Neonatal Phosphate Nutrition Alters in Vivo and in Vitro Satellite Cell Activity in Pigs
Abstract
:1. Introduction
2. Experimental Section
2.1. Piglets
| Base Diet 2,3 | |
|---|---|
| Ingredients | Composition, % |
| Whey | 21.6 |
| Whey protein concentrate | 5.0 |
| Edible lard | 24.3 |
| Soy protein isolate | 30.0 |
| Calcium carbonate | 1.2 |
| Dicalcium phosphate | 1.0 |
| Calcium chloride | 0.33 |
| Mineral premix 4 | 0.8 |
| Vitamin premix 5 | 0.8 |
| Potassium sorbate | 0.5 |
| Dextrose | 14.6 |
| D, L-Methionine | 0.18 |
2.2. Sera Analysis
2.3. Isolation of Satellite Cells and Determination of in Vivo Proliferation
2.4. Cell Culture
2.5. Analysis of Gene Expression
| Gene name | Gene ID | Primer sequence |
|---|---|---|
| PAX7 | 494466 | F: 5′ CAACCACATCCGCCACAAGATAGT 3′ |
| R: 5′ AGAGGATCTTGGAGACACAGCCAT 3′ | ||
| MYOD1 | 407604 | F: 5′ GCGTGCAAACGCAAGACCACTAA 3′ |
| R: 5′ AGTCTCGAAGGCCTCGTTGACTTT 3′ | ||
| MYOG | 497618 | F: 5′ TGACCCTACAGATGCCCACAATCT 3′ |
| R: 5′ GTTGGGCATGGTTTCATCTGGGAA 3′ | ||
| PTH | 399502 | F: 5′ ATGCATAACCTGGGCAAACACCTG 3′ |
| R: 5′ TAGAAGCTCCGAGGGCAACAAAGT 3′ | ||
| CALCA | 100579174 | F: 5′ TCCAGAGCTAAGCGGTFCAGTAAT 3′ |
| R: 5′ TTTCTTGCCAGGTGTTTCAGGC 3′ | ||
| CASR | 100520980 | F: 5′ CATCAAGTTCCGAAACACGCCCAT 3′ |
| R: 5′ GCAGAGCACGAAGCTAATGCCAAA 3′ | ||
| RPL35 | 397598 | F: 5′ AACCAGACCCAGAAAGAGAAC 3′ |
| R: 5′ TTCCGCTGCTGCTTCTTG 3′ | ||
| RPL4 | 6124 | F: 5′ CAA GAG TAA CTA CAA CCT TC 3′ |
| R: 5′ GAA CTC TAC GAT GAA TCT TC 3′ |
2.6. Statistical Analysis
3. Results
| PO4 Deficient | PO4 Adequate | PO4 Supra-adequate | |
|---|---|---|---|
| BW gain, g/day | 191 ± 5 | 194 ± 4 | 201 ± 4 |
| Feed intake, g/day | 146 ± 0.7 | 145 ± 0.6 | 144 ± 0.6 |
| Feed Efficiency | 0.77 ± 0.02 a | 0.75 ± 0.01 ab | 0.72 ± 0.01 b |
| Sera Ca, mM | |||
| day 6 | 2.91 ± 0.13 a | 2.48 ± 0.13 b | 2.57 ± 0.13 ab |
| day 12 | 2.80 ± 0.14 | 2.71 ± 0.14 | 2.61 ± 0.14 |
| Sera PO4, mM | |||
| day 6 | 1.70 ± 0.06 b | 1.98 ± 0.06 a | 2.05 ± 0.06 a |
| day 12 | 1.62 ± 0.08 b | 2.17 ± 0.08 a | 2.26 ± 0.08 a |
| Sera PTH, pg/mL | |||
| day 6 | 1.5 ± 2.6 b | 13.6 ± 2.6 a | 15.9 ± 2.6 a |
| day 12 | 5.0 ± 15.1 b | 37.6 ± 15.1 ab | 52.0 ± 15.1 a |
| Bone mineral content, g | 1.61 ± 0.08 | 1.72 ± 0.08 | 1.84 ± 0.08 |
| Bone Ash % | 32.48 ± 1.02 | 34.02 ± 1.02 | 34.16 ± 1.02 |
| Radial Length, mm | 7.12 ± 0.10 b | 7.25 ± 0.10 ab | 7.42 ± 0.10 a |
| Radial Width, mm | 0.94 ± 0.03 a | 0.81 ± 0.03 b | 0.84 ± 0.03 b |
| BrdU + Satellite Cell, % | 9.9 ± 1.4 b | 15.6 ± 1.5 a | 17.6 ± 1.7 a |
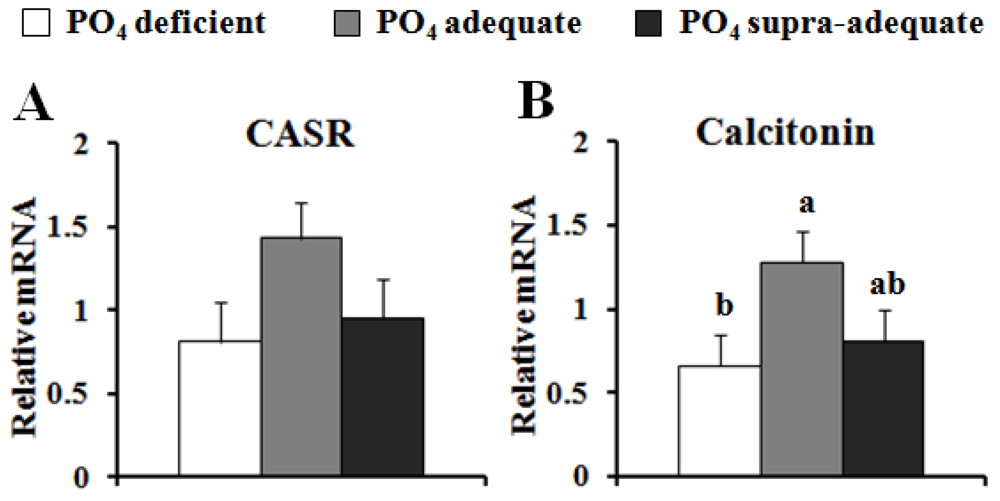
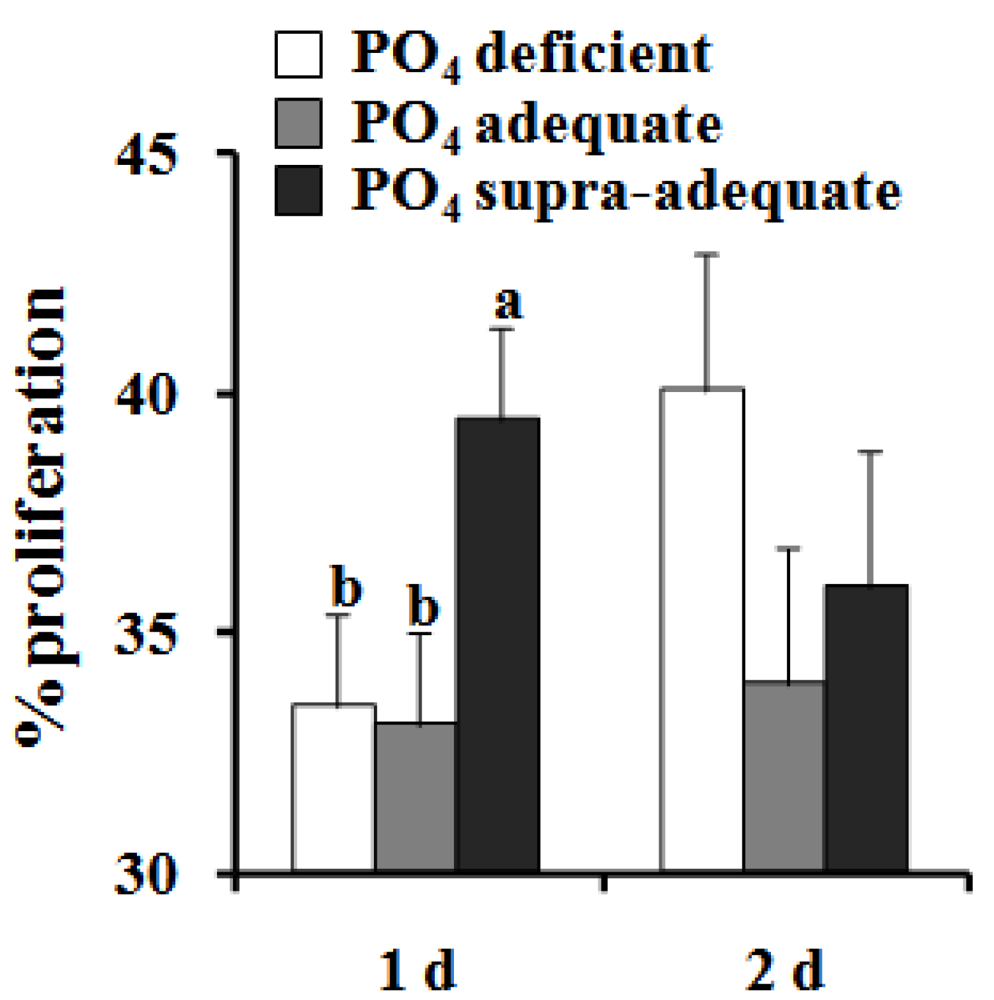
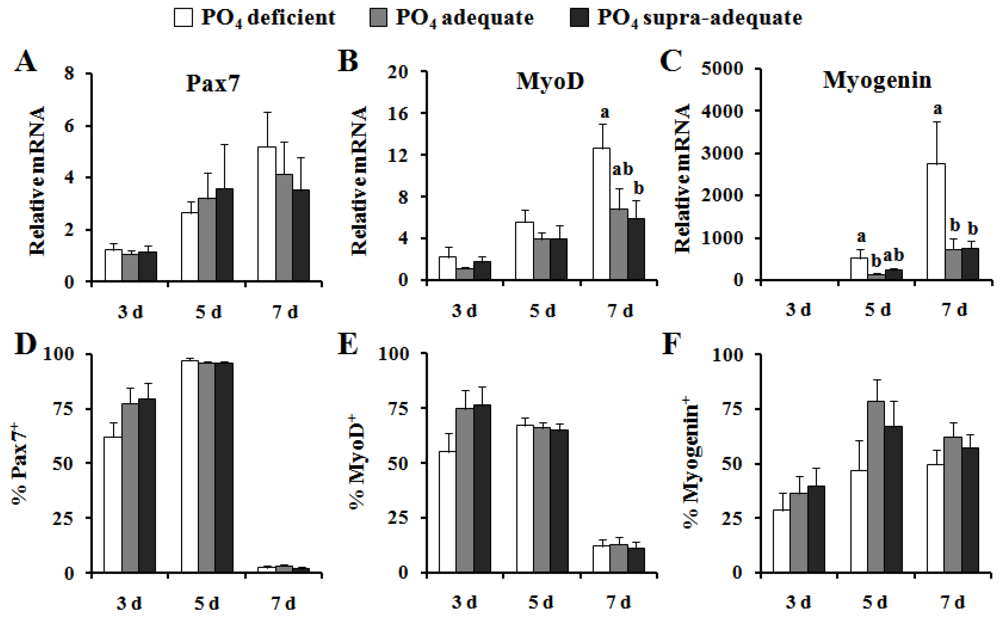
4. Discussion
5. Conclusions
Acknowledgments
Conflict of Interest
References
- Allen, R.E.; Merkel, R.A.; Young, R.B. Cellular aspects of muscle growth: myogenic cell proliferation. J. Anim. Sci. 1979, 49, 115–127. [Google Scholar]
- Campion, D.R.; Richardson, R.L.; Reagan, J.O.; Kraeling, R.R. Changes in the satellite cell population during postnatal growth of pig skeletal muscle. J. Anim. Sci. 1981, 52, 1014–1018. [Google Scholar]
- Winick, M.; Noble, A. Cellular response in rats during malnutrition at various ages. J. Nutr. 1966, 89, 300–306. [Google Scholar]
- Harbison, S.A.; Goll, D.E.; Parrish, F.C.; Wang, V.; Kline, E.A. Muscle growth in two genetically different lines of swine. Growth 1976, 40, 253–283. [Google Scholar]
- Swatland, H.J. Accumulation of myofiber nuclei in pigs with normal and arrested development. J. Anim. Sci. 1977, 44, 759–764. [Google Scholar]
- Trenkle, A. Relation of hormonal variations to nutritional studies and metabolism of ruminants. J. Dairy Sci. 1978, 61, 281–293. [Google Scholar] [CrossRef]
- Moss, F.P.; Leblond, C.P. Satellite cells as the source of nuclei in muscles of growing rats. Anat. Rec. 1971, 170, 421–435. [Google Scholar] [CrossRef]
- Chargé, S.B.; Rudnicki, M.A. Cellular and molecular regulation of muscle regeneration. Physiol. Rev. 2004, 84, 209–238. [Google Scholar] [CrossRef]
- Cardasis, C.A.; Cooper, G.W. An analysis of nuclear numbers in individual muscle fibers during differentiation and growth: A satellite cell-muscle fiber growth unit. J. Exp. Zool. 1975, 191, 347–358. [Google Scholar] [CrossRef]
- Campion, D.R. The muscle satellite cell: A review. Int. Rev. Cytol. 1984, 87, 225–251. [Google Scholar] [CrossRef]
- Mulvaney, D.R.; Marple, D.N.; Merkel, R.A. Proliferation of skeletal muscle satellite cells after castration and administration of testosterone propionate. Proc. Soc. Exp. Biol. Med. 1988, 188, 40–45. [Google Scholar]
- Mesires, N.T.; Doumit, M.E. Satellite cell proliferation and differentiation during postnatal growth of porcine skeletal muscle. Am. J. Physiol. Cell Physiol. 2002, 282, 899–906. [Google Scholar]
- Alexander, L.S.; Mahajan, A.; Odle, J.; Flann, K.L.; Rhoads, R.P.; Stahl, C.H. Dietary phosphate restriction decreases stem cell proliferation and subsequent growth potential in neonatal pigs. J. Nutr. 2010, 140, 477–482. [Google Scholar] [CrossRef]
- Moore, D.T.; Ferket, P.R.; Mozdziak, P.E. The effect of early nutrition on satellite cell dynamics in the young turkey. Poult. Sci. 2005, 84, 748–756. [Google Scholar]
- Nierobisz, L.S.; Felts, V.; Mozdziak, P.E. The effect of early dietary amino acid levels on muscle satellite cell dynamics in turkeys. Poult. Sci. 2007, 148, 286–294. [Google Scholar]
- Halevy, O.; Nadel, Y.; Barak, M.; Rozenboim, I.; Sklan, D. Early posthatch feeding stimulates satellite cell proliferation and skeletal muscle growth in turkey poults. J. Nutr. 2003, 133, 1376–1382. [Google Scholar]
- Jeanplong, F.; Bass, J.; Smith, H.K.; Kirk, S.P.; Kambadur, R.; Sharma, M.; Oldham, J.M. Prolonged underfeeding of sheep increases myostatin and myogenic regulatory factor Myf-5 in skeletal muscle while IGF-1 and myogenin are repressed. J. Endocrinol. 2003, 176, 425–437. [Google Scholar] [CrossRef]
- Abrams, S.A. What are the risks and benefits to increasing dietary bone minerals and vitamin D intake in infants and small children? Annu. Rev. Nutr. 2011, 31, 285–297. [Google Scholar] [CrossRef]
- Environmental Protection Agency. National pollutant discharge elimination system permit regulation. Fed Regist. 2003, 68, 7175–7274.
- Heidebrecht, A.A.; Mac, V.R.; Ross, O.B.; Whitehair, C.K. Composition of swine milk. I. Major constituents and carotene, vitamin A and vitamin C. J. Nutr. 1951, 44, 43–50. [Google Scholar]
- National Research Council, Nutrient Requirements of Swine, 10th ed; National Academy Press: Washington, DC, USA, 1998.
- Mathews, S.A.; Oliver, W.T.; Phillips, O.T.; Odle, J.; Diersen-Schade, D.A.; Harrell, R.J. Comparison of triglycerides and phospholipids as supplemental sources of dietary long-chain polyunsaturated fatty acids in piglets. J. Nutr. 2002, 132, 3081–3089. [Google Scholar]
- Gomori, G. A modification of the colorimetric phosphorus determination for use with the photoelectric colorimeter. J. Lab. Clin. Med. 1942, 27, 955–960. [Google Scholar]
- Allen, R.E.; Temm-Grove, C.J.; Sheehan, S.M.; Rice, G. Skeletal muscle satellite cell cultures. Methods Cell. Biol. 1997, 52, 155–176. [Google Scholar] [CrossRef]
- Rhoads, R.P.; Johnson, R.M.; Rathbone, C.R.; Liu, X.; Temm-Grove, C.; Sheehan, S.M.; Hoying, J.B.; Allen, R.E. Satellite cell mediated angiogenesis coincides with a functional hypoxia-inducible factor (HIF) pathway. Am. J. Physiol. Cell Physiol. 2009, 296, 1321–1328. [Google Scholar] [CrossRef]
- Doumit, M.E.; Merkel, R.A. Conditions for isolation and culture of porcine myogenic satellite cells. Tissue Cell 1992, 24, 253–262. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittigen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Slatopolsky, E.; Finch, J.; Denda, M.; Ritter, C.; Zhong, M.; Dusso, A.; MacDonald, P.N.; Brown, A.J. Phospohrus restriction prevents parathyroid gland growth: high phosphorus directly stimulates PTH secretion in vitro. J. Clin. Invest. 1996, 97, 2534–2540. [Google Scholar] [CrossRef]
- Mahajan, A.; Alexander, L.S.; Seabolt, B.S.; Catrambone, D.E.; McClung, J.P.; Odle, J.; Pfeiler, T.W.; Loboa, E.G.; Stahl, C.H. Dietary calcium restriction affects mesenchymal stem cell activity and bone development in neonatal pigs. J. Nutr. 2011, 141, 373–379. [Google Scholar] [CrossRef]
- Halloran, B.P.; DeLuca, H.F. Calcium transport in the small intestine during early development: role of vitamin D. Am. J. Physiol. 1980, 239, 473–479. [Google Scholar]
- Halloran, B.P.; DeLuca, H.F. Appearance of the intestinal cytosolic receptor for 1,25-dihydroxyvitamin D3 during neonatal development in the rat. J. Biol. Chem. 1981, 256, 7338–7342. [Google Scholar]
- Halevy, O.; Geyra, A.; Barak, M.; Uni, Z.; Sklan, D. Early posthatch starvation decreases satellite cell proliferation and skeletal muscle growth in chicks. J. Nutr. 2000, 130, 858–864. [Google Scholar]
- Davis, T.A.; Fiorotto, M.L. Regulation of muscle growth in neonates. Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 78–85. [Google Scholar] [CrossRef]
- Oliver, W.T.; Miles, J.R. A low-fat liquid diet increases protein accretion and alters cellular signaling for protein synthesis in 10-day-old pigs. J. Anim. Sci. 2010, 88, 2576–2584. [Google Scholar] [CrossRef]
- Chen, J.; Tao, Y.; Li, J.; Deng, Z.; Yan, Z.; Xiao, X.; Wang, D. microRNA-1 and microRNA-206 regulate skeletal muscle satellite cell proliferation and differentiation by repressing Pax7. J. Cell Biol. 2010, 190, 867–879. [Google Scholar] [CrossRef]
- Hirai, H.; Verma, M.; Watanabe, S.; Tastad, C.; Asakura, Y.; Asakura, A. MyoD regulates apoptosis of myoblasts through microRNA-mediated down-regulation of Pax3. J. Cell Biol. 2010, 191, 347–365. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Alexander, L.S.; Seabolt, B.S.; Rhoads, R.P.; Stahl, C.H. Neonatal Phosphate Nutrition Alters in Vivo and in Vitro Satellite Cell Activity in Pigs. Nutrients 2012, 4, 436-448. https://doi.org/10.3390/nu4060436
Alexander LS, Seabolt BS, Rhoads RP, Stahl CH. Neonatal Phosphate Nutrition Alters in Vivo and in Vitro Satellite Cell Activity in Pigs. Nutrients. 2012; 4(6):436-448. https://doi.org/10.3390/nu4060436
Chicago/Turabian StyleAlexander, Lindsey S., Brynn S. Seabolt, Robert P. Rhoads, and Chad H. Stahl. 2012. "Neonatal Phosphate Nutrition Alters in Vivo and in Vitro Satellite Cell Activity in Pigs" Nutrients 4, no. 6: 436-448. https://doi.org/10.3390/nu4060436
APA StyleAlexander, L. S., Seabolt, B. S., Rhoads, R. P., & Stahl, C. H. (2012). Neonatal Phosphate Nutrition Alters in Vivo and in Vitro Satellite Cell Activity in Pigs. Nutrients, 4(6), 436-448. https://doi.org/10.3390/nu4060436





