Gut Microbiota and Inflammation
Abstract
:1. Inflammation
- (i) Blood vessels widen, resulting in increased blood flow (causing the redness and increased heat);
- (ii) Permeability of the blood vessels is increased, which results in an outflow of fluid and plasma proteins into the tissue, manifesting itself as swelling;
- (iii) White blood cells are recruited from the blood circulation to the tissue;
- (iv) The metabolism is adjusted, for example by increased levels of glucose in the blood, and symptoms such as fever, fatigue and loss of appetite can occur.
2. Human Gastrointestinal Microbiota
2.1. Viable Count, Metagenomics and the Phylogenetic Core
| Phyla/Division | Class | Family | Genus | Gram (2) |
|---|---|---|---|---|
| Actinobacteria | Actinobacteria | Micrococcaceae | Rothia * | + |
| Actinobacteria | Actinobacteria | Bifidobacteriaceae | Bifidobacterium | + |
| Firmicutes | Bacilli | Streptoccaceae | Streptococcus | + |
| Firmicutes | Bacilli | Lactobacillaceae | Lactobacillus | + |
| Firmicutes | Bacilli | Enterococcaceae | Enterococcus | + |
| Firmicutes | Negativicutes | Veillonellaceae | Veillonella | (−) |
| Firmicutes | Negativicutes | Veillonellaceae | Dialiser | (−) |
| Firmicutes | Clostridia | unclassified Clostridiales | Mogibacterium * | + |
| Firmicutes | Clostridia | Peptostreptococcaceae | Peptostreptococcus * | + |
| Firmicutes | Clostridia | Lachnospiraceae | Coprococcus | + |
| Firmicutes | Clostridia | Lachnospiraceae | Dorea | + |
| Firmicutes | Clostridia | Lachnospiraceae | Roseburia | (−) |
| Firmicutes | Clostridia | Lachnospiraceae | Butyrivibrio | (−) |
| Firmicutes | Clostridia | Ruminococcaceae | Ruminococcus | + |
| Firmicutes | Clostridia | Ruminococcaceae | Faecalibacterium | + |
| Firmicutes | Clostridia | Ruminococcaceae | Anaerotruncus | + |
| Firmicutes | Clostridia | Ruminococcaceae | Subdoligranulum | + |
| Firmicutes | Clostridia | Clostridiaceae | Clostridium | + |
| Firmicutes | Clostridia | Clostridiaceae | Blautia | + |
| Firmicutes | Clostridia | Eubacteriaceae | Eubacterium | + |
| Firmicutes | Clostridia | unclassified | Collinsella | + |
| Firmicutes | Erysipelotrichia | Erysipelotrichaceae | Holdemania | + |
| Proteobacteria | Betaproteobacteria | Alcaligenaceae | Sutterella | - |
| Proteobacteria | Betaproteobacteria | Neisseriaceae | Neisseria | - |
| Proteobacteria | Deltaproteobacteria | Desulfovibrionaceae | Bilophila | - |
| Proteobacteria | Gammaproteobacteria | Pasteurellaceae | Haemophilus * | - |
| Proteobacteria | Gammaproteobacteria | Enterobacteriaceae | Enterobacter * | - |
| Proteobacteria | Gammaproteobacteria | Enterobacteriaceae | Serratia * | - |
| Proteobacteria | Gammaproteobacteria | Enterobacteriaceae | Escherichia | - |
| Proteobacteria | Gammaproteobacteria | Enterobacteriaceae | Klebsiella | - |
| Proteobacteria | Gammaproteobacteria | Moraxellaceae | Acinetobacter | - |
| Proteobacteria | Gammaproteobacteria | Pseudomonadaseae | Pseudomonas * | - |
| Proteobacteria | Gammaproteobacteria | Cardiobacteriaceae | Cardiobacterium | - |
| Bacteroidetes | Bacteroidia | Prevotellaceae | Prevotella * | - |
| Bacteroidetes | Bacteroidia | Porphyromonadaceae | Porphyromonas * | - |
| Bacteroidetes | Bacteroidia | Porphyromonadaceae | Parabacteroides | - |
| Bacteroidetes | Bacteroidia | Bacteroidaceae | Bacteroides | - |
| Bacteroidetes | Bacteroidia | Rikenellaceae | Alistipes | |
| Fusobacteria | Fusobacteria | Fusobacteriaceae | Fusobacterium | - |
| Spirochaetae | Spirochaetes | Brachyspiraceae | Brachyspira | - |
| Verrucomicrobia | Verrucomicrobiae | Verrucomicrobiaceae | Akkermansia | - |
2.2. Mouth
2.3. Stomach
2.4. Small Intestine
2.4.1. Jejunum
2.4.2. Ileum
2.5. Large Intestine
2.6. Inflammation Driving Capacity
2.7. Bacterial Neutralisation of Inflammation
3. Probiotics for Humans
3.1. Species Used as Probiotics
| Category of subjects | Strains | Major symptom affected | Systemic marker affected | Ref. |
|---|---|---|---|---|
| Healthy subjects | L. salivarius CECT5713 | - | NK-cells, monocytes, IgM, IgA, IgG, IL-10 | [ 84] |
| L. casei Shirota | - | NK-cells | [ 85] | |
| L. paracasei Lpc-37, L. acidophilus 74-2, B. animalis subsp. lactis DGCC 420 | - | CD57+, phagocytic activity oxidative burst | [ 86] | |
| L. acidophilus 74-2, B. animalis subsp. lactis DGCC 420 | - | phagocytic activity | [ 87] | |
| L. rhamnosus GG | - | Receptors CR1, CR3, FcγRI, IgαR | [ 88] | |
| L. plantarum WCSF1 | - | Occluding, ZO-1 | [ 89] | |
| Metabolic syndrome and low-grade inflammation | L. acidophilus 145, B. longum 913 | - | HDL-cholesterol | [ 90] |
| L. helveticus -, S. cerevisiae - | Blood pressure | - | [ 91] | |
| L. plantarum 299v | - | total cholesterol, LDL-cholesterol, fibrinogen | [ 92] | |
| L. plantarum 299v | Systolic blood pressure | leptin, fibrinogen, F2-isoprostanes, IL-6 | [ 93] | |
| B. lactis HN019 | - | CD3+, CD4+, CD25+, CD56+, phagocytic activity, tumoricidal activity of NK cells | [ 94] | |
| Non-alcoholic fatty liver disease (NAFLD) | Mixture (1) | - | alanine-aminotransferase (ALAT), γ-glutamyl-transpeptidase, 4-hydroxynonenal, TNF-α | [ 95] |
| VSL#3 (2) | - | S-nitrosothiols, malondialdehyde (MDA), 4-hydroxynonenal | [ 96] | |
| Alcohol-related liver injury | B. bifidum -, L. plantarum 8PA3 | - | ALAT, aspartate-aminotransferase (ASAT), gamma glutamyl transpeptidase, lactate dehydrogenase, bilirubin | [ 97] |
| L. casei Shirota | - | neutrophil phagocytic activity TLR4 | [ 98] | |
| Fibrosis, cirrhosis, liver transplantations and minimal hepatic encephalopathy (MHE) | P. pentoseceus 5-33:3, L. mesenteroides 32-77:1, L. paracasei 19, L. plantarum 2592 | Child-Turcotte-Pugh score | ammonia, endotoxin, bilirubin, ALAT, albumin, prothrombin activity | [ 99] |
| L. acidophilus - | Clinical status | ammonia | [ 100,101] | |
| S. thermophilus-, L. bulgaricus -, L. acidophilus -, bifidobacteria -, L. casei - | MHE reversal | - | [ 102] | |
| L. plantarum 299 | Incidence of postoperative infections | - | [ 103] | |
| P.pentosaceus 5-33:3, L. mesenteroides 77:1, L. paracasei F19, L. plantarum 2362 | Incidence of postoperative infections | - | [ 104] | |
| Acute pancreatitis | “Ecologic 641” (3) | -(4) | - | [ 82] |
| Acute pancreatitis | L. plantarum - | Clinical outcome | - | [ 83] |
| Critically ill patients | L. plantarum 299v | - | IL-6, intestinal translocation | [ 105] |
| L. plantarum 299v | - | intestinal translocation, IL-10 white blood cell count, lactate | [ 106] | |
| VSL#3 | - | IgA, IgG | [ 107] | |
| Allergy; infants | L. acidophilus LAVRI-A1 | - | - | [ 108] |
| L. rhamnosus GG | Atopic eczema | - | [ 109] | |
| B. lactis Bb-12 | SCORAD score | soluble CD4, eosinophilic protein X | [ 110] | |
| L. rhamnosus GG | SCORAD | soluble CD4, eosinophilic protein X | [ 110] | |
| L. acidophilus NCFM, B. lactis Bl-04 | Nasal symptoms | IgA | [ 111] | |
| L. rhamnosus GG | - | IgA, alpha1-antitrypsin | [ 112] | |
| mixture (5) | - | IgA | [ 112] | |
| L. gasseri CECT5714, L. coryniformis CECT5711 | - | IgE, IgA, CD4(+)CD25(+) T regulatory cells, NK-cells | [ 113] | |
| B. lactis Bb12 | Body weight | Calprotectin, IgA | [ 114] | |
| Allergy; adults | L.paracasei Lpc-37, L. acidophilus 74-2, B. animalis subsp. lactis DGCC 420 | - | CD4(+)CD54(+) | [ 86] |
| L. rhamnosus GG | - | Receptors CR1, CR3, FcγRI, IgαR | [ 88] | |
| Crohn’s disease | L. rhamnosus GG | None (6) | - | [ 115] |
| L. rhamnosus GG | None | - | [ 116] | |
| L. rhamnosus GG | None | - | [ 117] | |
| L. rhamnosus GG | Clinical outcome | - | [ 118] | |
| L. rhamnosus GG | Clinical activity | Intestinal permeability | [ 119] | |
| L. johnsonii LA1 | None | - | [ 120] | |
| L. johnsonii LA1 | None | - | [ 121] | |
| Ileal pouchitis, ulcerative colitis and colorectal cancer | VSL#3 | Disease activity | CD4+CD25high cells, CD4+ LAP+ cells, IL-1β mRNA, Foxp3 mRNA | [ 122] |
| VSL#3 | Disease activity index, remisson | - | [ 123] | |
| VSL#3 | Remission | - | [ 124] | |
| VSL#3 | Disease activity index, inflammatory bowel disease questionnaire, remission | - | [ 125] | |
| BIO-THREE (7) | Clinical symptoms, endoscopic findings | - | [ 126] | |
| E. coli Nissle 1917 | Clinical symptoms | - | [ 127] | |
| L. rhamnosus GR1, L. reuteri RC-14 | - | CD4+CD25high cells, IL-12, TNF-α/IL-12-producing monocytes, DCs | [ 128] | |
| L. rhamnosus GG | Remission | - | [ 129] | |
| B. breve Yakult, B. bifidum Yakult, L. acidophilus - | Clinical activity index, endoscopic activity index | - | [ 130] | |
| Bifidobacterium -, Lactobacillus -, Enterococcus - | Flare-ups | NF-κB, TNF-α, IL-1β, IL-10 | [ 131] | |
| Bifidobacterium - | Postoperative septic complications | SIgA, IgG, IgM, IgA, IL-6, C-reactive protein (CRP) | [ 132] | |
| Radiation-induced enteritis | VSL#3 | Diarrhea, bowel movements | - | [ 133] |
| L. rhamnosus - | Bowel movements, stool consistency | - | [ 134] | |
| L. rhamnosus GG | Diarrhea, abdominal discomfort | - | [ 135] | |
| L. acidophilus - | Diarrhea, flatulence | - | [ 136] | |
| L. casei DN-114 001 | Stool consistency | - | [ 137] |
3.2. Immune Modulation
3.2.1. T Regulatory Cells: A Key Factor in Several Dysfunctions
3.2.2. Healthy and Allergic Adults
3.2.3. Allergic Children
3.3. Metabolic Syndrome and Low-Grade Inflammation
3.4. Liver Injury
3.4.1. Liver Homeostasis
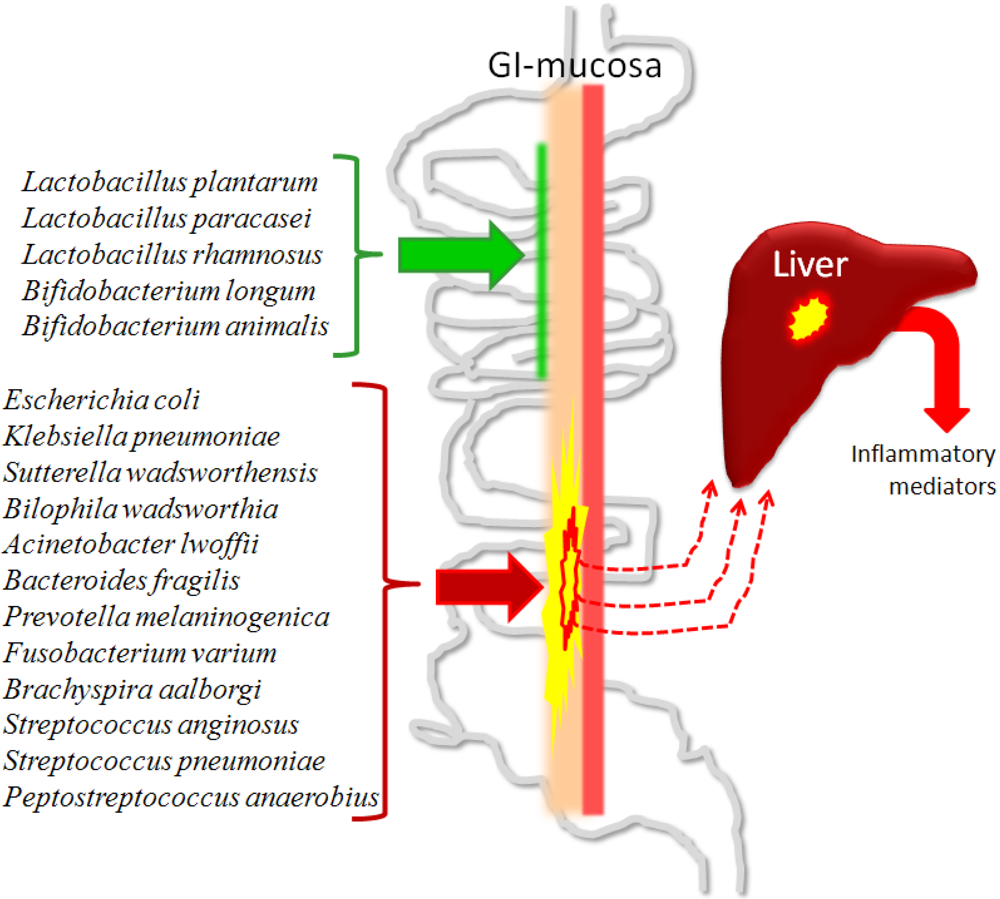
3.4.2. Fibrosis, Cirrhosis and Minimal Hepatic Encephalopathy
3.4.3. Alcohol-Related Liver Injury
3.4.4. Non-Alcoholic Fatty Liver Disease (NAFLD)
3.5. Ulcerative Colitis, Pouchitis and Colorectal Cancer
3.6. Crohn’s Disease
3.7. Radiation-Induced Enteritis
4. Conclusions
References
- Mölne, J.; Wold, A. Inflammation, 1st ed; Liber AB: Stockholm, Sweden, 2007. [Google Scholar]
- Pepys, M.B.; Baltz, M.L. Acute phase proteins with special reference to C-reactive and related proteins (pentaxins) and serum amyloid A protein. Adv. Immunol. 1983, 34, 141–212. [Google Scholar]
- Ridker, P.M. Clinical application of C-reactive protein for cardiovascular disease detection and prevention. Circulation 2003, 107, 363–369. [Google Scholar]
- Piéroni, L.; Bastard, J.P.; Piton, A.; Khalil, L.; Hainque, B.; Jardel, C. Interpretation of circulating C-reactive protein levels in adults: Body mass index and gender are a must. Diabetes Metab. 2003, 29, 133–138. [Google Scholar]
- Visser, M.; Bouter, L.M.; McQuillan, G.M.; Wener, M.H.; Harris, T.B. Elevated C-reactive protein levels in overweight and obese adults. JAMA 1999, 282, 2131–2135. [Google Scholar]
- Rescigno, M.; Urbano, M.; Valzasina, B.; Francolini, M.; Rotta, G.; Bonasio, R.; Granucci, F.; Kraehenbuhl, J.P.; Ricciardi-Castagnoli, P. Dendritic cells express tight junction proteins and penetrate gut epithelial monolayers to sample bacteria. Nat. Immunol. 2001, 2, 361–367. [Google Scholar]
- Newberry, R.D.; Lorenz, R.G. Organizing a mucosal defence. Immunol. Rev. 2005, 206, 6–21. [Google Scholar]
- Forchielli, M.L.; Walker, W.A. The role of gut-associated lymphoid tissues and mucosal defence. Br. J. Nutr. 2005, 93, S41–S48. [Google Scholar]
- Izcue, A.; Coombes, J.L.; Powrie, F. Regulatory lymphocytes and intestinal inflammation. Annu. Rev. Immunol. 2009, 27, 313–338. [Google Scholar]
- Palmblad, J. The role of granulocytes in inflammation. Scand. J. Rheumatol. 1984, 13, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, N.; Kobayashi, K. Macrophages in inflammation. Curr. Drug Targets Inflamm. Allergy 2005, 4, 281–286. [Google Scholar]
- Van Ginderachter, J.A.; Movahedi, K.; Ghassabeh, G.H.; Meerschaut, S.; Beschin, A.; Raes, G.; De Baetselier, P. Classical and alternative activation of mononuclear phagocytes: Picking the best of both worlds for tumor promotion. Immunobiology 2006, 211, 487–501. [Google Scholar]
- Anderson, C.F.; Mosser, D.M. A novel phenotype for an activated macrophage: The type 2 activated macrophage. J. Leukoc. Biol. 2002, 72, 101–106. [Google Scholar]
- Gordon, S. Alternative activation of macrophages. Nat. Rev. 2003, 3, 23–35. [Google Scholar]
- Mosser, D.M. The many faces of macrophage activation. J. Leukoc. Biol. 2003, 73, 209–212. [Google Scholar]
- Umesaki, Y.; Setoyama, H.; Matsumoto, S.; Okada, Y. Expansion of alpha beta T cell receptor-bearing intestinal intraepithelial lymphocytes after microbial colonization in germ-free mice and its independence from thymus. Immunology 1993, 79, 32–37. [Google Scholar]
- Helgeland, L.; Vaage, J.T.; Rolstad, B.; Midtvedt, T.; Brandtzaeg, P. Microbial colonization influences composition and T cell receptor V beta repertoire of intraepithelial lymphocytes in rat intestine. Immunology 1996, 89, 494–501. [Google Scholar]
- Cebra, J.J.; Periwal, S.B.; Lee, G.; Lee, F.; Shroff, K.E. Development and maintenance of the gut-associated lymphoid tissue (GALT): The roles of enteric bacteria and viruses. Dev. Immunol. 1998, 6, 13–18. [Google Scholar]
- Butler, J.E.; Sun, J.; Weber, P.; Navarro, P.; Francis, D. Antibody repertoire development in fetal and newborn piglets, III. Colonization of the gastrointestinal tract selectively diversifies the preimmune repertoire in mucosal lymphoid tissues. Immunology 2000, 100, 119–130. [Google Scholar] [CrossRef]
- Cario, E.; Brown, D.; McKee, M.; Lynch-Devaney, K.; Gerken, G.; Podolsky, D.K. Commensal associated molecular patterns induce selective toll-like receptor-trafficking from apical membrane to cytoplasmic compartments in polarized intestinal epithelium. Am. J. Pathol. 2002, 160, 165–173. [Google Scholar]
- Hershberg, R.M.; Mayer, L.F. Antigen processing and presentation by intestinal epithelial cells—polarity and complexity. Immunol. Today 2000, 21, 123–128. [Google Scholar]
- Testro, A.G.; Visvanathan, K. Toll-like receptors and their role in gastrointestinal disease. J. Gastroenterol. Hepatol. 2009, 24, 943–954. [Google Scholar]
- Zarember, K.A.; Godowski, P.J. Tissue expression of human Tolllike receptors and differential regulation of Toll-like receptor mRNAs in leukocytes in response to microbes, their products, and cytokines. J. Immunol. 2002, 168, 554–561. [Google Scholar] [PubMed]
- Fichorova, R.N.; Cronin, A.O.; Lien, E.; Anderson, D.J.; Ingalls, R.R. Response to Neisseria gonorrhoeae by cervicovaginal epithelial cells occurs in the absence of toll-like receptor 4-mediated signalling. J. Immunol. 2002, 168, 2424–2432. [Google Scholar]
- Poltorak, A.; He, X.; Smirnova, I.; Liu, M.Y.; Van Huffel, C.; Du, X.; Birdwell, D.; Alejos, E.; Silva, M.; Galanos, C.; et al. Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: Mutations in Tlr4 gene. Science 1998, 282, 2085–2088. [Google Scholar]
- Qureshi, S.T.; Lariviere, L.; Leveque, G.; Clermont, S.; Moore, K.J.; Gros, P.; Malo, D. Endotoxin-tolerant mice have mutations in Toll-like receptor 4 (Tlr4). J. Exp. Med. 1999, 189, 615–625. [Google Scholar]
- Schwandner, R.; Dziarski, R.; Wesche, H.; Rothe, M.; Kirschning, C.J. Peptidoglycan- and lipoteichoic acid-induced cell activation is mediated by Toll-like receptor 2. J. Biol. Chem. 1999, 274, 17406–17409. [Google Scholar]
- Takeuchi, O.; Kaufmann, A.; Grote, K.; Kawai, T.; Hoshino, K.; Morr, M.; Mühlradt, P.F.; Akira, S. Cutting edge: Preferentially the R-stereoisomer of the mycoplasmal lipopeptide macrophage-activating lipopeptide-2 activates immune cells through a toll-like receptor 2- and MyD88-dependent signaling pathway. J. Immunol. 2000, 164, 554–557. [Google Scholar]
- Philpott, D.J.; Girardin, S.E.; Sansonetti, P.J. Innate immune responses of epithelial cells following infection with bacterial pathogens. Curr. Opin. Immunol. 2001, 13, 410–416. [Google Scholar]
- Girardin, S.E.; Tournebize, R.; Mavris, M.; Page, A.L.; Li, X.; Stark, G.R.; Bertin, J.; DiStefano, P.S.; Yaniv, M.; Sansonetti, P.J.; et al. CARD4/Nod1 mediates NF-kappaB and JNK activation by invasive Shigella flexneri. EMBO Rep. 2001, 2, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Girardin, S.E.; Boneca, I.G.; Carneiro, L.A.; Antignac, A.; Jéhanno, M.; Viala, J.; Tedin, K.; Taha, M.K.; Labigne, A.; Zähringer, U.; et al. Nod1 detects a unique muropeptide from gram-negative bacterial peptidoglycan. Science 2003, 300, 1584–1587. [Google Scholar]
- Hasegawa, M.; Yang, K.; Hashimoto, M.; Park, J.H.; Kim, Y.G.; Fujimoto, Y.; Nuñez, G.; Fukase, K.; Inohara, N. Differential release and distribution of Nod1 and Nod2 immunostimulatory molecules among bacterial species and environments. J. Biol. Chem. 2006, 281, 29054–29063. [Google Scholar]
- Chen, G.; Shaw, M.H.; Kim, Y.G.; Nunez, G. NOD-like receptors: role in innate immunity and inflammatory disease. Annu. Rev. Pathol. Mech. Dis. 2009, 4, 365–398. [Google Scholar]
- Pickard, K.M.; Bremner, A.R.; Gordon, J.N.; MacDonald, T.T. Microbial gut interactions in health and disease. Immune responses. Best Pract. Res. Clin. Gastroenterol. 2004, 18, 271–285. [Google Scholar] [PubMed]
- Cheroutre, H. Starting at the beginning: new perspectives on the biology of mucosal T cells. Annu. Rev. Immunol. 2004, 22, 217–246. [Google Scholar]
- Cash, H.L.; Whitham, C.V.; Behrendt, C.L.; Hooper, L.V. Symbiotic bacteria direct expression of an intestinal bactericidal lectin. Science 2006, 313, 1126–1130. [Google Scholar]
- Cerutti, A.; Rescigno, M. The biology of intestinal immunoglobulin A responses. Immunity 2008, 28, 740–750. [Google Scholar]
- Eisen, J.A. Environmental shotgun sequencing: Its potential and challenges for studying the hidden world of microbes. PLoS Biol. 2007, 5, e82. [Google Scholar]
- Handelsman, J.; Rondon, M.R.; Brady, S.F.; Clardy, J.; Goodman, R.M. Molecular biological access to the chemistry of unknown soil microbes: a new frontier for natural products. Chem. Biol. 1998, 5, 245–249. [Google Scholar]
- Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T.; et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59–65. [Google Scholar]
- Tap, J.; Mondot, S.; Levenez, F.; Pelletier, E.; Caron, C.; Furet, J.P.; Ugarte, E.; Muñoz-Tamayo, R.; Paslier, D.L.E.; Nalin, R.; et al. Towards the human intestinal microbiota phylogenetic core. Environ. Microbiol. 2009, 11, 2574–2584. [Google Scholar]
- Costello, E.K.; Lauber, C.L.; Hamady, C.; Fierer, N.; Gordon, J.I.; Knight, R. Bacterial community variation in human body habitats across space and time. Science 2009, 326, 1694–1697. [Google Scholar]
- Wang, M.; Ahrné, S.; Jeppsson, B.; Molin, G. Comparison of bacterial diversity along the human intestinal tract by direct cloning and sequencing of 16S rRNA genes. FEMS Microbiol. Ecol. 2005, 54, 219–231. [Google Scholar]
- Cheng, K.J.; Costerton, J.W. Ultrastructure of Butyrivibrio fibrisolvens: A gram-positive bacterium? J. Bacteriol. 1977, 129, 1506–1512. [Google Scholar] [PubMed]
- Pettersson, B.; Ahrné, S.; Wang, M.; Jeppsson, B.; Uhlén, M.; Molin, G. The Mucosa-Associated Bacteria from the Sigmoid Colon of Nine Healthy 60-Year-Old Individuals,Identified by Bacterial 16S rDNA; Lund University Publications (LUP, Lund): Lund, Sweden, 2003. [Google Scholar]
- Hayashi, H.; Takahashi, R.; Nishi, T.; Sakamoto, M.; Benno, Y. Molecular analysis of jejunal, ileal, caecal and rectosigmoidal human colonic microbiota using 16S rRNA gene libraries and terminal restriction fragment length polymorphism. J. Med. Microbiol. 2005, 54, 1093–1101. [Google Scholar] [PubMed]
- Bik, E.M.; Eckburg, P.B.; Gill, S.R.; Nelson, K.E.; Purdom, E.A.; Francois, F.; Perez-Perez, G.; Blaser, M.J.; Relman, D.A. Molecular Analysis of the Bacterial Microbiota in the Human Stomach. Proc. Natl. Acad. Sci. USA 2006, 103, 732–737. [Google Scholar]
- Lazarevic, V.; Whiteson, K.; Huse, S.; Hernandez, D.; Farinelli, L.; Østerås, M.; Schrenzel, J.; François, P. Metagenomic study of the oral microbiota by Illumina high-throughput sequencing. J. Microbiol. Methods 2009, 79, 266–271. [Google Scholar] [CrossRef] [PubMed]
- Li, X.X.; Wong, G.L.H.; To, K.F.; Wong, V.W.S.; Lai, L.H.; Chow, D.K.L.; Lau, J.U.W.; Sung, J.J.Y.; Ding, C. Bacterial microbiota profiling in gastritis without Helicobacter pylori infection or non-steroidal anti-inflammatory drug use. PLoS One 2009, 4, e7985. [Google Scholar] [PubMed]
- Nasidze, I.; Li, J.; Quinque, D.; Tang, K.; Stoneking, M. Global diversity in the human salivary microbiome. Genome Res. 2009, 19, 636–643. [Google Scholar]
- Turnbaugh, P.J.; Hamady, M.; Yatsunenko, T.; Cantarel, B.L.; Duncan, A.; Ley, R.E.; Sogin, M.L.; Jones, W.J.; Roe, B.A.; Affourtit, J.P.; et al. A core gut microbiome in obese and lean twins. Nature 2009, 457, 480–484. [Google Scholar]
- Rheims, H.; Sproer, C.; Rainey, F.A.; Stackebrandt, E. Molecular biological evidence for the occurrence of uncultured members of the actinomycete line of descent in different environments and geographical locations. Microbiology 1996, 142, 2863–2870. [Google Scholar]
- Huyghe, A.; Francois, P.; Charbonnier, Y.; Tangomo-Bento, M.; Bonetti, E.J.; Paster, B.J.; Bolivar, I.; Baratti-Mayer, D.; Pittet, D.; Schrenzel, J. Novel microarray design strategy to study complex bacterial communities. Appl. Environ. Microbiol. 2008, 74, 1876–1885. [Google Scholar]
- Collins, M.D.; Lawson, P.A.; Willems, A.; Cordoba, J.J.; Fernandez-Garayzabal, J.; Garcia, P.; Cai, J.; Hippe, H.; Farrow, J.A. The phylogeny of the genus Clostridium: Proposal of five new genera and eleven new species combinations. Int. J. Syst. Bacteriol. 1994, 44, 812–826. [Google Scholar]
- Hold, G.L.; Pryde, S.E.; Russell, V.J.; Furrie, E.; Flint, H.J. Assessment of microbial diversity in human colonic samples by 16S rDNA sequence analysis. FEMS Microbiol. Ecol. 2002, 39, 33–39. [Google Scholar]
- Zhang, H.; DiBaise, J.K.; Zuccolo, A.; Kudrna, D.; Braidotti, M.; Yu, Y.; Parameswaran, P.; Crowell, M.D.; Wing, R.; Rittmann, B.E.; et al. Human gut microbiota in obesity and after gastric bypass. PNAS 2009, 106, 2365–2370. [Google Scholar]
- Friedrich, M.J. Microbiome project seeks to understand human body’s microscopic residents. JAMA 2008, 300, 777–778. [Google Scholar]
- Kleessen, B.; Kroesen, A.J.; Buhr, H.J.; Blaut, M. Mucosal and invading bacteria in patients with inflammatory bowel disease compared with controls. Scand. J. Gastroenterol. 2002, 37, 1034–1041. [Google Scholar]
- Swidsinski, A.; Weber, J.; Loening-Baucke, V.; Hale, L.P.; Lochs, H. Spatial organization and composition of the mucosal flora in patients with inflammatory bowel disease. J. Clin. Microbiol. 2005, 43, 3380–3389. [Google Scholar]
- Wang, M.; Molin, G.; Ahrné, S.; Adawi, D.; Jeppsson, B. High proportions of proinflammatory bacteria on the colonic mucosa in a young patient with ulcerative colitis as revealed by cloning and sequencing of 16S rRNA genes. Dig. Dis. Sci. 2007, 52, 620–627. [Google Scholar]
- Gangloff, S.C.; Hijiya, N.; Haziot, A.; Goyert, S.M. Lipopolysaccharide structure influences the macrophage response via CD14-independent and CD14-dependent pathways. Clin. Infect. Dis. 1999, 28, 491–496. [Google Scholar]
- Lindberg, A.A.; Weintraub, A.; Zahringer, U.; Rietschel, E.T. Structure-activity relationships in lipopolysaccharides of Bacteroides fragilis. Rev. Infect. Dis. 1990, 12, S133–S141. [Google Scholar] [CrossRef] [PubMed]
- Poxton, I.R.; Edmond, D.M. Biological activity of Bacteroides lipopolysaccharide-reappraisal. Clin. Infect. Dis. 1995, 20, S149–S153. [Google Scholar] [CrossRef] [PubMed]
- Berezowa, A.B.; Ernst, R.K.; Coats, S.R.; Brahamc, P.H.; Karimi-Naser, L.M.; Darveau, R.P. The structurally similar, penta-acylated lipopolysaccharides of Porphyromonas gingivalis and Bacteroides elicit strikingly different innate immune responses. Microb. Pathog. 2009, 47, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Kasper, D.L.; Hayes, M.E.; Reinap, B.G.; Craft, F.O.; Onderdonk, A.B.; Polk, B.F. Isolation and identification of encapsulated strains of Bacteroides fragilis. J. Infect. Dis. 1977, 136, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Amar, J.; Iglesias, M.A.; Poggi, M.; Knauf, C.; Bastelica, D.; Neyrinck, A.M.; Fava, F.; Tuohy, K.M.; Chabo, C.; et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 2007, 56, 1761–1772. [Google Scholar]
- Cani, P.D.; Bibiloni, R.; Knauf, C.; Waget, A.; Neyrinck, A.M.; Delzenne, N.M.; Burcelin, R. Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes 2008, 57, 1470–1481. [Google Scholar]
- González-Navajas, J.M.; Bellot, P.; Francés, R.; Zapater, P.; Munoz, C.; García-Pagán, J.C.; Pascual, S.; Pérez-Mateo, M.; Bosch, J.; Such, J. Presence of bacterial-DNA in cirrhosis identifies a subgroup of patients with marked inflammatory response not related to endotoxin. J. Hepatol. 2008, 48, 61–67. [Google Scholar]
- Biagi, B.; Nylund, L.; Candela, M.; Ostan, R.; Bucci, L.; Pini, E.; Nikkila, J.; Monti, D.; Satokari, R.; Franceschi, C.; et al. Through ageing, and beyond: Gut microbiota and inflammatory status in seniors and centenarians. PLoS One 2010, 5, e10667. [Google Scholar]
- Onderdonk, A.B. Animal models simulating anaerobic infections. Anaerobe 2005, 11, 189–195. [Google Scholar]
- Onderdonk, A.B.; Bartlett, J.G.; Louie, T.; Sullivan-Seigler, N.; Gorbach, S.L. Microbial synergy in experimental intra-abdominal abscess. Infect. Immun. 1976, 13, 22–26. [Google Scholar]
- Kim, J.M.; Kim, Y.J.; Cho, Y.J. Synergy of Bacteroides fragilis and Escherichia coli in the induction of KC gene expression in mouse peritoneal tissues. Scand. J. Infect. Dis. 2000, 32, 643–649. [Google Scholar] [PubMed]
- Rotstein, O.D.; Vittorini, T.; Kao, J.; McBurney, I.; Nasmith, P.; Grinstein, S.A. A soluble Bacteroides by-product impairs phagocytic killing of Escherichia coli by neutrophils. Infect. Immun. 1989, 57, 745–753. [Google Scholar] [PubMed]
- Montravers, P.; Mohler, J.; Saint Julien, L.; Carbon, C. Evidence of the proinflammatory role of Enterococcus faecalis in polymicrobial peritonitis in rats. Infect. Immun. 1997, 65, 144–149. [Google Scholar] [PubMed]
- Magnuson, D.K.; Weintraub, A.; Pohlman, H.; Maier, R.V. Human endothelial cell adhesiveness of neutrophils, induced by Escherichia coli lipopolysaccharide in vitro, is inhibited by Bacteroides fragilis lipopolysaccharid. J. Immunol. 1989, 143, 3025–3030. [Google Scholar] [PubMed]
- Sokol, H.; Pigneur, B.; Watterlot, L.; Lakhdari, O.; Bermudez-Humara, L.G.; Gratadoux, J.J.; Blugeon, S.; Bridonneau, C.; Furet, J.P.; Corthier, G.; et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. PNAS 2008, 105, 16731–16736. [Google Scholar] [PubMed]
- Forsythe, P.; Bienenstock, J. Immunomodulation by commensal and probiotic bacteria. Immunol. Invest. 2010, 39, 429–448. [Google Scholar]
- Lilley, D.M.; Stillwell, R.H. Probiotics: Growth promoting factors produced by microorganisms. Science 1965, 147, 747–748. [Google Scholar]
- FAO/WHO, Guidelines for the Evaluation of Probiotics in Food; Joint FAO/WHO Working Group Report, London, Ontario, Canada, April 30 and May 1, 2002. WHO: Geneva, Switzerland, 2002.
- Metchnikoff, E. The Prolongation of Life; Heinemann: London, UK, 1907. [Google Scholar]
- Vásquez, A.; Molin, G.; Pettersson, B.; Antonsson, M.; Ahrné, S. DNA-based classification and sequence heterogeneities in the 16S rRNA genes of Lactobacillus casei/paracasei and related species. Syst. Appl. Microbiol. 2005, 28, 430–441. [Google Scholar] [CrossRef]
- Besselink, M.G.H.; van Santvoort, H.C.; Buskens, E.; Boermeester, M.A.; van Goor, H.; Timmerman, H.M.; Nieuwenhuijs, V.B.; Bollen, T.L.; van Ramshorst, B.; Witteman, B.J.M.; et al. Probiotic prophylaxis in predicted severe acute pancreatitis: A randomised, double-blind, placebo-controlled trial. Lancet 2008, 371, 651–659. [Google Scholar] [PubMed]
- Qin, H.L.; Zheng, J.J.; Tong, D.N.; Chen, W.X.; Fan, X.B.; Hang, X.M.; Jiang, Y.Q. Effect of Lactobacillus plantarum enteral feeding on the gut permeability and septic complications in the patients with acute pancreatitis. Eur. J. Clin. Nutr. 2008, 62, 923–930. [Google Scholar] [PubMed]
- Sierra, S.; Lara-Villoslada, F.; Sempere, L.; Olivares, M.; Boza, J.; Xaus, J. Intestinal and immunological effects of daily oral administration of Lactobacillus salivarius CECT5713 to healthy adults. Anaerobe 2010, 16, 195–200. [Google Scholar]
- Takeda, K.; Suzuki, T.; Shimada, S.I.; Shida, K.; Nanno, M.; Okumura, K. Interleukin-12 is involved in the enhancement of human natural killer cell activity by Lactobacillus casei Shirota. Clin. Exp. Immunol. 2006, 146, 109–115. [Google Scholar]
- Roessler, A.; Friedrich, U.; Vogelsang, H.; Bauer, A.; Kaatz, M.; Hipler, U.C.; Schmidt, I.; Jahreis, G. The immune system in healthy adults and patients with atopic dermatitis seems to be affected differently by a probiotic intervention. Clin. Exp. Allergy 2008, 38, 93–102. [Google Scholar]
- Klein, A.; Friedrich, U.; Vogelsang, H.; Jahreis, G. Lactobacillus acidophilus 74-2 and Bifidobacterium animalis subsp. lactis DGCC 420 modulate unspecific cellular immune response in healthy adults. Eur. J. Clin. Nutr. 2008, 62, 584–593. [Google Scholar] [CrossRef] [PubMed]
- Pelto, L.; Isolauri, E.; Lilius, E.M.; Nuutila, J.; Salminen, S. Probiotic bacteria down-regulate the milk-induced inflammatory response in milk-hypersensitive subjects but have an immunostimulatory effect in healthy subjects. Clin. Exp. Allergy 1998, 28, 1474–1479. [Google Scholar]
- Karczewski, J.; Troost, F.J.; Konings, I.; Dekker, J.; Kleerebezem, M.; Brummer, R.J.M.; Wells, J.M. Regulation of human epithelial tight junction proteins by Lactobacillus plantarum in vivo and protective effects on the epithelial barrier. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 298, G851–G859. [Google Scholar]
- Kiessling, G.; Schneider, J.; Jahreis, G. Long-term consumption of fermented dairy products over 6 months increases HDL cholesterol. Eur. J. Clin. Nutr. 2002, 56, 843–849. [Google Scholar]
- Hata, Y.; Yamamoto, M.; Ohni, M.; Nakajima, K.; Nakamura, Y.; Takano, T. A placebo-controlled study of the effect of sour milk on blood pressure in hypertensive. Am. J. Clin. Nutr. 1996, 64, 767–771. [Google Scholar]
- Bukowska, H.; Pieczul-Mróz, J.; Jastrzebsk, K.; Chelstowski, K.; Naruszewicz, M. Significant decrease in fibrinogen and LDL-cholesterol levels upon supplementation of the diet with Lactobacillus plantarum (ProViva) in subjects with moderately elevated cholesterol concentrations. Atherosclerosis 1998, 137, 437–438. [Google Scholar] [CrossRef] [PubMed]
- Naruszewicz, M.; Johansson, M.L.; Zapolska-Downar, D.; Bukowska, H. Effect of Lactobacillus plantarum 299v on cardiovascular disease risk factors in smokers. Am. J. Clin. Nutr. 2002, 76, 1249–1255. [Google Scholar] [PubMed]
- Gill, H.S.; Rutherfurd, K.J.; Cross, M.L.; Gopal, P.K. Enhancement of immunity in the elderly by dietary supplementation with the probiotic Bifidobacterium lactis HN019. Am. J. Clin. Nutr. 2001, 74, 833–839. [Google Scholar] [PubMed]
- Loguercio, C.; De Simone, T.; Federico, A.; Terracciano, F.; Tuccillo, C.; Di Chicco, M.; Cartenì, M. Gut-liver axis: A new point of attack to treat chronic liver damage? Am. J. Gastroenterol. 2002, 97, 2144–2146. [Google Scholar] [PubMed]
- Loguercio, C.; Federico, A.; Tuccillo, C.; Terracciano, F.; D’Auria, M.V.; De Simone, C.; Del Vecchio Blanco, C. Beneficial effects of a probiotic VSL#3 on parameters of liver dysfunction in chronic liver diseases. J. Clin. Gastroenterol. 2005, 39, 540–543. [Google Scholar]
- Kirpich, I.A.; Solovieva, N.V.; Leikhter, S.N.; Shidakova, N.A.; Lebedeva, O.V.; Sidorov, P.I.; Bazhukova, T.A.; Soloviev, A.G.; Barve, S.S.; McClain, C.J.; et al. Probiotics restore bowel flora and improve liver enzymes in human alcohol-induced liver injury: A pilot study. Alcohol 2008, 42, 675–682. [Google Scholar] [PubMed]
- Stadlbauer, V.; Mookerjee, R.P.; Hodges, S.; Wright, G.A.K.; Davies, N.A.; Jalan, R. Effect of probiotic treatment on deranged neutrophil function and cytokine responses in patients with compensated alcoholic cirrhosis. J. Hepatol. 2008, 48, 945–951. [Google Scholar]
- Liu, Q.; Duan, Z.P.; Ha, D.K.; Bengmark, S.; Kurtovic, J.; Riordan, S.M. Synbiotic modulation of gut flora: Effect on minimal hepatic encephalopathy in patients with cirrhosis. Hepatology 2004, 39, 1441–1449. [Google Scholar]
- Macbeth, W.A.; Kass, E.H.; McDermott, W.V., Jr. Treatment of hepatic encephalopathy by alteration of intestinal flora with Lactobacillus acidophilus. Lancet 1965, 1, 399–403. [Google Scholar] [PubMed]
- Read, A.E.; McCarthy, C.F.; Heaton, K.W.; Laidlaw, J. Lactobacillus acidophilus (Enpac) in treatment of hepatic encephalopathy. BMJ 1966, 1, 1267–1269. [Google Scholar] [PubMed]
- Bajaj, J.S.; Saeian, K.; Christensen, K.M.; Hafeezullah, M.; Varma, R.R.; Franco, J.; Pleuss, J.A.; Krakower, G.; Hoffmann, R.G.; Binion, D.G. Probiotic yogurt for the treatment of minimal hepatic encephalopathy. Am. J. Gastroenterol. 2008, 103, 1707–1715. [Google Scholar]
- Rayes, N.; Seehofer, D.; Hansen, S.; Boucsein, K.; Müller, A.R.; Serke, S.; Bengmark, S.; Neuhaus, P. Early enteral supply of lactobacillus and fiber versus selective bowel decontamination: A controlled trial in liver transplant recipients. Transplantation 2002, 74, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Rayes, N.; Seehofer, D.; Theruvath, T.; Schiller, R.A.; Langrehr, J.M.; Jonas, S.; Bengmark, S.; Neuhaus, P. Supply of pre- and probiotics reduces bacterial infection rates after liver transplantation—a randomized, double-blind trial. Am. J. Transplant. 2005, 5, 125–130. [Google Scholar]
- McNaught, C.E.; Woodcook, N.P.; Andersson, A.D.G.; MacFie, J. A prospective randomized trial of probiotics in critically ill patients. Clin. Nutr. 2005, 24, 211–219. [Google Scholar]
- Klarin, B.; Wullt, M.; Palmquist, I.; Molin, G.; Larsson, A.; Jeppsson, B. Lactobacillus plantarum 299v reduces colonization of Clostridium difficile in critically ill patients treated with antibiotics. Acta Anaesthesiol. Scand. 2008, 52, 1096–1102. [Google Scholar] [CrossRef] [PubMed]
- Alberda, C.; Gramlich, L.; Meddings, J.; Field, C.; McCargar, L.; Kutsogiannis, D.; Fedorak, R.; Madsen, K. Effects of probiotic therapy in critically ill patients: A randomized, double-blind, placebo-controlled trial. Am. J. Clin. Nutr. 2007, 85, 816–823. [Google Scholar] [PubMed]
- Taylor, A.L.; Hale, J.; Hales, B.J.; Dunstan, J.A.; Thomas, W.R.; Prescott, S.L. FOXP3 mRNA expression at 6 months of age is higher in infants who develop atopic dermatitis, but is not affected by giving probiotics from birth. Pediatr. Allergy Immunol. 2007, 18, 10–19. [Google Scholar]
- Kalliomäki, M.; Salminen, S.; Arvilommi, H.; Kero, P.; Koskinen, P.; Isolauri, E. Probiotics in primary prevention of atopic disease: a randomised placebo-controlled trial. Lancet 2001, 357, 1076–1079. [Google Scholar]
- Isolauri, E.; Arvola, T.; Sütas, Y.; Moilanen, E.; Salminen, S. Probiotics in the management of atopic eczema. Clin. Exp. Allergy 2000, 30, 1604–1610. [Google Scholar]
- Ouwehand, A.C.; Nermes, M.; Collado, M.C.; Rautonen, N.; Salminen, S.; Isolauri, E. Specific probiotics alleviate allergic rhinitis during the birch pollen season. World J. Gastroenterol. 2009, 15, 3261–3268. [Google Scholar]
- Viljanen, M.; Kuitunen, M.; Haahtela, T.; Juntunen-Backman, K.; Korpela, R.; Savilahti, E. Probiotic effects on faecal inflammatory markers and on faecal IgA in food allergic atopic eczema/dermatitis syndrome infants. Pediatr. Allergy Immunol. 2005, 16, 65–71. [Google Scholar]
- Martínez-Cañavate, A.; Sierra, S.; Lara-Villoslada, F.; Romero, J.; Maldonado, J.; Boza, J.; Xaus, J.; Olivares, M. A probiotic dairy product containing L. gasseri CECT5714 and L. coryniformis CECT5711 induces immunological changes in children suffering from allergy. Pediatr. Allergy Immunol. 2009, 20, 592–600. [Google Scholar] [CrossRef] [PubMed]
- Mohan, R.; Koebnick, C.; Schildt, J.; Mueller, M.; Radke, M.; Blaut, M. Effects of Bifidobacterium lactis Bb12 supplementation on body weight, fecal pH, acetate, lactate, calprotectin, and IgA in preterm infants. Pediatr. Res. 2008, 64, 418–422. [Google Scholar] [CrossRef] [PubMed]
- Schultz, M.; Timmer, A.; Herfarth, H.H.; Sartor, R.B.; Vanderhoof, J.A.; Rath, H.C. Lactobacillus GG in inducing and maintaining remission of Crohn’s disease. BMC Gastroenterol. 2004, 4, 5. [Google Scholar] [CrossRef] [PubMed]
- Bousvaros, A.; Guandalini, S.; Baldassano, R.N.; Botelho, C.; Evans, J.; Ferry, G.D.; Goldin, B.; Hartigan, L.; Kugathasan, S.; Levy, J.; et al. A randomized, double-blind trial of Lactobacillus GG versus placebo in addition to standard maintenance therapy for children with Crohn’s disease. Inflamm. Bowel Dis. 2005, 11, 833–839. [Google Scholar] [PubMed]
- Prantera, C.; Scribano, M.L.; Falasco, G.; Andreoli, A.; Luzi, C. Ineffectiveness of probiotics in preventing recurrence after curative resection for Crohn’s disease: A randomised controlled trial with Lactobacillus GG. Gut 2002, 51, 405–409. [Google Scholar] [PubMed]
- Guandalini, S. Use of Lactobacillus-GG in paediatric Crohn’s disease. Dig. Liver Dis. 2002, 34, S63–S65. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.; Andrew, H.; Kirschne, B.S.; Guandalini, S. Is Lactobacillus GG helpful in children with Crohn’s disease? Results of a preliminary, open-label study. J. Pediatr. Gastroenterol. Nutr. 2000, 31, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Marteau, P.; Lemann, M.; Seksik, P.; Laharie, D.; Colombei, J.F.; Bouhnik, Y.; Cadiot, G.; Soulé, J.C.; Bourreille, A.; Metman, E.; et al. Ineffectiveness of Lactobacillus johnsonii LA1 for prophylaxis of postoperative recurrence in Crohn’s disease: A randomised, double blind, placebo controlled GETAID trial. Gut 2006, 55, 842–847. [Google Scholar] [PubMed]
- Van Gossum, A.; Dewit, O.; Louis, E.; de Hertogh, G.; Baert, F.; Fontaine, F.; DeVos, M.; Enslen, M.; Paintin, M.; Franchimont, D. Multicenter randomized-controlled clinical trial of probiotics (Lactobacillus johnsonii, LA1) on early endoscopic recurrence of Crohn’s disease after lleo-caecal resection. Inflamm. Bowel Dis. 2007, 13, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Pronio, A.; Montesani, C.; Butteroni, C.; Vecchione, S.; Mumolo, G.; Vestri, A.; Vitolo, D.; Boirivant, M. Probiotic administration in patients with ileal pouch-anal anastomosis for ulcerative colitis is associated with expansion of mucosal regulatory cells. Inflamm. Bowel Dis. 2008, 14, 662–668. [Google Scholar]
- Sood, A.; Midha, V.; Makharia, G.K.; Ahuja, V.; Singal, D.; Goswami, P.; Tandon, R.K. The probiotic preparation, VSL#3 induces remission in patients with mild-to-moderately active ulcerative colitis. Clin. Gastroenterol. Hepatol. 2009, 7, 1202–1209. [Google Scholar]
- Bibiloni, R.; Fedorak, R.N.; Tannock, G.W.; Madsen, K.L.; Gionchetti, P.; Campieri, M.; De Simone, C.; Sartor, R.B. VSL#3 probiotic-mixture induces remission in patients with active ulcerative colitis. Am. J. Gastroenterol. 2005, 100, 1539–1546. [Google Scholar]
- Gionchetti, P.; Rizzell, F.; Morselli, C.; Poggioli, G.; Tambasco, R.; Calabrese, C.; Brigidi, P.; Vitali, B.; Straforini, G.; Campieri, M. High-dose probiotics for the treatment of active pouchitis. Dis. Colon Rectum 2007, 50, 2075–2082. [Google Scholar]
- Tsuda, Y.; Yoshimatsu, Y.; Aoki, H.; Nakamura, K.; Irie, M.; Fukuda, K.; Hosoe, N.; Takada, N.; Shirai, K.; Suzuki, Y. Clinical effectiveness of probiotics therapy (BIO-THREE) in patients with ulcerative colitis refractory to conventional therapy. Scand. J. Gastroenterol. 2007, 42, 1306–1311. [Google Scholar]
- Matthes, H.; Krummenerl, T.; Giensch, M.; Wolff, C.; Schulze, J. Clinical trial: Probiotic treatment of acute distal ulcerative colitis with rectally administered Escherichia coli Nissle 1917 (EcN). BMC Complement. Altern. Med. 2010, 10, 13. [Google Scholar] [CrossRef] [PubMed]
- Lorea Baroja, M.; Kirjavainen, P.V.; Hekmat, S.; Reid, G. Anti-inflammatory effects of probiotic yogurt in inflammatory bowel disease patients. Clin. Exp. Immunol. 2007, 149, 470–479. [Google Scholar]
- Zocco, M.A.; dal Verme, L.Z.; Cremonini, F.; Piscaglia, A.C.; Nista, E.C.; Candelli, M.; Novi, M.; Rigante, D.; Cazzato, I.A.; Ojetti, V.; et al. Efficacy of Lactobacillus GG in maintaining remission of ulcerative colitis. Aliment. Pharmacol. Ther. 2006, 23, 1567–1574. [Google Scholar] [PubMed]
- Kato, K.; Mizuno, S.; Umesaki, Y.; Ishii, Y.; Sugitani, M.; Imaoka, A.; Otsuka, M.; Hasunuma, O.; Kurihara, R.; Iwasaki, A.; et al. Randomized placebo-controlled trial assessing the effect of bifidobacteria-fermented milk on active ulcerative colitis. Aliment. Pharmacol. Ther. 2004, 20, 1133–1141. [Google Scholar]
- Cui, H.H.; Chen, C.L.; Wang, J.D.; Yang, Y.J.; Cun, Y.; Wu, J.B.; Liu, Y.H.; Dan, H.L.; Jian, Y.T.; Chen, X.Q. Effects of probiotic on intestinal mucosa of patients with ulcerative colitis. World J. Gastroenterol. 2004, 10, 1521–1525. [Google Scholar]
- Zhang, J.W.; Du, P.; Chen, D.W.; Cui, L.; Ying, C.M. Effect of viable Bifidobacterium supplement on the immune status and inflammatory response in patients undergoing resection for colorectal cancer. Zhonghua Wei Chang Wai Ke Za Zhi 2010, 13, 40–43. (in Chinese).. [Google Scholar] [PubMed]
- Delia, P.; Sansotta, G.; Donato, V.; Frosina, P.; Messina, G.; De Renzis, C.; Famularo, G. Use of probiotics for prevention of radiation-induced diarrhea. World J. Gastroenterol. 2007, 13, 912–915. [Google Scholar]
- Urbancsek, H.; Kazar, T.; Mezes, I.; Neumann, K. Results of a double-blind, randomized study to evaluate the efficacy and safety of Antibiophilus in patients with radiation-induced diarrhoea. Eur. J. Gastroenterol. Hepatol. 2001, 13, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Osterlund, P.; Ruotsalainen, T.; Korpela, R.; Saxelin, M.; Ollus, A.; Valta, P.; Kour, M.; Elomaa, I.; Joensuu, H. Lactobacillus supplementation for diarrhoea related to chemotherapy of colorectal cancer: A randomised study. Br. J. Cancer 2007, 97, 1028–1034. [Google Scholar] [CrossRef] [PubMed]
- Salminen, E.; Elomaa, I.; Minkkinen, J.; Vapaatalo, H.; Salminen, S. Preservation of intestinal integrity during radiotherapy using live Lactobacillus acidophilus cultures. Clin. Radiol. 1988, 39, 435–437. [Google Scholar] [CrossRef] [PubMed]
- Giralt, J.; Regadera, J.P.; Verges, R.; Romero, J.; de la Fuente, I.; Biete, A.; Villoria, J.; Cobo, J.M.; Guarner, F. Effects of probiotic Lactobacillus casei DN-114 001 in prevention of radiation induced diarrhea: results from multicenter, randomized, placebo-controlled nutritional trial. Int. J. Radiat. Oncol. Biol. Phys. 2008, 71, 1213–1219. [Google Scholar] [CrossRef] [PubMed]
- Pandolfi, F.; Cianci, R.; Pagliari, D.; Landolfi, R.; Cammarota, G. Cellular mediators of inflammation: Tregs and TH17 cells in gastrointestinal diseases. Mediators Inflamm. 2009, 2009, 132028. [Google Scholar] [PubMed]
- Tang, Q.; Bluestone, J.A. The Foxp3+ regulatory T cell: A jack of all trades, master of regulation. Nat. Immunol. 2008, 9, 239–244. [Google Scholar]
- Baecher-Allan, C.; Brown, J.A.; Freeman, G.J.; Hafler, D.A. CD4+CD25 high regulatory cells in human peripheral blood. J. Immunol. 2001, 167, 1245–1253. [Google Scholar] [PubMed]
- Maul, J.; Loddenkemper, C.; Mundt, P.; Berg, E.; Giese, T.; Stallmach, A.; Zeitz, M.; Duchmann, R. Peripheral and intestinal regulatory CD4+CD25high T cells in inflammatory bowel disease. Gastroenterology 2005, 128, 1868–1878. [Google Scholar]
- McIntyre, T.M.; Strober, W. Gut-associated Lymphoid Tissue. Regulation of IgA B-cell Development. In Mucosal Immunology; Ogra, L.P., Mestecky, J., Lamm, M.E., Strober, W., Bienenstock, J., McGhee, J.R., Eds.; Academic Press: New York, NY, USA, 1999; pp. 319–356. [Google Scholar]
- Benson, E.B.; Strober, W. Regulation of IgA secretion by T cell clones derived from the human gastrointestinal tract. J. Immunol. 1988, 140, 1874–1882. [Google Scholar]
- Ogata, K.; An, E.; Shioi, Y.; Nakamura, K.; Luo, S.; Yokose, N.; Minami, S.; Dan, K. Association between natural killer cell activity and infection in immunologically normal elderly people. Clin. Exp. Immunol. 2001, 124, 392–397. [Google Scholar]
- Penders, J.; Thijs, C.; van den Brandt, P.A.; Kummeling, I.; Snijders, B.; Stelma, F.; Adams, H.; van Ree, R.; Stobberingh, E.E. Gut microbiota composition and development of atopic manifestations in infancy: The KOALA Birth Cohort Study. Gut 2007, 56, 661–667. [Google Scholar]
- Wang, M.; Karlsson, C.; Olsson, C.; Adlerberth, I.; Wold, A.; Strachan, D.P.; Martricardi, P.M.; et al. Reduced diversity in the early fecal microbiota of infants with atopic eczema. J. Allergy Clin. Immunol. 2008, 121, 129–134. [Google Scholar] [PubMed]
- Saarinen, K.M.; Sarnesto, A.; Savilahti, E. Markers of inflammation in the feces of infants with cow’s milk allergy. Pediatr. Allergy Immunol. 2002, 13, 188–194. [Google Scholar]
- Majamaa, H.; Miettinen, A.; Laine, S.; Isolauri, E. Intestinal inflammation in children with atopic eczema: Faecal eosinophil cationic protein and tumour necrosis factor-alpha as non-invasive indicators of food allergy. Clin. Exp. Allergy 1996, 26, 181–187. [Google Scholar]
- Ahrne, S.; Johansson Hagslatt, M.-L. Effect of lactobacilli on paracellular permeability in the gut. Nutrients 2011, 3, 104–117. [Google Scholar]
- Backhed, F.; Ding, H.; Wang, T.; Hooper, L.V.; Koh, G.Y.; Nagy, A.; Semenkovich, C.F.; Gordon, J.I. The gut microbiota as an environmental factor that regulates fat storage. Proc. Natl. Acad. Sci. USA 2004, 101, 15718–15723. [Google Scholar]
- Ley, R.E.; Turnbaugh, P.J.; Klein, S.; Gordon, J.I. Microbial ecology: Human gut microbes associated with obesity. Nature 2006, 444, 1022–1023. [Google Scholar]
- Raoult, D. Obesity pandemics and the modification of digestive bacterial flora. Eur. J. Clin. Microbiol. Infect. Dis. 2008, 27, 631–634. [Google Scholar]
- Raoult, D. Probiotics and obesity: A link? Nat. Rev. Microbiol. 2009, 7, 616. [Google Scholar] [CrossRef] [PubMed]
- Ehrlich, S.D. Probiotics—little evidence for a link to obesity. Nat. Rev. Microbiol. 2009, 7, 901. [Google Scholar] [CrossRef]
- Delzenne, N.; Reid, G. No causal link between obesity and probiotics. Nat. Rev. Microbiol. 2009, 7, 901. [Google Scholar] [CrossRef]
- Kannel, W.B.; Wolf, P.A.; Castelli, W.P.; D’Agnostino, R.B. Fibrinogen and risk of cardiovascular disease. The Framingham Study. JAMA 1987, 258, 1183–1186. [Google Scholar] [PubMed]
- Church, D.F.; Pryor, W.A. Free-radical chemistry of cigarette smoke and its toxicological implications. Environ. Health Perspect. 1985, 64, 111–126. [Google Scholar] [CrossRef] [PubMed]
- Tappia, P.S.; Troughton, K.L.; Langley-Evans, S.; Grimble, R.F. Cigarette smoking influences cytokine production and antioxidant defences. Clin. Sci. 1995, 88, 485–489. [Google Scholar]
- Goodwin, J.S. Decreased immunity and increased morbidity in the elderly. Nutr. Rev. 1995, 53, S41–S44. [Google Scholar]
- Kalache, A. Active ageing makes the difference. Bull. World Health Organ. 1999, 77, 299. [Google Scholar]
- Guigoz, Y.; Doré, J.; Schiffrin, E.J. The inflammatory status of old age can be nurtured from the intestinal environment. Curr. Opin. Clin. Nutr. Metab. Care 2008, 11, 13–20. [Google Scholar]
- Mikelsaar, M.; Stsepetova, J.; Hütt, P.; Kolk, H.; Sepp, E.; Lõivukene, K.; Zilmer, K.; Zilmer, M. Intestinal Lactobacillus sp. is associated with some cellular and metabolic characteristics of blood in elderly people. Anaerobe 2010, 16, 240–246. [Google Scholar] [PubMed]
- Zeuzem, S. Gut-liver axis. Int. J. Colorectal Dis. 2000, 15, 59–82. [Google Scholar]
- Berg, R.D.; Garlinton, A.W. Translocation of certain indigenous bacteria from the gastrointestinal tract to the mesenteric lymph nodes and other organs in a gnotobiotic mouse model. Infect. Immun. 1979, 23, 403–411. [Google Scholar]
- Jacob, A.I.; Goldberg, P.H.; Bloom, D.; Degenshein, G.A.; Korin, P.J. Endotoxin and bacteria in portal blood. Gastroenterology 1977, 72, 1268–1270. [Google Scholar]
- Nolan, J.P. Endotoxin, reticuloendothelial function and liver injury. Hepatology 1981, 1, 458–465. [Google Scholar]
- Mathison, J.C.; Ulevitch, R.J. The clearance, tissue distribution and cellular localization of intravenously injected lipopolysaccharide in rabbits. J. Immunol. 1979, 123, 2133–2143. [Google Scholar]
- Ruiter, D.J.; van der Meulen, J.; Brouwer, A.; Hummel, M.J.; Mauw, B.J.; van der Ploeg, J.C.; Wisse, E. Uptake by liver cells of endotoxin following its intravenous injection. Lab. Invest. 1981, 45, 38–45. [Google Scholar]
- Ishibashi, H.; Nakamura, M.; Komori, A.; Migita, K.; Shimoda, S. Liver architecture, cell function, and disease. Semin. Immunopathol. 2009, 31, 399–409. [Google Scholar] [CrossRef] [PubMed]
- Racanelli, V.; Rehermann, B. The liver as an immunological organ. Hepatology 2006, 43, 54–62. [Google Scholar]
- Van Leeuwen, P.A.; Boermeester, M.A.; Houdijk, A.P.; Ferwerda, C.C.; Cuesta, M.A.; Meyer, S.; Wesdorp, R.I. Clinical significance of translocation. Gut 1994, 35, 28–34. [Google Scholar]
- Akira, S.; Takeda, K.; Kaisho, T. Toll-like receptors: Critical proteins linking innate and acquired immunity. Nat. Immunol. 2001, 2, 675–680. [Google Scholar]
- Mencin, A.; Kluwe, J.; Schwabe, R.F. Toll-like receptors as targets in chronic liver diseases. Gut 2009, 58, 704–720. [Google Scholar]
- Szabo, G.; Bala, S. Alcoholic liver disease and the gut-liver axis. World J. Gastroenterol. 2010, 16, 1321–1329. [Google Scholar]
- Bataller, R.; Brenner, D.A. Liver fibrosis. J. Clin. Invest. 2005, 115, 209–218. [Google Scholar]
- Seki, E.; De Minicis, S.; Osterreicher, C.H.; Kluwe, J.; Osawa, Y.; Brenner, D.A.; Schwabe, R.F. TLR4 enhances TGF-beta signaling and hepatic fibrosis. Nat. Med. 2007, 13, 1324–1332. [Google Scholar]
- Lockwood, A.H.; Yap, E.W.; Wong, W.H. Cerebral ammonia metabolism in patients with severe liver disease and minimal hepatic encephalopathy. J. Cereb. Blood Flow Metab. 1991, 11, 337–341. [Google Scholar]
- Cordoba, J.; Alonso, J.; Rovira, A.; Jacas, C.; Sanpedro, F.; Castells, L.; Vargas, V.; Margarit, C.; Kulisewsky, J.; Esteban, R.; et al. The development of low-grade cerebral edema in cirrhosis is supported by the evolution of (1)H-magnetic resonance abnormalities after liver transplantation. J. Hepatol. 2001, 35, 598–604. [Google Scholar] [CrossRef] [PubMed]
- Stewart, S.; Jones, D.; Day, C.P. Alcoholic liver disease: New insights into mechanisms and preventative strategies. Trends Mol. Med. 2001, 7, 408–413. [Google Scholar]
- Morgan, T.R.; Mandayam, S.; Jamal, M.M. Alcohol and hepatocellular carcinoma. Gastroenterology 2004, 127, S87–S96. [Google Scholar]
- Apte, U.M.; Banerjee, A.; McRee, R.; Wellberg, E.; Ramaiah, S.K. Role of osteopontin in hepatic neutrophil infiltration during alcoholic steatohepatitis. Toxicol. Appl. Pharmacol. 2005, 207, 25–38. [Google Scholar]
- Ramaiah, S.K.; Jaeschke, H. Hepatic neutrophil infiltration in the pathogenesis of alcohol-induced liver injury. Toxicol. Mech. Methods 2007, 17, 431–440. [Google Scholar]
- McClain, C.J.; Barve, S.; Deaciuc, I.; Kugelmas, M.; Hill, D. Cytokines in alcoholic liver disease. Semin. Liver Dis. 1999, 19, 205–220. [Google Scholar]
- Such, J.; Guardiola, J.V.; de Juan, J.; Casellas, J.A.; Pascual, S.; Aparicio, J.R.; Solá-Vera, J.; Pérez-Mateo, M. Ultrastructural characteristics of distal duodenum mucosa in patients with cirrhosis. Eur. J. Gastroenterol. Hepatol. 2002, 14, 371–376. [Google Scholar]
- Zuckerman, M.J.; Menzies, I.S.; Ho, H.; Gregory, G.G.; Casner, N.A.; Crane, R.S.; Hernandez, J.A. Assessment of intestinal permeability and absorption in cirrhotic patients with ascites using combined sugar probes. Dig. Dis. Sci. 2004, 49, 621–626. [Google Scholar]
- Rao, R.K.; Seth, A.; Sheth, P. Recent advances in alcoholic liver disease. I. Role of intestinal permeability and endotoxiemia in alcoholic liver disease. Am. J. Physiol. Gastrointest. Liver Physiol. 2004, 286, G881–G884. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.H.; Wu, X.J.; Li, J.S. Influence of portal pressure change on intestinal permeability in patients with portal hypertension. HepatobiliaryPancreat. Dis. Int. 2002, 1, 510–514. [Google Scholar]
- Medina, J.; Fernández-Salazar, L.I.; García-Buey, L.; Moreno-Otero, R. Approach to the pathogenesis and treatment of nonalcoholic steatohepatitis. Diabetes Care 2004, 27, 2057–2066. [Google Scholar]
- Younossi, Z.M.; Diehl, A.M.; Ong, J.P. Nonalcoholic fatty liver disease: An agenda for clinical research. Hepatology 2002, 35, 746–752. [Google Scholar]
- Lirussi, F.; Mastropasqua, E.; Orando, S.; Orlando, R. Probiotics for non-alcoholic fatty liver disease and/or steatohepatitis. Cochrane DatabaseSyst. Rev. 2007. [Google Scholar]
- Hwang, S.T.; Cho, Y.K.; Park, J.H.; Kim, H.J.; Park, D.I.; Sohn, C.I.; Jeon, W.K.; Kim, B.I.; Won, K.H.; Jin, W. Relationship of non-alcoholic fatty liver disease to colorectal adenomatous polyps. J. Gastroenterol. Hepatol. 2010, 25, 562–567. [Google Scholar]
- Farrell, G.C. Is bacterial ash the flash that ignites NASH? Gut 2001, 48, 148–149. [Google Scholar] [CrossRef] [PubMed]
- Nair, S.; Cope, K.; Risby, T.H.; Diehl, A.M. Obesity and female gender increase breath ethanol concentration: Potential implications for the pathogenesis of nonalcoholic steatohepatitis. Am. J. Gastroenterol. 2001, 96, 1200–1204. [Google Scholar]
- Wigg, A.J.; Roberts-Thomson, I.C.; Dymock, R.B.; McCarthy, P.J.; Grose, R.H.; Cummins, A.G. The role of small intestinal bacterial overgrowth, intestinal permeability, endotoxaemia, and tumour necrosis factor alpha in the pathogenesis of non-alcoholic steatohepatitis. Gut 2001, 48, 206–211. [Google Scholar] [CrossRef] [PubMed]
- Pessayre, D.; Mansouri, A.; Fromenty, B. Nonalcoholic steatosis and steatohepatitis. V. Mitochondrial dysfunction in steatohepatitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2002, 282, G193–G199. [Google Scholar] [PubMed]
- Neuman, M.G. Immune dysfunction in inflammatory bowel disease. Transl. Res. 2007, 149, 173–186. [Google Scholar]
- Asakura, H.; Suzuki, A.; Ohtsuka, K.; Hasegawa, K.; Sugimura, K. Gut-associated lymphoid tissues in ulcerative colitis. J. Parenter. Enteral. Nutr. 1999, 23, 25–28. [Google Scholar]
- Sasakawa, T.; Takizawa, H.; Bannai, H.; Narisawa, R.; Asakura, H. Activated CD4+ and CD8+ cells in the colonic mucosa of ulcerative colitis patients: their relationship to HLA-DR antigen expression on the colonic epithelium and serum soluble CD25 levels. Digestion 1995, 56, 516–522. [Google Scholar]
- Blumberg, R.S. Inflammation in the intestinal tract: Pathogenesis and treatment. Dig. Dis. 2009, 27, 455–464. [Google Scholar]
- Dharmani, P.; Chadee, K. Biologic therapies against inflammatory bowel disease: A dysregulated immune system and the cross talk with gastrointestinal mucosa hold the key. Curr. Mol. Pharmacol. 2008, 1, 195–212. [Google Scholar]
- Abreu, M.T.; Arnold, E.T.; Thomas, L.S.; Gonsky, R.; Zhou, Y.; Hu, B.; Arditi, M. TLR4 and MD-2 expression is regulated by immune-mediated signals in human intestinal epithelial cells. J. Biol. Chem. 2002, 277, 20431–20437. [Google Scholar]
- Cario, E.; Podolsky, D.K. Differential alteration in intestinal epithelial cell expression of toll-like receptor 3 (TLR3) and TLR4 in inflammatory bowel disease. Infect. Immun. 2000, 68, 7010–7017. [Google Scholar] [CrossRef] [PubMed]
- Baumgart, D.C.; Thomas, S.; Przesdzing, I.; Metzke, D.; Bielecki, C.; Lehmann, S.M.; Lehnardt, S.; Dörffel, Y.; Sturm, A.; Scheffold, A.; et al. Exaggerated inflammatory response of primary human myeloid dendritic cells to lipopolysaccharide in patients with inflammatory bowel disease. Clin. Exp. Immunol. 2009, 157, 423–436. [Google Scholar]
- Kanai, K.; Watanabe, M. Clinical application of human CD4+CD25+ regulatory T cells for the treatment of inflammatory bowel disease. Expert. Opin. Biol. Ther. 2005, 5, 451–462. [Google Scholar]
- Pullan, R.D.; Thomas, G.A.O.; Rhodes, M.; Newcombe, R.G.; Williams, G.T.; Allen, A.; Rhodes, J. Thickness of adherent mucus gel on colonic mucosa in humans and its relevance to colitis. Gut 1994, 35, 353–359. [Google Scholar]
- Corfield, A.P.; Myerscough, N.; Bradfield, N.; Corfield, C.D.A.; Gough, M.; Clamp, J.R.; Durdey, P.; Warren, B.F.; Bartolo, D.C.C.; King, K.R.; et al. Colonic mucins in ulcerative colitis: Evidence for loss of sulphation. Glycoconj. J. 1996, 13, 809–822. [Google Scholar]
- Nishikawa, J.; Kudo, T.; Sakata, S.; Benno, Y.; Sugiyama, T. Diversity of mucosa-associated microbiota in active and inactive ulcerative colitis. Scand. J. Gastroenterol. 2009, 44, 180–186. [Google Scholar]
- Kotlowski, R.; Bernstein, C.N.; Sepehri, S.; Krause, D.O. High prevalence of Escherichia coli belonging to the B2+D phylogenetic group in inflammatory bowel disease. Gut 2007, 56, 669–675. [Google Scholar] [PubMed]
- Rowan, F.E.; Docherty, N.G.; Coffey, J.C.; O’Connell, P.R. Sulphate-reducing bacteria and hydrogen sulphide in the aetiology of ulcerative colitis. Br. J. Surg. 2009, 96, 151–158. [Google Scholar]
- Pitcher, M.C.; Beatty, E.R.; Cummings, J.H. The contribution of sulphate reducing bacteria and 5-aminosalicylic acid to faecal sulphide in patients with ulcerative colitis. Gut 2000, 46, 64–72. [Google Scholar]
- Fabia, R.; Ar’Rajab, A.; Johansson, M.L.; Andersson, R.; Willén, R.; Jeppsson, B.; Molin, G.; Bengmark, S. Impairment of bacterial flora in human ulcerative colitis and experimental colitis in the rat. Digestion 1993, 54, 248–255. [Google Scholar]
- Andoh, A.; Sakata, S.; Koizumi, Y.; Mitsuyama, K.; Fujiyama, Y.; Benno, Y. Terminal restriction fragment length polymorphism analysis of the diversity of fecal microbiota in patients with ulcerative colitis. Inflamm. Bowel. Dis. 2007, 13, 955–962. [Google Scholar]
- Zoetendal, E.G.; von Wright, A.; Vilpponen-Salmela, T.; Ben-Amor, K.; Akkermans, A.D.; de Vos, W.M. Mucosa-associated bacteria in the human gastrointestinal tract are uniformly distributed along the colon and differ from the community recovered from feces. Appl. Environ. Microbiol. 2002, 68, 3401–3407. [Google Scholar]
- Zhang, M.; Liu, B.; Zhang, Y.; Wei, H.; Lei, Y.; Zhao, L. Structural shifts of mucosa-associated lactobacilli and Clostridium leptum subgroup in patients with ulcerative colitis. J. Clin. Microbiol. 2007, 45, 496–500. [Google Scholar]
- Sang, L.X.; Chang, B.; Zhang, W.L.; Wu, X.M.; Li, X.H.; Jiang, M. Remission induction and maintenance effect of probiotics on ulcerative colitis: a meta-analysis. World J. Gastroenterol. 2010, 16, 1908–1915. [Google Scholar]
- Sandborn, W.J. Pouchitis following ileal pouch-anal anastomosis: Definition, pathogenesis, and treatment. Gastroenterology 1994, 107, 1856–1860. [Google Scholar] [PubMed]
- Elahi, B.; Nikfar, S.; Derakhshani, S.; Vafaie, M.; Abdollahi, M. On the benefit of probiotics in the management of pouchitis in patients underwent ileal pouch anal anastomosis: A meta-analysis of controlled clinical trials. Dig. Dis. Sci. 2008, 53, 1278–1284. [Google Scholar]
- Kuhbacher, T.; Ott, S.J.; Helwig, U.; Mimura, T.; Rizzello, F.; Kleessen, B.; Gionchetti, P.; Blaut, M.; Campieri, M.; Fölsch, U.R.; et al. Bacterial and fungal microbiota in relation to probiotic therapy (VSL#3) in pouchitis. Gut 2006, 55, 833–841. [Google Scholar]
- Williams, S.M.; Harned, R.K. Hepatobiliary complications of inflammatory bowel disease. Radiol. Clin. North Am. 1987, 25, 175–188. [Google Scholar]
- Masubuchi, Y.; Horie, T. Endotoxin-mediated disturbance of hepatic cytochrome P450 function and development of endotoxin tolerance in the rat model of dextran sulfate sodium-induced experimental colitis. Drug Metab. Dispos. 2004, 32, 437–441. [Google Scholar]
- Rutter, M.; Saunders, B.; Wilkins, K.; Rumbles, S.; Schofield, G.; Kamm, M.; Williams, C.; Price, A.; Talbot, I.; Forbes, A. Severity of inflammation is a risk factor for colorectal neoplasia in ulcerative colitis. Gastroenterology 2004, 126, 451–459. [Google Scholar]
- Gianotti, L.; Morelli, L.; Galbiati, F.; Rocchetti, S.; Coppola, S.; Beneduce, A.; Gilardini, C.; Zonenschain, D.; Nespoli, A.; Braga, M. A randomized double-blind trial on perioperative administration of probiotics in colorectal cancer patients. World J. Gastroenterol. 2010, 16, 167–175. [Google Scholar]
- Podolsky, D.K. Inflammatory bowel disease. N. Engl. J. Med. 2002, 347, 417–429. [Google Scholar]
- Kornbluth, A.; Sacher, D.B. Ulcerative colitis practice guidelines in adults. Am. J. Gastroenterol. 2004, 99, 1371–1385. [Google Scholar]
- Friedman, S.; Blumberg, R. Inflammatory Bowel Disease. In Harrison’s Principles of Internal Medicine, 17th; Fauci, A.S., Braunwald, E., Kasper, D.L., Hauser, S.L., Longo, D.L., Jameson, J.L., Loscalzo, J., Eds.; McGraw-Hill Companies: New York, NY, USA, 2008; pp. 1777–1789. [Google Scholar]
- Ott, S.J.; Musfeldt, M.; Wenderoth, D.F.; Hampe, J.; Brant, O.; Fölsch, U.R.; Timmis, K.N.; Schreiber, S. Reduction in diversity of the colonic mucosa associated bacterial microflora in patients with active inflammatory bowel disease. Gut 2004, 53, 685–693. [Google Scholar]
- Swidsinski, A.; Ladhoff, A.; Pernthaler, A.; Swidsinski, S.; Loening-Baucke, V.; Ortner, M.; Weber, J.; Hoffmann, U.; Schreiber, S.; Dietel, M.; et al. Mucosal flora in inflammatory bowel disease. Gastroenterology 2002, 122, 44–54. [Google Scholar]
- Butterworth, A.D.; Thomas, A.G.; Akobeng, A.K. Probiotics for induction of remission in Crohn’s disease. Cochrane Database Syst. Rev. 2008, CD006634. [Google Scholar] [PubMed]
- Weiss, J.F. Pharmacologic approaches to protection against radiation-induced lethality and other damage. Environ. Health Perspect. 1997, 105, 1473–1478. [Google Scholar]
- Weiss, J.F.; Landauer, M.R. Radioprotection by antioxidants. Ann. N. Y. Acad. Sci. 2000, 899, 44–60. [Google Scholar]
- Berthrong, M. Pathologic changes secondary to radiation. World J. Surg. 1986, 10, 155–170. [Google Scholar]
- Bismar, M.M.; Sinicrope, F.A. Radiation enteritis. Curr. Gastroenterol. Rep. 2002, 4, 361–365. [Google Scholar]
- Molla, M.; Gironella, M.; Salas, A.; Miquel, R.; Pérez-del-Pulgar, S.; Conill, C.; Engel, P.; Biete, A.; Piqué, J.M.; Panés, J. Role of P-selectin in radiation-induced intestinal inflammatory damage. Int. J. Cancer 2001, 96, 99–109. [Google Scholar]
- Novak, J.M.; Collins, J.T.; Donowitz, M.; Farman, J.; Sheahan, D.G.; Spiro, H.M. Effects of radiation on the human gastrointestinal tract. J. Clin. Gastroenterol. 1979, 1, 9–39. [Google Scholar]
- Summers, R.W.; Flatt, A.J.; Prihoda, M.J.; Mitros, F.A. Effect of irradiation on morphology and motility of canine small intestine. Dig. Dis. Sci. 1987, 32, 1402–1410. [Google Scholar]
- Fernandez-Banares, F.; Villa, S.; Esteve, M.; Roca, M.; Cabré, E.; Abad-Lacruz, A.; Martín-Comín, J.; Gassull, M.A. Acute effects of abdominopelvic irradiation on the orocecal transit time: Its relation to clinical symptoms, and bile salt and lactose malabsorption. Am. J. Gastroenterol. 1991, 86, 1771–1777. [Google Scholar]
- Yeoh, E.K.; Horowit, M.; Russo, A.; Muecke, T.; Robb, T.; Chatterton, B.E. Gastrointestinal function in chronic radiation enteritis—effects of loperamide-N-oxide. Gut 1993, 34, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Indaram, A.V.; Visvalingam, V.; Locke, M.; Bank, S. Mucosal cytokine production in radiation induced proctosigmoiditis compared with inflammatory bowel disease. Am. J. Gastroenterol. 2000, 95, 1221–1225. [Google Scholar]
- Mikelsaar, M.; Türi, M.; Lencner, H.; Kolts, K.; Kirch, R.; Lencner, A. Interrelations between mucosal and luminal microflora of gastrointestine. Nahrung 1987, 31, 449-456, 637-638. [Google Scholar]
- Deitch, E.A. The role of bacterial failure and bacterial translocation in the development of systemic infection and multiple organ failure. Arch. Surg. 1990, 125, 403–404. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hakansson, A.; Molin, G. Gut Microbiota and Inflammation. Nutrients 2011, 3, 637-682. https://doi.org/10.3390/nu3060637
Hakansson A, Molin G. Gut Microbiota and Inflammation. Nutrients. 2011; 3(6):637-682. https://doi.org/10.3390/nu3060637
Chicago/Turabian StyleHakansson, Asa, and Goran Molin. 2011. "Gut Microbiota and Inflammation" Nutrients 3, no. 6: 637-682. https://doi.org/10.3390/nu3060637
APA StyleHakansson, A., & Molin, G. (2011). Gut Microbiota and Inflammation. Nutrients, 3(6), 637-682. https://doi.org/10.3390/nu3060637




