1. Introduction
Protein is the main ingredient in milk, and 80% of it is CN [
1]. Based on the homology of the AA sequences, there are four different types of CN: αs1-CN, αs2-CN, β-CN, and κ-CN [
2]. Among them, β-CN is the main protein in the CN fraction, accounting for about 30–40%. There are 13 genetic variants of β-CN: A1, A2, A3, B, C, D, E, F, G, H1, H2, I, and J [
3]. A1 and A2 are the main variants, differing only by the 67th AA, which is His in A1 and Pro in A2 [
4]. Structural differences between variants can impact milk properties and bioactivity. According to the literature [
5], A2 β-CN is less hydrophobic and its dairy products have a tighter micellar structure, and A2 β-CN also shows higher molecular chaperone activity against heat-induced whey protein aggregation during heat treatment than A1 β-CN. Therefore, dairy products containing A2 β-CN have a better stability than those containing A1 β-CN [
6]. Other studies showed that the His67 in A1 β-CN confers reduced sensitivity to proteolytic cleavage by digestive enzymes, leading to the release of β-casomorphin-7 [
7,
8]. Numerous gastrointestinal conditions, including stomach pain and changes in stool consistency, have been linked to β-casomorphin-7 [
9,
10], and may cause other health conditions such as immune disorders [
11,
12]. Ingestion of milk or dairy products containing A2 β-CN results in little or no intestinal release of β-casomorphin-7 [
13].
In most studies on A1 and A2 β-CN, animal models have been employed to compare their functional properties. For example, one study [
9] found that an A2 β-CN milk-based diet accelerated gastrointestinal motility in rats in contrast to A1 β-CN, which showed the opposite trend and induced localized inflammation. Another study [
14] found similar results and showed that consumption of A1 variants (A1A1 and A1A2) induced an intestinal inflammatory response through activation of the T helper 2 pathway, whereas no changes occurred with consumption of A2 β-CN variants (A2A2). Additionally, another study [
15] reported that consuming A2 milk lowered cholesterol levels and prevented ischemic heart disease.
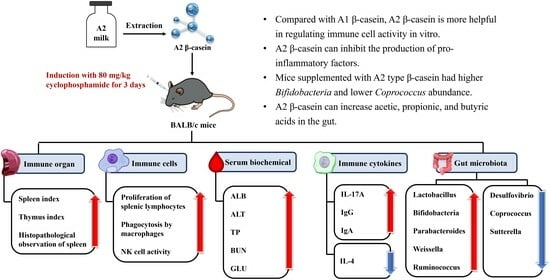
In summary, although studies have shown that the physicochemical properties of A2 β-CN milk are more stable and more beneficial to human gastrointestinal health than A1 β-CN milk, the possible relationship between the digestive properties of A2 β-CN and human health needs to be further explored in preclinical tests, which allow us to gather more direct evidence and establish functional tests to evaluate the in vivo immunomodulatory aspects of A2 β-CN. Therefore, the present study aimed to evaluate the immunoreactivity of A2 β-CN digested both in vitro and in cyclophosphamide (CTX)-induced immunosuppressed mice in vivo. The immune organs, immune cells, serum biochemical indexes, immunoglobulins, and other indexes of A2 β-CN-containing dietary intervention were measured, and the changes in the structure of intestinal flora and short-chain fatty acids (SCFAs) in the cecum contents of mice were detected via 16S RNA and gas chromatography (GC) in order to assess the immune-modulating ability of A2 β-CN in immunosuppressed mice. This information will provide a theoretical basis and reference value for other functional foods.
2. Materials and Methods
2.1. Milk Samples
A2 milk was obtained from purebred A2 genotype cows located on a farm in northeast China. A1 milk was purchased from shopping malls and supermarkets in northeast China.
2.2. Extraction of A2 β-CN
Milk samples of 100 mL were taken, homogenized for 1 min, and then centrifuged at 5000× g for 20 min at 4 °C to remove most of the fat. The pH was increased to 4.6–4.7. After leaving the solution to stand for 30 min, β-CN precipitation could be observed. Precipitated CN was neutralized by washing with distilled water and it was transferred to dialysis bags. For 48 h, the dialysis bags were submerged in deionized water at 4 °C; the dialysate was replaced every 6 to 8 h. After dialysis, the protein solution was freeze-dried and stored at −20 °C. The β-CN in the crude extracted casein was separated and purified using a DEAE Bestarose Fast Flow (Bio-Rad, Hercules, CA, USA)) anion-exchange column using a GE AKTA Pure protein purifier (Bio-Rad, Hercules, CA, USA). Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) was used to detect and identify the protein purification solution with the aim of identifying the type and purity of the protein. It was then desalted, and a BCA protein concentration kit was used to measure the protein content.
2.3. Preparation of Digestion Products
β-CN digests were prepared using a static in vitro digestion system according to the method in [
16] with some modifications. A 4% A2 β-CN solution was used, and saliva, gastric juice, and intestinal juice were prepared simultaneously. The specific operation methods of the test were as follows:
Oral phase: 4% β-CN solution was mixed with simulated saliva, and the pH value was adjusted to 6.8. The reaction system was placed in an incubator at 37 °C and 130 rpm for 10 min.
Gastric phase: 0.2 g Nacl and 0.7 mL Hcl were mixed, and the volume was fixed to 100 mL. The sample solution was mixed 1:1 with the simulated gastric fluid, and 0.064 g of pepsin (3000 U/mg) was added. The pH value was adjusted to 2.5 and the culture conditions were the same as above. Samples were removed at 0.5 h, 1.0 h, 1.5 h, and 2.0 h.
Intestinal phase: Cacl2 at 36.7 mg/mL and Nacl at 219.1 mg/mL were prepared. Then, 1.5 mL Cacl2 and Nacl were added, and 1.8 g bile salt was added. The pH value was adjusted to 7.0, and 1.2 g trypsin (10,000 U/mg) was added. The culture conditions were the same as above and samples were removed at 2.5 h, 3.0 h, 3.5 h, 4.0 h, and 5.0 h.
2.4. A2 β-CN Immune Cell Assay In Vitro
2.4.1. Cells
We obtained mice lymphoma cells (YAC-1) and mononuclear macrophages (RAW 264.7) from Shanghai Biological Technology Co., Ltd. (EK, Shanghai, China).
2.4.2. Determination of Splenic Lymphocyte Proliferation
Cells were aseptically extracted [
17], and the cell concentration was finally adjusted to 2 × 10
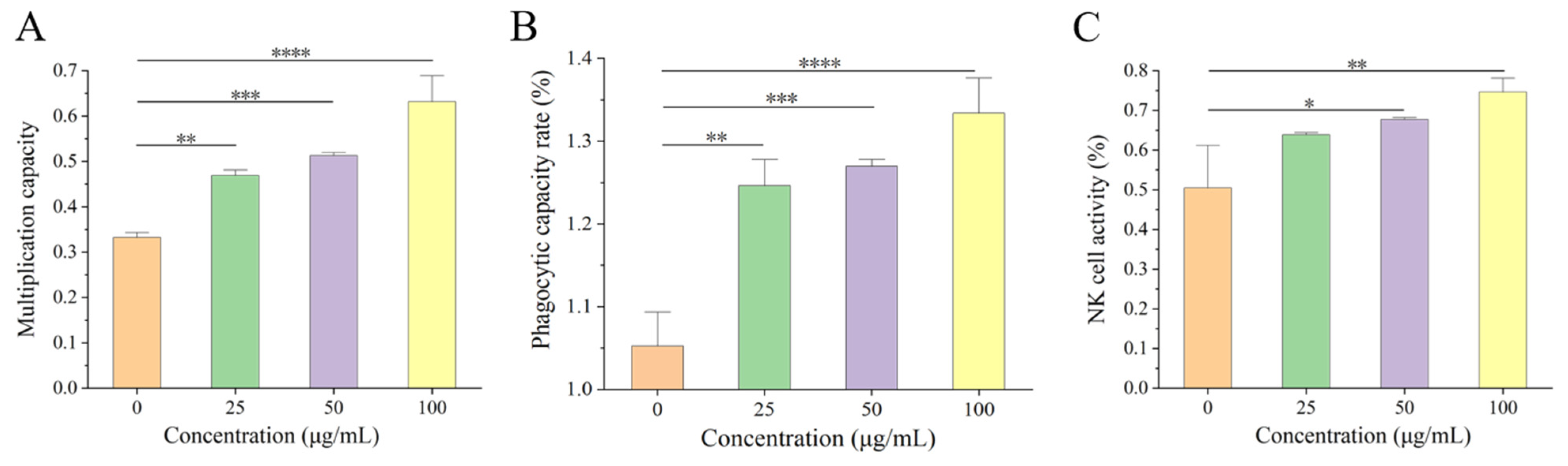
7 cells/mL. Aliquots of 100 μL of this spleen lymphocyte suspension were inoculated in 96-well plates, and 100 μL of β-CN digest was added to obtain final concentrations of 0, 25, 50, and 100 μg/mL. Concanavalin A (ConA) was added, along with RPMI-1640 (Aladdin, Shanghai, China ), as a blank control well, and cultured for 48 h. After adding the Cell Counting Kit-8 (Aladdin, Shanghai, China) solution, the cells were grown for 4 h. The optical density value was determined at a wavelength of 570 nm, and the proliferation index (PI) of splenic lymphocytes was determined according to Equation (1):
2.4.3. Phagocytosis Experiments of Macrophages
In 96-well plates, RAW 264.7 cells were seeded at a density of 2 × 105 cells/mL. After 4 h, the cell supernatant was drained, and diluted β-CN digest was added to achieve final concentrations of 0, 25, 50, and 100 μg/mL. After 48 h, the dye solution (Neutral Red) (Amresco, OH, USA) was added, and the cells were rinsed twice or three times with PBS. After adding the cell lysis solution, the absorbance value at 540 nm was measured two hours later.
2.4.4. Determination of Natural Killer Cell Activity
The concentration of YAC-1 cells was set to 1 × 10
4 cells/mL and the concentration of spleen cells was set to 2 × 10
7 cells/mL [
18]. Effector cells and target cells were added at an effector/target ratio of 50:1. A total of 50 μL spleen cells and 50 μL target cells were added into the test well group X, 50 μL spleen cells into the effector cell control well group Y, and 50 μL target cells into the target cell control well group Z; three parallel groups were set up. An amount of 50 μL A2 β-CN digest was added to obtain final concentrations of 0, 25, 50, and 100 μg/mL. The absorbance values were measured at 450 nm after continued incubation for 4 h. NK cell activity was calculated according to Equation (2):
2.5. Regulatory Effect of A2 β-CN in Immunosuppressed Mice
2.5.1. Animals
Specific pathogen-free (SPF) BALB/c mice (6–8 wk old, n = 64, BW 18–20 g; Vital River, Beijing, China) were chosen to harvest the primary splenic lymphocyte suspension. The mice were adaptively raised for 7 d starting on the day of purchase at a relative temperature of 21 ± 1 °C and a relative humidity of 55 ± 5%. Animal use and care guidelines and ethical standards for animal protection were closely followed during animal research. The Animal Management Rules of the Ministry of Health, People’s Republic of China (documentation number 55, 2001), were strictly followed when using the animals in our experiment. The Animal Ethics Committee of Northeast Agriculture University approved the study’s experimental protocol (approved number: NEAUEC 20230418).
2.5.2. Establishment and Treatment of Immunosuppressed BALB/c Mice Model
During the experiment, all mice were regularly gavaged every day with ad libitum access to drinking water. The mice were randomly divided into six groups, with 10 mice per group (12 mice in the model group (M)): the normal group (NC), the M group (M), the low-dose A2 β-CN group (LA2), the medium-dose A2 β-CN group (MA2), the high-dose A2 β-CN group (HA2), and the high-dose A1 β-CN group (HA1). From the 8th to the 10th d, the NC group was fed with normal saline. Other groups were given 80 mg/kg CTX for 3 d, and from the 11th to 35th d, groups NC and M were given normal saline by gavage. The LA2, MA2, and HA2 groups were treated by gavage with different doses of A2 β-CN (300, 600, and 900 mg/kg BW), and the CN group was treated with the same dose of A1 β-CN as the HA2 group. The dose by gavage was chosen according to the standard [
19]. The specific grouping and gavage methods were carried out as described in
Table 1.
To ascertain whether the modeling was successful, two mice from the NC and M groups were randomly euthanized three days into the experiment. (1) Compared with the mice in the NC group, the BW of mice in group M decreased by 15% ± 5%. (2) The number of white blood cells, neutrophils, and lymphocytes in peripheral blood decreased by 50%. (3) The spleen and thymus were taken and weighed to calculate the organ coefficients. A statistically significant difference in all the above indicators indicated successful modeling. Mice were weighed weekly and recorded. After the last gavage for 24 h, the mice were weighed, and except for the mice that needed to undergo the immunity-related experiments on the same day, the remaining mice were sacrificed, and the samples were collected, put into sterile freezing tubes, frozen in liquid nitrogen immediately, and kept in a refrigerator at −80 °C for spare use.
2.5.3. Determination of Immune Organ Index
Mice were sacrificed according to the method in [
20]. After eyeball blood collection, the mice were sacrificed by cervical dislocation, and the spleen and thymus were dissected in a sterile environment. The spleen and thymus tissues of the mice were weighed, and the organ index was calculated using Equation (3):
2.5.4. Histopathological Observation of Spleen
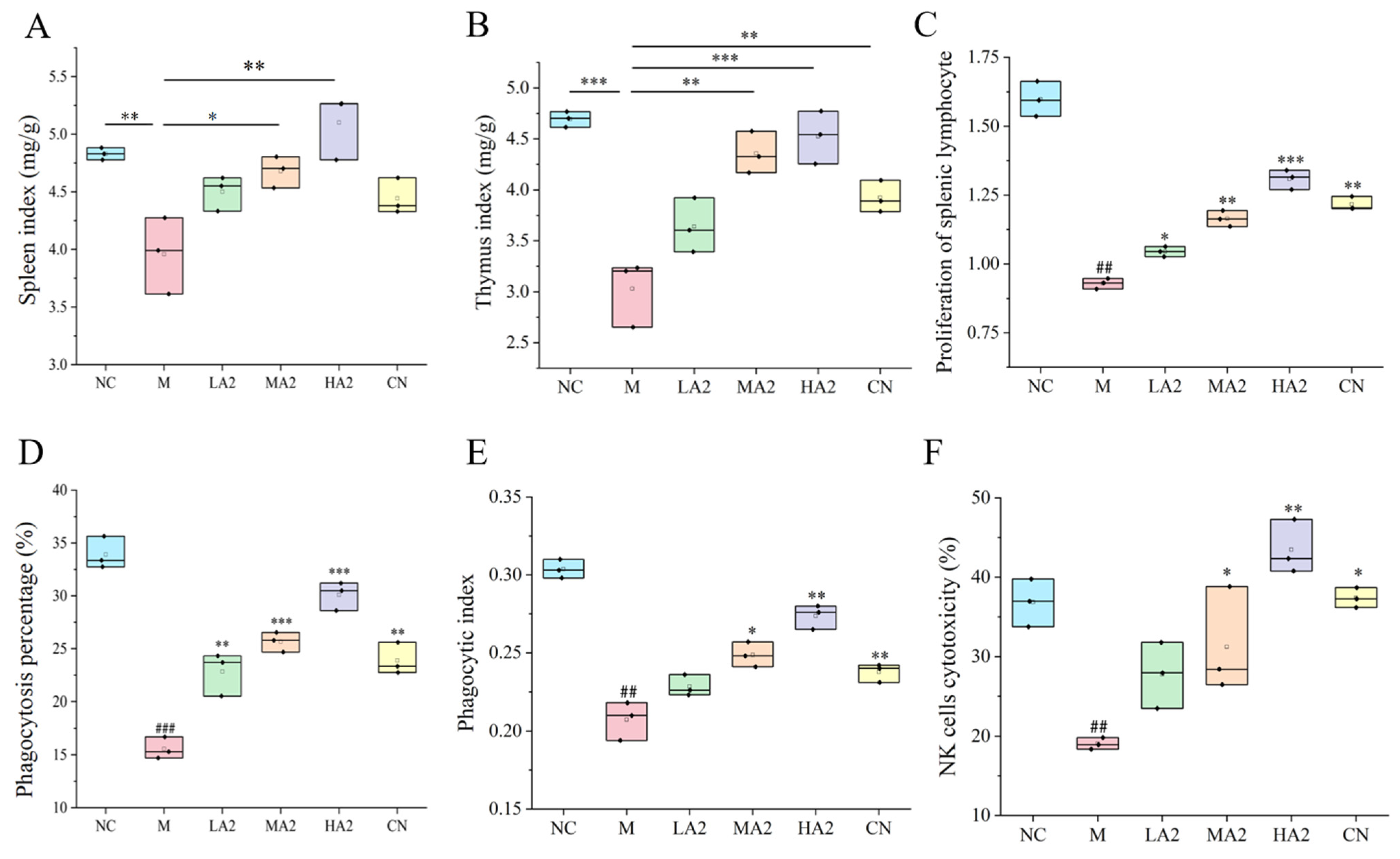
After the mice were sacrificed and the spleens were removed, the surface color and texture of the spleen were observed and judged by touching the surface of the spleen. Following this overall observation, hematoxylin–eosin staining was performed. Paraffin sections were deparaffinized in water in order to achieve this. Hematoxylin was then used to stain the nuclei, and eosin was used to stain the cytoplasm. Lastly, the histopathological morphology and seal dewatering were noted.
2.5.5. Spleen Lymphocyte PI
Spleens were harvested aseptically (see
Section 2.4.2), and the cell concentration was 3 × 10
6 cells/mL. Two wells of a 24-well culture plate were filled with the spleen cell suspension; one well received ConA solution (7.5 μg/mL) and the other well served as a control and was cultured for 3 d. The supernatant from each well was discarded 4 h prior to the completion of the culture. Next, RPMI-1640 medium without calf serum was added, followed by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (5 mg/mL). Dimethyl sulfoxide was added and mixed, and aliquots were loaded into 96-well culture plates; three parallel wells were repeated. Absorbance values were measured at 570 nm, and the lymphocyte stimulation index was calculated using Equation (4):
2.5.6. Phagocytosis Index and Percentage of Peritoneal Macrophages
Before the immunization test, the mice were i.p. injected with 0.2 mL of 4% sheep red blood cells (RBCs). Mice were sacrificed on the day of the test and injected with Hank’s solution supplemented with calf serum. The abdominal wash was sucked into the test tube along the abdomen, and 0.5 mL was added to the test tube with the same volume of chicken RBC suspension. Then, 0.5 mL of the mixed liquid was sucked into the agar circle on the cover glass, incubated at 37 °C for approximately 15 min to dry, and then rinsed to remove the cells that did not adhere to the wall, fixed with propanol/methanol solution, and stained. The time of staining was determined according to the actual situation. After completion, the specimens were rinsed with distilled water, dried, and observed using a microscope at 100× magnification for counting. The phagocytosis rate (%) and phagocytic index were calculated according to Equations (5) and (6), respectively:
2.5.7. Determination of NK Cell Activity
Following the procedure outlined in
Section 2.4.2, a splenocyte suspension was created, and the cell concentration was increased to 2 × 10
7 cells/mL. Then, 100 μL of target cells (YAC-1 cells) and effector cells (splenocytes) were added to U-type 96-well culture plates. RPMI-1640 culture medium and target cells were added to the natural release well, and 2.5% Triton and target cells were added to the maximum release well. After being aspirated, the supernatant was combined with the LDH matrix solution. A 1 M HCl solution was added after 3–10 min. At 490 nm, absorbance values were measured, and Equation (7) was used to determine the NK cell activity:
2.5.8. Determination of Immune Cytokines and Immunoglobulins
An appropriate amount of spleen was weighed and blended with normal saline in a ratio of 1:9. The spleen was homogenized using a tissue grinder over an ice water bath. The contents of immune cytokines IL-4 and IL-17A were determined by an ELISA kit (Hengyuan biological, Shanghai, China) at 4 °C.
After eyeball blood sampling, serum samples were collected to detect IgG and IgA levels via the corresponding kits.
2.5.9. Determination of Serum Biochemical Indicators
Blood samples were extracted from the mice’s eyes and centrifuged for 10 min at 2000× g. An automatic biochemical analyzer was used to measure 5 serum biochemical parameters.
2.5.10. 16S rDNA Sequencing and Analysis
Each mouse had its feces sampled, and the samples were kept at −80 °C. An OMEGA Soil DNA Kit (M5636-02) (Omega Bio-Tek, Norcross, GA, USA) was utilized to extract DNA. Using agarose gel electrophoresis and a NanoDrop NC2000 spectrophotometer (Molecular Devices, San Jose, CA, USA), the amount and quality of isolated DNA were determined. Using the reverse primer 806R (GGACTACHVGGGTWTCTAAT) and forward primer 338F (ACTCCTACGGGAGGCAGCA), the V3–V4 region of the bacterial 16S rRNA genes was amplified by PCR. The process of thermal cycling involved 5 min of initial denaturation at 98 °C, 25 cycles of denaturation at 98 °C, 30 s of annealing at 5 °C, 45 s of extension at 72 °C, and finally 5 min of extension at 72 °C. PCR amplicons were measured via the Invitrogen Quant-iT PicoGreen dsDNA Assay Kit (Invitrogen, Waltham, MA, USA). Following equal pooling of amplicons, pair-end 2–250 bp sequencing was carried out on the Illumina platform with the 500 cycle NovaSeq 6000 SP Reagent Kit (Illumina, San Diego, CA, USA).
2.5.11. Determination of SCFAs
GC was used to determine the levels of SCFAs in stool samples. A fecal sample of 0.1 g was weighed into a centrifuge tube containing a saturated NaCl solution of 0.5 mL, homogenized, acidified by adding H2SO4, and spun for 30 s. Anhydrous ether was added, and the vessel stirred for 30 s. SCFAs were extracted at 14,000 rpm/min for 15 min. Anhydrous sodium sulfate solution was added to the supernatant, and the supernatant was centrifuged under the above conditions, filtered with a filter membrane, and transferred to a gas flask for analysis and determination.
2.6. Data Analysis
SPSS 27.0.1 software was used to perform a one-way ANOVA. Other statistical analyses were performed using Excel 2018. Data visualization and plotting were achieved using PeakFit v4.12 and GraphPad Prism 8.02. Every experiment was repeated three times, and the results are expressed as means ± SD. p < 0.05 indicates a significant difference, and p < 0.01 is considered highly significant.
4. Discussion
As one of the body’s many defense mechanisms, the immune system plays a vital physiological role. By monitoring and recognizing self and non-self components, it protects the body by recognizing and eliciting an appropriate response to threats, such as toxins and invasion by pathogenic microorganisms, as well as removing internally damaged cells and maintaining body health [
21]. The immune system of the spleen guards the body against pathogen invasion, so an analysis of the immune reactivity of spleen cells reveals the immune capacity of the host [
22]. Proliferation of splenic lymphocytes is the most accurate measure of the immunological state of the host [
23]. When infections are met, natural killer cells (NK cells) react promptly by secreting cytokines, including TNF-α and IFN-γ [
24]. In addition, ingredients in food, including CN hydrolysate, whey protein and its enzymatic hydrolysate, and plant polysaccharides, have in vitro immune effects [
25]. CN, a component with important nutritional value in food, plays a crucial role in immune regulation. On this basis, we found that β-CN digest dietary intervention at 25–100 μg/mL led to noticeably increased splenic lymphocyte proliferation, phagocytic capacity of macrophages, and NK cell activity in a dose-dependent pattern compared to no dietary intervention.
CTX is an alkylating agent that is used as an antineoplastic drug and an immunosuppressant [
26]. It can cause significant immunosuppressive effects on the gastrointestinal tract, genital system, and immune system of animals and has been revealed to induce liver function damage and intestinal microbiota disorder and destroy intestinal mucosal immunity [
27]. In the current study, CTX was used as an immunosuppressant to establish an immunosuppressive mice model, and the immune status of the mice was observed via examination of spleen tissue sections to determine the immunosuppressive effect.
Important immune system organs include the spleen and thymus, whose organ indices can provide insight into how well the body’s immune system is functioning [
28]. Therefore, a decrease in the immune organ index is one of the representative symptoms of immune suppression in mice. We found that CTX induced immunosuppression in BALB/c mice, resulting in a decrease in the spleen index and thymus index in group M, while different doses of A2 β-CN noticeably augmented these two indexes and improved the growth and development of immunodeficient mice. The immunomodulatory effect of A1 β-CN was lower than that of A2 β-CN at the same dose. Compared with the NC group, the immunosuppressed mice showed a physiological state of low immune function. Hematoxylin–eosin staining indicated that the morphological structure of the spleen was severely scattered, the organizational structure was disordered, and the pathological damage was intense, which explained the decrease in the spleen index.
The main immune cells are NK cells, T cells, and macrophages. T cells are essential for cellular immunity [
29]. The basis for the T lymphocyte proliferation experiment is the lymphocytes’ reactivity to mitogens. ConA has the ability to stimulate T cell transformation and proliferation, which modulates immunity. As a result, the capacity of spleen cells to proliferate when triggered by ConA may indicate how well the cells are functioning immunologically. The two main effector cells in innate immunity, macrophages and natural killer cells, are crucial in antitumor and anti-infection processes. In this study, we found significant reductions in the proliferation of splenic lymphocytes, phagocytosis of macrophages, and activity of NK cells, demonstrating that CTX noticeably inhibited the immune function of mice and destroyed the stability of the body’s immune system. However, different doses of A2 β-CN significantly increased the proliferation of splenic lymphocytes, the phagocytosis of macrophages, and NK cell activity, and the immune status was improved.
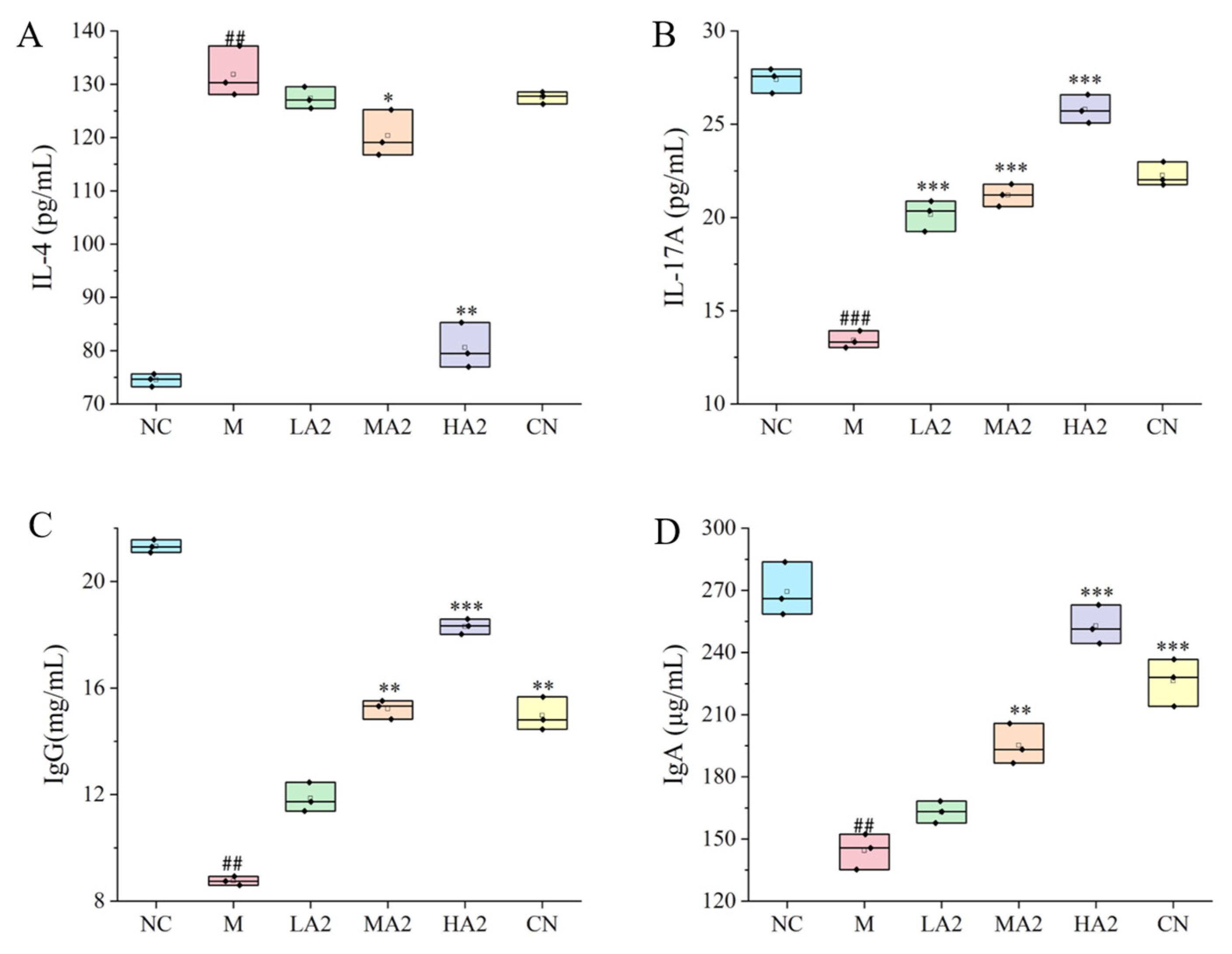
In the process of immunosuppression, the body will produce a series of inflammatory responses through the interaction of anti-inflammatory and pro-inflammatory factors. IL-4 is a multifunctional cytokine with an important role in cells and is involved in innate and acquired immunity in vivo [
30]. Depending on the model of intestinal inflammation, it can play either a pro- or anti-inflammatory role as a key mediator of intestinal inflammation. One significant pro-inflammatory cytokine that has a role in the immune response is IL-17A. We found that the levels of pro-inflammatory cytokines in group M were observably lower than in group NC. Compared with group M, the serum level of IL-17A in the protein intervention group was significantly increased, and compared with group NC, the serum level of IL-17A in group M observably increased. In addition, the content of IL-4 in serum was significantly reduced after the gavage of medium and high doses of protein, which is consistent with other research results [
31].
The serum biochemical indexes of experimental animals generally reflect the absorption and distribution of nutrients and their amount in the blood and further reflect the physiological state of experimental animals. Some studies have shown an increase in allergic or inflammatory responses after consumption of A1 milk compared with A2 milk. A study reported elevated levels of IgE and IgG in the serum of mice fed with A1 milk, and the intake of A1 β-CN compared with A2 β-CN induced an intestinal inflammatory response by activating the T helper 2 pathway [
18]. However, another study found no difference in serum IgG levels between the groups of mice fed a diet containing lyophilized A1 milk and lyophilized A2 milk and the control group, and the level of intestinal inflammation was comparable among all three groups [
32]. We found that our A2 β-CN dietary intervention could significantly increase the levels of IgG and IgA in the serum of mice, promote the production of proteins by lymphocytes in the immune system of mice, and induce the conversion of antigens to antibodies. Meanwhile, A2 β-CN showed a stronger immune-enhancing effect than A1 β-CN at the same dose.
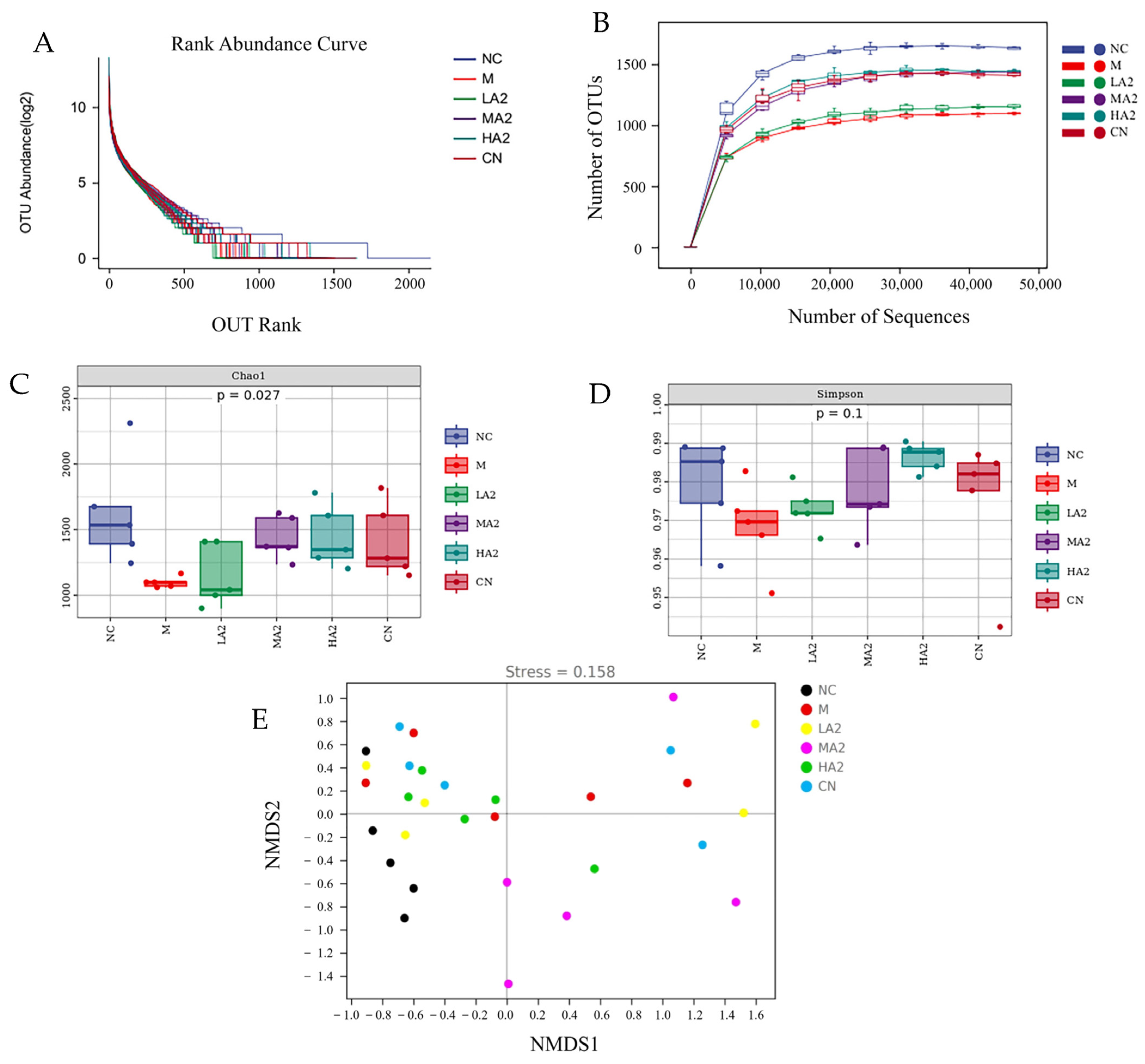
With the development of gut microbiota research, diet is being increasingly considered as a key factor in the regulation of the gut microbiota. Many studies have reported the effect of proteins on the gut microbiota. For instance, they discovered that A2 β-CN altered the composition of the gut microbiota in aging mice [
32]. Although A2 β-CN has attracted much attention, few studies have discussed the effect of A2 β-CN on intestinal health. In the current study, we explored the effects of A2 β-CN on intestinal health in immunosuppressed mice by measuring the alpha and beta diversity of the gut microbiota. At the family level, we found that an A2 β-CN diet significantly inhibited the colonization of harmful bacteria. At the genus level, we found that compared with the NC group, the relative abundance of the harmful bacteria
Coprococcus and
Desulfovibrio increased. Compared with group M, the A2 β-CN diet could increase the relative abundance of
Lactobacillus and
Anobacterium and reduce the relative abundance of
Coprococcus and
Desulfovibrio in mice.
Oscillospira is widely distributed in the intestinal tract of animals and humans. At other levels, it has been shown to be associated with diseases such as chronic constipation, emaciation, and gallstones, as well as positive or negative changes in pathogenesis.
Alistipes, a Gram-negative bacterium in the phylum
Bacteroidetes and a relatively new genus, is found in the healthy human gut [
33].
A heat map analysis of the species composition at the genus level showed that CTX induction observably affected the NC mice and even changed the colony abundance, and the intestinal microecology was destroyed. However, the A2 β-CN diet improved the CTX-induced flora imbalance to a certain extent, regulated the gut microbiota imbalance in immunosuppressed mice, and augmented the colonization of salutary bacteria in the gastrointestinal tract to make it closer to the level in NC mice and achieve the purpose of alleviating the reduced immune ability.
Acetic acid, propionic acid, butyric acid, valeric acid, caproic acid, and their isomers are examples of SCFAs, which are SFAs with a carbon chain consisting of one to six carbon atoms [
34]. Their possible functions include immune system regulation, lipid metabolism, weight control, glucose homeostasis, and inflammatory responses [
29,
35]. It has been demonstrated that butyric and acetic acids control the body’s immunological response primarily by attaching to various receptors to encourage immune cell development and activate intestinal immune cells [
36]. Moreover, it has been demonstrated that modifications to the gut microbiota directly impact the synthesis of SCFAs and quicken the host’s antibody responses [
37].
In this study, we found that the content of SCFAs in the intestine of the group M mice was noticeably reduced, which is consistent with other research results [
38]. It may be due to the reduced variety of gut microbiota in mice, as induced by CTX, resulting in a reduction in the species or the number of SCFA-producing microorganisms. On the other hand, the A2 β-CN diet has the potential to mitigate this decline, comparatively elevate the SCFA content, and markedly elevate the levels of acetic acid, propionic acid, butyric acid, and total acid.