Effects of Zinc Supplementation on Inflammatory and Cognitive Parameters in Middle-Aged Women with Overweight or Obesity
Abstract
:1. Introduction
2. Materials and Methods
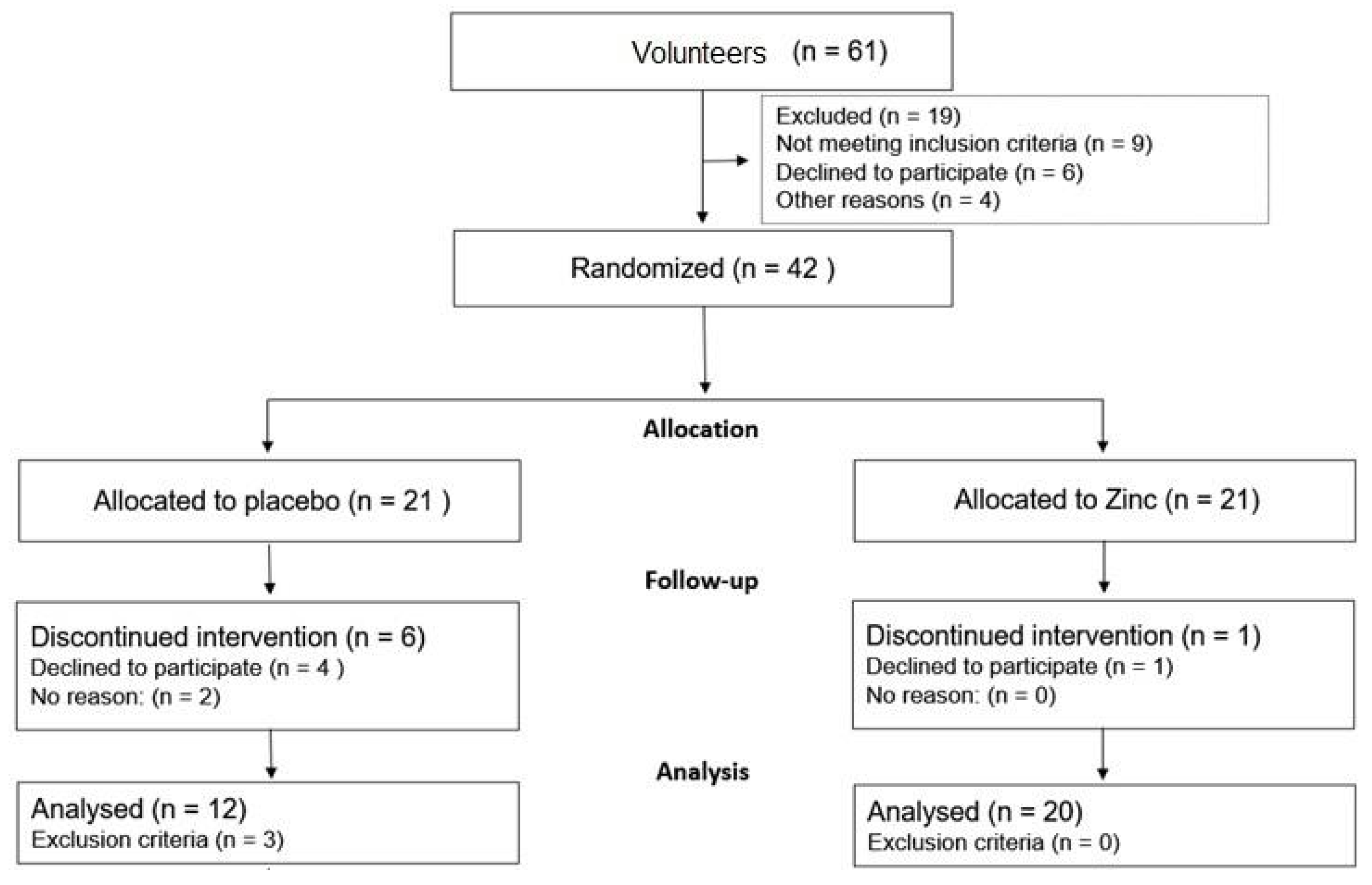
2.1. Participants and Randomization
2.2. Participant Interview and Sample Collection Procedures
2.3. Salivary Sample Collection and Analysis
2.4. Zn Supplement
2.5. Cognitive Assessment
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hara, T.; Takeda, T.; Takagishi, T.; Fukue, K.; Kambe, T.; Fukada, T. Physiological roles of zinc transporters: Molecular and genetic importance in zinc homeostasis. J. Physiol. Sci. 2017, 67, 283–301. [Google Scholar] [CrossRef] [PubMed]
- Vashum, K.P.; McEvoy, M.; Milton, A.H.; Islam, M.R.; Hancock, S.; Attia, J. Is Serum Zinc Associated with Pancreatic Beta Cell Function and Insulin Sensitivity in Pre-Diabetic and Normal Individuals? Findings from the Hunter Community Study. PLoS ONE 2014, 9, e83944. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Dischler, A.; Glover, K.; Qin, Y. Neuronal signalling of zinc: From detection and modulation to function. Open Biol. 2022, 12, 220188. [Google Scholar] [CrossRef] [PubMed]
- Maares, M.; Haase, H. A Guide to Human Zinc Absorption: General Overview and Recent Advances of In Vitro Intestinal Models. Nutrients 2020, 12, 762. [Google Scholar] [CrossRef]
- Wang, B.; Fang, T.; Chen, H. Zinc and Central Nervous System Disorders. Nutrients 2023, 15, 2140. [Google Scholar] [CrossRef]
- de Luis, D.A.; Pacheco, D.; Izaola, O.; Terroba, M.C.; Cuellar, L.; Cabezas, G. Micronutrient status in morbidly obese women before bariatric surgery. Surg. Obes. Relat. Dis. 2013, 9, 323–327. [Google Scholar] [CrossRef]
- Razzaque, M.S. COVID-19 pandemic: Can zinc supplementation provide an additional shield against the infection? Comput. Struct. Biotechnol. J. 2021, 19, 1371–1378. [Google Scholar] [CrossRef]
- Warthon-Medina, M.; Moran, V.H.; Stammers, A.-L.; Dillon, S.; Qualter, P.; Nissensohn, M.; Serra-Majem, L.; Lowe, N.M. Zinc intake, status and indices of cognitive function in adults and children: A systematic review and meta-analysis. Eur. J. Clin. Nutr. 2015, 69, 649–661. [Google Scholar] [CrossRef]
- Fukunaka, A.; Fujitani, Y. Role of Zinc Homeostasis in the Pathogenesis of Diabetes and Obesity. Int. J. Mol. Sci. 2018, 19, 476. [Google Scholar] [CrossRef]
- Gu, K.; Xiang, W.; Zhang, Y.; Sun, K.; Jiang, X. The association between serum zinc level and overweight/obesity: A meta-analysis. Eur. J. Nutr. 2019, 58, 2971–2982. [Google Scholar] [CrossRef]
- Yerlikaya, F.H.; Toker, A.; Arıbaş, A. Serum trace elements in obese women with or without diabetes. Indian J. Med. Res. 2013, 137, 339–345. [Google Scholar] [PubMed]
- Payahoo, L.; Ostadrahimi, A.; Mobasseri, M.; Bishak, Y.K.; Farrin, N.; Jafarabadi, M.A.; Mahluji, S. Effects of zinc supplementation on the anthropometric measurements, lipid profiles and fasting blood glucose in the healthy obese adults. Adv. Pharm. Bull. 2013, 3, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Abdollahi, S.; Toupchian, O.; Jayedi, A.; Meyre, D.; Tam, V.; Soltani, S. Zinc Supplementation and Body Weight: A Systematic Review and Dose–Response Meta-analysis of Randomized Controlled Trials. Adv. Nutr. 2020, 11, 398–411. [Google Scholar] [CrossRef] [PubMed]
- Olechnowicz, J.; Tinkov, A.; Skalny, A.; Suliburska, J. Zinc status is associated with inflammation, oxidative stress, lipid, and glucose metabolism. J. Physiol. Sci. 2018, 68, 19. [Google Scholar] [CrossRef] [PubMed]
- Baltaci, A.K.; Mogulkoc, R.; Baltaci, S.B. Review: The role of zinc in the endocrine system. Pak. J. Pharm. Sci. 2019, 32, 231–239. [Google Scholar] [PubMed]
- Habib, S.A.; Saad, E.A.; Elsharkawy, A.A.; Attia, Z.R. Pro-inflammatory adipocytokines, oxidative stress, insulin, Zn and Cu: Interrelations with obesity in Egyptian non-diabetic obese children and adolescents. Adv. Med. Sci. 2015, 60, 179–185. [Google Scholar] [CrossRef]
- Salas-Venegas, V.; Flores-Torres, R.P.; Rodríguez-Cortés, Y.M.; Rodríguez-Retana, D.; Ramírez-Carreto, R.J.; Concepción-Carrillo, L.E.; Pérez-Flores, L.J.; Alarcón-Aguilar, A.; López-Díazguerrero, N.E.; Gómez-González, B.; et al. The Obese Brain: Mechanisms of Systemic and Local Inflammation, and Interventions to Reverse the Cognitive Deficit. Front. Integr. Neurosci. 2022, 16, 798995. [Google Scholar] [CrossRef]
- Quaye, E.; Galecki, A.T.; Tilton, N.; Whitney, R.; Briceño, E.M.; Elkind, M.S.V.; Fitzpatrick, A.L.; Gottesman, R.F.; Griswold, M.; Gross, A.L.; et al. Association of Obesity With Cognitive Decline in Black and White Americans. Neurology 2023, 100, e220–e231. [Google Scholar] [CrossRef]
- Liu, Z.; Yang, H.; Chen, S.; Cai, J.; Huang, Z. The association between body mass index, waist circumference, waist–hip ratio and cognitive disorder in older adults. J. Public Health 2019, 41, 305–312. [Google Scholar] [CrossRef]
- Tang, X.; Zhao, W.; Lu, M.; Zhang, X.; Zhang, P.; Xin, Z.; Sun, R.; Tian, W.; Cardoso, M.A.; Yang, J.; et al. Relationship between Central Obesity and the incidence of Cognitive Impairment and Dementia from Cohort Studies Involving 5,060,687 Participants. Neurosci. Biobehav. Rev. 2021, 130, 301–313. [Google Scholar] [CrossRef]
- Akintoye, O.; Ajibare, A.; Folawiyo, M.; Jimoh-Abdulghaffaar, H.; Asuku, A.; Owolabi, G.; Babalola, K. Zinc supplement reverses short-term memory deficit in sodium benzoate-induced neurotoxicity in male Wistar rats by enhancing anti-oxidative capacity via Nrf 2 up-regulation. Behav. Brain Res. 2023, 437, 114163. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, H.; Talebi, S.; Ghavami, A.; Rafiei, M.; Sharifi, S.; Faghihimani, Z.; Ranjbar, G.; Miraghajani, M.; Askari, G. Effects of zinc supplementation on inflammatory biomarkers and oxidative stress in adults: A systematic review and meta-analysis of randomized controlled trials. J. Trace Elem. Med. Biol. 2021, 68, 126857. [Google Scholar] [CrossRef]
- You, Y.X.; Shahar, S.; Rajab, N.F.; Haron, H.; Yahya, H.M.; Mohamad, M.; Din, N.C.; Maskat, M.Y. Effects of 12 Weeks Cosmos caudatus Supplement among Older Adults with Mild Cognitive Impairment: A Randomized, Double-Blind and Placebo-Controlled Trial. Nutrients 2021, 13, 434. [Google Scholar] [CrossRef] [PubMed]
- Chiappin, S.; Antonelli, G.; Gatti, R.; De Palo, E.F. Saliva specimen: A new laboratory tool for diagnostic and basic investigation. Clin. Chim. Acta 2007, 383, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Miranda, K.M.; Espey, M.G.; Wink, D.A. A Rapid, Simple Spectrophotometric Method for Simultaneous Detection of Nitrate and Nitrite. Nitric Oxide 2001, 5, 62–71. [Google Scholar] [CrossRef]
- de Oliveira, S.; Feijó, G.d.S.; Neto, J.; Jantsch, J.; Braga, M.F.; Castro, L.F.d.S.; Giovenardi, M.; Porawski, M.; Guedes, R.P. Zinc Supplementation Decreases Obesity-Related Neuroinflammation and Improves Metabolic Function and Memory in Rats. Obesity 2021, 29, 116–124. [Google Scholar] [CrossRef]
- Almeida, O.P. Mini exame dos estado mental e o diagnóstico de demência no Brasil. Arq. Neuropsiquiatr. 1998, 56, 605–612. [Google Scholar] [CrossRef]
- Shulman, K.I.; Pushkar Gold, D.; Cohen, C.A.; Zucchero, C.A. Clock-drawing and dementia in the community: A longitudinal study. Int. J. Geriatr. Psychiatry 1993, 8, 487–496. [Google Scholar] [CrossRef]
- Brucki, S.M.D.; Malheiros, S.M.F.; Okamoto, I.H.; Bertolucci, P.H.F. Dados normativos para o teste de fluência verbal categoria animais em nosso meio. Arq. Neuropsiquiatr. 1997, 55, 56–61. [Google Scholar] [CrossRef]
- Stroop, J.R. Studies of interference in serial verbal reactions. J. Exp. Psychol. 1935, 18, 643–662. [Google Scholar] [CrossRef]
- Beck, A.T.; Steer, R.A.; Brown, G.K. Beck Depression Inventory; Harcourt Brace Jovanovich: San Diego, CA, USA, 1987. [Google Scholar]
- Beck, A.T.; Epstein, N.; Brown, G.; Steer, R. Beck anxiety inventory. J. Consult. Clin. Psychol. 1993. [Google Scholar] [CrossRef]
- Kalantari, S.; Rounds, J.D.; Kan, J.; Tripathi, V.; Cruz-Garza, J.G. Comparing physiological responses during cognitive tests in virtual environments vs. in identical real-world environments. Sci. Rep. 2021, 11, 10227. [Google Scholar] [CrossRef] [PubMed]
- Frank, C.; John, P.S.; Molnar, F. Screening tools for virtual assessment of cognition. Can. Fam. Physician 2020, 66, 502–503. [Google Scholar]
- Jarosz, M.; Olbert, M.; Wyszogrodzka, G.; Młyniec, K.; Librowski, T. Antioxidant and anti-inflammatory effects of zinc. Zinc-dependent NF- j B signaling. Inflammopharmacology 2017, 25, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.S. Zinc is an antioxidant and anti-inflammatory agent: Its role in human health. Front. Nutr. 2014, 1, 14. [Google Scholar] [CrossRef]
- Rivers-Auty, J.; Tapia, V.S.; White, C.S.; Daniels, M.J.; Drinkall, S.; Kennedy, P.T.; Spence, H.G.; Yu, S.; Green, J.P.; Hoyle, C.; et al. Zinc Status Alters Alzheimer’s Disease Progression through NLRP3-Dependent Inflammation. J. Neurosci. 2021, 41, 3025–3038. [Google Scholar] [CrossRef]
- Lee, J.; Park, S. Serum zinc deficiency could be associated with dementia conversion in Parkinson’ s disease. Front. Aging Neurosci. 2023, 15, 1132907. [Google Scholar] [CrossRef]
- Xie, Z.; Wu, H.; Zhao, J. Multifunctional roles of zinc in Alzheimer’s disease. Nerotoxicology 2020, 80, 112–123. [Google Scholar] [CrossRef]
- Ghoreishy, S.M.; Ebrahimi Mousavi, S.; Asoudeh, F.; Mohammadi, H. Zinc status in attention-deficit/hyperactivity disorder: A systematic review and meta-analysis of observational studies. Sci. Rep. 2021, 11, 14612. [Google Scholar] [CrossRef]
- Petrilli, M.A.; Kranz, T.M.; Kleinhaus, K.; Joe, P.; Getz, M.; Johnson, P.; Chao, M.; Malaspina, D. The Emerging Role for Zinc in Depression and Psychosis. Front. Pharmacol. 2017, 8, 414. [Google Scholar] [CrossRef]
- Nam, S.M.; Kim, J.W.; Kwon, H.J.; Yoo, D.Y.; Jung, H.Y.; Kim, D.W.; Hwang, I.K.; Seong, J.K.; Yoon, Y.S. Differential Effects of Low- and High-dose Zinc Supplementation on Synaptic Plasticity and Neurogenesis in the Hippocampus of Control and High-fat Diet-fed Mice. Neurochem. Res. 2017, 42, 3149–3159. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Kumar, A.; Singh, K.; Avasthi, K.; Kim, J.-J. Neurobiology of zinc and its role in neurogenesis. Eur. J. Nutr. 2021, 60, 55–64. [Google Scholar] [CrossRef]
- Hafez, L.M.; Aboudeya, H.M.; Matar, N.A.; El-Sebeay, A.S.; Nomair, A.M.; El-Hamshary, S.A.; Nomeir, H.M.; Ibrahim, F.A.R. Ameliorative effects of zinc supplementation on cognitive function and hippocampal leptin signaling pathway in obese male and female rats. Sci. Rep. 2023, 13, 5072. [Google Scholar] [CrossRef] [PubMed]
- Solati, Z.; Jazayeri, S.; Tehrani-Doost, M.; Mahmoodianfard, S.; Gohari, M.R. Zinc monotherapy increases serum brain-derived neurotrophic factor (BDNF) levels and decreases depressive symptoms in overweight or obese subjects: A double-blind, randomized, placebo-controlled trial. Nutr. Neurosci. 2015, 18, 162–168. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Obesity an Overweight n.d. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 2 March 2023).
- Cook, R.L.; O’Dwyer, N.J.; Donges, C.E.; Parker, H.M.; Cheng, H.L.; Steinbeck, K.S.; Cox, E.P.; Franklin, J.L.; Garg, M.L.; Rooney, K.B.; et al. Relationship between Obesity and Cognitive Function in Young Women: The Food, Mood and Mind Study. J. Obes. 2017, 2017, 5923862. [Google Scholar] [CrossRef] [PubMed]
- Rochette, A.D.; Spitznagel, M.B.; Strain, G.; Devlin, M.; Crosby, R.D.; Mitchell, J.E.; Courcoulas, A.; Gunstad, J. Mild cognitive impairment is prevalent in persons with severe obesity. Obesity 2016, 24, 1427–1429. [Google Scholar] [CrossRef] [PubMed]
- van den Berg, E.; Kloppenborg, R.P.; Kessels, R.P.C.; Kappelle, L.J.; Biessels, G.J. Type 2 diabetes mellitus, hypertension, dyslipidemia and obesity: A systematic comparison of their impact on cognition. Biochim. Biophys. Acta-Mol. Basis Dis. 2009, 1792, 470–481. [Google Scholar] [CrossRef]
- Cheke, L.G.; Simons, J.S.; Clayton, N.S. Higher Body Mass Index is Associated with Episodic Memory Deficits in Young Adults. Q. J. Exp. Psychol. 2016, 69, 2305–2316. [Google Scholar] [CrossRef]
- Gómez-Apo, E.; Mondragón-Maya, A.; Ferrari-Díaz, M.S.-P.J. Structural Brain Changes Associated with Overweight and Obesity. J. Obes. 2021, 2021, 6613385. [Google Scholar] [CrossRef]
- Andrade, M.M.; Fernandes, C.; Forny-Germano, L.; A Gonçalves, R.; Gomes, M.; Castro-Fonseca, E.; Ramos-Lobo, A.M.; Tovar-Moll, F.; Andrade-Moraes, C.H.; Donato, J.; et al. Alteration in the number of neuronal and non-neuronal cells in mouse models of obesity. Brain Commun. 2023, 5, fcad059. [Google Scholar] [CrossRef]
- Guillemot-Legris, O.; Muccioli, G.G. Obesity-Induced Neuroinflammation: Beyond the Hypothalamus. Trends Neurosci. 2017, 40, 237–253. [Google Scholar] [CrossRef] [PubMed]
- Singh-Manoux, A.; Czernichow, S.; Elbaz, A.; Dugravot, A.; Sabia, S.; Hagger-Johnson, G.; Kaffashian, S.; Zins, M.; Brunner, E.J.; Nabi, H.; et al. Obesity phenotypes in midlife and cognition in early old age: The Whitehall II cohort study. Neurology 2012, 79, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Chávez-Manzanera, E.; Ramírez-Flores, M.; Duran, M.; Torres, M.; Ramírez, M.; Kaufer-Horwitz, M.; Stephano, S.; Quiroz-Casian, L.; Cantú-Brito, C.; Chiquete, E. Influence of Weight Loss on Cognitive Functions: A Pilot Study of a Multidisciplinary Intervention Program for Obesity Treatment. Brain Sci. 2022, 12, 509. [Google Scholar] [CrossRef] [PubMed]
- Khorsandi, H.; Nikpayam, O.; Yousefi, R.; Parandoosh, M.; Hosseinzadeh, N.; Saidpour, A.; Ghorbani, A. Zinc supplementation improves body weight management, inflammatory biomarkers and insulin resistance in individuals with obesity: A randomized, placebo-controlled, double-blind trial. Diabetol. Metab. Syndr. 2019, 11, 101. [Google Scholar] [CrossRef] [PubMed]
- Te, L.; Liu, J.; Ma, J.; Wang, S. Correlation between serum zinc and testosterone: A systematic review. J. Trace Elem. Med. Biol. 2023, 76, 127124. [Google Scholar] [CrossRef]
- Prasad, A.S.; Beck, F.W.; Bao, B.; Fitzgerald, J.T.; Snell, D.C.; Steinberg, J.D.; Cardozo, L.J. Zinc supplementation decreases incidence of infections in the elderly: Effect of zinc on generation of cytokines and oxidative stress. Am. J. Clin. Nutr. 2007, 85, 837–844. [Google Scholar] [CrossRef]
- Jaattela, M.; Mouritzen, H.; Elling, F.; Bastholm, L. A20 zinc finger protein inhibits TNF and IL-1 signaling. J. Immunol. 1996, 156, 1166–1173. [Google Scholar] [CrossRef]
- Driessen, C.; Hirv, K.; Kirchner, H.; Rink, L. Zinc regulates cytokine induction by superantigens and lipopolysaccharide. Immunology 1995, 84, 272–277. [Google Scholar]
- Squizani, S.; Jantsch, J.; Rodrigues, F.d.S.; Braga, M.F.; Eller, S.; de Oliveira, T.F.; Silveira, A.K.; Moreira, J.C.F.; Giovenardi, M.; Porawski, M.; et al. Zinc Supplementation Partially Decreases the Harmful Effects of a Cafeteria Diet in Rats but Does Not Prevent Intestinal Dysbiosis. Nutrients 2022, 14, 3921. [Google Scholar] [CrossRef]
- McNicholas, K.; François, M.; Liu, J.-W.; Doecke, J.D.; Hecker, J.; Faunt, J.; Maddison, J.; Johns, S.; Pukala, T.L.; Rush, R.A.; et al. Salivary inflammatory biomarkers are predictive of mild cognitive impairment and Alzheimer’s disease in a feasibility study. Front. Aging Neurosci. 2022, 14, 1019296. [Google Scholar] [CrossRef]
- Zyśk, B.; Ostrowska, L.; Smarkusz-Zarzecka, J. Salivary Adipokine and Cytokine Levels as Potential Markers for the Development of Obesity and Metabolic Disorders. Int. J. Mol. Sci. 2021, 22, 11703. [Google Scholar] [CrossRef] [PubMed]

| Placebo Group | Zinc Group | p-Value | |
|---|---|---|---|
| Age a | 51.1 ± 6.9 | 48.4 ± 6.0 | 0.224 |
| ≥12 years of education b n (%) | 6 (37.5) | 13 (65.0) | 0.101 |
| Body weight (kg) b | 87.56 ± 15.26 | 91.08 ± 15.45 | 0.499 |
| Height a | 1.62 ± 0.07 | 1.63 ± 0.05 | 0.616 |
| BMI (kg/m2) b | 33.70 ± 5.58 | 34.51 ± 6.56 | 0.698 |
| Physical activity (2 times/week) b n (%) | 2 (12.5) | 8 (40.0) | 0.133 |
| Hours of sleep a | 7.0 ± 1.0 | 6.8 ± 1.8 | 0.690 |
| Scores obtained in the Cognitive tests | |||
| MMSE b | 26.81 ± 1.11 | 27.60 ± 1.47 | 0.084 |
| TFV a | 18.94 ± 5.35 | 20.65 ± 4.76 | 0.317 |
| Clock test b | 8.60 ± 1.92 | 9.10 ± 1.21 | 0.352 |
| Stroop test b | 78.19 ± 10.77 | 77.85 ± 21.09 | 0.954 |
| BDI-II b | 12.55 ± 7.09 | 9.11 ± 6.34 | 0.187 |
| BECK a | 16.64 ± 13.73 | 11.53 ± 7.86 | 0.226 |
| Biomarkers b | |||
| Zinc (µg/dL) | 50.8 ± 9.8 | 49.5 ± 7.7 | 0.703 |
| Nitrite (μMol/L) | 9.5 ± 3.1 | 10.8 ± 4.2 | 0.190 |
| IL-1β (pg/mL) | 187.1 ± 162.7 | 150.3 ± 117.2 | 0.922 |
| IL-6 (pg/mL) | 18.4 ± 5.8 | 20.1 ± 6.3 | 0.337 |
| Insulin (pg/mL) | 387.4 ± 575.6 | 406.0 ± 344.7 | 0.076 |
| MCP-1 (pg/mL) | 519.8 ± 1061.2 | 2327.7 ± 1806.8 | 0.009 * |
| TNFα (pg/mL) | 3.20 ± 1.90 | 4.10 ± 3.7 | 0.909 |
| Food record b | |||
| Energy (kcal) | 1365.9 ± 323.6 | 1452.4 ± 417.4 | 0.547 |
| Carbohydrate (kcal) | 625.4 ± 201.7 | 675.4 ± 225.9 | 0.537 |
| Protein (kcal) | 280.3 ± 46.9 | 299.8 ± 79.5 | 0.450 |
| Fat (kcal) | 457.9 ± 144.2 | 455.5 ± 196.9 | 0.971 |
| Zinc (mg) | 7.12 ± 1.83 | 7.01 ± 2.3 | 0.0893 |
| Group | Adjusted Mean | CI 95% | Δ | CI 95% Δ | p-Value | |
|---|---|---|---|---|---|---|
| Weight (kg) | Placebo | 87.69 | (85.72–89.65) | −0.300 | (−2.79–2.19) | 0.807 |
| Zinc | 87.39 | (85.87–88.90) | ||||
| BMI (kg/m2) | Placebo | 33.26 | (32.47–34.05) | −0.026 | (−1.02–0.97) | 0.958 |
| Zinc | 33.23 | (32.62–33.84) | ||||
| Energy (kcal) | Placebo | 1245.70 | (1036.95–1454.44) | 145.34 | (−118.1–408.7) | 0.265 |
| Zinc | 1391.03 | (1235.64–1546.43) | ||||
| Carbohydrate (kcal) | Placebo | 547.67 | (457.49–637.84) | 75.75 | (−38.40–189.90) | 0.183 |
| Zinc | 623.41 | (556.46–690.37) | ||||
| Protein (kcal) | Placebo | 270.02 | (215.41–324.63) | 22.46 | (−46.08–91.00) | 0.504 |
| Zinc | 292.48 | (251.65–333.31) | ||||
| Fat (kcal) | Placebo | 378.60 | (280.83–476.37) | 48.66 | (−73.63–170.96) | 0.418 |
| Zinc | 427.26 | (353.97–500.55) | ||||
| Zinc (mg) | Placebo | 6.47 | (4.98–7.97) | 1.13 | (−0.736–3.01) | 0.222 |
| Zinc | 7.61 | (6.49–8.73) |
| Group | Adjusted Mean | CI 95% | Δ | CI 95% Δ | p-Value | |
|---|---|---|---|---|---|---|
| MMSE | Placebo | 27.57 | (26.91–28.24) | 0.885 * | (0.03–1.74) | 0.044 * |
| Zinc | 28.46 | (27.95–28.97) | ||||
| VFT | Placebo | 21.23 | (19.23–23.24) | 0.877 | (−1.69–3.44) | 0.490 |
| Zinc | 22.11 | (20.57–23.65) | ||||
| Clock Test | Placebo | 9.20 | (8.76–9.64) | 0.283 | (−0.27–0.84) | 0.306 |
| Zinc | 9.48 | (9.14–9.82) | ||||
| Stroop test a | Placebo | 68.13 | (64.39–71.87) | −6.156 | (−10.90–−1.42) | 0.013 * |
| Zinc | 61.97 | (59.08–64.86) | ||||
| BDI-II a | Placebo | 12.86 | (9.85–15.88) | 2.276 | (1.89–0.24) | 0.239 |
| Zinc | 15.14 | (12.80–17.48) | ||||
| Beck scale a | Placebo | 12.59 | (7.08–18.10) | −2.340 | (−9.37–4.69) | 0.500 |
| Zinc | 10.25 | (5.95–14.55) |
| Group | Adjusted Mean | CI 95% | Δ | CI 95% Δ | p-Value | |
|---|---|---|---|---|---|---|
| IL-1β (pg/mL) | Placebo | 84.38 | (20.76–148.01) | 118.234 * | (32.06–204.41) | 0.013 * |
| Zinc | 202.62 | (144.54–260.69) | ||||
| IL-6 (pg/mL) | Placebo | 18.88 | (16.32–21.43) | −2.271 | (−5.53–0.99) | 0.163 |
| Zinc | 16.60 | (14.58–18.62) | ||||
| Insulin (pg/mL) | Placebo | 308.07 | (90.40–525.73) | 132.579 | (−211.92–477.08) | 0.393 |
| Zinc | 440.65 | (173.98–707.318) | ||||
| MCP-1 (pg/mL) | Placebo | 332.23 | (164.90–499.55) | −2.277 | (−223.87–−219.31) | 0.983 |
| Zinc | 329.95 | (199.23–460.66) | ||||
| TNFα (pg/mL) | Placebo | 3.58 | (1.81–5.35) | 0.223 | (−2.03–2.48) | 0.840 |
| Zinc | 3.80 | (2.41–5.20) | ||||
| Nitrite (μMol/L) | Placebo | 12.95 | (8.96–16.95) | −1.078 | (−6.10–−3.95) | 0.663 |
| Zinc | 11.88 | (8.84–14.91) | ||||
| Zinc (µg/dL) | Placebo | 35.37 | (26.95–43.78) | 11.678 | (0.71–22.65) | 0.038 * |
| Zinc | 47.05 | (40.05–54.04) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Vargas, L.d.S.; Jantsch, J.; Fontoura, J.R.; Dorneles, G.P.; Peres, A.; Guedes, R.P. Effects of Zinc Supplementation on Inflammatory and Cognitive Parameters in Middle-Aged Women with Overweight or Obesity. Nutrients 2023, 15, 4396. https://doi.org/10.3390/nu15204396
de Vargas LdS, Jantsch J, Fontoura JR, Dorneles GP, Peres A, Guedes RP. Effects of Zinc Supplementation on Inflammatory and Cognitive Parameters in Middle-Aged Women with Overweight or Obesity. Nutrients. 2023; 15(20):4396. https://doi.org/10.3390/nu15204396
Chicago/Turabian Stylede Vargas, Liziane da Silva, Jeferson Jantsch, Juliana Ribeiro Fontoura, Gilson Pires Dorneles, Alessandra Peres, and Renata Padilha Guedes. 2023. "Effects of Zinc Supplementation on Inflammatory and Cognitive Parameters in Middle-Aged Women with Overweight or Obesity" Nutrients 15, no. 20: 4396. https://doi.org/10.3390/nu15204396
APA Stylede Vargas, L. d. S., Jantsch, J., Fontoura, J. R., Dorneles, G. P., Peres, A., & Guedes, R. P. (2023). Effects of Zinc Supplementation on Inflammatory and Cognitive Parameters in Middle-Aged Women with Overweight or Obesity. Nutrients, 15(20), 4396. https://doi.org/10.3390/nu15204396






