Effect of Continuous Ingestion of Bifidobacteria and Inulin on Reducing Body Fat: A Randomized, Double-Blind, Placebo-Controlled, Parallel-Group Comparison Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Test Foods
2.3. Experimental Design
2.4. Abdominal Fat Area
2.5. Anthropometric Measures and Body Composition
2.6. Clinical Parameters
2.7. Fecal Samples
2.8. Fecal DNA Extraction
2.9. Fecal Bifidobacteria
2.10. Statistical Analysis
3. Results
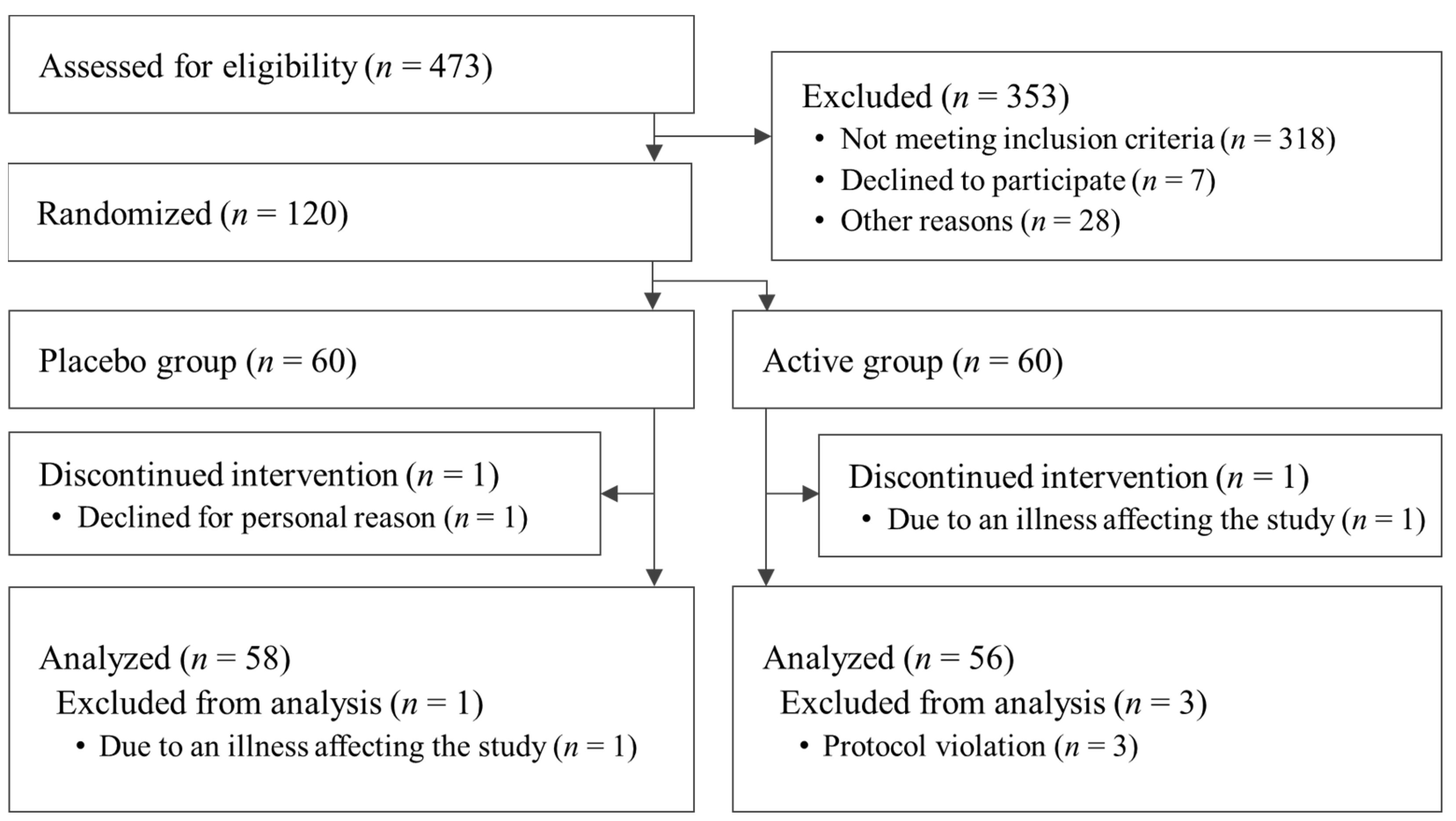
3.1. Analysis of the Participant Population
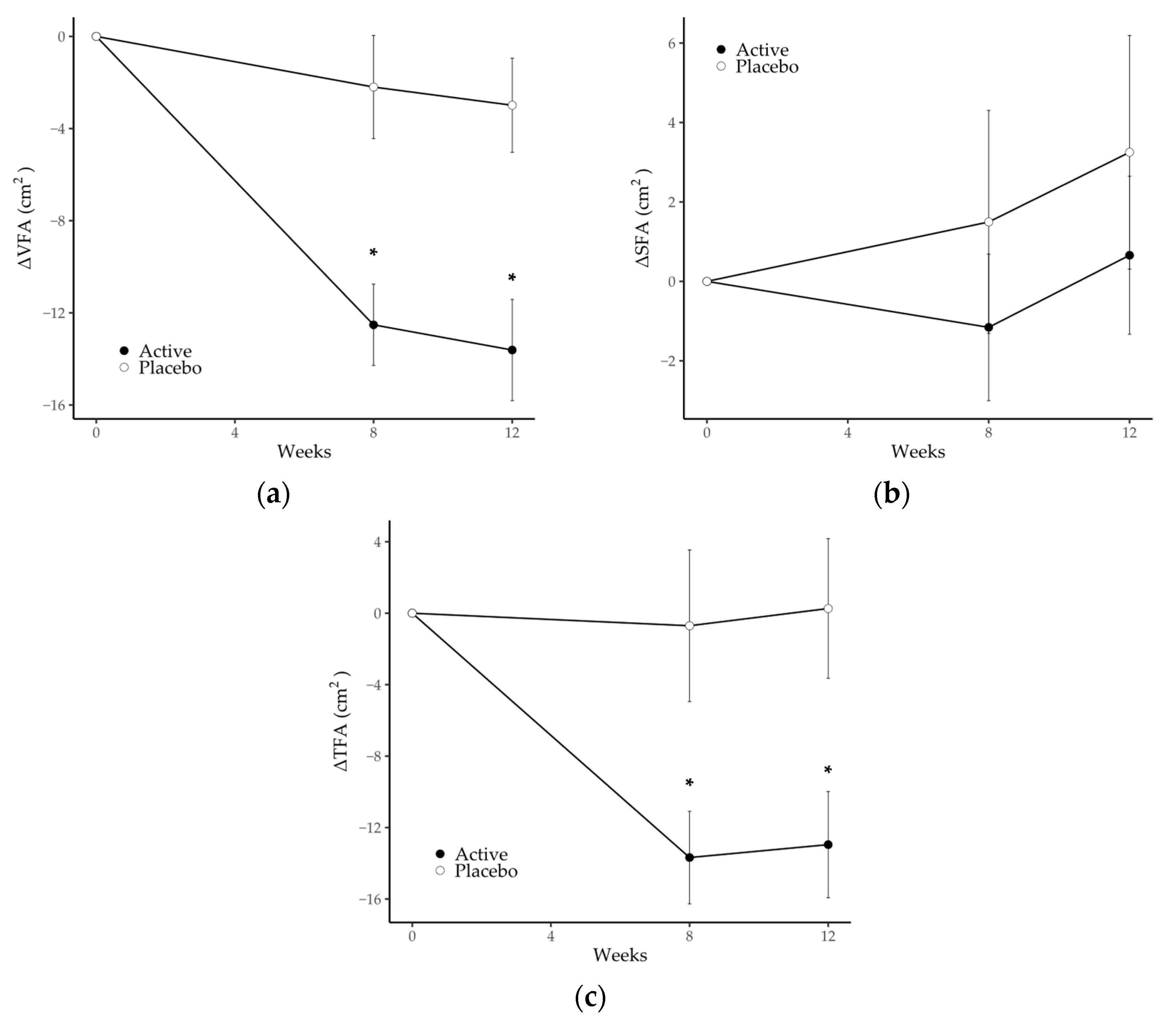
3.2. Abdominal Fat Area
3.3. Anthropometric Parameters
3.4. Biochemical Parameters in Plasma
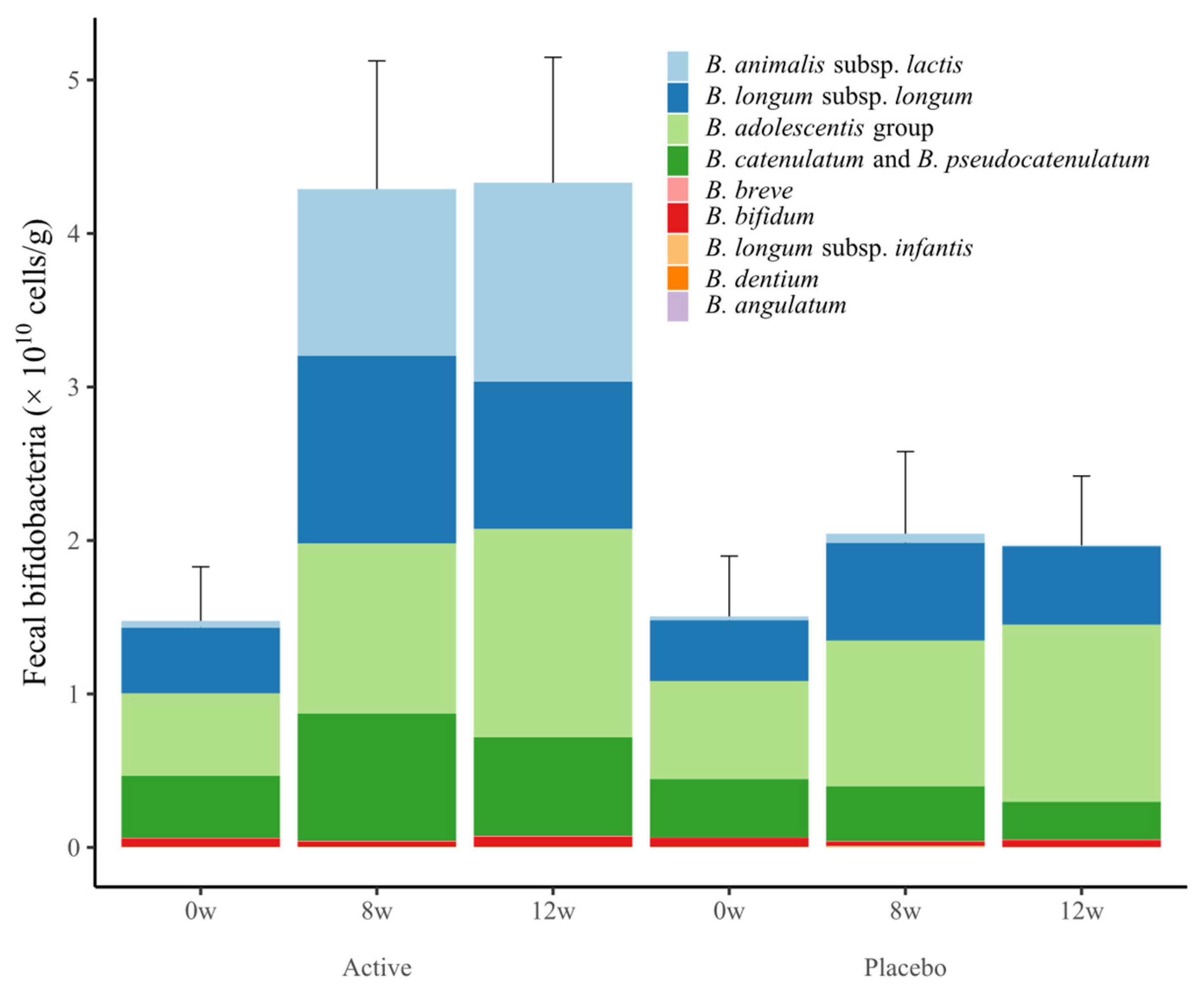
3.5. Fecal Bifidobacteria
3.6. Regression Analysis with Fecal Bifidobacteria and Measured Parameters
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 19 October 2023).
- Shuster, A.; Patlas, M.; Pinthus, J.H.; Mourtzakis, M. The clinical importance of visceral adiposity: A critical review of methods for visceral adipose tissue analysis. Br. J. Radiol. 2012, 85, 1-e25. [Google Scholar] [CrossRef] [PubMed]
- Green, M.; Arora, K.; Prakash, S. Microbial medicine: Prebiotic and probiotic functional foods to target obesity and metabolic syndrome. Int. J. Mol. Sci. 2020, 21, 2890. [Google Scholar] [CrossRef] [PubMed]
- Pedroza Matute, S.; Iyavoo, S. Exploring the Gut Microbiota: Lifestyle Choices, Disease Associations, and Personal Genomics. Front. Nutr. 2023, 10, 1225120. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. The International Scientific Association for Probiotics and Prebiotics consensus statement on the Scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Kadooka, Y.; Sato, M.; Imaizumi, K.; Ogawa, A.; Ikuyama, K.; Akai, Y.; Okano, M.; Kagoshima, M.; Tsuchida, T. Regulation of abdominal adiposity by probiotics (Lactobacillus gasseri SBT2055) in adults with obese tendencies in a randomized controlled trial. Eur. J. Clin. Nutr. 2010, 64, 636–643. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, A.; Kadooka, Y.; Kato, K.; Shirouchi, B.; Sato, M. Lactobacillus gasseri SBT2055 reduces postprandial and fasting serum non-esterified fatty acid levels in Japanese hypertriacylglycerolemic subjects. Lipids Health Dis. 2014, 13, 36. [Google Scholar] [CrossRef]
- Kim, J.; Yun, J.M.; Kim, M.K.; Kwon, O.; Cho, B. Lactobacillus gasseri BNR17 supplementation reduces the visceral fat accumulation and waist circumference in obese adults: A randomized, double-blind, placebo-controlled trial. J. Med. Food 2018, 21, 454–461. [Google Scholar] [CrossRef]
- Rahayu, E.S.; Mariyatun, M.; Putri Manurung, N.E.; Hasan, P.N.; Therdtatha, P.; Mishima, R.; Komalasari, H.; Mahfuzah, N.A.; Pamungkaningtyas, F.H.; Yoga, W.K.; et al. Effect of probiotic Lactobacillus plantarum Dad-13 powder consumption on the gut microbiota and intestinal health of overweight adults. World J. Gastroenterol. 2021, 27, 107–128. [Google Scholar] [CrossRef]
- Sohn, M.; Na, G.Y.; Chu, J.; Joung, H.; Kim, B.-K.; Lim, S. Efficacy and safety of Lactobacillus plantarum K50 on lipids in Koreans with obesity: A randomized, double-blind controlled clinical trial. Front. Endocrinol. 2021, 12, 790046. [Google Scholar] [CrossRef]
- Sohn, M.; Jung, H.; Lee, W.S.; Kim, T.H.; Lim, S. Effect of Lactobacillus plantarum LMT1-48 on body fat in overweight subjects: A randomized, double-blind, placebo-controlled trial. Diabetes Metab. J. 2023, 47, 92–103. [Google Scholar] [CrossRef]
- Minami, J.; Iwabuchi, N.; Tanaka, M.; Yamauchi, K.; Xiao, J.-Z.; Abe, F.; Sakane, N. Effects of Bifidobacterium breve B-3 on body fat reductions in pre-obese adults: A randomized, double-blind, placebo-controlled trial. Biosci. Microbiota Food Health 2018, 37, 67–75. [Google Scholar] [CrossRef]
- Pedret, A.; Valls, R.M.; Calderón-Pérez, L.; Llauradó, E.; Companys, J.; Pla-Pagà, L.; Moragas, A.; Martín-Luján, F.; Ortega, Y.; Giralt, M.; et al. Effects of daily consumption of the probiotic Bifidobacterium animalis subsp. lactis CECT 8145 on anthropometric adiposity biomarkers in abdominally obese subjects: A randomized controlled trial. Int. J. Obes. 2019, 43, 1863–1868. [Google Scholar] [CrossRef]
- Takahashi, S.; Anzawa, D.; Takami, K.; Ishizuka, A.; Mawatari, T.; Kamikado, K.; Sugimura, H.; Nishijima, T. Effect of Bifidobacterium animalis ssp. lactis GCL2505 on visceral fat accumulation in healthy Japanese adults: A randomized controlled trial. Biosci. Microbiota Food Health 2016, 35, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Buys, N. Effects of probiotics consumption on lowering lipids and CVD risk factors: A systematic review and meta-analysis of randomized controlled trials. Ann. Med. 2015, 47, 430–440. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Wu, Y.; Fei, X. Effect of probiotics on body weight and body-mass index: A systematic review and meta-analysis of randomized, controlled trials. Int. J. Food Sci. Nutr. 2015, 67, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Koutnikova, H.; Genser, B.; Monteiro-Sepulveda, M.; Faurie, J.-M.; Rizkalla, S.; Schrezenmeir, J.; Clément, K. Impact of bacterial probiotics on obesity, diabetes and mon-alcoholic fatty liver disease related variables: A systematic review and meta-analysis of randomised controlled trials. BMJ Open 2019, 9, e017995. [Google Scholar] [CrossRef] [PubMed]
- Pineiro, M.; Asp, N.-G.; Reid, G.; Macfarlane, S.; Morelli, L.; Brunser, O.; Tuohy, K. FAO Technical Meeting on Prebiotics. J. Clin. Gastroenterol. 2008, 42 Pt 2 (Suppl. S3), S156–S159. [Google Scholar] [CrossRef]
- Beserra, B.T.S.; Fernandes, R.; do Rosario, V.A.; Mocellin, M.C.; Kuntz, M.G.F.; Trindade, E.B.S.M. A systematic review and meta-analysis of the prebiotics and synbiotics effects on glycaemia, insulin concentrations and lipid parameters in adult patients with overweight or obesity. Clin. Nutr. 2015, 34, 845–858. [Google Scholar] [CrossRef]
- Thompson, S.V.; Hannon, B.A.; An, R.; Holscher, H.D. Effects of isolated soluble fiber supplementation on body weight, glycemia, and insulinemia in adults with overweight and obesity: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2017, 106, 1514–1528. [Google Scholar] [CrossRef] [PubMed]
- Huwiler, V.V.; Schönenberger, K.A.; Segesser von Brunegg, A.; Reber, E.; Mühlebach, S.; Stanga, Z.; Balmer, M.L. Prolonged isolated soluble dietary fibre supplementation in overweight and obese patients: A systematic review with meta-analysis of randomised controlled trials. Nutrients 2022, 14, 2627. [Google Scholar] [CrossRef]
- Larsen, I.S.; Choi, B.S.-Y.; Föh, B.; Kristensen, N.N.; Ouellette, A.; Haller, R.F.; Olsen, P.B.; Saulnier, D.; Sina, C.; Jensen, B.A.H.; et al. Experimental diets dictate the metabolic benefits of probiotics in obesity. Gut Microbes 2023, 15, 2192547. [Google Scholar] [CrossRef]
- Wastyk, H.C.; Perelman, D.; Topf, M.; Fragiadakis, G.K.; Robinson, J.L.; Sonnenburg, J.L.; Gardner, C.D.; Sonnenburg, E.D. Randomized controlled trial demonstrates response to a probiotic intervention for metabolic syndrome that may correspond to diet. Gut Microbes 2023, 15, 2178794. [Google Scholar] [CrossRef]
- Ojima, M.N.; Yoshida, K.; Sakanaka, M.; Jiang, L.; Odamaki, T.; Katayama, T. Ecological and molecular perspectives on responders and non-responders to probiotics and prebiotics. Curr. Opin. Biotechnol. 2022, 73, 108–120. [Google Scholar] [CrossRef]
- Kovatcheva-Datchary, P.; Nilsson, A.; Akrami, R.; Lee, Y.S.; De Vadder, F.; Arora, T.; Hallen, A.; Martens, E.; Björck, I.; Bäckhed, F. Dietary fiber-induced improvement in glucose metabolism is associated with increased abundance of prevotella. Cell Metab. 2015, 22, 971–982. [Google Scholar] [CrossRef] [PubMed]
- Swanson, K.S.; Gibson, G.R.; Hutkins, R.; Reimer, R.A.; Reid, G.; Verbeke, K.; Scott, K.P.; Holscher, H.D.; Azad, M.B.; Delzenne, N.M.; et al. The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of synbiotics. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 687–701. [Google Scholar] [CrossRef] [PubMed]
- Childs, C.E.; Röytiö, H.; Alhoniemi, E.; Fekete, A.A.; Forssten, S.D.; Hudjec, N.; Lim, Y.N.; Steger, C.J.; Yaqoob, P.; Tuohy, K.M.; et al. Xylo-oligosaccharides alone or in synbiotic combination with Bifidobacterium animalis subsp. lactis induce bifidogenesis and modulate markers of immune function in healthy adults: A double-blind, placebo-controlled, randomised, factorial cross-over study. Br. J. Nutr. 2014, 111, 1945–1956. [Google Scholar] [CrossRef] [PubMed]
- Krumbeck, J.A.; Rasmussen, H.E.; Hutkins, R.W.; Clarke, J.; Shawron, K.; Keshavarzian, A.; Walter, J. Probiotic Bifidobacterium strains and galactooligosaccharides improve intestinal barrier function in obese adults but show no synergism when used together as synbiotics. Microbiome 2018, 6, 121. [Google Scholar] [CrossRef] [PubMed]
- Anzawa, D.; Mawatari, T.; Tanaka, Y.; Yamamoto, M.; Genda, T.; Takahashi, S.; Nishijima, T.; Kamasaka, H.; Suzuki, S.; Kuriki, T. Effects of synbiotics containing Bifidobacterium animalis subsp. lactis GCL2505 and inulin on intestinal bifidobacteria: A randomized, placebo-controlled, crossover study. Food Sci. Nutr. 2019, 7, 1828–1837. [Google Scholar] [CrossRef] [PubMed]
- Oh, Y.J.; Kim, H.J.; Kim, T.S.; Yeo, I.H.; Ji, G.E. Effects of Lactobacillus plantarum PMO 08 alone and combined with chia seeds on metabolic syndrome and parameters related to gut health in high-fat diet-induced obese mice. J. Med. Food 2019, 22, 1199–1207. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, M.; Liu, H.; Wei, X.; Su, X.; Li, M.; Yuan, J. Oral supplements of combined Bacillus licheniformis Zhengchangsheng® and xylooligosaccharides improve high-fat diet-induced obesity and modulate the gut microbiota in rats. Biomed. Res. Int. 2020, 2020, 9067821. [Google Scholar] [CrossRef]
- Rogha, M.; Esfahani, M.Z.; Zargarzadeh, A.H. The efficacy of a synbiotic containing Bacillus coagulans in treatment of irritable bowel syndrome: A randomized placebo-controlled trial. Gastroenterol. Hepatol. Bed Bench 2014, 7, 156–163. [Google Scholar]
- Skrzydło-Radomańska, B.; Prozorow-Król, B.; Cichoż-Lach, H.; Majsiak, E.; Bierła, J.B.; Kosikowski, W.; Szczerbiński, M.; Gantzel, J.; Cukrowska, B. The effectiveness of synbiotic preparation containing Lactobacillus and Bifidobacterium probiotic strains and short chain fructooligosaccharides in patients with diarrhea predominant irritable bowel syndrome-a randomized double-blind, placebo-controlled study. Nutrients 2020, 12, 1999. [Google Scholar]
- Ipar, N.; Aydogdu, S.D.; Yildirim, G.K.; Inal, M.; Gies, I.; Vandenplas, Y.; Dinleyici, E.C. Effects of synbiotic on anthropometry, lipid profile and oxidative stress in obese children. Benef. Microbes 2015, 6, 775–782. [Google Scholar] [CrossRef]
- Mohammadi-Sartang, M.; Bellissimo, N.; Totosy de Zepetnek, J.O.; Brett, N.R.; Mazloomi, S.M.; Fararouie, M.; Bedeltavana, A.; Famouri, M.; Mazloom, Z. The effect of daily fortified yogurt consumption on weight loss in adults with metabolic syndrome: A 10-week randomized controlled trial. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 565–574. [Google Scholar] [CrossRef]
- Eslamparast, T.; Poustchi, H.; Zamani, F.; Sharafkhah, M.; Malekzadeh, R.; Hekmatdoost, A. Synbiotic supplementation in nonalcoholic fatty liver disease: A randomized, double-blind, placebo-controlled pilot study. Am. J. Clin. Nutr. 2014, 99, 535–542. [Google Scholar] [CrossRef] [PubMed]
- Lalitsuradej, E.; Sirilun, S.; Sittiprapaporn, P.; Sivamaruthi, B.S.; Pintha, K.; Tantipaiboonwong, P.; Khongtan, S.; Fukngoen, P.; Peerajan, S.; Chaiyasut, C. The effects of synbiotics administration on stress-related parameters in Thai subjects-a preliminary study. Foods 2022, 11, 759. [Google Scholar] [CrossRef] [PubMed]
- Valle, M.C.P.R.; Vieira, I.A.; Fino, L.C.; Gallina, D.A.; Esteves, A.M.; da Cunha, D.T.; Cabral, L.; Benatti, F.B.; Marostica Junior, M.R.; Batista, Â.G.; et al. Immune status, well-being and gut microbiota in military supplemented with synbiotic ice cream and submitted to field training: A randomised clinical trial. Br. J. Nutr. 2021, 126, 1794–1808. [Google Scholar] [CrossRef] [PubMed]
- Chiu, Y.-H.; Lin, S.-L.; Tsai, J.-J.; Lin, M.-Y. Probiotic actions on diseases: Implications for therapeutic treatments. Food Funct. 2014, 5, 625–634. [Google Scholar] [CrossRef] [PubMed]
- Taipale, T.J.; Pienihäkkinen, K.; Isolauri, E.; Jokela, J.T.; Söderling, E.M. Bifidobacterium animalis subsp. lactis BB-12 in reducing the risk of infections in early childhood. Pediatr. Res. 2016, 79, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Ishizuka, A.; Tomizuka, K.; Aoki, R.; Nishijima, T.; Saito, Y.; Inoue, R.; Ushida, K.; Mawatari, T.; Ikeda, T. Effects of administration of Bifidobacterium animalis subsp. lactis GCL2505 on defecation frequency and bifidobacterial microbiota composition in humans. J. Biosci. Bioeng. 2012, 113, 587–591. [Google Scholar] [CrossRef]
- Aoki, R.; Tsuchida, S.; Arai, Y.; Ohno, K.; Nishijima, T.; Mawatari, T.; Mikami, Y.; Ushida, K. Effect of Bifidobacterium animalis subsp. lactis GCL2505 on the physiological function of intestine in a rat model. Food Sci. Nutr. 2016, 4, 782–790. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.; Moon, J.H.; Shin, C.M.; Jeong, D.; Kim, B. Effect of Lactobacillus sakei, a probiotic derived from kimchi, on body fat in Koreans with obesity: A randomized controlled study. Endocrinol. Metab. 2020, 35, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.G.; Yang, Y.J.; Yoon, Y.S.; Lee, E.S.; Lee, J.H.; Jeong, Y.; Kang, C.H. Effect of MED-02 containing two probiotic strains, Limosilactobacillus fermentum MG4231 and MG4244, on body fat reduction in overweight or obese subjects: A randomized, multicenter, double-blind, placebo-controlled study. Nutrients 2022, 14, 3583. [Google Scholar] [CrossRef]
- Ryo, M.; Kishida, K.; Nakamura, T.; Yoshizumi, T.; Funahashi, T.; Shimomura, I. Clinical significance of visceral adiposity assessed by computed tomography: A Japanese perspective. World J. Radiol. 2014, 6, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Tourlousse, D.M.; Narita, K.; Miura, T.; Sakamoto, M.; Ohashi, A.; Shiina, K.; Matsuda, M.; Miura, D.; Shimamura, M.; Ohyama, Y.; et al. Validation and standardization of DNA extraction and library construction methods for metagenomics-based human fecal microbiome measurements. Microbiome 2021, 9, 95. [Google Scholar] [CrossRef]
- Tanaka, Y.; Takami, K.; Nishijima, T.; Aoki, R.; Mawatari, T.; Ikeda, T. Short- and long-term dynamics in the intestinal microbiota following ingestion of Bifidobacterium animalis subsp. lactis GCL2505. Biosci. Microbiota Food Health 2015, 34, 77–85. [Google Scholar] [CrossRef]
- Ministry of Health, Labour and Welfare. Dietary Reference Intakes for Japanese (2020); Ministry of Health, Labour and Welfare: Tokyo, Japan, 2019; Available online: https://www.mhlw.go.jp/content/001151422.pdf (accessed on 26 October 2023).
- Ibrahim, M.M. Subcutaneous and visceral adipose tissue: Structural and functional differences. Obes. Rev. 2010, 11, 11–18. [Google Scholar] [CrossRef]
- Chaston, T.B.; Dixon, J.B. Factors associated with percent change in visceral versus subcutaneous abdominal fat during weight loss: Findings from a systematic review. Int. J. Obes. 2008, 32, 619–628. [Google Scholar] [CrossRef]
- Musazadeh, V.; Mohammadi Anilou, M.; Vajdi, M.; Karimi, A.; Sedgh Ahrabi, S.; Dehghan, P. Effects of synbiotics supplementation on anthropometric and lipid profile parameters: Finding from an umbrella meta-analysis. Front. Nutr. 2023, 10, 1121541. [Google Scholar] [CrossRef]
- Aoki, R.; Kamikado, K.; Suda, W.; Takii, H.; Mikami, Y.; Suganuma, N.; Hattori, M.; Koga, Y. A proliferative probiotic Bifidobacterium strain in the gut ameliorates progression of metabolic disorders via microbiota modulation and acetate elevation. Sci. Rep. 2017, 7, 43522. [Google Scholar] [CrossRef]
- Horiuchi, H.; Kamikado, K.; Aoki, R.; Suganuma, N.; Nishijima, T.; Nakatani, A.; Kimura, I. Bifidobacterium animalis subsp. lactis GCL2505 modulates host energy metabolism via the short-chain fatty acid receptor GPR43. Sci. Rep. 2020, 10, 4158. [Google Scholar] [CrossRef] [PubMed]
- Birkeland, E.; Gharagozlian, S.; Birkeland, K.I.; Valeur, J.; Måge, I.; Rud, I.; Aas, A.-M. Prebiotic effect of inulin-type fructans on faecal microbiota and short-chain fatty acids in type 2 diabetes: A randomised controlled Trial. Eur. J. Nutr. 2020, 59, 3325–3338. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, H.; Nakanishi, N.; Miyoshi, T.; Okamura, T.; Hashimoto, Y.; Senmaru, T.; Majima, S.; Ushigome, E.; Asano, M.; Yamaguchi, M.; et al. Inulin reduces visceral adipose tissue mass and improves glucose tolerance through altering gut metabolites. Nutr. Metab. 2022, 19, 50. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Shi, B. Gut microbiota as a potential target of metabolic syndrome: The role of probiotics and prebiotics. Cell Biosci. 2017, 7, 54. [Google Scholar] [CrossRef]
- Ogawa, A.; Kobayashi, T.; Sakai, F.; Kadooka, Y.; Kawasaki, Y. Lactobacillus gasseri SBT2055 suppresses fatty acid release through enlargement of fat emulsion size in vitro and promotes fecal fat excretion in healthy Japanese subjects. Lipids Health Dis. 2015, 14, 20. [Google Scholar] [CrossRef]
- Azuma, N.; Mawatari, T.; Saito, Y.; Tsukamoto, M.; Sampei, M.; Iwama, Y. Effect of continuous ingestion of bifidobacteria and dietary fiber on improvement in cognitive function: A randomized, double-blind, placebo-controlled trial. Nutrients 2023, 15, 4175. [Google Scholar] [CrossRef]
- Azuma, N.; Saito, Y.; Nishijima, T.; Aoki, R.; Nishihira, J. Effect of daily ingestion of Bifidobacterium and dietary fiber on vascular endothelial function: A randomized, double-blind, placebo-controlled, parallel-group comparison study. Biosci. Biotechnol. Biochem. 2023, zbad148. [Google Scholar] [CrossRef]
| Parameter | Placebo Group | Active Group |
|---|---|---|
| Energy, kcal/100 g | 47.0 | 52.0 |
| Moisture, g/100 g | 87.0 | 84.8 |
| Protein, g/100 g | 2.8 | 2.8 |
| Fat, g/100 g | 0.1 | 0.1 |
| Carbohydrate, g/100 g | 9.0 | 11.2 |
| Ash, g/100 g | 1.1 | 1.1 |
| Characteristic | Placebo Group | Active Group | p-Value | |
|---|---|---|---|---|
| Clinical findings | Age, years | 50.6 (8.0) | 50.6 (8.4) | 0.973 |
| Height, cm | 165.9 (8.2) | 167.8 (8.5) | 0.221 | |
| Body weight, kg | 73.6 (7.9) | 73.8 (8.2) | 0.873 | |
| Body mass index, kg/m2 | 26.7 (1.5) | 26.2 (2.0) | 0.129 | |
| Waist circumference, cm | 92.5 (5.1) | 92.4 (6.2) | 0.902 | |
| Hip circumference, cm | 98.0 (3.6) | 98.2 (4.5) | 0.840 | |
| Waist-to-hip ratio | 0.9 (0.04) | 0.9 (0.04) | 0.720 | |
| Visceral fat area, cm2 | 124.9 (31.0) | 124.0 (31.3) | 0.864 | |
| Subcutaneous fat area, cm2 | 206.0 (58.1) | 207.7 (65.2) | 0.881 | |
| Total fat area, cm2 | 330.9 (61.6) | 331.7 (69.8) | 0.953 | |
| Systolic blood pressure, mmHg | 127.7 (11.2) | 132.4 (15.1) | 0.055 | |
| Diastolic blood pressure, mmHg | 79.0 (10.6) | 82.3 (11.7) | 0.108 | |
| Heartbeat, bpm | 77.5 (10.5) | 78.4 (10.8) | 0.644 | |
| Laboratory findings | White blood cell count, /µL | 6185.0 (1449) | 6076.7 (1077) | 0.643 |
| Red blood cell count, ×104/µL | 495.8 (45.4) | 489.0 (43.8) | 0.405 | |
| Hemoglobin, g/dL | 15.0 (1.3) | 14.7 (1.2) | 0.214 | |
| Hematocrit, % | 47.2 (3.6) | 46.6 (3.2) | 0.351 | |
| Platelet count, ×104/μL | 26.7 (4.5) | 28.0 (6.0) | 0.164 | |
| Neutrophil ratio, % | 58.3 (7.2) | 57.8 (5.9) | 0.675 | |
| Lymphocyte ratio, % | 32.2 (6.8) | 32.6 (5.7) | 0.678 | |
| Monocyte ratio, % | 5.6 (1.3) | 5.3 (1.0) | 0.193 | |
| Eosinophil ratio, % | 3.2 (2.0) | 3.4 (2.7) | 0.652 | |
| Basophil ratio, % | 0.7 (0.3) | 0.9 (0.3) | 0.047 | |
| Total serum protein, g/dL | 7.2 (0.4) | 7.3 (0.3) | 0.237 | |
| Albumin, g/dL | 4.4 (0.3) | 4.5 (0.3) | 0.120 | |
| Aspartate aminotransferase, U/L | 24.8 (10.8) | 23.5 (6.7) | 0.430 | |
| Alanine aminotransferase, U/L | 30.4 (21.2) | 26.6 (17.1) | 0.280 | |
| Lactate dehydrogenase, U/L | 183.0 (29.6) | 188.9 (32.7) | 0.308 | |
| Total bilirubin, mg/dL | 0.8 (0.2) | 0.9 (0.3) | 0.228 | |
| Alkaline phosphatase, U/L | 75.7 (19.9) | 76.8 (17.3) | 0.758 | |
| Laboratory findings | γ-Glutamyl transpeptidase, U/L | 40.4 (31.3) | 44.7 (36.6) | 0.485 |
| Blood urea nitrogen, mg/dL | 13.3 (2.8) | 13.8 (3.2) | 0.356 | |
| Creatinine, mg/dL | 0.8 (0.15) | 0.8 (0.16) | 0.894 | |
| Uric acid, mg/dL | 5.8 (1.3) | 5.8 (1.3) | 0.746 | |
| Sodium (Na), mEq/L | 141.3 (1.7) | 141.4 (1.4) | 0.726 | |
| Chlorine (Cl), mEq/L | 104.1 (2.2) | 103.9 (1.7) | 0.645 | |
| Potassium (K), mEq/L | 4.2 (0.3) | 4.2 (0.2) | 0.145 | |
| Calcium (Ca), mg/dL | 9.5 (0.3) | 9.5 (0.3) | 0.294 | |
| Total cholesterol, mg/dL | 218.0 (30.2) | 217.8 (31.9) | 0.967 | |
| LDL cholesterol, mg/dL | 140.0 (27.5) | 137.9 (30.0) | 0.694 | |
| HDL cholesterol, mg/dL | 55.3 (12.4) | 58.6 (14.7) | 0.185 | |
| Triglycerides, mg/dL | 131.6 (65.8) | 121.4 (49.7) | 0.338 | |
| Glucose, mg/dL | 89.8 (9.2) | 89.1 (9.6) | 0.677 | |
| HbA1c (NGSP), % | 5.5 (0.3) | 5.5 (0.3) | 0.926 | |
| Free fatty acid, mEq/L | 0.4 (0.2) | 0.5 (0.2) | 0.077 | |
| Urine pH | 6.2 (0.5) | 6.2 (0.6) | 0.560 | |
| Urine specific gravity | 1.0 (0.007) | 1.0 (0.007) | 0.491 | |
| Compliance rate of the test sample, % * | 99.80 (0.50) | 99.88(0.42) | 0.349 |
| Parameter | 0 Weeks | 4 Weeks | 8 Weeks | 12 Weeks | |
|---|---|---|---|---|---|
| Energy, kcal | Active | 1820.9 (385.4) | 1816.5 (428.4) | 1792.3 (399.5) | 1704.8 (413.2) * |
| Placebo | 1925.3 (360.3) | 1929.8 (411.4) | 1880.7 (414.8) | 1820.1 (366.6) * | |
| Protein, g | Active | 68.7 (15.7) | 67.3 (17.8) | 66.0 (16.2) | 64.9 (17.5) * |
| Placebo | 72.5 (15.3) | 72.1 (15.4) | 69.7 (19.0) | 68.6 (14.9) * | |
| Fat, g | Active | 63.8 (17.9) | 61.4 (19.2) | 60.7 (20.0) | 55.9 (19.1) *# |
| Placebo | 67.9 (20.0) | 66.9 (20.4) | 65.3 (20.6) | 64.1 (18.7) | |
| Carbohydrate, g | Active | 229.7 (53.0) | 236.2 (57.4) | 233.3 (52.8) | 222.6 (54.8) |
| Placebo | 243.3 (56.2) | 247.1 (59.4) | 240.4 (53.5) | 230.2 (50.6) * | |
| Dietary fiber, g | Active | 10.8 (3.3) | 10.7 (3.1) | 10.6 (3.1) | 10.1 (3.2) |
| Placebo | 10.9 (2.9) | 11.0 (3.0) | 10.4 (3.0) | 9.9 (2.7) * | |
| Parameter | 0 Weeks | 8 Weeks | 12 Weeks | Time × Group † | |
|---|---|---|---|---|---|
| Visceral fat area, cm2 | Active | 124.0 (4.1) | 111.1 (4.2) * | 106.8 (3.6) * | <0.0001 |
| Placebo | 119.8 (3.9) | 117.5 (4.9) | 114.9 (4.0) | ||
| Subcutaneous fat area, cm2 | Active | 206.9 (8.3) | 207.2 (8.3) | 206.7 (8.6) | 0.379 |
| Placebo | 211.9 (8.8) | 212.2 (8.6) | 215.8 (9.4) | ||
| Total fat area, cm2 | Active | 331.0 (9.3) | 318.4 (9.9) * | 313.5 (9.5) * | 0.001 |
| Placebo | 331.7 (9.1) | 329.7 (10.2) | 330.7 (10.3) | ||
| Parameter | 0 Weeks | 4 Weeks | 8 Weeks | 12 Weeks | |
|---|---|---|---|---|---|
| Body weight, kg | Active | 74.9 (1.1) | 74.8 (1.1) | 74.7 (1.1) | 74.6 (1.1) * |
| Placebo | 74.8 (1.1) | 74.8 (1.1) | 74.6 (1.1) | 74.3 (1.1) | |
| Body mass index, kg/m2 | Active | 26.5 (0.3) | 26.4 (0.3) | 26.4 (0.3) | 26.3 (0.3) * |
| Placebo | 27.0 (0.2) | 27.0 (0.2) | 26.9 (0.2) | 26.9 (0.2) | |
| Waist-to-hip ratio | Active | 0.9 (0.005) | 0.9 (0.005) | 0.9 (0.005) | 0.9 (0.005) |
| Placebo | 0.9 (0.006) | 0.9 (0.005) | 0.9 (0.005) | 0.9 (0.005) | |
| Parameter | 0 Weeks | 4 Weeks | 8 Weeks | 12 Weeks | |
|---|---|---|---|---|---|
| Total cholesterol, mg/dL | Active | 219.4 (4.2) | 217.6 (4.1) | 211.6 (4.2) * | 217.5 (4.6) |
| Placebo | 219.2 (4.2) | 219.0 (3.7) | 216.2 (4.2) | 219.4 (4.3) | |
| LDL cholesterol, mg/dL | Active | 139.9 (4.2) | 136.3 (3.8) | 135.3 (3.9) * | 136.5 (4.3) |
| Placebo | 136.3 (3.8) | 139.0 (3.4) | 138.7 (3.7) | 138.1 (4.0) | |
| HDL cholesterol, mg/dL | Active | 57.9 (2.1) | 59.0 (1.9) | 55.8 (1.7) * | 56.2 (1.7) |
| Placebo | 55.5 (1.7) | 54.6 (1.8) | 54.2 (1.9) | 54.5 (1.8) | |
| Triglycerides, mg/dL | Active | 113.6 (7.0) | 116.2 (6.3) # | 113.6 (5.4) # | 118.3 (8.8) |
| Placebo | 130.2 (8.3) | 141.7 (11.1) | 138.5 (10.1) | 143.1 (15.8) | |
| Free fatty acid, mEq/L | Active | 0.53 (0.03) | 0.50 (0.02) | 0.53 (0.02) | 0.61 (0.03) * |
| Placebo | 0.48 (0.03) | 0.50 (0.02) | 0.48 (0.02) | 0.55 (0.03) * | |
| Bifidobacteria | 0 Weeks | 8 Weeks | 12 Weeks | |
|---|---|---|---|---|
| Total bifidobacteria | Active | 9.62 (0.14) | 10.35 (0.08) *# | 10.41 (0.07) *# |
| Placebo | 9.48 (0.17) | 9.60 (0.17) | 9.61 (0.17) | |
| B. animalis subsp. lactis | Active | 5.70 (0.15) | 9.67 (0.09) *# | 9.77 (0.08) *# |
| Placebo | 5.70 (0.14) | 5.72 (0.15) | 5.50 (0.10) | |
| B. longum subsp. longum | Active | 8.62 (0.20) | 8.86 (0.23) * | 8.96 (0.23) * |
| Placebo | 8.26 (0.23) | 8.35 (0.24) | 8.37 (0.23) | |
| B. adolescentis group | Active | 7.81 (0.28) | 8.14 (0.31) * | 8.18 (0.31) * |
| Placebo | 7.73 (0.29) | 8.09 (0.29) | 8.19 (0.30) | |
| B. catenulatum and B. pseudocatenulatum | Active | 8.22 (0.25) | 8.40 (0.27) *# | 8.37 (0.27) *# |
| Placebo | 7.89 (0.26) | 7.87 (0.26) | 7.90 (0.25) | |
| B. breve | Active | 5.66 (0.11) | 5.77 (0.12) | 5.74 (0.12) |
| Placebo | 5.62 (0.11) | 5.62 (0.11) | 5.68 (0.12) | |
| B. bifidum | Active | 6.02 (0.20) | 6.08 (0.20) | 6.14 (0.21) |
| Placebo | 5.89 (0.18) | 5.98 (0.19) | 5.98 (0.19) | |
| B. longum subsp. infantis | Active | 5.53 (0.09) | 5.59 (0.11) | 5.57 (0.10) |
| Placebo | 5.51 (0.09) | 5.51 (0.10) | 5.52 (0.09) | |
| B. dentium | Active | 5.64 (0.11) | 5.66 (0.10) | 5.63 (0.10) |
| Placebo | 5.63 (0.09) | 5.71 (0.10) | 5.64 (0.09) | |
| B. angulatum | Active | n.d. | n.d. | n.d. |
| Placebo | n.d. | n.d. | n.d. | |
| Endogenous bifidobacteria | Active | 9.60 (0.14) | 9.89 (0.16) * | 9.92 (0.16) * |
| Placebo | 9.45 (0.17) | 9.57 (0.17) | 9.61 (0.17) | |
| Object Variable | Ratio of Change from Week 0 to 12 | Explanatory Variable | Change from Week 0 to 12, % | p-Value |
|---|---|---|---|---|
| Total bifidobacteria | 33.46 (12.51) | BMI | −0.5 (0.2) | 0.010 |
| VFA | −7.2 (1.3) | 0.012 | ||
| Endogenous bifidobacteria | 27,875.92 (5977.64) | BMI | −0.5 (0.2) | 0.025 |
| B. animalis subsp. lactis | 8.07 (2.63) | BMI | −0.5 (0.2) | 0.025 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baba, Y.; Saito, Y.; Kadowaki, M.; Azuma, N.; Tsuge, D. Effect of Continuous Ingestion of Bifidobacteria and Inulin on Reducing Body Fat: A Randomized, Double-Blind, Placebo-Controlled, Parallel-Group Comparison Study. Nutrients 2023, 15, 5025. https://doi.org/10.3390/nu15245025
Baba Y, Saito Y, Kadowaki M, Azuma N, Tsuge D. Effect of Continuous Ingestion of Bifidobacteria and Inulin on Reducing Body Fat: A Randomized, Double-Blind, Placebo-Controlled, Parallel-Group Comparison Study. Nutrients. 2023; 15(24):5025. https://doi.org/10.3390/nu15245025
Chicago/Turabian StyleBaba, Yuhei, Yasuo Saito, Mei Kadowaki, Naoki Azuma, and Daisuke Tsuge. 2023. "Effect of Continuous Ingestion of Bifidobacteria and Inulin on Reducing Body Fat: A Randomized, Double-Blind, Placebo-Controlled, Parallel-Group Comparison Study" Nutrients 15, no. 24: 5025. https://doi.org/10.3390/nu15245025
APA StyleBaba, Y., Saito, Y., Kadowaki, M., Azuma, N., & Tsuge, D. (2023). Effect of Continuous Ingestion of Bifidobacteria and Inulin on Reducing Body Fat: A Randomized, Double-Blind, Placebo-Controlled, Parallel-Group Comparison Study. Nutrients, 15(24), 5025. https://doi.org/10.3390/nu15245025





