A Prospective, Observational Study of the Effect of a High-Calorie, High-Protein Oral Nutritional Supplement with HMB in an Old and Malnourished or at-Risk-of-Malnutrition Population with Hip Fractures: A FracNut Study
Abstract
1. Introduction
2. Subjects and Methods
2.1. Inclusion Criteria
2.2. Exclusion Criteria
2.3. Nutritional Intervention
2.4. Study Variables
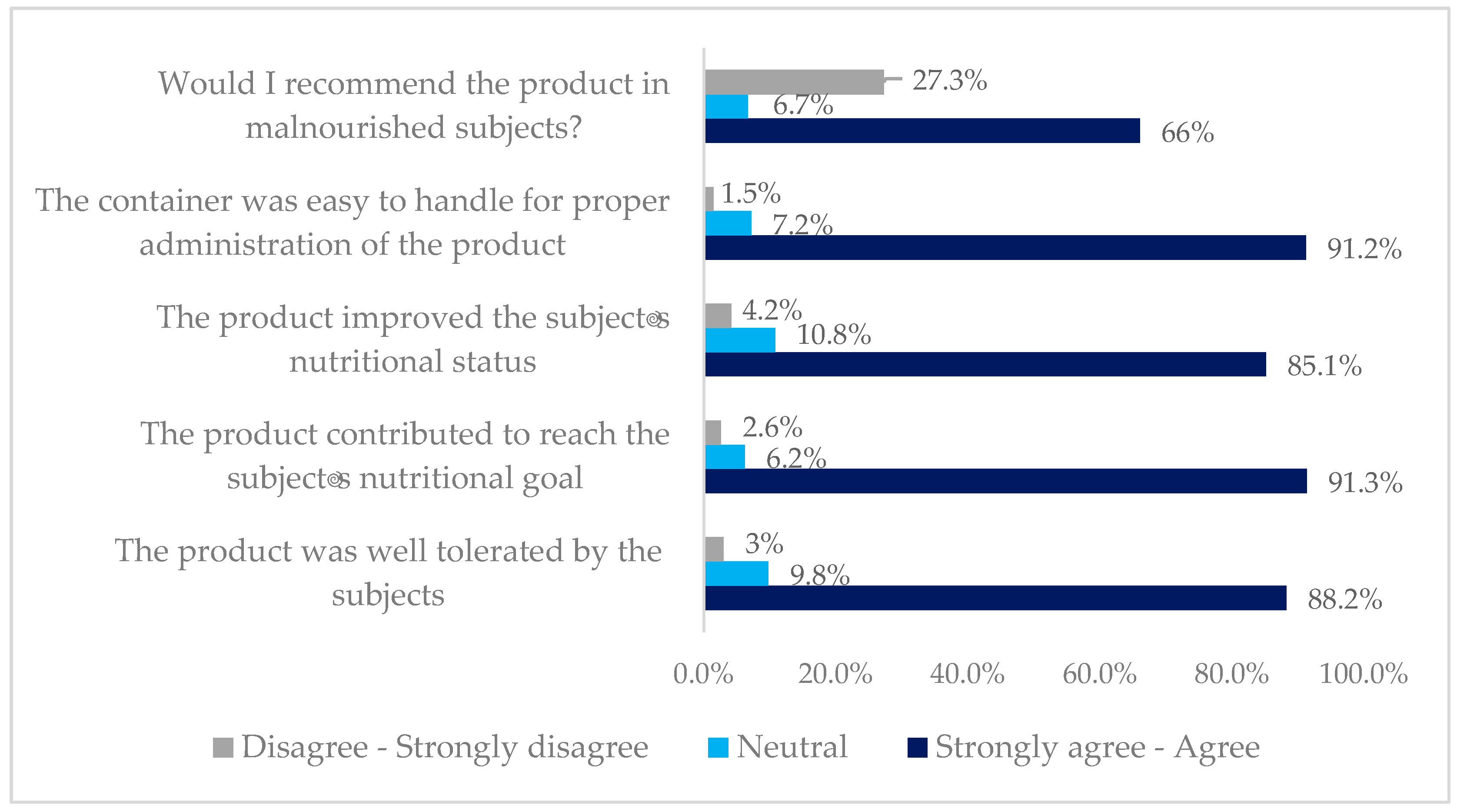
3. Results
4. Discussion
5. Limitations and Strengths
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Correction Statement
References
- Woolf, A.D.; Pfleger, B. Burden of major musculoskeletal conditions. Bull. World Health Organ. 2003, 81, 646–656. [Google Scholar]
- Kanis, J.A.; Oden, A.; McCloskey, E.V.; Johansson, H.; Wahl, D.A.; Cooper, C.; on behalf of the IOF Working Group on Epidemiology and Quality of Life. A systematic review of hip fracture incidence and probability of fracture worldwide. Osteoporos. Int. 2012, 23, 2239–2256. [Google Scholar] [CrossRef]
- Bardales Mas, Y.; Gonzalez Montalvo, J.I.; Abizanda Soler, P.; Alarcon Alarcon, M.T. Hip fracture guidelines. A comparison of the main recommendations. Rev. Esp. Geriatr. Gerontol. 2012, 47, 220–227. [Google Scholar] [CrossRef]
- Li, L.; Bennett-Brown, K.; Morgan, C.; Dattani, R. Hip fractures. Br. J. Hosp. Med. 2020, 81, 1–10. [Google Scholar] [CrossRef]
- Veronese, N.; Maggi, S. Epidemiology and social costs of hip fracture. Injury 2018, 49, 1458–1460. [Google Scholar] [CrossRef]
- Perez Durillo, F.T.; Torio Durantez, J.; Villarejo Villar, A.B.; Sanchez Vico, A.B.; Cueto Camarero Mdel, M.; Durillo, J.P. Comparative study of dietary intake and nutritional status in elderly women with and without hip fracture. Aten. Primaria 2011, 43, 362–368. [Google Scholar]
- Curtis, E.; Litwic, A.; Cooper, C.; Dennison, E. Determinants of Muscle and Bone Aging. J. Cell Physiol. 2015, 230, 2618–2625. [Google Scholar] [CrossRef]
- Wunderle, C.; Gomes, F.; Schuetz, P.; Stumpf, F.; Austin, P.; Ballesteros-Pomar, M.D.; Cederholm, T.; Fletcher, J.; Laviano, A.; Norman, K.; et al. ESPEN guideline on nutritional support for polymorbid medical inpatients. Clin. Nutr. 2023, 42, 1545–1568. [Google Scholar] [CrossRef]
- Fitschen, P.J.; Wilson, G.J.; Wilson, J.M.; Wilund, K.R. Efficacy of beta-hydroxy-beta-methylbutyrate supplementation in elderly and clinical populations. Nutrition 2013, 29, 29–36. [Google Scholar] [CrossRef]
- Kuriyan, R.; Lokesh, D.P.; Selvam, S.; Jayakumar, J.; Philip, M.G.; Shreeram, S.; Kurpad, A.V. The relationship of endogenous plasma concentrations of beta-Hydroxy beta-Methyl Butyrate (HMB) to age and total appendicular lean mass in humans. Exp. Gerontol. 2016, 81, 13–18. [Google Scholar] [CrossRef]
- Rizzoli, R.; Stevenson, J.C.; Bauer, J.M.; van Loon, L.J.; Walrand, S.; Kanis, J.A.; Cooper, C.; Brandi, M.-L.; Diez-Perez, A.; Reginster, J.-Y. The role of dietary protein and vitamin D in maintaining musculoskeletal health in postmenopausal women: A consensus statement from the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO). Maturitas 2014, 79, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Rizzoli, R.; Boonen, S.; Brandi, M.L.; Bruyere, O.; Cooper, C.; Kanis, J.A.; Kaufman, J.-M.; Ringe, J.D.; Weryha, G.; Reginster, J.-Y. Vitamin D supplementation in elderly or postmenopausal women: A 2013 update of the 2008 recommendations from the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO). Curr. Med. Res. Opin. 2013, 29, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Eriksen, E.F.; Glerup, H. Vitamin D deficiency and aging: Implications for general health and osteoporosis. Biogerontology 2002, 3, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Houston, D.K.; Tooze, J.A.; Garcia, K.; Visser, M.; Rubin, S.; Harris, T.B.; Newman, A.B.; Kritchevsky, S.B.; the Health ABC Study. Protein Intake and Mobility Limitation in Community-Dwelling Older Adults: The Health ABC Study. J. Am. Geriatr. Soc. 2017, 65, 1705–1711. [Google Scholar] [CrossRef] [PubMed]
- Gerdhem, P.; Ringsberg, K.A.; Obrant, K.J.; Akesson, K. Association between 25-hydroxy vitamin D levels, physical activity, muscle strength and fractures in the prospective population-based OPRA Study of Elderly Women. Osteoporos. Int. 2005, 16, 1425–1431. [Google Scholar] [CrossRef] [PubMed]
- Morley, J.E.; Argiles, J.M.; Evans, W.J.; Bhasin, S.; Cella, D.; Deutz, N.E.; Doehner, W.; Fearon, K.C.; Ferrucci, L.; Hellerstein, M.K.; et al. Nutritional recommendations for the management of sarcopenia. J. Am. Med. Dir. Assoc. 2010, 11, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Bjelakovic, G.; Gluud, L.L.; Nikolova, D.; Whitfield, K.; Krstic, G.; Wetterslev, J.; Gluud, C. Vitamin D supplementation for prevention of cancer in adults. Cochrane Database Syst. Rev. 2014, 6, CD007469. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Fang, F.; Tang, J.; Jia, L.; Feng, Y.; Xu, P.; Faramand, A. Association between vitamin D supplementation and mortality: Systematic review and meta-analysis. BMJ 2019, 366, l4673. [Google Scholar] [CrossRef] [PubMed]
- Bouillon, R.; Manousaki, D.; Rosen, C.; Trajanoska, K.; Rivadeneira, F.; Richards, J.B. The health effects of vitamin D supplementation: Evidence from human studies. Nat. Rev. Endocrinol. 2022, 18, 96–110. [Google Scholar] [CrossRef]
- Vellas, B.; Villars, H.; Abellan, G.; Soto, M.E.; Rolland, Y.; Guigoz, Y.; Garry, P. Overview of the MNA—Its history and challenges. J. Nutr. Health Aging 2006, 10, 456–463; discussion 463–465. [Google Scholar]
- Mahoney, F.I.; Barthel, D.W. Functional Evaluation: The Barthel Index. Md. State Med. J. 1965, 14, 61–65. [Google Scholar] [PubMed]
- Guralnik, J.M.; Simonsick, E.M.; Ferrucci, L.; Glynn, R.J.; Berkman, L.F.; Blazer, D.G.; Scherr, P.A.; Wallace, R.B. A short physical performance battery assessing lower extremity function: Association with self-reported disability and prediction of mortality and nursing home admission. J. Gerontol. 1994, 49, M85–M94. [Google Scholar] [CrossRef] [PubMed]
- Subra, J.; Gillette-Guyonnet, S.; Cesari, M.; Oustric, S.; Vellas, B.; Platform, T. The integration of frailty into clinical practice: Preliminary results from the Gerontopole. J. Nutr. Health Aging 2012, 16, 714–720. [Google Scholar] [CrossRef] [PubMed]
- Jebb, A.T.; Ng, V.; Tay, L. A Review of Key Likert Scale Development Advances: 1995–2019. Front. Psychol. 2021, 12, 637547. [Google Scholar] [CrossRef] [PubMed]
- Alexiou, K.I.; Roushias, A.; Varitimidis, S.E.; Malizos, K.N. Quality of life and psychological consequences in elderly patients after a hip fracture: A review. Clin. Interv. Aging 2018, 13, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Peeters, C.M.; Visser, E.; Van de Ree, C.L.; Gosens, T.; Den Oudsten, B.L.; De Vries, J. Quality of life after hip fracture in the elderly: A systematic literature review. Injury 2016, 47, 1369–1382. [Google Scholar] [CrossRef] [PubMed]
- Koren-Hakim, T.; Weiss, A.; Hershkovitz, A.; Otzrateni, I.; Grosman, B.; Frishman, S.; Salai, M.; Beloosesky, Y. The relationship between nutritional status of hip fracture operated elderly patients and their functioning, comorbidity and outcome. Clin. Nutr. 2012, 31, 917–921. [Google Scholar] [CrossRef]
- Frandsen, C.F.; Glassou, E.N.; Stilling, M.; Hansen, T.B. Malnutrition, poor function and comorbidities predict mortality up to one year after hip fracture: A cohort study of 2800 patients. Eur. Geriatr. Med. 2022, 13, 433–443. [Google Scholar] [CrossRef]
- Malafarina, V.; Reginster, J.Y.; Cabrerizo, S.; Bruyère, O.; Kanis, J.A.; Martinez, J.A.; Zulet, M.A. Nutritional Status and Nutritional Treatment Are Related to Outcomes and Mortality in Older Adults with Hip Fracture. Nutrients 2018, 10, 555. [Google Scholar] [CrossRef]
- Avenell, A.; Smith, T.O.; Curtain, J.P.; Mak, J.C.; Myint, P.K. Nutritional supplementation for hip fracture aftercare in older people. Cochrane Database Syst. Rev. 2016, 11, CD001880. [Google Scholar] [CrossRef]
- Arkley, J.; Dixon, J.; Wilson, F.; Charlton, K.; Ollivere, B.J.; Eardley, W. Assessment of Nutrition and Supplementation in Patients With Hip Fractures. Geriatr. Orthop. Surg. Rehabil. 2019, 10, 2151459319879804. [Google Scholar] [CrossRef] [PubMed]
- Myint, M.W.; Wu, J.; Wong, E.; Chan, S.P.; To, T.S.J.; Chau, M.W.R.; Ting, K.H.; Fung, P.M.; Au, K.S.D. Clinical benefits of oral nutritional supplementation for elderly hip fracture patients: A single blind randomised controlled trial. Age Ageing 2013, 42, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.G.A.; Ohnuma, T.; Haines, K.L.; Krishnamoorthy, V.; Raghunathan, K.; Sulo, S.; Cassady, B.A.; Hegazi, R.; Wischmeyer, P.E. Association between early postoperative nutritional supplement utilisation and length of stay in malnourished hip fracture patients. Br. J. Anaesth. 2021, 126, 730–737. [Google Scholar] [CrossRef] [PubMed]
- Gunnarsson, A.K.; Lonn, K.; Gunningberg, L. Does nutritional intervention for patients with hip fractures reduce postoperative complications and improve rehabilitation? J. Clin. Nurs. 2009, 18, 1325–1333. [Google Scholar] [CrossRef]
- Volkert, D.; Berner, Y.N.; Berry, E.; Cederholm, T.; Bertrand, P.C.; Milne, A.; Palmblad, J.; Schneider, S.; Sobotka, L.; Stanga, Z.; et al. ESPEN Guidelines on Enteral Nutrition: Geriatrics. Clin. Nutr. 2006, 25, 330–360. [Google Scholar] [CrossRef] [PubMed]
- Guigoz, Y.; Vellas, B. Nutritional Assessment in Older Adults: MNA(R) 25 years of a Screening Tool and a Reference Standard for Care and Research; What Next? J. Nutr. Health Aging 2021, 25, 528–583. [Google Scholar] [CrossRef] [PubMed]
- Nishioka, S.; Wakabayashi, H.; Momosaki, R. Nutritional Status Changes and Activities of Daily Living after Hip Fracture in Convalescent Rehabilitation Units: A Retrospective Observational Cohort Study from the Japan Rehabilitation Nutrition Database. J. Acad. Nutr. Diet. 2018, 118, 1270–1276. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Zhang, J.; Zheng, H.; Wang, X.; Liu, Z.; Sun, T. Prognostic Role of Serum Albumin, Total Lymphocyte Count, and Mini Nutritional Assessment on Outcomes after Geriatric Hip Fracture Surgery: A Meta-Analysis and Systematic Review. J Arthroplast. 2019, 34, 1287–1296. [Google Scholar] [CrossRef] [PubMed]
- Wengstrom, Y.; Wahren, L.K.; Grodzinsky, E. Importance of dietary advice, nutritional supplements and compliance for maintaining body weight and body fat after hip fracture. J. Nutr. Health Aging 2009, 13, 632–638. [Google Scholar] [CrossRef]
- Hubbard, G.P.; Elia, M.; Holdoway, A.; Stratton, R.J. A systematic review of compliance to oral nutritional supplements. Clin. Nutr. 2012, 31, 293–312. [Google Scholar] [CrossRef]
- Bruce, D.; Laurance, I.; McGuiness, M.; Ridley, M.; Goldswain, P. Nutritional supplements after hip fracture: Poor compliance limits effectiveness. Clin. Nutr. 2003, 22, 497–500. [Google Scholar] [CrossRef] [PubMed]
- Deutz, N.E.; Matheson, E.M.; Matarese, L.E.; Luo, M.; Baggs, G.E.; Nelson, J.L.; Hegazi, R.A.; Tappenden, K.A.; Ziegler, T.R. Readmission and mortality in malnourished, older, hospitalized adults treated with a specialized oral nutritional supplement: A randomized clinical trial. Clin. Nutr. 2016, 35, 18–26. [Google Scholar] [CrossRef] [PubMed]
- de Luis, D.A.; Izaola, O.; Lopez, L.; Blanco, B.; Colato, C.A.; Kelly, O.J.; Sanz, R. AdNut study: Effectiveness of a high calorie and protein oral nutritional supplement with beta-hydroxy-beta-methylbutyrate in an older malnourished population in usual clinical practice. Eur. Geriatr. Med. 2018, 9, 809–817. [Google Scholar] [CrossRef]
- Habibi Ghahfarrokhi, S.; Mohammadian-Hafshejani, A.; Sherwin, C.M.T.; Heidari-Soureshjani, S. Relationship between serum vitamin D and hip fracture in the elderly: A systematic review and meta-analysis. J. Bone Min. Metab. 2022, 40, 541–553. [Google Scholar] [CrossRef] [PubMed]
- Dauny, V.; Thietart, S.; Cohen-Bittan, J.; Riou, B.; Khiami, F.; Meziere, A.; Verny, M.; Boddaert, J.; Zerah, L.; Villain, C. Association between Vitamin D Deficiency and Prognosis after Hip Fracture Surgery in Older Patients in a Dedicated Orthogeriatric Care Pathway. J. Nutr. Health Aging 2022, 26, 324–331. [Google Scholar] [CrossRef] [PubMed]
- Sprague, S.; Slobogean, G.P.; Bogoch, E.; Petrisor, B.; Garibaldi, A.; O’Hara, N.; Bhandari, M.; FAITH Investigators. Vitamin D Use and Health Outcomes after Surgery for Hip Fracture. Orthopedics 2017, 40, e868–e875. [Google Scholar] [CrossRef] [PubMed]
- Pass, B.; Malek, F.; Rommelmann, M.; Aigner, R.; Knauf, T.; Eschbach, D.; Hussmann, B.; Maslaris, A.; Lendemans, S.; Schoeneberg, C. The Influence of Malnutrition Measured by Hypalbuminemia and Body Mass Index on the Outcome of Geriatric Patients with a Fracture of the Proximal Femur. Medicina 2022, 58, 1610. [Google Scholar] [CrossRef]
- O’Daly, B.J.; Walsh, J.C.; Quinlan, J.F.; Falk, G.A.; Stapleton, R.; Quinlan, W.R.; O’Rourke, S.K. Serum albumin and total lymphocyte count as predictors of outcome in hip fractures. Clin. Nutr. 2010, 29, 89–93. [Google Scholar] [CrossRef]
- Chen, Y.H.; Chou, C.H.; Su, H.H.; Tsai, Y.-T.; Chiang, M.-H.; Kuo, Y.-J. Correlation between neutrophil-to-lymphocyte ratio and postoperative mortality in elderly patients with hip fracture: A meta-analysis. J. Orthop. Surg. Res. 2021, 16, 681. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, H.; Yang, L.; Jiang, W.; Chen, X.; Liu, Y. High platelet-to-lymphocyte ratio predicts poor survival of elderly patients with hip fracture. Int. Orthop. 2021, 45, 13–21. [Google Scholar] [CrossRef]
- Pareja Sierra, T.; Bartolome Martin, I.; Rodriguez Solis, J.; Sanz, M.D.M.; de Suso, M.T.G.; Goitiandia, L.Á.B.; Calvo, M.H. Results of an anaemia treatment protocol complementary to blood transfusion in elderly patients with hip fracture. Rev. Esp. Geriatr. Gerontol. 2019, 54, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Elite Learning. The Original Barthel Index of ADLs. Available online: https://www.elitecme.com/resource-center/rehabilitation-therapy/the-original-barthel-index-of-adls/ (accessed on 30 October 2023).
- Malafarina, V.; Uriz-Otano, F.; Malafarina, C.; Martinez, J.A.; Zulet, M.A. Effectiveness of nutritional supplementation on sarcopenia and recovery in hip fracture patients. A multi-centre randomized trial. Maturitas 2017, 101, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Western, M.J.; Malkowski, O.S. Associations of the Short Physical Performance Battery (SPPB) with Adverse Health Outcomes in Older Adults: A 14-Year Follow-Up from the English Longitudinal Study of Ageing (ELSA). Int. J. Environ. Res. Public. Health 2022, 19, 16319. [Google Scholar] [CrossRef] [PubMed]
- Beckmann, M.; Bruun-Olsen, V.; Pripp, A.H.; Bergland, A.; Smith, T.; Heiberg, K.E. Recovery and prediction of physical function 1 year following hip fracture. Physiother. Res. Int. 2022, 27, e1947. [Google Scholar] [CrossRef] [PubMed]
| Variable | No. of Subjects | |
|---|---|---|
| Age, years (mean ± SEM) | 270 | 87.3 ± 0.3 |
| Female sex, n (%) | 270 | 214 (79.3) |
| Residential status, n (%) | ||
| Community dwelling | 270 | 236 (87.4) |
| Nursing home | 34 (12.6) | |
| Weight, kg (mean ± SEM) | 266 | 58.1 ± 0.7 |
| Height, cm (mean ± SEM) | 267 | 158.4 ± 0.5 |
| Body mass index, kg/m2 (mean ± SEM) | 265 | 23.1 ± 0.2 |
| Time from hip surgery to study baseline, days (mean ± SEM) | 270 | 1.8 ± 0.1 |
| Charlson Comorbidity Index (mean ± SEM) | 270 | 2.0 ± 0.1 |
| MNA score, (mean ± SEM) | ||
| Screening score | 8.4 ± 0.1 | |
| Assessment | 270 | 10.1 ± 0.1 |
| Total assessment | 18.3 ± 0.1 | |
| Co-morbidities, n (%) | ||
| Hypertension | 203 (75.2) | |
| Dyslipidemia | 106 (39.3) | |
| Smoker | 13 (4.8) | |
| Cardiovascular disease | 127 (47) | |
| Renal disease | 31 (11.5) | |
| Hepatobiliary disease | 13 (4.8) | |
| Cancer (current or past) | 39 (14.4) | |
| Diabetes | 270 | |
| Insulin-dependent | 0 (0.0) | |
| Not insulin-dependent | 49 (18.1) | |
| Other diseases | 183 (67.8) | |
| Barthel ADL index, (mean ± SEM) | 177 | 68.2 ± 2.1 |
| Anemia (hemoglobin < 11 g/dL), n (%) | 138 | 86 (62.3) |
| Reasons for ONS-HC-HP-HMB Discontinuation, n (%) | |
|---|---|
| Investigator’s decision due to AEs | 1 (0.4) |
| Subject’s decision due to AEs | 4 (1.5) |
| Investigator’s decision for reasons other than AEs | 4 (1.5) |
| Subject’s decision for reasons other than AEs | 6 (2.2) |
| Non-compliance | 32 (11.9) |
| Lost to follow-up | 22 (8.1) |
| Other | 8 (3.0) |
| Reasons for premature study discontinuation, n (%) | |
| Investigator’s decision due to AEs/change in medical status | 5 (1.9) |
| Subject’s decision due to AEs/change in medical status | 4 (1.5) |
| Subject’s decision for reasons other than AEs/change in medical status | 5 (1.9) |
| Non-compliance | 30 (11.1) |
| Lost to follow-up | 22 (8.1) |
| Exitus | 10 (3.7) |
| Other | 2 (0.7) |
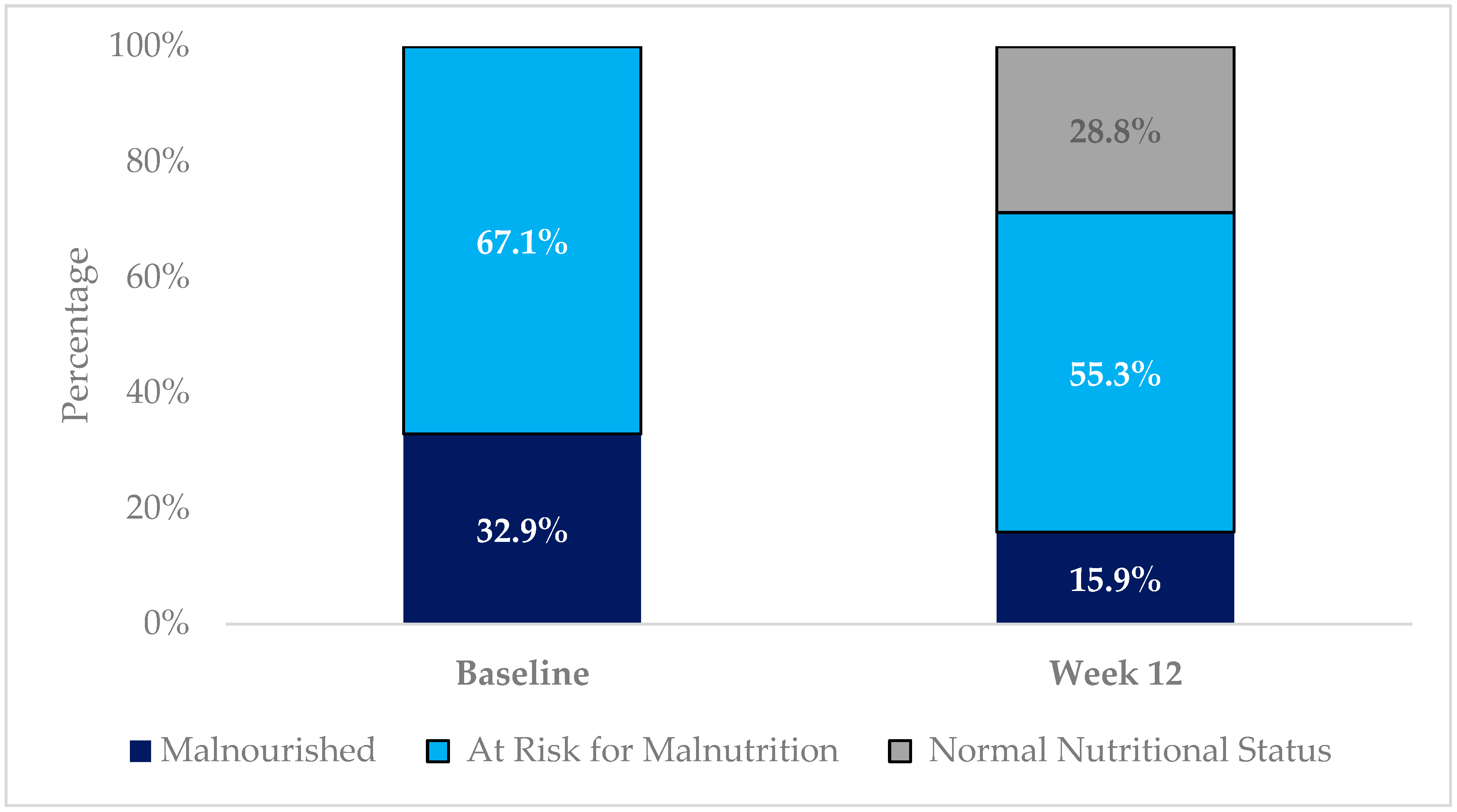
| MNA Change in Status | n (%) |
|---|---|
| Improved | 80 (46.8) |
| Maintained | 82 (48.0) |
| Worsened | 9 (5.3) |
| Variable | n | Baseline | Week 12 | Difference | p-Value |
|---|---|---|---|---|---|
| Total cholesterol (mg/dL) | 137 | 137.2 ± 3.3 | 173.5 ± 3.8 | 36.34 | <0.001 |
| LDL-cholesterol (mg/dL) | 95 | 76.2 ± 3.2 | 97.3 ± 3.6 | 21.07 | <0.001 |
| HDL-cholesterol (mg/dL) | 100 | 46.6 ± 1.3 | 53.5 ± 1.4 | 6.93 | <0.001 |
| Triglycerides (mg/dL) | 131 | 97.0 ± 3.7 | 112.3 ± 4.5 | 16.33 | <0.001 |
| Lymphocytes (%) | 132 | 16.8 ± 0.9 | 27.0 ± 0.9 | 10.20 | <0.001 |
| Hemoglobin (g/dL) | 144 | 10.48 ± 0.15 | 12.79 ± 0.28 | 2.31 | <0.001 |
| Total proteins (g/dL) | 126 | 5.59 ± 0.6 | 6.65 ± 0.7 | 1.06 | <0.001 |
| Albumin (g/dL) | 137 | 3.2 ± 0.03 | 3.82 ± 0.04 | 0.62 | <0.001 |
| Creatinine (mg/dL) | 148 | 0.95 ± 0.03 | 0.85 ± 0.02 | −0.10 | <0.001 |
| Glucose (mg/dL) | 148 | 111.12 ± 3.41 | 100.30 ± 2.17 | −10.82 | <0.001 |
| Vitamin D (ng/mL) | 90 | 16.4 ± 1.1 | 36.5 ± 1.8 | 20.05 | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pareja Sierra, T.; Hünicken Torrez, F.L.; Pablos Hernández, M.C.; López Velasco, R.; Ortés Gómez, R.; Cervera Díaz, M.d.C.; Hormigo Sánchez, A.I.; Perdomo Ramírez, B.; Mora Fernández, J.; Jiménez Mola, S.; et al. A Prospective, Observational Study of the Effect of a High-Calorie, High-Protein Oral Nutritional Supplement with HMB in an Old and Malnourished or at-Risk-of-Malnutrition Population with Hip Fractures: A FracNut Study. Nutrients 2024, 16, 1223. https://doi.org/10.3390/nu16081223
Pareja Sierra T, Hünicken Torrez FL, Pablos Hernández MC, López Velasco R, Ortés Gómez R, Cervera Díaz MdC, Hormigo Sánchez AI, Perdomo Ramírez B, Mora Fernández J, Jiménez Mola S, et al. A Prospective, Observational Study of the Effect of a High-Calorie, High-Protein Oral Nutritional Supplement with HMB in an Old and Malnourished or at-Risk-of-Malnutrition Population with Hip Fractures: A FracNut Study. Nutrients. 2024; 16(8):1223. https://doi.org/10.3390/nu16081223
Chicago/Turabian StylePareja Sierra, Teresa, Flavia Lorena Hünicken Torrez, María Carmen Pablos Hernández, Rosario López Velasco, Raquel Ortés Gómez, María del Carmen Cervera Díaz, Ana Isabel Hormigo Sánchez, Beatriz Perdomo Ramírez, Jesús Mora Fernández, Sonia Jiménez Mola, and et al. 2024. "A Prospective, Observational Study of the Effect of a High-Calorie, High-Protein Oral Nutritional Supplement with HMB in an Old and Malnourished or at-Risk-of-Malnutrition Population with Hip Fractures: A FracNut Study" Nutrients 16, no. 8: 1223. https://doi.org/10.3390/nu16081223
APA StylePareja Sierra, T., Hünicken Torrez, F. L., Pablos Hernández, M. C., López Velasco, R., Ortés Gómez, R., Cervera Díaz, M. d. C., Hormigo Sánchez, A. I., Perdomo Ramírez, B., Mora Fernández, J., Jiménez Mola, S., Rodriguez Piñera, M. A., Condorhuaman Alvarado, P. Y., Sanchez Juan, C., Ramos Clemente, J. I., Veses Martín, S., Rodríguez Manzano, I., González-Colaço Harmand, M., Camprubí Robles, M., Martín Aguilar, A., & Saez Lopez, P. (2024). A Prospective, Observational Study of the Effect of a High-Calorie, High-Protein Oral Nutritional Supplement with HMB in an Old and Malnourished or at-Risk-of-Malnutrition Population with Hip Fractures: A FracNut Study. Nutrients, 16(8), 1223. https://doi.org/10.3390/nu16081223






