Casein-Derived Lactotripeptides Reduce Systolic and Diastolic Blood Pressure in a Meta-Analysis of Randomised Clinical Trials
Abstract
:1. Introduction
2. Experimental Section
2.1. Protocol
2.2. Eligibility Criteria for Meta-Analysis
2.3. Information Sources and Search Strategy
2.4. Study Selection, Data Collection Process and Data Items
2.5. Risk of Bias in Individual Studies
2.6. Summary Measures and Synthesis of Results
3. Results
3.1. Study Selection
3.2. Study Characteristics
3.3. Summary Results of the Effect of LTP Intervention
| Reference | Subject Group, n (M/F), Average Age and BMI | Study Design and Duration | Country | Treatment | Tripeptide (mg/day) | Baseline | BP Measure-Ment 3 | ||
|---|---|---|---|---|---|---|---|---|---|
| IPP | VPP | SBP (mmHg) 2 | DBP (mmHg) 2 | ||||||
| Hata et al. 1996 [55] | hypertensive, 30 (8/22), (26 on BP ↓ medicine), 74.8 years, 20.5 kg/m2 | R, NR, C, PAL; 8 weeks | Japan | L. helveticus and S. cerevisiae; 1 × 100 mL milk drink | 1.10 | 1.50 | T: 158.5 ± 45.8 C: 150.9 ± 34.2 | 88.7 ± 38.8 87.0 ± 32.8 | Office |
| Kajimoto et al. 2001 [56] | normotensive, 43 (20/23), 29.7 years, 21.5 kg/m2 | R, D, C, PAL; 2 weeks | Japan | L. helveticus CM4; 1 × 6 tablets | 4.50 | 8.10 | T: 113.6 ± 12.0 C: 114.1 ± 10.5 | 67.3 ± 8.7 68.1 ± 7.6 | Office |
| Kajimoto et al. 2002 [57] | mild hypertensive, 64 (33/31), 50 years, 25.1 kg/m2 | R, D, C, PAL; 8 weeks | Japan | L. helveticus and S. cerevisiae, L. helveticus CM4, L. delbrueckii bulgaricus, S. thermophilus 2 × 150 mL yoghurt drink | 1.58 | 2.24 | T: 148 ± 9 C: 148 ± 9 | 94 ± 7 95 ± 7 | Office |
| Seppo et al. 2002 [68] | mild hypertensive, 17 (5/12), 47.5 years, 27.2 kg/m2 | R, D, C, PAL; 8 weeks | Finland | L. helveticus LBK-16 H; 1 × 150 mL milk drink | 2.25 | 3–3.75 | T: 148 ± 12.6 C: 148 ± 13.3 | 94 ± 6.3 93 ± 2.7 | Office |
| Seppo et al. 2003 [66] | hypertensive, 39 (19/20), (16 on BP ↓ medicine), 49.4 years, NR | R, D, C, PAL; 21 weeks | Finland | L. helveticus LBK-16 H; 1 × 150 mL milk drink | 2.25 | 3.00 | T: 152 ± 12.7 C: 149 ± 11.1 | 96 ± 5.2 95 ± 5.8 | Home |
| Nakamura et al. 2004 [59] | high-normotensive, 106 (34/72), 38.5 years, 22.1 kg/m2 | R, D, C, PAL; 12 weeks | Japan | L. helveticus and S. cerevisiae, L. helveticus CM4, L. delbrueckii bulgaricus, S. thermophilus 2 × 150 mL yoghurt drink | 1.48 | 2.26 | T: 134 ± 5 C: 135 ± 4 | 79 ± 5 78 ± 5 | Office |
| Mizushima et al. 2004 [60] | hypertensive, 46 M, 46.5 years, 25 kg/m2 | R, D, C, PAL; 4 weeks | Japan | L. helveticus and S. cerevisiae; 1 × 160 mL milk drink | 1.15 | 1.98 | T: 147.6 ± 9.6 C: 145.3 ± 13.0 | 95.3 ± 9.9 91.5 ± 9.6 | Office |
| Toulomilehto et al. 2004 [52] | mild hypertensive, 59 (36/23), 52.8 years, 28.6 kg/m2 | R, D, C, CO; 13–17 weeks | Finland | L. helveticus LBK-16 H; 1 × 150 mL milk drink | 2.4–2.7 | 2.4–2.7 | T: 152.7 ± 10.0 C: 156.6 ± 11.4 | 98.1 ± 7.2 98.1 ± 7.0 | Office |
| Aihara et al. 2005 [61] | high-normotensive, 40 (26/14), 51.4 years, 24.2 kg/m2 | R, D, C, PAL; 4 weeks | Japan | L. helveticus CM4; 1 × 6 tablets | 4.70 | 8.30 | T: 137.4 ± 5.1 C: 136.8 ± 6.1 | 85.1 ± 4.9 84.8 ± 11.0 | Office |
| Jauhiainen et al. 2005 [67] | hypertensive, 108 (69/39), 53 years, 28.5 kg/m2 | R, D, C, PAL; 10 weeks | Finland | L. helveticus LBK-16 H; 2 × 150 mL milk drink | IPP | VPP | SBP (mmHg)2 | DBP (mmHg)2 | Office |
| Mizuno et al. 2005 4 [48] | high-normotensive, 24 (6/18), 42.8 years, 25 kg/m2 | R, S, C, PAL; 6 weeks | Japan | A. oryzae hydrolysate; 1 × 2 tablets | 1.76 | 1.86 | T: 133.9 ± 6.7 C: 132.8 ± 3.4 | 81.2 ± 4.7 80.2 ± 2.7 | Office |
| Sano et al. 2005 [62] | hypertensive, 144 (57/87), 50.5 years, 24 kg/m2 | R, D, C, PAL; 12 weeks | Japan | A. oryzae hydrolysate; 1 × 200 mL vegetable-fruit juice | 1.60 | 1.47 | T: 138.2 ± 6.5 C: 138.5 ± 6.7 | 84.4 ± 5.3 85.2 ± 5.7 | Office |
| Sano et al. 2005 [63] | normotensive and hypertensive, 43 (21/22), 46.9 years, 23.6 kg/m2 | R, D, C, PAL; 4 weeks | Japan | A. oryzae hydrolysate; 3 × 200 mL vegetable-fruit juice | 4.80 | 4.41 | T: 132.3 ± 17.0 C: 133.1 ± 15.3 | 80.6 ± 9.2 82.3 ± 9.5 | Office |
| Ishida et al. 2006 [65] | normotensive and hypertensive, 54 (16/38), 51.9 years, 24.9 kg/m2 | R, D, C, PAL; 4 weeks | Japan | A. oryzae hydrolysate; 1 × 20 tablets | 10.10 | 5.65 | T: 131.4 ± 15.2 C: 133.4 ± 15.9 | 81.9 ± 10.8 83.7 ± 10.9 | Office |
| Engberink et al. 2008 [69] | hypertensive, 35 (23/12), 58.8 years, 26.9 kg/m2 | R, D, C, PAL; 8 weeks | the Netherlands | fermentation; 1 × 200 mL low-fat yoghurt drink | 4.20 | 5.80 | 142.0 ± 11.3 | 83.2 ± 8.3 | Office |
| van der Zander et al. 2008 [70] | mild hypertensive, 275 (153/122), 60 years, 26.7 kg/m2 | R, D, C, PAL; 8 weeks | the Netherlands | A. oryzae hydrolysate; 1 × 200 mL yoghurt drink | 5.78 | 4.40 | 150 ± 12.8 150 ± 12.9 | 85.2 ± 9.2 86.2 ± 9.3 | Office |
| van Mierlo et al. 2009 5 [46] | mild hypertensive, 69 (45/24), 61.7 years, 26.9 kg/m2 | R, D, C, CO; 8 weeks | Scotland | A. oryzae hydrolysate; 1 × 200 mL yoghurt drink | 5.78 | 4.40 | 147.4 ± 9.0 | 89.0 ± 5.9 | Office |
| de Leeuw et al. 2009 2 [49] | mild hypertensive, 41 (21/20), 59.5 years, 25 kg/m2 | R, D, C, PAL; 8 weeks | the Netherlands | A. oryzae hydrolysate; 1 × 200 mL yoghurt drink | 4.56 | 4.47 | 149 ± 15 148 ± 13 | 85 ± 8 85 ± 9 | Office |
| Yoshizawa et al. 2009 6 [43] | normotensive, 28 F, 58 years, 21.7 kg/m2 | R, S, C, PAL; 8 weeks | Japan | enzymatic hydrolysis; 1 × 8 tablets | 4.30 | 2.40 | 116 ± 20.8 110 ± 19.0 | 68 ± 10.4 66 ± 12.6 | Office |
| Yoshizawa et al. 2010 6 [44] | normotensive, 22 F, 57.5 years, 22 kg/m2 | R, S, C, PAL; 8 weeks | Japan | enzymatic hydrolysis; 1 × 8 capsules | 4.30 | 2.40 | 112 ± 23.4 104 ± 14.4 | 76 ± 11.7 72 ± 10.8 | Office |
| Cicero et al. 2010 [53] | normo- and high-normotensive, 55 (30/25), 40.3 years, 25 kg/m2 | R, D, C, CO; 4 weeks | Italy | enzymatic hydrolysis; 2 × 250 mL vegetable and fruit juice | 2.00 | 4.00 | 126.0 ± 15.5 | 80.3 ± 8.7 | Office |
| Jauhiainen et al. 2010 4 [50] | hypertensive, 89 (54/35), 49 years, 28.1 kg/m2 | R, D, C, PAL; 12 weeks | Finland | L. helveticus LBK-16 H; 2 × 200 mL fermented milk | 11.60 | 13.20 | 151.3 ± 14.8 154.6 ± 13.9 | 95.2 ± 12.2 94.2 ± 8.8 | Office |
| Boelsma, Kloek 2010 7 [ 45] | stage 1 hypertensive, 26 (17/9), 59 years, 26.5 kg/m2 | R, D, C, CO; 4 weeks | the Netherlands | enzymatic hydrolysis; 2 ×1 capsules | 15.00 | - | 148.7 ± 11.5 | 89.2 ± 10.1 | Office |
| Usinger et al. 2010 4 [47] | mild hypertensive, 60 (28/32), 54 years, 26.5 kg/m2 | R, D, C, PAL; 8 weeks | Denmark | fermentation; 1 × 300 mL fermented milk | 2.50 | 1.10 | 145 ± 10.9 136 ± 8.8 | 92.6 ± 6.4 89.4 ± 7.6 | Office |
| Germino et al. 2010 [72] | hypertensive, 81 (45/36), 57.1 years, 29.5 kg/m2 | R, D, C, PAL; 6 weeks | USA | 2 × 1 (75 mg) powder dissolved in apple juice | NR | NR | 156.6 ± 6 154.6 ± 6 | 92.5 ± 8.3 91.6 ± 9.6 | Office |
| Ishida et al. 2011 [64] | normo- and mild hypertensive, 48 (24/24), 49.3 years, 22.4 kg/m2 | R, D, C, PAL; 4 weeks | Japan | A. oryzae hydrolysate; 1 × 20 tablets | 9.60 | 7.50 | 130.6 ± 16.3 131.8 ± 14.8 | 80.4 ± 10.6 81.1 ± 9.6 | Office |
| Nakamura et al. 2011 [58] | hypertensive, 70 (47/23), 57.8 years, 23.9 kg/m2 | R, D, C, PAL; 8 weeks | Japan | A. oryzae hydrolysate; 1 × 4 tablets | 1.90 | 1.50 | 146.8 ± 4.4 146.9 ± 4.3 | 87.5 ± 7.1 88.0 ± 7.7 | Office |
| Cicero et al. 2011 [71] | high-normotensive and mild hypertensive, 50 (29/21), 51.2 years, 26.8 kg/m2 | R, D, C, PAL; 6 weeks | Italy | enzymatic hydrolysis; 1 × 250 mL fruit juice | 1.00 | 2.00 | 142.0 ± 10.9 141.2 ± 9.6 | 86.4 ± 8.2 86.6 ± 7.7 | ABPM |
| Cicero 2012 [54] | high-normotensive and mild hypertensive, 164 (101/63), 43.9 years, 25.7 kg/m2 | R, D, C, CO; 4 weeks | Italy | enzymatic hydrolysis; 1 × 250 mL fruit juice | 1.00 | 2.00 | T: 133.5 ± 12.9 C: 132.7 ± 12.5 | 83.3 ± 8.8 82.8 ± 8.3 | Office |
| Jauhiainen 2012 [51] | mild hypertensive, 89 (54/35), 49 years, 28.1 kg/m2 | R, D, C, PAL; 12 weeks | Finland | L. helveticus LBK-16 H; 1 × 200 mL fermented milk | 2.40 | 2.60 | T: 148 ± 7 C: 147 ± 5 | 95 ± 5 94 ± 4 | ABPM |
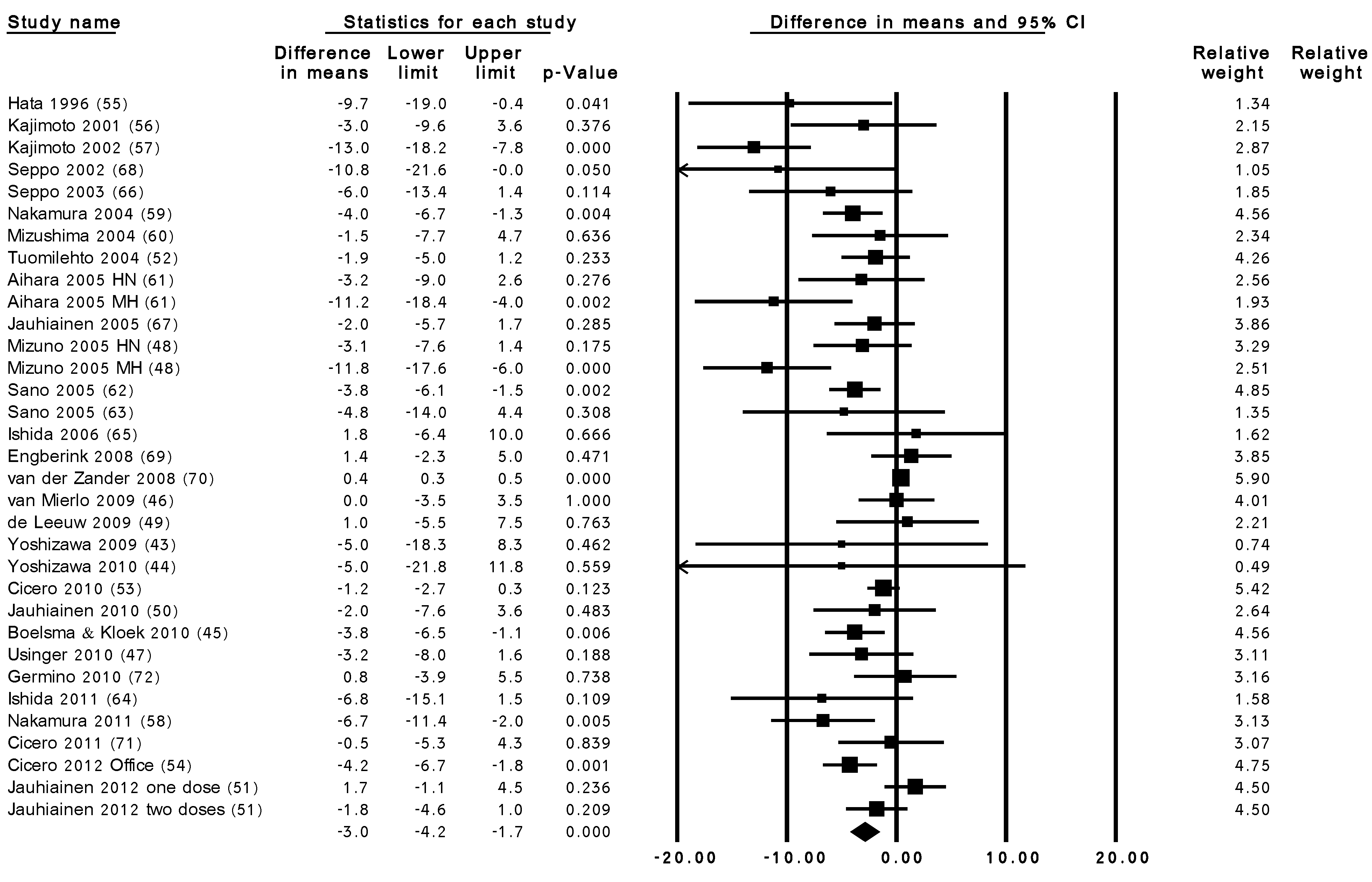
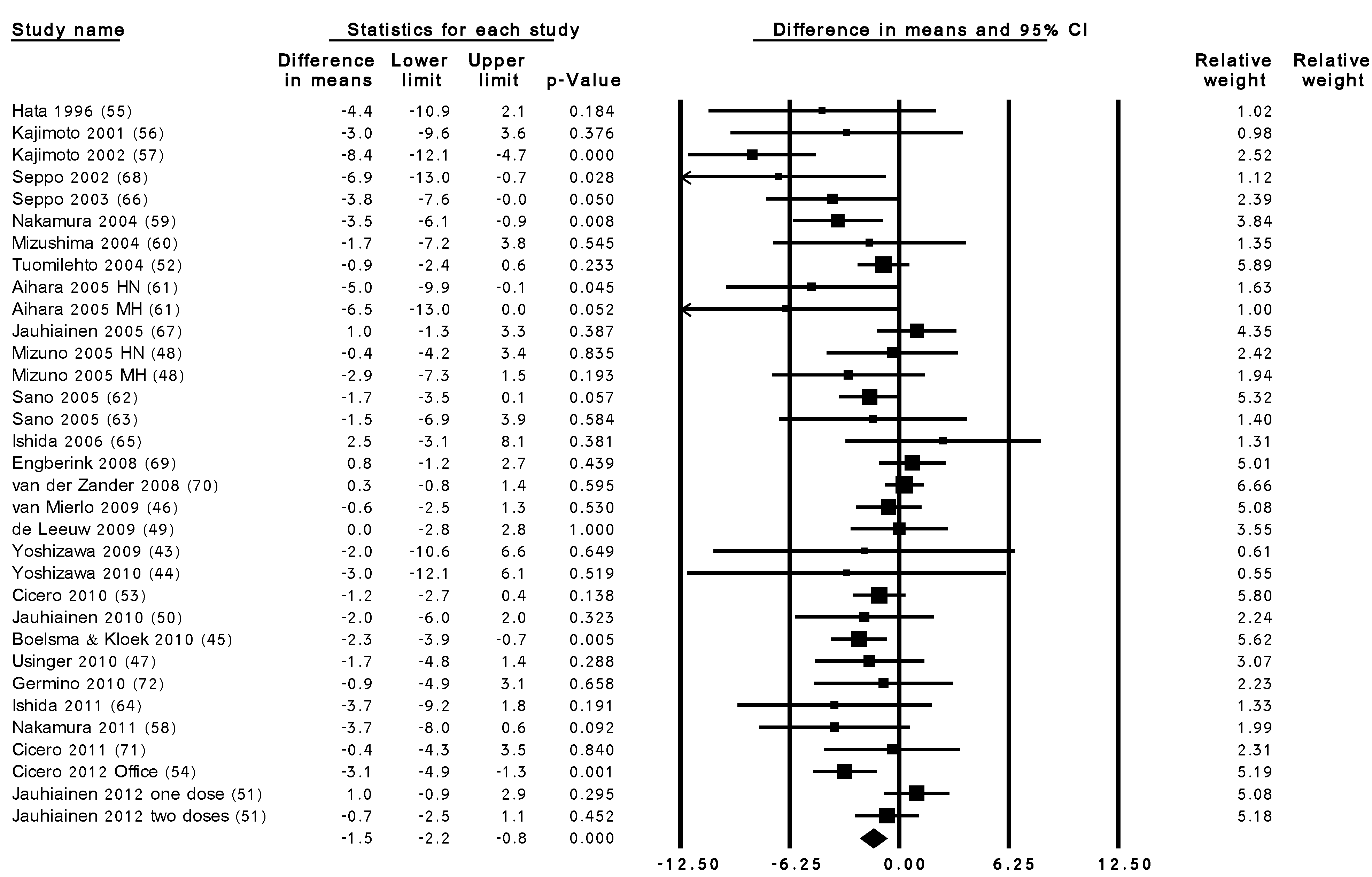
| Sub-group Title | No. of Trials | Effect Size (mmHg) (95% CI) | |
|---|---|---|---|
| SBP | DBP | ||
| Baseline BP | |||
| above 140/90 mmHg | 20/12 | −3.00 (−4.71, −1.29) | −1.90 (−3.38, −0.42) |
| below 140/90 mmHg | 13/21 | −2.68 (−3.61, −1.74) | −1.38 (−2.13, −0.63) |
| p-value * | 0.748 | 0.541 | |
| Duration of intervention | |||
| ≤8 weeks | 25 | −3.35 (−4.91, −1.79) | −1.80 (−2.72, −0.89) |
| >8 weeks | 8 | −2.18 (−3.72, −0.64) | −1.00 (−2.10, −0.09) |
| p-value * | 0.966 | 0.921 | |
| Dose of LTP | |||
| ≤10 mg/day | 21 | −3.81 (−5.30, −2.32) | −2.02 (−2.87, −1.16) |
| >10 mg/day | 11 | −1.32 (−2.94, 0.30) | −0.41 (−1.38, 0.57) |
| p-value * | 0.027 | 0.015 | |
| Production of LTP | |||
| Enzymatic hydrolysis | 14 | −2.77 (−4.48, −1.05) | −1.25 (−2.03, −0.47) |
| Fermentation | 15 | −3.82 (−5.80, −1.84) | −2.38 (−3.77, −1.00) |
| p-value * | 0.433 | 0.164 | |
| Country of studies | |||
| Japan | 16 | −5.54 (−7.43, −3.65) | −3.01 (−4.25, −1.78) |
| European countries | 16 | −1.36 (−2.53, −0.20) | −0.83 (−1.57, −0.10) |
| p-value * | 0.002 | <0.001 | |
| Trial size | |||
| <50 participants | 15 | −4.93 (−6.78, −3.07) | −2.40 (−3.42, −1.39) |
| ≥50 participants | 18 | −1.99 (−3.31, −0.66) | −1.11 (−1.96, −0.26) |
| p-value * | 0.011 | 0.057 | |
3.4. Risk of Bias within and across Studies
4. Discussion
5. Strengths of the Meta-Analysis
6. Limitation of the Meta-Analysis
7. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- World Health Organisation (WHO). Cardiovascular Diseases (CVDs) Fact Sheet No 317 Internet. Available online: http://www.who.int/mediacentre/factsheets/fs317/en/index.html (accessed on 21 January 2012).
- World Health Organisation (WHO). Global Health Risk: Mortality and Burden of Disease Attributable to Selected Major Risks. Available online: http://www.who.int/healthinfo/global_burden_disease/GlobalHealthRisks_report_full.pdf (accessed on 21 January 2012).
- Appel, L.; Brands, M.; Daniels, S.; Karanja, N.; Elmer, P.; Sacks, F. Dietary approaches to prevent and treat hypertension: A scientific statement from the American Heart Association. Hypertension 2006, 47, 296–308. [Google Scholar] [CrossRef] [PubMed]
- Whitworth, J. World Health Organization (WHO)/International Society of Hypertension (ISH) statement on management of hypertension. J. Hypertens. 2003, 21, 1983–1992. [Google Scholar] [CrossRef] [PubMed]
- Korhonen, H.; Pihlanto, A. Bioactive peptides: Production and functionality. Int. Dairy J. 2006, 16, 945–960. [Google Scholar] [CrossRef]
- Xu, J.-Y.; Qin, L.-Q.; Wang, P.-Y.; Li, W.; Chang, C. Effect of milk tripeptides on blood pressure: A meta-analysis of randomised controlled trials. Nutrition 2008, 24, 933–940. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.; Gerocarni, B.; Laghi, L.; Borghi, C. Blood pressure lowering effect of lactotripeptides assumed as functional foods: A meta-analysis of current available clinical trials. J. Hum. Hypertens. 2011, 25, 425–436. [Google Scholar] [CrossRef] [PubMed]
- Turpeinen, A.M.; Jarvenpaa, S.; Kautiainen, H.; Korpela, R.; Vapaatalo, H. Antihypertensive effects of bioactive tripeptides—Arandom effects meta-analysis. Ann. Med. 2013, 45, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.-Q.; Xu, J.-Y.; Dong, J.-Y.; Zhao, Y.; van Bladeren, P.; Zhang, W. Lactotripeptides intake and blood pressure management: A meta-analysis of randomised controlled trials. Nutr. Metab. Cardiovas. 2013, 23, 395–402. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.; Ioannidis, J.; Clarke, M.; Devereaux, P.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. BMJ 2009, 151, 65–94. [Google Scholar]
- The Cochrane Collaboration. Cochrane Handbook for Systematic Reviews of Interventions; Higgins, J., Green, S., Eds.; Version 5.1.0. Wiley: Hoboken, NJ, USA, 2011. Available online: http://www.cochrane-handbook.org (accessed on 21 January 2012).
- Follmann, D.; Elliott, P.; Suh, I.; Cutler, J. Variance imputation for overviews of clinical trials with continuous response. J. Clin. Epidemiol. 1992, 45, 769–773. [Google Scholar] [CrossRef] [PubMed]
- Borenstein, M.; Hedges, L.; Higgins, J.; Rothstein, H. Comprehensive Meta-analysis; Version 2; Biostat: Englewood, NJ, USA, 2005. [Google Scholar]
- Borenstein, M.; Hedges, L.; Higgins, J.; Rothstein, H. Subgroup analyses. In Introduction to Meta-Analysis, 1st ed.; John Wiley & Sons, Ltd.: Croydon, UK, 2009; pp. 149–186. [Google Scholar]
- Kawase, M.; Hashimoto, H.; Hosoda, M.; Morita, H.; Hosono, A. Effect of administration of fermented milk containing whey protein concentrate to rats and healthy men on serum lipids and blood pressure. J. Dairy Sci. 2000, 83, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Pins, J.; Keenan, J. Effects of whey peptides on cardiovascular disease risk factors. J. Clin. Hypertens. 2006, 8, 775–782. [Google Scholar] [CrossRef]
- Lee, Y.; Skurk, T.; Hennig, M.; Hauner, H. Effect of a milk drink supplemented with whey peptides on blood pressure in patients with mild hypertension. Eur. J. Nutr. 2007, 46, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Ballard, K.; Bruno, R.; Seip, R.; Quann, E.; Volk, B.; Freidenreich, D.; Kawiecki, D.; Kupchak, B.; Chung, M.; Kraemer, W.; et al. Acute ingestion of a novel whey-derived peptide improves vascular endothelial responses in healthy individuals: A randomised, placebo controlled trial. Nutr. J. 2009, 8. [Google Scholar] [CrossRef] [PubMed]
- Fluegel, S.; Shultz, T.; Powers, J.; Stephanie, C.; Barbosa-Leiker, C.; Wright, B.; Freson, T.; Fluegel, H.; Minch, J.; Schwarzkopf, L. Whey beverages decrease blood pressure in prehypertensive and hypertensive young men and women. Int. Dairy J. 2010, 20, 753–760. [Google Scholar] [CrossRef]
- Pal, S.; Ellis, V. Acute effects of whey protein isolate on blood pressure, vascular function and inflammatory markers in overweight postmenopausal women. Br. J. Nutr. 2011, 105, 1512–1519. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Ellis, V. The chronic effects of whey proteins on blood pressure, vascular function, and inflammatory markers in overweight individuals. Obesity 2010, 18, 1354–1359. [Google Scholar] [CrossRef] [PubMed]
- Cadee, J.; Chang, C.; Chen, C.; Huang, C.; Chen, S.; Wang, C. Bovine casein hydrolysate (c12 Peptide) reduces blood pressure in prehypertensive subjects. Am. J. Hypertens. 2007, 20, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Townsend, R.; McFadden, C.; Ford, V.; Cadee, J. A randomzed, double-blind, placebo-controlled trial of casein protein hydrolysate (c12 peptide) in human essential hypertension. Am. J. Hypertens. 2004, 17, 1056–1058. [Google Scholar] [CrossRef] [PubMed]
- Sekiya, S.; Kobayashi, Y.; Kita, E.; Imamura, Y.; Toyama, S. Antihypertensive effects of tryptic hydolysate of casein on normotensive and hypertensive volunteers. J. Jpn. Soc. Nutr. Food Sci. 1992, 45, 513–551. [Google Scholar] [CrossRef]
- Sugai, R. ACE inhibitors and functional foods. Bull. IDF 1998, 336, 17–20. [Google Scholar]
- Ashar, M.; Chand, R. Fermented milk containing ACE-inhibitory peptides reduces blood pressure in middle ages hypertensive subjects. Milchwissenschaft 2004, 59, 363–366. [Google Scholar]
- Kajimoto, O.; Aihara, K.; Hirata, H.; Takahashi, R.; Nakamura, Y. Hypotensive effects of the tablets containing “Lactotripeptides (VPP, IPP)”. J. Nutr. Food 2001, 4, 51–61. [Google Scholar]
- Kajimoto, O.; Nakamura, Y.; Yada, H.; Moriguchi, S.; Hirata, H.; Takahashi, T. Hypotensive effect of sour milk in subjects with mild or moderate hypertension. J. Jpn. Soc. Nutr. Food Sci. 2001, 54, 347–354. [Google Scholar] [CrossRef]
- Hirata, H.; Nakamura, Y.; Yada, H.; Moriguchi, S.; Kajimoto, O.; Takahashi, T. Clinical effect of new sour milk drink on mild or moderate hypertensive subjects. J. New Rem Clin. 2002, 51, 61–69. [Google Scholar]
- Van der Zander, K.; Jakel, M.; Bianco, V.; Koning, M. Fermented lactotripeptides-containing milk lowers daytime blood pressure in high normal-to-mild hypertensive subjects. J. Hum. Hypertens. 2008, 22, 804–806. [Google Scholar] [CrossRef] [PubMed]
- Turpeinen, A.; Ehlers, P.; Kivimaki, A.; Jarvenpaa, S.; Filler, I.; Wiegert, E.; Janchen, E.; Vapaatalo, H.; Korpela, R.; Wagner, F. Ile-Pro-Pro and Val-Pro-Pro tripeptide-containing milk product has acute blood pressure lowering effects in mildly hypertensive subjects. Clin. Exp. Hypertens. 2011, 33, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Mizutani, J.; Sasaki, K.; Yamamoto, N.; Takazawa, N. Beneficial potential of casein hydrolysate containing Val-Pro-Pro and Ile-Pro-Pro on central blood pressure and hemodynamic Index: A preliminary Study. J. Med. Food 2009, 6, 1–6. [Google Scholar]
- Kurosawa, M.; Nakamura, Y.; Yamamoto, N.; Yamada, K.; Iketani, T. Effects of Val-Pro-Pro and Ile-Pro-Pro on nondipper patients: A preliminary study. J. Med. Food 2011, 14, 538–542. [Google Scholar] [CrossRef] [PubMed]
- Yamasue, K.; Morikawa, N.; Mizushima, S.; Tochikub, O. The blood pressure lowering effect of lactotripeptides and salt intake in 24-h ambulatory blood pressure measurements. Clin. Exp. Hypertens. 2010, 32, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Turpeinen, A.; Kumpu, M.; Rönnback, M.; Seppo, L.; Kautiainen, H.; Jauhiainen, T.; Vapaatalo, H.; Korpela, R. Antihypertensive and cholesterol-lowering effects of a spread containing bioactive peptides IPP and VPP and plant sterols. J. Funct. Food 2009, 1, 260–265. [Google Scholar] [CrossRef]
- Turpeinen, A.; Ikonen, M.; Kivimaki, A.S.; Kautiainen, H.; Vapaatalo, H.; Korpela, R. A spread containing bioactive milk peptides Ile-Pro-Pro and Val-Pro-Pro, and plant sterols has antuhypertensive and cholesterol-lowering effects. Food Funct. 2012, 3, 621–627. [Google Scholar] [CrossRef] [PubMed]
- Jauhiainen, T.; Rönnback, M.; Vapaatalo, H.; Wuolle, K.; Kautiainen, H.; Korpela, R. Lactobacillus helveticus fermented milk reduced arterial stiffness in hypertensive subjects. Int. Dairy J. 2007, 17, 1209–1211. [Google Scholar] [CrossRef]
- Usinger, L.; Ibsen, H.; Linneberg, A.; Azizi, M.; Flambard, B.; Jensen, L. Human in vivo study of the renin-angiotensin-aldosterone system and the sympathetic activity after 8 weeks daily intake of fermented milk. Clin. Physiol. Funct. Imaging 2010, 30, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Itakura, H.; Ikemoto, S.; Terada, S.; Konodo, K. The effect of sour milk on blood pressure in untreated hypertensive and normotensive subjects. J. Jpn. Soc. Clin. Nutr. 2001, 23, 26–31. [Google Scholar]
- Hirota, T.; Ohki, K.; Kawagishi, R.; Kajimoto, Y.; Mizuno, S.; Nakamura, Y.; Kitakaze, M. Casein hydrolysate containing the antihypertensive tripeptides Val-Pro-Pro and Ile-Pro-Pro improves vascular endothelial function independent of blood pressure-lowering effects: Contribution of the inhibitory action of angiotensin-converting enzyme. Hypertens. Res. 2007, 30, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, K.; Aihara, K.; Komazaki, K.; Mochii, M.; Nakamura, Y. Effect of large intake of tablets containing “lactotripeptides (VPP, IPP)” on blood pressure, pulse rate and clinical parameters in healthy volunteers. J. Nutr. Food 2001, 4, 63–72. [Google Scholar]
- Ishida, Y.; Sagitani, A.; Kaneko, K.; Nakamura, Y.; Mizutani, J.; Masuda, O.; Watanabe, M.; Sato, S.; Shioya, N. Antihypertensive effects of the tablet containing “lactotripeptide (IPP, VPP)” in subjects with high normal blood pressure or mild hypertension. J. Pharmacol. Ther. 2007, 35, 1249–1260. [Google Scholar]
- Yoshizawa, M.; Maeda, S.; Miyaki, A.; Misono, M.; Choi, Y.; Shimojo, N.; Ajisaka, R.; Tanaka, H. Additive beneficial effects of lactotripeptides and aerobic exercise on arterial compliance in postmenopausal women. Am. J. Physiol. Heart Circ. Physiol. 2009, 297, 1899–1903. [Google Scholar] [CrossRef]
- Yoshizawa, M.; Maeda, S.; Miyaki, A.; Misono, M.; Choi, Y.; Shimojo, N.; Ajisaka, R.; Tanaka, H. Additive beneficial effects of lactotripeptides intake with regular exercise on endothelium-dependent dilatation in postmenopausal women. Am. J. Hypertens. 2010, 23, 368–372. [Google Scholar] [CrossRef] [PubMed]
- Boelsma, E.; Kloek, J. IPP-rich milk protein hydrolysate lowers blood pressure in subjects with stage 1 hypertension, a randomised controlled trial. Nutr. J. 2010, 9. [Google Scholar] [CrossRef]
- Van Mierlo, L.A.; Koning, M.M.; van der Zander, K.; Draijer, R. Lactotripeptides do not lower ambulatory blood pressure in untreated whites: Results from 2 controlled multicenter crossover studies. Am. J. Clin. Nutr. 2009, 89, 617–623. [Google Scholar] [CrossRef] [PubMed]
- Usinger, L.; Jensen, L.T.; Flambard, B.; Linneberg, A.; Ibsen, H. The antihypertensive effect of fermented milk in individuals with prehypertension or borderline hypertension. J. Hum. Hypertens. 2010, 24, 678–683. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mizuno, S.; Matsuura, K.; Gotou, T.; Nishimura, S.; Kajimoto, O.; Yabune, M.; Kajimoto, Y.; Yamamoto, N. Antihypertensive effect of casein hydrolysate in a placebo-controlled study in subjects with high-normal blood pressure and mild hypertension. Br. J. Nutr. 2005, 94, 84–91. [Google Scholar] [CrossRef] [PubMed]
- De Leeuw, P.W.; van der Zander, K.; Kroon, A.A.; Rennenberg, R.M.; Koning, M.M. Dose-dependent lowering of blood pressure by dairy peptides in mildly hypertensive subjects. Blood Press. 2009, 18, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Jauhiainen, T.; Ronnback, M.; Vapaatalo, H.; Wuolle, K.; Kautiainen, H.; Groop, P.H.; Korpela, R. Long-term intervention with Lactobacillus helveticus fermented milk reduces augmentation index in hypertensive subjects. Eur. J. Clin. Nutr. 2010, 64, 424–431. [Google Scholar] [CrossRef] [PubMed]
- Jauhiainen, T.; Niittynen, L.; Oresic, M.; Jarvenpaa, S.; Hiltunen, T.P.; Ronnback, M.; Vapaatalo, H.; Korpela, R. Effects of long-term intake of lactotripeptides on cardiovascular risk factors in hypertensive subjects. Eur. J. Clin. Nutr. 2012, 66, 843–849. [Google Scholar] [CrossRef] [PubMed]
- Tuomilehto, J.; Lindstrom, J.; Hyyrynen, J.; Korpela, R.; Karhunen, M.L.; Mikkola, L.; Jauhiainen, T.; Seppo, L.; Nissinen, A. Effect of ingesting sour milk fermented using Lactobacillus helveticus bacteria producing tripeptides on blood pressure in subjects with mild hypertension. J. Hum. Hypertens. 2004, 18, 795–802. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.; Rosticci, M.; Veronesi, M.; Bacchelli, S.; Strocchi, E.; Melegari, C.; Grandi, E.; Borghi, C. Hemodynamic effects of lactotripeptides from casein hydrolysate in Mediterranean normotensive subjects and patients with high-normal blood pressure: A randomised, double-blind, crossover clinical trial. J. Med. Food 2010, 13, 1363–1368. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.; Rosticci, M.; Ferroni, A.; Bacchelli, S.; Veronesi, M.; Strocchi, E.; Borghi, C. Predictors of the short-term effect of isoleucine-proline-proline/valine-proline-proline lactotripeptdies from casein on office and ambulatory blood pressure in subjects with pharmacologically untreated high-normal blood pressure of first-degree hypertension. Clin. Exp. Hypertens. 2012, 34, 601–605. [Google Scholar] [CrossRef] [PubMed]
- Hata, Y.; Yamamoto, M.; Ohni, M.; Nakajima, K.; Nakamura, Y.; Takano, T. A placebo-controlled study of the effect of sour milk on blood pressure in hypertensive subjects. Am. J. Clin. Nutr. 1996, 64, 767–771. [Google Scholar] [PubMed]
- Kajimoto, O.; Aihara, K.; Hirata, H.; Takahashi, R.; Nakamura, Y. Safety evaluation of the tablet containing “Lactotripeptides (VPP, IPP)” on healthy volunteers. J. Nutr. Food 2001, 4, 37–46. [Google Scholar]
- Kajimoto, O.; Kurosaki, T.; Mizutani, J.; Ikeda, N.; Kaneko, K.; Aihara, K.; Yabune, M.; Nakamura, Y. Antihypertensive effects of liquid yogurts containing “lactotripeptides (VPP, IPP)” in mild hypertensive subjects. J. Nutr. Food 2002, 5, 55–66. [Google Scholar]
- Nakamura, T.; Mizutani, J.; Ohki, K.; Yamada, K.; Yamamoto, N.; Takeshi, M.; Takazawa, K. Casein hydrolysate containing Val-Pro-Pro and Ile-Pro-Pro improves central blood pressure and arterial stiffness in hypertensive subjects: A randomised, double-blind, placebo-controlled trial. Atherosclerosis 2011, 219, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Kajimoto, O.; Kaneko, K.; Aihara, K.; Mizutani, J.; Ikeda, N.; Nishimura, A.; Kajimoto, Y. Effects of the liquid yoghurts containing “lactotripeptide (VPP, IPP)” on high-normal blood pressure. J. Nutr. Food 2004, 7, 123–137. [Google Scholar]
- Mizushima, S.; Ohshige, K.; Watanabe, J.; Kimura, M.; Kadowaki, T.; Nakamura, Y.; Tochikubo, O.; Ueshima, H. Randomised controlled trial of sour milk on blood pressure in borderline hypertensive men. Am. J. Hypertens. 2004, 17, 701–706. [Google Scholar] [CrossRef] [PubMed]
- Aihara, K.; Kajimoto, O.; Hirata, H.; Takahashi, R.; Nakamura, Y. Effect of powdered fermented milk with Lactobacillus helveticus on subjects with high-normal blood pressure or mild hypertension. J. Am. Coll. Nutr. 2005, 24, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Sano, J.; Ohki, K.; Higuchi, T.; Aihara, K.; Mizuno, S.; Kajimoto, O.; Nakagawa, S.; Kajimoto, Y.; Nakamura, Y. Effect of casein hydrolysate, prepared with protease derived from Aspergillus oryzae, on subjects with high-normal blood pressure or mild hypertension. J. Med. Food 2005, 8, 423–430. [Google Scholar] [CrossRef] [PubMed]
- Sano, J.; Ohki, K.; Higuchi, T.; Aihara, K.; Mizuno, S.; Kajimoto, O.; Nakagawa, S.; Kajimoto, Y.; Nakamura, Y. Safety evaluation of excessive intake of drink containing “lactotripeptides (VPP, IPP)” in subjects with normal blood pressure to mild hypertension. J. Nutr. Food 2005, 7, 17–30. [Google Scholar]
- Ishida, Y.; Shibata, Y.; Fukuhara, I.; Yano, Y.; Takehara, I.; Kaneko, K. Effect of an excess intake of casein hydrolysate containing Val-Pro-Pro and Ile-Pro-Pro in subjects with normal blood pressure, high-normal blood pressure, or mild hypertension. Biosci. Biotechnol. Biochem. 2011, 75, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Ishida, Y.; Aihara, K.; Sagitani, A.; Kaneko, K.; Mizutani, J.; Nakamura, K.; Shimura, T.; Yagasaki, K.; Nakamura, Y. Safety evaluation of excessive intake of the tablet containing “lactotripeptides (VPP, IPP)” in subject with normal blood pressure to mild hypertension. J. Pharmacol. Ther. 2006, 34, 1107–1117. [Google Scholar]
- Seppo, L.; Jauhiainen, T.; Poussa, T.; Korpela, R. A fermented milk high in bioactive peptides has a blood pressure-lowering effect in hypertensive subjects. Am. J. Clin. Nutr. 2003, 77, 326–330. [Google Scholar] [PubMed]
- Jauhiainen, T.; Vapaatalo, H.; Poussa, T.; Kyronpalo, S.; Rasmussen, M.; Korpela, R. Lactobacillus helveticus fermented milk lowers blood pressure in hypertensive subjects in 24-h ambulatory blood pressure measurement. Am. J. Hypertens. 2005, 18, 1600–1605. [Google Scholar] [CrossRef] [PubMed]
- Seppo, L.; Kerojoki, O.; Suomalainen, T.; Korpela, R. The effect of a Lactobacillus helveticus LBK-16 H fermented milk on hypertension—A pilot study on humans. Milchwissenschaft 2002, 57, 124–127. [Google Scholar]
- Engberink, M.F.; Schouten, E.G.; Kok, F.J.; van Mierlo, L.A.; Brouwer, I.A.; Geleijnse, J.M. Lactotripeptides show no effect on human blood pressure: Results from a double-blind randomised controlled trial. Hypertension 2008, 51, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Van der Zander, K.; Bots, M.L.; Bak, A.A.; Koning, M.M.; de Leeuw, P.W. Enzymatically hydrolyzed lactotripeptides do not lower blood pressure in mildly hypertensive subjects. Am. J. Clin. Nutr. 2008, 88, 1697–1702. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.; Rosticci, M.; Gerocarni, B.; Bacchelli, S.; Veronesi, M.; Strocchi, E.; Borghi, C. Lactotripeptides effect on office and 24-h ambulatory blood pressure, blood pressure stress response, pulse wave velocity and cardiac output in patients with high-normal blood pressure or first-degree hypertension: A randomised double-blind clinical trial. Hypertens. Res. 2011, 34, 1035–1040. [Google Scholar] [CrossRef]
- Germino, F.W.; Neutel, J.; Nonaka, M.; Hendler, S.S. The impact of lactotripeptides on blood pressure response in stage 1 and stage 2 hypertensives. J. Clin. Hypertens. 2010, 12, 153–159. [Google Scholar] [CrossRef]
- Sterne, J.; Sutton, A.; Ioannidis, J.; Terrin, N.; Jones, D.; Lau, J.; Carpenter, J.; Rücker, G.; Harbord, R.; Schmid, C.; et al. Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomised controlled trials. BMJ 2011, 343. [Google Scholar] [CrossRef]
- Mancia, G.; Grassi, G. Mechanisms and clinical implications of blood pressure variability. J. Cardiovasc. Pharmacol. 2000, 35, 15–19. [Google Scholar] [CrossRef]
- Cicero, A.F.G.; Aubin, F.; Azais-Braesco, V.; Borghi, C. Do the lactotripeptides Isoleucin-Proline-Proline and Valin-Proline-Proline reduce systolic blood pressure in European subjects? A meta-analysis of randomised controlled trial. Am. J. Hypertens. 2013, 26, 442–449. [Google Scholar] [PubMed]
- Johnson, J. Ethnic differences in cardiovascular drug response: Potential contribution of pharmacogenetics. Circulation 2008, 118, 1383–1393. [Google Scholar] [CrossRef] [PubMed]
- Campo, I.; Beghin, J. Dairy Food Consumption, Production, and Policy in Japan. Available online: http://ageconsearch.umn.edu/bitstream/18596/1/wp050401.pdf (accessed on 14 September 2012).
- Bulletin of the International Dairy Federation. The World Dairy Situation 2010. Available online: http://www.milksa.co.za/sites/default/files/KORINL070_world_dairy_situation_2010.pdf (accessed on 15 October 2012).
- Siltari, A.; Viitanen, R.; Kukkurainen, S.; Vapaatalo, H.; Valjakka, J. Does the cis/trans configuration of peptide bonds in bioactive tripeptides play a role in ACE-1 enzyme inhibition? Biologics 2014, 8, 59–65. [Google Scholar] [PubMed]
- Stamler, J. Epidemiologic findings on body mass and blood pressure in adults. Ann. Epidemiol. 1991, 1, 347–362. [Google Scholar] [CrossRef] [PubMed]
- Thompson, S.G.; Higgins, J.P. How should meta-regression analyses be undertaken and interpreted? Stat. Med. 2002, 21, 1559–1573. [Google Scholar] [CrossRef] [PubMed]
- Berlin, J.A.; Santanna, J.; Schmid, C.H.; Szczech, L.A.; Feldman, H.I. Anti-Lymphocyte Antibody Induction Therapy Study Group. Individual patient- versus group-level data meta-regressions for the investigation of treatment effect modifiers: Ecological bias rears its ugly head. Stat. Med. 2002, 21, 371–387. [Google Scholar]
- Baguet, J.P.; Legallicier, B.; Auquier, P.; Robitail, S. Updated meta-analytical approach to the efficacy of antihypertensive drugs in reducing blood pressure. Clin. Drug Investig. 2007, 27, 735–753. [Google Scholar] [CrossRef] [PubMed]
- Joshi, V.D.; Dahake, A.P.; Suthar, A.P. Adverse effects associated with the use of antihypertesive drugs: An overview. Int. J. Pharm. Tech. Res. 2010, 2, 10–13. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fekete, Á.A.; Givens, D.I.; Lovegrove, J.A. Casein-Derived Lactotripeptides Reduce Systolic and Diastolic Blood Pressure in a Meta-Analysis of Randomised Clinical Trials. Nutrients 2015, 7, 659-681. https://doi.org/10.3390/nu7010659
Fekete ÁA, Givens DI, Lovegrove JA. Casein-Derived Lactotripeptides Reduce Systolic and Diastolic Blood Pressure in a Meta-Analysis of Randomised Clinical Trials. Nutrients. 2015; 7(1):659-681. https://doi.org/10.3390/nu7010659
Chicago/Turabian StyleFekete, Ágnes A., D. Ian Givens, and Julie A. Lovegrove. 2015. "Casein-Derived Lactotripeptides Reduce Systolic and Diastolic Blood Pressure in a Meta-Analysis of Randomised Clinical Trials" Nutrients 7, no. 1: 659-681. https://doi.org/10.3390/nu7010659
APA StyleFekete, Á. A., Givens, D. I., & Lovegrove, J. A. (2015). Casein-Derived Lactotripeptides Reduce Systolic and Diastolic Blood Pressure in a Meta-Analysis of Randomised Clinical Trials. Nutrients, 7(1), 659-681. https://doi.org/10.3390/nu7010659





