Biometabolites of Citrus unshiu Peel Enhance Intestinal Permeability and Alter Gut Commensal Bacteria
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Mice and Natural Products
2.3. Extraction of CUP
2.4. Component Extraction from Natural Products
2.5. Antibodies and Reagents
2.6. DSS-Induced Colitis
2.7. Histological Analysis
2.8. cDNA Synthesis and Real-Time PCR
2.9. Cell Isolation and Analysis
2.10. Mucin2 (MUC2) Immunofluorescence Staining
2.11. Immunoblotting
2.12. Microbiome Analysis
2.13. Statistical Analysis
3. Results
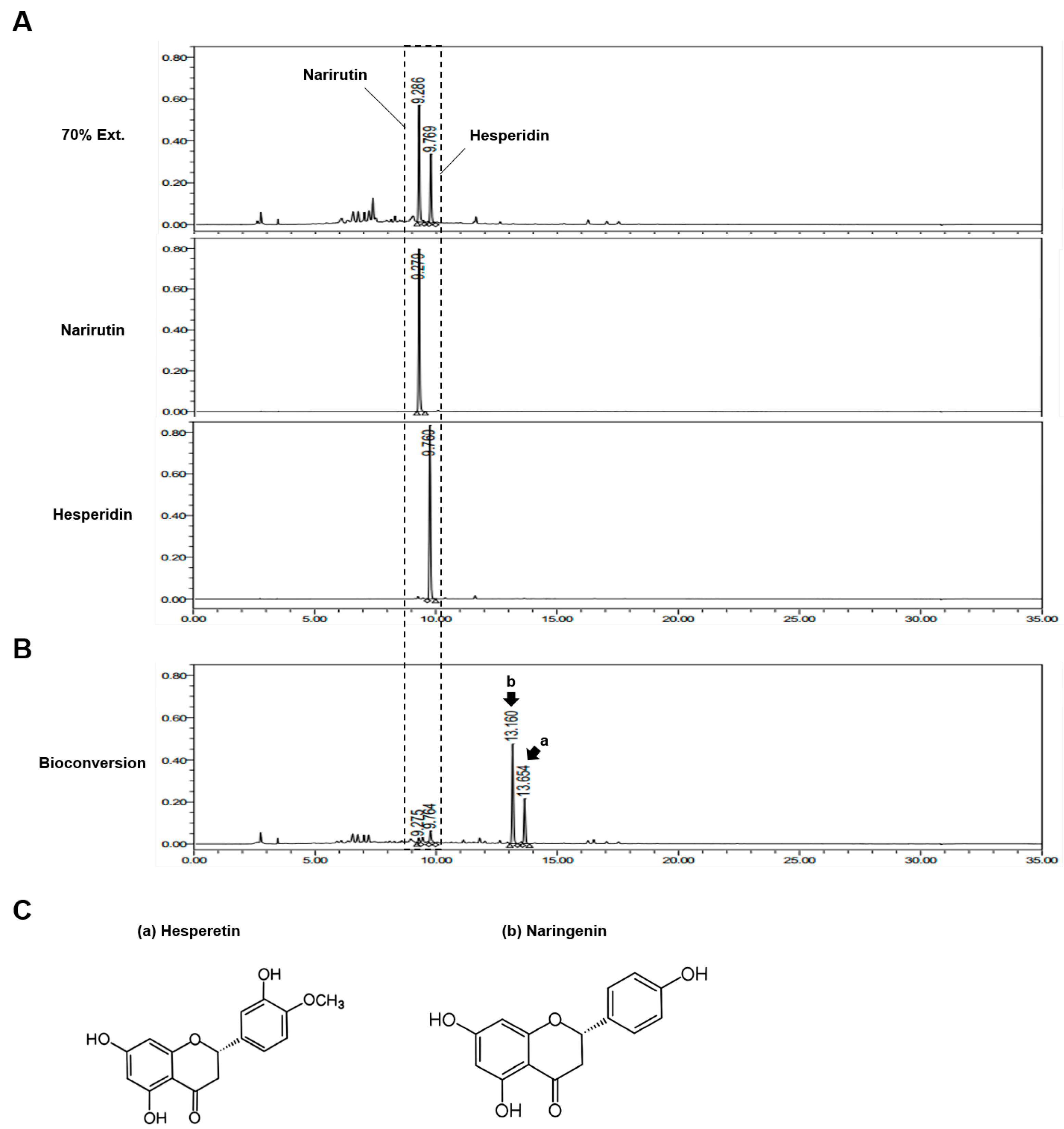
3.1. Bioconversion Metabolites in CUP Mainly Consist of Hesperetin and Naringenin
3.2. Bioconversion Metabolites in CUP Can Reduce Intestinal Inflammation Mediated by IκB Degradation
3.3. Bioconversion of CUP Ameliorates the Inflammatory Phenotypes in the DSS-Induced Colitis Model
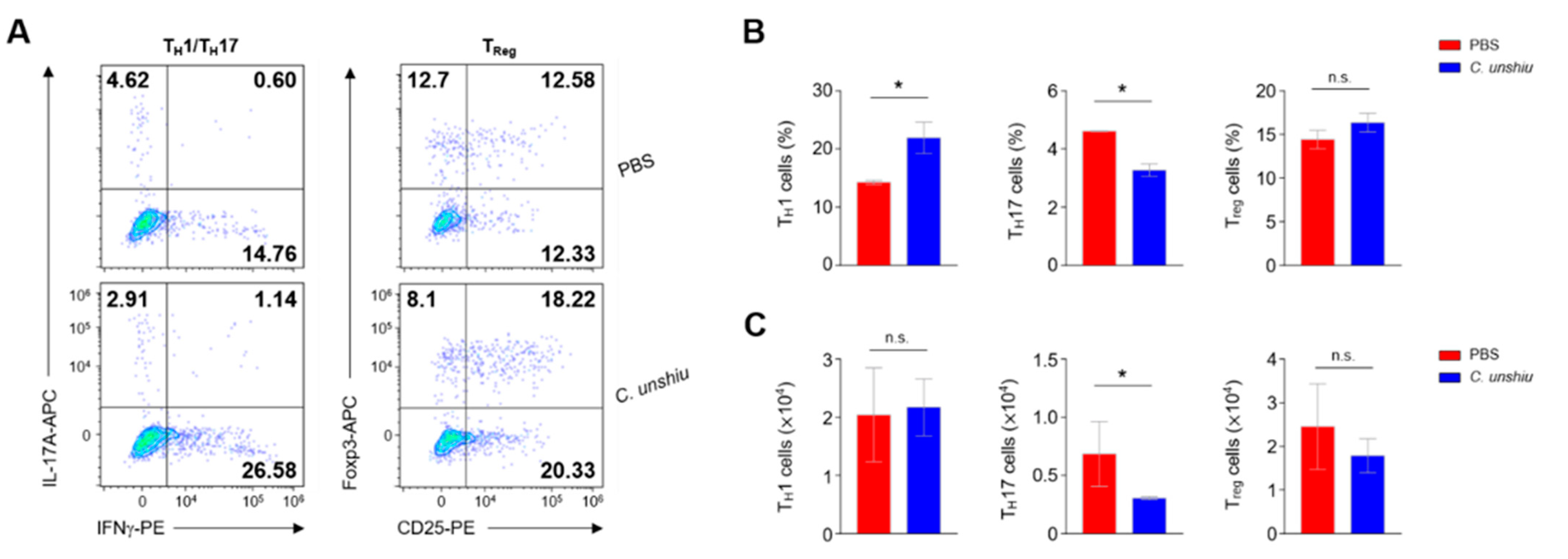
3.4. Bioconversion of CUP Decreases the Frequency of Th17 Cells in the Lamina Propria of Large Intestine
3.5. Bioconversion of CUP Enhances Intestinal Integrity by Upregulating the Levels of Tight Junction Proteins
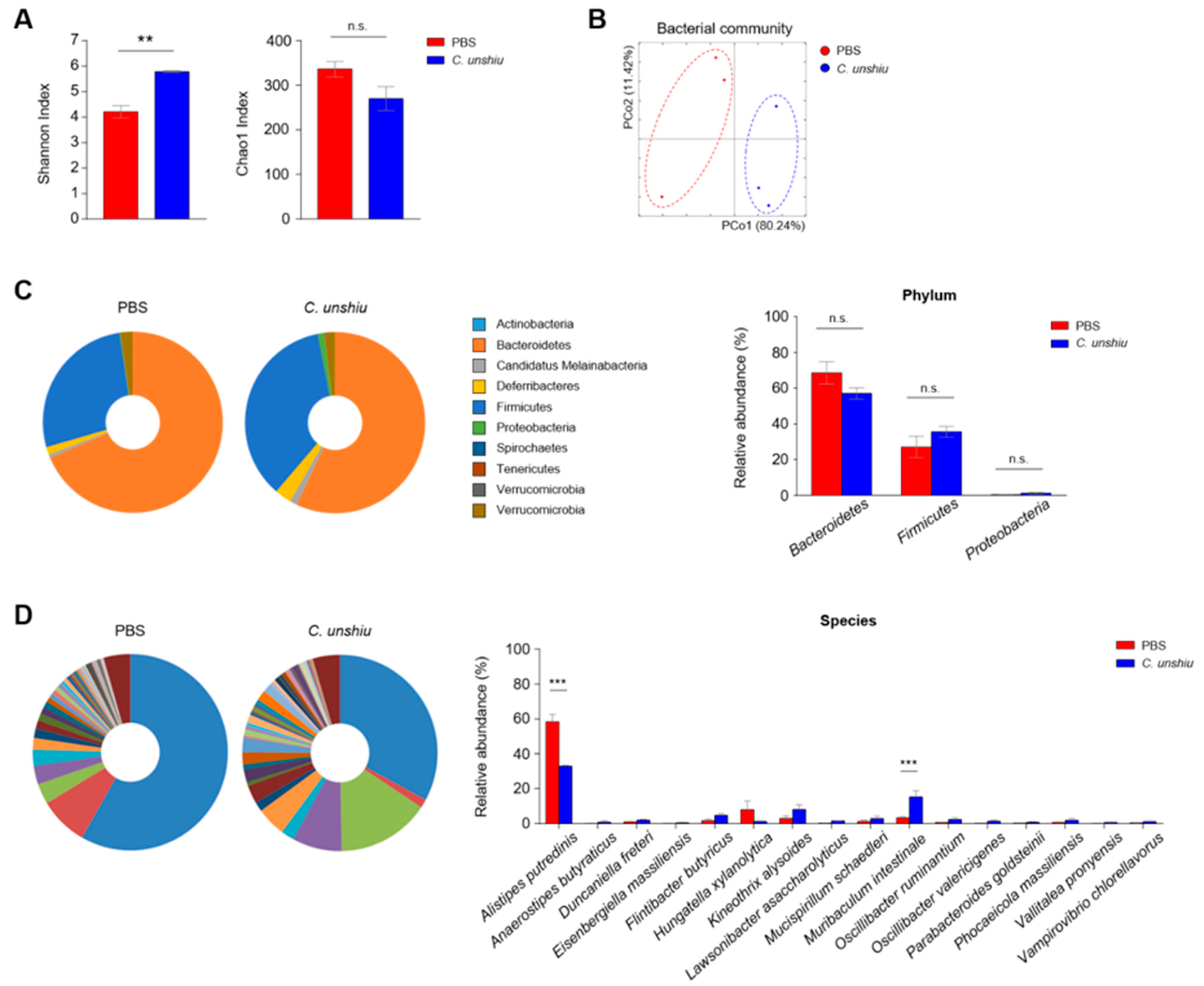
3.6. Bioconversion of CUP Alters the Composition of Gut Commensal Bacteria
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Ademosun, A.O.; Popoola, T.V.; Oboh, G.; Fasakin, O.W. Parquetina nigrescens and Spondias mombin protects against neurochemical alterations in the scopolamine model of cognitive dysfunction. J. Food. Biochem. 2022, 46, e14213. [Google Scholar] [CrossRef] [PubMed]
- Demir, S.; Atayoglu, A.T.; Galeotti, F.; Garzarella, E.U.; Zaccaria, V.; Volpi, N.; Karagoz, A.; Sahin, F. Antiviral activity of different extracts of standardized propolis preparations against HSV. Antivir. Ther. 2020, 25, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.F.; Wen, Z.H.; Bakheit, A.H.; Basudan, O.A.; Ghabbour, H.A.; Al-Ahmari, A.; Feng, C.W. A Major Diplotaxis harra-Derived Bioflavonoid Glycoside as a Protective Agent against Chemically Induced Neurotoxicity and Parkinson’s Models; In Silico Target Prediction; and Biphasic HPTLC-Based Quantification. Plants 2022, 11, 648. [Google Scholar] [CrossRef] [PubMed]
- Askin, S.; Askin, H.; Dursun, E.; Palabiyik, E.; Uguz, H.; Cakmak, O.; Koc, K. The hepato-renal protective potential of walnut seed skin extract against acute renal ischemia/reperfusion damage. Cytokine 2022, 153, 155861. [Google Scholar] [CrossRef] [PubMed]
- Rai, V.; Pogu, S.V.; Bhatnagar, R.; Bomzan, P.; Dutta, A.; Mandal, A.; Roy, M.N.; Kumar, A.; Ghosh, S. Biological evaluation of a natural steroid ester, Stigmasta-5(6), 22(23)-dien-3-beta-yl acetate isolated from the Himalayan herb Astilbe rivularis as potential antitumor agent. Chem. Biol. Interact. 2022, 360, 109935. [Google Scholar] [CrossRef]
- Heard, S.C.; Wu, G.; Winter, J.M. Antifungal natural products. Curr. Opin. Biotechnol. 2021, 69, 232–241. [Google Scholar] [CrossRef]
- Koychev, S.; Dommisch, H.; Chen, H.; Pischon, N. Antimicrobial Effects of Mastic Extract Against Oral and Periodontal Pathogens. J. Periodontol. 2017, 88, 511–517. [Google Scholar] [CrossRef]
- Nocerino, R.; Cecere, G.; Micillo, M.; De Marco, G.; Ferri, P.; Russo, M.; Bedogni, G.; Canani, R.B. Efficacy of ginger as antiemetic in children with acute gastroenteritis: A randomised controlled trial. Aliment. Pharm. Ther. 2021, 54, 24–31. [Google Scholar] [CrossRef]
- Zare, R.; Nadjarzadeh, A.; Zarshenas, M.M.; Shams, M.; Heydari, M. Efficacy of cinnamon in patients with type II diabetes mellitus: A randomized controlled clinical trial. Clin. Nutr. 2019, 38, 549–556. [Google Scholar] [CrossRef]
- Chen, I.J.; Liu, C.Y.; Chiu, J.P.; Hsu, C.H. Therapeutic effect of high-dose green tea extract on weight reduction: A randomized, double-blind, placebo-controlled clinical trial. Clin. Nutr. 2016, 35, 592–599. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from January 1981 to September 2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef]
- Chaves, J.O.; de Souza, M.C.; da Silva, L.C.; Lachos-Perez, D.; Torres-Mayanga, P.C.; Machado, A.; Forster-Carneiro, T.; Vazquez-Espinosa, M.; Gonzalez-de-Peredo, A.V.; Barbero, G.F.; et al. Extraction of Flavonoids from Natural Sources Using Modern Techniques. Front. Chem. 2020, 8, 507887. [Google Scholar] [CrossRef]
- Czech, A.; Zarycka, E.; Yanovych, D.; Zasadna, Z.; Grzegorczyk, I.; Klys, S. Mineral Content of the Pulp and Peel of Various Citrus Fruit Cultivars. Biol. Trace Elem. Res. 2020, 193, 555–563. [Google Scholar] [CrossRef]
- Barros, H.R.; Ferreira, T.A.; Genovese, M.I. Antioxidant capacity and mineral content of pulp and peel from commercial cultivars of citrus from Brazil. Food Chem. 2012, 134, 1892–1898. [Google Scholar] [CrossRef]
- Jang, A.; Choi, G.E.; Kim, Y.J.; Lee, G.H.; Hyun, K.Y. Neuroprotective properties of ethanolic extract of Citrus unshiu Markovich peel through NADPH oxidase 2 inhibition in chemotherapy-induced neuropathic pain animal model. Phytother. Res. 2021, 35, 6918–6931. [Google Scholar] [CrossRef]
- Choi, Y.S.; Han, J.M.; Kang, Y.J.; Jung, H.J. Chloroform extract of Citrus unshiu Markovich peel induces apoptosis and inhibits stemness in HeLa human cervical cancer cells. Mol. Med. Rep. 2021, 23, 86. [Google Scholar] [CrossRef]
- Shin, M.R.; Shin, S.H.; Roh, S.S. Diospyros kaki and Citrus unshiu Mixture Improves Disorders of Lipid Metabolism in Nonalcoholic Fatty Liver Disease. Can. J. Gastroenterol. Hepatol. 2020, 2020, 8812634. [Google Scholar] [CrossRef]
- Park, H.J.; Jung, U.J.; Cho, S.J.; Jung, H.K.; Shim, S.; Choi, M.S. Citrus unshiu peel extract ameliorates hyperglycemia and hepatic steatosis by altering inflammation and hepatic glucose- and lipid-regulating enzymes in db/db mice. J. Nutr. Biochem. 2013, 24, 419–427. [Google Scholar] [CrossRef]
- Lee, J.; Lee, J.; Kim, M.; Kim, J.H. Fermented Extraction of Citrus unshiu Peel Inhibits Viability and Migration of Human Pancreatic Cancers. J. Med. Food 2018, 21, 5–12. [Google Scholar] [CrossRef]
- Nishi, K.; Ito, T.; Kadota, A.; Ishida, M.; Nishiwaki, H.; Fukuda, N.; Kanamoto, N.; Nagata, Y.; Sugahara, T. Aqueous Extract from Leaves of Citrus unshiu Attenuates Lipopolysaccharide-Induced Inflammatory Responses in a Mouse Model of Systemic Inflammation. Plants 2021, 10, 1708. [Google Scholar] [CrossRef]
- Lee, S.H.; Lee, J.A.; Shin, M.R.; Park, H.J.; Roh, S.S. Citrus unshiu Peel Attenuates Dextran Sulfate Sodium-Induced Ulcerative Colitis in Mice due to Modulation of the PI3K/Akt Signaling Pathway and MAPK and NF-kappaB. Evid. Based Complement. Alternat. Med. 2022, 2022, 4041402. [Google Scholar] [CrossRef] [PubMed]
- Braune, A.; Blaut, M. Bacterial species involved in the conversion of dietary flavonoids in the human gut. Gut Microbes 2016, 7, 216–234. [Google Scholar] [CrossRef] [PubMed]
- Stevens, Y.; Rymenant, E.V.; Grootaert, C.; Camp, J.V.; Possemiers, S.; Masclee, A.; Jonkers, D. The Intestinal Fate of Citrus Flavanones and Their Effects on Gastrointestinal Health. Nutrients 2019, 11, 1464. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Moon, K.M.; Kim, C.Y. Tight Junction in the Intestinal Epithelium: Its Association with Diseases and Regulation by Phytochemicals. J. Immunol. Res. 2018, 2018, 2645465. [Google Scholar] [CrossRef] [PubMed]
- Vijay-Kumar, M.; Aitken, J.D.; Carvalho, F.A.; Cullender, T.C.; Mwangi, S.; Srinivasan, S.; Sitaraman, S.V.; Knight, R.; Ley, R.E.; Gewirtz, A.T. Metabolic syndrome and altered gut microbiota in mice lacking Toll-like receptor 5. Science 2010, 328, 228–231. [Google Scholar] [CrossRef]
- Elinav, E.; Strowig, T.; Kau, A.L.; Henao-Mejia, J.; Thaiss, C.A.; Booth, C.J.; Peaper, D.R.; Bertin, J.; Eisenbarth, S.C.; Gordon, J.I.; et al. NLRP6 inflammasome regulates colonic microbial ecology and risk for colitis. Cell 2011, 145, 745–757. [Google Scholar] [CrossRef]
- Carvalho, F.A.; Koren, O.; Goodrich, J.K.; Johansson, M.E.; Nalbantoglu, I.; Aitken, J.D.; Su, Y.; Chassaing, B.; Walters, W.A.; Gonzalez, A.; et al. Transient inability to manage proteobacteria promotes chronic gut inflammation in TLR5-deficient mice. Cell Host Microbe 2012, 12, 139–152. [Google Scholar] [CrossRef]
- Rawat, M.; Nighot, M.; Al-Sadi, R.; Gupta, Y.; Viszwapriya, D.; Yochum, G.; Koltun, W.; Ma, T.Y. IL1B Increases Intestinal Tight Junction Permeability by Up-regulation of MIR200C-3p, Which Degrades Occludin mRNA. Gastroenterology 2020, 159, 1375–1389. [Google Scholar] [CrossRef]
- Atarashi, K.; Tanoue, T.; Shima, T.; Imaoka, A.; Kuwahara, T.; Momose, Y.; Cheng, G.; Yamasaki, S.; Saito, T.; Ohba, Y.; et al. Induction of colonic regulatory T cells by indigenous Clostridium species. Science 2011, 331, 337–341. [Google Scholar] [CrossRef]
- Omenetti, S.; Bussi, C.; Metidji, A.; Iseppon, A.; Lee, S.; Tolaini, M.; Li, Y.; Kelly, G.; Chakravarty, P.; Shoaie, S.; et al. The Intestine Harbors Functionally Distinct Homeostatic Tissue-Resident and Inflammatory Th17 Cells. Immunity 2019, 51, 77–89.e76. [Google Scholar] [CrossRef]
- Conrad, K.; Roggenbuck, D.; Laass, M.W. Diagnosis and classification of ulcerative colitis. Autoimmun. Rev. 2014, 13, 463–466. [Google Scholar] [CrossRef]
- Qin, X. Etiology of inflammatory bowel disease: A unified hypothesis. World J. Gastroenterol. 2012, 18, 1708–1722. [Google Scholar] [CrossRef]
- Khan, I.; Ullah, N.; Zha, L.; Bai, Y.; Khan, A.; Zhao, T.; Che, T.; Zhang, C. Alteration of Gut Microbiota in Inflammatory Bowel Disease (IBD): Cause or Consequence? IBD Treatment Targeting the Gut Microbiome. Pathogens 2019, 8, 126. [Google Scholar] [CrossRef]
- Glassner, K.L.; Abraham, B.P.; Quigley, E.M.M. The microbiome and inflammatory bowel disease. J. Allergy Clin. Immunol. 2020, 145, 16–27. [Google Scholar] [CrossRef]
- Pasparakis, M. IKK/NF-kappaB signaling in intestinal epithelial cells controls immune homeostasis in the gut. Mucosal. Immunol. 2008, 1, S54–S57. [Google Scholar] [CrossRef]
- Sano, T.; Kageyama, T.; Fang, V.; Kedmi, R.; Martinez, C.S.; Talbot, J.; Chen, A.; Cabrera, I.; Gorshko, O.; Kurakake, R.; et al. Redundant cytokine requirement for intestinal microbiota-induced Th17 cell differentiation in draining lymph nodes. Cell Rep. 2021, 36, 109608. [Google Scholar] [CrossRef]
- Selek, S.; Koyuncu, I.; Caglar, H.G.; Bektas, I.; Yilmaz, M.A.; Gonel, A.; Akyuz, E. The evaluation of antioxidant and anticancer effects of Lepidium Sativum Subsp Spinescens L. methanol extract on cancer cells. Cell Mol. Biol. 2018, 64, 72–80. [Google Scholar] [CrossRef]
- Deme, P.; Aluganti Narasimhulu, C.; Parthasarathy, S. Evaluation of Anti-Inflammatory Properties of Herbal Aqueous Extracts and Their Chemical Characterization. J. Med. Food 2019, 22, 861–873. [Google Scholar] [CrossRef]
- Dorjsembe, B.; Nho, C.W.; Choi, Y.; Kim, J.C. Extract from Black Soybean Cultivar A63 Extract Ameliorates Atopic Dermatitis-like Skin Inflammation in an Oxazolone-Induced Murine Model. Molecules 2022, 27, 2751. [Google Scholar] [CrossRef]
- Jiao, C.; Liang, H.; Liu, L.; Li, S.; Chen, J.; Xie, Y. Transcriptomic analysis of the anti-inflammatory effect of Cordyceps militaris extract on acute gouty arthritis. Front. Pharmacol. 2022, 13, 1035101. [Google Scholar] [CrossRef]
- Shiu, P.H.; Li, J.; Zheng, C.; Rangsinth, P.; Li, R.; Cheung, Q.T.; Lau, A.H.; Chan, J.C.; Kwan, Y.W.; Cheung, T.M.; et al. Amauroderma rugosum Extract Suppresses Inflammatory Responses in Tumor Necrosis Factor Alpha/Interferon Gamma-Induced HaCaT Keratinocytes. Molecules 2022, 27, 6533. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Cheng, C.; Han, Q.; Chen, Y.; Guo, J.; Wu, Q.; Zhu, B.; Shan, J.; Shi, L. Flos Abelmoschus manihot extract attenuates DSS-induced colitis by regulating gut microbiota and Th17/Treg balance. Biomed. Pharmacother. 2019, 117, 109162. [Google Scholar] [CrossRef] [PubMed]
- Qiao, L.; Fang, L.; Zhu, J.; Xiang, Y.; Xu, H.; Sun, X.; Chen, H.; Yang, B. Total Flavone of Abelmoschus manihot Ameliorates TNBS-Induced Colonic Fibrosis by Regulating Th17/Treg Balance and Reducing Extracellular Matrix. Front. Pharmacol. 2021, 12, 769793. [Google Scholar] [CrossRef] [PubMed]
- Wen, S.; He, L.; Zhong, Z.; Zhao, R.; Weng, S.; Mi, H.; Liu, F. Stigmasterol Restores the Balance of Treg/Th17 Cells by Activating the Butyrate-PPARgamma Axis in Colitis. Front. Immunol. 2021, 12, 741934. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Lin, J.; Zhang, C.; Gao, H.; Lu, H.; Gao, X.; Zhu, R.; Li, Z.; Li, M.; Liu, Z. Microbiota metabolite butyrate constrains neutrophil functions and ameliorates mucosal inflammation in inflammatory bowel disease. Gut Microbes 2021, 13, 1968257. [Google Scholar] [CrossRef]
- Zhong, W.; Chen, K.; Yang, L.; Tang, T.; Jiang, S.; Guo, J.; Gao, Z. Essential Oils from Citrus unshiu Marc. Effectively Kill Aeromonas hydrophila by Destroying Cell Membrane Integrity, Influencing Cell Potential, and Leaking Intracellular Substances. Front. Microbiol. 2022, 13, 869953. [Google Scholar] [CrossRef]
- Yang, J.Y.; Kim, M.S.; Kim, E.; Cheon, J.H.; Lee, Y.S.; Kim, Y.; Lee, S.H.; Seo, S.U.; Shin, S.H.; Choi, S.S.; et al. Enteric Viruses Ameliorate Gut Inflammation via Toll-like Receptor 3 and Toll-like Receptor 7-Mediated Interferon-beta Production. Immunity 2016, 44, 889–900. [Google Scholar] [CrossRef]
- Herath, H.; Piao, M.J.; Kang, K.A.; Zhen, A.X.; Fernando, P.; Kang, H.K.; Yi, J.M.; Hyun, J.W. Hesperidin Exhibits Protective Effects against PM2.5-Mediated Mitochondrial Damage, Cell Cycle Arrest, and Cellular Senescence in Human HaCaT Keratinocytes. Molecules 2022, 27, 4800. [Google Scholar] [CrossRef]
- Liu, F.; Qu, Y.K.; Geng, C.; Wang, A.M.; Zhang, J.H.; Chen, K.J.; Liu, B.; Tian, H.Y.; Yang, W.P.; Yu, Y.B. Effects of hesperidin on the growth performance, antioxidant capacity, immune responses and disease resistance of red swamp crayfish (Procambarus clarkii). Fish Shellfish Immunol. 2020, 99, 154–166. [Google Scholar] [CrossRef]
- Guazelli, C.F.S.; Fattori, V.; Ferraz, C.R.; Borghi, S.M.; Casagrande, R.; Baracat, M.M.; Verri, W.A., Jr. Antioxidant and anti-inflammatory effects of hesperidin methyl chalcone in experimental ulcerative colitis. Chem. Biol. Interact. 2021, 333, 109315. [Google Scholar] [CrossRef]
- Guo, K.; Ren, J.; Gu, G.; Wang, G.; Gong, W.; Wu, X.; Ren, H.; Hong, Z.; Li, J. Hesperidin Protects Against Intestinal Inflammation by Restoring Intestinal Barrier Function and Up-Regulating Treg Cells. Mol. Nutr. Food Res. 2019, 63, e1800975. [Google Scholar] [CrossRef]
- Polat, F.R.; Karaboga, I.; Polat, M.S.; Erboga, Z.; Yilmaz, A.; Guzel, S. Effect of hesperetin on inflammatory and oxidative status in trinitrobenzene sulfonic acid-induced experimental colitis model. Cell Mol. Biol. 2018, 64, 58–65. [Google Scholar] [CrossRef]
- Guven, B.; Can, M.; Piskin, O.; Aydin, B.G.; Karakaya, K.; Elmas, O.; Acikgoz, B. Flavonoids protect colon against radiation induced colitis. Regul. Toxicol. Pharmacol. 2019, 104, 128–132. [Google Scholar] [CrossRef]
- Chen, Z.; Wu, H.; Fan, W.; Zhang, J.; Yao, Y.; Su, W.; Wang, Y.; Li, P. Naringenin suppresses BEAS-2B-derived extracellular vesicular cargoes disorder caused by cigarette smoke extract thereby inhibiting M1 macrophage polarization. Front. Immunol. 2022, 13, 930476. [Google Scholar] [CrossRef]
- Lin, C.; Zeng, Z.; Lin, Y.; Wang, P.; Cao, D.; Xie, K.; Luo, Y.; Yang, H.; Yang, J.; Wang, W.; et al. Naringenin suppresses epithelial ovarian cancer by inhibiting proliferation and modulating gut microbiota. Phytomedicine 2022, 106, 154401. [Google Scholar] [CrossRef]
- Hua, Y.Q.; Zeng, Y.; Xu, J.; Xu, X.L. Naringenin alleviates nonalcoholic steatohepatitis in middle-aged Apoe(-/-)mice: Role of SIRT1. Phytomedicine 2021, 81, 153412. [Google Scholar] [CrossRef]
- Li, J.; Wang, T.; Liu, P.; Yang, F.; Wang, X.; Zheng, W.; Sun, W. Hesperetin ameliorates hepatic oxidative stress and inflammation via the PI3K/AKT-Nrf2-ARE pathway in oleic acid-induced HepG2 cells and a rat model of high-fat diet-induced NAFLD. Food Funct. 2021, 12, 3898–3918. [Google Scholar] [CrossRef]
- Al-Sadi, R.; Guo, S.; Ye, D.; Rawat, M.; Ma, T.Y. TNF-alpha Modulation of Intestinal Tight Junction Permeability Is Mediated by NIK/IKK-alpha Axis Activation of the Canonical NF-kappaB Pathway. Am. J. Pathol. 2016, 186, 1151–1165. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-kappaB signaling in inflammation. Signal Transduct. Target Ther. 2017, 2, 17023. [Google Scholar] [CrossRef]
- Viatour, P.; Merville, M.P.; Bours, V.; Chariot, A. Phosphorylation of NF-kappaB and IkappaB proteins: Implications in cancer and inflammation. Trends Biochem. Sci. 2005, 30, 43–52. [Google Scholar] [CrossRef]
- Dave, S.H.; Tilstra, J.S.; Matsuoka, K.; Li, F.; Karrasch, T.; Uno, J.K.; Sepulveda, A.R.; Jobin, C.; Baldwin, A.S.; Robbins, P.D.; et al. Amelioration of chronic murine colitis by peptide-mediated transduction of the IkappaB kinase inhibitor NEMO binding domain peptide. J. Immunol. 2007, 179, 7852–7859. [Google Scholar] [CrossRef] [PubMed]
- Eckmann, L.; Nebelsiek, T.; Fingerle, A.A.; Dann, S.M.; Mages, J.; Lang, R.; Robine, S.; Kagnoff, M.F.; Schmid, R.M.; Karin, M.; et al. Opposing functions of IKKbeta during acute and chronic intestinal inflammation. Proc. Natl. Acad. Sci. USA 2008, 105, 15058–15063. [Google Scholar] [CrossRef] [PubMed]
- Oh, Y.C.; Cho, W.K.; Jeong, Y.H.; Im, G.Y.; Yang, M.C.; Hwang, Y.H.; Ma, J.Y. Anti-inflammatory effect of Citrus Unshiu peel in LPS-stimulated RAW 264.7 macrophage cells. Am. J. Chin. Med. 2012, 40, 611–629. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.; Ji, J.; Ho Baek, S.; Lee, J.H.; Ha, I.J.; Lim, S.S.; Yoon, H.J.; Je Nam, Y.; Ahn, K.S. Fermented dried Citrus unshiu peel extracts exert anti-inflammatory activities in LPS-induced RAW264.7 macrophages and improve skin moisturizing efficacy in immortalized human HaCaT keratinocytes. Pharm. Biol. 2019, 57, 392–402. [Google Scholar] [CrossRef]
- Marwaha, A.K.; Leung, N.J.; McMurchy, A.N.; Levings, M.K. Th17 Cells in Autoimmunity and Immunodeficiency: Protective or Pathogenic? Front. Immunol. 2012, 3, 129. [Google Scholar] [CrossRef]
- Palmieri, V.; Ebel, J.F.; Ngo Thi Phuong, N.; Klopfleisch, R.; Vu, V.P.; Adamczyk, A.; Zoller, J.; Riedel, C.; Buer, J.; Krebs, P.; et al. Interleukin-33 signaling exacerbates experimental infectious colitis by enhancing gut permeability and inhibiting protective Th17 immunity. Mucosal Immunol. 2021, 14, 923–936. [Google Scholar] [CrossRef]
- Shimano, K.; Maeda, Y.; Kataoka, H.; Murase, M.; Mochizuki, S.; Utsumi, H.; Oshita, K.; Sugahara, K. Amiselimod (MT-1303), a novel sphingosine 1-phosphate receptor-1 functional antagonist, inhibits progress of chronic colitis induced by transfer of CD4+CD45RBhigh T cells. PLoS ONE 2019, 14, e0226154. [Google Scholar] [CrossRef]
- Yang, W.; Gibson, S.A.; Yan, Z.; Wei, H.; Tao, J.; Sha, B.; Qin, H.; Benveniste, E.N. Protein kinase 2 (CK2) controls CD4(+) T cell effector function in the pathogenesis of colitis. Mucosal Immunol. 2020, 13, 788–798. [Google Scholar] [CrossRef]
- Zhao, X.; Yang, W.; Yu, T.; Yu, Y.; Cui, X.; Zhou, Z.; Yang, H.; Yu, Y.; Bilotta, A.J.; Yao, S.; et al. Th17 Cell-Derived Amphiregulin Promotes Colitis-Associated Intestinal Fibrosis Through Activation of mTOR and MEK in Intestinal Myofibroblasts. Gastroenterology 2022, 164, 89–102. [Google Scholar] [CrossRef]
- Jung, S.H.; Bae, C.H.; Kim, J.H.; Park, S.D.; Shim, J.J.; Lee, J.L. Lactobacillus casei HY2782 and Pueraria lobata Root Extract Complex Ameliorates Particulate Matter-Induced Airway Inflammation in Mice by Inhibiting Th2 and Th17 Immune Responses. Prev. Nutr. Food Sci. 2022, 27, 188–197. [Google Scholar] [CrossRef]
- Pei, R.; Liu, X.; Bolling, B. Flavonoids and gut health. Curr. Opin. Biotechnol. 2020, 61, 153–159. [Google Scholar] [CrossRef]
- Schirmer, M.; Denson, L.; Vlamakis, H.; Franzosa, E.A.; Thomas, S.; Gotman, N.M.; Rufo, P.; Baker, S.S.; Sauer, C.; Markowitz, J.; et al. Compositional and Temporal Changes in the Gut Microbiome of Pediatric Ulcerative Colitis Patients Are Linked to Disease Course. Cell Host Microbe 2018, 24, 600–610 e604. [Google Scholar] [CrossRef]
- Ott, S.J.; Musfeldt, M.; Wenderoth, D.F.; Hampe, J.; Brant, O.; Folsch, U.R.; Timmis, K.N.; Schreiber, S. Reduction in diversity of the colonic mucosa associated bacterial microflora in patients with active inflammatory bowel disease. Gut 2004, 53, 685–693. [Google Scholar] [CrossRef]
- De Meij, T.G.J.; de Groot, E.F.J.; Peeters, C.F.W.; de Boer, N.K.H.; Kneepkens, C.M.F.; Eck, A.; Benninga, M.A.; Savelkoul, P.H.M.; van Bodegraven, A.A.; Budding, A.E. Variability of core microbiota in newly diagnosed treatment-naive paediatric inflammatory bowel disease patients. PLoS ONE 2018, 13, e0197649. [Google Scholar] [CrossRef]
- Parker, B.J.; Wearsch, P.A.; Veloo, A.C.M.; Rodriguez-Palacios, A. The Genus Alistipes: Gut Bacteria with Emerging Implications to Inflammation, Cancer, and Mental Health. Front. Immunol. 2020, 11, 906. [Google Scholar] [CrossRef]
- Dobranowski, P.A.; Tang, C.; Sauve, J.P.; Menzies, S.C.; Sly, L.M. Compositional changes to the ileal microbiome precede the onset of spontaneous ileitis in SHIP deficient mice. Gut Microbes 2019, 10, 578–598. [Google Scholar] [CrossRef]
- Hasan, R.; Bose, S.; Roy, R.; Paul, D.; Rawat, S.; Nilwe, P.; Chauhan, N.K.; Choudhury, S. Tumor tissue-specific bacterial biomarker panel for colorectal cancer: Bacteroides massiliensis, Alistipes species, Alistipes onderdonkii, Bifidobacterium pseudocatenulatum, Corynebacterium appendicis. Arch. Microbiol. 2022, 204, 348. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, S.-H.; Seo, D.; Lee, K.-H.; Park, S.-J.; Park, S.; Kim, H.; Kim, T.; Joo, I.H.; Park, J.-M.; Kang, Y.-H.; et al. Biometabolites of Citrus unshiu Peel Enhance Intestinal Permeability and Alter Gut Commensal Bacteria. Nutrients 2023, 15, 319. https://doi.org/10.3390/nu15020319
Lee S-H, Seo D, Lee K-H, Park S-J, Park S, Kim H, Kim T, Joo IH, Park J-M, Kang Y-H, et al. Biometabolites of Citrus unshiu Peel Enhance Intestinal Permeability and Alter Gut Commensal Bacteria. Nutrients. 2023; 15(2):319. https://doi.org/10.3390/nu15020319
Chicago/Turabian StyleLee, Se-Hui, Dongju Seo, Kang-Hee Lee, So-Jung Park, Sun Park, Hyeyun Kim, Taekyung Kim, In Hwan Joo, Jong-Min Park, Yun-Hwan Kang, and et al. 2023. "Biometabolites of Citrus unshiu Peel Enhance Intestinal Permeability and Alter Gut Commensal Bacteria" Nutrients 15, no. 2: 319. https://doi.org/10.3390/nu15020319
APA StyleLee, S.-H., Seo, D., Lee, K.-H., Park, S.-J., Park, S., Kim, H., Kim, T., Joo, I. H., Park, J.-M., Kang, Y.-H., Lim, G.-H., Kim, D. H., & Yang, J.-Y. (2023). Biometabolites of Citrus unshiu Peel Enhance Intestinal Permeability and Alter Gut Commensal Bacteria. Nutrients, 15(2), 319. https://doi.org/10.3390/nu15020319






