Effects of Isoflavonoid and Vitamin D Synergism on Bone Mineral Density—A Systematic and Critical Review
Abstract
:1. Introduction
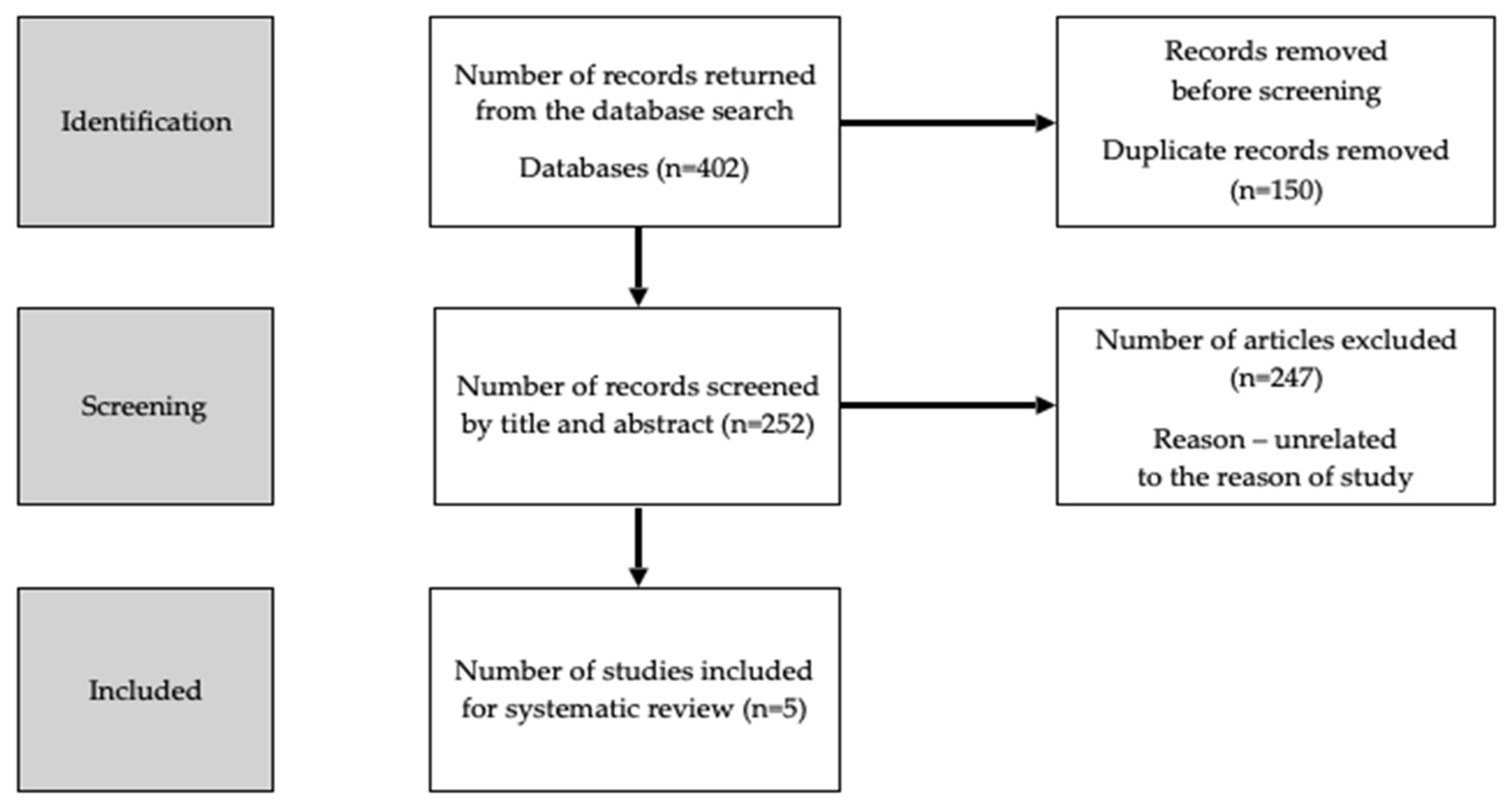
2. Materials and Methods
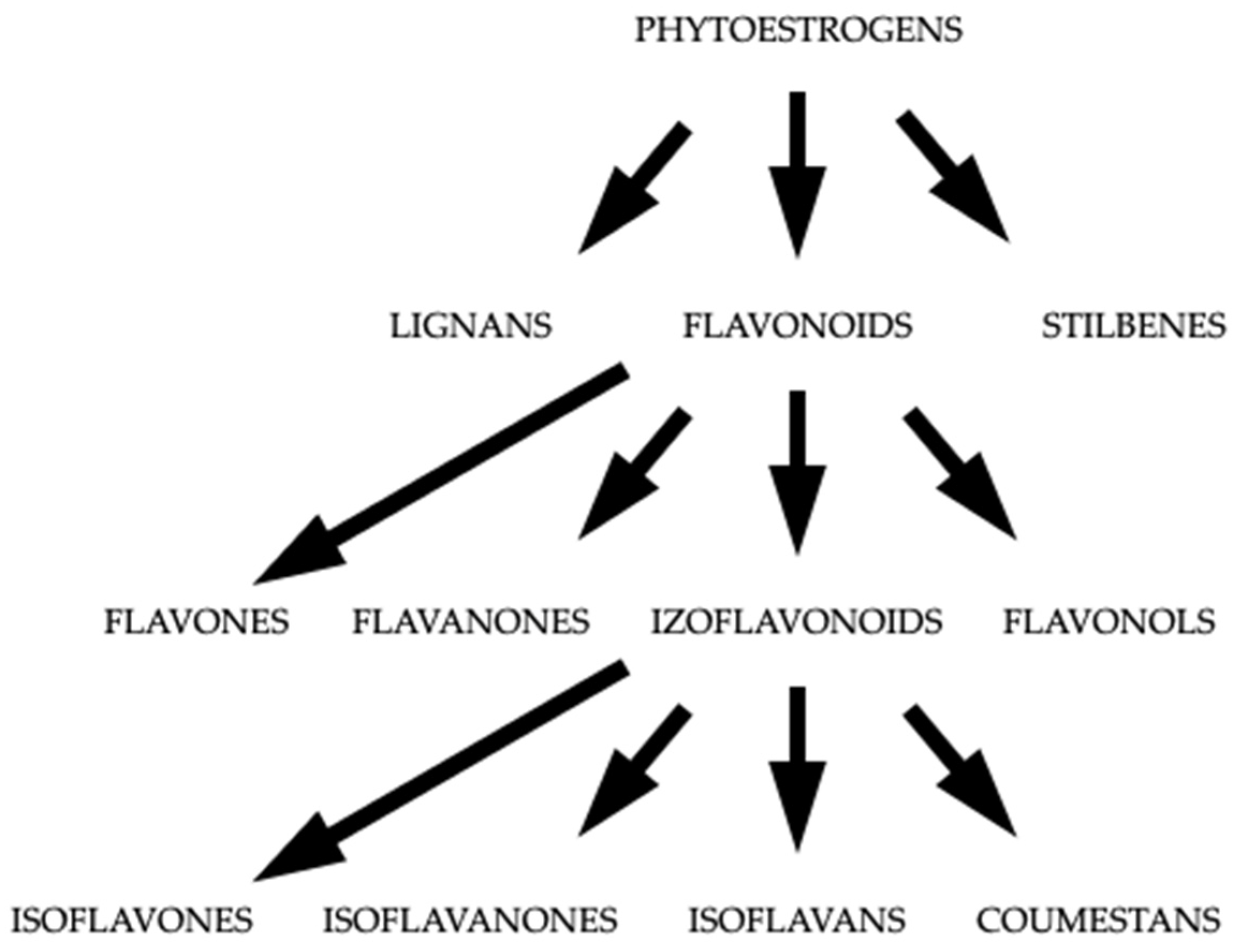
3. Isoflavonoids
3.1. Impact of Isoflavonoids on Bone Mineral Density
| Author | Year | Country | Studied Group | Patients/Controls | Intervention | Time | BMD/Fracture Risk (+) Reduction Bone Loss/Reduction Fracture Risk (−) No Reduction Bone Loss/No Reduction Fracture Risk | Other Parameters | Type of the Study |
|---|---|---|---|---|---|---|---|---|---|
| Agnusdei et al. [39] | 1997 | Italy | Postmenopausal women (65–79 y) | 41/43 | 200 mg ipriflavone Control group: 1 g calcium | 24 months | Forearm BMD (+) | Not studied | Randomized, double-blind, placebo-controlled |
| Khalil et al. [34] | 2002 | USA | Healthy men (59.2 ± 17.6 y) | 24/22 | 88 mg/day of isoflavones | 3 months | Not studied | Bone turnover markers, IGF-1 | Randomized, double-blind, controlled |
| Chen et al. [40] | 2003 | China | Postmenopausal women (48–62 y) | 68/67 | 1 g soy extracts and 80 mg isoflavones | 12 months | Femoral neck BMD (−) lumbar spine BMD (−) | Not studied | Randomized, double-blind, placebo-controlled |
| Bunout et al. [24] | 2006 | Chile | Elderly subjects (both genders) with femoral osteoporosis | 45/39 | 90 mg/day of isoflavones (together with other supplements) | 12 months | Femoral neck BMD (−) lumbar spine BMD (−) | Bone turnover markers, 25(OH)D, PTH, osteocalcin | Randomized, controlled |
| Newton et al. [35] | 2006 | USA | Men and women (50–80 y) | 9F, 45M/7F, 54M | 45.6 mg genistein, 31.7 mg daidzein, 5.5 mg glycitein daily | 12 months | Proximal femur BMD (−) lumbar spine BMD (+, only women) | Not studied | Randomized, double-blind, placebo-controlled |
| Marini et al. [38] | 2007 | Italy | Postmenopausal women with low femoral neck BMD (osteopenia or osteoporosis) | 198/191 | 54 mg of genistein aglycone daily | 24 months | Femoral neck BMD (+) lumbar spine BMD (+) | Bone turnover markers | Randomized, double-blind, placebo-controlled |
| Marini et al. [22] | 2008 | Italy | Woman with postmenopausal osteopenia | 71/67 | 54 mg of genistein aglycone daily | 36 months | Femoral neck BMD (+) lumbar spine BMD (+) | Bone turnover markers, sRANKL, osteoprotegrin, IGF-1 | Randomized, double-blind, placebo-controlled |
| Marini et al. [41] | 2008 | Italy | Woman with postmenopausal osteopenia (49–67 y) | 198/191 | 54 mg of genistein aglycone daily | 24 months | Not studied | Bone turnover markers | Randomized, double-blind, placebo-controlled |
| Brink et al. [42] | 2008 | Netherlands | Postmenopausal women (53 ± 3 y) | 119/118 | 110 mg of soy isoflavones | 12 months | Lumbar spine BMD (−) | Bone turnover markers | Randomized, double-blind, placebo-controlled |
| Wong et al. [43] | 2009 | USA | Postmenopausal women (40–60 y) | 134/134 | 120 mg of soy hypocotyl aglycone isoflavones Control group: without treatment | 24 months | Whole body BMD (+) | Bone turnover markers | Randomized, double-blind, placebo-controlled |
| Alekel et al. [44] | 2009 | USA | Postmenopausal women (45.8–65.0 y) | 73/74 | 120 mg of soy isoflavones Control group: without treatment | 36 months | Femoral neck BMD (−) lumbar spine BMD (−) | Not studied | Randomized, double-blind, placebo-controlled |
| Tai et al. [23] | 2012 | Taiwan-China | Postmenopausal women (45–65 y) | 200/199 | 172.5 mg genistein + 127.5 mg daidzein daily | 24 months | Proximal femur BMD (−) lumbar spine BMD (−) | Bone turnover markers | Randomized, double-blind, placebo-controlled |
| Chilibeck et al. [45] | 2013 | Canada | Postmenopausal women | 76/73 | 165 mg of soy isoflavones | 24 months | Lumbar spine BMD (−) | Not studied | Randomized, double-blind, placebo-controlled |
| Arcoraci et al. [25] | 2017 | Italy | Postmenopausal women with femoral neck osteoporosis | 62/59 | 54 mg of genistein aglycone daily | 24 months | Femoral neck BMD (+) | Bone turnover markers | Post hoc analysis, randomized, double-blind, placebo-controlled |
| Zhang et al. [46] | 2020 | China | Postmenopausal women (40–55 y) | 38/37 | 15 mg of soy isoflavones | 6 months | BMD (−) | Not studied | Randomized, double-blind, placebo-controlled |
| Cui et al. [36] | 2021 | China | Men (40–74 y) | 61,025 population-based cohort | Soy isoflavone intake | Median follow-up ~9.5 years | Osteoporotic fracture risk (−) | Not studied | Population based, prospective, observational |
| Cui et al. [26] | 2022 | China | Postmenopausal women (43–76 y) | 48,584 population-based cohort | Soy isoflavone intake | Median follow-up ~10 years | Osteoporotic fracture risk (+, only in bone fracture history group) Non-osteoporotic fracture risk (−) | Not studied | Population based, prospective, observational |
3.2. Effects of Isoflavones in Men
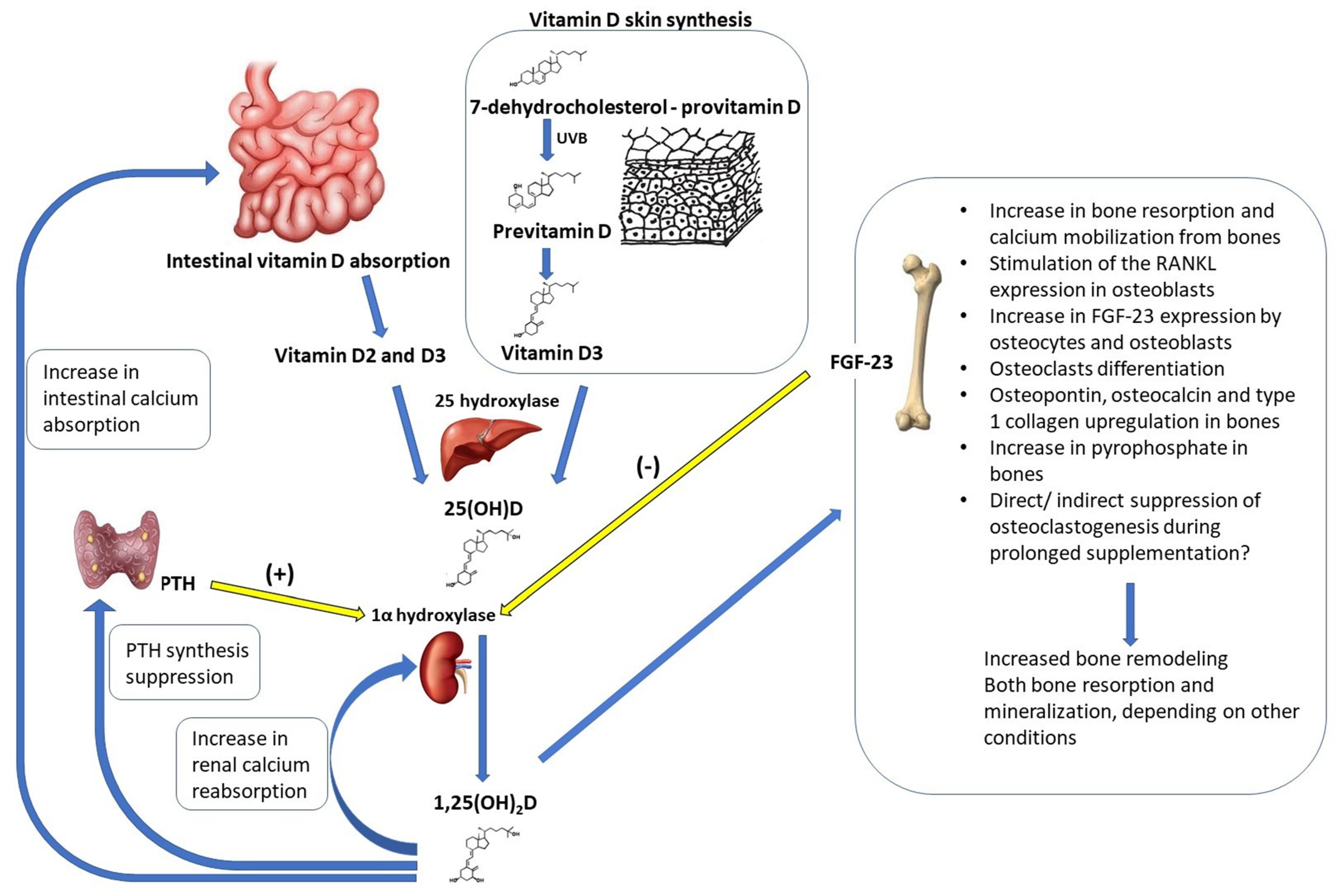
4. Vitamin D
4.1. Impact of Vitamin D on Bone Mineral Density
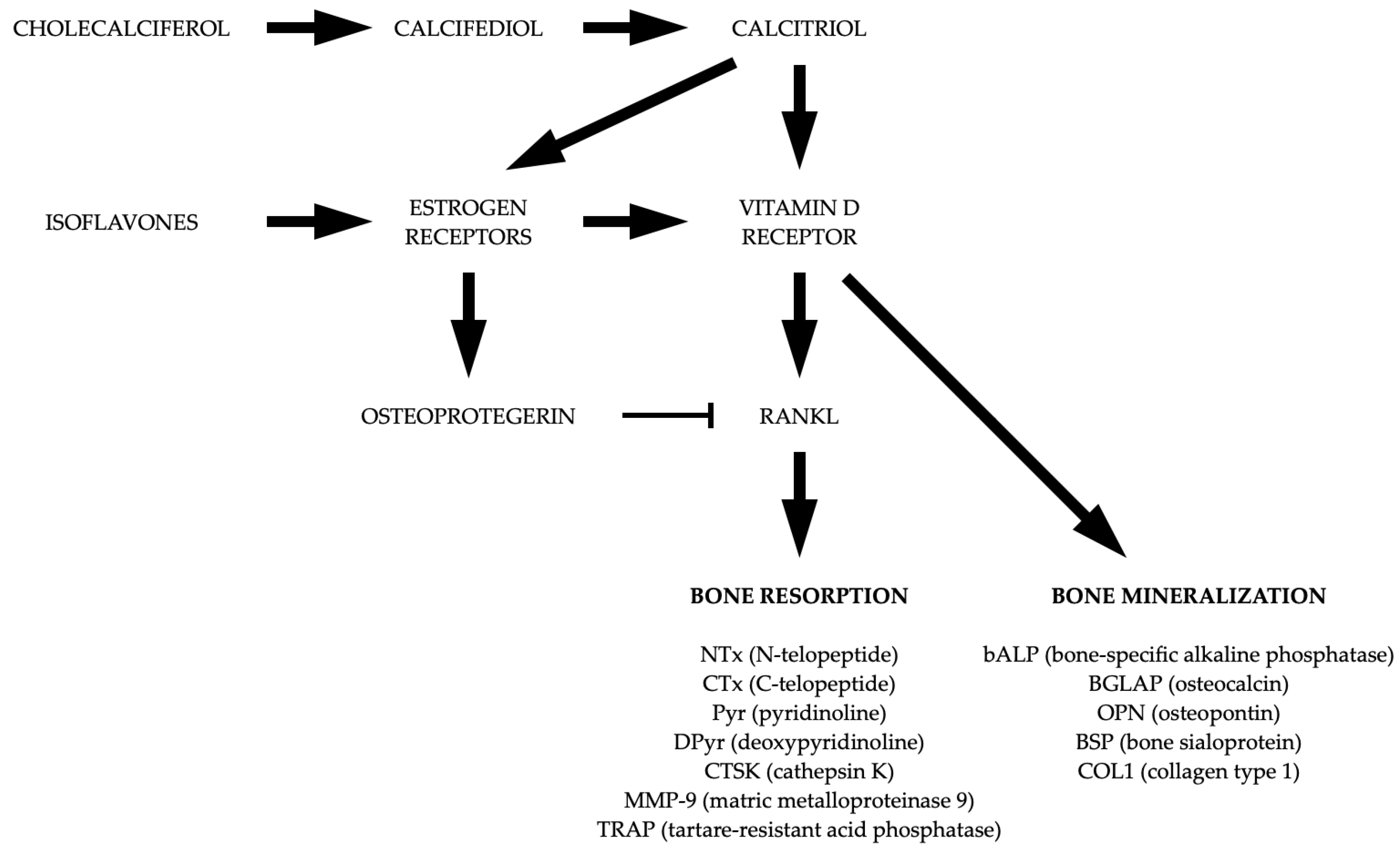
4.2. The Synergistic Effects of Isoflavonoids and Vitamin D
| Author | Year | Country | Studied Group | Patients/Controls | Intervention | Time | BMD/Fracture Risk (+) Reduction Bone Loss/Reduction Fracture Risk (−) No Reduction Bone Loss/No Reduction Fracture Risk | Other Parameters | Type of the Study |
|---|---|---|---|---|---|---|---|---|---|
| Ushiroyama et al. [100] | 1994 | Japan | Postmenopausal women (45–65 y) | 20/35 | Ipriflavone 600 mg/day, alfacalcidol 1 µg/day Control group: without treatment | 18 months | Vertebral BMD (+) | Bone turnover markers | Randomized, placebo-controlled |
| Lappe et al. [89] | 2013 | USA | Postmenopausal women (45–55 y) | 30/28 | Genistein 30 mg/day and vitamin D3 800 IU/day (together with other supplements) Control group: vitamin D3 800 IU/day (together with other supplements) | 6 months | Femoral neck BMD (+) Ward’s triangle BMD (+) lumbar spine BMD (−) | Bone turnover markers | Randomized, double-blind, placebo-controlled |
| Bevilacqua et al. [101] | 2013 | Italy | Postmenopausal women (36–84 y) | 28 for study group, without control group | Soy isoflavones 40 mg/day, inulin 3 g/day, vitamin D 300 IU/day, calcium 500 mg/day | 3 months | Not studied | Bone turnover markers | Retrospective study |
| Perez-Alonso et al. [102] | 2017 | Spain | Postmenopausal women (55 ± 4 y) | 102 in 2 groups (no more detailed information) | Genistein 90 mg/day and vitamin D3 800 IU/day and calcium 1000 mg/day Control group: vitamin D3 800 IU/day and calcium 1000 mg/day | 3 months | Not studied | Bone turnover markers | Randomized, double-blind, placebo-controlled |
| Perez-Alonso et al. [103] | 2023 | Spain | Postmenopausal women (55 ± 4 y) | 150 in 3 groups (no more detailed information) | Genistein 90 mg/day and vitamin D3 800 IU/day and calcium 1000 mg/day Control group: vitamin D3 800 IU/day and calcium 1000 mg/day Second control group: without treatment | 3 months | Not studied | Bone turnover markers | Randomized, double-blind, placebo-controlled |
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Napoli, N.; Faccio, R.; Shrestha, V.; Bucchieri, S.; Rini, G.B.; Armamento-Villareal, R. Estrogen metabolism modulates bone density in men. Calcif. Tissue Int. 2007, 80, 227–232. [Google Scholar] [CrossRef]
- DeLoughery, E.P.; Dow, M.L. Decreased bone mineral density and reproductive axis dysfunction: More than oestrogen. Neth. J. Med. 2020, 78, 50–54. [Google Scholar] [PubMed]
- Rozenberg, S.; Al-Daghri, N.; Aubertin-Leheudre, M.; Brandi, M.L.; Cano, A.; Collins, P.; Cooper, C.; Genazzani, A.R.; Hillard, T.; Kanis, J.A.; et al. Is there a role for menopausal hormone therapy in the management of postmenopausal osteoporosis? Osteoporos. Int. 2020, 31, 2271–2286. [Google Scholar] [CrossRef] [PubMed]
- Hwang, C.S.; Kwak, H.S.; Lim, H.J.; Lee, S.H.; Kang, Y.S.; Choe, T.B.; Hur, H.G.; Han, K.O. Isoflavone metabolites and their in vitro dual functions: They can act as an estrogenic agonist or antagonist depending on the estrogen concentration. J. Steroid Biochem. Mol. Biol. 2006, 101, 246–253. [Google Scholar] [CrossRef] [PubMed]
- van Duursen, M.B.M. Modulation of estrogen synthesis and metabolism by phytoestrogens in vitro and the implications for women’s health. Toxicol. Res. 2017, 6, 772–794. [Google Scholar] [CrossRef] [PubMed]
- Farkas, S.; Szabó, A.; Hegyi, A.E.; Török, B.; Fazekas, C.L.; Ernszt, D.; Kovács, T.; Zelena, D. Estradiol and Estrogen-like Alternative Therapies in Use: The Importance of the Selective and Non-Classical Actions. Biomedicines 2022, 10, 861. [Google Scholar] [CrossRef] [PubMed]
- Poluzzi, E.; Piccinni, C.; Raschi, E.; Rampa, A.; Recanatini, M.; De Ponti, F. Phytoestrogens in postmenopause: The state of the art from a chemical, pharmacological and regulatory perspective. Curr. Med. Chem. 2014, 21, 417–436. [Google Scholar] [CrossRef]
- Wyse, J.; Latif, S.; Gurusinghe, S.; McCormick, J.; Weston, L.A.; Stephen, C.P. Phytoestrogens: A Review of Their Impacts on Reproductive Physiology and Other Effects upon Grazing Livestock. Animals 2022, 12, 2709. [Google Scholar] [CrossRef]
- Bingham, S.A.; Atkinson, C.; Liggins, J.; Bluck, L.; Coward, A. Phyto-oestrogens: Where are we now? Br. J. Nutr. 1998, 79, 393–406. [Google Scholar] [CrossRef]
- Zheng, X.; Lee, S.K.; Chun, O.K. Soy Isoflavones and Osteoporotic Bone Loss: A Review with an Emphasis on Modulation of Bone Remodeling. J. Med. Food. 2016, 19, 1–14. [Google Scholar] [CrossRef]
- Schmid, A.; Walther, B. Natural vitamin D content in animal products. Adv. Nutr. 2013, 4, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Bikle, D.D. Vitamin D and bone. Curr. Osteoporos. Rep. 2012, 10, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.L.; Hu, Y.C.; Hsieh, B.S.; Cheng, H.L.; Hsu, H.W.; Huang, L.W.; Su, S.J. Combined effect of soy isoflavones and vitamin D3 on bone loss in ovariectomized rats. Nutrition 2013, 29, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Sahin, N.; Balci, T.A.; Kucuk, O.; Smith, M.O.; Sahin, K. Effects of 25-hydroxycholecalciferol and soy isoflavones supplementation on bone mineralisation of quail. Br. Poult. Sci. 2009, 50, 709–715. [Google Scholar] [CrossRef]
- Jalili, M.; Hekmatdoost, A.; Vahedi, H.; Poustchi, H.; Khademi, B.; Saadi, M.; Zemestani, M.; Janani, L. Co-Administration of Soy Isoflavones and Vitamin D in Management of Irritable Bowel Disease. PLoS ONE 2016, 11, e0158545. [Google Scholar] [CrossRef] [PubMed]
- Martiniakova, M.; Babikova, M.; Omelka, R. Pharmacological agents and natural compounds: Available treatments for osteoporosis. J. Physiol. Pharmacol. 2020, 71, 307–320. [Google Scholar] [CrossRef]
- Spence, L.A.; Lipscomb, E.R.; Cadogan, J.; Martin, B.; Wastney, M.E.; Peacock, M.; Weaver, C.M. The effect of soy protein and soy isoflavones on calcium metabolism in postmenopausal women: A randomized crossover study. Am. J. Clin. Nutr. 2005, 81, 916–922. [Google Scholar] [CrossRef]
- Roughead, Z.K.; Hunt, J.R.; Johnson, L.K.; Badger, T.M.; Lykken, G.I. Controlled substitution of soy protein for meat protein: Effects on calcium retention, bone, and cardiovascular health indices in postmenopausal women. J. Clin. Endocrinol. Metab. 2005, 90, 181–189. [Google Scholar] [CrossRef]
- Lu, L.W.; Chen, N.W.; Nayeem, F.; Ramanujam, V.S.; Kuo, Y.F.; Brunder, D.G.; Nagamani, M.; Anderson, K.E. Novel effects of phytoestrogenic soy isoflavones on serum calcium and chloride in premenopausal women: A 2-year double-blind, randomized, placebo-controlled study. Clin. Nutr. 2018, 37, 1862–1870. [Google Scholar] [CrossRef]
- Pawlowski, J.W.; Martin, B.R.; McCabe, G.P.; McCabe, L.; Jackson, G.S.; Peacock, M.; Barnes, S.; Weaver, C.M. Impact of equol-producing capacity and soy-isoflavone profiles of supplements on bone calcium retention in postmenopausal women: A randomized crossover trial. Am. J. Clin. Nutr. 2015, 102, 695–703. [Google Scholar] [CrossRef]
- Park, C.Y.; Weaver, C.M. Vitamin D interactions with soy isoflavones on bone after menopause: A review. Nutrients 2012, 4, 1610–1621. [Google Scholar] [CrossRef] [PubMed]
- Marini, H.; Bitto, A.; Altavilla, D.; Burnett, B.P.; Polito, F.; Di Stefano, V.; Minutoli, L.; Atteritano, M.; Levy, R.M.; D’Anna, R.; et al. Breast safety and efficacy of genistein aglycone for postmenopausal bone loss: A follow-up study. J. Clin. Endocrinol. Metab. 2008, 93, 4787–4796. [Google Scholar] [CrossRef] [PubMed]
- Tai, T.Y.; Tsai, K.S.; Tu, S.T.; Wu, J.S.; Chang, C.I.; Chen, C.L.; Shaw, N.S.; Peng, H.Y.; Wang, S.Y.; Wu, C.H. The effect of soy isoflavone on bone mineral density in postmenopausal Taiwanese women with bone loss: A 2-year randomized double-blind placebo-controlled study. Osteoporos. Int. 2012, 23, 1571–1580. [Google Scholar] [CrossRef] [PubMed]
- Bunout, D.; Barrera, G.; Leiva, L.; Gattas, V.; de la Maza, M.P.; Haschke, F.; Steenhout, P.; Klassen, P.; Hager, C.; Offord, E.; et al. Effect of a nutritional supplementation on bone health in Chilean elderly subjects with femoral osteoporosis. J. Am. Coll. Nutr. 2006, 25, 170–177. [Google Scholar] [CrossRef]
- Arcoraci, V.; Atteritano, M.; Squadrito, F.; D’Anna, R.; Marini, H.; Santoro, D.; Minutoli, L.; Messina, S.; Altavilla, D.; Bitto, A. Antiosteoporotic Activity of Genistein Aglycone in Postmenopausal Women: Evidence from a Post-Hoc Analysis of a Multicenter Randomized Controlled Trial. Nutrients 2017, 9, 179. [Google Scholar] [CrossRef]
- Cui, Y.; Cai, H.; Gao, Y.; Dai, Q.; Yang, G.; Zheng, W.; Shu, X.O. Associations of dietary intakes of calcium, magnesium and soy isoflavones with osteoporotic fracture risk in postmenopausal women: A prospective study. J. Nutr. Sci. 2022, 11, e62. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.F.; Qin, L.Q.; Wang, P.Y.; Katoh, R. Soy isoflavone intake inhibits bone resorption and stimulates bone formation in menopausal women: Meta-analysis of randomized controlled trials. Eur. J. Clin. Nutr. 2008, 62, 155–161. [Google Scholar] [CrossRef]
- Taku, K.; Melby, M.K.; Kurzer, M.S.; Mizuno, S.; Watanabe, S.; Ishimi, Y. Effects of soy isoflavone supplements on bone turnover markers in menopausal women: Systematic review and meta-analysis of randomized controlled trials. Bone 2010, 47, 413–423. [Google Scholar] [CrossRef]
- Akhlaghi, M.; Ghasemi Nasab, M.; Riasatian, M.; Sadeghi, F. Soy isoflavones prevent bone resorption and loss, a systematic review and meta-analysis of randomized controlled trials. Crit. Rev. Food Sci. Nutr. 2020, 60, 2327–2341. [Google Scholar] [CrossRef]
- Kanadys, W.; Barańska, A.; Błaszczuk, A.; Polz-Dacewicz, M.; Drop, B.; Malm, M.; Kanecki, K. Effects of Soy Isoflavones on Biochemical Markers of Bone Metabolism in Postmenopausal Women: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Int. J. Environ. Res. Public Health 2021, 18, 5346. [Google Scholar] [CrossRef]
- Barańska, A.; Kanadys, W.; Bogdan, M.; Stępień, E.; Barczyński, B.; Kłak, A.; Augustynowicz, A.; Szajnik, M.; Religioni, U. The Role of Soy Isoflavones in the Prevention of Bone Loss in Postmenopausal Women: A Systematic Review with Meta-Analysis of Randomized Controlled Trials. J. Clin. Med. 2022, 11, 4676. [Google Scholar] [CrossRef]
- Lydeking-Olsen, E.; Beck-Jensen, J.E.; Setchell, K.D.; Holm-Jensen, T. Soymilk or progesterone for prevention of bone loss—A 2 year randomized, placebo-controlled trial. Eur. J. Nutr. 2004, 43, 246–257. [Google Scholar] [CrossRef]
- Inpan, R.; Na Takuathung, M.; Sakuludomkan, W.; Dukaew, N.; Teekachunhatean, S.; Koonrungsesomboon, N. Isoflavone intervention and its impact on bone mineral density in postmenopausal women: A systematic review and meta-analysis of randomized controlled trials. Osteoporos. Int. 2023. [Google Scholar] [CrossRef]
- Khalil, D.A.; Lucas, E.A.; Juma, S.; Smith, B.J.; Payton, M.E.; Arjmandi, B.H. Soy protein supplementation increases serum insulin-like growth factor-I in young and old men but does not affect markers of bone metabolism. J. Nutr. 2002, 132, 2605–2608. [Google Scholar] [CrossRef]
- Newton, K.M.; LaCroix, A.Z.; Levy, L.; Li, S.S.; Qu, P.; Potter, J.D.; Lampe, J.W. Soy protein and bone mineral density in older men and women: A randomized trial. Maturitas 2006, 5, 270–277. [Google Scholar] [CrossRef]
- Cui, Y.; Cai, H.; Zheng, W.; Shu, X.O. Associations of Dietary Intakes of Calcium, Magnesium, and Soy Isoflavones with Bone Fracture Risk in Men: A Prospective Study. JBMR Plus 2021, 6, e10563. [Google Scholar] [CrossRef]
- Marini, H.; Bitto, A.; Altavilla, D.; Burnett, B.P.; Polito, F.; Di Stefano, V.; Minutoli, L.; Atteritano, M.; Levy, R.M.; Frisina, N.; et al. Efficacy of genistein aglycone on some cardiovascular risk factors and homocysteine levels: A follow-up study. Nutr. Metab. Cardiovasc. Dis. 2010, 20, 332–340. [Google Scholar] [CrossRef]
- Marini, H.; Minutoli, L.; Polito, F.; Bitto, A.; Altavilla, D.; Atteritano, M.; Gaudio, A.; Mazzaferro, S.; Frisina, A.; Frisina, N.; et al. Effects of the phytoestrogen genistein on bone metabolism in osteopenic postmenopausal women: A randomized trial. Ann. Intern. Med. 2007, 146, 839–847. [Google Scholar] [CrossRef]
- Agnusdei, D.; Bufalino, L. Efficacy of ipriflavone in established osteoporosis and long-term safety. Calcif. Tissue Int. 1997, 61, 23–27. [Google Scholar] [CrossRef]
- Chen, Y.M.; Ho, S.C.; Lam, S.S.; Ho, S.S.; Woo, J.L. Soy isoflavones have a favorable effect on bone loss in Chinese postmenopausal women with lower bone mass: A double-blind, randomized, controlled trial. J. Clin. Endocrinol. Metab. 2003, 88, 4740–4747. [Google Scholar] [CrossRef] [PubMed]
- Marini, H.; Minutoli, L.; Polito, F.; Bitto, A.; Altavilla, D.; Atteritano, M.; Gaudio, A.; Mazzaferro, S.; Frisina, A.; Frisina, N.; et al. OPG and sRANKL serum concentrations in osteopenic, postmenopausal women after 2-year genistein administration. J. Bone Miner. Res. 2008, 23, 715–720. [Google Scholar] [CrossRef] [PubMed]
- Brink, E.; Coxam, V.; Robins, S.; Wahala, K.; Cassidy, A.; Branca, F.; PHYTOS Investigators. Long-term consumption of isoflavone-enriched foods does not affect bone mineral density, bone metabolism, or hormonal status in early postmenopausal women: A randomized, double-blind, placebo controlled study. Am. J. Clin. Nutr. 2008, 87, 761–770. [Google Scholar] [CrossRef] [PubMed]
- Wong, W.W.; Lewis, R.D.; Steinberg, F.M.; Murray, M.J.; Cramer, M.A.; Amato, P.; Young, R.L.; Barnes, S.; Ellis, K.J.; Shypailo, R.J.; et al. Soy isoflavone supplementation and bone mineral density in menopausal women: A 2-y multicenter clinical trial. Am. J. Clin. Nutr. 2009, 90, 1433–1439. [Google Scholar] [CrossRef] [PubMed]
- Alekel, D.L.; Van Loan, M.D.; Koehler, K.J.; Hanson, L.N.; Stewart, J.W.; Hanson, K.B.; Kurzer, M.S.; Peterson, C.T. The soy isoflavones for reducing bone loss (SIRBL) study: A 3-y randomized controlled trial in postmenopausal women. Am. J. Clin. Nutr. 2010, 91, 218–230. [Google Scholar] [CrossRef] [PubMed]
- Chilibeck, P.D.; Vatanparast, H.; Pierson, R.; Case, A.; Olatunbosun, O.; Whiting, S.J.; Beck, T.J.; Pahwa, P.; Biem, H.J. Effect of exercise training combined with isoflavone supplementation on bone and lipids in postmenopausal women: A randomized clinical trial. J. Bone Miner. Res. 2013, 28, 780–793. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Liu, Y.; Xu, Q.; Zhang, Y.; Liu, L.; Li, H.; Li, F.; Liu, Z.; Yang, X.; Yu, X.; et al. The effect of soy isoflavone combined with calcium on bone mineral density in perimenopausal Chinese women: A 6-month randomised double-blind placebo-controlled study. Int. J. Food Sci. Nutr. 2020, 71, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Somjen, D.; Katzburg, S.; Kohen, F.; Gayer, B.; Sharon, O.; Hendel, D.; Posner, G.H.; Kaye, A.M. Responsiveness to phytoestrogens in primary human osteoblasts is modulated differentially by a “less-calcemic” analog of 1,25 dihydroxyvitamin D(3): JK 1624F(2)-2 (JKF). J. Steroid Biochem. Mol. Biol. 2006, 98, 139–146. [Google Scholar] [CrossRef]
- Laird, E.; Ward, M.; McSorley, E.; Strain, J.J.; Wallace, J. Vitamin D and bone health: Potential mechanisms. Nutrients 2010, 2, 693–724. [Google Scholar] [CrossRef]
- Miedziaszczyk, M.; Idasiak-Piechocka, I.; Wiśniewski, O.W.; Lacka, K. A systematic review of the pharmacotherapy of secondary hyperparathyroidism (SHPT) in grades 3–5 Chronic Kidney Disease (CKD). Eur. Rev. Med. Pharmacol. Sci. 2022, 26, 232–239. [Google Scholar]
- Miedziaszczyk, M.; Lacka, K.; Tomczak, O.; Bajon, A.; Primke, M.; Idasiak-Piechocka, I. Systematic Review of the Treatment of Persistent Hyperparathyroidism Following Kidney Transplantation. Biomedicines 2022, 11, 25. [Google Scholar] [CrossRef]
- Siddiqui, J.A.; Partridge, N.C. Physiological Bone Remodeling: Systemic Regulation and Growth Factor Involvement. Physiology 2016, 31, 233–245. [Google Scholar] [CrossRef] [PubMed]
- Raggatt, L.J.; Partridge, N.C. Cellular and molecular mechanisms of bone remodeling. J. Biol. Chem. 2010, 285, 25103–25108. [Google Scholar] [CrossRef] [PubMed]
- Wacker, M.; Holick, M.F. Sunlight and Vitamin D: A global perspective for health. Dermatoendocrinol 2013, 5, 51–108. [Google Scholar] [CrossRef] [PubMed]
- Benedik, E. Sources of vitamin D for humans. Int. J. Vitam. Nutr. Res. 2022, 92, 118–125. [Google Scholar] [CrossRef] [PubMed]
- McCourt, A.F.; O’Sullivan, A.M. Using food fortification to improve vitamin D bioaccessibility and intakes. Proc. Nutr. Soc. 2022, 81, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Cashman, K.D. Global differences in vitamin D status and dietary intake: A review of the data. Endocr. Connect. 2022, 11, e210282. [Google Scholar] [CrossRef]
- Man, P.W.; van der Meer, I.M.; Lips, P.; Middelkoop, B.J. Vitamin D status and bone mineral density in the Chinese population: A review. Arch. Osteoporos. 2016, 11, 14. [Google Scholar] [CrossRef]
- Cauley, J.A.; Lacroix, A.Z.; Wu, L.; Horwitz, M.; Danielson, M.E.; Bauer, D.C.; Lee, J.S.; Jackson, R.D.; Robbins, J.A.; Wu, C.; et al. Serum 25-hydroxyvitamin D concentrations and risk for hip fractures. Ann. Intern. Med. 2008, 149, 242–250. [Google Scholar] [CrossRef]
- Zmijewski, M.A. Vitamin D and Human Health. Int. J. Mol. Sci. 2019, 20, 145. [Google Scholar] [CrossRef]
- Pludowski, P.; Grant, W.B.; Konstantynowicz, J.; Holick, M.F. Editorial: Classic and Pleiotropic Actions of Vitamin D. Front. Endocrinol. 2019, 10, 341. [Google Scholar] [CrossRef]
- Cashman, K.D.; Dowling, K.G.; Škrabáková, Z.; Gonzalez-Gross, M.; Valtueña, J.; De Henauw, S.; Moreno, L.; Damsgaard, C.T.; Michaelsen, K.F.; Mølgaard, C.; et al. Vitamin D deficiency in Europe: Pandemic? Am. J. Clin. Nutr. 2016, 103, 1033–1044. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M.; Endocrine Society. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Płudowski, P.; Kos-Kudła, B.; Walczak, M.; Fal, A.; Zozulińska-Ziółkiewicz, D.; Sieroszewski, P.; Peregud-Pogorzelski, J.; Lauterbach, R.; Targowski, T.; Lewiński, A.; et al. Guidelines for Preventing and Treating Vitamin D Deficiency: A 2023 Update in Poland. Nutrients 2023, 15, 695. [Google Scholar] [CrossRef] [PubMed]
- Płudowski, P.; Ducki, C.; Konstantynowicz, J.; Jaworski, M. Vitamin D status in Poland. Pol. Arch. Med. Wewn. 2016, 126, 530–539. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Wang, J.; Song, M.; Giovannucci, E.L.; Ma, H.; Jin, G.; Hu, Z.; Shen, H.; Hang, D. Vitamin D Status and Risk of All-Cause and Cause-Specific Mortality in a Large Cohort: Results from the UK Biobank. J. Clin. Endocrinol. Metab. 2020, 105, dgaa432. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Fang, F.; Tang, J.; Jia, L.; Feng, Y.; Xu, P.; Faramand, A. Association between vitamin D supplementation and mortality: Systematic review and meta-analysis. BMJ 2019, 366, l4673. [Google Scholar] [CrossRef]
- Cranney, A.; Horsley, T.; O’Donnell, S.; Weiler, H.; Puil, L.; Ooi, D.; Atkinson, S.; Ward, L.; Moher, D.; Hanley, D.; et al. Effectiveness and safety of vitamin D in relation to bone health. Evid. Rep. Technol. Assess. (Full Rep.) 2007, 158, 1–235. [Google Scholar]
- Kuchuk, N.O.; van Schoor, N.M.; Pluijm, S.M.; Chines, A.; Lips, P. Vitamin D status, parathyroid function, bone turnover, and BMD in postmenopausal women with osteoporosis: Global perspective. J. Bone Miner. Res. 2009, 24, 693–701. [Google Scholar] [CrossRef]
- Reid, I.R. Vitamin D Effect on Bone Mineral Density and Fractures. Endocrinol. Metab. Clin. N. Am. 2017, 46, 935–945. [Google Scholar] [CrossRef]
- Sanders, K.M.; Nicholson, G.C.; Ebeling, P.R. Is high dose vitamin D harmful? Calcif. Tissue Int. 2013, 92, 191–206. [Google Scholar] [CrossRef]
- Burt, L.A.; Billington, E.O.; Rose, M.S.; Raymond, D.A.; Hanley, D.A.; Boyd, S.K. Effect of High-Dose Vitamin D Supplementation on Volumetric Bone Density and Bone Strength: A Randomized Clinical Trial. JAMA 2019, 322, 736–745. [Google Scholar] [CrossRef] [PubMed]
- Anagnostis, P.; Bosdou, J.K.; Kenanidis, E.; Potoupnis, M.; Tsiridis, E.; Goulis, D.G. Vitamin D supplementation and fracture risk: Evidence for a U-shaped effect. Maturitas 2020, 141, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Dhaliwal, R.; Islam, S.; Mikhail, M.; Ragolia, L.; Aloia, J.F. Effect of vitamin D on bone strength in older African Americans: A randomized controlled trial. Osteoporos. Int. 2020, 31, 1105–1114. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.Y.; Zhao, M.; Hou, Y.; Zhang, C.; Oh, J.; Sun, Z.; Sun, B.L. Circulating serum vitamin D levels and total body bone mineral density: A Mendelian randomization study. J. Cell Mol. Med. 2019, 23, 2268–2271. [Google Scholar] [CrossRef] [PubMed]
- Bischoff-Ferrari, H.A.; Willett, W.C.; Orav, E.J.; Lips, P.; Meunier, P.J.; Lyons, R.A.; Flicker, L.; Wark, J.; Jackson, R.D.; Cauley, J.A.; et al. A pooled analysis of vitamin D dose requirements for fracture prevention. N. Engl. J. Med. 2012, 367, 40–49. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Willett, W.C.; Wong, J.B.; Stuck, A.E.; Staehelin, H.B.; Orav, E.J.; Thoma, A.; Kiel, D.P.; Henschkowski, J. Prevention of nonvertebral fractures with oral vitamin D and dose dependency: A meta-analysis of randomized controlled trials. Arch. Intern. Med. 2009, 169, 551–561. [Google Scholar] [CrossRef]
- Zhao, J.G.; Zeng, X.T.; Wang, J.; Liu, L. Association Between Calcium or Vitamin D Supplementation and Fracture Incidence in Community-Dwelling Older Adults: A Systematic Review and Meta-analysis. JAMA 2017, 318, 2466–2482. [Google Scholar] [CrossRef]
- Ramirez, N.; Ortiz-Fullana, J.L.; Arciniegas, N.; Fullana, A.; Valentin, P.; Orengo, J.C.; Iriarte, I.; Carlo, S. Vitamin D levels and fracture risk among Hispanic children. Eur. J. Orthop. Surg. Traumatol. 2019, 29, 531–536. [Google Scholar] [CrossRef]
- Bolland, M.J.; Grey, A.; Avenell, A. Effects of vitamin D supplementation on musculoskeletal health: A systematic review, meta-analysis, and trial sequential analysis. Lancet Diabetes Endocrinol. 2018, 6, 847–858. [Google Scholar] [CrossRef]
- Chakhtoura, M.; Bacha, D.S.; Gharios, C.; Ajjour, S.; Assaad, M.; Jabbour, Y.; Kahale, F.; Bassatne, A.; Antoun, S.; Akl, E.A.; et al. Vitamin D Supplementation and Fractures in Adults: A Systematic Umbrella Review of Meta-Analyses of Controlled Trials. J. Clin. Endocrinol. Metab. 2022, 107, 882–898. [Google Scholar] [CrossRef]
- Wang, J.; Shu, B.; Li, C.G.; Xie, X.W.; Liang, D.; Chen, B.L.; Lin, X.C.; Wei, X.; Wang, L.; Leng, X.Y.; et al. Polymorphisms of genes related to vitamin D metabolism and transportation and its relationship with the risk of osteoporosis: Protocol for a multicentre prospective cohort study in China. BMJ Open 2019, 9, e028084. [Google Scholar] [CrossRef] [PubMed]
- Pakpahan, C.; Wungu, C.D.K.; Agustinus, A.; Darmadi, D. Do Vitamin D receptor gene polymorphisms affect bone mass density in men?: A meta-analysis of observational studies. Ageing Res. Rev. 2022, 75, 101571. [Google Scholar] [CrossRef]
- Bao, L.; Chen, M.; Lei, Y.; Zhou, Z.; Shen, H.; Le, F. Association between vitamin D receptor BsmI polymorphism and bone mineral density in pediatric patients: A meta-analysis and systematic review of observational studies. Medicine 2017, 96, e6718. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Xi, B.; Li, K.; Wang, C. Association between vitamin D receptor gene polymorphisms and bone mineral density in Chinese women. Mol. Biol. Rep. 2012, 39, 5709–5717. [Google Scholar] [CrossRef] [PubMed]
- Spinek, A.E.; Majsterek, I.; Walczak, A.; Rosset, I.; Sewerynek, E.; Żądzińska, E. VDR polymorphisms effect on bone mineral density in Polish postmenopausal women. Homo 2021, 72, 239–260. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Ai, Z.; Song, M.; Yan, P.; Li, J.; Wang, S. The association between vitamin D receptor FokI gene polymorphism and osteoporosis in postmenopausal women: A meta-analysis. Climacteric 2021, 24, 74–79. [Google Scholar] [CrossRef]
- Jiang, L.L.; Zhang, C.; Zhang, Y.; Ma, F.; Guan, Y. Associations between polymorphisms in VDR gene and the risk of osteoporosis: A meta-analysis. Arch. Physiol. Biochem. 2022, 128, 1637–1644. [Google Scholar] [CrossRef]
- Mu, Y.Y.; Liu, B.; Chen, B.; Zhu, W.F.; Ye, X.H.; Li, H.Z.; He, X.F. Evaluation of Association Studies and an Updated Meta-Analysis of VDR Polymorphisms in Osteoporotic Fracture Risk. Front. Genet. 2022, 12, 791368. [Google Scholar] [CrossRef]
- Lappe, J.; Kunz, I.; Bendik, I.; Prudence, K.; Weber, P.; Recker, R.; Heaney, R.P. Effect of a combination of genistein, polyunsaturated fatty acids and vitamins D3 and K1 on bone mineral density in postmenopausal women: A randomized, placebo-controlled, double-blind pilot study. Eur. J. Nutr. 2013, 52, 203–215. [Google Scholar] [CrossRef]
- Lai, C.Y.; Yang, J.Y.; Rayalam, S.; Della-Fera, M.A.; Ambati, S.; Lewis, R.D.; Hamrick, M.W.; Hartzell, D.L.; Baile, C.A. Preventing bone loss and weight gain with combinations of vitamin D and phytochemicals. J. Med. Food. 2011, 14, 1352–1362. [Google Scholar] [CrossRef]
- Bitto, A.; Marini, H.; Burnett, B.P.; Polito, F.; Levy, R.M.; Irrera, N.; Minutoli, L.; Adamo, E.B.; Squadrito, F.; Altavilla, D. Genistein aglycone effect on bone loss is not enhanced by supplemental calcium and vitamin D3: A dose ranging experimental study. Phytomedicine 2011, 18, 879–886. [Google Scholar] [CrossRef] [PubMed]
- Morito, K.; Hirose, T.; Kinjo, J.; Hirakawa, T.; Okawa, M.; Nohara, T.; Ogawa, S.; Inoue, S.; Muramatsu, M.; Masamune, Y. Interaction of phytoestrogens with estrogen receptors alpha and beta. Biol. Pharm. Bull. 2001, 24, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Wietzke, J.A.; Welsh, J. Phytoestrogen regulation of a Vitamin D3 receptor promoter and 1,25-dihydroxyvitamin D3 actions in human breast cancer cells. J. Steroid Biochem. Mol. Biol. 2003, 84, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Farhan, H.; Wähälä., K.; Cross, H.S. Genistein inhibits vitamin D hydroxylases CYP24 and CYP27B1 expression in prostate cells. J. Steroid Biochem. Mol. Biol. 2003, 84, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Santos-Martínez, N.; Díaz, L.; Ortiz-Ortega, V.M.; Ordaz-Rosado, D.; Prado-Garcia, H.; Avila, E.; Larrea, F.; García-Becerra, R. Calcitriol induces estrogen receptor α expression through direct transcriptional regulation and epigenetic modifications in estrogen receptor-negative breast cancer cells. Am. J. Cancer Res. 2021, 11, 5951–5964. [Google Scholar] [PubMed]
- Ziemińska, M.; Sieklucka, B.; Pawlak, K. Vitamin K and D Supplementation and Bone Health in Chronic Kidney Disease-Apart or Together? Nutrients 2021, 13, 809. [Google Scholar] [CrossRef]
- Locantore, P.; Del Gatto, V.; Gelli, S.; Paragliola, R.M.; Pontecorvi, A. The Interplay between Immune System and Microbiota in Osteoporosis. Mediat. Inflamm. 2020, 2020, 3686749. [Google Scholar] [CrossRef] [PubMed]
- Mori, G.; D’Amelio, P.; Faccio, R.; Brunetti, G. The Interplay between the bone and the immune system. Clin. Dev. Immunol. 2013, 2013, 720504. [Google Scholar] [CrossRef]
- Aranow, C. Vitamin D and the immune system. J. Investig. Med. 2011, 59, 881–886. [Google Scholar] [CrossRef]
- Ushiroyama, T.; Okamura, S.; Ikeda, A.; Ueki, M. Efficacy of ipriflavone and 1 alpha vitamin D therapy for the cessation of vertebral bone loss. Int. J. Gynaecol. Obstet. 1995, 48, 283–288. [Google Scholar] [CrossRef]
- Bevilacqua, M.; Righini, V.; Certan, D.; Gandolini, G.; Alemanni, M. Effect of a mixture of calcium, vitamin D, inulin and soy isoflavones on bone metabolism in post-menopausal women: A retrospective analysis. Aging Clin. Exp. Res. 2013, 25, 611–617. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Alonso, M.; Briongos, L.S.; Ruiz-Mambrilla, M.; Velasco, E.A.; Linares, L.; Cuellar, L.; Olmos, J.M.; De Luis, D.; Dueñas-Laita, A.; Pérez-Castrillón, J.L. The Effect of Genistein Supplementation on Vitamin D Levels and Bone Turnover Markers during the Summer in Healthy Postmenopausal Women: Role of Genotypes of Isoflavone Metabolism. J. Nutr. Nutr. 2017, 10, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Alonso, M.; Calero-Paniagua, I.; Usategui-Martin, R.; Briongos, L.S.; Ruiz-Mambrilla, M.; Olmos, J.M.; González-Sagrado, M.; De Luis, D.; Dueñas-Laita, A.; Pérez-Castrillón, J.L. Genistein supplementation has no effects on vitamin D levels in healthy Spanish postmenopausal women. Int. J. Vitam. Nutr. Res. 2023. [Google Scholar] [CrossRef] [PubMed]
- Messina, M.; Duncan, A.; Messina, V.; Lynch, H.; Kiel, J.; Erdman, J.W., Jr. The health effects of soy: A reference guide for health professionals. Front. Nutr. 2022, 9, 970364. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.W.; Chen, N.W.; Brunder, D.G.; Nayeem, F.; Nagamani, M.; Nishino, T.K.; Anderson, K.E.; Khamapirad, T. Soy isoflavones decrease fibroglandular breast tissue measured by magnetic resonance imaging in premenopausal women: A 2-year randomized double-blind placebo controlled clinical trial. Clin. Nutr. ESPEN 2022, 52, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, J.; Wang, T.; Wang, Y.; Zhao, L.; Fang, Y. The effect of genistein on glucose control and insulin sensitivity in postmenopausal women: A meta-analysis. Maturitas 2017, 97, 44–52. [Google Scholar] [CrossRef]
- Li, J.; Liu, Y.; Wang, T.; Zhao, L.; Feng, W. Does genistein lower plasma lipids and homocysteine levels in postmenopausal women? A meta-analysis. Climacteric 2016, 19, 440–447. [Google Scholar] [CrossRef]
- Yi, X.Y.; Wang, Z.H.; Wang, Y. Genistein for glycolipid metabolism in postmenopausal women: A meta-analysis. Climacteric 2021, 24, 267–274. [Google Scholar] [CrossRef]
- Muñoz-Garach, A.; García-Fontana, B.; Muñoz-Torres, M. Nutrients and Dietary Patterns Related to Osteoporosis. Nutrients 2020, 12, 1986. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miedziaszczyk, M.; Maciejewski, A.; Idasiak-Piechocka, I.; Karczewski, M.; Lacka, K. Effects of Isoflavonoid and Vitamin D Synergism on Bone Mineral Density—A Systematic and Critical Review. Nutrients 2023, 15, 5014. https://doi.org/10.3390/nu15245014
Miedziaszczyk M, Maciejewski A, Idasiak-Piechocka I, Karczewski M, Lacka K. Effects of Isoflavonoid and Vitamin D Synergism on Bone Mineral Density—A Systematic and Critical Review. Nutrients. 2023; 15(24):5014. https://doi.org/10.3390/nu15245014
Chicago/Turabian StyleMiedziaszczyk, Miłosz, Adam Maciejewski, Ilona Idasiak-Piechocka, Marek Karczewski, and Katarzyna Lacka. 2023. "Effects of Isoflavonoid and Vitamin D Synergism on Bone Mineral Density—A Systematic and Critical Review" Nutrients 15, no. 24: 5014. https://doi.org/10.3390/nu15245014
APA StyleMiedziaszczyk, M., Maciejewski, A., Idasiak-Piechocka, I., Karczewski, M., & Lacka, K. (2023). Effects of Isoflavonoid and Vitamin D Synergism on Bone Mineral Density—A Systematic and Critical Review. Nutrients, 15(24), 5014. https://doi.org/10.3390/nu15245014