Interleukin-6 Gene Polymorphisms, Dietary Fat Intake, Obesity and Serum Lipid Concentrations in Black and White South African Women
Abstract
:1. Introduction
2. Experimental Section
2.1. Subjects
2.2. Measurements
2.2.1. Body Composition
2.2.2. Blood Sampling and Analysis
2.2.3. Polymorphism Selection
2.2.4. DNA Extraction and Genotype Analysis
2.2.5. IL-6 IVS3 +281 G>T, rs1554606
2.2.6. IL-6 IVS4 +869 A>G, rs2069845
2.2.7. Dietary Assessment
2.3. Statistical Analysis
3. Results
3.1. Subject Characteristics
3.2. Genotype and Allelic Frequency
| Subject characteristics | Black, n = 147 | White, n = 121 | p-values | ||||
|---|---|---|---|---|---|---|---|
| Normal-weight | Obese | Normal-weight | Obese | BMI Group | Ethnicity | Interaction | |
| n | 73 | 74 | 73 | 48 | - | - | - |
| Age, years | 24 ± 6 | 30 ± 8 | 28 ± 7 | 34 ± 9 | <0.001 | <0.001 | 0.969 |
| Body Composition a | |||||||
| Height, m | 1.61 ± 0.06 | 1.59 ± 0.06 | 1.67 ± 0.07 | 1.67 ± 0.07 | 0.009 | <0.001 | 0.350 |
| Weight, kg | 58 ± 6 | 91 ± 13 | 61 ± 6 | 97 ± 14 | <0.001 | 0.001 | 0.272 |
| BMI, kg/m2 | 22 ± 2 | 36 ± 5 | 21 ± 2 | 35 ± 4 | <0.001 | 0.005 | 0.596 |
| Fat Mass, kg | 17 ± 4 | 41 ± 9 | 17 ± 4 | 44 ± 9 | <0.001 | 0.190 | 0.045 |
| Body fat, % | 31 ± 5 | 45 ± 4 | 28 ± 5 | 45 ± 4 | <0.001 | 0.006 | 0.004 |
| Waist, cm | 74 ± 6 | 103 ± 12 | 77 ± 6 | 106 ± 10 | <0.001 | 0.004 | 0.972 |
| WHR | 0.75 ± 0.06 | 0.83 ± 0.08 | 0.78 ± 0.05 | 0.85 ± 0.06 | <0.001 | <0.001 | 0.426 |
| Metabolic Outcomes b | |||||||
| TAG, mmol/L | 0.50 (0.40–0.70) | 0.80 (0.60–1.04) | 0.80 (0.60–1.08) | 1.10 (0.78–1.50) | <0.001 | <0.001 | 0.339 |
| T-C, mmol/L | 3.8 (3.3–4.2) | 4.1 (3.6–4.5) | 4.5 (4.0–5.0) | 5.0 (4.4–5.7) | 0.031 | <0.001 | 0.357 |
| HDL-C, mmol/L | 1.5 (1.2–1.7) | 1.1 (1.0–1.3) | 1.8 (1.6–2.0) | 1.5 (1.3–1.6) | <0.001 | < 0.001 | 0.638 |
| LDL-C, mmol/L | 2.1 (1.6–2.5) | 2.5 (2.0–2.9) | 2.3 (1.9–2.7) | 2.9 (2.6–3.5) | <0.001 | < 0.001 | 0.215 |
| T-C:HDL–C ratio | 2.6 (2.2–3.1) | 3.5 (2.9–4.1) | 2.6 (2.2–2.8) | 3.5 (3.0–4.2) | <0.001 | 0.520 | 0.260 |
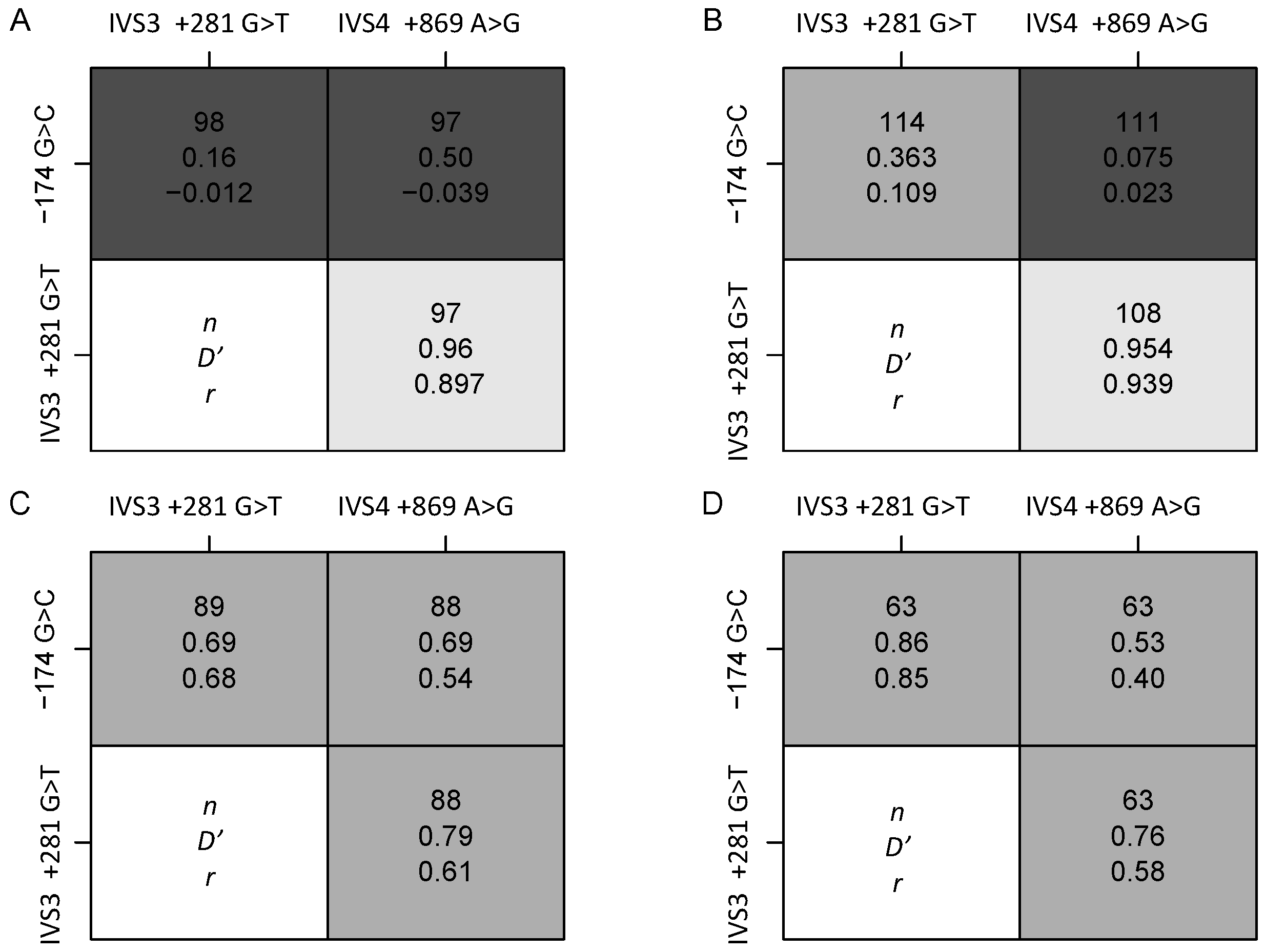
| Subject characteristics | Normal-weight | Obese | * p-values | Ensemble 1000 Genomes: Phase 1 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Black | White | Black | White | * Ethnicity | ** BMI Group | EUR | GBR | AFR | ASW | |
| IL-6 174 G>C rs1800795 | ||||||||||
| n | 72 | 74 | 72 | 48 | - | - | - | - | - | - |
| G/G | 0.97 | 0.28 | 0.97 | 0.25 | - | - | 0.36 | 0.39 | 0.95 | 0.78 |
| G/C | 0.03 | 0.61 | 0.03 | 0.50 | - | - | 0.44 | 0.42 | 0.05 | 0.21 |
| C/C | 0.00 | 0.11 | 0.00 | 0.25 | <0.001 | 0.104 | 0.20 | 0.19 | 0.0 | 0.0 |
| C | 0.01 | 0.41 | 0.01 | 0.50 | <0.001 | 0.515 | 0.41 | 0.40 | 0.02 | 0.11 |
| *** HWE p-value | 1.000 | 0.053 | 1.000 | 1.000 | - | - | - | - | - | - |
| IL-6 281 G>T Dde I rs1554606 | ||||||||||
| n | 66 | 74 | 69 | 48 | - | - | - | - | - | - |
| G/G | 0.59 | 0.35 | 0.59 | 0.25 | - | - | 0.34 | 0.36 | 0.48 | 0.39 |
| AG/T | 0.35 | 0.47 | 0.36 | 0.54 | - | - | 0.45 | 0.44 | 0.46 | 0.57 |
| T/T | 0.06 | 0.18 | 0.04 | 0.21 | <0.001 | 0.914 | 0.20 | 0.19 | 0.05 | 0.03 |
| T | 0.23 | 0.41 | 0.22 | 0.48 | <0.001 | 0.950 | 0.43 | 0.41 | 0.28 | 0.32 |
| *** HWE p-value | 0.740 | 0.814 | 1.000 | 0.772 | - | - | - | - | - | - |
| IL-6 869 A>G Msp I rs2069845 | ||||||||||
| n | 65 | 73 | 65 | 48 | - | - | - | - | - | - |
| A/A | 0.55 | 0.38 | 0.58 | 0.29 | - | - | 0.34 | 0.36 | 0.46 | 0.37 |
| G/A | 0.35 | 0.60 | 0.37 | 0.71 | - | - | 0.45 | 0.44 | 0.47 | 0.57 |
| G/G | 0.09 | 0.01 | 0.05 | 0.00 | <0.001 | 0.293 | 0.20 | 0.19 | 0.05 | 0.04 |
| G | 0.27 | 0.32 | 0.23 | 0.35 | 0.688 | 0.474 | 0.43 | 0.41 | 0.29 | 0.33 |
| *** HWE p-value | 0.526 | 0.001 | 1.000 | <0.001 | - | - | - | - | - | - |
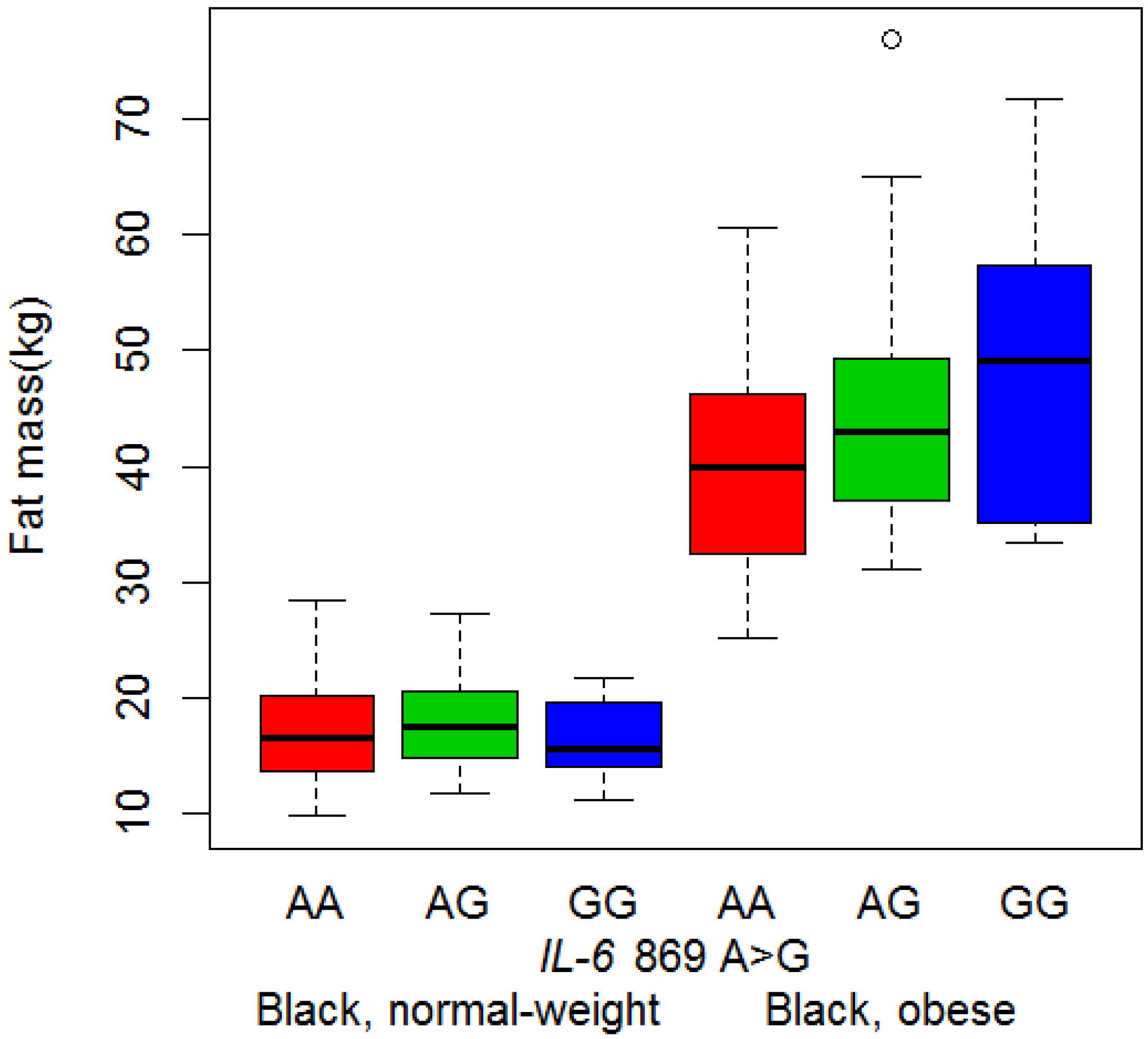
3.3. Body Composition and Genotype
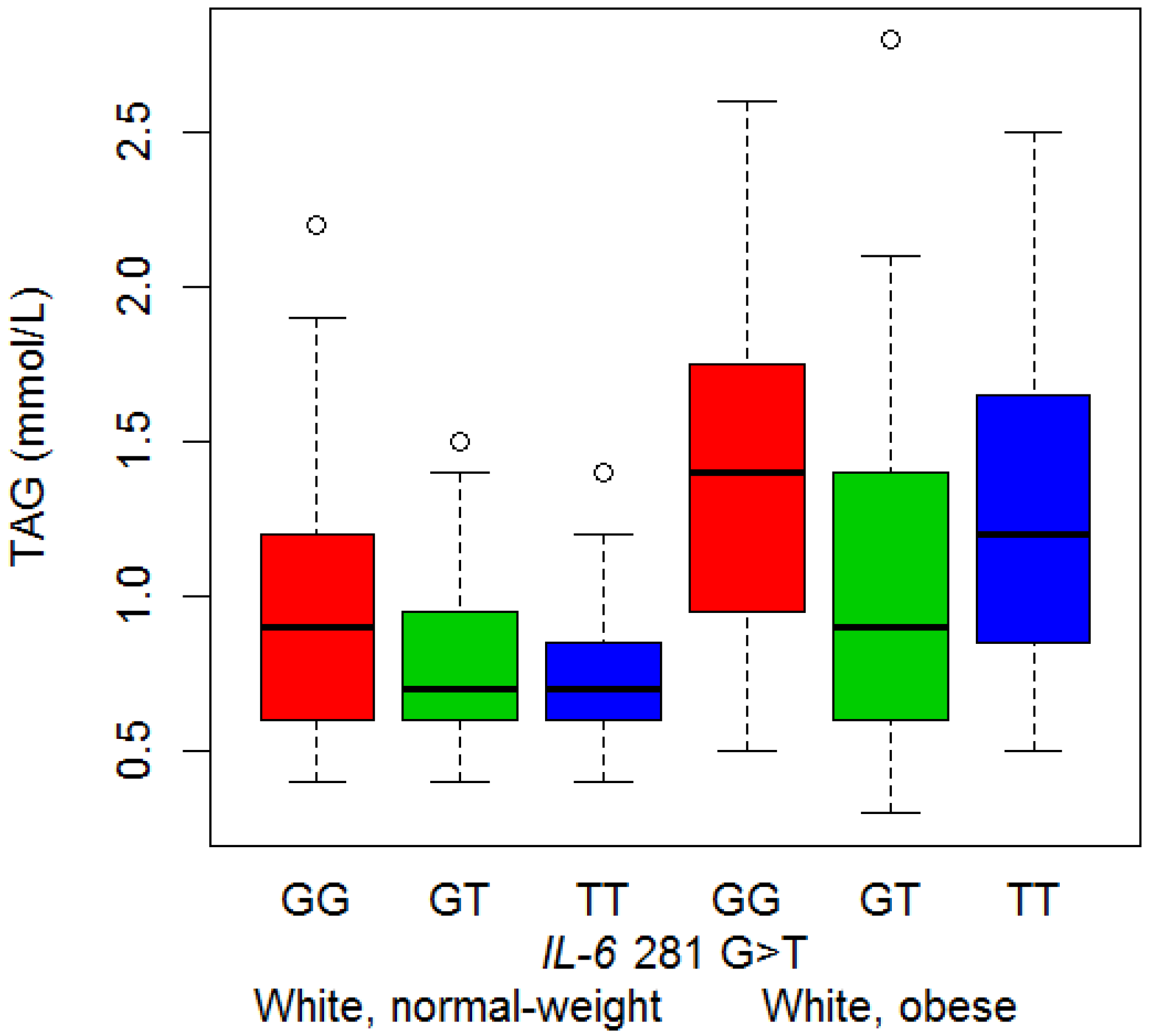
3.4. Serum Lipid Concentrations and Genotype
3.5. Dietary Intake
| Dietary variable | Black | White | p-value | |||
|---|---|---|---|---|---|---|
| Normal-weight | Obese | Normal-weight | Obese | BMI Group | Ethnic Group | |
| n | 73 | 74 | 73 | 48 | - | - |
| Fat (%E) | 33.7 (28.3–37.2) | 34.7 (31.1–38.5) | 29.6 (25.7–32.5) | 30.6 (26.3–33.9) | 0.023 | 0.002 |
| SFA (%E) | 9.2 (7.9–10.7) | 9.5 (8.3–10.9) | 9.7 (8.3–12.0) | 11.2 (9.1–12.3) | 0.041 | <0.001 |
| MUFA (%E) | 10.3 (8.8–11.8) | 11.4 (9.8–12.9) | 10.2 (8.7–11.6) | 10.4 (8.4–12.1) | 0.05 | 0.844 |
| PUFA (%E) | 8.4 (6.8–10.2) | 9.5 (7.7–11.0) | 6.1 (4.9–7.9) | 6.1 (4.8–7.6) | 0.007 | <0.001 |
| P:S ratio | 0.91 (0.73–1.17) | 1 (0.81–1.21) | 0.62 (0.46–0.78) | 0.56 (0.48–0.72) | 0.205 | <0.001 |
| (n-3) PUFA (%E) | 0.28 (0.22–0.34) | 0.36 (0.27–0.46) | 0.33 (0.26–0.41) | 0.32 (0.27–0.39) | 0.296 | 0.997 |
| (n-6) PUFA (%E) | 7.9 (6.6–10.3) | 9.1 (7.3–10.7) | 5.5 (4.2–7.5) | 5.5 (4.4–7.2) | 0.01 | <0.001 |
| (n-6):(n-3) PUFA ratio | 27 (20.0–39.9) | 25.7 (18.3–36.5) | 15.9 (12.3–22.5) | 16.4 (13.0–24.9) | 0.203 | <0.001 |
| ALA (%E) | 0.21 (0.18–0.25) | 0.22 (0.18–0.27) | 0.26 (0.21–0.30) | 0.25 (0.21–0.29) | 0.372 | <0.001 |
| LA (%E) | 8.1 (6.70–10.6) | 9.3 (7.50–10.9) | 5.5 (4.20–7.40) | 5.5 (4.40–7.20) | 0.011 | <0.001 |
| AA (%E) | 0.04 (0.030–0.060) | 0.06 (0.040–0.070) | 0.03 (0.020–0.040) | 0.04 (0.030–0.050) | <0.001 | <0.001 |
| EPA (%E) | 0.017 (0.009–0.035) | 0.037 (0.016–0.065) | 0.014 (0.009–0.023) | 0.018 (0.009–0.025) | 0.037 | <0.001 |
| DHA (%E) | 0.037 (0.023–0.076) | 0.079 (0.038–0.128) | 0.047 (0.029–0.063) | 0.045 (0.028–0.066) | 0.043 | 0.001 |
3.6. Diet-Genotype Interactions on BMI and Body Composition
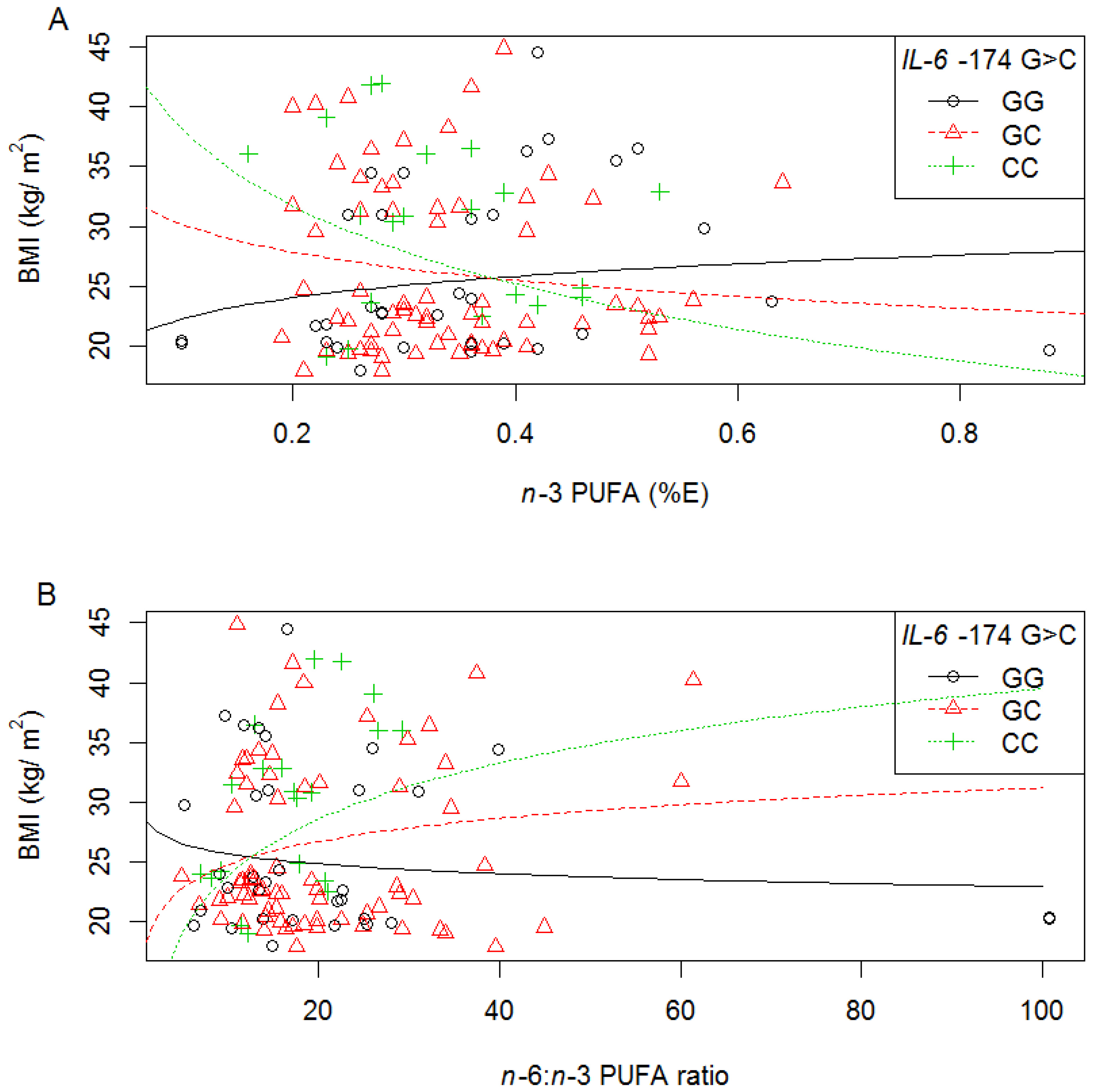
3.6.1. IL-6 −174 G>C
| Dietary variable | SNP | Black | White | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Weight, kg | BMI, kg/m2 | Fat Mass, kg | Waist, cm | BMI, kg/m2 | Fat Mass, kg | Body Fat % | Waist, cm | WHR | ||
| Total fat | IVS3 +281 G>T | ↑ GG or GT ↓ TT | ↑ GG or GT ↓ TT | ↑ GG or GT ↓ TT | ↑ GG or GT ↓ TT | - | - | - | - | - |
| IVS4 +869 A>G | ↑ AG or AA a ↓ GG a | ↑ AG or AA a ↓ GG a | ↑ AG or AA a ↓ GG a | ↑ AG or AA ↓ GG a | - | - | - | - | - | |
| SFA | IVS3 +281 G>T | - | ↑ GG or G b ↓ TT b | ↑ GG or GT ↓ TT | - | - | - | - | - | - |
| IVS4 +869 A>G | ↑ AG or AA a ↓ GG a | ↑ AG or AA a ↓ GG a | ↑ AG or AA a ↓ GG a | ↑ AG or AA a ↓ GG a | - | - | - | - | - | |
| MUFA | IVS3 +281 G>T | - | ↓ TT | ↑ GG or GT ↓ TT | - | - | - | - | - | - |
| IVS4 +869 A>G | ↑ AG or AA a ↓ GG a | ↑ AG or AA a ↓ GG a | ↑ AG or AA a ↓ GG a | ↑ AG or AA a ↓ GG a | - | - | - | - | - | |
| PUFA | IVS3 +281 G>T | ↑ GG or GT ↓ TT | ↑ GG or GT b ↓ TT b | ↑ GG or GT ↓ TT | ↑ GG or GT ↓ TT | - | - | - | - | - |
| n-6 PUFA | IVS3 +281 G>T | ↑ GG or GT ↓ TT | ↑ GG or G b ↓ TT b | ↑ GG or GT ↓ TT | - | - | - | - | - | - |
| LA | IVS3 +281 G>T | ↑ GG or GT ↓ TT | ↑ GG or G b ↓ TT b | ↑ GG or GT ↓ TT | - | - | - | - | - | - |
| AA | IVS3 +281 G>T | ↑ GG or GT ↓ TT | ↑ GG or GT b ↓ TT b | ↑ GG or GT ↓ TT | - | - | - | - | - | - |
| IVS4 +869 A>G | ↑ AG or AA | ↑ AG or AA | ↑ AG or AA | - | - | - | - | - | - | |
| n-3 PUFA | −174 G>C | - | - | - | - | ↓ C allele | - | - | - | - |
| IVS4 +869 A>G | - | - | - | - | - | ↓ AG or GG | - | - | - | |
| EPA | −174 G>C | - | - | - | - | ↓ C allele | - | - | - | - |
| DHA | −174 G>C | - | - | - | - | ↓ C allele | - | - | - | - |
| ALA | IVS3 +281 G>T | - | - | - | - | ↓ T allele | - | - | ↓ T allele | ↓ T allele |
| IVS4 +869 A>G | - | - | - | - | - | ↓ AG or GG b ↑ AA ab | - | - | - | |
| n-6:n-3 ratio | −174 G>C | - | - | - | - | ↑ C allele | - | - | - | - |
| IVS3 +281 G>T | - | - | - | - | ↑ T allele b | - | ↑ T allele | ↑ T allele b | - | |
| IVS4 +869 A>G | - | - | - | - | - | ↑ AG or GG c | - | - | - | |
3.6.2. IVS3 +281 G>T
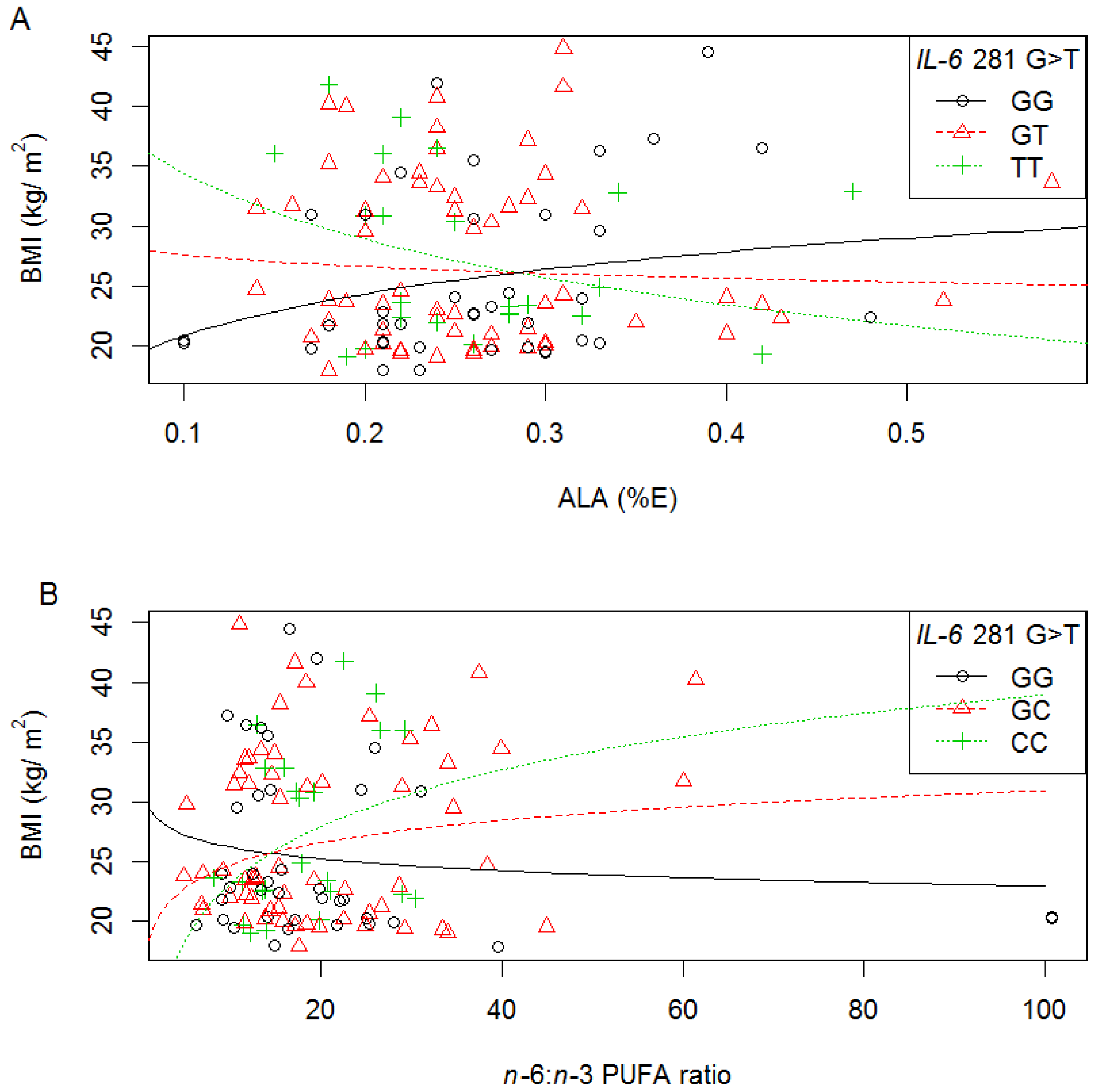
3.6.3. IVS4 +869 A>G
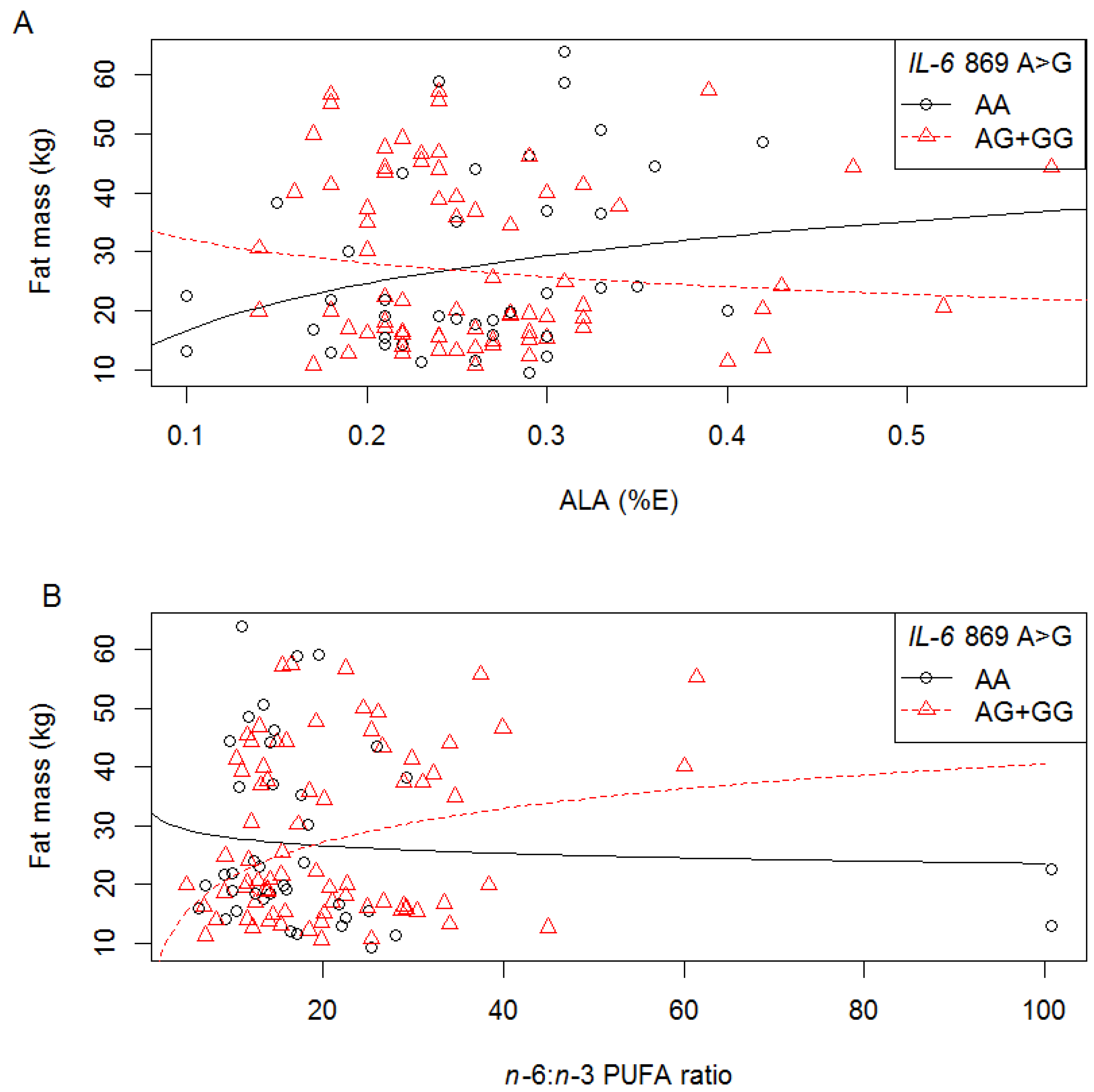
3.7. Diet-Genotype Interactions on Serum Lipid Concentrations
3.7.1. IL-6 −174 G>C
| Dietary variable | SNP | Black | White | ||||
|---|---|---|---|---|---|---|---|
| TAG, mmol/L | LDL-C, mmol/L | T-C:HDL-C Ratio | TAG, mmol/L | HDL-C, mmol/L | T-C:HDL-C Ratio | ||
| Total fat | IVS3 +281 G>T | - | ↓ VGT | ↓ GT | - | - | - |
| IVS4 +869 A>G | ↑ GG ↓ AA | - | ↑ GG a ↓ AA a | - | - | - | |
| MUFA | −174 G>C | - | - | - | ↓ C allele | - | - |
| IVS3 +281 G>T | - | ↓ GT | ↓ GT | - | - | - | |
| PUFA | IVS3 +281 G>T | - | ↓ GT b | ↓ GT | - | - | - |
| n -6 PUFA | IVS3 +281 G>T | - | ↓ GT b | ↓ GT | - | - | - |
| LA | IVS3 +281 G>T | - | ↓ GT b | ↓ GT | - | - | - |
| n -3 PUFA | −174 G>C | - | - | - | - | ↑ C allele | - |
| EPA | −174 G>C | - | - | - | ↓ C allele | - | ↓ CC |
| IVS3 +281 G>T | - | - | - | - | - | ↓ TT | |
| IVS4 +869 A>G | - | - | - | - | ↓ G allele a | - | |
| DHA | −174 G>C | - | - | - | - | ↑ C allele | ↓ CC |
| IVS4 +869 A>G | - | - | - | - | ↓ G allele a | - | |
| ALA | −174 G>C | - | - | - | - | ↑ C allele | - |
| IVS3 +281 G>T | - | - | - | - | ↑ T allele | ↓ TT b | |
| n -6: n -3 ratio | IVS4 +869 A>G | ↑ GG ↓ AA | - | ↑ GG ↓ AA | - | - | - |
3.7.2. IVS3 +281 G>T
3.7.3. IVS4 +869 A>G
4. Discussion
5. Conclusions
Supplementary Files
Abbreviations
| AA | arachidonic acid |
| ALA | α-linolenic acid |
| CVD | cardiovascular disease |
| CHO | carbohydrate |
| DHA | docosahexaenoic acid |
| EPA | eicosapentaenoic acid |
| HDL-C | high density lipoprotein-cholesterol |
| hsCRP | high sensitivity C-reactive protein |
| HOMA-IR | homeostasis model assessment of insulin resistance |
| IL-6 | interleukin 6 |
| IL-6 | interleukin six gene |
| LA | linoleic acid |
| LDL-C | low density lipoprotein-cholesterol |
| MUFA | mono-unsaturated fat |
| %E | percentage energy |
| (n-6) PUFA | (omega-6) polyunsaturated fatty acids |
| (n-3) PUFA | (omega-3) polyunsaturated fatty acid |
| (n-6):(n-3) PUFA ratio | (omega-6):(omega-3) polyunsaturated fatty acids ratio |
| P:S ratio | polyunsaturated:saturated fat ratio |
| SFA | saturated fat |
| SNP | single nucleotide polymorphism |
| SA | South African |
| T-C | total cholesterol |
| T-C:HDL-C ratio | total cholesterol:high density lipoprotein-cholesterol ratio |
| TAG | triacylglycerol |
| VAT | visceral adipose tissue |
| VLDL-C | very low density lipoprotein-cholesterol |
| WHR | waist hip ratio |
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Eder, K.; Baffy, N.; Falus, A.; Fulop, A.K. The major inflammatory mediator interleukin-6 and obesity. Inflamm. Res. 2009, 58, 727–736. [Google Scholar] [CrossRef]
- Vozarova, B.; Weyer, C.; Hanson, K.; Tataranni, P.A.; Bogardus, C.; Pratley, R.E. Circulating interleukin-6 in relation to adiposity, insulin action, and insulin secretion. Obes. Res. 2001, 9, 414–417. [Google Scholar] [CrossRef]
- Shoelson, S.E.; Herrero, L.; Naaz, A. Obesity, inflammation, and insulin resistance. Gastroenterology 2007, 132, 2169–2180. [Google Scholar] [CrossRef]
- Rexrode, K.M.; Pradhan, A.; Manson, J.E.; Buring, J.E.; Ridker, P.M. Relationship of total and abdominal adiposity with CRp and IL-6 in women. Ann. Epidemiol. 2003, 13, 674–682. [Google Scholar] [CrossRef]
- Jenny, N.S.; Tracy, R.P.; Ogg, M.S.; Luong, L.A.; Kuller, L.H.; Arnold, A.M.; Sharrett, A.R.; Humphries, S.E. In the elderly, interleukin-6 plasma levels and the −174G>C polymorphism are associated with the development of cardiovascular disease. Arterioscler. Thromb. Vasc. Biol. 2002, 22, 2066–2071. [Google Scholar] [CrossRef]
- Henningsson, S.; Hakansson, A.; Westberg, L.; Baghaei, F.; Rosmond, R.; Holm, G.; Ekman, A.; Nissbrandt, H.; Eriksson, E. Interleukin-6 gene polymorphism –174G/C influences plasma lipid levels in women. Obesity 2006, 14, 1868–1873. [Google Scholar]
- Humphries, S.E.; Luong, L.A.; Ogg, M.S.; Hawe, E.; Miller, G.J. The interleukin-6 −174 G/C promoter polymorphism is associated with risk of coronary heart disease and systolic blood pressure in healthy men. Eur. Heart J. 2001, 22, 2243–2252. [Google Scholar] [CrossRef]
- Riikola, A.; Sipila, K.; Kahonen, M.; Jula, A.; Nieminen, M.S.; Moilanen, L.; Kesaniemi, Y.A.; Lehtimaki, T.; Hulkkonen, J. Interleukin-6 promoter polymorphism and cardiovascular risk factors: The Health 2000 Survey. Atherosclerosis 2009, 207, 466–470. [Google Scholar] [CrossRef]
- Curti, M.L.; Jacob, P.; Borges, M.C.; Rogero, M.M.; Ferreira, S.R. Studies of gene variants related to inflammation, oxidative stress, dyslipidemia, and obesity: Implications for a nutrigenetic approach. J. Obes. 2011, 2011, ID497401. [Google Scholar]
- Qi, L.; Zhang, C.; van Dam, R.M.; Hu, F.B. Interleukin-6 genetic variability and adiposity: Associations in two prospective cohorts and systematic review in 26,944 individuals. J. Clin. Endocrinol. Metab. 2007, 92, 3618–3625. [Google Scholar] [CrossRef]
- Sie, M.P.; Sayed-Tabatabaei, F.A.; Oei, H.H.; Uitterlinden, A.G.; Pols, H.A.; Hofman, A.; van Duijn, C.M.; Witteman, J.C. Interleukin 6 −174 G/C promoter polymorphism and risk of coronary heart disease: Results from the rotterdam study and a meta-analysis. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 212–217. [Google Scholar] [CrossRef]
- Walston, J.D.; Fallin, M.D.; Cushman, M.; Lange, L.; Psaty, B.; Jenny, N.; Browner, W.; Tracy, R.; Durda, P.; Reiner, A. IL-6 gene variation is associated with IL-6 and C-reactive protein levels but not cardiovascular outcomes in the Cardiovascular Health Study. Hum. Genet. 2007, 122, 485–494. [Google Scholar] [CrossRef]
- Yu, Z.; Han, S.; Cao, X.; Zhu, C.; Wang, X.; Guo, X. Genetic Polymorphisms in Adipokine Genes and the Risk of Obesity: A Systematic Review and Meta-Analysis. Obesity 2012, 20, 396–406. [Google Scholar]
- Huth, C.; Illig, T.; Herder, C.; Gieger, C.; Grallert, H.; Vollmert, C.; Rathmann, W.; Hamid, Y.H.; Pedersen, O.H.; Hansen, T.; et al. Joint analysis of individual participants’ data from 17 studies on the association of the IL6 variant −174 G>C with circulating glucose levels, interleukin-6 levels, and body mass index. Ann. Med. 2009, 41, 128–138. [Google Scholar] [CrossRef]
- Fernandez-Real, J.M.; Broch, M.; Vendrell, J.; Richart, C.; Ricart, W. Interleukin-6 gene polymorphism and lipid abnormalities in healthy subjects. J. Clin. Endocrinol. Metab. 2000, 85, 1334–1339. [Google Scholar]
- Hulkkonen, J.; Lehtimaki, T.; Mononen, N.; Juonala, M.; Hutri-Kahonen, N.; Taittonen, L.; Marniemi, J.; Nieminen, T.; Viikari, J.; Raitakari, O.; et al. Polymorphism in the IL6 promoter region is associated with the risk factors and markers of subclinical atherosclerosis in men: The Cardiovascular Risk in Young Finns Study. Atherosclerosis 2009, 203, 454–458. [Google Scholar] [CrossRef]
- Joffe, Y.T.; van der Merwe, L.; Carstens, M.; Collins, M.; Jennings, C.; Levitt, N.S.; Lambert, E.V.; Goedecke, J.H. Tumor necrosis factor-alpha gene −308 G/A polymorphism modulates the relationship between dietary fat intake, serum lipids, and obesity risk in black South African women. J. Nutr. 2010, 140, 901–907. [Google Scholar]
- Joffe, Y.T.; van der Merwe, L.; Collins, M.; Carstens, M.; Evans, J.; Lambert, E.V.; Goedecke, J.H. The –308 G/A polymorphism of the tumour necrosis factor-alpha gene modifies the association between saturated fat intake and serum total cholesterol levels in white South African women. Genes Nutr. 2011, 6, 353–359. [Google Scholar]
- Joffe, Y.T.; van der Merwe, L.; Evans, J.; Collins, M.; Lambert, E.V.; September, A.; Goedecke, J.H. The tumor necrosis factor-alpha gene −238 G>A polymorphism, dietary fat intake, obesity risk and serum lipid concentrations in black and white South African women. Eur. J. Clin. Nutr. 2012, 66, 1295–1302. [Google Scholar]
- Nieters, A.; Becker, N.; Linseisen, J. Polymorphisms in candidate obesity genes and their interaction with dietary intake of n-6 polyunsaturated fatty acids affect obesity risk in a sub-sample of the EPIC-Heidelberg cohort. Eur. J. Nutr. 2002, 41, 210–221. [Google Scholar]
- Fontaine-Bisson, B.; El-Sohemy, A. Genetic polymorphisms of tumor necrosis factor-alpha modify the association between dietary polyunsaturated fatty acids and plasma high-density lipoprotein-cholesterol concentration in a population of young adults. J. Nutr. Nutr. 2008, 1, 215–223. [Google Scholar] [CrossRef]
- Fontaine-Bisson, B.; Wolever, T.M.; Chiasson, J.L.; Rabasa-Lhoret, R.; Maheux, P.; Josse, R.G.; Leiter, L.A.; Rodger, N.W.; Ryan, E.A.; Connelly, P.W.; et al. Genetic polymorphisms of tumor necrosis factor-alpha modify the association between dietary polyunsaturated fatty acids and fasting HDL-cholesterol and apo A-I concentrations. Am. J. Clin. Nutr. 2007, 86, 768–774. [Google Scholar]
- Corpeleijn, E.; Petersen, L.; Holst, C.; Saris, W.H.; Astrup, A.; Langin, D.; MacDonald, I.; Martinez, J.A.; Oppert, J.M.; Polak, J.; et al. obesity-related polymorphisms and their associations with the ability to regulate fat oxidation in obese Europeans: The NUGENOB study. Obesity 2010, 18, 1369–1377. [Google Scholar] [CrossRef]
- Razquin, C.; Martinez, J.A.; Martinez-Gonzalez, M.A.; Fernandez-Crehuet, J.; Santos, J.M.; Marti, A. A Mediterranean diet rich in virgin olive oil may reverse the effects of the −174G/C IL6 gene variant on 3-year body weight change. Mol. Nutr. Food Res. 2010, 54 (Suppl. 1), S75–S82. [Google Scholar]
- Belfer, I.; Buzas, B.; Hipp, H.; Dean, M.; Evans, C.; Lorincz, I.; Max, M.B.; Goldman, D. Haplotype structure of inflammatory cytokines genes (IL1B, IL6 and TNF/LTA) in US Caucasians and African Americans. Genes Immun. 2004, 5, 505–512. [Google Scholar] [CrossRef]
- Friedewald, W.T.; Levy, R.I.; Fredrickson, D.S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin. Chem. 1972, 18, 499–502. [Google Scholar]
- Martin, A.M.; Athanasiadis, G.; Greshock, J.D.; Fisher, J.; Lux, M.P.; Calzone, K.; Rebbeck, T.R.; Weber, B.L. Population frequencies of single nucleotide polymorphisms (SNPs) in immuno-modulatory genes. Hum. Hered. 2003, 55, 171–178. [Google Scholar] [CrossRef]
- Lahiri, D.K.; Nurnberger, J.I., Jr. A rapid non-enzymatic method for the preparation of HMW DNA from blood for RFLp studies. Nucleic Acids Res. 1991, 19, 5444. [Google Scholar] [CrossRef]
- Steyn, N.P.; Senekal, M. Dietary Assessment and Education Kit; Medical Research Council: Cape Town, South Africa, 2004. [Google Scholar]
- De Villiers, A.; Dugas, L.; Lambert, E.V.; Senekal, M. Relative Validity of a Quantified Food Frequency Questionnaire in Black South African Women. BSc (Med)(Hon) in Nutrition and Dietetics; University of Cape Town: Cape Town, South Africa, 2006. [Google Scholar]
- Grant, K.I.; Langenhoven, M.L.; Stockton, M.A. Foodfinder Dietary Analysis Software. Release 1.10; Medical Research Council: Cape Town, South Africa, 1992. [Google Scholar]
- Black, A.E. Critical evaluation of energy intake using the Goldberg cut-off for energy intake:Basal metabolic rate. A practical guide to its calculation, use and limitations. Int. J. Obes. 2000, 24, 1119–1130. [Google Scholar] [CrossRef]
- Tooze, J.A.; Krebs-Smith, S.M.; Troiano, R.P.; Subar, A.F. The accuracy of the Goldberg method for classifying misreporters of energy intake on a food frequency questionnaire and 24-h recalls: Comparison with doubly labeled water. Eur. J. Clin. Nutr. 2012, 66, 569–576. [Google Scholar]
- FAO/WHO/UNU. Energy and Protein Requirements; Technical Report Series 724; World Health Organisation: Geneva, Switzerland, 1985. [Google Scholar]
- Nyholt, D.R. A simple correction for multiple testing for single-nucleotide polymorphisms in linkage disequilibrium with each other. Am. J. Hum. Genet. 2004, 74, 765–769. [Google Scholar] [CrossRef]
- Perneger, T.V. What’s wrong with Bonferroni adjustments. BMJ 1998, 316, 1236–1238. [Google Scholar] [CrossRef]
- Nyholt, D.R. Evaluation of Nyholt’s procedure for multiple testing correction—Author’s reply. Hum. Hered. 2005, 60, 61–62. [Google Scholar] [CrossRef]
- Warnes, G.; Gorjanc, G.; Leisch, F.; Man, M. Genetics: Population Genetics. R package version 1.3.8.1., R package version 1.3.6. 2013. Available online: http://cran.r-project.org/package=genetics (accessed on 4 June 2014).
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing: Vienna, Austria, 2013. Available online: http://www.r-project.org/ (accessed on 4 June 2014).
- Flicek, P.; Amode, M.R.; Barrell, D.; Beal, K.; Brent, S.; Chen, Y.; Clapham, P.; Coates, G.; Fairley, S.; Fitzgerald, S.; et al. Ensembl 2011. Nucleic Acids Res. 2011, 39, D800–D806. [Google Scholar] [CrossRef]
- Chan, K.H.; Brennan, K.; You, N.C.; Lu, X.; Song, Y.; Hsu, Y.H.; Chaudhuri, G.; Nathan, L.; Tinker, L.; Liu, S. Common variations in the genes encoding C-reactive protein, tumor necrosis factor-alpha, and interleukin-6, and the risk of clinical diabetes in the Women’s Health Initiative Observational Study. Clin. Chem. 2011, 57, 317–325. [Google Scholar] [CrossRef]
- Ness, R.B.; Haggerty, C.L.; Harger, G.; Ferrell, R. Differential distribution of allelic variants in cytokine genes among African Americans and White Americans. Am. J. Epidemiol. 2004, 160, 1033–1038. [Google Scholar]
- Puoane, T.; Steyn, K.; Bradshaw, D.; Laubscher, R.; Fourie, J.; Lambert, V.; Mbananga, N. Obesity in South Africa: The South African demographic and health survey. Obes. Res. 2002, 10, 1038–1048. [Google Scholar] [CrossRef]
- Goedecke, J.H.; Levitt, N.S.; Lambert, E.V.; Utzschneider, K.M.; Faulenbach, M.V.; Dave, J.A.; West, S.; Victor, H.; Evans, J.; Olsson, T.; et al. Differential effects of abdominal adipose tissue distribution on insulin sensitivity in black and white South African women. Obesity 2009, 17, 1506–1512. [Google Scholar] [CrossRef]
- Evans, J.; Goedecke, J.H.; Soderstrom, I.; Buren, J.; Alvehus, M.; Blomquist, C.; Jonsson, F.; Hayes, P.M.; Adams, K.; Dave, J.A.; et al. Depot- and ethnic-specific differences in the relationship between adipose tissue inflammation and insulin sensitivity. Clin. Endocrinol. 2010, 74, 51–59. [Google Scholar]
- Goedecke, J.H.; Utzschneider, K.; Faulenbach, M.V.; Rizzo, M.; Berneis, K.; Spinas, G.A.; Dave, J.A.; Levitt, N.S.; Lambert, E.V.; Olsson, T.; et al. Ethnic differences in serum lipoproteins and their determinants in South African women. Metabolism 2010, 59, 1341–1350. [Google Scholar] [CrossRef]
- Despres, J.P.; Couillard, C.; Gagnon, J.; Bergeron, J.; Leon, A.S.; Rao, D.C.; Skinner, J.S.; Wilmore, J.H.; Bouchard, C. Race, visceral adipose tissue, plasma lipids, and lipoprotein lipase activity in men and women: The Health, Risk Factors, Exercise Training, and Genetics (HERITAGE) family study. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 1932–1938. [Google Scholar] [CrossRef]
- Arora, P.; Garcia-Bailo, B.; Dastani, Z.; Brenner, D.; Villegas, A.; Malek, S.; Spector, T.D.; Richards, B.; El-Sohemy, A.; Karmali, M.; et al. Genetic polymorphisms of innate immunity-relatedinflammatory pathways and their association with factors related to type 2 diabetes. BMC Med. Genet. 2011, 12, 95. [Google Scholar]
- Liu, Y.; Berthier-Schaad, Y.; Fallin, M.D.; Fink, N.E.; Tracy, R.P.; Klag, M.J.; Smith, M.W.; Coresh, J. IL-6 haplotypes, inflammation, and risk for cardiovascular disease in a multiethnic dialysis cohort. J. Am. Soc. Nephrol. 2006, 17, 863–870. [Google Scholar] [CrossRef]
- Shen, J.; Arnett, D.K.; Perez-Martinez, P.; Parnell, L.D.; Lai, C.Q.; Peacock, J.M.; Hixson, J.E.; Tsai, M.Y.; Straka, R.J.; Hopkins, P.N.; et al. The effect of IL6–174C/G polymorphism on postprandial triglyceride metabolism in the GOLDN studyboxs. J. Lipid Res. 2008, 49, 1839–1845. [Google Scholar] [CrossRef]
- Shisana, O.; Labadarios, D.; Rehle, T.; Simbayi, L.; Zuma, K.; Dhansay, A.; Reddy, P.; Parker, W.; Hoosain, E.; Naidoo, P.; et al. South African National Health and Nutrition Examination Survey (SANHANES-1); HSRC Press: Cape Town, South Africa, 2013. [Google Scholar]
- Chronic Diseases of Lifestyle in South Africa: 1995–2005; South African Medical Research Council Technical Report; South African Medical Research Council: Cape Town, South Africa, 2006.
- Fenech, M.; El-Sohemy, A.; Cahill, L.; Ferguson, L.R.; French, T.A.; Tai, E.S.; Milner, J.; Koh, W.P.; Xie, L.; Zucker, M.; et al. Nutrigenetics and nutrigenomics: Viewpoints on the current status and applications in nutrition research and practice. J. Nutr. Nutr. 2011, 4, 69–89. [Google Scholar] [CrossRef]
- MacIntyre, U.E.; Kruger, H.S.; Venter, C.S.; Vorster, H.H. Dietary intakes of an African population in different stages of transition in the North West Province, South Africa: The THUSA study. Nutr Res. 2002, 22, 239–256. [Google Scholar] [CrossRef]
- Calder, P.C. N-3 polyunsaturated fatty acids and inflammation: From molecular biology to the clinic. Lipids 2003, 38, 343–352. [Google Scholar] [CrossRef]
- Calder, P.C. n-3 polyunsaturated fatty acids, inflammation, and inflammatory diseases. Am. J. Clin. Nutr. 2006, 83, 1505S–1519S. [Google Scholar]
- Calder, P.C.; Ahluwalia, N.; Brouns, F.; Buetler, T.; Clement, K.; Cunningham, K.; Esposito, K.; Jonsson, L.S.; Kolb, H.; Lansink, M.; et al. Dietary factors and low-grade inflammation in relation to overweight and obesity. Br. J. Nutr. 2011, 106, S5–S78. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Exp. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef]
- Stryjecki, C.; Mutch, D.M. Fatty acid-gene interactions, adipokines and obesity. Eur. J. Clin. Nutr. 2011, 65, 285–297. [Google Scholar] [CrossRef]
- He, K.; Liu, K.; Daviglus, M.L.; Jenny, N.S.; Mayer-Davis, E.; Jiang, R.; Steffen, L.; Siscovick, D.; Tsai, M.; Herrington, D. Associations of dietary long-chain n-3 polyunsaturated fatty acids and fish with biomarkers of inflammation and endothelial activation (from the Multi-Ethnic Study of Atherosclerosis [MESA]). Am. J. Cardiol. 2009, 103, 1238–1243. [Google Scholar] [CrossRef]
- Ferrucci, L.; Cherubini, A.; Bandinelli, S.; Bartali, B.; Corsi, A.; Lauretani, F.; Martin, A.; Andres-Lacueva, C.; Senin, U.; Guralnik, J.M. Relationship of plasma polyunsaturated fatty acids to circulating inflammatory markers. J. Clin. Endocrinol. Metab. 2006, 91, 439–446. [Google Scholar] [CrossRef]
- Garcia-Escobar, E.; Rodriguez-Pacheco, F.; Garcia-Serrano, S.; Gomez-Zumaquero, J.M.; Haro-Mora, J.J.; Soriguer, F.; Rojo-Martinez, G. Nutritional regulation of interleukin-6 release from adipocytes. Int. J. Obes. 2010, 34, 1328–1332. [Google Scholar] [CrossRef]
- Van Dijk, S.J.; Feskens, E.J.; Bos, M.B.; Hoelen, D.W.; Heijligenberg, R.; Bromhaar, M.G.; de Groot, L.C.; de Vries, J.H.; Muller, M.; Afman, L.A. A saturated fatty acid-rich diet induces an obesity-linked proinflammatory gene expression profile in adipose tissue of subjects at risk of metabolic syndrome. Am. J. Clin. Nutr. 2009, 90, 1656–1664. [Google Scholar] [CrossRef]
- Czernichow, S.; Thomas, D.; Bruckert, E. n-6 Fatty acids and cardiovascular health: A review of the evidence for dietary intake recommendations. Br. J. Nutr. 2010, 104, 788–796. [Google Scholar] [CrossRef]
- Wall, R.; Ross, R.P.; Fitzgerald, G.F.; Stanton, C. Fatty acids from fish: The anti-inflammatory potential of long-chain omega-3 fatty acids. Nutr. Rev. 2010, 68, 280–289. [Google Scholar] [CrossRef]
- Lu, Y.; Feskens, E.J.; Dolle, M.E.; Imholz, S.; Verschuren, W.M.; Muller, M.; Boer, J.M. Dietary n-3 and n-6 polyunsaturated fatty acid intake interacts with FADS1 genetic variation to affect total and HDL-cholesterol concentrations in the Doetinchem Cohort Study. Am. J. Clin. Nutr. 2010, 92, 258–265. [Google Scholar]
- Visscher, P.M.; Brown, M.A.; McCarthy, M.I.; Yang, J. Five years of GWAS discovery. Am. J. Hum. Genet. 2012, 90, 7–24. [Google Scholar] [CrossRef]
- Genome-wide association study of 14,000 cases of seven common diseases and 3,000 shared controls. Nature 2007, 447, 661–678.
- Pirruccello, J.; Kathiresan, S. Genetics of lipid disorders. Curr. Opin. Cardiol. 2010, 25, 238–242. [Google Scholar] [CrossRef]
- Liu, C.-T.; Monda, K.L.; Taylor, K.C.; Lange, L.; Demerath, E.W.; Palmas, W.; Wojczynski, M.K.; Ellis, J.C.; Vitolins, M.Z.; Liu, S.; et al. Genome-Wide Association of Body Fat Distribution in African Ancestry Populations Suggests New Loci. PLoS Genet. 2013, 9, e1003681. [Google Scholar]
- Kang, S.J.; Chiang, C.W.; Palmer, C.D.; Tayo, B.O.; Lettre, G.; Butler, J.L.; Hackett, R.; Adeyemo, A.A.; Guiducci, C.; Berzins, I.; et al. Genome-wide association of anthropometric traits in African- and African-derived populations. Hum. Mol. Genet. 2010, 19, 2725–2738. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Joffe, Y.T.; Van der Merwe, L.; Evans, J.; Collins, M.; Lambert, E.V.; September, A.V.; Goedecke, J.H. Interleukin-6 Gene Polymorphisms, Dietary Fat Intake, Obesity and Serum Lipid Concentrations in Black and White South African Women. Nutrients 2014, 6, 2436-2465. https://doi.org/10.3390/nu6062436
Joffe YT, Van der Merwe L, Evans J, Collins M, Lambert EV, September AV, Goedecke JH. Interleukin-6 Gene Polymorphisms, Dietary Fat Intake, Obesity and Serum Lipid Concentrations in Black and White South African Women. Nutrients. 2014; 6(6):2436-2465. https://doi.org/10.3390/nu6062436
Chicago/Turabian StyleJoffe, Yael T., Lize Van der Merwe, Juliet Evans, Malcolm Collins, Estelle V. Lambert, Alison V. September, and Julia H. Goedecke. 2014. "Interleukin-6 Gene Polymorphisms, Dietary Fat Intake, Obesity and Serum Lipid Concentrations in Black and White South African Women" Nutrients 6, no. 6: 2436-2465. https://doi.org/10.3390/nu6062436
APA StyleJoffe, Y. T., Van der Merwe, L., Evans, J., Collins, M., Lambert, E. V., September, A. V., & Goedecke, J. H. (2014). Interleukin-6 Gene Polymorphisms, Dietary Fat Intake, Obesity and Serum Lipid Concentrations in Black and White South African Women. Nutrients, 6(6), 2436-2465. https://doi.org/10.3390/nu6062436




