Nutrigenomics of Body Weight Regulation: A Rationale for Careful Dissection of Individual Contributors
Abstract
:1. Introduction: Obesity and Body Weight Loss
2. Energy Balance
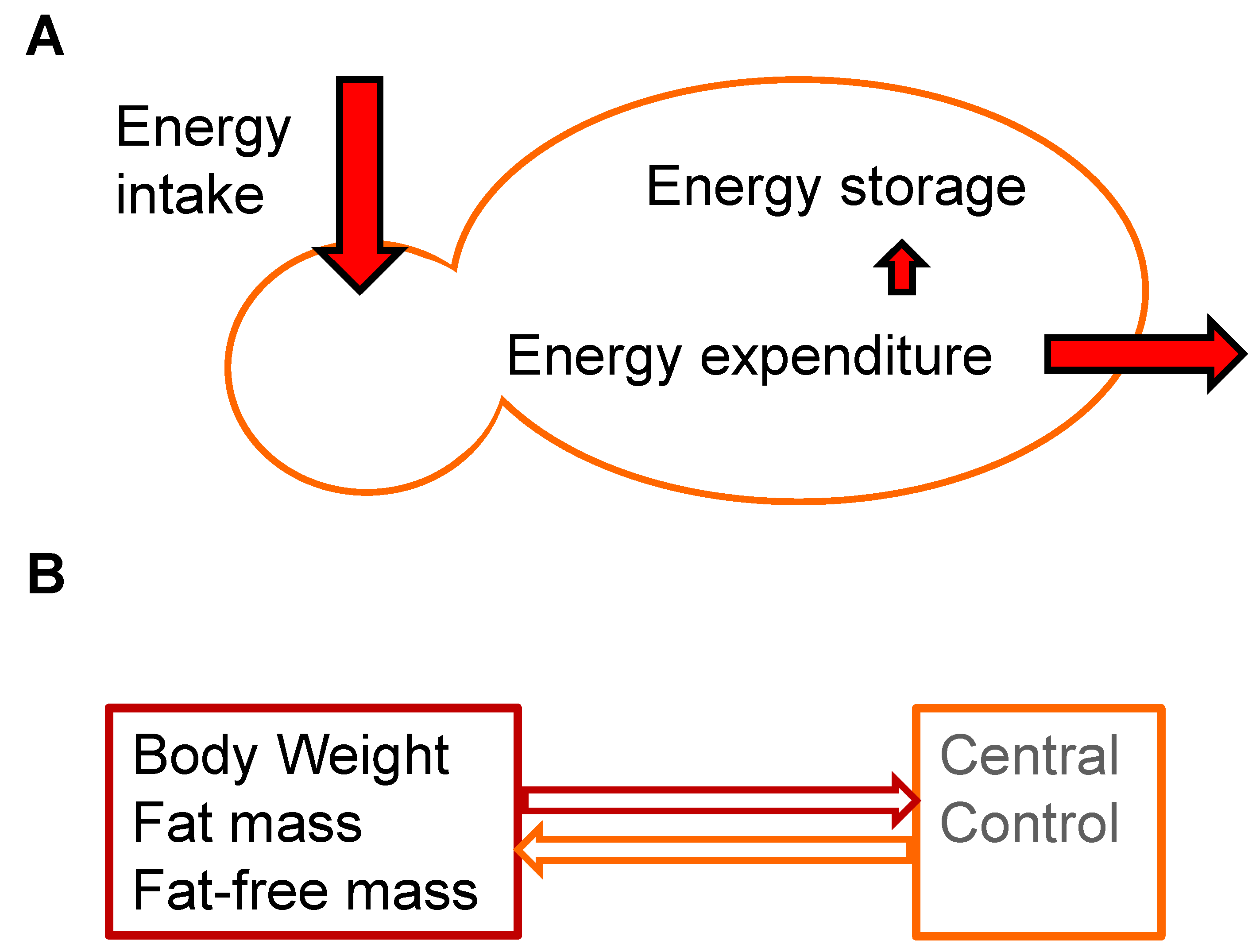
3. Contributors to Body Weight Gain
4. Setpoint
5. Settling Point
6. The Rise in Obesity Questions a Body Weight Setpoint
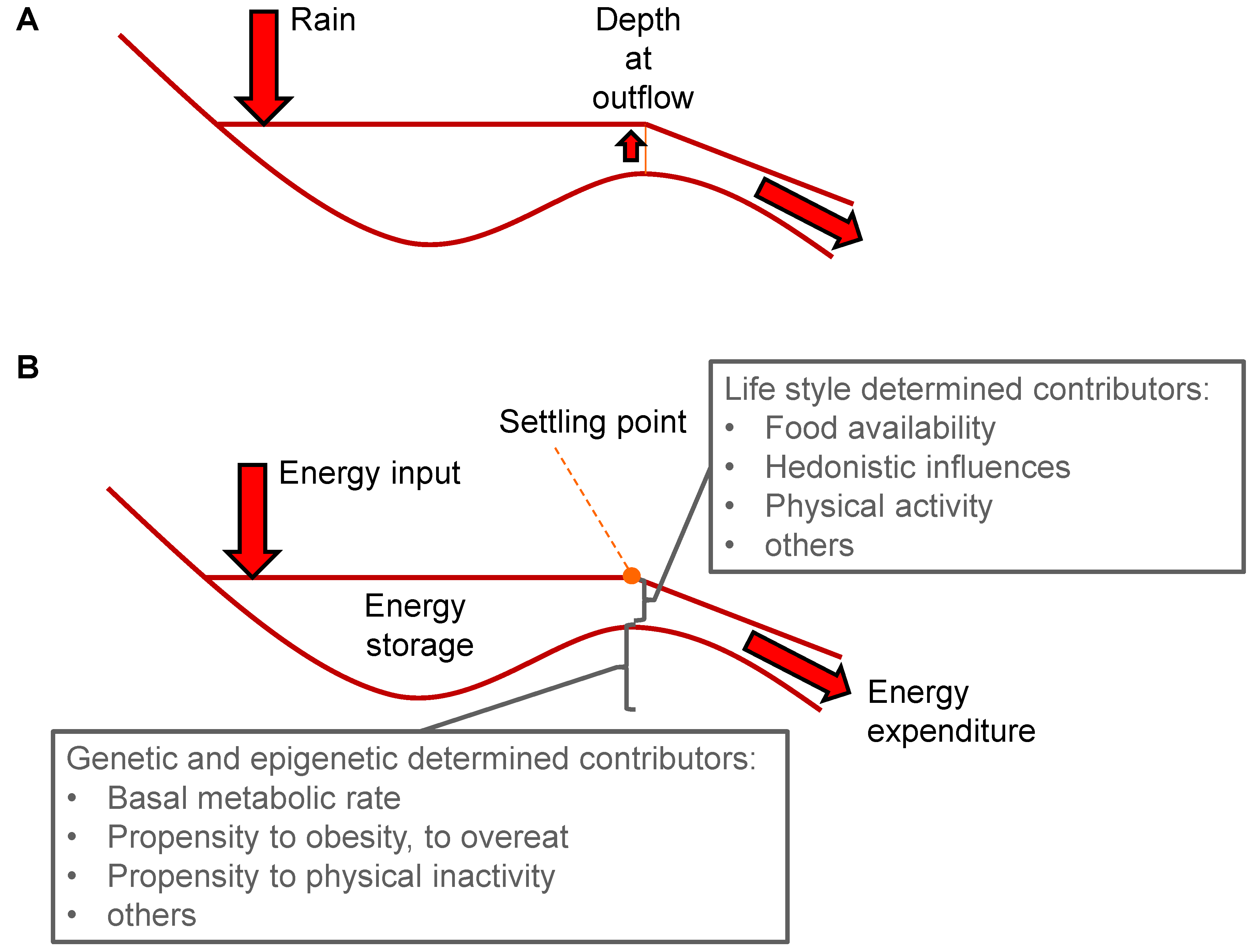
7. Setpoint versus Settling Point
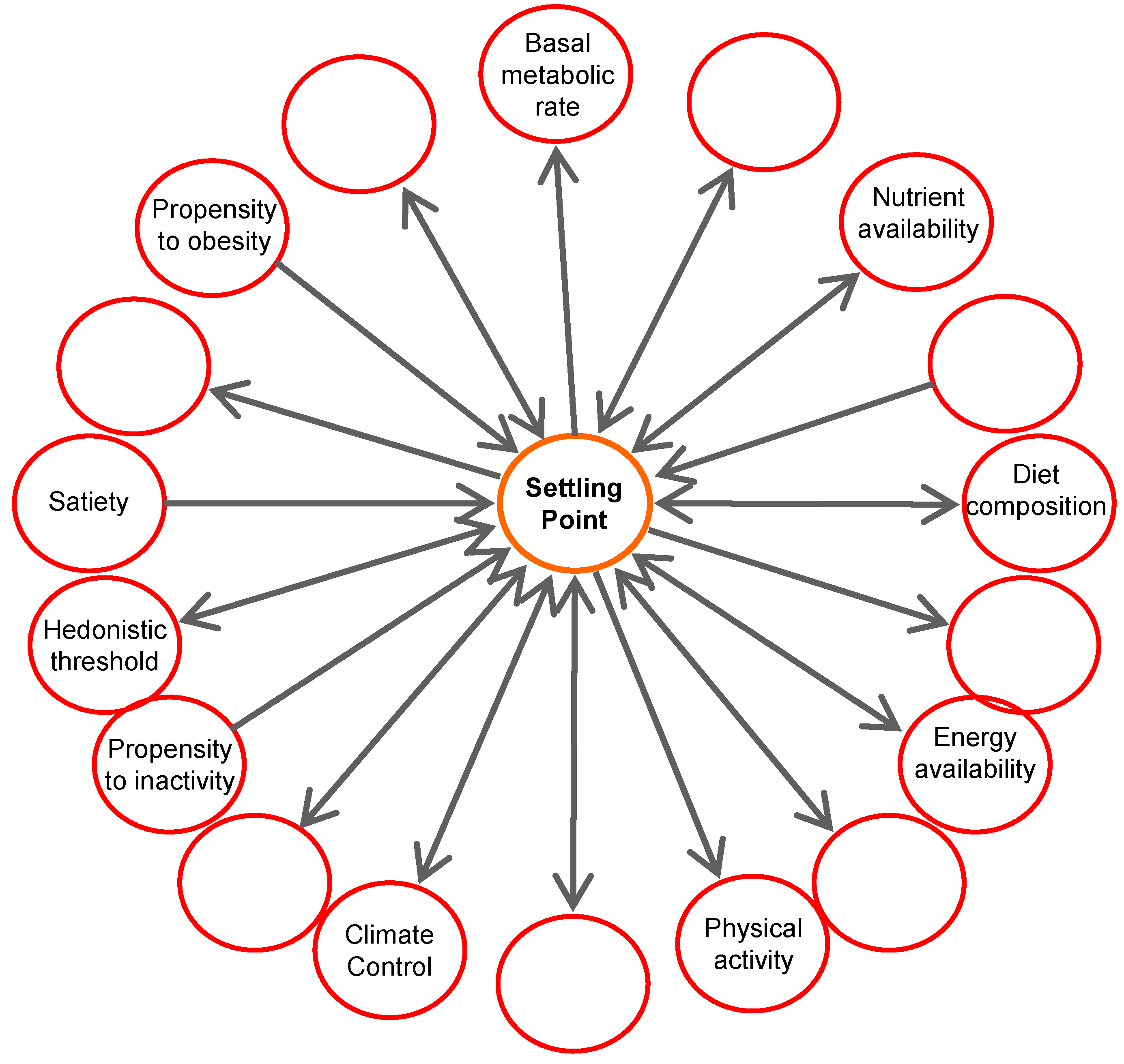
8. Forward, Taking the Settling Point Perspective
9. Study Design; Detailes Matter
10. Weight Loss, Metabolic Rate and Leptin
11. Metabolic Flexibility
12. Hedonistic Thresholds
13. Nutrigenomics Approaches
14. The Next Steps
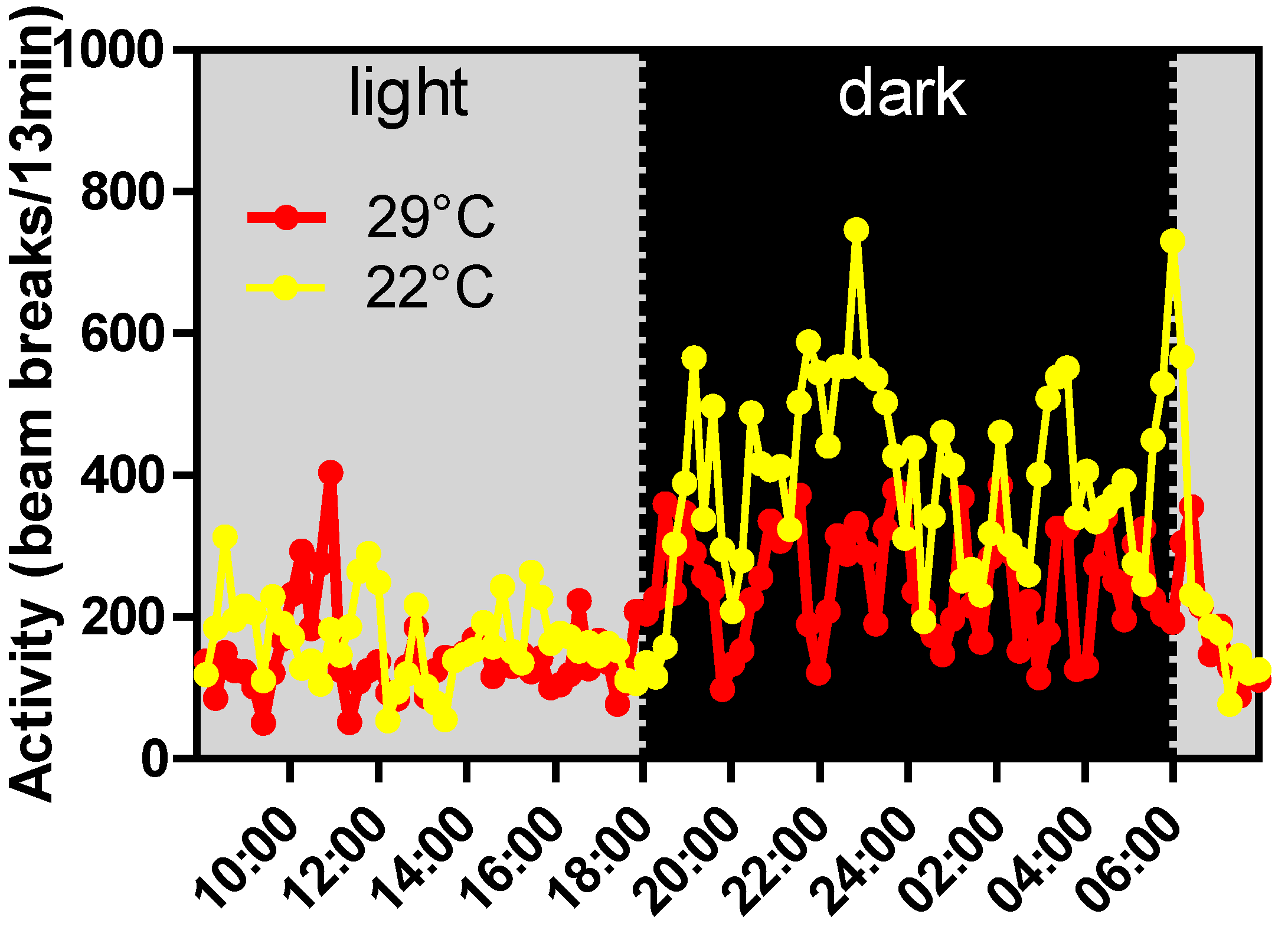
15. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Van den Brink, C.; Blokstra, A. Hoeveel Mensen Hebben Overgewicht. Available online: http://nationaalkompas.nl/gezondheidsdeterminanten/persoonsgebonden/overgewicht/hoeveel-mensen-hebben-overgewicht/ (accessed on 28 November 2013).
- WHO. Global Status Report on Noncommunicable Diseases 2010; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Ng, M.; Fleming, T.; Robinson, M.; Thomson, B.; Graetz, N.; Margono, C.; Mullany, E.C.; Biryukov, S.; Abbafati, C.; Abera, S.F.; et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980–2013: A systematic analysis for the global burden of disease study 2013. Lancet 2014, 384, 766–781. [Google Scholar] [PubMed]
- Franz, M.J.; VanWormer, J.J.; Crain, A.L.; Boucher, J.L.; Histon, T.; Caplan, W.; Bowman, J.D.; Pronk, N.P. Weight-Loss outcomes: A systematic review and meta-analysis of weight-loss clinical trials with a minimum 1-year follow-up. J. Am. Diet. Assoc. 2007, 107, 1755–1767. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.D.; Ryan, D.H.; Donato, K.A.; Apovian, C.M.; Ard, J.D.; Comuzzie, A.G.; Hu, F.B.; Hubbard, V.S.; Jakicic, J.M.; Kushner, R.F.; et al. Guidelines (2013) for managing overweight and obesity in adults. Obesity 2014, 22, S1–S410. [Google Scholar] [PubMed]
- Atkins, R.C. Atkinsʼ New Diet Revolution; M. Evans and Company Inc.: New York, NY, USA, 1992; p. 562. [Google Scholar]
- Astrup, A.; Grunwald, G.K.; Melanson, E.L.; Saris, W.H.; Hill, J.O. The role of low-fat diets in body weight control: A meta-analysis of ad libitum dietary intervention studies. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 1545–1552. [Google Scholar] [CrossRef] [PubMed]
- Samaha, F.F.; Iqbal, N.; Seshadri, P.; Chicano, K.L.; Daily, D.A.; McGrory, J.; Williams, T.; Williams, M.; Gracely, E.J.; Stern, L. A low-carbohydrate as compared with a low-fat diet in severe obesity. N. Engl. J. Med. 2003, 348, 2074–2081. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, D.J. Beneficial health effects of modest weight loss. Int. J. Obes. Relat. Metab. Disord. 1992, 16, 397–415. [Google Scholar] [PubMed]
- Van Gaal, L.; Mertens, I.; Ballaux, D. What is the relationship between risk factor reduction and degree of weight loss? Eur. Heart J. Suppl. 2005, 7, L21–L26. [Google Scholar]
- Langeveld, M.; de Vries, J.H. The mediocre results of dieting. Ned. Tijdschr. Geneesk. 2013, 157, A6017. [Google Scholar]
- Jeffery, R.W.; Wing, R.R. Long-term effects of interventions for weight loss using food provision and monetary incentives. J. Consult. Clin. Psychol. 1995, 63, 793–796. [Google Scholar] [CrossRef] [PubMed]
- Weiss, E.C.; Galuska, D.A.; Kettel Khan, L.; Gillespie, C.; Serdula, M.K. Weight regain in U.S. adults who experienced substantial weight loss, 1999–2002. Am. J. Prev. Med. 2007, 33, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Sumithran, P.; Proietto, J. The defence of body weight: A physiological basis for weight regain after weight loss. Clin. Sci. 2013, 124, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Goran, M.I. Energy metabolism and obesity. Med. Clin. N. Am. 2000, 84, 347–362. [Google Scholar] [CrossRef] [PubMed]
- Rolfe, D.F.; Brown, G.C. Cellular energy utilization and molecular origin of standard metabolic rate in mammals. Physiol. Rev. 1997, 77, 731–758. [Google Scholar] [PubMed]
- Wang, P.; Mariman, E.; Renes, J.; Keijer, J. The secretory function of adipocytes in the physiology of white adipose tissue. J. Cell Physiol. 2008, 216, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Keijer, J.; van Schothorst, E.M. Adipose tissue failure and mitochondria as a possible target for improvement by bioactive food components. Curr. Opin. Lipidol. 2008, 19, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Lissner, L.; Odell, P.M.; D’Agostino, R.B.; Stokes, J., III; Kreger, B.E.; Belanger, A.J.; Brownell, K.D. Variability of body weight and health outcomes in the framingham population. N. Engl. J. Med. 1991, 324, 1839–1844. [Google Scholar] [CrossRef]
- Khosla, T.; Billewicz, W.Z. Measurement of change in body-weight. Br. J. Nutr. 1964, 18, 227–239. [Google Scholar] [CrossRef]
- Leibel, R.L.; Rosenbaum, M.; Hirsch, J. Changes in energy expenditure resulting from altered body weight. N. Engl. J. Med. 1995, 332, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Rossen, L.M.; Talih, M. Social determinants of disparities in weight among us children and adolescents. Ann. Epidemiol. 2014, 24, 705–713. [Google Scholar] [CrossRef] [PubMed]
- Steyn, N.P.; McHiza, Z.J. Obesity and the nutrition transition in sub-saharan Africa. Ann. N. Y. Acad. Sci. 2014, 1311, 88–101. [Google Scholar] [CrossRef] [PubMed]
- Gordon-Larsen, P.; Wang, H.; Popkin, B.M. Overweight dynamics in Chinese children and adults. Obes. Rev. 2014, 15, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Baker, P.; Friel, S. Processed foods and the nutrition transition: Evidence from Asia. Obes. Rev. 2014, 15, 564–577. [Google Scholar] [CrossRef] [PubMed]
- Zhai, F.Y.; Du, S.F.; Wang, Z.H.; Zhang, J.G.; Du, W.W.; Popkin, B.M. Dynamics of the Chinese diet and the role of urbanicity, 1991–2011. Obes. Rev. 2014, 15, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Neel, J.V. Diabetes mellitus: A “thrifty” genotype rendered detrimental by “progress”? Am. J. Hum. Genet. 1962, 14, 353–362. [Google Scholar]
- Bouchard, C.; Tremblay, A.; Despres, J.P.; Nadeau, A.; Lupien, P.J.; Theriault, G.; Dussault, J.; Moorjani, S.; Pinault, S.; Fournier, G. The response to long-term overfeeding in identical twins. N. Engl. J. Med. 1990, 322, 1477–1482. [Google Scholar] [CrossRef] [PubMed]
- Wirtshafter, D.; Davis, J.D. Set points, settling points, and the control of body weight. Physiol. Behav. 1977, 19, 75–78. [Google Scholar]
- Montague, C.T.; Farooqi, I.S.; Whitehead, J.P.; Soos, M.A.; Rau, H.; Wareham, N.J.; Sewter, C.P.; Digby, J.E.; Mohammed, S.N.; Hurst, J.A.; et al. Congenital leptin deficiency is associated with severe early-onset obesity in humans. Nature 1997, 387, 903–908. [Google Scholar] [CrossRef]
- Kennedy, G.C. The role of depot fat in the hypothalamic control of food intake in the rat. Proc. R. Soc. Lond. 1953, 140, 578–596. [Google Scholar] [CrossRef] [PubMed]
- Speakman, J.R.; Levitsky, D.A.; Allison, D.B.; Bray, M.S.; de Castro, J.M.; Clegg, D.J.; Clapham, J.C.; Dulloo, A.G.; Gruer, L.; Haw, S.; et al. Set points, settling points and some alternative models: Theoretical options to understand how genes and environments combine to regulate body adiposity. Dis. Model Mech. 2011, 4, 733–745. [Google Scholar] [CrossRef]
- Parekh, P.I.; Petro, A.E.; Tiller, J.M.; Feinglos, M.N.; Surwit, R.S. Reversal of diet-induced obesity and diabetes in C57BL/6J mice. Metabolism 1998, 47, 1089–1096. [Google Scholar] [CrossRef] [PubMed]
- Hoevenaars, F.P.; Keijer, J.; Swarts, H.J.; Snaas-Alders, S.; Bekkenkamp-Grovenstein, M.; van Schothorst, E.M. Effects of dietary history on energy metabolism and physiological parameters in C57BL/6J mice. Exp. Physiol. 2013, 98, 1053–1062. [Google Scholar] [CrossRef]
- Swinburn, B.A.; Metcalf, P.A.; Ley, S.J. Long-term (5-year) effects of a reduced-fat diet intervention in individuals with glucose intolerance. Diabetes Care 2001, 24, 619–624. [Google Scholar] [CrossRef] [PubMed]
- Bouchard, C.; Tremblay, A.; Despres, J.P.; Nadeau, A.; Lupien, P.J.; Moorjani, S.; Theriault, G.; Kim, S.Y. Overfeeding in identical twins: 5-Year postoverfeeding results. Metabolism 1996, 45, 1042–1050. [Google Scholar] [CrossRef] [PubMed]
- Dubois, L.; Ohm Kyvik, K.; Girard, M.; Tatone-Tokuda, F.; Perusse, D.; Hjelmborg, J.; Skytthe, A.; Rasmussen, F.; Wright, M.J.; Lichtenstein, P.; et al. Genetic and environmental contributions to weight, height, and bmi from birth to 19 years of age: An international study of over 12,000 twin pairs. PLoS One 2012, 7, e30153. [Google Scholar] [CrossRef]
- Guo, J.; Jou, W.; Gavrilova, O.; Hall, K.D. Persistent diet-induced obesity in male C57BL/6 mice resulting from temporary obesigenic diets. PLoS One 2009, 4, e5370. [Google Scholar] [CrossRef] [PubMed]
- Fine, E.J.; Feinman, R.D. Thermodynamics of weight loss diets. Nutr. Metab 2004, 1, 15. [Google Scholar] [CrossRef]
- Feinman, R.D.; Fine, E.J. Nonequilibrium thermodynamics and energy efficiency in weight loss diets. Theor. Biol. Med. Model. 2007, 4, 27. [Google Scholar] [CrossRef] [PubMed]
- Nye, C.; Kim, J.; Kalhan, S.C.; Hanson, R.W. Reassessing triglyceride synthesis in adipose tissue. Trends Endocrinol. Metab. 2008, 19, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Stern, J.S.; Hirsch, J.; Drewnowski, A.; Sullivan, A.C.; Johnson, P.R.; Cohn, C.K. Glycerol kinase activity in adipose tissue of obese rats and mice: Effects of diet composition. J. Nutr. 1983, 113, 714–720. [Google Scholar] [PubMed]
- Guan, H.P.; Li, Y.; Jensen, M.V.; Newgard, C.B.; Steppan, C.M.; Lazar, M.A. A futile metabolic cycle activated in adipocytes by antidiabetic agents. Nat. Med. 2002, 8, 1122–1128. [Google Scholar] [CrossRef] [PubMed]
- Flachs, P.; Rossmeisl, M.; Kuda, O.; Kopecky, J. Stimulation of mitochondrial oxidative capacity in white fat independent of UCP1: A key to lean phenotype. Biochim. Biophys. Acta 2013, 1831, 986–1003. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, G.R.; Kemp, B.E. AMPK in health and disease. Physiol. Rev. 2009, 89, 1025–1078. [Google Scholar] [CrossRef]
- Bessesen, D.H.; Bull, S.; Cornier, M.A. Trafficking of dietary fat and resistance to obesity. Physiol. Behav. 2008, 94, 681–688. [Google Scholar] [CrossRef] [PubMed]
- Leonhardt, M.; Langhans, W. Fatty acid oxidation and control of food intake. Physiol. Behav. 2004, 83, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Delavari, M.; Sonderlund, A.L.; Swinburn, B.; Mellor, D.; Renzaho, A. Acculturation and obesity among migrant populations in high income countries—A systematic review. BMC Public Health 2013, 13, 458. [Google Scholar] [CrossRef] [PubMed]
- Hoevenaars, F.P.; Keijer, J.; Herreman, L.; Palm, I.; Hegeman, M.A.; Swarts, H.J.; van Schothorst, E.M. Adipose tissue metabolism and inflammation are differently affected by weight loss in obese mice due to either a high-fat diet restriction or change to a low-fat diet. Genes Nutr. 2014, 9, 391. [Google Scholar] [CrossRef] [PubMed]
- Cameron, K.M.; Speakman, J.R. Reduction of dietary energy density reduces body mass regain following energy restriction in female mice. J. Nutr. 2011, 141, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Hoevenaars, F.P.; van Schothorst, E.M.; Horakova, O.; Voigt, A.; Rossmeisl, M.; Pico, C.; Caimari, A.; Kopecky, J.; Klaus, S.; Keijer, J. Bioclaims standard diet (BIOsd): A reference diet for nutritional physiology. Genes Nutr. 2012, 7, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Tschop, M.H.; Speakman, J.R.; Arch, J.R.; Auwerx, J.; Bruning, J.C.; Chan, L.; Eckel, R.H.; Farese, R.V., Jr.; Galgani, J.E.; Hambly, C.; et al. A guide to analysis of mouse energy metabolism. Nat. Methods 2012, 9, 57–63. [Google Scholar] [CrossRef]
- Ravussin, Y.; Gutman, R.; Diano, S.; Shanabrough, M.; Borok, E.; Sarman, B.; Lehmann, A.; LeDuc, C.A.; Rosenbaum, M.; Horvath, T.L.; et al. Effects of chronic weight perturbation on energy homeostasis and brain structure in mice. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 300, R1352–R1362. [Google Scholar] [CrossRef] [PubMed]
- MacLean, P.S.; Higgins, J.A.; Johnson, G.C.; Fleming-Elder, B.K.; Donahoo, W.T.; Melanson, E.L.; Hill, J.O. Enhanced metabolic efficiency contributes to weight regain after weight loss in obesity-prone rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2004, 287, R1306–R1315. [Google Scholar] [CrossRef] [PubMed]
- Taylor, H.L.; Keys, A. Adaptation to caloric restriction. Science 1950, 112, 215–218. [Google Scholar] [CrossRef] [PubMed]
- Doucet, E.; St-Pierre, S.; Almeras, N.; Despres, J.P.; Bouchard, C.; Tremblay, A. Evidence for the existence of adaptive thermogenesis during weight loss. Br. J. Nutr. 2001, 85, 715–723. [Google Scholar] [CrossRef] [PubMed]
- Dulloo, A.G.; Jacquet, J. Adaptive reduction in basal metabolic rate in response to food deprivation in humans: A role for feedback signals from fat stores. Am. J. Clin. Nutr. 1998, 68, 599–606. [Google Scholar] [PubMed]
- Martin, J.E.; Sheaff, M.T. The pathology of ageing: Concepts and mechanisms. J. Pathol. 2007, 211, 111–113. [Google Scholar] [CrossRef] [PubMed]
- Johannsen, D.L.; Knuth, N.D.; Huizenga, R.; Rood, J.C.; Ravussin, E.; Hall, K.D. Metabolic slowing with massive weight loss despite preservation of fat-free mass. J. Clin. Endocrinol. Metab. 2012, 97, 2489–2496. [Google Scholar] [CrossRef]
- Camps, S.G.; Verhoef, S.P.; Westerterp, K.R. Weight loss, weight maintenance, and adaptive thermogenesis. Am. J. Clin. Nutr. 2013, 97, 990–994. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, M.; Hirsch, J.; Gallagher, D.A.; Leibel, R.L. Long-term persistence of adaptive thermogenesis in subjects who have maintained a reduced body weight. Am. J. Clin. Nutr. 2008, 88, 906–912. [Google Scholar] [PubMed]
- Froidevaux, F.; Schutz, Y.; Christin, L.; Jequier, E. Energy expenditure in obese women before and during weight loss, after refeeding, and in the weight-relapse period. Am. J. Clin. Nutr. 1993, 57, 35–42. [Google Scholar]
- Leibel, R.L.; Hirsch, J. Diminished energy requirements in reduced-obese patients. Metabolism 1984, 33, 164–170. [Google Scholar] [CrossRef]
- Weinsier, R.L.; Nagy, T.R.; Hunter, G.R.; Darnell, B.E.; Hensrud, D.D.; Weiss, H.L. Do adaptive changes in metabolic rate favor weight regain in weight-reduced individuals? An examination of the set-point theory. Am. J. Clin. Nutr. 2000, 72, 1088–1094. [Google Scholar] [PubMed]
- Dore, C.; Hesp, R.; Wilkins, D.; Garrow, J.S. Prediction of energy requirements of obese patients after massive weight loss. Hum. Nutr. Clin. Nutr. 1982, 36C, 41–48. [Google Scholar]
- Elliot, D.L.; Goldberg, L.; Kuehl, K.S.; Bennett, W.M. Sustained depression of the resting metabolic rate after massive weight loss. Am. J. Clin. Nutr. 1989, 49, 93–96. [Google Scholar]
- Bouwman, F.G.; Claessens, M.; van Baak, M.A.; Noben, J.P.; Wang, P.; Saris, W.H.; Mariman, E.C. The physiologic effects of caloric restriction are reflected in the in vivo adipocyte-enriched proteome of overweight/obese subjects. J. Proteome Res. 2009, 8, 5532–5540. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, M.; Goldsmith, R.; Bloomfield, D.; Magnano, A.; Weimer, L.; Heymsfield, S.; Gallagher, D.; Mayer, L.; Murphy, E.; Leibel, R.L. Low-dose leptin reverses skeletal muscle, autonomic, and neuroendocrine adaptations to maintenance of reduced weight. J. Clin. Investig. 2005, 115, 3579–3586. [Google Scholar] [CrossRef]
- Jackman, M.R.; Steig, A.; Higgins, J.A.; Johnson, G.C.; Fleming-Elder, B.K.; Bessesen, D.H.; MacLean, P.S. Weight regain after sustained weight reduction is accompanied by suppressed oxidation of dietary fat and adipocyte hyperplasia. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 294, R1117–R1129. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.C.; Nunez, N.P.; Perkins, S.N.; Barrett, J.C.; Hursting, S.D. Effects of energy balance on cancer in genetically altered mice. J. Nutr. 2004, 134, 3394S–3398S. [Google Scholar] [PubMed]
- MacLean, P.S.; Higgins, J.A.; Johnson, G.C.; Fleming-Elder, B.K.; Peters, J.C.; Hill, J.O. Metabolic adjustments with the development, treatment, and recurrence of obesity in obesity-prone rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2004, 287, R288–R297. [Google Scholar] [CrossRef] [PubMed]
- Dhurandhar, E.J.; Dawson, J.; Alcorn, A.; Larsen, L.H.; Thomas, E.A.; Cardel, M.; Bourland, A.C.; Astrup, A.; St-Onge, M.P.; Hill, J.O.; et al. The effectiveness of breakfast recommendations on weight loss: A randomized controlled trial. Am. J. Clin. Nutr. 2014. [Google Scholar] [CrossRef]
- Rothschild, J.; Hoddy, K.K.; Jambazian, P.; Varady, K.A. Time-restricted feeding and risk of metabolic disease: A review of human and animal studies. Nutr. Rev. 2014, 72, 308–318. [Google Scholar] [CrossRef] [PubMed]
- Arguin, H.; Dionne, I.J.; Senechal, M.; Bouchard, D.R.; Carpentier, A.C.; Ardilouze, J.L.; Tremblay, A.; Leblanc, C.; Brochu, M. Short- and long-term effects of continuous versus intermittent restrictive diet approaches on body composition and the metabolic profile in overweight and obese postmenopausal women: A pilot study. Menopause 2012, 19, 870–876. [Google Scholar] [CrossRef] [PubMed]
- Harvie, M.N.; Pegington, M.; Mattson, M.P.; Frystyk, J.; Dillon, B.; Evans, G.; Cuzick, J.; Jebb, S.A.; Martin, B.; Cutler, R.G.; et al. The effects of intermittent or continuous energy restriction on weight loss and metabolic disease risk markers: A randomized trial in young overweight women. Int. J. Obes. 2011, 35, 714–727. [Google Scholar] [CrossRef]
- Storlien, L.; Oakes, N.D.; Kelley, D.E. Metabolic flexibility. Proc. Nutr. Soc. 2004, 63, 363–368. [Google Scholar] [CrossRef]
- Anonymous. What is health? The ability to adapt. Lancet 2009, 373, 781. [Google Scholar]
- Horakova, O.; Medrikova, D.; van Schothorst, E.M.; Bunschoten, A.; Flachs, P.; Kus, V.; Kuda, O.; Bardova, K.; Janovska, P.; Hensler, M.; et al. Preservation of metabolic flexibility in skeletal muscle by a combined use of n-3 pufa and rosiglitazone in dietary obese mice. PLoS One 2012, 7, e43764. [Google Scholar] [CrossRef] [PubMed]
- Huffman, K.M.; Redman, L.M.; Landerman, L.R.; Pieper, C.F.; Stevens, R.D.; Muehlbauer, M.J.; Wenner, B.R.; Bain, J.R.; Kraus, V.B.; Newgard, C.B.; et al. Caloric restriction alters the metabolic response to a mixed-meal: Results from a randomized, controlled trial. PLoS One 2012, 7, e28190. [Google Scholar] [CrossRef] [PubMed]
- Malin, S.K.; Haus, J.M.; Solomon, T.P.; Blaszczak, A.; Kashyap, S.R.; Kirwan, J.P. Insulin sensitivity and metabolic flexibility following exercise training among different obese insulin-resistant phenotypes. Am. J. Physiol. Endocrinol. Metab. 2013, 305, E1292–E1298. [Google Scholar] [CrossRef] [PubMed]
- Blundell, J.E.; Gillett, A. Control of food intake in the obese. Obes. Res. 2001, 9, 263S–270S. [Google Scholar] [CrossRef]
- Lenoir, M.; Serre, F.; Cantin, L.; Ahmed, S.H. Intense sweetness surpasses cocaine reward. PLoS One 2007, 2, e698. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.D.; Wang, G.J.; Baler, R.D. Reward, dopamine and the control of food intake: Implications for obesity. Trend Cogn. Sci. 2011, 15, 37–46. [Google Scholar] [CrossRef]
- Sampey, B.P.; Vanhoose, A.M.; Winfield, H.M.; Freemerman, A.J.; Muehlbauer, M.J.; Fueger, P.T.; Newgard, C.B.; Makowski, L. Cafeteria diet is a robust model of human metabolic syndrome with liver and adipose inflammation: Comparison to high-fat diet. Obesity 2011, 19, 1109–1117. [Google Scholar] [CrossRef] [PubMed]
- Caimari, A.; Oliver, P.; Rodenburg, W.; Keijer, J.; Palou, A. Feeding conditions control the expression of genes involved in sterol metabolism in peripheral blood mononuclear cells of normoweight and diet-induced (cafeteria) obese rats. J. Nutr. Biochem. 2010, 21, 1127–1133. [Google Scholar] [CrossRef] [PubMed]
- Lalanza, J.F.; Caimari, A.; del Bas, J.M.; Torregrosa, D.; Cigarroa, I.; Pallas, M.; Capdevila, L.; Arola, L.; Escorihuela, R.M. Effects of a post-weaning cafeteria diet in young rats: Metabolic syndrome, reduced activity and low anxiety-like behaviour. PLoS One 2014, 9, e85049. [Google Scholar] [CrossRef] [PubMed]
- Desmarchelier, C.; Ludwig, T.; Scheundel, R.; Rink, N.; Bader, B.L.; Klingenspor, M.; Daniel, H. Diet-Induced obesity in ad libitum-fed mice: Food texture overrides the effect of macronutrient composition. Br. J. Nutr. 2013, 109, 1518–1527. [Google Scholar] [PubMed]
- Chen, G.C.; Huang, C.Y.; Chang, M.Y.; Chen, C.H.; Chen, S.W.; Huang, C.J.; Chao, P.M. Two unhealthy dietary habits featuring a high fat content and a sucrose-containing beverage intake, alone or in combination, on inducing metabolic syndrome in wistar rats and C57BL/6J mice. Metabolism 2011, 60, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Light, H.R.; Tsanzi, E.; Gigliotti, J.; Morgan, K.; Tou, J.C. The type of caloric sweetener added to water influences weight gain, fat mass, and reproduction in growing sprague-dawley female rats. Exp. Biol. Med. 2009, 234, 651–661. [Google Scholar] [CrossRef]
- Nieuwenhuizen, A.; Swarts, H.; Keijer, J.; Human and Animal Physiology, Wageningen University, Wageningen, The Netherlands. Unpublished work. 2014.
- Rolls, E.T. Taste, olfactory and food texture reward processing in the brain and obesity. Int. J. Obes. 2011, 35, 550–561. [Google Scholar] [CrossRef]
- Rolls, B.J. Dietary strategies for weight management. Nestle Nutr. Inst. Workshop Ser. 2012, 73, 37–48. [Google Scholar] [PubMed]
- Mattes, R.D.; Campbell, W.W. Effects of food form and timing of ingestion on appetite and energy intake in lean young adults and in young adults with obesity. J. Am. Diet. Assoc. 2009, 109, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Rudkowska, I.; Perusse, L. Individualized weight management: What can be learned from nutrigenomics and nutrigenetics? Prog. Mol. Biol. Transl. Sci. 2012, 108, 347–382. [Google Scholar]
- Elliott, R.M. Transcriptomics and micronutrient research. Br. J. Nutr. 2008, 99, S59–S65. [Google Scholar] [CrossRef] [PubMed]
- Keijer, J.; van Helden, Y.G.; Bunschoten, A.; van Schothorst, E.M. Transcriptome analysis in benefit-risk assessment of micronutrients and bioactive food components. Mol. Nutr. Food Res. 2010, 54, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Yu, S.; Li, H.; Xiang, H.; Peng, J.; Jiang, S. Micrornas: Emerging roles in adipogenesis and obesity. Cell Sign. 2014, 26, 1888–1896. [Google Scholar] [CrossRef]
- Palmer, J.D.; Soule, B.P.; Simone, B.A.; Zaorsky, N.G.; Jin, L.; Simone, N.L. Microrna expression altered by diet: Can food be medicinal? Ageing Res. Rev. 2014, 17, 16–24. [Google Scholar]
- Kornfeld, J.W.; Bruning, J.C. Regulation of metabolism by long, non-coding rnas. Front. Genet. 2014, 5, 57. [Google Scholar] [CrossRef] [PubMed]
- Malkaram, S.A.; Hassan, Y.I.; Zempleni, J. Online tools for bioinformatics analyses in nutrition sciences. Adv. Nutr. 2012, 3, 654–665. [Google Scholar] [CrossRef] [PubMed]
- McGettigan, P.A. Transcriptomics in the RNA-seq era. Curr. Opin. Chem. Biol. 2013, 17, 4–11. [Google Scholar] [CrossRef] [PubMed]
- Hirschey, M.D.; Shimazu, T.; Goetzman, E.; Jing, E.; Schwer, B.; Lombard, D.B.; Grueter, C.A.; Harris, C.; Biddinger, S.; Ilkayeva, O.R.; et al. Sirt3 regulates mitochondrial fatty-acid oxidation by reversible enzyme deacetylation. Nature 2010, 464, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Rardin, M.J.; Newman, J.C.; Held, J.M.; Cusack, M.P.; Sorensen, D.J.; Li, B.; Schilling, B.; Mooney, S.D.; Kahn, C.R.; Verdin, E.; et al. Label-free quantitative proteomics of the lysine acetylome in mitochondria identifies substrates of SIRT3 in metabolic pathways. Proc. Natl. Acad. Sci. USA 2013, 110, 6601–6606. [Google Scholar] [CrossRef] [PubMed]
- De Roos, B.; Duthie, S.J.; Polley, A.C.; Mulholland, F.; Bouwman, F.G.; Heim, C.; Rucklidge, G.J.; Johnson, I.T.; Mariman, E.C.; Daniel, H.; et al. Proteomic methodological recommendations for studies involving human plasma, platelets, and peripheral blood mononuclear cells. J. Proteome Res. 2008, 7, 2280–2290. [Google Scholar] [CrossRef] [PubMed]
- Ibanez, C.; Simo, C.; Garcia-Canas, V.; Cifuentes, A.; Castro-Puyana, M. Metabolomics, peptidomics and proteomics applications of capillary electrophoresis-mass spectrometry in foodomics: A review. Anal. Chim. Acta 2013, 802, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Putri, S.P.; Nakayama, Y.; Matsuda, F.; Uchikata, T.; Kobayashi, S.; Matsubara, A.; Fukusaki, E. Current metabolomics: Practical applications. J. Biosci. Bioeng. 2013, 115, 579–589. [Google Scholar] [CrossRef] [PubMed]
- Di Girolamo, F.; Lante, I.; Muraca, M.; Putignani, L. The role of mass spectrometry in the “omics” era. Curr. Org. Chem. 2013, 17, 2891–2905. [Google Scholar] [CrossRef] [PubMed]
- Mulvihill, M.M.; Nomura, D.K. Metabolomic strategies to map functions of metabolic pathways. Am. J. Physiol. Endocrinol. Metab. 2014, 307, E237–E244. [Google Scholar] [CrossRef] [PubMed]
- Kussmann, M.; Rezzi, S.; Daniel, H. Profiling techniques in nutrition and health research. Curr. Opin. Biotech. 2008, 19, 83–99. [Google Scholar] [CrossRef] [PubMed]
- Norheim, F.; Gjelstad, I.M.; Hjorth, M.; Vinknes, K.J.; Langleite, T.M.; Holen, T.; Jensen, J.; Dalen, K.T.; Karlsen, A.S.; Kielland, A.; et al. Molecular nutrition research: The modern way of performing nutritional science. Nutrients 2012, 4, 1898–1944. [Google Scholar] [CrossRef] [PubMed]
- Van Iersel, M.P.; Sokolovic, M.; Lenaerts, K.; Kutmon, M.; Bouwman, F.G.; Lamers, W.H.; Mariman, E.C.; Evelo, C.T. Integrated visualization of a multi-omics study of starvation in mouse intestine. J. Int. Bioinform. 2014, 11, 235. [Google Scholar]
- Duivenvoorde, L.P.M.; van Schothorst, E.M.; Derous, D.; van der Stelt, I.; Masania, J.; Rabbani, N.; Thornalley, P.J.; Keijer, J. Oxygen restriction as challenge test reveals early high-fat diet-induced changes in glucose and lipid metabolism. Pflügers Arch. 2014. [Google Scholar] [CrossRef]
- De Mello, V.D.; Kolehmanien, M.; Schwab, U.; Pulkkinen, L.; Uusitupa, M. Gene expression of peripheral blood mononuclear cells as a tool in dietary intervention studies: What do we know so far? Mol. Nutr. Food Res. 2012, 56, 1160–1172. [Google Scholar]
- Joseph, P.; Umbright, C.; Sellamuthu, R. Blood transcriptomics: Applications in toxicology. J. Appl. Toxicol. 2013. [Google Scholar] [CrossRef]
- Mariadason, J.M.; Arango, D.; Corner, G.A.; Aranes, M.J.; Hotchkiss, K.A.; Yang, W.; Augenlicht, L.H. A gene expression profile that defines colon cell maturation in vitro. Cancer Res. 2002, 62, 4791–4804. [Google Scholar] [PubMed]
- Wielinga, P.Y.; Yakala, G.K.; Heeringa, P.; Kleemann, R.; Kooistra, T. Beneficial effects of alternate dietary regimen on liver inflammation, atherosclerosis and renal activation. PLoS One 2011, 6, e18432. [Google Scholar] [PubMed]
- Cannon, B.; Nedergaard, J. Nonshivering thermogenesis and its adequate measurement in metabolic studies. J. Exp. Biol. 2011, 214, 242–253. [Google Scholar] [CrossRef]
- Speakman, J.; Keijer, J. Not so hot: Optimal housing temperatures for mice to mimic the thermal environment of humans. Mol. Metab. 2013, 2, 5–9. [Google Scholar] [CrossRef]
- Feldmann, H.M.; Golozoubova, V.; Cannon, B.; Nedergaard, J. Ucp1 ablation induces obesity and abolishes diet-induced thermogenesis in mice exempt from thermal stress by living at thermoneutrality. Cell Metab. 2009, 9, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Van Schothorst, E.M.; Hoevenaars, F.P.M.; van der Stelt, I.; Keijer, J.; Human and Animal Physiology, Wageningen University, Wageningen, The Netherlands. Unpublished work. 2014.
- Kus, V.; Prazak, T.; Brauner, P.; Hensler, M.; Kuda, O.; Flachs, P.; Janovska, P.; Medrikova, D.; Rossmeisl, M.; Jilkova, Z.; et al. Induction of muscle thermogenesis by high-fat diet in mice: Association with obesity-resistance. Am. J. Physiol. Endocr. Metab. 2008, 295, E356–E367. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Keijer, J.; Hoevenaars, F.P.M.; Nieuwenhuizen, A.; Van Schothorst, E.M. Nutrigenomics of Body Weight Regulation: A Rationale for Careful Dissection of Individual Contributors. Nutrients 2014, 6, 4531-4551. https://doi.org/10.3390/nu6104531
Keijer J, Hoevenaars FPM, Nieuwenhuizen A, Van Schothorst EM. Nutrigenomics of Body Weight Regulation: A Rationale for Careful Dissection of Individual Contributors. Nutrients. 2014; 6(10):4531-4551. https://doi.org/10.3390/nu6104531
Chicago/Turabian StyleKeijer, Jaap, Femke P. M. Hoevenaars, Arie Nieuwenhuizen, and Evert M. Van Schothorst. 2014. "Nutrigenomics of Body Weight Regulation: A Rationale for Careful Dissection of Individual Contributors" Nutrients 6, no. 10: 4531-4551. https://doi.org/10.3390/nu6104531
APA StyleKeijer, J., Hoevenaars, F. P. M., Nieuwenhuizen, A., & Van Schothorst, E. M. (2014). Nutrigenomics of Body Weight Regulation: A Rationale for Careful Dissection of Individual Contributors. Nutrients, 6(10), 4531-4551. https://doi.org/10.3390/nu6104531





