Cellular Mechanisms of Zinc Dysregulation: A Perspective on Zinc Homeostasis as an Etiological Factor in the Development and Progression of Breast Cancer
Abstract
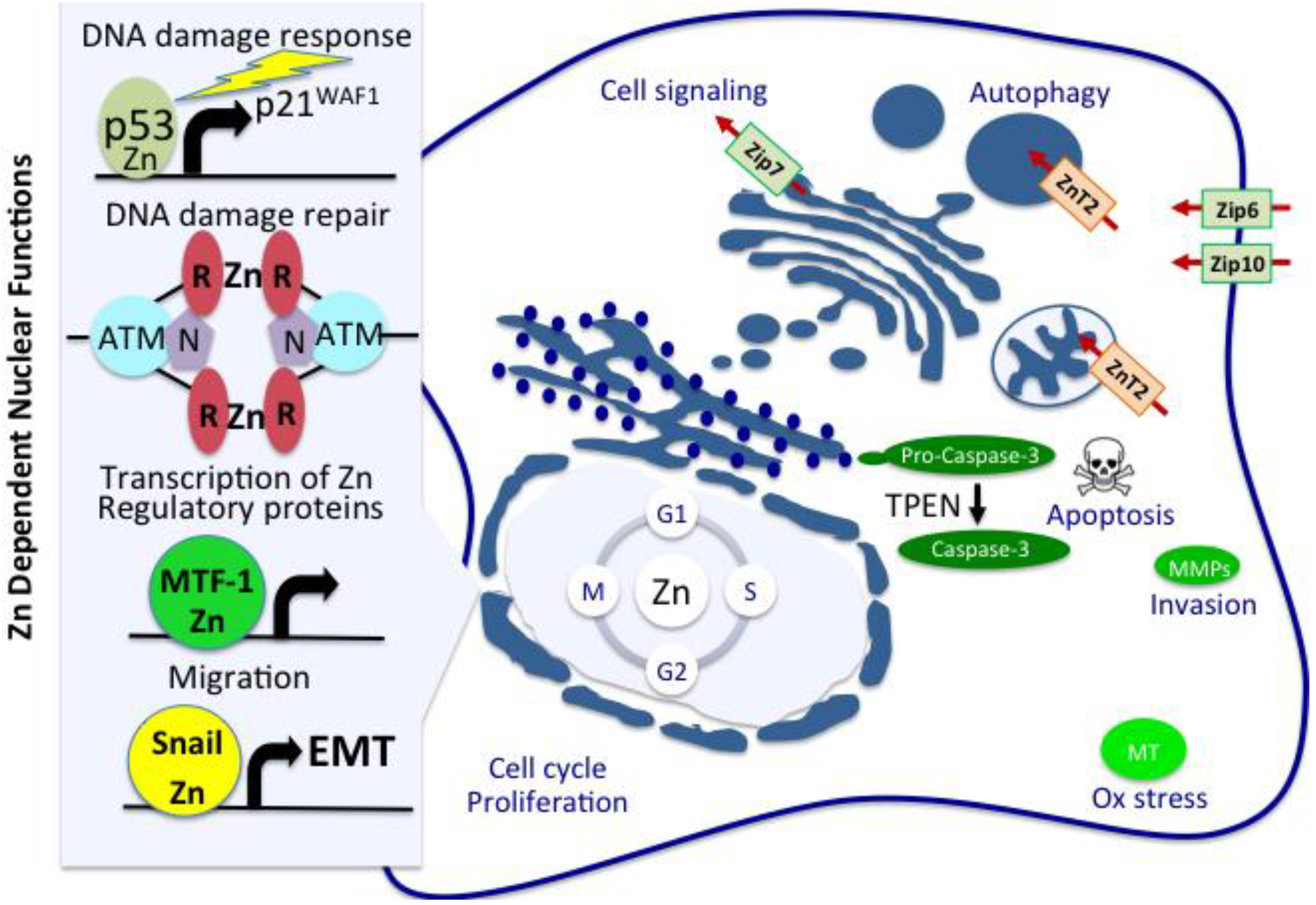
:1. Introduction
2. Comparative Plasma and Tumor Zinc Levels and Breast Cancer Risk
3. Zinc and Oxidative Stress
4. Zinc-Metallothionein Regulation of Oxidative Stress and Chemoresistance in Breast Cancer
5. Zinc Regulation of p53 Activation, ATM/MRN Mediated DNA Damage Response and Repair
6. Zinc Regulation of Cell Proliferation, Signaling and Apoptosis
7. Zinc Regulation of MMPs in Mammary Gland Development and Cancer
8. Regulation of Cellular Zinc Homeostasis
9. Zinc Transporters Associated with Breast Tissue Carcinogenesis
10. Dysregulation of Zinc Metabolism in Other Hormonally Regulated Cancers
11. Perspective
Acknowledgments
Conflict of Interest
References
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA Cancer J.Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. The hallmarks of cancer. Cell 2000, 100, 57–70. [Google Scholar] [CrossRef]
- Garcia, A.G.; Rodriguez-Rocha, H.; Madayiputhiya, N.; Pappa, A.; Panayiotidis, M.I.; Franco, R. Biomarkers of Protein oxidation in human disease. Curr. Mol. Med. 2012, 12, 681–697. [Google Scholar] [CrossRef]
- Acharya, A.; Das, I.; Chandhok, D.; Saha, T. Redox regulation in cancer: A double-edged sword with therapeutic potential. Oxid. Med. Cell. Longev. 2010, 3, 23–34. [Google Scholar] [CrossRef]
- Milner, J.A. Molecular targets for bioactive food components. J. Nutr. 2004, 134, 2492S–2498S. [Google Scholar]
- Finley, J.W. Proposed criteria for assessing the efficacy of cancer reduction by plant foods enriched in carotenoids, glucosinolates, polyphenols and selenocompounds. Ann. Bot. 2005, 95, 1075–1096. [Google Scholar] [CrossRef]
- Fenech, M.; Ferguson, L.R. Vitamins/minerals and genomic stability in humans. Mut. Res. 2001, 475, 1–6. [Google Scholar] [CrossRef]
- Eide, D.J. The oxidative stress of zinc deficiency. Metallomics 2011, 3, 1124–1129. [Google Scholar] [CrossRef]
- Leone, N.; Courbon, D.; Ducimetiere, P.; Zureik, M. Zinc, copper, and magnesium and risks for all-cause, cancer, and cardiovascular mortality. Epidemiology 2006, 17, 308–314. [Google Scholar]
- Brown, K.H.; Rivera, J.A.; Bhutta, Z.; Gibson, R.S.; King, J.C.; Lonnerdal, B.; Ruel, M.T.; Sandtrom, B.; Wasantwisut, E.; Hotz, C. International Zinc Nutrition Consultative Group (IZiNCG). International Zinc Nutrition Consultative Group (IZiNCG) technical document #1. Assessment of the risk of zinc deficiency in populations and options for its control. Food Nutr. Bull. 2004, 25, S99–S203. [Google Scholar]
- Andreini, C.; Banci, L.; Bertini, I.; Rosato, A. Counting the zinc-proteins encoded in the human genome. J. Proteome Res. 2006, 5, 196–201. [Google Scholar] [CrossRef]
- Rink, L.; Gabriel, P. Zinc and the immune system. Proc. Nutr. Soc. 2000, 59, 541–552. [Google Scholar] [CrossRef]
- Theocharis, S.E.; Margeli, A.P.; Klijanienko, J.T.; Kouraklis, G.P. Metallothionein expression in human neoplasia. Histopathology 2004, 45, 103–118. [Google Scholar] [CrossRef]
- Oteiza, P.I.; Clegg, M.S.; Keen, C.L. Short-term zinc deficiency affects nuclear factor-kappab nuclear binding activity in rat testes. J. Nutr. 2001, 131, 21–26. [Google Scholar]
- Prasad, A.S. Zinc deficiency in humans: A neglected problem. J. Am. Coll. Nutr. 1998, 17, 542–543. [Google Scholar]
- Prasad, A.S. Zinc deficiency. BMJ 2003, 326, 409–410. [Google Scholar] [CrossRef]
- Song, Y.; Chung, C.S.; Bruno, R.S.; Traber, M.G.; Brown, K.H.; King, J.C.; Ho, E. Dietary zinc restriction and repletion affects DNA integrity in healthy men. Am. J. Clin. Nutr. 2009, 90, 321–328. [Google Scholar] [CrossRef]
- Song, Y.; Leonard, S.W.; Traber, M.G.; Ho, E. Zinc deficiency affects DNA damage, oxidative stress, antioxidant defenses, and DNA repair in rats. J. Nutr. 2009, 139, 1626–1631. [Google Scholar] [CrossRef]
- Vallee, B.L.; Auld, D.S. Cocatalytic zinc motifs in enzyme catalysis. Proc. Natl. Acad. Sci. USA 1993, 90, 2715–2718. [Google Scholar] [CrossRef]
- Prasad, A.S. Zinc: An overview. Nutrition 1995, 11, 93–99. [Google Scholar]
- Brown, K.H.; Peerson, J.M.; Rivera, J.; Allen, L.H. Effect of supplemental zinc on the growth and serum zinc concentrations of prepubertal children: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2002, 75, 1062–1071. [Google Scholar]
- Prasad, A.S.; Beck, F.W.; Snell, D.C.; Kucuk, O. Zinc in cancer prevention. Nutr. Cancer 2009, 61, 879–887. [Google Scholar] [CrossRef]
- Lin, C.Y.; Tsai, P.H.; Kandaswami, C.C.; Lee, P.P.; Huang, C.J.; Hwang, J.J.; Lee, M.T. Matrix metalloproteinase-9 cooperates with transcription factor Snail to induce epithelial-mesenchymal transition. Cancer Sci. 2011, 102, 815–827. [Google Scholar] [CrossRef]
- Yamasaki, S.; Sakata-Sogawa, K.; Hasegawa, A.; Suzuki, T.; Kabu, K.; Sato, E.; Kurosaki, T.; Yamashita, S.; Tokunaga, M.; Nishida, K.; et al. Zinc is a novel intracellular second messenger. J. Cell Biol. 2007, 177, 637–645. [Google Scholar] [CrossRef]
- Sharif, R.; Thomas, P.; Zalewski, P.; Fenech, M. The role of zinc in genomic stability. Mutat. Res. 2011, 733, 111–121. [Google Scholar]
- Chasapis, C.T.; Loutsidou, A.C.; Spiliopoulou, C.A.; Stefanidou, M.E. Zinc and human health: An update. Arch. Toxicol. 2011, 86, 521–534. [Google Scholar]
- Ho, E.; Courtemanche, C.; Ames, B.N. Zinc deficiency induces oxidative DNA damage and increases p53 expression in human lung fibroblasts. J. Nutr. 2003, 133, 2543–2548. [Google Scholar]
- Ho, E. Zinc deficiency, DNA damage and cancer risk. J. Nutr. Biochem. 2004, 15, 572–578. [Google Scholar] [CrossRef]
- Beyersmann, D.; Haase, H. Functions of zinc in signaling, proliferation and differentiation of mammalian cells. Biometals 2001, 14, 331–341. [Google Scholar] [CrossRef]
- John, E.; Laskow, T.C.; Buchser, W.J.; Pitt, B.R.; Basse, P.H.; Butterfield, L.H.; Kalinski, P.; Lotze, M.T. Zinc in innate and adaptive tumor immunity. J. Transl. Med. 2010, 8, 118. [Google Scholar] [CrossRef]
- Poo, J.L.; Romero, R.R.; Robles, J.A.; Montemayor, A.C.; Isoard, F.; Estanes, A.; Uribe, M. Diagnostic value of the copper/zinc ratio in digestive cancer: A case control study. Arch. Med. Res. 1997, 28, 259–263. [Google Scholar]
- Goel, T.; Sankhwar, S.N. Comparative study of zinc levels in benign and malignant lesions of the prostate. Scand. J. Urol. Nephrol. 2006, 40, 108–112. [Google Scholar] [CrossRef]
- Gal, D.; Lischinsky, S.; Friedman, M.; Zinder, O. Prediction of the presence of ovarian cancer at surgery by an immunochemical panel: CA 125 and copper-to-zinc ratio. Gynecol. Oncol. 1989, 35, 246–250. [Google Scholar] [CrossRef]
- Gupta, S.K.; Singh, S.P.; Shukla, V.K. Copper, zinc, and Cu/Zn ratio in carcinoma of the gallbladde. J. Surg. Oncol. 2005, 91, 204–208. [Google Scholar] [CrossRef]
- Issell, B.F.; MacFadyen, B.V.; Gum, E.T.; Valdivieso, M.; Dudrick, S.J.; Bodey, G.P. Serum zinc levels in lung cancer patients. Cancer 1981, 47, 1845–1848. [Google Scholar] [CrossRef]
- Buntzel, J.; Bruns, F.; Glatzel, M.; Garayev, A.; Mucke, R.; Kisters, K.; Schafer, U.; Schonekaes, K.; Micke, O. Zinc concentrations in serum during head and neck cancer progression. Anticancer Res. 2007, 27, 1941–1943. [Google Scholar]
- Schlag, P.; Seeling, W.; Merkle, P.; Betzler, M. Changes of serum-zinc in breast cancer (author’s transl) (in German). Langenbecks Arch. Chir. 1978, 346, 129–133. [Google Scholar] [CrossRef]
- Kuo, H.W.; Chen, S.F.; Wu, C.C.; Chen, D.R.; Lee, J.H. Serum and tissue trace elements in patients with breast cancer in Taiwan. Biol. TraceElem. Res. 2002, 89, 1–11. [Google Scholar] [CrossRef]
- Adzersen, K.H.; Jess, P.; Freivogel, K.W.; Gerhard, I.; Bastert, G. Raw and cooked vegetables, fruits, selected micronutrients, and breast cancer risk: A case-control study in Germany. Nutr. Cancer 2003, 46, 131–137. [Google Scholar] [CrossRef]
- Oyama, T.; Kawamoto, T.; Matsuno, K.; Osaki, T.; Matsumoto, A.; Isse, T.; Nakata, S.; Ozaki, S.; Sugaya, M.; Yasuda, M.; et al. A case-case study comparing the usefulness of serum trace elements (Cu, Zn and Se) and tumor markers (CEA, SCC and SLX) in non-small cell lung cancer patients. Anticancer Res. 2003, 23, 605–612. [Google Scholar]
- Oyama, T.; Matsuno, K.; Kawamoto, T.; Mitsudomi, T.; Shirakusa, T.; Kodama, Y. Efficiency of serum copper/zinc ratio for differential diagnosis of patients with and without lung cancer. Biol. Trace Elem. Res. 1994, 42, 115–127. [Google Scholar] [CrossRef]
- Kopanski, Z.; Piekoszewski, W.; Habiniak, J.; Wojewoda, T.; Wojewoda, A.; Schlegel-Zawadzka, M.; Sibiga, W. The clinical value of the determinations in the serum of zinc concentration in women with breast cancer. Folia Histochem. Cytobiol. 2001, 39, 84–86. [Google Scholar]
- Gibson, R.S. Assessment of trace Element Status. In Principles of Nutritional Assessment; Gibson, R.S., Ed.; Oxford University Press: New York, NY, USA, 1990. [Google Scholar]
- Tinoco-Veras, C.M.; Bezerra Sousa, M.S.; da Silva, B.B.; Franciscato Cozzolino, S.M.; Viana Pires, L.; Coelho Pimentel, J.A.; do Nascimento-Nogueira, N.; do Nascimento-Marreiro, D. Analysis of plasma and erythrocyte zinc levels in premenopausal women with breast cancer. Nutr. Hosp. 2011, 26, 293–297. [Google Scholar]
- Geraki, K.; Farquharson, M.J.; Bradley, D.A. Concentrations of Fe, Cu and Zn in breast tissue: A synchrotron XRF study. Phys. Med. Biol. 2002, 47, 2327–2339. [Google Scholar] [CrossRef]
- Geraki, K.; Farquharson, M.J.; Bradley, D.A. X-ray fluorescence and energy dispersive X-ray diffraction for the quantification of elemental concentrations in breast tissue. Phys. Med. Biol. 2004, 49, 99–110. [Google Scholar]
- Margalioth, E.J.; Schenker, J.G.; Chevion, M. Copper and zinc levels in normal and malignant tissues. Cancer 1983, 52, 868–872. [Google Scholar] [CrossRef]
- Santoliquido, P.M.; Southwick, H.W.; Olwin, J.H. Trace metal levels in cancer of the breast. Surg. Gynecol. Obstet. 1976, 142, 65–70. [Google Scholar]
- Cui, Y.; Vogt, S.; Olson, N.; Glass, A.G.; Rohan, T.E. Levels of zinc, selenium, calcium, and iron in benign breast tissue and risk of subsequent breast cancer. Cancer Epidemiol. Biomark. Prev. 2007, 16, 1682–1685. [Google Scholar] [CrossRef]
- Kagara, N.; Tanaka, N.; Noguchi, S.; Hirano, T. Zinc and its transporter ZIP10 are involved in invasive behavior of breast cancer cells. Cancer Sci. 2007, 98, 692–697. [Google Scholar] [CrossRef]
- Taylor, K.M. LIV-1 breast cancer protein belongs to new family of histidine-rich membrane proteins with potential to control intracellular Zn2+ homeostasis. IUBMB Life 2000, 49, 249–253. [Google Scholar] [CrossRef]
- Taylor, K.M.; Vichova, P.; Jordan, N.; Hiscox, S.; Hendley, R.; Nicholson, R.I. ZIP7-mediated intracellular zinc transport contributes to aberrant growth factor signaling in antihormone-resistant breast cancer cells. Endocrinology 2008, 149, 4912–4920. [Google Scholar] [CrossRef]
- Lipman, T.O.; Diamond, A.; Mellow, M.H.; Patterson, K.Y. Esophageal zinc content in human squamous esophageal cancer. J. Am. Coll. Nutr. 1987, 6, 41–46. [Google Scholar]
- Mellow, M.H.; Layne, E.A.; Lipman, T.O.; Kaushik, M.; Hostetler, C.; Smith, J.C., Jr. Plasma zinc and vitamin A in human squamous carcinoma of the esophagus. Cancer 1983, 51, 1615–1620. [Google Scholar]
- Chakravarty, P.K.; Ghosh, A.; Chowdhury, J.R. Zinc in human malignancies. Neoplasma 1986, 33, 85–90. [Google Scholar]
- Costello, L.C.; Feng, P.; Milon, B.; Tan, M.; Franklin, R.B. Role of zinc in the pathogenesis and treatment of prostate cancer: Critical issues to resolve. Prostate Cancer Prostatic Dis. 2004, 7, 111–117. [Google Scholar] [CrossRef]
- Li, M.; Zhang, Y.; Liu, Z.; Bharadwaj, U.; Wang, H.; Wang, X.; Zhang, S.; Liuzzi, J.P.; Chang, S.M.; Cousins, R.J.; et al. Aberrant expression of zinc transporter ZIP4 (SLC39A4) significantly contributes to human pancreatic cancer pathogenesis and progression. Proc. Natl. Acad. Sci. USA 2007, 104, 18636–18641. [Google Scholar]
- Wu, T.; Sempos, C.T.; Freudenheim, J.L.; Muti, P.; Smit, E. Serum iron, copper and zinc concentrations and risk of cancer mortality in US adults. Ann. Epidemiol. 2004, 14, 195–201. [Google Scholar] [CrossRef]
- Golub, M.S.; Gershwin, M.E.; Hurley, L.S.; Hendrickx, A.G.; Saito, W.Y. Studies of marginal zinc deprivation in rhesus monkeys: Infant behavior. Am. J. Clin. Nutr. 1985, 42, 1229–1239. [Google Scholar]
- Oteiza, P.I.; Olin, K.L.; Fraga, C.G.; Keen, C.L. Zinc deficiency causes oxidative damage to proteins, lipids and DNA in rat testes. J. Nutr. 1995, 125, 823–829. [Google Scholar]
- Oteiza, P.I.; Clegg, M.S.; Zago, M.P.; Keen, C.L. Zinc deficiency induces oxidative stress and AP-1 activation in 3T3 cells. Free Radic. Biol. Med. 2000, 28, 1091–1099. [Google Scholar] [CrossRef]
- Dreosti, I.E. Zinc and the gene. Mutat. Res. 2001, 475, 161–167. [Google Scholar] [CrossRef]
- Ames, B.N.; Gold, L.S. Paracelsus to parascience: The environmental cancer distraction. Mutat. Res. 2000, 447, 3–13. [Google Scholar] [CrossRef]
- Blount, B.C.; Mack, M.M.; Wehr, C.M.; MacGregor, J.T.; Hiatt, R.A.; Wang, G.; Wickramasinghe, S.N.; Everson, R.B.; Ames, B.N. Folate deficiency causes uracil misincorporation into human DNA and chromosome breakage: Implications for cancer and neuronal damage. Proc. Natl. Acad. Sci. USA 1997, 94, 3290–3295. [Google Scholar]
- Prasad, A.S.; Beck, F.W.; Bao, B.; Fitzgerald, J.T.; Snell, D.C.; Steinberg, J.D.; Cardozo, L.J. Zinc supplementation decreases incidence of infections in the elderly: Effect of zinc on generation of cytokines and oxidative stress. Am. J. Clin. Nutr. 2007, 85, 837–844. [Google Scholar]
- Ho, E.; Ames, B.N. Low intracellular zinc induces oxidative DNA damage, disrupts p53, NFkappa B, and AP1 DNA binding, and affects DNA repair in a rat glioma cell line. Proc. Natl. Acad. Sci. USA 2002, 99, 16770–16775. [Google Scholar]
- Vallee, B.L.; Auld, D.S. Zinc metallochemistry in biochemistry. EXS 1995, 73, 259–277. [Google Scholar]
- Maret, W. Zinc coordination environments in proteins as redox sensors and signal transducers. Antioxid. RedoxSignal. 2006, 8, 1419–1441. [Google Scholar] [CrossRef]
- Zeng, J.; Heuchel, R.; Schaffner, W.; Kagi, J.H. Thionein (apometallothionein) can modulate DNA binding and transcription activation by zinc finger containing factor Sp1. FEBSLett. 1991, 279, 310–312. [Google Scholar] [CrossRef]
- Zeng, J.; Vallee, B.L.; Kagi, J.H. Zinc transfer from transcription factor IIIA fingers to thionein clusters. Proc. Natl. Acad. Sci. USA 1991, 88, 9984–9988. [Google Scholar]
- Zangger, K.; Oz, G.; Haslinger, E.; Kunert, O.; Armitage, I.M. Nitric oxide selectively releases metals from the amino-terminal domain of metallothioneins: Potential role at inflammatory sites. FASEB J. 2001, 15, 1303–1305. [Google Scholar]
- Sato, M.; Bremner, I. Oxygen free radicals and metallothionein. Free Radic. Biol. Med. 1993, 14, 325–337. [Google Scholar] [CrossRef]
- Cai, L.; Klein, J.B.; Kang, Y.J. Metallothionein inhibits peroxynitrite-induced DNA and lipoprotein damage. J. Biol. Chem. 2000, 275, 38957–38960. [Google Scholar] [CrossRef]
- Surowiak, P.; Matkowski, R.; Materna, V.; Gyorffy, B.; Wojnar, A.; Pudelko, M.; Dziegiel, P.; Kornafel, J.; Zabel, M. Elevated metallothionein (MT) expression in invasive ductal breast cancers predicts tamoxifen resistance. Histol. Histopathol. 2005, 20, 1037–1044. [Google Scholar]
- Petros, W.P.; Hopkins, P.J.; Spruill, S.; Broadwater, G.; Vredenburgh, J.J.; Colvin, O.M.; Peters, W.P.; Jones, R.B.; Hall, J.; Marks, J.R. Associations between drug metabolism genotype, chemotherapy pharmacokinetics, and overall survival in patients with breast cancer. J. Clin. Oncol. 2005, 23, 6117–6125. [Google Scholar]
- Cherian, M.G.; Howell, S.B.; Imura, N.; Klaassen, C.D.; Koropatnick, J.; Lazo, J.S.; Waalkes, M.P. Role of metallothionein in carcinogenesis. Toxicol. Appl. Pharmacol. 1994, 126, 1–5. [Google Scholar] [CrossRef]
- Theocharis, S.E.; Margeli, A.P.; Koutselinis, A. Metallothionein: A multifunctional protein from toxicity to cancer. Int. J. Biol. Markers 2003, 18, 162–169. [Google Scholar]
- Yap, X.; Tan, H.Y.; Huang, J.; Lai, Y.; Yip, G.W.; Tan, P.H.; Bay, B.H. Over-expression of metallothionein predicts chemoresistance in breast cancer. J. Pathol. 2009, 217, 563–570. [Google Scholar] [CrossRef]
- Jin, R.; Bay, B.H.; Chow, V.T.; Tan, P.H. Metallothionein 1F mRNA expression correlates with histological grade in breast carcinoma. Breast Cancer Res. Treat. 2001, 66, 265–272. [Google Scholar] [CrossRef]
- Bakka, A.; Endresen, L.; Johnsen, A.B.; Edminson, P.D.; Rugstad, H.E. Resistance against cis-dichlorodiammineplatinum in cultured cells with a high content of metallothionein. Toxicol. Appl. Pharmacol. 1981, 61, 215–226. [Google Scholar] [CrossRef]
- Satoh, M.; Cherian, M.G.; Imura, N.; Shimizu, H. Modulation of resistance to anticancer drugs by inhibition of metallothionein synthesis. Cancer Res. 1994, 54, 5255–5257. [Google Scholar]
- Bier, B.; Douglas-Jones, A.; Totsch, M.; Dockhorn-Dworniczak, B.; Bocker, W.; Jasani, B.; Schmid, K.W. Immunohistochemical demonstration of metallothionein in normal human breast tissue and benign and malignant breast lesions. Breast Cancer Res. Treat. 1994, 30, 213–221. [Google Scholar] [CrossRef]
- Fresno, M.; Wu, W.; Rodriguez, J.M.; Nadji, M. Localization of metallothionein in breast carcinomas. An immunohistochemical study. Virchows Arch. APathol. Anat. Histopathol. 1993, 423, 215–219. [Google Scholar] [CrossRef]
- Oyama, T.; Take, H.; Hikino, T.; Iino, Y.; Nakajima, T. Immunohistochemical expression of metallothionein in invasive breast cancer in relation to proliferative activity, histology and prognosis. Oncology 1996, 53, 112–117. [Google Scholar] [CrossRef]
- Witkiewicz-Kucharczyk, A.; Bal, W. Damage of zinc fingers in DNA repair proteins, a novel molecular mechanism in carcinogenesis. Toxicol. Lett. 2006, 162, 29–42. [Google Scholar]
- Harris, C.C. Structure and function of the p53 tumor suppressor gene: Clues for rational cancer therapeutic strategies. J. Natl. Cancer Inst. 1996, 88, 1442–1455. [Google Scholar] [CrossRef]
- Gasco, M.; Shami, S.; Crook, T. The p53 pathway in breast cancer. Breast Cancer Res. 2002, 4, 70–76. [Google Scholar]
- Bartek, J.; Lukas, J. Pathways governing G1/S transition and their response to DNA damage. FEBS Lett. 2001, 490, 117–122. [Google Scholar] [CrossRef]
- Hainaut, P.; Milner, J. A structural role for metal ions in the “wild-type” conformation of the tumor suppressor protein p53. Cancer Res. 1993, 53, 1739–1742. [Google Scholar]
- Cho, Y.; Gorina, S.; Jeffrey, P.D.; Pavletich, N.P. Crystal structure of a p53 tumor suppressor-DNA complex: Understanding tumorigenic mutations. Science 1994, 265, 346–355. [Google Scholar]
- Meplan, C.; Richard, M.J.; Hainaut, P. Metalloregulation of the tumor suppressor protein p53: Zinc mediates the renaturation of p53 after exposure to metal chelators in vitro and in intact cells. Oncogene 2000, 19, 5227–5236. [Google Scholar] [CrossRef]
- Meplan, C.; Richard, M.J.; Hainaut, P. Redox signalling and transition metals in the control of the p53 pathway. Biochem. Pharmacol. 2000, 59, 25–33. [Google Scholar]
- Abdel-Mageed, A.; Agrawal, K.C. Antisense down-regulation of metallothionein induces growth arrest and apoptosis in human breast carcinoma cells. Cancer Gene Ther. 1997, 4, 199–207. [Google Scholar]
- Chae, B.J.; Bae, J.S.; Lee, A.; Park, W.C.; Seo, Y.J.; Song, B.J.; Kim, J.S.; Jung, S.S. p53 as a specific prognostic factor in triple-negative breast cancer. Jpn. J. Clin. Oncol. 2009, 39, 217–224. [Google Scholar] [CrossRef]
- Rossner, P., Jr.; Gammon, M.D.; Zhang, Y.J.; Terry, M.B.; Hibshoosh, H.; Memeo, L.; Mansukhani, M.; Long, C.M.; Garbowski, G.; Agrawal, M.; et al. Mutations in p53, p53 protein overexpression and breast cancer survival. J. Cell. Mol. Med. 2009, 13, 3847–3857. [Google Scholar] [CrossRef]
- Guo, Z.; Kozlov, S.; Lavin, M.F.; Person, M.D.; Paull, T.T. ATM activation by oxidative stress. Science 2010, 330, 517–521. [Google Scholar]
- So, S.; Davis, A.J.; Chen, D.J. Autophosphorylation at serine 1981 stabilizes ATM at DNA damage sites. J. Cell Biol. 2009, 187, 977–990. [Google Scholar] [CrossRef]
- Lavin, M.F. ATM and the Mre11 complex combine to recognize and signal DNA double-strand breaks. Oncogene 2007, 26, 7749–7758. [Google Scholar] [CrossRef]
- Venkitaraman, A.R. Cancer susceptibility and the functions of BRCA1 and BRCA2. Cell 2002, 108, 171–182. [Google Scholar] [CrossRef]
- MacLachlan, T.K.; El-Deiry, W.S. Pointing (zinc) fingers at BRCA1 targets. Nat. Med. 2000, 6, 1318–1319. [Google Scholar] [CrossRef]
- Roehm, P.C.; Berg, J.M. Sequential metal binding by the RING finger domain of BRCA1. Biochemistry 1997, 36, 10240–10245. [Google Scholar] [CrossRef]
- Chesters, J.K.; Petrie, L.; Vint, H. Specificity and timing of the Zn2+ requirement for DNA synthesis by 3T3 cells. Exp. CellRes. 1989, 184, 499–508. [Google Scholar] [CrossRef]
- Chesters, J.K.; Petrie, L.; Travis, A.J. A requirement for Zn2+ for the induction of thymidine kinase but not ornithine decarboxylase in 3T3 cells stimulated from quiescence. Biochem. J. 1990, 272, 525–527. [Google Scholar]
- Chesters, J.K.; Boyne, R. Nature of the Zn2+ requirement for DNA synthesis by 3T3 cells. Exp. CellRes. 1991, 192, 631–634. [Google Scholar] [CrossRef]
- Chesters, J.K.; Petrie, L. A possible role for cyclins in the zinc requirements during G1 and G2 phases of the cell cycle. J. Nutr. Biochem. 1999, 10, 279–290. [Google Scholar] [CrossRef]
- Prasad, A.S.; Beck, F.W.; Endre, L.; Handschu, W.; Kukuruga, M.; Kumar, G. Zinc deficiency affects cell cycle and deoxythymidine kinase gene expression in HUT-78 cells. J. Lab. Clin. Med. 1996, 128, 51–60. [Google Scholar] [CrossRef]
- Wu, F.Y.; Huang, W.J.; Sinclair, R.B.; Powers, L. The structure of the zinc sites of Escherichia coli DNA-dependent RNA polymerase. J. Biol. Chem. 1992, 267, 25560–25567. [Google Scholar]
- Springgate, C.F.; Mildvan, A.S.; Abramson, R.; Engle, J.L.; Loeb, L.A. Escherichia coli deoxyribonucleic acid polymerase I, a zinc metalloenzyme. Nuclear quadrupolar relaxation studies of the role of bound zinc. J. Biol. Chem. 1973, 248, 5987–5993. [Google Scholar]
- Hicks, S.E.; Wallwork, J.C. Effect of dietary zinc deficiency on protein synthesis in cell-free systems isolated from rat liver. J. Nutr. 1987, 117, 1234–1240. [Google Scholar]
- Hard, T.; Rak, A.; Allard, P.; Kloo, L.; Garber, M. The solution structure of ribosomal protein L36 from Thermus thermophilus reveals a zinc-ribbon-like fold. J. Mol. Biol. 2000, 296, 169–180. [Google Scholar] [CrossRef]
- Ducruet, A.P.; Vogt, A.; Wipf, P.; Lazo, J.S. Dual specificity protein phosphatases: Therapeutic targets for cancer and Alzheimer’s disease. Annu. Rev. Pharmacol. Toxicol. 2005, 45, 725–750. [Google Scholar] [CrossRef]
- Sun, L.; Chai, Y.; Hannigan, R.; Bhogaraju, V.K.; Machaca, K. Zinc regulates the ability of Cdc25C to activate MPF/cdk1. J. Cell. Physiol. 2007, 213, 98–104. [Google Scholar] [CrossRef]
- Li, Y.; Maret, W. Transient fluctuations of intracellular zinc ions in cell proliferation. Exp. CellRes. 2009, 315, 2463–2470. [Google Scholar] [CrossRef]
- Hansson, A. Extracellular zinc ions induces mitogen-activated protein kinase activity and protein tyrosine phosphorylation in bombesin-sensitive Swiss 3T3 fibroblasts. Arch. Biochem. Biophys. 1996, 328, 233–238. [Google Scholar] [CrossRef]
- Kim, Y.M.; Reed, W.; Wu, W.; Bromberg, P.A.; Graves, L.M.; Samet, J.M. Zn2+-induced IL-8 expression involves AP-1, JNK, and ERK activities in human airway epithelial cells. Am. J. Physiol. Lung Cell. Mol. Physiol. 2006, 290, L1028–L1035. [Google Scholar] [CrossRef]
- Ho, Y.; Samarasinghe, R.; Knoch, M.E.; Lewis, M.; Aizenman, E.; DeFranco, D.B. Selective inhibition of mitogen-activated protein kinase phosphatases by zinc accounts for extracellular signal-regulated kinase 1/2-dependent oxidative neuronal cell death. Mol. Pharmacol. 2008, 74, 1141–1151. [Google Scholar]
- Haase, H.; Maret, W. Intracellular zinc fluctuations modulate protein tyrosine phosphatase activity in insulin/insulin-like growth factor-1 signaling. Exp. CellRes. 2003, 291, 289–298. [Google Scholar] [CrossRef]
- Wu, W.; Graves, L.M.; Gill, G.N.; Parsons, S.J.; Samet, J.M. Src-dependent phosphorylation of the epidermal growth factor receptor on tyrosine 845 is required for zinc-induced Ras activation. J. Biol. Chem. 2002, 277, 24252–24257. [Google Scholar]
- Wu, W.; Graves, L.M.; Jaspers, I.; Devlin, R.B.; Reed, W.; Samet, J.M. Activation of the EGF receptor signaling pathway in human airway epithelial cells exposed to metals. Am. J. Physiol. 1999, 277, L924–L931. [Google Scholar]
- Bourdeau, A.; Dube, N.; Tremblay, M.L. Cytoplasmic protein tyrosine phosphatases, regulation and function: The roles of PTP1B and TC-PTP. Curr. Opin. CellBiol. 2005, 17, 203–209. [Google Scholar]
- McCusker, R.H.; Kaleko, M.; Sackett, R.L. Multivalent cations and ligand affinity of the type 1 insulin-like growth factor receptor on P2A2-LISN muscle cells. J. Cell. Physiol. 1998, 176, 392–401. [Google Scholar]
- Cossack, Z.T. Decline in somatomedin-C (insulin-like growth factor-1) with experimentally induced zinc deficiency in human subjects. Clin. Nutr. 1991, 10, 284–291. [Google Scholar] [CrossRef]
- Lefebvre, D.; Boney, C.M.; Ketelslegers, J.M.; Thissen, J.P. Inhibition of insulin-like growth factor-I mitogenic action by zinc chelation is associated with a decreased mitogen-activated protein kinase activation in RAT-1 fibroblasts. FEBS Lett. 1999, 449, 284–288. [Google Scholar] [CrossRef]
- Krezel, A.; Maret, W. Thionein/metallothionein control Zn(II) availability and the activity of enzymes. J. Biol. Inorg. Chem. 2008, 13, 401–409. [Google Scholar] [CrossRef]
- Clegg, M.S.; Hanna, L.A.; Niles, B.J.; Momma, T.Y.; Keen, C.L. Zinc deficiency-induced cell death. IUBMB Life 2005, 57, 661–669. [Google Scholar] [CrossRef]
- Claerhout, S.; Decraene, D.; Van Laethem, A.; Van Kelst, S.; Agostinis, P.; Garmyn, M. AKT delays the early-activated apoptotic pathway in UVB-irradiated keratinocytes via BAD translocation. J. Invest. Dermatol. 2007, 127, 429–438. [Google Scholar]
- Yang, X.; Cao, W.; Zhang, L.; Zhang, W.; Zhang, X.; Lin, H. Targeting 14-3-3zeta in cancer therapy. Cancer Gene Ther. 2012, 19, 153–159. [Google Scholar]
- King, L.E.; Osati-Ashtiani, F.; Fraker, P.J. Apoptosis plays a distinct role in the loss of precursor lymphocytes during zinc deficiency in mice. J. Nutr. 2002, 132, 974–979. [Google Scholar]
- Duffy, J.Y.; Miller, C.M.; Rutschilling, G.L.; Ridder, G.M.; Clegg, M.S.; Keen, C.L.; Daston, G.P. A decrease in intracellular zinc level precedes the detection of early indicators of apoptosis in HL-60 cells. Apoptosis 2001, 6, 161–172. [Google Scholar] [CrossRef]
- Kolenko, V.M.; Uzzo, R.G.; Dulin, N.; Hauzman, E.; Bukowski, R.; Finke, J.H. Mechanism of apoptosis induced by zinc deficiency in peripheral blood T lymphocytes. Apoptosis 2001, 6, 419–429. [Google Scholar] [CrossRef]
- Chimienti, F.; Seve, M.; Richard, S.; Mathieu, J.; Favier, A. Role of cellular zinc in programmed cell death: Temporal relationship between zinc depletion, activation of caspases, and cleavage of Sp family transcription factors. Biochem. Pharmacol. 2001, 62, 51–62. [Google Scholar]
- Takahashi, A.; Alnemri, E.S.; Lazebnik, Y.A.; Fernandes-Alnemri, T.; Litwack, G.; Moir, R.D.; Goldman, R.D.; Poirier, G.G.; Kaufmann, S.H.; Earnshaw, W.C. Cleavage of lamin A by Mch2 alpha but not CPP32: Multiple interleukin 1 beta-converting enzyme-related proteases with distinct substrate recognition properties are active in apoptosis. Proc. Natl. Acad. Sci. USA 1996, 93, 8395–8400. [Google Scholar]
- Jankowski-Hennig, M.A.; Clegg, M.S.; Daston, G.P.; Rogers, J.M.; Keen, C.L. Zinc-deficient rat embryos have increased caspase 3-like activity and apoptosis. Biochem. Biophys. Res. Commun. 2000, 271, 250–256. [Google Scholar]
- Hashemi, M.; Ghavami, S.; Eshraghi, M.; Booy, E.P.; Los, M. Cytotoxic effects of intra and extracellular zinc chelation on human breast cancer cells. Eur. J. Pharmacol. 2007, 557, 9–19. [Google Scholar] [CrossRef]
- Mackenzie, G.G.; Keen, C.L.; Oteiza, P.I. Zinc status of human IMR-32 neuroblastoma cells influences their susceptibility to iron-induced oxidative stress. Dev. Neurosci. 2002, 24, 125–133. [Google Scholar] [CrossRef]
- Cadenas, E. Mitochondrial free radical production and cell signaling. Mol. Aspects Med. 2004, 25, 17–26. [Google Scholar] [CrossRef]
- Gumulec, J.; Masarik, M.; Krizkova, S.; Adam, V.; Hubalek, J.; Hrabeta, J.; Eckschlager, T.; Stiborova, M.; Kizek, R. Insight to physiology and pathology of zinc(II) ions and their actions in breast and prostate carcinoma. Curr. Med. Chem. 2011, 18, 5041–5051. [Google Scholar] [CrossRef]
- Ostrakhovitch, E.A.; Cherian, M.G. Role of p53 and reactive oxygen species in apoptotic response to copper and zinc in epithelial breast cancer cells. Apoptosis 2005, 10, 111–121. [Google Scholar] [CrossRef]
- Hwang, J.J.; Kim, H.N.; Kim, J.; Cho, D.H.; Kim, M.J.; Kim, Y.S.; Kim, Y.; Park, S.J.; Koh, J.Y. Zinc(II) ion mediates tamoxifen-induced autophagy and cell death in MCF-7 breast cancer cell line. Biometals 2010, 23, 997–1013. [Google Scholar] [CrossRef]
- Hadler-Olsen, E.; Fadnes, B.; Sylte, I.; Uhlin-Hansen, L.; Winberg, J.O. Regulation of matrix metalloproteinase activity in health and disease. FEBS J. 2011, 278, 28–45. [Google Scholar] [CrossRef]
- Benaud, C.; Dickson, R.B.; Thompson, E.W. Roles of the matrix metalloproteinases in mammary gland development and cancer. Breast Cancer Res. Treat. 1998, 50, 97–116. [Google Scholar] [CrossRef]
- Radisky, E.S.; Radisky, D.C. Matrix metalloproteinase-induced epithelial-mesenchymal transition in breast cancer. J. Mammary Gland Biol. Neoplasia 2010, 15, 201–212. [Google Scholar] [CrossRef]
- Overall, C.M. Molecular determinants of metalloproteinase substrate specificity: Matrix metalloproteinase substrate binding domains, modules, and exosite. Mol. Biotechnol. 2002, 22, 51–86. [Google Scholar] [CrossRef]
- Birkedal-Hansen, H.; Moore, W.G.; Bodden, M.K.; Windsor, L.J.; Birkedal-Hansen, B.; DeCarlo, A.; Engler, J.A. Matrix metalloproteinases: A review. Crit. Rev. Oral Biol. Med. 1993, 4, 197–250. [Google Scholar]
- Folgueras, A.R.; Pendas, A.M.; Sanchez, L.M.; Lopez-Otin, C. Matrix metalloproteinases in cancer: From new functions to improved inhibition strategies. Int. J. Dev. Biol. 2004, 48, 411–424. [Google Scholar] [CrossRef]
- Klein, T.; Bischoff, R. Physiology and pathophysiology of matrix metalloproteases. Amino Acids 2011, 41, 271–290. [Google Scholar] [CrossRef]
- Kambe, T. An overview of a wide range of functions of ZnT and Zip zinc transporters in the secretory pathway. Biosci. Biotechnol. Biochem. 2011, 75, 1036–1043. [Google Scholar] [CrossRef]
- Kelleher, S.L.; Seo, Y.A.; Lopez, V. Mammary gland zinc metabolism: Regulation and dysregulation. Genes Nutr. 2009, 4, 83–94. [Google Scholar] [CrossRef]
- Seo, Y.A.; Lopez, V.; Kelleher, S.L. A histidine-rich motif mediates mitochondrial localization of ZnT2 to modulate mitochondrial function. Am. J. Physiol. Cell Physiol. 2011, 300, C1479–C1489. [Google Scholar] [CrossRef]
- Kelleher, S.L.; McCormick, N.H.; Velasquez, V.; Lopez, V. Zinc in specialized secretory tissues: Roles in the pancreas, prostate, and mammary gland. Adv. Nutr. 2011, 2, 101–111. [Google Scholar] [CrossRef]
- Xue, L.; Li, G.; Yu, C.; Jiang, H. A ratiometric and targetable fluorescent sensor for quantification of mitochondrial zinc ions. Chemistry 2012, 18, 1050–1054. [Google Scholar]
- Zalewski, P.D.; Forbes, I.J.; Betts, W.H. Correlation of apoptosis with change in intracellular labile Zn(II) using zinquin [(2-methyl-8-p-toluenesulphonamido-6-quinolyloxy)acetic acid], a new specific fluorescent probe for Zn(II). Biochem. J. 1993, 296, 403–408. [Google Scholar]
- Haase, H.; Beyersmann, D. Uptake and intracellular distribution of labile and total Zn(II) in C6 rat glioma cells investigated with fluorescent probes and atomic absorption. Biometals 1999, 12, 247–254. [Google Scholar] [CrossRef]
- Dunn, K.W.; Mayor, S.; Myers, J.N.; Maxfield, F.R. Applications of ratio fluorescence microscopy in the study of cell physiology. FASEB J. 1994, 8, 573–582. [Google Scholar]
- Ma, H.; Gibson, E.A.; Dittmer, P.J.; Jimenez, R.; Palmer, A.E. High-throughput examination of fluorescence resonance energy transfer-detected metal-ion response in mammalian cells. J. Am. Chem. Soc. 2012, 134, 2488–2491. [Google Scholar]
- Haase, H.; Beyersmann, D. Intracellular zinc distribution and transport in C6 rat glioma cells. Biochem. Biophys. Res. Commun. 2002, 296, 923–928. [Google Scholar] [CrossRef]
- Lichten, L.A.; Cousins, R.J. Mammalian zinc transporters: Nutritional and physiologic regulation. Annu. Rev. Nutr. 2009, 29, 153–176. [Google Scholar] [CrossRef]
- MacDonald, R.S. The role of zinc in growth and cell proliferation. J. Nutr. 2000, 130, 1500S–1508S. [Google Scholar]
- Lopez, V.; Foolad, F.; Kelleher, S.L. ZnT2-overexpression represses the cytotoxic effects of zinc hyper-accumulation in malignant metallothionein-null T47D breast tumor cells. Cancer Lett. 2011, 304, 41–51. [Google Scholar] [CrossRef]
- Schneider, J.; Ruschhaupt, M.; Buness, A.; Asslaber, M.; Regitnig, P.; Zatloukal, K.; Schippinger, W.; Ploner, F.; Poustka, A.; Sultmann, H. Identification and meta-analysis of a small gene expression signature for the diagnosis of estrogen receptor status in invasive ductal breast cancer. Int. J. Cancer 2006, 119, 2974–2979. [Google Scholar]
- Tozlu, S.; Girault, I.; Vacher, S.; Vendrell, J.; Andrieu, C.; Spyratos, F.; Cohen, P.; Lidereau, R.; Bieche, I. Identification of novel genes that co-cluster with estrogen receptor alpha in breast tumor biopsy specimens, using a large-scale real-time reverse transcription-PCR approach. Endocr. Relat. Cancer 2006, 13, 1109–1120. [Google Scholar] [CrossRef]
- Taylor, K.M. A distinct role in breast cancer for two LIV-1 family zinc transporters. Biochem. Soc. Trans. 2008, 36, 1247–1251. [Google Scholar] [CrossRef]
- El-Tanani, M.K.; Green, C.D. Interaction between estradiol and growth factors in the regulation of specific gene expression in MCF-7 human breast cancer cells. J. Steroid Biochem. Mol. Biol. 1997, 60, 269–276. [Google Scholar] [CrossRef]
- Manning, D.L.; Daly, R.J.; Lord, P.G.; Kelly, K.F.; Green, C.D. Effects of oestrogen on the expression of a 4.4 kb mRNA in the ZR-75-1 human breast cancer cell line. Mol. Cell. Endocrinol. 1988, 59, 205–212. [Google Scholar] [CrossRef]
- McClelland, R.A.; Manning, D.L.; Gee, J.M.; Willsher, P.; Robertson, J.F.; Ellis, I.O.; Blamey, R.W.; Nicholson, R.I. Oestrogen-regulated genes in breast cancer: Association of pLIV1 with response to endocrine therapy. Br. J. Cancer 1998, 77, 1653–1656. [Google Scholar] [CrossRef]
- Taylor, K.M.; Nicholson, R.I. The LZT proteins; the LIV-1 subfamily of zinc transporters. Biochim. Biophysic. Acta. 2003, 1611, 16–30. [Google Scholar]
- Manning, D.L.; McClelland, R.A.; Gee, J.M.; Chan, C.M.; Green, C.D.; Blamey, R.W.; Nicholson, R.I. The role of four oestrogen-responsive genes, pLIV1, pS2, pSYD3 and pSYD8, in predicting responsiveness to endocrine therapy in primary breast cancer. Eur. J. Cancer 1993, 29A, 1462–1468. [Google Scholar]
- Manning, D.L.; Robertson, J.F.; Ellis, I.O.; Elston, C.W.; McClelland, R.A.; Gee, J.M.; Jones, R.J.; Green, C.D.; Cannon, P.; Blamey, R.W.; et al. Oestrogen-regulated genes in breast cancer: Association of pLIV1 with lymph node involvement. Eur. J. Cancer 1994, 30A, 675–678. [Google Scholar]
- Cousins, R.J.; Liuzzi, J.P.; Lichten, L.A. Mammalian zinc transport, trafficking, and signal. J. Biol. Chem. 2006, 281, 24085–24089. [Google Scholar]
- Yamashita, S.; Miyagi, C.; Fukada, T.; Kagara, N.; Che, Y.S.; Hirano, T. Zinc transporter LIVI controls epithelial-mesenchymal transition in zebrafish gastrula organizer. Nature 2004, 429, 298–302. [Google Scholar]
- Cano, A.; Perez-Moreno, M.A.; Rodrigo, I.; Locascio, A.; Blanco, M.J.; del Barrio, M.G.; Portillo, F.; Nieto, M.A. The transcription factor snail controls epithelial-mesenchymal transitions by repressing E-cadherin expression. Nat. Cell Biol. 2000, 2, 76–83. [Google Scholar]
- Zhou, B.P.; Hung, M.C. Wnt, hedgehog and snail: Sister pathways that control by GSK-3beta and beta-Trcp in the regulation of metastasis. Cell Cycle 2005, 4, 772–776. [Google Scholar] [CrossRef]
- Devarajan, E.; Huang, S. STAT3 as a central regulator of tumor metastases. Curr. Mol. Med. 2009, 9, 626–633. [Google Scholar] [CrossRef]
- Sorlie, T.; Perou, C.M.; Tibshirani, R.; Aas, T.; Geisler, S.; Johnsen, H.; Hastie, T.; Eisen, M.B.; van de Rijn, M.; Jeffrey, S.S.; et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl. Acad. Sci. USA 2001, 98, 10869–10874. [Google Scholar]
- Van’t Veer, L.J.; Paik, S.; Hayes, D.F. Gene expression profiling of breast cancer: A new tumor marker. J. Clin. Oncol. 2005, 23, 1631–1635. [Google Scholar] [CrossRef]
- Wang, Y.; Klijn, J.G.; Zhang, Y.; Sieuwerts, A.M.; Look, M.P.; Yang, F.; Talantov, D.; Timmermans, M.; Meijer-van Gelder, M.E.; Yu, J.; et al. Gene-expression profiles to predict distant metastasis of lymph-node-negative primary breast cancer. Lancet 2005, 365, 671–679. [Google Scholar]
- Shen, H.; Qin, H.; Guo, J. Concordant correlation of LIV-1 and E-cadherin expression in human breast cancer cell MCF-7. Mol. Biol. Rep. 2009, 36, 653–659. [Google Scholar] [CrossRef]
- Lopez, V.; Kelleher, S.L. Zip6-attenuation promotes epithelial-to-mesenchymal transition in ductal breast tumor (T47D) cells. Exp. CellRes. 2010, 316, 366–375. [Google Scholar] [CrossRef]
- Kasper, G.; Weiser, A.A.; Rump, A.; Sparbier, K.; Dahl, E.; Hartmann, A.; Wild, P.; Schwidetzky, U.; Castanos-Velez, E.; Lehmann, K. Expression levels of the putative zinc transporter LIV-1 are associated with a better outcome of breast cancer patients. Int. J. Cancer 2005, 117, 961–973. [Google Scholar] [CrossRef]
- Liuzzi, J.P.; Cousins, R.J. Mammalian zinc transporters. Annu. Rev. Nutr. 2004, 24, 151–172. [Google Scholar] [CrossRef]
- Taylor, K.M.; Morgan, H.E.; Smart, K.; Zahari, N.M.; Pumford, S.; Ellis, I.O.; Robertson, J.F.; Nicholson, R.I. The emerging role of the LIV-1 subfamily of zinc transporters in breast cancer. Mol. Med. 2007, 13, 396–406. [Google Scholar]
- Egeblad, M.; Werb, Z. New functions for the matrix metalloproteinases in cancer progression. Nat. Rev. Cancer 2002, 2, 161–174. [Google Scholar] [CrossRef]
- Orlichenko, L.S.; Radisky, D.C. Matrix metalloproteinases stimulate epithelial-mesenchymal transition during tumor development. Clin. Exp. Metastasis 2008, 25, 593–600. [Google Scholar] [CrossRef]
- McGuire, J.K.; Li, Q.; Parks, W.C. Matrilysin (matrix metalloproteinase-7) mediates E-cadherin ectodomain shedding in injured lung epithelium. Am. J. Pathol. 2003, 162, 1831–1843. [Google Scholar] [CrossRef]
- Sternlicht, M.D.; Werb, Z. How matrix metalloproteinases regulate cell behavior. Annu. Rev. Cell Dev. Biol. 2001, 17, 463–516. [Google Scholar] [CrossRef]
- Wu, Y.; Zhou, B.P. New insights of epithelial-mesenchymal transition in cancer metastasis. ActaBiochim. Biophys. Sin. 2008, 40, 643–650. [Google Scholar]
- Radisky, D.C.; Levy, D.D.; Littlepage, L.E.; Liu, H.; Nelson, C.M.; Fata, J.E.; Leake, D.; Godden, E.L.; Albertson, D.G.; Nieto, M.A.; et al. Rac1b and reactive oxygen species mediate MMP-3-induced EMT and genomic instability. Nature 2005, 436, 123–127. [Google Scholar]
- Thiery, J.P.; Sleeman, J.P. Complex networks orchestrate epithelial-mesenchymal transitions. Nat. Rev. Mol. Cell Biol. 2006, 7, 131–142. [Google Scholar] [CrossRef]
- Fukada, T.; Yamasaki, S.; Nishida, K.; Murakami, M.; Hirano, T. Zinc homeostasis and signaling in health and diseases: Zinc signaling. J. Biol. Inorg. Chem. 2011, 16, 1123–1134. [Google Scholar] [CrossRef]
- Taylor, K.M.; Hiscox, S.; Nicholson, R.I.; Hogstrand, C.; Kille, P. Protein kinase CK2 triggers cytosolic zinc signaling pathways by phosphorylation of zinc channel ZIP7. Sci. Signal. 2012, 5. [Google Scholar]
- Barr, S.; Thomson, S.; Buck, E.; Russo, S.; Petti, F.; Sujka-Kwok, I.; Eyzaguirre, A.; Rosenfeld-Franklin, M.; Gibson, N.W.; Miglarese, M.; et al. Bypassing cellular EGF receptor dependence through epithelial-to-mesenchymal-like transitions. Clin. Exp. Metastasis 2008, 25, 685–693. [Google Scholar] [CrossRef]
- Hogstrand, C.; Kille, P.; Nicholson, R.I.; Taylor, K.M. Zinc transporters and cancer: A potential role for ZIP7 as a hub for tyrosine kinase activation. Trends Mol. Med. 2009, 15, 101–111. [Google Scholar] [CrossRef]
- Chowanadisai, W.; Lonnerdal, B.; Kelleher, S.L. Identification of a mutation in SLC30A2 (ZnT-2) in women with low milk zinc concentration that results in transient neonatal zinc deficiency. J. Biol. Chem. 2006, 281, 39699–39707. [Google Scholar]
- Kelleher, S.L.; Lonnerdal, B. Zn transporter levels and localization change throughout lactation in rat mammary gland and are regulated by Zn in mammary cells. J. Nutr. 2003, 133, 3378–3385. [Google Scholar]
- Truong-Tran, A.Q.; Ho, L.H.; Chai, F.; Zalewski, P.D. Cellular zinc fluxes and the regulation of apoptosis/gene-directed cell death. J. Nutr. 2000, 130, 1459–1466. [Google Scholar]
- Ho, E.; Song, Y. Zinc and prostatic cancer. Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 640–645. [Google Scholar] [CrossRef]
- Costello, L.C.; Liu, Y.; Zou, J.; Franklin, R.B. Evidence for a zinc uptake transporter in human prostate cancer cells which is regulated by prolactin and testosterone. J. Biol. Chem. 1999, 274, 17499–17504. [Google Scholar]
- Franklin, R.B.; Ma, J.; Zou, J.; Guan, Z.; Kukoyi, B.I.; Feng, P.; Costello, L.C. Human ZIP1 is a major zinc uptake transporter for the accumulation of zinc in prostate cells. J. Inorg. Biochem. 2003, 96, 435–442. [Google Scholar] [CrossRef]
- Desouki, M.M.; Geradts, J.; Milon, B.; Franklin, R.B.; Costello, L.C. hZip2 and hZip3 zinc transporters are down regulated in human prostate adenocarcinomatous glands. Mol. Cancer 2007, 6, 37. [Google Scholar]
- Franklin, R.B.; Feng, P.; Milon, B.; Desouki, M.M.; Singh, K.K.; Kajdacsy-Balla, A.; Bagasra, O.; Costello, L.C. hZIP1 zinc uptake transporter down regulation and zinc depletion in prostate cancer. Mol. Cancer 2005, 4, 32. [Google Scholar] [CrossRef]
- Franklin, R.B.; Costello, L.C. The important role of the apoptotic effects of zinc in the development of cancers. J. Cell. Biochem. 2009, 106, 750–757. [Google Scholar] [CrossRef]
- Hershfinkel, M.; Moran, A.; Grossman, N.; Sekler, I. A zinc-sensing receptor triggers the release of intracellular Ca2+ and regulates ion transport. Proc. Natl. Acad. Sci. USA 2001, 98, 11749–11754. [Google Scholar]
- Zhang, Y.; Zhao, H.; Peng, H.; Hu, Z.; Ning, L.; Cao, Y.; Tan, Y.; Duan, E. GPR39, a putative receptor of Zn2+, is region specifically localized in different lobes of the mouse prostate. Urology 2011, 77, 1010.e1–1010.e6. [Google Scholar]
- Sharir, H.; Zinger, A.; Nevo, A.; Sekler, I.; Hershfinkel, M. Zinc released from injured cells is acting via the Zn2+-sensing receptor, ZnR, to trigger signaling leading to epithelial repair. J. Biol. Chem. 2010, 285, 26097–26106. [Google Scholar]
- Costello, L.C.; Franklin, R.B. The clinical relevance of the metabolism of prostate cancer; zinc and tumor suppression: Connecting the dots. Mol. Cancer 2006, 5, 17. [Google Scholar] [CrossRef] [Green Version]
- Franklin, R.B.; Costello, L.C. Zinc as an anti-tumor agent in prostate cancer and in other cancers. Arch. Biochem. Biophys. 2007, 463, 211–217. [Google Scholar] [CrossRef]
- Costello, L.C.; Franklin, R.B. Why do tumour cells glycolyse?”: From glycolysis through citrate to lipogenesis. Mol. Cell. Biochem. 2005, 280, 1–8. [Google Scholar]
- Franklin, R.B.; Milon, B.; Feng, P.; Costello, L.C. Zinc and zinc transporters in normal prostate and the pathogenesis of prostate cancer. Front. Biosci. 2005, 10, 2230–2239. [Google Scholar] [CrossRef]
- Costello, L.C.; Franklin, R.B.; Feng, P. Mitochondrial function, zinc, and intermediary metabolism relationships in normal prostate and prostate cancer. Mitochondrion 2005, 5, 143–153. [Google Scholar] [CrossRef]
- Costello, L.C.; Guan, Z.; Kukoyi, B.; Feng, P.; Franklin, R.B. Terminal oxidation and the effects of zinc in prostate versus liver mitochondria. Mitochondrion 2004, 4, 331–338. [Google Scholar] [CrossRef]
- Costello, L.C.; Franklin, R.; Stacey, R. Mitochondrial isocitrate dehydrogenase and isocitrate oxidation of rat ventral prostate. Enzyme 1976, 21, 495–506. [Google Scholar]
- Dhar, N.K.; Goel, T.C.; Dube, P.C.; Chowdhury, A.R.; Kar, A.B. Distribution and concentration of zinc in the subcellular fractions of benign hyperplastic and malignant neoplastic human prostate. Exp. Mol. Pathol. 1973, 19, 139–142. [Google Scholar] [CrossRef]
- Ye, B.; Maret, W.; Vallee, B.L. Zinc metallothionein imported into liver mitochondria modulates respiration. Proc. Natl. Acad. Sci. USA 2001, 98, 2317–2322. [Google Scholar] [CrossRef]
- Maret, W. Oxidative metal release from metallothionein via zinc-thiol/disulfide interchange. Proc. Natl. Acad. Sci. USA. 1994, 91, 237–241. [Google Scholar] [CrossRef]
- Costello, L.C.; Guan, Z.; Franklin, R.B.; Feng, P. Metallothionein can function as a chaperone for zinc uptake transport into prostate and liver mitochondria. J. Inorg. Biochem. 2004, 98, 664–666. [Google Scholar] [CrossRef]
- Guan, Z.; Kukoyi, B.; Feng, P.; Kennedy, M.C.; Franklin, R.B.; Costello, L.C. Kinetic identification of a mitochondrial zinc uptake transport process in prostate cells. J. Inorg. Biochem. 2003, 97, 199–206. [Google Scholar] [CrossRef]
- Singh, K.K.; Desouki, M.M.; Franklin, R.B.; Costello, L.C. Mitochondrial aconitase and citrate metabolism in malignant and nonmalignant human prostate tissues. Mol. Cancer 2006, 5, 14. [Google Scholar] [CrossRef]
- Feng, P.; Liang, J.Y.; Li, T.L.; Guan, Z.X.; Zou, J.; Franklin, R.; Costello, L.C. Zinc induces mitochondria apoptogenesis in prostate cells. Mol. Urol. 2000, 4, 31–36. [Google Scholar]
- Feng, P.; Li, T.L.; Guan, Z.X.; Franklin, R.B.; Costello, L.C. Direct effect of zinc on mitochondrial apoptogenesis in prostate cells. Prostate 2002, 52, 311–318. [Google Scholar] [CrossRef]
- Feng, P.; Li, T.; Guan, Z.; Franklin, R.B.; Costello, L.C. The involvement of Bax in zinc-induced mitochondrial apoptogenesis in malignant prostate cells. Mol. Cancer 2008, 7, 25. [Google Scholar] [CrossRef]
- Uzzo, R.G.; Crispen, P.L.; Golovine, K.; Makhov, P.; Horwitz, E.M.; Kolenko, V.M. Diverse effects of zinc on NF-kappaB and AP-1 transcription factors: Implications for prostate cancer progression. Carcinogenesis 2006, 27, 1980–1990. [Google Scholar] [CrossRef]
- Ishii, K.; Usui, S.; Sugimura, Y.; Yoshida, S.; Hioki, T.; Tatematsu, M.; Yamamoto, H.; Hirano, K. Aminopeptidase N regulated by zinc in human prostate participates in tumor cell invasion. Int. J. Cancer. 2001, 92, 49–54. [Google Scholar]
- Lightman, A.; Brandes, J.M.; Binur, N.; Drugan, A.; Zinder, O. Use of the serum copper/zinc ratio in the differential diagnosis of ovarian malignancy. Clin. Chem. 1986, 32, 101–103. [Google Scholar]
- Bae, S.N.; Lee, Y.S.; Kim, M.Y.; Kim, J.D.; Park, L.O. Antiproliferative and apoptotic effects of zinc-citrate compound (CIZAR(R)) on human epithelial ovarian cancer cell line, OVCAR-3. Gynecol. Oncol. 2006, 103, 127–136. [Google Scholar] [CrossRef]
- Ding, W.Q.; Yu, H.J.; Lind, S.E. Zinc-binding compounds induce cancer cell death via distinct modes of action. Cancer Lett. 2008, 271, 251–259. [Google Scholar] [CrossRef]
- Cousins, R.J. Gastrointestinal factors influencing zinc absorption and homeostasis. Int. J. Vitam. Nutr. Res. 2010, 80, 243–248. [Google Scholar]
- Weaver, B.P.; Zhang, Y.; Hiscox, S.; Guo, G.L.; Apte, U.; Taylor, K.M.; Sheline, C.T.; Wang, L.; Andrews, G.K. Zip4 (Slc39a4) expression is activated in hepatocellular carcinomas and functions to repress apoptosis, enhance cell cycle and increase migration. PLoS One 2010, 5, e13158. [Google Scholar]
- Ebara, M.; Fukuda, H.; Hatano, R.; Saisho, H.; Nagato, Y.; Suzuki, K.; Nakajima, K.; Yukawa, M.; Kondo, F.; Nakayama, A.; et al. Relationship between copper, zinc and metallothionein in hepatocellular carcinoma and its surrounding liver parenchyma. J. Hepatol. 2000, 33, 415–422. [Google Scholar] [CrossRef]
- Costello, L.C.; Levy, B.A.; Desouki, M.M.; Zou, J.; Bagasra, O.; Johnson, L.A.; Hanna, N.; Franklin, R.B. Decreased zinc and downregulation of ZIP3 zinc uptake transporter in the development of pancreatic adenocarcinoma. Cancer Biol. Ther. 2011, 12, 297–303. [Google Scholar] [CrossRef]
- Donadelli, M.; Dalla Pozza, E.; Costanzo, C.; Scupoli, M.T.; Scarpa, A.; Palmieri, M. Zinc depletion efficiently inhibits pancreatic cancer cell growth by increasing the ratio of antiproliferative/proliferative genes. J. Cell. Biochem. 2008, 104, 202–212. [Google Scholar]
- Donadelli, M.; Dalla Pozza, E.; Scupoli, M.T.; Costanzo, C.; Scarpa, A.; Palmieri, M. Intracellular zinc increase inhibits p53(−/−) pancreatic adenocarcinoma cell growth by ROS/AIF-mediated apoptosis. Biochim. Biophys. Acta 2009, 1793, 273–280. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Alam, S.; Kelleher, S.L. Cellular Mechanisms of Zinc Dysregulation: A Perspective on Zinc Homeostasis as an Etiological Factor in the Development and Progression of Breast Cancer. Nutrients 2012, 4, 875-903. https://doi.org/10.3390/nu4080875
Alam S, Kelleher SL. Cellular Mechanisms of Zinc Dysregulation: A Perspective on Zinc Homeostasis as an Etiological Factor in the Development and Progression of Breast Cancer. Nutrients. 2012; 4(8):875-903. https://doi.org/10.3390/nu4080875
Chicago/Turabian StyleAlam, Samina, and Shannon L. Kelleher. 2012. "Cellular Mechanisms of Zinc Dysregulation: A Perspective on Zinc Homeostasis as an Etiological Factor in the Development and Progression of Breast Cancer" Nutrients 4, no. 8: 875-903. https://doi.org/10.3390/nu4080875
APA StyleAlam, S., & Kelleher, S. L. (2012). Cellular Mechanisms of Zinc Dysregulation: A Perspective on Zinc Homeostasis as an Etiological Factor in the Development and Progression of Breast Cancer. Nutrients, 4(8), 875-903. https://doi.org/10.3390/nu4080875



