Asthma and Multi-Food Allergy Are Risk Factors for Oral Food Challenge Failure—A Single-Center Experience
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Data Collection
2.2. Laboratory Tests
2.3. Oral Food Challenges (OFCs)
2.4. Statistical Method
3. Results
3.1. General Information
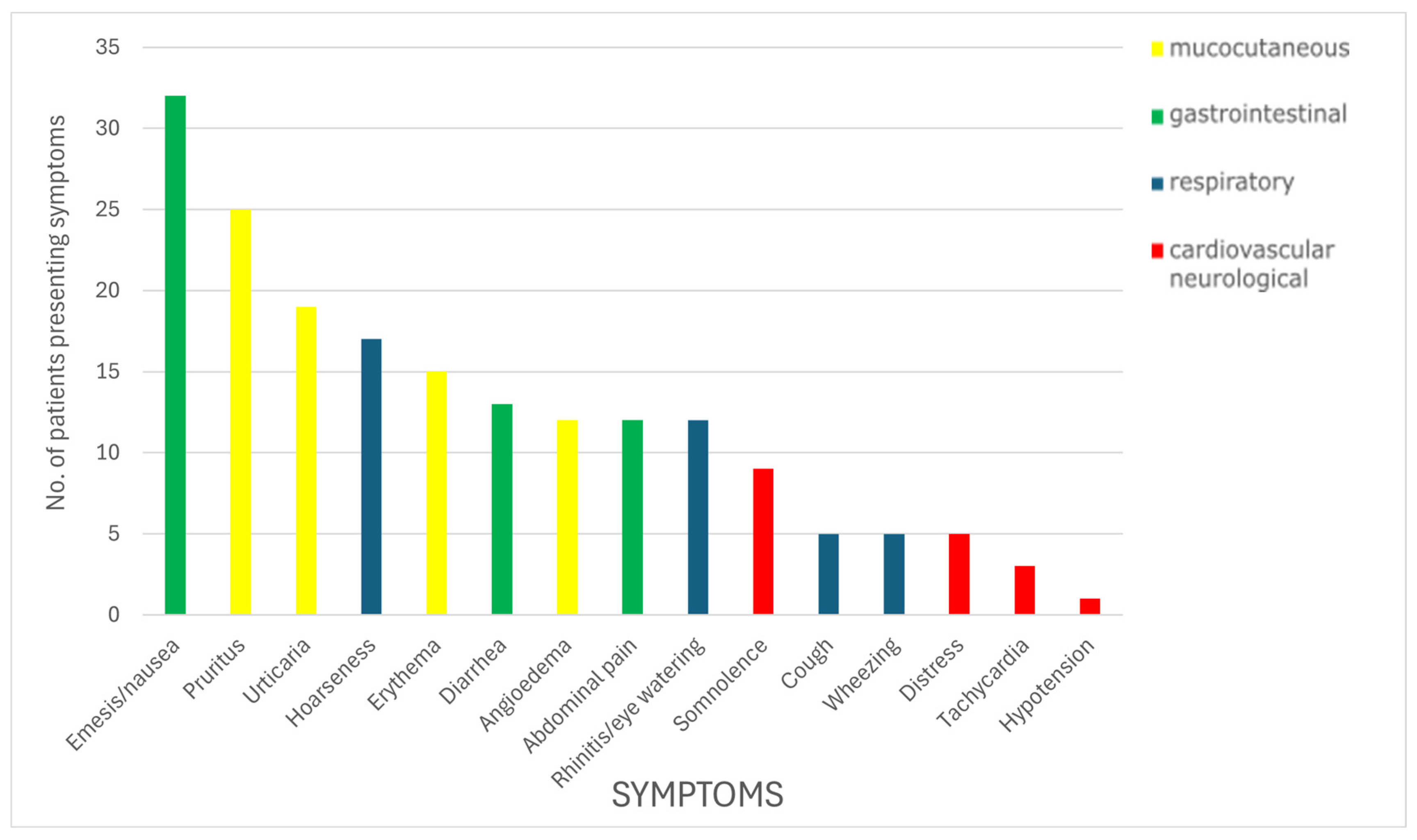
3.2. Symptoms Reported During Failed OFCs and Treatment
3.3. Factors Influencing OFC Outcome
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| FA | food allergy |
| OFC | oral food challenge |
| CMP | cow’s milk protein |
| HEWP | hen’s egg white protein |
| ROC | receiver operating characteristic |
| DBPCFC | double-blind, placebo-controlled food challenge |
| sIgE | specific IgE |
| AUC | area under curve |
| CRD | component-resolved diagnostics |
References
- Muraro, A.; de Silva, D.; Halken, S.; Worm, M.; Khaleva, E.; Arasi, S.; Dunn-Galvin, A.; Nwaru, B.I.; De Jong, N.W.; Rodríguez Del Río, P.; et al. Managing Food Allergy: GA2LEN Guideline 2022. World Allergy Organ. J. 2022, 15, 100687. [Google Scholar] [CrossRef]
- Barni, S.; Liccioli, G.; Sarti, L.; Giovannini, M.; Novembre, E.; Mori, F. Immunoglobulin E (IgE)-Mediated Food Allergy in Children: Epidemiology, Pathogenesis, Diagnosis, Prevention, and Management. Medicina 2020, 56, 111. [Google Scholar] [CrossRef]
- Sampath, V.; Abrams, E.M.; Adlou, B.; Akdis, C.; Akdis, M.; Brough, H.A.; Chan, S.; Chatchatee, P.; Chinthrajah, R.S.; Cocco, R.R.; et al. Food Allergy across the Globe. J. Allergy Clin. Immunol. 2021, 148, 1347–1364. [Google Scholar] [CrossRef]
- Sicherer, S.H.; Sampson, H.A. Food Allergy: A Review and Update on Epidemiology, Pathogenesis, Diagnosis, Prevention, and Management. J. Allergy Clin. Immunol. 2018, 141, 41–58. [Google Scholar] [CrossRef] [PubMed]
- Botha, M.; Basera, W.; Facey-Thomas, H.E.; Gaunt, B.; Gray, C.L.; Ramjith, J.; Watkins, A.; Levin, M.E. Rural and Urban Food Allergy Prevalence from the South African Food Allergy (SAFFA) Study. J. Allergy Clin. Immunol. 2019, 143, 662–668.e2. [Google Scholar] [CrossRef]
- Sigurdardottir, S.T.; Jonasson, K.; Clausen, M.; Lilja Bjornsdottir, K.; Sigurdardottir, S.E.; Roberts, G.; Grimshaw, K.; Papadopoulos, N.G.; Xepapadaki, P.; Fiandor, A.; et al. Prevalence and Early-Life Risk Factors of School-Age Allergic Multimorbidity: The EuroPrevall-IFAAM Birth Cohort. Allergy Eur. J. Allergy Clin. Immunol. 2021, 76, 2855–2865. [Google Scholar] [CrossRef] [PubMed]
- Grabenhenrich, L.; Trendelenburg, V.; Bellach, J.; Yürek, S.; Reich, A.; Fiandor, A.; Rivero, D.; Sigurdardottir, S.; Clausen, M.; Papadopoulos, N.G.; et al. Frequency of Food Allergy in School-Aged Children in Eight European Countries—The EuroPrevall-IFAAM Birth Cohort. Allergy Eur. J. Allergy Clin. Immunol. 2020, 75, 2294–2308. [Google Scholar] [CrossRef] [PubMed]
- Spolidoro, G.C.I.; Amera, Y.T.; Ali, M.M.; Nyassi, S.; Lisik, D.; Ioannidou, A.; Rovner, G.; Khaleva, E.; Venter, C.; van Ree, R.; et al. Frequency of Food Allergy in Europe: An Updated Systematic Review and Meta-Analysis. Allergy Eur. J. Allergy Clin. Immunol. 2023, 78, 351–368. [Google Scholar] [CrossRef]
- Knyziak-Mędrzycka, I.; Majsiak, E.; Gromek, W.; Kozłowska, D.; Swadźba, J.; Beata, B.J.; Kurzawa, R.; Cukrowska, B. The Sensitization Profile for Selected Food Allergens in Polish Children Assessed with the Use of a Precision Allergy Molecular Diagnostic Technique. Int. J. Mol. Sci. 2024, 25, 825. [Google Scholar] [CrossRef]
- Lyons, S.A.; Clausen, M.; Knulst, A.C.; Ballmer-Weber, B.K.; Fernandez-Rivas, M.; Barreales, L.; Bieli, C.; Dubakiene, R.; Fernandez-Perez, C.; Jedrzejczak-Czechowicz, M.; et al. Prevalence of Food Sensitization and Food Allergy in Children Across Europe. J. Allergy Clin. Immunol. Pract. 2020, 8, 2736–2746.e9. [Google Scholar] [CrossRef]
- Krzych-Fałta, E.; Białek, S.; Sybilski, A.J.; Tylewicz, A.; Samoliński, B.; Wojas, O. Differential Diagnostics of Food Allergy as Based on Provocation Tests and Laboratory Diagnostic Assays. Postep. Dermatol. Alergol. 2023, 40, 709–715. [Google Scholar] [CrossRef]
- Riggioni, C.; Ricci, C.; Moya, B.; Wong, D.; van Goor, E.; Bartha, I.; Buyuktiryaki, B.; Giovannini, M.; Jayasinghe, S.; Jaumdally, H.; et al. Systematic Review and Meta-Analyses on the Accuracy of Diagnostic Tests for IgE-Mediated Food Allergy. Allergy Eur. J. Allergy Clin. Immunol. 2024, 79, 324–352. [Google Scholar] [CrossRef]
- Calvani, M.; Bianchi, A.; Reginelli, C.; Peresso, M.; Testa, A. Oral Food Challenge. Medicina 2019, 55, 651. [Google Scholar] [CrossRef] [PubMed]
- Hsu, E.; Soller, L.; Abrams, E.M.; Protudjer, J.L.P.; Mill, C.; Chan, E.S. Oral Food Challenge Implementation: The First Mixed-Methods Study Exploring Barriers and Solutions. J. Allergy Clin. Immunol. Pract. 2020, 8, 149–156.e1. [Google Scholar] [CrossRef]
- Greiwe, J. Oral Food Challenges in Infants and Toddlers. Immunol. Allergy Clin. N. Am. 2019, 39, 481–493. [Google Scholar] [CrossRef] [PubMed]
- Santos, A.F.; Riggioni, C.; Agache, I.; Akdis, C.A.; Akdis, M.; Alvarez-Perea, A.; Alvaro-Lozano, M.; Ballmer-Weber, B.; Barni, S.; Beyer, K.; et al. EAACI Guidelines on the Diagnosis of IgE-Mediated Food Allergy. Allergy Eur. J. Allergy Clin. Immunol. 2023, 78, 3057–3076. [Google Scholar] [CrossRef]
- Eiwegger, T.; Hung, L.; San Diego, K.E.; O’Mahony, L.; Upton, J. Recent Developments and Highlights in Food Allergy. Allergy Eur. J. Allergy Clin. Immunol. 2019, 74, 2355–2367. [Google Scholar] [CrossRef]
- Yanagida, N.; Sato, S.; Asaumi, T.; Ogura, K.; Ebisawa, M. Risk Factors for Severe Reactions during Double-Blind Placebo-Controlled Food Challenges. Int. Arch. Allergy Immunol. 2017, 172, 173–182. [Google Scholar] [CrossRef]
- Abrams, E.M.; Becker, A.B. Oral Food Challenge Outcomes in a Pediatric Tertiary Care Center. Allergy Asthma Clin. Immunol. 2017, 13, 43. [Google Scholar] [CrossRef] [PubMed]
- Sindher, S.; Long, A.J.; Purington, N.; Chollet, M.; Slatkin, S.; Andorf, S.; Tupa, D.; Kumar, D.; Woch, M.A.; O’Laughlin, K.L.; et al. Analysis of a Large Standardized Food Challenge Data Set to Determine Predictors of Positive Outcome across Multiple Allergens. Front. Immunol. 2018, 9, 2689. [Google Scholar] [CrossRef]
- Bird, J.A.; Leonard, S.; Groetch, M.; Assa’ad, A.; Cianferoni, A.; Clark, A.; Crain, M.; Fausnight, T.; Fleischer, D.; Green, T.; et al. Conducting an Oral Food Challenge: An Update to the 2009 Adverse Reactions to Foods Committee Work Group Report. J. Allergy Clin. Immunol. Pract. 2020, 8, 75–90.e17. [Google Scholar] [CrossRef]
- Sampson, H.A.; Gerth Van Wijk, R.; Bindslev-Jensen, C.; Sicherer, S.; Teuber, S.S.; Burks, A.W.; Dubois, A.E.J.; Beyer, K.; Eigenmann, P.A.; Spergel, J.M.; et al. Standardizing Double-Blind, Placebo-Controlled Oral Food Challenges: American Academy of Allergy, Asthma & Immunology-European Academy of Allergy and Clinical Immunology PRACTALL Consensus Report. J. Allergy Clin. Immunol. 2012, 130, 1260–1274. [Google Scholar] [CrossRef]
- Nowak-Wegrzyn, A.; Assa’ad, A.H.; Bahna, S.L.; Bock, S.A.; Sicherer, S.H.; Teuber, S.S. Work Group Report: Oral Food Challenge Testing. J. Allergy Clin. Immunol. 2009, 123, S365–S383. [Google Scholar] [CrossRef]
- Leonard, S.A.; Caubet, J.C.; Kim, J.S.; Groetch, M.; Nowak-Wegrzyn, A. Baked Milk- and Egg-Containing Diet in the Management of Milk and Egg Allergy. J. Allergy Clin. Immunol. Pract. 2015, 3, 13–23. [Google Scholar] [CrossRef]
- Sampson, H.A. Anaphylaxis and Emergency Treatment. Pediatrics 2003, 111, 1601–1608. [Google Scholar] [CrossRef] [PubMed]
- Dölle-Bierke, S.; Höfer, V.; Francuzik, W.; Näher, A.F.; Bilo, M.B.; Cichocka-Jarosz, E.; Lopes de Oliveira, L.C.; Fernandez-Rivas, M.; García, B.E.; Hartmann, K.; et al. Food-Induced Anaphylaxis: Data from the European Anaphylaxis Registry. J. Allergy Clin. Immunol. Pract. 2023, 11, 2069–2079.e7. [Google Scholar] [CrossRef]
- de Almeida Kotchetkoff, E.C.; de Oliveira, L.C.L.; Sarni, R.O.S. Elimination Diet in Food Allergy: Friend or Foe? J. Pediatr. (Rio J.) 2024, 100, S65–S73. [Google Scholar] [CrossRef] [PubMed]
- Şengül Emeksiz, Z.; Ertuğrul, A.; Uygun, S.D.; Özmen, S. Evaluation of Emotional, Behavioral, and Clinical Characteristics of Children Aged 1–5 with a History of Food-Related Anaphylaxis. Pediatr. Neonatol. 2023, 64, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Assa’ad, A.H. Oral Food Challenges. J. Food Allergy 2020, 2, 31–34. [Google Scholar] [CrossRef]
- Murai, H.; Irahara, M.; Sugimoto, M.; Takaoka, Y.; Takahashi, K.; Wada, T.; Yamamoto-Hanada, K.; Okafuji, I.; Yamada, Y.; Futamura, M.; et al. Is Oral Food Challenge Useful to Avoid Complete Elimination in Japanese Patients Diagnosed with or Suspected of Having IgE-Dependent Hen’s Egg Allergy? A Systematic Review. Allergol. Int. 2022, 71, 221–229. [Google Scholar] [CrossRef]
- Maeda, M.; Kuwabara, Y.; Tanaka, Y.; Nishikido, T.; Hiraguchi, Y.; Yamamoto-Hanada, K.; Okafuji, I.; Yamada, Y.; Futamura, M.; Ebisawa, M. Is Oral Food Challenge Test Useful for Avoiding Complete Elimination of Cow’s Milk in Japanese Patients with or Suspected of Having IgE-Dependent Cow’s Milk Allergy? Allergol. Int. 2022, 71, 214–220. [Google Scholar] [CrossRef]
- Correa, N.; Protudjer, J.L.P.; Hsu, E.; Soller, L.; Chan, E.S.; Kim, H.; Jeimy, S. Canadian Parent Perceptions of Oral Food Challenges: A Qualitative Analysis. Pediatr. Allergy Immunol. 2022, 33, e13698. [Google Scholar] [CrossRef]
- Ballini, G.; Gavagni, C.; Guidotti, C.; Ciolini, G.; Liccioli, G.; Giovannini, M.; Sarti, L.; Ciofi, D.; Novembre, E.; Mori, F.; et al. Frequency of Positive Oral Food Challenges and Their Outcomes in the Allergy Unit of a Tertiary-Care Pediatric Hospital. Allergol. Immunopathol. 2021, 49, 120–130. [Google Scholar] [CrossRef]
- Ogata, M.; Kido, J.; Watanabe, S.; Yoshida, T.; Nishi, N.; Shimomura, S.; Hirai, N.; Tanaka, K.; Mizukami, T.; Yanai, M.; et al. The Efficacy and Safety of Stepwise Oral Food Challenge in Children with Cow’s Milk Allergy. Int. Arch. Allergy Immunol. 2024, 186, 232–242. [Google Scholar] [CrossRef] [PubMed]
- Ünsal, H.; Gökçe, Ö.B.G.; Ocak, M.; Akarsu, A.; Şahiner, Ü.M.; Soyer, Ö.; Şekerel, B.E. Oral Food Challenge in IgE Mediated Food Allergy in Eastern Mediterranean Children. Allergol. Immunopathol. 2021, 49, 185–192. [Google Scholar] [CrossRef]
- Jacob, J.G.; Fernando, S.L.; Nickolls, C.; Li, J. Oral Food Challenge Outcomes in Children and Adolescents in a Tertiary Centre: A 5-Year Experience. J. Paediatr. Child. Health 2023, 59, 822–826. [Google Scholar] [CrossRef] [PubMed]
- Emeksiz, Z.S.; Ertugrul, A.; Ozmen, S.; Cavkaytar, O.; Ercan, N.; Bostancl, I.B. Is Oral Food Challenge as Safe Enough as It Seems? J. Trop. Pediatr. 2021, 67, fmab065. [Google Scholar] [CrossRef] [PubMed]
- Leffler, J.; Stumbles, P.A.; Strickland, D.H. Immunological Processes Driving IgE Sensitisation and Disease Development in Males and Females. Int. J. Mol. Sci. 2018, 19, 1554. [Google Scholar] [CrossRef]
- Uekert, S.J.; Akan, G.; Evans, M.D.; Li, Z.; Roberg, K.; Tisler, C.; DaSilva, D.; Anderson, E.; Gangnon, R.; Allen, D.B.; et al. Sex-Related Differences in Immune Development and the Expression of Atopy in Early Childhood. J. Allergy Clin. Immunol. 2006, 118, 1375–1381. [Google Scholar] [CrossRef]
- Su, Y.; Wen, J.; Zhang, H.; Zou, Z.; Cai, Y.; Zhang, C. Clinical Characteristics of Anaphylaxis in Children Aged 0–16 Years in Xi’an, China. Int. Arch. Allergy Immunol. 2023, 184, 220–227. [Google Scholar] [CrossRef]
- Gaspar, Â.; Santos, N.; Faria, E.; Pereira, A.M.; Gomes, E.; Câmara, R.; Rodrigues-Alves, R.; Borrego, L.M.; Carrapatoso, I.; Carneiro-Leão, L.; et al. Anaphylaxis in Children and Adolescents: The Portuguese Anaphylaxis Registry. Pediatr. Allergy Immunol. 2021, 32, 1278–1286. [Google Scholar] [CrossRef]
- Honda, A.; Imai, T.; Okada, Y.; Maeda, M.; Kamiya, T. Severe Anaphylaxis Requiring Continuous Adrenaline Infusion during Oral Food Challenge: A Case Series. Ann. Allergy Asthma Immunol. 2024, 133, 606–608. [Google Scholar] [CrossRef]
- Itazawa, T.; Adachi, Y.; Takahashi, Y.; Miura, K.; Uehara, Y.; Kameda, M.; Kitamura, T.; Kuzume, K.; Tezuka, J.; Ito, K.; et al. The Severity of Reaction after Food Challenges Depends on the Indication: A Prospective Multicenter Study. Pediatr. Allergy Immunol. 2020, 31, 167–174. [Google Scholar] [CrossRef]
- Koutlas, N.; Stallings, A.; Hall, G.; Zhou, C.; Kim-Chang, J.; Mousallem, T. Pediatric Oral Food Challenges in the Outpatient Setting: A Single-Center Experience. J. Allergy Clin. Immunol. Glob. 2024, 3, 100187. [Google Scholar] [CrossRef]
- Yagmur, I.T.; Celik, I.K.; Topal, O.Y.; Toyran, M.; Civelek, E.; Misirlioglu, E.D. Development of Anaphylaxis upon Oral Food Challenge and Drug Provocation Tests in Pediatric Patients. Allergy Asthma Proc. 2023, 44, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Jeong, K.; Park, M.; Roh, Y.Y.; Jung, J.H.; Kim, S.Y.; Kim, J.D.; Kim, M.J.; Kim, Y.H.; Sohn, M.H.; et al. Predicting the Outcome of Pediatric Oral Food Challenges for Determining Tolerance Development. Allergy Asthma Immunol. Res. 2024, 16, 179–190. [Google Scholar] [CrossRef]
- Pouessel, G.; Jean-Bart, C.; Deschildre, A.; Van der Brempt, X.; Tanno, L.K.; Beaumont, P.; Dumond, P.; Sabouraud-Leclerc, D.; Beaudouin, E.; Ramdane, N.; et al. Food-Induced Anaphylaxis in Infancy Compared to Preschool Age: A Retrospective Analysis. Clin. Exp. Allergy 2020, 50, 74–81. [Google Scholar] [CrossRef]
- Kennedy, K.; Alfaro, M.K.C.; Spergel, Z.C.; Dorris, S.L.; Spergel, J.M.; Capucilli, P. Differences in Oral Food Challenge Reaction Severity Based on Increasing Age in a Pediatric Population. Ann. Allergy Asthma Immunol. 2021, 127, 562–567.e1. [Google Scholar] [CrossRef] [PubMed]
- Capucilli, P.; Cianferoni, A.; Fiedler, J.; Gober, L.; Pawlowski, N.; Ram, G.; Saltzman, R.; Spergel, J.M.; Heimall, J. Differences in Egg and Milk Food Challenge Outcomes Based on Tolerance to the Baked Form. Ann. Allergy Asthma Immunol. 2018, 121, 580–587. [Google Scholar] [CrossRef] [PubMed]
- Aquilante, B.P.; Castro, A.P.B.M.; Yonamine, G.H.; de Barros Dorna, M.; Barp, M.F.; Martins, T.P.d.R.; Pastorino, A.C. IgE-Mediated Cow’s Milk Allergy in Brazilian Children: Outcomes of Oral Food Challenge. World Allergy Organ. J. 2023, 16, 100781. [Google Scholar] [CrossRef]
- Esteban, C.A.; Shreffler, W.G.; Virkud, Y.V.; Pistiner, M. Oral Food Challenge Outcomes in Children under 3 Years of Age. J. Allergy Clin. Immunol. Pract. 2020, 8, 3653–3656.e3. [Google Scholar] [CrossRef] [PubMed]
- Yanagida, N.; Sato, S.; Nagakura, K.; Takahashi, K.; Fusayasu, N.; Miura, Y.; Itonaga, T.; Ogura, K.; Ebisawa, M. Relationship between Serum Allergen-Specific Immunoglobulin E and Threshold Dose in an Oral Food Challenge. Pediatr. Allergy Immunol. 2023, 34, e13926. [Google Scholar] [CrossRef] [PubMed]
- Triggiani, M.; Patella, V.; Staiano, R.I.; Granata, F.; Marone, G. Allergy and the Cardiovascular System. Clin. Exp. Immunol. 2008, 153, 7–11. [Google Scholar] [CrossRef]
- Yanagida, N.; Minoura, T.; Sato, S.; Takahashi, K.; Nagakura, K.; Ogura, K.; Itonaga, T.; Miura, Y.; Fusayasu, N.; Ebisawa, M. Timing of Initial Symptom Onset during Milk and Wheat Challenges: A Retrospective Study. Immun. Inflamm. Dis. 2024, 12, e1174. [Google Scholar] [CrossRef]
- Shin, M. Food Allergies and Food-Induced Anaphylaxis: Role of Cofactors. Clin. Exp. Pediatr. 2021, 64, 393–399. [Google Scholar] [CrossRef]
- Benedé, S.; Garrido-Arandia, M.; Martín-Pedraza, L.; Bueno, C.; Díaz-Perales, A.; Villalba, M. Multifactorial Modulation of Food-Induced Anaphylaxis. Front. Immunol. 2017, 8, 552. [Google Scholar] [CrossRef]
- Upton, J.E.M.; Bird, J.A. Oral Food Challenges: Special Considerations. Ann. Allergy Asthma Immunol. 2020, 124, 451–458. [Google Scholar] [CrossRef]
- Chinthrajah, R.S.; Purington, N.; Andorf, S.; Rosa, J.S.; Mukai, K.; Hamilton, R.; Smith, B.M.; Gupta, R.; Galli, S.J.; Desai, M.; et al. Development of a Tool Predicting Severity of Allergic Reaction during Peanut Challenge. Ann. Allergy Asthma Immunol. 2018, 121, 69–76.e2. [Google Scholar] [CrossRef]
- Purington, N.; Chinthrajah, R.S.; Long, A.; Sindher, S.; Andorf, S.; O’Laughlin, K.; Woch, M.A.; Scheiber, A.; Assa’Ad, A.; Pongracic, J.; et al. Eliciting Dose and Safety Outcomes from a Large Dataset of Standardized Multiple Food Challenges. Front. Immunol. 2018, 9, 2057. [Google Scholar] [CrossRef] [PubMed]
- Olabarri, M.; Vazquez, P.; Gonzalez-Posada, A.; Sanz, N.; Gonzalez-Peris, S.; Diez, N.; Vinuesa, A.; Martinez-Indart, L.; Benito, J.; Mintegi, S. Risk Factors for Severe Anaphylaxis in Children. J. Pediatr. 2020, 225, 193–197.e5. [Google Scholar] [CrossRef]
- Cichocka-Jarosz, E.; Dölle-Bierke, S.; Jedynak-Wąsowicz, U.; Sabouraud-Leclerc, D.; Köhli, A.; Lange, L.; Papadopoulos, N.G.; Hourihane, J.; Nemat, K.; Scherer Hofmeier, K.; et al. Cow’s Milk and Hen’s Egg Anaphylaxis: A Comprehensive Data Analysis from the European Anaphylaxis Registry. Clin. Transl. Allergy 2023, 13, e12228. [Google Scholar] [CrossRef]
- Nishino, M.; Yanagida, N.; Sato, S.; Nagakura, K.; Takahashi, K.; Ogura, K.; Ebisawa, M. Risk Factors for Failing a Repeat Oral Food Challenge in Preschool Children with Hen’s Egg Allergy. Pediatr. Allergy Immunol. 2022, 33, e13895. [Google Scholar] [CrossRef]
- Taniuchi, S.; Sakai, R.; Nishida, T.; Goma, M.; Mitomori, M.; Imaide, A.; Enomoto, M.; Nishino, M.; Okizuka, Y.; Kido, H. The Combination of Binding Avidity of Ovomucoid-Specific IgE Antibody and Specific IgG4 Antibody Can Predict Positive Outcomes of Oral Food Challenges during Stepwise Slow Oral Immunotherapy in Children with Hen’s Egg Allergy. Nutrients 2023, 15, 2770. [Google Scholar] [CrossRef]
- Cunico, D.; Giannì, G.; Scavone, S.; Buono, E.V.; Caffarelli, C. The Relationship Between Asthma and Food Allergies in Children. Children 2024, 11, 1295. [Google Scholar] [CrossRef]
- Yonkof, J.R.; Mikhail, I.J.; Prince, B.T.; Stukus, D. Delayed and Severe Reactions to Baked Egg and Baked Milk Challenges. J. Allergy Clin. Immunol. Pract. 2021, 9, 283–289.e2. [Google Scholar] [CrossRef]
- Savage, J.; Sicherer, S.; Wood, R. The Natural History of Food Allergy. J. Allergy Clin. Immunol. Pract. 2016, 4, 196–203. [Google Scholar] [CrossRef]
- Ogata, M.; Kido, J.; Yoshida, T.; Nishi, N.; Shimomura, S.; Hirai, N.; Mizukami, T.; Yanai, M.; Nakamura, K. The Efficacy and Safety of Stepwise Oral Food Challenge in Children with Hen’s Egg Allergy. Allergy Asthma Clin. Immunol. 2024, 20, 67. [Google Scholar] [CrossRef] [PubMed]
- Hill, S.A.; Nurmatov, U.; DunnGalvin, A.; Reese, I.; Vieira, M.C.; Rommel, N.; Dupont, C.; Venter, C.; Cianferoni, A.; Walsh, J.; et al. Feeding Difficulties in Children with Food Allergies: An EAACI Task Force Report. Pediatr. Allergy Immunol. 2024, 35, e14119. [Google Scholar] [CrossRef] [PubMed]
- Domínguez, O.; Plaza, A.M.; Alvaro, M. Relationship Between Atopic Dermatitis and Food Allergy. Curr. Pediatr. Rev. 2019, 16, 115–122. [Google Scholar] [CrossRef]
- Mohammad Tariq, S.; Marie Matthews, S.; Abe Hakim, E.; Stevens, M.; Hasan Arshad, S.; Wallace Hide, D. The Prevalence of and Risk Factors for Atopy in Early Childhood: A Whole Population Birth Cohort Study. J. Allergy Clin. Immunol. 1998, 101, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Manti, S.; Galletta, F.; Bencivenga, C.L.; Bettini, I.; Klain, A.; D’Addio, E.; Mori, F.; Licari, A.; Miraglia Del Giudice, M.; Indolfi, C. Food Allergy Risk: A Comprehensive Review of Maternal Interventions for Food Allergy Prevention. Nutrients 2024, 16, 1087. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, P.M.; Cassin, A.M.; Durban, R.; Upton, J.E.M. Effects of Food Processing on Allergenicity. Curr. Allergy Asthma Rep. 2025, 25, 9. [Google Scholar] [CrossRef]
- Nowak-Wegrzyn, A.; Bloom, K.A.; Sicherer, S.H.; Shreffler, W.G.; Noone, S.; Wanich, N.; Sampson, H.A. Tolerance to Extensively Heated Milk in Children with Cow’s Milk Allergy. J. Allergy Clin. Immunol. 2008, 122, 342–347.e2. [Google Scholar] [CrossRef]
- Gantulga, P.; Lee, J.; Jeong, K.; Jeon, S.A.; Lee, S. Variation in the Allergenicity of Scrambled, Boiled, Short-Baked and Long-Baked Egg White Proteins. J. Korean Med. Sci. 2024, 39, e54. [Google Scholar] [CrossRef]
- Gruzelle, V.; Juchet, A.; Martin-Blondel, A.; Michelet, M.; Chabbert-Broue, A.; Didier, A. Benefits of Baked Milk Oral Immunotherapy in French Children with Cow’s Milk Allergy. Pediatr. Allergy Immunol. 2020, 31, 364–370. [Google Scholar] [CrossRef]
- Kim, J.S.; Nowak-Wgrzyn, A.; Sicherer, S.H.; Noone, S.; Moshier, E.L.; Sampson, H.A. Dietary Baked Milk Accelerates the Resolution of Cow’s Milk Allergy in Children. J. Allergy Clin. Immunol. 2011, 128, 125–131.e2. [Google Scholar] [CrossRef] [PubMed]
- Giannetti, A.; Toschi Vespasiani, G.; Ricci, G.; Miniaci, A.; Di Palmo, E.; Pession, A. Cow’s Milk Protein Allergy as a Model of Food Allergies. Nutrients 2021, 13, 1525. [Google Scholar] [CrossRef]
- Leonard, S.A.; Sampson, H.A.; Sicherer, S.H.; Noone, S.; Moshier, E.L.; Godbold, J.; Nowak-Wegrzyn, A. Dietary Baked Egg Accelerates Resolution of Egg Allergy in Children. J. Allergy Clin. Immunol. 2012, 130, 473–480.e1. [Google Scholar] [CrossRef]
- Upton, J.E.M.; Wong, D.; Nowak-Wegrzyn, A. Baked Milk and Egg Diets Revisited. Ann. Allergy Asthma Immunol. 2024, 132, 328–336.e5. [Google Scholar] [CrossRef] [PubMed]
- Buyuktiryaki, B.; Soyer, O.; Bingol, G.; Can, C.; Nacaroglu, H.T.; Bingol, A.; Arik Yilmaz, E.; Aydogan, M.; Sackesen, C. Milk Ladder: Who? When? How? Where? With the Lowest Risk of Reaction. Front. Allergy 2024, 5, 1516774. [Google Scholar] [CrossRef]
- Meyer, R.; Venter, C.; Bognanni, A.; Szajewska, H.; Shamir, R.; Nowak-Wegrzyn, A.; Fiocchi, A.; Vandenplas, Y. World Allergy Organization (WAO) Diagnosis and Rationale for Action against Cow’s Milk Allergy (DRACMA) Guideline Update—VII—Milk Elimination and Reintroduction in the Diagnostic Process of Cow’s Milk Allergy. World Allergy Organ. J. 2023, 16, 100785. [Google Scholar] [CrossRef]
- De Boer, R.; Cartledge, N.; Lazenby, S.; Tobias, A.; Chan, S.; Fox, A.T.; Santos, A.F. Specific IgE as the Best Predictor of the Outcome of Challenges to Baked Milk and Baked Egg. J. Allergy Clin. Immunol. Pract. 2020, 8, 1459–1461.e5. [Google Scholar] [CrossRef]
- Yanagida, N.; Sato, S.; Ebisawa, M. Relationship between Eliciting Doses and the Severity of Allergic Reactions to Food. Curr. Opin. Allergy Clin. Immunol. 2023, 23, 226–232. [Google Scholar] [CrossRef]
- Yanagida, N.; Sato, S.; Takahashi, K.; Nagakura, K.I.; Asaumi, T.; Ogura, K.; Ebisawa, M. Increasing Specific Immunoglobulin E Levels Correlate with the Risk of Anaphylaxis during an Oral Food Challenge. Pediatr. Allergy Immunol. 2018, 29, 417–424. [Google Scholar] [CrossRef]
- Coimbra, M.R.; Araújo, L.M.L.; Filho, N.A.R. Oral Food Challenge in Children with Contact Urticaria in Reaction to Cow’s Milk. Allergol. Immunopathol. 2023, 51, 93–98. [Google Scholar] [CrossRef]
- Sasaki, Y.; Matsunami, K.; Kondo, M.; Matsukuma, E.; Imamura, A.; Kaneko, H. Oral Food Challenge Test Results of Patients with Food Allergy with Specific IgE Levels >100 UA/Ml. Biomed. Rep. 2024, 21, 178. [Google Scholar] [CrossRef] [PubMed]
- Gawryjołek, J.; Wycech, A.; Smyk, A.; Krogulska, A. Difficulties in Interpretation of Oral Food Challenge Results. Postep. Dermatol. Alergol. 2021, 38, 721–726. [Google Scholar] [CrossRef]
- Valluzzi, R.L.; Riccardi, C.; Urbani, S.; Ursi, D.; Zavettieri, D.; Di Girolamo, F.; Dahdah, L.; Calandrelli, V.; Fierro, V.; Fiocchi, A. The Baked Side: Cow’s Milk and Egg Protein Threshold Dose Distributions in Children Reacting to Baked Milk and Baked Egg. World Allergy Organ. J. 2025, 18, 101012. [Google Scholar] [CrossRef]
- Valluzzi, R.L.; Riccardi, C.; Arasi, S.; Piscitelli, A.L.; Calandrelli, V.; Dahdah, L.; Fierro, V.; Mennini, M.; Fiocchi, A. Cow’s Milk and Egg Protein Threshold Dose Distributions in Children Tolerant to Beef, Baked Milk, and Baked Egg. Allergy Eur. J. Allergy Clin. Immunol. 2022, 77, 3052–3060. [Google Scholar] [CrossRef] [PubMed]
- Tosca, M.A.; Schiavetti, I.; Olcese, R.; Trincianti, C.; Ciprandi, G. Molecular Allergy Diagnostics in Children with Cow’s Milk Allergy: Prediction of Oral Food Challenge Response in Clinical Practice. J. Immunol. Res. 2023, 2023, 1129449. [Google Scholar] [CrossRef] [PubMed]
- Upton, J.; Alvaro, M.; Nadeau, K. A Perspective on the Pediatric Death from Oral Food Challenge Reported from the Allergy Vigilance Network. Allergy Eur. J. Allergy Clin. Immunol. 2019, 74, 1035–1036. [Google Scholar] [CrossRef] [PubMed]
- Sampson, H.A.; Arasi, S.; Bahnson, H.T.; Ballmer-Weber, B.; Beyer, K.; Bindslev-Jensen, C.; Bird, J.A.; Blumchen, K.; Davis, C.; Ebisawa, M.; et al. AAAAI–EAACI PRACTALL: Standardizing Oral Food Challenges—2024 Update. Pediatr. Allergy Immunol. 2024, 35, e14276. [Google Scholar] [CrossRef]
- Anagnostou, K. Safety of Oral Food Challenges in Early Life. Children 2018, 5, 65. [Google Scholar] [CrossRef] [PubMed]
- Sirin Kose, S.; Asilsoy, S.; Uzuner, N.; Karaman, O.; Anal, O. Outcomes of Baked Milk and Egg Challenge in Cow’s Milk and Hen’s Egg Allergy: Can Tolerance Be Predicted with Allergen-Specific IgE and Prick-to-Prick Test? Int. Arch. Allergy Immunol. 2019, 180, 264–273. [Google Scholar] [CrossRef] [PubMed]
| Feature | Value |
|---|---|
| Number of patients | 205 * |
| Mean age, SD; age range (years) | 5.7 ± 3.1; 1.2–17.8 |
| Gender, n (%) | |
| Male | 135 (65.9%) |
| Female | 70 (34.1%) |
| Allergen challenged in the OFC, n (%) | |
| Cow’s milk protein | 103 (50.2%) |
| Hen’s egg white protein | 84 (41.0%) |
| Peanuts and tree nuts (hazelnut, cashew, and walnut) | 7 (3.4%) |
| Peanuts | 3 (1.5%) |
| Hazelnuts | 2 (1.0%) |
| Cashew | 1 (0.5%) |
| Walnut | 1 (0.5%) |
| Gluten | 3 (1.5%) |
| Hen’s egg yolk | 4 (2.0%) |
| Other (chicken, cod, banana, and potato) | 4 (2.0%) |
| Mean time from food consumption to reaction occurrence, SD, range (minutes) | 94.1 ± 50.4; 0–230 |
| Accompanying atopic diseases | |
| Multi-food allergy | 152 (74.1%) |
| Atopic dermatitis | 124 (60.5%) |
| Asthma | 101 (49.3%) |
| Family history of atopy | 85 (41.5%) |
| Previous history of anaphylaxis to any food | 158 (77.1%) |
| Previous history of anaphylaxis to challenged food | 110 (53.7%) |
| Feature | CMP (n = 103) * | HEWP (n = 84) * | p |
|---|---|---|---|
| Mean age, SD; age range (years) | 5.41 | 5.54 | 0.52 |
| Min | 1.18 | 1.54 | |
| Max | 17.84 | 16.54 | |
| SD | 3.03 | 2.91 | |
| Gender, n (%) | |||
| Male | 70 (67.96%) | 55 (65.48%) | 0.72 |
| Female | 33 (32.04%) | 29 (34.52%) | |
| Mean time from consumption to reaction occurrence, SD, range (minutes) | 97.28 | 95.86 | 0.88 |
| Min | 0 | 0 | |
| Max | 230 | 180 | |
| SD | 56.3 | 43.96 | |
| Accompanying atopic diseases | |||
| Multi-food allergy | 74 (71.84%) | 66 (78.57%) | 0.29 |
| Atopic dermatitis | 55(53.4%) | 58 (69.05%) | 0.03 |
| Asthma | 54 (52.43%) | 38 (45.24%) | 0.33 |
| Family history of atopy | 44 (42.72%) | 36 (42.86%) | 0.99 |
| Previous history of anaphylaxis to any food | 79 (76.7%) | 64 (79.19%) | 0.94 |
| Previous history of anaphylaxis to challenged food | 63 (61.17%) | 40 (47.62%) | 0.06 |
| Allergen Challenged | Total No. of OFCs * | No. of Failed OFCs (%) |
|---|---|---|
| Milk: | 103 | 32 (31.1%) |
| Baked milk | 66 | 20 (30.3%) |
| Non-baked milk: | 37 | 12 (32.4%) |
| Fermented milk | 14 | 5 (35.7%) |
| Plain fresh milk | 7 | 1 (14.3%) |
| Modified infant formula | 7 | 3 (42.9%) |
| Heated fresh milk | 6 | 1 (16.7%) |
| Other forms | 3 | 2 (66.7%) |
| Hen’s Egg White: | 84 | 29 (34.5%) |
| Baked | 60 | 21 (35.0%) |
| Cooked | 24 | 8 (33.3%) |
| Hen’s Egg Yolk: | 4 | 1 (25.0%) |
| Cooked | 3 | 1 (33.3%) |
| Baked | 1 | 0 (0.0%) |
| Peanuts and tree nuts: | 7 | 1 (14.3%) |
| Peanuts | 3 | 1 (33.3%) |
| Hazelnuts | 2 | 0 (0.0%) |
| Cashew | 1 | 0 (0.0%) |
| Walnut | 1 | 0 (0.0%) |
| Gluten | 3 | 2 (66.7%) |
| Other: | 4 | 1 (25.0%) |
| Chicken | 1 | 1 (100.0%) |
| Cod | 1 | 0 (0.0%) |
| Banana | 1 | 0 (0.0%) |
| Potato | 1 | 0 (0.0%) |
| N | Cow’s Milk Protein [g] | N | Hen’s Egg White Protein [g] | p Value | |
|---|---|---|---|---|---|
| Baked | 20 | 0.27 [0.135–0.54] | 21 | 0.58 [0.29–1.17] | 0.01 |
| Non-baked | 12 | 1.16 [0.12–1.66] | 8 | 2.68 [0.765–3.83] | 0.06 |
| Variables | Coef. | Std. Err. | z | p > |z| | [0.025 | 0.975] |
|---|---|---|---|---|---|---|
| Asthma | 0.67 | 0.31 | 2.19 | 0.03 | 0.07 | 1.27 |
| Atopic dermatitis | 0.13 | 0.31 | 0.43 | 0.67 | −0.47 | 0.74 |
| Previous history of anaphylaxis to any food | 0.61 | 0.36 | 1.69 | 0.09 | −0.10 | 1.31 |
| Previous history of anaphylaxis to challenged food | 0.49 | 0.29 | 1.66 | 0.10 | −0.09 | 1.07 |
| Multi-food allergy | 0.90 | 0.39 | 2.31 | 0.02 | 0.14 | 1.67 |
| Baked | Non-Baked | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| N | Passed | N | Failed | p Value | N | Passed | N | Failed | p Value | |
| CMP-specific IgE [kU/L] | 34 | 18.80 [5.69–35.90] | 17 | 45.00 [6.77–65.90] | 0.20 | 19 | 1.53 [0.25–4.05] | 8 | 1.28 [0.39–5.88] | 0.83 |
| HEWP-specific IgE [kU/L] | 28 | 6.48 [3.28–18.90] | 13 | 18.90 [5.87–35.40] | 0.01 | 12 | 1.12 [0.53–3.45] | 7 | 23.90 [3.22–39.00] | 0.12 |
| N | CMP-Specific IgE [kU/L] | N | HEWP-Specific IgE [kU/L] | p Value | |
|---|---|---|---|---|---|
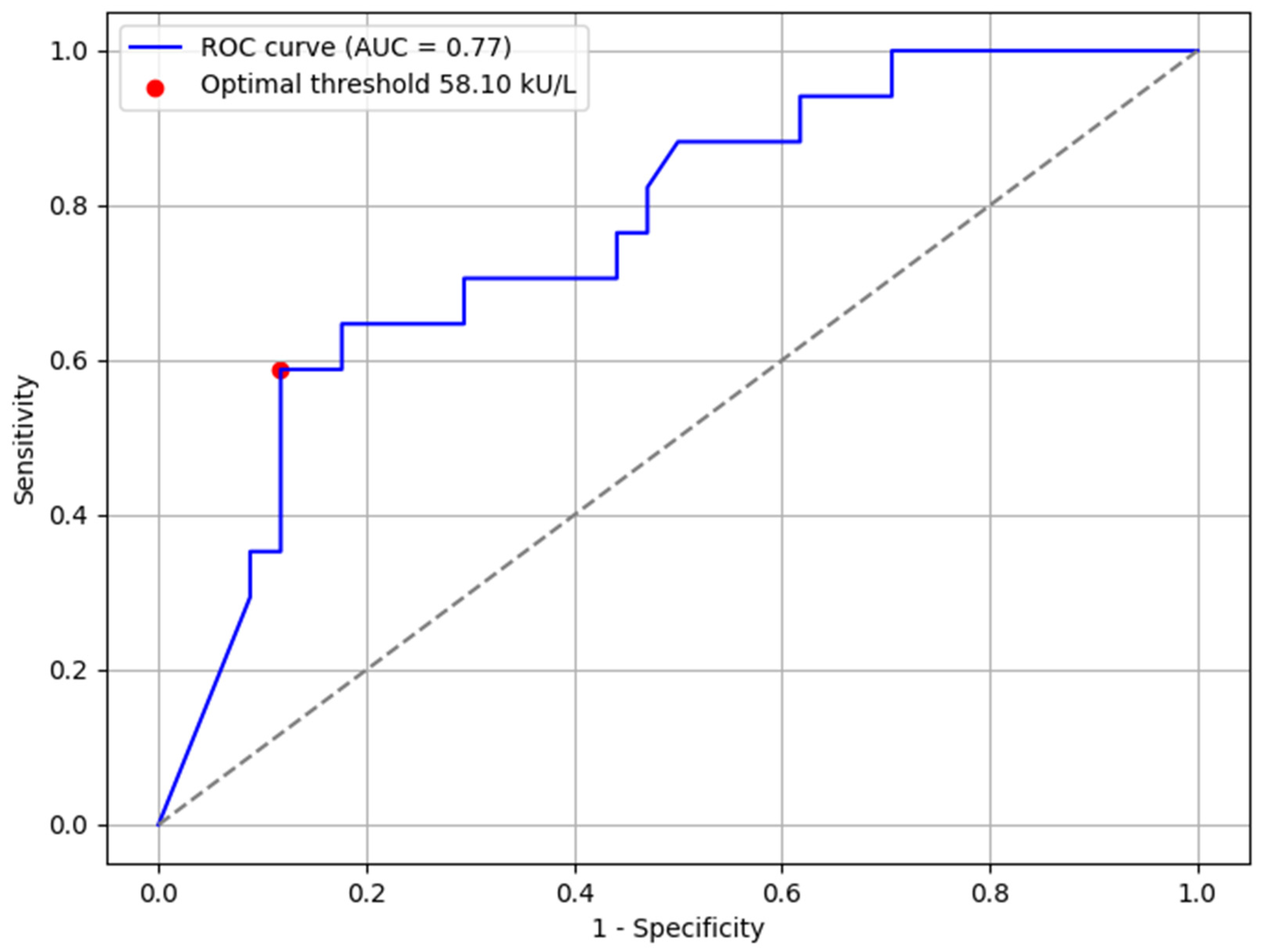
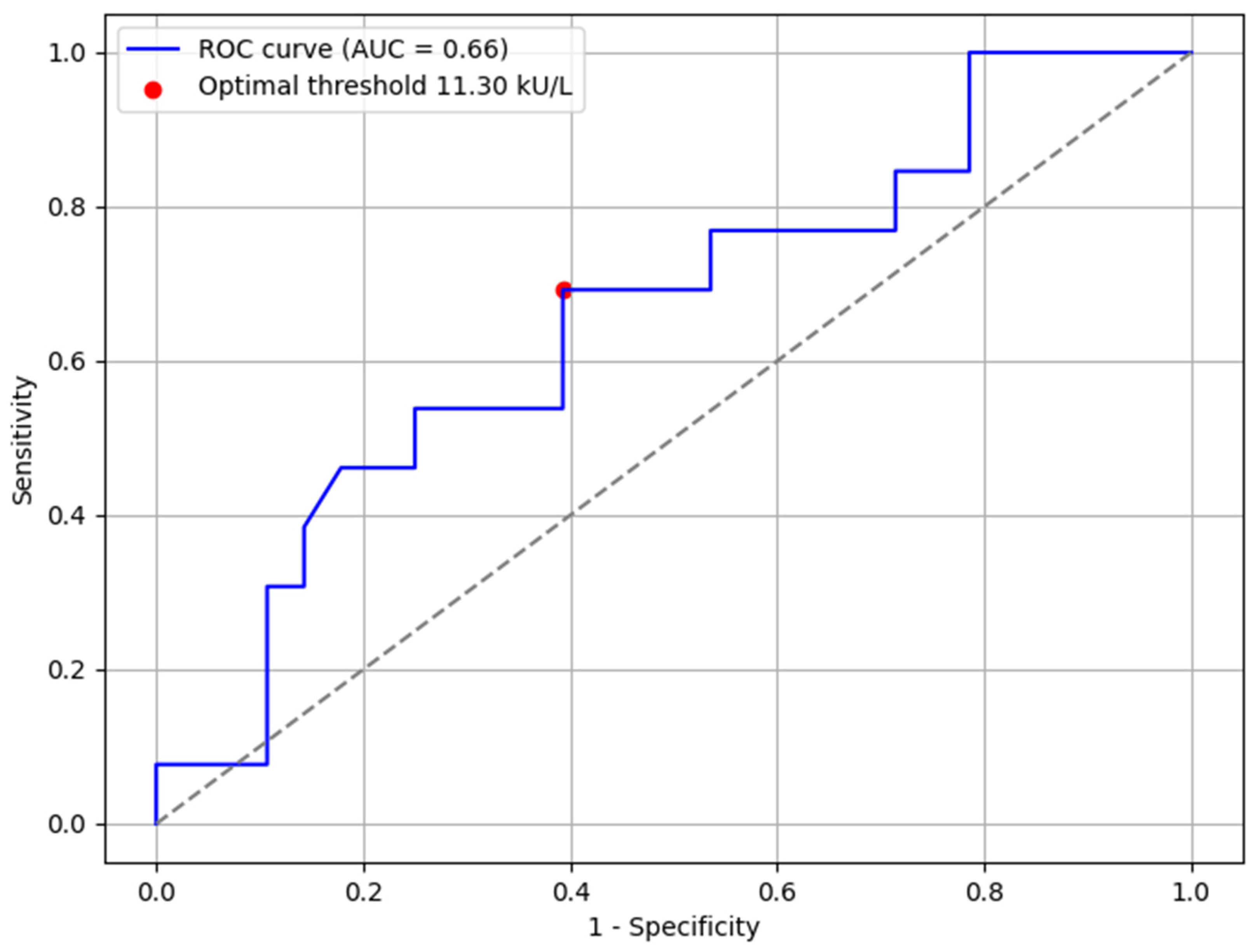
| Baked | 17 | 58.1 [19.4–100.0] | 13 | 18.9 [5.87–35.4] | 0.02 |
| Non-baked | 8 | 3.53 [1.13–8.58] | 7 | 23.9 [3.22–39.0] | 0.06 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klim, L.; Michalik, M.; Cichocka-Jarosz, E.; Jedynak-Wąsowicz, U. Asthma and Multi-Food Allergy Are Risk Factors for Oral Food Challenge Failure—A Single-Center Experience. Nutrients 2025, 17, 2769. https://doi.org/10.3390/nu17172769
Klim L, Michalik M, Cichocka-Jarosz E, Jedynak-Wąsowicz U. Asthma and Multi-Food Allergy Are Risk Factors for Oral Food Challenge Failure—A Single-Center Experience. Nutrients. 2025; 17(17):2769. https://doi.org/10.3390/nu17172769
Chicago/Turabian StyleKlim, Liliana, Maria Michalik, Ewa Cichocka-Jarosz, and Urszula Jedynak-Wąsowicz. 2025. "Asthma and Multi-Food Allergy Are Risk Factors for Oral Food Challenge Failure—A Single-Center Experience" Nutrients 17, no. 17: 2769. https://doi.org/10.3390/nu17172769
APA StyleKlim, L., Michalik, M., Cichocka-Jarosz, E., & Jedynak-Wąsowicz, U. (2025). Asthma and Multi-Food Allergy Are Risk Factors for Oral Food Challenge Failure—A Single-Center Experience. Nutrients, 17(17), 2769. https://doi.org/10.3390/nu17172769






