Effects of a Personalized Diet on Nutritional Status and Renal Function Outcome in Nephrectomized Patients with Renal Cancer
Abstract
1. Introduction
2. Materials and Methods
2.1. Assessment of Nutritional Status
2.2. Statistical Analysis
3. Results
3.1. Clinical and Demographic Findings of the Patients
3.2. Overall Improvement in Anthropometric Indices following the Nephrological–Nutritional Combined Approach of an LNPHC Regimen for Nephrectomized Patients
3.3. Nephrological Clinical Parameters before and after LNPHC Diet
3.4. Statistical Correlations between Delta eGFR Values and Anthropometric Indices Using BIA
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Capitanio, U.; Montorsi, F. Does preoperative nutritional status affect survival in renal cell carcinoma? The debate continues. Eur. Urol. 2011, 59, 929–930. [Google Scholar] [CrossRef] [PubMed]
- Coca, S.G.; Singanamala, S.; Parikh, C.R. Chronic kidney disease after acute kidney injury: A systematic review and meta-analysis. Kidney Int. 2012, 81, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Hsu, R.K.; Hsu, C.Y. The Role of Acute Kidney Injury in Chronic Kidney Disease. Semin. Nephrol. 2016, 36, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Dalmartello, M.; Bravi, F.; Serraino, D.; Crispo, A.; Ferraroni, M.; La Vecchia, C.; Edefonti, V. Dietary Patterns in Italy and the Risk of Renal Cell Carcinoma. Nutrients 2020, 12, 134. [Google Scholar] [CrossRef] [PubMed]
- Levine, M.E.; Suarez, J.A.; Brandhorst, S.; Balasubramanian, P.; Cheng, C.W.; Madia, F.; Fontana, L.; Mirisola, M.G.; Guevara-Aguirre, J.; Wan, J.; et al. Low protein intake is associated with a major reduction in IGF-1, cancer, and overall mortality in the 65 and younger but not older population. Cell Metab. 2014, 19, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Kalantar-Zadeh, K.; Fouque, D. Nutritional Management of Chronic Kidney Disease. N. Engl. J. Med. 2017, 377, 1765–1776. [Google Scholar] [CrossRef] [PubMed]
- Arends, J.; Bachmann, P.; Baracos, V.; Barthelemy, N.; Bertz, H.; Bozzetti, F.; Fearon, K.; Hütterer, E.; Isenring, E.; Kaasa, S.; et al. ESPEN guidelines on nutrition in cancer patients. Clin. Nutr. 2017, 36, 11–48. [Google Scholar] [CrossRef] [PubMed]
- D’Alessandro, C.; Giannese, D.; Panichi, V.; Cupisti, A. Mediterranean Dietary Pattern Adjusted for CKD Patients: The MedRen Diet. Nutrients 2023, 15, 1256. [Google Scholar] [CrossRef]
- Bettiga, A.; Fiorio, F.; Liguori, F.; Marco, F.D.; Quattrini, G.; Vago, R.; Giannese, D.; Salonia, A.; Montorsi, F.; Trevisani, F. The Impact of a Mediterranean-like Diet with Controlled Protein Intake on the Onco-Nephrological Scenario: Time for a New Perspective. Nutrients 2022, 23, 5193. [Google Scholar] [CrossRef]
- Ljungberg, B.; Albiges, L.; Abu-Ghanem, Y.; Bedke, J.; Capitanio, U.; Dabestani, S.; Fernández-Pello, S.; Giles, R.H.; Hofmann, F.; Hora, M.; et al. EAU Guidelines. In Proceedings of the EAU Annual Congress, Milan, Italy, 10–13 March 2023; ISBN 978-94-92671-19-6. Available online: https://d56bochluxqnz.cloudfront.net/documents/full-guideline/EAU-Guidelines-on-Urological-infections-2023.pdf (accessed on 18 January 2024).
- Mickisch, G.; Carballido, J.; Hellsten, S.; Schulze, H.; Mensink, H. Guidelines on renal cell cancer. Eur. Urol. 2001, 40, 252–255. [Google Scholar] [CrossRef]
- Ljungberg, B.; Albiges, L.; Abu-Ghanem, Y.; Bedke, J.; Capitanio, U.; Dabestani, S.; Fernández-Pello, S.; Giles, R.H.; Hofmann, F.; Hora, M. European Association of Urology Guidelines on Renal Cell Carcinoma: The 2022 Update. Eur. Urol. 2022, 82, 399–410. [Google Scholar] [CrossRef] [PubMed]
- Kalantar-Zadeh, K.; Kramer, H.M.; Fouque, D. High-protein diet is bad for kidney health: Unleashing the taboo. Nephrol. Dial. Transplant. 2020, 35, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Chapman, D.; Moore, R.; Klarenbach, S.; Braam, B. Residual renal function after partial or radical nephrectomy for renal cell carcinoma. Can. Urol. Assoc. J. 2010, 4, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Ghavamian, R.; Cheville, J.C.; Lohse, C.M.; Weaver, A.L.; Zincke, H.; Blute, M.L. Renal cell carcinoma in the solitary kidney: An analysis of complications and outcome after nephron sparing surgery. J. Urol. 2002, 168, 454–459. [Google Scholar] [CrossRef] [PubMed]
- Provet, J.; Tessler, A.; Brown, J.; Golimbu, M.; Bosniak, M.; Morales, P. Partial nephrectomy for renal cell carcinoma: Indications, results and implications. J. Urol. 1991, 145, 472–476. [Google Scholar] [CrossRef] [PubMed]
- Minervini, A.; Grosso, A.A.; Di Maida, F. Partial Nephrectomy for Metastatic Renal Cell Carcinoma: Referee. Eur. Urol. Open Sci. 2022, 30, 78–80. [Google Scholar] [CrossRef] [PubMed]
- Porta, C.; Cosmai, L.; Leibovich, B.C.; Powles, T.; Gallieni, M.; Bex, A. The adjuvant treatment of kidney cancer: A multidisciplinary outlook. Nat. Rev. Nephrol. 2019, 15, 423–433. [Google Scholar] [CrossRef] [PubMed]
- Molina, A.M. A multidisciplinary approach for the management of earlier stage renal cell carcinoma. Urol. Oncol. 2018, 36, 15–16. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Zhu, S.; Wang, Z.; Chen, J.; Dai, J.; Liu, Z.; Sun, G.; Liang, J.; Zhang, X.; Wang, Z.; et al. Multidisciplinary Team (MDT) Discussion Improves Overall Survival Outcomes for Metastatic Renal Cell Carcinoma Patients. J. Multidiscip. Healthc. 2023, 16, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Cupisti, A.; Gallieni, M.; Avesani, C.M.; D’Alessandro, C.; Carrero, J.J.; Piccoli, G.B. Medical Nutritional Therapy for Patients with Chronic Kidney Disease not on Dialysis: The Low Protein Diet as a Medication. J. Clin. Med. 2020, 9, 3644. [Google Scholar] [CrossRef]
- Massini, G.; Caldiroli, L.; Molinari, P.; Carminati, F.M.I.; Castellano, G.; Vettoretti, S. Nutritional Strategies to Prevent Muscle Loss and Sarcopenia in Chronic Kidney Disease: What Do We Currently Know? Nutrients 2023, 15, 3107. [Google Scholar] [CrossRef]
- Naghshi, S.; Sadeghi, O.; Willett, W.C.; Esmaillzadeh, A. Dietary intake of total, animal, and plant proteins and risk of all cause, cardiovascular, and cancer mortality: Systematic review and dose-response meta-analysis of prospective cohort studies. BMJ 2020, 370, m2412. [Google Scholar] [CrossRef]
- Chauveau, P.; Aparicio, M.; Bellizzi, V.; Campbell, K.; Hong, X.; Johansson, L.; Kolko, A.; Molina, P.; Sezer, S.; Wanner, C.; et al. European Renal Nutrition (ERN) Working Group of the European Renal Association–European Dialysis Transplant Association (ERA-EDTA). Mediterranean diet as the diet of choice for patients with chronic kidney disease. Nephrol. Dial. Transplant. 2018, 33, 725–735. [Google Scholar] [CrossRef]
- Piercy, K.L.; Troiano, R.P.; Ballard, R.M.; Carlson, S.A.; Fulton, J.E.; Galuska, D.A.; George, S.M.; Olson, R.D. The Physical Activity Guidelines for Americans. JAMA 2018, 320, 2020–2028. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.; Robyak, K.; Zhu, Y. The CKD-EPI 2021 Equation and Other Creatinine-Based Race-Independent eGFR Equations in Chronic Kidney Disease Diagnosis and Staging. J. Appl. Lab. Med. 2023, 8, 952–961. [Google Scholar] [CrossRef] [PubMed]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B (Methodol.) 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Moore, L.W.; Byham-Gray, L.D.; Scott Parrott, J.; Rigassio-Radler, D.; Mandayam, S.; Jones, S.L.; Mitch, W.E.; Osama Gaber, A. The mean dietary protein intake at different stages of chronic kidney disease is higher than current guidelines. Kidney Int. 2013, 83, 724–732. [Google Scholar] [CrossRef]
- Vitale, M.; Giosuè, A.; Vaccaro, O.; Riccardi, G. Recent Trends in Dietary Habits of the Italian Population: Potential Impact on Health and the Environment. Nutrients 2021, 13, 476. [Google Scholar] [CrossRef] [PubMed]
- Dinu, M.; Pagliai, G.; Giangrandi, I.; Colombini, B.; Toniolo, L.; Gensini, G.; Sofi, F. Adherence to the Mediterranean diet among Italian adults: Results from the web-based Medi-Lite questionnaire. Int. J. Food Sci. Nutr. 2021, 72, 271–279. [Google Scholar] [CrossRef]
- Marventano, S.; Godos, J.; Platania, A.; Galvano, F.; Mistretta, A.; Grosso, G. Mediterranean diet adherence in the Mediterranean healthy eating, aging and lifestyle (MEAL) study cohort. Int. J. Food Sci. Nutr. 2018, 69, 100–107. [Google Scholar] [CrossRef]
- Vitale, M.; Racca, E.; Izzo, A.; Giacco, A.; Parente, E.; Riccardi, G.; Giacco, R. Adherence to the traditional Mediterranean diet in a population of South of Italy: Factors involved and proposal of an educational field-based survey tool. Int. J. Food Sci. Nutr. 2019, 70, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Iwasaka, C.; Yamada, Y.; Nishida, Y.; Hara, M.; Yasukata, J.; Miyoshi, N.; Shimanoe, C.; Nanri, H.; Furukawa, T.; Koga, K.; et al. Association of appendicular extracellular-to-intracellular water ratio with age, muscle strength, and physical activity in 8018 community-dwelling middle-aged and older adults. Arch. Gerontol. Geriatr. 2023, 108, 104931. [Google Scholar] [CrossRef]
- Lin, W.; Liu, M.; Wu, X.; Meng, S.; Yu, K.; Su, H.; Liang, Q.; Chen, F.; Li, J.; Xiao, W.; et al. Altered Cerebral Blood Flow in the Progression of Chronic Kidney Disease. J. Pers. Med. 2023, 13, 142. [Google Scholar] [CrossRef] [PubMed]
- Di Marco, F.; Pani, A.; Floris, M.; Martini, A.; Dell’Antonio, G.; Capitanio, U.; Bettiga, A.; Larcher, A.; Cinque, A.; Bertini, R.; et al. Unexpected Outcomes of Renal Function after Radical Nephrectomy: Histology Relevance along with Clinical Aspects. J. Clin. Med. 2021, 10, 3322. [Google Scholar] [CrossRef] [PubMed]
- Capitanio, U.; Montorsi, F. Renal cancer. Lancet 2016, 387, 894–906. [Google Scholar] [CrossRef] [PubMed]
- Capitanio, U.; Larcher, A.; Terrone, C.; Antonelli, A.; Volpe, A.; Fiori, C.; Furlan, M.; Dehò, F.; Minervini, A.; Serni, S.; et al. End-Stage Renal Disease After Renal Surgery in Patients with Normal Preoperative Kidney Function: Balancing Surgical Strategy and Individual Disorders at Baseline. Eur. Urol. 2016, 70, 558–561. [Google Scholar] [CrossRef] [PubMed]
- Capitanio, U.; Larcher, A.; Kriegmair, M.C.; Bertolo, R.; Salagierski, M.; Campi, R.; Klatte, T.; Trevisani, F.; Montorsi, F.; Mir, M.C.; et al. Kidney Cancer Working Group of the Young Academic Urologists Working Party of the European Association of Urology. Do We Truly Care About the Functional Outcomes for Renal Cancer Patients? Multidisciplinarity Is Still Far Away. Eur. Urol. 2019, 75, 349–350. [Google Scholar] [CrossRef] [PubMed]
- Rymarz, A.; Szamotulska, K.; Niemczyk, S. Comparison of Skinfold Thicknesses and Bioimpedance Spectroscopy to Dual-Energy X-Ray Absorptiometry for the Body Fat Measurement in Patients With Chronic Kidney Disease. Nutr. Clin. Pract. 2017, 32, 533–538. [Google Scholar] [CrossRef]
- Seltzer, C.C.; Goldman, R.F.; Mayer, J. The triceps skinfold as a predictive measure of body density and body fat in obese adolescent girls. Pediatrics 1965, 36, 212–218. [Google Scholar] [CrossRef]
- Agarwal, R.; Bills, J.E.; Light, R.P. Diagnosing obesity by body mass index in chronic kidney disease: An explanation for the “obesity paradox?”. Hypertension 2010, 56, 893–900. [Google Scholar] [CrossRef]
- Xu, X.; Eales, J.M.; Jiang, X.; Sanderson, E.; Drzal, M.; Saluja, S.; Scannali, D.; Williams, B.; Morris, A.P.; Guzik, T.J.; et al. Contributions of obesity to kidney health and disease: Insights from Mendelian randomization and the human kidney transcriptomics. Cardiovasc. Res. 2022, 118, 3151–3161. [Google Scholar] [CrossRef] [PubMed]
- Nam, G.E.; Cho, K.H.; Han, K.; Kim, C.M.; Han, B.; Cho, S.J.; Jung, S.J.; Kwon, Y.; Kim, Y.H.; Kim, D.H.; et al. Obesity, abdominal obesity and subsequent risk of kidney cancer: A cohort study of 23.3 million East Asians. Br. J. Cancer. 2019, 121, 271–277. [Google Scholar] [CrossRef]
- Yu, Z.; Grams, M.E.; Ndumele, C.E.; Wagenknecht, L.; Boerwinkle, E.; North, K.E.; Rebholz, C.M.; Giovannucci, E.L.; Coresh, J. Association Between Midlife Obesity and Kidney Function Trajectories: The Atherosclerosis Risk in Communities (ARIC) Study. Am. J. Kidney Dis. 2021, 77, 376–385. [Google Scholar] [CrossRef] [PubMed]
- Brenner, B.M.; Lawler, E.V.; Mackenzie, H.S. The hyperfiltration theory: A paradigm shift in nephrology. Kidney Int. 1996, 49, 1774–1777. [Google Scholar] [CrossRef]
- D’Agati, V.D.; Chagnac, A.; de Vries, A.P.; Levi, M.; Porrini, E.; Herman-Edelstein, M.; Praga, M. Obesity-related glomerulopathy: Clinical and pathologic characteristics and pathogenesis. Nat. Rev. Nephrol. 2016, 12, 453–471. [Google Scholar] [CrossRef]
- Aurilio, G.; Piva, F.; Santoni, M.; Cimadamore, A.; Sorgentoni, G.; Lopez-Beltran, A.; Cheng, L.; Battelli, N.; Nolè, F.; Montironi, R. The Role of Obesity in Renal Cell Carcinoma Patients: Clinical-Pathological Implications. Int. J. Mol. Sci. 2019, 20, 5683. [Google Scholar] [CrossRef]
- VanItallie, T.B.; Yang, M.U.; Heymsfield, S.B.; Funk, R.C.; Boileau, R.A. Height-normalized indices of the body’s fat-free mass and fat mass: Potentially useful indicators of nutritional status. Am. J. Clin. Nutr. 1990, 52, 953–959. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, R.; Tanisawa, K.; Ito, T.; Usui, C.; Miyachi, M.; Torii, S.; Midorikawa, T.; Ishii, K.; Muraoka, I.; Suzuki, K.; et al. Fat-Free Mass Index as a Surrogate Marker of Appendicular Skeletal Muscle Mass Index for Low Muscle Mass Screening in Sarcopenia. J. Am. Med. Dir. Assoc. 2022, 23, 1955–1961.e3. [Google Scholar] [CrossRef]
- Moriconi, D.; D’Alessandro, C.; Giannese, D.; Panichi, V.; Cupisti, A. Diagnosis, Prevalence and Significance of Obesity in a Cohort of CKD Patients. Metabolites 2023, 13, 196. [Google Scholar] [CrossRef]
- Wu, L.W.; Lin, Y.Y.; Kao, T.W.; Lin, C.M.; Liaw, F.Y.; Wang, C.C.; Peng, T.C.; Chen, W.L. Mid-arm muscle circumference as a significant predictor of all-cause mortality in male individuals. PLoS ONE 2017, 12, e0171707. [Google Scholar] [CrossRef]
- Noori, N.; Kopple, J.D.; Kovesdy, C.P.; Feroze, U.; Sim, J.J.; Murali, S.B.; Luna, A.; Gomez, M.; Luna, C.; Bross, R.; et al. Mid-arm muscle circumference and quality of life and survival in maintenance hemodialysis patients. Clin. J. Am. Soc. Nephrol. 2010, 5, 2258–2268. [Google Scholar] [CrossRef] [PubMed]
- Gibney, M.J.; Ljungqvist, J.E.M. Clinical Nutrition: Anthropometric Assessment of Body Composition, 3rd ed.; Blackwell: Oxford, UK, 2005; p. 20. [Google Scholar]
- Cano, N.J.; Miolane-Debouit, M.; Léger, J.; Heng, A.E. Assessment of body protein: Energy status in chronic kidney disease. Semin. Nephrol. 2009, 29, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Viggiano, D.; Wagner, C.A.; Martino, G.; Nedergaard, M.; Zoccali, C.; Unwin, R.; Capasso, G. Mechanisms of cognitive dysfunction in CKD. Nat. Rev. Nephrol. 2020, 16, 452–469. [Google Scholar] [CrossRef] [PubMed]
- Yan, B.; Su, X.; Xu, B.; Qiao, X.; Wang, L. Effect of diet protein restriction on progression of chronic kidney disease: A systematic review and meta-analysis. PLoS ONE 2018, 13, e0206134. [Google Scholar] [CrossRef]
| Characteristics | Number |
|---|---|
| Number of patients | 40 |
| Age, median (range) | 64 (44–86) |
| Gender, male (%) | 32 (80) |
| Gender, female (%) | 8 (20) |
| BMI (range) | 25.8 (23.1–28.7) |
| Underweight (≤18.5) (%) | 0 (0) |
| Healthy weight (>18.6 ≤ 24.9) (%) | 16 (40.0) |
| Overweight (>24.9 ≤ 30) (%) | 16 (40.0) |
| Obese (>30) (%) | 8 (20.0) |
| Diabetes mellitus (%) | 7 (17.5) |
| Hypertension (%) | 22 (55.0) |
| CKD stage | |
| CKD-I (%) | 1 (2.5) |
| CKD-II (%) | 6 (15.0) |
| CKD-IIIA (%) | 18 (45) |
| CKD-IIIB (%) | 5 (12.5) |
| CKD-IV (%) | 10 (25.0) |
| CKD-V (%) | 0 (0) |
| Variables | Before Dietary (Visit 1) Median [Range] | After Dietary (Visit 2) Median [Range] | p-Value |
|---|---|---|---|
| PA | 6.2° [4.1–8.7] | 6.3° [4.4–8.8] | 0.2530 |
| ECM/BCM | 0.81 [0.6–1.4] | 0.78 [0.6–1.3] | 0.0182 * |
| BCM/h2 | 12.2 kg/m2 [6.9–16.8] | 12.5 kg/m2 [6.8–16.0] | 0.4845 |
| BMI | 25.8 kg/m2 [19.7–35.4] | 25.1 kg/m2 [19.6–36.4] | 0.0503 |
| Waist Circumference | 92.0 cm [74.0–133.0] | 90.0 cm [68.0–120.0] | 0.0129 * |
| Waist–Hip Ratio | 0.93 [0.68–1.3] | 0.91 [0.68–1.1] | 0.2464 |
| Skinfold Thickness | 15 mm [5.0–32.0] | 13 mm [5.0–27.0] | 0.0390 * |
| MAMC | 26.3 (23.3–27.6) | 26.8 (24.2–28.5) | 0.0016 ** |
| ECW/ICW | 0.84 [0.57–1.3] | 0.82 [0.51–1.1] | 0.0178 * |
| TBW % | 59.6 [42.7–67.1] | 61.9 [41.2–67.1] | 0.0104 * |
| FM/h2 | 4.7 kg/m2 [1.4–14.1] | 3.7 kg/m2 [1.5–14.9] | 0.0044 ** |
| FFM/h2 | 21.2 kg/m2 [16.3–27.4] | 21.3 kg/m2 [13.3–28.2] | 0.5960 |
| Before Dietary Intervention | After Dietary Intervention | p Value | |
|---|---|---|---|
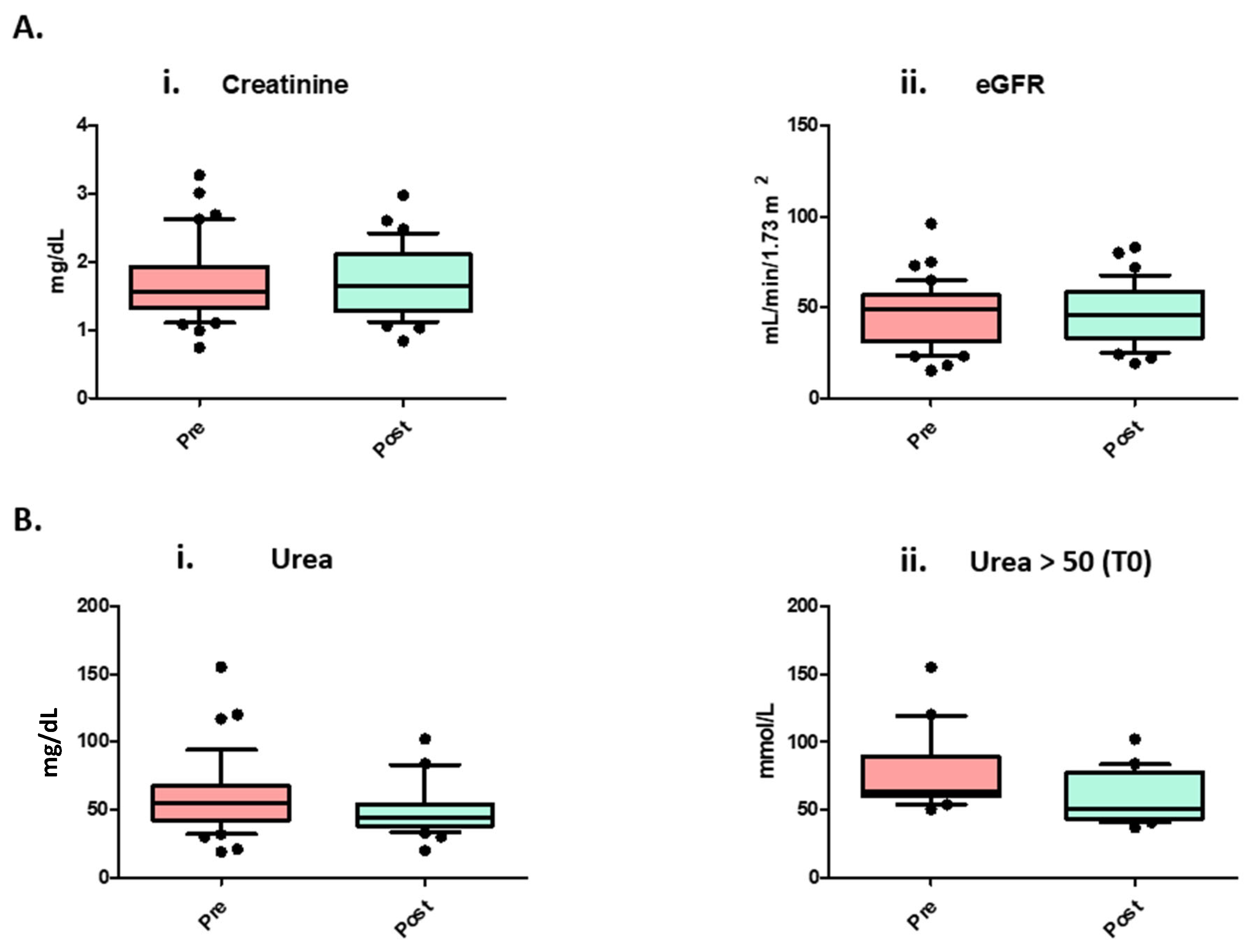
| Creatinine | 1.6 [0.7–3.3] | 1.7 [0.8–3.0] | 0.6847 |
| eGFR (EPI 2021) | 48.5 [15.0–96.0] | 46.0 [19.0–83.0] | 0.9812 |
| Urea | 55 [19–155] | 44 [20–102] | 0.0006 ** |
| Urea > 50 mg/dL (To) | 64 [50.5–155] | 51 [37–102] | 0.0001 ** |
| Correlations | |||||||
|---|---|---|---|---|---|---|---|
| Delta CDK-EPI 2021 | MAMC T1 | ECW/ICW T1 | FM/h2 T1 | ECM/BCM T1 | TBW% T1 | ||
| Delta CDK-EPI 2021 | PC | 1 | −0.020 | −0.264 | −0.170 | −0.216 | 0.190 |
| S2T | / | 0.911 | 0.115 | 0.315 | 0.205 | 0.266 | |
| MAMC T1 | PC | −0.020 | 1 | 0.031 | 0.182 | 0.222 | −0.140 |
| S2T | 0.911 | / | 0.864 | 0.302 | 0.214 | 0.436 | |
| ECW/ICW T1 | PC | −0.264 | 0.031 | 1 | 0.523 ** | 0.855 ** | −0.586 ** |
| S2T | 0.115 | 0.864 | / | 0.001 | 0.000 | 0.000 | |
| FM/h2 T1 | PC | −0.170 | 0.182 | 0.523 ** | 1 | 0.481 ** | −0.960 ** |
| S2T | 0.315 | 0.302 | 0.001 | / | 0.002 | 0.000 | |
| ECM/BCM T1 | PC | −0.216 | 0.222 | 0.855 ** | 0.481 ** | 1 | −0.600 ** |
| S2T | 0.205 | 0.214 | 0.000 | 0.002 | / | 0.000 | |
| TBW % T1 | PC | 0.190 | −0.140 | −0.586 ** | −0.960 ** | −0.600 ** | 1 |
| S2T | 0.266 | 0.436 | 0.000 | 0.000 | 0.000 | / | |
| Univariate Tests | |||||||
|---|---|---|---|---|---|---|---|
| Sum of Squares | df | Mean Square | F | Sig. | Partial Eta Squared | ||
| Delta ECM/BCM | Contrast | 0.003 | 1 | 0.003 | 0.885 | 0.354 | 0.028 |
| Error | 0.089 | 31 | 0.003 | ||||
| Delta ECW/ICW | Contrast | 0.013 | 1 | 0.013 | 5.174 | 0.030 | 0.143 |
| Error | 0.077 | 31 | 0.002 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trevisani, F.; Laurenti, F.; Fiorio, F.; Paccagnella, M.; Floris, M.; Capitanio, U.; Ghidini, M.; Garrone, O.; Abbona, A.; Salonia, A.; et al. Effects of a Personalized Diet on Nutritional Status and Renal Function Outcome in Nephrectomized Patients with Renal Cancer. Nutrients 2024, 16, 1386. https://doi.org/10.3390/nu16091386
Trevisani F, Laurenti F, Fiorio F, Paccagnella M, Floris M, Capitanio U, Ghidini M, Garrone O, Abbona A, Salonia A, et al. Effects of a Personalized Diet on Nutritional Status and Renal Function Outcome in Nephrectomized Patients with Renal Cancer. Nutrients. 2024; 16(9):1386. https://doi.org/10.3390/nu16091386
Chicago/Turabian StyleTrevisani, Francesco, Fabiana Laurenti, Francesco Fiorio, Matteo Paccagnella, Matteo Floris, Umberto Capitanio, Michele Ghidini, Ornella Garrone, Andrea Abbona, Andrea Salonia, and et al. 2024. "Effects of a Personalized Diet on Nutritional Status and Renal Function Outcome in Nephrectomized Patients with Renal Cancer" Nutrients 16, no. 9: 1386. https://doi.org/10.3390/nu16091386
APA StyleTrevisani, F., Laurenti, F., Fiorio, F., Paccagnella, M., Floris, M., Capitanio, U., Ghidini, M., Garrone, O., Abbona, A., Salonia, A., Montorsi, F., & Bettiga, A. (2024). Effects of a Personalized Diet on Nutritional Status and Renal Function Outcome in Nephrectomized Patients with Renal Cancer. Nutrients, 16(9), 1386. https://doi.org/10.3390/nu16091386








