2.1. Materials and Chemicals
The soybean (SB) fermented paste and brown rice were purchased from a local market in Daegu, Korea. The rice bran was provided by Rice Processing Complex (RPC, Gimcheon, Korea), while the red ginseng marc was obtained from Punggi Ginseng Farming Corporation (YEongju, Gyeongbuk, Korea). The Aspergillus oryzae was procured from NUC Electronics Co., Ltd. (Daegu, Korea). The chemicals such as ethanol, ketamine-HCl, potassium phosphate buffer, trichloroacetic acid, and thiobarbituric acid were purchased from Merck KGaA (Darmstadt, Germany). All other chemicals used were obtained from Sigma-Aldrich, Inc. (Steinhein, Germany).
2.2. Preparation of Brown Rice Fermented Paste Samples
The brown rice (BR) fermented paste was prepared according to the manufacturing process for commercial fermented paste with some modifications [
17]. Briefly, the rice grains were washed, soaked in water for 3 h at 4 °C, and cooked using an electric rice cooker. After cooling, the cooked rice was inoculated with
A. oryzae (0.2%, w/w) and incubated for 3 days at 30 °C. Salt was added to the resulting koji (12%, w/w), mixed using a food processor, and fermented and ripened for 30 days at 30 °C. The same method was used for the preparation of the brown rice-rice bran (BRB) and brown rice-red ginseng marc (BRG) fermented paste samples. The rice bran and red ginseng marc were washed and steamed separately for 40 min. Prior to inoculation with
A. oryzae, the cooked brown rice was added with rice bran (20%, w/w) or red ginseng (30%, w/w). All fermented paste samples were freeze-dried at −70 °C prior to use. Their proximate compositions are presented in
Table 1.
Table 1.
Proximate composition (% dry basis) of the fermented paste samples.
Table 1.
Proximate composition (% dry basis) of the fermented paste samples.
| Composition | Fermented Paste (1) |
|---|
| SB | BR | BRB | BRG |
|---|
| Moisture | 4.52 | 4.91 | 4.96 | 5.23 |
| Carbohydrate | 45.55 | 74.72 | 68.77 | 70.79 |
| Crude protein | 23.42 | 7.82 | 9.45 | 8.15 |
| Crude fat | 0.91 | 0.41 | 1.13 | 0.16 |
| Dietary fiber | 4.36 | 2.47 | 4.16 | 3.04 |
| Ash | 21.24 | 9.67 | 11.53 | 12.63 |
2.3. Animals and Diets
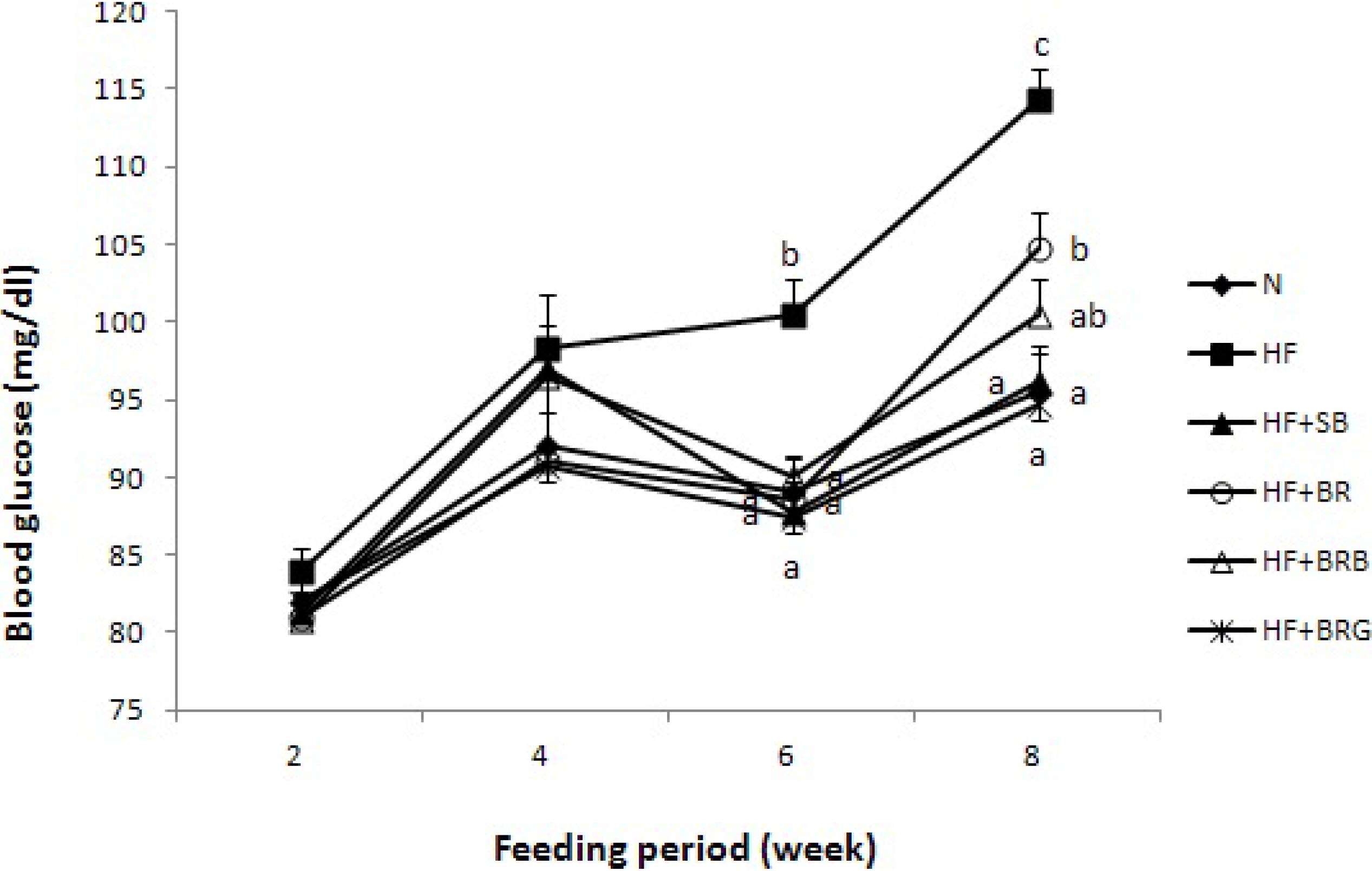
Forty-eight male C57BL/6N mice (4-week-old), weighing 12 g, were obtained from Orient Inc. (Seoul, Korea). Each mouse was housed in a stainless steel cage in a room maintained at 25 °C with 50% relative humidity and 12/12 h light/dark cycle. Upon arrival, the animals were fed with pelletized chow diet for 2 weeks. They were then randomly divided into 6 dietary groups (
n = 8). The first and second groups were fed with normal control (NC group) and high fat (20%, w/w, HF group) diets, respectively. The other 4 groups were fed with a high fat diet supplemented with soybean fermented paste (HF + SB group), brown rice fermented paste (HF + BR group), brown rice-rice bran fermented paste (HF + BRB group), or brown rice-red ginseng marc fermented paste (HF + BRG group). The composition of the experimental diet (
Table 2) was based on the AIN-76 semisynthetic diet [
18]. The diets for all animal groups, except for the NC group, were added with 17% (w/w) lard to make the diet high fat. The mice were fed for eight weeks and allowed free access to food and water. The feed intake and weight gain were measured daily and weekly, respectively. At the end of the experimental period, the mice were anaesthetized with ketamine-HCl following a 12-h fast. The blood samples were drawn from the inferior vena cava into a heparin-coated tube and centrifuged at 1000×
g for 15 min at 4 °C to obtain the plasma and erythrocyte. The plasma and buffy coat were removed after centrifugation and the erythrocytes were washed with physiological saline, followed by hemolysis with distilled water [
19]. The concentration of hemoglobin was determined using a commercial assay kit (Asan Pharmaceutical, Seoul, Korea). The liver and adipose tissues (epididymal, perirenal, and inguinal) were removed, rinsed with physiological saline, weighed, and stored at −70 °C until analysis. The current study protocol was approved by the Ethics Committee of Kyungpook National University for animal studies.
Table 2.
Composition of the experimental diets (% w/w).
Table 2.
Composition of the experimental diets (% w/w).
| Component | NC (1) | HF | HF + SB | HF + BR | HF + BRB | HF + BRG |
|---|
| Casein | 20 | 20 | 17.58 | 19.12 | 18.96 | 19.09 |
| dl-Methionine | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 |
| Sucrose | 50 | 50 | 45.24 | 42.32 | 42.91 | 42.70 |
| Corn starch | 15 | - | - | - | - | - |
| Cellulose | 5 | 5 | 4.54 | 4.73 | 4.56 | 4.67 |
| Corn oil | 5 | 3 | 3 | 3 | 3 | 3 |
| Cholinbitartrate | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 |
| Mineral mixture (2) | 3.5 | 3.5 | 1.37 | 2.52 | 2.34 | 2.23 |
| Vitamin mixture (3) | 1 | 1 | 1 | 1 | 1 | 1 |
| Lard | - | 17 | 16.83 | 16.88 | 16.80 | 16.90 |
| SB | - | - | 10 | - | - | - |
| BR | - | - | - | 10 | - | - |
| BRB | - | - | - | - | 10 | - |
| BRG | - | - | - | - | - | 10 |
| Total (%) | 100 | 100 | 100 | 100 | 100 | 100 |
2.5. Determination of Hepatic Glycogen and Plasma Insulin Levels
The concentration of glycogen was determined according to the method described by Seifter
et al. [
20]. A 100 mg of fresh liver was mixed with 30% KOH and heated at 100 °C for 30 min. The mixture was added with 1.5 mL ethanol (95%) and kept overnight at 4 °C. The pellet was then mixed with 4 mL distilled water. The mixture (500 µL) was added with 0.2% anthrone (in 95% H
2SO
4) and its absorbance was measured at 620 nm. The results were calculated on the basis of a standard calibration curve of glucose. The insulin concentration was measured using enzyme-linked immunosorbent assay (ELISA) kits (TMB Mouse Insulin ELISA kit, Sibayagi, Japan).
2.7. Measurement of Hepatic Glucose-Regulating Enzyme and Antioxidant Enzyme Activities
The hepatic enzyme source was prepared based on the method of Hulcher and Oleson [
22]. A 0.3 g liver was homogenized in buffer solution (0.1 M triethanolamine, 0.2 M EDTA, and 0.002 M dithiothreitol) and centrifuged at 1000×
g for 15 min at 4 °C. The supernatant was centrifuged at 10,000×
g for 15 min at 4 °C and the resulting precipitate served as the mitochondrial fraction, while the supernatant was further centrifuged at 105,000×
g for 1 h at 4 °C. The resulting supernatant and precipitate were the cytosol and microsome fractions, respectively.
The glucokinase (GK) activity was measured according to the method described by Davidson and Arion [
23] with slight modifications. A 0.98 mL of the reaction mixture (50 mM Hepes-NaOH (pH 7.4), 100 mM KCl, 7.5 mM MgCl
2, 2.5 mM dithioerythritol, 10 mg/mL albumin, 10 mM glucose, 4 units of glucose-6-phosphate dehydrogenase, 50 mM NAD
+, and 10 µL cytosol) was pre-incubated at 37 °C for 10 min. The reaction was initiated with the addition of 10 µL of 5 mM ATP and the mixture was incubated at 37 °C for 10 min. The change in absorbance at 340 nm was recorded.
The glucose-6-phosphatase (G6pase) activity was measured based on the method of Alegre
et al. [
24]. The reaction mixture contained 765 µL of 131.58 mM Hepes-NaOH (pH 6.5), 100 µL of 18 mM EDTA (pH 6.5), 100 µL of 265 mM glucose-6-phosphate, 10 µL of 0.2 M NADP
+, 0.6 IU/mL mutarotase, and 0.6 IU/mL glucose dehydrogenase. After pre-incubation at 37 °C for 3 min, the mixture was added with 5 µL microsome and incubated at 37 °C for 4 min. The change in absorbance at 340 nm was measured.
The phosphoenolpyruvate carboxykinase (PEPCK) activity was measured using the method of Bentle and Lardy [
25]. The reaction mixture contained 72.92 mM sodium Hepes (pH 7.0), 10 mM dithiothreitol, 500 mM NaHCO
3, 10 mM MnCl
2, 25 mM NADH, 100 mM IDP, 200 mM PEP, 7.2 unit of malic dehydrogenase, and 10 µL cytosol. The enzyme activity was determined based from the decrease in the absorbance of the mixture at 340 nm at 25 °C.
The superoxide dismutase (SOD) activity was spectrophotometrically measured according to the method of Marklund and Marklund [
26]. The SOD was detected based on its ability to inhibit superoxide-mediated reduction. The reaction mixture (50 mM Tris-HCl buffer (pH 8.5), 10 mM EDTA, 0.1 mL cytosol or erythrocyte, and 7.2 mM pyrogallol) was incubated at 25 °C for 10 min and added with 50 µL of 1 N HCl. The absorbance was measured at 420 nm and the activity was expressed as unit/mg protein, wherein one unit represents the amount of enzyme that inhibited the oxidation of pyrogallol by 50%. The amount of protein was determined using Bradford protein assay [
27].
The glutathione peroxidase (GPx) activity was measured using the method of Paglia and Valentine [
28] with slight modifications. A 0.1 mL of the cytosolic supernatant or erythrocyte was added to the reaction mixture (6 mM glutathione, 1.2 mM NADPH, and 1.25 µM H
2O
2 in 20mM Tris-HCl, pH 7.0) that was pre-warmed at 25 °C for 5 min. The mixture was further incubated at 25 °C for 5 min and the absorbance was measured at 340 nm. A molar extinction coefficient of 6.22/mM/cm was used to determine the activity, which was expressed as nmol oxidized NADPH /min/mg protein.
The catalase (CAT) activity was measured according to the method of Aebi [
29]. A mixture of 50 mM potassium phosphate buffer (pH 7.4) and 10 µL of mitochondrial fraction or erythrocyte was pre-incubated at 25 °C for 5 min and added with 0.1 mL of 30 mM H
2O
2. The disappearance of hydrogen peroxide was monitored spectrophotometrically at 240 nm for 5 min. A molar extinction coefficient of 0.041/mM/cm was used to determine the CAT activity. The activity was defined as the µmol decreased H
2O
2/min/mg protein.
The glutathione reductase (GR) activity was determined based on the method of Mize and Langdon [
30]. A 10 µL of cytosol or erythrocyte was added to the reaction mixture (1 mM EDTA and 1 mM GSSG in a 0.1 M potassium phosphate buffer, pH 7.4) and the oxidation of NADPH was monitored at 340 nm. The activity was expressed as nmol oxidized NADPH/min/mg protein.
The paraoxonase (PON) activity was measured using the method described by Mackness
et al. [
31]. The microsome or erythrocyte (50 µL) was added to 1 mL Tris/HCl buffer (100 mM, pH 8.0) containing 2 mM CaCl
2 and 5.5 mM paraoxon. The absorbance of the mixture was measured at 412 nm at 25 °C to determine the generation rate of 4-nitrophenol. The enzymatic activity was calculated using the molar extinction coefficient of 17,100/M/cm.