The Power of Vitamin D: Is the Future in Precision Nutrition through Personalized Supplementation Plans?
Abstract
1. Introduction
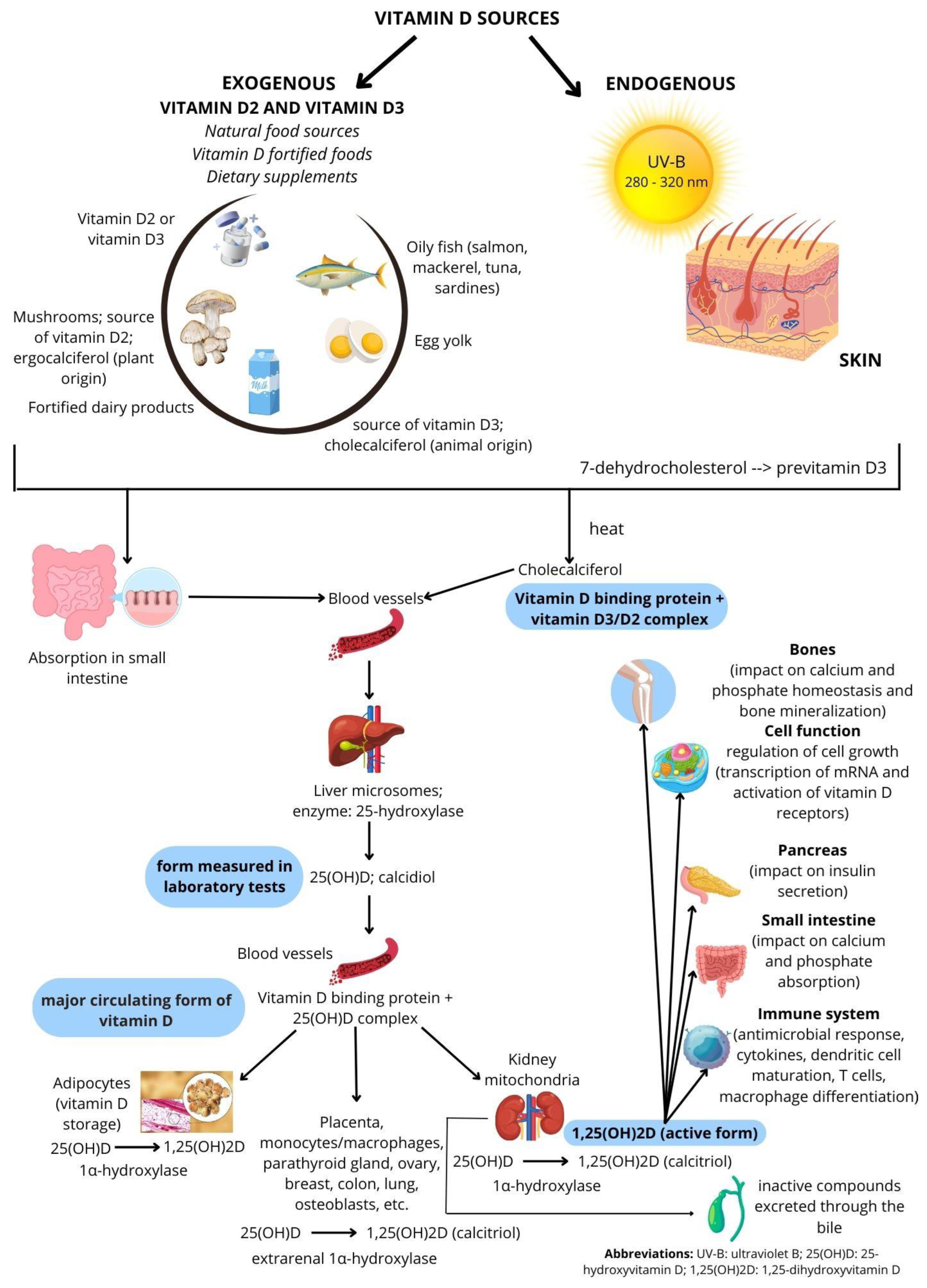
2. Vitamin D Metabolism
3. Vitamin D and Its Positive Health Effects
3.1. An Inseparable Bond: Vitamin D and Vitamin D Receptors
3.2. Vitamin D and Chronic Obstructive Pulmonary Disease
3.3. Vitamin D and the Coronavirus Disease 2019
3.4. Vitamin D and Polycystic Ovary Syndrome
3.5. Vitamin D and Bone Health
3.6. Vitamin D and Cancer
3.7. Vitamin D and Mental Health
3.8. Vitamin D, Immune System, and Autoimmune Diseases
3.9. Vitamin D and Diabetes
3.10. Vitamin D and Cardiovascular Disease
4. Shedding Light on the Global Pandemic of Vitamin D Deficiency—Do We Need Urgent Action?
5. Is It Possible to Obtain Sufficient Amounts of Vitamin D from Your Diet?
+ (1.74 × serum albumin) − (0.62 × starting 25[OH]D concentration)]/(−0.002)’
6. Can You Take Too Much of Vitamin D?
7. Future Developments in Vitamin D Therapy and the Potential Role of Personalized Supplementation Plans
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jones, G. The discovery and synthesis of the nutritional factor vitamin D. Int. J. Paleopathol. 2018, 23, 96–99. [Google Scholar] [CrossRef]
- Jones, G. 100 years of vitamin D: Historical aspects of vitamin D. Endocr. Connect. 2022, 11, e210594. [Google Scholar] [CrossRef]
- Carlberg, C. Vitamin D in the Context of Evolution. Nutrients 2022, 14, 3018. [Google Scholar] [CrossRef]
- Hess, A.F. The history of rickets. In Rickets, Including Osteomalacia and Tetany; Lea & Febiger: Philadelphia, PA, USA, 1929; pp. 22–37. [Google Scholar]
- MacIntyre, I.; Evans, A.; Larkins, R.G. Vitamin D. Clin. Endocrinol. 1977, 6, 65–79. [Google Scholar] [CrossRef]
- McCollum, E.V. The Paths to the Discovery of Vitamins A and D. J. Nutr. 1967, 91 (Suppl. S1), 11–16. [Google Scholar] [CrossRef]
- McCollum, E.V.; Simmonds, N.; Becker, J.E.; Shipley, P.G. Studies on experimental rickets: XXI. An experimental demonstration of the existence of a vitamin which promotes calcium deposition. J. Biol. Chem. 1922, 53, 293–312. [Google Scholar] [CrossRef]
- Mellanby, E. An experimental investigation on rickets. Lancet 1919, 193, 407–412. [Google Scholar] [CrossRef]
- Steenbock, H. The Induction of Growth-Promoting and Calcifying Properties in a Ration by Exposure to Ultra-Violet Light. Science 1924, 60, 224–225. [Google Scholar] [CrossRef]
- Steenbock, H.; Black, A. Fat-Soluble Vitamins: XVII. The Induction of Growth-Promoting and Calcifying Properties in a Ration by Exposure to Ultra-Violet Light. J. Biol. Chem. 1924, 61, 405–422. [Google Scholar] [CrossRef]
- Silva, M.C.; Furlanetto, T.W. Intestinal absorption of vitamin D: A systematic review. Nutr. Rev. 2018, 76, 60–76. [Google Scholar] [CrossRef]
- Williams, S.E. Vitamin D supplementation: Pearls for practicing clinicians. Clevel. Clin. J. Med. 2022, 89, 154–160. [Google Scholar] [CrossRef]
- Wacker, M.; Holick, M.F. Sunlight and Vitamin D. Derm.-Endocrinol. 2013, 5, 51–108. [Google Scholar] [CrossRef]
- Ramasamy, I. Vitamin D Metabolism and Guidelines for Vitamin D Supplementation. Clin. Biochem. Rev. 2020, 41, 103–126. [Google Scholar] [CrossRef]
- Dominguez, L.J.; Farruggia, M.; Veronese, N.; Barbagallo, M. Vitamin D Sources, Metabolism, and Deficiency: Available Compounds and Guidelines for Its Treatment. Metabolites 2021, 11, 255. [Google Scholar] [CrossRef]
- Bikle, D.D. Vitamin D Metabolism, Mechanism of Action, and Clinical Applications. Chem. Biol. 2014, 21, 319–329. [Google Scholar] [CrossRef]
- Bikle, D.D. Vitamin D: Production, Metabolism and Mechanisms of Action. In Endotext; Feingold, K.R., Anawalt, B., Blackman, M.R., Boyce, A., Chrousos, G., Corpas, E., de Herder, W.W., Dhatariya, K., Dungan, K., Hofland, J., et al., Eds.; MDText.com, Inc.: South Dartmouth, MA, USA, 2021. [Google Scholar]
- Bikle, D.D.; Patzek, S.; Wang, Y. Physiologic and pathophysiologic roles of extra renal CYP27b1: Case report and review. Bone Rep. 2018, 8, 255–267. [Google Scholar] [CrossRef]
- Norman, A.W.; Henry, H.H. Vitamin D. In Present Knowledge in Nutrition, 10th ed.; Erdman, J.W., Macdonald, I.A., Zeisel, S.H., Eds.; Wiley-Blackwell: Washington, DC, USA, 2012; p. 207. [Google Scholar]
- Jeon, S.M.; Shin, E.A. Exploring vitamin D metabolism and function in cancer. Exp. Mol. Med. 2018, 50, 1–14. [Google Scholar] [CrossRef]
- Szymczak-Pajor, I.; Miazek, K.; Selmi, A.; Balcerczyk, A.; Śliwińska, A. The Action of Vitamin D in Adipose Tissue: Is There the Link between Vitamin D Deficiency and Adipose Tissue-Related Metabolic Disorders? Int. J. Mol. Sci. 2022, 23, 956. [Google Scholar] [CrossRef]
- Raymond-Lezman, J.R.; Riskin, S.I. Benefits and Risks of Sun Exposure to Maintain Adequate Vitamin D Levels. Cureus 2023, 15, e38578. [Google Scholar] [CrossRef]
- Battistini, C.; Ballan, R.; Herkenhoff, M.E.; Saad, S.M.I.; Sun, J. Vitamin D Modulates Intestinal Microbiota in Inflammatory Bowel Diseases. Int. J. Mol. Sci. 2020, 22, 362. [Google Scholar] [CrossRef]
- Voltan, G.; Cannito, M.; Ferrarese, M.; Ceccato, F.; Camozzi, V. Vitamin D: An Overview of Gene Regulation, Ranging from Metabolism to Genomic Effects. Genes 2023, 14, 1691. [Google Scholar] [CrossRef]
- Sun, J.; Zhang, Y.G. Vitamin D Receptor Influences Intestinal Barriers in Health and Disease. Cells 2022, 11, 1129. [Google Scholar] [CrossRef]
- Arora, J.; Wang, J.; Weaver, V.; Zhang, Y.; Cantorna, M.T. Novel insight into the role of the vitamin D receptor in the development and function of the immune system. J. Steroid Biochem. Mol. Biol. 2022, 219, 106084. [Google Scholar] [CrossRef]
- Li, X.; He, J.; Yu, M.; Sun, J. The efficacy of vitamin D therapy for patients with COPD: A meta-analysis of randomized controlled trials. Ann. Palliat. Med. 2020, 9, 286–297. [Google Scholar] [CrossRef]
- Martineau, A.R.; James, W.Y.; Hooper, R.L.; Barnes, N.C.; Jolliffe, D.A.; Greiller, C.L.; Islam, K.; McLaughlin, D.; Bhowmik, A.; Timms, P.M.; et al. Vitamin D3 supplementation in patients with chronic obstructive pulmonary disease (ViDiCO): A multicentre, double-blind, randomised controlled trial. Lancet Respir. Med. 2015, 3, 120–130. [Google Scholar] [CrossRef]
- Donastin, A.; Amin, M.; Yulistiani. Mechanism of High Dosage Vitamin D Supplementation on The Lung Function and Quality of Life of Stable COPD Patients. Pharmacogn. J. 2023, 15, 274–278. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, J.; Chen, L.; Zhang, H.; Yu, L.; Chi, Y.; Chen, M.; Cai, Y. Efficacy of vitamin D supplementation on COPD and asthma control: A systematic review and meta-analysis. J. Glob. Health 2022, 12, 04100. [Google Scholar] [CrossRef]
- Sanjari, M.; Soltani, A.; Habibi Khorasani, A.; Zareinejad, M. The effect of vitamin D on COPD exacerbation: A double blind randomized placebo-controlled parallel clinical trial. J. Diabetes Metab. Disord. 2016, 15, 33. [Google Scholar] [CrossRef]
- Ferrari, R.; Caram, L.M.O.; Tanni, S.E.; Godoy, I.; Rupp de Paiva, S.A. The relationship between Vitamin D status and exacerbation in COPD patients– a literature review. Respir. Med. 2018, 139, 34–38. [Google Scholar] [CrossRef]
- Argano, C.; Mallaci Bocchio, R.; Lo Monaco, M.; Scibetta, S.; Natoli, G.; Cavezzi, A.; Troiani, E.; Corrao, S. An Overview of Systematic Reviews of the Role of Vitamin D on Inflammation in Patients with Diabetes and the Potentiality of Its Application on Diabetic Patients with COVID-19. Int. J. Mol. Sci. 2022, 23, 2873. [Google Scholar] [CrossRef]
- Argano, C.; Mallaci Bocchio, R.; Natoli, G.; Scibetta, S.; Lo Monaco, M.; Corrao, S. Protective Effect of Vitamin D Supplementation on COVID-19-Related Intensive Care Hospitalization and Mortality: Definitive Evidence from Meta-Analysis and Trial Sequential Analysis. Pharmaceuticals 2023, 16, 130. [Google Scholar] [CrossRef]
- Cicero, A.F.G.; Fogacci, F.; Borghi, C. Vitamin D Supplementation and COVID-19 Outcomes: Mounting Evidence and Fewer Doubts. Nutrients 2022, 14, 3584. [Google Scholar] [CrossRef]
- Hadizadeh, F. Supplementation with vitamin D in the COVID-19 pandemic? Nutr. Rev. 2021, 79, 200–208. [Google Scholar] [CrossRef]
- Kolls, J.K.; Garry, R.F. Role of the T cell vitamin D receptor in severe COVID-19. Nat. Immunol. 2022, 23, 5–6. [Google Scholar] [CrossRef]
- Azmi, H.; Hassou, N.; Ennaji, M.M. Vitamin D Immunomodulatory Role in Chronic and Acute Viral Diseases. In Emerging and Reemerging Viral Pathogens; Ennaji, M.M., Ed.; Elsevier: London, UK, 2020; pp. 489–506. [Google Scholar]
- Han, J.E.; Jones, J.L.; Tangpricha, V.; Brown, M.A.; Hao, L.; Hebbar, G.; Lee, M.J.; Liu, S.; Brown, L.A.S.; Ziegler, T.R.; et al. High Dose Vitamin D Administration in Ventilated Intensive Care Unit Patients: A Pilot Double Blind Randomized Controlled Trial. J. Clin. Transl. Endocrinol. 2016, 4, 59–65. [Google Scholar] [CrossRef]
- Mutt, S.J.; Hyppönen, E.; Saarnio, J.; Järvelin, M.R.; Herzig, K.H. Vitamin D and adipose tissue—More than storage. Front. Physiol. 2014, 5, 228. [Google Scholar] [CrossRef]
- Charoenngam, N.; Holick, M.F. Immunologic Effects of Vitamin D on Human Health and Disease. Nutrients 2020, 12, 2097. [Google Scholar] [CrossRef]
- Dhawan, M.; Priyanka; Choudhary, O.P. Immunomodulatory and therapeutic implications of vitamin D in the management of COVID-19. Hum. Vaccines Immunother. 2022, 18, 2025734. [Google Scholar] [CrossRef]
- Tang, W.; Chen, L.; Ma, W.; Chen, D.; Wang, C.; Gao, Y.; Ran, X. Association between vitamin D status and diabetic foot in patients with type 2 diabetes mellitus. J. Diabetes Investig. 2022, 13, 1213–1221. [Google Scholar] [CrossRef]
- Alcala-Diaz, J.F.; Limia-Perez, L.; Gomez-Huelgas, R.; Martin-Escalante, M.D.; Cortes-Rodriguez, B.; Zambrana-Garcia, J.L.; Entrenas-Castillo, M.; Perez-Caballero, A.I.; López-Carmona, M.D.; Garcia-Alegria, J.; et al. Calcifediol Treatment and Hospital Mortality Due to COVID-19: A Cohort Study. Nutrients 2021, 13, 1760. [Google Scholar] [CrossRef]
- Annweiler, C.; Hanotte, B.; Grandin de l’Eprevier, C.; Sabatier, J.M.; Lafaie, L.; Célarier, T. Vitamin D and survival in COVID-19 patients: A quasi-experimental study. J. Steroid Biochem. Mol. Biol. 2020, 204, 105771. [Google Scholar] [CrossRef] [PubMed]
- Annweiler, G.; Corvaisier, M.; Gautier, J.; Dubée, V.; Legrand, E.; Sacco, G.; Annweiler, C. Vitamin D Supplementation Associated to Better Survival in Hospitalized Frail Elderly COVID-19 Patients: The GERIA-COVID Quasi-Experimental Study. Nutrients 2020, 12, 3377. [Google Scholar] [CrossRef] [PubMed]
- Leclair, T.; Zakai, N.A.; Bunn, J.Y.; Gianni, M.; Heyland, D.K.; Ardren, S.S.; Stapleton, R.D. Vitamin D Supplementation in Mechanically Ventilated Patients in the Medical Intensive Care Unit. JPEN J. Parenter. Enter. Nutr. 2019, 43, 1037–1043. [Google Scholar] [CrossRef] [PubMed]
- Ling, S.F.; Broad, E.; Murphy, R.; Pappachan, J.M.; Pardesi-Newton, S.; Kong, M.F.; Jude, E.B. High-Dose Cholecalciferol Booster Therapy is Associated with a Reduced Risk of Mortality in Patients with COVID-19: A Cross-Sectional Multi-Centre Observational Study. Nutrients 2020, 12, 3799. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, A.; Bhansali, A.; Khare, N.; Suri, V.; Yaddanapudi, N.; Sachdeva, N.; Puri, G.D.; Malhotra, P. Short term, high-dose vitamin D supplementation for COVID-19 disease: A randomised, placebo-controlled, study (SHADE study). Postgrad. Med. J. 2022, 98, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Sabico, S.; Enani, M.A.; Sheshah, E.; Aljohani, N.J.; Aldisi, D.A.; Alotaibi, N.H.; Alshingetti, N.; Alomar, S.Y.; Alnaami, A.M.; Amer, O.E.; et al. Effects of a 2-Week 5000 IU versus 1000 IU Vitamin D3 Supplementation on Recovery of Symptoms in Patients with Mild to Moderate Covid-19: A Randomized Clinical Trial. Nutrients 2021, 13, 2170. [Google Scholar] [CrossRef] [PubMed]
- Malaguarnera, L. Vitamin D3 as Potential Treatment Adjuncts for COVID-19. Nutrients 2020, 12, 3512. [Google Scholar] [CrossRef] [PubMed]
- Borsche, L.; Glauner, B.; von Mendel, J. COVID-19 Mortality Risk Correlates Inversely with Vitamin D3 Status, and a Mortality Rate Close to Zero Could Theoretically Be Achieved at 50 ng/mL 25(OH)D3: Results of a Systematic Review and Meta-Analysis. Nutrients 2021, 13, 3596. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.B.; Lahore, H.; McDonnell, S.L.; Baggerly, C.A.; French, C.B.; Aliano, J.L.; Bhattoa, H.P. Evidence that Vitamin D Supplementation Could Reduce Risk of Influenza and COVID-19 Infections and Deaths. Nutrients 2020, 12, 988. [Google Scholar] [CrossRef]
- Raperport, C.; Homburg, R. The Source of Polycystic Ovarian Syndrome. Clin. Med. Insights Reprod. Health 2019, 13, 1179558119871467. [Google Scholar] [CrossRef]
- Thomson, R.L.; Spedding, S.; Buckley, J.D. Vitamin D in the aetiology and management of polycystic ovary syndrome. Clin. Endocrinol. 2012, 77, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Morgante, G.; Darino, I.; Spanò, A.; Luisi, S.; Luddi, A.; Piomboni, P.; Governini, L.; De Leo, V. PCOS Physiopathology and Vitamin D Deficiency: Biological Insights and Perspectives for Treatment. J. Clin. Med. 2022, 11, 4509. [Google Scholar] [CrossRef]
- Fatemi, F.; Mohammadzadeh, A.; Sadeghi, M.R.; Akhondi, M.M.; Mohammadmoradi, S.; Kamali, K.; Lackpour, N.; Jouhari, S.; Zafadoust, S.; Mokhtar, S.; et al. Role of vitamin E and D 3 supplementation in Intra-Cytoplasmic Sperm Injection outcomes of women with polycystic ovarian syndrome: A double blinded randomized placebo-controlled trial. Clin. Nutr. ESPEN 2017, 18, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Irani, M.; Merhi, Z. Role of vitamin D in ovarian physiology and its implication in reproduction: A systematic review. Fertil. Steril. 2014, 102, 460–468. [Google Scholar] [CrossRef] [PubMed]
- Fox, J.D.; Benesh, G.; Abrouk, M.; Kirsner, R.S. Controversies in Sunscreens: A Practical Approach. Am. J. Med. 2020, 133, 1378–1379. [Google Scholar] [CrossRef] [PubMed]
- Laird, E.; Ward, M.; McSorley, E.; Strain, J.J.; Wallace, J. Vitamin D and Bone Health; Potential Mechanisms. Nutrients 2010, 2, 693–724. [Google Scholar] [CrossRef] [PubMed]
- Lips, P.; van Schoor, N.M. The effect of vitamin D on bone and osteoporosis. Best Pract. Res. Clin. Endocrinol. Metab. 2011, 25, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Yao, P.; Bennett, D.; Mafham, M.; Lin, X.; Chen, Z.; Armitage, J.; Clarke, R. Vitamin D and Calcium for the Prevention of Fracture: A Systematic Review and Meta-analysis. JAMA Netw. Open 2019, 2, e1917789. [Google Scholar] [CrossRef] [PubMed]
- Weaver, C.M.; Alexander, D.D.; Boushey, C.J.; Dawson-Hughes, B.; Lappe, J.M.; LeBoff, M.S.; Liu, S.; Looker, A.C.; Wallace, T.C.; Wang, D.D. Calcium plus vitamin D supplementation and risk of fractures: An updated meta-analysis from the National Osteoporosis Foundation. Osteoporos. Int. 2016, 27, 367–376. [Google Scholar] [CrossRef]
- Zhao, J.G.; Zeng, X.T.; Wang, J.; Liu, L. Association Between Calcium or Vitamin D Supplementation and Fracture Incidence in Community-Dwelling Older Adults. JAMA 2017, 318, 2466. [Google Scholar] [CrossRef]
- DIPART (Vitamin D Individual Patient Analysis of Randomized Trials) Group. Patient level pooled analysis of 68 500 patients from seven major vitamin D fracture trials in US and Europe. BMJ 2010, 340, b5463. [Google Scholar] [CrossRef]
- Reid, I.R.; Bolland, M.J.; Grey, A. Effects of vitamin D supplements on bone mineral density: A systematic review and meta-analysis. Lancet 2014, 383, 146–155. [Google Scholar] [CrossRef]
- Henn, M.; Martin-Gorgojo, V.; Martin-Moreno, J.M. Vitamin D in Cancer Prevention: Gaps in Current Knowledge and Room for Hope. Nutrients 2022, 14, 4512. [Google Scholar] [CrossRef]
- Muñoz, A.; Grant, W.B. Vitamin D and Cancer: An Historical Overview of the Epidemiology and Mechanisms. Nutrients 2022, 14, 1448. [Google Scholar] [CrossRef]
- Hossain, S.; Beydoun, M.A.; Beydoun, H.A.; Chen, X.; Zonderman, A.B.; Wood, R.J. Vitamin D and breast cancer: A systematic review and meta-analysis of observational studies. Clin. Nutr. ESPEN 2019, 30, 170–184. [Google Scholar] [CrossRef] [PubMed]
- Voutsadakis, I.A. Vitamin D baseline levels at diagnosis of breast cancer: A systematic review and meta-analysis. Hematol. Oncol. Stem Cell Ther. 2021, 14, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, S.; Che, X.; Li, X. Vitamin D and lung cancer risk: A comprehensive review and meta-analysis. Cell. Physiol. Biochem. 2015, 36, 299–305. [Google Scholar] [CrossRef]
- Zhao, J.; Wang, H.; Zhang, Z.; Zhou, X.; Yao, J.; Zhang, R.; Liao, L.; Dong, J. Vitamin D deficiency as a risk factor for thyroid cancer: A meta-analysis of case-control studies. Nutrition 2019, 57, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Keum, N.; Chen, Q.Y.; Lee, D.H.; Manson, J.E.; Giovannucci, E. Vitamin D supplementation and total cancer incidence and mortality by daily vs. infrequent large-bolus dosing strategies: A meta-analysis of randomised controlled trials. Br. J. Cancer 2022, 127, 872–878. [Google Scholar] [CrossRef]
- Kuznia, S.; Zhu, A.; Akutsu, T.; Buring, J.E.; Camargo, C.A., Jr.; Cook, N.R.; Chen, L.J.; Cheng, T.Y.D.; Hantunen, S.; Lee, I.M.; et al. Efficacy of vitamin D3 supplementation on cancer mortality: Systematic review and individual patient data meta-analysis of randomised controlled trials. Ageing Res. Rev. 2023, 87, 101923. [Google Scholar] [CrossRef]
- Akpınar, Ş.; Karadağ, M.G. Is Vitamin D Important in Anxiety or Depression? What Is the Truth? Curr. Nutr. Rep. 2022, 11, 675–681. [Google Scholar] [CrossRef] [PubMed]
- Cuomo, A.; Fagiolini, A. Vitamin D and psychiatric illnesses. Vitamin D UpDates 2019, 2, 3–10. [Google Scholar] [CrossRef]
- Gowda, U.; Mutowo, M.P.; Smith, B.J.; Wluka, A.E.; Renzaho, A.M.N. Vitamin D supplementation to reduce depression in adults: Meta-analysis of randomized controlled trials. Nutrition 2015, 31, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Mbuagbaw, L.; Samaan, Z.; Falavigna, M.; Zhang, S.; Adachi, J.D.; Cheng, J.; Papaioannou, A.; Thabane, L. Efficacy of Vitamin D Supplementation in Depression in Adults: A Systematic Review. J. Clin. Endocrinol. Metab. 2014, 99, 757–767. [Google Scholar] [CrossRef]
- Shaffer, J.A.; Edmondson, D.; Wasson, L.T.; Falzon, L.; Homma, K.; Ezeokoli, N.; Li, P.; Davidson, K.W. Vitamin D supplementation for depressive symptoms: A systematic review and meta-analysis of randomized controlled trials. Psychosom. Med. 2014, 76, 190–196. [Google Scholar] [CrossRef]
- Spedding, S. Vitamin D and depression: A systematic review and meta-analysis comparing studies with and without biological flaws. Nutrients 2014, 6, 1501–1518. [Google Scholar] [CrossRef]
- Vellekkatt, F.; Menon, V. Efficacy of vitamin D supplementation in major depression: A meta-analysis of randomized controlled trials. J. Postgrad. Med. 2019, 65, 74–80. [Google Scholar] [CrossRef]
- Robinson, S.L.; Marín, C.; Oliveros, H.; Mora-Plazas, M.; Lozoff, B.; Villamor, E. Vitamin D Deficiency in Middle Childhood Is Related to Behavior Problems in Adolescence. J. Nutr. 2020, 150, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Tolppanen, A.M.; Sayers, A.; Fraser, W.D.; Lewis, G.; Zammit, S.; Lawlor, D.A. The association of serum 25-hydroxyvitamin D3 and D2 with depressive symptoms in childhood—A prospective cohort study. J. Child Psychol. Psychiatry 2012, 53, 757–766. [Google Scholar] [CrossRef]
- Chan, R.; Chan, D.; Woo, J.; Ohlsson, C.; Mellström, D.; Kwok, T.; Leung, P. Association between serum 25-hydroxyvitamin D and psychological health in older Chinese men in a cohort study. J. Affect. Disord. 2011, 130, 251–259. [Google Scholar] [CrossRef]
- Lee, D.M.; Tajar, A.; O’Neill, T.W.; O’Connor, D.B.; Bartfai, G.; Boonen, S.; Bouillon, R.; Casanueva, F.F.; Finn, J.D.; Forti, G.; et al. Lower vitamin D levels are associated with depression among community-dwelling European men. J. Psychopharmacol. 2011, 25, 1320–1328. [Google Scholar] [CrossRef] [PubMed]
- May, H.T.; Bair, T.L.; Lappé, D.L.; Anderson, J.L.; Horne, B.D.; Carlquist, J.F.; Muhlestein, J.B. Association of vitamin D levels with incident depression among a general cardiovascular population. Am. Heart J. 2010, 159, 1037–1043. [Google Scholar] [CrossRef] [PubMed]
- Milaneschi, Y.; Hoogendijk, W.; Lips, P.; Heijboer, A.C.; Schoevers, R.; van Hemert, A.M.; Beekman, A.T.F.; Smit, J.H.; Penninx, B.W.J.H. The association between low vitamin D and depressive disorders. Mol. Psychiatry 2014, 19, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Ford, E.S.; Li, C. Associations of Serum Concentrations of 25-Hydroxyvitamin D and Parathyroid Hormone with Surrogate Markers of Insulin Resistance Among U.S. Adults Without Physician-Diagnosed Diabetes: NHANES, 2003–2006. Diabetes Care 2010, 33, 344–347. [Google Scholar] [CrossRef] [PubMed]
- Menon, V.; Kar, S.K.; Suthar, N.; Nebhinani, N. Vitamin D and Depression: A Critical Appraisal of the Evidence and Future Directions. Indian J. Psychol. Med. 2020, 42, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Zhang, Y.; Wang, T.; Lin, Y.; Yu, J.; Xia, Q.; Zhu, P.; Zhu, D.M. Vitamin D supplementation improves anxiety but not depression symptoms in patients with vitamin D deficiency. Brain Behav. 2020, 10, e01760. [Google Scholar] [CrossRef] [PubMed]
- Guzek, D.; Kołota, A.; Lachowicz, K.; Skolmowska, D.; Stachoń, M.; Głąbska, D. Effect of Vitamin D Supplementation on Depression in Adults: A Systematic Review of Randomized Controlled Trials (RCTs). Nutrients 2023, 15, 951. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; McGrath, J.J.; Burne, T.H.J.; Eyles, D.W. Vitamin D and schizophrenia: 20 years on. Mol. Psychiatry 2021, 26, 2708–2720. [Google Scholar] [CrossRef]
- Cereda, G.; Enrico, P.; Ciappolino, V.; Delvecchio, G.; Brambilla, P. The role of vitamin D in bipolar disorder: Epidemiology and influence on disease activity. J. Affect. Disord. 2021, 278, 209–217. [Google Scholar] [CrossRef]
- Saedisomeolia, A.; Samadi, M.; Gholami, F.; Seyedi, M.; Effatpanah, M.; Hashemi, R.; Abdolahi, M.; Honarvar, N.M. Vitamin D’s Molecular Action Mechanism in Attention-deficit/Hyperactivity Disorder: A Review of Evidence. CNS Neurol. Disord. Drug Targets 2018, 17, 280–290. [Google Scholar] [CrossRef]
- Krivoy, A.; Onn, R.; Vilner, Y.; Hochman, E.; Weizman, S.; Paz, A.; Hess, S.; Sagy, R.; Kimhi-Nesher, S.; Kalter, E.; et al. Vitamin D Supplementation in Chronic Schizophrenia Patients Treated with Clozapine: A Randomized, Double-Blind, Placebo-controlled Clinical Trial. EbioMedicine 2017, 26, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Ghaderi, A.; Banafshe, H.R.; Mirhosseini, N.; Moradi, M.; Karimi, M.A.; Mehrzad, F.; Bahmani, F.; Asemi, Z. Clinical and metabolic response to vitamin D plus probiotic in schizophrenia patients. BMC Psychiatry 2019, 19, 77. [Google Scholar] [CrossRef] [PubMed]
- Aranow, C. Vitamin D and the Immune System. J. Investig. Med. 2011, 59, 881–886. [Google Scholar] [CrossRef] [PubMed]
- Sîrbe, C.; Rednic, S.; Grama, A.; Pop, T.L. An Update on the Effects of Vitamin D on the Immune System and Autoimmune Diseases. Int. J. Mol. Sci. 2022, 23, 9784. [Google Scholar] [CrossRef] [PubMed]
- Fatima, M.; Lamis, A.; Siddiqui, S.W.; Ashok, T.; Patni, N.; Fadiora, O.E. Therapeutic Role of Vitamin D in Multiple Sclerosis: An Essentially Contested Concept. Cureus 2022, 14, e26186. [Google Scholar] [CrossRef] [PubMed]
- Pierrot-Deseilligny, C.; Souberbielle, J.C. Vitamin D and multiple sclerosis: An update. Mult. Scler. Relat. Disord. 2017, 14, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Scazzone, C.; Agnello, L.; Bivona, G.; Lo Sasso, B.; Ciaccio, M. Vitamin D and Genetic Susceptibility to Multiple Sclerosis. Biochem. Genet. 2021, 59, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Alfredsson, L.; Olsson, T. Lifestyle and Environmental Factors in Multiple Sclerosis. Cold Spring Harb. Perspect. Med. 2019, 9, a028944. [Google Scholar] [CrossRef]
- Thompson, A.J.; Baranzini, S.E.; Geurts, J.; Hemmer, B.; Ciccarelli, O. Multiple sclerosis. Lancet 2018, 391, 1622–1636. [Google Scholar] [CrossRef]
- Feinstein, A.; Freeman, J.; Lo, A.C. Treatment of progressive multiple sclerosis: What works, what does not, and what is needed. Lancet Neurol. 2015, 14, 194–207. [Google Scholar] [CrossRef]
- Feige, J.; Moser, T.; Bieler, L.; Schwenker, K.; Hauer, L.; Sellner, J. Vitamin D Supplementation in Multiple Sclerosis: A Critical Analysis of Potentials and Threats. Nutrients 2020, 12, 783. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, F.; Jhaveri, S.; Avanthika, C.; Singh, A.; Jain, N.; Gulraiz, A.; Shah, P.; Nasir, F. Impact of Vitamin D Supplementation on Multiple Sclerosis. Cureus 2021, 13, e18487. [Google Scholar] [CrossRef]
- McLaughlin, L.; Clarke, L.; Khalilidehkordi, E.; Butzkueven, H.; Taylor, B.; Broadley, S.A. Vitamin D for the treatment of multiple sclerosis: A meta-analysis. J. Neurol. 2018, 265, 2893–2905. [Google Scholar] [CrossRef] [PubMed]
- Simpson-Yap, S.; Jelinek, P.; Weiland, T.; Nag, N.; Neate, S.; Jelinek, G. Self-reported use of vitamin D supplements is associated with higher physical quality of life scores in multiple sclerosis. Mult. Scler. Relat. Disord. 2021, 49, 102760. [Google Scholar] [CrossRef]
- Beckmann, Y.; Türe, S.; Duman, S.U. Vitamin D deficiency and its association with fatigue and quality of life in multiple sclerosis patients. EPMA J. 2020, 11, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Jagannath, V.A.; Filippini, G.; Borges do Nascimento, I.J.; Di Pietrantonj, C.; Robak, E.W.; Whamond, L. Vitamin D for the management of multiple sclerosis. Cochrane Database Syst. Rev. 2018, 9, CD008422. [Google Scholar] [CrossRef] [PubMed]
- Kim, D. The Role of Vitamin D in Thyroid Diseases. Int. J. Mol. Sci. 2017, 18, 1949. [Google Scholar] [CrossRef] [PubMed]
- Turashvili, N.; Javashvili, L.; Giorgadze, E. Vitamin D Deficiency Is More Common in Women with Autoimmune Thyroiditis: A Retrospective Study. Int. J. Endocrinol. 2021, 2021, 4465563. [Google Scholar] [CrossRef] [PubMed]
- Camurdan, O.M.; Döğer, E.; Bideci, A.; Celik, N.; Cinaz, P. Vitamin D status in children with Hashimoto thyroiditis. J. Pediatr. Endocrinol. Metab. 2012, 25, 467–470. [Google Scholar] [CrossRef]
- Aktaş, H.S. Vitamin B12 and Vitamin D Levels in Patients with Autoimmune Hypothyroidism and Their Correlation with Anti-Thyroid Peroxidase Antibodies. Med. Princ. Pract. 2020, 29, 364–370. [Google Scholar] [CrossRef]
- Unal, A.D.; Tarcin, O.; Parildar, H.; Cigerli, O.; Eroglu, H.; Demirag, N.G. Vitamin D deficiency is related to thyroid antibodies in autoimmune thyroiditis. Cent. Eur. J. Immunol. 2014, 39, 493–497. [Google Scholar] [CrossRef]
- Li, B.; Liu, B.; Bao, W.; Rong, S. Association between Serum 25-Hydroxyvitamin D Concentrations and Respiratory Infection among United States Adults. J. Nutr. 2023, 153, 260–267. [Google Scholar] [CrossRef]
- Carr, A.C.; Maggini, S. Vitamin C and Immune Function. Nutrients 2017, 9, 1211. [Google Scholar] [CrossRef]
- Maddaloni, E.; Cavallari, I.; Napoli, N.; Conte, C. Vitamin D and Diabetes Mellitus. Front. Horm. Res. 2018, 50, 161–176. [Google Scholar] [CrossRef]
- Mitri, J.; Pittas, A.G. Vitamin D and diabetes. Endocrinol. Metab. Clin. N. Am. 2014, 43, 205–232. [Google Scholar] [CrossRef]
- Wu, J.; Atkins, A.; Downes, M.; Wei, Z. Vitamin D in Diabetes: Uncovering the Sunshine Hormone’s Role in Glucose Metabolism and Beyond. Nutrients 2023, 15, 1997. [Google Scholar] [CrossRef]
- Denos, M.; Mai, X.M.; Åsvold, B.O.; Sørgjerd, E.P.; Chen, Y.; Sun, Y.Q. Vitamin D status and risk of type 2 diabetes in the Norwegian HUNT cohort study: Does family history or genetic predisposition modify the association? BMJ Open Diabetes Res. Care 2021, 9, e001948. [Google Scholar] [CrossRef]
- Kwak, J.H.; Choi, Y.H.; Paik, J.K. Vitamin D Status, Fiber Intake, and Type 2 Diabetes in U.S. Adults. J. Med. Food 2020, 23, 711–718. [Google Scholar] [CrossRef]
- Wang, M.; Zhou, T.; Li, X.; Ma, H.; Liang, Z.; Fonseca, V.; Heianza, Y.; Qi, L. Baseline Vitamin D Status, Sleep Patterns, and the Risk of Incident Type 2 Diabetes in Data from the UK Biobank Study. Diabetes Care 2020, 43, 2776–2784. [Google Scholar] [CrossRef]
- Todorova, A.S.; Jude, E.B.; Dimova, R.B.; Chakarova, N.Y.; Serdarova, M.S.; Grozeva, G.G.; Tsarkova, P.V.; Tankova, T.I. Vitamin D Status in a Bulgarian Population with Type 2 Diabetes and Diabetic Foot Ulcers. Int. J. Low. Extrem. Wounds 2022, 21, 506–512. [Google Scholar] [CrossRef]
- Li, X.; Liu, Y.; Zheng, Y.; Wang, P.; Zhang, Y. The Effect of Vitamin D Supplementation on Glycemic Control in Type 2 Diabetes Patients: A Systematic Review and Meta-Analysis. Nutrients 2018, 10, 375. [Google Scholar] [CrossRef]
- Lee, C.J.; Iyer, G.; Liu, Y.; Kalyani, R.R.; Bamba, N.; Ligon, C.B.; Varma, S.; Mathioudakis, N. The effect of vitamin D supplementation on glucose metabolism in type 2 diabetes mellitus: A systematic review and meta-analysis of intervention studies. J. Diabetes Complicat. 2017, 31, 1115–1126. [Google Scholar] [CrossRef]
- Pittas, A.G.; Jorde, R.; Kawahara, T.; Dawson-Hughes, B. Vitamin D Supplementation for Prevention of Type 2 Diabetes Mellitus: To D or Not to D? J. Clin. Endocrinol. Metab. 2020, 105, 3721–3733. [Google Scholar] [CrossRef]
- Zhang, Y.; Tan, H.; Tang, J.; Li, J.; Chong, W.; Hai, Y.; Feng, Y.; Lunsford, L.D.; Xu, P.; Jia, D.; et al. Effects of Vitamin D Supplementation on Prevention of Type 2 Diabetes in Patients with Prediabetes: A Systematic Review and Meta-analysis. Diabetes Care 2020, 43, 1650–1658. [Google Scholar] [CrossRef]
- Manousaki, D.; Harroud, A.; Mitchell, R.E.; Ross, S.; Forgetta, V.; Timpson, N.J.; Smith, G.D.; Polychronakos, C.; Richards, J.B. Vitamin D levels and risk of type 1 diabetes: A Mendelian randomization study. PLoS Med. 2021, 18, e1003536. [Google Scholar] [CrossRef]
- Najjar, L.; Sutherland, J.; Zhou, A.; Hyppönen, E. Vitamin D and Type 1 Diabetes Risk: A Systematic Review and Meta-Analysis of Genetic Evidence. Nutrients 2021, 13, 4260. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Song, A.; Jin, Y.; Xia, Q.; Song, G.; Xing, X. A dose–response meta-analysis between serum concentration of 25-hydroxy vitamin D and risk of type 1 diabetes mellitus. Eur. J. Clin. Nutr. 2021, 75, 1010–1023. [Google Scholar] [CrossRef]
- Nascimento, B.F.; Moreira, C.F.F.; da Fonseca, E.R.; Fedeszen, P.M.K.; de Paula, T.P.; de Sena, A.S.S.; de Almeida, N.F.A.; Bandeira Filho, O.C.S.; Curval, D.R.; Padilha, P.C. Effects of vitamin D supplementation on glycemic control of children and adolescents with type 1 diabetes mellitus: A systematic review. J. Pediatr. Endocrinol. Metab. 2022, 35, 973–988. [Google Scholar] [CrossRef] [PubMed]
- Bogdanou, D.; Penna-Martinez, M.; Filmann, N.; Chung, T.L.; Moran-Auth, Y.; Wehrle, J.; Cappel, C.; Huenecke, S.; Herrmann, E.; Koehl, U.; et al. T-lymphocyte and glycemic status after vitamin D treatment in type 1 diabetes: A randomized controlled trial with sequential crossover. Diabetes Metab. Res. Rev. 2017, 33, e2865. [Google Scholar] [CrossRef]
- Cosentino, N.; Campodonico, J.; Milazzo, V.; De Metrio, M.; Brambilla, M.; Camera, M.; Marenzi, G. Vitamin D and Cardiovascular Disease: Current Evidence and Future Perspectives. Nutrients 2021, 13, 3603. [Google Scholar] [CrossRef]
- Zittermann, A.; Trummer, C.; Theiler-Schwetz, V.; Lerchbaum, E.; März, W.; Pilz, S. Vitamin D and Cardiovascular Disease: An Updated Narrative Review. Int. J. Mol. Sci. 2021, 22, 2896. [Google Scholar] [CrossRef]
- Ford, J.A.; MacLennan, G.S.; Avenell, A.; Bolland, M.; Grey, A.; Witham, M. Cardiovascular disease and vitamin D supplementation: Trial analysis, systematic review, and meta-analysis. Am. J. Clin. Nutr. 2014, 100, 746–755. [Google Scholar] [CrossRef] [PubMed]
- Abboud, M. Vitamin D Supplementation and Blood Pressure in Children and Adolescents: A Systematic Review and Meta-Analysis. Nutrients 2020, 12, 1163. [Google Scholar] [CrossRef] [PubMed]
- Barbarawi, M.; Kheiri, B.; Zayed, Y.; Barbarawi, O.; Dhillon, H.; Swaid, B.; Yelangi, A.; Sundus, S.; Bachuwa, G.; Alkotob, M.L.; et al. Vitamin D Supplementation and Cardiovascular Disease Risks in More Than 83,000 Individuals in 21 Randomized Clinical Trials. JAMA Cardiol. 2019, 4, 765–776. [Google Scholar] [CrossRef]
- Dibaba, D.T. Effect of vitamin D supplementation on serum lipid profiles: A systematic review and meta-analysis. Nutr. Rev. 2019, 77, 890–902. [Google Scholar] [CrossRef] [PubMed]
- Wimalawansa, S.J. Causes, Benefits and Consequences of Vitamin D Deficiency. J. Community Med. Public Health 2019, 2, 110. [Google Scholar]
- Garland, C.F.; Kim, J.J.; Mohr, S.B.; Gorham, E.D.; Grant, W.B.; Giovannucci, E.L.; Baggerly, L.; Hofflich, H.; Ramsdell, J.W.; Zeng, K.; et al. Meta-analysis of all-cause mortality according to serum 25-hydroxyvitamin D. Am. J. Public Health 2014, 104, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Papadimitriou, D.T. The Big Vitamin D Mistake. J. Prev. Med. Public Health 2017, 50, 278–281. [Google Scholar] [CrossRef] [PubMed]
- Guzek, D.; Kołota, A.; Lachowicz, K.; Skolmowska, D.; Stachoń, M.; Głąbska, D. Association between Vitamin D Supplementation and Mental Health in Healthy Adults: A Systematic Review. J. Clin. Med. 2021, 10, 5156. [Google Scholar] [CrossRef]
- Parva, N.R.; Tadepalli, S.; Singh, P.; Qian, A.; Joshi, R.; Kandala, H.; Nookala, V.K.; Cheriyath, P. Prevalence of Vitamin D Deficiency and Associated Risk Factors in the US Population (2011–2012). Cureus 2018, 10, e2741. [Google Scholar] [CrossRef]
- Department of Economic and Social Affairs Population Division. World Population Ageing 2017. Available online: https://www.un.org/en/development/desa/population/publications/pdf/ageing/WPA2017_Report.pdf (accessed on 12 April 2023).
- National Institutes of Health. Vitamin D. Available online: https://ods.od.nih.gov/factsheets/VitaminD-HealthProfessional/ (accessed on 12 April 2023).
- Sizar, O.; Khare, S.; Goyal, A.; Givler, A. Vitamin D Deficiency. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2024. [Google Scholar]
- Healthline. 7 Nutritionus Foods That Are High in Vitamin D. Available online: https://www.healthline.com/nutrition/9-foods-high-in-vitamin-d#7.-Vitamin-D-fortified-foods (accessed on 15 July 2023).
- U. S. Department of Agriculture. FoodData Central. Available online: https://fdc.nal.usda.gov/ (accessed on 10 August 2023).
- Vaes, A.M.M.; Brouwer-Brolsma, E.M.; van der Zwaluw, N.L.; van Wijngaarden, J.P.; Berendsen, A.A.M.; van Schoor, N.; van der Velde, N.; Uitterlinden, A.; Lips, P.; Dhonukshe-Rutten, R.A.M.; et al. Food sources of vitamin D and their association with 25-hydroxyvitamin D status in Dutch older adults. J. Steroid Biochem. Mol. Biol. 2017, 173, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Cashman, K.D. 100 years of vitamin D: Global differences in vitamin D status and dietary intake: A review of the data. Endocr. Connect. 2022, 11, e210282. [Google Scholar] [CrossRef] [PubMed]
- Hribar, M.; Hristov, H.; Lavriša, Ž.; Koroušić Seljak, B.; Gregorič, M.; Blaznik, U.; Žmitek, K.; Pravst, I. Vitamin D Intake in Slovenian Adolescents, Adults, and the Elderly Population. Nutrients 2021, 13, 3528. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.; Plourde, H.; Bouzo, V.; Kilgour, R.D.; Cohen, T.R. Validity and Usability of a Smartphone Image-Based Dietary Assessment App Compared to 3-Day Food Diaries in Assessing Dietary Intake Among Canadian Adults: Randomized Controlled Trial. JMIR mHealth uHealth 2020, 8, e16953. [Google Scholar] [CrossRef]
- Vernia, F.; Burrelli Scotti, G.; Bertetti, N.S.; Donato, G.; Necozione, S.; Vernia, P.; Pallotta, N. Low Vitamin K and Vitamin D Dietary Intake in Patients with Inflammatory Bowel Diseases. Nutrients 2023, 15, 1678. [Google Scholar] [CrossRef] [PubMed]
- Głąbska, D.; Uroić, V.; Guzek, D.; Pavić, E.; Bival, S.; Jaworska, K.; Giljević, Z.; Lange, E. The Possibility of Applying the Vitamin D Brief Food Frequency Questionnaire as a Tool for a Country with No Vitamin D Data in Food Composition Tables. Nutrients 2018, 10, 1278. [Google Scholar] [CrossRef] [PubMed]
- Mattila, P.; Lehikoinen, K.; Kiiskinen, T.; Piironen, V. Cholecalciferol and 25-hydroxycholecalciferol content of chicken egg yolk as affected by the cholecalciferol content of feed. J. Agric. Food Chem. 1999, 47, 4089–4092. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Bonham, A.J. A predictive equation to guide vitamin D replacement dose in patients. J. Am. Board Fam. Med. 2014, 27, 495–509. [Google Scholar] [CrossRef]
- Webb, A.; Kazantzidis, A.; Kift, R.; Farrar, M.; Wilkinson, J.; Rhodes, L. Colour Counts: Sunlight and Skin Type as Drivers of Vitamin D Deficiency at UK Latitudes. Nutrients 2018, 10, 457. [Google Scholar] [CrossRef]
- World Health Organization. Nutrient Adequacy of Exclusive Breastfeeding for the Term Infant during the First Six Months of Life. Available online: https://apps.who.int/iris/bitstream/handle/10665/42519/9241562110.pdf;jsessionid=9D68A014855719F0F0E8D70C888186D6?sequence=1 (accessed on 15 April 2023).
- Martin, C.R.; Ling, P.R.; Blackburn, G.L. Review of Infant Feeding: Key Features of Breast Milk and Infant Formula. Nutrients 2016, 8, 279. [Google Scholar] [CrossRef]
- Wagner, C.L.; Greer, F.R.; American Academy of Pediatrics Section on Breastfeeding; American Academy of Pediatrics Committee on Nutrition. Prevention of Rickets and Vitamin D Deficiency in Infants, Children, and Adolescents. Pediatrics 2008, 122, 1142–1152. [Google Scholar] [CrossRef] [PubMed]
- Montoro-Huguet, M.A.; Belloc, B.; Domínguez-Cajal, M. Small and Large Intestine (I): Malabsorption of Nutrients. Nutrients 2021, 13, 1254. [Google Scholar] [CrossRef] [PubMed]
- Thani, N.S.I.A.; Khairudin, R.; Ho, J.J.; Muhamad, N.A.; Ismail, H. Vitamin D supplementation for overweight or obese adults. Cochrane Database Syst. Rev. 2019, 2019, CD011629. [Google Scholar]
- Alshahrani, F.; Aljohani, N. Vitamin D: Deficiency, sufficiency and toxicity. Nutrients 2013, 5, 3605–3616. [Google Scholar] [CrossRef] [PubMed]
- Neufingerl, N.; Eilander, A. Nutrient Intake and Status in Adults Consuming Plant-Based Diets Compared to Meat-Eaters: A Systematic Review. Nutrients 2021, 14, 29. [Google Scholar] [CrossRef] [PubMed]
- Marcus, J.F.; Shalev, S.M.; Harris, C.A.; Goodin, D.S.; Josephson, S.A. Severe Hypercalcemia Following Vitamin D Supplementation in a Patient with Multiple Sclerosis. Arch. Neurol. 2012, 69, 129–132. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.N.; Davies, J.S. A review of the growing risk of vitamin D toxicity from inappropriate practice. Br. J. Clin. Pharmacol. 2018, 84, 1121–1127. [Google Scholar] [CrossRef] [PubMed]
- Tebben, P.J.; Singh, R.J.; Kumar, R. Vitamin D-Mediated Hypercalcemia: Mechanisms, Diagnosis, and Treatment. Endocr. Rev. 2016, 37, 521–547. [Google Scholar] [CrossRef] [PubMed]
- Williams, S.; Malatesta, K.; Norris, K. Vitamin D and Chronic Kidney Disease. Ethn. Dis. 2009, 19 (Suppl. S5), S5–8-11. Available online: http://www.ncbi.nlm.nih.gov/pmc/articles/pmc2878736/ (accessed on 11 August 2023).
- Iruzubieta, P.; Terán, Á.; Crespo, J.; Fábrega, E. Vitamin D deficiency in chronic liver disease. World J. Hepatol. 2014, 6, 901–915. [Google Scholar] [CrossRef]
- Efird, J.T.; Anderson, E.J.; Jindal, C.; Redding, T.S.; Thompson, A.D.; Press, A.M.; Upchurch, J.; Williams, C.D.; Choi, Y.M.; Suzuki, A. The Interaction of Vitamin D and Corticosteroids: A Mortality Analysis of 26,508 Veterans Who Tested Positive for SARS-CoV-2. Int. J. Environ. Res. Public Health 2022, 19, 447. [Google Scholar] [CrossRef] [PubMed]
- Baughman, R.P.; Lower, E.E. Goldilocks, vitamin D and sarcoidosis. Arthritis Res. Ther. 2014, 16, 111. [Google Scholar] [CrossRef] [PubMed]
- Tripkovic, L.; Lambert, H.; Hart, K.; Smith, C.P.; Bucca, G.; Penson, S.; Chope, G.; Hyppönen, E.; Berry, J.; Vieth, R.; et al. Comparison of vitamin D2 and vitamin D3 supplementation in raising serum 25-hydroxyvitamin D status: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2012, 95, 1357–1364. [Google Scholar] [CrossRef] [PubMed]
- Vento, K.A.; Wardenaar, F.C. Third-Party Testing Nutritional Supplement Knowledge, Attitudes, and Use Among an NCAA I Collegiate Student-Athlete Population. Front. Sports Act. Living 2020, 2, 115. [Google Scholar] [CrossRef] [PubMed]
- Maroof, S.U.; Shaukat, F.; Aslam, J.; Jawaid, M. Use of Oral Vitamin-D Glass ampoule and tablet: Experience of patients and physicians. Pak. J. Med. Sci. 2017, 33, 498–501. [Google Scholar] [CrossRef] [PubMed]
- Wagner, C.L.; Shary, J.R.; Nietert, P.J.; Wahlquist, A.E.; Ebeling, M.D.; Hollis, B.W. Bioequivalence Studies of Vitamin D Gummies and Tablets in Healthy Adults: Results of a Cross-Over Study. Nutrients 2019, 11, 1023. [Google Scholar] [CrossRef]
- Dawson-Hughes, B.; Harris, S.S.; Lichtenstein, A.H.; Dolnikowski, G.; Palermo, N.J.; Rasmussen, H. Dietary Fat Increases Vitamin D-3 Absorption. J. Acad. Nutr. Diet. 2015, 115, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Rudrapal, M.; Chetia, D. Calcium and vitamin D through diet for healthy bones. Everyman’s Sci. 2017, 52, 190–195. [Google Scholar]
- Uwitonze, A.M.; Razzaque, M.S. Role of Magnesium in Vitamin D Activation and Function. J. Am. Osteopath. Assoc. 2018, 118, 181–189. [Google Scholar] [CrossRef]
- Çaykara, B.; Öztürk, G.; Mutlu, H.H.; Arslan, E. Relationship Between Vitamin D, Calcium, and Phosphorus Levels. J. Acad. Res. Med. 2020, 10, 252–257. [Google Scholar] [CrossRef]
- van Ballegooijen, A.J.; Pilz, S.; Tomaschitz, A.; Grübler, M.R.; Verheyen, N. The Synergistic Interplay between Vitamins D and K for Bone and Cardiovascular Health: A Narrative Review. Int. J. Endocrinol. 2017, 2017, 7454376. [Google Scholar] [CrossRef] [PubMed]
- Ahsan, N.; Imran, M.; Mohammed, Y.; Al Anouti, F.; Khan, M.I.; Banerjee, T.; Adnan, M.; Ashfaq, F.; Kieliszek, M.; Ashraf, S.A.; et al. Mechanistic Insight into the role of Vitamin D and Zinc in Modulating Immunity Against COVID-19: A View from an Immunological Standpoint. Biol. Trace Elem. Res. 2023, 201, 5546–5560. [Google Scholar] [CrossRef] [PubMed]
- Yee, M.M.F.; Chin, K.Y.; Ima-Nirwana, S.; Wong, S.K. Vitamin A and Bone Health: A Review on Current Evidence. Molecules 2021, 26, 1757. [Google Scholar] [CrossRef] [PubMed]
- Chlebna-Sokół, D.; Konstantynowicz, J.; Abramowicz, P.; Kulik-Rechberger, B.; Niedziela, M.; Obuchowicz, A.; Ziora, K.; Karalus-Gach, J.; Golec, J.; Michałus, I.; et al. Evidence of a significant vitamin D deficiency among 9–13-year-old Polish children: Results of a multicentre study. Eur. J. Nutr. 2019, 58, 2029–2036. [Google Scholar] [CrossRef] [PubMed]
- Płudowski, P.; Kos-Kudła, B.; Walczak, M.; Fal, A.; Zozulińska-Ziółkiewicz, D.; Sieroszewski, P.; Peregud-Pogorzelski, J.; Lauterbach, R.; Targowski, T.; Lewiński, A.; et al. Guidelines for Preventing and Treating Vitamin D Deficiency: A 2023 Update in Poland. Nutrients 2023, 15, 695. [Google Scholar] [CrossRef] [PubMed]
- Sewerynek, E.; Cieślak, K.; Janik, M.; Gowin, E.; Stuss, M. Evaluation of vitamin D concentration in a population of young, healthy women—The effects of vitamin D supplementation. Endokrynol. Pol. 2017, 68, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.B.; Boucher, B.J.; Al Anouti, F.; Pilz, S. Comparing the Evidence from Observational Studies and Randomized Controlled Trials for Nonskeletal Health Effects of Vitamin D. Nutrients 2022, 14, 3811. [Google Scholar] [CrossRef] [PubMed]
- Jolliffe, D.A.; Camargo, C.A.; Sluyter, J.D.; Aglipay, M.; Aloia, J.F.; Ganmaa, D.; Bergman, P.; Bischoff-Ferrari, H.A.; Borzutzky, A.; Damsgaard, C.T.; et al. Vitamin D supplementation to prevent acute respiratory infections: A systematic review and meta-analysis of aggregate data from randomised controlled trials. Lancet Diabetes Endocrinol. 2021, 9, 276–292. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, J.P.; Zhou, A.; Hyppönen, E. Vitamin D Deficiency Increases Mortality Risk in the UK Biobank: A Nonlinear Mendelian Randomization Study. Ann. Intern. Med. 2022, 175, 1552–1559. [Google Scholar] [CrossRef]
- Burt, L.A.; Billington, E.O.; Rose, M.S.; Raymond, D.A.; Hanley, D.A.; Boyd, S.K. Effect of High-Dose Vitamin D Supplementation on Volumetric Bone Density and Bone Strength: A Randomized Clinical Trial. JAMA 2019, 322, 736–745. [Google Scholar] [CrossRef]
- Manson, J.E.; Cook, N.R.; Lee, I.M.; Christen, W.; Bassuk, S.S.; Mora, S.; Gibson, H.; Gordon, D.; Copeland, T.; D’Agostino, D.; et al. Vitamin D Supplements and Prevention of Cancer and Cardiovascular Disease. N. Engl. J. Med. 2019, 380, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Pittas, A.G.; Dawson-Hughes, B.; Sheehan, P.; Ware, J.H.; Knowler, W.C.; Aroda, V.R.; Brodsky, I.; Ceglia, L.; Chadha, C.; Chatterjee, R.; et al. Vitamin D Supplementation and Prevention of Type 2 Diabetes. N. Engl. J. Med. 2019, 381, 520–530. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.B.; Bhattoa, H.P.; Boucher, B.J. Seasonal variations of U.S. mortality rates: Roles of solar ultraviolet-B doses, vitamin D, gene expression, and infections. J. Steroid Biochem. Mol. Biol. 2017, 173, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.B.; Boucher, B.J. An Exploration of How Solar Radiation Affects the Seasonal Variation of Human Mortality Rates and the Seasonal Variation in Some Other Common Disorders. Nutrients 2022, 14, 2519. [Google Scholar] [CrossRef] [PubMed]
- Crowe, F.L.; Steur, M.; Allen, N.E.; Appleby, P.N.; Travis, R.C.; Key, T.J. Plasma concentrations of 25-hydroxyvitamin D in meat eaters, fish eaters, vegetarians and vegans: Results from the EPIC–Oxford study. Public Health Nutr. 2011, 14, 340–346. [Google Scholar] [CrossRef] [PubMed]
- Hyppönen, E.; Power, C. Hypovitaminosis D in British adults at age 45 y: Nationwide cohort study of dietary and lifestyle predictors. Am. J. Clin. Nutr. 2007, 85, 860–868. [Google Scholar] [CrossRef] [PubMed]
- Kroll, M.H.; Bi, C.; Garber, C.C.; Kaufman, H.W.; Liu, D.; Caston-Balderrama, A.; Zhang, K.; Clarke, N.; Xie, M.; Reitz, R.E.; et al. Temporal Relationship between Vitamin D Status and Parathyroid Hormone in the United States. PLoS ONE 2015, 10, e0118108. [Google Scholar] [CrossRef] [PubMed]
- Kmieć, P.; Żmijewski, M.; Lizakowska-Kmieć, M.; Sworczak, K. Widespread vitamin D deficiency among adults from northern Poland (54° N) after months of low and high natural UVB radiation. Endokrynol. Pol. 2015, 66, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Mason, R.S.; Rybchyn, M.S.; Abboud, M.; Brennan-Speranza, T.C.; Fraser, D.R. The Role of Skeletal Muscle in Maintaining Vitamin D Status in Winter. Curr. Dev. Nutr. 2019, 3, nzz087. [Google Scholar] [CrossRef]
- Rybchyn, M.S.; Abboud, M.; Puglisi, D.A.; Gordon-Thomson, C.; Brennan-Speranza, T.C.; Mason, R.S.; Fraser, D.R. Skeletal Muscle and the Maintenance of Vitamin D Status. Nutrients 2020, 12, 3270. [Google Scholar] [CrossRef]
- Karlic, H.; Krammer, U.; Haslberger, A. Nutritional supplements for athletes and personalization; a short review. Funct. Food Sci. 2022, 2, 224–241. [Google Scholar] [CrossRef]
- Seçkin, A.Ç.; Ateş, B.; Seçkin, M. Review on Wearable Technology in Sports: Concepts, Challenges, and Opportunities. Appl. Sci. 2023, 13, 10399. [Google Scholar] [CrossRef]
- Umpierrez, G.E.; Klonoff, D.C. Diabetes Technology Update: Use of Insulin Pumps and Continuous Glucose Monitoring in the Hospital. Diabetes Care 2018, 41, 1579–1589. [Google Scholar] [CrossRef] [PubMed]
- Dalle Carbonare, L.; Valenti, M.; del Forno, F.; Caneva, E.; Pietrobelli, A. Vitamin D: Daily vs. Monthly Use in Children and Elderly—What Is Going On? Nutrients 2017, 9, 652. [Google Scholar] [CrossRef]
- Dałek, P.; Drabik, D.; Wołczańska, H.; Foryś, A.; Jagas, M.; Jędruchniewicz, N.; Przybyło, M.; Witkiewicz, W.; Langner, M. Bioavailability by design—Vitamin D3 liposomal delivery vehicles. Nanomedicine 2022, 43, 102552. [Google Scholar] [CrossRef]
| Food Source | The Amount of Vitamin D (μg/100 g) |
|---|---|
| Fish oil, cod liver Fish, mackerel, Atlantic, raw Fish, swordfish, raw Fish, salmon. Pink, raw Fish oil, sardine Fish, sardines, canned Fish, trout, mixed species, raw Fish, tuna, raw Fish, shark, mixed species, raw Liver, beef Mushrooms, shiitake, raw Egg, yolk, raw, fresh Milk, low-fat (1% milk fat), with added vitamin D Yogurt, nonfat milk, plain Soy milk Orange juice, chilled, includes from concentrate, with added calcium and vitamin D Margarine, NFS Butter, NFS Cheese, cheddar Cheese, feta | 250.0 16.1 13.9 10.9 8.3 4.8 3.9 1.7 0.6 1.2 0.5 5.5 1.0 1.2 0.7 1.0 3.7 0.4 0.6 0.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mavar, M.; Sorić, T.; Bagarić, E.; Sarić, A.; Matek Sarić, M. The Power of Vitamin D: Is the Future in Precision Nutrition through Personalized Supplementation Plans? Nutrients 2024, 16, 1176. https://doi.org/10.3390/nu16081176
Mavar M, Sorić T, Bagarić E, Sarić A, Matek Sarić M. The Power of Vitamin D: Is the Future in Precision Nutrition through Personalized Supplementation Plans? Nutrients. 2024; 16(8):1176. https://doi.org/10.3390/nu16081176
Chicago/Turabian StyleMavar, Mladen, Tamara Sorić, Ena Bagarić, Ana Sarić, and Marijana Matek Sarić. 2024. "The Power of Vitamin D: Is the Future in Precision Nutrition through Personalized Supplementation Plans?" Nutrients 16, no. 8: 1176. https://doi.org/10.3390/nu16081176
APA StyleMavar, M., Sorić, T., Bagarić, E., Sarić, A., & Matek Sarić, M. (2024). The Power of Vitamin D: Is the Future in Precision Nutrition through Personalized Supplementation Plans? Nutrients, 16(8), 1176. https://doi.org/10.3390/nu16081176






