Immunonutrition in Acute Geriatric Care: Clinical Outcomes, Inflammatory Profiles, and Immune Responses
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects and Study Design
2.1.1. Primary Objective (Primary Endpoint)
2.1.2. Secondary Objectives (Secondary Endpoint)
2.2. Study Products
2.3. Clinical and Multidimensional Assessment
2.4. Analytical Method
2.5. Multiplex Assay
2.6. Flow Cytometric Evaluation of Immune System
2.7. Telomerase Activity in Peripheral Blood Mononuclear Cells (PBMC)
2.8. Statistical Analysis
3. Results
3.1. Clinical Variables
3.2. Plasma Inflammatory Molecules Profile
3.3. Immune System Profile
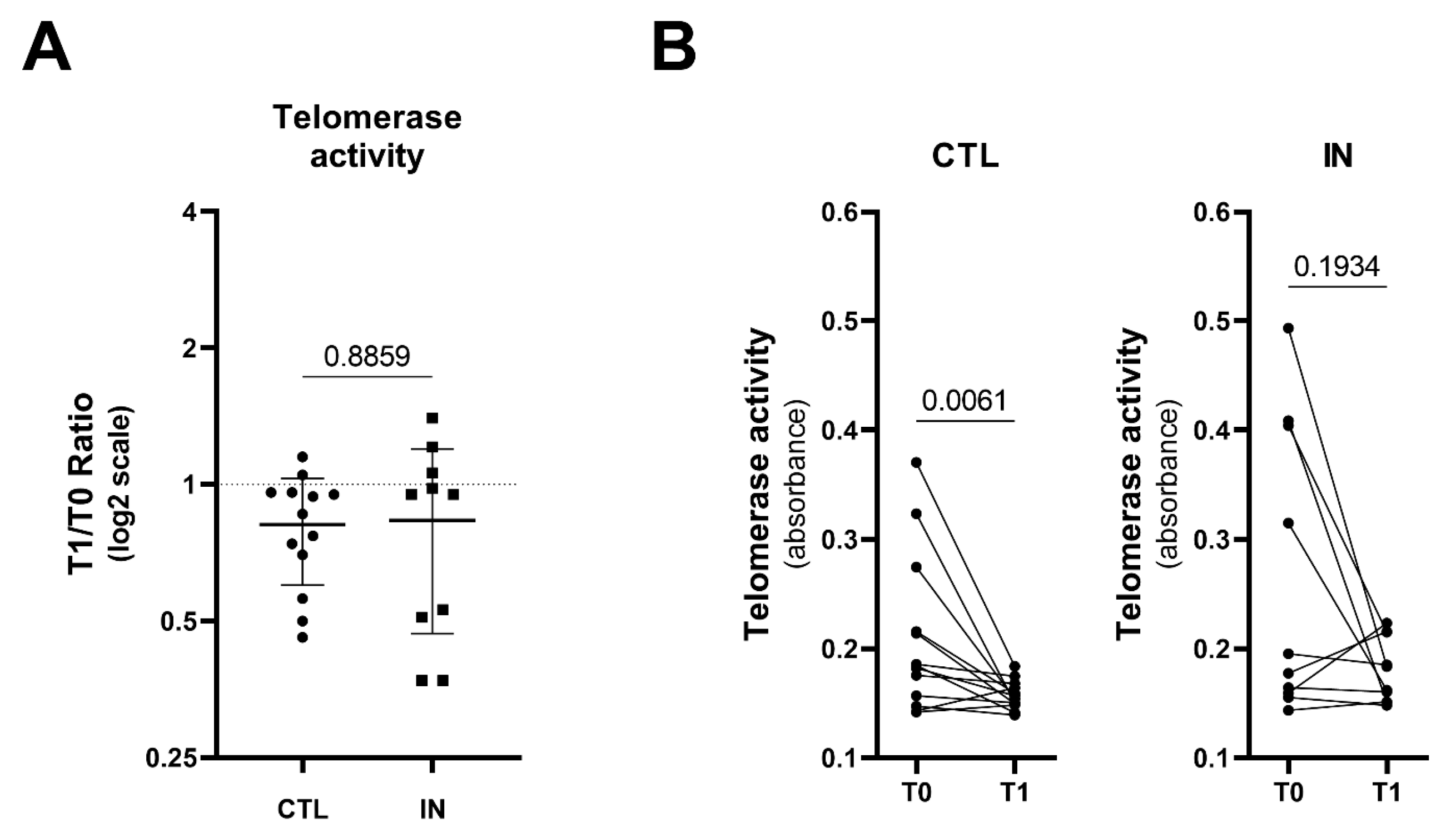
3.4. Telomerase Activity in PBMC
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dent, E.; Wright, O.R.L.; Woo, J.; Hoogendijk, E.O. Malnutrition in older adults. Lancet 2023, 401, 951–966. [Google Scholar] [CrossRef] [PubMed]
- Elia, M.; Stratton, R.J. How Much Undernutrition Is There in Hospitals? Br. J. Nutr. 2000, 84, 257–259. [Google Scholar] [CrossRef] [PubMed]
- Saunders, J.; Smith, T. Malnutrition: Causes and Consequences. Clin. Med. 2010, 10, 624. [Google Scholar] [CrossRef] [PubMed]
- Norman, K.; Haß, U.; Pirlich, M. Malnutrition in Older Adults—Recent Advances and Remaining Challenges. Nutrients 2021, 13, 2764. [Google Scholar] [CrossRef]
- Padilla, P.F.; Martínez, G.; Vernooij, R.W.M.; Urrútia, G.; Figuls, M.R.I.; Cosp, X.B. Early Enteral Nutrition (Within 48 Hours) versus Delayed Enteral Nutrition (after 48 Hours) with or without Supplemental Parenteral Nutrition in Critically Ill Adults. Cochrane Database Syst. Rev. 2019, 2019. [Google Scholar] [CrossRef]
- Dijkink, S.; Meier, K.; Krijnen, P.; Yeh, D.D.; Velmahos, G.C.; Schipper, I.B. Malnutrition and Its Effects in Severely Injured Trauma Patients. Eur. J. Trauma Emerg. Surg. 2020, 46, 993. [Google Scholar] [CrossRef]
- Chen, L.; Deng, H.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; Wang, X.; Zhao, L. Inflammatory Responses and Inflammation-Associated Diseases in Organs. Oncotarget 2018, 9, 7204. [Google Scholar] [CrossRef]
- Chakraborty, R.K.; Burns, B. Systemic Inflammatory Response Syndrome; StatPearls: Treasure Island, FL, USA, 2023. [Google Scholar]
- Boccardi, V.; Marano, L. Improving Geriatric Outcomes through Nutritional and Immunonutritional Strategies: Focus on Surgical Setting by a Comprehensive Evidence Review. Ageing Res. Rev. 2024, 96, 102272. [Google Scholar] [CrossRef]
- Braga, M.; Wischmeyer, P.E.; Drover, J.; Heyland, D.K. Clinical Evidence for Pharmaconutrition in Major Elective Surgery. JPEN J. Parenter. Enter. Nutr. 2013, 37, 66S–72S. [Google Scholar] [CrossRef]
- De Felice, F.; Cattaneo, C.G.; Poto, G.E.; Antropoli, C.; Brillantino, A.; Carbone, L.; Brunetti, O.; De Luca, R.; Desideri, I.; Incorvaia, L.; et al. Mapping the Landscape of Immunonutrition and Cancer Research: A Comprehensive Bibliometric Analysis on Behalf of NutriOnc Research Group. Int. J. Surg. 2024, 110, 395–405. [Google Scholar] [CrossRef]
- Gupta, R.; Senagore, A.; Perioperative, S. Immunonutrition within Enhanced Recovery after Surgery (ERAS): An Unresolved Matter. Perioper. Med. 2017, 6, 24. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Li, N.; Zhang, J.; Fu, Q.; Qiu, Y.; Chen, Y. The Effect of Immunonutrition in Patients Undergoing Pancreaticoduodenectomy: A Systematic Review and Meta-Analysis. BMC Cancer 2023, 23, 351. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, C.; Vieira, P.; Sá, H.; Malva, J.; Castelo-Branco, M.; Reis, F.; Viana, S. Polyphenols: Immunonutrients Tipping the Balance of Immunometabolism in Chronic Diseases. Front. Immunol. 2024, 15, 1360065. [Google Scholar] [CrossRef] [PubMed]
- Apaza, C.J.; Cerezo, J.F.; García-Tejedor, A.; Giménez-Bastida, J.A.; Laparra-Llopis, J.M. Revisiting the Immunometabolic Basis for the Metabolic Syndrome from an Immunonutritional View. Biomedicines 2024, 12, 1825. [Google Scholar] [CrossRef] [PubMed]
- Mohsen, G.; Stroemer, A.; Mayr, A.; Kunsorg, A.; Stoppe, C.; Wittmann, M.; Velten, M. Effects of Omega-3 Fatty Acids on Postoperative Inflammatory Response: A Systematic Review and Meta-Analysis. Nutrients 2023, 15, 3414. [Google Scholar] [CrossRef]
- Tao, K.M.; Li, X.Q.; Yang, L.Q.; Yu, W.F.; Lu, Z.J.; Sun, Y.M.; Wu, F.X. Glutamine Supplementation for Critically Ill Adults. Cochrane Database Syst. Rev. 2014, 2014. [Google Scholar] [CrossRef]
- Ortiz-Reyes, L.; Lee, Z.Y.; Chin Han Lew, C.; Hill, A.; Jeschke, M.G.; Turgeon, A.F.; Cancio, L.; Stoppe, C.; Patel, J.J.; Day, A.G.; et al. The Efficacy of Glutamine Supplementation in Severe Adult Burn Patients: A Systematic Review with Trial Sequential Meta-Analysis. Crit. Care Med. 2023, 51, 1086–1095. [Google Scholar] [CrossRef]
- Garrel, D.; Patenaude, J.; Nedelec, B.; Samson, L.; Dorais, J.; Champoux, J.; D’Elia, M.; Bernier, J. Decreased Mortality and Infectious Morbidity in Adult Burn Patients given Enteral Glutamine Supplements: A Prospective, Controlled, Randomized Clinical Trial. Crit. Care Med. 2003, 31, 2444–2449. [Google Scholar] [CrossRef]
- Compher, C.; Bingham, A.L.; McCall, M.; Patel, J.; Rice, T.W.; Braunschweig, C.; McKeever, L. Guidelines for the Provision of Nutrition Support Therapy in the Adult Critically Ill Patient: The American Society for Parenteral and Enteral Nutrition. J. Parenter. Enter. Nutr. 2022, 46, 12–41. [Google Scholar] [CrossRef]
- McClave, S.A.; Taylor, B.E.; Martindale, R.G.; Warren, M.M.; Johnson, D.R.; Braunschweig, C.; McCarthy, M.S.; Davanos, E.; Rice, T.W.; Cresci, G.A.; et al. Guidelines for the Provision and Assessment of Nutrition Support Therapy in the Adult Critically Ill Patient. J. Parenter. Enter. Nutr. 2016, 40, 159–211. [Google Scholar] [CrossRef]
- Fried, L.P.; Cohen, A.A.; Xue, Q.L.; Walston, J.; Bandeen-Roche, K.; Varadhan, R. The Physical Frailty Syndrome as a Transition from Homeostatic Symphony to Cacophony. Nature Aging 2021, 1, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Cunha, A.I.L.; Veronese, N.; de Melo Borges, S.; Ricci, N.A. Frailty as a Predictor of Adverse Outcomes in Hospitalized Older Adults: A Systematic Review and Meta-Analysis. Ageing Res. Rev. 2019, 56, 100960. [Google Scholar] [CrossRef] [PubMed]
- Volkert, D.; Beck, A.M.; Cederholm, T.; Cruz-Jentoft, A.; Hooper, L.; Kiesswetter, E.; Maggio, M.; Raynaud-Simon, A.; Sieber, C.; Sobotka, L.; et al. ESPEN Practical Guideline: Clinical Nutrition and Hydration in Geriatrics. Clin. Nutr. 2022, 41, 958–989. [Google Scholar] [CrossRef] [PubMed]
- Katz, S.; Ford, A.B.; Moskowitz, R.W.; Jackson, B.A.; Jaffe, M.W. Studies of Illness in the Aged. The Index of Adl: A Standardized Measure of Biological and Psychosocial Function. JAMA 1963, 185, 914–919. [Google Scholar] [CrossRef]
- Katz, S. Assessing Self-Maintenance: Activities of Daily Living, Mobility, and Instrumental Activities of Daily Living. J. Am. Geriatr. Soc. 1983, 31, 721–727. [Google Scholar] [CrossRef]
- Guigoz, Y.; Vellas, B. The Mini Nutritional Assessment (MNA) for Grading the Nutritional State of Elderly Patients: Presentation of the MNA, History and Validation. Nestle Nutr. Workshop Ser. Clin. Perform. Programme 1999, 1. [Google Scholar] [CrossRef]
- Blaylock, A.; Cason, C.L. Discharge Planning Predicting Patients’ Needs. J. Gerontol. Nurs. 1992, 18, 5–10. [Google Scholar] [CrossRef]
- Veronese, N.; Fazzari, A.; Armata, M.; Parisi, A.; Parrinello, A.; Petralia, V.; Saccaro, C.; Vesco, M.; Tagliaferri, F.; Fittipaldo, V.A.; et al. Clinical Prognostic Factors for Older People: A Systematic Review and Meta-Analysis. Ageing Res. Rev. 2024, 98, 102345. [Google Scholar] [CrossRef]
- Pottel, H.; Hoste, L.; Dubourg, L.; Ebert, N.; Schaeffner, E.; Eriksen, B.O.; Melsom, T.; Lamb, E.J.; Rule, A.D.; Turner, S.T.; et al. An Estimated Glomerular Filtration Rate Equation for the Full Age Spectrum. Nephrol. Dial. Transplant. 2016, 31, 798–806. [Google Scholar] [CrossRef]
- Ricci, C.; Serbassi, F.; Alberici, L.; Ingaldi, C.; Gaetani, L.; De Raffele, E.; Pironi, L.; Sasdelli, A.S.; Mosconi, C.; Di Marco, M.C.; et al. Immunonutrition in Patients Who Underwent Major Abdominal Surgery: A Comprehensive Systematic Review and Component Network Metanalysis Using GRADE and CINeMA Approaches. Surgery 2023, 174, 1401–1409. [Google Scholar] [CrossRef]
- Montejo, J.C.; Zarazaga, A.; López-Martínez, J.; Urrútia, G.; Roqué, M.; Blesa, A.L.; Celaya, S.; Conejero, R.; Galbán, C.; García de Lorenzo, A.; et al. Immunonutrition in the Intensive Care Unit. A Systematic Review and Consensus Statement. Clin. Nutr. 2003, 22, 221–233. [Google Scholar] [CrossRef] [PubMed]
- Arykan, N.; Shestopalov, A.; Evdokimov, E. Immunonutrition as a component of immunomodulating program in the elderly patients in non-Covid-19 hospital. Clin. Nutr. ESPEN 2020, 40, 600. [Google Scholar] [CrossRef]
- Cetean, S.; Căinap, C.; Constantin, A.M.; Căinap, S.; Gherman, A.; Oprean, L.; Hangan, A.; Oprean, R. The Importance of the Granulocyte-Colony Stimulating Factor in Oncology. Clujul Med. 2015, 88, 468. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.J.; Ashkar, A.A. The Dual Nature of Type I and Type II Interferons. Front. Immunol. 2018, 9, 2061. [Google Scholar] [CrossRef] [PubMed]
- Verma, N.D.; Hall, B.M.; Plain, K.M.; Robinson, C.M.; Boyd, R.; Tran, G.T.; Wang, C.; Bishop, G.A.; Hodgkinson, S.J. Interleukin-12 (IL-12p70) Promotes Induction of Highly Potent Th1-Like CD4+CD25+ T Regulatory Cells That Inhibit Allograft Rejection in Unmodified Recipients. Front. Immunol. 2014, 5, 190. [Google Scholar] [CrossRef]
- Cai, M.; Huang, X.; Huang, X.; Ju, D.; Zhu, Y.Z.; Ye, L. Research Progress of Interleukin-15 in Cancer Immunotherapy. Front. Pharmacol. 2023, 14, 1184703. [Google Scholar] [CrossRef]
- Ross, S.H.; Cantrell, D.A. Signaling and Function of Interleukin-2 in T Lymphocytes. Annu. Rev. Immunol. 2018, 36, 411. [Google Scholar] [CrossRef]
- Yadav, P.; Vats, R.; Bano, A.; Bhardwaj, R. Hematopoietic Stem Cells Culture, Expansion and Differentiation: An Insight into Variable and Available Media. Int. J. Stem Cells 2020, 13, 326–334. [Google Scholar] [CrossRef]
- Schellnegger, M.; Hofmann, E.; Carnieletto, M.; Kamolz, L.P. Unlocking Longevity: The Role of Telomeres and Its Targeting Interventions. Front. Aging 2024, 5, 1339317. [Google Scholar] [CrossRef]

| Nutrient | Impact® Enteral | Novasource® GIControl |
|---|---|---|
| Energy | 101 Kcal | 110 Kcal |
| Protein | 5.6 g | 4.1 g |
| Carbohydrates | 13.4 g | 14.0 g |
| Fat | 2.8 g | 3.5 g |
| Saturated Fatty Acids | 1.6 g | 1.2 g |
| Monounsaturated Fatty Acids | 0.5 g | 1.1 g |
| Polyunsaturated Fatty Acids | 0.5 g | 0.9 g |
| Osmolarity | 298 mOsm/L | 286 mOsm/L |
| Omega-3 Fatty Acids (EPA/DHA) | 0.3 g | - |
| RNA | 0.1 g | - |
| Arginine | 1.3 g | - |
| Vitamins (selected) | ||
| - Vitamin A | 100 µg | 125 µg |
| - Vitamin D | 0.67 µg | 1.7 µg |
| - Vitamin E | 3 mg | 1.7 mg |
| - Vitamin C | 6.7 mg | 11 mg |
| Total Sample n = 36 | CTL n = 17 | IN n = 19 | p | |
|---|---|---|---|---|
| Sex (F/M) | 28/8 | 14/3 | 14/5 | 0.414 |
| Age, y | 88.67 ± 4.90 | 87.47 ± 5.26 | 89.74 ± 4.42 | 0.170 |
| ADL | 1.14 ± 1.80 | 1.06 ± 1.91 | 1.21 ± 1.75 | 0.813 |
| IADL | 0.63 ± 1.91 | 0.75 ± 2.01 | 0.53 ± 1.86 | 0.736 |
| MNA | 17.19 ± 4.83 | 16.25 ± 4.65 | 17.90 ± 4.98 | 0.379 |
| BRASS | 26.07 ± 3.84 | 26.43 ± 4.09 | 25.73 ± 3.71 | 0.635 |
| MPI | 0.72 ± 0.08 | 0.74 ± 0.11 | 0.71 ± 0.06 | 0.536 |
| N drugs | 6.06 ± 3.71 | 6.69 ± 3.89 | 5.53 ± 3.56 | 0.364 |
| LOS | 13.94 ± 8.57 | 16.82 ± 10.83 | 11.37 ± 4.87 | 0.050 |
| CTL n = 17 | IN n = 19 | p | |
|---|---|---|---|
| Hemoglobin (mg/dL) | 11.19 ± 2.43 | 11.56 ± 2.02 | 0.622 |
| WBC (×10³/µL) | 10.75 ± 4.92 | 12.03 ± 5.70 | 0.486 |
| Glucose (mg/dL) | 121.38 ± 36.41 | 113.94 ± 38.08 | 0.577 |
| Total Protein (mg/dL) | 5.95 ± 0.58 | 5.97 ± 0.86 | 0.937 |
| Albumin (mg/dL) | 3.00 ± 0.30 | 3.09 ± 0.44 | 0.511 |
| Total-Cholesterol (mg/dL) | 155.63 ± 3.93 | 172.08 ± 67.4 | 0.527 |
| Transferrin (mg/dL) | 207.46 ± 55.25 | 201.36 ± 60.10 | 0.786 |
| Vitamin B12 (pg/mL) | 325.63 ± 146.76 | 354.17 ± 288.86 | 0.572 |
| Folic acid (ng/mL) | 10.14 ± 6.49 | 8.56 ± 5.91 | 0.500 |
| CRP (mg/L) | 7.82 ± 7.25 | 8.71 ± 8.57 | 0.751 |
| Clearance creatinine (BIS1) | 54.79 ± 27.20 | 40.03 ± 18.36 | 0.065 |
| CTL n = 13 | IN n = 10 | p | |
|---|---|---|---|
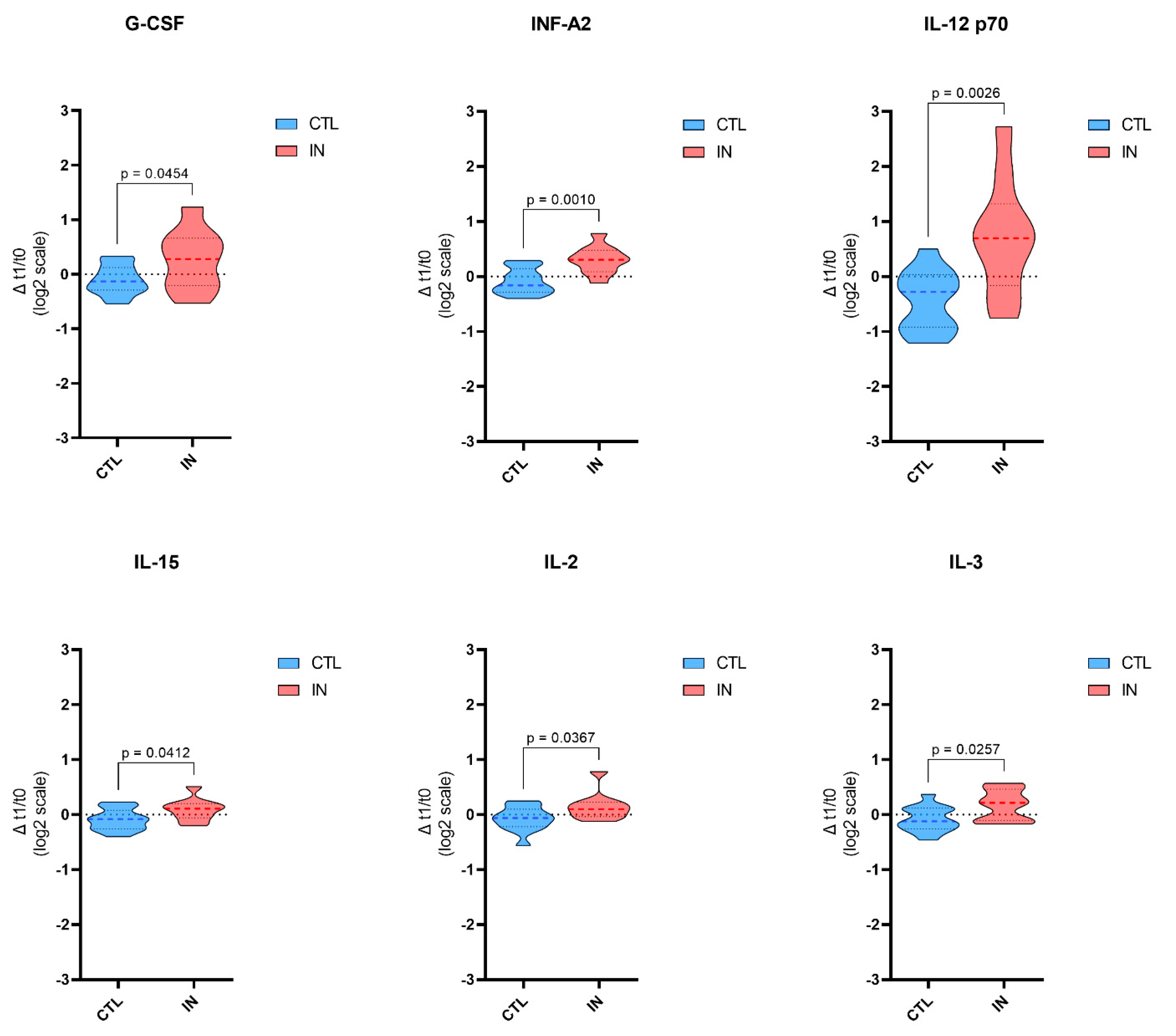
| EGF | 0.153 ± 0.464 | −0.020 ± 0.511 | 0.4050 |
| Eotaxin | −0.289 ± 0.495 | 0.004 ± 0.800 | 0.2924 |
| G-CSF | −0.105 ± 0.271 | 0.264 ± 0.547 | 0.0454 |
| GM-CSF | −0.042 ± 0.266 | 0.206 ± 0.703 | 0.3840 |
| INF-α2 | −0.085 ± 0.232 | 0.303 ± 0.253 | 0.0010 |
| INF-γ | 0.107 ± 0.214 | 0.232 ± 0.631 | 0.5100 |
| IL-10 | 0.025 ± 0.415 | 0.194 ± 0.479 | 0.3739 |
| IL-12 p 40 | 0.113 ± 0.283 | 0.035 ± 0.255 | 0.5015 |
| IL-12 p 70 | −0.435 ± 0.542 | 0.710 ± 1.044 | 0.0026 |
| IL-13 | −0.005 ± 0.379 | 0.033 ± 0.208 | 0.4186 |
| IL-15 | −0.092 ± 0.195 | 0.089 ± 0.202 | 0.0412 |
| IL-17 | −0.045 ± 0.462 | 0.358 ± 0.589 | 0.0799 |
| IL-1 RA | 0.020 ± 0.611 | 0.151 ± 1.688 | 0.7715 |
| IL-1 α | 0.000 ± 0.287 | 0.127 ± 0.265 | 0.2921 |
| IL-1 β | −0.092 ± 0.419 | 0.187 ± 0.723 | 0.6152 |
| IL-2 | −0.074 ± 0.223 | 0.148 ± 0.253 | 0.0367 |
| IL-3 | −0.085 ± 0.231 | 0.172 ± 0.284 | 0.0257 |
| IL−4 | −0.001 ± 0.337 | 0.039 ± 0.227 | 0.7517 |
| IL-5 | −0.084 ± 0.766 | 0.304 ± 0.660 | 0.2157 |
| IL-6 | −0.3585 ± 0.845 | −0.176 ± 1.15 | 0.6662 |
| IL-7 | −0.285 ± 1.280 | 0.222 ± 0.442 | 0.4833 |
| IL-8 | −0.223 ± 0.534 | 0.010 ± 0.827 | 0.4209 |
| IP-10 | −0.208 ± 1.170 | 0.460 ± 1.192 | 0.1925 |
| MCP-1 | −0.288 ± 0.652 | 0.109 ± 0.935 | 0.2434 |
| MIP-1 α | −0.003 ± 0.238 | 0.107 ± 0.245 | 0.2247 |
| MIP-1 β | −0.102 ± 0.470 | 0.091 ± 0.669 | 0.4263 |
| TNF-α | 0.006 ± 0.452 | 0.177 ± 0.739 | 0.5000 |
| TNF-β | −0.055 ± 0.193 | 0.085 ± 0.225 | 0.3350 |
| VEGF | −0.013 ± 0.144 | 0.125 ± 0.263 | 0.2017 |
| CTL n = 13 | IN n = 10 | p | |
|---|---|---|---|
| B cells (CD19+) | −0.118 ± 0.507 | 0.112 ± 1.024 | 0.4871 |
| T cells (CD3+) | 0.190 ± 0.386 | −0.553 ± 1.215 | 0.0494 |
| B-T cell ratio | −0.305 ± 0.653 | 0.668 ± 1.397 | 0.0371 |
| T helper cells (CD3+CD4+) | 0.004 ± 0.336 | −0.072 ± 0.493 | 0.6649 |
| T cytotoxic cells (CD3+CD8+) | −0.084 ± 0.248 | 0.216 ± 0.451 | 0.0543 |
| CD4-CD8 T cell ratio | 0.088 ± 0.440 | −0.288 ± 0.932 | 0.2126 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boccardi, V.; Cari, L.; Naghavi Alhosseini, M.; Bastiani, P.; Scamosci, M.; Caironi, G.; Aprea, G.; Mancinetti, F.; Cecchetti, R.; Ruggiero, C.; et al. Immunonutrition in Acute Geriatric Care: Clinical Outcomes, Inflammatory Profiles, and Immune Responses. Nutrients 2024, 16, 4211. https://doi.org/10.3390/nu16234211
Boccardi V, Cari L, Naghavi Alhosseini M, Bastiani P, Scamosci M, Caironi G, Aprea G, Mancinetti F, Cecchetti R, Ruggiero C, et al. Immunonutrition in Acute Geriatric Care: Clinical Outcomes, Inflammatory Profiles, and Immune Responses. Nutrients. 2024; 16(23):4211. https://doi.org/10.3390/nu16234211
Chicago/Turabian StyleBoccardi, Virginia, Luigi Cari, Mahdieh Naghavi Alhosseini, Patrizia Bastiani, Michela Scamosci, Giulia Caironi, Giulia Aprea, Francesca Mancinetti, Roberta Cecchetti, Carmelinda Ruggiero, and et al. 2024. "Immunonutrition in Acute Geriatric Care: Clinical Outcomes, Inflammatory Profiles, and Immune Responses" Nutrients 16, no. 23: 4211. https://doi.org/10.3390/nu16234211
APA StyleBoccardi, V., Cari, L., Naghavi Alhosseini, M., Bastiani, P., Scamosci, M., Caironi, G., Aprea, G., Mancinetti, F., Cecchetti, R., Ruggiero, C., Nocentini, G., & Mecocci, P. (2024). Immunonutrition in Acute Geriatric Care: Clinical Outcomes, Inflammatory Profiles, and Immune Responses. Nutrients, 16(23), 4211. https://doi.org/10.3390/nu16234211








