The Impact of Combined Nutrition and Exercise Interventions in Patients with Chronic Kidney Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Protocol
2.2. Search Strategy
2.3. In and Exclusion Criteria
2.4. Outcome Measures
- Nutritional status;
- Dietary protein- and energy intake (24-h recall or dietary record),
- Body weight and body composition (BODPOD, dual-energy X-ray absorptiometry or bio-impedance spectroscopy),
- Acute phase proteins (C-reactive protein (CRP) and (pre)albumin).
- Muscle strength (HGS; dynamometer), knee or leg extension (one-repetition maximum (1 RM) or dynamometer) and leg press (1 RM);
- Physical performance (6-min walk test (6-MWT), timed up-and-go (TUG), short physical performance battery (SPPB), gait speed and sit-to-stand (STS));
- QoL (mental composite score (MCS) and physical composite score (PCS)).
2.5. Screening and Data Extraction
2.6. Risk of Bias and Quality Assessment
2.7. Data Analysis and Statistical Methods
3. Results
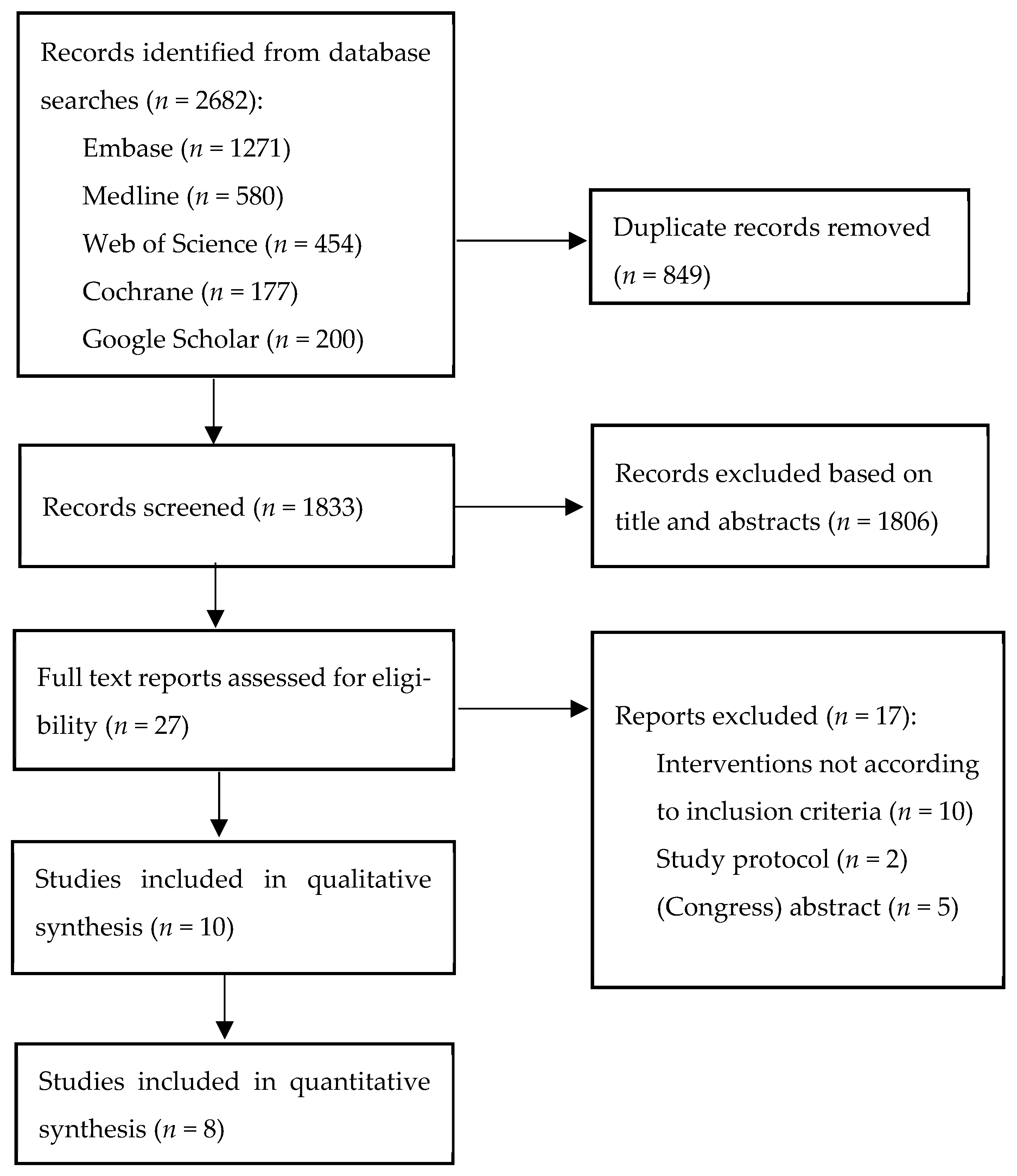
3.1. Study Selection
3.2. Study Characteristics
3.2.1. General Characteristics of Studies
3.2.2. Intervention Groups
3.2.3. Control Groups
3.3. Risk of Bias
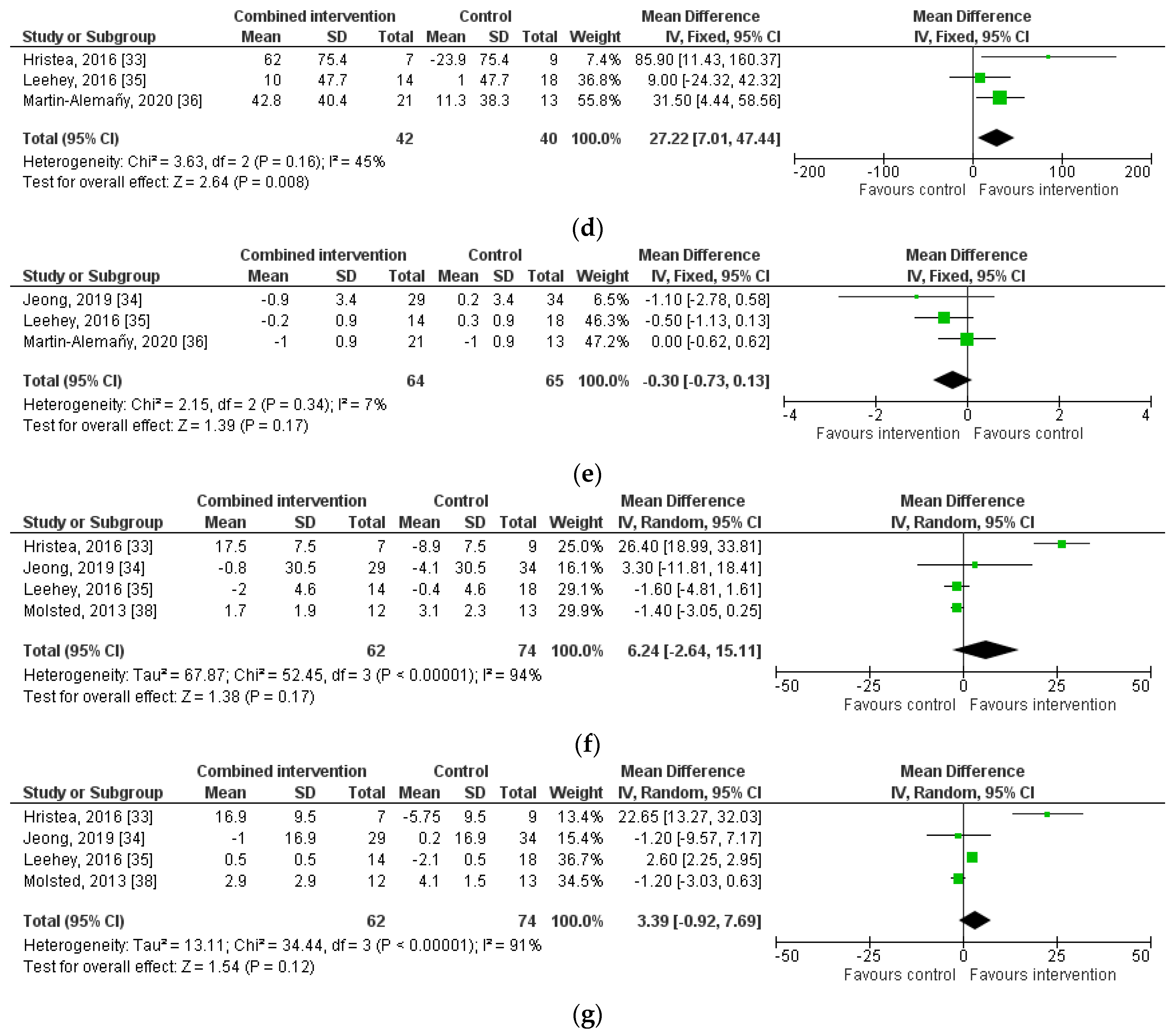
3.4. The Effect of Combined Nutritional and Exercise Interventions on Nutritional Status
3.4.1. Nutritional Intake
3.4.2. Body Weight and Body Composition
3.4.3. Acute Phase Proteins
3.5. The Effect of Combined Nutritional and Exercise Interventions on Muscle Strength
3.6. The Effect of Combined Nutritional and Exercise Interventions on Physical Performance
3.7. The Effect of Combined Nutritional and Exercise Interventions on QoL
4. Discussion
4.1. Limitations
4.2. Effect of Combined Nutritional and Exercise Interventions on Lean Body Mass and Muscle Strength
4.3. Effect of Combined Nutritional and Exercise Interventions on Physical Performance
4.4. Effect of Combined Nutritional and Exercise Interventions on QoL
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Souweine, J.S.; Kuster, N.; Chenine, L.; Rodriguez, A.; Patrier, L.; Morena, M.; Badia, E.; Chalabi, L.; Raynal, N.; Ohresser, I.; et al. Physical inactivity and protein energy wasting play independent roles in muscle weakness in maintenance haemodialysis patients. PLoS ONE 2018, 13, e0200061. [Google Scholar] [CrossRef]
- Kopple, J.D. Physical performance and all-cause mortality in CKD. J. Am. Soc. Nephrol. 2013, 24, 689–690. [Google Scholar] [CrossRef][Green Version]
- Tsai, Y.C.; Chen, H.M.; Hsiao, S.M.; Chen, C.S.; Lin, M.Y.; Chiu, Y.W.; Hwang, S.J.; Kuo, M.C. Association of physical activity with cardiovascular and renal outcomes and quality of life in chronic kidney disease. PLoS ONE 2017, 12, e0183642. [Google Scholar] [CrossRef]
- Kim, J.C.; Kalantar-Zadeh, K.; Kopple, J.D. Frailty and Protein-Energy Wasting in Elderly Patients with End Stage Kidney Disease. J. Am. Soc. Nephrol. 2013, 24, 337–351. [Google Scholar] [CrossRef] [PubMed]
- Cederholm, T.; Jensen, G.L.; Correia, M.I.T.D.; Gonzalez, M.C.; Fukushima, R.; Higashiguchi, T.; Baptista, G.; Barazzoni, R.; Blaauw, R.; Coats, A.J.S.; et al. GLIM criteria for the diagnosis of malnutrition—A consensus report from the global clinical nutrition community. J. Cachexia Sarcopeni 2019, 10, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Ikizler, T.A.; Cano, N.J.; Franch, H.; Fouque, D.; Himmelfarb, J.; Kalantar-Zadeh, K.; Kuhlmann, M.K.; Stenvinkel, P.; TerWee, P.; Teta, D.; et al. Prevention and treatment of protein energy wasting in chronic kidney disease patients: A consensus statement by the International Society of Renal Nutrition and Metabolism. Kidney Int. 2013, 84, 1096–1107. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.Z.C.; Cederholm, T.; Gonzalez, M.C.; Lindholm, B.; Avesani, C.M. GLIM in chronic kidney disease: What do we need to know? Clin. Nutr. 2023, 42, 937–943. [Google Scholar] [CrossRef] [PubMed]
- Obi, Y.; Qader, H.; Kovesdy, C.P.; Kalantar-Zadeh, K. Latest consensus and update on protein-energy wasting in chronic kidney disease. Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Sundell, M.B.; Cavanaugh, K.L.; Wu, P.S.; Shintani, A.; Hakim, R.M.; Ikizler, T.A. Oral Protein Supplementation Alone Improves Anabolism in a Dose-Dependent Manner in Chronic Hemodialysis Patients. J. Renal Nutr. 2009, 19, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Kalantar-Zadeh, K.; Cano, N.J.; Budde, K.; Chazot, C.; Kovesdy, C.P.; Mak, R.H.; Mehrotra, R.; Raj, D.S.; Sehgal, A.R.; Stenvinkel, P.; et al. Diets and enteral supplements for improving outcomes in chronic kidney disease. Nat. Rev. Nephrol. 2011, 7, 369–384. [Google Scholar] [CrossRef] [PubMed]
- Fouque, D.; McKenzie, J.; de Mutsert, R.; Azar, R.; Teta, D.; Plauth, M.; Cano, N.; Renilon Multicentre Trial Study, G. Use of a renal-specific oral supplement by haemodialysis patients with low protein intake does not increase the need for phosphate binders and may prevent a decline in nutritional status and quality of life. Nephrol. Dial. Transplant. 2008, 23, 2902–2910. [Google Scholar] [CrossRef]
- Lu, Y.; Wang, Y.J.; Lu, Q. The effect of oral nutritional supplement on muscle fitness of patients undergoing dialysis: A systematic review and meta-analysis. J. Adv. Nurs. 2021, 77, 1716–1730. [Google Scholar] [CrossRef]
- Wang, X.N.H.; Mitch, W.E. Mechanisms of muscle wasting in chronic kidney disease. Nat. Rev. Nephrol. 2014, 10, 504–516. [Google Scholar] [CrossRef]
- Zelle, D.M.; Klaassen, G.; van Adrichem, E.; Bakker, S.J.L.; Corpeleijn, E.; Navis, G. Physical inactivity: A risk factor and target for intervention in renal care. Nat. Rev. Nephrol. 2017, 13, 152–168. [Google Scholar] [CrossRef]
- Sheshadri, A.; Kittiskulnam, P.; Lazar, A.A.; Johansen, K.L. A Walking Intervention to Increase Weekly Steps in Dialysis Patients: A Pilot Randomized Controlled Trial. Am. J. Kidney Dis. 2020, 75, 488–496. [Google Scholar] [CrossRef] [PubMed]
- Ekramzadeh, M.; Santoro, D.; Kopple, J.D. The Effect of Nutrition and Exercise on Body Composition, Exercise Capacity, and Physical Functioning in Advanced CKD Patients. Nutrients 2022, 14, 2129. [Google Scholar] [CrossRef] [PubMed]
- Heiwe, S.; Jacobson, S.H. Exercise training in adults with CKD: A systematic review and meta-analysis. Am. J. Kidney Dis. 2014, 64, 383–393. [Google Scholar] [CrossRef] [PubMed]
- Song, W.J.; Sohng, K.Y. Effects of progressive resistance training on body composition, physical fitness and quality of life of patients on hemodialysis. J. Korean Acad. Nurs. 2012, 42, 947–956. [Google Scholar] [CrossRef] [PubMed]
- Andersen, L.L.; Tufekovic, G.; Zebis, M.K.; Crameri, R.M.; Verlaan, G.; Kjaer, M.; Suetta, C.; Magnusson, P.; Aagaard, P. The effect of resistance training combined with timed ingestion of protein on muscle fiber size and muscle strength. Metabolism 2005, 54, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Biolo, G.; Tipton, K.D.; Klein, S.; Wolfe, R.R. An abundant supply of amino acids enhances the metabolic effect of exercise on muscle protein. Am. J. Physiol. 1997, 273, E122–E129. [Google Scholar] [CrossRef] [PubMed]
- Esmarck, B.; Andersen, J.L.; Olsen, S.; Richter, E.A.; Mizuno, M.; Kjaer, M. Timing of postexercise protein intake is important for muscle hypertrophy with resistance training in elderly humans. J. Physiol. 2001, 535, 301–311. [Google Scholar] [CrossRef]
- Liao, C.D.; Tsauo, J.Y.; Wu, Y.T.; Cheng, C.P.; Chen, H.C.; Huang, Y.C.; Chen, H.C.; Liou, T.H. Effects of protein supplementation combined with resistance exercise on body composition and physical function in older adults: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2017, 106, 1078–1091. [Google Scholar] [CrossRef] [PubMed]
- Nunes, E.A.; Colenso-Semple, L.; McKellar, S.R.; Yau, T.; Ali, M.U.; Fitzpatrick-Lewis, D.; Sherifali, D.; Gaudichon, C.; Tome, D.; Atherton, P.J.; et al. Systematic review and meta-analysis of protein intake to support muscle mass and function in healthy adults. J. Cachexia Sarcopenia Muscle 2022, 13, 795–810. [Google Scholar] [CrossRef]
- Pupim, L.B.; Flakoll, P.J.; Levenhagen, D.K.; Ikizler, T.A. Exercise augments the acute anabolic effects of intradialytic parenteral nutrition in chronic hemodialysis patients. Am. J. Physiol. Endocrinol. Metab. 2004, 286, E589–E597. [Google Scholar] [CrossRef] [PubMed]
- Majchrzak, K.M.; Pupim, L.B.; Flakoll, P.J.; Ikizler, T.A. Resistance exercise augments the acute anabolic effects of intradialytic oral nutritional supplementation. Nephrol. Dial. Transplant. 2008, 23, 1362–1369. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Ikizler, T.A.; Burrowes, J.D.; Byham-Gray, L.D.; Campbell, K.L.; Carrero, J.J.; Chan, W.; Fouque, D.; Friedman, A.N.; Ghaddar, S.; Goldstein-Fuchs, D.J.; et al. KDOQI Clinical Practice Guideline for Nutrition in CKD: 2020 Update. Am. J. Kidney Dis. 2020, 76, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Fouque, D.; Kalantar-Zadeh, K.; Kopple, J.; Cano, N.; Chauveau, P.; Cuppari, L.; Franch, H.; Guarnieri, G.; Ikizler, T.A.; Kaysen, G.; et al. A proposed nomenclature and diagnostic criteria for protein-energy wasting in acute and chronic kidney disease. Kidney Int. 2008, 73, 391–398. [Google Scholar] [CrossRef]
- Cumpston, M.; Li, T.J.; Page, M.J.; Chandler, J.; Welch, V.A.; Higgins, J.P.T.; Thomas, J. Updated guidance for trusted systematic reviews: A new edition of the Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Database Syst. Rev. 2019, 10, ED000142. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Savovic, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ. 2019, 366, l4898. [Google Scholar] [CrossRef]
- Review Manager [RevMan], version 5.4.5.3; The Nordic Cochrane Centre, The Cochrane Collaboraion: Copenhaven, Denmark, 2022.
- Dong, J.; Sundell, M.B.; Pupim, L.B.; Wu, P.; Shintani, A.; Ikizler, T.A. The effect of resistance exercise to augment long-term benefits of intradialytic oral nutritional supplementation in chronic hemodialysis patients. J. Ren. Nutr. 2011, 21, 149–159. [Google Scholar] [CrossRef]
- Hristea, D.; Deschamps, T.; Paris, A.; Lefrancois, G.; Collet, V.; Savoiu, C.; Ozenne, S.; Coupel, S.; Testa, A.; Magnard, J. Combining intra-dialytic exercise and nutritional supplementation in malnourished older haemodialysis patients: Towards better quality of life and autonomy. Nephrology 2016, 21, 785–790. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.H.; Biruete, A.; Tomayko, E.J.; Wu, P.T.; Fitschen, P.; Chung, H.R.; Ali, M.; McAuley, E.; Fernhall, B.; Phillips, S.A.; et al. Results from the randomized controlled IHOPE trial suggest no effects of oral protein supplementation and exercise training on physical function in hemodialysis patients. Kidney Int. 2019, 96, 777–786. [Google Scholar] [CrossRef] [PubMed]
- Leehey, D.J.; Collins, E.; Kramer, H.J.; Cooper, C.; Butler, J.; McBurney, C.; Jelinek, C.; Reda, D.; Edwards, L.; Garabedian, A.; et al. Structured Exercise in Obese Diabetic Patients with Chronic Kidney Disease: A Randomized Controlled Trial. Am. J. Nephrol. 2016, 44, 54–62. [Google Scholar] [CrossRef]
- Martin-Alemany, G.; Espinosa-Cuevas, M.L.A.; Perez-Navarro, M.; Wilund, K.R.; Miranda-Alatriste, P.; Cortes-Perez, M.; Garcia-Villalobos, G.; Gomez-Guerrero, I.; Cantu-Quintanilla, G.; Ramirez-Mendoza, M.; et al. Effect of Oral Nutritional Supplementation With and Without Exercise on Nutritional Status and Physical Function of Adult Hemodialysis Patients: A Parallel Controlled Clinical Trial (AVANTE-HEMO Study). J. Ren. Nutr. 2020, 30, 126–136. [Google Scholar] [CrossRef]
- Martin-Alemany, G.; Valdez-Ortiz, R.; Olvera-Soto, G.; Gomez-Guerrero, I.; Aguire-Esquivel, G.; Cantu-Quintanilla, G.; Lopez-Alvarenga, J.C.; Miranda-Alatriste, P.; Espinosa-Cuevas, A. The effects of resistance exercise and oral nutritional supplementation during hemodialysis on indicators of nutritional status and quality of life. Nephrol. Dial. Transplant. 2016, 31, 1712–1720. [Google Scholar] [CrossRef]
- Molsted, S.; Harrison, A.P.; Eidemak, I.; Andersen, J.L. The effects of high-load strength training with protein- or nonprotein-containing nutritional supplementation in patients undergoing dialysis. J. Ren. Nutr. 2013, 23, 132–140. [Google Scholar] [CrossRef]
- Castaneda, C.; Gordon, P.L.; Uhlin, K.L.; Levey, A.S.; Kehayias, J.J.; Dwyer, J.T.; Fielding, R.A.; Roubenoff, R.; Singh, M.F. Resistance training to counteract the catabolism of a low-protein diet in patients with chronic renal insufficiency. A randomized, controlled trial. Ann. Intern. Med. 2001, 135, 965–976. [Google Scholar] [CrossRef] [PubMed]
- Castaneda, C.; Gordon, P.L.; Parker, R.C.; Uhlin, K.L.; Roubenoff, R.; Levey, A.S. Resistance training to reduce the malnutrition-inflammation complex syndrome of chronic kidney disease. Am. J. Kidney Dis. 2004, 43, 607–616. [Google Scholar] [CrossRef]
- Martin-Alemany, G.; Perez-Navarro, M.; Wilund, K.R.; Garcia-Villalobos, G.; Gomez-Guerrero, I.; Cantu-Quintanilla, G.; Reyes-Caldelas, M.A.; Espinosa-Cuevas, A.; Escobedo, G.; Medeiros, M.; et al. Effect of Intradialytic Oral Nutritional Supplementation with or without Exercise Improves Muscle Mass Quality and Physical Function in Hemodialysis Patients: A Pilot Study. Nutrients 2022, 14, 2946. [Google Scholar] [CrossRef]
- Carrero, J.J.; Stenvinkel, P.; Cuppari, L.; Ikizler, T.A.; Kalantar-Zadeh, K.; Kaysen, G.; Mitch, W.E.; Price, S.R.; Wanner, C.; Wang, A.Y.; et al. Etiology of the protein-energy wasting syndrome in chronic kidney disease: A consensus statement from the International Society of Renal Nutrition and Metabolism (ISRNM). J. Ren. Nutr. 2013, 23, 77–90. [Google Scholar] [CrossRef]
- Watson, E.L.; Greening, N.J.; Viana, J.L.; Aulakh, J.; Bodicoat, D.H.; Barratt, J.; Feehally, J.; Smith, A.C. Progressive Resistance Exercise Training in CKD: A Feasibility Study. Am. J. Kidney Dis. 2015, 66, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Headley, S.; Germain, M.; Milch, C.; Pescatello, L.; Coughlin, M.A.; Nindl, B.C.; Cornelius, A.; Sullivan, S.; Gregory, S.; Wood, R. Exercise Training Improves HR Responses and V˙O2peak in Predialysis Kidney Patients. Med. Sci. Sport. Exer. 2012, 44, 2392–2399. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Perez de Villar, L.; Martinez-Olmos, F.J.; Junque-Jimenez, A.; Amer-Cuenca, J.J.; Martinez-Gramage, J.; Mercer, T.; Segura-Orti, E. Test-retest reliability and minimal detectable change scores for the short physical performance battery, one-legged standing test and timed up and go test in patients undergoing hemodialysis. PLoS ONE 2018, 13, e0201035. [Google Scholar] [CrossRef] [PubMed]
- Painter, P.; Carlson, L.; Carey, S.; Paul, S.M.; Myll, J. Physical functioning and health-related quality-of-life changes with exercise training in hemodialysis patients. Am. J. Kidney Dis. 2000, 35, 482–492. [Google Scholar] [CrossRef]
- Campbell, K.L.; Ash, S.; Bauer, J.D. The impact of nutrition intervention on quality of life in pre-dialysis chronic kidney disease patients. Clin. Nutr. 2008, 27, 537–544. [Google Scholar] [CrossRef]
- Kalantar-Zadeh, K.; Unruh, M. Health related quality of life in patients with chronic kidney disease. Int. Urol. Nephrol. 2005, 37, 367–378. [Google Scholar] [CrossRef]


| First Author, Year | Study Design | Trial Duration (mo), Sample Size | Study Participants, Mean Age | Methods of Intervention | Relevant Outcomes Measures | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Nutrition | Exercise | Nutritional Status | Muscle Strength | Physical Performance | QoL | |||||
| Type | Frequency | |||||||||
| Castaneda, 2001 & 2004 [39,40] | RCT | 3 I: 14 C: 12 | CKD, non-dialysis 65 ± 10 y | I: Supervised LPD 0.6 g/kg C: Same as intervention | I: Supervised resistance training C: Supervised low-intensity exercises | I: 3 times/wk C: 3 times/wk | Protein intake Energy intake Body weight BMI CRP Albumin Pre albumin | Leg press Knee extension | Not reported | Not reported |
| Dong, 2011 [32] | RCT | 6 I: 15 C: 17 | HD (3 times/wk) 43 ± 13 y | I: ONS (2 × 480 kcal, 17 g protein) taken prior to, during or after HD C: Same as intervention | I: Supervised resistance training C: No exercise | I: 3 times/wk | Protein intake Energy intake BMI LBM CRP Albumin Pre albumin | Leg press | Not reported | Not reported |
| Hristea, 2016 [33] | RCT | 6 I: 7 C: 9 | HD (3 times/wk) diagnosed with PEW 70 ± 15 y | I: Dietary counseling by a dietitian (aiming 30–40 kcal/kg and >1.1 g/kg ideal weight/day) C: Same as intervention | I: Supervised aerobic training C: No exercise | I: 3 times/wk | Protein intake Energy intake BMI LTI CRP Albumin Pre albumin | Knee extension | 6-MWT | SF-36 PCS SF-36 MCS |
| Jeong, 2019 [34] | RCT | 12, I1: 38 I2: 29 C: 34 | HD (3 times/wk) 55 ± 12 y | I1: 30 g whey protein supplement during HD I2: Same as intervention 1 C: ±150 g of non-caloric, non-protein containing beverage during HD | I1: No exercise I2: Supervised aerobic training C: No exercise | I2: 3 times/wk | Protein intake Energy intake BMI Lean mass Albumin CRP | Knee extension | SWT TUG Normal gait speed STS | SF-36 PCS SF-36 MCS |
| Leehey, 2016 [35] | RCT | 12 I: 18 C: 18 | CKD (stage 2–4) with type 2 diabetes and BMI > 30 m2/kg 66 ± 8 y | I: Dietary counseling at baseline aiming for a 200–250 calorie deficit with 9 follow-up calls C: Same as intervention | I: Resistance and aerobic training C: No exercise | I: 3 times/wk | Lean body weight BMI CRP | Knee extension | 6-MWT TUG | SF-36 PCS SF-36 MCS |
| Martin-Alemañy, 2022 [41] | RCT | 24 I: 10 C: 14 | HD (2–3 times/wk) 34 ± 11 | I: ONS (2 × 434 kcal, 19 g protein) C: ONS (2 × 434 kcal, 19 g protein) | I: Supervised resistance and aerobic training C: No exercise | I: 2–3 times/wk | Body weight CRP Albumin | HGS | 6-MWT TUG STS Gait speed | KDQOL-SF |
| Martin-Alemañy, 2020 [36] | RCT | 3 I1: 9 I2: 12 C: 13 | HD (2–3 times/wk) 29 ± 9 | I1: ONS (1 × 480 kcal, 20 g protein) taken during HD I2: Same as intervention C: Same as intervention | I1: Supervised resistance training I2: Supervised aerobic training C: No exercise | I1: 2–3 times/wk I2: 2–3 times/wk | Body weight BMI CRP Albumin | HGS | 6-MWT TUG STS | KDQOL-SF |
| Martin-Alemañy, 2016 [37] | RCT | 3 I: 17 C: 19 | HD (2 times/wk) 34 (25–43) | I: ONS (1 × 430 kcal, 19 g protein) before and during HD C: Same as intervention | I: Supervised resistance training C: No exercise | I: 3 times/wk | Protein intake Energy intake Body weight BMI Albumin | HGS | Not reported | KDQOL-SF |
| Molsted, 2013 [38] | RCT | 4 I: 16 C: 13 | HD (24), PD (5) 55 ± 14 | I: ONS (1 × 250 kcal, 9 g protein, 25 g carbohydrates) C: ONS (1 × 250 kcal, 0 g protein, 2 g carbohydrates) | I: Supervised resistance training C: Same as intervention | I: 3 times/wk C: 3 times/wk | Not reported | Knee extension right | CTS | SF-36 PCS SF-36 MCS |
| Author | Outcomes | Results * | ||||
|---|---|---|---|---|---|---|
| Intervention | Control | p-Value | ||||
| Baseline | Last Follow-Up | Baseline | Last Follow-Up | |||
| Castenada [39,40] | Protein intake (g/kg) | 0.6 ± 0.1 | 0.6 ± 0.1 | 0.7 ± 0.1 | 0.6 ± 0.1 | p > 0.2 |
| Energy intake (J/kg) | 68 ± 27 | 76 ± 32 | 87 ± 28 | 98 ± 25 | p > 0.2 | |
| Body weight (kg) | 85 ± 16 | 85 ± 16 | 76 ± 14 | 73 ± 9 | p = 0.05 | |
| CRP (mg/L) | 8 ± 6 | 6 ± 6 | 6 ± 6 | 8 ± 6 | p = 0.05 (group effect) | |
| Albumin (g/dL) | 4 ± 0.3 | 4 ± 0.2 | 4 ± 0.4 | 4 ± 0.4 | p = 0.09 | |
| Prealbumin (mg/dL) | 253 ± 46 | 276 ± 42 | 232 ± 60 | 234 ± 50 | p = 0.05 | |
| Dong [32] | Protein intake (g/kg) | 0.8 ± 0.2 | 1.0 ± 0.3 | 0.8 ± 0.3 | 1.1 ± 0.4 | p > 0.05 |
| Energy intake (kcal/kg) | 24 ± 7 | 27 ± 7 | 22 ± 9 | 28 ± 12 | p > 0.05 | |
| Body weight (kg) | 76 ± 15 | 75 ± 13 | 84 ± 17 | 86 ± 21 | p = 0.02 (↑ overall time effect group) | |
| CRP (mg/L) | 4 (2–13) | 3 (1.3–8.3) | 4 (1–12) | 7 (6–12) | p > 0.05 | |
| Albumin (mg/L) | 41 ± 3 | 42 ± 4 | 42 ± 3 | 42 ± 2 | p > 0.05 | |
| Prealbumin (mg/dL) | 40 ± 11 | 42 ± 12 | 38 ± 10 | 42 ± 7 | p > 0.05 | |
| Hristea [33] | Protein intake (g/kg) | 1.1 ± 0.2 | 1.2 ± 03 | 0.9 ± 0.2 | 1.2 ± 0.4 | p = 0.01 (overall time effect group) |
| Energy intake (kcal/kg) | 27 ± 4 | 30 ± 7 | 21 ± 4 | 28 ± 8 | p = 0.03 (main group effect, I ↑) | |
| CRP (mg/L) | 6 ± 8 | 2 ± 2 | 6 ± 6 | 5 ± 6 | Not reported | |
| Albumin (mg/L) | 38 ± 3 | 39 ± 3 | 40 ± 4 | 39 ± 4 | p = 0.03 (time*group, I ↑, C ↓) | |
| Prealbumin (g/L) | 226 ± 45 | 232 ± 27 | 251 ± 69 | 227 ± 56 | Not reported | |
| Jeong [34] | Protein intake (g/kg) | 0.8 ± 0.5 0.8 ± 0.4 | 0.9 ± 0.3 (I1) 1.0 ± 0.5 (I2) | 0.8 ± 0.4 | 0.7 ± 0.4 | p = 0.02 (time*group) |
| Energy intake (kcal/kg) | 18 ± 8 17 ± 7 | 19 ± 9 (I1) 20 ± 12 (I2) | 19 ± 11 | 17 ± 8 | p = 0.16 (time*group) | |
| CRP (mg/L) | 18 ± 21 15 ± 14 | 11 ± 8 (I1) 13 ± 12 (I2) | 7 ± 6 | 11 ± 11 | p = 0.40 (time*group) | |
| Albumin (g/L) | 4 ± 0.4 4 ± 0.4 | 4 ± 0.3 (I1) 4 ± 0.5 (I2) | 4 ± 0.3 | 4 ± 0.3 | p = 0.71 (time*group) | |
| Leehey [35] | CRP (mg/L) | 6 ± 8 | 8 ± 14 | 9 ± 11 | 7 ± 8 | p = 0.23 |
| Martin-Alemañy [41] | Body weight (kg) | 56 ± 9 | 58 ± 9 | 55 ± 7 | 56 ± 7 | p = 0.46 (time*group) |
| CRP (mg/L) | 5 (1–13) | 3 (3–9) | 6 (3–9) | 4 (2–7) | p = 0.78 (time*group) | |
| Albumin (g/dL) | 4 ± 0.4 | 4 ± 0.5 | 4 ± 0.5 | 4 ± 0.3 | p = 0.4 (time*group) | |
| Martin-Alemañy [36] | Body weight (kg) | 53 ± 6 52 ± 9 | 55 ± 5 (I1) 53 ± 8 (I2) | 52 ± 10 | 53 ± 9 | p = 0.22 (time*group) |
| CRP (mg/L) | 3 (3–9) 7 (3–13) | 6 (4–9) (I1) 5 (3–17) (I2) | 4 (2–4) | 3 (2–6) | p = 0.44 (time*group) | |
| Albumin (g/L) | 4 ± 0.5 4 ± 0.3 | 4 ± 0.4 (I1) 3 ± 0.5 (I2) | 4 ± 0.5 | 4 ± 0.4 | p = 0.42 (time*group) | |
| Martin-Alemañy [37] | Protein intake (g/kg) | 1.0 ± 0.4 | 1.6 ± 0.5 | 1.0 ± 0.6 | 1.6 ± 0.5 | ↑ (no p reported) |
| Energy intake (kcal/kg) | 27 ± 11 | 36 ± 15 | 27 ± 11 | 35 ± 16 | ↑ (no p reported) | |
| Body weight (kg) | 51 (46–57) | 52 (47–58) | 47 (43–52) | 49 (45–54) | ↑ (no p reported) | |
| Albumin (g/dL) | 3 ± 0.3 | 4 ± 0.3 | 4 ± 0.3 | 4 ± 0.4 | ↑ (no p reported) | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Geus, M.; Dam, M.; Visser, W.J.; Ipema, K.J.R.; de Mik-van Egmond, A.M.E.; Tieland, M.; Weijs, P.J.M.; Kruizenga, H.M. The Impact of Combined Nutrition and Exercise Interventions in Patients with Chronic Kidney Disease. Nutrients 2024, 16, 406. https://doi.org/10.3390/nu16030406
de Geus M, Dam M, Visser WJ, Ipema KJR, de Mik-van Egmond AME, Tieland M, Weijs PJM, Kruizenga HM. The Impact of Combined Nutrition and Exercise Interventions in Patients with Chronic Kidney Disease. Nutrients. 2024; 16(3):406. https://doi.org/10.3390/nu16030406
Chicago/Turabian Stylede Geus, Manon, Manouk Dam, Wesley J. Visser, Karin J. R. Ipema, Anneke M. E. de Mik-van Egmond, Michael Tieland, Peter J. M. Weijs, and Hinke M. Kruizenga. 2024. "The Impact of Combined Nutrition and Exercise Interventions in Patients with Chronic Kidney Disease" Nutrients 16, no. 3: 406. https://doi.org/10.3390/nu16030406
APA Stylede Geus, M., Dam, M., Visser, W. J., Ipema, K. J. R., de Mik-van Egmond, A. M. E., Tieland, M., Weijs, P. J. M., & Kruizenga, H. M. (2024). The Impact of Combined Nutrition and Exercise Interventions in Patients with Chronic Kidney Disease. Nutrients, 16(3), 406. https://doi.org/10.3390/nu16030406





