Potential Drug–Nutrient Interactions of 45 Vitamins, Minerals, Trace Elements, and Associated Dietary Compounds with Acetylsalicylic Acid and Warfarin—A Review of the Literature
Abstract
1. Introduction
1.1. Drug–Nutrient Interactions (DNIs)
1.2. Quality of Evidence–Clinical Speculation or Theoretical Major Burden?
1.3. Acetylsalicylic Acid (ASA)—An Overview
1.4. Warfarin—An Overview
2. Methods
3. Defining the “Hidden Hunger” Essentiality of Micronutrients
4. ASA and DNIs
4.1. Reported ASA DNIs
4.1.1. Water-Soluble Vitamins
Thiamine (B1)
Niacin (B3)
Folate (B9)
Cobalamins (B12)
Ascorbic Acid (C)
4.1.2. Fat-Soluble Vitamins
Tocopherols/Tocotrienols (E)
4.1.3. Minerals
Sodium
4.1.4. Trace Elements
Iron
4.1.5. Associated Dietary Compounds
Taurine
| Nutriment | Effect on Nutrient Status or Function | Human Studies | References | ||||
|---|---|---|---|---|---|---|---|
| Number | Study Design | Number of Patients | Dosage | Result | |||
| thiamine (B1) | ↑ excretion | 1 | case report | 5 | 647–1943 mg/day aspirin (ASA) for 5 days | mean urinary excretion thiamine ↑ 50% | [154] |
| niacin (B3) | ↓ flush (PGD2/PGE2 modulation) | 2 | Interventional—4 groups, 1 control | 31 | placebo—placebo; 80 mg ASA—500 mg B3; 325 mg ASA—500 mg B3; Placebo—500 mg B3 | ↓ warmth, ↓ flushing, ↓ itching, ↓ tingling, no difference between 325 mg and 650 mg ASA | [164] |
| interventional—3 groups, 1 control | 42 | Placebo—500 mg B3; 325 mg ASA—500 mg B3; 650 mg ASA—500 mg B3 | [165] | ||||
| folate (B9) | ↑ excretion | 3 | observational—37 patients, 59 controls | 37 | non indicated high-dose ASA (rhumatoid arthritis treatment) | 65% (24/37) subnormal folate serum level (<150 mcg/mL) | [166] |
| observational | 1 | 650 mg ASA every 4 h, for 3 days | subnormal serum folate level | [169] | |||
| observational | 18 | 2.1–3 g/day ASA, from 1 day to chronic use | subnormal serum folate level (<5 ng/mL) | [171] | |||
| cobalamins (B12) | ↓ absorption | 1 | descriptive cross-sectional observational study | 255 | low dose ASA for secondary prevention of ischemic heart disease | ①14% patients <150 pmol/L serum B12 ② 30% patients 150–250 pmol/L serum B12 | [176] |
| ascorbic acid (C) | ↓ C intragastric concentration | 1 | interventional—randomized, double-blind, parallel group | 45 | 3 × 80 mg ASA for 6 days | ↓ gastric mucosa concentration per 10% | [188] |
| ↑ urinary excretion | 1 | case report | 3 | 162 mg ASA 2 times at 3 days interval | ↑ urinary excretion | [193] | |
| ↓ C leukocyte concentration | 1 | interventional | 10 | 600 mg ASA, 500 mg C | ↓ C leukocyte concentration by 114% | [195] | |
| tocopherols/tocotrienols (E) | antiplatelet potentiation of ASA | 1 | interventional | 100 | 325 mg ASA and 400 IU α-tocopherol during 2 years | platelet adhesion reduced by 40% ASA + α-tocopherol group | [205] |
| sodium | ↓ urinary excretion | 1 | interventional—2 groups, 1 control | 16 | placebo and 160 mg ASA Group 1, 80 mg and 320 mg ASA Group 2 | interacting with ACE Inhibitors from 80 mg | [214] |
| iron | ↓ serum ferritin | 2 | Interventional—from Framingham heart study, 4 groups | 913 | number of ASA per week: non-user, 1–6, 7, >7 | ↓ 25% serum ferritin from >7 ASA per week than non users | [215] |
| interventional—multiple cohorts | 170 on ASA arm; 1146 placebo arm | dose ASA unavailable—based on medical history | ↓Lower serum ferritin (median 136 mcg/L ASA, 169 mcg/L) | [216] | |||
| ↑ anemia | 1 | interventional | 19,114 | placebo and 100 mg ASA | increased incidence of anemia and decline in ferritin | [217] | |
| taurine | potentiation of ASA | 1 | interventional | 49 | 400 mg and 1600 mg/day taurine for 14 days | decreased aggregability through alteration in TXA2 release and GSH | [223] |
| ↓ urinary excretion | 1 | interventional | 6 | high dose ASA for rhumatoid arthritis | Increased excretion of taurine | [227] | |
4.2. Questionable ASA DNIs
4.2.1. Water-Soluble Vitamins
Riboflavin (B2)
Niacin (B3)
Pantothenic Acid (B5)
4.2.2. Fat-Soluble Vitamins
Retinol (A)
4.2.3. Minerals
Phosphorous
Calcium
Magnesium
Potassium
4.2.4. Trace Elements
Copper
Zinc
Selenium
Chromium
4.2.5. Associated Dietary Compounds
Lycopene
α-Carotene
β-Carotene
β-Cryptoxanthin
Astaxanthin
4.3. ASA and Metabolic Misuse of Micronutrients
4.4. Unstudied DNIs with ASA
5. Warfarin and DNIs
5.1. Reported Warfarin DNIs
5.1.1. Water-Soluble Vitamins
Niacin (B3)
Folate (B9)
Ascorbic Acid (C)
5.1.2. Fat-Soluble Vitamins
Retinol (A)
Tocopherols/Tocotrienols (E)
Calciferols (D)
K Vitamin
5.1.3. Minerals
Magnesium
Potassium
5.1.4. Associated Dietary Compounds
Astaxanthin
| Nutriment | Effect on Nutrient Status or Function | Human Studies | References | ||||
|---|---|---|---|---|---|---|---|
| Number | Study Design | Number of Patients | Dosage | Result | |||
| niacin (B3) | synergistic effect | 1 | case report | 1 | 2.5 mg warfarin/day + 1000 mg Niacin/day | INR jumped from 18 months stable INR 2.0–2.9 to 12.3 in a week | [271] |
| folate (B9) | no association with bleeding | 1 | longitudinal cohort | 719 | 86% patients in INR 2.0–3.5 | no association | [277] |
| ↑ clearance of S-7-hydroxywarfarin | 1 | interventional | 24 | 5 mg/day B9 supplementation | non significant changes in dose and INR | [278] | |
| dietary-induced Folate deficiency | 1 | observational | 114 | dose unavailable | impaired folate status in as little as 6 months | [275] | |
| ascorbic acid (C) | no interaction | 8 | observational | 57 | mean 3.3 mg/day warfarin | no significant INR change between C vitamin and warfarin dose | [287] |
| retinol (A) | possible interaction | 2 | case reports | 13 | daily mango intake from 1–6 in 2 days to one month | exact mechanism unknown, suspected A vitamin intake through mango | [289] |
| tocopherols/tocotrienols (E) | potentiate with vitamin E | 3 | observational | 566 | dose unavailable | higher serum E predictive of hemorrhagic events | [294] |
| calciferol (D) | low 25-OH D status | 1 | observational | 89 | INR 2.0–3.5 for 3 months from at least 3 consecutive visits | 25/89 normal (>30 ng/mL); 43/89 subclinical deficiency (21–29 ng/mL); 21/89 clinical deficiency (<20 ng/mL) | [298] |
| K vitamin | vascular calcification | 4 quoted in McCann/Ames | MGLA inhibition can cause vascular calcification | [313] | |||
| bone density reduction—pediatrics | 1 | observational | 70 | more than a year on warfarin | 13% of patients with BMD < 2.0 | [309] | |
| bone density reduction—adults | 1 | observational | 70 | more than a year, 1.25–8.75 mg warfarin/day | Significant decrease in lumbar spine BMD | [310] | |
| MK7 | influence INR | interventional | 18 | 10, 20, 45 mcg/day K2 MK-7 | mean lowering of INR of 40% at 10 microg, 60% at 20 microg | [319] | |
| magnesium | stabilize INR | 1 | observational—2 groups, stable and unstable INR | 169 | various | Mg significantly lower in unstable patients, most influential INR stabilization factor in the study | [321] |
| astaxanthin | influence INR | 1 | case report | 1 | warfarin 3 mg, astaxanthin 16 mg | INR jumped from 1.4 to 10.38. Probable relationship through the scale | [327] |
5.2. Questionable Warfarin DNIs
5.2.1. Water-Soluble Vitamins
Riboflavin (B2)
Cobalamins (B12)
5.2.2. Minerals
Calcium
Magnesium
Sodium
5.2.3. Trace Elements
Iron
5.2.4. Associated Dietary Compounds
Lutein
Zeaxanthin
Lycopene
β-Carotene
β-Cryptoxanthin
5.3. Coenzyme Q10
5.4. Unstudied DNIs with Warfarin
6. Discussion
7. Limitations
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bushra, R.; Aslam, N.; Khan, A. Food Drug Interactions. Oman Med. J. 2011, 26, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Vuong, M.; González Aragón, C.; Montarroyos, S.S. Common Food and Drug Interactions. Pediatr. Rev. 2023, 44, 68–80. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Zhu, X.; Chen, Z.; Fan, C.H.; Kwan, H.S.; Wong, C.H.; Shek, K.Y.; Zuo, Z.; Lam, T.N. A Review of Food–Drug Interactions on Oral Drug Absorption. Drugs 2017, 77, 1833–1855. [Google Scholar] [CrossRef] [PubMed]
- Roe, D.A. Diet and Drug Interactions; Springer: Dordrecht, The Netherlands, 1989. [Google Scholar] [CrossRef]
- Fugh-Berman, A. Herb-Drug Interactions. Lancet 2000, 355, 134–138. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Yang, X.; Ho, P.C.L.; Chan, S.Y.; Heng, P.W.S.; Chan, E.; Duan, W.; Koh, H.L.; Zhou, S. Herb-Drug Interactions: A Literature Review. Drugs 2005, 65, 1239–1282. [Google Scholar] [CrossRef] [PubMed]
- Izzo, A.A.; Ernst, E. Interactions Between Herbal Medicines and Prescribed Drugs: A Systematic Review. Drugs 2001, 61, 2163–2175. [Google Scholar] [CrossRef] [PubMed]
- Izzo, A.A.; Di Carlo, G.; Borrelli, F.; Ernst, E. Cardiovascular Pharmacotherapy and Herbal Medicines: The Risk of Drug Interaction. Int. J. Cardiol. 2005, 98, 1–14. [Google Scholar] [CrossRef]
- Karadima, V.; Kraniotou, C.; Bellos, G.; Tsangaris, G.T. Drug-Micronutrient Interactions: Food for Thought and Thought for Action. EPMA J. 2016, 7, 10. [Google Scholar] [CrossRef] [PubMed]
- Samaras, D.; Samaras, N.; Lang, P.-O.; Genton, L.; Frangos, E.; Pichard, C. Effects of Widely Used Drugs on Micronutrients: A Story Rarely Told. Nutrition 2013, 29, 605–610. [Google Scholar] [CrossRef]
- Chan, L. Drug-Nutrient Interactions. J. Parenter. Enter. Nutr. 2013, 37, 450–459. [Google Scholar] [CrossRef]
- Santos, C.A.; Boullata, J.I. An Approach to Evaluating Drug-Nutrient Interactions. Pharmacotherapy 2005, 25, 1789–1800. [Google Scholar] [CrossRef] [PubMed]
- Mohn, E.; Kern, H.; Saltzman, E.; Mitmesser, S.; McKay, D. Evidence of Drug–Nutrient Interactions with Chronic Use of Commonly Prescribed Medications: An Update. Pharmaceutics 2018, 10, 36. [Google Scholar] [CrossRef] [PubMed]
- Roe, D.A. Drug and Food Interactions as They Affect the Nutrition of Older Individuals. Aging Milan Italy 1993, 5, 51–53. [Google Scholar] [PubMed]
- Chan, L.-N. Drug-Nutrient Interaction in Clinical Nutrition. Curr. Opin. Clin. Nutr. Metab. Care 2002, 5, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Gröber, U. Interactions between drugs and micronutrients. Med. Monatsschr. Pharm. 2006, 29, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Gunturu, S.G.; Dharmarajan, T.S.; Dharmarajan, T.S.; Dharmarajan, T.S.; Dharmarajan, T.S. Drug–Nutrient Interactions. In Geriatric Gastroenterology; Pitchumoni, C.S., Dharmarajan, T.S., Eds.; Springer: New York, NY, USA, 2012; pp. 89–98. [Google Scholar] [CrossRef]
- Boullata, J.I. Influence of Medication on Nutritional Status. In Preventive Nutrition; Bendich, A., Deckelbaum, R.J., Eds.; Humana Press: Totowa, NJ, USA, 2005; pp. 833–868. [Google Scholar] [CrossRef]
- Said, H.M.; Redha, R.; Nylander, W. Biotin Transport in the Human Intestine: Inhibition by Anticonvulsant Drugs. Am. J. Clin. Nutr. 1989, 49, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Mock, D.M.; Dyken, M.E. Biotin Catabolism Is Accelerated in Adults Receiving Long-Term Therapy with Anticonvulsants. Neurology 1997, 49, 1444–1447. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, L.R.; Kreys, E.; Gerry, S.; Boullata, J.I. Nutrition in the Age of Polypharmacy. In Preventive Nutrition; Bendich, A., Deckelbaum, R.J., Eds.; Humana Press: Totowa, NJ, USA, 2010; pp. 79–123. [Google Scholar] [CrossRef]
- Prescott, J.D.; Drake, V.J.; Stevens, J.F. Medications and Micronutrients: Identifying Clinically Relevant Interactions and Addressing Nutritional Needs. J. Pharm. Technol. 2018, 34, 216–230. [Google Scholar] [CrossRef] [PubMed]
- Boullata, J.I. Drug and Nutrition Interactions: Not Just Food for Thought. J. Clin. Pharm. Ther. 2013, 38, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Genser, D. Food and Drug Interaction: Consequences for the Nutrition/Health Status. Ann. Nutr. Metab. 2008, 52 (Suppl. S1), 29–32. [Google Scholar] [CrossRef] [PubMed]
- White, R. Drugs and Nutrition: How Side Effects Can Influence Nutritional Intake. Proc. Nutr. Soc. 2010, 69, 558–564. [Google Scholar] [CrossRef] [PubMed]
- Wallace, K.B. Drug-Induced Mitochondrial Neuropathy in Children: A Conceptual Framework for Critical Windows of Development. J. Child Neurol. 2014, 29, 1241–1248. [Google Scholar] [CrossRef]
- Mason, P. Important Drug–Nutrient Interactions. Proc. Nutr. Soc. 2010, 69, 551–557. [Google Scholar] [CrossRef] [PubMed]
- Thurnham, D.I. An Overview of Interactions between Micronutrients and of Micronutrients with Drugs, Genes and Immune Mechanisms. Nutr. Res. Rev. 2004, 17, 211–240. [Google Scholar] [CrossRef] [PubMed]
- Polk, R.E.; Healy, D.P.; Sahai, J.; Drwal, L.; Racht, E. Effect of Ferrous Sulfate and Multivitamins with Zinc on Absorption of Ciprofloxacin in Normal Volunteers. Antimicrob. Agents Chemother. 1989, 33, 1841–1844. [Google Scholar] [CrossRef] [PubMed]
- Campbell, N.; Kara, M.; Hasinoff, B.; Haddara, W.; McKay, D. Norfloxacin Interaction with Antacids and Minerals. Br. J. Clin. Pharmacol. 1992, 33, 115–116. [Google Scholar] [CrossRef] [PubMed]
- Btaiche, I.F.; Sweet, B.V.; Kraft, M.D. Positive Drug–Nutrient Interactions. In Handbook of Drug-Nutrient Interactions; Boullata, J.I., Armenti, V.T., Eds.; Humana Press: Totowa, NJ, USA, 2009; pp. 303–339. [Google Scholar] [CrossRef]
- Proell, P.; Congpuong, K.; Reinthaler, F.F.; Wernsdorfer, G.; Wiedermann, U.; Wernsdorfer, W.H. Synergism between Pyronaridine and Retinol in Plasmodium Falciparum in Vitro. Wien. Klin. Wochenschr. 2009, 121 (Suppl. S3), 32. [Google Scholar] [CrossRef] [PubMed]
- Byshchevskił, A.S.; Galiyan, S.L.; Vakulin, A.A.; Dement’eva, I.A.; SAbirov, A.X. [Influence of vitamins-antioxidants on antiaggregative effect of acetylsalicylic acid]. Ukr. Biokhimicheskii Zhurnal 1978 1997, 69, 93–98. [Google Scholar]
- Drain, P.K.; Kupka, R.; Mugusi, F.; Fawzi, W.W. Micronutrients in HIV-Positive Persons Receiving Highly Active Antiretroviral Therapy. Am. J. Clin. Nutr. 2007, 85, 333–345. [Google Scholar] [CrossRef] [PubMed]
- Nose, S.; Wasa, M.; Tazuke, Y.; Owari, M.; Fukuzawa, M. Cisplatin Upregulates Glutamine Transport in Human Intestinal Epithelial Cells: The Protective Mechanism of Glutamine on Intestinal Mucosa After Chemotherapy. J. Parenter. Enter. Nutr. 2010, 34, 530–537. [Google Scholar] [CrossRef] [PubMed]
- Bailey, R.L.; West, K.P., Jr.; Black, R.E. The Epidemiology of Global Micronutrient Deficiencies. Ann. Nutr. Metab. 2015, 66 (Suppl. S2), 22–33. [Google Scholar] [CrossRef] [PubMed]
- Kirkwood, T.B.L. Understanding Ageing from an Evolutionary Perspective: 4th Key Symposium: Understanding Ageing. J. Intern. Med. 2008, 263, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Kirkwood, T.B.L.; Austad, S.N. Why Do We Age? Nature 2000, 408, 233–238. [Google Scholar] [CrossRef]
- Ames, B.N. Low Micronutrient Intake May Accelerate the Degenerative Diseases of Aging through Allocation of Scarce Micronutrients by Triage. Proc. Natl. Acad. Sci. USA 2006, 103, 17589–17594. [Google Scholar] [CrossRef] [PubMed]
- McCann, J.C.; Ames, B.N. Vitamin K, an Example of Triage Theory: Is Micronutrient Inadequacy Linked to Diseases of Aging? Am. J. Clin. Nutr. 2009, 90, 889–907. [Google Scholar] [CrossRef] [PubMed]
- Kaźmierczak-Barańska, J.; Karwowski, B.T. Vitamin K Contribution to DNA Damage—Advantage or Disadvantage? A Human Health Response. Nutrients 2022, 14, 4219. [Google Scholar] [CrossRef] [PubMed]
- McCann, J.C.; Ames, B.N. Adaptive Dysfunction of Selenoproteins from the Perspective of the Triage Theory: Why Modest Selenium Deficiency May Increase Risk of Diseases of Aging. FASEB J. 2011, 25, 1793–1814. [Google Scholar] [CrossRef]
- Péter, S.; Navis, G.; de Borst, M.H.; von Schacky, C.; van Orten-Luiten, A.C.B.; Zhernakova, A.; Witkamp, R.F.; Janse, A.; Weber, P.; Bakker, S.J.L.; et al. Public Health Relevance of Drug–Nutrition Interactions. Eur. J. Nutr. 2017, 56 (Suppl. S2), 23–36. [Google Scholar] [CrossRef] [PubMed]
- Boullata, J.I.; Hudson, L.M. Drug–Nutrient Interactions: A Broad View with Implications for Practice. J. Acad. Nutr. Diet. 2012, 112, 506–517. [Google Scholar] [CrossRef] [PubMed]
- Boullata, J.I.; Armenti, V.T. (Eds.) Handbook of Drug-Nutrient Interactions; Humana Press: Totowa, NJ, USA, 2010. [Google Scholar] [CrossRef]
- Sjöqvist, F.; Böttiger, Y. Historical Perspectives: Drug Interactions—It All Began with Cheese. J. Intern. Med. 2010, 268, 512–515. [Google Scholar] [CrossRef] [PubMed]
- Drummond, D.; Kirby, A.N.; Gamston, C.E.; Westrick, S.; Saunders, J.; Lloyd, K.B. Impact of Interdisciplinary Education on Pharmacy Student Knowledge and Comfort with Counseling on Drug-Nutrient Interactions. Curr. Pharm. Teach. Learn. 2021, 13, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Desborough, M.J.R.; Keeling, D.M. The Aspirin Story—From Willow to Wonder Drug. Br. J. Haematol. 2017, 177, 674–683. [Google Scholar] [CrossRef] [PubMed]
- Tanasescu, S.; Lévesque, H.; Thuillez, C. Pharmacologie de l’aspirine. Rev. Médecine Interne 2000, 21, S18–S26. [Google Scholar] [CrossRef]
- Bjorkman, D.J. The Effect of Aspirin and Nonsteroidal Anti-Inflammatory Drugs on Prostaglandins. Am. J. Med. 1998, 105, 8S–12S. [Google Scholar] [CrossRef] [PubMed]
- Shimokawa, T.; Smith, W.L. Prostaglandin Endoperoxide Synthase. The Aspirin Acetylation Region. J. Biol. Chem. 1992, 267, 12387–12392. [Google Scholar] [CrossRef] [PubMed]
- Tacconelli, S.; Contursi, A.; Falcone, L.; Mucci, M.; D’Agostino, I.; Fullone, R.; Sacco, A.; Zucchelli, M.; Bruno, A.; Ballerini, P.; et al. Characterization of Cyclooxygenase-2 Acetylation and Prostanoid Inhibition by Aspirin in Cellular Systems. Biochem. Pharmacol. 2020, 178, 114094. [Google Scholar] [CrossRef] [PubMed]
- Schrör, K. Aspirin and Platelets: The Antiplatelet Action of Aspirin and Its Role in Thrombosis Treatment and Prophylaxis. Semin. Thromb. Hemost. 1997, 23, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Vane, J.R. Inhibition of Prostaglandin Synthesis as a Mechanism of Action for Aspirin-like Drugs. Nat. New Biol. 1971, 231, 232–235. [Google Scholar] [CrossRef] [PubMed]
- Hochgesang, G.P.; Rowlinson, S.W.; Marnett, L.J. Tyrosine-385 Is Critical for Acetylation of Cyclooxygenase-2 by Aspirin. J. Am. Chem. Soc. 2000, 122, 6514–6515. [Google Scholar] [CrossRef]
- Ajjan, R.A.; Standeven, K.F.; Khanbhai, M.; Phoenix, F.; Gersh, K.C.; Weisel, J.W.; Kearney, M.T.; Ariëns, R.A.S.; Grant, P.J. Effects of Aspirin on Clot Structure and Fibrinolysis Using a Novel In Vitro Cellular System. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 712–717. [Google Scholar] [CrossRef] [PubMed]
- Undas, A.; Brummel-Ziedins, K.E.; Mann, K.G. Antithrombotic Properties of Aspirin and Resistance to Aspirin: Beyond Strictly Antiplatelet Actions. Blood 2007, 109, 2285–2292. [Google Scholar] [CrossRef]
- Oberle, S.; Polte, T.; Abate, A.; Podhaisky, H.-P.; Schröder, H. Aspirin Increases Ferritin Synthesis in Endothelial Cells: A Novel Antioxidant Pathway. Circ. Res. 1998, 82, 1016–1020. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Wang, Y. Combined Influence of ABCB1 Genetic Polymorphism and DNA Methylation on Aspirin Resistance in Chinese Ischemic Stroke Patients. Acta Neurol. Belg. 2022, 122, 1057–1064. [Google Scholar] [CrossRef] [PubMed]
- Angiolillo, D.J.; Capodanno, D. Aspirin for Primary Prevention of Cardiovascular Disease in the 21st Century: A Review of the Evidence. Am. J. Cardiol. 2021, 144, S15–S22. [Google Scholar] [CrossRef]
- Undas, A.; Sydor, W.J.; Brummel, K.; Musial, J.; Mann, K.G.; Szczeklik, A. Aspirin Alters the Cardioprotective Effects of the Factor XIII Val34Leu Polymorphism. Circulation 2003, 107, 17–20. [Google Scholar] [CrossRef][Green Version]
- Dubois, R.W.; Melmed, G.Y.; Henning, J.M.; Bernal, M. Risk of Upper Gastrointestinal Injury and Events in Patients Treated with Cyclooxygenase (COX)-1/COX-2 Nonsteroidal Antiinflammatory Drugs (NSAIDs), COX-2 Selective NSAIDs, and Gastroprotective Cotherapy: An Appraisal of the Literature. JCR J. Clin. Rheumatol. 2004, 10, 178–189. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, A.; Pineda, M.; Venkata, K. Comprehension of Top 200 Prescribed Drugs in the US as a Resource for Pharmacy Teaching, Training and Practice. Pharmacy 2018, 6, 43. [Google Scholar] [CrossRef] [PubMed]
- Ansell, J.; Hirsh, J.; Hylek, E.; Jacobson, A.; Crowther, M.; Palareti, G. Pharmacology and Management of the Vitamin K Antagonists. Chest 2008, 133, 160S–198S. [Google Scholar] [CrossRef]
- Esmon, C.T.; Vigano-D’Angelo, S.; D’Angelo, A.; Comp, P.C. Anticoagulation Proteins C and S. Adv. Exp. Med. Biol. 1987, 214, 47–54. [Google Scholar] [CrossRef]
- Lim, G.B. Warfarin: From Rat Poison to Clinical Use. Nat. Rev. Cardiol. 2017. [Google Scholar] [CrossRef]
- Jaffer, I.H.; Weitz, J.I. Antithrombotic Drugs. In Hematology; Elsevier: Amsterdam, The Netherlands, 2018; pp. 2168–2188. [Google Scholar] [CrossRef]
- Kaminsky, L.S.; Zhang, Z.-Y. Human P450 Metabolism of Warfarin. Pharmacol. Ther. 1997, 73, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Echizen, H. Pharmacogenetics of Warfarin Elimination and Its Clinical Implications. Clin. Pharmacokinet. 2001, 40, 587–603. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Kashima, T.; Nomizo, Y.; Muramoto, N.; Shimizu, T.; Nasu, K.; Kubota, T.; Kimura, S.; Echizen, H. Metabolism of Warfarin Enantiomers in Japanese Patients with Heart Disease Having Different CYP2C9 and CYP2C19 Genotypes*. Clin. Pharmacol. Ther. 1998, 63, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Guengerich, F.P. Characterization of Human Cytochrome P450 Enzymes. FASEB J. 1992, 6, 745–748. [Google Scholar] [CrossRef] [PubMed]
- Norwood, D.A.; Parke, C.K.; Rappa, L.R. A Comprehensive Review of Potential Warfarin-Fruit Interactions. J. Pharm. Pract. 2015, 28, 561–571. [Google Scholar] [CrossRef] [PubMed]
- Okey, A.B. Enzyme Induction in the Cytochrome P-450 System. Pharmacol. Ther. 1990, 45, 241–298. [Google Scholar] [CrossRef] [PubMed]
- Rettie, A.E.; Korzekwa, K.R.; Kunze, K.L.; Lawrence, R.F.; Eddy, A.C.; Aoyama, T.; Gelboin, H.V.; Gonzalez, F.J.; Trager, W.F. Hydroxylation of Warfarin by Human cDNA-Expressed Cytochrome P-450: A Role for P-4502C9 in the Etiology of (S)-Warfarin-Drug Interactions. Chem. Res. Toxicol. 1992, 5, 54–59. [Google Scholar] [CrossRef]
- Eble, J.N.; West, B.D.; Link, K.P. A Comparison of the Isomers of Warfarin. Biochem. Pharmacol. 1966, 15, 1003–1006. [Google Scholar] [CrossRef]
- Ingelman-Sundberg, M. Polymorphism of Cytochrome P450 and Xenobiotic Toxicity. Toxicology 2002, 181–182, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Preissner, S.C.; Hoffmann, M.F.; Preissner, R.; Dunkel, M.; Gewiess, A.; Preissner, S. Polymorphic Cytochrome P450 Enzymes (CYPs) and Their Role in Personalized Therapy. PLoS ONE 2013, 8, e82562. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Wang, J.; Huang, S.-Q.; Su, H.-H.; Zhou, S.-F. Genetic Polymorphism of the Human Cytochrome P450 2C9 Gene and Its Clinical Significance. Curr. Drug Metab. 2009, 10, 781–834. [Google Scholar] [CrossRef] [PubMed]
- Herman, D.; Locatelli, I.; Grabnar, I.; Peternel, P.; Stegnar, M.; Mrhar, A.; Breskvar, K.; Dolzan, V. Influence of CYP2C9 Polymorphisms, Demographic Factors and Concomitant Drug Therapy on Warfarin Metabolism and Maintenance Dose. Pharmacogenomics J. 2005, 5, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Lindh, J.; Lundgren, S.; Holm, L.; Alfredsson, L.; Rane, A. Several-Fold Increase in Risk of Overanticoagulation by CYP2C9 Mutations. Clin. Pharmacol. Ther. 2005, 78, 540–550. [Google Scholar] [CrossRef] [PubMed]
- Uno, T.; Sugimoto, K.; Sugawara, K.; Tateishi, T. The Effect of CYP2C19 Genotypes on the Pharmacokinetics of Warfarin Enantiomers. J. Clin. Pharm. Ther. 2008, 33, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Karl, J.P.; Meydani, M.; Barnett, J.B.; Vanegas, S.M.; Barger, K.; Fu, X.; Goldin, B.; Kane, A.; Rasmussen, H.; Vangay, P.; et al. Fecal Concentrations of Bacterially Derived Vitamin K Forms Are Associated with Gut Microbiota Composition but Not Plasma or Fecal Cytokine Concentrations in Healthy Adults. Am. J. Clin. Nutr. 2017, 106, 1052–1061. [Google Scholar] [CrossRef] [PubMed]
- Aydin, S. Can Vitamin K Synthesis Altered by Dysbiosis of Microbiota Be Blamed in the Etiopathogenesis of Venous Thrombosis? Biosci. Microbiota Food Health 2017, 36, 73–74. [Google Scholar] [CrossRef] [PubMed]
- Caldwell, M.D.; Awad, T.; Johnson, J.A.; Gage, B.F.; Falkowski, M.; Gardina, P.; Hubbard, J.; Turpaz, Y.; Langaee, T.Y.; Eby, C.; et al. CYP4F2 Genetic Variant Alters Required Warfarin Dose. Blood 2008, 111, 4106–4112. [Google Scholar] [CrossRef] [PubMed]
- Wadelius, M.; Sörlin, K.; Wallerman, O.; Karlsson, J.; Yue, Q.-Y.; Magnusson, P.K.E.; Wadelius, C.; Melhus, H. Warfarin Sensitivity Related to CYP2C9, CYP3A5, ABCB1 (MDR1) and Other Factors. Pharmacogenomics J. 2004, 4, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Kuruvilla, M.; Gurk-Turner, C. A Review of Warfarin Dosing and Monitoring. Bayl. Univ. Med. Cent. Proc. 2001, 14, 305–306. [Google Scholar] [CrossRef] [PubMed]
- Davidson, M.H. Does Differing Metabolism by Cytochrome P450 Have Clinical Importance? Curr. Atheroscler. Rep. 2000, 2, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Gage, B.; Eby, C.; Johnson, J.; Deych, E.; Rieder, M.; Ridker, P.; Milligan, P.; Grice, G.; Lenzini, P.; Rettie, A.; et al. Use of Pharmacogenetic and Clinical Factors to Predict the Therapeutic Dose of Warfarin. Clin. Pharmacol. Ther. 2008, 84, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Di Minno, A.; Frigerio, B.; Spadarella, G.; Ravani, A.; Sansaro, D.; Amato, M.; Kitzmiller, J.P.; Pepi, M.; Tremoli, E.; Baldassarre, D. Old and New Oral Anticoagulants: Food, Herbal Medicines and Drug Interactions. Blood Rev. 2017, 31, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Holbrook, A.M. Systematic Overview of Warfarin and Its Drug and Food Interactions. Arch. Intern. Med. 2005, 165, 1095. [Google Scholar] [CrossRef] [PubMed]
- Hurren, K.M.; Lewis, C.L. Probable Interaction between Warfarin and Bee Pollen. Am. J. Health Syst. Pharm. 2010, 67, 2034–2037. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.; Geng, J.; Zeiler, M.; Nieckula, E.; Sandalinas, F.; Williams, A.; Young, M.F.; Suchdev, P.S. A Practical Guide to Adjust Micronutrient Biomarkers for Inflammation Using the BRINDA Method. J. Nutr. 2023, 153, 1265–1272. [Google Scholar] [CrossRef] [PubMed]
- Morris, A.L.; Mohiuddin, S.S. Biochemistry, Nutrients. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- McCormick, D.B. Micronutrient Cofactor Research with Extensions to Applications. Nutr. Res. Rev. 2002, 15, 245–262. [Google Scholar] [CrossRef] [PubMed]
- Biesalski Hans, K.; Jana, T. Micronutrients in the Life Cycle: Requirements and Sufficient Supply. NFS J. 2018, 11, 1–11. [Google Scholar] [CrossRef]
- Hrubša, M.; Siatka, T.; Nejmanová, I.; Vopršalová, M.; Kujovská Krčmová, L.; Matoušová, K.; Javorská, L.; Macáková, K.; Mercolini, L.; Remião, F.; et al. Biological Properties of Vitamins of the B-Complex, Part 1: Vitamins B1, B2, B3, and B5. Nutrients 2022, 14, 484. [Google Scholar] [CrossRef] [PubMed]
- Lonsdale, D. Thiamin(e): The Spark of Life. In Water Soluble Vitamins; Stanger, O., Ed.; Subcellular Biochemistry; Springer: Dordrecht, The Netherlands, 2012; Volume 56, pp. 199–227. [Google Scholar] [CrossRef]
- Marrs, C.; Lonsdale, D. Hiding in Plain Sight: Modern Thiamine Deficiency. Cells 2021, 10, 2595. [Google Scholar] [CrossRef] [PubMed]
- Thiamine Deficiency Disease, Dysautonomia, and High Calorie Malnutrition. ScienceDirect. Available online: http://www.sciencedirect.com:5070/book/9780128103876/thiamine-deficiency-disease-dysautonomia-and-high-calorie-malnutrition (accessed on 4 October 2023).
- Lienhart, W.-D.; Gudipati, V.; Macheroux, P. The Human Flavoproteome. Arch. Biochem. Biophys. 2013, 535, 150–162. [Google Scholar] [CrossRef]
- Balasubramaniam, S.; Christodoulou, J.; Rahman, S. Disorders of Riboflavin Metabolism. J. Inherit. Metab. Dis. 2019, 42, 608–619. [Google Scholar] [CrossRef] [PubMed]
- Hellmann, H.; Mooney, S. Vitamin B6: A Molecule for Human Health? Molecules 2010, 15, 442–459. [Google Scholar] [CrossRef] [PubMed]
- Zempleni, J.; Wijeratne, S.S.K.; Hassan, Y.I. Biotin. BioFactors 2009, 35, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Ebara, S. Nutritional Role of Folate. Congenit. Anom. 2017, 57, 138–141. [Google Scholar] [CrossRef] [PubMed]
- Shane, B. Folate and Vitamin B12 Metabolism: Overview and Interaction with Riboflavin, Vitamin B6, and Polymorphisms. Food Nutr. Bull. 2008, 29 (Suppl. S2), S5–S16. [Google Scholar] [CrossRef]
- O’Leary, F.; Samman, S. Vitamin B12 in Health and Disease. Nutrients 2010, 2, 299–316. [Google Scholar] [CrossRef] [PubMed]
- Chambial, S.; Dwivedi, S.; Shukla, K.K.; John, P.J.; Sharma, P. Vitamin C in Disease Prevention and Cure: An Overview. Indian J. Clin. Biochem. 2013, 28, 314–328. [Google Scholar] [CrossRef]
- Carr, A.; Maggini, S. Vitamin C and Immune Function. Nutrients 2017, 9, 1211. [Google Scholar] [CrossRef] [PubMed]
- Naidu, K.A. Vitamin C in Human Health and Disease Is Still a Mystery? An Overview. Nutr. J. 2003, 2, 7. [Google Scholar] [CrossRef] [PubMed]
- Peh, H.Y.; Tan, W.S.D.; Liao, W.; Wong, W.S.F. Vitamin E Therapy beyond Cancer: Tocopherol versus Tocotrienol. Pharmacol. Ther. 2016, 162, 152–169. [Google Scholar] [CrossRef]
- Sen, C.K.; Khanna, S.; Roy, S. Tocotrienols: Vitamin E beyond Tocopherols. Life Sci. 2006, 78, 2088–2098. [Google Scholar] [CrossRef] [PubMed]
- Sen, C.K.; Khanna, S.; Rink, C.; Roy, S. Tocotrienols: The Emerging Face of Natural Vitamin E. In Vitamins & Hormones; Elsevier: Amsterdam, The Netherlands, 2007; Volume 76, pp. 203–261. [Google Scholar] [CrossRef]
- Jackson, M.J. Diagnosis and Detection of Deficiencies of Micronutrients: Minerals. Br. Med. Bull. 1999, 55, 634–642. [Google Scholar] [CrossRef] [PubMed]
- Peacock, M. Calcium Metabolism in Health and Disease. Clin. J. Am. Soc. Nephrol. 2010, 5 (Suppl. S1), S23–S30. [Google Scholar] [CrossRef]
- Peacock, M. Phosphate Metabolism in Health and Disease. Calcif. Tissue Int. 2021, 108, 3–15. [Google Scholar] [CrossRef]
- Swaminathan, R. Magnesium Metabolism and Its Disorders. Clin. Biochem. Rev. 2003, 24, 47–66. [Google Scholar] [PubMed]
- Strazzullo, P.; Leclercq, C. Sodium. Adv. Nutr. 2014, 5, 188–190. [Google Scholar] [CrossRef] [PubMed]
- Weaver, C.M. Potassium and Health. Adv. Nutr. 2013, 4, 368S–377S. [Google Scholar] [CrossRef] [PubMed]
- Berend, K.; Van Hulsteijn, L.H.; Gans, R.O.B. Chloride: The Queen of Electrolytes? Eur. J. Intern. Med. 2012, 23, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Dev, S.; Babitt, J.L. Overview of Iron Metabolism in Health and Disease. Hemodial. Int. 2017, 21 (Suppl. S1), S6–S20. [Google Scholar] [CrossRef] [PubMed]
- Abbaspour, N.; Hurrell, R.; Kelishadi, R. Review on Iron and Its Importance for Human Health. J. Res. Med. Sci. Off. J. Isfahan Univ. Med. Sci. 2014, 19, 164–174. [Google Scholar]
- Uriu-Adams, J.Y.; Keen, C.L. Copper, Oxidative Stress, and Human Health. Mol. Aspects Med. 2005, 26, 268–298. [Google Scholar] [CrossRef] [PubMed]
- DiNicolantonio, J.J.; Mangan, D.; O’Keefe, J.H. Copper Deficiency May Be a Leading Cause of Ischaemic Heart Disease. Open Heart 2018, 5, e000784. [Google Scholar] [CrossRef] [PubMed]
- Wapnir, R.A. Copper Absorption and Bioavailability. Am. J. Clin. Nutr. 1998, 67, 1054S–1060S. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Han, P.; Liu, J.; Li, R.; Yin, W.; Wang, T.; Zhang, W.; James Kang, Y. Role of Copper in Regression of Cardiac Hypertrophy. Pharmacol. Ther. 2015, 148, 66–84. [Google Scholar] [CrossRef] [PubMed]
- Chasapis, C.T.; Loutsidou, A.C.; Spiliopoulou, C.A.; Stefanidou, M.E. Zinc and Human Health: An Update. Arch. Toxicol. 2012, 86, 521–534. [Google Scholar] [CrossRef]
- Maret, W.; Sandstead, H.H. Zinc Requirements and the Risks and Benefits of Zinc Supplementation. J. Trace Elem. Med. Biol. 2006, 20, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.; Arthur, J. Selenium, Selenoproteins and Human Health: A Review. Public Health Nutr. 2001, 4, 593–599. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P. The Importance of Selenium to Human Health. Lancet 2000, 356, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Hatch-McChesney, A.; Lieberman, H.R. Iodine and Iodine Deficiency: A Comprehensive Review of a Re-Emerging Issue. Nutrients 2022, 14, 3474. [Google Scholar] [CrossRef] [PubMed]
- Spence, J.D.; Yi, Q.; Hankey, G.J.B. Vitamins in Stroke Prevention: Time to Reconsider. Lancet Neurol. 2017, 16, 750–760. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine, Food and Nutrition Board; Subcommittee on Upper Reference Levels of Nutrients; Standing Committee on the Scientific Evaluation of Dietary Reference Intakes and its Panel on Folate, Other B Vitamins, and Choline. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline; National Academies Press: Washington, DC, USA, 1998; p. 6015. [Google Scholar] [CrossRef]
- Panel on Micronutrients, Food and Nutrition Board. Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc; National Academies Press: Washington, DC, USA, 2001; p. 10026. [Google Scholar] [CrossRef]
- Panel on Dietary Antioxidants and Related Compounds; Subcommittee on Upper Reference Levels of Nutrients; Subcommittee on Interpretation and Uses of Dietary Reference Intakes; Standing Committee on the Scientific Evaluation of Dietary Reference Intakes; Food and Nutrition Board; Institute of Medicine. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids; National Academies Press: Washington, DC, USA, 2000; p. 9810. [Google Scholar] [CrossRef]
- Ames, B.N. Prolonging Healthy Aging: Longevity Vitamins and Proteins. Proc. Natl. Acad. Sci. USA 2018, 115, 10836–10844. [Google Scholar] [CrossRef] [PubMed]
- Huxtable, R.J. Physiological Actions of Taurine. Physiol. Rev. 1992, 72, 101–163. [Google Scholar] [CrossRef] [PubMed]
- Jong, C.J.; Azuma, J.; Schaffer, S. Mechanism Underlying the Antioxidant Activity of Taurine: Prevention of Mitochondrial Oxidant Production. Amino Acids 2012, 42, 2223–2232. [Google Scholar] [CrossRef] [PubMed]
- Hansen, S.; Andersen, M.; Cornett, C.; Gradinaru, R.; Grunnet, N. A Role for Taurine in Mitochondrial Function. J. Biomed. Sci. 2010, 17 (Suppl. S1), S23. [Google Scholar] [CrossRef] [PubMed]
- Melville, D.B. Ergothioneine. In Vitamins & Hormones; Elsevier: Amsterdam, The Netherlands, 1959; Volume 17, pp. 155–204. [Google Scholar] [CrossRef]
- Cheah, I.K.; Tang, R.M.Y.; Yew, T.S.Z.; Lim, K.H.C.; Halliwell, B. Administration of Pure Ergothioneine to Healthy Human Subjects: Uptake, Metabolism, and Effects on Biomarkers of Oxidative Damage and Inflammation. Antioxid. Redox Signal. 2017, 26, 193–206. [Google Scholar] [CrossRef] [PubMed]
- Cheah, I.K.; Halliwell, B. Ergothioneine, Recent Developments. Redox Biol. 2021, 42, 101868. [Google Scholar] [CrossRef] [PubMed]
- Jonscher, K.R.; Chowanadisai, W.; Rucker, R.B. Pyrroloquinoline-Quinone Is More Than an Antioxidant: A Vitamin-like Accessory Factor Important in Health and Disease Prevention. Biomolecules 2021, 11, 1441. [Google Scholar] [CrossRef] [PubMed]
- Harris, C.B.; Chowanadisai, W.; Mishchuk, D.O.; Satre, M.A.; Slupsky, C.M.; Rucker, R.B. Dietary Pyrroloquinoline Quinone (PQQ) Alters Indicators of Inflammation and Mitochondrial-Related Metabolism in Human Subjects. J. Nutr. Biochem. 2013, 24, 2076–2084. [Google Scholar] [CrossRef] [PubMed]
- Fergus, C.; Barnes, D.; Alqasem, M.; Kelly, V. The Queuine Micronutrient: Charting a Course from Microbe to Man. Nutrients 2015, 7, 2897–2929. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Concepcion, M.; Avalos, J.; Bonet, M.L.; Boronat, A.; Gomez-Gomez, L.; Hornero-Mendez, D.; Limon, M.C.; Meléndez-Martínez, A.J.; Olmedilla-Alonso, B.; Palou, A.; et al. A Global Perspective on Carotenoids: Metabolism, Biotechnology, and Benefits for Nutrition and Health. Prog. Lipid Res. 2018, 70, 62–93. [Google Scholar] [CrossRef] [PubMed]
- Przybylska, S. Lycopene—A Bioactive Carotenoid Offering Multiple Health Benefits: A Review. Int. J. Food Sci. Technol. 2020, 55, 11–32. [Google Scholar] [CrossRef]
- Burri, B.J.; La Frano, M.R.; Zhu, C. Absorption, Metabolism, and Functions of β-Cryptoxanthin. Nutr. Rev. 2016, 74, 69–82. [Google Scholar] [CrossRef] [PubMed]
- Burri, B.J. Beta-Cryptoxanthin as a Source of Vitamin A: Beta-Cryptoxanthin as a Source of Vitamin A. J. Sci. Food Agric. 2015, 95, 1786–1794. [Google Scholar] [CrossRef] [PubMed]
- Higuera-Ciapara, I.; Félix-Valenzuela, L.; Goycoolea, F.M. Astaxanthin: A Review of Its Chemistry and Applications. Crit. Rev. Food Sci. Nutr. 2006, 46, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Donoso, A.; González-Durán, J.; Muñoz, A.A.; González, P.A.; Agurto-Muñoz, C. Therapeutic Uses of Natural Astaxanthin: An Evidence-Based Review Focused on Human Clinical Trials. Pharmacol. Res. 2021, 166, 105479. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H.; Da Costa, K.-A. Choline: An Essential Nutrient for Public Health. Nutr. Rev. 2009, 67, 615–623. [Google Scholar] [CrossRef]
- Wallace, T.C.; Blusztajn, J.K.; Caudill, M.A.; Klatt, K.C.; Natker, E.; Zeisel, S.H.; Zelman, K.M. Choline: The Underconsumed and Underappreciated Essential Nutrient. Nutr. Today 2018, 53, 240–253. [Google Scholar] [CrossRef]
- Kumar, N.; Goel, N. Phenolic Acids: Natural Versatile Molecules with Promising Therapeutic Applications. Biotechnol. Rep. 2019, 24, e00370. [Google Scholar] [CrossRef] [PubMed]
- Das, U. Essential Fatty Acids—A Review. Curr. Pharm. Biotechnol. 2006, 7, 467–482. [Google Scholar] [CrossRef] [PubMed]
- Das, U.N. Essential Fatty Acids: Biochemistry, Physiology and Pathology. Biotechnol. J. 2006, 1, 420–439. [Google Scholar] [CrossRef] [PubMed]
- Cholewski, M.; Tomczykowa, M.; Tomczyk, M. A Comprehensive Review of Chemistry, Sources and Bioavailability of Omega-3 Fatty Acids. Nutrients 2018, 10, 1662. [Google Scholar] [CrossRef]
- Wu, G. Amino Acids: Metabolism, Functions, and Nutrition. Amino Acids 2009, 37, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Babygirija, R.; Lamming, D.W. The Regulation of Healthspan and Lifespan by Dietary Amino Acids. Transl. Med. Aging 2021, 5, 17–30. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Wu, G. Nutritionally Nonessential Amino Acids: A Misnomer in Nutritional Sciences. Adv. Nutr. 2017, 8, 137–139. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Wang, Z.; Tang, W.H.W.; Hazen, S.L. Gut Microbe-Generated Trimethylamine N-Oxide from Dietary Choline Is Prothrombotic in Subjects. Circulation 2017, 135, 1671–1673. [Google Scholar] [CrossRef] [PubMed]
- Kyselova, Z. Toxicological Aspects of the Use of Phenolic Compounds in Disease Prevention. Interdiscip. Toxicol. 2011, 4, 173–183. [Google Scholar] [CrossRef]
- Cleland, J.B. The Effect of Salicylates (A) on the estimation of thiamine by the thiochrome method, (B) on the excretion of thiamine. Aust. J. Exp. Biol. Med. Sci. 1943, 21, 153–158. [Google Scholar] [CrossRef]
- Cleland, J.B. The effect of salicylates and thiamine deficiency on the thiamine content of rat tissue. Aust. J. Exp. Biol. Med. Sci. 1946, 24, 227–230. [Google Scholar] [CrossRef]
- Mehlman, M.A.; Tobin, R.B.; Madappally, M.M.; Hahn, H.K.J. Mode of Action of Aspirin. J. Biol. Chem. 1971, 246, 1618–1622. [Google Scholar] [CrossRef]
- Maggioni, G.F. Salicylate Therapy in Children. Arch. Dis. Child. 1948, 23, 40–47. [Google Scholar] [CrossRef][Green Version]
- Phillips, W. Is cutaneous flushing prostaglandin mediated? Lancet 1981, 317, 754–756. [Google Scholar] [CrossRef]
- Parson, H.K.; Harati, H.; Cooper, D.; Vinik, A.I. Role of Prostaglandin D2 and the Autonomic Nervous System in Niacin-Induced Flushing: PGD2 and ANS in Niacin-Induced Flushing. J. Diabetes 2013, 5, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.; Wu, T.-J.; Wu, K.K.; Sturino, C.; Metters, K.; Gottesdiener, K.; Wright, S.D.; Wang, Z.; O’Neill, G.; Lai, E.; et al. Antagonism of the Prostaglandin D2 Receptor 1 Suppresses Nicotinic Acid-Induced Vasodilation in Mice and Humans. Proc. Natl. Acad. Sci. USA 2006, 103, 6682–6687. [Google Scholar] [CrossRef] [PubMed]
- Jiang, P.; Parreno, R.; Thakkar, R.B.; Krause, S.; Doan, T.T.; Padley, R.J. Abstract P59: Aspirin Improves the Impact of Niacin-Associated Flushing on Quality of Life Measures. Circ. Cardiovasc. Qual. Outcomes 2011, 4 (Suppl. S1), AP59. [Google Scholar] [CrossRef]
- Offermanns, S. Heating Up the Cutaneous Flushing Response. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 1122–1123. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kamanna, V.S.; Ganji, S.H.; Kashyap, M.L. The Mechanism and Mitigation of Niacin-Induced Flushing. Int. J. Clin. Pract. 2009, 63, 1369–1377. [Google Scholar] [CrossRef] [PubMed]
- Whelan, A.M.; Price, S.O.; Fowler, S.F.; Hainer, B.L. The Effect of Aspirin on Niacin-Induced Cutaneous Reactions. J. Fam. Pract. 1992, 34, 165–168. [Google Scholar]
- Jungnickel, P.W.; Maloley, P.A.; Tuin, E.L.; Peddicord, T.E.; Campbell, J.R. Effect of Two Aspirin Pretreatment Regimens on Niacin-Induced Cutaneous Reactions. J. Gen. Intern. Med. 1997, 12, 591–596. [Google Scholar] [CrossRef] [PubMed]
- Omer, A.; Mowat, A.G. Nature of Anaemia in Rheumatoid Arthritis. IX. Folate Metabolism in Patients with Rheumatoid Arthritis. Ann. Rheum. Dis. 1968, 27, 414–424. [Google Scholar] [CrossRef] [PubMed]
- Deller, D.J.; Urban, E.; Ibbotson, R.N.; Horwood, J.; Milazzo, S.; Robson, H.N. Folic-Acid Deficiency in Rheumatoid Arthritis: Relation of Levels of Serum Folic-Acid Activity of Treatment with Phenylbutazone. BMJ 1966, 1, 765–767. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gough, K.R.; McCarthy, C.; Read, A.E.; Mollin, D.L.; Waters, A.H. Folic-Acid Deficiency in Rheumatoid Arthritis. BMJ 1964, 1, 212–217. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lawrence, V.A.; Loewenstein, J.E.; Eichner, E.R. Aspirin and Folate Binding: In Vivo and in Vitro Studies of Serum Binding and Urinary Excretion of Endogenous Folate. J. Lab. Clin. Med. 1984, 103, 944–948. [Google Scholar]
- Alonso-Aperte, E.; Varela-Moreiras, G. Drugs-Nutrient Interactions: A Potential Problem during Adolescence. Eur. J. Clin. Nutr. 2000, 54 (Suppl. S1), S69–S74. [Google Scholar] [CrossRef] [PubMed]
- Alter, H.J.; Zvaifler, N.J.; Rath, C.E. Interrelationship of Rheumatoid Arthritis, Folic Acid, and Aspirin. Blood 1971, 38, 405–416. [Google Scholar] [CrossRef] [PubMed]
- Baggott, J.E.; Morgan, S.L.; Ha, T.; Vaughn, W.H.; Hine, R.J. Inhibition of Folate-Dependent Enzymes by Non-Steroidal Anti-Inflammatory Drugs. Biochem. J. 1992, 282, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Ha, T.; Morgan, S.L.; Vaughn, W.H.; Eto, I.; Baggott, J.E. Detection of Inhibition of 5-Aminoimidazole-4-Carboxamide Ribotide Transformylase by Thioinosinic Acid and Azathioprine by a New Colorimetric Assay. Biochem. J. 1990, 272, 339–342. [Google Scholar] [CrossRef] [PubMed]
- Ho, G.Y.F.; Xue, X.; Cushman, M.; McKeown-Eyssen, G.; Sandler, R.S.; Ahnen, D.J.; Barry, E.L.; Saibil, F.; Bresalier, R.S.; Rohan, T.E.; et al. Antagonistic Effects of Aspirin and Folic Acid on Inflammation Markers and Subsequent Risk of Recurrent Colorectal Adenomas. JNCI J. Natl. Cancer Inst. 2009, 101, 1650–1654. [Google Scholar] [CrossRef] [PubMed]
- McCarty, M.F. High-Dose Folate May Improve Platelet Function in Acute Coronary Syndrome and Other Pathologies Associated with Increased Platelet Oxidative Stress. Med. Hypotheses 2007, 69, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Van Oijen, M.G.H.; Laheij, R.J.F.; Peters, W.H.M.; Jansen, J.B.M.J.; Verheugt, F.W.A. Association of Aspirin Use with Vitamin B12 Deficiency (Results of the BACH Study). Am. J. Cardiol. 2004, 94, 975–977. [Google Scholar] [CrossRef] [PubMed]
- Hunt, A.; Harrington, D.; Robinson, S. Vitamin B12 Deficiency. BMJ 2014, 349 (Suppl. S1), g5226. [Google Scholar] [CrossRef] [PubMed]
- Carabotti, M.; Annibale, B.; Lahner, E. Common Pitfalls in the Management of Patients with Micronutrient Deficiency: Keep in Mind the Stomach. Nutrients 2021, 13, 208. [Google Scholar] [CrossRef] [PubMed]
- Carmel, R. Malabsorption of Food Cobalamin. Baillieres Clin. Haematol. 1995, 8, 639–655. [Google Scholar] [CrossRef] [PubMed]
- Andres, E. Vitamin B12 (Cobalamin) Deficiency in Elderly Patients. Can. Med. Assoc. J. 2004, 171, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Annibale, B.; Capurso, G.; Delle Fave, G. Consequences of Helicobacter Pylori Infection on the Absorption of Micronutrients. Dig. Liver Dis. 2002, 34, S72–S77. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Smith, A.D.; Refsum, H.; Kwok, T. Effectiveness of B Vitamins and Their Interactions with Aspirin in Improving Cognitive Functioning in Older People with Mild Cognitive Impairment: Pooled Post-Hoc Analyses of Two Randomized Trials. J. Nutr. Health Aging 2021, 25, 1154–1160. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhu, Z.; Ni, Y. Interaction between Aspirin and Vitamin C with Human Serum Albumin as Binary and Ternary Systems. Spectrochim. Acta. A Mol. Biomol. Spectrosc. 2020, 236, 118356. [Google Scholar] [CrossRef] [PubMed]
- Basu, T.K. Vitamin C-Aspirin Interactions. Int. J. Vitam. Nutr. Res. Suppl. Int. Z. Vitam.-Ernahrungsforschung Suppl. 1982, 23, 83–90. [Google Scholar]
- Hollander, D.; Dadufalza, V.D.; Fairchild, P.A. Intestinal Absorption of Aspirin. Influence of pH, Taurocholate, Ascorbate, and Ethanol. J. Lab. Clin. Med. 1981, 98, 591–598. [Google Scholar] [PubMed]
- Nelson, E.W.; Lane, H.; Fabri, P.J.; Scott, B. Demonstration of Saturation Kinetics in the Intestinal Absorption of Vitamin C in Man and the Guinea Pig. J. Clin. Pharmacol. 1978, 18, 325–335. [Google Scholar] [CrossRef]
- Sahud, M. Effect of aspirin ingestion on ascorbic-acid levels in rheumatoid arthritis. Lancet 1971, 297, 937–938. [Google Scholar] [CrossRef]
- Schulz, H.-U.; Schürer, M.; Krupp, S.; Dammann, H.-G.; Timm, J.; Gessner, U. Effects of Acetylsalicylic Acid on Ascorbic Acid Concentrations in Plasma, Gastric Mucosa, Gastric Juice and Urine a Double-Blind Study in Healthy Subjects. Int. J. Clin. Pharmacol. Ther. 2004, 42, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Konturek, P.C.; Kania, J.; Hahn, E.G.; Konturek, J.W. Ascorbic Acid Attenuates Aspirin-Induced Gastric Damage: Role of Inducible Nitric Oxide Synthase. J. Physiol. Pharmacol. Off. J. Pol. Physiol. Soc. 2006, 57 (Suppl. S5), 125–136. [Google Scholar]
- Pohle, T.; Brzozowski, T.; Becker, J.C.; Van der Voort, I.R.; Markmann, A.; Konturek, S.J.; Moniczewski, A.; Domschke, W.; Konturek, J.W. Role of Reactive Oxygen Metabolites in Aspirin-Induced Gastric Damage in Humans: Gastroprotection by Vitamin C. Aliment. Pharmacol. Ther. 2001, 15, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Daş, N.; Nebioğlu, S. Vitamin C Aspirin Interactions in Laboratory Animals. J. Clin. Pharm. Ther. 1992, 17, 343–346. [Google Scholar] [CrossRef] [PubMed]
- Spitzer, J.M.; Shapiro, S. The Effect of Salicylate Medication upon the Urinary Excretion of Vitamin C. Am. J. Dig. Dis. 1948, 15, 80–84. [Google Scholar] [CrossRef] [PubMed]
- Daniels, A.L.; Everson, G.J. Influence of Acetylsalicylic Acid (Aspirin) on Urinary Excretion of Ascorbic Acid. Exp. Biol. Med. 1936, 35, 20–24. [Google Scholar] [CrossRef]
- Loh, H.S.; Watters, K.; Wilson, C.W.M. The Effects of Aspirin on the Metabolic Availability of Ascorbic Acid in Human Beings. J. Clin. Pharmacol. New Drugs 1973, 13, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Oiness, K.N. Nutritional Consequences of Drugs Used in Pediatrics. Clin. Pediatr. 1985, 24, 417–420. [Google Scholar] [CrossRef] [PubMed]
- Candelario-Jalil, E.; Akundi, R.S.; Bhatia, H.S.; Lieb, K.; Appel, K.; Muñoz, E.; Hüll, M.; Fiebich, B.L. Ascorbic Acid Enhances the Inhibitory Effect of Aspirin on Neuronal Cyclooxygenase-2-Mediated Prostaglandin E2 Production. J. Neuroimmunol. 2006, 174, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Han, S.-S.; Kim, K.; Hahm, E.-R.; Lee, S.J.; Surh, Y.-J.; Park, H.K.; Kim, W.S.; Jung, C.W.; Lee, M.H.; Park, K.; et al. L-Ascorbic Acid Represses Constitutive Activation of NF-kappaB and COX-2 Expression in Human Acute Myeloid Leukemia, HL-60. J. Cell. Biochem. 2004, 93, 257–270. [Google Scholar] [CrossRef]
- Sánchez-Moreno, C.; Paniagua, M.; Madrid, A.; Martín, A. Protective Effect of Vitamin C against the Ethanol Mediated Toxic Effects on Human Brain Glial Cells. J. Nutr. Biochem. 2003, 14, 606–613. [Google Scholar] [CrossRef] [PubMed]
- Freedman, J.E.; Farhat, J.H.; Loscalzo, J.; Keaney, J.F. α-Tocopherol Inhibits Aggregation of Human Platelets by a Protein Kinase C–Dependent Mechanism. Circulation 1996, 94, 2434–2440. [Google Scholar] [CrossRef] [PubMed]
- Steiner, M.; Anastasi, J. Vitamin E. An Inhibitor of the Platelet Release Reaction. J. Clin. Investig. 1976, 57, 732–737. [Google Scholar] [CrossRef] [PubMed]
- Agradi, E.; Petroni, A.; Socini, A.; Galli, C. In Vitro Effects of Synthetic Antioxidants and Vitamin E on Arachidonic Acid Metabolism and Thromboxane Formation in Human Platelets and on Platelet Aggregation. Prostaglandins 1981, 22, 255–266. [Google Scholar] [CrossRef] [PubMed]
- González-Correa, J.A.; Arrebola, M.M.; Guerrero, A.; Cañada, M.J.; Muñoz Marín, J.; De La Cuesta, F.S.; De La Cruz, J.P. Antioxidant and Antiplatelet Effects of the Alpha-Tocopherol–Aspirin Combination in Type 1-like Diabetic Rats. Life Sci. 2006, 79, 1405–1412. [Google Scholar] [CrossRef]
- Vatassery, G.; Morley, J.; Kuskowski, M. Vitamin E in Plasma and Platelets of Human Diabetic Patients and Control Subjects. Am. J. Clin. Nutr. 1983, 37, 641–644. [Google Scholar] [CrossRef] [PubMed]
- Liede, K.E.; Haukka, J.K.; Saxén, L.M.; Heinonen, O.P. Increased Tendency towards Gingival Bleeding Caused by Joint Effect of α-Tocopherol Supplementation and Acetylsalicylic Acid. Ann. Med. 1998, 30, 542–546. [Google Scholar] [CrossRef] [PubMed]
- Steiner, M.; Glantz, M.; Lekos, A. Vitamin E plus Aspirin Compared with Aspirin Alone in Patients with Transient Ischemic Attacks. Am. J. Clin. Nutr. 1995, 62, 1381S–1384S. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Wallmon, A.; Olsson-Mortlock, C.; Wallin, R.; Saldeen, T. Mixed Tocopherols Inhibit Platelet Aggregation in Humans: Potential Mechanisms123. Am. J. Clin. Nutr. 2003, 77, 700–706. [Google Scholar] [CrossRef] [PubMed]
- Podszun, M.; Frank, J. Vitamin E—Drug Interactions: Molecular Basis and Clinical Relevance. Nutr. Res. Rev. 2014, 27, 215–231. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, N.; Yoshida, N.; Yoshikawa, T.; Nakamuara, Y.; Ichikawa, H.; Naito, Y.; Kondo, M. Effect of Vitamin E on Aspirin-Induced Gastric Mucosal Injury in Rats. Dig. Dis. Sci. 2000, 45, 599–605. [Google Scholar] [CrossRef] [PubMed]
- Nafeeza, M.I.; Fauzee, A.M.; Kamsiah, J.; Gapor, M.T. Comparative Effects of a Tocotrienol-Rich Fraction and Tocopherol in Aspirin-Induced Gastric Lesions in Rats. Asia Pac. J. Clin. Nutr. 2002, 11, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q.; Moreland, M.; Ames, B.N.; Yin, X. A Combination of Aspirin and γ-Tocopherol Is Superior to That of Aspirin and α-Tocopherol in Anti-Inflammatory Action and Attenuation of Aspirin-Induced Adverse Effects. J. Nutr. Biochem. 2009, 20, 894–900. [Google Scholar] [CrossRef] [PubMed]
- Susic, D.; Sparks, J.C. Effects of Aspirin on Renal Sodium Excretion, Blood Pressure, and Plasma and Extracellular Fluid Volume in Salt-Loaded Rats. Prostaglandins 1975, 10, 825–831. [Google Scholar] [CrossRef] [PubMed]
- Kadokawa, T.; Hosoki, K.; Takeyama, K.; Minato, H.; Shimizu, M. Effects of Nonsteroidal Anti-Inflammatory Drugs (NSAID) on Renal Excretion of Sodium and Water, and on Body Fluid Volume in Rats. J. Pharmacol. Exp. Ther. 1979, 209, 219–224. [Google Scholar] [PubMed]
- Costa, A.C.; Reina-Couto, M.; Albino-Teixeira, A.; Sousa, T. Aspirin and Blood Pressure: Effects When Used Alone or in Combination with Antihypertensive Drugs. Rev. Port. Cardiol. 2017, 36, 551–567. [Google Scholar] [CrossRef] [PubMed]
- Juhlin, T.; Jönsson, B.A.G.; Höglund, P. Renal Effects of Aspirin Are Clearly Dose-Dependent and Are of Clinical Importance from a Dose of 160 Mg. Eur. J. Heart Fail. 2008, 10, 892–898. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, S.S.; Levy, D.; Vasan, R.S.; Wang, T.J. The Framingham Heart Study and the Epidemiology of Cardiovascular Disease: A Historical Perspective. Lancet 2014, 383, 999–1008. [Google Scholar] [CrossRef]
- Fleming, D.J.; Jacques, P.F.; Massaro, J.M.; D’Agostino, R.B., Sr.; Wilson, P.W.; Wood, R.J. Aspirin Intake and the Use of Serum Ferritin as a Measure of Iron Status. Am. J. Clin. Nutr. 2001, 74, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Milman, N.; Ovesen, L.; Byg, K.-E.; Graudal, N. Iron Status in Danes Updated 1994. I: Prevalence of Iron Deficiency and Iron Overload in 1332 Men Aged 40–70 Years. Influence of Blood Donation, Alcohol Intake, and Iron Supplementation. Ann. Hematol. 1999, 78, 393–400. [Google Scholar] [CrossRef] [PubMed]
- McQuilten, Z.K.; Thao, L.T.P.; Pasricha, S.-R.; Artz, A.S.; Bailey, M.; Chan, A.T.; Cohen, H.J.; Lockery, J.E.; Murray, A.M.; Nelson, M.R.; et al. Effect of Low-Dose Aspirin Versus Placebo on Incidence of Anemia in the Elderly: A Secondary Analysis of the Aspirin in Reducing Events in the Elderly Trial. Ann. Intern. Med. 2023, 176, 913–921. [Google Scholar] [CrossRef] [PubMed]
- Gaskell, H.; Derry, S.; Moore, R.A. Is There an Association between Low Dose Aspirin and Anemia (without Overt Bleeding)?: Narrative Review. BMC Geriatr. 2010, 10, 71. [Google Scholar] [CrossRef] [PubMed]
- Mascitelli, L.; Goldstein, M.R. Aspirin-Associated Iron Loss: An Anticancer Mechanism Even in the Short Term? Am. J. Med. 2013, 126, e11. [Google Scholar] [CrossRef] [PubMed]
- Martell, A.E.; Smith, R.M. Other Organic Ligands; Springer: Boston, MA, USA, 1977. [Google Scholar] [CrossRef]
- Davis, M.L.; Seaborn, C.D.; Stoecker, B.J. Effects of Over-the-Counter Drugs on 51chromium Retention and Urinary Excretion in Rats. Nutr. Res. 1995, 15, 201–210. [Google Scholar] [CrossRef]
- Franconi, F.; Miceli, M.; Bennardini, F.; Mattana, A.; Covarrubias, J.; Seghieri, G. Taurine Potentiates the Antiaggregatory Action of Aspirin and Indomethacin. In Taurine; Lombardini, J.B., Schaffer, S.W., Azuma, J., Eds.; Advances in Experimental Medicine and Biology; Springer: Boston, MA, USA, 1992; Volume 315, pp. 181–186. [Google Scholar] [CrossRef]
- Kurachi, M.; Hongoh, K.; Watanabe, A.; Aihara, H. Suppression of Bronchial Response to Platelet Activating Factor Following Taurine Administration. In The Biology of Taurine; Huxtable, R.J., Franconi, F., Giotti, A., Eds.; Advances in Experimental Medicine and Biology; Springer: Boston, MA, USA, 1987; Volume 217, pp. 189–198. [Google Scholar] [CrossRef]
- Roşca, A.E.; Vlădăreanu, A.-M.; Mirica, R.; Anghel-Timaru, C.-M.; Mititelu, A.; Popescu, B.O.; Căruntu, C.; Voiculescu, S.E.; Gologan, Ş.; Onisâi, M.; et al. Taurine and Its Derivatives: Analysis of the Inhibitory Effect on Platelet Function and Their Antithrombotic Potential. J. Clin. Med. 2022, 11, 666. [Google Scholar] [CrossRef]
- Kimura, T.; Yamashita, S.; Kim, K.S.; Sezaki, H. Electrophysiological Approach to the Action of Taurine on Rat Gastric Mucosa. J. Pharmacobiodyn. 1982, 5, 495–500. [Google Scholar] [CrossRef] [PubMed]
- Rylance, H.J.; Myhal, D.R. Taurine Excretion and the Influence of Drugs. Clin. Chim. Acta 1971, 35, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Baranovskii, S.F.; Bolotin, P.A. Association of Riboflavin, Caffeine, and Sodium Salicylate in Aqueous Solution. J. Appl. Spectrosc. 2007, 74, 211–218. [Google Scholar] [CrossRef]
- Scurachio, R.S.; Mattiucci, F.; Santos, W.G.; Skibsted, L.H.; Cardoso, D.R. Caffeine Metabolites Not Caffeine Protect against Riboflavin Photosensitized Oxidative Damage Related to Skin and Eye Health. J. Photochem. Photobiol. B 2016, 163, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Schoenen, J.; Lenaerts, M.; Bastings, E. High-Dose Riboflavin as a Prophylactic Treatment of Migraine: Results of an Open Pilot Study. Cephalalgia 1994, 14, 328–329. [Google Scholar] [CrossRef] [PubMed]
- Norgard, N.R.B.; Bacon, N.; Agosti, M. Oral Administration of Sustained Release Niacin Inhibits Platelet Aggregation. Curr. Clin. Pharmacol. 2016, 11, 43–46. [Google Scholar] [CrossRef] [PubMed]
- Chesney, C.M.; Elam, M.B.; Herd, J.A.; Davis, K.B.; Garg, R.; Hunninghake, D.; Kennedy, J.W.; Applegate, W.B. Effect of Niacin, Warfarin, and Antioxidant Therapy on Coagulation Parameters in Patients with Peripheral Arterial Disease in the Arterial Disease Multiple Intervention Trial (ADMIT). Am. Heart J. 2000, 140, 631–636. [Google Scholar] [CrossRef] [PubMed]
- Philipp, C.S.; Cisar, L.A.; Saidi, P.; Kostis, J.B. Effect of Niacin Supplementation on Fibrinogen Levels in Patients with Peripheral Vascular Disease. Am. J. Cardiol. 1998, 82, 697–699. [Google Scholar] [CrossRef] [PubMed]
- Rumberger, J.A.; Napolitano, J.; Azumano, I.; Kamiya, T.; Evans, M. Pantethine, a Derivative of Vitamin B5 Used as a Nutritional Supplement, Favorably Alters Low-Density Lipoprotein Cholesterol Metabolism in Low– to Moderate–Cardiovascular Risk North American Subjects: A Triple-Blinded Placebo and Diet-Controlled Investigation. Nutr. Res. 2011, 31, 608–615. [Google Scholar] [CrossRef] [PubMed]
- Hiramatsu, K.; Nozaki, H.; Arimori, S. Influence of pantethine on platelet volume, microviscosity, lipid composition and functions in diabetes mellitus with hyperlipidemia. Tokai J. Exp. Clin. Med. 1981, 6, 49–57. [Google Scholar] [PubMed]
- Maqbool, T.; Hashmi, A.S.; Shah, B.H. Effect of Chronic Administration of Aspirin, Phenobarbitone and Oxytetracycline on the Plasma Levels of Vitamin A in Albino Rats (Partially Retrieved on a Server of the World Health Organization). JPMA J. Pak. Med. Assoc. 1990, 40, 89–90. [Google Scholar] [PubMed]
- Jávor, T.; Bata, M.; Lovász, L.; Morón, F.; Nagy, L.; Patty, I.; Szabolcs, J.; Tárnok, F.; Tóth, G.; Mózsik, G. Gastric Cytoprotective Effects of Vitamin A and Other Carotenoids. Int. J. Tissue React. 1983, 5, 289–296. [Google Scholar] [PubMed]
- Mózsik, G.; Jávor, T.; Tóth, G.; Zsoldos, T.; Tigyi, A. Interrelationships between the Gastric Cytoprotective Effects of Vitamin A and Beta-Carotene and the Gastric Mucosal Superoxide Dismutase Activity in Rats. Acta Physiol. Hung. 1984, 64, 315–318. [Google Scholar] [PubMed]
- Jávor, T.; Tárnok, F.; Past, T.; Nagy, S. Cytoprotective Effect of Free Radical Scavengers against Mucosal Damage Produced by Different Antirheumatic Drugs. Int. J. Tissue React. 1986, 8, 35–40. [Google Scholar] [PubMed]
- Moses, F.M.; Kikendall, J.W.; Bowen, P.; Young, T.R. Chronic β-Carotene Supplementation Does Not Alter the Gastric Mucosal Response to Acute Aspirin Ingestion. J. Clin. Gastroenterol. 1990, 12, 267–270. [Google Scholar] [CrossRef] [PubMed]
- Olson, J.A. Provitamin A Function of Carotenoids: The Conversion of β-Carotene into Vitamin A. J. Nutr. 1989, 119, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Gomaa, A.A.; Hassan, H.A.; Ghaneimah, S.A. Effect of Aspirin and Indomethacin on the Serum and Urinary Calcium, Magnesium and Phosphate. Pharmacol. Res. 1990, 22, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Bennati, G.; Cirino, M.; Benericetti, G.; Maximova, N.; Zanier, M.; Pigato, F.; Parzianello, A.; Maestro, A.; Barbi, E.; Zanon, D. Compounded Effervescent Magnesium for Familial Hypomagnesemia: A Case Report. Pharmaceuticals 2023, 16, 785. [Google Scholar] [CrossRef] [PubMed]
- Think Healthy Group, Inc. Bioavailability of Single-Dose Magnesium Salts; Clinical Trial Registration NCT04139928; Think Healthy Group, Inc.: Washington, DC, USA, 2020. Available online: https://clinicaltrials.gov/ct2/show/NCT04139928 (accessed on 11 July 2022).
- Brumas, V. Copper(II) Interactions with Nonsteroidal Antiinflammatory Agents. I. Salicylic Acid and Acetylsalicylic Acid. J. Inorg. Biochem. 1995, 57, 191–207. [Google Scholar] [CrossRef] [PubMed]
- Chohan, Z.H.; Iqbal, M.S.; Iqbal, H.S.; Scozzafava, A.; Supuran, C.T. Transition Metal Acetylsalicylates and Their Anti-Inflammatory Activity. J. Enzyme Inhib. Med. Chem. 2002, 17, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Pearson, D.J.; Suarez-Mendez, V.J.; Day, J.P.; Miller, P.F. Selenium Status in Relation to Reduced Glutathione Peroxidase Activity in Aspirin-Sensitive Asthma. Clin. Exp. Allergy 1991, 21, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Mattmiller, S.A.; Carlson, B.A.; Sordillo, L.M. Regulation of Inflammation by Selenium and Selenoproteins: Impact on Eicosanoid Biosynthesis. J. Nutr. Sci. 2013, 2, e28. [Google Scholar] [CrossRef] [PubMed]
- Birt, D.F.; Julius, A.D. Dietary Fat and Selenium Effects on Ex Vivo Prostaglandin Production in Rat Colon, Kidney, and Blood. Nutr. Cancer 1986, 8, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Eisenmann, C.J.; Miller, R.K. The Effect of Selenium Compounds (Selenite, Selenate, Ebselen) on the Production of Thromboxane and Prostacyclin by the Human Term Placenta in Vitro. Toxicol. Appl. Pharmacol. 1995, 135, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Perona, G.; Schiavon, R.; Guidi, G.C.; Veneri, D.; Minuz, P. Selenium Dependent Glutathione Peroxidase: A Physiological Regulatory System for Platelet Function. Thromb. Haemost. 1990, 64, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Ducros, V. Chromium Metabolism: A Literature Review. Biol. Trace Elem. Res. 1992, 32, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, L.L.; Schwarz, K. Chromium (III) Binding to Serum Proteins, Specifically Siderophilin. Biochim. Biophys. Acta BBA-Gen. Subj. 1964, 90, 484–491. [Google Scholar] [CrossRef] [PubMed]
- Sparks, K.D.; Whitenack, M.L.; Stoecker, B.J.; Adeleye, B.O.; Sangiah, S. Aspirin and Buffering Compounds Affect the Absorption and Retention of 51chromium. J. Am. Diet. Assoc. 1995, 95, A18. [Google Scholar] [CrossRef]
- Yamamoto, J.; Taka, T.; Yamada, K.; Ijiri, Y.; Murakami, M.; Hirata, Y.; Naemura, A.; Hashimoto, M.; Yamashita, T.; Oiwa, K.; et al. Tomatoes Have Natural Anti-Thrombotic Effects. Br. J. Nutr. 2003, 90, 1031–1038. [Google Scholar] [CrossRef]
- Das, R.K.; Datta, T.; Biswas, D.; Duss, R.; O’Kennedy, N.; Duttaroy, A.K. Evaluation of the Equivalence of Different Intakes of Fruitflow in Affecting Platelet Aggregation and Thrombin Generation Capacity in a Randomized, Double-Blinded Pilot Study in Male Subjects. BMC Nutr. 2021, 7, 80. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Zhang, S.; Wang, H.; Bao, L.; Wu, W.; Qi, R. Fruitflow Inhibits Platelet Function by Suppressing Akt/GSK3β, Syk/PLCγ2 and P38 MAPK Phosphorylation in Collagen-Stimulated Platelets. BMC Complement. Med. Ther. 2022, 22, 75. [Google Scholar] [CrossRef] [PubMed]
- Sawardekar, S.; Patel, T.; Uchil, D. Comparative Evaluation of Antiplatelet Effect of Lycopene with Aspirin and the Effect of Their Combination on Platelet Aggregation: An in Vitro Study. Indian J. Pharmacol. 2016, 48, 26. [Google Scholar] [CrossRef] [PubMed]
- Mayne, S.T.; Cartmel, B.; Silva, F.; Kim, C.S.; Fallon, B.G.; Briskin, K.; Zheng, T.; Baum, M.; Shor-Posner, G.; Goodwin, W.J. Plasma Lycopene Concentrations in Humans Are Determined by Lycopene Intake, Plasma Cholesterol Concentrations and Selected Demographic Factors. J. Nutr. 1999, 129, 849–854. [Google Scholar] [CrossRef] [PubMed]
- Cooperstone, J.L.; Goetz, H.J.; Riedl, K.M.; Harrison, E.H.; Schwartz, S.J.; Kopec, R.E. Relative Contribution of α-Carotene to Postprandial Vitamin A Concentrations in Healthy Humans after Carrot Consumption. Am. J. Clin. Nutr. 2017, 106, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Serebruany, V.; Malinin, A.; Goodin, T.; Pashkow, F. The In Vitro Effects of Xancor, a Synthetic Astaxanthine Derivative, on Hemostatic Biomarkers in Aspirin-Naïve and Aspirin-Treated Subjects with Multiple Risk Factors for Vascular Disease. Am. J. Ther. 2010, 17, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Neupane, P.; Bhuju, S.; Thapa, N.; Bhattarai, H.K. ATP Synthase: Structure, Function and Inhibition. Biomol. Concepts 2019, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Tewari, D.; Majumdar, D.; Vallabhaneni, S.; Bera, A.K. Aspirin Induces Cell Death by Directly Modulating Mitochondrial Voltage-Dependent Anion Channel (VDAC). Sci. Rep. 2017, 7, 45184. [Google Scholar] [CrossRef] [PubMed]
- Liesa, M.; Shirihai, O.S. Mitochondrial Dynamics in the Regulation of Nutrient Utilization and Energy Expenditure. Cell Metab. 2013, 17, 491–506. [Google Scholar] [CrossRef] [PubMed]
- Wesselink, E.; Koekkoek, W.A.C.; Grefte, S.; Witkamp, R.F.; Van Zanten, A.R.H. Feeding Mitochondria: Potential Role of Nutritional Components to Improve Critical Illness Convalescence. Clin. Nutr. 2019, 38, 982–995. [Google Scholar] [CrossRef] [PubMed]
- Ames, B.N. Delaying the Mitochondrial Decay of Aging. Ann. N. Y. Acad. Sci. 2004, 1019, 406–411. [Google Scholar] [CrossRef] [PubMed]
- Mackerer, C.R.; Mehlman, M.A.; Tobin, R.B. Effects of Chronic Acetylsalicylate Administration on Several Nutritional and Biochemical Parameters in Rats Fed Diets of Varied Thiamin Content. Biochem. Med. 1973, 8, 51–60. [Google Scholar] [CrossRef]
- Ames, B.N.; Atamna, H.; Killilea, D.W. Mineral and Vitamin Deficiencies Can Accelerate the Mitochondrial Decay of Aging. Mol. Aspects Med. 2005, 26, 363–378. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.L.; Sobel, B.E.; Fujii, S. Attenuation of the Synthesis of Plasminogen Activator Inhibitor Type 1 by Niacin: A Potential Link Between Lipid Lowering and Fibrinolysis. Circulation 1995, 92, 767–772. [Google Scholar] [CrossRef]
- Johansson, J.O.; Egberg, N.; Asplund-Carlson, A.; Carlson, L.A. Nicotinic Acid Treatment Shifts the Fibrinolytic Balance Favourably and Decreases Plasma Fibrinogen in Hypertriglyceridaemic Men. J. Cardiovasc. Risk 1997, 4, 165–171. [Google Scholar] [CrossRef]
- Christopher, A. Critically Elevated INR in a Patient on Warfarin After Increase in Extended-Release Niacin Dose. Ann. Pharmacother. 2011, 45, 1450. [Google Scholar] [CrossRef]
- Shohag, M.J.I.; Wei, Y.; Yu, N.; Lu, L.; Zhang, J.; He, Z.; Patring, J.; Yang, X. Folate Content and Composition of Vegetables Commonly Consumed in China. J. Food Sci. 2012, 77, H239–H245. [Google Scholar] [CrossRef] [PubMed]
- Tulipani, S.; Romandini, S.; Suarez, J.M.A.; Capocasa, F.; Mezzetti, B.; Battino, M.; Busco, F.; Bamonti, F.; Novembrino, C. Folate Content in Different Strawberry Genotypes and Folate Status in Healthy Subjects after Strawberry Consumption. BioFactors 2008, 34, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Krajcovicová-Kudlácková, M.; Valachovicová, M.; Blazícek, P. Seasonal Folate Serum Concentrations at Different Nutrition. Cent. Eur. J. Public Health 2013, 21, 36–38. [Google Scholar] [CrossRef] [PubMed]
- Leblanc, C.; Presse, N.; Lalonde, G.; Dumas, S.; Ferland, G. Higher Vitamin K Intake Is Associated with Better INR Control and a Decreased Need for INR Tests in Long-Term Warfarin Therapy. Thromb. Res. 2014, 134, 210–212. [Google Scholar] [CrossRef] [PubMed]
- Ferland, G.; Chahine, S.; Presse, N.; Dube, M.-P.; Nigam, A.; Blostein, M.; deDenus, S.; Perreault, S.; Tardif, J.-C. Increasing Dietary Vitamin K Intake Stabilizes Anticoagulation Therapy in Warfarin-Treated Patients with a History of Instability: A 24-Week Randomized Controlled Trial (OR36-04-19). Curr. Dev. Nutr. 2019, 3 (Suppl. S1), nzz035.OR36-04-19. [Google Scholar] [CrossRef]
- Leblanc, C.; Dubé, M.-P.; Presse, N.; Dumas, S.; Nguyen, M.; Rouleau-Mailloux, É.; Perreault, S.; Ferland, G. Avoidance of Vitamin K-Rich Foods Is Common among Warfarin Users and Translates into Lower Usual Vitamin K Intakes. J. Acad. Nutr. Diet. 2016, 116, 1000–1007. [Google Scholar] [CrossRef] [PubMed]
- Sobczyńska-Malefora, A.J.; Harrington, D.; Rangarajan, S.; Kovacs, J.-A.; Shearer, M.J.; Savidge, G.F. Hyperhomocysteinemia and B-Vitamin Status after Discontinuation of Oral Anticoagulation Therapy in Patients with a History of Venous Thromboembolism. Clin. Chem. Lab. Med. 2003, 41, 1493–1497. [Google Scholar] [CrossRef] [PubMed]
- Sobczyńska-Malefora, A.; Harrington, D.J.; Lomer, M.C.; Pettitt, C.; Hamilton, S.; Rangarajan, S.; Shearer, M.J. Erythrocyte Folate and 5-Methyltetrahydrofolate Levels Decline during 6 Months of Oral Anticoagulation with Warfarin. Blood Coagul. Fibrinolysis 2009, 20, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Lind, M.; Jansson, J.-H.; Nilsson, T.K.; Johansson, L. High Homocysteine and Low Folate Plasma Concentrations Are Associated with Cardiovascular Events but Not Bleeding during Warfarin Treatment. Clin. Chem. Lab. Med. CCLM 2016, 54, 1981–1986. [Google Scholar] [CrossRef] [PubMed]
- Muszkat, M.; Bialer, O.; Blotnick, S.; Adar, L.; Xie, H.-G.; Ufer, M.; Cascorbi, I.; Caraco, Y. Effects of Folic Acid Supplementation on the Pharmacokinetics and Anticoagulant Effect of Warfarin: An Open-Label, Prospective Study of Long-Term Administration in Adults. Clin. Ther. 2010, 32, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.E. Interaction of Dietary Factors with Oral Anticoagulants: Review and Applications. J. Am. Diet. Assoc. 1995, 95, 580–584. [Google Scholar] [CrossRef] [PubMed]
- Deckert, F.W. Ascorbic Acid and Warfarin. JAMA 1973, 223, 440. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, G. Interaction of Ascorbic Acid and Warfarin. JAMA J. Am. Med. Assoc. 1971, 215, 1671. [Google Scholar] [CrossRef]
- Smith, E.C.; Skalski, R.J.; Johnson, G.C.; Rossi, G.V. Interaction of Ascorbic Acid and Warfarin. JAMA 1972, 221, 1166. [Google Scholar] [CrossRef]
- Hume, R.; Johnstone, J.M.; Weyers, E. Interaction of Ascorbic Acid and Warfarin. JAMA 1972, 219, 1479. [Google Scholar] [CrossRef] [PubMed]
- Dedichen, J. The Effect of Ascorbic Acid given to Patients on Chronic Anticoagulant Therapy. Boll. Soc. Ital. Cardiol. 1973, 18, 690–692. [Google Scholar] [PubMed]
- Weintraub, M.; Griner, P.F. Warfarin and Ascorbic Acid: Lack of Evidence for a Drug Interaction. Toxicol. Appl. Pharmacol. 1974, 28, 53–56. [Google Scholar] [CrossRef] [PubMed]
- Feetam, C.L.; Leach, R.H.; Meynell, M.J. Lack of a Clinically Important Interaction between Warfarin and Ascorbic Acid. Toxicol. Appl. Pharmacol. 1975, 31, 544–547. [Google Scholar] [CrossRef]
- Wynne, H.; Khan, T.; Avery, P.; Wood, P.; Ward, A.; Kamali, F. Dietary Related Plasma Vitamin C Concentration Has No Effect on Anticoagulation Response to Warfarin. Thromb. Res. 2006, 118, 501–504. [Google Scholar] [CrossRef] [PubMed]
- Sattar, A.; Willman, J.E.; Kolluri, R. Possible Warfarin Resistance Due to Interaction with Ascorbic Acid: Case Report and Literature Review. Am. J. Health-Syst. Pharm. AJHP Off. J. Am. Soc. Health-Syst. Pharm. 2013, 70, 782–786. [Google Scholar] [CrossRef] [PubMed]
- Monterrey-Rodríguez, J.; Feliú, J.F.; Rivera-Miranda, G.C. Interaction between Warfarin and Mango Fruit. Ann. Pharmacother. 2002, 36, 940–941. [Google Scholar] [CrossRef] [PubMed]
- Vitamin Supplements. Med. Lett. Drugs Ther. 1985, 27, 66–68.
- Yamazaki, H. Effects of Arachidonic Acid, Prostaglandins, Retinol, Retinoic Acid and Cholecalciferol on Xenobiotic Oxidations Catalysed by Human Cytochrome P450 Enzymes. Xenobiotica 1999, 29, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Garewal, H.S.; Diplock, A.T. How ???Safe??? Are Antioxidant Vitamins? Drug Saf. 1995, 13, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.M.; White, R.H. Effect of Vitamin E on the Anticoagulant Response to Warfarin. Am. J. Cardiol. 1996, 77, 545–546. [Google Scholar] [CrossRef] [PubMed]
- Pastori, D.; Carnevale, R.; Cangemi, R.; Saliola, M.; Nocella, C.; Bartimoccia, S.; Vicario, T.; Farcomeni, A.; Violi, F.; Pignatelli, P. Vitamin E Serum Levels and Bleeding Risk in Patients Receiving Oral Anticoagulant Therapy: A Retrospective Cohort Study. J. Am. Heart Assoc. 2013, 2, e000364. [Google Scholar] [CrossRef] [PubMed]
- Corrigan, J.J. The effect of vitamin e on warfarin-induced vitamin k deficiency*. Ann. N. Y. Acad. Sci. 1982, 393, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Traber, M.G. Vitamin E and K Interactions--a 50-Year-Old Problem. Nutr. Rev. 2008, 66, 624–629. [Google Scholar] [CrossRef]
- Hejazi, M.E.; Modarresi-Ghazani, F.; Hamishehkar, H.; Mesgari-Abbasi, M.; Dousti, S.; Entezari-Maleki, T. The Effect of Treatment of Vitamin D Deficiency on the Level of P-Selectin and Hs-CRP in Patients with Thromboembolism: A Pilot Randomized Clinical Trial. J. Clin. Pharmacol. 2017, 57, 40–47. [Google Scholar] [CrossRef]
- Khansari, N.; Bagheri, M.; Homayounfar, S.; Poorolajal, J.; Mehrpooya, M. Influence of Vitamin D Status on the Maintenance Dose of Warfarin in Patients Receiving Chronic Warfarin Therapy. Cardiol. Ther. 2022, 11, 421–432. [Google Scholar] [CrossRef] [PubMed]
- Mohammad, S.; Mishra, A.; Ashraf, M.Z. Emerging Role of Vitamin D and Its Associated Molecules in Pathways Related to Pathogenesis of Thrombosis. Biomolecules 2019, 9, 649. [Google Scholar] [CrossRef] [PubMed]
- Margier, M.; Collet, X.; le May, C.; Desmarchelier, C.; André, F.; Lebrun, C.; Defoort, C.; Bluteau, A.; Borel, P.; Lespine, A.; et al. ABCB1 (P-Glycoprotein) Regulates Vitamin D Absorption and Contributes to Its Transintestinal Efflux. FASEB J. 2019, 33, 2084–2094. [Google Scholar] [CrossRef] [PubMed]
- Price, P.A.; Faus, S.A.; Williamson, M.K. Warfarin-Induced Artery Calcification Is Accelerated by Growth and Vitamin D. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Card, D.J.; Gorska, R.; Harrington, D.J. Laboratory Assessment of Vitamin K Status. J. Clin. Pathol. 2020, 73, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, C.J.; Harrington, D.J. Therapeutic Warfarin Use and the Extrahepatic Functions of Vitamin K-Dependent Proteins. Br. J. Biomed. Sci. 2017, 74, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Theuwissen, E.; Smit, E.; Vermeer, C. The Role of Vitamin K in Soft-Tissue Calcification. Adv. Nutr. 2012, 3, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Roumeliotis, S.; Dounousi, E.; Eleftheriadis, T.; Liakopoulos, V. Association of the Inactive Circulating Matrix Gla Protein with Vitamin K Intake, Calcification, Mortality, and Cardiovascular Disease: A Review. Int. J. Mol. Sci. 2019, 20, 628. [Google Scholar] [CrossRef] [PubMed]
- Cirilli, I.; Orlando, P.; Silvestri, S.; Marcheggiani, F.; Dludla, P.V.; Kaesler, N.; Tiano, L. Carboxylative Efficacy of Trans and Cis MK7 and Comparison with Other Vitamin K Isomers. BioFactors Oxf. Engl. 2022, 48, 1129–1136. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Inaba, N.; Yamashita, T. MK-7 and Its Effects on Bone Quality and Strength. Nutrients 2020, 12, 965. [Google Scholar] [CrossRef]
- Margier, M.; Le May, C.; Antoine, T.; Halimi, C.; Nowicki, M.; Lespine, A.; Reboul, E. P-Glycoprotein (ABCB1) Is Involved in Vitamin K Efflux. Food Chem. 2021, 343, 128510. [Google Scholar] [CrossRef] [PubMed]
- Fawzy, A.M.; Lip, G.Y.H. Warfarin and Increased Fracture Risk? Answering the Big Question. Age Ageing 2022, 51, afab263. [Google Scholar] [CrossRef] [PubMed]
- Trost, S.; Tesfaye, N.; Harindhanavudhi, T. The Interplay between Bone and Heart Health as Reflected in Medication Effects: A Narrative Review. Womens Health 2023, 19, 174550572311655. [Google Scholar] [CrossRef] [PubMed]
- Avila, M.L.; Pullenayegum, E.; Williams, S.; Shammas, A.; Stimec, J.; Sochett, E.; Marr, K.; Brandão, L.R. Timing of Low Bone Mineral Density and Predictors of Bone Mineral Density Trajectory in Children on Long-Term Warfarin: A Longitudinal Study. Osteoporos. Int. 2016, 27, 1547–1557. [Google Scholar] [CrossRef] [PubMed]
- Rezaieyazdi, Z.; Falsoleiman, H.; Khajehdaluee, M.; Saghafi, M.; Mokhtari-Amirmajdi, E. Reduced Bone Density in Patients on Long-Term Warfarin. Int. J. Rheum. Dis. 2009, 12, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.-K.; Peng, C.C.-H.; Lin, S.-M.; Munir, K.M.; Chang, R.H.-E.; Wu, B.B.-C.; Liu, P.P.-S.; Hsu, J.-Y.; Loh, C.-H.; Tu, Y.-K. Fracture Risks in Patients Treated with Different Oral Anticoagulants: A Systematic Review and Meta-Analysis. J. Am. Heart Assoc. 2021, 10, e019618. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, T.; Kugimiya, F.; Kono, S.; Kim, Y.T.; Oda, H. Warfarin Use and Fracture Risk: An Evidence-Based Mechanistic Insight. Osteoporos. Int. 2015, 26, 1231–1232. [Google Scholar] [CrossRef] [PubMed]
- Poterucha, T.J.; Goldhaber, S.Z. Warfarin and Vascular Calcification. Am. J. Med. 2016, 129, 635.e1–635.e4. [Google Scholar] [CrossRef]
- Lee, S.J.; Lee, I.-K.; Jeon, J.-H. Vascular Calcification—New Insights into Its Mechanism. Int. J. Mol. Sci. 2020, 21, 2685. [Google Scholar] [CrossRef]
- Elango, K.; Javaid, A.; Khetarpal, B.K.; Ramalingam, S.; Kolandaivel, K.P.; Gunasekaran, K.; Ahsan, C. The Effects of Warfarin and Direct Oral Anticoagulants on Systemic Vascular Calcification: A Review. Cells 2021, 10, 773. [Google Scholar] [CrossRef] [PubMed]
- Anticoagulation Algorithm for Fontan Patients. American College of Cardiology. Available online: https://www.acc.org/Latest-in-Cardiology/Articles/2023/04/05/14/10/Anticoagulation-Algorithm-For-Fontan-Patients (accessed on 10 October 2023).
- Liu, J.; Liu, C.; Qian, C.; Abela, G.; Sun, W.; Kong, X. Ginkgo Biloba Extract EGB761 Alleviates Warfarin-Induced Aortic Valve Calcification Through the BMP2/Smad1/5/Runx2 Signaling Pathway. J. Cardiovasc. Pharmacol. 2021, 78, 411–421. [Google Scholar] [CrossRef] [PubMed]
- Haemorrhage Due to Ginkgo Biloba? Prescrire Int. 2008, 17, 19.
- Theuwissen, E.; Teunissen, K.J.; Spronk, H.M.H.; Hamulyák, K.; Ten Cate, H.; Shearer, M.J.; Vermeer, C.; Schurgers, L.J. Effect of Low-Dose Supplements of Menaquinone-7 (Vitamin K2) on the Stability of Oral Anticoagulant Treatment: Dose-Response Relationship in Healthy Volunteers. J. Thromb. Haemost. JTH 2013, 11, 1085–1092. [Google Scholar] [CrossRef] [PubMed]
- Stenton, S.B.; Bungard, T.J.; Ackman, M.L. Interactions between Warfarin and Herbal Products, Minerals, and Vitamins: A Pharmacist’s Guide. Can. J. Hosp. Pharm. 2001, 54, 186–192. [Google Scholar] [CrossRef]
- Ardahanli, I.; Akhan, O.; Celik, M. The Effect of Serum Magnesium Level on Stable Anticoagulation in Patients Using Warfarin for Various Cardiac Indications. Biol. Trace Elem. Res. 2022, 200, 4297–4302. [Google Scholar] [CrossRef] [PubMed]
- Sekiya, F.; Yoshida, M.; Yamashita, T.; Morita, T. Magnesium(II) Is a Crucial Constituent of the Blood Coagulation Cascade. J. Biol. Chem. 1996, 271, 8541–8544. [Google Scholar] [CrossRef] [PubMed]
- Gajsiewicz, J.M.; Nuzzio, K.M.; Rienstra, C.M.; Morrissey, J.H. Tissue Factor Residues That Modulate Magnesium-Dependent Rate Enhancements of the Tissue Factor/Factor VIIa Complex. Biochemistry 2015, 54, 4665–4671. [Google Scholar] [CrossRef] [PubMed]
- Han, H. The Importance of Vitamin K Monitoring with Warfarin (Coumadin) Use in Chronic Kidney Disease Patients. J. Ren. Nutr. 2015, 25, e7–e8. [Google Scholar] [CrossRef] [PubMed]
- Brookes, E.M.; Snider, J.; Hart, G.K.; Robbins, R.; Power, D.A. Serum Potassium Abnormalities in Chronic Kidney Disease: Prevalence, Patient Characteristics and Clinical Outcomes. Intern. Med. J. 2021, 51, 1906–1918. [Google Scholar] [CrossRef] [PubMed]
- Santiyanon, N.; Yeephu, S. Interaction between Warfarin and Astaxanthin: A Case Report. J. Cardiol. Cases 2019, 19, 173–175. [Google Scholar] [CrossRef]
- Zheng, Y.F.; Bae, S.H.; Kwon, M.J.; Park, J.B.; Choi, H.D.; Shin, W.G.; Bae, S.K. Inhibitory Effects of Astaxanthin, β-Cryptoxanthin, Canthaxanthin, Lutein, and Zeaxanthin on Cytochrome P450 Enzyme Activities. Food Chem. Toxicol. 2013, 59, 78–85. [Google Scholar] [CrossRef]
- Suttie, J.W.; Jackson, C.M. Prothrombin Structure, Activation, and Biosynthesis. Physiol. Rev. 1977, 57, 1–70. [Google Scholar] [CrossRef] [PubMed]
- Preusch, P.C.; Suttie, J.W. Vitamin K-Dependent Reactions in Rat Liver: Role of Flavoproteins. J. Nutr. 1981, 111, 2087–2097. [Google Scholar] [CrossRef]
- Worcester, E.M.; Sebastian, J.L.; Hiatt, J.G.; Beshensky, A.M.; Sadowski, J.A. The Effect of Warfarin on Urine Calcium Oxalate Crystal Growth Inhibition and Urinary Excretion of Calcium and Nephrocalcin. Calcif. Tissue Int. 1993, 53, 242–248. [Google Scholar] [CrossRef] [PubMed]
- McElnay, J.C.; Harron, D.W.; D’Arcy, P.F.; Collier, P.S. The Interaction of Warfarin with Antacid Constituents in the Gut. Experientia 1979, 35, 1359–1360. [Google Scholar] [CrossRef] [PubMed]
- Ambre, J.J.; Fischer, L.J. Effect of Coadministration of Aluminum and Magnesium Hydroxides on Absorption of Anticoagulants in Man. Clin. Pharmacol. Ther. 1973, 14, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Blancquaert, L.; Vervaet, C.; Derave, W. Predicting and Testing Bioavailability of Magnesium Supplements. Nutrients 2019, 11, 1663. [Google Scholar] [CrossRef] [PubMed]
- PubChem. Warfarin Sodium. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/16204922 (accessed on 10 October 2023).
- DailyMed—WARFARIN SODIUM Tablet. Available online: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=36a919ce-7a21-40c0-9af5-2299bf2b1f97 (accessed on 10 October 2023).
- Flint, S.; Taylor, E.; Beavis, J.; Becker, G.J.; Pedagogos, E. Increased Iron Requirement in Hemodialysis Patients on Antiplatelet Agents or Warfarin. Nephron Clin. Pract. 2009, 113, c38–c45. [Google Scholar] [CrossRef] [PubMed]
- Nicholls, D.P.; Shepherd, A.M.M. Altered warfarin metabolism in iron-deficient patient. Lancet 1978, 312, 215–216. [Google Scholar] [CrossRef] [PubMed]
- Gilardi, G.; Di Nardo, G. Heme Iron Centers in Cytochrome P450: Structure and Catalytic Activity. Rend. Lincei 2017, 28 (Suppl. S1), 159–167. [Google Scholar] [CrossRef]
- Hernández-Camacho, J.D.; Bernier, M.; López-Lluch, G.; Navas, P. Coenzyme Q10 Supplementation in Aging and Disease. Front. Physiol. 2018, 9, 44. [Google Scholar] [CrossRef]
- Bentinger, M.; Brismar, K.; Dallner, G. The Antioxidant Role of Coenzyme Q. Mitochondrion 2007, 7, S41–S50. [Google Scholar] [CrossRef] [PubMed]
- Sangsefidi, Z.S.; Yaghoubi, F.; Hajiahmadi, S.; Hosseinzadeh, M. The Effect of Coenzyme Q10 Supplementation on Oxidative Stress: A Systematic Review and Meta-Analysis of Randomized Controlled Clinical Trials. Food Sci. Nutr. 2020, 8, 1766–1776. [Google Scholar] [CrossRef] [PubMed]
- Combs, A.B.; Porter, T.H.; Folkers, K. Anticoagulant Activity of a Naphthoquinone Analog of Vitamin K and an Inhibitor of Coenzyme Q10- Enzyme Systems. Res. Commun. Chem. Pathol. Pharmacol. 1976, 13, 109–114. [Google Scholar] [PubMed]
- Morton, R.A. Ubiquinones, plastoquinones and vitamins K. Biol. Rev. 1971, 46, 47–96. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Chan, E. Effect of Ubidecarenone on Warfarin Anticoagulation and Pharmacokinetics of Warfarin Enantiomers in Rats. Drug Metabol. Drug Interact. 2001, 18, 99–122. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Zhou, S.; Chan, E. Effect of Coenzyme Q10 on Warfarin Hydroxylation in Rat and Human Liver Microsomes. Curr. Drug Metab. 2005, 6, 67–81. [Google Scholar] [CrossRef] [PubMed]
- Heck, A.M.; DeWitt, B.A.; Lukes, A.L. Potential Interactions between Alternative Therapies and Warfarin. Am. J. Health-Syst. Pharm. AJHP Off. J. Am. Soc. Health-Syst. Pharm. 2000, 57, 1221–1227; quiz 1228–1230. [Google Scholar] [CrossRef]
- Spigset, O. Reduced Effect of Warfarin Caused by Ubidecarenone. Lancet 1994, 344, 1372–1373. [Google Scholar] [CrossRef] [PubMed]
- Landbo, C.; Almdal, T.P. [Interaction between warfarin and coenzyme Q10]. Ugeskr. Laeger 1998, 160, 3226–3227. [Google Scholar] [PubMed]
- Engelsen, J.; Nielsen, J.D.; Hansen, K.F.W. [Effect of Coenzyme Q10 and Ginkgo biloba on warfarin dosage in patients on long-term warfarin treatment. A randomized, double-blind, placebo-controlled cross-over trial]. Ugeskr. Laeger 2003, 165, 1868–1871. [Google Scholar] [PubMed]
- Odén, A.; Fahlén, M. Oral Anticoagulation and Risk of Death: A Medical Record Linkage Study. BMJ 2002, 325, 1073–1075. [Google Scholar] [CrossRef] [PubMed]
- Fiddian-Green, R.G. Did the Anticoagulants Cause Deaths by Inducing Mitochondrial Dysfuinction? 2023. Available online: https://www.bmj.com/rapid-response/2011/10/29/did-anticoagulants-cause-deaths-inducing-mitochondrial-dysfuinction (accessed on 20 March 2024).
- Bețiu, A.M.; Noveanu, L.; Hâncu, I.M.; Lascu, A.; Petrescu, L.; Maack, C.; Elmér, E.; Muntean, D.M. Mitochondrial Effects of Common Cardiovascular Medications: The Good, the Bad and the Mixed. Int. J. Mol. Sci. 2022, 23, 13653. [Google Scholar] [CrossRef] [PubMed]
- Szabo, R.; Bodolea, C.; Mocan, T. Iron, Copper, and Zinc Homeostasis: Physiology, Physiopathology, and Nanomediated Applications. Nanomaterials 2021, 11, 2958. [Google Scholar] [CrossRef]
- Hitchings, R.; Kelly, L. Predicting and Understanding the Human Microbiome’s Impact on Pharmacology. Trends Pharmacol. Sci. 2019, 40, 495–505. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Buschmann, M.M.; Gilbert, J.A. Pharmacomicrobiomics: The Holy Grail to Variability in Drug Response? Clin. Pharmacol. Ther. 2019, 106, 317–328. [Google Scholar] [CrossRef]
- Pryor, R.; Martinez-Martinez, D.; Quintaneiro, L.; Cabreiro, F. The Role of the Microbiome in Drug Response. Annu. Rev. Pharmacol. Toxicol. 2020, 60, 417–435. [Google Scholar] [CrossRef]
- Masnoon, N.; Shakib, S.; Kalisch-Ellett, L.; Caughey, G.E. What Is Polypharmacy? A Systematic Review of Definitions. BMC Geriatr. 2017, 17, 230. [Google Scholar] [CrossRef] [PubMed]
- Bakaki, P.M.; Horace, A.; Dawson, N.; Winterstein, A.; Waldron, J.; Staley, J.; Pestana Knight, E.M.; Meropol, S.B.; Liu, R.; Johnson, H.; et al. Defining Pediatric Polypharmacy: A Scoping Review. PLoS ONE 2018, 13, e0208047. [Google Scholar] [CrossRef] [PubMed]
- Kok, W.E.; Haverkort, E.B.; Algra, Y.A.; Mollema, J.; Hollaar, V.R.Y.; Naumann, E.; Schueren, M.A.E.; de van der Jerković-Ćosić, K. The Association between Polypharmacy and Malnutrition(Risk) in Older People: A Systematic Review. Clin. Nutr. ESPEN 2022, 49, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Doan, J.; Zakrzewski-Jakubiak, H.; Roy, J.; Turgeon, J.; Tannenbaum, C. Prevalence and Risk of Potential Cytochrome P450-Mediated Drug-Drug Interactions in Older Hospitalized Patients with Polypharmacy. Ann. Pharmacother. 2013, 47, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Kaganov, B.; Caroli, M.; Mazur, A.; Singhal, A.; Vania, A. Suboptimal Micronutrient Intake among Children in Europe. Nutrients 2015, 7, 3524–3535. [Google Scholar] [CrossRef]
- Kordas, K.; Lönnerdal, B.; Stoltzfus, R.J. Interactions between Nutrition and Environmental Exposures: Effects on Health Outcomes in Women and Children. J. Nutr. 2007, 137, 2794–2797. [Google Scholar] [CrossRef] [PubMed]
- Avitabile, C.M.; Leonard, M.B.; Zemel, B.S.; Brodsky, J.L.; Lee, D.; Dodds, K.; Hayden-Rush, C.; Whitehead, K.K.; Goldmuntz, E.; Paridon, S.M.; et al. Lean Mass Deficits, Vitamin D Status and Exercise Capacity in Children and Young Adults after Fontan Palliation. Heart 2014, 100, 1702–1707. [Google Scholar] [CrossRef] [PubMed]
- Bruun, D.E.R. Vitamin D and Calcium Intake in Children with Fontan Circulation—In Relationship to Vitamin D Status and Bone Mineral Density. Master’s Thesis, University of Oslo, Oslo, Norway, 2021. [Google Scholar]
- Charpie, J.R. Vitamin C May Improve Endothelial Function and Exercise Capacity in Functional Single Ventricle Patients after Fontan Palliation; Clinical Ttrial Registration NCT00974025. 2017. Available online: https://clinicaltrials.gov/ct2/show/NCT00974025 (accessed on 8 May 2022).
- Diab, S.G.; Godang, K.; Müller, L.O.; Almaas, R.; Lange, C.; Brunvand, L.; Hansen, K.M.; Myhre, A.G.; Døhlen, G.; Thaulow, E.; et al. Progressive Loss of Bone Mass in Children with Fontan Circulation. Congenit. Heart Dis. 2019, 14, 996–1004. [Google Scholar] [CrossRef]
- Renaud, D.; Scholl-Bürgi, S.; Karall, D.; Michel, M. Comparative Metabolomics in Single Ventricle Patients after Fontan Palliation: A Strong Case for a Targeted Metabolic Therapy. Metabolites 2023, 13, 932. [Google Scholar] [CrossRef] [PubMed]
- Michel, M.; Dubowy, K.-O.; Entenmann, A.; Karall, D.; Adam, M.G.; Zlamy, M.; Odri Komazec, I.; Geiger, R.; Niederwanger, C.; Salvador, C.; et al. Targeted Metabolomic Analysis of Serum Amino Acids in the Adult Fontan Patient with a Dominant Left Ventricle. Sci. Rep. 2020, 10, 8930. [Google Scholar] [CrossRef] [PubMed]
- Michel, M.; Dubowy, K.-O.; Zlamy, M.; Karall, D.; Adam, M.G.; Entenmann, A.; Keller, M.A.; Koch, J.; Odri Komazec, I.; Geiger, R.; et al. Targeted Metabolomic Analysis of Serum Phospholipid and Acylcarnitine in the Adult Fontan Patient with a Dominant Left Ventricle. Ther. Adv. Chronic Dis. 2020, 11, 204062232091603. [Google Scholar] [CrossRef] [PubMed]
- Schranz, D.; Voelkel, N.F. “Nihilism” of Chronic Heart Failure Therapy in Children and Why Effective Therapy Is Withheld. Eur. J. Pediatr. 2016, 175, 445–455. [Google Scholar] [CrossRef]
- Nutescu, E.A.; Shapiro, N.L.; Ibrahim, S.; West, P. Warfarin and Its Interactions with Foods, Herbs and Other Dietary Supplements. Expert Opin. Drug Saf. 2006, 5, 433–451. [Google Scholar] [CrossRef] [PubMed]
- Mallet, L.; Spinewine, A.; Huang, A. The Challenge of Managing Drug Interactions in Elderly People. Lancet 2007, 370, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Lonsdale, D. Thiamine and Magnesium Deficiencies: Keys to Disease. Med. Hypotheses 2015, 84, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Stevens, G.A.; Beal, T.; Mbuya, M.N.N.; Luo, H.; Neufeld, L.M.; Addo, O.Y.; Adu-Afarwuah, S.; Alayón, S.; Bhutta, Z.; Brown, K.H.; et al. Micronutrient Deficiencies among Preschool-Aged Children and Women of Reproductive Age Worldwide: A Pooled Analysis of Individual-Level Data from Population-Representative Surveys. Lancet Glob. Health 2022, 10, e1590–e1599. [Google Scholar] [CrossRef] [PubMed]
- Kirkwood, T.B.; Rose, M.R. Evolution of Senescence: Late Survival Sacrificed for Reproduction. Philos. Trans. R. Soc. Lond. B Biol. Sci. 1991, 332, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Lamers, Y. Approaches to Improving Micronutrient Status Assessment at the Population Level. Proc. Nutr. Soc. 2019, 78, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Ervin, R.B.; Kennedy-Stephenson, J. Mineral Intakes of Elderly Adult Supplement and Non-Supplement Users in the Third National Health and Nutrition Examination Survey. J. Nutr. 2002, 132, 3422–3427. [Google Scholar] [CrossRef] [PubMed]
- Grüne, S. Anamnese und körperliche Untersuchung. DMW-Dtsch. Med. Wochenschr. 2015, 141, 24–27. [Google Scholar] [CrossRef] [PubMed]
- Horn, J.R.; Hansten, P.D.; Chan, L.-N. Proposal for a New Tool to Evaluate Drug Interaction Cases. Ann. Pharmacother. 2007, 41, 674–680. [Google Scholar] [CrossRef] [PubMed]
- Komperda, K.E. Potential Interaction Between Pomegranate Juice and Warfarin. Pharmacotherapy 2009, 29, 1002–1006. [Google Scholar] [CrossRef] [PubMed]
- Hanselin, M.R.; Vande Griend, J.P.; Linnebur, S.A. INR Elevation with Maitake Extract in Combination with Warfarin. Ann. Pharmacother. 2010, 44, 223–224. [Google Scholar] [CrossRef] [PubMed]
- Sadkovsky, I.A.; Golubnitschaja, O.; Mandrik, M.A.; Studneva, M.A.; Abe, H.; Schroeder, H.; Antonova, E.N.; Betsou, F.; Bodrova, T.A.; Payne, K.; et al. PPPM (Predictive, Preventive and Personalized Medicine) as a New Model of the National and International Healthcare Services and Thus a Promising Strategy to Prevent a Disease: From Basics to Practice. Int. J. Clin. Med. 2014, 05, 855–870. [Google Scholar] [CrossRef]
- McColl, E.R.; Asthana, R.; Paine, M.F.; Piquette-Miller, M. The Age of Omics-Driven Precision Medicine. Clin. Pharmacol. Ther. 2019, 106, 477–481. [Google Scholar] [CrossRef] [PubMed]
- Krier, J.B.; Kalia, S.S.; Green, R.C. Genomic Sequencing in Clinical Practice: Applications, Challenges, and Opportunities. Dialogues Clin. Neurosci. 2016, 18, 299–312. [Google Scholar] [CrossRef] [PubMed]
- Al-Amrani, S.; Al-Jabri, Z.; Al-Zaabi, A.; Alshekaili, J.; Al-Khabori, M. Proteomics: Concepts and Applications in Human Medicine. World J. Biol. Chem. 2021, 12, 57–69. [Google Scholar] [CrossRef] [PubMed]
- Kirwan, J.A. Translating Metabolomics into Clinical Practice. Nat. Rev. Bioeng. 2023, 1, 228–229. [Google Scholar] [CrossRef]
- Archibald, J. Genomics: A Very Short Introduction; Oxford University Press: Oxford, UK, 2018; Volume 1. [Google Scholar] [CrossRef]
- Ghosh, D.; Skinner, M.A.; Laing, W.A. Pharmacogenomics and Nutrigenomics: Synergies and Differences. Eur. J. Clin. Nutr. 2007, 61, 567–574. [Google Scholar] [CrossRef]
- Subbiah, M.T.R. Nutrigenetics and Nutraceuticals: The next Wave Riding on Personalized Medicine. Transl. Res. J. Lab. Clin. Med. 2007, 149, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Wilkins, M.R.; Sanchez, J.-C.; Gooley, A.A.; Appel, R.D.; Humphery-Smith, I.; Hochstrasser, D.F.; Williams, K.L. Progress with Proteome Projects: Why All Proteins Expressed by a Genome Should Be Identified and How to Do It. Biotechnol. Genet. Eng. Rev. 1996, 13, 19–50. [Google Scholar] [CrossRef] [PubMed]
- Clish, C.B. Metabolomics: An Emerging but Powerful Tool for Precision Medicine. Mol. Case Stud. 2015, 1, a000588. [Google Scholar] [CrossRef]
- Michel, M.; Laser, K.T.; Dubowy, K.-O.; Scholl-Bürgi, S.; Michel, E. Metabolomics and Random Forests in Patients with Complex Congenital Heart Disease. Front. Cardiovasc. Med. 2022, 9, 994068. [Google Scholar] [CrossRef]
- Patient-Specific Modelling & Bioinformatics in PPPM. EPMA J. 2011, 2 (Suppl. S1), 181–187. [CrossRef][Green Version]
- Steg, A.; Oczkowicz, M.; Smołucha, G. Omics as a Tool to Help Determine the Effectiveness of Supplements. Nutrients 2022, 14, 5305. [Google Scholar] [CrossRef] [PubMed]
- McClorry, S.; Slupsky, C.M.; Lind, T.; Karlsland Åkeson, P.; Hernell, O.; Öhlund, I. Effectiveness of Vitamin D Supplementation in Swedish Children May Be Negatively Impacted by BMI and Serum Fructose. J. Nutr. Biochem. 2020, 75, 108251. [Google Scholar] [CrossRef] [PubMed]
- von Arnim, C.A.F.; Dismar, S.; Ott-Renzer, C.S.; Noeth, N.; Ludolph, A.C.; Biesalski, H.K. Micronutrients Supplementation and Nutritional Status in Cognitively Impaired Elderly Persons: A Two-Month Open Label Pilot Study. Nutr. J. 2013, 12, 148. [Google Scholar] [CrossRef] [PubMed]
- Ortea, I. Foodomics in Health: Advanced Techniques for Studying the Bioactive Role of Foods. TrAC Trends Anal. Chem. 2022, 150, 116589. [Google Scholar] [CrossRef]
- Stouras, I.; Papaioannou, T.G.; Tsioufis, K.; Eliopoulos, A.G.; Sanoudou, D. The Challenge and Importance of Integrating Drug–Nutrient–Genome Interactions in Personalized Cardiovascular Healthcare. J. Pers. Med. 2022, 12, 513. [Google Scholar] [CrossRef] [PubMed]
- Saito, R.; Takeda, K.; Yamamoto, K.; Nakagawa, A.; Aoki, H.; Fujibayashi, K.; Wakasa, M.; Motoyama, A.; Iwadare, M.; Ishida, R.; et al. Nutri-Pharmacogenomics of Warfarin Anticoagulation Therapy: VKORC1 Genotype-Dependent Influence of Dietary Vitamin K Intake. J. Thromb. Thrombolysis 2014, 38, 105–114. [Google Scholar] [CrossRef] [PubMed]
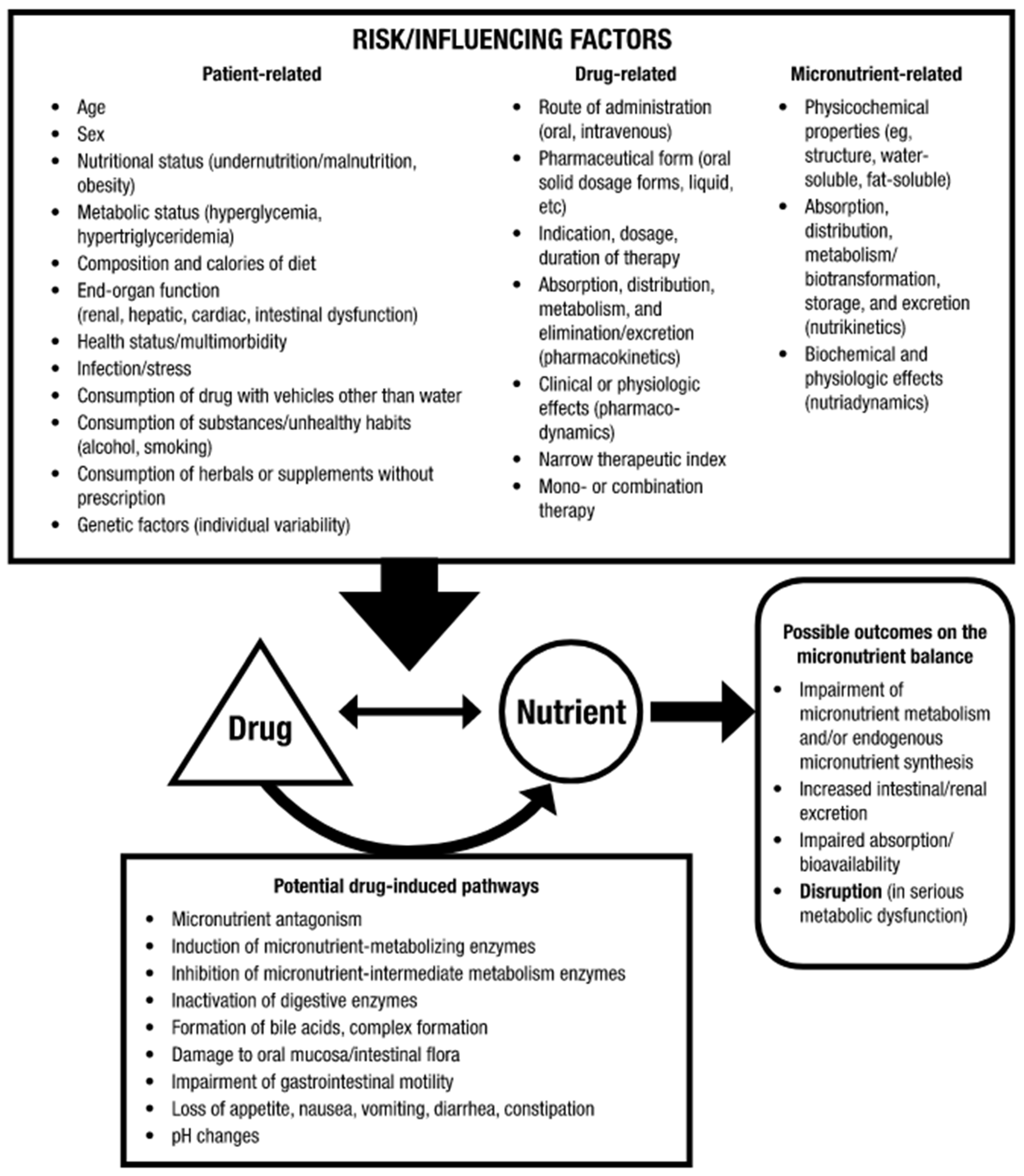
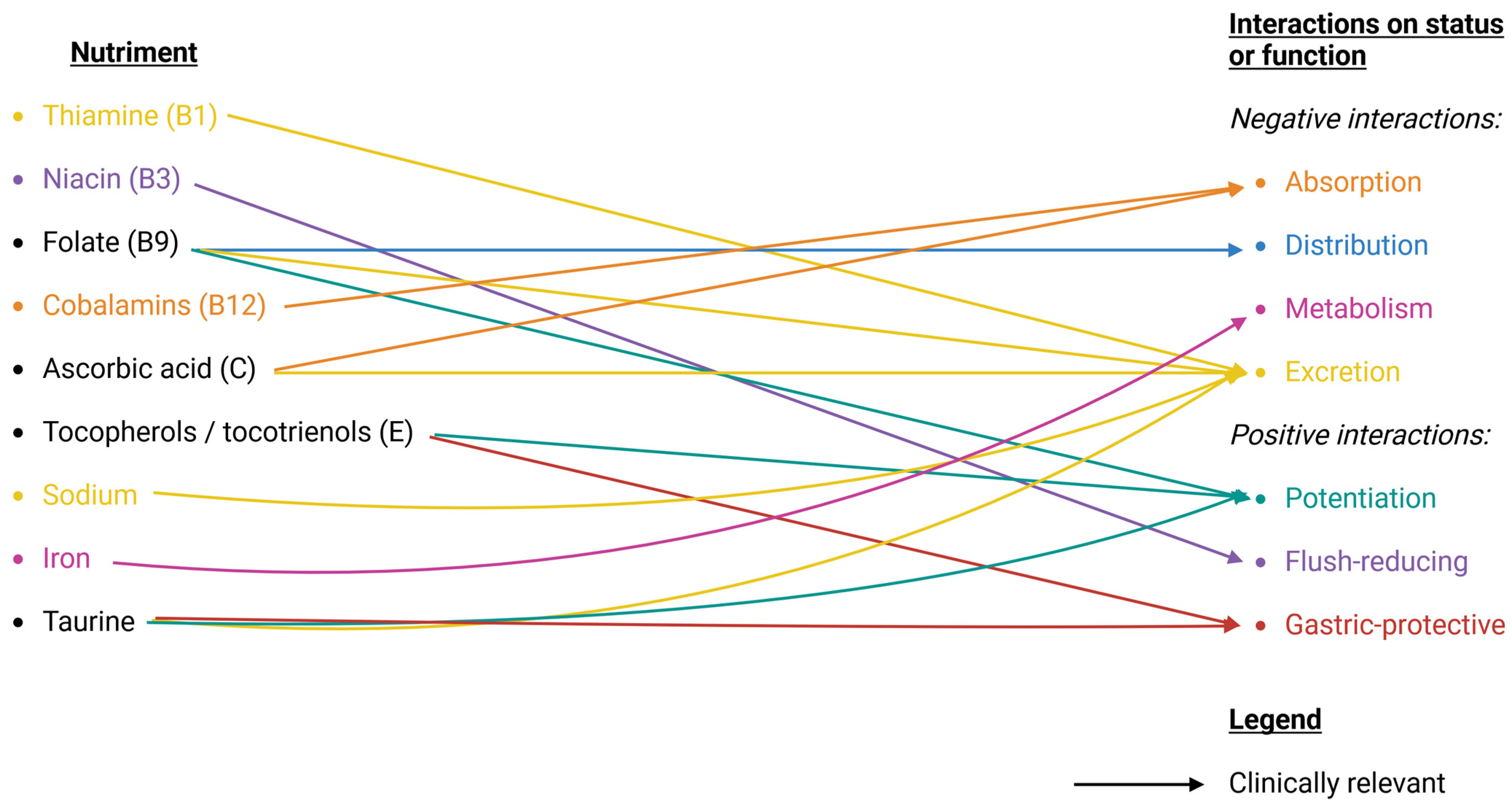

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Renaud, D.; Höller, A.; Michel, M. Potential Drug–Nutrient Interactions of 45 Vitamins, Minerals, Trace Elements, and Associated Dietary Compounds with Acetylsalicylic Acid and Warfarin—A Review of the Literature. Nutrients 2024, 16, 950. https://doi.org/10.3390/nu16070950
Renaud D, Höller A, Michel M. Potential Drug–Nutrient Interactions of 45 Vitamins, Minerals, Trace Elements, and Associated Dietary Compounds with Acetylsalicylic Acid and Warfarin—A Review of the Literature. Nutrients. 2024; 16(7):950. https://doi.org/10.3390/nu16070950
Chicago/Turabian StyleRenaud, David, Alexander Höller, and Miriam Michel. 2024. "Potential Drug–Nutrient Interactions of 45 Vitamins, Minerals, Trace Elements, and Associated Dietary Compounds with Acetylsalicylic Acid and Warfarin—A Review of the Literature" Nutrients 16, no. 7: 950. https://doi.org/10.3390/nu16070950
APA StyleRenaud, D., Höller, A., & Michel, M. (2024). Potential Drug–Nutrient Interactions of 45 Vitamins, Minerals, Trace Elements, and Associated Dietary Compounds with Acetylsalicylic Acid and Warfarin—A Review of the Literature. Nutrients, 16(7), 950. https://doi.org/10.3390/nu16070950





