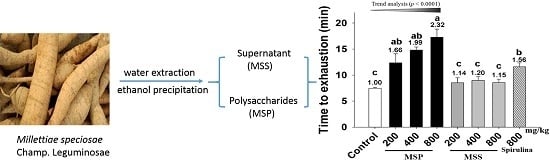
Anti-Fatigue and Antioxidant Activity of the Polysaccharides Isolated from Millettiae speciosae Champ. Leguminosae
Abstract
:1. Introduction
2. Experimental Section
2.1. Plant Material and Preparation
2.2. Determination of Total Polysaccharides in MSP and MSS
2.3. Chemicals and Reagents
2.4. Animals and Treatments
2.5. Exhaustive Swimming Exercise (ESE) Test
2.6. Mechanistic Study of the Anti-Fatigue Activity
2.7. In Vitro Antioxidant Experiment
2.7.1. ·OH Scavenging Activity
2.7.2. DPPH Scavenging Activity
2.8. Statistical Analysis
3. Results
3.1. Quantification of Total Polysaccharides
3.2. Effect of MSP and MSS on Body Weights of Mice
| Group | N | Dose(mg/kg/day) | Initial(g) | Final(g) |
|---|---|---|---|---|
| Control | 8 | 0 | 16.3 ± 1.1 | 28.7 ± 1.4 |
| positive | 8 | 800 | 16.4 ± 1.8 | 28.2 ± 0.9 |
| MSP | 8 | 200 | 16.2 ± 0.9 | 29.0 ± 1.3 |
| 8 | 400 | 16.2 ± 1.0 | 30.0 ± 1.8 | |
| 8 | 800 | 16.4 ± 1.2 | 29.5 ± 2.2 | |
| MSS | 8 | 200 | 16.3 ± 1.4 | 29.9 ± 1.4 |
| 8 | 400 | 15.8 ± 0.9 | 28.1 ± 1.7 | |
| 8 | 800 | 16.1 ± 1.1 | 30.0 ± 1.6 |
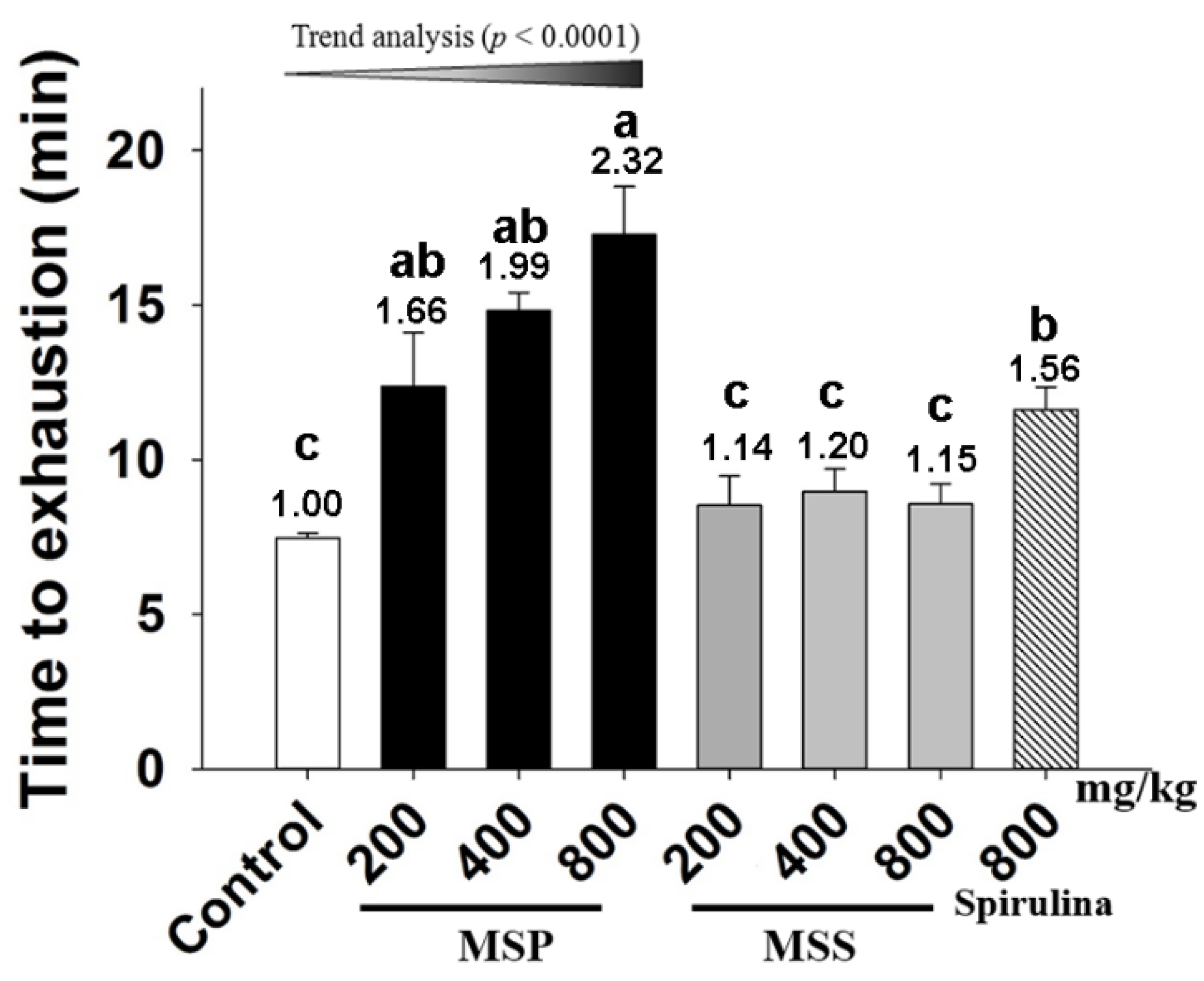
3.3. Effects of MSP and MSS on the Performance of Mice in ESE Test
3.4. Effects of MSP on Biochemical Parameters Related to Fatigue in Mice
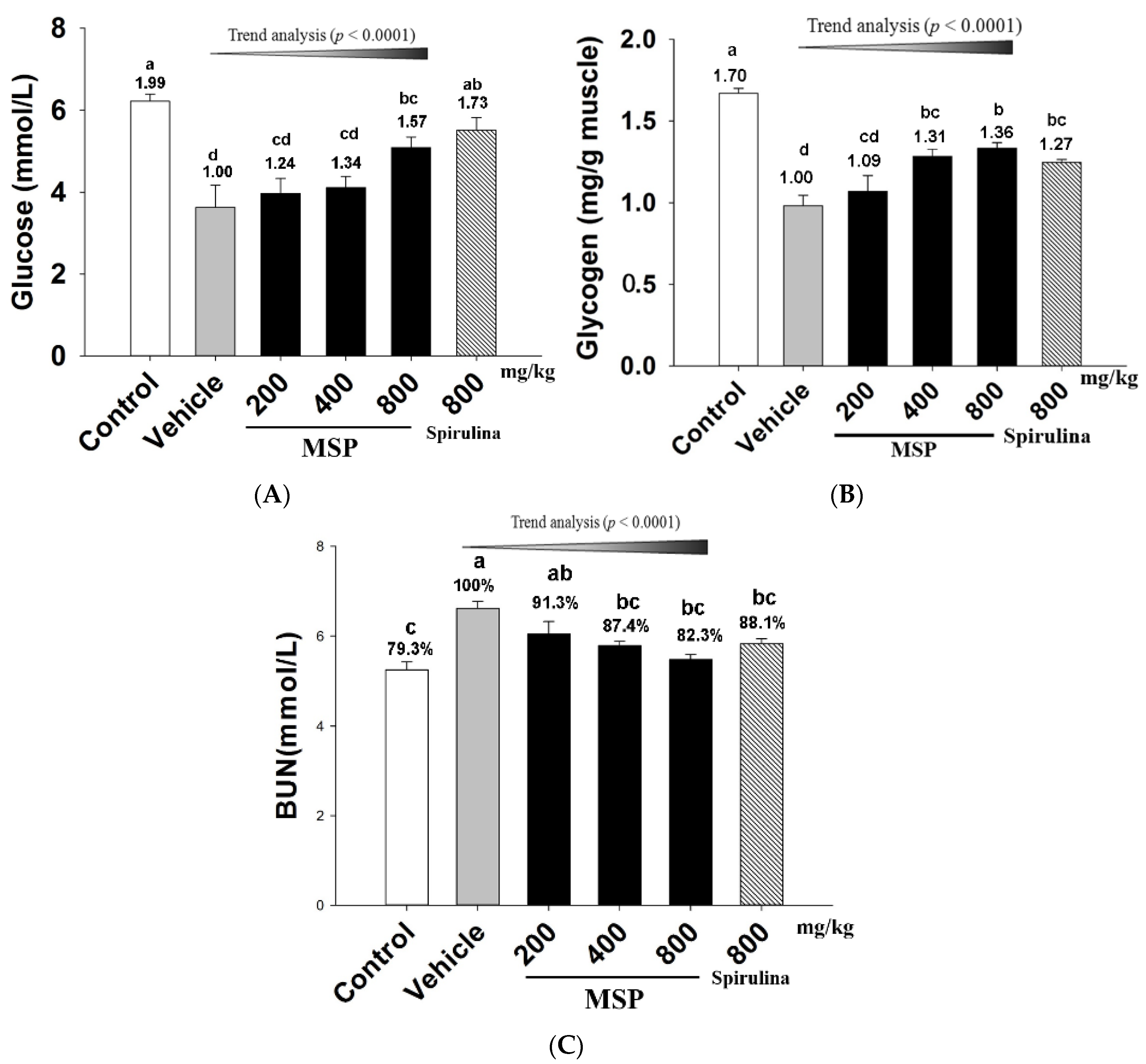
3.4.1. Effects of MSP on Energy Consumption
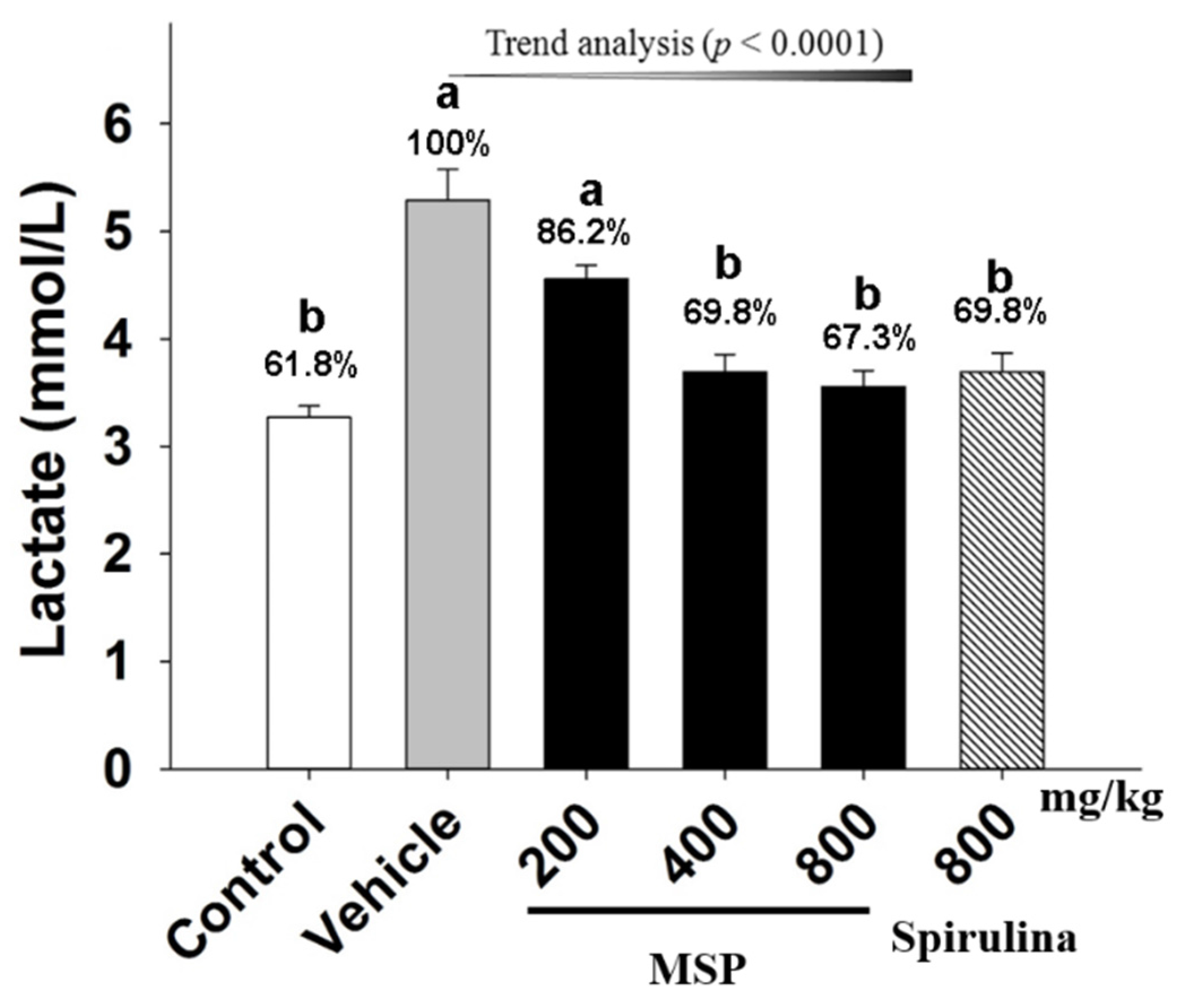
3.4.2. Effect of MSP on the Accumulation of Metabolites
3.4.3. The Protective Effects of MSP
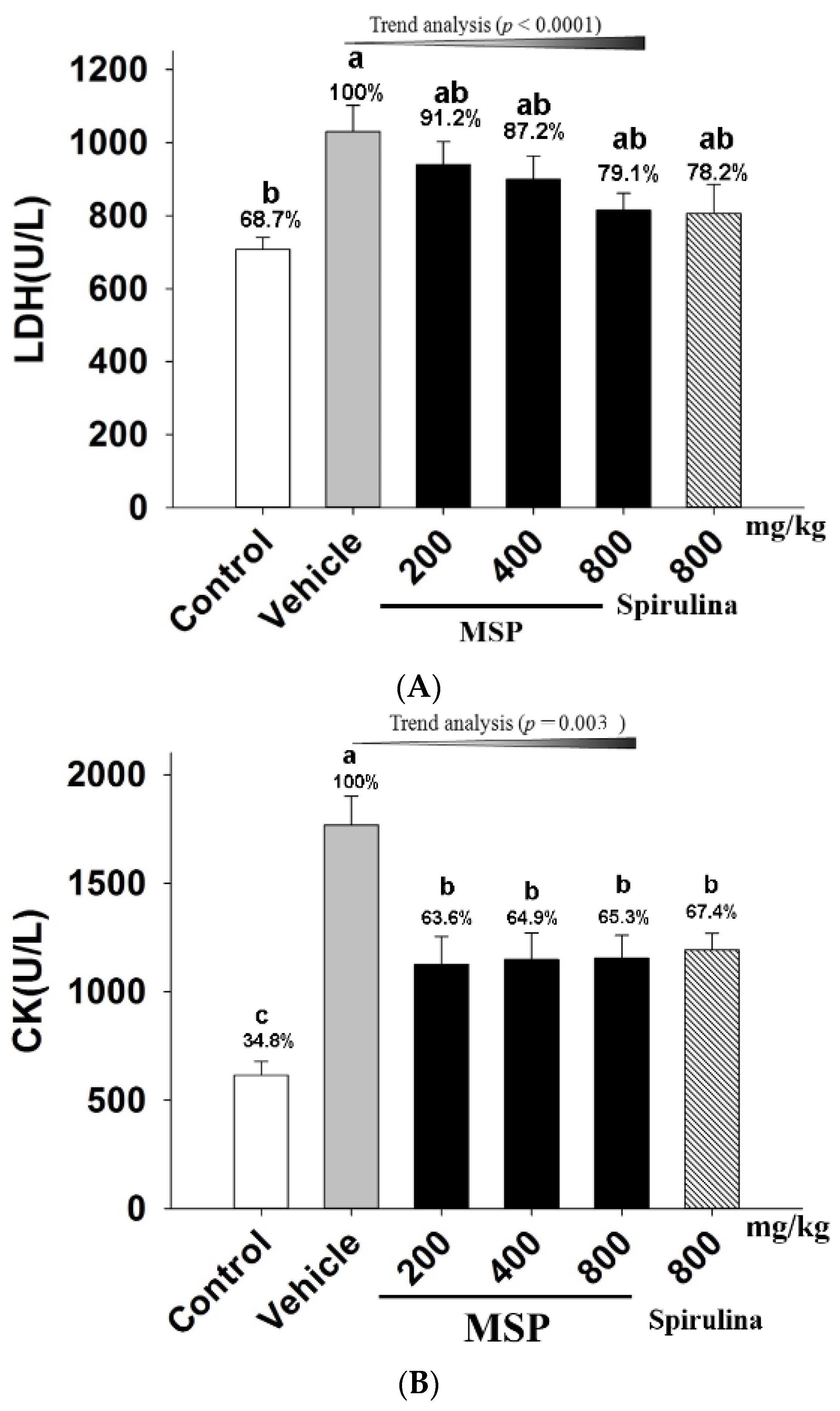
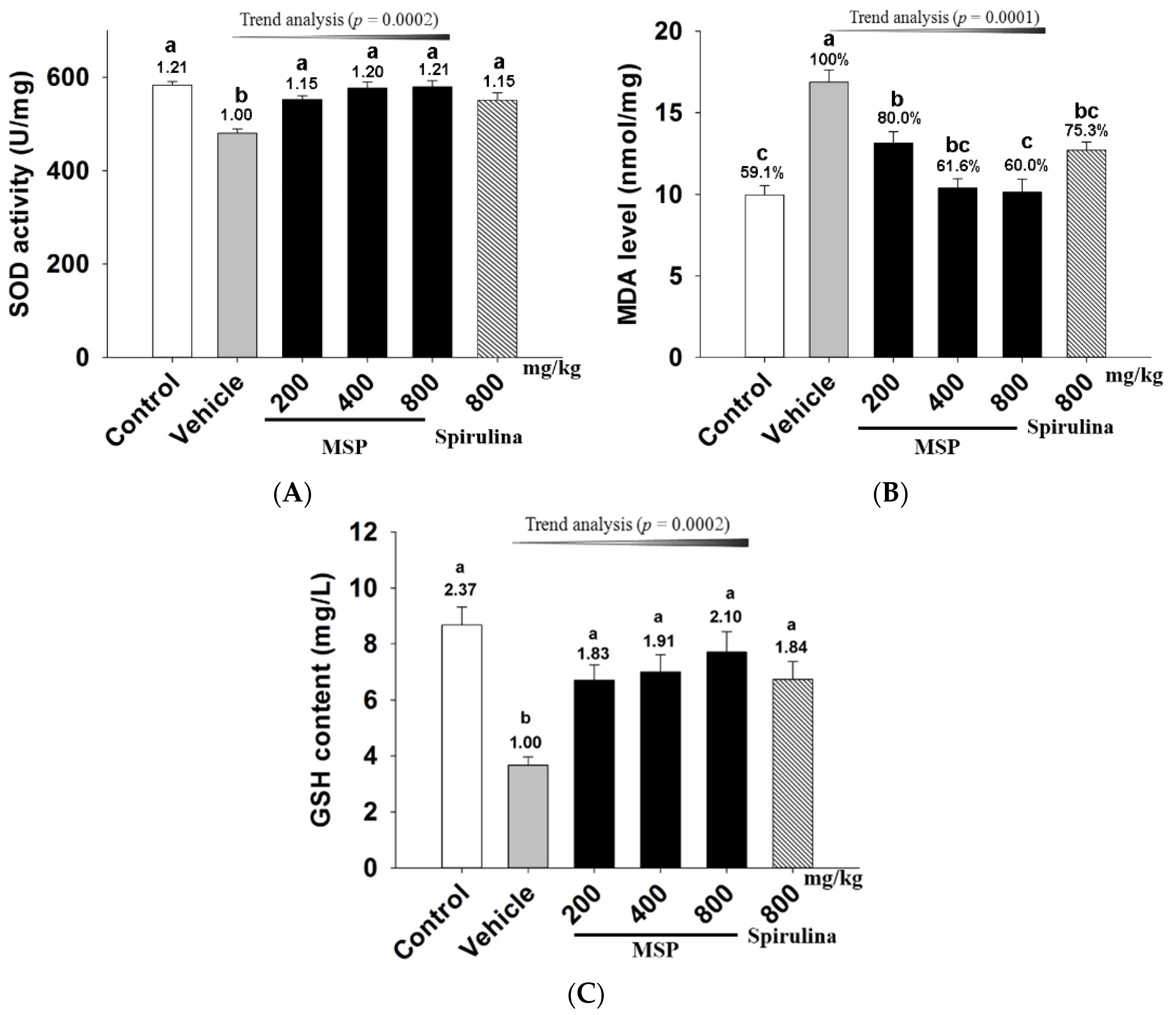
3.4.4. In Vivo Antioxidative Activity of MSP
3.5. Free Radical Scavenging Activity of MSP
3.6. Recommended Use of MSP for Human
| Group | Concentration (μg/mL) | HSR (%) | DSR (%) |
|---|---|---|---|
| Vc | 200 | 93.57 ± 1.42 | 96.71 ± 0.12 |
| MSP | 50 | 14.72 ± 0.31 | 15.64 ± 0.81 |
| 100 | 90.16 ± 1.00 | 88.43 ± 1.70 | |
| 200 | 92.71 ± 2.63 | 93.14 ± 0.42 | |
| MSS | 50 | 2.99 ± 0.57 | 2.29 ± 0.23 |
| 100 | 11.68 ± 0.43 | 10.93 ± 0.78 | |
| 200 | 13.06 ± 0.58 | 13.00 ± 0.85 |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Davis, J.M.; Bailey, S.P. Possible mechanisms of central nervous system fatigue during exercise. Med. Sci. Sports Exer. 1997, 29, 45–57. [Google Scholar] [CrossRef]
- Huang, L.Z.; Huang, B.K.; Qin, L.P. Progress on the mechanism and anti-fatigue function of traditional Chinese medicine. Drugs Clin. 2010, 25, 161–166. [Google Scholar]
- Huang, L.Z.; Huang, B.K.; Ye, Q.; Qin, L.P. Bioactivity-guided fractionation for anti-fatigue property of Acanthopanax senticosus. J. Ethnopharmacol. 2011, 133, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.d.; Zhai, F.; Shi, D.X.; Qiao, X.F.; Fu, X.L.; Li, H.P. Evaluation of antioxidant properties and anti-fatigue effect of green tea polyphenols. Sci. Res. Essays 2011, 6, 2624–2629. [Google Scholar]
- Zhang, Z.D.; Pang, X.G. Discussion on Blocking-up Hypothesis of Sports Fatigue. Sports Sci. Res. 2002, 23, 26–28. [Google Scholar]
- Li, R.R.; Chen, Z.K.; Gao, S.; Liang, W.S. Study Progress of Millettiae Speciosae. Asia-Pac. Tradit. Med. 2010, 6, 165–167. [Google Scholar]
- Zhao, X.N.; Wang, X.F.; Liao, J.B.; Guo, H.Z.; Yu, X.D.; Liang, J.L.; Zhang, X.; Su, Z.R.; Zhang, X.J.; Zeng, H.F. Antifatigue Effect of Millettiae speciosae Champ (Leguminosae) Extract in Mice. Trop. J. Pharm. Res. 2015, 14, 1319–1326. [Google Scholar] [CrossRef]
- Li, T.; Li, W. Impact of Polysaccharides from Cordyceps on Anti-fatigue in mice. Acad. J. 2009, 4, 705–709. [Google Scholar]
- Wang, J.; Li, S.; Fan, Y.; Chen, Y.; Liu, D.; Cheng, H.; Gao, X.; Zhou, Y. Anti-fatigue activity of the water-soluble polysaccharides isolated from Panax gineng C.A. Meyer. J. Ethnopharmacol. 2010, 130, 421–423. [Google Scholar] [CrossRef]
- Huang, W.C.; Lin, C.I.; Chiu, C.C.; Lin, Y.T.; Huang, C.Y.; Wen, K.C.; Chiang, H.M.; Huang, C.C.; Hsu, M.C. Hepatoprotective effects of Ixora parviflora extract against exhaustive exercise-induced oxidative stress in mice. Molecules 2013, 18, 10721–10732. [Google Scholar]
- Huang, W.C.; Lin, C.I.; Chiu, C.C.; Lin, Y.T.; Huang, W.K.; Huang, H.Y.; Huang, C.C. Chicken essence improves exercise performance and ameliorates physical fatigue. Nutrients 2014, 6, 2681–2696. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Li, C.Y.; Li, J.P.; Pan, J.; Xiang, D.X. Determination of polysaccharide in rhizome of Panax japonicus by anthrone sulfuric acid method and phenol sulfuric method. Central South Pharm. 2012, 10, 421–424. [Google Scholar]
- Institute of Laboratory Animal Research; Commission on Life Sciences; National Research Council. Guide for the Care and Use of Laboratory Animals; National Academy Press: Washington, DC, USA, 1996. [Google Scholar]
- Wang, S.Y.; Huang, W.C.; Liu, C.C.; Wang, M.F.; Ho, C.S.; Huang, W.P.; Hou, C.C.; Chuang, H.L.; Huang, C.C. Pumpkin (Cucurbita moschata) fruit extract improves physical fatigue and exercise performance in mice. Molecules 2012, 17, 11864–11876. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.C.; Chiu, W.C.; Chuang, H.L.; Tang, D.W.; Lee, Z.M.; Wei, L.; Huang, C.C. Effect of curcumin supplementation on physiological fatigue and physical performance in mice. Nutrients 2015, 7, 905–921. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.P.; Chan, S.W.; Chan, A.S.C.; Chen, S.L.; Ma, X.J.; Xu, H.X. Puerarin decreases serum total cholesterol and enhances thoracic aorta endothelial nitric oxide synthase expression in diet-induced hypercholesterolemic rats. Life Sci. 2006, 79, 324–330. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.P.; Anand, T.; Singsit, D.; Khanum, F.; Anilakumar, K.R. Evaluation of antioxidant and anti-fatigue properties of Trigonella foenum-graecum L. in rats subjected to weight loaded forced swim test. Pharmacogn. J. 2013, 5, 66–71. [Google Scholar] [CrossRef]
- Chen, W.C.; Huang, W.C.; Chiu, C.C.; Chang, Y.K.; Huang, C.C. Whey protein improves exercise performance and biochemical profiles in trained mice. Med. Sci. Sports Exerc. 2014, 46, 1517–1524. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, K.; Ishihara, K.; Tanaka, K.; Inoue, K.; Fushiki, T. An adjustable-current swimming pool for the evaluation of endurance capacity of mice. J. Appl. Physiol. 1996, 81, 1843–1849. [Google Scholar] [PubMed]
- Belluardo, N.; Westerblad, H.; Mudo, G.; Casabona, A.; Bruton, J.; Caniglia, G.; Pastoris, O.; Grassi, F.; Ibanez, C.F. Neuromuscular junction disassembly and muscle fatigue in mice lacking neurotrophin-4. Mol. Cell. Neurosci. 2001, 18, 56–67. [Google Scholar] [CrossRef] [PubMed]
- Bergstrom, J.; Hermansen, L.; Hultman, E.; Saltin, B. Diet, muscle glycogen and physical performance. Acta Physiol. Scand. 1967, 71, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.J.; Shieh, M.J.; Kuo, S.L.; Lee, C.L.; Pan, T.M. Effect of red mold rice on antifatigue and exercise-related changes in lipid peroxidation in endurance exercise. Appl. Microbiol. Biotechnol. 2006, 70, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.A.; Stannard, S.R.; Thompson, M.W. Muscle triglyceride and glycogen in endurance exercise: Implications for performance. Sports Med. 2004, 34, 151–164. [Google Scholar] [CrossRef] [PubMed]
- Jia, J.M.; Wu, C.F. Antifatigue activity of tissue culture extracts of Saussurea involucrata. Pharm. Biol. 2008, 46, 433–436. [Google Scholar] [CrossRef]
- Chen, Y.M.; Tsai, Y.H.; Tsai, T.Y.; Chiu, Y.S.; Wei, L.; Chen, W.C.; Huang, C.C. Fucoidan supplementation improves exercise performance and exhibits anti-fatigue action in mice. Nutrients 2014, 7, 239–252. [Google Scholar] [CrossRef] [PubMed]
- Murase, T.; Haramizu, S.; Shimotoyodome, A.; Tokimitsu, I.; Hase, T. Green tea extract improves running endurance in mice by stimulating lipid utilization during exercise. Am. J. Physiol.Regul. Integr. 2006, 290, 1550–1556. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.F.; Li, Y.Y.; Xu, J.J.; Su, X.R.; Gao, X.; Yue, F.P. Study on effect of jellyfish collagen hydrolysate on anti-fatigue and anti-oxidation. Food Hydrocoll. 2011, 25, 1350–1353. [Google Scholar] [CrossRef]
- Brancaccio, P.; Maffulli, N.; Limongelli, F.M. Creatine kinase monitoring in sport medicine. Br. Med. Bull. 2007, 81, 209–230. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.M.; Wei, P. Anti-fatigue properties of tartary buckwheat extracts in mice. Int. J. Mol. Sci. 2011, 12, 4770–4780. [Google Scholar] [CrossRef]
- Anand, T.; Kumar, G.P.; Pandareesh, M.D.; Swamy, M.S.; Khanum, F.; Bawa, A.S. Effect of bacoside extract from Bacopa monniera on physical fatigue induced by forced swimming. Phytother. Res. 2012, 26, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Warren, G.L.; Ingalls, C.P.; Lowe, D.A.; Armstrong, R.B. Excitation-contraction uncoupling: Major role in contraction-induced muscle injury. Exerc. Sports Sci. Rev. 2001, 29, 82–87. [Google Scholar] [CrossRef]
- Lee, S.H.; Kim, I.B.; Kim, J.B.; Parkc, D.H.; Min, K.J. The effects of Korean mistletoe extract on endurance during exercise in mice. Anim. Cells Syst. 2014, 18, 34–40. [Google Scholar] [CrossRef] [Green Version]
- Logan, A.C.; Wong, C. Chronic fatigue syndrome: oxidative stress and dietary modifications. Altern. Med. Rev. 2001, 6, 450–459. [Google Scholar] [PubMed]
- Liu, L.P. The relation of the change in SOD, MDA and mitochondrial membrane potential to apoptosis in hepatocyte of rats after swimming training. Chin. J. Sports Med. 2002, 21, 10–15. [Google Scholar]
- Mestre, A.A.; Ferrer, M.D.; Sureda, A.; Tauler, P.; Martínez, E.; Bibiloni, M.M.; Micol, V.; Tur, J.A.; Pons, A. Phytoestrogens enhance antioxidant enzymes after swimming exercise andmodulate sex hormone plasma levels in female swimmers. Eur. J. Appl. Physiol. 2011, 111, 2281–2294. [Google Scholar] [CrossRef] [PubMed]
- Dringen, R. Metabolism and functions of glutathione in brain. Prog. Neuro-Psychoph. 2000, 62, 649–671. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, X.-N.; Liang, J.-L.; Chen, H.-B.; Liang, Y.-E.; Guo, H.-Z.; Su, Z.-R.; Li, Y.-C.; Zeng, H.-F.; Zhang, X.-J. Anti-Fatigue and Antioxidant Activity of the Polysaccharides Isolated from Millettiae speciosae Champ. Leguminosae. Nutrients 2015, 7, 8657-8669. https://doi.org/10.3390/nu7105422
Zhao X-N, Liang J-L, Chen H-B, Liang Y-E, Guo H-Z, Su Z-R, Li Y-C, Zeng H-F, Zhang X-J. Anti-Fatigue and Antioxidant Activity of the Polysaccharides Isolated from Millettiae speciosae Champ. Leguminosae. Nutrients. 2015; 7(10):8657-8669. https://doi.org/10.3390/nu7105422
Chicago/Turabian StyleZhao, Xiao-Ning, Jia-Li Liang, Han-Bin Chen, Ye-Er Liang, Hui-Zhen Guo, Ze-Ren Su, Yu-Cui Li, Hui-Fang Zeng, and Xiao-Jun Zhang. 2015. "Anti-Fatigue and Antioxidant Activity of the Polysaccharides Isolated from Millettiae speciosae Champ. Leguminosae" Nutrients 7, no. 10: 8657-8669. https://doi.org/10.3390/nu7105422
APA StyleZhao, X.-N., Liang, J.-L., Chen, H.-B., Liang, Y.-E., Guo, H.-Z., Su, Z.-R., Li, Y.-C., Zeng, H.-F., & Zhang, X.-J. (2015). Anti-Fatigue and Antioxidant Activity of the Polysaccharides Isolated from Millettiae speciosae Champ. Leguminosae. Nutrients, 7(10), 8657-8669. https://doi.org/10.3390/nu7105422