Scytosiphon lomentaria Extract Ameliorates Obesity and Modulates Gut Microbiota in High-Fat-Diet-Fed Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of S. lomentaria Extract and LC-MS Analysis
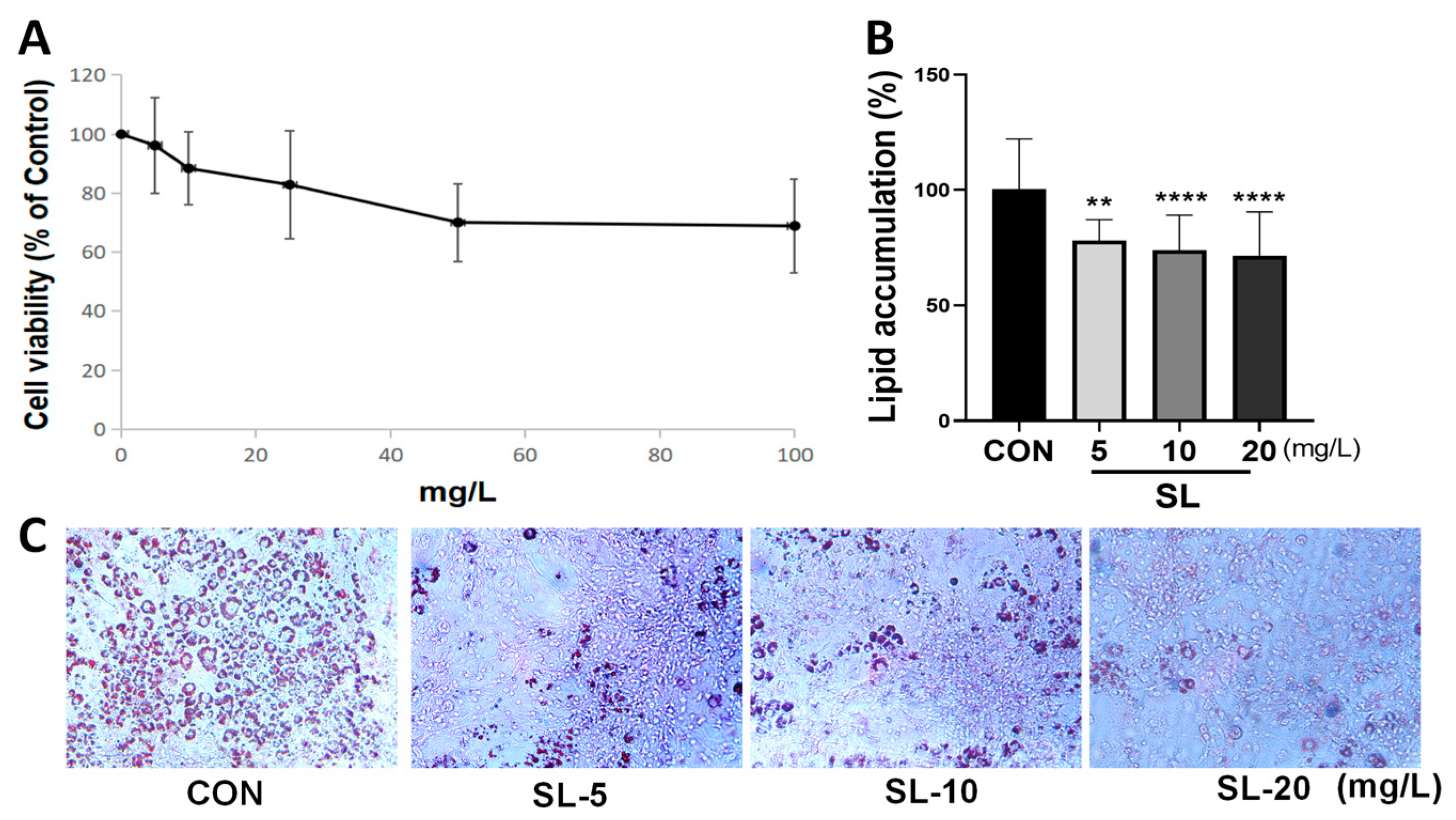
2.2. Cell Viability Assay
2.3. Cell Differentiation and Oil Red O Staining
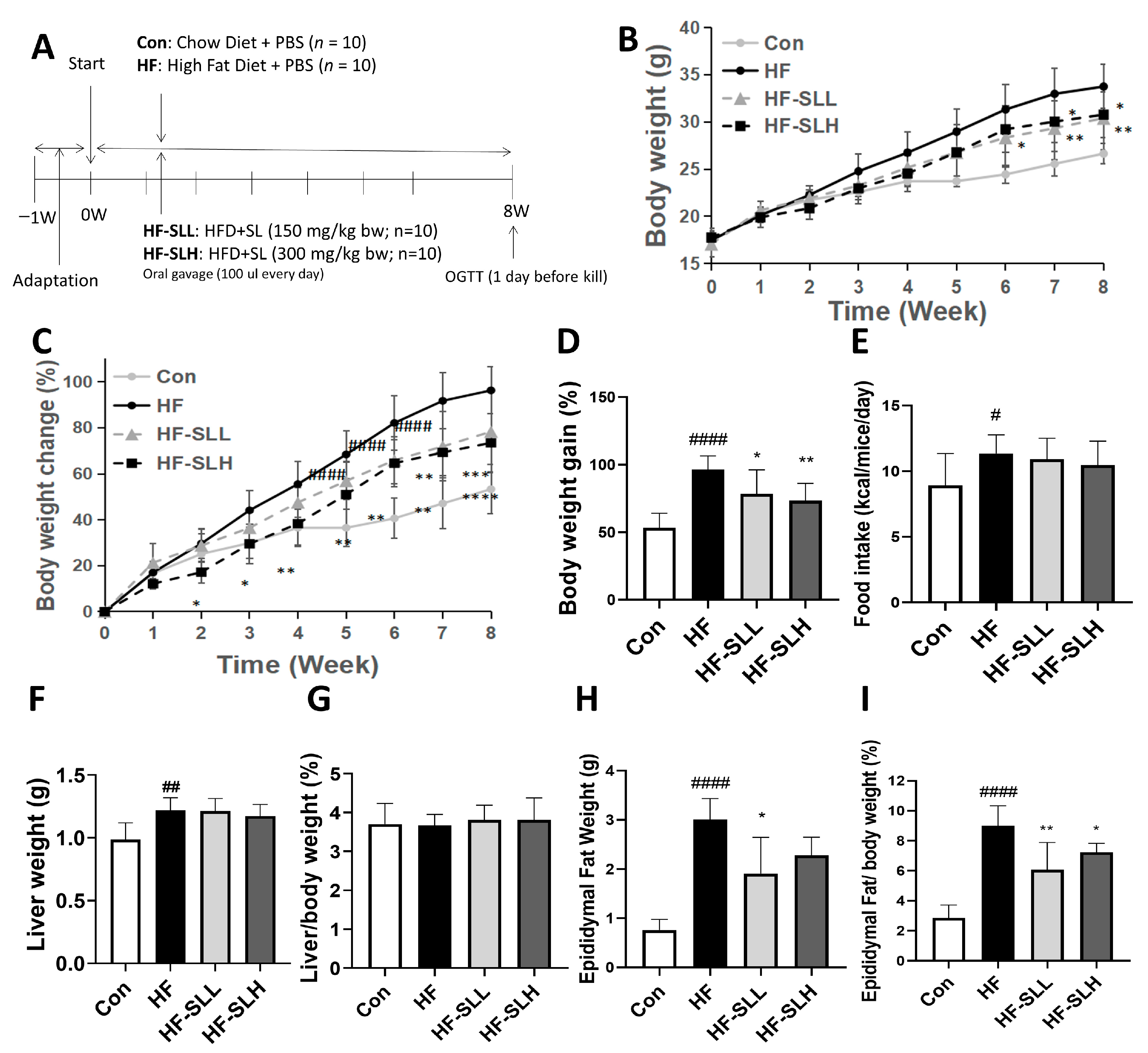
2.4. Animal Studies
2.5. Histolopathology of Adipose Tissues and Liver
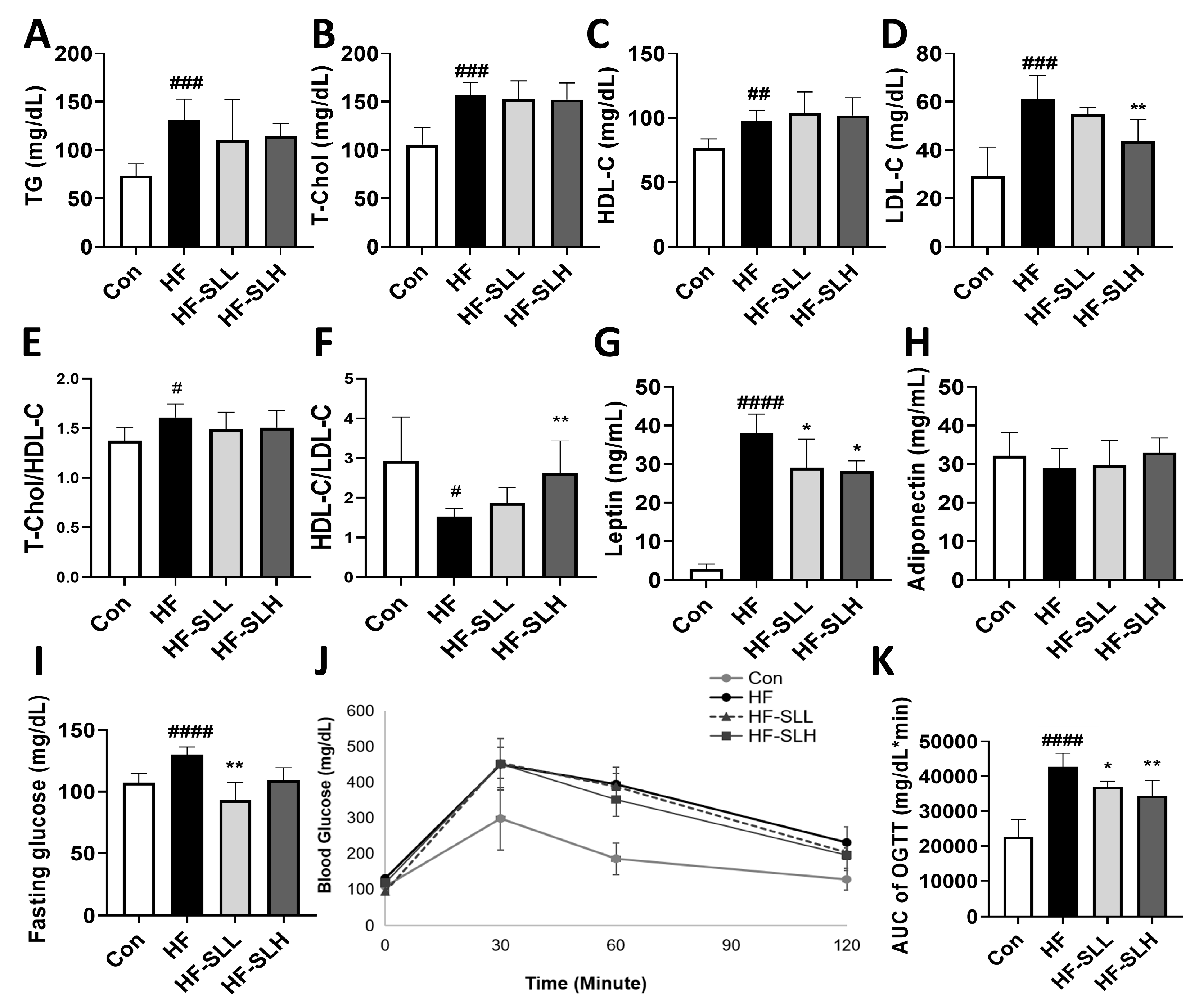
2.6. Blood Analysis
2.7. RT-PCR Analysis
2.8. Bacterial DNA Extraction, qPCR, and 16S rRNA Gene Sequencing
2.9. Western Blot
2.10. Statistical Analysis
3. Results
3.1. Identification of Phytochemical Compounds in S. lomentaria Extract
3.2. Effects of S. lomentaria Extract on the Differentiation of 3T3-L1 Cells
3.3. Effects of S. lomentaria Extract on Gene and Protein Expressions Related to Lipogenesis, Adipogenesis, and Thermogenesis in 3T3-L1 Cells
3.4. Effects of S. lomentaria Extract on Body Weight and Organ Weight in High-Fat-Fed Mice
3.5. Effects of S. lomentaria Extract on the Adipose Tissue and Liver Histology in High-Fat-Fed Mice
3.6. Effects of S. lomentaria Extract on Serum Profiles
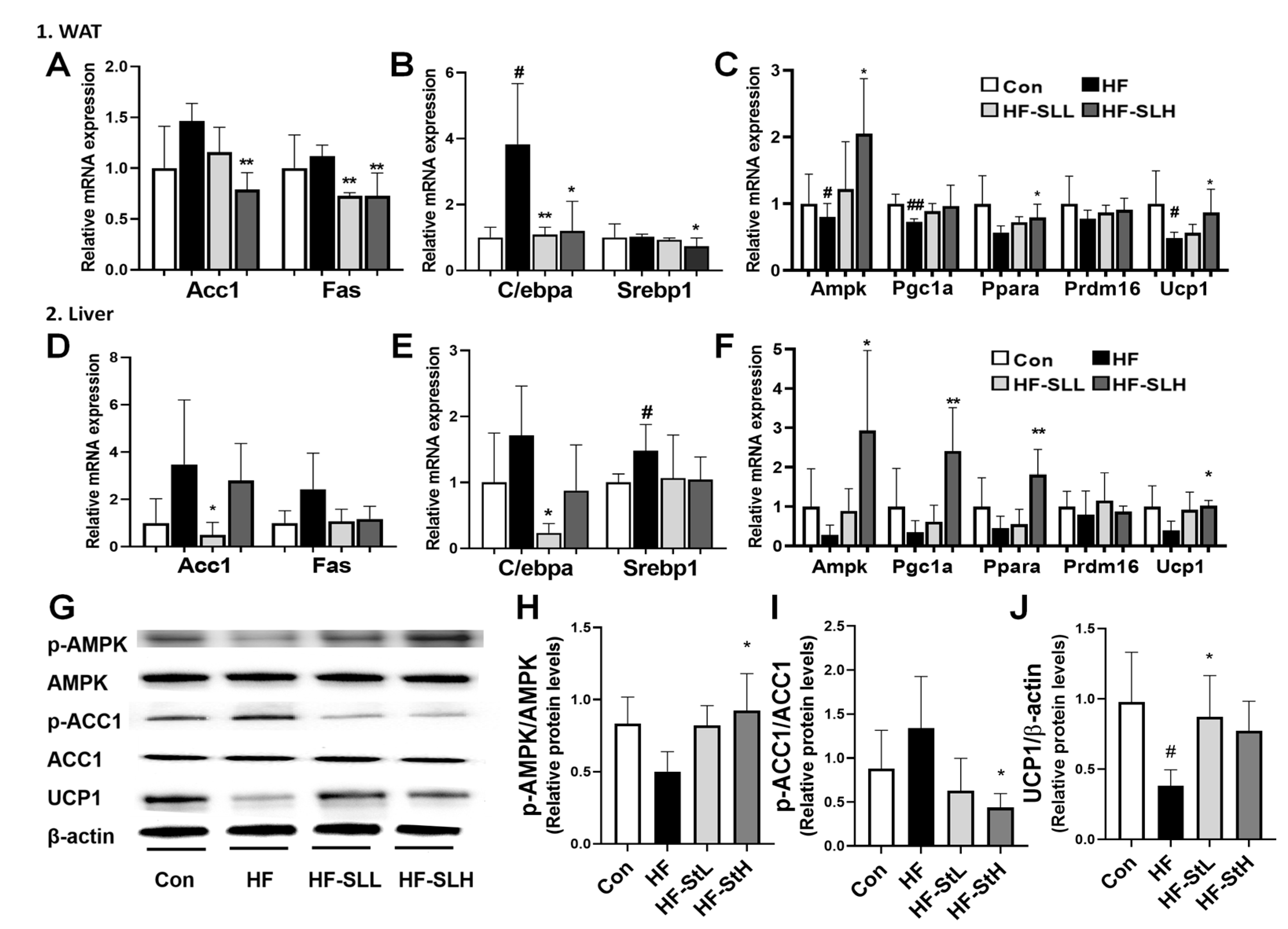
3.7. Effects of S. lomentaria Extract on the Gene and Protein Expression Related to Lipogenesis, Adipogenesis, and Thermogenesis in WAT and Liver in High-Fat-Fed Mice
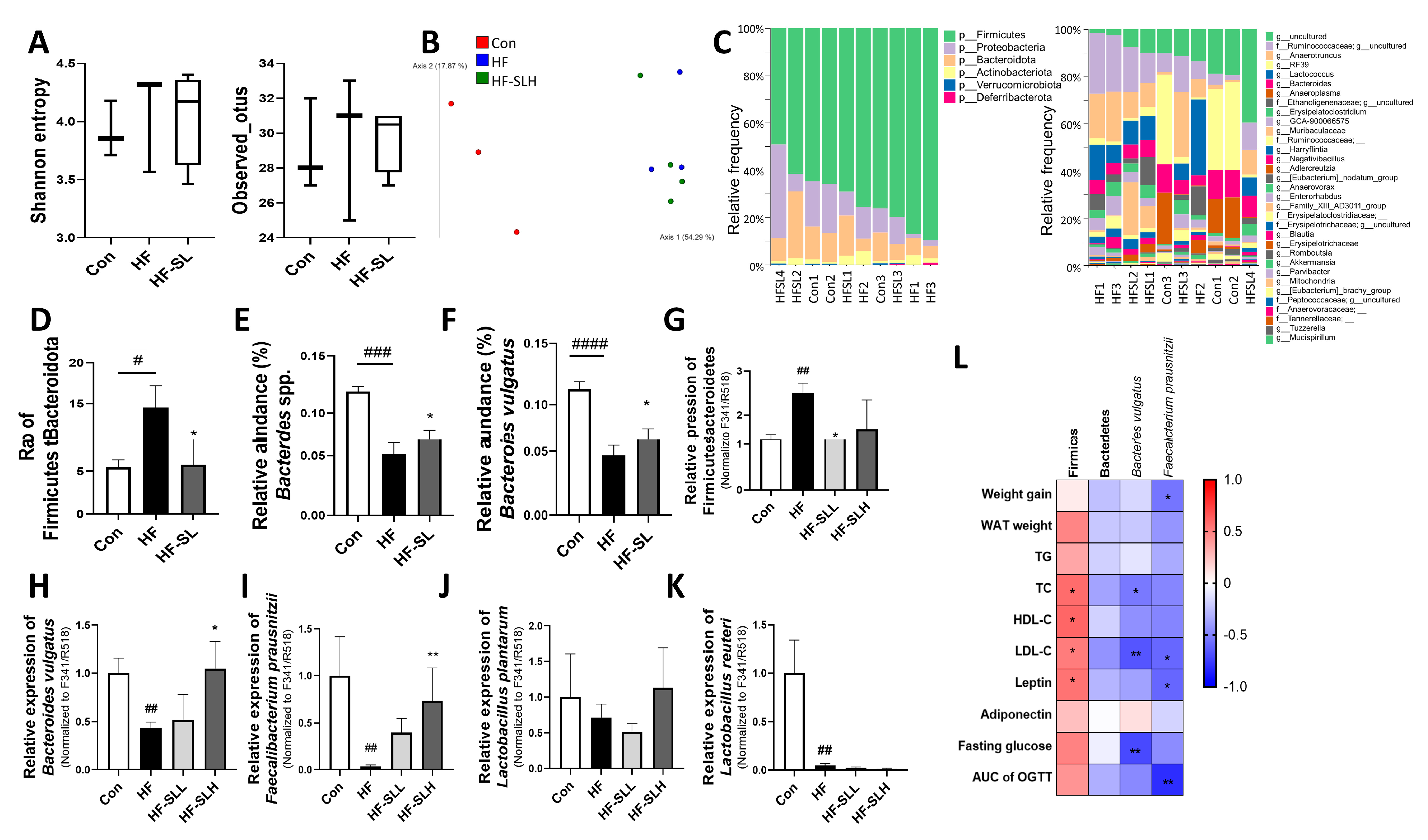
3.8. Effects of S. lomentaria Extract on the Gut Microbiota Composition in High-Fat-Fed Mice
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lafontan, M. Adipose tissue and adipocyte dysregulation. Diabetes Metab. 2014, 40, 16–28. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Shen, Y.; Zhang, H.; Cao, M.; Guo, M.; He, J.; Zhang, B.; Xiao, C. Gut Microbiota Characteristics of People with Obesity by Meta-Analysis of Existing Datasets. Nutrients 2022, 14, 2993. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.-F.; Wang, L.; Pan, A. Epidemiology and determinants of obesity in China. Lancet Diabetes Endocrinol. 2021, 9, 373–392. [Google Scholar] [CrossRef]
- Saltiel, A.R. New therapeutic approaches for the treatment of obesity. Sci. Transl. Med. 2016, 8, 323rv2. [Google Scholar] [CrossRef]
- Srivastava, G.; Apovian, C.M. Current pharmacotherapy for obesity. Nat. Rev. Endocrinol. 2018, 14, 12–24. [Google Scholar] [CrossRef]
- Su, H.; Wang, W.J.; Zheng, G.D.; Yin, Z.P.; Li, J.E.; Chen, L.L.; Zhang, Q.F. The anti-obesity and gut microbiota modulating effects of taxifolin in C57BL/6J mice fed with a high-fat diet. J. Sci. Food Agric. 2022, 102, 1598–1608. [Google Scholar] [CrossRef]
- Kishida, R.; Yamagishi, K.; Muraki, I.; Sata, M.; Tamakoshi, A.; Iso, H.; Group, J.S. Frequency of seaweed intake and its association with cardiovascular disease mortality: The JACC Study. J. Atheroscler. Thromb. 2020, 27, 1340–1347. [Google Scholar] [CrossRef]
- Vaughan, K.; Ranawana, V.; Cooper, D.; Aceves-Martins, M. Effect of brown seaweed on plasma glucose in healthy, at-risk, and type 2 diabetic individuals: Systematic review and meta-analysis. Nutr. Rev. 2022, 80, 1194–1205. [Google Scholar] [CrossRef]
- Peng, J.; Yuan, J.-P.; Wu, C.-F.; Wang, J.-H. Fucoxanthin, a marine carotenoid present in brown seaweeds and diatoms: Metabolism and bioactivities relevant to human health. Mar. Drugs 2011, 9, 1806–1828. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Cai, C.; Yang, C.; Li, J.; Sun, T.; Yu, G. Recent advances in pharmaceutical potential of brown algal polysaccharides and their derivatives. Curr. Pharm. Des. 2019, 25, 1290–1311. [Google Scholar] [CrossRef]
- Sharifuddin, Y.; Chin, Y.-X.; Lim, P.-E.; Phang, S.-M. Potential bioactive compounds from seaweed for diabetes management. Mar. Drugs 2015, 13, 5447–5491. [Google Scholar] [CrossRef] [PubMed]
- Kuda, T.; Tsunekawa, M.; Hishi, T.; Araki, Y. Antioxidant properties of driedkayamo-nori', a brown alga Scytosiphon lomentaria (Scytosiphonales, Phaeophyceae). Food Chem. 2005, 89, 617–622. [Google Scholar] [CrossRef]
- Kim, S.-C.; Park, S.-Y.; Hyoun, J.-H.; Cho, H.; Kang, J.-H.; Lee, Y.-K.; Park, D.-B.; Yoo, E.-S.; Kang, H.-K. The cytotoxicity of Scytosiphon lomentaria against HL-60 promyelocytic leukemia cells. Cancer Biother. Radiopharm. 2004, 19, 641–648. [Google Scholar] [CrossRef]
- Xu, X.J.; Zhang, Y.; Lin, H.; Kang, C.; Fu, X.T. Antioxidant and Antimicrobial Properties of a Brown Alga Scytosiphon lomentaria. Chiang Mai J. Sci. 2017, 44, 595–604. [Google Scholar]
- Pan, C.-H.; Kim, E.-S.; Um, B.-H.; Lee, J.-K. Anti-inflammatory mechanism of seaweeds in murine macrophage. Food Sci. Biotechnol. 2009, 18, 813–817. [Google Scholar]
- Jia, J.; Zheng, W.; Zhang, C.; Zhang, P.; Guo, X.; Song, S.; Ai, C. Fucoidan from Scytosiphon lomentaria protects against destruction of intestinal barrier, inflammation and lipid abnormality by modulating the gut microbiota in dietary fibers-deficient mice. Int. J. Biol. Macromol. 2023, 224, 556–567. [Google Scholar] [CrossRef]
- Jung, J.G.; Kim, J.H.; Kim, J.H.; Kim, Y.S.; Jin, D.-H.; Jin, H.-J. Effect of Scytosiphon lomentaria Ethanol Extracts on Myostatin Activity and Zebrafish Obesity Induced by High Feeding. J. Life Sci. 2021, 31, 699–709. [Google Scholar]
- Jakab, J.; Miškić, B.; Mikšić, Š.; Juranić, B.; Ćosić, V.; Schwarz, D.; Včev, A. Adipogenesis as a Potential Anti-Obesity Target: A Review of Pharmacological Treatment and Natural Products. Diabetes Metab. Syndr. Obes. 2021, 14, 67–83. [Google Scholar] [CrossRef]
- Batchuluun, B.; Pinkosky, S.L.; Steinberg, G.R. Lipogenesis inhibitors: Therapeutic opportunities and challenges. Nat. Rev. Drug Discov. 2022, 21, 283–305. [Google Scholar] [CrossRef]
- Sung, Y.Y.; Kim, S.H.; Kim, D.S. Combined Phyllostachys pubescens and Scutellaria baicalensis Prevent High-Fat Diet-Induced Obesity via Upregulating Thermogenesis and Energy Expenditure by UCP1 in Male C57BL/6J Mice. Nutrients 2022, 14, 446. [Google Scholar] [CrossRef] [PubMed]
- Cooper, M.P.; Uldry, M.; Kajimura, S.; Arany, Z.; Spiegelman, B.M. Modulation of PGC-1 coactivator pathways in brown fat differentiation through LRP130. J. Biol. Chem. 2008, 283, 31960–31967. [Google Scholar] [CrossRef] [PubMed]
- Cuevas-Sierra, A.; Ramos-Lopez, O.; Riezu-Boj, J.I.; Milagro, F.I.; Martinez, J.A. Diet, gut microbiota, and obesity: Links with host genetics and epigenetics and potential applications. Adv. Nutr. 2019, 10, S17–S30. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Xu, Z.; Li, Y.; Gan, L.; Wu, P.; Wu, R.; Jin, J.; Zheng, X.; Zhang, K.; Ma, H.; et al. Polysaccharides from Callerya speciosa alleviate metabolic disorders and gut microbiota dysbiosis in diet-induced obese C57BL/6 mice. Food Funct. 2022, 13, 8662–8675. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.J.; Lin, C.S.; Lu, C.C.; Martel, J.; Ko, Y.F.; Ojcius, D.M.; Tseng, S.F.; Wu, T.R.; Chen, Y.Y.; Young, J.D.; et al. Ganoderma lucidum reduces obesity in mice by modulating the composition of the gut microbiota. Nat. Commun. 2015, 6, 7489. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.N.; Liu, X.T.; Liang, Z.H.; Wang, J.H. Gut microbiota in obesity. World J. Gastroenterol. 2021, 27, 3837–3850. [Google Scholar] [CrossRef]
- Swain, T.; Hillis, W. The phenolic constituents of Prunus domestica. I.—The quantitative analysis of phenolic constituents. J. Sci. Food Agric. 1959, 10, 63–68. [Google Scholar] [CrossRef]
- Cassano, A.; Conidi, C.; Ruby-Figueroa, R. Recovery of flavonoids from orange press liquor by an integrated membrane process. Membranes 2014, 4, 509–524. [Google Scholar] [CrossRef]
- Kim, D.; Yan, J.; Bak, J.; Park, J.; Lee, H.; Kim, H. Sargassum thunbergii Extract Attenuates High-Fat Diet-Induced Obesity in Mice by Modulating AMPK Activation and the Gut Microbiota. Foods 2022, 11, 2529. [Google Scholar] [CrossRef]
- Zhao, D.; Pan, Y.; Yu, N.; Bai, Y.; Ma, R.; Mo, F.; Zuo, J.; Chen, B.; Jia, Q.; Zhang, D. Curcumin improves adipocytes browning and mitochondrial function in 3T3-L1 cells and obese rodent model. Royal Soc. Open Sci. 2021, 8, 200974. [Google Scholar] [CrossRef]
- Zhu, Y.; Guo, J.; Hu, X.; Liu, J.; Li, S.; Wang, J. Eckol protects against acute experimental colitis in mice: Possible involvement of Reg3g. J. Funct. Foods 2020, 73, 104088. [Google Scholar] [CrossRef]
- Li, K.K.; Liu, C.L.; Shiu, H.T.; Wong, H.L.; Siu, W.S.; Zhang, C.; Han, X.Q.; Ye, C.X.; Leung, P.C.; Ko, C.H. Cocoa tea (Camellia ptilophylla) water extract inhibits adipocyte differentiation in mouse 3T3-L1 preadipocytes. Sci. Rep. 2016, 6, 20172. [Google Scholar] [CrossRef]
- Won, S.-M.; Chen, S.; Park, K.W.; Yoon, J.-H. Isolation of lactic acid bacteria from kimchi and screening of Lactobacillus sakei ADM14 with anti-adipogenic effect and potential probiotic properties. LWT 2020, 126, 109296. [Google Scholar] [CrossRef]
- Lane, M.D. Adipogenesis. In Encyclopedia of Biological Chemistry, 2nd ed.; Lennarz, W.J., Lane, M.D., Eds.; Academic Press: Waltham, MA, USA, 2013; pp. 52–56. [Google Scholar]
- Zhao, J.; Zhou, A.; Qi, W. The Potential to Fight Obesity with Adipogenesis Modulating Compounds. Int. J. Mol. Sci. 2022, 23, 2299. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.; Lee, H.; Kim, S.; Park, J.; Ha, T. The anti-obesity effect of quercetin is mediated by the AMPK and MAPK signaling pathways. Biochem. Biophys. Res. Commun. 2008, 373, 545–549. [Google Scholar] [CrossRef]
- Choi, K.-M.; Lee, Y.-S.; Kim, W.; Kim, S.J.; Shin, K.-O.; Yu, J.-Y.; Lee, M.K.; Lee, Y.-M.; Hong, J.T.; Yun, Y.-P. Sulforaphane attenuates obesity by inhibiting adipogenesis and activating the AMPK pathway in obese mice. J. Nutr. Biochem. 2014, 25, 201–207. [Google Scholar] [CrossRef]
- Fryer, L.; Carling, D. AMP-activated protein kinase and the metabolic syndrome. Biochem. Soc. Trans. 2005, 33, 362–366. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, C.; Wu, J.; Liu, W.; Li, D.; Xu, J. Harmane ameliorates obesity though inhibiting lipid accumulation and inducing adipocyte browning. RSC Adv. 2020, 10, 4397–4403. [Google Scholar] [CrossRef]
- Lin, S.; Wang, Z.; Lin, Y.; Ge, S.; Hamzah, S.S.; Hu, J. Bound phenolics from fresh lotus seeds exert anti-obesity effects in 3T3-L1 adipocytes and high-fat diet-fed mice by activation of AMPK. J. Funct. Foods 2019, 58, 74–84. [Google Scholar] [CrossRef]
- Pan, R.; Zhu, X.; Maretich, P.; Chen, Y. Combating obesity with thermogenic fat: Current challenges and advancements. Front. Endocrinol. 2020, 11, 185. [Google Scholar] [CrossRef]
- Singh, M.; Thrimawithana, T.; Shukla, R.; Adhikari, B. Managing obesity through natural polyphenols: A review. Future Foods 2020, 1, 100002. [Google Scholar] [CrossRef]
- Jeon, S.-M.; Kim, H.-J.; Woo, M.-N.; Lee, M.-K.; Shin, Y.C.; Park, Y.B.; Choi, M.-S. Fucoxanthin-rich seaweed extract suppresses body weight gain and improves lipid metabolism in high-fat-fed C57BL/6J mice. Biotechnol. J. 2010, 5, 961–969. [Google Scholar] [CrossRef] [PubMed]
- Ponce, N.M.A.; Flores, M.L.; Pujol, C.A.; Becerra, M.B.; Navarro, D.A.; Córdoba, O.; Damonte, E.B.; Stortz, C.A. Fucoidans from the phaeophyta Scytosiphon lomentaria: Chemical analysis and antiviral activity of the galactofucan component. Carbohydr. Res. 2019, 478, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Mori, K.; Ooi, T.; Hiraoka, M.; Oka, N.; Hamada, H.; Tamura, M.; Kusumi, T. Fucoxanthin and Its Metabolites in Edible Brown Algae Cultivated in Deep Seawater. Mar. Drugs 2004, 2, 63–72. [Google Scholar] [CrossRef]
- John, C.M.; Arockiasamy, S. Sinapic acid prevents adipogenesis by regulating transcription factors and exerts an anti-ROS effect by modifying the intracellular anti-oxidant system in 3T3-L1 adipocytes. Iran. J. Basic Med. Sci. 2022, 25, 611–620. [Google Scholar] [PubMed]
- Hossain, M.; Imran, K.M.; Rahman, M.S.; Yoon, D.; Marimuthu, V.; Kim, Y.S. Sinapic acid induces the expression of thermogenic signature genes and lipolysis through activation of PKA/CREB signaling in brown adipocytes. BMB Rep. 2020, 53, 142–147. [Google Scholar] [CrossRef]
- Bae, I.-S.; Kim, S.H. Sinapic Acid Promotes Browning of 3T3-L1 Adipocytes via p38 MAPK/CREB Pathway. Biomed. Res. Int. 2020, 2020, 5753623. [Google Scholar] [CrossRef]
- Tokimitsu, I. Effects of tea catechins on lipid metabolism and body fat accumulation. Biofactors 2004, 22, 141–143. [Google Scholar] [CrossRef] [PubMed]
- Pei, Y.; Otieno, D.; Gu, I.; Lee, S.-O.; Parks, J.S.; Schimmel, K.; Kang, H.W. Effect of quercetin on nonshivering thermogenesis of brown adipose tissue in high-fat diet-induced obese mice. J. Nutr. Biochem. 2021, 88, 108532. [Google Scholar] [CrossRef]
- Liou, C.J.; Wu, S.J.; Shen, S.C.; Chen, L.C.; Chen, Y.L.; Huang, W.C. Acacetin Protects against Non-Alcoholic Fatty Liver Disease by Regulating Lipid Accumulation and Inflammation in Mice. Int. J. Mol. Sci. 2022, 23, 4687. [Google Scholar] [CrossRef]
- Liou, C.J.; Wu, S.J.; Chen, L.C.; Yeh, K.W.; Chen, C.Y.; Huang, W.C. Acacetin from Traditionally Used Saussurea involucrata Kar. et Kir. Suppressed Adipogenesis in 3T3-L1 Adipocytes and Attenuated Lipid Accumulation in Obese Mice. Front. Pharmacol. 2017, 8, 589. [Google Scholar] [CrossRef]
- Rosen, E.D.; MacDougald, O.A. Adipocyte differentiation from the inside out. Nat. Rev. Mol. Cell Biol. 2006, 7, 885–896. [Google Scholar] [CrossRef] [PubMed]
- Boughanem, H.; Cabrera-Mulero, A.; Millán-Gómez, M.; Garrido-Sánchez, L.; Cardona, F.; Tinahones, F.J.; Moreno-Santos, I.; Macías-González, M. Transcriptional Analysis of FOXO1, C/EBP-α and PPAR-γ2 Genes and Their Association with Obesity-Related Insulin Resistance. Genes 2019, 10, 706. [Google Scholar] [CrossRef] [PubMed]
- Guru, A.; Issac, P.K.; Velayutham, M.; Saraswathi, N.T.; Arshad, A.; Arockiaraj, J. Molecular mechanism of down-regulating adipogenic transcription factors in 3T3-L1 adipocyte cells by bioactive anti-adipogenic compounds. Mol. Biol. Rep. 2021, 48, 743–761. [Google Scholar] [CrossRef]
- Bu, S.; Yuan, C.Y.; Xue, Q.; Chen, Y.; Cao, F. Bilobalide Suppresses Adipogenesis in 3T3-L1 Adipocytes via the AMPK Signaling Pathway. Molecules 2019, 24, 3503. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, G.R.; Carling, D. AMP-activated protein kinase: The current landscape for drug development. Nat. Rev. Drug Discov. 2019, 18, 527–551. [Google Scholar] [CrossRef]
- Singh, P.; Zhang, Y.; Sharma, P.; Covassin, N.; Soucek, F.; Friedman, P.A.; Somers, V.K. Statins decrease leptin expression in human white adipocytes. Physiol. Rep. 2018, 6, e13566. [Google Scholar] [CrossRef]
- Hardie, D.G.; Pan, D.A. Regulation of fatty acid synthesis and oxidation by the AMP-activated protein kinase. Biochem. Soc. Trans. 2002, 30, 1064–1070. [Google Scholar] [CrossRef]
- Cherng, Y.-G.; Tsai, C.-C.; Chung, H.-H.; Lai, Y.-W.; Kuo, S.-C.; Cheng, J.-T. Antihyperglycemic Action of Sinapic Acid in Diabetic Rats. J. Agric. Food Chem. 2013, 61, 12053–12059. [Google Scholar] [CrossRef]
- Mottillo, E.P.; Desjardins, E.M.; Crane, J.D.; Smith, B.K.; Green, A.E.; Ducommun, S.; Henriksen, T.I.; Rebalka, I.A.; Razi, A.; Sakamoto, K.; et al. Lack of Adipocyte AMPK Exacerbates Insulin Resistance and Hepatic Steatosis through Brown and Beige Adipose Tissue Function. Cell Metab. 2016, 24, 118–129. [Google Scholar] [CrossRef]
- Koliada, A.; Syzenko, G.; Moseiko, V.; Budovska, L.; Puchkov, K.; Perederiy, V.; Gavalko, Y.; Dorofeyev, A.; Romanenko, M.; Tkach, S.; et al. Association between body mass index and Firmicutes/Bacteroidetes ratio in an adult Ukrainian population. BMC Microbiol. 2017, 17, 120. [Google Scholar] [CrossRef]
- Sokol, H.; Pigneur, B.; Watterlot, L.; Lakhdari, O.; Bermúdez-Humarán, L.G.; Gratadoux, J.-J.; Blugeon, S.; Bridonneau, C.; Furet, J.-P.; Corthier, G.; et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc. Natl. Acad. Sci. USA 2008, 105, 16731–16736. [Google Scholar] [CrossRef] [PubMed]
- Turck, D.; Bresson, J.L.; Burlingame, B.; Dean, T.; Fairweather-Tait, S.; Heinonen, M.; Hirsch-Ernst, K.I.; Mangelsdorf, I.; McArdle, H.J.; Naska, A.; et al. Safety of Ecklonia cava phlorotannins as a novel food pursuant to Regulation (EC) No 258/97. Efsa J. 2017, 15, e05003. [Google Scholar]
- Kim, I.H.; Choi, J.W.; Lee, M.K.; Kwon, C.J.; Nam, T.J. Anti-obesity effects of pectinase and cellulase enzyme-treated Ecklonia cava extract in high-fat diet-fed C57BL/6N mice. Int. J. Mol. Med. 2018, 41, 924–934. [Google Scholar] [PubMed]
- Abbas, M.A.; Boby, N.; Lee, E.B.; Hong, J.H.; Park, S.C. Anti-Obesity Effects of Ecklonia cava Extract in High-Fat Diet-Induced Obese Rats. Antioxidants 2022, 11, 310. [Google Scholar] [CrossRef] [PubMed]
- Park, E.Y.; Choi, H.; Yoon, J.Y.; Lee, I.Y.; Seo, Y.; Moon, H.S.; Hwang, J.H.; Jun, H.S. Polyphenol-Rich Fraction of Ecklonia cava Improves Nonalcoholic Fatty Liver Disease in High Fat Diet-Fed Mice. Mar. Drugs 2015, 13, 6866–6883. [Google Scholar] [CrossRef]
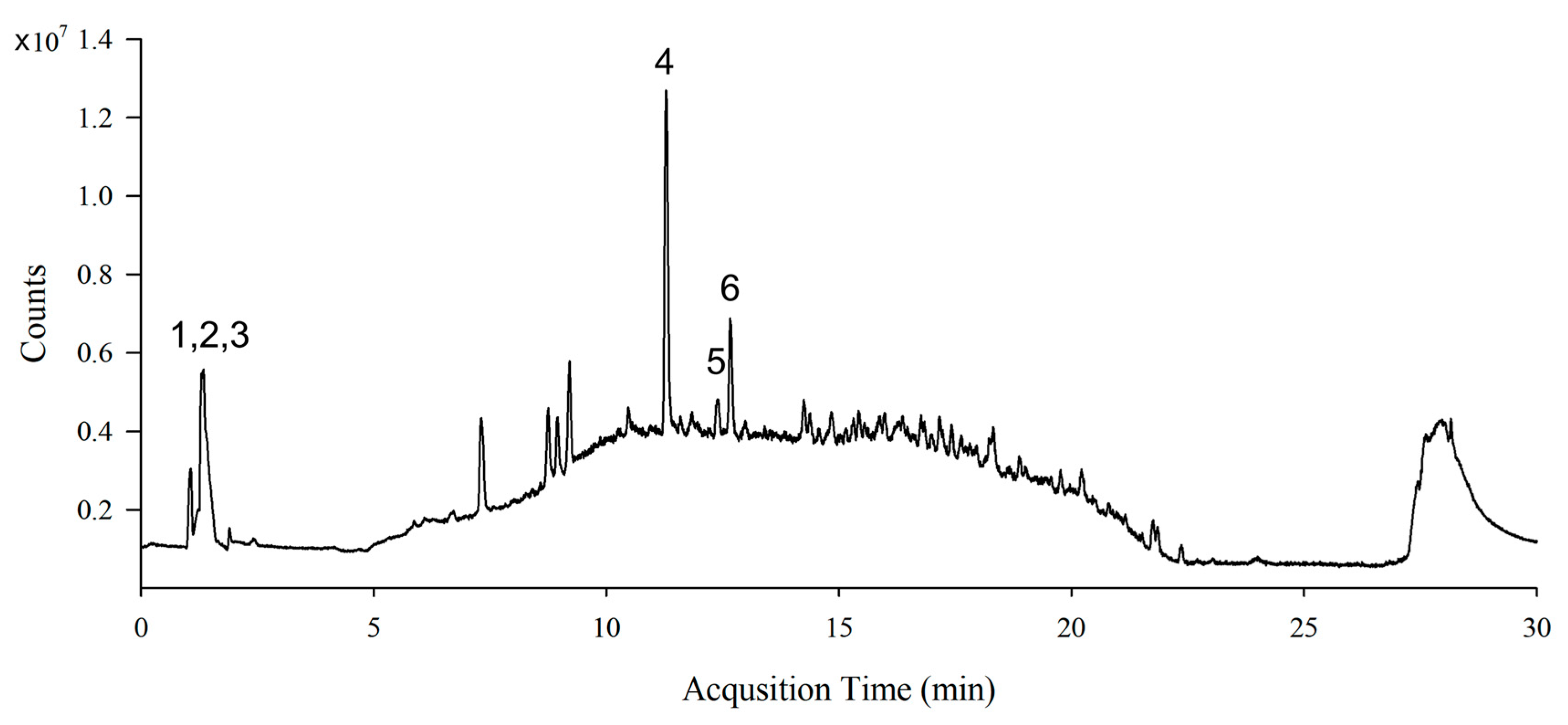


| Peak | Compound Name | Molecular Formula | Retention Time (min) | [M+H]+ | Fragment Ion (m/z) | Content * |
|---|---|---|---|---|---|---|
| 1 | Sinapic acid | C11H12O5 | 1.348 | 225.0757 | 208.1340, 207.0781, 206.0697 | 13.00 |
| 2 | Catechin | C15H14O6 | 1.348 | 291.0863 | 207.0781, 140.0687, 122.0968 | 13.00 |
| 3 | Quercetin | C15H10O7 | 1.348 | 303.0499 | 302.1982, 258.1327, 202.1808 | 13.00 |
| 4 | Hydroxytrifuhalol A | C18H14O11 | 11.289 | 407.0609 | 407.2007, 391.2078, 361.1700, 249.1461 | 16.44 |
| 5 | Acacetin | C16H12O5 | 12.381 | 285.0757 | 243.1123, 242.1096, 241.1044 | 1.99 |
| 6 | Caffeic acid | C9H8O4 | 12.669 | 181.0495 | 182.1262, 180.1135, 181.1230, | 5.33 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yan, J.; Bak, J.; Go, Y.; Park, J.; Park, M.; Lee, H.-J.; Kim, H. Scytosiphon lomentaria Extract Ameliorates Obesity and Modulates Gut Microbiota in High-Fat-Diet-Fed Mice. Nutrients 2023, 15, 815. https://doi.org/10.3390/nu15040815
Yan J, Bak J, Go Y, Park J, Park M, Lee H-J, Kim H. Scytosiphon lomentaria Extract Ameliorates Obesity and Modulates Gut Microbiota in High-Fat-Diet-Fed Mice. Nutrients. 2023; 15(4):815. https://doi.org/10.3390/nu15040815
Chicago/Turabian StyleYan, Jing, Jinwoo Bak, Yula Go, Jumin Park, Minkyoung Park, Hae-Jeung Lee, and Hyemee Kim. 2023. "Scytosiphon lomentaria Extract Ameliorates Obesity and Modulates Gut Microbiota in High-Fat-Diet-Fed Mice" Nutrients 15, no. 4: 815. https://doi.org/10.3390/nu15040815
APA StyleYan, J., Bak, J., Go, Y., Park, J., Park, M., Lee, H.-J., & Kim, H. (2023). Scytosiphon lomentaria Extract Ameliorates Obesity and Modulates Gut Microbiota in High-Fat-Diet-Fed Mice. Nutrients, 15(4), 815. https://doi.org/10.3390/nu15040815






