.T.; Glassbrook, N.; Mizuta, H.; Luo, W.; Baltzegar, D.A.; Williams, V.N.; Todo, T.; et al. Lrp13 is a novel vertebrate lipoprotein receptor that binds vitellogenins in teleost fishes. J. Lipid Res. 2014, 55, 2287–2295. [Google Scholar] [CrossRef] [PubMed]
- Reading, B.J.; Hiramatsu, N.; Sullivan, C.V. Disparate binding of three types of vitellogenin to multiple forms of vitellogenin receptor in white perch. Biol. Reprod. 2011, 84, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Williams, V.N.; Reading, B.J.; Amano, H.; Hiramatsu, N.; Schilling, J.; Salger, S.A.; Williams, T.I.; Gross, K.; Sullivan, C.V. Proportional accumulation of yolk proteins derived from multiple vitellogenins is precisely regulated during vitellogenesis in striped bass (Morone saxatilis). J. Exp. Zool. A Ecol. Genet. Physiol. 2014, 321, 301–315. [Google Scholar] [CrossRef] [PubMed]
- Romano, M.; Rosanova, P.; Anteo, C.; Limatola, E. Vertebrate yolk proteins: A review. Mol. Reprod. Dev. 2004, 69, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Carnevali, O.; Carletta, R.; Cambi, A.; Vita, A.; Bromage, N. Yolk formation and degradation during oocyte maturation in seabream Sparus aurata: Involvement of two lysosomal proteinases. Biol. Reprod. 1999, 60, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Carnevali, O.; Cionna, C.; Tosti, L.; Lubzens, E.; Maradonna, F. Role of cathepsins in ovarian follicle growth and maturation. Gen. Comp. Endocrinol. 2006, 146, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Hiramatsu, N.; Ichikawa, N.; Fukada, H.; Fujita, T.; Sullivan, C.V.; Hara, A. Identification and characterization of proteases involved in specific proteolysis of vitellogenin and yolk proteins in salmonids. J. Exp. Zool. 2002, 292, 11–25. [Google Scholar] [CrossRef] [PubMed]
- Fabra, M.; Cerda, J. Ovarian cysteine proteinases in the teleost Fundulus heteroclitus: Molecular cloning and gene expression during vitellogenesis and oocyte maturation. Mol. Reprod. Dev. 2004, 67, 282–294. [Google Scholar] [CrossRef] [PubMed]
- Opresko, L.K.; Karpf, R.A. Specific proteolysis regulates fusion between endocytic compartments in Xenopus oocytes. Cell 1987, 51, 557–568. [Google Scholar] [CrossRef]
- Sire, M.F.; Babin, P.J.; Vernier, J.M. Involvement of the lysosomal system in yolk protein deposit and degradation during vitellogenesis and embryonic development in trout. J. Exp. Zool. 1994, 269, 69–83. [Google Scholar] [CrossRef]
- Retzek, H.; Steyrer, E.; Sanders, E.J.; Nimpf, J.; Schneider, W.J. Molecular cloning and functional characterization of chicken cathepsin D, a key enzyme for yolk formation. DNA Cell Biol. 1992, 11, 661–672. [Google Scholar] [CrossRef] [PubMed]
- Babin, P.J. Apolipoproteins and the association of egg yolk proteins with plasma high density lipoproteins after ovulation and follicular atresia in the rainbow trout (Salmo gairdneri). J. Biol. Chem. 1987, 262, 4290–4296. [Google Scholar] [PubMed]
- Matsubara, T.; Ohkubo, N.; Andoh, T.; Sullivan, C.V.; Hara, A. Two forms of vitellogenin, yielding two distinct lipovitellins, play different roles during oocyte maturation and early development of barfin flounder, Verasper moseri, a marine teleost that spawns pelagic eggs. Dev. Biol. 1999, 213, 18–32. [Google Scholar] [CrossRef] [PubMed]
- Tyler, C.R.; Sumpter, J.P.; Bromage, N.R. In vivo ovarian uptake and processing of vitellogenin in the rainbow trout, Salmo gairdneri. J. Exp. Zool. 1988, 246, 171–179. [Google Scholar] [CrossRef]
- Yilmaz, O.; Prat, F.; Ibanez, A.J.; Amano, H.; Koksoy, S.; Sullivan, C.V. Estrogen-induced yolk precursors in European sea bass, Dicentrarchus labrax: Status and perspectives on multiplicity and functioning of vitellogenins. Gen. Comp. Endocrinol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Finn, R.N.; Fyhn, H.J. Requirement for amino acids in ontogeny of fish. Aquac. Res. 2010, 41, 684–716. [Google Scholar] [CrossRef]
- Arukwe, A.; Goksoyr, A. Eggshell and egg yolk proteins in fish: Hepatic proteins for the next generation: Oogenetic, population, and evolutionary implications of endocrine disruption. Comp. Hepatol. 2003, 2. [Google Scholar] [CrossRef]
- Pan, M.L.; Bell, W.J.; Telfer, W.H. Vitellogenic blood protein synthesis by insect fat body. Science 1969, 165, 393–394. [Google Scholar] [CrossRef] [PubMed]
- Nath, P.; Sundararaj, B.I. Isolation and identification of female-specific serum lipophosphoprotein (vitellogenin) in the catfish, Heteropneustes fossilis (Bloch). Gen. Comp. Endocrinol. 1981, 43, 184–190. [Google Scholar] [CrossRef]
- Shyu, A.B.; Raff, R.A.; Blumenthal, T. Expression of the vitellogenin gene in female and male sea urchin. Proc. Natl. Acad. Sci. USA 1986, 83, 3865–3869. [Google Scholar] [CrossRef] [PubMed]
- Piulachs, M.D.; Guidugli, K.R.; Barchuk, A.R.; Cruz, J.; Simoes, Z.L.; Belles, X. The vitellogenin of the honey bee, Apis mellifera: Structural analysis of the cdna and expression studies. Insect Biochem. Mol. Biol. 2003, 33, 459–465. [Google Scholar] [CrossRef]
- Scharf, M.E.; Wu-Scharf, D.; Zhou, X.; Pittendrigh, B.R.; Bennett, G.W. Gene expression profiles among immature and adult reproductive castes of the termite Reticulitermes flavipes. Insect Mol. Biol. 2005, 14, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Amdam, G.V.; Norberg, K.; Hagen, A.; Omholt, S.W. Social exploitation of vitellogenin. Proc. Natl. Acad. Sci. USA 2003, 100, 1799–1802. [Google Scholar] [CrossRef] [PubMed]
- Amdam, G.V.; Norberg, K.; Page, R.E., Jr.; Erber, J.; Scheiner, R. Downregulation of vitellogenin gene activity increases the gustatory responsiveness of honey bee workers (Apis mellifera). Behav. Brain Res. 2006, 169, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Guidugli, K.R.; Nascimento, A.M.; Amdam, G.V.; Barchuk, A.R.; Omholt, S.; Simoes, Z.L.; Hartfelder, K. Vitellogenin regulates hormonal dynamics in the worker caste of a eusocial insect. FEBS Lett. 2005, 579, 4961–4965. [Google Scholar] [CrossRef] [PubMed]
- Nelson, C.M.; Ihle, K.E.; Fondrk, M.K.; Page, R.E.; Amdam, G.V. The gene vitellogenin has multiple coordinating effects on social organization. PLoS Biol. 2007, 5, e62. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Sun, Y.; Pang, Q.; Shi, X. Hemagglutinating and antibacterial activities of vitellogenin. Fish Shellfish Immunol. 2005, 19, 93–95. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Zhang, S.; Pang, Q. Vitellogenin is a novel player in defense reactions. Fish Shellfish Immunol. 2006, 20, 769–772. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.H.; Zhang, S.C.; Li, Z.J.; Gao, C.R. Characterization of a pattern recognition molecule vitellogenin from carp (Cyprinus carpio). Immunobiology 2009, 214, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Liu, Z.; Zhou, L.; Ji, G.; Yang, A. Molecular cloning, expression, purification and characterization of vitellogenin in scallop Patinopecten yessoensis with special emphasis on its antibacterial activity. Deve. Comp. Immunol. 2015, 49, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Fischer, M.; Regitz, C.; Kull, R.; Boll, M.; Wenzel, U. Vitellogenins increase stress resistance of Caenorhabditis elegans after Photorhabdus luminescens infection depending on the steroid-signaling pathway. Microbes Infect. 2013, 15, 569–578. [Google Scholar] [CrossRef] [PubMed]
- Fischer, M.; Regitz, C.; Kahl, M.; Werthebach, M.; Boll, M.; Wenzel, U. Phytoestrogens genistein and daidzein affect immunity in the nematode Caenorhabditis elegans via alterations of vitellogenin expression. Mol. Nutr. Food Res. 2012, 56, 957–965. [Google Scholar] [CrossRef] [PubMed]
- Lu, A.; Hu, X.; Wang, Y.; Shen, X.; Zhu, A.; Shen, L.; Ming, Q.; Feng, Z. Comparative analysis of the acute response of zebrafish Danio rerio skin to two different bacterial infections. J. Aquat. Anim. Health 2013, 25, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Lu, A.; Hu, X.; Xue, J.; Zhu, J.; Wang, Y.; Zhou, G. Gene expression profiling in the skin of zebrafish infected with Citrobacter freundii. Fish Shellfish Immunol. 2012, 32, 273–283. [Google Scholar] [CrossRef] [PubMed]
- Nachappa, P.; Levy, J.; Tamborindeguy, C. Transcriptome analyses of Bactericera cockerelli adults in response to “Candidatus Liberibacter solanacearum” infection. Mol. Genet. Genom. 2012, 287, 803–817. [Google Scholar] [CrossRef] [PubMed]
- Tong, Z.; Li, L.; Pawar, R.; Zhang, S. Vitellogenin is an acute phase protein with bacterial-binding and inhibiting activities. Immunobiology 2010, 215, 898–902. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zhang, S.; Liu, Q. Vitellogenin functions as a multivalent pattern recognition receptor with an opsonic activity. PLoS ONE 2008, 3, e1940. [Google Scholar] [CrossRef] [PubMed]
- Udompetcharaporn, A.; Junkunlo, K.; Senapin, S.; Roytrakul, S.; Flegel, T.W.; Sritunyalucksana, K. Identification and characterization of a QM protein as a possible peptidoglycan recognition protein (PGRP) from the giant tiger shrimp Penaeus monodon. Dev. Comp. Immunol. 2014, 46, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zhang, S.; Zhang, J.; Liu, M.; Liu, Z. Vitellogenin is a cidal factor capable of killing bacteria via interaction with lipopolysaccharide and lipoteichoic acid. Mol. Immunol. 2009, 46, 3232–3239. [Google Scholar] [CrossRef] [PubMed]
- Bouchard, B.; Gagne, F.; Fortier, M.; Fournier, M. An in-situ study of the impacts of urban wastewater on the immune and reproductive systems of the freshwater mussel Elliptio complanata. Comp. Biochem. Physiol. C Toxicol. Pharm. 2009, 150, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Pan, J.; Ji, H.; Zhao, B.; Zhang, S. Vitellogenin mediates phagocytosis through interaction with FcγR. Mol. Immunol. 2011, 49, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Garcia, J.; Munro, E.S.; Monte, M.M.; Fourrier, M.C.; Whitelaw, J.; Smail, D.A.; Ellis, A.E. Atlantic salmon (Salmo salar L.) serum vitellogenin neutralises infectivity of infectious pancreatic necrosis virus (IPNV). Fish Shellfish Immunol. 2010, 29, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Rono, M.K.; Whitten, M.M.; Oulad-Abdelghani, M.; Levashina, E.A.; Marois, E. The major yolk protein vitellogenin interferes with the anti-plasmodium response in the malaria mosquito Anopheles gambiae. PLoS Biol. 2010, 8, e1000434. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, S. Lipovitellin is a non-self recognition receptor with opsonic activity. Mar. Biotechnol. 2011, 13, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wang, Y.; Ma, J.; Ding, Y.; Zhang, S. Phosvitin plays a critical role in the immunity of zebrafish embryos via acting as a pattern recognition receptor and an antimicrobial effector. J. Biol. Chem. 2011, 286, 22653–22664. [Google Scholar] [CrossRef] [PubMed]
- Sattar Khan, M.A.; Nakamura, S.; Ogawa, M.; Akita, E.; Azakami, H.; Kato, A. Bactericidal action of egg yolk phosvitin against Escherichia coli under thermal stress. J. Agric. Food Chem. 2000, 48, 1503–1506. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Wang, H.; Wang, Y.; Zhang, S. Endotoxin-neutralizing activity of hen egg phosvitin. Mol. Immunol. 2013, 53, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Liu, X.; Bu, L.; Li, H.; Zhang, S. Antimicrobial-immunomodulatory activities of zebrafish phosvitin-derived peptide Pt5. Peptides 2012, 37, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Sun, C.; Wang, S.; Su, F.; Zhang, S. Lipopolysaccharide neutralization by a novel peptide derived from phosvitin. Int. J. Biochem. Cell Biol. 2013, 45, 2622–2631. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Hu, L.; Liu, S.; Hu, G.; Zhang, S. Antiviral activity of phosvitin from zebrafish Danio rerio. Deve. Comp. Immunol. 2013, 40, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Ando, S.; Yanagida, K. Susceptibility to oxidation of copper-induced plasma lipoproteins from Japanese eel: Protective effect of vitellogenin on the oxidation of very low density lipoprotein. Comp. Biochem. Physiol. Part C Pharmacol. Toxicol. Endocrinol. 1999, 123, 1–7. [Google Scholar] [CrossRef]
- Nakamura, A.; Yasuda, K.; Adachi, H.; Sakurai, Y.; Ishii, N.; Goto, S. Vitellogenin-6 is a major carbonylated protein in aged nematode, Caenorhabditis elegans. Biochem. Biophys. Res. Commun. 1999, 264, 580–583. [Google Scholar] [CrossRef] [PubMed]
- Seehuus, S.C.; Norberg, K.; Gimsa, U.; Krekling, T.; Amdam, G.V. Reproductive protein protects functionally sterile honey bee workers from oxidative stress. Proc. Natl. Acad. Sci. USA 2006, 103, 962–967. [Google Scholar] [CrossRef] [PubMed]
- Corona, M.; Velarde, R.A.; Remolina, S.; Moran-Lauter, A.; Wang, Y.; Hughes, K.A.; Robinson, G.E. Vitellogenin, juvenile hormone, insulin signaling, and queen honey bee longevity. Proc. Natl. Acad. Sci. USA 2007, 104, 7128–7133. [Google Scholar] [CrossRef] [PubMed]
- Havukainen, H.; Munch, D.; Baumann, A.; Zhong, S.; Halskau, O.; Krogsgaard, M.; Amdam, G.V. Vitellogenin recognizes cell damage through membrane binding and shields living cells from reactive oxygen species. J. Biol. Chem. 2013, 288, 28369–28381. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.L.; Baker, R.C. Characteristics of egg yolk phosvitin as an antioxidant for inhibiting metal-catalyzed phospholipid oxidations. Poult. Sci. 1986, 65, 2065–2070. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, S.; Yano, Y.; Arihara, K.; Itoh, M. Egg yolk phosvitin inhibits hydroxyl radical formation from the fenton reaction. Biosci. Biotechnol. Biochem. 2004, 68, 1324–1331. [Google Scholar] [CrossRef] [PubMed]
- Guérin-Dubiard, C.; Anton, M.; Dhene-Garcia, A.; Martinet, V.; Brulé, G. Hen egg and fish egg phosvitins: Composition and iron binding properties. Eur. Food Res. Technol. 2002, 214, 460–464. [Google Scholar] [CrossRef]
- Hu, L.; Sun, C.; Luan, J.; Lu, L.; Zhang, S. Zebrafish phosvitin is an antioxidant with non-cytotoxic activity. Acta Biochim. Biophys. Sin. 2015, 47, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Maria-Neto, S.; de Almeida, K.C.; Macedo, M.L.; Franco, O.L. Understanding bacterial resistance to antimicrobial peptides: From the surface to deep inside. Biochim. Biophys. Acta 2015. [Google Scholar] [CrossRef] [PubMed]
- Grundmann, H.; Klugman, K.P.; Walsh, T.; Ramon-Pardo, P.; Sigauque, B.; Khan, W.; Laxminarayan, R.; Heddini, A.; Stelling, J. A framework for global surveillance of antibiotic resistance. Drug Resist. Updates 2011, 14, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Wiesner, J.; Vilcinskas, A. Antimicrobial peptides: The ancient arm of the human immune system. Virulence 2010, 1, 440–464. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.L.; Hancock, R.E. Cationic host defense (antimicrobial) peptides. Curr. Opin. Immunol. 2006, 18, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Uppu, D.S.; Ghosh, C.; Haldar, J. Surviving sepsis in the era of antibiotic resistance: Are there any alternative approaches to antibiotic therapy? Microb. Pathog. 2015, 80, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Knight, J.A. The biochemistry of aging. Adv. Clin. Chem. 2000, 35, 1–62. [Google Scholar] [PubMed]
- Lupo, M.P. Antioxidants and vitamins in cosmetics. Clin. Dermatol. 2001, 19, 467–473. [Google Scholar] [CrossRef]
- Bonilla, J.; Atares, L.; Chiralt, A.; Vargas, M. Recent patents on the use of antioxidant agents in food. Recent Pat. Food Nutr. Agric. 2011, 3, 123–132. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, C.; Zhang, S. Immune-Relevant and Antioxidant Activities of Vitellogenin and Yolk Proteins in Fish. Nutrients 2015, 7, 8818-8829. https://doi.org/10.3390/nu7105432
Sun C, Zhang S. Immune-Relevant and Antioxidant Activities of Vitellogenin and Yolk Proteins in Fish. Nutrients. 2015; 7(10):8818-8829. https://doi.org/10.3390/nu7105432
Chicago/Turabian StyleSun, Chen, and Shicui Zhang. 2015. "Immune-Relevant and Antioxidant Activities of Vitellogenin and Yolk Proteins in Fish" Nutrients 7, no. 10: 8818-8829. https://doi.org/10.3390/nu7105432
APA StyleSun, C., & Zhang, S. (2015). Immune-Relevant and Antioxidant Activities of Vitellogenin and Yolk Proteins in Fish. Nutrients, 7(10), 8818-8829. https://doi.org/10.3390/nu7105432




