AMPA Receptor Modulation Through Medium-Chain Triglycerides and Decanoic Acid Supports Nutritional Intervention in Pediatric Epilepsy
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy and Eligibility Criteria
- Database Searched: The search was performed on PubMed, and the last search was carried out on 31 January 2025. The keywords used included: “ketogenic diet”, “medium-chain triglycerides”, “decanoic acid”, “AMPA receptor”, “drug-resistant epilepsy”, “pediatric epilepsy”, and “MCT supplementation”. The systematic literature search was conducted employing the Boolean operator AND to combine pairs of selected keywords, thereby restricting the results to studies containing all specified terms within each query. As part of the search strategy, in accordance with PRISMA guidelines, we applied predefined filters on PubMed to restrict results based on the inclusion criteria. These filters included publication type (e.g., original articles, clinical studies, RCT etc.), language (English), and publication date range to ensure that only studies relevant to the objectives of the systematic review were considered.
- Study Period: We included articles published from January 2000 to January 2025 in order to collect the latest scientific evidence.
- Inclusion Criteria:
- Preclinical Studies: Investigations using cellular/in vitro models or animal models that assessed seizure thresholds, seizure frequency, receptor-binding characteristics, or relevant mechanistic insights tied to MCTs or decanoic acid.
- Clinical Studies: Research on pediatric DRE or adults where mechanistic insights are relevant to pediatrics (including observational studies, case series, pilot/feasibility studies, randomized controlled trials (RCTs), and meta-analyses).
- AMPA Receptor Modulation Mechanisms: Studies that provided mechanistic or functional data on the interaction between decanoic acid (or MCTs) and excitatory glutamatergic pathways.
- Articles in English.
- Exclusion Criteria:
- Reviews, editorials, and conference abstracts without original data.
- Studies focused exclusively on adult epilepsy or other pathologies without translational relevance.
- Studies unrelated to MCTs or ketogenic diets.
2.2. Study Selection and Data Extraction
- Study Type and Design: Preclinical (in vitro, animal), observational (retrospective/prospective cohorts), RCT, meta-analysis.
- Population: Age range, epilepsy etiology/subtypes, DRE status.
- Dietary Intervention: Classic KD, MCT-based KD, or free diet with MCT supplementation; specific dosing regimens if provided.
- Endpoints: Seizure frequency/severity, metabolic biomarkers (ketone levels, lipid profiles), tolerability/adverse events, adherence rates, neurocognitive outcomes, and putative mechanistic measures (e.g., receptor binding, neuroinflammatory markers).
- Results: Efficacy measures (e.g., % seizure reduction, seizure-freedom rates), mechanistic findings (AMPA receptor modulation), and study limitations.
2.3. Data Synthesis and Analysis
- Preclinical models elucidating the mechanistic underpinnings of MCT/decanoic acid actions.
- Clinical trials and observational studies detailing dietary interventions in pediatric populations with DRE.
- Mechanistic or structural analyses of AMPA receptor antagonism relevant to MCTs.
3. Results
3.1. Preclinical Evidence Supporting AMPA Receptor Modulation by MCTs
- Demonstrated that MCTs (e.g., decanoic acid) reduce seizure activity in rodent models.
- Proposed inhibitory action at the AMPA receptor, backed by electrophysiological data.
- Proposed Butyl Cyclohexane Carboxylic Acid (4-BCCA), a derivative of the octanoic acid, as an alternative in cases of poor tolerability of KD, since it has shown potent antiseizure activity.
- Yelshanskaya et al. (2016) [11]:
- Explored structural bases of noncompetitive inhibition of AMPA-subtype ionotropic glutamate receptors by antiepileptic drugs.
- While not specific to MCT supplementation, the structural insights provide a mechanistic framework to understand how molecules like decanoic acid might interact with AMPA receptor sites.
- Augustin et al. (2018) [12]:
- Showed synergistic effects of perampanel (an AMPA receptor antagonist) and decanoic acid on seizure reduction in animal models, reinforcing the concept of decanoic acid as an AMPA receptor modulator.
- Berk et al. (2022) [13]:
- Although conducted in dogs with idiopathic epilepsy, this study reported metabolic shifts linked to MCT oil supplementation. The authors posited that improved seizure control could be partly derived from MCT-induced changes in neurotransmitter balance or receptor modulation.
3.2. Clinical Studies on MCT-Based KD
- Neal et al. (2009) [14]:
- Compared classical KD vs. MCT-KD in pediatric epilepsy. Both diets reduced seizures, but MCT-KD allowed for slightly higher carbohydrate intake, potentially improving dietary tolerance.
- Lambrechts et al. (2015) [15]:
- A prospective 2-year follow-up study in children on MCT-KD reported sustained seizure reduction. Compliance over the long term, though challenging, remained feasible for a subset of families, highlighting that MCTs might improve palatability relative to classic KD.
- Henderson et al. (2006) [16]:
- Meta-analysis of KD efficacy in epilepsy (not exclusively MCT-based diets), but included data supporting the potential advantages of MCT-KD in certain subgroups.
- Shin et al. (2025) [17]:
- Investigated a decanoic acid-enriched KD in children with refractory epilepsy. Reported notable seizure reduction in a significant proportion of participants and underscored the correlation between higher plasma decanoic acid levels and AMPA receptor modulation.
3.3. Trials and Observational Studies on MCT Supplementation in Less Restrictive Diets
- Borges et al. (2019) [18]:
- Randomized trial comparing triheptanoin vs. standard MCT oil as add-on therapy in adults with refractory epilepsy. Demonstrated partial seizure frequency reductions with MCT, offering proof of concept for less restrictive diets. Though adult-based, the rationale can be extrapolated to pediatrics.
- Schoeler et al. (2021) [19]:
- Investigated a blend of MCT oils as an adjunct in drug-resistant epilepsy. Showed feasibility and some seizure reduction benefits, albeit with variable individual responses. This open-label design in a less restrictive dietary context highlights the potential of partial dietary modification.
- Rasmussen et al. (2022) [20]:
- This preliminary study suggests that MCT oil supplementation may significantly reduce seizure frequency in adults with intractable epilepsy, with a 42% reduction observed. The intervention was generally well tolerated, with only mild gastrointestinal side effects.
3.4. AMPA Receptor Modulation Mechanisms
- Chang et al. (2016) [7]
- Demonstrated that decanoic acid directly inhibits AMPA receptors.
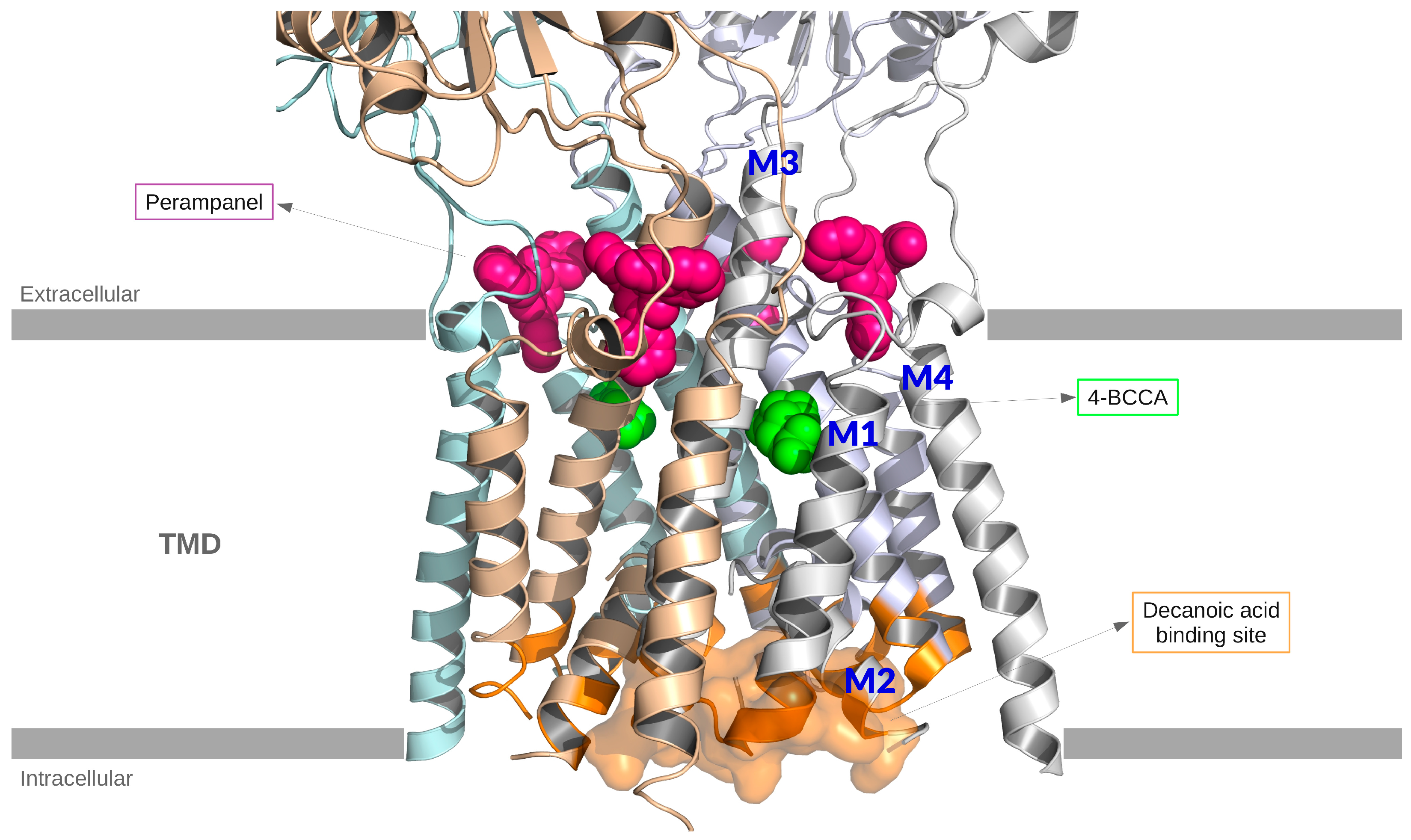
- With an in silico docking approach, they found that the most frequent residues interacting with decanoic acid are located in the M3 helix of the transmembrane domain, which is involved in gating.
- They showed that non-competitive voltage- and subunit-dependent inhibition offers a distinct mechanism from ketone bodies, as decanoic acid is binding to a different site than the typical antagonists used in epilepsy treatment, such as perampanel.
- Narangoda et al. (2019) [21]
- Employed molecular dynamics simulations and thermodynamic integration to investigate how three structurally diverse noncompetitive inhibitors—perampanel, GYKI 53655, and CP 465022—interact with AMPA receptors.
- By using the crystal structures as initial binding configurations, they observed that all three inhibitors undergo several adjustments, but their binding remains stable over time.
- They found stable alternative binding modes to those observed in crystal structures, suggesting that the binding site is quite flexible and capable of accommodating multiple ligands in a variety of poses.
- Yelshanskaya et al. (2022) [22]
- Studied the interaction between the rat AMPA receptor GluA2 and 4-BCCA.
- Located the 4-BCCA binding sites in AMPA transmembrane domain (TMD) using X-ray crystallography, showing two 4-BCCA molecules bound in different orientations to the TMD of the channel near the pore lining M3 helices.
- Identified 4-BCCA binding region close to those previously predicted for decanoic acid (Chang et al. 2016 [7]), but further up in the channel and closer to the SYTANLAAF motif in M3.
- Showed via MD simulations and mutagenesis analysis that the 4-BCCA molecule adopts alternative orientations within its binding site; a behavior consistent with its low affinity.
- 4-BCCA binding is proposed to interfere with ion flow and receptor gating, hinting at possible synergy with other AMPA receptor modulators.
- Authors suggested that the identified 4-BCCA binding sites are probable binding regions for the whole group of medium-chain fatty acids and their branched derivatives that share the inhibitory mechanism.
4. Discussion
5. Conclusions
6. Limits
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Jehi, L. Advances in Therapy for Refractory Epilepsy. Annu. Rev. Med. 2025, 76, 389–402. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.H.; Tsai, C.Y.; Chou, I.C.; Tsai, J.D. The impact of parenting stress on parents of school-age children with drug-resistant epilepsy. Front. Pediatr. 2022, 10, 948286. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pizzo, F.; Collotta, A.D.; Di Nora, A.; Costanza, G.; Ruggieri, M.; Falsaperla, R. Ketogenic diet in pediatric seizures: A randomized controlled trial review and meta-analysis. Expert Rev. Neurother. 2022, 22, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Dyńka, D.; Kowalcze, K.; Paziewska, A. The Role of Ketogenic Diet in the Treatment of Neurological Diseases. Nutrients 2022, 14, 5003. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Borowicz-Reutt, K.; Krawczyk, M.; Czernia, J. Ketogenic Diet in the Treatment of Epilepsy. Nutrients 2024, 16, 1258. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Augustin, K.; Khabbush, A.; Williams, S.; Eaton, S.; Orford, M.; Cross, J.H.; Heales, S.J.R.; Walker, M.C.; Williams, R.S.B. Mechanisms of action for the medium-chain triglyceride ketogenic diet in neurological and metabolic disorders. Lancet Neurol. 2018, 17, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.; Augustin, K.; Boddum, K.; Williams, S.; Sun, M.; Terschak, J.A.; Hardege, J.D.; Chen, P.E.; Walker, M.C.; Williams, R.S. Seizure control by decanoic acid through direct AMPA receptor inhibition. Brain 2016, 139 Pt 2, 431–443. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wright, S.K.; Wilson, M.A.; Walsh, R.; Lo, W.B.; Mundil, N.; Agrawal, S.; Philip, S.; Seri, S.; Greenhill, S.D.; Woodhall, G.L. Abolishing spontaneous epileptiform activity in human brain tissue through AMPA receptor inhibition. Ann. Clin. Transl. Neurol. 2020, 7, 883–890. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chang, P.; Terbach, N.; Plant, N.; Chen, P.E.; Walker, M.C.; Williams, R.S. Seizure control by ketogenic diet-associated medium chain fatty acids. Neuropharmacology 2013, 69, 105–114. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chang, P.; Zuckermann, A.M.; Williams, S.; Close, A.J.; Cano-Jaimez, M.; McEvoy, J.P.; Spencer, J.; Walker, M.C.; Williams, R.S. Seizure control by derivatives of medium chain fatty acids associated with the ketogenic diet show novel branching-point structure for enhanced potency. J. Pharmacol. Exp. Ther. 2015, 352, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Yelshanskaya, M.V.; Singh, A.K.; Sampson, J.M.; Narangoda, C.; Kurnikova, M.; Sobolevsky, A.I. Structural Bases of Noncompetitive Inhibition of AMPA-Subtype Ionotropic Glutamate Receptors by Antiepileptic Drugs. Neuron 2016, 91, 1305–1315. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Augustin, K.; Williams, S.; Cunningham, M.; Devlin, A.M.; Friedrich, M.; Jayasekera, A.; Hussain, M.A.; Holliman, D.; Mitchell, P.; Jenkins, A.; et al. Perampanel and decanoic acid show synergistic action against AMPA receptors and seizures. Epilepsia 2018, 59, e172–e178. [Google Scholar] [CrossRef] [PubMed]
- Berk, B.A.; Ottka, C.; Hong Law, T.; Packer, R.M.A.; Wessmann, A.; Bathen-Nöthen, A.; Jokinen, T.S.; Knebel, A.; Tipold, A.; Lohi, H.; et al. Metabolic fingerprinting of dogs with idiopathic epilepsy receiving a ketogenic medium-chain triglyceride (MCT) oil. Front. Vet. Sci. 2022, 9, 935430. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Neal, E.G.; Chaffe, H.; Schwartz, R.H.; Lawson, M.S.; Edwards, N.; Fitzsimmons, G.; Whitney, A.; Cross, J.H. A randomized trial of classical and medium-chain triglyceride ketogenic diets in the treatment of childhood epilepsy. Epilepsia 2009, 50, 1109–1117. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, D.A.; de Kinderen, R.J.; Vles, H.S.; de Louw, A.J.; Aldenkamp, A.P.; Majoie, M.J. The MCT-ketogenic diet as a treatment option in refractory childhood epilepsy: A prospective study with 2-year follow-up. Epilepsy Behav. 2015, 51, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Henderson, C.B.; Filloux, F.M.; Alder, S.C.; Lyon, J.L.; Caplin, D.A. Efficacy of the ketogenic diet as a treatment option for epilepsy: Meta-analysis. J. Child Neurol. 2006, 21, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.J.; Ryu, S.; Lee, N.; Lee, E.; Ko, A.; Kang, H.C.; Lee, J.S.; Kim, S.H.; Kim, H.D. Decanoic acid-enriched ketogenic diet in refractory epilepsy. Front. Neurol. 2025, 16, 1524799. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Borges, K.; Kaul, N.; Germaine, J.; Kwan, P.; O’Brien, T.J. Randomized trial of add-on triheptanoin vs medium chain triglycerides in adults with refractory epilepsy. Epilepsia Open 2019, 4, 153–163. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Schoeler, N.E.; Orford, M.; Vivekananda, U.; Simpson, Z.; Van de Bor, B.; Smith, H.; Balestrini, S.; Rutherford, T.; Brennan, E.; McKenna, J.; et al. K.Vita: A feasibility study of a blend of medium chain triglycerides to manage drug-resistant epilepsy. Brain Commun. 2021, 3, fcab160. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rasmussen, E.; Patel, V.; Tideman, S.; Frech, R.; Frigerio, R.; Narayanan, J. Efficacy of supplemental MCT oil on seizure reduction of adult drug-resistant epilepsy—A single-center open-label pilot study. Nutr. Neurosci. 2023, 26, 535–539. [Google Scholar] [CrossRef] [PubMed]
- Narangoda, C.; Sakipov, S.N.; Kurnikova, M.G. AMPA Receptor Noncompetitive Inhibitors Occupy a Promiscuous Binding Site. ACS Chem. Neurosci. 2019, 10, 4511–4521. [Google Scholar] [CrossRef] [PubMed]
- Yelshanskaya, M.V.; Singh, A.K.; Narangoda, C.; Williams, R.S.B.; Kurnikova, M.G.; Sobolevsky, A.I. Structural basis of AMPA receptor inhibition by trans-4-butylcyclohexane carboxylic acid. Br. J. Pharmacol. 2022, 179, 3628–3644. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ko, A.; Kwon, H.E.; Kim, H.D. Updates on the ketogenic diet therapy for pediatric epilepsy. Biomed. J. 2022, 45, 19–26. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Falsaperla, R.; Sortino, V.; Striano, P.; Kluger, G.; Ramantani, G.; Ruggieri, M.; the Network for Therapy in Rare Epilepsies (NETRE). Is ketogenic diet a ‘precision medicine’? Recent developments and future challenges. Eur. J. Paediatr. Neurol. 2024, 48, 13–16. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, D.; Garg, D.; Sharma, S. Emerging Role of the Ketogenic Dietary Therapies beyond Epilepsy in Child Neurology. Ann. Indian Acad. Neurol. 2021, 24, 470–480. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Akiyama, M.; Akiyama, T.; Saigusa, D.; Hishinuma, E.; Matsukawa, N.; Shibata, T.; Tsuchiya, H.; Mori, A.; Fujii, Y.; Mogami, Y.; et al. Comprehensive study of metabolic changes induced by a ketogenic diet therapy using GC/MS- and LC/MS-based metabolomics. Seizure 2023, 107, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Griffen, C.; Schoeler, N.E.; Browne, R.; Cameron, T.; Kirkpatrick, M.; Thowfeek, S.; Munn, J.; Champion, H.; Mills, N.; Phillips, S.; et al. Tolerance, adherence, and acceptability of a ketogenic 2.5:1 ratio, nutritionally complete, medium chain triglyceride-containing liquid feed in children and adults with drug-resistant epilepsy following a ketogenic diet. Epilepsia Open 2024, 9, 727–738. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]


| Author/Year | Study Type | Population/Model | Diet/Intervention | Key Outcomes | AMPA Receptor Link | Limitations | MCT Dosage | Ketone Levels | Side Effect |
|---|---|---|---|---|---|---|---|---|---|
| Shin, 2025 [17] | Clinical (prospective) | Pediatric refractory epilepsy (n = 15) | Decanoic acid enriched KD | Reduced seizures in a significant subset. High decanoic acid levels correlate with improved seizure control. | Direct: Decanoic acid shown to inhibit AMPA receptors | High cost; potential poor adherence in some families | 4 g/100 mL | NA | Transient hypoglycemia and metabolic acidosis; hypercalciuria; transient gastrointestinal symptoms |
| Chang, 2013 [9] | Preclinical (rodent) | Rodent seizure model | MCT supplementation (lab diet) | Reduced seizure susceptibility in rodents Identified MCFAs’ capacity to modulate excitability | Noncompetitive inhibition of AMPA receptors | Animal study; translation to humans requires caution | 100 mM | NA | Sedaction |
| Chang, 2015 [10] | Preclinical (rodent) | Rodent seizure model | MCFA derivatives | Enhanced potency against seizures through chain structure modifications | Supports decanoic acid as partial AMPA antagonist | Synthetic derivatives may differ from dietary MCT formulations | 100–125 mg/kg | NA | Teratogenicity |
| Yelshanskaya, 2016 [11] | Structural in vitro | Human/rodent AMPA receptors (in vitro) | Drug binding studies (AMPA receptor) | Clarified architecture of noncompetitive inhibition; provided insight into how molecules (including decanoic acid analogs) may bind | Mechanistic blueprint for decanoic acid activity | Not an in vivo or dietary study | NA | NA | NA |
| Neal, 2009 [14] | Clinical (RCT) | Children with epilepsy (n = 145) | Classical KD vs. MCT-KD | Comparable seizure reduction; MCT-KD slightly more flexible with carbs | MCT-KD hypothesized to modulate AMPA via decanoic acid production | Adherence challenges; short to mid-term follow-up | started at 40–45% of energy, increased up to 60% if necessary | 0.696 ± 0.475 mmol/L (3 months) | Gastrointestinal |
| Lambrechts, 2015 [15] | Clinical (prospective) | Children with refractory epilepsy (2-year follow-up) | MCT-KD | Sustained seizure reduction; better tolerability vs. classic KD in some patients | Likely partial AMPA receptor modulation via decanoic acid | Non-randomized; potential selection bias | 10 g/die | 3.1 mmol/L (12 months) | Gastrointestinal, fatigue |
| Augustin, 2018 [12] | Preclinical (rodent) | Rodent seizure model and in vitro assays | Perampanel and decanoic acid | Synergistic anticonvulsant effect; enhanced blockade of AMPA receptors | Confirmed synergy with known AMPA antagonist (perampanel) | Lack of direct translational data to pediatric populations | ~157 μmol/L (plasma) | NA | NA |
| Henderson, 2006 [16] | Meta-analysis | Pediatric KD studies (various designs) | Classical KD and MCT-KD | KD (including MCT-KD) shown to be effective across multiple studies | Potential mechanism includes AMPA modulation by decanoic acid | Heterogeneous study designs; older references | NA | NA | Gastrointestinal, weight loss, irritability, increased serum cholesterol or triglyceride and liver anzymes, lethargy, hypercalciuria, renal stones, hypoglicemia |
| Berk, 2022 [13] | Clinical (observational) | Dogs with idiopathic epilepsy | Free diet + MCT | Reduced seizure frequency; metabolic fingerprints | Possibly relevant to AMPA receptors, but not directly tested | Veterinary study; extrapolation to humans limited | 9% of metabolic energy | 0.06 mmol/L | Not reported |
| Borges, 2019 [18] | Clinical (RCT) | Adults with DRE | Add-on triheptanoin vs. MCT | Demonstrated partial seizure frequency reduction; proof-of-concept for less restrictive approach | Decanoic acid among possible MCFA mediators | Adult population; short follow-up | 35% of energy | NA | Gastrointestinal, headache, disturbed sleep pattern |
| Schoeler, 2021 [19] | Clinical (feasibility) | Adults and adolescents with DRE | MCT as add-on | Feasibility and partial seizure control benefits; high inter-individual variability | Potential for AMPA receptor modulation via C10:0 and related MCFA | Non-randomized; open-label design | Adult: 240 mL; children: 19% daily energy (120 mL) | >1 mmol | Gastrointestinal |
| Rasmussen, 2022 [20] | Clinical (RCT) | Adults with DRE | Free diet + MCT | Seizure reduction | Potential mechanism includes AMPA modulation | Adult population, small cohort | 15 mL up to 60 mL daily (14 g of MCT/15 mL) | Presence in urine | Gastrointestinal |
| Feature | Classic Ketogenic Diet | MCT Supplementation |
|---|---|---|
| Efficacy | High in selected patients; often requires sustained ketosis | Moderate; some benefit even with low ketosis |
| Dietary flexibility | Very restrictive; low carbohydrate allowance | Less restrictive; more flexible food choices |
| Palatability | May be challenging for children | Improved palatability with oils or emulsion |
| Tolerability | Gastrointestinal side effects common | Generally well tolerated, but varies by dose |
| Adherence | Difficult to maintain long-term | Potentially better due to lower restriction |
| Aspect | Recommendation/Findings |
|---|---|
| MCT dosage (pediatric) | 0.5–1.5 g/kg/day (divided dose), based on tolerance and metabolic response |
| Duration | Minimum 3 months for efficacy assessment; some protocols extend to 12+ months |
| Administration form | Emulsified oil, flavored liquids, or incorporated into meals/snack |
| Common side effects | Gastrointestinal discomfort (bloating, diarrhea), lipid profile alterations |
| Monitoring parameters | Growth charts (weight, height), lipid panels, liver enzymes, ketone levels |
| Adherence tips | Use of palatable formulations, caregiver education, involvement of dietitians |
| Contraindications | Fat metabolism disorders, pancreatitis, certain liver diseases |
| Co-therapies | Can be combined with AEDs; interactions generally minimal, but monitoring is advised |
| Follow-up frequency | Monthly in the first 3 months; every 3–6 months thereafter |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Falsaperla, R.; Sortino, V.; Soler, M.A.; Spatuzza, M.; Fortuna, S.; Salpietro, V. AMPA Receptor Modulation Through Medium-Chain Triglycerides and Decanoic Acid Supports Nutritional Intervention in Pediatric Epilepsy. Nutrients 2025, 17, 1805. https://doi.org/10.3390/nu17111805
Falsaperla R, Sortino V, Soler MA, Spatuzza M, Fortuna S, Salpietro V. AMPA Receptor Modulation Through Medium-Chain Triglycerides and Decanoic Acid Supports Nutritional Intervention in Pediatric Epilepsy. Nutrients. 2025; 17(11):1805. https://doi.org/10.3390/nu17111805
Chicago/Turabian StyleFalsaperla, Raffaele, Vincenzo Sortino, Miguel Angel Soler, Michela Spatuzza, Sara Fortuna, and Vincenzo Salpietro. 2025. "AMPA Receptor Modulation Through Medium-Chain Triglycerides and Decanoic Acid Supports Nutritional Intervention in Pediatric Epilepsy" Nutrients 17, no. 11: 1805. https://doi.org/10.3390/nu17111805
APA StyleFalsaperla, R., Sortino, V., Soler, M. A., Spatuzza, M., Fortuna, S., & Salpietro, V. (2025). AMPA Receptor Modulation Through Medium-Chain Triglycerides and Decanoic Acid Supports Nutritional Intervention in Pediatric Epilepsy. Nutrients, 17(11), 1805. https://doi.org/10.3390/nu17111805





