Pre-Pregnancy Adherence to Mediterranean Diet and Risk of Gestational Diabetes Mellitus: A Prospective Cohort Study in Greece
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Variables for Assessment
2.3. Assessment of Diet
2.4. Statistical Analysis
3. Results
4. Discussions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Paulo, M.S.; Abdo, N.M.; Bettencourt-Silva, R.; Al-Rifai, R.H. Gestational diabetes mellitus in Europe: A systematic review and meta-analysis of prevalence studies. Front. Endocrinol. 2021, 12, 691033. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Zhang, C. Prevalence of gestational diabetes and risk of progression to type 2 diabetes: A global perspective. Curr. Diab. Rep. 2016, 16, 7. [Google Scholar] [CrossRef] [PubMed]
- Malaza, N.; Masete, M.; Adam, S.; Dias, S.; Nyawo, T.; Pheiffer, C. A Systematic Review to Compare Adverse Pregnancy Outcomes in Women with Pregestational Diabetes and Gestational Diabetes. Int. J. Env. Res. Public Health 2022, 19, 10846. [Google Scholar] [CrossRef] [PubMed]
- Lauenborg, J.; Crusell, M.; Mathiesen, E.R.; Damm, P. Maternal Long-Term Outcomes after a Pregnancy Complicated by Gestational Diabetes Mellitus. In Gestational Diabetes. A Decade after the HAPO Study; Lapolla, A., Metzger, B.E., Eds.; Karger Publishers: Basel, Switzerland, 2020; pp. 223–233. [Google Scholar]
- Dassanayake, M.; Langen, E.; Davis, M.B. Pregnancy complications as a window to future cardiovascular disease. Cardiol. Rev. 2020, 28, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Zhu, Y.; Yeung, E.; Chavarro, J.E.; Yuan, C.; Field, A.E.; Missmer, S.A.; Mills, J.L.; Hu, F.B.; Zhang, C. Offspring risk of obesity in childhood, adolescence and adulthood in relation to gestational diabetes mellitus: A sex-specific association. Int. J. Epidemiol. 2017, 46, 1533–1541. [Google Scholar] [CrossRef] [PubMed]
- Damm, P.; Houshmand-Oeregaard, A.; Kelstrup, L.; Lauenborg, J.; Mathiesen, E.R.; Clausen, T.D. Gestational diabetes mellitus and long-term consequences for mother and offspring: A view from Denmark. Diabetologia 2016, 59, 1396–1399. [Google Scholar] [CrossRef] [PubMed]
- Tranidou, A.; Dagklis, T.; Tsakiridis, I.; Siargkas, A.; Apostolopoulou, A.; Mamopoulos, A.; Goulis, D.G.; Chourdakis, M. Risk of developing metabolic syndrome after gestational diabetes mellitus-a systematic review and meta-analysis. J. Endocrinol. Investig. 2021, 44, 1139–1149. [Google Scholar] [CrossRef] [PubMed]
- Tsakiridis, I.; Giouleka, S.; Mamopoulos, A.; Kourtis, A.; Athanasiadis, A.; Filopoulou, D.; Dagklis, T. Diagnosis and management of gestational diabetes mellitus: An overview of national and international guidelines. Obstet. Gynecol. Surv. 2021, 76, 367–381. [Google Scholar] [CrossRef]
- Ramakrishnan, U.; Grant, F.; Goldenberg, T.; Zongrone, A.; Martorell, R. Effect of women’s nutrition before and during early pregnancy on maternal and infant outcomes: A systematic review. Paediatr. Perinat. Epidemiol. 2012, 26, 285–301. [Google Scholar] [CrossRef]
- Olmedo-Requena, R.; Gómez-Fernández, J.; Amezcua-Prieto, C.; Mozas-Moreno, J.; Khan, K.S.; Jiménez-Moleón, J.J. Pre-pregnancy adherence to the Mediterranean diet and gestational diabetes mellitus: A case-control study. Nutrients 2019, 11, 1003. [Google Scholar] [CrossRef]
- Assaf-Balut, C.; García de la Torre, N.; Durán, A.; Fuentes, M.; Bordiú, E.; Del Valle, L.; Familiar, C.; Ortolá, A.; Jiménez, I.; Herraiz, M.A. A Mediterranean diet with additional extra virgin olive oil and pistachios reduces the incidence of gestational diabetes mellitus (GDM): A randomized controlled trial: The St. Carlos GDM prevention study. PLoS ONE 2017, 12, e0185873. [Google Scholar] [CrossRef]
- Izadi, V.; Tehrani, H.; Haghighatdoost, F.; Dehghan, A.; Surkan, P.J.; Azadbakht, L. Adherence to the DASH and Mediterranean diets is associated with decreased risk for gestational diabetes mellitus. Nutrition 2016, 32, 1092–1096. [Google Scholar] [CrossRef] [PubMed]
- Colberg, S.R.; Castorino, K.; Jovanovič, L. Prescribing physical activity to prevent and manage gestational diabetes. World J. Diabetes 2013, 4, 256. [Google Scholar] [CrossRef] [PubMed]
- Doi, S.A.; Furuya-Kanamori, L.; Toft, E.; Musa, O.A.; Mohamed, A.M.; Clark, J.; Thalib, L. Physical activity in pregnancy prevents gestational diabetes: A meta-analysis. Diabetes Res. Clin. Pract. 2020, 168, 108371. [Google Scholar] [CrossRef] [PubMed]
- Schoenaker, D.A.; De Jersey, S.; Willcox, J.; Francois, M.E.; Wilkinson, S. Prevention of gestational diabetes: The role of dietary intake, physical activity, and weight before, during, and between pregnancies. Semin. Reprod. Med. 2020, 38, 352–365. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Vizcaíno, V.; Sanabria-Martínez, G.; Fernández-Rodríguez, R.; Cavero-Redondo, I.; Pascual-Morena, C.; Álvarez-Bueno, C.; Martínez-Hortelano, J.A. Exercise during pregnancy for preventing gestational diabetes mellitus and hypertensive disorders: An umbrella review of randomised controlled trials and an updated meta-analysis. BJOG 2023, 130, 264–275. [Google Scholar] [CrossRef] [PubMed]
- Esposito, K.; Giugliano, D. Mediterranean diet and type 2 diabetes. Diabetes Metab. Res. Rev. 2014, 30, 34–40. [Google Scholar] [CrossRef]
- Donazar-Ezcurra, M.; Lopez-del Burgo, C.; Martinez-Gonzalez, M.A.; Basterra-Gortari, F.J.; de Irala, J.; Bes-Rastrollo, M. Pre-pregnancy adherences to empirically derived dietary patterns and gestational diabetes risk in a Mediterranean cohort: The Seguimiento Universidad de Navarra (SUN) project. Br. J. Nutr. 2017, 118, 715–721. [Google Scholar] [CrossRef]
- Tobias, D.K.; Zhang, C.; Chavarro, J.; Bowers, K.; Rich-Edwards, J.; Rosner, B.; Mozaffarian, D.; Hu, F.B. Prepregnancy adherence to dietary patterns and lower risk of gestational diabetes mellitus. Am. J. Clin. Nutr. 2012, 96, 289–295. [Google Scholar] [CrossRef]
- Schoenaker, D.A.; Soedamah-Muthu, S.S.; Callaway, L.K.; Mishra, G.D. Pre-pregnancy dietary patterns and risk of gestational diabetes mellitus: Results from an Australian population-based prospective cohort study. Diabetologia 2015, 58, 2726–2735. [Google Scholar] [CrossRef]
- Xiao, S.; Zhang, Q.; Zhang, M.; Hu, R.; Liu, R. A modified Mediterranean diet against gestational diabetes mellitus. STEMedicine 2022, 3, e129. [Google Scholar] [CrossRef]
- Gicevic, S.; Gaskins, A.J.; Fung, T.T.; Rosner, B.; Tobias, D.K.; Isanaka, S.; Willett, W.C. Evaluating pre-pregnancy dietary diversity vs. dietary quality scores as predictors of gestational diabetes and hypertensive disorders of pregnancy. PLoS ONE 2018, 13, e0195103. [Google Scholar] [CrossRef] [PubMed]
- Panagiotakos, D.B.; Pitsavos, C.; Stefanadis, C. Dietary patterns: A Mediterranean diet score and its relation to clinical and biological markers of cardiovascular disease risk. Nutr. Metab. Cardiovasc. Dis. 2006, 16, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Bach-Faig, A.; Berry, E.M.; Lairon, D.; Reguant, J.; Trichopoulou, A.; Dernini, S.; Medina, F.X.; Battino, M.; Belahsen, R.; Miranda, G. Mediterranean diet pyramid today. Science and cultural updates. Public Health Nutr. 2011, 14, 2274–2284. [Google Scholar] [CrossRef] [PubMed]
- Papadaki, A.; Johnson, L.; Toumpakari, Z.; England, C.; Rai, M.; Toms, S.; Penfold, C.; Zazpe, I.; Martínez-González, M.A.; Feder, G. Validation of the English version of the 14-item Mediterranean diet adherence screener of the PREDIMED study, in people at high cardiovascular risk in the UK. Nutrients 2018, 10, 138. [Google Scholar] [CrossRef] [PubMed]
- Hutchins-Wiese, H.L.; Bales, C.W.; Starr, K.N.P. Mediterranean diet scoring systems: Understanding the evolution and applications for Mediterranean and non-Mediterranean countries. Br. J. Nutr. 2021, 128, 1371–1392. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean Diet and Survival in a Greek Population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [CrossRef]
- Gomez-Arango, L.F.; Barrett, H.L.; Wilkinson, S.A.; Callaway, L.K.; McIntyre, H.D.; Morrison, M.; Dekker Nitert, M. Low dietary fiber intake increases Collinsella abundance in the gut microbiota of overweight and obese pregnant women. Gut Microbes 2018, 9, 189–201. [Google Scholar] [CrossRef]
- Merra, G.; Noce, A.; Marrone, G.; Cintoni, M.; Tarsitano, M.G.; Capacci, A.; De Lorenzo, A. Influence of mediterranean diet on human gut microbiota. Kompass Nutr. Diet. 2022, 2, 19–25. [Google Scholar] [CrossRef]
- Wang, X.; Liu, H.; Li, Y.; Huang, S.; Zhang, L.; Cao, C.; Baker, P.N.; Tong, C.; Zheng, P.; Qi, H. Altered gut bacterial and metabolic signatures and their interaction in gestational diabetes mellitus. Gut Microbes 2020, 12, 1840765. [Google Scholar] [CrossRef]
- Rold, L.S.; Bundgaard-Nielsen, C.; Niemann Holm-Jacobsen, J.; Glud Ovesen, P.; Leutscher, P.; Hagstrøm, S.; Sørensen, S. Characteristics of the gut microbiome in women with gestational diabetes mellitus: A systematic review. PLoS ONE 2022, 17, e0262618. [Google Scholar] [CrossRef] [PubMed]
- Pinto, Y.; Frishman, S.; Turjeman, S.; Eshel, A.; Nuriel-Ohayon, M.; Shrossel, O.; Ziv, O.; Walters, W.; Parsonnet, J.; Ley, C. Gestational diabetes is driven by microbiota-induced inflammation months before diagnosis. Gut 2023. [Google Scholar] [CrossRef] [PubMed]
- Ezra-Nevo, G.; Henriques, S.F.; Ribeiro, C. The diet-microbiome tango: How nutrients lead the gut brain axis. Curr. Opin. Neurobiol. 2020, 62, 122–132. [Google Scholar] [CrossRef] [PubMed]
- De Filippis, F.; Pellegrini, N.; Vannini, L.; Jeffery, I.B.; La Storia, A.; Laghi, L.; Serrazanetti, D.I.; Di Cagno, R.; Ferrocino, I.; Lazzi, C. High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut 2016, 65, 1812–1821. [Google Scholar] [CrossRef] [PubMed]
- Hellenic-Society-of-Obstetrics-and-Gynecology. Gestational Diabetes and Pregnancy: Gestational Diabetes. Guideline No 36, May 2020. EMGE. Available online: https://hsog.gr/wp-content/uploads/2020/06/EMGE-No-36.pdf (accessed on 2 February 2023).
- Coustan, D.R.; Lowe, L.P.; Metzger, B.E.; Dyer, A.R. The Hyperglycemia and Adverse Pregnancy Outcome (HAPO) study: Paving the way for new diagnostic criteria for gestational diabetes mellitus. Am. J. Obstet. Gynecol. 2010, 202, 654.e1–654.e6. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Classification and diagnosis of diabetes: Standards of medical care in diabetes—2019. Diabetes Care 2019, 42 (Suppl. S1), S13–S28. [Google Scholar] [CrossRef]
- Weir, C.B.; Jan, A. BMI Classification Percentile and Cut Off Points; StatPearls Publishing: Treasure Island, FL, USA, 2019. [Google Scholar]
- Apostolopoulou, A.; Magriplis, E.; Tsekitsidi, E.; Oikonomidou, A.C.; Papaefstathiou, E.; Tsakiridis, I.; Dagklis, T.; Chourdakis, M. Development and validation of a short culture specific Food Frequency Questionnaire for Greek pregnant women and adherence to the Mediterranean Diet. Nutrition 2021, 90, 111357. [Google Scholar] [CrossRef]
- Schröder, H.; Fitó, M.; Estruch, R.; Martínez-González, M.A.; Corella, D.; Salas-Salvadó, J.; Lamuela-Raventós, R.; Ros, E.; Salaverría, I.; Fiol, M. A short screener is valid for assessing Mediterranean diet adherence among older Spanish men and women. J. Nutr. 2011, 141, 1140–1145. [Google Scholar] [CrossRef]
- Radd-Vagenas, S.; Fiatarone Singh, M.A.; Daniel, K.; Noble, Y.; Jain, N.; O’Leary, F.; Mavros, Y.; Heffernan, M.; Meiklejohn, J.; Guerrero, Y. Validity of the Mediterranean diet and culinary index (MediCul) for online assessment of adherence to the ‘traditional’ diet and aspects of cuisine in older adults. Nutrients 2018, 10, 1913. [Google Scholar] [CrossRef]
- Athanasiadou, E.; Kyrkou, C.; Fotiou, M.; Tsakoumaki, F.; Dimitropoulou, A.; Polychroniadou, E.; Menexes, G.; Athanasiadis, A.P.; Biliaderis, C.G.; Michaelidou, A.-M. Development and validation of a Mediterranean oriented culture-specific semi-quantitative food frequency questionnaire. Nutrients 2016, 8, 522. [Google Scholar] [CrossRef]
- Leighton, F.; Polic, G.; Strobel, P.; Pérez, D.; Martínez, C.; Vásquez, L.; Castillo, O.; Villarroel, L.; Echeverría, G.; Urquiaga, I. Health impact of Mediterranean diets in food at work. Public Health Nutr. 2009, 12, 1635–1643. [Google Scholar] [CrossRef]
- Guasch-Ferré, M.; Willett, W. The Mediterranean diet and health: A comprehensive overview. J. Intern. Med. 2021, 290, 549–566. [Google Scholar] [CrossRef]
- Chicco, F.; Magrì, S.; Cingolani, A.; Paduano, D.; Pesenti, M.; Zara, F.; Tumbarello, F.; Urru, E.; Melis, A.; Casula, L. Multidimensional impact of Mediterranean diet on IBD patients. Inflamm. Bowel Dis. 2021, 27, 1–9. [Google Scholar] [CrossRef]
- Kastorini, C.-M.; Milionis, H.J.; Esposito, K.; Giugliano, D.; Goudevenos, J.A.; Panagiotakos, D.B. The effect of Mediterranean diet on metabolic syndrome and its components: A meta-analysis of 50 studies and 534,906 individuals. J. Am. Coll. Cardiol. 2011, 57, 1299–1313. [Google Scholar] [CrossRef] [PubMed]
- Di Daniele, N.; Noce, A.; Vidiri, M.F.; Moriconi, E.; Marrone, G.; Annicchiarico-Petruzzelli, M.; D’Urso, G.; Tesauro, M.; Rovella, V.; De Lorenzo, A. Impact of Mediterranean diet on metabolic syndrome, cancer and longevity. Oncotarget 2017, 8, 8947. [Google Scholar] [CrossRef]
- Walczak, P.; Walczak, K.; Zdun, S.; Nemeczek, S.; Merkisz, K.; Grzybowski, J.; Marciniak, A.; Grzywna, N.; Jaskuła, K.; Orłowski, W. Effect of mediterranean diet on non-alcoholic fatty liver disease (NAFLD). J. Educ. Health Sport 2023, 13, 58–64. [Google Scholar] [CrossRef]
- Bakaloudi, D.R.; Chrysoula, L.; Kotzakioulafi, E.; Theodoridis, X.; Chourdakis, M. Impact of the level of adherence to Mediterranean diet on the parameters of metabolic syndrome: A systematic review and meta-analysis of observational studies. Nutrients 2021, 13, 1514. [Google Scholar] [CrossRef] [PubMed]
- Makarem, N.; Chau, K.; Miller, E.C.; Gyamfi-Bannerman, C.; Tous, I.; Booker, W.; Catov, J.M.; Haas, D.M.; Grobman, W.A.; Levine, L.D. Association of a Mediterranean Diet Pattern With Adverse Pregnancy Outcomes among US Women. JAMA Netw. Open 2022, 5, e2248165. [Google Scholar] [CrossRef] [PubMed]
- Karamanos, B.; Thanopoulou, A.; Anastasiou, E.; Assaad-Khalil, S.; Albache, N.; Bachaoui, M.; Slama, C.B.; El Ghomari, H.; Jotic, A.; Lalic, N. Relation of the Mediterranean diet with the incidence of gestational diabetes. Eur. J. Clin. Nutr. 2014, 68, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Chatzi, L.; Leventakou, V.; Vafeiadi, M.; Koutra, K.; Roumeliotaki, T.; Chalkiadaki, G.; Karachaliou, M.; Daraki, V.; Kyriklaki, A.; Kampouri, M. Cohort profile: The mother-child cohort in Crete, Greece (Rhea Study). Int. J. Epidemiol. 2017, 46, 1392–1393k. [Google Scholar] [CrossRef]
- Marí-Sanchis, A.; Díaz-Jurado, G.; Basterra-Gortari, F.J.; de la Fuente-Arrillaga, C.; Martínez-González, M.A.; Bes-Rastrollo, M. Association between pre-pregnancy consumption of meat, iron intake, and the risk of gestational diabetes: The SUN project. Eur. J. Nutr. 2018, 57, 939–949. [Google Scholar] [CrossRef]
- Schoenaker, D.A.; Mishra, G.D.; Callaway, L.K.; Soedamah-Muthu, S.S. The role of energy, nutrients, foods, and dietary patterns in the development of gestational diabetes mellitus: A systematic review of observational studies. Diabetes Care 2016, 39, 16–23. [Google Scholar] [CrossRef]
- Liang, Y.; Gong, Y.; Zhang, X.; Yang, D.; Zhao, D.; Quan, L.; Zhou, R.; Bao, W.; Cheng, G. Dietary protein intake, meat consumption, and dairy consumption in the year preceding pregnancy and during pregnancy and their associations with the risk of gestational diabetes mellitus: A prospective cohort study in southwest China. Front. Endocrinol. 2018, 9, 596. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Franco, O.H.; Lamballais, S.; Ikram, M.A.; Schoufour, J.D.; Muka, T.; Voortman, T. Associations of specific dietary protein with longitudinal insulin resistance, prediabetes and type 2 diabetes: The Rotterdam Study. Clin. Nutr. 2020, 39, 242–249. [Google Scholar] [CrossRef]
- Alamolhoda, S.-H.; Zare, E.; Mirabi, P. Diet and Gestational Diabetes Mellitus: A Systematic Review Study. Curr. Women’s Health Rev. 2023, 19, 42–48. [Google Scholar] [CrossRef]
- Tsakiridis, I.; Kasapidou, E.; Dagklis, T.; Leonida, I.; Leonida, C.; Bakaloudi, D.R.; Chourdakis, M. Nutrition in pregnancy: A comparative review of major guidelines. Obstet. Gynecol. Surv. 2020, 75, 692–702. [Google Scholar] [CrossRef]
- Killeen, S.L.; Geraghty, A.A.; O’Brien, E.C.; O’Reilly, S.L.; Yelverton, C.A.; McAuliffe, F.M. Addressing the gaps in nutritional care before and during pregnancy. Proc. Nutr. Soc. 2022, 81, 87–98. [Google Scholar] [CrossRef] [PubMed]

| Maternal Characteristics | Total Population | GDM (n = 112, 15.1%) | Non GDM (n = 631, 84.9%) | p Value | |
|---|---|---|---|---|---|
| Maternal age in years, mean ± sd | 32.1 ± 4.85 | 33.7 ± 4.54 | 31.9 ± 4.85 | <0.001 | |
| Maternal age > 35 years, n (%) | 207 (27.9) | 45 (40.2) | 162 (25.7) | 0.003 | |
| Smoking before pregnancy, n (%) | 272 (36.6) | 40 (35.7) | 232 (36.8) | 0.92 | |
| Never smoked, n (%) | 397 (53.4) | 52 (46.4) | 345 (54.7) | 0.12 | |
| Smoking during pregnancy, n (%) | 74 (10) | 20 (17.9) | 54 (8.6) | 0.05 | |
| Body mass index (kg/m2), mean ± sd | 24.3 ± 4.82 | 25.8 ± 5.93 | 24 ± 4.55 | 0.006 | |
| Weight status pre-pregnancy, n (%) | Underweight | 29 (3.9) | 2 (1.8) | 27 (4.3) | 0.29 |
| Normal weight | 469 (63.1) | 66 (58.9) | 403 (63.9) | 0.34 | |
| Overweight | 154 (20.7) | 20 (17.9) | 134 (21.2) | 0.45 | |
| Obese | 91 (12.3) | 24 (21.4) | 67 (10.6) | 0.003 | |
| Gravidity, n (%) | 0 | 303 (40.8) | 38 (33.9) | 265 (42) | 0.12 |
| 1 | 256 (34.4) | 50 (44.6) | 206 (32.7) | 0.02 | |
| 2 | 116 (15.6) | 14 (12.5) | 102 (16.2) | 0.4 | |
| 3 | 49 (6.6) | 7 (6.3) | 42 (6.7) | 1 | |
| 4 | 16 (2.1) | 3 (2.7) | 13 (2.1) | 0.72 | |
| 5 | 3 (0.4) | 0 (0) | 3 (0.5) | 1 | |
| Parity, n (%) | 0 | 395 (53.2) | 59 (52.7) | 336 (53.3) | 0.91 |
| 1 | 266 (35.8) | 43 (38.4) | 223 (35.3) | 0.6 | |
| 2 | 72 (9.7) | 9 (8) | 63 (10) | 0.6 | |
| 3 | 10 (1.4) | 1 (0.9) | 9 (1.5) | 1 | |
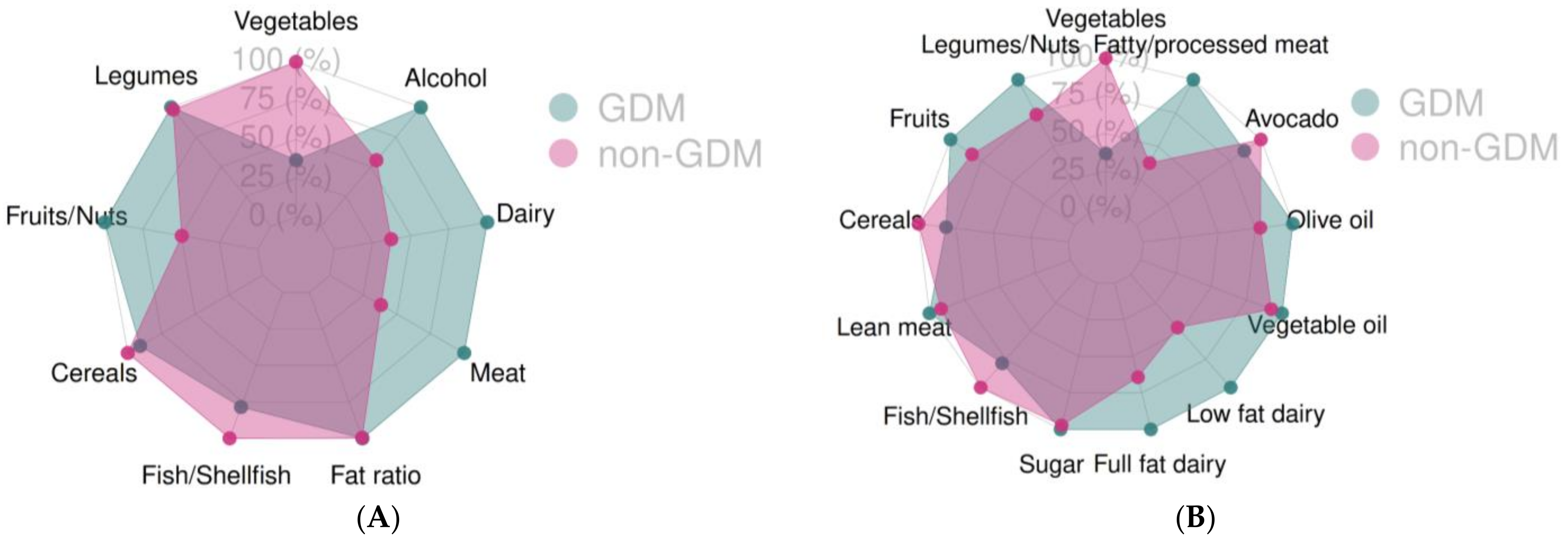
| Food Group Categories of the Mediterranean Diet (g/Day) | GDM (n = 112) Median 95% CI | Non GDM (n = 631) Median 95% CI | p-Value |
|---|---|---|---|
| p25, p50, p75 | p25, p50, p75 | ||
| Vegetables | 88.3 83.3, 88.3 60.3, 88.3, 117.9 | 92.7 88.3–98.8 63, 92.7, 133.3 | 0.08 |
| Legumes | 26.7 26.7–26.7 26.78, 26.7, 26.7 | 26.7 26.7–26.7 26.78, 26.7, 26.7 | 0.86 |
| Fruits and nuts | 130 92.8–134.5 51.3, 130, 175.2 | 119.5 94.3–130 55.7, 119.5, 155.8 | 0.86 |
| Cereals | 12.1 6.09–18.2 0, 12.1, 24.3 | 12.1 12.1–12.1 0, 12.1, 18.2 | 0.93 |
| Fish | 18.2 12.7–18.2 8.5, 18.2, 18.2 | 18.2 18.2–18.2 8.5, 18.2, 18.2 | 0.34 |
| Dairy products | 182.7 120.8–246.1 120.8, 182.7, 285.5 | 164.7 144.3–167.8 120.8, 164.7, 241.6 | 0.24 |
| Meat and derivatives | 52.1 36–56 36, 52.1, 73.1 | 41.4 36–46.1 36, 41.4, 56 | 0.008 |
| Monounsaturated/saturated fat ratio | 1.53 1.4–1.7 1.2, 1.5, 1.9 | 1.53 1.48–1.58 1.28, 1.53, 1.8 | 0.86 |
| Alcohol | 0 0–11 0, 0, 29.5 | 0 0–0 0, 0, 23.6 | 0.11 |
| MDS score, mean (sd) | 4.4 (1.8) | 4.7 (1.7) | 0.18 |
| Food Group Categories of the Mediterranean Diet (g/Day) | GDM (n = 112) Median 95% CI | Non GDM (n = 631) Median 95% CI | p Value |
|---|---|---|---|
| p25, p50, p75 | p25, p50, p75 | ||
| Vegetables (without potatoes) | 88.3 83.3–88.3 60.3, 88.3, 117.9 | 92.7 88.3–98.8 63, 92.7, 133.3 | 0.07 |
| Legumes and nuts | 33.2 26.7–45.2 26.7, 33.2, 56.9 | 33.2 31.3–39.7 26.7, 33.2, 52.6 | 0.46 |
| Fruits | 132 115.4–174.5 53.5, 132, 255 | 130 130–134.1 64.2, 130, 201.4 | 0.95 |
| Whole grain cereals | 16.7 12.1–18.2 0, 16.7, 24.3 | 12.1 12.1–18.2 1.4, 12.1, 24.3 | 0.89 |
| Lean meat | 16 16–16 16.07, 16.07, 32.14 | 16 16–16 16, 16, 32.1 | 0.67 |
| Fish and shellfish | 18.2 12.7–18.2 8.5, 18.21, 18.21 | 18.2 18.2–18.2 8.5, 18.21, 18.21 | 0.34 |
| Fatty meat and processed meat | 47 40–58.4 31.9, 47.04, 72.2 | 40 38.4–40 22.8, 40, 60 | 0.004 |
| Full fat dairy products not fermented | 120.8 120.8–120.8 34.5, 120.8, 241.6 | 120.8 120.8–120.8 51.77, 120.8, 241.6 | 0.72 |
| Low fat and fermented dairy products | 0 0–0 0, 0, 123.56 | 0 0–0 0, 0, 70.60 | 0.78 |
| Vegetable oils | 0 0–0 0, 0, 0 | 0 0–0 0, 0, 0 | 0.33 |
| Olive and canola oil | 30 30–30 15, 30, 30 | 30 30–30 15, 30, 45 | 0.32 |
| Avocado | 1.3 0–2.6 0, 1.33, 5.71 | 2.6 1.3–2.6 0, 2.6, 11.4 | 0.40 |
| Sugar | 19.8 13.2–26.4 4.6, 19.8, 46.3 | 19.8 19.8–19.8 6.6, 19.8, 46.3 | 0.86 |
| Wine | ND | ND | ND |
| Modified MDS, mean (sd) | 4.6 (1.1) | 4.7 (1.2) | 0.62 |
| Adherence to Mediterranean Diet (Points) | Total Population n (%) | GDM n (%) | Non GDM n (%) | aOR | 95% CI | p Value |
|---|---|---|---|---|---|---|
| Low (0–3) | 202 (27.2) | 38 (33.9) | 164 (26) | 1 | Reference | Reference |
| Middle (4) | 152 (20.5) | 23 (20.5) | 129 (20.44) | 0.72 | 0.40–1.28 | 0.27 |
| High (5–9) | 389 (52.4) | 51 (45.5) | 338 (53.6) | 0.57 | 0.36–0.94 | 0.02 |
| Adherence to Mediterranean Diet (Points) | Total Population n (%) | GDM (%) | Non GDM n (%) | aOR | 95% CI | p Value |
|---|---|---|---|---|---|---|
| Low (0–3.5) | 157 (21.13) | 23 (20.54) | 134 (21.23) | 1 | Reference | Reference |
| Middle (4–4.5) | 341 (45.9) | 53 (47.32) | 288 (45.64) | 1.02 | 0.59–1.78 | 0.94 |
| High (5–14) | 245 (32.97) | 36 (32.14) | 209 (33.12) | 0.85 | 0.47–1.55 | 0.59 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tranidou, A.; Dagklis, T.; Magriplis, E.; Apostolopoulou, A.; Tsakiridis, I.; Chroni, V.; Tsekitsidi, E.; Kalaitzopoulou, I.; Pazaras, N.; Chourdakis, M. Pre-Pregnancy Adherence to Mediterranean Diet and Risk of Gestational Diabetes Mellitus: A Prospective Cohort Study in Greece. Nutrients 2023, 15, 848. https://doi.org/10.3390/nu15040848
Tranidou A, Dagklis T, Magriplis E, Apostolopoulou A, Tsakiridis I, Chroni V, Tsekitsidi E, Kalaitzopoulou I, Pazaras N, Chourdakis M. Pre-Pregnancy Adherence to Mediterranean Diet and Risk of Gestational Diabetes Mellitus: A Prospective Cohort Study in Greece. Nutrients. 2023; 15(4):848. https://doi.org/10.3390/nu15040848
Chicago/Turabian StyleTranidou, Antigoni, Themistoklis Dagklis, Emmanuella Magriplis, Aikaterini Apostolopoulou, Ioannis Tsakiridis, Violeta Chroni, Eirini Tsekitsidi, Ioustini Kalaitzopoulou, Nikolaos Pazaras, and Michail Chourdakis. 2023. "Pre-Pregnancy Adherence to Mediterranean Diet and Risk of Gestational Diabetes Mellitus: A Prospective Cohort Study in Greece" Nutrients 15, no. 4: 848. https://doi.org/10.3390/nu15040848
APA StyleTranidou, A., Dagklis, T., Magriplis, E., Apostolopoulou, A., Tsakiridis, I., Chroni, V., Tsekitsidi, E., Kalaitzopoulou, I., Pazaras, N., & Chourdakis, M. (2023). Pre-Pregnancy Adherence to Mediterranean Diet and Risk of Gestational Diabetes Mellitus: A Prospective Cohort Study in Greece. Nutrients, 15(4), 848. https://doi.org/10.3390/nu15040848








