The Association between Lifestyle Factors and COVID-19: Findings from Qatar Biobank
Abstract
1. Introduction
2. Materials and Methods
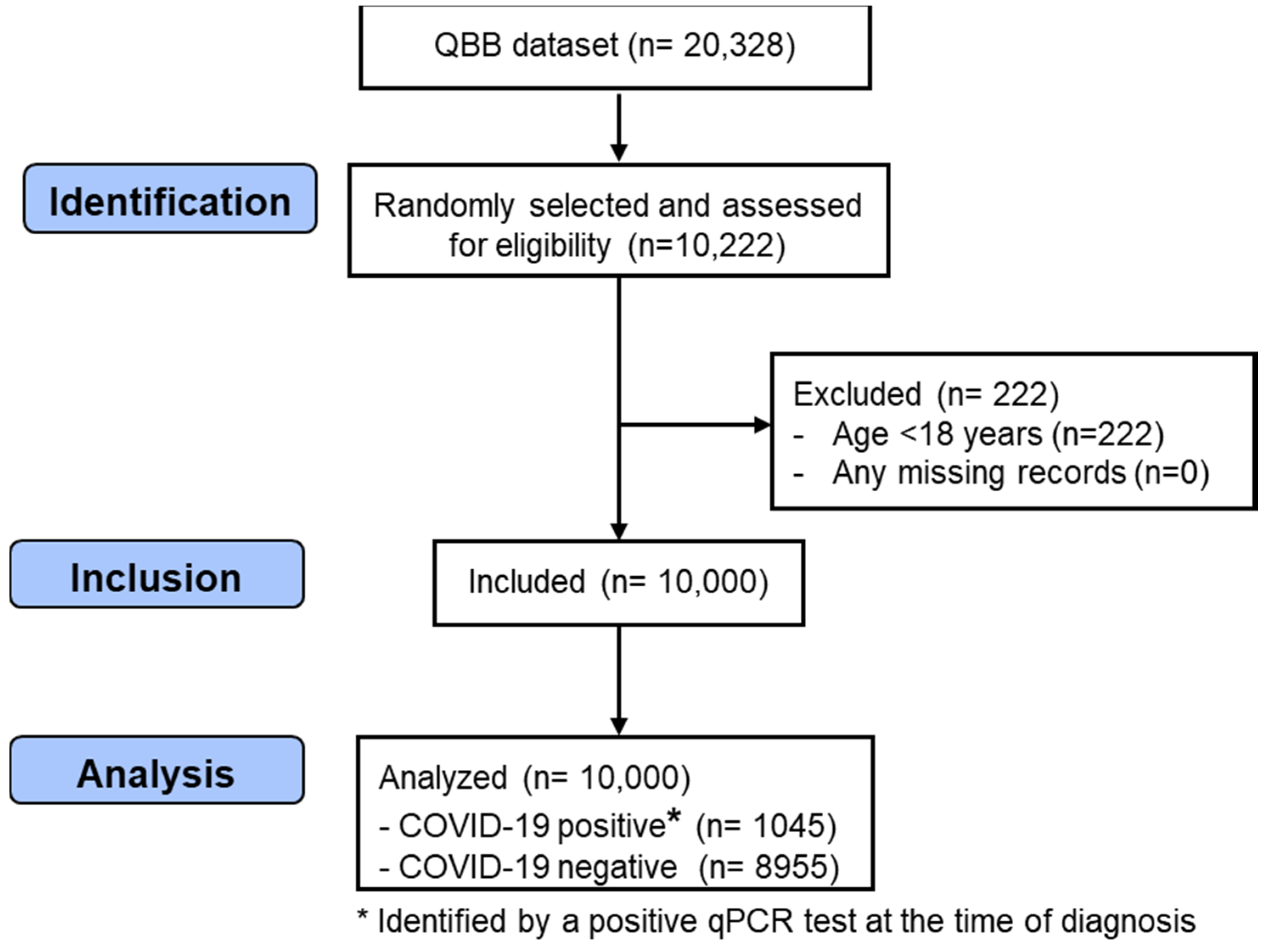
2.1. Design of Study and Sample Collection
2.2. Outcome Variable: COVID-19 Infection
2.3. Exposure Variables: Sociodemographic and Lifestyle Factors
2.4. Statistical Analysis
2.5. Ethical Approval
3. Results
3.1. Sample Characteristics
3.2. Sociodemographic and Lifestyle Factors and COVID-19 Infection
3.3. Vitamin D and COVID-19 Infection
4. Discussion
4.1. Smoking Status and COVID-19 Susceptibility
4.2. Vitamin D Supplementation and COVID-19 Susceptibility
4.3. Obesity and History of Bariatric Surgery and COVID-19 Susceptibility
4.4. Modern Dietary Pattern
4.5. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- WHO Coronavirus (COVID-19) Dashboard. Available online: https://covid19.who.int/ (accessed on 5 November 2023).
- Wiersinga, W.J.; Rhodes, A.; Cheng, A.C.; Peacock, S.J.; Prescott, H.C. Pathophysiology, Transmission, Diagnosis, and Treatment of Coronavirus Disease 2019 (COVID-19): A Review. JAMA 2020, 324, 782. [Google Scholar] [CrossRef] [PubMed]
- Aslan, A.; Aslan, C.; Zolbanin, N.M.; Jafari, R. Acute Respiratory Distress Syndrome in COVID-19: Possible Mechanisms and Therapeutic Management. Pneumonia 2021, 13, 14. [Google Scholar] [CrossRef] [PubMed]
- Brodin, P. Immune Determinants of COVID-19 Disease Presentation and Severity. Nat. Med. 2021, 27, 28–33. [Google Scholar] [CrossRef] [PubMed]
- Adams, K.K.; Baker, W.L.; Sobieraj, D.M. Myth Busters: Dietary Supplements and COVID-19. Ann. Pharmacother. 2020, 54, 820–826. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, F. Vitamin-D and COVID-19: Do Deficient Risk a Poorer Outcome? Lancet Diabetes Endocrinol. 2020, 8, 570. [Google Scholar] [CrossRef] [PubMed]
- Theodore, D.A.; Branche, A.R.; Zhang, L.; Graciaa, D.S.; Choudhary, M.; Hatlen, T.J.; Osman, R.; Babu, T.M.; Robinson, S.T.; Gilbert, P.B.; et al. Clinical and Demographic Factors Associated with COVID-19, Severe COVID-19, and SARS-CoV-2 Infection in Adults: A Secondary Cross-Protocol Analysis of 4 Randomized Clinical Trials. JAMA Netw. Open 2023, 6, e2323349. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Diaz, C.E.; Guilamo-Ramos, V.; Mena, L.; Hall, E.; Honermann, B.; Crowley, J.S.; Baral, S.; Prado, G.J.; Marzan-Rodriguez, M.; Beyrer, C.; et al. Risk for COVID-19 Infection and Death among Latinos in the United States: Examining Heterogeneity in Transmission Dynamics. Ann. Epidemiol. 2020, 52, 46. [Google Scholar] [CrossRef]
- Holt, H.; Talaei, M.; Greenig, M.; Zenner, D.; Symons, J.; Relton, C.; Young, K.S.; Davies, M.R.; Thompson, K.N.; Ashman, J.; et al. Risk Factors for Developing COVID-19: A Population-Based Longitudinal Study (COVIDENCE UK). Thorax 2022, 77, 900–912. [Google Scholar] [CrossRef]
- Kaeuffer, C.; Le Hyaric, C.; Fabacher, T.; Mootien, J.; Dervieux, B.; Ruch, Y.; Hugerot, A.; Zhu, Y.-J.; Pointurier, V.; Clere-Jehl, R.; et al. Clinical Characteristics and Risk Factors Associated with Severe COVID-19: Prospective Analysis of 1045 Hospitalised Cases in North-Eastern France, March 2020. Eurosurveillance 2020, 25, 2000895. [Google Scholar] [CrossRef]
- Louca, P.; Murray, B.; Klaser, K.; Graham, M.S.; Mazidi, M.; Leeming, E.R.; Thompson, E.; Bowyer, R.; Drew, D.A.; Nguyen, L.H.; et al. Modest Effects of Dietary Supplements during the COVID-19 Pandemic: Insights from 445 850 Users of the COVID-19 Symptom Study App. BMJ Nutr. Prev. Health 2021, 4, 149–157. [Google Scholar] [CrossRef]
- Rahmati, M.; Fatemi, R.; Yon, D.K.; Lee, S.W.; Koyanagi, A.; Shin, J.I.; Smith, L. The Effect of Adherence to High-quality Dietary Pattern on COVID-19 Outcomes: A Systematic Review and Meta-analysis. J. Med. Virol. 2023, 95, e28298. [Google Scholar] [CrossRef] [PubMed]
- Ho, J.S.Y.; Fernando, D.I.; Chan, M.Y.; Sia, C.-H. Obesity in COVID-19: A Systematic Review and Meta-Analysis. Ann. Acad. Med. Singapore 2020, 49, 996–1008. [Google Scholar] [CrossRef] [PubMed]
- Najafabadi, B.T.; Rayner, D.G.; Shokraee, K.; Shokraie, K.; Panahi, P.; Rastgou, P.; Seirafianpour, F.; Landi, F.M.; Alinia, P.; Parnianfard, N.; et al. Obesity as an Independent Risk Factor for COVID-19 Severity and Mortality. Cochrane Database Syst. Rev. 2023, 5, CD015201. [Google Scholar]
- Wander, P.L.; Lowy, E.; Beste, L.A.; Tulloch-Palomino, L.; Korpak, A.; Peterson, A.C.; Kahn, S.E.; Boyko, E.J. The Incidence of Diabetes among 2,808,106 Veterans with and without Recent SARS-CoV-2 Infection. Diabetes Care 2022, 45, 782. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Behl, T.; Sachdeva, M.; Sehgal, A.; Kumari, S.; Kumar, A.; Kaur, G.; Yadav, H.N.; Bungau, S. Implicating the Effect of Ketogenic Diet as a Preventive Measure to Obesity and Diabetes Mellitus. Life Sci. 2021, 264, 118661. [Google Scholar] [CrossRef] [PubMed]
- Annual Report 2022–2023; Qatar Biobank: Doha, Qatar, 2023.
- Al Thani, A.; Fthenou, E.; Paparrodopoulos, S.; Al Marri, A.; Shi, Z.; Qafoud, F.; Afifi, N. Qatar Biobank Cohort Study: Study Design and First Results. Am. J. Epidemiol. 2019, 188, 1420–1433. [Google Scholar] [CrossRef] [PubMed]
- Afifi, N.; Fthenou, E.; El Deeb, M.; Frenneaux, M.; Al Thani, A.; Al Khal, A.L.; Biobank, Q.J. Qatar Biobank: COVID-19 Biorepository Project. Emerg. Med. Trauma Acute Care 2021. [Google Scholar] [CrossRef]
- Bawadi, H.; Akasheh, R.T.; Kerkadi, A.; Haydar, S.; Tayyem, R.; Shi, Z. Validity and Reproducibility of a Food Frequency Questionnaire to Assess Macro and Micro-Nutrient Intake among a Convenience Cohort of Healthy Adult Qataris. Nutrients 2021, 13, 2002. [Google Scholar] [CrossRef]
- StataCorp. Stata Statistical Software: Release 17; StataCorp LLC: College Station, TX, USA, 2023. [Google Scholar]
- WMA Declaration of Helsinki–Ethical Principles for Medical Research Involving Human Subjects. Available online: https://www.wma.net/policies-post/wma-declaration-of-helsinki-ethical-principles-for-medical-research-involving-human-subjects/ (accessed on 27 November 2023).
- Arab, A.; Rafie, N.; Hadi, A.; Khorvash, F.; Heidari, Z.; Askari, G. Empirically Derived Dietary Patterns and Their Association with Mental Health: A Cross-Sectional Sample of Iranian Migraine Patients (2019–2020). BMC Nutr. 2022, 8, 28. [Google Scholar] [CrossRef]
- Fituri, S.; Shi, Z. Association between Dietary Patterns and Cognitive Function among Qatari Adults: A Cross-Sectional Analysis of the Qatar Biobank Study. Nutrients 2023, 15, 4053. [Google Scholar] [CrossRef]
- Tayyem, R.; Al-Qawasmeh, R.; Khawaja, N. Dietary Patterns and Nutrients Associated with Metabolic Syndrome. Nutr. Food Sci. 2019, 50, 253–268. [Google Scholar] [CrossRef]
- Zhao, J.; Li, Z.; Gao, Q.; Zhao, H.; Chen, S.; Huang, L.; Wang, W.; Wang, T. A Review of Statistical Methods for Dietary Pattern Analysis. Nutr. J. 2021, 20, 37. [Google Scholar] [CrossRef] [PubMed]
- de Lusignan, S.; Dorward, J.; Correa, A.; Jones, N.; Akinyemi, O.; Amirthalingam, G.; Andrews, N.; Byford, R.; Dabrera, G.; Elliot, A.; et al. Risk Factors for SARS-CoV-2 among Patients in the Oxford Royal College of General Practitioners Research and Surveillance Centre Primary Care Network: A Cross-Sectional Study. Lancet Infect. Dis. 2020, 20, 1034. [Google Scholar] [CrossRef] [PubMed]
- Paleiron, N.; Mayet, A.; Marbac, V.; Perisse, A.; Barazzutti, H.; Brocq, F.-X.; Janvier, F.; Dautzenberg, B.; Bylicki, O. Impact of Tobacco Smoking on the Risk of COVID-19: A Large Scale Retrospective Cohort Study. Nicotine Tob. Res. 2021, 23, 1398. [Google Scholar] [CrossRef] [PubMed]
- Simons, D.; Shahab, L.; Brown, J.; Perski, O. The Association of Smoking Status with SARS-CoV-2 Infection, Hospitalization and Mortality from COVID-19: A Living Rapid Evidence Review with Bayesian Meta-Analyses (Version 7). Addiction 2021, 116, 1319. [Google Scholar] [CrossRef] [PubMed]
- Maggi, F.; Rosellini, A.; Spezia, P.G.; Focosi, D.; Macera, L.; Lai, M.; Pistello, M.; de Iure, A.; Tomino, C.; Bonassi, S.; et al. Nicotine Upregulates ACE2 Expression and Increases Competence for SARS-CoV-2 in Human Pneumocytes. ERJ Open Res. 2021, 7, 00713. [Google Scholar] [CrossRef] [PubMed]
- Salehi, Z.; Ghoochani, B.F.N.M.; Nourian, Y.H.; Jamalkandi, S.A.; Ghanei, M. The Controversial Effect of Smoking and Nicotine in SARS-CoV-2 Infection. Allergy Asthma Clin. Immunol. 2023, 19, 49. [Google Scholar] [CrossRef]
- Tindle, H.A.; Newhouse, P.A.; Freiberg, M.S. Beyond Smoking Cessation: Investigating Medicinal Nicotine to Prevent and Treat COVID-19. Nicotine Tob. Res. 2020, 22, 1669. [Google Scholar] [CrossRef]
- Mohammadi, S.; Heidarizadeh, M.; Entesari, M.; Esmailpour, A.; Esmailpour, M.; Moradi, R.; Sakhaee, N.; Doustkhah, E. In Silico Investigation on the Inhibiting Role of Nicotine/Caffeine by Blocking the S Protein of SARS-CoV-2 versus ACE2 Receptor. Microorganisms 2020, 8, 1600. [Google Scholar] [CrossRef]
- Farsalinos, K.; Eliopoulos, E.; Leonidas, D.D.; Papadopoulos, G.E.; Tzartos, S.; Poulas, K. Nicotinic Cholinergic System and COVID-19: In Silico Identification of an Interaction between SARS-CoV-2 and Nicotinic Receptors with Potential Therapeutic Targeting Implications. Int. J. Mol. Sci. 2020, 21, 5807. [Google Scholar] [CrossRef]
- Leitzke, M. Is the Post-COVID-19 Syndrome a Severe Impairment of Acetylcholine-Orchestrated Neuromodulation That Responds to Nicotine Administration? Bioelectron. Med. 2023, 9, 2. [Google Scholar] [CrossRef] [PubMed]
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider Cytokine Storm Syndromes and Immunosuppression. Lancet 2020, 395, 1033–1034. [Google Scholar] [CrossRef] [PubMed]
- Tracey, K.J. The Inflammatory Reflex. Nature 2002, 420, 853. [Google Scholar] [CrossRef] [PubMed]
- Clift, A.K.; von Ende, A.; Tan, P.S.; Sallis, H.M.; Lindson, N.; Coupland, C.A.C.; Munafò, M.R.; Aveyard, P.; Hippisley-Cox, J.; Hopewell, J.C. Smoking and COVID-19 Outcomes: An Observational and Mendelian Randomisation Study Using the UK Biobank Cohort. Thorax 2022, 77, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Gallus, S.; Scala, M.; Possenti, I.; Jarach, C.M.; Clancy, L.; Fernandez, E.; Gorini, G.; Carreras, G.; Malevolti, M.C.; Commar, A.; et al. The Role of Smoking in COVID-19 Progression: A Comprehensive Meta-Analysis. Eur. Respir. Rev. 2023, 32, 220191. [Google Scholar] [CrossRef] [PubMed]
- Bilezikian, J.P.; Binkley, N.; De Luca, H.F.; Fassio, A.; Formenti, A.M.; Fuleihan, G.E.-H.; Heijboer, A.C.; Giustina, A. Consensus and Controversial Aspects of Vitamin D and COVID-19. J. Clin. Endocrinol. Metab. 2023, 108, 1034–1042. [Google Scholar] [CrossRef] [PubMed]
- Gibbons, J.B.; Norton, E.C.; McCullough, J.S.; Meltzer, D.O.; Lavigne, J.; Fiedler, V.C.; Gibbons, R.D. Association between Vitamin D Supplementation and COVID-19 Infection and Mortality. Sci. Rep. 2022, 12, 19397. [Google Scholar] [CrossRef]
- Villasis-Keever, M.A.; López-Alarcón, M.G.; Miranda-Novales, G.; Zurita-Cruz, J.N.; Barrada-Vázquez, A.S.; González-Ibarra, J.; Martínez-Reyes, M.; Grajales-Muñiz, C.; Santacruz-Tinoco, C.E.; Martínez-Miguel, B.; et al. Efficacy and Safety of Vitamin D Supplementation to Prevent COVID-19 in Frontline Healthcare Workers. A Randomized Clinical Trial. Arch. Med. Res. 2022, 53, 423. [Google Scholar] [CrossRef]
- Jolliffe, D.A.; Holt, H.; Greenig, M.; Talaei, M.; Perdek, N.; Pfeffer, P.; Vivaldi, G.; Maltby, S.; Symons, J.; Barlow, N.L.; et al. Effect of a Test-and-Treat Approach to Vitamin D Supplementation on Risk of All Cause Acute Respiratory Tract Infection and Covid-19: Phase 3 Randomised Controlled Trial (CORONAVIT). BMJ 2022, 378, e071230. [Google Scholar] [CrossRef]
- Greiller, C.; Martineau, A. Modulation of the Immune Response to Respiratory Viruses by Vitamin D. Nutrients 2015, 7, 4240–4270. [Google Scholar] [CrossRef]
- Keutmann, M.; Hermes, G.; Meinberger, D.; Roth, A.; Stemler, J.; Cornely, O.A.; Klatt, A.R.; Streichert, T. The Ratio of Serum LL-37 Levels to Blood Leucocyte Count Correlates with COVID-19 Severity. Sci. Rep. 2022, 12, 9447. [Google Scholar] [CrossRef] [PubMed]
- Kudryashova, E.; Zani, A.; Vilmen, G.; Sharma, A.; Lu, W.; Yount, J.S.; Kudryashov, D.S. Inhibition of SARS-CoV-2 Infection by Human Defensin HNP1 and Retrocyclin RC-101. J. Mol. Biol. 2022, 434, 167225. [Google Scholar] [CrossRef] [PubMed]
- Bilezikian, J.P.; Bikle, D.; Hewison, M.; Lazaretti-Castro, M.; Formenti, A.M.; Gupta, A.; Madhavan, M.V.; Nair, N.; Babalyan, V.; Hutchings, N.; et al. Mechanisms In Endocrinology: Vitamin D and COVID-19. Eur. J. Endocrinol. 2020, 183, R133. [Google Scholar] [CrossRef] [PubMed]
- Michalakis, K.; Ilias, I. SARS-CoV -2 Infection and Obesity: Common Inflammatory and Metabolic Aspects. Diabetes Metab. Syndr. 2020, 14, 469. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Li, P.; Dai, S.; Wang, G.; Li, W.; Song, Z.; Zhu, L.; Zhu, S. Is Prior Bariatric Surgery Associated with Poor COVID-19 Outcomes? A Systematic Review and Meta-Analysis of Case-Control Studies. J. Glob. Health 2023, 13, 06012. [Google Scholar] [CrossRef] [PubMed]
- Wood, G.C.; Benotti, P.N.; Fano, R.M.; Dove, J.T.; Rolston, D.D.; Petrick, A.T.; Still, C.D. Prior Metabolic Surgery Reduced COVID-19 Severity: Systematic Analysis from Year One of the COVID-19 Pandemic. Heliyon 2023, 9, e15824. [Google Scholar] [CrossRef] [PubMed]
- Iannelli, A.; Bouam, S.; Schneck, A.-S.; Frey, S.; Zarca, K.; Gugenheim, J.; Alifano, M. The Impact of Previous History of Bariatric Surgery on Outcome of COVID-19. A Nationwide Medico-Administrative French Study. Obes. Surg. 2021, 31, 1455. [Google Scholar] [CrossRef] [PubMed]
- Lewis, C.-A.; de Jersey, S.; Seymour, M.; Hopkins, G.; Hickman, I.; Osland, E. Iron, Vitamin B12, Folate and Copper Deficiency after Bariatric Surgery and the Impact on Anaemia: A Systematic Review. Obes. Surg. 2020, 30, 4542–4591. [Google Scholar] [CrossRef]
- Bal, B.S.; Finelli, F.C.; Shope, T.R.; Koch, T.R. Nutritional Deficiencies after Bariatric Surgery. Nat. Rev. Endocrinol. 2012, 8, 544. [Google Scholar] [CrossRef]
- Giusti, V.; Suter, M.; Héraïef, E.; Gaillard, R.C.; Burckhardt, P. Effects of Laparoscopic Gastric Banding on Body Composition, Metabolic Profile and Nutritional Status of Obese Women: 12-Months Follow-Up. Obes. Surg. 2004, 14, 239–245. [Google Scholar] [CrossRef]
- Morales-Marroquin, E.; Xie, L.; Uppuluri, M.; Almandoz, J.P.; de la Cruz-Muñoz, N.; Messiah, S.E. Immunosuppression and Clostridioides (Clostridium) Difficile Infection Risk in Metabolic and Bariatric Surgery Patients. J. Am. Coll. Surg. 2021, 233, 223. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimzadeh, A.; Taghizadeh, M.; Milajerdi, A. Major Dietary Patterns in Relation to Disease Severity, Symptoms, and Inflammatory Markers in Patients Recovered from COVID-19. Front. Nutr. 2022, 9, 929384. [Google Scholar] [CrossRef] [PubMed]
- Merino, J.; Joshi, A.D.; Nguyen, L.H.; Leeming, E.R.; Mazidi, M.; Drew, D.A.; Gibson, R.; Graham, M.S.; Lo, C.-H.; Capdevila, J.; et al. Diet Quality and Risk and Severity of COVID-19: A Prospective Cohort Study. Gut 2021, 70, 2096–2104. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Rebholz, C.M.; Hegde, S.; LaFiura, C.; Raghavan, M.; Lloyd, J.F.; Cheng, S.; Seidelmann, S.B.; Diets, P.-B. Pescatarian Diets and COVID-19 Severity: A Population-Based Case–Control Study in Six Countries. BMJ Nutr. Prev. Health 2021, 4, 257. [Google Scholar] [CrossRef]
- Salazar-Robles, E.; Kalantar-Zadeh, K.; Badillo, H.; Calderón-Juárez, M.; García-Bárcenas, C.A.; Ledesma-Pérez, P.D.; Lerma, A.; Lerma, C. Association between Severity of COVID-19 Symptoms and Habitual Food Intake in Adult Outpatients. BMJ Nutr. Prev. Health 2021, 4, 469–478. [Google Scholar] [CrossRef]
- Calder, P.C. Nutrition, Immunity and COVID-19. BMJ Nutr. Prev. Health 2020, 3, 74. [Google Scholar] [CrossRef]
- Belanger, M.J.; Hill, M.A.; Angelidi, A.M.; Dalamaga, M.; Sowers, J.R.; Mantzoros, C.S. COVID-19 and Disparities in Nutrition and Obesity. N. Engl. J. Med. 2020, 383, e69. [Google Scholar] [CrossRef]
- Yan, B.; Chu, H.; Yang, D.; Sze, K.-H.; Lai, P.-M.; Yuan, S.; Shuai, H.; Wang, Y.; Kao, R.Y.-T.; Chan, J.F.-W.; et al. Characterization of the Lipidomic Profile of Human Coronavirus-Infected Cells: Implications for Lipid Metabolism Remodeling upon Coronavirus Replication. Viruses 2019, 11, 73. [Google Scholar] [CrossRef]
- Eiser, A.R. Could Dietary Factors Reduce COVID-19 Mortality Rates? Moderating the Inflammatory State. J. Altern. Complement. Med. 2021, 27, 176–178. [Google Scholar] [CrossRef]
- Butler, M.J.; Barrientos, R.M. The Impact of Nutrition on COVID-19 Susceptibility and Long-Term Consequences. Brain Behav. Immun. 2020, 87, 53–54. [Google Scholar] [CrossRef]
- Soltani, A.; Jaam, M.; Nazar, Z.; Stewart, D.; Shaito, A. Attitudes and Beliefs Regarding the Use of Herbs and Supplementary Medications with COVID-19: A Systematic Review. Res. Social Adm. Pharm. 2023, 19, 343–355. [Google Scholar] [CrossRef]
- Hassan, S.S.U.; Abdel-Daim, M.M.; Behl, T.; Bungau, S. Natural Products for Chronic Diseases: A Ray of Hope. Molecules 2022, 27, 5573. [Google Scholar] [CrossRef]
| Total | No | Yes | p-Value | |
|---|---|---|---|---|
| N = 10,000 | N = 8955 | N = 1045 | ||
| Age (years) | 40.3 ± 13.1 | 40.4 ± 13.1 | 39.6 ± 12.7 | 0.083 |
| Gender | <0.001 | |||
| Male | 4780 (47.8%) | 4351 (48.6%) | 429 (41.1%) | |
| Female | 5220 (52.2%) | 4604 (51.4%) | 616 (58.9%) | |
| Educational level | 0.020 | |||
| Low | 1796 (18.0%) | 1594 (17.8%) | 202 (19.3%) | |
| Medium | 3040 (30.4%) | 2694 (30.1%) | 346 (33.1%) | |
| High | 5164 (51.6%) | 4667 (52.1%) | 497 (47.6%) | |
| Smoking status | <0.001 | |||
| None | 6468 (64.7%) | 5695 (63.6%) | 773 (74.0%) | |
| Smoker | 1866 (18.7%) | 1735 (19.4%) | 131 (12.5%) | |
| Ex-smoker | 1666 (16.7%) | 1525 (17.0%) | 141 (13.5%) | |
| Leisure time physical activity (MET hours/week) | 24.9 ± 52.5 | 24.9 ± 51.0 | 24.3 ± 64.2 | 0.74 |
| Modern dietary pattern score | 0.0 ± 1.00 | −0.01 ± 1.0 | 0.1 ± 1.1 | 0.001 |
| Prudent dietary pattern score | 0.0 ± 1.00 | −0.0 ± 1.0 | 0.02 ± 0.9 | 0.45 |
| Convenience dietary pattern score | 0.0 ± 1.00 | −0.01 ± 1.0 | 0.1 ± 1.1 | 0.032 |
| Serum vitamin D (ng/mL) | 19.3 ± 11.1 | 19.3 ± 1.1 | 18.9 ± 10.9 | 0.22 |
| Hypertension | 1638 (16.4%) | 1479 (16.5%) | 159 (15.2%) | 0.28 |
| Diabetes | 2026 (20.3%) | 1808 (20.3%) | 218 (21.0%) | 0.60 |
| History of bariatric surgery | 1213 (12.1%) | 1058 (11.8%) | 155 (14.9%) | 0.004 |
| BMI categories | 0.051 | |||
| Normal | 2112 (21.1%) | 1905 (21.3%) | 207 (19.8%) | |
| Overweight | 3462 (34.7%) | 3123 (34.9%) | 339 (32.4%) | |
| Obese | 4417 (44.2%) | 3918 (43.8%) | 499 (47.8%) | |
| Dietary supplement use | ||||
| Multivitamin/minerals | 3565 (35.6%) | 3174 (35.4%) | 391 (37.4%) | 0.21 |
| Calcium | 726 (7.3%) | 649 (7.2%) | 77 (7.4%) | 0.89 |
| Folic acid | 316 (3.2%) | 276 (3.1%) | 40 (3.8%) | 0.19 |
| Iron | 1366 (13.7%) | 1208 (13.5%) | 158 (15.1%) | 0.15 |
| Vitamin B | 806 (8.1%) | 728 (8.1%) | 78 (7.5%) | 0.45 |
| Vitamin C | 665 (6.7%) | 604 (6.7%) | 61 (5.8%) | 0.27 |
| Vitamin D | 2402 (24.0%) | 2166 (24.2%) | 236 (22.6%) | 0.25 |
| Other supplements | 3315 (33.1%) | 2968 (33.1%) | 347 (33.2%) | 0.97 |
| Any supplement use | 5977 (59.8%) | 5339 (59.6%) | 638 (61.1%) | 0.37 |
| Model 1 | Model 2 | |||
|---|---|---|---|---|
| OR [95% CI] | p-Value | OR [95% CI] | p-Value | |
| Vitamin D | 0.81 (0.68–0.96) | 0.018 | 0.82 (0.69–0.97) | 0.022 |
| Age (years) | 0.99 (0.99–1.00) | 0.050 | 0.99 (0.99–1.00) | 0.084 |
| Gender | ||||
| Male | 1.00 | 1.00 | ||
| Female | 1.03 (0.88–1.21) | 0.715 | 1.03 (0.88–1.21) | 0.704 |
| Education | ||||
| Low | 1.00 | 1.00 | ||
| Medium | 0.95 (0.77–1.17) | 0.641 | 0.94 (0.76–1.17) | 0.588 |
| High | 0.86 (0.71–1.04) | 0.124 | 0.85 (0.70–1.03) | 0.096 |
| Smoking | ||||
| Non | 1.00 | 1.00 | ||
| Smoker | 0.55 (0.44–0.68) | <0.001 | 0.55 (0.44–0.68) | <0.001 |
| Ex-smoker | 0.70 (0.57–0.86) | <0.001 | 0.70 (0.57–0.86) | <0.001 |
| Leisure time PA (MET hours/week) | ||||
| T1 | 1.00 | 1.00 | ||
| T2 | 1.02 (0.87–1.20) | 0.817 | 1.03 (0.88–1.21) | 0.737 |
| T3 | 0.94 (0.79–1.11) | 0.472 | 0.93 (0.79–1.10) | 0.404 |
| BMI level | ||||
| Normal | 1.00 | |||
| Overweight | 1.09 (0.90–1.32) | 0.379 | ||
| Obese | 1.20 (1.00–1.45) | 0.049 | ||
| Diabetes | ||||
| No | 1.00 | 1.00 | ||
| Yes | 1.09 (0.91–1.31) | 0.338 | 1.10 (0.92–1.32) | 0.308 |
| Hypertension | ||||
| No | 1.00 | 1.00 | ||
| Yes | 0.94 (0.76–1.16) | 0.547 | 0.96 (0.78–1.18) | 0.693 |
| Modern dietary pattern | 1.09 (1.02–1.16) | 0.012 | 1.08 (1.01–1.16) | 0.018 |
| Prudent dietary pattern | 1.03 (0.97–1.10) | 0.344 | 1.03 (0.97–1.10) | 0.307 |
| Convenience dietary pattern | 1.05 (0.99–1.12) | 0.100 | 1.05 (0.99–1.12) | 0.092 |
| Any supplement use | 1.12 (0.96–1.30) | 0.155 | 1.10 (0.95–1.28) | 0.205 |
| History of bariatric surgery | ||||
| No | 1.00 | |||
| Yes | 1.24 (1.03–1.50) | 0.022 | ||
| Model 1 | Model 2 | Model 3 | ||||
|---|---|---|---|---|---|---|
| OR (95% CI) | p-Value | OR (95% CI) | p-Value | OR (95% CI) | p-Value | |
| Any supplement use | 1.00 (0.87–1.14) | 0.979 | 1.03 (0.90–1.19) | 0.652 | 1.02 (0.89–1.18) | 0.749 |
| Multivitamin/minerals | 1.07 (0.94–1.22) | 0.322 | 1.17 (0.99–1.40) | 0.067 | 1.16 (0.97–1.38) | 0.096 |
| Calcium | 0.96 (0.75–1.22) | 0.723 | 0.97 (0.75–1.25) | 0.792 | 0.96 (0.75–1.24) | 0.775 |
| Folic acid | 1.13 (0.80–1.59) | 0.485 | 1.18 (0.83–1.66) | 0.356 | 1.17 (0.82–1.65) | 0.385 |
| Iron | 1.01 (0.84–1.22) | 0.886 | 1.02 (0.84–1.24) | 0.869 | 1.00 (0.82–1.22) | 1.000 |
| Other supplements | 0.94 (0.81–1.08) | 0.357 | 0.90 (0.76–1.06) | 0.197 | 0.90 (0.76–1.06) | 0.216 |
| Vitamin B | 0.87 (0.68–1.12) | 0.281 | 0.89 (0.69–1.14) | 0.348 | 0.87 (0.68–1.12) | 0.288 |
| Vitamin C | 0.84 (0.64–1.10) | 0.210 | 0.86 (0.65–1.14) | 0.288 | 0.86 (0.65–1.13) | 0.276 |
| Vitamin D | 0.85 (0.73–1.00) | 0.046 | 0.81 (0.68–0.96) | 0.018 | 0.82 (0.69–0.97) | 0.022 |
| Quartiles of Serum Vitamin D | |||||
|---|---|---|---|---|---|
| Q1 | Q2 | Q3 | Q4 | p-Value | |
| Model 1 | 1.00 | 1.19 (0.99–1.42) | 1.04 (0.85–1.26) | 0.94 (0.77–1.15) | 0.324 |
| Model 2 | 1.00 | 1.19 (1.00–1.43) | 1.06 (0.87–1.29) | 0.98 (0.80–1.20) | 0.552 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akbar, Z.; Kunhipurayil, H.H.; Saliba, J.; Ahmad, J.; Al-Mansoori, L.; Al-Khatib, H.A.; Al Thani, A.A.; Shi, Z.; Shaito, A.A. The Association between Lifestyle Factors and COVID-19: Findings from Qatar Biobank. Nutrients 2024, 16, 1037. https://doi.org/10.3390/nu16071037
Akbar Z, Kunhipurayil HH, Saliba J, Ahmad J, Al-Mansoori L, Al-Khatib HA, Al Thani AA, Shi Z, Shaito AA. The Association between Lifestyle Factors and COVID-19: Findings from Qatar Biobank. Nutrients. 2024; 16(7):1037. https://doi.org/10.3390/nu16071037
Chicago/Turabian StyleAkbar, Zoha, Hasna H. Kunhipurayil, Jessica Saliba, Jamil Ahmad, Layla Al-Mansoori, Hebah A. Al-Khatib, Asmaa A. Al Thani, Zumin Shi, and Abdullah A. Shaito. 2024. "The Association between Lifestyle Factors and COVID-19: Findings from Qatar Biobank" Nutrients 16, no. 7: 1037. https://doi.org/10.3390/nu16071037
APA StyleAkbar, Z., Kunhipurayil, H. H., Saliba, J., Ahmad, J., Al-Mansoori, L., Al-Khatib, H. A., Al Thani, A. A., Shi, Z., & Shaito, A. A. (2024). The Association between Lifestyle Factors and COVID-19: Findings from Qatar Biobank. Nutrients, 16(7), 1037. https://doi.org/10.3390/nu16071037









