Comparative Bioavailability of Vitamin C After Short-Term Consumption of Raw Fruits and Vegetables and Their Juices: A Randomized Crossover Study
Highlights
- Vitamin C bioavailability from whole fruits, fresh juices, and supplements was evaluated under equivalent intake using a randomized crossover design.
- Plasma vitamin C responses varied depending on the form of intake, with differences observed in AUC under certain fresh juice conditions.
- Metabolomic analysis revealed significant alterations in amino acid-related pathways, including glycine and taurine metabolism.
- These findings suggest that the food matrix influences vitamin C absorption dynamics.
- The results support the use of fresh fruit and vegetable juices as an effective dietary source of vitamin C.
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Equivalent Vitamin C Sources: Raw Fruits and Vegetables and Their Juice
2.3. Vitamin C Concentration of Raw Fruits and Vegetables and Their Juice
2.4. Subjects and Study Design
2.5. Blood and Urine Sample Collection
2.6. Quantitative Analysis of Vitamin C in Plasma and Urine
2.7. 1H NMR Analysis
2.8. Plasma Antioxidant Activity
2.8.1. Oxygen Radical Absorbance Capacity (ORAC) Assay
2.8.2. Total Radical-Trapping Antioxidant Parameter (TRAP) Assay
2.9. Statistical Analysis
3. Results
3.1. Vitamin C Concentration in Raw Fruits and Vegetables and Their Juices
3.2. Characteristics of Subjects
3.3. Plasma and Urine Vitamin C Concentration
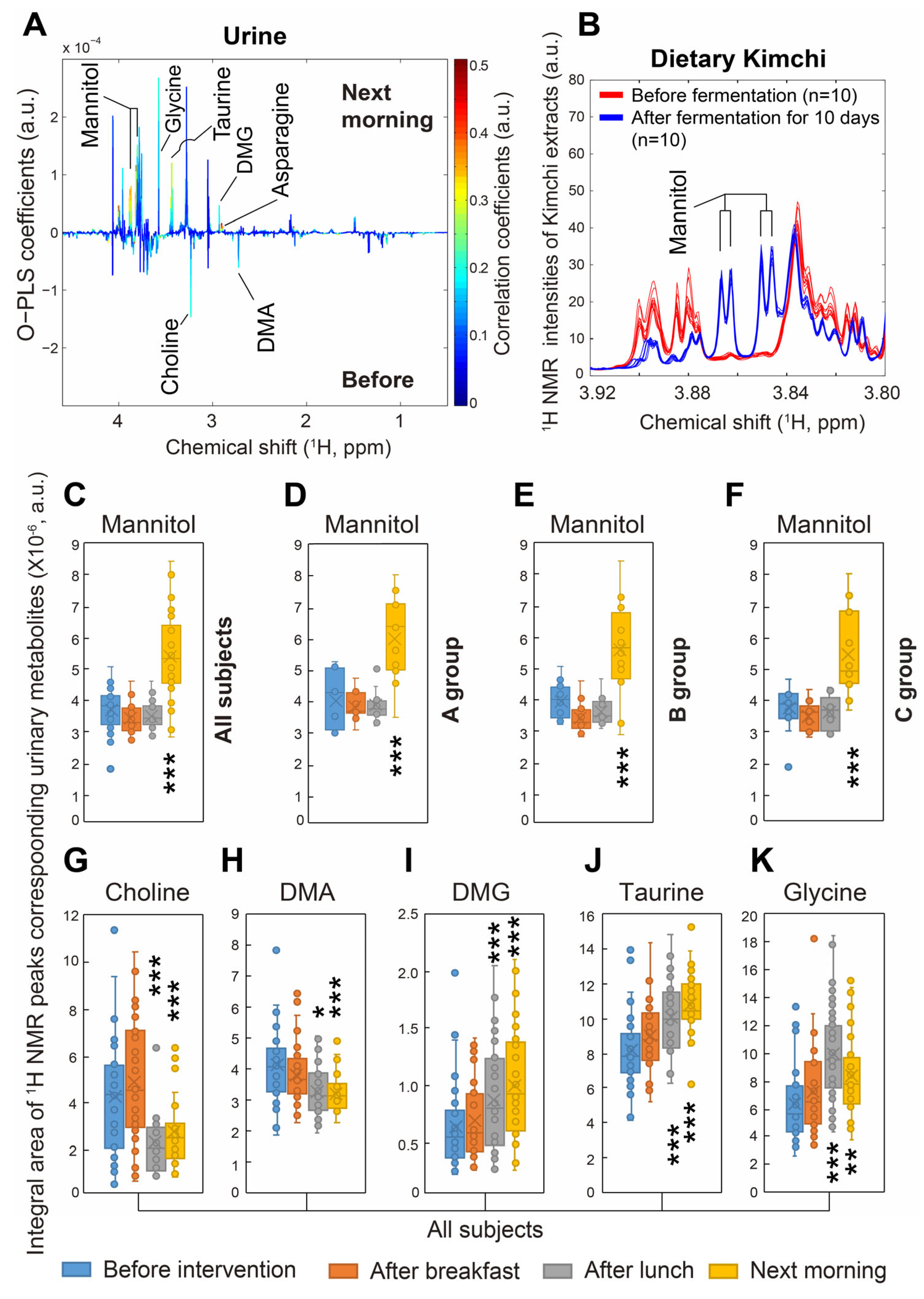
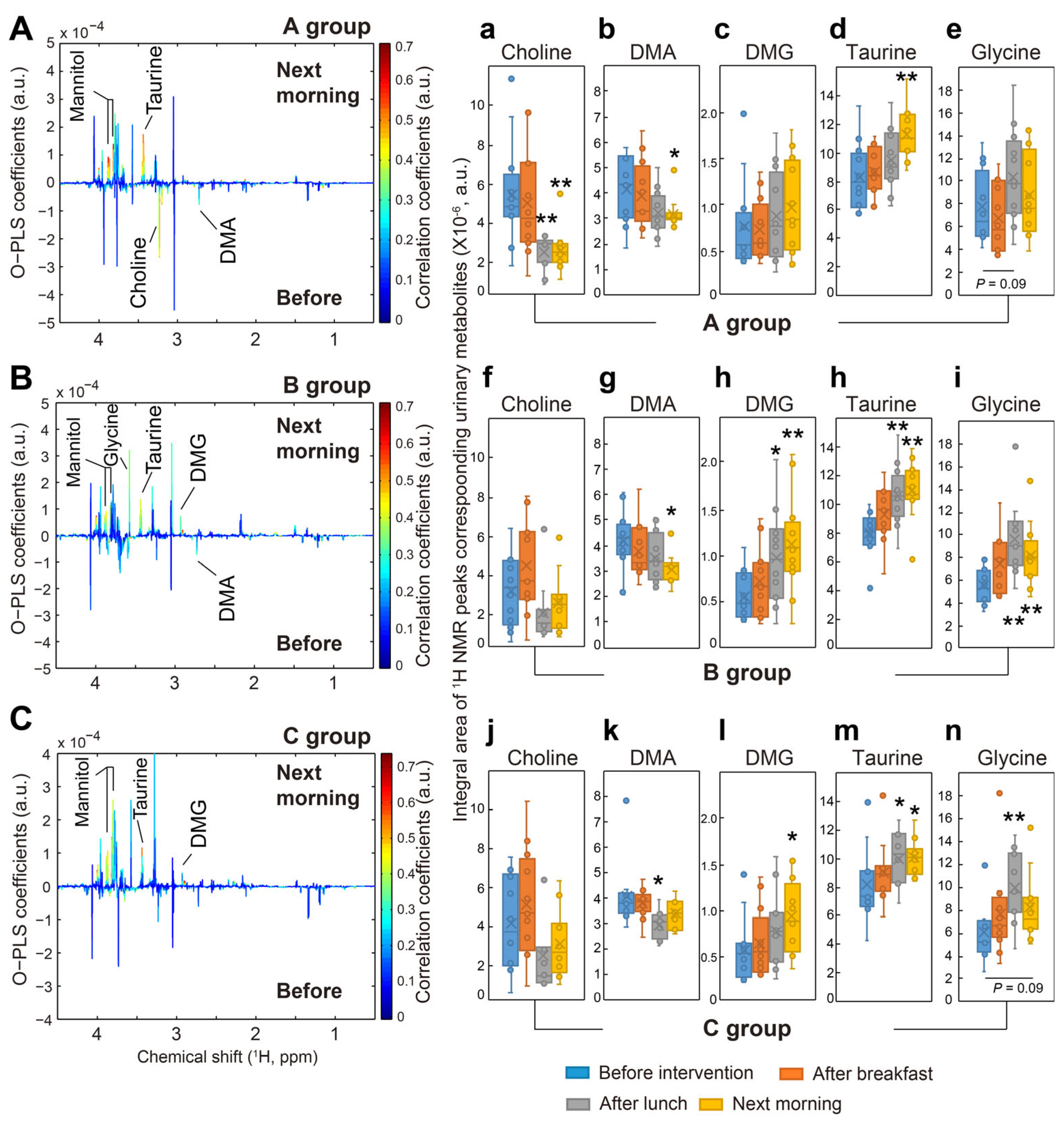
3.4. 1H NMR Analysis of Participants’ Urinary Metabolites
3.5. Plasma Antioxidant Activity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nishikimi, M.; Koshizaka, T.; Ozawa, T.; Yagi, K. Occurrence in Humans and Guinea Pigs of the Gene Related to Their Missing Enzyme L-Gulono-γ-Lactone Oxidase. Arch. Biochem. Biophys. 1988, 267, 842–846. [Google Scholar] [CrossRef]
- Chambial, S.; Dwivedi, S.; Shukla, K.K.; John, P.J.; Sharma, P. Vitamin C in Disease Prevention and Cure: An Overview. Indian J. Clin. Biochem. 2013, 28, 314–328. [Google Scholar] [CrossRef]
- Frei, B.; England, L.; Ames, B.N. Ascorbate Is an Outstanding Antioxidant in Human Blood Plasma. Proc. Natl. Acad. Sci. USA 1989, 86, 6377–6381. [Google Scholar] [CrossRef]
- Buettner, G.R. The Pecking Order of Free Radicals and Antioxidants: Lipid Peroxidation, α-Tocopherol, and Ascorbate. Arch. Biochem. Biophys. 1993, 300, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Jacob, R.A.; Sotoudeh, G. Vitamin C Function and Status in Chronic Disease. Nutr. Clin. Care 2002, 5, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Rozman, B.; Gašperlin, M. Stability of Vitamins C and E in Topical Microemulsions for Combined Antioxidant Therapy. Drug Deliv. 2007, 14, 235–245. [Google Scholar] [CrossRef]
- Li, Y.; Schellhorn, H.E. New Developments and Novel Therapeutic Perspectives for Vitamin C. J. Nutr. 2007, 137, 2171–2184. [Google Scholar] [CrossRef]
- Carr, A.C.; Frei, B. Toward a New Recommended Dietary Allowance for Vitamin C Based on Antioxidant and Health Effects in Humans. Am. J. Clin. Nutr. 1999, 69, 1086–1107. [Google Scholar] [CrossRef]
- Savini, I.; Rossi, A.; Pierro, C.; Avigliano, L.; Catani, M. SVCT1 and SVCT2: Key Proteins for Vitamin C Uptake. Amino Acids 2008, 34, 347–355. [Google Scholar] [CrossRef]
- Vera, J.C.; Rivas, C.I.; Velásquez, F.V.; Zhang, R.H.; Concha, I.I.; Golde, D.W. Resolution of the Facilitated Transport of Dehydroascorbic Acid from Its Intracellular Accumulation as Ascorbic Acid. J. Biol. Chem. 1995, 270, 23706–23712. [Google Scholar] [CrossRef]
- Graumlich, J.F.; Ludden, T.M.; Conry-Cantilena, C.; Cantilena, L.R.; Wang, Y.; Levine, M. Pharmacokinetic Model of Ascorbic Acid in Healthy Male Volunteers during Depletion and Repletion. Pharm. Res. 1997, 14, 1133–1139. [Google Scholar] [CrossRef]
- Padayatty, S.J.; Sun, H.; Wang, Y.; Riordan, H.D.; Hewitt, S.M.; Katz, A.; Wesley, R.A.; Levine, M. Vitamin C Pharmacokinetics: Implications for Oral and Intravenous Use. Ann. Intern. Med. 2004, 140, 533–537. [Google Scholar] [CrossRef] [PubMed]
- Levine, M.; Conry-Cantilena, C.; Wang, Y.; Welch, R.W.; Washko, P.W.; Dhariwal, K.R.; Park, J.B.; Lazarev, A.; Graumlich, J.F.; King, J.; et al. Vitamin C Pharmacokinetics in Healthy Volunteers: Evidence for a Recommended Dietary Allowance. Proc. Natl. Acad. Sci. USA 1996, 93, 3704–3709. [Google Scholar] [CrossRef] [PubMed]
- Shimpo, M.; Akamatsu, R.; Kojima, Y. Impact of the COVID-19 Pandemic on Food and Drink Consumption and Related Factors: A Scoping Review. Nutr. Health 2022, 28, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.; Mendiola, J.A.; Oliveira, M.B.; Ibáñez, E.; Herrero, M. Sequential Determination of Fat- and Water-Soluble Vitamins in Green Leafy Vegetables during Storage. J. Chromatogr. A 2012, 1261, 179–188. [Google Scholar] [CrossRef]
- Kurihara, H.; Fukama, H.; Asami, S.; Totoda, Y.; Nakai, M.; Shibata, H.; Yao, X.S. Effects of Oolong Tea on Plasma Antioxidative Capacity in Mice Loaded with Restraint Stress Assessed Using the Oxygen Radical Absorbance Capacity (ORAC) Assay. Biol. Pharm. Bull. 2004, 27, 1093–1098. [Google Scholar] [CrossRef]
- Ou, B.; Hampsch-Woodill, M.; Prior, R.L. Development and Validation of an Improved Oxygen Radical Absorbance Capacity Assay Using Fluorescein as the Fluorescent Probe. J. Agric. Food Chem. 2001, 49, 4619–4626. [Google Scholar] [CrossRef]
- Rice-Evans, C.; Miller, N.J. Total Antioxidant Status in Plasma and Body Fluids. Methods Enzymol. 1994, 234, 279–293. [Google Scholar] [CrossRef]
- Cloarec, O.; Dumas, M.E.; Craig, A.; Barton, R.H.; Trygg, J.; Hudson, J.; Blancher, C.; Gauguier, D.; Lindon, J.C.; Holmes, E.; et al. Statistical Total Correlation Spectroscopy: An Exploratory Approach for Latent Biomarker Identification from Metabolic 1H NMR Data Sets. Anal. Chem. 2005, 77, 1282–1290. [Google Scholar] [CrossRef]
- Walsh, M.C.; Brennan, L.; Pujos-Guillot, E.; Sébédio, J.-L.; Scalbert, A.; Fagan, A.; Higgins, D.G.; Gibney, M.J. Influence of Acute Phytochemical Intake on Human Urinary Metabolomic Profiles. Am. J. Clin. Nutr. 2007, 86, 1687–1693. [Google Scholar] [CrossRef]
- Vinson, J.A.; Bose, P. Comparative Bioavailability to Humans of Ascorbic Acid Alone or in a Citrus Extract. Am. J. Clin. Nutr. 1988, 48, 501–504. [Google Scholar] [CrossRef]
- Carr, A.C.; Bozonet, S.M.; Pullar, J.M.; Simcock, J.W.; Vissers, M.C.M. A Randomized Steady-State Bioavailability Study of Synthetic versus Natural (Kiwifruit-Derived) Vitamin C. Nutrients 2013, 5, 4451–4461. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Vissers, M.C.M. Synthetic or Food-Derived Vitamin C—Are They Equally Bioavailable? Nutrients 2013, 5, 4284–4304. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.Y.; Lee, S.H.; Kim, J.M.; Park, M.S.; Bae, J.W.; Hahn, Y.; Madsen, E.L.; Jeon, C.O. Metagenomic Analysis of Kimchi, a Traditional Korean Fermented Food. Appl. Environ. Microbiol. 2011, 77, 2264–2274. [Google Scholar] [CrossRef]
- Louis, P.; Flint, H.J. Formation of Propionate and Butyrate by the Human Colonic Microbiota. Environ. Microbiol. 2017, 19, 29–41. [Google Scholar] [CrossRef]
- Qian, W.; Li, M.; Yu, L.; Tian, F.; Zhao, J.; Zhai, Q. Effects of Taurine on Gut Microbiota Homeostasis: An Evaluation Based on Two Models of Gut Dysbiosis. Biomedicines 2023, 11, 1048. [Google Scholar] [CrossRef]
- Otten, A.T.; Bourgonje, A.R.; Harmsen, H.J.M.; Alizadeh, B.Z.; Dijkstra, G.; Faber, K.N. Vitamin C Supplementation in Healthy Individuals Leads to Shifts of Bacterial Populations in the Gut—A Pilot Study. Antioxidants 2021, 10, 1278. [Google Scholar] [CrossRef]
- Hazan, S.; Dave, S.; Papoutsis, A.J.; Deshpande, N.; Howell, M.C.; Martin, L.M. Vitamin C Improves Gut Bifidobacteria in Humans. Future Microbiol. 2022; online ahead of print. [Google Scholar] [CrossRef]
- Koeth, R.A.; Wang, Z.; Levison, B.S.; Buffa, J.A.; Org, E.; Sheehy, B.T.; Britt, E.B.; Fu, X.; Wu, Y.; Li, L.; et al. Intestinal Microbiota Metabolism of L-Carnitine, a Nutrient in Red Meat, Promotes Atherosclerosis. Nat. Med. 2013, 19, 576–585. [Google Scholar] [CrossRef]
- Wang, Z.; Klipfell, E.; Bennett, B.J.; Koeth, R.; Levison, B.S.; Dugar, B.; Feldstein, A.E.; Britt, E.B.; Fu, X.; Chung, Y.M.; et al. Gut Flora Metabolism of Phosphatidylcholine Promotes Cardiovascular Disease. Nature 2011, 472, 57–63. [Google Scholar] [CrossRef]
- Moolenaar, S.H.; Poggi-Bach, J.; Engelke, U.F.; Corstiaensen, J.M.; Heerschap, A.; de Jong, J.G.; Binzak, B.A.; Vockley, J.; Wevers, R.A. Defect in Dimethylglycine Dehydrogenase, a New Inborn Error of Metabolism: NMR Spectroscopy Study. Clin. Chem. 1999, 45, 459–464. [Google Scholar] [CrossRef]
- Guzior, D.V.; Quinn, R.A. Review: Microbial Transformations of Human Bile Acids. Microbiome 2021, 9, 140. [Google Scholar] [CrossRef]
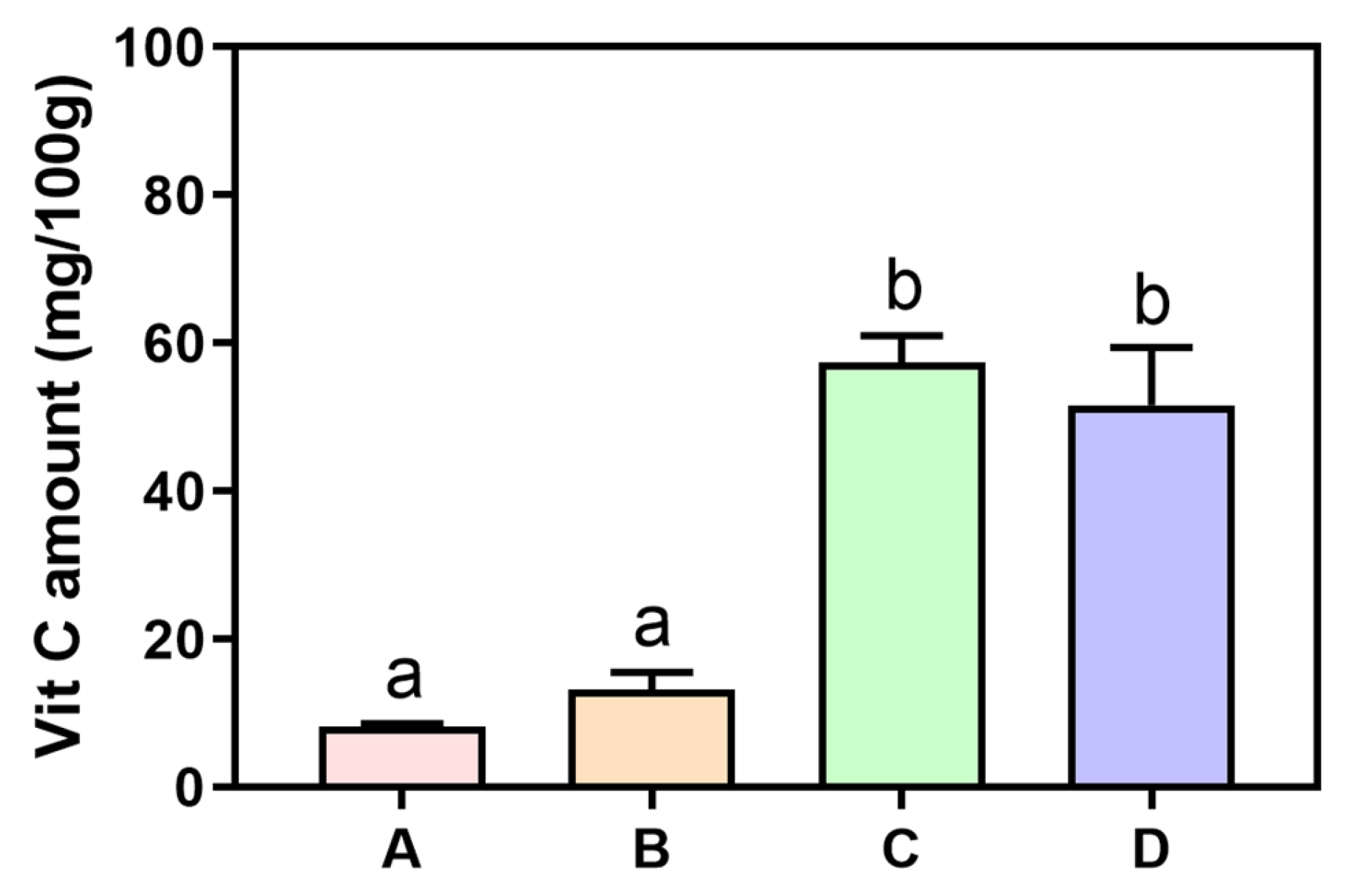

| Amount | Vitamin C Content | |
|---|---|---|
| Vitamin C supplements | 101.7 mg | 101.7 mg |
| Raw fruits and vegetables | ||
| Tangerine | 74.7 g | 101.7 mg |
| Cherry tomato | 37.4 g | |
| Orange bell pepper | 74.7 g | |
| Fruit and vegetable juices | 202.5 g (200 mL) | 101.7 mg |
| Menu | |
|---|---|
| Meal types | |
| Lunch | Cooked rice Fried egg Grilled chicken drumsticks (boneless thigh meat) Seasoned dried shredded fish (Ilmi muchim) Seasoned dried filefish strips Fried pollock skin Braised quail eggs |
| Afternoon snacks | Steamed white rice cake Energy bar |
| Dinner | Cooked rice Aged kimchi stew with thinly sliced beef brisket Korean-style rolled omelet grilled chicken wings Braised peanuts Seasoned dried shredded fish (Ilmi muchim) Seasoned dried filefish strips Fried pollock skin Braised quail eggs |
| Evening snacks | Grilled chicken Carbonated water |
| Nutrient contents | |
| Energy | |
| Calorie (kcal) | 3363.2 |
| Carbohydrate (g) | 329.0 |
| Protein (g) | 318.6 |
| Fats (g) | 120.3 |
| Vitamin | |
| Vitamin C (mg) | 0.0 |
| A Group (n = 12) (1) | B Group (n = 12) | C Group (n = 12) | |
|---|---|---|---|
| Sex | |||
| Male (n) | 6 | 6 | 6 |
| Female (n) | 6 | 6 | 6 |
| Age (years) | 22.0 ± 0.5 (3), ns(4) | 22.0 ± 0.5 | 21.8 ± 0.7 |
| Hight (kg) | 167.5 ± 1.9 ns | 167.3 ± 1.8 | 167.91 ± 1.5 |
| Weight (kg) | 78.5 ± 6.7 ns | 78.9 ± 6.8 | 72.45 ± 5.4 |
| BMI (kg/m2) (2) | 27.8 ± 2.2 ns | 28.0 ± 2.2 | 25.55 ± 1.7 |
| SBP (mmHg) | 130.3 ± 6.1 ns | 127.8 ± 3.9 | 123.83 ± 3.7 |
| DBP (mmHg) | 80.6 ± 4.9 ns | 75.3 ± 3.1 | 73.67 ± 3.3 |
| WC (cm) | 90.8 ± 5.0 ns | 91.5 ± 5.0 | 83.72 ± 4.1 |
| HC (cm) | 103.6 ± 3.6 ns | 103.3 ± 3.6 | 100.75 ± 3.4 |
| Time | A Group (n = 12) (1) | B Group (n = 12) | C Group (n = 12) |
|---|---|---|---|
| −2 h | 1.41 ± 0.16 (2), a(3), NS(4) | 1.08 ± 0.06 a | 0.96 ± 0.05 a |
| +2 h | 0.92 ± 0.05 a, NS | 1.01 ± 0.06 a | 0.94 ± 0.06 a |
| +8 h | 0.91 ± 0.05 b, NS | 0.98 ± 0.08 b | 1.04 ± 0.05 b |
| +24 h | 0.98 ± 0.07 c, NS | 1.00 ± 0.05 c | 1.07 ± 0.06 c |
| A Group (n = 12) (1) | B Group (n = 12) | C Group (n = 12) | |
|---|---|---|---|
| ORAC (μM TE) (2) | |||
| 0 h | 342.0 ± 33.2 (3), a(4), ns(5) | 308.8 ± 23.2 a | 290.9 ± 16.1 a |
| 0.5 h | 465.2 ± 29.1 bc, B(6) | 339.7 ± 34.0 ab, A | 343.6 ± 31.7 ab, A |
| 1 h | 407.8 ± 35.7 abc, ns | 452.9 ± 45.9 c | 416.2 ± 33.2 bc |
| 2 h | 399.9 ± 23.1 abc, ns | 481.9 ± 32.9 c | 412.8 ± 48.6 bc |
| 4 h | 380.4 ± 33.0 ab, ns | 429.8 ± 34.7 bc | 395.0 ± 33.5 bc |
| 6 h | 453.2 ± 28.8 bc, ns | 489.7 ± 26.0 c | 446.6 ± 29.6 bc |
| 8 h | 469.5 ± 20.4 bc, ns | 468.7 ± 53.0 c | 447.9 ± 36.9 bc |
| 12 h | 479.1 ± 22.6 c, ns | 504.4 ± 22.6 c | 423.9 ± 39.0 bc |
| 24 h | 460.0 ± 34.3 bc, ns | 435.7 ± 23.5 bc | 459.4 ± 37.4 c |
| TRAP (mM) | |||
| 0 h | 1.04 ± 0.05 NS (7), ns | 1.08 ± 0.06 ns | 0.96 ± 0.05 ns |
| 0.5 h | 0.95 ± 0.06 NS | 1.02 ± 0.06 | 0.98 ± 0.07 |
| 1 h | 0.97 ± 0.06 NS | 1.05 ± 0.04 | 1.03 ± 0.07 |
| 2 h | 1.00 ± 0.06 NS | 0.99 ± 0.06 | 1.03 ± 0.07 |
| 4 h | 0.89 ± 0.06 NS | 0.98 ± 0.06 | 0.97 ± 0.07 |
| 6 h | 0.98 ± 0.06 NS | 1.08 ± 0.05 | 1.04 ± 0.03 |
| 8 h | 0.92 ± 0.05 NS | 1.01 ± 0.06 | 0.94 ± 0.06 |
| 12 h | 0.91 ± 0.05 NS | 0.98 ± 0.08 | 1.04 ± 0.05 |
| 24 h | 0.98 ± 0.07 NS | 1.00 ± 0.05 | 1.07 ± 0.06 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, M.; Baek, J.; Yun, J.-M.; Hong, Y.-S.; Park, E. Comparative Bioavailability of Vitamin C After Short-Term Consumption of Raw Fruits and Vegetables and Their Juices: A Randomized Crossover Study. Nutrients 2025, 17, 3331. https://doi.org/10.3390/nu17213331
Choi M, Baek J, Yun J-M, Hong Y-S, Park E. Comparative Bioavailability of Vitamin C After Short-Term Consumption of Raw Fruits and Vegetables and Their Juices: A Randomized Crossover Study. Nutrients. 2025; 17(21):3331. https://doi.org/10.3390/nu17213331
Chicago/Turabian StyleChoi, Mijoo, Juha Baek, Jung-Mi Yun, Young-Shick Hong, and Eunju Park. 2025. "Comparative Bioavailability of Vitamin C After Short-Term Consumption of Raw Fruits and Vegetables and Their Juices: A Randomized Crossover Study" Nutrients 17, no. 21: 3331. https://doi.org/10.3390/nu17213331
APA StyleChoi, M., Baek, J., Yun, J.-M., Hong, Y.-S., & Park, E. (2025). Comparative Bioavailability of Vitamin C After Short-Term Consumption of Raw Fruits and Vegetables and Their Juices: A Randomized Crossover Study. Nutrients, 17(21), 3331. https://doi.org/10.3390/nu17213331






