Selected Nutrients and Their Implications for Health and Disease across the Lifespan: A Roadmap
Abstract
:1. Introduction
2. Health Economics of Malnutrition
2.1. Measuring the Impact of Malnutrition
2.2. The Global Impact of Inadequate Micronutrient Intake
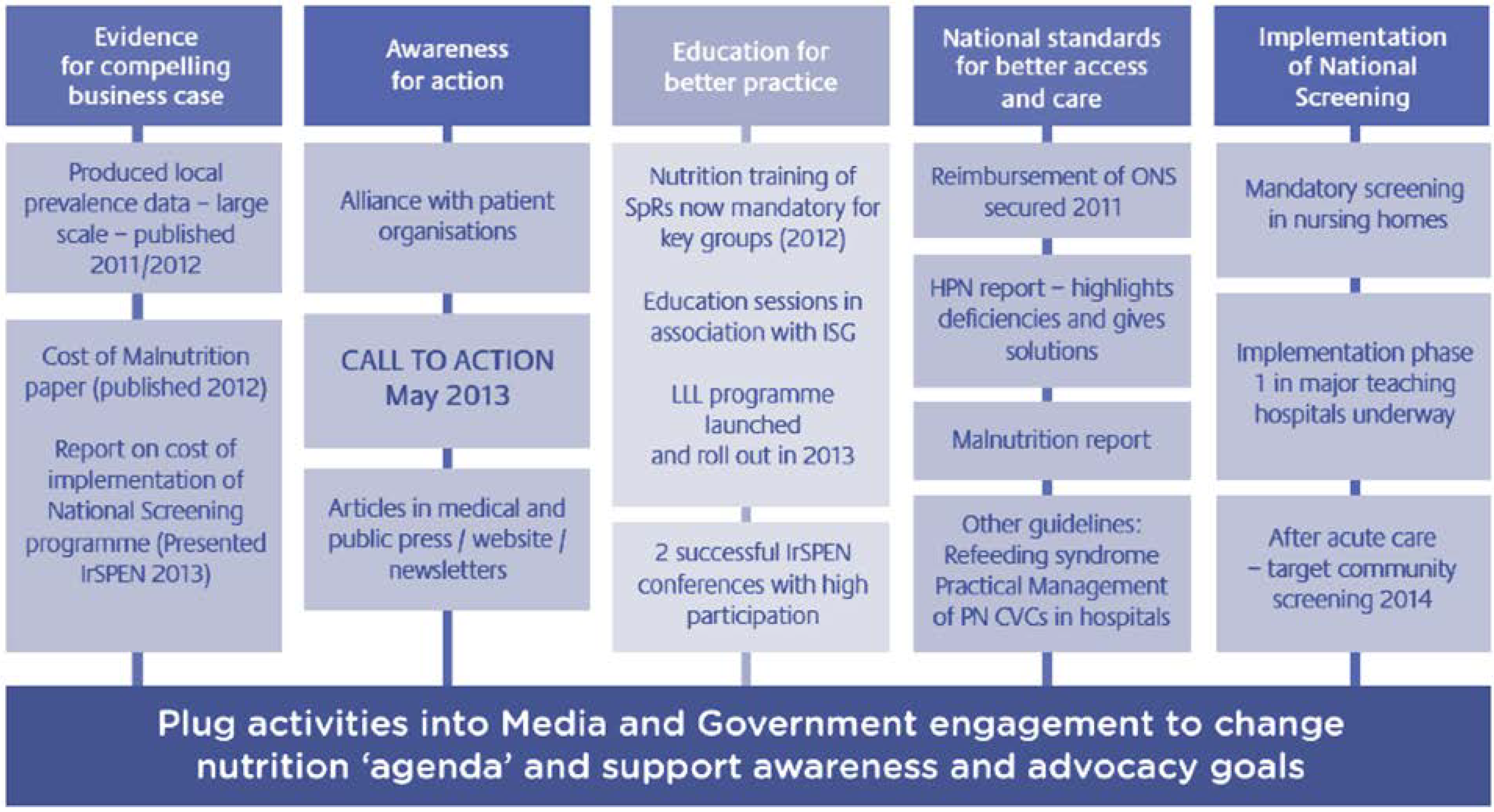
3. Healthy Ageing during Life Course
3.1. Translating Scientific Knowledge into Public Health Solutions
3.2. The Importance of Micronutrient Intake in the First 1000 Days of Life
3.3. Undernutrition in the Elderly
4. Established Evidence of Health and Economic Benefits of Selected Nutrients
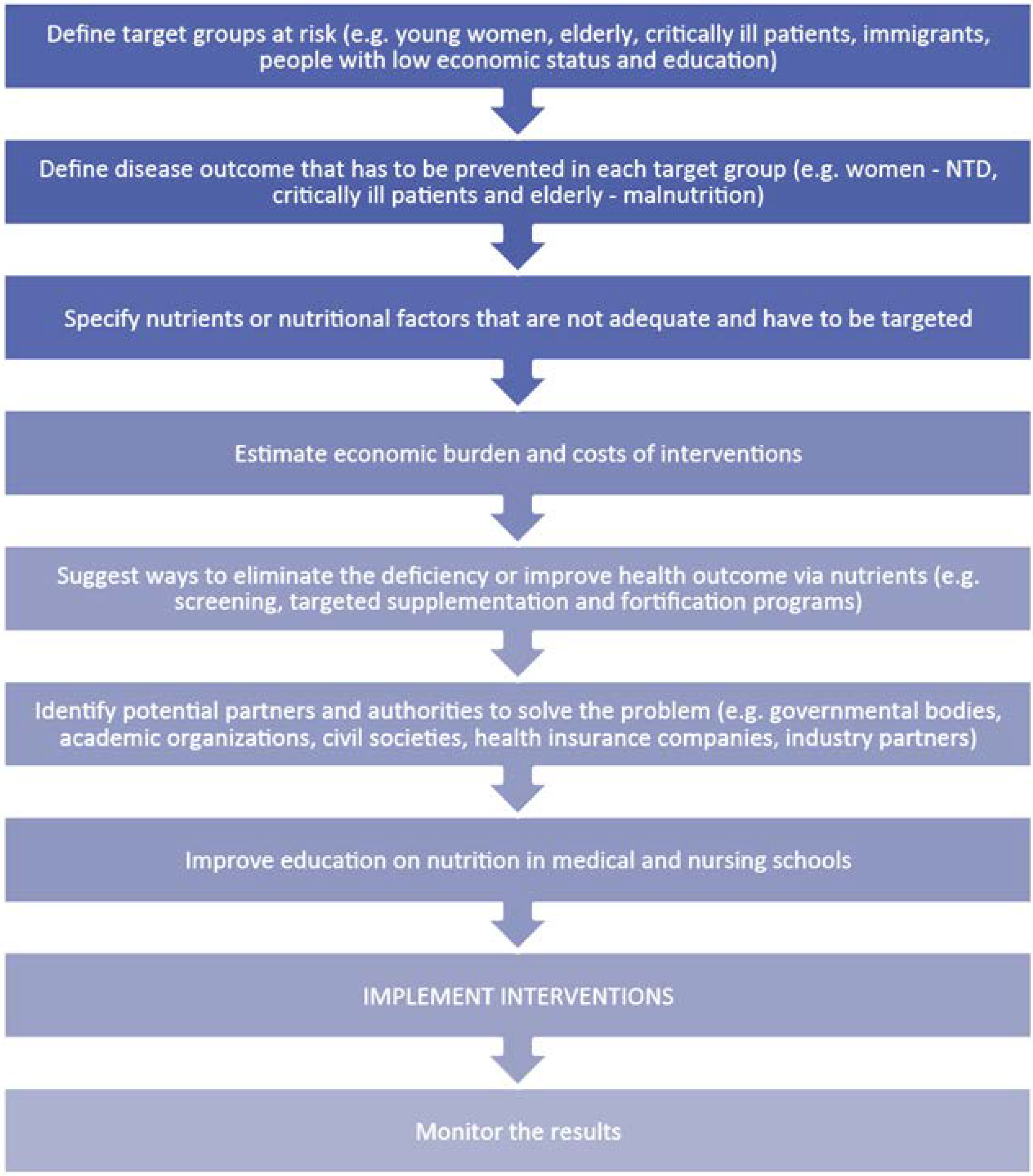
5. Pertinent Considerations in Providing Nutritional Solutions
5.1. Disease-Related Malnutrition and the Economic Value of Medical Nutrition
5.2. Fortification of Affordable Products for Emerging Economies
6. Conclusions and Pending Issues to Reduce Health Care Costs
Acknowledgements
Author Contributions
Conflicts of Interest
References
- World Health Organization. Global Age-Friendly Cities; World Health Organisation: Geneva, Switzerland, 2007; pp. 1–76. [Google Scholar]
- Lagiewka, K. European Innovation Partnership on Active and Healthy Ageing: Triggers of setting the headline target of 2 additional healthy life years at birth at EU average by 2020. Arch. Public. Health. 2012, 70. [Google Scholar] [CrossRef]
- Roman Vinas, B.; Ribas Barba, L.; Ngo, J.; Gurinovic, M.; Novakovic, R.; Cavelaars, A.; de Groot, L.C.; van’t Veer, P.; Matthys, C.; Serra Majem, L. Projected prevalence of inadequate nutrient intakes in Europe. Ann. Nutr. Metab. 2011, 59, 84–95. [Google Scholar]
- Lochs, H.; Allison, S.P.; Meier, R.; Pirlich, M.; Kondrup, J.; Schneider, S.; van den Berghe, G.; Pichard, C. Introductory to the ESPEN guidelines on enteral nutrition: Terminology, definitions and general topics. Clin. Nutr. 2006, 25, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Fight Malnutrition. Available online: http://www.fightmalnutrition.eu/malnutrition/general-information/ (accessed on 14 November 2014).
- National Collaborating Centre for Acute Care. Nutrition Support in Adults: Oral Nutrition Support, Enteral Tube Feeding and Parenteral Nutrition; National Collaborating Centre for Acute Care: London, UK, 2006; pp. 1–175. [Google Scholar]
- Rabovskaja, V.; Parkinson, B.; Goodall, S. The cost-effectiveness of mandatory folic acid fortification in Australia. J. Nutr. 2013, 143, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Shanahan, C.; de Lorimier, R. Smart Prevention—Health Care Cost Savings Resulting from the Targeted Use of Dietary Supplements; Frost & Sullivan: Mountain View, CA, USA, 2013; pp. 1–125. [Google Scholar]
- Fighting the Malnutrition Battle: The Power of Partnerships. Available online: http://www.espen.org/files/IRSPEN_entry_MNI_grant_2013_poster.pdf (accessed on 14 November 2014).
- Koletzko, B.; Symonds, M.E.; Olsen, S.F. Programming research: Where are we and where do we go from here? Am. J. Clin. Nutr. 2011, 94, 2036–2043. [Google Scholar] [CrossRef]
- Koletzko, B. Developmental origins of adult disease: Barker’s Or Dorner’s hypothesis? Am. J. Hum. Biol. 2005, 17, 381–382. [Google Scholar] [CrossRef] [PubMed]
- Aghajafari, F.; Nagulesapillai, T.; Ronksley, P.E.; Tough, S.C.; O’Beirne, M.; Rabi, D.M. Association between maternal serum 25-hydroxyvitamin D level and pregnancy and neonatal outcomes: Systematic review and meta-analysis of observational studies. BMJ 2013, 346. [Google Scholar] [CrossRef] [PubMed]
- Suren, P.; Roth, C.; Bresnahan, M.; Haugen, M.; Hornig, M.; Hirtz, D.; Lie, K.K.; Lipkin, W.I.; Magnus, P.; Reichborn-Kjennerud, T.; et al. Association between maternal use of folic acid supplements and risk of autism spectrum disorders in children. JAMA 2013, 309, 570–577. [Google Scholar] [CrossRef]
- Walker, S.P.; Wachs, T.D.; Gardner, J.M.; Lozoff, B.; Wasserman, G.A.; Pollitt, E.; Carter, J.A. Child development: Risk factors for adverse outcomes in developing countries. Lancet 2007, 369, 145–157. [Google Scholar] [CrossRef] [PubMed]
- Lozoff, B.; Jimenez, E.; Hagen, J.; Mollen, E.; Wolf, A.W. Poorer behavioral and developmental outcome more than 10 years after treatment for iron deficiency in infancy. Pediatrics 2000, 105, 51. [Google Scholar] [CrossRef]
- Commission Regulation (EU) No. 440/2011. Available online: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2011:119:0004:0009:en:PDF (accessed on 14 November 2014).
- Hibbeln, J.R.; Davis, J.M.; Steer, C.; Emmett, P.; Rogers, I.; Williams, C.; Golding, J. Maternal seafood consumption in pregnancy and neurodevelopmental outcomes in childhood (ALSPAC study): An observational cohort study. Lancet 2007, 369, 578–585. [Google Scholar] [CrossRef] [PubMed]
- Kohlboeck, G.; Glaser, C.; Tiesler, C.; Demmelmair, H.; Standl, M.; Romanos, M.; Koletzko, B.; Lehmann, I.; Heinrich, J.; Group, L.I.S. Effect of fatty acid status in cord blood serum on children’s behavioral difficulties at 10 y of age: Results from the LISAplus study. Am. J. Clin. Nutr. 2011, 94, 1592–1599. [Google Scholar] [CrossRef] [PubMed]
- Krauss-Etschmann, S.; Hartl, D.; Rzehak, P.; Heinrich, J.; Shadid, R.; Del Carmen Ramirez-Tortosa, M.; Campoy, C.; Pardillo, S.; Schendel, D.J.; Decsi, T.; et al. Decreased cord blood IL-4, IL-13, and CCR4 and increased TGF-Beta levels after fish oil supplementation of pregnant women. J. Allergy Clin. Immunol. 2008, 121, 464–470. [Google Scholar] [CrossRef]
- Imhoff-Kunsch, B.; Stein, A.D.; Martorell, R.; Parra-Cabrera, S.; Romieu, I.; Ramakrishnan, U. Prenatal docosahexaenoic acid supplementation and infant morbidity: Randomized controlled trial. Pediatrics 2011, 128, 505–512. [Google Scholar]
- Agostoni, C.; Braegger, C.; Decsi, T.; Kolacek, S.; Koletzko, B.; Michaelsen, K.F.; Mihatsch, W.; Moreno, L.A.; Puntis, J.; Shamir, R.; et al. Breast-feeding: A commentary by the ESPGHAN committee on nutrition. J. Pediatr. Gastroenterol. Nutr. 2009, 49, 112–125. [Google Scholar] [CrossRef] [PubMed]
- Straub, N.; Grunert, P.; von Kries, R.; Koletzko, B. Health economic potential of early nutrition programming: A model calculation of long-term reduction in blood pressure and related morbidity costs by use of long-chain polyunsaturated fatty acid-supplemented formula. Am. J. Clin. Nutr. 2011, 94, 2030–2035. [Google Scholar] [CrossRef]
- Symonds, M.E.; Mendez, M.A.; Meltzer, H.M.; Koletzko, B.; Godfrey, K.; Forsyth, S.; van der Beek, E.M. Early life nutritional programming of obesity: Mother-child cohort studies. Ann. Nutr. Metab. 2013, 62, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Krebs-Smith, S.M.; Guenther, P.M.; Subar, A.F.; Kirkpatrick, S.I.; Dodd, K.W. Americans Do not meet federal dietary recommendations. J. Nutr. 2010, 140, 1832–1838. [Google Scholar] [CrossRef] [PubMed]
- Schilp, J.; Kruizenga, H.M.; Wijnhoven, H.A.; Leistra, E.; Evers, A.M.; van Binsbergen, J.J.; Deeg, D.J.; Visser, M. High prevalence of undernutrition in Dutch community-dwelling older individuals. Nutrition 2012, 28, 1151–1156. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, M.J.; Bauer, J.M.; Ramsch, C.; Uter, W.; Guigoz, Y.; Cederholm, T.; Thomas, D.R.; Anthony, P.S.; Charlton, K.E.; Maggio, M.; et al. Frequency of malnutrition in older adults: A multinational perspective using the mini nutritional assessment. J. Am. Geriatr. Soc. 2010, 58, 1734–1738. [Google Scholar] [CrossRef]
- Schilling, S. Epidemic vitamin D deficiency among patients in an elderly care rehabilitation facility. Deutsch. Arztebl. Int. 2012, 109, 33–38. [Google Scholar]
- Lim, S.L.; Ong, K.C.; Chan, Y.H.; Loke, W.C.; Ferguson, M.; Daniels, L. Malnutrition and its impact on cost of hospitalization, length of stay, readmission and 3-year mortality. Clin. Nutr. 2012, 31, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Humphreys, J.; de la Maza, P.; Hirsch, S.; Barrera, G.; Gattas, V.; Bunout, D. Muscle strength as a predictor of loss of functional status in hospitalized patients. Nutrition 2002, 18, 616–620. [Google Scholar] [CrossRef] [PubMed]
- BAPEN: Malnutrition Matters. Available online: http://www.bapen.org.uk/professionals/publications-and-resources/bapen-reports/combating-malnutrition-recommendations-for-action/20-professionals/publications-and-resources/174-malnutrition-among-older-people-in-the-community (accessed on 14 November 2014).
- Montero-Fernandez, N.; Serra-Rexach, J.A. Role of exercise on Sarcopenia in the elderly. Eur. J. Phys. Rehabil. Med. 2013, 49, 131–143. [Google Scholar] [PubMed]
- Stratton, R.J.; King, C.L.; Stroud, M.A.; Jackson, A.A.; Elia, M. ‘Malnutrition Universal Screening Tool’ predicts mortality and length of hospital stay in acutely ill elderly. Br. J. Nutr. 2006, 95, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Ensrud, K.E.; Ewing, S.K.; Stone, K.L.; Cauley, J.A.; Bowman, P.J.; Cummings, S.R. Intentional and unintentional weight loss increase bone loss and hip fracture risk in older women. J. Am. Geriatr. Soc. 2003, 51, 1740–1747. [Google Scholar] [CrossRef] [PubMed]
- Neelemaat, F.; Lips, P.; Bosmans, J.E.; Thijs, A.; Seidell, J.C.; van Bokhorst-de van der Schueren, M.A. Short-term oral nutritional intervention with protein and vitamin D decreases falls in malnourished older adults. J. Am. Geriatr. Soc. 2012, 60, 691–699. [Google Scholar] [CrossRef] [PubMed]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, 146–156. [Google Scholar] [CrossRef]
- Freijer, K.; Tan, S.S.; Koopmanschap, M.A.; Meijers, J.M.; Halfens, R.J.; Nuijten, M.J. The economic costs of disease related malnutrition. Clin. Nutr. 2013, 32, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Fry, D.E.; Pine, M.; Jones, B.L.; Meimban, R.J. Patient characteristics and the occurrence of never events. Arch. Surg. 2010, 145, 148–151. [Google Scholar] [CrossRef] [PubMed]
- Ellis, G.; Whitehead, M.A.; Robinson, D.; O’Neill, D.; Langhorne, P. Comprehensive geriatric assessment for older adults admitted to hospital: Meta-analysis of randomised controlled trials. BMJ 2011, 343. [Google Scholar] [CrossRef]
- Inouye, S.K.; Bogardus, S.T., Jr.; Charpentier, P.A.; Leo-Summers, L.; Acampora, D.; Holford, T.R.; Cooney, L.M., Jr. A multicomponent intervention to prevent delirium in hospitalized older patients. N. Engl. J. Med. 1999, 340, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Van Asselt, D.Z.; van Bokhorst-de van der Schueren, M.A.; van der Cammen, T.J.; Disselhorst, L.G.; Janse, A.; Lonterman-Monasch, S.; Maas, H.A.; Popescu, M.E.; Scholzel-Dorenbos, C.J.; Sipers, W.M.; et al. Assessment and treatment of malnutrition in Dutch geriatric practice: Consensus through a modified Delphi study. Age Ageing 2012, 41, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Troesch, B.; Eggersdorfer, M.; Weber, P. 100 years of vitamins: Adequate intake in the elderly is still a matter of concern. J. Nutr. 2012, 142, 979–980. [Google Scholar] [CrossRef] [PubMed]
- Blencowe, H.; Cousens, S.; Modell, B.; Lawn, J. Folic acid to reduce neonatal mortality from neural tube disorders. Int. J. Epidemiol. 2010, 39 (Suppl. 1), 110–121. [Google Scholar]
- Wald, N.J.; Law, M.R.; Morris, J.K.; Wald, D.S. Quantifying the effect of folic acid. Lancet 2001, 358, 2069–2073. [Google Scholar] [CrossRef] [PubMed]
- Van Beynum, I.M.; Kapusta, L.; Bakker, M.K.; den Heijer, M.; Blom, H.J.; de Walle, H.E. Protective effect of periconceptional folic acid supplements on the risk of congenital heart defects: A registry-based case-control study in the Northern Netherlands. Eur. Heart J. 2010, 31, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Kelly, Y.; Sacker, A.; Marmot, M. Nutrition and respiratory health in adults: Findings from the health survey for Scotland. Eur. Respir. J. 2003, 21, 664–671. [Google Scholar] [CrossRef] [PubMed]
- Fekete, K.; Berti, C.; Trovato, M.; Lohner, S.; Dullemeijer, C.; Souverein, O.W.; Cetin, I.; Decsi, T. Effect of folate intake on health outcomes in pregnancy: A systematic review and meta-analysis on birth weight, placental weight and length of gestation. Nutr. J. 2012, 11. [Google Scholar] [CrossRef]
- Czeizel, A.E.; Puho, E.H.; Langmar, Z.; Acs, N.; Banhidy, F. Possible association of folic acid supplementation during pregnancy with reduction of preterm birth: A population-based study. Eur. J. Obstet. Gynecol. Reprod. Biol. 2010, 148, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.C.; Liu, N.; Wen, S.W.; Walker, M. Why do Canadian women fail to achieve optimal pre-conceptional folic acid supplementation? An observational study. J. Obstet. Gynaecol. Can. 2011, 33, 1116–1123. [Google Scholar] [PubMed]
- Pfeiffer, C.M.; Johnson, C.L.; Jain, R.B.; Yetley, E.A.; Picciano, M.F.; Rader, J.I.; Fisher, K.D.; Mulinare, J.; Osterloh, J.D. Trends in blood folate and vitamin B-12 concentrations in the United States, 1988–2004. Am. J. Clin. Nutr. 2007, 86, 718–727. [Google Scholar] [PubMed]
- Yi, Y.; Lindemann, M.; Colligs, A.; Snowball, C. Economic burden of neural tube defects and impact of prevention with folic acid: A literature review. Eur. J. Pediatr. 2011, 170, 1391–1400. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.; Cui, Y.; Shen, L.; Sun, N.; Zhang, Y.; Li, J.; Xu, X.; Wang, B.; Xu, X.; Huo, Y.; et al. Folic acid supplementation and cancer risk: A meta-analysis of randomized controlled trials. Int. J. Cancer 2013, 133, 1033–1041. [Google Scholar] [CrossRef] [PubMed]
- Vollset, S.E.; Clarke, R.; Lewington, S.; Ebbing, M.; Halsey, J.; Lonn, E.; Armitage, J.; Manson, J.E.; Hankey, G.J.; Spence, J.D.; et al. Effects of folic acid supplementation on overall and site-specific cancer incidence during the randomised trials: Meta-analyses of data on 50,000 individuals. Lancet 2013, 381, 1029–1036. [Google Scholar] [CrossRef] [PubMed]
- Lökk, J. News and views on folate and elderly persons. J. Gerontol. A. Biol. Sci. Med. Sci. 2003, 58, 354–361. [Google Scholar] [CrossRef] [PubMed]
- FFI Global Progress. Available online: http://www.ffinetwork.org/global_progress/index.php (accessed on 14 November 2014).
- Flynn, A.; Hirvonen, T.; Mensink, G.B.; Ocke, M.C.; Serra-Majem, L.; Stos, K.; Szponar, L.; Tetens, I.; Turrini, A.; Fletcher, R.; et al. Intake of selected nutrients from foods, from fortification and from supplements in various European countries. Food Nutr. Res. 2009, 53. [Google Scholar] [CrossRef]
- Obeid, R.; Koletzko, B.; Pietrzik, K. Critical evaluation of lowering the recommended dietary intake of folate. Clin. Nutr. 2014, 33, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Zittermann, A.; Gummert, J.F. Nonclassical vitamin D action. Nutrients 2010, 2, 408–425. [Google Scholar] [CrossRef] [PubMed]
- Rizzoli, R.; Boonen, S.; Brandi, M.L.; Bruyere, O.; Cooper, C.; Kanis, J.A.; Kaufman, J.M.; Ringe, J.D.; Weryha, G.; Reginster, J.Y. Vitamin D supplementation in elderly or postmenopausal women: A 2013 Update of the 2008 recommendations from the European society for clinical and economic aspects of osteoporosis and osteoarthritis (ESCEO). Curr. Med. Res. Opin. 2013, 29, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Zittermann, A.; Iodice, S.; Pilz, S.; Grant, W.B.; Bagnardi, V.; Gandini, S. Vitamin D deficiency and mortality risk in the general population: A meta-analysis of prospective cohort studies. Am. J. Clin. Nutr. 2012, 95, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Lotters, F.J.; Lenoir-Wijnkoop, I.; Fardellone, P.; Rizzoli, R.; Rocher, E.; Poley, M.J. Dairy foods and osteoporosis: An example of assessing the health-economic impact of food products. Osteoporos. Int. 2013, 24, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Ljungqvist, O.; Man, F. Under nutrition: A major health problem in Europe. Nutr. Hosp. 2009, 24, 369–370. [Google Scholar] [PubMed]
- Freijer, K.; Nuijten, M.J. Analysis of the health economic impact of medical nutrition in the Netherlands. Eur. J. Clin. Nutr. 2010, 64, 1229–1234. [Google Scholar] [CrossRef] [PubMed]
- Freijer, K.; Nuijten, M.J.; Schols, J.M. The budget impact of oral nutritional supplements for disease related malnutrition in elderly in the community setting. Front. Pharmacol. 2012, 3. [Google Scholar] [CrossRef]
- Lassen, K.O.; Olsen, J.; Grinderslev, E.; Kruse, F.; Bjerrum, M. Nutritional care of medical inpatients: A health technology assessment. BMC Health Serv. Res. 2006, 6. [Google Scholar] [CrossRef]
- Philipson, T.J.; Snider, J.T.; Lakdawalla, D.N.; Stryckman, B.; Goldman, D.P. Impact of oral nutritional supplementation on hospital outcomes. Am. J. Manag. Care 2013, 19, 121–128. [Google Scholar] [PubMed]
- Freijer, K.; Bours, M.J.; Nuijten, M.J.; Poley, M.J.; Meijers, J.M.; Halfens, R.J.; Schols, J.M. The economic value of enteral medical nutrition in the management of disease-related malnutrition: A systematic review. J. Am. Med. Dir. Assoc. 2014, 15, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Milte, R.K.; Ratcliffe, J.; Miller, M.D.; Crotty, M. Economic evaluation for protein and energy supplementation in adults: Opportunities to strengthen the evidence. Eur. J. Clin. Nutr. 2013, 67, 1243–1250. [Google Scholar] [CrossRef] [PubMed]
- Eichler, K.; Wieser, S.; Ruthemann, I.; Brugger, U. Effects of micronutrient fortified milk and cereal food for infants and children: A systematic review. BMC Public Health 2012, 12. [Google Scholar] [CrossRef] [PubMed]
- Medical Nutrition International Industry. Available online: http://www.medicalnutritionindustry.com/mni-publications/oral-nutritional-supplements-to-tackle-malnutriton-a-summary-of-the-evidence-base/ (accessed on 14 November 2014).
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Péter, S.; Eggersdorfer, M.; Van Asselt, D.; Buskens, E.; Detzel, P.; Freijer, K.; Koletzko, B.; Kraemer, K.; Kuipers, F.; Neufeld, L.; et al. Selected Nutrients and Their Implications for Health and Disease across the Lifespan: A Roadmap. Nutrients 2014, 6, 6076-6094. https://doi.org/10.3390/nu6126076
Péter S, Eggersdorfer M, Van Asselt D, Buskens E, Detzel P, Freijer K, Koletzko B, Kraemer K, Kuipers F, Neufeld L, et al. Selected Nutrients and Their Implications for Health and Disease across the Lifespan: A Roadmap. Nutrients. 2014; 6(12):6076-6094. https://doi.org/10.3390/nu6126076
Chicago/Turabian StylePéter, Szabolcs, Manfred Eggersdorfer, Dieneke Van Asselt, Erik Buskens, Patrick Detzel, Karen Freijer, Berthold Koletzko, Klaus Kraemer, Folkert Kuipers, Lynnette Neufeld, and et al. 2014. "Selected Nutrients and Their Implications for Health and Disease across the Lifespan: A Roadmap" Nutrients 6, no. 12: 6076-6094. https://doi.org/10.3390/nu6126076
APA StylePéter, S., Eggersdorfer, M., Van Asselt, D., Buskens, E., Detzel, P., Freijer, K., Koletzko, B., Kraemer, K., Kuipers, F., Neufeld, L., Obeid, R., Wieser, S., Zittermann, A., & Weber, P. (2014). Selected Nutrients and Their Implications for Health and Disease across the Lifespan: A Roadmap. Nutrients, 6(12), 6076-6094. https://doi.org/10.3390/nu6126076





