Targeting Glutamate Neurotoxicity through Dietary Manipulation: Potential Treatment for Migraine
Abstract
1. Introduction
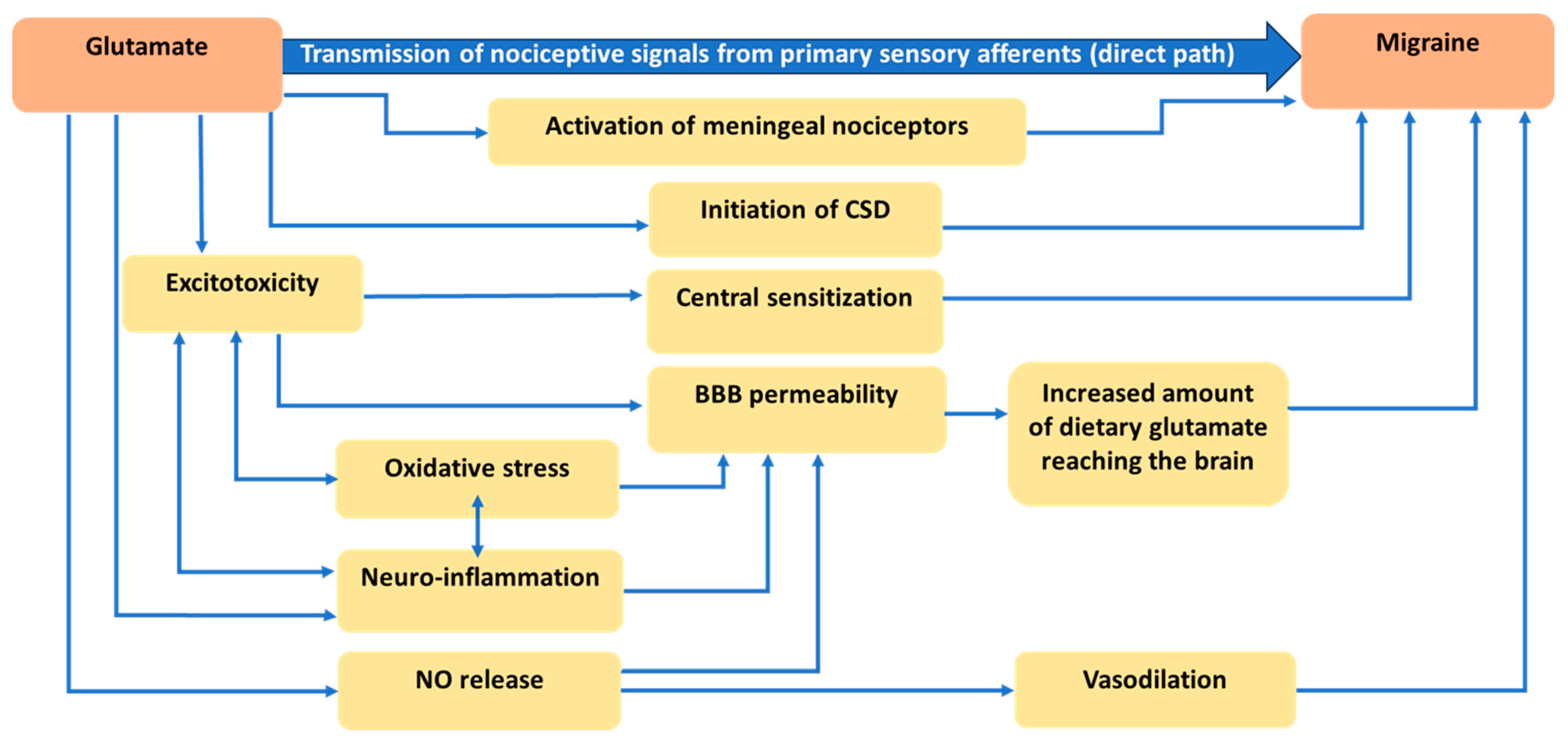
2. Glutamatergic Neurotransmission
3. Physiological and Anatomical Evidence Related to the Role of Glutamate in Migraine
3.1. Role of Glutamate in Nociception
3.2. Role of Glutamate in Cortical Spreading Depression
3.3. Role of Glutamate in Central Sensitization
3.4. Role of Glutamate in Disruption of the Blood-Brain Barrier (BBB)
3.5. Role of Glutamate in Nitric Oxide Release and Vasodilation
4. Glutamate Concentration in Migraineurs
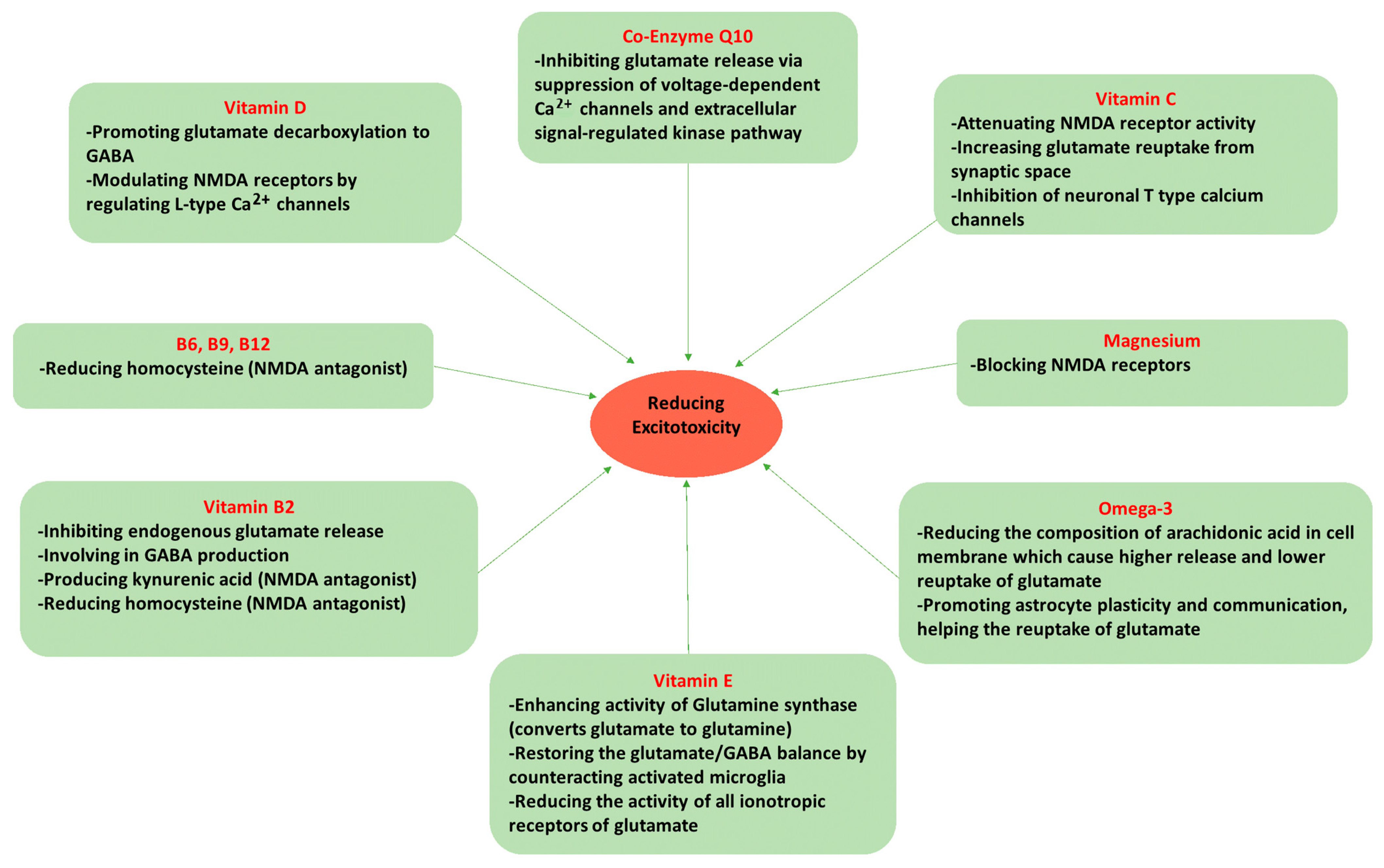
5. Dietary Components Affecting Glutamate Neurotoxicity and Migraine
5.1. Omega-3 Fatty Acids
5.2. Magnesium (Mg2+)
5.3. Vitamin D
5.4. Vitamin C
5.5. Vitamin E
5.6. Riboflavin (Vitamin B2)
5.7. Vitamin B6 (Pyridoxine), Folate (Vitamin B9), and Vitamin B12 (Cobalamin)
5.8. Coenzyme Q10 (CoQ10)
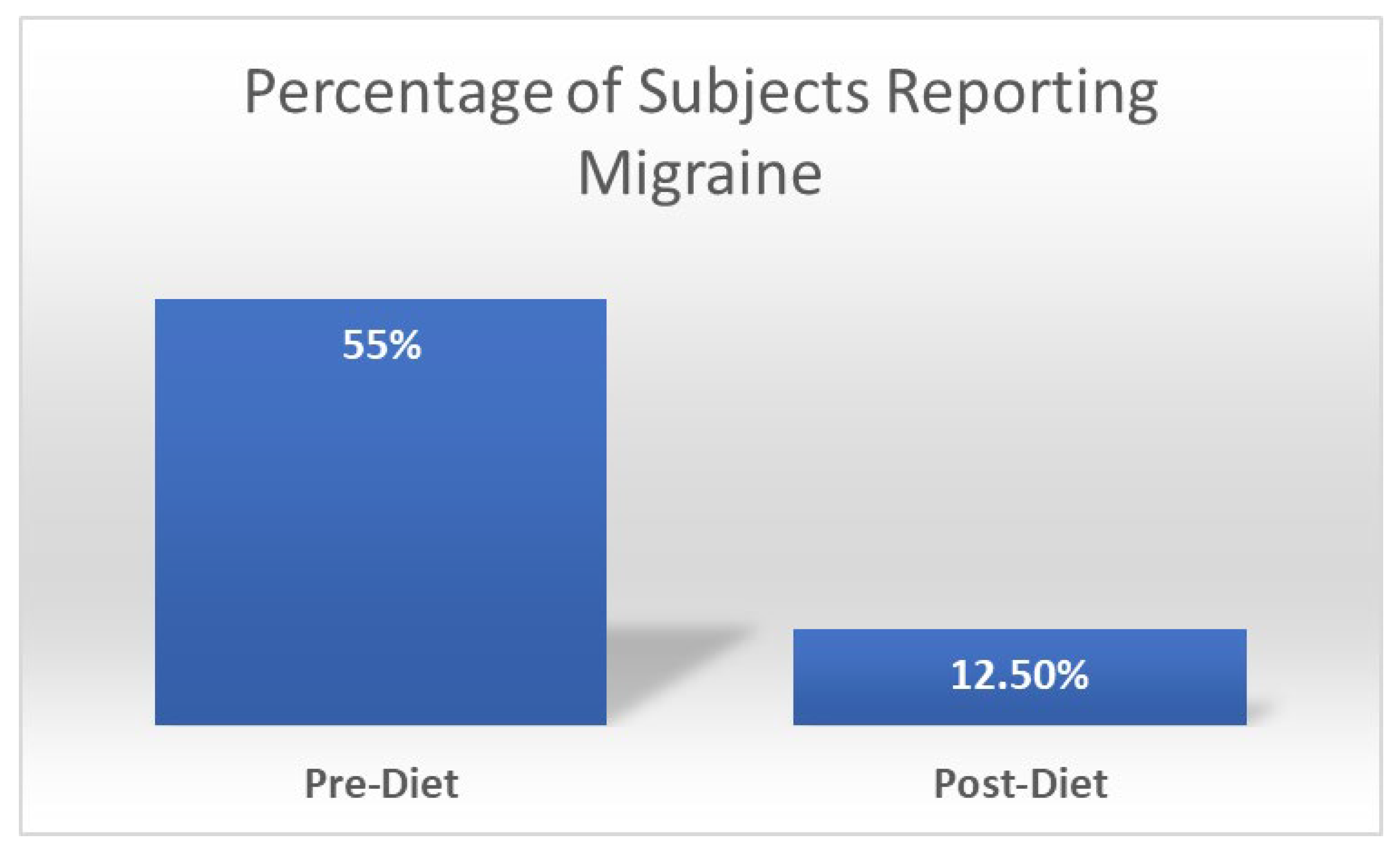
6. Gap between Pathophysiology of Migraine and Interventions: Where Do We Stand Now?
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Woldeamanuel, Y.W.; Cowan, R.P. Migraine affects 1 in 10 people worldwide featuring recent rise: A systematic review and meta-analysis of community-based studies involving 6 million participants. J. Neurol. Sci. 2017, 372, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Stovner, L.J.; Zwart, J.A.; Hagen, K.; Terwindt, G.; Pascual, J. Epidemiology of headache in Europe. Eur. J. Neurol. 2006, 13, 333–345. [Google Scholar] [CrossRef] [PubMed]
- Steiner, T.J.; Stovner, L.J.; Vos, T.; Jensen, R.; Katsarava, Z. Migraine is First Cause of Disability in Under 50s: Will Health Politicians Now Take Notice? Springer: Berlin/Heidelberg, Germany, 2018; Volume 19, pp. 1–4. [Google Scholar]
- Bonafede, M.; Sapra, S.; Shah, N.; Tepper, S.; Cappell, K.; Desai, P. Direct and indirect healthcare resource utilization and costs among migraine patients in the United States. Headache J. Head Face Pain 2018, 58, 700–714. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.-J.; Chen, P.-K.; Fuh, J.-L. Comorbidities of migraine. Front. Neurol. 2010, 1, 16. [Google Scholar] [CrossRef]
- Katsarava, Z.; Mania, M.; Lampl, C.; Herberhold, J.; Steiner, T.J. Poor medical care for people with migraine in Europe–evidence from the Eurolight study. J. Headache Pain 2018, 19, 10. [Google Scholar] [CrossRef]
- Ashina, M.; Katsarava, Z.; Do, T.P.; Buse, D.C.; Pozo-Rosich, P.; Özge, A.; Krymchantowski, A.V.; Lebedeva, E.R.; Ravishankar, K.; Yu, S. Migraine: Epidemiology and systems of care. Lancet 2021, 397, 1485–1495. [Google Scholar] [CrossRef]
- Ashina, M.; Hansen, J.M.; Do, T.P.; Melo-Carrillo, A.; Burstein, R.; Moskowitz, M.A. Migraine and the trigeminovascular system—40 years and counting. Lancet Neurol. 2019, 18, 795–804. [Google Scholar] [CrossRef]
- May, A.; Goadsby, P.J. The Trigeminovascular System in Humans: Pathophysiologic Implications for Primary Headache Syndromes of the Neural Influences on the Cerebral Circulation. J. Cereb. Blood Flow Metab. 1999, 19, 115–127. [Google Scholar] [CrossRef]
- Ramadan, N.M. The link between glutamate and migraine. CNS Spectr. 2003, 8, 446–449. [Google Scholar] [CrossRef]
- Vikelis, M.; Mitsikostas, D.D. The role of glutamate and its receptors in migraine. CNS Neurol. Disord.-Drug Targets (Former. Curr. Drug Targets-CNS Neurol. Disord.) 2007, 6, 251–257. [Google Scholar]
- Guerrero-Toro, C.; Koroleva, K.; Ermakova, E.; Gafurov, O.; Abushik, P.; Tavi, P.; Sitdikova, G.; Giniatullin, R. Testing the Role of Glutamate NMDA Receptors in Peripheral Trigeminal Nociception Implicated in Migraine Pain. Int. J. Mol. Sci. 2022, 23, 1529. [Google Scholar] [CrossRef] [PubMed]
- Lauritzen, M.; Hansen, A.J. The effect of glutamate receptor blockade on anoxic depolarization and cortical spreading depression. J. Cereb. Blood Flow Metab. 1992, 12, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Gallai, V.; Alberti, A.; Gallai, B.; Coppola, F.; Floridi, A.; Sarchielli, P. Glutamate and nitric oxide pathway in chronic daily headache: Evidence from cerebrospinal fluid. Cephalalgia 2003, 23, 166–174. [Google Scholar] [CrossRef]
- Sarchielli, P.; Filippo, M.; Nardi, K.; Calabresi, P. Sensitization, glutamate, and the link between migraine and fibromyalgia. Curr. Pain Headache Rep. 2007, 11, 343–351. [Google Scholar] [CrossRef]
- Ramachandran, R. Neurogenic inflammation and its role in migraine. In Seminars in Immunopathology; Springer: Berlin/Heidelberg, Germany, 2018; pp. 301–314. [Google Scholar]
- Landy, S.; Rice, K.; Lobo, B. Central sensitisation and cutaneous allodynia in migraine: Implications for treatment. CNS Drugs 2004, 18, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Latremoliere, A.; Woolf, C.J. Central sensitization: A generator of pain hypersensitivity by central neural plasticity. J. Pain 2009, 10, 895–926. [Google Scholar] [CrossRef]
- Woolf, C.J.; Thompson, S.W. The induction and maintenance of central sensitization is dependent on N-methyl-D-aspartic acid receptor activation; implications for the treatment of post-injury pain hypersensitivity states. Pain 1991, 44, 293–299. [Google Scholar] [CrossRef]
- Olney, J. Excitotoxicity: An overview. Can. Dis. Wkly. Rep.=Rapp. Hebd. Mal. Can. 1990, 16, 47–57, discussion 57. [Google Scholar]
- Holton, K.F. Micronutrients May Be a Unique Weapon Against the Neurotoxic Triad of Excitotoxicity, Oxidative Stress and Neuroinflammation: A Perspective. Front. Neurosci. 2021, 15, 726457. [Google Scholar] [CrossRef]
- Willard, S.S.; Koochekpour, S. Glutamate, glutamate receptors, and downstream signaling pathways. Int. J. Biol. Sci. 2013, 9, 948. [Google Scholar] [CrossRef]
- Riedel, G.; Platt, B.; Micheau, J. Glutamate receptor function in learning and memory. Behav. Brain Res. 2003, 140, 1–47. [Google Scholar] [CrossRef] [PubMed]
- Ohgi, Y.; Futamura, T.; Hashimoto, K. Glutamate signaling in synaptogenesis and NMDA receptors as potential therapeutic targets for psychiatric disorders. Curr. Mol. Med. 2015, 15, 206–221. [Google Scholar] [CrossRef] [PubMed]
- Reiner, A.; Levitz, J. Glutamatergic signaling in the central nervous system: Ionotropic and metabotropic receptors in concert. Neuron 2018, 98, 1080–1098. [Google Scholar] [CrossRef]
- Sattler, R.; Tymianski, M. Molecular mechanisms of glutamate receptor-mediated excitotoxic neuronal cell death. Mol. Neurobiol. 2001, 24, 107–129. [Google Scholar] [CrossRef] [PubMed]
- Skeberdis, V.A.; Lan, J.-y.; Opitz, T.; Zheng, X.; Bennett, M.V.; Zukin, R.S. mGluR1-mediated potentiation of NMDA receptors involves a rise in intracellular calcium and activation of protein kinase C. Neuropharmacology 2001, 40, 856–865. [Google Scholar] [CrossRef]
- Plitman, E.; Nakajima, S.; de la Fuente-Sandoval, C.; Gerretsen, P.; Chakravarty, M.M.; Kobylianskii, J.; Chung, J.K.; Caravaggio, F.; Iwata, Y.; Remington, G. Glutamate-mediated excitotoxicity in schizophrenia: A review. Eur. Neuropsychopharmacol. 2014, 24, 1591–1605. [Google Scholar] [CrossRef]
- Dong, X.-x.; Wang, Y.; Qin, Z.-h. Molecular mechanisms of excitotoxicity and their relevance to pathogenesis of neurodegenerative diseases. Acta Pharmacol. Sin. 2009, 30, 379–387. [Google Scholar] [CrossRef]
- Pitt, D.; Werner, P.; Raine, C.S. Glutamate excitotoxicity in a model of multiple sclerosis. Nat. Med. 2000, 6, 67–70. [Google Scholar] [CrossRef]
- Olney, J.; Collins, R.; Sloviter, R. Excitotoxic mechanisms of epileptic brain damage. Adv. Neurol. 1986, 44, 857–877. [Google Scholar]
- Petrenko, A.B.; Shimoji, K. A possible role for glutamate receptor-mediated excitotoxicity in chronic pain. J. Anesth. 2001, 15, 39–48. [Google Scholar] [CrossRef]
- Longoni, M.; Ferrarese, C. Inflammation and excitotoxicity: Role in migraine pathogenesis. Neurol. Sci. 2006, 27, s107–s110. [Google Scholar] [CrossRef]
- Storer, R.; Goadsby, P. Trigeminovascular nociceptive transmission involves N-methyl-D-aspartate and non-N-methyl-D-aspartate glutamate receptors. Neuroscience 1999, 90, 1371–1376. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Montoya, J.; Avendaño, C.; Negredo, P. The Glutamatergic System in Primary Somatosensory Neurons and Its Involvement in Sensory Input-Dependent Plasticity. Int. J. Mol. Sci. 2018, 19, 69. [Google Scholar] [CrossRef] [PubMed]
- Tallaksen-Greene, S.J.; Young, A.B.; Penney, J.B.; Beitz, A.J. Excitatory amino acid binding sites in the trigeminal principal sensory and spinal trigeminal nuclei of the rat. Neurosci. Lett. 1992, 141, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Quartu, M.; Serra, M.P.; Ambu, R.; Lai, M.L.; Del Fiacco, M. AMPA-type glutamate receptor subunits 2/3 in the human trigeminal sensory ganglion and subnucleus caudalis from prenatal ages to adulthood. Mech. Ageing Dev. 2002, 123, 463–471. [Google Scholar] [CrossRef]
- Yang, Z.; Wang, Y.; Luo, W.; Hua, X.; Wamalwa, P.; Wang, J.; Zhao, Z.; Lu, Y.; Liao, Z.; Lai, W. Trigeminal expression of N-methyl-D-aspartate receptor subunit 1 and behavior responses to experimental tooth movement in rats. Angle Orthod. 2009, 79, 951–957. [Google Scholar] [CrossRef]
- Sahara, Y.; Noro, N.; Iida, Y.; Soma, K.; Nakamura, Y. Glutamate receptor subunits GluR5 and KA-2 are coexpressed in rat trigeminal ganglion neurons. J. Neurosci. 1997, 17, 6611–6620. [Google Scholar] [CrossRef]
- Zhou, S.; Bonasera, L.; Carlton, S.M. Peripheral administration of NMDA, AMPA or KA results in pain behaviors in rats. Neuroreport 1996, 7, 895–900. [Google Scholar] [CrossRef]
- Lawand, N.B.; Willis, W.D.; Westlund, K.N. Excitatory amino acid receptor involvement in peripheral nociceptive transmission in rats. Eur. J. Pharmacol. 1997, 324, 169–177. [Google Scholar] [CrossRef]
- Raigorodsky, G.; Urca, G. Spinal antinociceptive effects of excitatory amino acid antagonists: Quisqualate modulates the action of N-methyl-D-aspartate. Eur. J. Pharmacol. 1990, 182, 37–47. [Google Scholar] [CrossRef]
- Crivellaro, G.; Tottene, A.; Vitale, M.; Melone, M.; Casari, G.; Conti, F.; Santello, M.; Pietrobon, D. Specific activation of GluN1-N2B NMDA receptors underlies facilitation of cortical spreading depression in a genetic mouse model of migraine with reduced astrocytic glutamate clearance. Neurobiol. Dis. 2021, 156, 105419. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.; Tozzi, A.; Rainero, I.; Cupini, L.M.; Calabresi, P.; Ayata, C.; Sarchielli, P. Cortical spreading depression as a target for anti-migraine agents. J. Headache Pain 2013, 14, 62. [Google Scholar] [PubMed]
- De Simone, R.; Ranieri, A.; Montella, S.; Bonavita, V. Cortical spreading depression and central pain networks in trigeminal nuclei modulation: Time for an integrated migraine pathogenesis perspective. Neurol. Sci. 2013, 34, 51–55. [Google Scholar] [CrossRef][Green Version]
- Footitt, D.R.; Newberry, N.R. Cortical spreading depression induces an LTP-like effect in rat neocortex in vitro. Brain Res. 1998, 781, 339–342. [Google Scholar] [CrossRef]
- Richter, F.; Lehmenkühler, A. Cortical spreading depression (CSD). Der Schmerz 2008, 22, 544–550. [Google Scholar] [CrossRef] [PubMed]
- Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia 2018, 38, 1–211. [CrossRef] [PubMed]
- Cutrer, F.M.; Huerter, K. Migraine aura. Neurologist 2007, 13, 118–125. [Google Scholar] [CrossRef]
- Dodick, D.; Silberstein, S. Central sensitization theory of migraine: Clinical implications. Headache J. Head Face Pain 2006, 46, S182–S191. [Google Scholar] [CrossRef]
- Nihi, M.A.; Santos, P.S.F.; Almeida, D.B. Central sensitization in episodic and chronic migraine. Headache Med. 2020, 11, 85–89. [Google Scholar] [CrossRef]
- Nijs, J.; Meeus, M.; Versijpt, J.; Moens, M.; Bos, I.; Knaepen, K.; Meeusen, R. Brain-derived neurotrophic factor as a driving force behind neuroplasticity in neuropathic and central sensitization pain: A new therapeutic target? Expert Opin. Ther. Targets 2015, 19, 565–576. [Google Scholar] [CrossRef]
- Park, C.G.; Chu, M.K. Interictal plasma glutamate levels are elevated in individuals with episodic and chronic migraine. Sci. Rep. 2022, 12, 6921. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Suzuki, S.; Shiina, T.; Kobayashi, S.; Hirata, K. Central sensitization in migraine: A narrative review. J. Pain Res. 2022, 15, 2673–2682. [Google Scholar] [CrossRef] [PubMed]
- Delussi, M.; Vecchio, E.; Libro, G.; Quitadamo, S.; De Tommaso, M. Failure of preventive treatments in migraine: An observational retrospective study in a tertiary headache center. BMC Neurol. 2020, 20, 256. [Google Scholar] [CrossRef]
- Barbanti, P.; Aurilia, C.; Cevoli, S.; Egeo, G.; Fofi, L.; Messina, R.; Salerno, A.; Torelli, P.; Albanese, M.; Carnevale, A. Long-term (48 weeks) effectiveness, safety, and tolerability of erenumab in the prevention of high-frequency episodic and chronic migraine in a real world: Results of the EARLY 2 study. Headache J. Head Face Pain 2021, 61, 1351–1363. [Google Scholar] [CrossRef] [PubMed]
- Bono, F.; Salvino, D.; Mazza, M.; Curcio, M.; Trimboli, M.; Vescio, B.; Quattrone, A. The influence of ictal cutaneous allodynia on the response to occipital transcutaneous electrical stimulation in chronic migraine and chronic tension-type headache: A randomized, sham-controlled study. Cephalalgia 2015, 35, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Campos-Bedolla, P.; Walter, F.R.; Veszelka, S.; Deli, M.A. Role of the blood–brain barrier in the nutrition of the central nervous system. Arch. Med. Res. 2014, 45, 610–638. [Google Scholar]
- Vazana, U.; Veksler, R.; Pell, G.S.; Prager, O.; Fassler, M.; Chassidim, Y.; Roth, Y.; Shahar, H.; Zangen, A.; Raccah, R.; et al. Glutamate-Mediated Blood–Brain Barrier Opening: Implications for Neuroprotection and Drug Delivery. J. Neurosci. 2016, 36, 7727–7739. [Google Scholar] [CrossRef]
- Toklu, H.Z.; Tümer, N. Oxidative Stress, Brain Edema, Blood–Brain Barrier Permeability, and Autonomic Dysfunction from Traumatic Brain Injury; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2015. [Google Scholar]
- Galea, I. The blood–brain barrier in systemic infection and inflammation. Cell. Mol. Immunol. 2021, 18, 2489–2501. [Google Scholar] [CrossRef]
- Jones, K.H.; Dechkovskaia, A.M.; Herrick, E.A.; Abdel-Rahman, A.A.; Khan, W.A.; Abou-Donia, M.B. Subchronic effects following a single sarin exposure on blood-brain and blood-testes barrier permeability, acetylcholinesterase, and acetylcholine receptors in the central nervous system of rat: A dose-response study. J. Toxicol. Environ. Health Part A 2000, 61, 695–707. [Google Scholar] [CrossRef]
- Xu, G.; Li, Y.; Ma, C.; Wang, C.; Sun, Z.; Shen, Y.; Liu, L.; Li, S.; Zhang, X.; Cong, B. Restraint stress induced hyperpermeability and damage of the blood-brain barrier in the amygdala of adult rats. Front. Mol. Neurosci. 2019, 12, 32. [Google Scholar] [CrossRef]
- Petty, M.A.; Lo, E.H. Junctional complexes of the blood–brain barrier: Permeability changes in neuroinflammation. Prog. Neurobiol. 2002, 68, 311–323. [Google Scholar] [CrossRef] [PubMed]
- Lochhead, J.J.; McCaffrey, G.; Quigley, C.E.; Finch, J.; DeMarco, K.M.; Nametz, N.; Davis, T.P. Oxidative stress increases blood–brain barrier permeability and induces alterations in occludin during hypoxia–reoxygenation. J. Cereb. Blood Flow Metab. 2010, 30, 1625–1636. [Google Scholar] [CrossRef]
- Rogoz, K.; Andersen, H.H.; Kullander, K.; Lagerström, M.C. Glutamate, substance P, and calcitonin gene-related peptide cooperate in inflammation-induced heat hyperalgesia. Mol. Pharmacol. 2014, 85, 322–334. [Google Scholar] [CrossRef] [PubMed]
- Ji, R.-R.; Xu, Z.-Z.; Gao, Y.-J. Emerging targets in neuroinflammation-driven chronic pain. Nat. Rev. Drug Discov. 2014, 13, 533–548. [Google Scholar] [CrossRef]
- Rueda, C.B.; Llorente-Folch, I.; Traba, J.; Amigo, I.; Gonzalez-Sanchez, P.; Contreras, L.; Juaristi, I.; Martinez-Valero, P.; Pardo, B.; Del Arco, A. Glutamate excitotoxicity and Ca2+-regulation of respiration: Role of the Ca2+ activated mitochondrial transporters (CaMCs). Biochim. Biophys. Acta (BBA)-Bioenerg. 2016, 1857, 1158–1166. [Google Scholar] [CrossRef] [PubMed]
- Lafon-Cazal, M.; Pietri, S.; Culcasi, M.; Bockaert, J. NMDA-dependent superoxide production and neurotoxicity. Nature 1993, 364, 535–537. [Google Scholar] [CrossRef]
- Schieber, M.; Chandel, N.S. ROS function in redox signaling and oxidative stress. Curr. Biol. 2014, 24, R453–R462. [Google Scholar] [CrossRef]
- Zorov, D.B.; Juhaszova, M.; Sollott, S.J. Mitochondrial ROS-induced ROS release: An update and review. Biochim. Biophys. Acta (BBA)-Bioenerg. 2006, 1757, 509–517. [Google Scholar] [CrossRef]
- Garthwaite, J.; Charles, S.L.; Chess-Williams, R. Endothelium-derived relaxing factor release on activation of NMDA receptors suggests role as intercellular messenger in the brain. Nature 1988, 336, 385–388. [Google Scholar] [CrossRef]
- Christopherson, K.S.; Hillier, B.J.; Lim, W.A.; Bredt, D.S. PSD-95 Assembles a Ternary Complex with theN-Methyl-D-aspartic Acid Receptor and a Bivalent Neuronal NO Synthase PDZ Domain. J. Biol. Chem. 1999, 274, 27467–27473. [Google Scholar] [CrossRef]
- LeMaistre, J.L.; Sanders, S.A.; Stobart, M.J.; Lu, L.; Knox, J.D.; Anderson, H.D.; Anderson, C.M. Coactivation of NMDA receptors by glutamate and-serine induces dilation of isolated middle cerebral arteries. J. Cereb. Blood Flow Metab. 2012, 32, 537. [Google Scholar] [CrossRef] [PubMed]
- Goadsby, P.J.; Holland, P.R.; Martins-Oliveira, M.; Hoffmann, J.; Schankin, C.; Akerman, S. Pathophysiology of migraine: A disorder of sensory processing. Physiol. Rev. 2017, 97, 553–622. [Google Scholar] [CrossRef] [PubMed]
- Shevel, E. The extracranial vascular theory of migraine—A great story confirmed by the facts. Headache J. Head Face Pain 2011, 51, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.D.; Cizkova, D. The role of nitric oxide in nociception. Curr. Rev. Pain 2000, 4, 459–466. [Google Scholar] [CrossRef]
- Thiel, V.E.; Audus, K.L. Nitric oxide and blood–brain barrier integrity. Antioxid. Redox Signal. 2001, 3, 273–278. [Google Scholar] [CrossRef]
- Mayhan, W.G. Nitric oxide donor-induced increase in permeability of the blood–brain barrier. Brain Res. 2000, 866, 101–108. [Google Scholar] [CrossRef]
- Ferrari, M.D.; Odink, J.; Bos, K.; Malessy, M.; Bruyn, G. Neuroexcitatory plasma amino acids are elevated in migraine. Neurology 1990, 40, 1582. [Google Scholar] [CrossRef]
- Alam, Z.; Coombes, N.; Waring, R.H.; Williams, A.C.; Steventon, G.B. Plasma levels of neuroexcitatory amino acids in patients with migraine or tension headache. J. Neurol. Sci. 1998, 156, 102–106. [Google Scholar] [CrossRef]
- Martínez, F.; Castillo, J.; Rodríguez, J.R.; Leira, R.; Noya, M. Neuroexcitatory amino acid levels in plasma and cerebrospinal fluid during migraine attacks. Cephalalgia 1993, 13, 89–93. [Google Scholar] [CrossRef]
- Peres, M.; Zukerman, E.; Soares, C.S.; Alonso, E.; Santos, B.; Faulhaber, M. Cerebrospinal fluid glutamate levels in chronic migraine. Cephalalgia 2004, 24, 735–739. [Google Scholar] [CrossRef]
- D’andrea, G.; Cananzi, A.; Joseph, R.; Morra, M.; Zamberlan, F.; Milone, F.F.; Grunfeld, S.; Welch, K. Platelet glycine, glutamate and aspartate in primary headache. Cephalalgia 1991, 11, 197–200. [Google Scholar] [CrossRef]
- Cananzi, A.; D’andrea, G.; Perini, F.; Zamberlan, F.; Welch, K. Platelet and plasma levels of glutamate and glutamine in migraine with and without aura. Cephalalgia 1995, 15, 132–135. [Google Scholar] [CrossRef] [PubMed]
- Vaccaro, M.; Riva, C.; Tremolizzo, L.; Longoni, M.; Aliprandi, A.; Agostoni, E.; Rigamonti, A.; Leone, M.; Bussone, G.; Ferrarese, C. Platelet glutamate uptake and release in migraine with and without aura. Cephalalgia 2007, 27, 35–40. [Google Scholar] [CrossRef]
- Vieira, D.S.d.; Naffah-Mazzacoratti, M.d.G.; Zukerman, E.; Soares, C.A.S.; Cavalheiro, E.A.; Peres, M.F.P. Glutamate levels in cerebrospinal fluid and triptans overuse in chronic migraine. Headache J. Head Face Pain 2007, 47, 842–847. [Google Scholar] [CrossRef]
- Zukerman, E.; Minatti-Hannuch, S.; Mazzacoratti, M.; dos Reis Filho, J.; Cavalheiro, E. Cerebrospinal fluid neurotransmitter amino acids in migraine. Cephalalgia 1993, 13, 92. [Google Scholar]
- Ferrari, A.; Spaccalopelo, L.; Pinetti, D.; Tacchi, R.; Bertolini, A. Effective prophylactic treatments of migraine lower plasma glutamate levels. Cephalalgia 2009, 29, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Peek, A.L.; Rebbeck, T.; Puts, N.A.; Watson, J.; Aguila, M.-E.R.; Leaver, A.M. Brain GABA and glutamate levels across pain conditions: A systematic literature review and meta-analysis of 1H-MRS studies using the MRS-Q quality assessment tool. Neuroimage 2020, 210, 116532. [Google Scholar] [CrossRef]
- Bathel, A.; Schweizer, L.; Stude, P.; Glaubitz, B.; Wulms, N.; Delice, S.; Schmidt-Wilcke, T. Increased thalamic glutamate/glutamine levels in migraineurs. J. Headache Pain 2018, 19, 55. [Google Scholar] [CrossRef]
- Tripathi, G.M.; Kalita, J.; Misra, U.K. Role of glutamate and its receptors in migraine with reference to amitriptyline and transcranial magnetic stimulation therapy. Brain Res. 2018, 1696, 31–37. [Google Scholar] [CrossRef]
- Sarchielli, P.; Mancini, M.L.; Floridi, A.; Coppola, F.; Rossi, C.; Nardi, K.; Acciarresi, M.; Pini, L.A.; Calabresi, P. Increased Levels of Neurotrophins Are Not Specific for Chronic Migraine: Evidence From Primary Fibromyalgia Syndrome. J. Pain 2007, 8, 737–745. [Google Scholar] [CrossRef]
- Zielman, R.; Wijnen, J.P.; Webb, A.; Onderwater, G.L.J.; Ronen, I.; Ferrari, M.D.; Kan, H.E.; Terwindt, G.M.; Kruit, M.C. Cortical glutamate in migraine. Brain 2017, 140, 1859–1871. [Google Scholar] [CrossRef]
- Gonzalez de la Aleja, J.; Ramos, A.; Mato-Abad, V.; Martínez-Salio, A.; Hernández-Tamames, J.A.; Molina, J.A.; Hernández-Gallego, J.; Álvarez-Linera, J. Higher glutamate to glutamine ratios in occipital regions in women with migraine during the interictal state. Headache J. Head Face Pain 2013, 53, 365–375. [Google Scholar] [CrossRef] [PubMed]
- Fayed, N.; Andrés, E.; Viguera, L.; Modrego, P.J.; Garcia-Campayo, J. Higher glutamate+ glutamine and reduction of N-acetylaspartate in posterior cingulate according to age range in patients with cognitive impairment and/or pain. Acad. Radiol. 2014, 21, 1211–1217. [Google Scholar] [CrossRef] [PubMed]
- Arzani, M.; Jahromi, S.R.; Ghorbani, Z.; Vahabizad, F.; Martelletti, P.; Ghaemi, A.; Sacco, S.; Togha, M.; School of Advanced Studies of the European Headache Federation (EHF-SAS). Gut-brain axis and migraine headache: A comprehensive review. J. Headache Pain 2020, 21, 15. [Google Scholar] [CrossRef] [PubMed]
- Czyż, K.; Bodkowski, R.; Herbinger, G.; Librowski, T. Omega-3 fatty acids and their role in central nervous system-a review. Curr. Med. Chem. 2016, 23, 816–831. [Google Scholar]
- McNamara, R.K.; Carlson, S.E. Role of omega-3 fatty acids in brain development and function: Potential implications for the pathogenesis and prevention of psychopathology. Prostaglandins Leukot. Essent. Fat. Acids 2006, 75, 329–349. [Google Scholar]
- Galán-Arriero, I.; Serrano-Muñoz, D.; Gómez-Soriano, J.; Goicoechea, C.; Taylor, J.; Velasco, A.; Ávila-Martín, G. The role of Omega-3 and Omega-9 fatty acids for the treatment of neuropathic pain after neurotrauma. Biochim. Biophys. Acta (BBA)-Biomembr. 2017, 1859, 1629–1635. [Google Scholar] [CrossRef]
- Silva, R.V.; Oliveira, J.T.; Santos, B.L.; Dias, F.C.; Martinez, A.M.; Lima, C.K.; Miranda, A.L. Long-chain omega-3 fatty acids supplementation accelerates nerve regeneration and prevents neuropathic pain behavior in mice. Front. Pharmacol. 2017, 8, 723. [Google Scholar] [CrossRef]
- Figueroa, J.D.; Cordero, K.; Serrano-Illan, M.; Almeyda, A.; Baldeosingh, K.; Almaguel, F.G.; De Leon, M. Metabolomics uncovers dietary omega-3 fatty acid-derived metabolites implicated in anti-nociceptive responses after experimental spinal cord injury. Neuroscience 2013, 255, 1–18. [Google Scholar] [CrossRef]
- Voskuyl, R.A.; Vreugdenhil, M.; Kang, J.X.; Leaf, A. Anticonvulsant effect of polyunsaturated fatty acids in rats, using the cortical stimulation model. Eur. J. Pharmacol. 1998, 341, 145–152. [Google Scholar] [CrossRef]
- Heurteaux, C.; Laigle, C.; Blondeau, N.; Jarretou, G.; Lazdunski, M. Alpha-linolenic acid and riluzole treatment confer cerebral protection and improve survival after focal brain ischemia. Neuroscience 2006, 137, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Blondeau, N. The nutraceutical potential of omega-3 alpha-linolenic acid in reducing the consequences of stroke. Biochimie 2016, 120, 49–55. [Google Scholar] [CrossRef]
- King, V.R.; Huang, W.L.; Dyall, S.C.; Curran, O.E.; Priestley, J.V.; Michael-Titus, A.T. Omega-3 fatty acids improve recovery, whereas omega-6 fatty acids worsen outcome, after spinal cord injury in the adult rat. J. Neurosci. 2006, 26, 4672–4680. [Google Scholar] [CrossRef]
- Relton, J.K.; Strijbos, P.J.L.M.; Cooper, A.L.; Rothwell, N.J. Dietary N-3 fatty acids inhibit ischaemic and excitotoxic brain damage in the rat. Brain Res. Bull. 1993, 32, 223–226. [Google Scholar] [CrossRef] [PubMed]
- Barbour, B.; Szatkowski, M.; Ingledew, N.; Attwell, D. Arachidonic acid induces a prolonged inhibition of glutamate uptake into glial cells. Nature 1989, 342, 918–920. [Google Scholar] [CrossRef]
- Lynch, M.A.; Voss, K.L. Arachidonic Acid Increases Inositol Phospholipid Metabolism and Glutamate Release in Synaptosomes Prepared from Hippocampal Tissue. J. Neurochem. 1990, 55, 215–221. [Google Scholar] [CrossRef]
- Gürgen, S.G.; Sayın, O.; Çetïn, F.; Sarsmaz, H.Y.; Yazıcı, G.N.; Umur, N.; Yücel, A.T. The Effect of Monosodium Glutamate on Neuronal Signaling Molecules in the Hippocampus and the Neuroprotective Effects of Omega-3 Fatty Acids. ACS Chem. Neurosci. 2021, 12, 3028–3037. [Google Scholar] [CrossRef]
- Hennebelle, M.; Champeil-Potokar, G.; Lavialle, M.; Vancassel, S.; Denis, I. Omega-3 polyunsaturated fatty acids and chronic stress-induced modulations of glutamatergic neurotransmission in the hippocampus. Nutr. Rev. 2014, 72, 99–112. [Google Scholar] [CrossRef]
- Latour, A.; Grintal, B.; Champeil-Potokar, G.; Hennebelle, M.; Lavialle, M.; Dutar, P.; Potier, B.; Billard, J.M.; Vancassel, S.; Denis, I. Omega-3 fatty acids deficiency aggravates glutamatergic synapse and astroglial aging in the rat hippocampal CA 1. Aging Cell 2013, 12, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi, O.; Maghsoudi, Z.; Khorvash, F.; Ghiasvand, R.; Askari, G. The relationship between different fatty acids intake and frequency of migraine attacks. Iran. J. Nurs. Midwifery Res. 2015, 20, 334. [Google Scholar]
- Evans, E.W.; Lipton, R.B.; Peterlin, B.L.; Raynor, H.A.; Thomas, J.G.; O’Leary, K.C.; Pavlovic, J.; Wing, R.R.; Bond, D.S. Dietary intake patterns and diet quality in a nationally representative sample of women with and without severe headache or migraine. Headache J. Head Face Pain 2015, 55, 550–561. [Google Scholar] [CrossRef] [PubMed]
- Tajmirriahi, M.; Sohelipour, M.; Basiri, K.; Shaygannejad, V.; Ghorbani, A.; Saadatnia, M. The effects of sodium valproate with fish oil supplementation or alone in migraine prevention: A randomized single-blind clinical trial. Iran. J. Neurol. 2012, 11, 21. [Google Scholar]
- Harel, Z.; Gascon, G.; Riggs, S.; Vaz, R.; Brown, W.; Exil, G. Supplementation with omega-3 polyunsaturated fatty acids in the management of recurrent migraines in adolescents. J. Adolesc. Health 2002, 31, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Abdolahi, M.; Jafarieh, A.; Sarraf, P.; Sedighiyan, M.; Yousefi, A.; Tafakhori, A.; Abdollahi, H.; Salehinia, F.; Djalali, M. The neuromodulatory effects of ω-3 fatty acids and nano-curcumin on the COX-2/iNOS network in migraines: A clinical trial study from gene expression to clinical symptoms. Endocr. Metab. Immune Disord.-Drug Targets (Former. Curr. Drug Targets-Immune Endocr. Metab. Disord.) 2019, 19, 874–884. [Google Scholar] [CrossRef] [PubMed]
- Maghsoumi-Norouzabad, L.; Mansoori, A.; Abed, R.; Shishehbor, F. Effects of omega-3 fatty acids on the frequency, severity, and duration of migraine attacks: A systematic review and meta-analysis of randomized controlled trials. Nutr. Neurosci. 2018, 21, 614–623. [Google Scholar] [CrossRef]
- Jahnen-Dechent, W.; Ketteler, M. Magnesium basics. Clin. Kidney J. 2012, 5, i3–i14. [Google Scholar] [CrossRef]
- Lambuk, L.; Jafri, A.J.A.; Arfuzir, N.N.N.; Iezhitsa, I.; Agarwal, R.; Rozali, K.N.B.; Agarwal, P.; Bakar, N.S.; Kutty, M.K.; Yusof, A.P.M. Neuroprotective effect of magnesium acetyltaurate against NMDA-induced excitotoxicity in rat retina. Neurotox. Res. 2017, 31, 31–45. [Google Scholar] [CrossRef]
- Veronese, N.; Zurlo, A.; Solmi, M.; Luchini, C.; Trevisan, C.; Bano, G.; Manzato, E.; Sergi, G.; Rylander, R. Magnesium status in Alzheimer’s disease: A systematic review. Am. J. Alzheimer’s Dis. Other Dement. ® 2016, 31, 208–213. [Google Scholar] [CrossRef]
- Stippler, M.; Fischer, M.R.; Puccio, A.M.; Wisniewski, S.R.; Carson-Walter, E.B.; Dixon, C.E.; Walter, K.A. Serum and cerebrospinal fluid magnesium in severe traumatic brain injury outcome. J. Neurotrauma 2007, 24, 1347–1354. [Google Scholar] [CrossRef]
- Altura, B.T.; Memon, Z.I.; Zhang, A.; Cheng, T.P.-O.; Silverman, R.; Cracco, R.Q.; Altura, B.M. Low levels of serum ionized magnesium are found in patients early after stroke which result in rapid elevation in cytosolic free calcium and spasm in cerebral vascular muscle cells. Neurosci. Lett. 1997, 230, 37–40. [Google Scholar] [CrossRef]
- Prasad, D.K.V.; Shaheen, U.; Satyanarayana, U.; Surya Prabha, T.; Jyothy, A.; Munshi, A. Association of Serum Trace Elements and Minerals with Genetic Generalized Epilepsy and Idiopathic Intractable Epilepsy. Neurochem. Res. 2014, 39, 2370–2376. [Google Scholar] [CrossRef] [PubMed]
- Oyanagi, K.; Hashimoto, T. Magnesium in Parkinson’s disease: An update in clinical and basic aspects. In Magnesium in the Central Nervous System; University of Adelaide Press: Adelaide, Australia, 2011. [Google Scholar]
- Botturi, A.; Ciappolino, V.; Delvecchio, G.; Boscutti, A.; Viscardi, B.; Brambilla, P. The role and the effect of magnesium in mental disorders: A systematic review. Nutrients 2020, 12, 1661. [Google Scholar] [CrossRef]
- Sarchielli, P.; Coata, G.; Firenze, C.; Morucci, P.; Abbritti, G.; Gallai, V. Serum and salivary magnesium levels in migraine and tension-type headache. Results in a group of adult patients. Cephalalgia 1992, 12, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Ramadan, N.; Halvorson, H.; Vande-Linde, A.; Levine, S.R.; Helpern, J.; Welch, K. Low brain magnesium in migraine. Headache J. Head Face Pain 1989, 29, 590–593. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, J.; Storer, R.J.; Park, J.W.; Goadsby, P.J. N-Methyl-d-aspartate receptor open-channel blockers memantine and magnesium modulate nociceptive trigeminovascular neurotransmission in rats. Eur. J. Neurosci. 2019, 50, 2847–2859. [Google Scholar] [CrossRef]
- Manet, S.; Gressens, P.; Gadisseux, J.F.; Evrard, P. Prevention by magnesium of exototoxic neuronal death in the developing brain: An animal model for clinical intervention studies. Dev. Med. Child Neurol. 1995, 37, 473–484. [Google Scholar] [CrossRef]
- Lin, J.-Y.; Chung, S.-Y.; Lin, M.-C.; Cheng, F.-C. Effects of magnesium sulfate on energy metabolites and glutamate in the cortex during focal cerebral ischemia and reperfusion in the gerbil monitored by a dual-probe microdialysis technique. Life Sci. 2002, 71, 803–811. [Google Scholar] [CrossRef]
- van der Hel, W.S.; van den Bergh, W.M.; Nicolay, K.; Tulleken, K.A.; Dijkhuizen, R.M. Suppression of cortical spreading depressions after magnesium treatment in the rat. Neuroreport 1998, 9, 2179–2182. [Google Scholar] [CrossRef]
- Mody, I.; Lambert, J.; Heinemann, U. Low extracellular magnesium induces epileptiform activity and spreading depression in rat hippocampal slices. J. Neurophysiol. 1987, 57, 869–888. [Google Scholar] [CrossRef]
- Van Harreveld, A.; Fifková, E. Mechanisms involved in spreading depression. J. Neurobiol. 1973, 4, 375–387. [Google Scholar] [CrossRef]
- Chiu, H.-Y.; Yeh, T.-H.; Yin-Cheng, H.; Pin-Yuan, C. Effects of intravenous and oral magnesium on reducing migraine: A meta-analysis of randomized controlled trials. Pain Physician 2016, 19, E97. [Google Scholar] [PubMed]
- Choi, H.; Parmar, N. The use of intravenous magnesium sulphate for acute migraine: Meta-analysis of randomized controlled trials. Eur. J. Emerg. Med. 2014, 21, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.S.; Hewison, M. Update in Vitamin D. J. Clin. Endocrinol. Metab. 2010, 95, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Garcion, E.; Wion-Barbot, N.; Montero-Menei, C.N.; Berger, F.; Wion, D. New clues about vitamin D functions in the nervous system. Trends Endocrinol. Metab. 2002, 13, 100–105. [Google Scholar] [CrossRef]
- Mpandzou, G.; Haddou, E.A.B.; Regragui, W.; Benomar, A.; Yahyaoui, M. Vitamin D deficiency and its role in neurological conditions: A review. Rev. Neurol. 2016, 172, 109–122. [Google Scholar] [CrossRef]
- Eyles, D.W.; Smith, S.; Kinobe, R.; Hewison, M.; McGrath, J.J. Distribution of the vitamin D receptor and 1α-hydroxylase in human brain. J. Chem. Neuroanat. 2005, 29, 21–30. [Google Scholar] [CrossRef]
- Taniura, H.; Ito, M.; Sanada, N.; Kuramoto, N.; Ohno, Y.; Nakamichi, N.; Yoneda, Y. Chronic vitamin D3 treatment protects against neurotoxicity by glutamate in association with upregulation of vitamin D receptor mRNA expression in cultured rat cortical neurons. J. Neurosci. Res. 2006, 83, 1179–1189. [Google Scholar] [CrossRef]
- Groves, N.J.; Kesby, J.P.; Eyles, D.W.; McGrath, J.J.; Mackay-Sim, A.; Burne, T.H.J. Adult vitamin D deficiency leads to behavioural and brain neurochemical alterations in C57BL/6J and BALB/c mice. Behav. Brain Res. 2013, 241, 120–131. [Google Scholar] [CrossRef]
- Brewer, L.D.; Thibault, V.; Chen, K.-C.; Langub, M.C.; Landfield, P.W.; Porter, N.M. Vitamin D hormone confers neuroprotection in parallel with downregulation of L-type calcium channel expression in hippocampal neurons. J. Neurosci. 2001, 21, 98–108. [Google Scholar] [CrossRef]
- Zandifar, A.; Masjedi, S.s.; Banihashemi, M.; Asgari, F.; Manouchehri, N.; Ebrahimi, H.; Haghdoost, F.; Saadatnia, M. Vitamin D Status in Migraine Patients: A Case-Control Study. BioMed Res. Int. 2014, 2014, 514782. [Google Scholar] [CrossRef]
- Kjærgaard, M.; Eggen, A.E.; Mathiesen, E.B.; Jorde, R. Association Between Headache and Serum 25-Hydroxyvitamin D; the Tromsø Study: Tromsø 6. Headache J. Head Face Pain 2012, 52, 1499–1505. [Google Scholar] [CrossRef]
- Hussein, M.; Fathy, W.; Abd Elkareem, R.M. The potential role of serum vitamin D level in migraine headache: A case–control study. J. Pain Res. 2019, 12, 2529–2536. [Google Scholar] [CrossRef] [PubMed]
- Togha, M.; Razeghi Jahromi, S.; Ghorbani, Z.; Martami, F.; Seifishahpar, M. Serum Vitamin D Status in a Group of Migraine Patients Compared With Healthy Controls: A Case–Control Study. Headache J. Head Face Pain 2018, 58, 1530–1540. [Google Scholar] [CrossRef] [PubMed]
- Liampas, I.; Siokas, V.; Brotis, A.; Dardiotis, E. Vitamin D serum levels in patients with migraine: A meta-analysis. Rev. Neurol. 2020, 176, 560–570. [Google Scholar] [CrossRef] [PubMed]
- Song, T.-J.; Chu, M.-K.; Sohn, J.-H.; Ahn, H.-Y.; Lee, S.H.; Cho, S.-J. Effect of Vitamin D Deficiency on the Frequency of Headaches in Migraine. J. Clin. Neurol. 2018, 14, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-F.; Xu, Z.-Q.; Zhou, H.-J.; Liu, Y.-Z.; Jiang, X.-J. The Efficacy of Vitamin D supplementation for migraine: A meta-analysis of randomized controlled studies. Clin. Neuropharmacol. 2021, 44, 5–8. [Google Scholar] [CrossRef]
- May, J.M. Vitamin C Transport and Its Role in the Central Nervous System. In Water Soluble Vitamins: Clinical Research and Future Application; Stanger, O., Ed.; Springer: Dordrecht, The Netherlands, 2012; pp. 85–103. [Google Scholar] [CrossRef]
- Majewska, M.D.; Bell, J.A. Ascorbic acid protects neurons from injury induced by glutamate and NMDA. Neuroreport 1990, 1, 194–196. [Google Scholar] [CrossRef]
- Lane, D.J.; Lawen, A. The glutamate aspartate transporter (GLAST) mediates L-glutamate-stimulated ascorbate-release via swelling-activated anion channels in cultured neonatal rodent astrocytes. Cell Biochem. Biophys. 2013, 65, 107–119. [Google Scholar] [CrossRef]
- Hashem, H.E.; El-Din Safwat, M.D.; Algaidi, S. The effect of monosodium glutamate on the cerebellar cortex of male albino rats and the protective role of vitamin C (histological and immunohistochemical study). J. Mol. Histol. 2012, 43, 179–186. [Google Scholar] [CrossRef]
- Nelson, M.T.; Joksovic, P.M.; Su, P.; Kang, H.-W.; Van Deusen, A.; Baumgart, J.P.; David, L.S.; Snutch, T.P.; Barrett, P.Q.; Lee, J.-H. Molecular mechanisms of subtype-specific inhibition of neuronal T-type calcium channels by ascorbate. J. Neurosci. 2007, 27, 12577–12583. [Google Scholar] [CrossRef]
- Visser, E.J.; Drummond, P.D.; Lee-Visser, J.L.A. Reduction in Migraine and Headache Frequency and Intensity With Combined Antioxidant Prophylaxis (N-acetylcysteine, Vitamin E, and Vitamin C): A Randomized Sham-Controlled Pilot Study. Pain Pract. 2020, 20, 737–747. [Google Scholar] [CrossRef] [PubMed]
- Brigelius-Flohé, R.; Traber, M.G. Vitamin E: Function and metabolism. FASEB J. 1999, 13, 1145–1155. [Google Scholar] [CrossRef] [PubMed]
- Friedman, J. Why is the nervous system vulnerable to oxidative stress? In Oxidative Stress in Applied Basic Research and Clinical Practice; Humana Press: Totowa, NJ, USA, 2011; pp. 19–27. [Google Scholar]
- Kim, H.K.; Kim, J.H.; Gao, X.; Zhou, J.-L.; Lee, I.; Chung, K.; Chung, J.M. Analgesic effect of vitamin E is mediated by reducing central sensitization in neuropathic pain. Pain 2006, 122, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Ambrogini, P.; Minelli, A.; Galati, C.; Betti, M.; Lattanzi, D.; Ciffolilli, S.; Piroddi, M.; Galli, F.; Cuppini, R. Post-Seizure α-Tocopherol Treatment Decreases Neuroinflammation and Neuronal Degeneration Induced by Status Epilepticus in Rat Hippocampus. Mol. Neurobiol. 2014, 50, 246–256. [Google Scholar] [CrossRef]
- Betti, M.; Minelli, A.; Ambrogini, P.; Ciuffoli, S.; Viola, V.; Galli, F.; Canonico, B.; Lattanzi, D.; Colombo, E.; Sestili, P. Dietary supplementation with α-tocopherol reduces neuroinflammation and neuronal degeneration in the rat brain after kainic acid-induced status epilepticus. Free Radic. Res. 2011, 45, 1136–1142. [Google Scholar] [CrossRef]
- Martinez-Hernandez, A.; Bell, K.P.; Norenberg, M.D. Glutamine synthetase: Glial localization in brain. Science 1977, 195, 1356–1358. [Google Scholar] [CrossRef]
- Ambrogini, P.; Albertini, M.C.; Betti, M.; Galati, C.; Lattanzi, D.; Savelli, D.; Di Palma, M.; Saccomanno, S.; Bartolini, D.; Torquato, P. Neurobiological correlates of alpha-tocopherol antiepileptogenic effects and microRNA expression modulation in a rat model of kainate-induced seizures. Mol. Neurobiol. 2018, 55, 7822–7838. [Google Scholar]
- Tilleux, S.; Hermans, E. Neuroinflammation and regulation of glial glutamate uptake in neurological disorders. J. Neurosci. Res. 2007, 85, 2059–2070. [Google Scholar]
- Upaganlawar, A.B.; Wankhede, N.L.; Kale, M.B.; Umare, M.D.; Sehgal, A.; Singh, S.; Bhatia, S.; Al-Harrasi, A.; Najda, A.; Nurzyńska-Wierdak, R.; et al. Interweaving epilepsy and neurodegeneration: Vitamin E as a treatment approach. Biomed. Pharmacother. 2021, 143, 112146. [Google Scholar] [CrossRef]
- Crowley, T.; Cryan, J.F.; Downer, E.J.; O’Leary, O.F. Inhibiting neuroinflammation: The role and therapeutic potential of GABA in neuro-immune interactions. Brain Behav. Immun. 2016, 54, 260–277. [Google Scholar] [CrossRef]
- Rideau Batista Novais, A.; Crouzin, N.; Cavalier, M.; Boubal, M.; Guiramand, J.; Cohen-Solal, C.; de Jesus Ferreira, M.-C.; Cambonie, G.; Vignes, M.; Barbanel, G. Tiagabine improves hippocampal long-term depression in rat pups subjected to prenatal inflammation. PLoS ONE 2014, 9, e106302. [Google Scholar] [CrossRef] [PubMed]
- Fatemi, S.H.; Stary, J.M.; Earle, J.A.; Araghi-Niknam, M.; Eagan, E. GABAergic dysfunction in schizophrenia and mood disorders as reflected by decreased levels of glutamic acid decarboxylase 65 and 67 kDa and Reelin proteins in cerebellum. Schizophr. Res. 2005, 72, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Lanoue, A.C.; Dumitriu, A.; Myers, R.H.; Soghomonian, J.-J. Decreased glutamic acid decarboxylase mRNA expression in prefrontal cortex in Parkinson’s disease. Exp. Neurol. 2010, 226, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Gruol, D.L.; Vo, K.; Bray, J.G. Increased astrocyte expression of IL-6 or CCL2 in transgenic mice alters levels of hippocampal and cerebellar proteins. Front. Cell. Neurosci. 2014, 8, 234. [Google Scholar] [CrossRef]
- Abedi, Z.; Khaza’ai, H.; Vidyadaran, S.; Mutalib, M.S.A. The Modulation of NMDA and AMPA/Kainate Receptors by Tocotrienol-Rich Fraction and A-Tocopherol in Glutamate-Induced Injury of Primary Astrocytes. Biomedicines 2017, 5, 68. [Google Scholar] [CrossRef]
- Ziaei, S.; Kazemnejad, A.; Sedighi, A. The effect of vitamin E on the treatment of menstrual migraine. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2009, 15, CR16–CR19. [Google Scholar]
- Shaik, M.M.; Gan, S.H. Vitamin Supplementation as Possible Prophylactic Treatment against Migraine with Aura and Menstrual Migraine. BioMed Res. Int. 2015, 2015, 469529. [Google Scholar] [CrossRef]
- Granella, F.; Sances, G.; Messa, G.; De Marinis, M.; Manzoni, G. Treatment of menstrual migraine. Cephalalgia 1997, 17, 35–38. [Google Scholar] [CrossRef]
- Lee, J.C.M.; Simonyi, A.; Sun, A.Y.; Sun, G.Y. Phospholipases A2 and neural membrane dynamics: Implications for Alzheimer’s disease. J. Neurochem. 2011, 116, 813–819. [Google Scholar] [CrossRef]
- Fleury, C.; Mignotte, B.; Vayssière, J.-L. Mitochondrial reactive oxygen species in cell death signaling. Biochimie 2002, 84, 131–141. [Google Scholar] [CrossRef]
- Pinto, J.T.; Zempleni, J. Riboflavin. Adv. Nutr. 2016, 7, 973–975. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.-J.; Wu, W.-M.; Yang, F.-L.; Hsu, G.-S.W.; Huang, C.-Y. Vitamin B2 inhibits glutamate release from rat cerebrocortical nerve terminals. Neuroreport 2008, 19, 1335–1338. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Desbois, A.; Jiang, S.; Hou, S.T. Group B vitamins protect murine cerebellar granule cells from glutamate/NMDA toxicity. Neuroreport 2004, 15, 2241–2244. [Google Scholar] [CrossRef] [PubMed]
- Adelekan, D.A.; Adekile, A.D.; Thurnham, D.I. Dependence of pyridoxine metabolism on riboflavin status in sickle cell patients. Am. J. Clin. Nutr. 1987, 46, 86–90. [Google Scholar] [CrossRef]
- Petroff, O.A.C. GABA and glutamate in the human brain. Neuroscientist 2002, 8, 562–573. [Google Scholar] [CrossRef]
- Theofylaktopoulou, D.; Ulvik, A.; Midttun, Ø.; Ueland, P.M.; Vollset, S.E.; Nygård, O.; Hustad, S.; Tell, G.S.; Eussen, S.J.P.M. Vitamins B2 and B6 as determinants of kynurenines and related markers of interferon-γ-mediated immune activation in the community-based Hordaland Health Study. Br. J. Nutr. 2014, 112, 1065–1072. [Google Scholar] [CrossRef]
- Zinger, A.; Barcia, C.; Herrero, M.T.; Guillemin, G.J. The Involvement of Neuroinflammation and Kynurenine Pathway in Parkinson’s Disease. Park. Dis. 2011, 2011, 716859. [Google Scholar] [CrossRef]
- Curto, M.; Lionetto, L.; Negro, A.; Capi, M.; Fazio, F.; Giamberardino, M.A.; Simmaco, M.; Nicoletti, F.; Martelletti, P. Altered kynurenine pathway metabolites in serum of chronic migraine patients. J. Headache Pain 2016, 17, 47. [Google Scholar] [CrossRef]
- Guillemin, G.J. Quinolinic acid, the inescapable neurotoxin. FEBS J. 2012, 279, 1356–1365. [Google Scholar] [CrossRef]
- Schoenen, J.; Lenaerts, M.; Bastings, E. High-dose riboflavin as a prophylactic treatment of migraine: Results of an open pilot study. Cephalalgia 1994, 14, 328–329. [Google Scholar] [CrossRef]
- Gaul, C.; Diener, H.-C.; Danesch, U.; Group, M.S. Improvement of migraine symptoms with a proprietary supplement containing riboflavin, magnesium and Q10: A randomized, placebo-controlled, double-blind, multicenter trial. J. Headache Pain 2015, 16, 21. [Google Scholar] [CrossRef] [PubMed]
- Maizels, M.; Blumenfeld, A.; Burchette, R. A combination of riboflavin, magnesium, and feverfew for migraine prophylaxis: A randomized trial. Headache J. Head Face Pain 2004, 44, 885–890. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-S.; Lee, H.-F.; Tsai, C.-H.; Hsu, Y.-Y.; Fang, C.-J.; Chen, C.-J.; Hung, Y.-H.; Hu, F.-W. Effect of Vitamin B2 supplementation on migraine prophylaxis: A systematic review and meta-analysis. Nutr. Neurosci. 2022, 25, 1801–1812. [Google Scholar] [CrossRef]
- Holland, S.; Silberstein, S.D.; Freitag, F.; Dodick, D.W.; Argoff, C.; Ashman, E. Evidence-based guideline update: NSAIDs and other complementary treatments for episodic migraine prevention in adults: [RETIRED]. Rep. Qual. Stand. Subcomm. Am. Acad. Neurol. Am. Headache Soc. 2012, 78, 1346–1353. [Google Scholar] [CrossRef] [PubMed]
- Liebman, T.N.; Crystal, S.C. What is the evidence that riboflavin can be used for migraine prophylaxis? Einstein J. Biol. Med. 2011, 27, 7–9. [Google Scholar] [CrossRef]
- Strain, J.J.; Dowey, L.; Ward, M.; Pentieva, K.; McNulty, H. B-vitamins, homocysteine metabolism and CVD. Proc. Nutr. Soc. 2004, 63, 597–603. [Google Scholar] [CrossRef] [PubMed]
- Boldyrev, A.A. Molecular mechanisms of homocysteine toxicity. Biochemistry 2009, 74, 589–598. [Google Scholar] [CrossRef]
- Ponce-Monter, H.A.; Ortiz, M.I.; Garza-Hernández, A.F.; Monroy-Maya, R.; Soto-Ríos, M.; Carrillo-Alarcón, L.; Reyes-García, G.; Fernández-Martínez, E. Effect of diclofenac with B vitamins on the treatment of acute pain originated by lower-limb fracture and surgery. Pain Res. Treat. 2012, 2012, 104782. [Google Scholar] [CrossRef]
- Marashly, E.T.; Bohlega, S.A. Riboflavin Has Neuroprotective Potential: Focus on Parkinson’s Disease and Migraine. Front. Neurol. 2017, 8, 333. [Google Scholar] [CrossRef]
- Lominadze, D.; Tyagi, N.; Sen, U.; Ovechkin, A.; Tyagi, S.C. Homocysteine alters cerebral microvascular integrity and causes remodeling by antagonizing GABA-A receptor. Mol. Cell. Biochem. 2012, 371, 89–96. [Google Scholar] [CrossRef][Green Version]
- Lominadze, D.; Roberts, A.M.; Tyagi, N.; Moshal, K.S.; Tyagi, S.C. Homocysteine causes cerebrovascular leakage in mice. Am. J. Physiol.-Heart Circ. Physiol. 2006, 290, H1206–H1213. [Google Scholar] [CrossRef] [PubMed]
- Tietjen, G.E.; Maly, E.F. Migraine and Ischemic Stroke in Women. A Narrative Review. Headache J. Head Face Pain 2020, 60, 843–863. [Google Scholar] [CrossRef] [PubMed]
- Lippi, G.; Mattiuzzi, C.; Meschi, T.; Cervellin, G.; Borghi, L. Homocysteine and migraine. A narrative review. Clin. Chim. Acta 2014, 433, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Liampas, I.; Siokas, V.; Mentis, A.-F.A.; Aloizou, A.-M.; Dastamani, M.; Tsouris, Z.; Aslanidou, P.; Brotis, A.; Dardiotis, E. Serum Homocysteine, Pyridoxine, Folate, and Vitamin B12 Levels in Migraine: Systematic Review and Meta-Analysis. Headache J. Head Face Pain 2020, 60, 1508–1534. [Google Scholar] [CrossRef]
- Askari, G.; Nasiri, M.; Mozaffari-Khosravi, H.; Rezaie, M.; Bagheri-Bidakhavidi, M.; Sadeghi, O. The effects of folic acid and pyridoxine supplementation on characteristics of migraine attacks in migraine patients with aura: A double-blind, randomized placebo-controlled, clinical trial. Nutrition 2017, 38, 74–79. [Google Scholar] [CrossRef]
- Sadeghi, O.; Nasiri, M.; Maghsoudi, Z.; Pahlavani, N.; Rezaie, M.; Askari, G. Effects of pyridoxine supplementation on severity, frequency and duration of migraine attacks in migraine patients with aura: A double-blind randomized clinical trial study in Iran. Iran. J. Neurol. 2015, 14, 74–80. [Google Scholar]
- Garrido-Maraver, J.; Cordero, M.D.; Oropesa-Avila, M.; Vega, A.F.; de la Mata, M.; Pavon, A.D.; Alcocer-Gomez, E.; Calero, C.P.; Paz, M.V.; Alanis, M. Clinical applications of coenzyme Q10. Front. Biosci. 2014, 19, 619–633. [Google Scholar] [CrossRef]
- Neergheen, V.; Chalasani, A.; Wainwright, L.; Yubero, D.; Montero, R.; Artuch, R.; Hargreaves, I. Coenzyme Q 10 in the treatment of mitochondrial disease. J. Inborn Errors Metab. Screen. 2019, 5, e160063. [Google Scholar]
- Deichmann, R.; Lavie, C.; Andrews, S. Coenzyme q10 and statin-induced mitochondrial dysfunction. Ochsner J. 2010, 10, 16–21. [Google Scholar]
- Kobori, Y.; Ota, S.; Sato, R.; Yagi, H.; Soh, S.; Arai, G.; Okada, H. Antioxidant cosupplementation therapy with vitamin C, vitamin E, and coenzyme Q10 in patients with oligoasthenozoospermia. Arch. Ital. Urol. E Androl. 2014, 86, 1–4. [Google Scholar] [CrossRef]
- Lee, D.; Shim, M.S.; Kim, K.Y.; Noh, Y.H.; Kim, H.; Kim, S.Y.; Weinreb, R.N.; Ju, W.K. Coenzyme Q10 inhibits glutamate excitotoxicity and oxidative stress-mediated mitochondrial alteration in a mouse model of glaucoma. Investig. Ophthalmol. Vis. Sci. 2014, 55, 993–1005. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Huang, S.-K.; Wang, S.-J. Coenzyme Q10 Inhibits the Release of Glutamate in Rat Cerebrocortical Nerve Terminals by Suppression of Voltage-Dependent Calcium Influx and Mitogen-Activated Protein Kinase Signaling Pathway. J. Agric. Food Chem. 2012, 60, 11909–11918. [Google Scholar] [CrossRef] [PubMed]
- Kumari, S.; Mehta, S.L.; Milledge, G.Z.; Huang, X.; Li, H.; Li, P.A. Ubisol-Q10 Prevents Glutamate-Induced Cell Death by Blocking Mitochondrial Fragmentation and Permeability Transition Pore Opening. Int. J. Biol. Sci. 2016, 12, 688–700. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sândor, P.S.; Di Clemente, L.; Coppola, G.; Saenger, U.; Fumal, A.; Magis, D.; Seidel, L.; Agosti, R.; Schoenen, J. Efficacy of coenzyme Q10 in migraine prophylaxis: A randomized controlled trial. Neurology 2005, 64, 713–715. [Google Scholar] [CrossRef]
- Rozen, T.; Oshinsky, M.; Gebeline, C.; Bradley, K.; Young, W.; Shechter, A.; Silberstein, S. Open label trial of coenzyme Q10 as a migraine preventive. Cephalalgia 2002, 22, 137–141. [Google Scholar] [CrossRef]
- Dahri, M.; Tarighat-Esfanjani, A.; Asghari-Jafarabadi, M.; Hashemilar, M. Oral coenzyme Q10 supplementation in patients with migraine: Effects on clinical features and inflammatory markers. Nutr. Neurosci. 2019, 22, 607–615. [Google Scholar] [CrossRef]
- Hajihashemi, P.; Askari, G.; Khorvash, F.; Reza Maracy, M.; Nourian, M. The effects of concurrent Coenzyme Q10, L-carnitine supplementation in migraine prophylaxis: A randomized, placebo-controlled, double-blind trial. Cephalalgia 2019, 39, 648–654. [Google Scholar] [CrossRef]
- Sazali, S.; Badrin, S.; Norhayati, M.N.; Idris, N.S. Coenzyme Q10 supplementation for prophylaxis in adult patients with migraine—A meta-analysis. BMJ Open 2021, 11, e039358. [Google Scholar] [CrossRef]
- Shanmugam, S.; Karunaikadal, K.; Varadarajan, S.; Krishnan, M. Memantine ameliorates migraine headache. Ann. Indian Acad. Neurol. 2019, 22, 286. [Google Scholar] [CrossRef]
- Lampl, C.; Katsarava, Z.; Diener, H.; Limmroth, V. Lamotrigine reduces migraine aura and migraine attacks in patients with migraine with aura. J. Neurol. Neurosurg. Psychiatry 2005, 76, 1730–1732. [Google Scholar] [CrossRef]
- Waung, M.W.; Akerman, S.; Wakefield, M.; Keywood, C.; Goadsby, P.J. Metabotropic glutamate receptor 5: A target for migraine therapy. Ann. Clin. Transl. Neurol. 2016, 3, 560–571. [Google Scholar] [CrossRef]
- Hoffmann, J.; Charles, A. Glutamate and Its Receptors as Therapeutic Targets for Migraine. Neurotherapeutics 2018, 15, 361–370. [Google Scholar] [CrossRef]
- Muir, K.W.; Lees, K.R. Clinical experience with excitatory amino acid antagonist drugs. Stroke 1995, 26, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Watkins, J.C.; Jane, D.E. The glutamate story. Br. J. Pharmacol. 2006, 147, S100–S108. [Google Scholar] [CrossRef] [PubMed]
- Assarzadegan, F.; Asgarzadeh, S.; Hatamabadi, H.R.; Shahrami, A.; Tabatabaey, A.; Asgarzadeh, M. Serum concentration of magnesium as an independent risk factor in migraine attacks: A matched case–control study and review of the literature. Int. Clin. Psychopharmacol. 2016, 31, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Holton, K.F.; Taren, D.L.; Thomson, C.A.; Bennett, R.M.; Jones, K.D. The effect of dietary glutamate on fibromyalgia and irritable bowel symptoms. Clin. Exp. Rheumatol. 2012, 30, 10–17. [Google Scholar]
- Smith, Q.R. Transport of glutamate and other amino acids at the blood-brain barrier. J. Nutr. 2000, 130, 1016S–1022S. [Google Scholar] [CrossRef] [PubMed]
- Kuriakose, M.; Younger, D.; Ravula, A.R.; Alay, E.; Rama Rao, K.V.; Chandra, N. Synergistic role of oxidative stress and blood-brain barrier permeability as injury mechanisms in the acute pathophysiology of blast-induced neurotrauma. Sci. Rep. 2019, 9, 7717. [Google Scholar] [CrossRef]
- Huber, J.; Witt, K.; Hom, S.; Egleton, R.; Mark, K.; Davis, T. Inflammatory pain alters blood-brain barrier permeability and tight junctional protein expression. Am. J. Physiol.-Heart Circ. Physiol. 2001, 280, H1241–H1248. [Google Scholar] [CrossRef]
- Alves, J.L. Blood–brain barrier and traumatic brain injury. J. Neurosci. Res. 2014, 92, 141–147. [Google Scholar] [CrossRef]
- Borkum, J.M. Migraine triggers and oxidative stress: A narrative review and synthesis. Headache J. Head Face Pain 2016, 56, 12–35. [Google Scholar]
- Waeber, C.; Moskowitz, M.A. Migraine as an inflammatory disorder. Neurology 2005, 64, S9–S15. [Google Scholar] [CrossRef] [PubMed]
- Baad-Hansen, L.; Cairns, B.; Ernberg, M.; Svensson, P. Effect of systemic monosodium glutamate (MSG) on headache and pericranial muscle sensitivity. Cephalalgia 2009, 55, 993–1005. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.H.; Drouin, M.A.; Herbert, M.; Mao, Y.; Karsh, J. The monosodium glutamate symptom complex: Assessment in a double-blind, placebo-controlled, randomized study. J. Allergy Clin. Immunol. 1997, 99, 757–762. [Google Scholar] [CrossRef]
- Benbow, T.; Ekbatan, M.R.; Wang, G.H.Y.; Teja, F.; Exposto, F.G.; Svensson, P.; Cairns, B.E. Systemic administration of monosodium glutamate induces sexually dimorphic headache-and nausea-like behaviours in rats. Pain 2022, 163, 1838–1853. [Google Scholar] [CrossRef]
- Finocchi, C.; Sivori, G. Food as trigger and aggravating factor of migraine. Neurol. Sci. 2012, 33, 77–80. [Google Scholar] [CrossRef]
- Hindiyeh, N.A.; Zhang, N.; Farrar, M.; Banerjee, P.; Lombard, L.; Aurora, S.K. The Role of Diet and Nutrition in Migraine Triggers and Treatment: A Systematic Literature Review. Headache J. Head Face Pain 2020, 60, 1300–1316. [Google Scholar] [CrossRef]
- Alpay, K.; Ertaş, M.; Orhan, E.K.; Üstay, D.K.; Lieners, C.; Baykan, B. Diet restriction in migraine, based on IgG against foods: A clinical double-blind, randomised, cross-over trial. Cephalalgia 2010, 30, 829–837. [Google Scholar] [CrossRef]
- Martin, V.T.; Vij, B. Diet and headache: Part 1. Headache J. Head Face Pain 2016, 56, 1543–1552. [Google Scholar] [CrossRef]
- Martin, V.T.; Behbehani, M.M. Toward a rational understanding of migraine trigger factors. Med. Clin. N. Am. 2001, 85, 911–941. [Google Scholar] [CrossRef]
- Gazerani, P. Migraine and diet. Nutrients 2020, 12, 1658. [Google Scholar] [CrossRef]
- Holton, K.F.; Kirkland, A.E.; Baron, M.; Ramachandra, S.S.; Langan, M.T.; Brandley, E.T.; Baraniuk, J.N. The Low Glutamate Diet Effectively Improves Pain and Other Symptoms of Gulf War Illness. Nutrients 2020, 12, 2593. [Google Scholar] [CrossRef] [PubMed]
- Holton, K.F.; Ndege, P.K.; Clauw, D.J. Dietary correlates of chronic widespread pain in Meru, Kenya. Nutrition 2018, 53, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.D.; Terpening, C.M.; Schmidt, S.O.; Gums, J.G. Relief of fibromyalgia symptoms following discontinuation of dietary excitotoxins. Ann. Pharmacother. 2001, 35, 702–706. [Google Scholar] [CrossRef] [PubMed]
| Migraine Patients | Healthy Controls | References | ||
|---|---|---|---|---|
| With Aura | Without Aura | |||
| Mean (SD) | Mean (SD) | Mean (SD) | ||
| Plasma | ||||
| Based on aura (nmol/mL) | 14 (6)–454 (98) | 13 (6)–485 (129) | 15 (8)–227 (87) | [53,81,82,85] |
| Without categorization (nmol/mL) | 23 (1)–75 (20) | 9 (2)–32 (20) | [80,89,92] | |
| Platelets | ||||
| (µmol/1010 plts) | 0.50 (0.22)–0.58 (0.12) | 0.43 (0.17)–0.45 (0.16) | 0.34 (0.09)–0.45 (0.11) | [84,85] |
| Cerebrospinal fluid | ||||
| Based on aura (nmol/mL) | 9.3 (1.5) | 7.5 (1.8) | 4.5 (1.8) | [82] |
| Without categorization (nmol/mL) | 2.18 (0.40) | 1.37 (0.30) | [93] | |
| Brain (MRS) | ||||
| Visual cortex (mmol/l) | 6.8 (0.5) | 7.0 (0.5) | 6.4 (0.8) | [94] |
| OCC | 7.20 (1.45) | 6.68 (1.25) | [95] | |
| APC | 6.98 (0.85) | 6.22 (0.97) | [95] | |
| PCC (mmol) | 7.21 (0.96) | 7.27 (0.94) | [96] | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martami, F.; Holton, K.F. Targeting Glutamate Neurotoxicity through Dietary Manipulation: Potential Treatment for Migraine. Nutrients 2023, 15, 3952. https://doi.org/10.3390/nu15183952
Martami F, Holton KF. Targeting Glutamate Neurotoxicity through Dietary Manipulation: Potential Treatment for Migraine. Nutrients. 2023; 15(18):3952. https://doi.org/10.3390/nu15183952
Chicago/Turabian StyleMartami, Fahimeh, and Kathleen F. Holton. 2023. "Targeting Glutamate Neurotoxicity through Dietary Manipulation: Potential Treatment for Migraine" Nutrients 15, no. 18: 3952. https://doi.org/10.3390/nu15183952
APA StyleMartami, F., & Holton, K. F. (2023). Targeting Glutamate Neurotoxicity through Dietary Manipulation: Potential Treatment for Migraine. Nutrients, 15(18), 3952. https://doi.org/10.3390/nu15183952






