Nutrient Patterns and Body Mass Index: A Comparative Longitudinal Analysis in Urban Black South African Adolescents and Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Design
2.2. Dietary Intake Assessment
2.3. Demographic Questionnaires
2.4. Anthropometry
2.5. Data Analysis
3. Results
3.1. Descriptive Characteristics of the Study Population
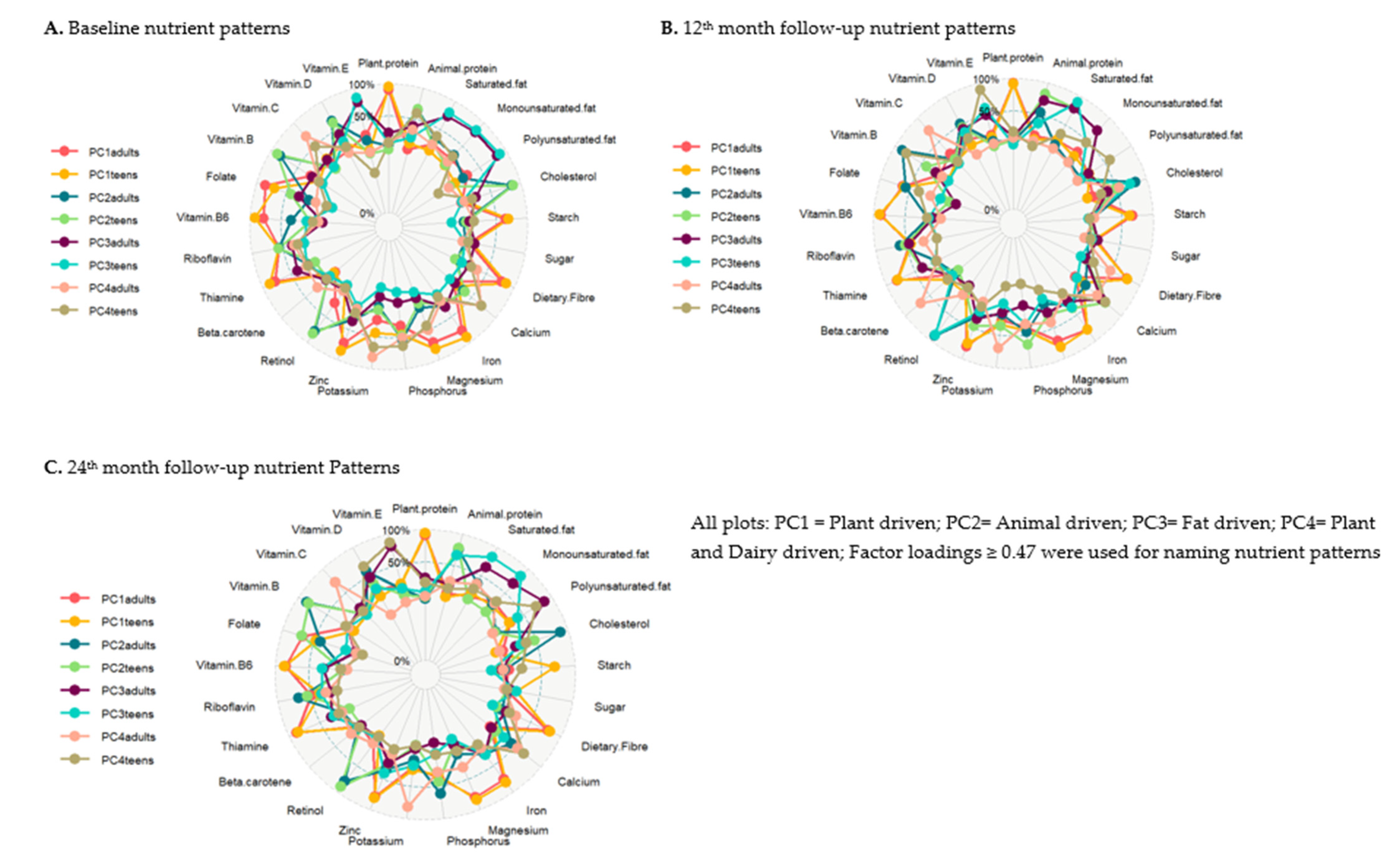
3.2. Nutrient Patterns
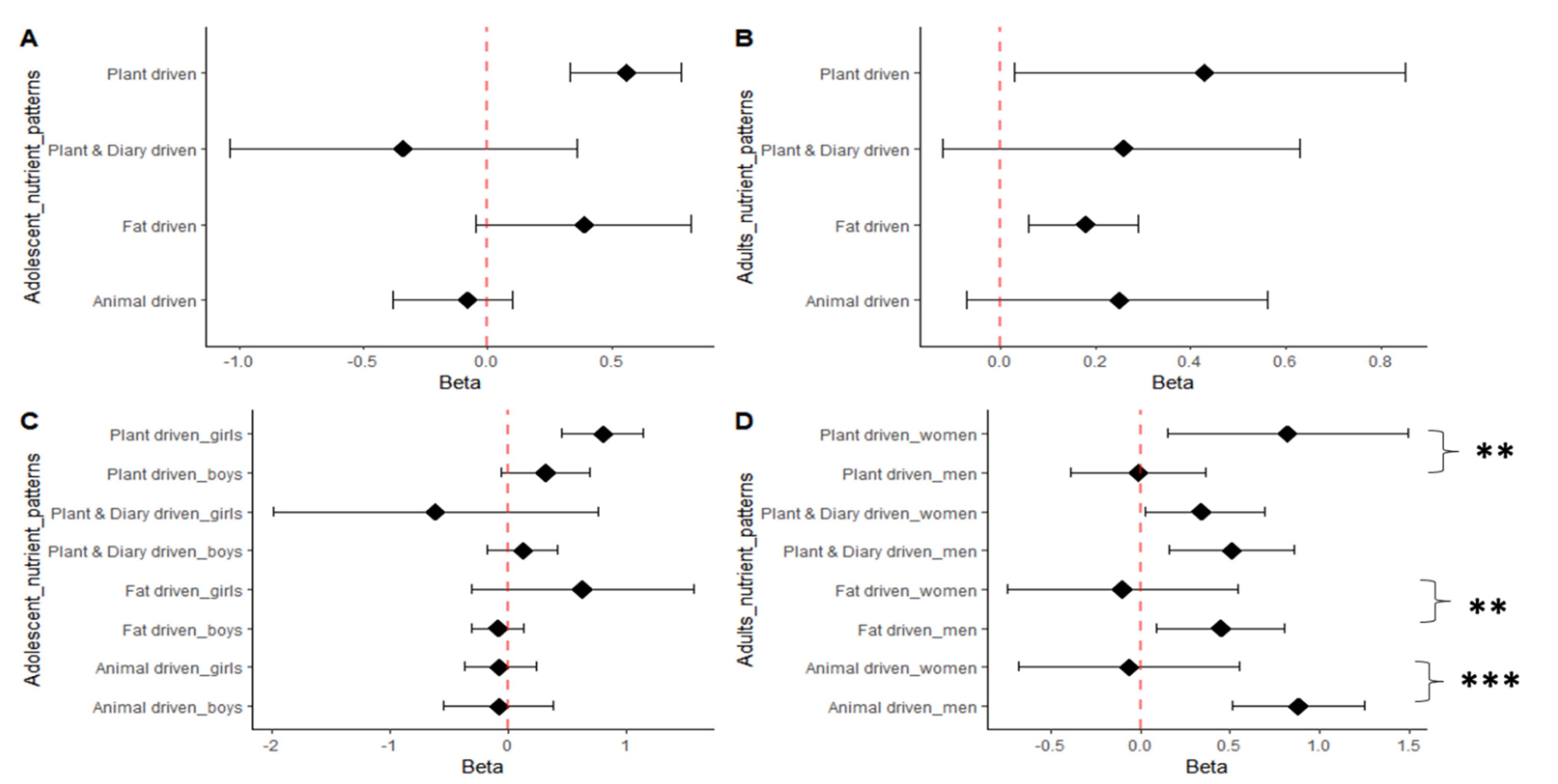
3.3. Nutrient Pattern Associations with BMI
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- GBD Obesity Collaborators. Health effects of overweight and obesity in 195 countries over 25 years. N. Engl. J. Med. 2017, 377, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Cois, A.; Day, C. Obesity trends and risk factors in the South African adult population. BMC Obes. 2015, 2, 42. [Google Scholar] [CrossRef] [PubMed]
- Maluleke, T.; Shisana, O.; Shisana, O. South African National Health and Nutrition Examination Survey (SANHANES-1); HSRC Press: Cape Town, South Africa, 2013. [Google Scholar]
- Ajayi, I.O.; Adebamowo, C.; Adami, H.O.; Dalal, S.; Diamond, M.B.; Bajunirwe, F.; Guwatudde, D.; Njelekela, M.; Nankya-Mutyoba, J.; Chiwanga, F.S.; et al. Urban-rural and geographic differences in overweight and obesity in four sub-Saharan African adult populations: A multi-country cross-sectional study. BMC Public Health 2016, 16, 1126. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.S.; Vos, T.; Flaxman, A.D.; Danaei, G.; Shibuya, K.; Adair-Rohani, H.; AlMazroa, M.A.; Amann, M.; Anderson, H.R.; Andrews, K.G.; et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2224–2260. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation (WHO). Obesity and Overweight Facts Sheet. 2021. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight#:~:text=Overall%2C%20about%2013%25%20of%20the,women)%20were%20obese%20in%202016 (accessed on 29 September 2022).
- Popkin, B.M.; Du, S.; Green, W.D.; Beck, M.A.; Algaith, T.; Herbst, C.H.; Alsukait, R.F.; Alluhidan, M.; Alazemi, N.; Shekar, M. Individuals with obesity and COVID-19: A global perspective on the epidemiology and biological relationships. Obes. Rev. 2020, 21, e13128. [Google Scholar] [CrossRef]
- National Department of Health (NDoH); Statistics South Africa; South African Medical Research Council; ICF. South Africa Demographic and Health Survey 2016; Statistics South Africa: Cape Town, South Africa, 2019. [Google Scholar]
- Ng, M.; Fleming, T.; Robinson, M.; Thomson, B.; Graetz, N.; Margono, C.; Mullany, E.C.; Biryukov, S.; Abbafati, C.; Abera, S.F.; et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2014, 384, 766–781. [Google Scholar] [CrossRef]
- Nyati, L.H.; Pettifor, J.M.; Norris, S.A. The prevalence of malnutrition and growth percentiles for urban South African children. BMC Public Health 2019, 19, 492. [Google Scholar] [CrossRef]
- Micklesfield, L.K.; Kagura, J.; Munthali, R.; Crowther, N.J.; Jaff, N.; Gradidge, P.; Ramsay, M.; Norris, S.A. Demographic, socio-economic and behavioural correlates of BMI in middle-aged black men and women from urban Johannesburg, South Africa. Glob. Health Action 2018, 11, 1448250. [Google Scholar] [CrossRef]
- Phaswana-Mafuya, N.; Peltzer, K.; Chirinda, W.; Musekiwa, A.; Kose, Z. Sociodemographic predictors of multiple non-communicable disease risk factors among older adults in South Africa. Glob. Health Action 2013, 6, 20680. [Google Scholar] [CrossRef]
- Micklesfield, L.K.; Pedro, T.M.; Kahn, K.; Kinsman, J.; Pettifor, J.M.; Tollman, S.; A Norris, S. Physical activity and sedentary behavior among adolescents in rural South Africa: Levels, patterns and correlates. BMC Public Health 2014, 14, 40. [Google Scholar] [CrossRef]
- Mukoma, G.; Wrottesley, S.V.; Kagura, J.; Oni, T.; Micklesfield, L.; Norris, S.A. The relationships between socioeconomic status, dietary knowledge and patterns, and physical activity with adiposity in urban South African women. S. Afr. J. Clin. Nutr. 2022, 1–7. [Google Scholar] [CrossRef]
- Malambo, P.; Kengne, A.P.; Lambert, E.V.; De Villiers, A.; Puoane, T. Prevalence and socio-demographic correlates of physical activity levels among South African adults in Cape Town and Mount Frere communities in 2008–2009. Arch. Public Health 2016, 74, 54. [Google Scholar] [CrossRef] [PubMed]
- Micklesfield, L.K.; Munthali, R.J.; Prioreschi, A.; Said-Mohamed, R.; Van Heerden, A.; Tollman, S.; Kahn, K.; Dunger, D.; Norris, S.A. Understanding the Relationship between Socio-Economic Status, Physical Activity and Sedentary Behaviour, and Adiposity in Young Adult South African Women Using Structural Equation Modelling. Int. J. Environ. Res. Public Health 2017, 14, 1271. [Google Scholar] [CrossRef] [PubMed]
- Gradidge, P.J.-L.; Norris, S.A.; Munthali, R.; Crowther, N.J. Influence of socioeconomic status on changes in body size and physical activity in ageing black South African women. Eur. Rev. Aging Phys. Act. 2018, 15, 6. [Google Scholar] [CrossRef]
- Chandrasekaran, S.; Neal-Perry, G. Long-term consequences of obesity on female fertility and the health of the offspring. Curr. Opin. Obstet. Gynecol. 2017, 29, 180–187. [Google Scholar] [CrossRef]
- Nicholas, L.M.; Morrison, J.L.; Rattanatray, L.; Zhang, S.; Ozanne, S.E.; McMillen, I.C. The early origins of obesity and insulin resistance: Timing, programming and mechanisms. Int. J. Obes. 2016, 40, 229–238. [Google Scholar] [CrossRef]
- Fleming, T.P.; Watkins, A.J.; Velazquez, M.A.; Mathers, J.C.; Prentice, A.M.; Stephenson, J.; Barker, M.; Saffery, R.; Yajnik, C.S.; Eckert, J.J.; et al. Origins of lifetime health around the time of conception: Causes and consequences. Lancet 2018, 391, 1842–1852. [Google Scholar] [CrossRef]
- Sinclair, K.D.; Watkins, A.J. Parental diet, pregnancy outcomes and offspring health: Metabolic determinants in developing oocytes and embryos. Reprod. Fertil. Dev. 2013, 26, 99–114. [Google Scholar] [CrossRef]
- Gaskins, A.J.; Colaci, D.S.; Mendiola, J.; Swan, S.H.; Chavarro, J.E. Dietary patterns and semen quality in young men. Hum. Reprod. 2012, 27, 2899–2907. [Google Scholar] [CrossRef]
- Agbaje, I.; Rogers, D.; McVicar, C.; McClure, N.; Atkinson, A.; Mallidis, C.; Lewis, S. Insulin dependant diabetes mellitus: Implications for male reproductive function. Hum. Reprod. 2007, 22, 1871–1877. [Google Scholar] [CrossRef]
- Mullins, E.; Davies, S. Annual Report of the Chief Medical Officer, 2014 The Health of the 51%: Women. 2015. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/595439/CMO_annual_report_2014.pdf (accessed on 16 February 2023).
- Berdanier, C.D.; Dwyer, J.T.; Feldman, E.B. Handbook of Nutrition and Food; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Kelly, O.J.; Gilman, J.C.; Kim, Y.; Ilich, J.Z. Micronutrient intake in the etiology, prevention and treatment of osteosarcopenic obesity. Curr. Aging Sci. 2016, 9, 260–278. [Google Scholar] [CrossRef] [PubMed]
- Hammad, S.S.; Eck, P.; Sihag, J.; Chen, X.; Connelly, P.W.; Lamarche, B.; Couture, P.; Guay, V.; Maltais-Giguère, J.; West, S.G.; et al. Common Variants in Lipid Metabolism–Related Genes Associate with Fat Mass Changes in Response to Dietary Monounsaturated Fatty Acids in Adults with Abdominal Obesity. J. Nutr. 2019, 149, 1749–1756. [Google Scholar] [CrossRef] [PubMed]
- Pisa, P.T.; Pedro, T.M.; Kahn, K.; Tollman, S.M.; Pettifor, J.M.; Norris, S.A. Nutrient patterns and their association with socio-demographic, lifestyle factors and obesity risk in rural South African adolescents. Nutrients 2015, 7, 3464–3482. [Google Scholar] [CrossRef]
- Chikowore, T.; Pisa, P.T.; Van Zyl, T.; Feskens, E.J.M.; Wentzel-Viljoen, E.; Conradie, K.R. Nutrient Patterns Associated with Fasting Glucose and Glycated Haemoglobin Levels in a Black South African Population. Nutrients 2017, 9, 9. [Google Scholar] [CrossRef]
- Ratshikombo, T.; Goedecke, J.H.; Soboyisi, M.; Kufe, C.; Makura-Kankwende, C.B.T.; Masemola, M.; Micklesfield, L.K.; Chikowore, T. Sex Differences in the Associations of Nutrient Patterns with Total and Regional Adiposity: A Study of Middle-Aged Black South African Men and Women. Nutrients 2021, 13, 4558. [Google Scholar] [CrossRef] [PubMed]
- Makura-Kankwende, C.B.; Gradidge, P.J.; Crowther, N.J.; Ratshikombo, T.; Goedecke, J.H.; Micklesfield, L.K.; Norris, S.A.; Chikowore, T. Association of Longitudinal Nutrient Patterns with Body Composition in Black Middle-Aged South African Women: A Five-Year Follow-Up Study. Int. J. Environ. Res. Public Health 2022, 19, 12792. [Google Scholar] [CrossRef] [PubMed]
- Makura-Kankwende, C.B.T.; Gradidge, P.J.; Crowther, N.J.; Norris, S.A.; Chikowore, T. Nutrient Patterns and Body Composition Parameters of Black South African Women. Nutrients 2021, 13, 6. [Google Scholar] [CrossRef] [PubMed]
- GBD 2015 Risk Factors Collaborators. Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1345–1422. [Google Scholar] [CrossRef]
- Newby, P.K.; Tucker, K.L. Empirically derived eating patterns using factor or cluster analysis: A review. Nutr. Rev. 2004, 62, 177–203. [Google Scholar] [CrossRef]
- Hu, F.B. Dietary pattern analysis: A new direction in nutritional epidemiology. Curr. Opin. Lipidol. 2002, 13, 3–9. [Google Scholar] [CrossRef]
- Zingoni, C.; Norris, S.A.; Griffiths, P.L.; Cameron, N. Studying a population undergoing nutrition transition: A practical case study of dietary assessment in urban South African adolescents. Ecol. Food Nutr. 2009, 48, 178–198. [Google Scholar] [CrossRef] [PubMed]
- Steyn, N.P.; Senekal, M.; Norris, S.A.; Whati, L.; MacKeown, J.M.; Nel, J.H. How well do adolescents determine portion sizes of foods and beverages? Asia Pac. J. Clin. Nutr. 2006, 15, 35–42. [Google Scholar]
- Wrottesley, S.V.; Pisa, P.T.; Norris, S.A. The Influence of Maternal Dietary Patterns on Body Mass Index and Gestational Weight Gain in Urban Black South African Women. Nutrients 2017, 9, 732. [Google Scholar] [CrossRef] [PubMed]
- Feeley, A.B.; Musenge, E.; Pettifor, J.M.; Norris, S.A. Investigation into longitudinal dietary behaviours and household socio-economic indicators and their association with BMI Z-score and fat mass in South African adolescents: The Birth to Twenty (Bt20) cohort. Public Health Nutr. 2013, 16, 693–703. [Google Scholar] [CrossRef]
- Jean-Luc Gradidge, P.; Norris, S.A.; Jaff, N.G.; Crowther, N.J. Metabolic and Body Composition Risk Factors Associated with Metabolic Syndrome in a Cohort of Women with a High Prevalence of Cardiometabolic Disease. PLoS ONE 2016, 11, e0162247. [Google Scholar] [CrossRef]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef]
- Vorster, H.H.; Kruger, A.; Wentzel-Viljoen, E.; Kruger, H.S.; Margetts, B.M. Added sugar intake in South Africa: Findings from the Adult Prospective Urban and Rural Epidemiology cohort study. Am. J. Clin. Nutr. 2014, 99, 1479–1486. [Google Scholar] [CrossRef]
- Griffiths, P.L.; Johnson, W.; Cameron, N.; Pettifor, J.M.; Norris, S.A. In urban South Africa, 16 year old adolescents experience greater health equality than children. Econ. Hum. Biol. 2013, 11, 502–514. [Google Scholar] [CrossRef]
- Kagura, J.; Adair, L.S.; Pisa, P.T.; Griffiths, P.L.; Pettifor, J.M.; Norris, S.A. Association of socioeconomic status change between infancy and adolescence, and blood pressure, in South African young adults: Birth to Twenty Cohort. BMJ Open 2016, 6, e008805. [Google Scholar] [CrossRef]
- Trumbo, P.; Schlicker, S.; Yates, A.A.; Poos, M. Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein and amino acids. J. Am. Diet. Assoc. 2002, 102, 1621–1630. [Google Scholar] [CrossRef]
- Steyn, N.P.; Mchiza, Z.J. Obesity and the nutrition transition in Sub-Saharan Africa. Ann. N. Y. Acad. Sci. 2014, 1311, 88–101. [Google Scholar] [CrossRef] [PubMed]
- Wrottesley, S.V.; Pedro, T.M.; Fall, C.H.; Norris, S.A. A review of adolescent nutrition in South Africa: Transforming adolescent lives through nutrition initiative. S. Afr. J. Clin. Nutr. 2020, 33, 94–132. [Google Scholar] [CrossRef]
- Prioreschi, A.; Wrottesley, S.V.; Cohen, E.; Reddy, A.; Said-Mohamed, R.; Twine, R.; Tollman, S.M.; Kahn, K.; Dunger, D.B.; Norris, S.A. Examining the relationships between body image, eating attitudes, BMI, and physical activity in rural and urban South African young adult females using structural equation modeling. PLoS ONE 2017, 12, e0187508. [Google Scholar] [CrossRef] [PubMed]
- Wentzel-Viljoen, E.; Lee, S.; Laubscher, R.; Vorster, H.H. Accelerated nutrition transition in the North West Province of South Africa: Results from the Prospective Urban and Rural Epidemiology (PURE-NWP-SA) cohort study, 2005 to 2010. Public Health Nutr. 2018, 21, 2630–2641. [Google Scholar] [CrossRef] [PubMed]
- Tydeman-Edwards, R.; Van Rooyen, F.C.; Walsh, C.M.J.H. Obesity, undernutrition and the double burden of malnutrition in the urban and rural southern Free State, South Africa. Heliyon 2018, 4, e00983. [Google Scholar] [CrossRef]
- Pringle, J.; Doi, L.; Jindal-Snape, D.; Jepson, R.; McAteer, J. Adolescents and health-related behaviour: Using a framework to develop interventions to support positive behaviours. Pilot Feasibility Stud. 2018, 4, 69. [Google Scholar] [CrossRef]

| Adolescents | Adults | |||||||
|---|---|---|---|---|---|---|---|---|
| p-Value a (M vs. F) | p-Value a (M vs. F) | |||||||
| Median (IQR) | Median (IQR) | |||||||
| Total (n = 250) | Male (n = 125) | Female (n = 125) | Total (n = 500) | Male (n = 250) | Female (n = 250) | |||
| Age (year) | 14 (13; 15) | 14 (14; 15) | 14 (13; 15) | 0.222 | 27 (27; 52) | 35 (27; 55) | 27 (27; 50) | 0.053 |
| Weight (kg) | 52.6 (46.6; 59.8) | 51.9 (44.6; 58.4) | 53 (48; 60.6) | 0.012 | 72.5 (60.5; 84.8) | 70.5 (59.4; 81.2) | 74.7 (62.8; 74.7) | 0.002 |
| Height (m) | 1.60 (1.54; 1.66) | 1.63 (1.55; 1.69) | 1.57 (1.53–1.62) | <0.001 | 1.64 (1.58; 1.71) | 1.68 (1.62; 1.73) | 1.61 (1.56; 1.66) | <0.001 |
| BMI (kg/m2) | 20.1 (18.2; 22.8) | 19.3 (17.7; 21.1) | 21.3 (19.2; 24.6) | <0.001 | 26.6 (22.2; 32.7) | 25.1 (21.2; 28.8) | 29.1 (23.6; 35.8) | <0.001 |
| Total energy (kJ/day) | 11,123 (8437; 14,284) | 11,305 (8709; 14,153) | 11,042 (8234; 14,705) | 0.878 | 9438 (6258; 14,686) | 9955 (6756; 15,799) | 8720 (5960; 12,284) | 0.001 |
| Total protein (g/day) | 65.6 (49.7; 86.3) | 68.9 (49.8; 88.3) | 64.3 (49.4; 83.9) | 0.227 | 63.2 (42.7; 90.9 | 67.4 (48.1; 96.6) | 55.4 (38.0; 82.2) | 0.025 |
| Plant protein (g/day) | 34.3 (25.6; 47.4) | 35.9 (27.6; 48.3) | 33.3 (24.7; 45.7) | 0.010 | 27.4 (18.5; 41.9) | 30.9 (19.5; 45.6) | 24.9 (17.9; 36.6) | 0.013 |
| Animal protein (g/day) | 29.5 (18.9; 41.8) | 29.8 (18.5; 39.9) | 29.5 (19.2; 43.3) | 0.842 | 31.1 (19.4; 48.0) | 34.6 (24.5; 48.5) | 27.6 (17.5; 45.7) | 0.318 |
| Total fat (g/day) | 97.2 (70.9; 142.9) | 95.7 (70.4; 134.6) | 100.8 (73.9; 144.6) | 0.024 | 85.6 (50.6; 135.1) | 86.7 (51.4; 139.0) | 84.2 (50.5; 130.3) | <0.001 |
| SFA (g/day) | 24.1 (16.9; 35.1) | 23.2 (16.6; 33.6) | 24.9 (17.2; 36.9) | 0.010 | 21.6 (13.3; 33.9) | 21.5 (13.8; 34.9) | 20.5 (12.7; 33.3) | <0.001 |
| MUFA (g/day) | 28.7 (19.1; 41.1) | 28.7 (19.1; 39.6) | 28.7 (19.1; 42.5) | 0.468 | 21.8 (15.7; 40.9) | 26.87 (16.9; 40.8) | 24.9 (15.2; 40.9) | 0.002 |
| PUFA (g/day) | 32.9 (20.1; 49.5) | 33.1 (20.3; 47.4) | 32.8 (20.1; 53.9) | 0.109 | 24.1 (14.9; 40.9) | 25.3 (15.0; 42.3) | 22.5 (14.9; 39.4) | 0.003 |
| Carbohydrate (g/day) | 352.3 (257.6; 436.6) | 366.0 (282.4; 436.7) | 341.3 (249.5; 433.5) | 0.031 | 284.4 (187.4; 416.1) | 315.3 (194.9; 465.9) | 264.3 (183.2; 366.4) | 0.460 |
| Added sugar (g/day) | 70.7 (44.0; 113.2) | 70.6 (45.9; 117.8) | 70.9 (42.6; 106.6) | 0.227 | 56.6 (32.1; 95.9) | 55.4 (28.9; 101.6) | 59.4 (35.7; 88.4) | 0.004 |
| EI/EER ratio (kJ/day) | 1.18 (0.88; 1.51) | 1.11 (0.82; 1.39) | 1.31 (0.96; 1.70) | 0.007 | 1.003 (0.64; 1.54) | 1.06 (0.64; 1.72) | 0.96 (1.34; 0.65) | 0.161 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mukoma, G.; Norris, S.A.; Chikowore, T. Nutrient Patterns and Body Mass Index: A Comparative Longitudinal Analysis in Urban Black South African Adolescents and Adults. Nutrients 2023, 15, 1075. https://doi.org/10.3390/nu15051075
Mukoma G, Norris SA, Chikowore T. Nutrient Patterns and Body Mass Index: A Comparative Longitudinal Analysis in Urban Black South African Adolescents and Adults. Nutrients. 2023; 15(5):1075. https://doi.org/10.3390/nu15051075
Chicago/Turabian StyleMukoma, Gudani, Shane A. Norris, and Tinashe Chikowore. 2023. "Nutrient Patterns and Body Mass Index: A Comparative Longitudinal Analysis in Urban Black South African Adolescents and Adults" Nutrients 15, no. 5: 1075. https://doi.org/10.3390/nu15051075
APA StyleMukoma, G., Norris, S. A., & Chikowore, T. (2023). Nutrient Patterns and Body Mass Index: A Comparative Longitudinal Analysis in Urban Black South African Adolescents and Adults. Nutrients, 15(5), 1075. https://doi.org/10.3390/nu15051075







