DHA in Pregnant and Lactating Women from Coastland, Lakeland, and Inland Areas of China: Results of a DHA Evaluation in Women (DEW) Study
Abstract
:1. Introduction
2. Subjects and Methods
2.1. Settings and Subjects
2.2. Data and Sample Collection
2.3. Sample Analysis
2.4. Statistical Analysis
3. Results
3.1. Maternal Characteristics
| Coastland | Lakeland | Inland | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Characteristic | MP | LP | LA | MP | LP | LA | MP | LP | LA |
| Number of participants | 136 | 127 | 136 | 133 | 134 | 135 | 138 | 136 | 136 |
| GA (week)/PP (day) at enrolment | |||||||||
| Mean | 16.9 | 37.5 | 42.7 | 17.0 | 38.0 | 41.7 | 16.7 | 38.6 | 42.1 |
| SD | 0.9 | 0.7 | 2.3 | 1.1 | 0.9 | 4.1 | 1.1 | 1.2 | 3.9 |
| Age (year) | |||||||||
| Mean | 27.9 | 28.4 | 28.3 | 26.5 | 27.1 | 27.1 | 27.9 | 28.5 | 28.1 |
| SD | 2.4 | 2.7 | 2.7 | 3.1 | 3.1 | 3.0 | 2.9 | 3.3 | 3.0 |
| Ethnics (%) | |||||||||
| Han | 97.8 | 99.2 | 97.1 | 98.5 | 97.8 | 97.8 | 83.3 | 94.1 | 91.9 |
| Mongolian | 0 | 0 | 0 | 0 | 0 | 0 | 12.3 | 4.4 | 4.4 |
| Hui | 0 | 0 | 0.7 | 0 | 0 | 0 | 0.7 | 0.7 | 2.2 |
| Others | 2.2 | 0.8 | 2.2 | 1.5 | 2.2 | 2.2 | 3.6 | 0.7 | 1.5 |
| Education (%) | |||||||||
| College or above | 67.7 | 66.2 | 65.4 | 47.4 | 61.3 | 62.2 | 79.0 | 78.4 | 73.5 |
| High school | 15.4 | 24.4 | 25.0 | 36.8 | 24.6 | 23.7 | 13.8 | 16.2 | 19.1 |
| Middle school or less | 16.9 | 9.4 | 9.6 | 15.8 | 14.1 | 14.1 | 7.2 | 15.4 | 7.4 |
| Height (cm) | |||||||||
| Mean | 163.6 | 163.7 | 163.2 | 159.3 | 159.8 | 159.8 | 164.1 | 162.8 | 163.1 |
| SD | 4.9 | 4.6 | 5.0 | 4.5 | 3.9 | 4.3 | 4.9 | 4.7 | 4.6 |
| Pre-pregnancy BMI (kg/m2) | |||||||||
| Mean | 21.1 | 21.3 | 21.9 | 20.2 | 19.8 | 20.1 | 21.3 | 21.6 | 21.2 |
| SD | 2.8 | 2.5 | 3.5 | 2.5 | 1.9 | 2.9 | 3.3 | 2.8 | 3.2 |
3.2. DHA Concentrations
| Inland | Lakeland | Coastland | P ANOVA | ||||
|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | ||
| wt. % | |||||||
| Plasma | |||||||
| Mid-pregnancy | 2.25 | 0.46 | 2.45 a | 0.44 | 3.19 a | 0.65 | <0.001 |
| Late pregnancy | 1.67 b | 0.35 | 1.95 a,b | 0.45 | 2.54 a,b | 0.60 | <0.001 |
| Lactation | 1.68 b | 0.48 | 2.26 a,b | 0.53 | 2.24 a,b | 0.70 | <0.001 |
| P ANOVA | <0.001 | <0.001 | <0.001 | ||||
| Erythrocyte | |||||||
| Mid-pregnancy | 5.85 | 1.06 | 6.34 a | 0.80 | 7.59 a | 1.46 | <0.001 |
| Late pregnancy | 5.06 b | 1.25 | 6.23 a | 1.09 | 7.09 a,b | 1.93 | <0.001 |
| Lactation | 5.20 b | 1.15 | 6.20 a | 0.92 | 6.07 a,b | 1.59 | <0.001 |
| P ANOVA | <0.001 | 0.45 | <0.001 | ||||
| μg/mL | |||||||
| Plasma | |||||||
| Mid-pregnancy | 93.4 | 25.2 | 86.2 a | 20.5 | 118.4 a | 30.8 | <0.001 |
| Late pregnancy | 101.9 b | 33.1 | 110.1 a,b | 28.3 | 146.2 a,b | 45.1 | <0.001 |
| Lactation | 60.5 b | 17.5 | 65.1 b | 20.9 | 75.7 a,b | 29.9 | <0.001 |
| P ANOVA | <0.001 | <0.001 | <0.001 | ||||
| Erythrocyte | |||||||
| Mid-pregnancy | 108.0 | 22.1 | 103.2 a | 13.6 | 127.7 a | 27.2 | <0.001 |
| Late pregnancy | 89.3 b | 25.2 | 106.6 a | 20.7 | 123.2 a | 37.5 | <0.001 |
| Lactation | 91.4 b | 22.1 | 97.0 a,b | 19.7 | 99.3 a,b | 29.6 | <0.05 |
| P ANOVA | <0.001 | <0.001 | <0.001 | ||||
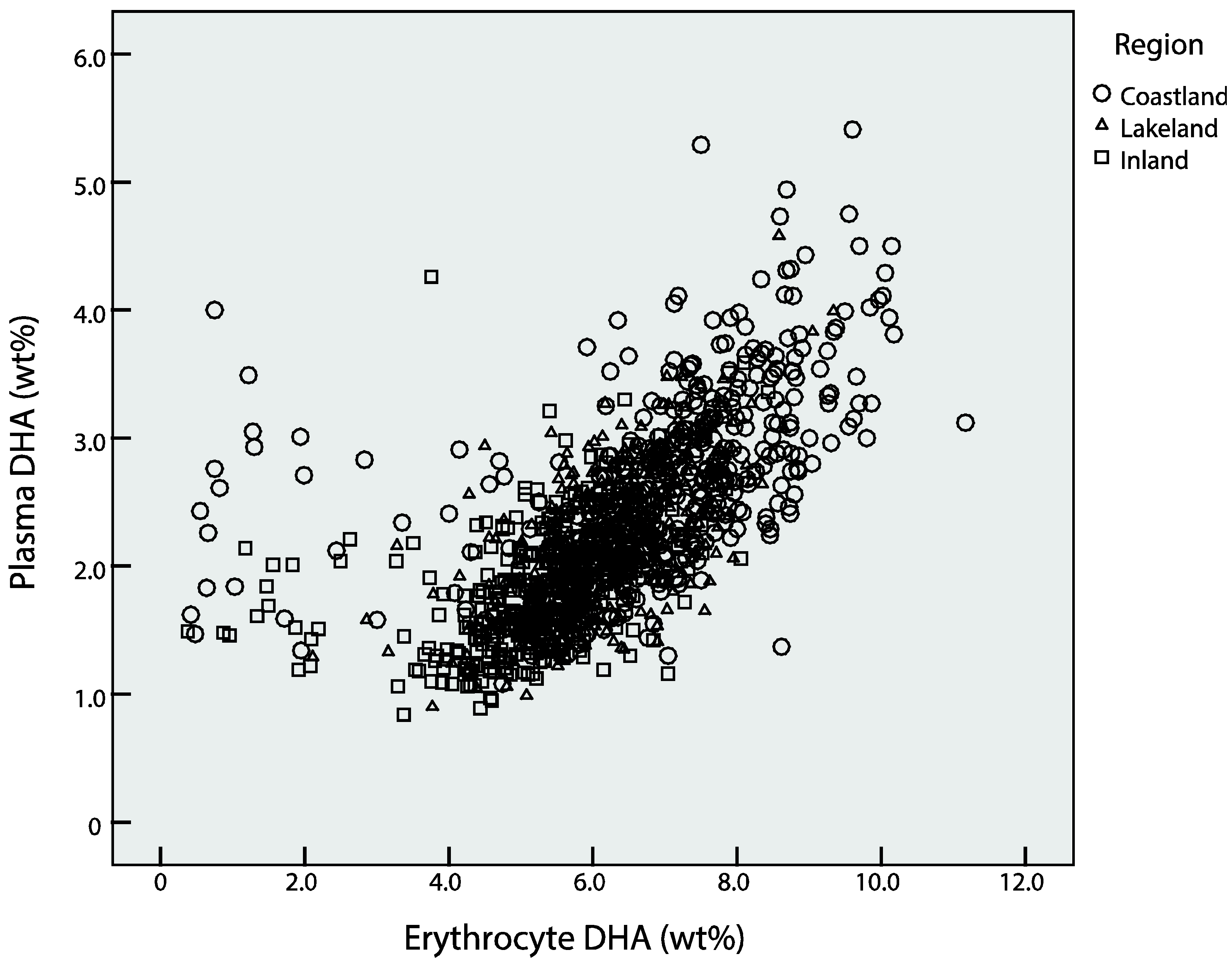
3.3. Correlation between DHA in Plasma and Erythrocyte
| Variables | Plasma DHA (wt. %) | Erythrocyte DHA (wt. %) | Plasma DHA (μg/mL) | Erythrocyte DHA (μg/mL) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | β | P | Mean | β | P | Mean | β | P | Mean | β | P | |
| Region | ||||||||||||
| Inland | 1.87 | 0 | Ref. | 5.37 | 0 | Ref. | 85.3 | 0 | Ref. | 96.3 | 0 | Ref. |
| Lakeland | 2.22 | 0.36 | <0.001 | 6.25 | 0.92 | <0.001 | 87.0 | 3.6 | 0.091 | 102.2 | 6.7 | <0.001 |
| Coastland | 2.66 | 0.80 | <0.001 | 6.91 | 1.55 | <0.001 | 112.7 | 28.4 | <0.001 | 116.6 | 20.4 | <0.001 |
| Participant group | ||||||||||||
| Mid-pregnancy | 2.63 | 0 | Ref. | 6.59 | 0 | Ref. | 99.4 | 0 | Ref. | 113.0 | 0 | Ref. |
| Late pregnancy | 2.04 | −0.59 | <0.001 | 6.10 | −0.49 | <0.001 | 118.8 | 19.4 | <0.001 | 106.0 | −7.0 | <0.001 |
| Lactation | 2.06 | −0.58 | <0.001 | 5.82 | −0.79 | <0.001 | 67.1 | −32.9 | <0.001 | 95.9 | −17.5 | <0.001 |
| Age(year) | ||||||||||||
| <25.0 | 2.15 | 0 | Ref. | 5.93 | 0 | Ref. | 88.2 | 0 | Ref. | 100.2 | 0 | Ref. |
| 25.0–29.9 | 2.27 | 0.02 | 0.623 | 6.24 | 0.17 | 0.129 | 94.7 | 1.7 | 0.499 | 106.1 | 2.8 | 0.191 |
| ≥30.0 | 2.25 | 0.12 | 0.029 | 6.15 | 0.25 | 0.050 | 100.6 | 7.9 | 0.006 | 105.3 | 4.0 | 0.108 |
| Education | ||||||||||||
| Middle school or less | 2.18 | 0 | Ref. | 5.93 | 0 | Ref. | 90.1 | 0 | Ref. | 100.2 | 0 | Ref. |
| High school | 2.24 | 0.07 | 0.187 | 6.20 | 0.29 | 0.033 | 94.2 | 7.2 | 0.016 | 105.0 | 5.6 | 0.034 |
| College or above | 2.26 | 0.12 | 0.014 | 6.21 | 0.36 | 0.003 | 96.0 | 8.5 | 0.002 | 105.8 | 6.4 | 0.006 |
| Pre-pregnancy BMI | ||||||||||||
| <18.5 | 2.26 | 0 | Ref. | 6.15 | 0 | Ref. | 90.7 | 0 | Ref. | 103.0 | 0 | Ref. |
| 18.5–23.9 | 2.27 | −0.07 | 0.073 | 6.22 | −0.06 | 0.546 | 96.0 | −0.3 | 0.901 | 105.9 | 0.1 | 0.970 |
| ≥24.0 | 2.12 | −0.21 | <0.001 | 5.97 | −0.20 | 0.143 | 95.3 | 1.6 | 0.612 | 103.2 | −1.7 | 0.523 |
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Heird, W.C.; Lapillonne, A. The role of essential fatty acids in development. Ann. Rev. Nutr. 2005, 25, 549–571. [Google Scholar] [CrossRef] [PubMed]
- Lauritzen, L.; Hansen, H.S.; Jorgensen, M.H.; Michaelsen, K.F. The essentiality of long chain n-3 fatty acids in relation to development and function of the brain and retina. Prog. Lipid Res. 2001, 40, 1–94. [Google Scholar] [CrossRef]
- FAO. Fats and Fatty Acids in Human Nutrition; Report of an Expert Consultation; Karger: Basel, Switzerland, 2010; Volume 91, pp. 1–166. [Google Scholar]
- Carlson, S.E.; Colombo, J.; Gajewski, B.J.; Gustafson, K.M.; Mundy, D.; Yeast, J.; Georgieff, M.K.; Markley, L.A.; Kerling, E.H.; Shaddy, D.J.; et al. DHA supplementation and pregnancy outcomes. Am. J. Clin. Nutr. 2013, 97, 808–815. [Google Scholar] [CrossRef] [PubMed]
- Hibbeln, J.R.; Davis, J.M.; Steer, C.; Emmett, P.; Rogers, I.; Williams, C.; Golding, J. Maternal seafood consumption in pregnancy and neurodevelopmental outcomes in childhood (ALSPAC study): An observational cohort study. Lancet 2007, 369, 578–585. [Google Scholar] [CrossRef]
- Makrides, M.; Gibson, R.A.; McPhee, A.J.; Yelland, L.; Quinlivan, J.; Ryan, P.; Team, D.O.I. Effect of DHA supplementation during pregnancy on maternal depression and neurodevelopment of young children: A randomized controlled trial. JAMA 2010, 304, 1675–1683. [Google Scholar] [CrossRef] [PubMed]
- Muthayya, S.; Dwarkanath, P.; Thomas, T.; Ramprakash, S.; Mehra, R.; Mhaskar, A.; Mhaskar, R.; Thomas, A.; Bhat, S.; Vaz, M.; et al. The effect of fish and omega-3 LCPUFA intake on low birth weight in Indian pregnant women. Eur. J. Clin. Nutr. 2009, 63, 340–346. [Google Scholar] [CrossRef] [PubMed]
- Luxwolda, M.F.; Kuipers, R.S.; Koops, J.H.; Muller, S.; de Graaf, D.; Dijck-Brouwer, D.A.; Muskiet, F.A. Interrelationships between maternal DHA in erythrocytes, milk and adipose tissue. Is 1 wt % DHA the optimal human milk content? Data from four tanzanian tribes differing in lifetime stable intakes of fish. Br. J. Nutr. 2014, 111, 854–866. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Van Eijsden, M.; Hornstra, G.; van der Wal, M.F.; Bonsel, G.J. Ethnic differences in early pregnancy maternal n-3 and n-6 fatty acid concentrations: An explorative analysis. Br. J. Nutr. 2009, 101, 1761–1768. [Google Scholar] [CrossRef] [PubMed]
- Otto, S.J.; Houwelingen, A.C.; Antal, M.; Manninen, A.; Godfrey, K.; Lopez-Jaramillo, P.; Hornstra, G. Maternal and neonatal essential fatty acid status in phospholipids: An international comparative study. Eur. J. Clin. Nutr. 1997, 51, 232–242. [Google Scholar] [CrossRef] [PubMed]
- Metherel, A.H.; Aristizabal Henao, J.J.; Stark, K.D. EPA and DHA levels in whole blood decrease more rapidly when stored at −20 °C as compared with room temperature, 4 and −75 °C. Lipids 2013, 48, 1079–1091. [Google Scholar] [CrossRef] [PubMed]
- Metherel, A.H.; Stark, K.D. Cryopreservation prevents iron-initiated highly unsaturated fatty acid loss during storage of human blood on chromatography paper at −20 °C. J. Nutr. 2015, 145, 654–660. [Google Scholar] [CrossRef] [PubMed]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [PubMed]
- Zhang, J.; Wang, Y.; Meng, L.; Wang, C.; Zhao, W.; Chen, J.; Ghebremeskel, K.; Crawford, M.A. Maternal and neonatal plasma n-3 and n-6 fatty acids of pregnant women and neonates from three regions of China with contrasting dietary patterns. Asia Pac. J. Clin. Nutr. 2009, 18, 377–388. [Google Scholar] [PubMed]
- Al, M.D.M.; van Houwelingen, A.C.; Hornstra, G. Relation between birth order and the maternal and neonatal docosahexaenoic acid status. Eur. J. Clin. Nutr. 1997, 51, 548–553. [Google Scholar] [CrossRef] [PubMed]
- Koletzko, B.; Larque, E.; Demmelmair, H. Placental transfer of long-chain polyunsaturated fatty acids (LC-PUFA). J. Perinat. Med. 2007, 35 (Suppl. S1), S5–S11. [Google Scholar] [CrossRef] [PubMed]
- Stark, K.D.; Beblo, S.; Murthy, M.; Buda-Abela, M.; Janisse, J.; Rockett, H.; Whitty, J.E.; Martier, S.S.; Sokol, R.J.; Hannigan, J.H.; et al. Comparison of bloodstream fatty acid composition from African-American women at gestation, delivery, and postpartum. J. Lipid Res. 2005, 46, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Stark, K.D.; Beblo, S.; Murthy, M.; Whitty, J.E.; Buda-Abela, M.; Janisse, J.; Rockett, H.; Martier, S.S.; Sokol, R.J.; Hannigan, J.H.; et al. Alcohol consumption in pregnant, black women is associated with decreased plasma and erythrocyte docosahexaenoic acid. Alcohol. Clin. Exp. Res. 2005, 29, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Wadhwani, N.; Patil, V.; Pisal, H.; Joshi, A.; Mehendale, S.; Gupte, S.; Wagh, G.; Joshi, S. Altered maternal proportions of long chain polyunsaturated fatty acids and their transport leads to disturbed fetal stores in preeclampsia. Prostaglandins Leukot. Essent. Fatty Acids 2014, 91, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Enke, U.; Jaudszus, A.; Schleussner, E.; Seyfarth, L.; Jahreis, G.; Kuhnt, K. Fatty acid distribution of cord and maternal blood in human pregnancy: Special focus on individual trans fatty acids and conjugated linoleic acids. Lipids Health Dis. 2011, 10, 247. [Google Scholar] [CrossRef] [PubMed]
- Markhus, M.W.; Skotheim, S.; Graff, I.E.; Froyland, L.; Braarud, H.C.; Stormark, K.M.; Malde, M.K. Low omega-3 index in pregnancy is a possible biological risk factor for postpartum depression. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Olsen, S.F.; Hansen, H.S.; Sommer, S.; Jensen, B.; Sorensen, T.I.; Secher, N.J.; Zachariassen, P. Gestational age in relation to marine n-3 fatty acids in maternal erythrocytes: A study of women in the Faroe Islands and Denmark. Am. J. Obstet. Gynecol. 1991, 164, 1203–1209. [Google Scholar] [CrossRef]
- Rioux, F.M.; Belanger-Plourde, J.; Leblanc, C.P.; Vigneau, F. Relationship between maternal DHA and iron status and infants’ cognitive performance. Can. J. Diet. Pract. Res. 2011, 72, 76. [Google Scholar] [PubMed]
- Stewart, F.; Rodie, V.A.; Ramsay, J.E.; Greer, I.A.; Freeman, D.J.; Meyer, B.J. Longitudinal assessment of erythrocyte fatty acid composition throughout pregnancy and post partum. Lipids 2007, 42, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Young, C.; Hikita, T.; Kaneko, S.; Shimizu, Y.; Hanaka, S.; Abe, T.; Shimasaki, H.; Ikeda, R.; Miyazawa, Y.; Nakajima, A. Fatty acid compositions of colostrum, cord blood, maternal blood and major infant formulas in Japan. Acta Paediatr. Jpn. 1997, 39, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Sanjurjo, P.; Matorras, R.; Ingunza, N.; Alonso, M.; Rodriguez-Alarcon, J.; Perteagudo, L. Cross-sectional study of percentual changes in total plasmatic fatty acids during pregnancy. Horm. Metab. Res. 1993, 25, 590–592. [Google Scholar] [CrossRef] [PubMed]
- Krasevec, J.M.; Jones, P.J.; Cabrera-Hernandez, A.; Mayer, D.L.; Connor, W.E. Maternal and infant essential fatty acid status in Havana, Cuba. Am. J. Clin. Nutr. 2002, 76, 834–844. [Google Scholar] [PubMed]
- Xie, L.; Innis, S.M. Genetic variants of the fads1 fads2 gene cluster are associated with altered (n-6) and (n-3) essential fatty acids in plasma and erythrocyte phospholipids in women during pregnancy and in breast milk during lactation. J. Nutr. 2008, 138, 2222–2228. [Google Scholar] [CrossRef] [PubMed]
- Koletzko, B.; Lattka, E.; Zeilinger, S.; Illig, T.; Steer, C. Genetic variants of the fatty acid desaturase gene cluster predict amounts of red blood cell docosahexaenoic and other polyunsaturated fatty acids in pregnant women: Findings from the avon longitudinal study of parents and children. Am. J. Clin. Nutr. 2011, 93, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Ghebremeskel, K.; Min, Y.; Crawford, M.A.; Nam, J.H.; Kim, A.; Koo, J.N.; Suzuki, H. Blood fatty acid composition of pregnant and nonpregnant Korean women: Red cells may act as a reservoir of arachidonic acid and docosahexaenoic acid for utilization by the developing fetus. Lipids 2000, 35, 567–574. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Li, H.-t.; Trasande, L.; Ge, H.; Yu, L.-x.; Xu, G.-s.; Bai, M.-x.; Liu, J.-m. DHA in Pregnant and Lactating Women from Coastland, Lakeland, and Inland Areas of China: Results of a DHA Evaluation in Women (DEW) Study. Nutrients 2015, 7, 8723-8732. https://doi.org/10.3390/nu7105428
Li Y, Li H-t, Trasande L, Ge H, Yu L-x, Xu G-s, Bai M-x, Liu J-m. DHA in Pregnant and Lactating Women from Coastland, Lakeland, and Inland Areas of China: Results of a DHA Evaluation in Women (DEW) Study. Nutrients. 2015; 7(10):8723-8732. https://doi.org/10.3390/nu7105428
Chicago/Turabian StyleLi, You, Hong-tian Li, Leonardo Trasande, Hua Ge, Li-xia Yu, Gao-sheng Xu, Man-xi Bai, and Jian-meng Liu. 2015. "DHA in Pregnant and Lactating Women from Coastland, Lakeland, and Inland Areas of China: Results of a DHA Evaluation in Women (DEW) Study" Nutrients 7, no. 10: 8723-8732. https://doi.org/10.3390/nu7105428
APA StyleLi, Y., Li, H.-t., Trasande, L., Ge, H., Yu, L.-x., Xu, G.-s., Bai, M.-x., & Liu, J.-m. (2015). DHA in Pregnant and Lactating Women from Coastland, Lakeland, and Inland Areas of China: Results of a DHA Evaluation in Women (DEW) Study. Nutrients, 7(10), 8723-8732. https://doi.org/10.3390/nu7105428




