Insights into the Chemical Compositions and Health Promoting Effects of Wild Edible Mushroom Chroogomphus rutilus
Abstract
1. Introduction
2. Description and Geographical Distribution of C. rutilus
3. Fungal Chemical Characteristics of C. rutilus
3.1. Primary Metabolites of C. rutilus
3.2. Secondary Metabolites of C. rutilus
4. Biological Activity of C. rutilus
4.1. Formation and Function of Antioxidant Activity
4.2. Anti-Tumor Activity of C. rutilus
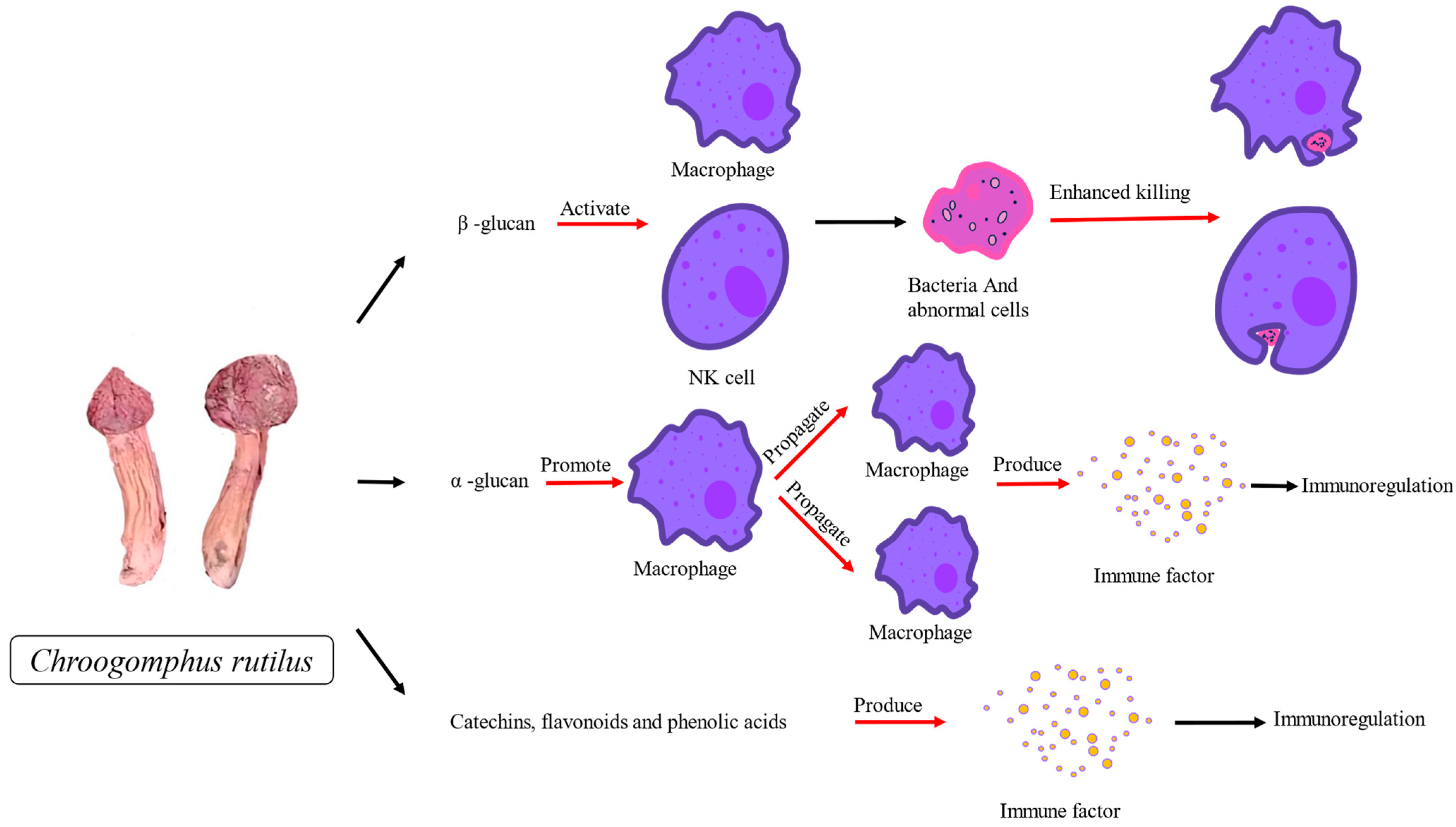
4.3. Immunomodulatory Activity of C. rutilus
4.4. Anti-Fatigue Activity of C. rutilus
4.5. Hypoglycemia Activity of C. rutilus
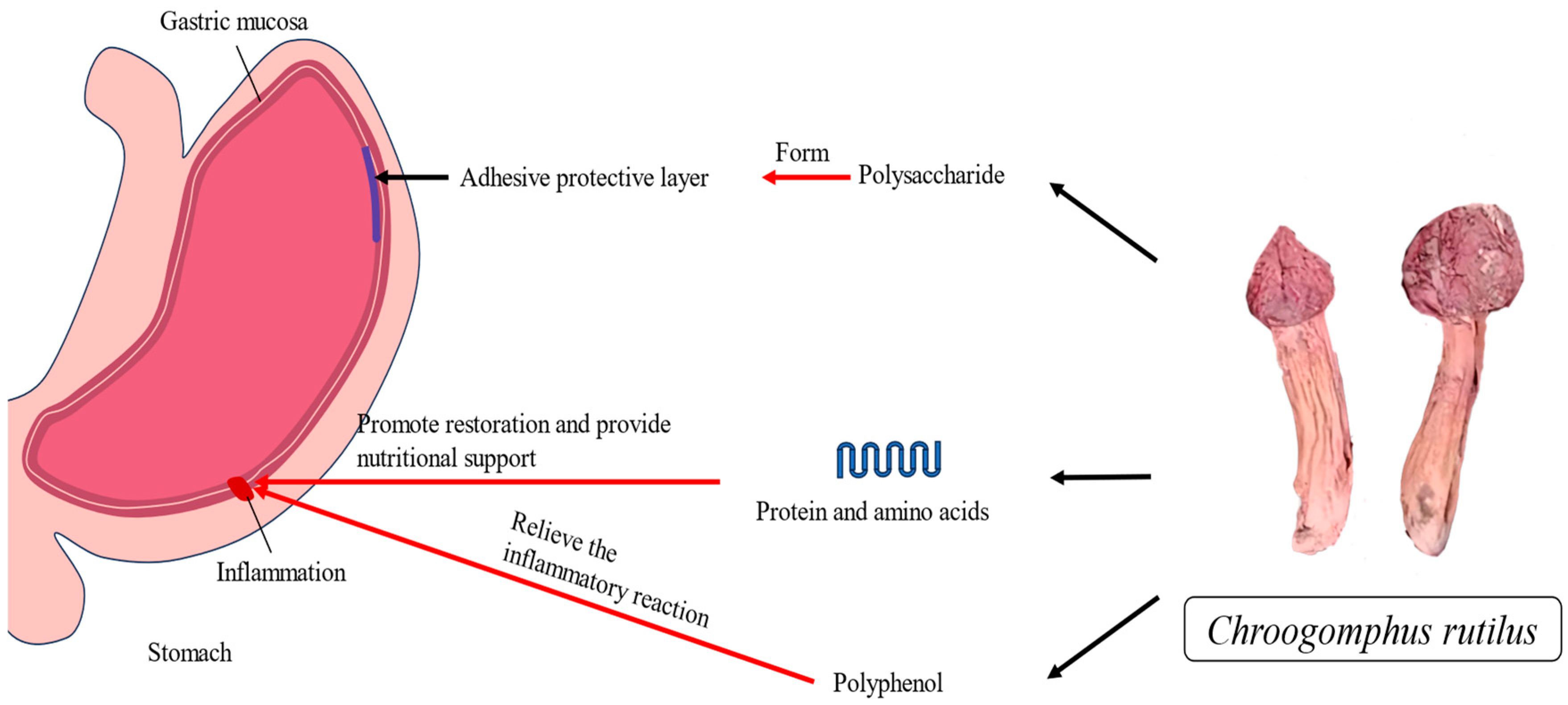
4.6. Gastric Protective Activity of C. rutilus
4.7. Hypolipidemic Activity of C. rutilus
4.8. Neuroprotective Effects of C. rutilus
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Habib, S.H.; Saha, S. Burden of non-communicable disease: Global overview. Diabetesi. Metab. Synd. 2010, 4, 41–47. [Google Scholar] [CrossRef]
- Munzel, T.; Hahad, O.; Sørensen, M.; Lelieveld, J.; Duerr, G.D.; Nieuwenhuijsen, M.; Daiber, A. Environmental risk factors and cardiovascular diseases: A comprehensive expert review. Cardiovasc. Res. 2011, 118, 2880–2902. [Google Scholar] [CrossRef]
- Buttar, H.S.; Li, T.; Ravi, N. Prevention of cardiovascular diseases: Role of exercise, dietary interventions, obesity and smoking cessation. Exp. Clin. Cardiol. 2005, 10, 229. [Google Scholar]
- Bray, G.A.; Kim, K.K.; Wilding, J.P.; World Obesity Federation. Obesity: A chronic relapsing progressive disease process. A position statement of the World Obesity Federation. Obes. Rev. 2017, 18, 715–723. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Zhao, J.K.; Hu, X.S.; Wang, P.H.; Qin, Y.; Lu, Y.C.; Yang, J.; Liu, A.M.; Wu, D.L.; Zhang, Z.F.; et al. Association of smoking, alcohol drinking and dietary factors with esophageal cancer in high-and low-risk areas of Jiangsu Province, China. World J. Gastroentero. 2006, 12, 1686. [Google Scholar] [CrossRef] [PubMed]
- Bach, J.F. Infections and autoimmune diseases. J. Autoimmun. 2005, 25, 74–80. [Google Scholar] [CrossRef]
- Sharkey, B.J.; Gaskill, S.E. Fitness & Health; Human Kinetics: Champaign, IL, USA, 2013. [Google Scholar]
- Dettmar, P.W.; Strugala, V.; Richardson, J.C. The key role alginates play in health. Food Hydrocoll. 2011, 25, 263–266. [Google Scholar] [CrossRef]
- Mudgil, D.; Barak, S. Functional Foods: Sources and Health Benefits; Scientific Publishers: London, UK, 2011. [Google Scholar]
- Kour, H.; Kour, D.; Kour, S.; Singh, S.; Hashmi, S.A.J.; Yadav, A.N.; Kumar, K.; Sharma, Y.P.; Ahluwalia, A.S. Bioactive compounds from mushrooms: An emerging bioresources of food and nutraceuticals. Food Bonsci. 2022, 50, 102124. [Google Scholar] [CrossRef]
- Lee, K.H.; Morris-Natschke, S.L.; Yang, X.; Huang, R.; Zhou, T.; Wu, S.F.; Shi, Q.; Itokawa, H. Recent progress of research on medicinal mushrooms, foods, and other herbal products used in traditional Chinese medicine. J. Tradit. Complement. Med. 2012, 2, 1–12. [Google Scholar] [CrossRef]
- Phan, C.W.; Tan, E.Y.Y.; Sabaratnam, V. Bioactive molecules in edible and medicinal mushrooms for human wellness. In Bioactive Molecules in Food; Springer: Cham, Switzerland, 2019; pp. 1597–1620. [Google Scholar]
- Wang, S.; Yang, B.; Zhou, Q.; Li, Z.; Li, W.; Zhang, J.; Tuo, F. Radionuclide content and risk analysis of edible mushrooms in northeast China. Radiat. Med. Prot. 2021, 2, 165–170. [Google Scholar] [CrossRef]
- Rathore, H.; Prasad, S.; Kapri, M.; Tiwari, A.; Sharma, S. Medicinal importance of mushroom mycelium: Mechanisms and applications. J. Funct. Foods 2019, 56, 182–193. [Google Scholar] [CrossRef]
- Şen, A.; Acevedo-Fani, A.; Dave, A.; Ye, A.; Husny, J.; Singh, H. Plant oil bodies and their membrane components: New natural materials for food applications. Crit. Rev. Food Sci. 2022, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Ma, G.; Yang, W.; Zhao, L.; Pei, F.; Fang, D.; Hu, Q. A critical review on the health promoting effects of mushrooms nutraceuticals. Food Sci. Hum. Wellness 2018, 7, 125–133. [Google Scholar] [CrossRef]
- Goswami, B.; Majumdar, S.; Das, A.; Barui, A.; Bhowal, J. Evaluation of bioactive properties of Pleurotus ostreatus mushroom protein hydrolysate of different degree of hydrolysis. LWT-Food Sci. Technol. 2021, 149, 111768. [Google Scholar] [CrossRef]
- Zhang, S.; Lei, L.; Zhou, Y.; Ye, F.Y.; Zhao, G.H. Roles of mushroom. polysaccharides in chronic disease management. J. Integr. Agric. 2022, 21, 1839–1866. [Google Scholar] [CrossRef]
- Saini, R.K.; Rauf, A.; Khalil, A.A.; Ko, E.Y.; Keum, Y.S.; Anwar, S.; Alamri, A.; Rengasamy, K.R. Edible mushrooms show significant differences in sterols and fatty acid compositions. S. Afr. J. Bot. 2021, 141, 344–356. [Google Scholar] [CrossRef]
- De Souza Lopes, L.; De Casssia Silva, M.; De Oliveira Faustino, A.; De Oliveira, L.L.; Kasuya, M.C.M. Bioaccessibility, oxidizing activity and co-accumulation of minerals in Li-enriched mushrooms. LWT-Food Sci. Technol. 2022, 155, 112989. [Google Scholar] [CrossRef]
- Pedrali, D.; Gallotti, F.; Proserpio, C.; Pagliarini, E.; Lavelli, V. Kinetic study of vitamin D2 degradation in mushroom powder to improve its applications in fortified foods. Lwt-Food Sci. Technol. 2020, 125, 109248. [Google Scholar] [CrossRef]
- Rathore, H.; Prasad, S.; Sharma, S. Mushroom nutraceuticals for improved nutrition and better human health: A review. Pharma Nutr. 2017, 5, 35–46. [Google Scholar] [CrossRef]
- Mwangi, R.W.; Macharia, J.M.; Wagara, I.N.; Bence, R.L. The antioxidant potential of different edible and medicinal mushrooms. Biomed. Pharmacother. 2022, 147, 112621. [Google Scholar] [CrossRef]
- Pathak, M.P.; Pathak, K.; Saikia, R.; Gogoi, U.; Ahmad, M.Z.; Patowary, P.; Das, A. Immunomodulatory effect of mushrooms and their bioactive compounds in cancer: A comprehensive review. Biomed. Pharmacother. 2022, 149, 112901. [Google Scholar] [CrossRef]
- McKnight, K.H.; McKnight, V.B. A Field Guide to Mushrooms: North America; Houghton Mifflin Harcourt: Boston, MA, USA, 1987; Volume 34. [Google Scholar]
- Sun, L.; He, W.; Xin, G.; Cai, P.; Zhang, Y.; Zhang, Z.; Wei, Y.; Sun, B.; Wen, X. Volatile components, total phenolic compounds, and antioxidant capacities of worm-infected Gomphidius rutilus. Food Sci. Hum. Wellness 2018, 7, 148–155. [Google Scholar] [CrossRef]
- Singer, R. New and interesting species of Basidiomycetes III. Sydowia 1950, 4, 130–157. [Google Scholar]
- Miller, O.K., Jr. Monograph of Chroogomphus (Gomphidiaceae). Mycologia 1964, 56, 526–549. [Google Scholar] [CrossRef]
- Miller, O.K., Jr.; Aime, M.C. Systematics, Ecology and World Distribution in the Genus Chroogomphus; Trichomycetes Other Fungal Groups: Boca Raton, FL, USA, 2001; p. 315. [Google Scholar]
- Miller, O.K., Jr. The Gomphidiaceae revisited: A worldwide perspective. Mycologia 2003, 95, 176–183. [Google Scholar] [CrossRef]
- Jiang, X. Study on the special niche of the Chroogomphus rutilus and its undergrowth. Liaoning Forest. Sci. Technol. 2022, 1, 54–57. [Google Scholar]
- Gao, C. Chroogomphus rutilus Polysaccharide Preparation and Bioactivity Analysis. Ph.D. Thesis, Liaoning Petrochemical University, Fushun, China, 2012. [Google Scholar]
- Luan, Q.S. Research status, development and utilization of Chroogomphus rutilus. Edible Fungi 2002, 24, 2–3. [Google Scholar]
- Jin, Z.M.; Chai, J.H.; He, T.T.; Peng, D. Study on antioxidant activity of polysaccharide from Gomphidius rutilus. Hubei Agric. Sci. 2020, 59, 123. [Google Scholar]
- Scambler, R.; Niskanen, T.; Assyov, B.; Ainsworth, A.M.; Bellanger, J.M.; Loizides, M.; Moreau, P.A.; Kirk, P.M.; Liimatainen, K. Diversity of Chroogomphus (Gomphidiaceae, Boletales) in Europe, and typification of C. rutilus. Ima. Fungus 2018, 9, 271–290. [Google Scholar] [CrossRef] [PubMed]
- Kuo, M. The Genus Chroogomphus; MushroomExpert. Com.: OH, USA, 2007; Available online: http://www.mushroomexpert.com/chroogomphus.html (accessed on 31 August 2023).
- Martín, M.P.; Siquier, J.L.; Salom, J.C.; Telleria, M.T.; Finschow, G. Barcoding sequences clearly separate Chroogomphus mediterraneus (Gomphidiaceae, Boletales) from C. rutilus, and allied species. Mycoscience 2016, 57, 384–392. [Google Scholar] [CrossRef]
- Mgbekem, M.A.; Lukpata, F.; Ndukaku, N.; Armon, M.; Uka, V.K.; Udosen, G.N.; Pricilla, A.B. Knowledge and utilization of mushroom as a food supplement among families in selected local government areas of Cross River State, Nigeria. Food Sci. Nutr. 2019, 10, 1287–1299. [Google Scholar] [CrossRef]
- Feng, J.; Qin, S.L.; Hu, B.; Zhao, X.J.; Wang, L. Preliminary study on chemical constituents and biological activities of fruiting bodies of Chroogomphus rutilus. Mycosystema 2014, 33, 355–364. [Google Scholar]
- Li, Z.; Bao, H. Overview of studies on chemical constituents and pharmacological activities of Chroogomphus rutilus. J. Fungal Res. 2015, 13, 181–186. [Google Scholar]
- Fulgoni, V.L., III; Agarwal, S. Nutritional impact of adding a serving of mushrooms on usual intakes and nutrient adequacy using National Health and Nutrition Examination Survey 2011–2016 data. Food Sci. Nutr. 2021, 9, 1504–1511. [Google Scholar] [CrossRef]
- Çayan, F.; Tel, G.; Duru, M.E.; Öztürk, M.; Türkoğlu, A.; Harmandar, M. Application of GC, GC-MSD, ICP-MS and spectrophotometric methods for the determination of chemical composition and in vitro bioactivities of Chroogomphus rutilus: The edible mushroom species. Food Anal. Method 2014, 7, 449–458. [Google Scholar] [CrossRef]
- Cheung, P.C.K. The nutritional and health benefits of mushrooms. Nutr. Bull. 2010, 35, 292–299. [Google Scholar] [CrossRef]
- Luo, J.; Zhou, W.; Cao, S.; Zhu, H.; Zhang, C.; Jin, M.; Li, G. Chemical constituents of Chroogomphus rutilus (Schaeff.) OK mill. Biochem. Syst. Ecol. 2015, 61, 203–207. [Google Scholar] [CrossRef]
- Zhang, J.; Zhao, X.; Zhao, L.Q.; Zhao, J.; Qi, Z.; Wang, L.A. A primary study of the antioxidant, hypoglycemic, hypolipidemic, and antitumor activities of ethanol extract of brown slimecap mushroom, Chroogomphus rutilus (Agaricomycetes). Int. J. Med. Mushrooms 2017, 19, 905–913. [Google Scholar] [CrossRef]
- Zhang, Y.; Lan, M.; Lü, J.P.; Li, J.F.; Zhang, K.Y.; Zhi, H.; Zhang, H.; Sun, J.M. Antioxidant, anti-inflammatory and cytotoxic activities of polyphenols extracted from Chroogomphus rutilus. Chem. Biodivers. 2020, 17, e1900479. [Google Scholar] [CrossRef]
- Nobsathian, S.; Tuchinda, P.; Sobhon, P.; Tinikul, Y.; Poljaroen, J.; Tinikul, R.; Sroyraya, M.; Poomton, T.; Chaichotranunt, S. An antioxidant activity of the whole body of Holothuria scabra. Chem. Biol. Technol. Agric. 2017, 4, 4. [Google Scholar] [CrossRef]
- Kunsch, C.; Luchoomun, J.; Chen, X.L.; Dodd, G.L.; Karu, K.S.; Meng, C.Q.; Marino, E.M.; Olliff, L.K.; Piper, J.D.; Qiu, F.H.; et al. AGIX-4207 [2-[4-[[1-[[3, 5-bis (1, 1-dimethylethyl)-4-hydroxyphenyl] thio]-1-methylethyl] thio]-2, 6-bis (1, 1-dimethylethyl) phenoxy] acetic acid], a novel antioxidant and anti-inflammatory compound: Cellular and biochemical characterization of antioxidant activity and inhibition of redox-sensitive inflammatory gene expression. J. Pharmacol. Exp. Ther. 2005, 313, 492–501. [Google Scholar]
- Akbas, E.; Ekin, S.; Ergan, E.; Karakus, Y. Synthesis, DFT calculations, spectroscopy and in vitro antioxidant activity studies on 4-hydroxyphenyl substituted thiopyrimidine derivatives. J. Mol. Struct. 2018, 1174, 177–183. [Google Scholar] [CrossRef]
- Choi, K.H.; Nam, K.C.; Lee, S.Y.; Cho, G.; Jung, J.S.; Kim, H.J.; Park, B.J. Antioxidant potential and antibacterial efficiency of caffeic acid-functionalized ZnO nanoparticles. Nanomaterials 2017, 7, 148. [Google Scholar] [CrossRef] [PubMed]
- Jamuna, S.; Karthika, K.; Paulsamy, S.; Thenmozhi, K.; Kathiravan, S.; Venkatesh, R. Confertin and scopoletin from leaf and root extracts of Hypochaeris radicata have anti-inflammatory and antioxidant activities. Ind. Crop. Prod. 2015, 70, 221–230. [Google Scholar] [CrossRef]
- Fernandez-Puntero, B.; Barroso, I.; Iglesias, I.; BENEDÍ, J.; VILLAR, A. Antioxidant activity of Fraxetin: In vivo and ex vivo parameters in normal situation versus induced stress. Biol. Pharm. Bull. 2001, 24, 777–784. [Google Scholar] [CrossRef][Green Version]
- Witaicenis, A.; Seito, L.N.; da Silveira Chagas, A.; de Almeida Junior, L.D.; Luchini, A.C.; Rodrigues-Orsi, P.; Helena Cestari, S.; Di Stasi, L.C. Antioxidant and intestinal anti-inflammatory effects of plant-derived coumarin derivatives. Phytomedicine 2014, 21, 240–246. [Google Scholar] [CrossRef]
- Zhuang, Y.-S. Studies on the Chemical Constituents and Biological Activities of the Thermophilic Fungus, Aspergillus terreus. Master’s Thesis, Taipei University of Technology, Taipei, Taiwan, 2011. [Google Scholar] [CrossRef]
- Shylaja, G.; Sathiavelu, A. Cytotoxicity of endophytic fungus Chaetomium cupreum from the plant Mussaenda luteola against breast cancer cell line MCF-7. Bangl. J. Pharmacol. 2017, 12, 373–375. [Google Scholar] [CrossRef]
- Rojas, J.O.H.N.; Londoño, C.E.S.A.R.; Ciro, Y. The health benefits of natural skin UVA photoprotective compounds found in botanical sources. Int. J. Pharm. Pharm. Sci. 2016, 8, 13–23. [Google Scholar]
- Alesiani, D.; Cicconi, R.; Mattei, M.; Bei, R.; Canini, A. Inhibition of Mek 1/2 kinase activity and stimulation of melanogenesis by 5, 7-dimethoxycoumarin treatment of melanoma cells. Int. J. Oncol. 2009, 34, 1727–1735. [Google Scholar]
- Ohta, A. A metabolic immune checkpoint: Adenosine in tumor microenvironment. Front. Immunol. 2016, 7, 109. [Google Scholar] [CrossRef]
- Huei-Chen, H.; Shu-Hsun, C.; Chao, P.D.L. Vasorelaxants from Chinese herbs, emodin and scoparone, possess immunosuppressive properties. Eur. J. Pharmacol. 1991, 198, 211–213. [Google Scholar] [CrossRef]
- Dobolyi, A.; Juhász, G.; Kovács, Z.; Kardos, J. Uridine function in the central nervous system. Curr. Top. Med. Chem. 2011, 11, 1058–1067. [Google Scholar] [CrossRef]
- Pallam, R.B.; Sarma, V.V. Metabolites of Ganoderma and their Applications in Medicine. In Fungal Biotechnology: Prospects and Avenues; CRC Press: Boca Raton, FL, USA, 2022; p. 175. [Google Scholar]
- Zhao, Y.Y.; Shen, X.; Chao, X.; Ho, C.C.; Cheng, X.L.; Zhang, Y.; Lin, R.C.; Du, K.J.; Luo, W.J.; Chen, J.Y.; et al. Ergosta-4, 6, 8 (14), 22-tetraen-3-one induces G2/M cell cycle arrest and apoptosis in human hepatocellular carcinoma HepG2 cells. Biochim. Biophys. Acta-Gen. Subj. 2011, 1810, 384–390. [Google Scholar] [CrossRef]
- Pereira, A.; Bester, M.; Soundy, P.; Apostolides, Z. Anti-proliferative properties of commercial Pelargonium sidoides tincture, with cell-cycle G0/G1 arrest and apoptosis in Jurkat leukaemia cells. Pharm. Biol. 2016, 54, 1831–1840. [Google Scholar] [CrossRef] [PubMed]
- Son, D.J.; Lee, G.R.; Oh, S.; Lee, S.E.; Choi, W.S. Gastroprotective efficacy and safety evaluation of scoparone derivatives on experimentally induced gastric lesions in rodents. Nutrients 2015, 7, 1945–1964. [Google Scholar] [CrossRef] [PubMed]
- Chodurek, E.; Orchel, A.; Orchel, J.; Kurkiewicz, S.; Gawlik, N.; Dzierżewicz, Z.; Stępień, K. Evaluation of melanogenesis in A-375 melanoma cells treated with 5, 7-dimethoxycoumarin and valproic acid. Cell Mol. Biol. Lett. 2012, 17, 616–632. [Google Scholar] [CrossRef] [PubMed]
- Fort, D.M.; Rao, K.; Jolad, S.D.; Luo, J.; Carlson, T.J.; King, S.R. Antihyperglycemic activity of Teramnus labialis (Fabaceae). Phytomedicine 2000, 6, 465–467. [Google Scholar] [CrossRef]
- Ding, Z.; Dai, Y.; Hao, H.; Pan, R.; Yao, X.; Wang, Z. Anti-inflammatory effects of scopoletin and underlying mechanisms. Pharm. Biol. 2008, 46, 854–860. [Google Scholar] [CrossRef]
- Marshall, M.E.; Kervin, K.; Benefield, C.; Umerani, A.; Albainy-Jenei, S.; Zhao, Q.; Khazaeli, M.B. Growth-inhibitory effects of coumarin (1, 2-benzopyrone) and 7-hydroxycoumarin on human malignant cell lines in vitro. J. Cancer Res. Clin. 1994, 120, S3–S10. [Google Scholar] [CrossRef]
- Bonaterra, G.A.; Schwarzbach, H.; Kelber, O.; Weiser, D.; Kinscherf, R. Anti-inflammatory effects of Phytodolor®(STW 1) and components (poplar, ash and goldenrod) on human monocytes/macrophages. Phytomedicine 2019, 58, 152868. [Google Scholar] [CrossRef]
- Chu, C.Y.; Tsai, Y.Y.; Wang, C.J.; Lin, W.L.; Tseng, T.H. Induction of apoptosis by esculetin in human leukemia cells. Eur. J. Pharmacol. 2001, 416, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Kalyoncu, F.; Oskay, M.; Kayalar, H. Antioxidant activity of the mycelium of 21 wild mushroom species. Mycology 2010, 1, 195–199. [Google Scholar] [CrossRef]
- Wang, J.; Hu, S.; Nie, S.; Yu, Q.; Xie, M. Reviews on mechanisms of in vitro antioxidant activity of polysaccharides. Oxid. Med. Cell Longev. 2016, 2016, 5692852. [Google Scholar] [CrossRef]
- López-Alarcón, C.; Denicola, A. Evaluating the antioxidant capacity of natural products: A review on chemical and cellular-based assays. Anal. Chim. Acta. 2013, 763, 1–10. [Google Scholar] [CrossRef]
- Valko, M.; Rhodes, C.J.B.; Moncol, J.; Izakovic, M.M.; Mazur, M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem-Biol. Interact. 2006, 160, 1–40. [Google Scholar] [CrossRef] [PubMed]
- Li, Z. Research on Chemical Constituents and Anti-Tumor Activity of Chroogomphus rutilus. Master’s Thesis, Jilin Agricultural University, Changchun, China, 2014. [Google Scholar]
- Mohamed, S.I.A.; Jantan, I.; Haque, M.A. Naturally occurring immunomodulators with antitumor activity: An insight on their mechanisms of action. Int. Immunopharmacol. 2017, 50, 291–304. [Google Scholar] [CrossRef]
- Park, H.J. Current uses of mushrooms in cancer treatment and their anticancer mechanisms. Int. J. Mol. Sci. 2022, 23, 10502. [Google Scholar] [CrossRef]
- Jana, P.; Acharya, K. Mushroom: A new resource for anti-angiogenic therapeutics. Food Rev. Int. 2022, 38, 88–109. [Google Scholar] [CrossRef]
- Luo, C.; Xu, X.; Wei, X.; Feng, W.; Huang, H.; Liu, H.; Xu, R.; Lin, J.; Han, L.; Zhang, D. Natural medicines for the treatment of fatigue: Bioactive components, pharmacology, and mechanisms. Pharmacol. Res. 2019, 148, 104409. [Google Scholar] [CrossRef]
- Zhou, Y.; El-Seedi, H.R.; Xu, B. Insights into health promoting effects and myochemical profiles of pine mushroom Tricholoma matsutake. Crit. Rev. Food Sci. 2021, 63, 5698–5723. [Google Scholar] [CrossRef] [PubMed]
- Villaño, D.; Fernández-Pachón, M.S.; Moyá, M.L.; Troncoso, A.M.; García-Parrilla, M.C. Radical scavenging ability of polyphenolic compounds towards DPPH free radical. Talanta 2007, 71, 230–235. [Google Scholar] [CrossRef] [PubMed]
- Losada-Barreiro, S.; Bravo-Diaz, C. Free radicals and polyphenols: The redox chemistry of neurodegenerative diseases. Eur. J. Med. Chem. 2017, 133, 379–402. [Google Scholar] [CrossRef] [PubMed]
- Tsivileva, O.M.; Koftin, O.V.; Evseeva, N.V. Coumarins as fungal metabolites with potential medicinal properties. Antibiotics 2022, 11, 1156. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, J.Q.; Zhang, J.; Li, Z.M.; Liu, H.G.; Wang, Y.Z. Traditional uses, chemical components and pharmacological activities of the genus Ganoderma P. Karst.: A review. Rsc. Adv. 2020, 10, 42084–42097. [Google Scholar] [CrossRef] [PubMed]
- Roosta, Z.; Hajimoradloo, A.; Ghorbani, R.; Hoseinifar, S.H. The effects of dietary vitamin C on mucosal immune responses and growth performance in Caspian roach (Rutilus rutilus caspicus) fry. Fish Physiol. Biochem. 2014, 40, 1601–1607. [Google Scholar] [CrossRef]
- Kaur, C.; Kapoor, H.C. Antioxidants in fruits and vegetables–the millennium’s health. Int. J. Food Sci. Tech. 2001, 36, 703–725. [Google Scholar] [CrossRef]
- Vázquez, E.L.; García, F.P.; Canales, M.G. Major and trace minerals present in wild mushrooms. Am.-Eurasian J. Agric. Environ. Sci. 2016, 16, 1145–1158. [Google Scholar]
- Aslani, B.A.; Ghobadi, S. Studies on oxidants and antioxidants with a brief glance at their relevance to the immune system. Life Sci. 2016, 146, 163–173. [Google Scholar] [CrossRef]
- Zhang, H.; Yang, X.; Li, Z.; Yu, H. Study on antioxidant activity of Chroogomphus rutilus flavonoids in vitro. Food Res. Dev. 2016, 37, 109–113. [Google Scholar]
- Petrushanko, I.; Bogdanov, N.; Bulygina, E.; Grenacher, B.; Leinsoo, T.; Boldyrev, A.; Gassmann, M.; Bogdanova, A. Na-K-ATPase in rat cerebellar granule cells is redox sensitive. Am. J. Physiol-Reg. I 2006, 290, R916–R925. [Google Scholar] [CrossRef]
- Jayathilake, C.; Rizliya, V.; Liyanage, R. Antioxidant and free radical scavenging capacity of extensively used medicinal plants in Sri Lanka. Procedia Food Sci. 2016, 6, 123–126. [Google Scholar] [CrossRef]
- Wolfe, K.L.; Liu, R.H. Cellular antioxidant activity (CAA) assay for assessing antioxidants, foods, and dietary supplements. J. Agric. Food Chem. 2007, 55, 8896–8907. [Google Scholar] [CrossRef] [PubMed]
- Ifeanyi, O.E. A review on free radicals and antioxidants. Int. J. Curr. Res. Med. Sci. 2018, 4, 123–133. [Google Scholar]
- Betteridge, D.J. What is oxidative stress? Metabolism 2000, 49, 3–8. [Google Scholar] [CrossRef]
- Engwa, G.A. Free radicals and the role of plant phytochemicals as antioxidants against oxidative stress-related diseases. In Phytochemicals: Source of Antioxidants and Role in Disease Prevention; BoD–Books on Demand: Norderstedt, Germany, 2018; Volume 7, pp. 49–74. [Google Scholar]
- Liu, Q.; Zhu, M.; Geng, X.; Wang, H.; Ng, T.B. Characterization of polysaccharides with antioxidant and hepatoprotective activities from the edible mushroom Oudemansiella radicata. Molecules 2017, 22, 234. [Google Scholar] [CrossRef]
- Rajendran, K.; Karthikeyan, A.; Krishnan, U.M. Emerging trends in nano-bioactive-mediated mitochondria-targeted therapeutic stratagems using polysaccharides, proteins and lipidic carriers. Int. J. Biol. Macromol. 2022, 208, 627–641. [Google Scholar] [CrossRef]
- Lv, J.; Han, R.; Huang, Z.; Luo, L.; Cao, D.; Zhang, S. Relationship between molecular components and reducing capacities of humic substances. Acs. Earth Space Chem. 2018, 2, 330–339. [Google Scholar] [CrossRef]
- Zhang, H.; Tsao, R. Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. Curr. Opin. Food Sci. 2016, 8, 33–42. [Google Scholar] [CrossRef]
- Zhang, X.; Sun, H.; Wang, L.; Wang, Y.; Liu, C.; Bai, Y.; Li, J.; Guan, Z. Protective effect of crude polysaccharide from Chroogomphus rutilus on MPTP injury in mouse DA neurons. Mycosystema 2011, 30, 77–84. [Google Scholar]
- Zhao, B.; Zhao, J.; Lv, M.; Li, X.; Wang, J.; Yue, Z.; Shi, J.; Zhang, G.; Sui, G. Comparative study of structural properties and biological activities of polysaccharides extracted from Chroogomphus rutilus by four different approaches. Int. J. Biol. Macromol. 2021, 188, 215–225. [Google Scholar] [CrossRef]
- Ghiselli, A.; Serafini, M.; Natella, F.; Scaccini, C. Total antioxidant capacity as a tool to assess redox status: Critical view and experimental data. Free Radical Bio. Med. 2000, 29, 1106–1114. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.E.; Hwang, H.J.; Ha, J.S.; Jeong, H.S.; Kim, J.H. Screening of medicinal plant extracts for antioxidant activity. Life Sci. 2003, 73, 167–179. [Google Scholar] [CrossRef]
- Du, J.; Zhang, H.; Chen, P. GC-MS analysis of chemical constituents of fruiting body of Chroogomphus rutilus and screening of anti-tumor active components of petroleum ether extract. Jiangsu Agric. Sci. 2019, 10, 197–201. [Google Scholar]
- Ouyang, F.; Wang, G.; Guo, W.; Zhang, Y.; Xiang, W.; Zhao, M. AKT signalling and mitochondrial pathways are involved in mushroom polysaccharide-induced apoptosis and G1 or S phase arrest in human hepatoma cells. Food Chem. 2013, 138, 2130–2139. [Google Scholar] [CrossRef]
- Chen, J.H.; Cao, J.L.; Chu, Y.L.; Wang, Z.L.; Yang, Z.T.; Wang, H.L. T-2 toxin-induced apoptosis involving Fas, p53, Bcl-xL, Bcl-2, Bax and caspase-3 signaling pathways in human chondrocytes. J. Zhejiang Univ. Sci. B 2008, 9, 455–463. [Google Scholar] [CrossRef]
- SEVİNDİK, M. Antioxidant and oxidant potantials and element contents of Chroogomphus rutilus (Agaricomycetes). Mantar Dergisi 2021, 12, 29–32. [Google Scholar]
- Hicklin, D.J.; Ellis, L.M. Role of the vascular endothelial growth factor pathway in tumor growth and angiogenesis. J. Clin. Oncol. 2005, 23, 1011–1027. [Google Scholar] [CrossRef]
- Pickard, J.M.; Zeng, M.Y.; Caruso, R.; Núñez, G. Gut microbiota: Role in pathogen colonization, immune responses, and inflammatory disease. Immunol. Rev. 2017, 279, 70–89. [Google Scholar] [CrossRef]
- Sun, M.; He, C.; Cong, Y.; Liu, Z. Regulatory immune cells in regulation of intestinal inflammatory response to microbiota. Mucosal Immunol. 2015, 8, 969–978. [Google Scholar] [CrossRef] [PubMed]
- Asadullah, K.; Sterry, W.; Trefzer, U. Cytokines: Interleukin and interferon therapy in dermatology. Clin. Exp. Dermatol. 2002, 27, 578–584. [Google Scholar] [CrossRef] [PubMed]
- Akpi, U.K. In Vitro Antimicrobial Activities of Some Selected Edible and Non-Edible Mushrooms. Ph.D. Thesis, Universiti Kebangsaan Malaysia, Bangi, Malaysia, 2017. [Google Scholar]
- Geng, P.; Siu, K.C.; Wang, Z.; Wu, J.Y. Antifatigue functions and mechanisms of edible and medicinal mushrooms. Biomed. Res. Int. 2017, 2017, 9648496. [Google Scholar] [CrossRef]
- Zhao, S.; Gao, Q.; Rong, C.; Wang, S.; Zhao, Z.; Liu, Y.; Xu, J. Immunomodulatory effects of edible and medicinal mushrooms and their bioactive immunoregulatory products. J. Fungus 2020, 6, 269. [Google Scholar] [CrossRef]
- Jiang, W.; Xu, J. Immune modulation by mesenchymal stem cells. Cell Proliferat. 2020, 53, e12712. [Google Scholar] [CrossRef]
- Cifaldi, L.; Prencipe, G.; Caiello, I.; Bracaglia, C.; Locatelli, F.; De Benedetti, F.; Strippoli, R. Inhibition of natural killer cell cytotoxicity by interleukin-6: Implications for the pathogenesis of macrophage activation syndrome. Arthritis Rheumatol. 2015, 67, 3037–3046. [Google Scholar] [CrossRef]
- Rizza, P.; Moretti, F.; Belardelli, F. Recent advances on the immunomodulatory effects of IFN-α: Implications for cancer immunotherapy and autoimmunity. Autoimmunity 2010, 43, 204–209. [Google Scholar] [CrossRef]
- Ganeshpurkar, A.; Saluja, A. Immunomodulatory effect of rutin, catechin, and hesperidin on macrophage function. Indian J. Biochem. Bio. 2020, 57, 58–63. [Google Scholar]
- Lewis, E.D.; Meydani, S.N.; Wu, D. Regulatory role of vitamin E in the immune system and inflammation. Iubmb. Life 2019, 71, 487–494. [Google Scholar] [CrossRef]
- Mousavi, S.; Bereswill, S.; Heimesaat, M.M. Immunomodulatory and antimicrobial effects of vitamin C. Eur. J. Microbiol. Immunol. 2019, 9, 73–79. [Google Scholar] [CrossRef] [PubMed]
- El-Senousey, H.K.; Chen, B.; Wang, J.Y.; Atta, A.M.; Mohamed, F.R.; Nie, Q.H. Effects of dietary vitamin C, vitamin E, and alpha-lipoic acid supplementation on the antioxidant defense system and immune-related gene expression in broilers exposed to oxidative stress by dexamethasone. Poultry Sci. 2018, 97, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Li, Z. Extraction and Alleviate Physical Fatigue of Polysaccharides from Gomphidius rutilus. Master’s Thesis, Changchun University of Technology, Changchun, China, 2016. [Google Scholar]
- Malaguti, M.; Angeloni, C.; Hrelia, S. Polyphenols in exercise performance and prevention of exercise-induced muscle damage. Oxid. Med. Cell. Longev. 2013, 2013, 825928. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Lv, J.; Lo, Y.M.; Cui, S.W.; Hu, X.; Fan, M. Effects of oat β-glucan on endurance exercise and its anti-fatigue properties in trained rats. Carbohyd. Polym. 2013, 92, 1159–1165. [Google Scholar] [CrossRef] [PubMed]
- Filippin, L.I.; Moreira, A.J.; Marroni, N.P.; Xavier, R.M. Nitric oxide and repair of skeletal muscle injury. Nitric Oxide-Biol. Ch. 2009, 21, 157–163. [Google Scholar] [CrossRef]
- Yu, W.; Zeng, D.; Xiong, Y.; Shan, S.; Yang, X.; Zhao, H.; Lu, W. Health benefits of functional plant polysaccharides in metabolic syndrome: An overview. J. Funct. Foods 2022, 95, 105154. [Google Scholar] [CrossRef]
- Gong, L.; Feng, D.; Wang, T.; Ren, Y.; Liu, Y.; Wang, J. Inhibitors of α-amylase and α-glucosidase: Potential linkage for whole cereal foods on prevention of hyperglycemia. Food Sci. Nutr. 2020, 8, 6320–6337. [Google Scholar] [CrossRef]
- Deveci, E.; Çayan, F.; Tel-Çayan, G.; Duru, M.E. Inhibitory activities of medicinal mushrooms on α-amylase and α-glucosidase-enzymes related to type 2 diabetes. S. Afr. J. Bot. 2021, 137, 19–23. [Google Scholar] [CrossRef]
- Lv, M.S. Structure Characterization and Biological Activities of Polysaccharide from Chroogomphis rutilus. Master’s Thesis, Northeast Forestry University, Harbin, China, 2021. [Google Scholar]
- He, X.; Wang, X.; Fang, J.; Chang, Y.; Ning, N.; Guo, H.; Huang, L.; Huang, X.; Zhao, Z. Structures, biological activities, and industrial applications of the polysaccharides from Hericium erinaceus (Lion’s Mane) mushroom: A review. Int. J. Biol. Macromol. 2017, 97, 228–237. [Google Scholar] [CrossRef]
- Chen, W.; Wu, D.; Jin, Y.; Li, Q.; Liu, Y.; Qiao, X.; Zhang, J.; Dong, G.; Li, Z.; Li, T.; et al. Pre-protective effect of polysaccharides purified from Hericium erinaceus against ethanol-induced gastric mucosal injury in rats. Int. J. Biol. Macromol. 2020, 159, 948–956. [Google Scholar] [CrossRef] [PubMed]
- Bequette, B.J. Amino acid metabolism in animals: An overview. Amino Acids Anim. Nutr. 2003, 2, 103–124. [Google Scholar]
- Sirisinha, S. The pleiotropic role of vitamin A in regulating mucosal immunity. Asian Pac. J. Allergy Immunol. 2015, 33, 71–89. [Google Scholar]
- Wu, Q.; Wang, Q.; Fu, J.; Ren, R. Polysaccharides derived from natural sources regulate triglyceride and cholesterol metabolism: A review of the mechanisms. Food Funct. 2019, 10, 2330–2339. [Google Scholar] [CrossRef] [PubMed]
- Ganesan, K.; Xu, B. Anti-diabetic effects and mechanisms of dietary polysaccharides. Molecules 2019, 24, 2556. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Moreno, J.; Martínez-Reyes, M. Edible ectomycorrhizal mushrooms: Biofactories for sustainable development. In Biosystems Engineering: Biofactories for Food Production in the Century XXI; Springer International Publishing: Cham, Switzerland, 2014; pp. 151–233. [Google Scholar]
- Ghorbanpour, M.; Omidvari, M.; Abbaszadeh-Dahaji, P.; Omidvar, R.; Kariman, K. Mechanisms underlying the protective effects of beneficial fungi against plant diseases. Biol. Control 2018, 117, 147–157. [Google Scholar] [CrossRef]
- Amor, S.; Puentes, F.; Baker, D.; Van Der Valk, P. Inflammation in neurodegenerative diseases. Immunology 2010, 129, 154–169. [Google Scholar] [CrossRef]
- Leszek, J.; E Barreto, G.; Gasiorowski, K.; Koutsouraki, E.; Aliev, G. Inflammatory mechanisms and oxidative stress as key factors responsible for progression of neurodegeneration: Role of brain innate immune system. Cns. Neurol. Disord-Drug 2016, 15, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.L.; Chen, Z.G.; Dai, H.L.; Ding, J.X.; Guo, J.S.; Han, N.; Jiang, B.G.; Jiang, H.J.; Li, J.; Li, S.P.; et al. Repair, protection and regeneration of peripheral nerve injury. Neural. Regen. Res. 2015, 10, 1777. [Google Scholar] [CrossRef] [PubMed]

| Components | Classification | Structure | Function | References |
|---|---|---|---|---|
| 4-Hydroxybenzaldehyde | hydroxybenzaldehyde | 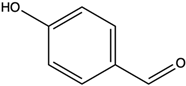 | Scavenged free radicals and promoted antioxidation | [47] |
| (4-hydroxyphenyl) acetic acid | monocarboxylic acid | 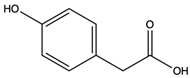 | Selectively inhibited tumor necrosis factor (TNF)-α-inducible levels of the redox-sensitive genes, vascular cell adhesion molecule-1, and monocyte chemoattractant protein-1 | [48] |
| Methyl (4-hydroxyphenyl) acetate | methyl ester |  | Inhibited phenyl hydrazine-induced hemolysis of erythrocytes to scavenge most of the free radicals generated | [49] |
| 3-(3,4-Dihydroxyphenyl)-2-propenoic acid | monocarboxylic acid |  | Scavenged free radicals and promoted antioxidation | [50] |
| Scopoletin | coumarin |  | Scavenged free radicals and promoted antioxidation. Activated some key antioxidant enzymes, such as superoxide dismutase (SOD), glutathione peroxidase (GPx), and glutathione -S- transferase (GST) to enhance the antioxidant defense system of cells | [51] |
| Fraxetin | coumarin | 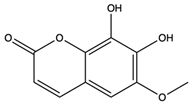 | Scavenged free radicals and promoted antioxidation | [52] |
| Esculin | coumarin | 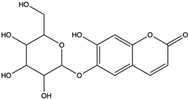 | Scavenged free radicals and promoted antioxidation | [53] |
| 5a, 8a-Epidioxyergosta-6, 22-dien-3b-ol | ergosterol peroxide | 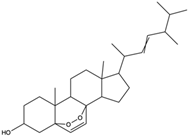 | Induced a cytotoxic effect on the OECM-1 cell strain and exerted an anti-tumor role | [54] |
| 3b,5a-Dihydroxy-(22E, 24R)-ergosta-7,22-dien-6-one | ergostanoid |  | Exerted a cytotoxic effect on the MCF-7 cell strain and fulfilled an anti-tumor role | [55] |
| 6-Hydroxy-5,7-dimethoxycoumarin | coumarins |  | Induced a cytotoxic effect on the L1210 cell strain and played an anti-tumor role | [56] |
| 5,7-Dimethoxycoumarin | coumarins | 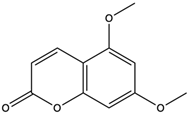 | Inhibited Mek 1/2 kinase activity and stimulated melanin production to inhibit melanoma | [57] |
| Adenosine | nucleoside | 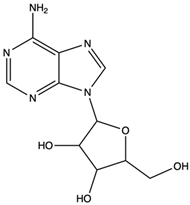 | Inhibited the activities of T cells (proliferation, cytokine production, and cytotoxicity), NK cells (cytotoxicity), NKT cells (cytokine production and CD40L up-regulation), macrophages/dendritic cells (antigen presentation and cytokine production), and neutrophils (oxidative burst) | [58] |
| Scoparone | coumarins | 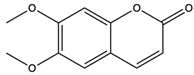 | Suppressed the responses of human mononuclear cells to phytohemagglutinin and mixed lymphocyte reaction for use against transplantation rejection and autoimmune disease | [59] |
| Uridine | nucleoside | 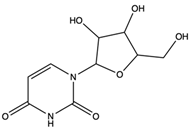 | Uridine is phosphorylated into nucleotides for the synthesis of DNA and RNA, as well as the synthesis of membrane components and glycosylation. Uridine nucleotides and UDP sugars may be released from neurons and glial cells. Used as neuroprotective agent for treating neurodegenerative diseases | [60] |
| 5a-Ergosta-7,22-dien-3-one (Stellasterin) | lanostanoids | 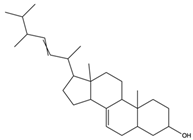 | (External α-sialidase) inhibitors and antifungal agents, which play an immunomodulatory role. Hydroxyl groups can separate polar lipids and reduce blood fat and cholesterol | [61] |
| Ergosta-4,6,8(14),22tetraene-3-one | ergostanoid | 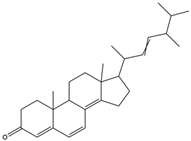 | Induced G2/M cell cycle arrest and apoptosis in human hepatocellular carcinoma HepG2 cells | [62] |
| 6-Hydroxy-5,7-dimethoxycoumarin (Fraxinol) | coumarins |  | Inhibited the growth of Jurkat cell line tumor cells | [63] |
| 6,7,8-Trimethoxycoumarin | coumarins | 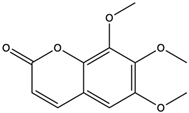 | The gastric protective activity of GU induced by HCl/ethanol and indomethacin was improved, resulting in more than 90% reversal of GU | [64] |
| 5,7-Dimethoxycoumarin (citropten) | coumarins | 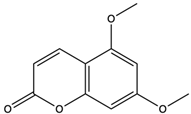 | Inhibited the growth of A-375 melanoma cells | [65] |
| Fraxidin | coumarins | 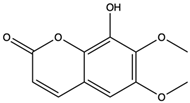 | Exerted inhibitory effects towards aldose reductase activity and platelet aggregation | [66] |
| Scopoletin | coumarins | 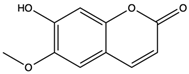 | Scopoletin has obvious anti-inflammatory activity in inhibiting the overproduction of PGE2 and TNF-α and neutrophil infiltration | [67] |
| 7-Hydroxycoumarin | coumarins |  | Inhibited [3H]thymidine, [3H]uridine, and [3H]leucine incorporation. Inhibited the intracellular production of prostate-specific antigen by LNCaP cells. Have direct antitumor (cytostatic) activity as well as immunomodulatory activity | [68] |
| Phytodolor | coumarins |  | The intracellular content of TNF-α and PTGS2 protein and the expression of TNF-α and PTGS2 gene were inhibited, and the induced apoptosis of LPS-activated human monocytes was inhibited in the absence of serum. In addition, phytodolor inhibited the translocation of p65 subunit of redox-regulated NF-κB in LPS-activated human macrophage nuclei. Played an anti-tumor role | [69] |
| Esculetin | coumarins | 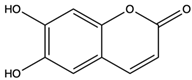 | Exerted a cytotoxic effect on the Leukemia HL-60 cell strain and fulfilled an anti-tumor role | [70] |
| 3b,5a,9a-Trihydroxy-(22E,24R)-ergosta-7,22-dien-6-one | ergostanoid |  | / | / |
| 5a-Ergosta-7-en-3b-ol | ergostanoid | 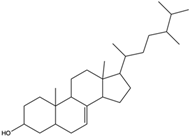 | / | / |
| Compounds or Extracts | Biological Activity | Method | References |
|---|---|---|---|
| 2-Methoxyadenine nucleoside, flavone | Antioxidant activity | Separating and purifying fruiting bodies and preparing macroporous resin | [39] |
| Polysaccharide | Antifatigue activity | Mice were given low (100 mg/kg/d), medium (250 mg/kg/d), and high (625 mg/kg/d) doses of Pleurotus eryngii polysaccharide | [75] |
| Polysaccharides (β-glucan and α-glucan) | Immunomodulatory activity | Organic extraction | [46] |
| Crude extract | Antitumor activity | Organic extraction (95% ethanol) | [45] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, B.; Luo, J.; Xu, B. Insights into the Chemical Compositions and Health Promoting Effects of Wild Edible Mushroom Chroogomphus rutilus. Nutrients 2023, 15, 4030. https://doi.org/10.3390/nu15184030
Han B, Luo J, Xu B. Insights into the Chemical Compositions and Health Promoting Effects of Wild Edible Mushroom Chroogomphus rutilus. Nutrients. 2023; 15(18):4030. https://doi.org/10.3390/nu15184030
Chicago/Turabian StyleHan, Bincheng, Jinhai Luo, and Baojun Xu. 2023. "Insights into the Chemical Compositions and Health Promoting Effects of Wild Edible Mushroom Chroogomphus rutilus" Nutrients 15, no. 18: 4030. https://doi.org/10.3390/nu15184030
APA StyleHan, B., Luo, J., & Xu, B. (2023). Insights into the Chemical Compositions and Health Promoting Effects of Wild Edible Mushroom Chroogomphus rutilus. Nutrients, 15(18), 4030. https://doi.org/10.3390/nu15184030








