Improving Adherence to the Mediterranean Diet in Early Pregnancy Using a Clinical Decision Support System; A Randomised Controlled Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics and Participants
2.2. Study Design
2.3. Screening and Assessments
2.4. Statistical Analysis and Primary Outcomes
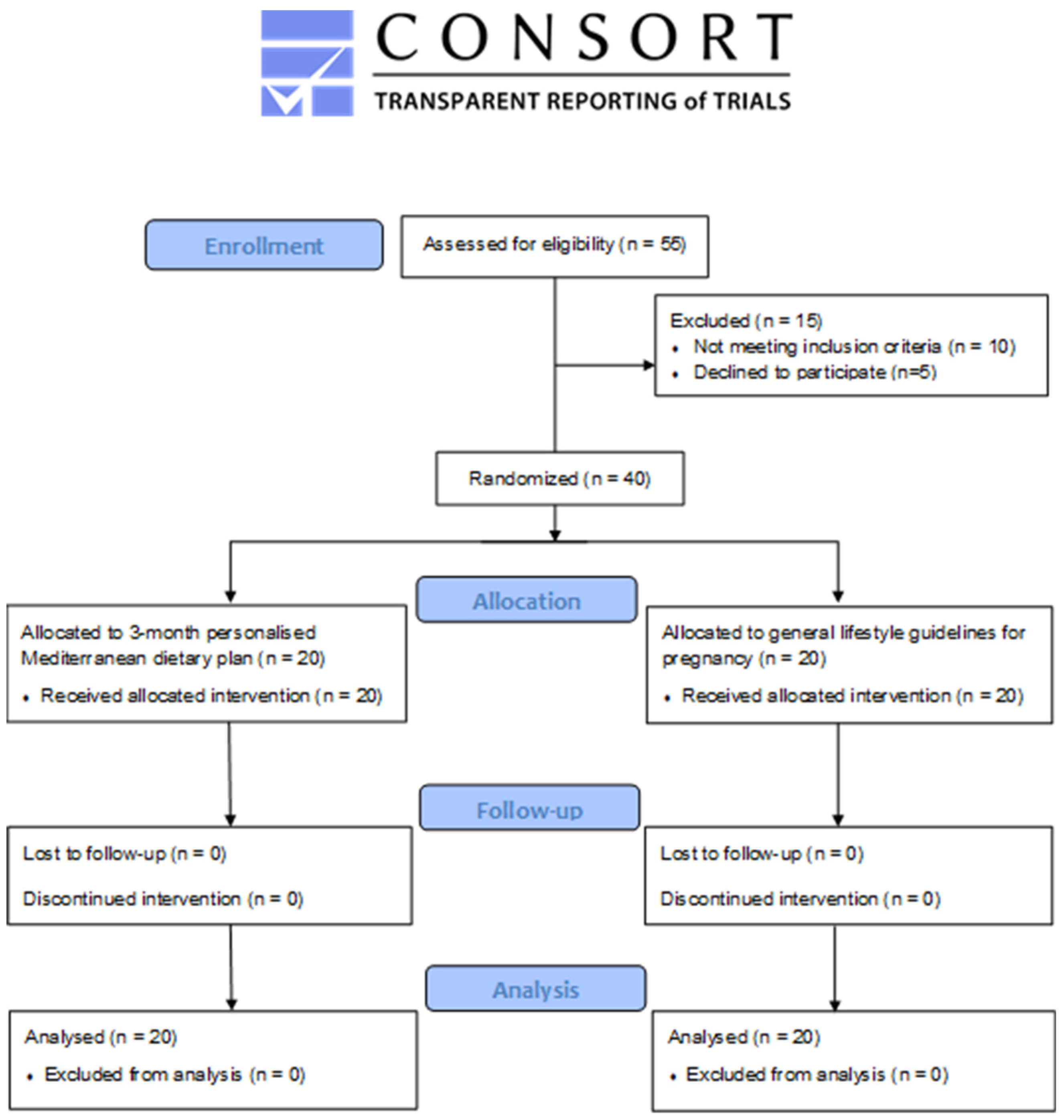
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chen, X.; Zhao, D.; Mao, X.; Xia, Y.; Baker, P.N.; Zhang, H. Maternal Dietary Patterns and Pregnancy Outcome. Nutrients 2016, 8, 351. [Google Scholar] [CrossRef]
- Abu-Saad, K.; Fraser, D. Maternal Nutrition and Birth Outcomes. Epidemiol. Rev. 2010, 32, 5–25. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Schulze, M.B.; Solomon, C.G.; Hu, F.B. A prospective study of dietary patterns, meat intake and the risk of gestational diabetes mellitus. Diabetologia 2006, 49, 2604–2613. [Google Scholar] [CrossRef]
- Kampmann, U. Gestational diabetes: A clinical update. World J. Diabetes 2015, 6, 1065. [Google Scholar] [CrossRef]
- Rasmussen, M.A.; Maslova, E.; Halldórsson, T.I.; Olsen, S.F. Characterization of Dietary Patterns in the Danish National Birth Cohort in Relation to Preterm Birth. PLoS ONE 2014, 9, e93644. [Google Scholar] [CrossRef]
- Hajianfar, H.; Esmaillzadeh, A.; Feizi, A.; Shahshahan, Z.; Azadbakht, L. Major Maternal Dietary Patterns during Early Pregnancy and Their Association with Neonatal Anthropometric Measurement. BioMed Res. Int. 2018, 2018, 4692193. [Google Scholar] [CrossRef]
- Yajnik, C.S. Early Life Origins of Insulin Resistance and Type 2 Diabetes in India and Other Asian Countries. J. Nutr. 2004, 134, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Agnoux, A.M.; Antignac, J.-P.; Simard, G.; Poupeau, G.; Darmaun, D.; Parnet, P.; Alexandre-Gouabau, M.-C. Time window-dependent effect of perinatal maternal protein restriction on insulin sensitivity and energy substrate oxidation in adult male offspring. Am. J. Physiol. Integr. Comp. Physiol. 2014, 307, R184–R197. [Google Scholar] [CrossRef]
- Pereira, T.J.; Moyce, B.L.; Kereliuk, S.M.; Dolinsky, V.W. Influence of maternal overnutrition and gestational diabetes on the programming of metabolic health outcomes in the offspring: Experimental evidence. Biochem. Cell Biol. 2015, 93, 438–451. [Google Scholar] [CrossRef]
- Portrait, F.; Teeuwiszen, E.; Deeg, D. Early life undernutrition and chronic diseases at older ages: The effects of the Dutch famine on cardiovascular diseases and diabetes. Soc. Sci. Med. 2011, 73, 711–718. [Google Scholar] [CrossRef]
- Toemen, L.; Gishti, O.; van Osch-Gevers, L.; Steegers, E.A.P.; Helbing, W.A.; Felix, J.F.; Reiss, I.K.M.; Duijts, L.; Gaillard, R.; Jaddoe, V.W.V. Maternal obesity, gestational weight gain and childhood cardiac outcomes: Role of childhood body mass index. Int. J. Obes. 2016, 40, 1070–1078. [Google Scholar] [CrossRef] [PubMed]
- Venter, C.; Brown, K.R.; Maslin, K.; Palmer, D.J. Maternal dietary intake in pregnancy and lactation and allergic disease outcomes in offspring. Pediatr. Allergy Immunol. 2017, 28, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Erkkola, M.; Nwaru, B.I.; Kaila, M.; Kronberg-Kippilä, C.; Ilonen, J.; Simell, O.; Veijola, R.; Knip, M.; Virtanen, S.M. Risk of asthma and allergic outcomes in the offspring in relation to maternal food consumption during pregnancy: A Finnish birth cohort study. Pediatr. Allergy Immunol. 2012, 23, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Devereux, G.; Seaton, A. Diet as a risk factor for atopy and asthma. J. Allergy Clin. Immunol. 2005, 115, 1109–1117. [Google Scholar] [CrossRef]
- Amati, F.; Hassounah, S.; Swaka, A. The Impact of Mediterranean Dietary Patterns During Pregnancy on Maternal and Offspring Health. Nutrients 2019, 11, 1098. [Google Scholar] [CrossRef]
- Assaf-Balut, C.; de la Torre, N.G.; Fuentes, M.; Durán, A.; Bordiú, E.; del Valle, L.; Valerio, J.; Jiménez, I.; Herraiz, M.A.; Izquierdo, N.; et al. A High Adherence to Six Food Targets of the Mediterranean Diet in the Late First Trimester is Associated with a Reduction in the Risk of Materno-Foetal Outcomes: The St. Carlos Gestational Diabetes Mellitus Prevention Study. Nutrients 2018, 11, 66. [Google Scholar] [CrossRef]
- Willett, W.C.; Sacks, F.; Trichopoulou, A.; Drescher, G.; Ferro-Luzzi, A.; Helsing, E.; Trichopoulos, D. Mediterranean diet pyramid: A cultural model for healthy eating. Am. J. Clin. Nutr. 1995, 61 (Suppl. 6), 1402S–1406S. [Google Scholar] [CrossRef]
- Panagiotakos, D.B.; Pitsavos, C.; Stefanadis, C. Dietary patterns: A Mediterranean diet score and its relation to clinical and biological markers of cardiovascular disease risk. Nutr. Metab. Cardiovasc. Dis. 2006, 16, 559–568. [Google Scholar] [CrossRef]
- Bach, A.; Serra-Majem, L.; Carrasco, J.L.; Roman, B.; Ngo, J.; Bertomeu, I.; Obrador, B. The use of indexes evaluating the adherence to the Mediterranean diet in epidemiological studies: A review. Public Health Nutr. 2006, 9, 132–146. [Google Scholar] [CrossRef]
- Spadafranca, A.; Piuri, G.; Bulfoni, C.; Liguori, I.; Battezzati, A.; Bertoli, S.; Speciani, A.F.; Ferrazzi, E. Adherence to the Mediterranean Diet and Serum Adiponectin Levels in Pregnancy: Results from a Cohort Study in Normal Weight Caucasian Women. Nutrients 2018, 10, 928. [Google Scholar] [CrossRef]
- Shortliffe, E.H.; Buchanan, B.G. A model of inexact reasoning in medicine. Math. Biosci. 1975, 23, 351–379. [Google Scholar] [CrossRef]
- Wyatt, J.; Spiegelhalter, D. Field trials of medical decision-aids: Potential problems and solutions. Proc. Annu. Symp. Comput. Appl. Med. Care 1991, 3–7. [Google Scholar]
- Dias, D.; Paulo Silva Cunha, J. Wearable Health Devices—Vital Sign Monitoring, Systems and Technologies. Sensors 2018, 18, 2414. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.X.; Adhikari, N.K.J.; McDonald, H.; Rosas-Arellano, M.P.; Devereaux, P.J.; Beyene, J.; Sam, J.; Haynes, R.B. Effects of Computerized Clinical Decision Support Systems on Practitioner Performance and Patient Outcomes. JAMA 2005, 293, 1223–1238. [Google Scholar] [CrossRef] [PubMed]
- Papandreou, P.; Gioxari, A.; Daskalou, E.; Vasilopoulou, A.; Skouroliakou, M. Personalized Nutritional Intervention to Improve Mediterranean Diet Adherence in Female Patients with Multiple Sclerosis: A Randomized Controlled Study. Dietetics 2022, 1, 25–38. [Google Scholar] [CrossRef]
- Gioxari, A.; Tzanos, D.; Kostara, C.; Papandreou, P.; Mountzios, G.; Skouroliakou, M. Mediterranean Diet Implementation to Protect against Advanced Lung Cancer Index (ALI) Rise: Study Design and Preliminary Results of a Randomised Controlled Trial. Int. J. Environ. Res. Public Health 2021, 18, 3700. [Google Scholar] [CrossRef]
- Papandreou, P.; Gioxari, A.; Nimee, F.; Skouroliakou, M. Application of Clinical Decision Support System to Assist Breast Cancer Patients with Lifestyle Modifications during the COVID-19 Pandemic: A Randomised Controlled Trial. Nutrients 2021, 13, 2115. [Google Scholar] [CrossRef]
- Nimee, F.; Gioxari, A.; Steier, J.; Skouroliakou, M. Bridging the Gap: Community Pharmacists’ Burgeoning Role as Point-of-Care Providers during the COVID-19 Pandemic Through the Integration of Emerging Technologies. J. Nutr. Health Food Sci. 2021, 9, 1–9. [Google Scholar] [CrossRef]
- National Dietary Guidelines for Women, Including Women in Pregnancy, Lactation & Menopause. Available online: http://www.diatrofikoiodigoi.gr/?page=summary-women (accessed on 3 November 2022).
- Katsouyanni, K.; Rimm, E.B.; Gnardellis, C.; Trichopoulos, D.; Polychronopoulos, E.; Trichopoulou, A. Reproducibility and relative validity of an extensive semi-quantitative food frequency questionnaire using dietary records and biochemical markers among Greek schoolteachers. Int. J. Epidemiol. 1997, 26, S118–S127. [Google Scholar] [CrossRef]
- Zigmond, A.S.; Snaith, R.P. The Hospital Anxiety and Depression Scale. Acta Psychiatr. Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef]
- WHO. Global Database on Body Mass Index (BMI). Available online: http://www.who.int/nutrition/databases/bmi/en (accessed on 26 April 2021).
- Zaragoza-Martí, A.; Ruiz-Ródenas, N.; Herranz-Chofre, I.; Sánchez-SanSegundo, M.; Delgado, V.d.l.C.S.; Hurtado-Sánchez, J.A. Adherence to the Mediterranean Diet in Pregnancy and Its Benefits on Maternal-Fetal Health: A Systematic Review of the Literature. Front. Nutr. 2022, 9, 813942. [Google Scholar] [CrossRef] [PubMed]
- Minhas, A.S.; Hong, X.; Wang, G.; Rhee, D.K.; Liu, T.; Zhang, M.; Michos, E.D.; Wang, X.; Mueller, N.T. Mediterranean-Style Diet and Risk of Preeclampsia by Race in the Boston Birth Cohort. J. Am. Heart Assoc. 2022, 11, e022589. [Google Scholar] [CrossRef]
- Usmanova, G.; Lalchandani, K.; Srivastava, A.; Joshi, C.S.; Bhatt, D.C.; Bairagi, A.K.; Jain, Y.; Afzal, M.; Dhoundiyal, R.; Benawri, J.; et al. The role of digital clinical decision support tool in improving quality of intrapartum and postpartum care: Experiences from two states of India. BMC Pregnancy Childbirth 2021, 21, 278. [Google Scholar] [CrossRef]
- Assaf-Balut, C.; de la Torre, N.G.; Durán, A.; Fuentes, M.; Bordiú, E.; Del Valle, L.; Valerio, J.; Familiar, C.; Jiménez, I.; Herraiz, M.A.; et al. Medical nutrition therapy for gestational diabetes mellitus based on Mediterranean Diet principles: A subanalysis of the St Carlos GDM Prevention Study. BMJ Open Diabetes Res. Care 2018, 6, e000550. [Google Scholar] [CrossRef]
- Al Wattar, B.H.; Dodds, J.; Placzek, A.; Beresford, L.; Spyreli, E.; Moore, A.; Carreras, F.J.G.; Austin, F.; Murugesu, N.; Roseboom, T.J.; et al. Mediterranean-style diet in pregnant women with metabolic risk factors (ESTEEM): A pragmatic multicentrerandomised trial. PLOS Med. 2019, 16, e1002857. [Google Scholar] [CrossRef] [PubMed]
- Flor-Alemany, M.; Nestares, T.; Alemany-Arrebola, I.; Marín-Jiménez, N.; Borges-Cosic, M.; Aparicio, V.A. Influence of Dietary Habits and Mediterranean Diet Adherence on Sleep Quality during Pregnancy. The GESTAFIT Project. Nutrients 2020, 12, 3569. [Google Scholar] [CrossRef] [PubMed]
- Chatzi, L.; Rifas-Shiman, S.L.; Georgiou, V.; Joung, K.E.; Koinaki, S.; Chalkiadaki, G.; Margioris, A.; Sarri, K.; Vassilaki, M.; Vafeiadi, M.; et al. Adherence to the Mediterranean diet during pregnancy and offspring adiposity and cardiometabolic traits in childhood. Pediatr. Obes. 2017, 12, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Melero, V.; Assaf-Balut, C.; De La Torre, N.G.; Jiménez, I.; Bordiú, E.; Del Valle, L.; Valerio, J.; Familiar, C.; Durán, A.; Runkle, I.; et al. Benefits of Adhering to a Mediterranean Diet Supplemented with Extra Virgin Olive Oil and Pistachios in Pregnancy on the Health of Offspring at 2 Years of Age. Results of the San Carlos Gestational Diabetes Mellitus Prevention Study. J. Clin. Med. 2020, 9, 1454. [Google Scholar] [CrossRef] [PubMed]
- Chatzi, L.; Torrent, M.; Romieu, I.; Garcia-Esteban, R.; Ferrer, C.; Vioque, J.; Kogevinas, M.; Sunyer, J. Mediterranean diet in pregnancy is protective for wheeze and atopy in childhood. Thorax 2008, 63, 507–513. [Google Scholar] [CrossRef] [PubMed]
- Schetter, C.D.; Tanner, L. Anxiety, depression and stress in pregnancy: Implications for mothers, children, research, and practice. Curr. Opin. Psychiatry 2012, 25, 141–148. [Google Scholar] [CrossRef]
- Rubertsson, C.; Hellström, J.; Cross, M.; Sydsjö, G. Anxiety in early pregnancy: Prevalence and contributing factors. Arch. Women’s Ment. Health 2014, 17, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Matthey, S.; Ross-Hamid, C. Repeat testing on the Edinburgh Depression Scale and the HADS-A in pregnancy: Differentiating between transient and enduring distress. J. Affect. Disord. 2012, 141, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Flor-Alemany, M.; Migueles, J.H.; Alemany-Arrebola, I.; Aparicio, V.A.; Baena-García, L. Exercise, Mediterranean Diet Adherence or Both during Pregnancy to Prevent Postpartum Depression—GESTAFIT Trial Secondary Analyses. Int. J. Environ. Res. Public Health 2022, 19, 14450. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Villegas, A.; Delgado-Rodríguez, M.; Alonso, A.; Schlatter, J.; Lahortiga, F.; Majem, L.S.; Martínez-González, M.A. Association of the Mediterranean dietary pattern with the incidence of depression: The Seguimiento Universidad de Navarra/University of Navarra follow-up (SUN) cohort. Arch. Gen. Psychiatry 2009, 66, 1090–1098. [Google Scholar] [CrossRef] [PubMed]
- Altun, A.; Brown, H.; Szoeke, C.; Goodwill, A.M. The Mediterranean dietary pattern and depression risk: A systematic review. Neurol. Psychiatry Brain Res. 2019, 33, 1–10. [Google Scholar] [CrossRef]
- Sánchez-Villegas, A.; Martínez-González, M.A.; Estruch, R.; Salas-Salvadó, J.; Corella, D.; Covas, M.I.; Arós, F.; Romaguera, D.; Gómez-Gracia, E.; Lapetra, J.; et al. Mediterranean dietary pattern and depression: The PREDIMED randomized trial. BMC Med. 2013, 11, 208. [Google Scholar] [CrossRef] [PubMed]
- Parletta, N.; Zarnowiecki, D.; Cho, J.; Wilson, A.; Bogomolova, S.; Villani, A.; Itsiopoulos, C.; Niyonsenga, T.; Blunden, S.; Meyer, B.; et al. A Mediterranean-style dietary intervention supplemented with fish oil improves diet quality and mental health in people with depression: A randomized controlled trial (HELFIMED). Nutr. Neurosci. 2017, 22, 474–487. [Google Scholar] [CrossRef] [PubMed]
| Inclusion Criteria | Exclusion Criteria |
|---|---|
| Adult women (≥18 years old) in the first trimester of pregnancy. | Adolescent pregnancy. |
| Women in the second semester of pregnancy. | |
| - Without pregnancy complications. | Women with pregnancy complications, e.g., infections, hypertension, pre-eclampsia, gestational diabetes. |
| - Without medical conditions before pregnancy. | Women with allergies or food intolerances. |
| - Who provided signed participation consent. | Women with pre-pregnancy chronic diseases (e.g., malignancy, cardiovascular diseases), inherited metabolic diseases, malabsorption, or cognitive disorders. |
| Women with psychiatric conditions. | |
| Women with alcoholism or drug addiction. | |
| Women following a vegan or macrobiotic diet ≤ 5 years prior to intervention. | |
| Women with vitamin or mineral supplementation ≤ 6 months prior to fetus conception. | |
| Inability to read and understand the consent information. |
| Characteristics | Enrolled Pregnant Participants (n = 40) | Control Group (n = 20) | CDSS Group (n = 20) | p-Value |
|---|---|---|---|---|
| Age (years) mean (SD) | 30.8 (6.4) | 29.1 (6.1) | 32.5 (6.4) | 0.099 |
| Pre-pregnancy BMI (kg/m2) n (%) | ||||
| <18.5 | 1 (2.5) | 1 (5.0) | 0 (0.0) | 0.549 |
| 18.5–24.9 | 34 (85.0) | 17 (85.0) | 17 (85.0) | |
| 25–29.9 | 5 (12.5) | 2 (10.0) | 3 (15.0) | |
| >30 | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| Fat (%) mean (SD) | 29.4 (6.9) | 29.2 (6.9) | 29.6 (7.0) | 0.862 |
| Fat Free Mass (%) mean (SD) | 70.6 (6.9) | 70.8 (6.9) | 70.4 (7.0) | 0.862 |
| Profession, n (%) | 0.060 | |||
| Private sector | 13 (32.5) | 5 (25.0) | 8 (40.0) | |
| State employee | 4 (10.0) | 1 (5.0) | 3 (15.0) | |
| Housewifery | 14 (35.0) | 6 (30.0) | 8 (40.0) | |
| Other | 9 (22.5) | 8 (40.0) | 1 (5.0) | |
| Smoking before pregnancy, n (%) | 0.901 | |||
| No | 21 (52.5) | 11 (55.0) | 10 (50.0) | |
| Yes | 10 (25.0) | 5 (25.0) | 5 (25.0) | |
| N/A | 9 (22.5) | 4 (20.0 | 5 (25.0) | |
| Alcohol consumption | 0.962 | |||
| No | 29 (72.5) | 15 (75.0) | 14 (70.0) | |
| Yes | 2 (5.0) | 1 (5.0) | 1 (5.0) | |
| N/A | 9 (22.5) | 4 (20.0) | 5 (25.0) | |
| HADS: anxiety n (%) | ||||
| 0–7 (%) | 16 (40.0) | 7 (35.0) | 9 (45.0) | 0.807 |
| 8–10 (%) | 9 (22.5) | 5 (25.0) | 4 (20.0) | |
| 11–21 (%) | 15 (37.5) | 8 (40.0) | 7 (35.0) | |
| HADS: depression n (%) | - | |||
| 0–7 (%) | 40 (100.0) | 20 (0.0) | 20 (0.0) | |
| 8–10 (%) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| 11–21 (%) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Characteristics | Group | Baseline (n = 20) | Follow-Up (n = 20) | p2 | p3 |
|---|---|---|---|---|---|
| Body weight (kg) mean (SD) | control | 64.3 (8.3) | 67.8 (9.0) | <0.001 | 0.941 |
| CDSS | 64.3 (8.1) | 68.2 (7.9) | <0.001 | ||
| p1 | 1.000 | 0.885 | |||
| Fat (%) mean (SD) | control | 29.2 (6.9) | 31.9 (7.5) | <0.001 | 0.640 |
| CDSS | 29.6 (7.0) | 29.4 (7.1) | 0.431 | ||
| p1 | 0.862 | 0.289 | |||
| Triacylglycerols (mg/dL) a median (IQR) | control | 100.0 (51.0) | 89.5 (69.5) | 0.552 | 0.341 |
| CDSS | 67.0 (52.0) | 59.0 (42.5) | 0.001 | ||
| p1 | 0.435 | 0.061 | |||
| Glucose (mg/dL) mean (SD) | control | 97.1 (21.3) | 95.4 (20.1) | 0.212 | 0.679 |
| CDSS | 101.3 (6.8) | 95.2 (4.8) | <0.001 | ||
| p1 | 0.415 | 0.955 | |||
| Total Cholesterol (mg/dL) mean (SD) | control | 192.6 (38.3) | 195.0 (37.4) | 0.687 | 0.127 |
| CDSS | 182.7 (41.0) | 167.9 (39.6) | 0.002 | ||
| p1 | 0.435 | 0.032 | |||
| LDL (mg/dL) mean (SD) | control | 114.7 (33.3) | 115.5 (34.7) | 0.870 | 0.702 |
| CDSS | 115.9 (40.7) | 105.8 (35.3) | 0.002 | ||
| p1 | 0.923 | 0.387 | |||
| HDL (mg/dL) a median (IQR) | control | 55.0 (15.0) | 58.0 (26.8) | 0.095 | 0.433 |
| CDSS | 63.0 (15.0) | 71.0 (9.8) | 0.035 | ||
| p1 | 0.174 | 0.583 | |||
| CRP (m/L) a median (IQR) | control | 0.8 (0.2) | 0.7 (0.2) | 0.082 | 0.096 |
| CDSS | 0.7 (0.1) | 0.7 (0.1) | 0.705 | ||
| p1 | 0.086 | 0.478 |
| Characteristics | Group | Baseline (n = 20) | Follow-Up (n = 20) | p2 | p3 |
|---|---|---|---|---|---|
| MetDietScore a median (IQR) | control | 32.0 (3.8) | 34.0 (2.0) | 0.007 | <0.001 |
| CDSS | 35.0 (3.8) | 38.0 (2.8) | <0.001 | ||
| p1 | <0.001 | <0.001 | |||
| Fibre Intake (g) mean (SD) | control | 20.1 (3.8) | 20.9 (3.4) | 0.075 | 0.978 |
| CDSS | 17.9 (3.8) | 23.2 (4.4) | <0.001 | ||
| p1 | 0.072 | 0.074 | |||
| Protein Intake (%) a median (IQR) | control | 18.0 (5.0) | 19.5 (2.8) | 0.063 | 0.713 |
| CDSS | 21.0 (4.0) | 20.0 (3.0) | 0.503 | ||
| p1 | 0.231 | 0.355 | |||
| Carbohydrates (%) a median (IQR) | control | 54.0 (8.0) | 52.0 (5.8) | 0.777 | 0.374 |
| CDSS | 54.0 (6.0) | 52.0 (3.5) | 0.023 | ||
| p1 | 0.429 | 0.583 | |||
| Fat Intake (%) a median (IQR) | control | 29.0 (4.0) | 29.0 (3.0) | 0.419 | 0.051 |
| CDSS | 24.0 (6.0) | 28.0 (3.8) | 0.009 | ||
| p1 | 0.011 | 0.779 | |||
| Energy Intake (kcal) a median (IQR) | control | 2300.0 (800.0) | 2375.0 (900.0) | 0.715 | <0.001 |
| CDSS | 2000.0 (100.0) | 2000.0 (150.0) | 0.707 | ||
| p1 | <0.001 | 0.017 |
| Characteristics | Group | Baseline (n = 20) | Follow-Up (n = 20) | p2 | p3 |
|---|---|---|---|---|---|
| HADS (anxiety) mean (SD) | control | 8.7 (4.3) | 7.1 (3.1) | 0.036 | 0.048 |
| CDSS | 8.2 (4.2) | 3.5 (2.2) | <0.001 | ||
| p1 | 0.711 | <0.001 | |||
| HADS (depression) a median (IQR) | control | 3.0 (2.8) | 1.0 (1.0) | 0.002 | 0.006 |
| CDSS | 3.0 (5.5) | 3.0 (4.5) | 0.054 | ||
| p1 | 0.968 | 0.192 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Papandreou, P.; Amerikanou, C.; Vezou, C.; Gioxari, A.; Kaliora, A.C.; Skouroliakou, M. Improving Adherence to the Mediterranean Diet in Early Pregnancy Using a Clinical Decision Support System; A Randomised Controlled Clinical Trial. Nutrients 2023, 15, 432. https://doi.org/10.3390/nu15020432
Papandreou P, Amerikanou C, Vezou C, Gioxari A, Kaliora AC, Skouroliakou M. Improving Adherence to the Mediterranean Diet in Early Pregnancy Using a Clinical Decision Support System; A Randomised Controlled Clinical Trial. Nutrients. 2023; 15(2):432. https://doi.org/10.3390/nu15020432
Chicago/Turabian StylePapandreou, Panos, Charalampia Amerikanou, Chara Vezou, Aristea Gioxari, Andriana C. Kaliora, and Maria Skouroliakou. 2023. "Improving Adherence to the Mediterranean Diet in Early Pregnancy Using a Clinical Decision Support System; A Randomised Controlled Clinical Trial" Nutrients 15, no. 2: 432. https://doi.org/10.3390/nu15020432
APA StylePapandreou, P., Amerikanou, C., Vezou, C., Gioxari, A., Kaliora, A. C., & Skouroliakou, M. (2023). Improving Adherence to the Mediterranean Diet in Early Pregnancy Using a Clinical Decision Support System; A Randomised Controlled Clinical Trial. Nutrients, 15(2), 432. https://doi.org/10.3390/nu15020432








