Insights into Nutritional Strategies in Psoriasis
Abstract
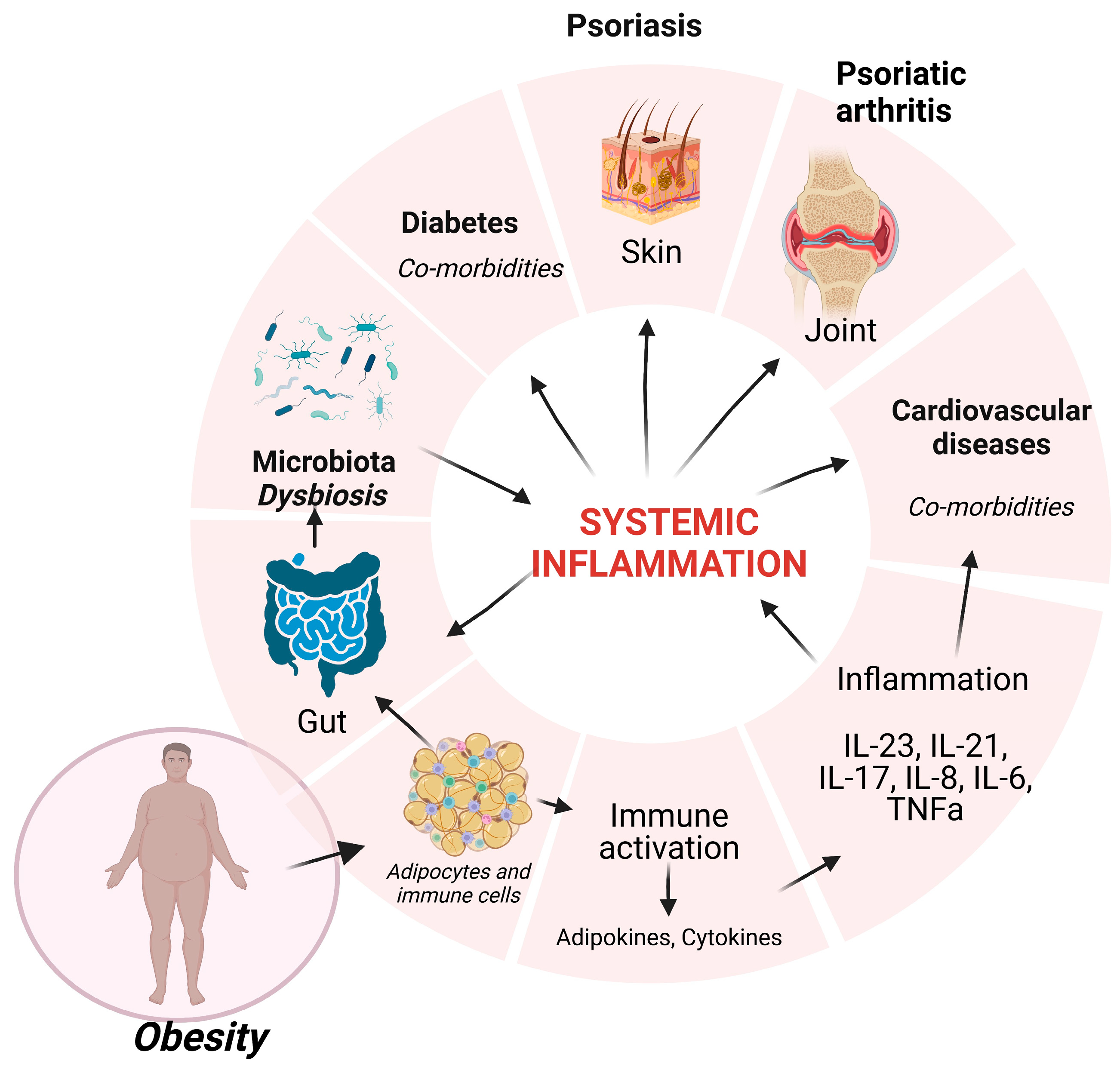
1. Introduction
2. Psoriatic Patients and Obesity
2.1. Obesity in Psoriasis Onset
2.2. Adipose Tissue Dysregulation in Psoriatic Patients
2.3. Microbiota in Obese Psoriatic Patients
3. Animal Experimental Models—Direct Proof of the Link between Psoriasis and a High-Fat Diet
Gut and Skin Microbiome in Animal Models of Psoriasis
4. Nutritional Therapeutical Strategies in Psoriasis
4.1. Skin–Gut Link—Inflammation Is the Trigger of Deregulation
4.2. Nutritional Strategies—Reducing Obesity and Inflammatory Status Is the Main Target
4.2.1. Low-Calorie Diet
4.2.2. Gluten-Free Diet
4.2.3. Supplements
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rachakonda, T.D.; Schupp, C.W.; Armstrong, A.W. Psoriasis prevalence among adults in the United States. J. Am. Acad. Dermatol. 2014, 70, 512–516. [Google Scholar] [CrossRef]
- De Arruda, L.; de Moraes, A. The impact of psoriasis on quality of life. Br. J. Dermatol. 2001, 144, 33–36. [Google Scholar] [CrossRef] [PubMed]
- Schadler, E.D.; Ortel, B.; Mehlis, S.L. Biologics for the primary care physician: Review and treatment of psoriasis. Dis. Mon. 2018, 65, 51–90. [Google Scholar] [CrossRef]
- Brandon, A.; Mufti, A.; Sibbald, R.G. Diagnosis and Management of Cutaneous Psoriasis: A Review. Adv. Ski. Wound Care 2019, 32, 58–69. [Google Scholar] [CrossRef]
- Salgado-Boquete, L.; Carrascosa, J.M.; Llamas-Velasco, M.; Ruiz-Villaverde, R.; de la Cueva, P.; Belinchón, I. A New Classification of the Severity of Psoriasis: What’s Moderate Psoriasis? Life 2021, 11, 627. [Google Scholar] [CrossRef]
- Budu-Aggrey, A.; Brumpton, B.; Tyrrell, J.; Watkins, S.; Modalsli, E.H.; Celis-Morales, C.; Ferguson, L.D.; Vie, G.Å.; Palmer, T.; Fritsche, L.G.; et al. Evidence of a causal relationship between body mass index and psoriasis: A mendelian randomization study. PLoS Med. 2019, 16, e1002739. [Google Scholar] [CrossRef] [PubMed]
- Castaldo, G.; Pagano, I.; Grimaldi, M.; Marino, C.; Molettieri, P.; Santoro, A.; Stillitano, I.; Romano, R.; Montoro, P.; D’ursi, A.M.; et al. Effect of Very-Low-Calorie Ketogenic Diet on Psoriasis Patients: A Nuclear Magnetic Resonance-Based Metabolomic Study. J. Proteome Res. 2021, 20, 1509–1521. [Google Scholar] [CrossRef]
- Barrea, L.; Megna, M.; Cacciapuoti, S.; Frias-Toral, E.; Fabbrocini, G.; Savastano, S.; Colao, A.; Muscogiuri, G. Very low-calorie ketogenic diet (VLCKD) in patients with psoriasis and obesity: An update for dermatologists and nutritionists. Crit. Rev. Food Sci. Nutr. 2022, 62, 398–414. [Google Scholar] [CrossRef]
- Kunz, M.; Simon, J.C.; Saalbach, A. Psoriasis: Obesity and Fatty Acids. Front. Immunol. 2019, 10, 1807. [Google Scholar] [CrossRef] [PubMed]
- Giles, J.T.; Ogdie, A.; Gomez-Reino, J.J.; Helliwell, P.S.; Germino, R.; Stockert, L.; Young, P.; Joseph, W.; Mundayat, R.; Graham, D.; et al. Impact of baseline body mass index on the efficacy and safety of tofacitinib in patients with psoriatic arthritis. RMD Open 2021, 7, e001486. [Google Scholar] [CrossRef]
- Boutari, C.; Mantzoros, C.S. A 2022 update on the epidemiology of obesity and a call to action: As its twin COVID-19 pandemic appears to be receding, the obesity and dysmetabolism pandemic continues to rage on. Metabolism 2022, 133, 155217. [Google Scholar] [CrossRef]
- Barros, G.; Duran, P.; Vera, I.; Bermúdez, V. Exploring the Links between Obesity and Psoriasis: A Comprehensive Review. Int. J. Mol. Sci. 2022, 23, 7499. [Google Scholar] [CrossRef]
- Armstrong, A.W.; Harskamp, C.T.; Armstrong, E.J. The association between psoriasis and obesity: A systematic review and meta-analysis of observational studies. Nutr. Diabetes 2012, 2, e54. [Google Scholar] [CrossRef]
- Kumthekar, A.; Ogdie, A. Obesity and Psoriatic Arthritis: A Narrative Review. Rheumatol. Ther. 2020, 7, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Liu, K.; Duan, X.; Xu, L.; Yang, Q.; Liu, F. A Comparison of Clinical Characteristics in Overweight/Obese and Normal Weight Patients with Psoriasis Vulgaris: A Bicentric Retrospective Observational Study. Clin. Cosmet. Investig. Dermatol. 2023, 16, 1377–1385. [Google Scholar] [CrossRef] [PubMed]
- Zanesco, S.; Hall, W.; Gibson, R.; Griffiths, C.; Maruthappu, T. Approaches to nutrition intervention in plaque psoriasis, a multi-system inflammatory disease-The Diet and Psoriasis Project (DIEPP). Nutr. Bull. 2022, 47, 524–537. [Google Scholar] [CrossRef] [PubMed]
- Blake, T.; Gullick, N.J.; Hutchinson, C.E.; Barber, T.M. Psoriatic disease and body composition: A systematic review and narrative synthesis. PLoS ONE. 2020, 15, e0237598. [Google Scholar] [CrossRef]
- Galluzzo, M.; Talamonti, M.; Perino, F.; Servoli, S.; Giordano, D.; Chimenti, S.; De Simone, C.; Peris, K. Bioelectrical impedance analysis to define an excess of body fat: Evaluation in patients with psoriasis. J. Dermatol. Treat. 2017, 28, 299–303. [Google Scholar] [CrossRef]
- Kanda, N.; Hoashi, T.; Saeki, H. Nutrition and Psoriasis. Int. J. Mol. Sci. 2020, 21, 5405. [Google Scholar] [CrossRef]
- Tilg, H.; Moschen, A.R. Adipocytokines: Mediators linking adipose tissue, inflammation and immunity. Nat. Rev. Immunol. 2006, 6, 772–783. [Google Scholar] [CrossRef]
- Haczeyni, F.; Bell-Anderson, K.S.; Farrell, G.C. Causes and mechanisms of adipocyte enlargement and adipose expansion. Obes. Rev. 2018, 19, 406–420. [Google Scholar] [CrossRef]
- Guo, Z.; Yang, Y.; Liao, Y.; Shi, Y.; Zhang, L.-J. Emerging Roles of Adipose Tissue in the Pathogenesis of Psoriasis and Atopic Dermatitis in Obesity. JID Innov. 2021, 2, 100064. [Google Scholar] [CrossRef]
- Caiazzo, G.; Fabbrocini, G.; Di Caprio, R.; Raimondo, A.; Scala, E.; Balato, N.; Balato, A. Psoriasis, Cardiovascular Events, and Biologics: Lights and Shadows. Front. Immunol. 2018, 9, 1668. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Autieri, M.V.; Scalia, R. Adipose tissue inflammation and metabolic dysfunction in obesity. Am. J. Physiol. Cell Physiol. 2021, 320, C375–C391. [Google Scholar] [CrossRef] [PubMed]
- Clemente-Suárez, V.J.; Redondo-Flórez, L.; Beltrán-Velasco, A.I.; Martín-Rodríguez, A.; Martínez-Guardado, I.; Navarro-Jiménez, E.; Laborde-Cárdenas, C.C.; Tornero-Aguilera, J.F. The Role of Adipokines in Health and Disease. Biomedicines 2023, 11, 1290. [Google Scholar] [CrossRef]
- Chyl-Surdacka, K.M.; Gerkowicz, A.; Bartosińska, J.; Kowal, M.; Przepiórka-Kosińska, J.; Surdacki, G.; Krasowska, D.; Chodorowska, G. Analysis of serum chemerin concentrations in psoriatic patients in relation to metabolic abnormalities. Postepy. Dermatol. Alergol. 2019, 36, 531–537. [Google Scholar] [CrossRef]
- Jakab, J.; Miškić, B.; Mikšić, Š.; Juranić, B.; Ćosić, V.; Schwarz, D.; Včev, A. Adipogenesis as a Potential Anti-Obesity Target: A Review of Pharmacological Treatment and Natural Products. Diabetes Metab. Syndr. Obes. 2021, 14, 67–83. [Google Scholar] [CrossRef] [PubMed]
- Gisondi, P.; Fostini, A.C.; Fossà, I.; Girolomoni, G.; Targher, G. Psoriasis and the metabolic syndrome. Clin. Dermatol. 2018, 36, 21–28. [Google Scholar] [CrossRef]
- Zhu, K.J.; Shi, G.; Zhang, C.; Li, M.; Zhu, C.Y.; Fan, Y.M. Adiponectin levels in patients with psoriasis: A meta-analysis. J. Dermatol. 2013, 40, 438–442. [Google Scholar] [CrossRef] [PubMed]
- Fantuzzi, G. Adipose tissue, adipokines, and inflammation. J. Allergy. Clin. Immunol. 2005, 115, 911–919. [Google Scholar] [CrossRef] [PubMed]
- Raucci, R.; Rusolo, F.; Sharma, A.; Colonna, G.; Castello, G.; Costantini, S. Functional and structural features of adipokine family. Cytokine 2013, 61, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Słuczanowska-Głabowska, S.; Staniszewska, M.; Marchlewicz, M.; Duchnik, E.; Łuczkowska, K.; Safranow, K.; Machaliński, B.; Pawlik, A. Adiponectin, Leptin and Resistin in Patients with Psoriasis. J. Clin. Med. 2023, 12, 663. [Google Scholar] [CrossRef] [PubMed]
- Bavoso, N.C.; Pinto, J.M.; Soares, M.M.S.; Diniz, M.D.S.; Júnior, A.L.T. Psoriasis in obesity: Comparison of serum levels of leptin and adiponectin in obese subjects—Cases and controls. An. Bras. Dermatol. 2019, 94, 192–197. [Google Scholar] [CrossRef]
- Kong, Y.; Zhang, S.; Wu, R.; Su, X.; Peng, D.; Zhao, M.; Su, Y. New insights into different adipokines in linking the pathophysiology of obesity and psoriasis. Lipids Health Dis. 2019, 18, 171. [Google Scholar] [CrossRef]
- Neagu, M.; Surcel, M.; Constantin, C. Systemic Circulating Leptin—Aiding New Dimension of Immune-Related Skin Carcinogenesis and Lipid Metabolism. SEE J. Immunol. 2023, 6, 1–6. [Google Scholar] [CrossRef]
- Dopytalska, K.; Baranowska-Bik, A.; Roszkiewicz, M.; Bik, W.; Walecka, I. The role of leptin in selected skin diseases. Lipids Health Dis. 2020, 19, 215. [Google Scholar] [CrossRef] [PubMed]
- Kyriakou, A.; Patsatsi, A.; Sotiriadis, D.; Goulis, D.G. Serum Leptin, Resistin, and Adiponectin Concentrations in Psoriasis: A Meta-Analysis of Observational Studies. Dermatology 2017, 233, 378–389. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.; Yoo, J.A.; Yoon, H.; Han, T.; Yoon, J.; An, S.; Cho, J.Y.; Lee, J. The Role of Leptin in the Association between Obesity and Psoriasis. Biomol. Ther. 2021, 29, 11–21. [Google Scholar] [CrossRef]
- Tripathi, D.; Kant, S.; Pandey, S.; Ehtesham, N.Z. Resistin in metabolism, inflammation, and disease. FEBS J. 2020, 287, 3141–3149. [Google Scholar] [CrossRef]
- Stockenhuber, K.; Hegazy, A.N.; West, N.; Ilott, N.E.; Stockenhuber, A.; Bullers, S.J.; Thornton, E.E.; Arnold, I.C.; Tucci, A.; Waldmann, H.; et al. Foxp3+ T reg cells control psoriasiform inflammation by restraining an IFN-I–driven CD8+ T cell response. J. Exp. Med. 2018, 215, 1987–1998. [Google Scholar] [CrossRef]
- Paroutoglou, K.; Papadavid, E.; Christodoulatos, G.S.; Dalamaga, M. Deciphering the Association between Psoriasis and Obesity: Current Evidence and Treatment Considerations. Curr. Obes. Rep. 2020, 9, 165–178. [Google Scholar] [CrossRef]
- Krautbauer, S.; Wanninger, J.; Eisinger, K.; Hader, Y.; Beck, M.; Kopp, A.; Schmid, A.; Weiss, T.S.; Dorn, C.; Buechler, C. Chemerin is highly expressed in hepatocytes and is induced in non-alcoholic steatohepatitis liver. Exp. Mol. Pathol. 2013, 95, 199–205. [Google Scholar] [CrossRef]
- Chiricozzi, A.; Raimondo, A.; Lembo, S.; Fausti, F.; Dini, V.; Costanzo, A.; Monfrecola, G.; Balato, N.; Ayala, F.; Romanelli, M.; et al. Crosstalk between skin inflammation and adipose tissue-derived products: Pathogenic evidence linking psoriasis to increased adiposity. Expert Rev. Clin. Immunol. 2016, 12, 1299–1308. [Google Scholar] [CrossRef]
- Ernst, M.C.; Sinal, C.J. Chemerin: At the crossroads of inflammation and obesity. Trends Endocrinol. Metab. 2010, 21, 660–667. [Google Scholar] [CrossRef]
- Buechler, C.; Feder, S.; Haberl, E.M.; Aslanidis, C. Chemerin Isoforms and Activity in Obesity. Int. J. Mol. Sci. 2019, 20, 1128. [Google Scholar] [CrossRef]
- Helfer, G.; Wu, Q.-F. Chemerin: A multifaceted adipokine involved in metabolic disorders. J. Endocrinol. 2018, 238, R79–R94. [Google Scholar] [CrossRef]
- Antal, D.; Alimohammadi, S.; Bai, P.; Szöllősi, A.G.; Szántó, M. Antigen-Presenting Cells in Psoriasis. Life 2022, 12, 234. [Google Scholar] [CrossRef]
- Albanesi, C.; Scarponi, C.; Pallotta, S.; Daniele, R.; Bosisio, D.; Madonna, S.; Fortugno, P.; Gonzalvo-Feo, S.; Franssen, J.-D.; Parmentier, M.; et al. Chemerin expression marks early psoriatic skin lesions and correlates with plasmacytoid dendritic cell recruitment. J. Exp. Med. 2009, 206, 249–258. [Google Scholar] [CrossRef]
- Kong, S.-M.; Sun, X.-Y.; Cui, W.-Y.; Cao, Y.-C. Chemerin Exacerbates Psoriasis by Stimulating Keratinocyte Proliferation and Cytokine Production. Curr. Med. Sci. 2023, 43, 399–408. [Google Scholar] [CrossRef]
- Bremner, J.D.; Moazzami, K.; Wittbrodt, M.T.; Nye, J.A.; Lima, B.B.; Gillespie, C.F.; Rapaport, M.H.; Pearce, B.D.; Shah, A.J.; Vaccarino, V. Diet, Stress and Mental Health. Nutrients 2020, 12, 2428. [Google Scholar] [CrossRef]
- Hölsken, S.; Krefting, F.; Schedlowski, M.; Sondermann, W. Common Fundamentals of Psoriasis and Depression. Acta Derm. Venereol. 2021, 101, adv00609. [Google Scholar] [CrossRef]
- Chen, L.; Li, J.; Zhu, W.; Kuang, Y.; Liu, T.; Zhang, W.; Chen, X.; Peng, C. Skin and Gut Microbiome in Psoriasis: Gaining Insight into the Pathophysiology of It and Finding Novel Therapeutic Strategies. Front. Microbiol. 2020, 11, 589726. [Google Scholar] [CrossRef]
- Sikora, M.; Stec, A.; Chrabaszcz, M.; Knot, A.; Waskiel-Burnat, A.; Rakowska, A.; Olszewska, M.; Rudnicka, L. Gut Microbiome in Psoriasis: An Updated Review. Pathogens 2020, 9, 463. [Google Scholar] [CrossRef]
- Vetrani, C.; Di Nisio, A.; Paschou, S.A.; Barrea, L.; Muscogiuri, G.; Graziadio, C.; Savastano, S.; Colao, A. On Behalf of the Obesity Programs of Nutrition Education Research and Assessment Opera Group. From Gut Microbiota through Low-Grade Inflammation to Obesity: Key Players and Potential Targets. Nutrients 2022, 14, 2103. [Google Scholar] [CrossRef]
- Crovesy, L.; Masterson, D.; Rosado, E.L. Profile of the gut microbiota of adults with obesity: A systematic review. Eur. J. Clin. Nutr. 2020, 74, 1251–1262. [Google Scholar] [CrossRef]
- Stojanov, S.; Berlec, A.; Štrukelj, B. The Influence of Probiotics on the Firmicutes/Bacteroidetes Ratio in the Treatment of Obesity and Inflammatory Bowel disease. Microorganisms 2020, 8, 1715. [Google Scholar] [CrossRef]
- Thye, A.Y.-K.; Bah, Y.-R.; Law, J.W.-F.; Tan, L.T.-H.; He, Y.-W.; Wong, S.-H.; Thurairajasingam, S.; Chan, K.-G.; Lee, L.-H.; Letchumanan, V. Gut–Skin Axis: Unravelling the Connection between the Gut Microbiome and Psoriasis. Biomedicines 2022, 10, 1037. [Google Scholar] [CrossRef]
- Tan, X.; Wang, Y.; Gong, T. The interplay between oral microbiota, gut microbiota and systematic diseases. J. Oral Microbiol. 2023, 15, 2213112. [Google Scholar] [CrossRef]
- Al Bander, Z.; Nitert, M.D.; Mousa, A.; Naderpoor, N. The Gut Microbiota and Inflammation: An Overview. Int. J. Environ. Res. Public Health 2020, 17, 7618. [Google Scholar] [CrossRef]
- Al-Mansoori, L.; Al-Jaber, H.; Prince, M.S.; Elrayess, M.A. Role of Inflammatory Cytokines, Growth Factors and Adipokines in Adipogenesis and Insulin Resistance. Inflammation 2022, 45, 31–44. [Google Scholar] [CrossRef]
- Wilchowski, S.M. The Role of the Gut Microbiome in Psoriasis: From Pathogens to Pathology. J. Clin. Aesthet. Dermatol. 2022, 15, S25–S28. [Google Scholar]
- Chen, G.; Chen, Z.-M.; Fan, X.-Y.; Jin, Y.-L.; Li, X.; Wu, S.-R.; Ge, W.-W.; Lv, C.-H.; Wang, Y.-K.; Chen, J.-G. Gut–Brain–Skin Axis in Psoriasis: A Review. Dermatol. Ther. 2020, 11, 25–38. [Google Scholar] [CrossRef]
- Surcel, M.; Huică, R.-I.; Munteanu, A.N.; Isvoranu, G.; Ciotaru, D.; Constantin, C.; Bratu, O.; Căruntu, C.; Neagu, M.; Ursaciuc, C. Phenotypic changes of lymphocyte populations in psoriasiform dermatitis animal model. Exp. Ther. Med. 2018, 17, 1030–1038. [Google Scholar] [CrossRef]
- Flutter, B.; Nestle, F.O. TLRs to cytokines: Mechanistic insights from the imiquimod mouse model of psoriasis. Eur. J. Immunol. 2013, 43, 3138–3146. [Google Scholar] [CrossRef]
- Zanvit, P.; Konkel, J.E.; Jiao, X.; Kasagi, S.; Zhang, D.; Wu, R.; Chia, C.; Ajami, N.J.; Smith, D.P.; Petrosino, J.F.; et al. Antibiotics in neonatal life increase murine susceptibility to experimental psoriasis. Nat. Commun. 2015, 6, 8424. [Google Scholar] [CrossRef]
- Kiyohara, H.; Sujino, T.; Teratani, T.; Miyamoto, K.; Arai, M.M.; Nomura, E.; Harada, Y.; Aoki, R.; Koda, Y.; Mikami, Y.; et al. Toll-Like Receptor 7 Agonist–Induced Dermatitis Causes Severe Dextran Sulfate Sodium Colitis by Altering the Gut Microbiome and Immune Cells. Cell. Mol. Gastroenterol. Hepatol. 2018, 7, 135–156. [Google Scholar] [CrossRef]
- Stehlikova, Z.; Kostovcikova, K.; Kverka, M.; Rossmann, P.; Dvorak, J.; Novosadova, I.; Kostovcik, M.; Coufal, S.; Srutkova, D.; Prochazkova, P.; et al. Crucial Role of Microbiota in Experimental Psoriasis Revealed by a Gnotobiotic Mouse Model. Front. Microbiol. 2019, 10, 236. [Google Scholar] [CrossRef]
- Slominski, A.T.; Manna, P.R.; Tuckey, R.C. On the role of skin in the regulation of local and systemic steroidogenic activities. Steroids 2015, 103, 72–88. [Google Scholar] [CrossRef]
- Slominski, A.T.; Slominski, R.M.; Raman, C.; Chen, J.Y.; Athar, M.; Elmets, C. Neuroendocrine signaling in the skin with a special focus on the epidermal neuropeptides. Am. J. Physiol. Cell Physiol. 2022, 323, C1757–C1776. [Google Scholar] [CrossRef]
- Shinno-Hashimoto, H.; Hashimoto, Y.; Wei, Y.; Chang, L.; Fujita, Y.; Ishima, T.; Matsue, H.; Hashimoto, K. Abnormal composition of microbiota in the gut and skin of imiquimod-treated mice. Sci. Rep. 2021, 11, 11265. [Google Scholar] [CrossRef]
- Fine, R.L.; Mubiru, D.L.; Kriegel, M.A. Friend or foe? Lactobacillus in the context of autoimmune disease. Adv. Immunol. 2020, 146, 29–56. [Google Scholar] [CrossRef]
- Zákostelská, Z.; Málková, J.; Klimešová, K.; Rossmann, P.; Hornová, M.; Novosádová, I.; Stehlíková, Z.; Kostovčík, M.; Hudcovic, T.; Štepánková, R.; et al. Intestinal Microbiota Promotes Psoriasis-Like Skin Inflammation by Enhancing Th17 Response. PLoS ONE 2016, 11, e0159539. [Google Scholar] [CrossRef]
- Alekseyenko, A.V.; Perez-Perez, G.I.; De Souza, A.; Strober, B.; Gao, Z.; Bihan, M.; Li, K.; Methé, B.A.; Blaser, M.J. Community differentiation of the cutaneous microbiota in psoriasis. Microbiome 2013, 1, 31. [Google Scholar] [CrossRef]
- Chen, H.L.; Zeng, Y.B.; Zhang, Z.Y.; Kong, C.Y.; Zhang, S.L.; Li, Z.M.; Huang, J.T.; Xu, Y.Y.; Mao, Y.Q.; Cai, P.R.; et al. Gut and Cutaneous Microbiome Featuring Abundance of Lactobacillus reuteri Protected Against Psoriasis-Like Inflammation in Mice. J. Inflamm. Res. 2021, 14, 6175–6190. [Google Scholar] [CrossRef]
- Gangwar, R.S.; Gudjonsson, J.E.; Ward, N.L. Mouse Models of Psoriasis: A Comprehensive Review. J. Investig. Dermatol. 2022, 142, 884–897. [Google Scholar] [CrossRef]
- Schön, M.P.; Manzke, V.; Erpenbeck, L. Animal models of psoriasis—Highlights and drawbacks. J. Allergy Clin. Immunol. 2021, 147, 439–455. [Google Scholar] [CrossRef]
- Parab, S.; Doshi, G. The Experimental Animal Models in Psoriasis Research: A Comprehensive Review. Int. Immunopharmacol. 2023, 117, 109897. [Google Scholar] [CrossRef]
- Brand, A.; Diener, N.; Zahner, S.P.; Tripp, C.; Backer, R.A.; Karram, K.; Jiang, A.; Mellman, I.; Stoitzner, P.; Clausen, B.E. E-Cadherin is Dispensable to Maintain Langerhans Cells in the Epidermis. J. Investig. Dermatol. 2020, 140, 132–142. [Google Scholar] [CrossRef]
- Cibrian, D.; Castillo-González, R.; Fernández-Gallego, N.; de la Fuente, H.; Jorge, I.; Saiz, M.L.; Punzón, C.; Ramírez-Huesca, M.; Vicente-Manzanares, M.; Fresno, M.; et al. Targeting L-type amino acid transporter 1 in innate and adaptive T cells efficiently controls skin inflammation. J. Allergy Clin. Immunol. 2020, 145, 199–214.e11. [Google Scholar] [CrossRef]
- Kiss, B.; Szántó, M.; Hegedűs, C.; Antal, D.; Szödényi, A.; Márton, J.; Méhes, G.; Virág, L.; Szegedi, A.; Bai, P. Poly(ADP-ribose) polymerase-1 depletion enhances the severity of inflammation in an imiquimod-induced model of psoriasis. Exp. Dermatol. 2020, 29, 79–85. [Google Scholar] [CrossRef]
- Schuster, C.; Huard, A.; Sirait-Fischer, E.; Dillmann, C.; Brüne, B.; Weigert, A. S1PR4-dependent CCL2 production promotes macrophage recruitment in a murine psoriasis model. Eur. J. Immunol. 2020, 50, 839–845. [Google Scholar] [CrossRef]
- Soley, B.d.S.; Silva, L.M.; Mendes, D.A.G.B.; Báfica, A.; Pesquero, J.B.; Bader, M.; Witherden, D.A.; Havran, W.L.; Calixto, J.B.; Otuki, M.F.; et al. B1 and B2 kinin receptor blockade improves psoriasis-like disease. Br. J. Pharmacol. 2020, 177, 3535–3551. [Google Scholar] [CrossRef]
- Hovhannisyan, Z.; Liu, N.; Khalil-Aguero, S.; Panea, C.; VanValkenburgh, J.; Zhang, R.; Lim, W.K.; Bai, Y.; Fury, W.; Huang, T.; et al. Enhanced IL-36R signaling promotes barrier impairment and inflammation in skin and intestine. Sci. Immunol. 2020, 5, eaax1686. [Google Scholar] [CrossRef]
- Karamani, C.; Antoniadou, I.T.; Dimou, A.; Andreou, E.; Kostakis, G.; Sideri, A.; Vitsos, A.; Gkavanozi, A.; Sfiniadakis, I.; Skaltsa, H.; et al. Optimization of psoriasis mouse models. J. Pharmacol. Toxicol. Methods 2021, 108, 107054. [Google Scholar] [CrossRef]
- Moreno-Sosa, T.; Sánchez, M.B.; Pietrobon, E.O.; Fernandez-Muñoz, J.M.; Zoppino, F.C.M.; Neira, F.J.; Germanó, M.J.; Cargnelutti, D.E.; Innocenti, A.C.; Jahn, G.A.; et al. Desmoglein-4 Deficiency Exacerbates Psoriasiform Dermatitis in Rats While Psoriasis Patients Displayed a Decreased Gene Expression of DSG4. Front. Immunol. 2021, 12, 625617. [Google Scholar] [CrossRef]
- Billi, A.C.; Ludwig, J.E.; Fritz, Y.; Rozic, R.; Swindell, W.R.; Tsoi, L.C.; Gruzska, D.; Abdollahi-Roodsaz, S.; Xing, X.; Diaconu, D.; et al. KLK6 expression in skin induces PAR1-mediated psoriasiform dermatitis and inflammatory joint disease. J. Clin. Investig. 2020, 130, 3151–3157. [Google Scholar] [CrossRef]
- Chan, J.R.; Blumenschein, W.; Murphy, E.; Diveu, C.; Wiekowski, M.; Abbondanzo, S.; Lucian, L.; Geissler, R.; Brodie, S.; Kimball, A.B.; et al. IL-23 stimulates epidermal hyperplasia via TNF and IL-20R2–dependent mechanisms with implications for psoriasis pathogenesis. J. Exp. Med. 2006, 203, 2577–2587. [Google Scholar] [CrossRef]
- Wang, M.; Zhang, S.; Zheng, G.; Huang, J.; Songyang, Z.; Zhao, X.; Lin, X. Gain-of-Function Mutation of Card14 Leads to Spontaneous Psoriasis-like Skin Inflammation through Enhanced Keratinocyte Response to IL-17A. Immunity 2018, 49, 66–79.e5. [Google Scholar] [CrossRef]
- Croxford, A.L.; Karbach, S.; Kurschus, F.C.; Wörtge, S.; Nikolaev, A.; Yogev, N.; Klebow, S.; Schüler, R.; Reissig, S.; Piotrowski, C.; et al. IL-6 Regulates Neutrophil Microabscess Formation in IL-17A-Driven Psoriasiform Lesions. J. Investig. Dermatol. 2014, 134, 728–735. [Google Scholar] [CrossRef]
- Karbach, S.; Croxford, A.L.; Oelze, M.; Schüler, R.; Minwegen, D.; Wegner, J.; Koukes, L.; Yogev, N.; Nikolaev, A.; Reißig, S.; et al. Interleukin 17 Drives Vascular Inflammation, Endothelial Dysfunction, and Arterial Hypertension in Psoriasis-Like Skin Disease. Arter. Thromb. Vasc. Biol. 2014, 34, 2658–2668. [Google Scholar] [CrossRef]
- Jordan, C.T.; Cao, L.; Roberson, E.D.; Duan, S.; Helms, C.A.; Nair, R.P.; Duffin, K.C.; Stuart, P.E.; Goldgar, D.; Hayashi, G.; et al. Rare and Common Variants in CARD14, Encoding an Epidermal Regulator of NF-kappaB, in Psoriasis. Am. J. Hum. Genet. 2012, 90, 796–808. [Google Scholar] [CrossRef]
- Mellett, M.; Meier-Schiesser, B.; Mohanan, D.; Schairer, R.; Cheng, P.; Satoh, T.; Kiefer, B.; Ospelt, C.; Nobbe, S.; Thome, M.; et al. CARD14 Gain-of-Function Mutation Alone Is Sufficient to Drive IL-23/IL-17–Mediated Psoriasiform Skin Inflammation In Vivo. J. Investig. Dermatol. 2018, 138, 2010–2023. [Google Scholar] [CrossRef]
- Zhu, H.; Lou, F.; Yin, Q.; Gao, Y.; Sun, Y.; Bai, J.; Xu, Z.; Liu, Z.; Cai, W.; Ke, F.; et al. RIG-I antiviral signaling drives interleukin-23 production and psoriasis-like skin disease. EMBO Mol. Med. 2017, 9, 589–604. [Google Scholar] [CrossRef]
- Rebholz, B.; Haase, I.; Eckelt, B.; Paxian, S.; Flaig, M.J.; Ghoreschi, K.; Nedospasov, S.A.; Mailhammer, R.; Debey-Pascher, S.; Schultze, J.L.; et al. Crosstalk between Keratinocytes and Adaptive Immune Cells in an IκBα Protein-Mediated Inflammatory Disease of the Skin. Immunity 2007, 27, 296–307. [Google Scholar] [CrossRef]
- Moos, S.; Mohebiany, A.N.; Waisman, A.; Kurschus, F.C. Imiquimod-Induced Psoriasis in Mice Depends on the IL-17 Signaling of Keratinocytes. J. Investig. Dermatol. 2019, 139, 1110–1117. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Lu, J.; Bao, X.; Yi, X.; Peng, C.; Chen, W.; Zhen, T.; Shi, Y.; Xing, K.; Zhu, S.; et al. Inhibition of phospholipases suppresses progression of psoriasis through modulation of inflammation. Exp. Biol. Med. 2021, 246, 1253–1262. [Google Scholar] [CrossRef]
- Yang, L.; Zhang, T.; Zhang, C.; Xiao, C.; Bai, X.; Wang, G. Upregulated E3 ligase tripartite motif-containing protein 21 in psoriatic epidermis ubiquitylates nuclear factor-κB p65 subunit and promotes inflammation in keratinocytes. Br. J. Dermatol. 2020, 184, 111–122. [Google Scholar] [CrossRef]
- Qu, S.; Liu, Z.; Wang, B. EZH2 is involved in psoriasis progression by impairing miR-125a-5p inhibition of SFMBT1 and leading to inhibition of the TGFβ/SMAD pathway. Ther. Adv. Chronic Dis. 2021, 12, 2040622320987348. [Google Scholar] [CrossRef]
- Shi, Z.; Wu, X.; Yu, S.; Huynh, M.; Jena, P.K.; Nguyen, M.; Wan, Y.-J.Y.; Hwang, S.T. Short-Term Exposure to a Western Diet Induces Psoriasiform Dermatitis by Promoting Accumulation of IL-17A–Producing γδ T Cells. J. Investig. Dermatol. 2020, 140, 1815–1823. [Google Scholar] [CrossRef]
- Le, S.T.; Toussi, A.; Maverakis, N.; Marusina, A.I.; Barton, V.R.; Merleev, A.A.; Luxardi, G.; Raychaudhuri, S.P.; Maverakis, E. The cutaneous and intestinal microbiome in psoriatic disease. Clin. Immunol. 2020, 218, 108537. [Google Scholar] [CrossRef]
- Szántó, M.; Dózsa, A.; Antal, D.; Szabó, K.; Kemény, L.; Bai, P. Targeting the gut-skin axis—Probiotics as new tools for skin disorder management? Exp. Dermatol. 2019, 28, 1210–1218. [Google Scholar] [CrossRef]
- Zheng, D.; Liwinski, T.; Elinav, E. Interaction between microbiota and immunity in health and disease. Cell Res. 2020, 30, 492–506. [Google Scholar] [CrossRef]
- Toor, D.; Wsson, M.K.; Kumar, P.; Karthikeyan, G.; Kaushik, N.K.; Goel, C.; Singh, S.; Kumar, A.; Prakash, H. Dysbiosis disrupts gut immune homeostasis and promotes gastric diseases. Int. J. Mol. Sci. 2019, 20, 2432. [Google Scholar] [CrossRef]
- Nishida, A.; Inoue, R.; Inatomi, O.; Bamba, S.; Naito, Y.; Andoh, A. Gut microbiota in the pathogenesis of inflammatory bowel disease. Clin. J. Gastroenterol. 2018, 11, 1–10. [Google Scholar] [CrossRef]
- Korman, N.J. Management of psoriasis as a systemic disease: What is the evidence? Br. J. Dermatol. 2020, 182, 840–848. [Google Scholar] [CrossRef]
- Surcel, M.; Munteanu, A.; Isvoranu, G.; Ibram, A.; Caruntu, C.; Constantin, C.; Neagu, M. Unconventional Therapy with IgY in a Psoriatic Mouse Model Targeting Gut Microbiome. J. Pers. Med. 2021, 11, 841. [Google Scholar] [CrossRef]
- Kim, H.-N.; Han, K.; Park, Y.-G.; Lee, J.H. Metabolic syndrome is associated with an increased risk of psoriasis: A nationwide population-based study. Metabolism 2019, 99, 19–24. [Google Scholar] [CrossRef]
- Alinaghi, F.; Tekin, H.G.; Burisch, J.; Wu, J.J.; Thyssen, J.P.; Egeberg, A. Global prevalence and bidirectional association between psoriasis and inflammatory bowel disease—A systematic review and meta-analysis. J. Crohns Colitis 2020, 14, 351–360. [Google Scholar] [CrossRef]
- O’Neill, C.A.; Monteleone, G.; McLaughlin, J.T.; Paus, R. The gut-skin axis in health and disease: A paradigm with therapeutic implications. Bioessays 2016, 38, 1167–1176. [Google Scholar] [CrossRef]
- Ramírez-Boscá, A.; Navarro-López, V.; Martínez-Andrés, A.; Such, J.; Francés, R.; de la Parte, J.H.; Asín-Llorca, M. Identification of bacterial DNA in the peripheral blood of patients with active psoriasis. JAMA Dermatol. 2015, 151, 670–671. [Google Scholar] [CrossRef]
- Sacchetti, L.; Nardelli, C. Gut microbiome investigation in celiac disease: From methods to its pathogenetic role. Clin. Chem. Lab. Med. 2020, 58, 340–349. [Google Scholar] [CrossRef]
- Ellis, S.R.; Nguyen, M.; Vaughn, A.R.; Notay, M.; Burney, W.A.; Sandhu, S.; Sivamani, R.K. The skin and gut microbiome and its role in common dermatologic conditions. Microorganisms 2019, 7, 550. [Google Scholar] [CrossRef]
- Mowat, A.M.; Agace, W.W. Regional specialization within the intestinal immune system. Nat. Rev. Immunol. 2014, 14, 667–685. [Google Scholar] [CrossRef]
- Bäckhed, F.; Roswall, J.; Peng, Y.; Feng, Q.; Jia, H.; Kovatcheva-Datchary, P.; Li, Y.; Xia, Y.; Xie, H.; Zhong, H.; et al. Dynamics and Stabilization of the Human Gut Microbiome during the First Year of Life. Cell Host Microbe 2015, 17, 690–703. [Google Scholar] [CrossRef]
- McKenzie, C.; Tan, J.; Macia, L.; Mackay, C.R. The nutrition-gut microbiome-physiology axis and allergic diseases. Immunol. Rev. 2017, 278, 277–295. [Google Scholar] [CrossRef]
- Lee, J.K.-F.; Hern Tan, L.T.; Ramadas, A.; Ab Mutalib, N.-S.; Lee, L.-H. Exploring the Role of Gut Bacteria in Health and Disease in Preterm Neonates. Int. J. Environ. Res. Public Health 2020, 17, 6963. [Google Scholar] [CrossRef]
- Lau, A.W.Y.; Tan, L.T.-H.; Ab Mutalib, N.-S.; Wong, S.H.; Letchumanan, V.; Lee, L.-H. The chemistry of gut microbiome in health and diseases. Prog. Microbes Mol. Biol. 2021, 4. [Google Scholar] [CrossRef]
- Stevens, B.R.; Roesch, L.; Thiago, P.; Russell, J.T.; Pepine, C.J.; Holbert, R.C.; Raizada, M.K.; Triplett, E.W. Depression phenotype identified by using single nucleotide exact amplicon sequence variants of the human gut microbiome. Mol. Psychiatry 2021, 26, 4277–4287. [Google Scholar] [CrossRef]
- Vallianou, N.; Stratigou, T.; Christodoulatos, G.S.; Dalamaga, M. Understanding the role of the gut microbiome and microbial metabolites in obesity and obesity-associated metabolic disorders: Current evidence and perspectives. Curr. Obes. Rep. 2019, 8, 317–332. [Google Scholar] [CrossRef]
- O’neill, A.M.; Gallo, R.L. Host-microbiome interactions and recent progress into understanding the biology of acne vulgaris. Microbiome 2018, 6, 177. [Google Scholar] [CrossRef]
- Gloster, H.; Gebauer, L.; Mistur, R. Cutaneous manifestations of gastrointestinal disease. In Absolute Dermatology Review; Springer: Cham, Switzerland, 2016; pp. 171–179. [Google Scholar] [CrossRef]
- Kim, H.-J.; Lee, S.-H.; Hong, S.-J. Antibiotics-Induced Dysbiosis of Intestinal Microbiota Aggravates Atopic Dermatitis in Mice by Altered Short-Chain Fatty Acids. Allergy, Asthma Immunol. Res. 2020, 12, 137–148. [Google Scholar] [CrossRef]
- Polkowska-Pruszyńska, B.; Gerkowicz, A.; Krasowska, D. The gut microbiome alterations in allergic and inflammatory skin diseases—An update. J. Eur. Acad. Dermatol. Venereol. 2020, 34, 455–464. [Google Scholar] [CrossRef]
- Sirisinha, S. The potential impact of gut microbiota on your health:Current status and future challenges. Asian Pac. J. Allergy Immunol. 2016, 34, 249–264. [Google Scholar] [CrossRef] [PubMed]
- Samuelson, D.R.; Welsh, D.A.; Shellito, J.E. Regulation of lung immunity and host defense by the intestinal microbiota. Front. Microbiol. 2015, 6, 1085. [Google Scholar] [CrossRef]
- Miyazaki, K.; Masuoka, N.; Kano, M.; Iizuka, R. Bifidobacterium fermented milk and galacto-oligosaccharides lead to improved skin health by decreasing phenols production by gut microbiota. Benef. Microbes 2014, 5, 121–128. [Google Scholar] [CrossRef]
- Spencer, S.P.; Fragiadakis, G.K.; Sonnenburg, J.L. Pursuing Human-Relevant Gut Microbiota-Immune Interactions. Immunity 2019, 51, 225–239. [Google Scholar] [CrossRef]
- Kosiewicz, M.M.; Dryden, G.W.; Chhabra, A.; Alard, P. Relationship between gut microbiota and development of T cell associated disease. FEBS Lett. 2014, 588, 4195–4206. [Google Scholar] [CrossRef]
- Scher, J.U.; Littman, D.R.; Abramson, S.B. Microbiome in Inflammatory Arthritis and Human Rheumatic Diseases. Arthritis Rheumatol. 2016, 68, 35–45. [Google Scholar] [CrossRef]
- Georgescu, S.-R.; Tampa, M.; Caruntu, C.; Sarbu, M.-I.; Mitran, C.-I.; Mitran, M.-I.; Matei, C.; Constantin, C.; Neagu, M. Advances in Understanding the Immunological Pathways in Psoriasis. Int. J. Mol. Sci. 2019, 20, 739. [Google Scholar] [CrossRef]
- Robertson, S.J.; Zhou, J.Y.; Geddes, K.; Rubino, S.J.; Cho, J.H.; Girardin, S.E.; Philpott, D.J. Nod1 and Nod2 signaling does not alter the composition of intestinal bacterial communities at homeostasis. Gut Microbes 2013, 4, 222–231. [Google Scholar] [CrossRef]
- Clevers, H.C.; Bevins, C.L. Paneth Cells: Maestros of the Small Intestinal Crypts. Annu. Rev. Physiol. 2013, 75, 289–311. [Google Scholar] [CrossRef]
- Hugot, J.-P.; Chamaillard, M.; Zouali, H.; Lesage, S.; Cézard, J.-P.; Belaiche, J.; Almer, S.; Tysk, C.; O’Morain, C.A.; Gassull, M.; et al. Association of NOD2 leucine-rich repeat variants with susceptibility to Crohn’s disease. Nature 2001, 411, 599–603. [Google Scholar] [CrossRef]
- Ogura, Y.; Bonen, D.K.; Inohara, N.; Nicolae, D.L.; Chen, F.F.; Ramos, R.; Britton, H.; Moran, T.; Karaliuskas, R.; Duerr, R.H.; et al. A frameshift mutation in NOD2 associated with susceptibility to Crohn’s disease. Nature 2001, 411, 603–606. [Google Scholar] [CrossRef]
- Suzuki, K.; Kawamoto, S.; Maruya, M.; Fagarasan, S. GALT: Organization and dynamics leading to IgA synthesis. Adv. Immunol. 2010, 107, 153–185. [Google Scholar] [CrossRef]
- Adarsh, M.B.; Dogra, S.; Vaiphei, K.; Vaishnavi, C.; Sinha, S.; Sharma, A. Evaluation of subclinical gut inflammation using faecal calprotectin levels and colonic mucosal biopsy in patients with psoriasis and psoriatic arthritis. Br. J. Dermatol. 2019, 181, 401–402. [Google Scholar] [CrossRef]
- Bassukas, I.D.; Gaitanis, G.; Katsanos, K.H.; Christodoulou, D.K.; Tsianos, E.; Vlachos, C. Psoriasis and inflammatory bowel disease: Links and risks. Psoriasis: Targets Ther. 2016, 6, 73–92. [Google Scholar] [CrossRef]
- Visser, M.J.E.; Kell, D.B.; Pretorius, E. Bacterial Dysbiosis and Translocation in Psoriasis Vulgaris. Front. Cell. Infect. Microbiol. 2019, 9, 7. [Google Scholar] [CrossRef]
- Mahmud, R.; Akter, S.; Tamanna, S.K.; Mazumder, L.; Esti, I.Z.; Banerjee, S.; Akter, S.; Hasan, R.; Acharjee, M.; Hossain, S.; et al. Impact of gut microbiome on skin health: Gut-Skin axis observed through the lenses of therapeutics and skin diseases. Gut Microbes 2022, 14, 2096995. [Google Scholar] [CrossRef]
- Chung, M.; Bartholomew, E.; Yeroushalmi, S.; Hakimi, M.; Bhutani, T.; Liao, W. Dietary Intervention and Supplements in the Management of Psoriasis: Current Perspectives. Psoriasis 2022, 12, 151–176. [Google Scholar] [CrossRef]
- Pona, A.; Haidari, W.; Kolli, S.S.; Feldman, S.R. Diet and psoriasis. Dermatol. Online J. 2019, 25. [Google Scholar] [CrossRef]
- Gelfand, J.M.; Abuabara, K. Diet and Weight Loss as a Treatment for Psoriasis. Arch. Dermatol. 2010, 146, 544–546. [Google Scholar] [CrossRef][Green Version]
- Aronson, P.J. Cases of psoriasis improved by lowering homocysteine using 4-7 mg folic acid, vitamins B6 and B12 previously worsened using 1-2 mg daily folic acid, B6 and B12 folic acid. J. Transl. Sci. 2017, 3, 1–6. [Google Scholar] [CrossRef]
- Balta, S.; Balta, I.; Mikhailidis, D.P.; Ozturk, C.; Demirkol, S.; Celik, T.; Kilic, S.; Demir, M.; Iyisoy, A. Bilirubin levels and their association with carotid intima media thickness and high-sensitivity Creactive protein in patients with psoriasis vulgaris. Am. J. Clin. Dermatol. 2014, 15, 137–142. [Google Scholar] [CrossRef]
- Evers, A.; Verhoeven, E.; Kraaimaat, F.; De Jong, E.; De Brouwer, S.; Schalkwijk, J.; Sweep, F.; Van De Kerkhof, P. How stress gets under the skin: Cortisol and stress reactivity in psoriasis. Br. J. Dermatol. 2010, 163, 986–991. [Google Scholar] [CrossRef]
- Jensen, P.; Zachariae, C.; Christensen, R.; Geiker, N.R.; Schaadt, B.K.; Stender, S.; Astrup, A.; Hansen, P.R.; Skov, L. Effect of weight loss on the cardiovascular risk profile of obese patients with psoriasis. Acta Derm Venereol. 2014, 94, 691–694. [Google Scholar] [CrossRef]
- Korovesi, A.; Dalamaga, M.; Kotopouli, M.; Papadavid, E. Adherence to the Mediterranean diet is independently associated with psoriasis risk, severity, and quality of life: A cross-sectional observational study. Int J Dermatol. 2019, 58, e164–e165. [Google Scholar] [CrossRef]
- Passali, M.; Josefsen, K.; Frederiksen, J.L.; Antvorskov, J.C. Current evidence on the efficacy of gluten-free diets in multiple sclerosis, psoriasis, type 1 diabetes and autoimmune thyroid diseases. Nutrients 2020, 12, 2316. [Google Scholar] [CrossRef]
- Drucker, A.M.; Qureshi, A.A.; Thompson, J.M.; Li, T.; Cho, E. Gluten intake and risk of psoriasis, psoriatic arthritis, and atopic dermatitis among United States women. J. Am. Acad. Dermatol. 2020, 82, 661–665. [Google Scholar] [CrossRef]
- Bhatia, B.K.; Millsop, J.W.; Debbaneh, M.; Koo, J.; Linos, E.; Liao, W. Diet and psoriasis, part II: Celiac disease and role of a gluten-free diet. J. Am. Acad. Dermatol. 2014, 71, 350–358. [Google Scholar] [CrossRef]
- Michaëlsson, G.; Gerdén, B.; Hagforsen, E.; Nilsson, B.; Pihl-Lundin, I.; Kraaz, W.; Hjelmquist, G.; Lööf, L. Psoriasis patients with antibodies to gliadin can be improved by a gluten-free diet. Br. J. Dermatol. 2000, 142, 44–51. [Google Scholar] [CrossRef]
- Michaëlsson, G.; Ahs, S.; Hammarström, I.; Lundin, I.P.; Hagforsen, E. Gluten-free diet in psoriasis patients with antibodies to gliadin results in decreased expression of tissue transglutaminase and fewer Ki67 + cells in the dermis. Acta Derm Venereol. 2003, 83, 425–429. [Google Scholar] [CrossRef]
- Michaëlsson, G.; Kristjánsson, G.; Lundin, I.P.; Hagforsen, E. Palmoplantar pustulosis and gluten sensitivity: A study of serum antibodies against gliadin and tissue transglutaminase, the duodenal mucosa and effects of gluten-free diet. Br. J. Dermatol. 2007, 156, 659–666. [Google Scholar] [CrossRef]
- Kolchak, N.A.; Tetarnikova, M.K.; Theodoropoulou, M.S.; Michalopoulou, A.P.; Theodoropoulos, D.S. Prevalence of antigliadin IgA antibodies in psoriasis vulgaris and response of seropositive patients to a gluten-free diet. J. Multidiscip. Healthc. 2018, 11, 13–19. [Google Scholar] [CrossRef]
- Millsop, J.W.; Bhatia, B.K.; Debbaneh, M.; Koo, J.; Liao, W. Diet and psoriasis, part III: Role of nutritional supplements. J. Am. Acad. Dermatol. 2014, 71, 561–569. [Google Scholar] [CrossRef]
- Hahn, J.; Cook, N.R.; Alexander, E.K.; Friedman, S.; Walter, J.; Bubes, V.; Kotler, G.; Lee, I.M.; Manson, J.E.; Costenbader, K.H. Vitamin D and marine omega 3 fatty acid supplementation and incident autoimmune disease: VITAL randomized controlled trial. BMJ 2022, 376, e066452. [Google Scholar] [CrossRef]
- Luo, Y.; Chen, J.; Kuai, L.; Zhang, Y.; Ding, X.; Luo, Y.; Ru, Y.; Xing, M.; Li, H.; Sun, X.; et al. Chinese Herbal Medicine for Psoriasis: Evidence From 11 High-Quality Randomized Controlled Trials. Front. Pharmacol. 2021, 11, 599433. [Google Scholar] [CrossRef]
- Lv, M.; Deng, J.; Tang, N.; Zeng, Y.; Lu, C. Efficacy and safety of Tripterygium wilfordii Hook F on psoriasis vulgaris: A systematic review and meta-analysis of randomized controlled trials. Evid. Based Complement. Altern. Med. 2018, 2018, 2623085. [Google Scholar] [CrossRef]
- Stern, R.S.; Nijsten, T.; Feldman, S.R.; Margolis, D.J.; Rolstad, T. Psoriasis Is Common, Carries a Substantial Burden Even When Not Extensive, and Is Associated with Widespread Treatment Dissatisfaction. J. Investig. Dermatol. Symp. Proc. 2004, 9, 136–139. [Google Scholar] [CrossRef]
- Gelfand, J.M.; Feldman, S.R.; Stern, R.S.; Thomas, J.; Rolstad, T.; Margolis, D.J. Determinants of quality of life in patients with psoriasis: A study from the US population. J. Am. Acad. Dermatol. 2004, 51, 704–708. [Google Scholar] [CrossRef]
- Armstrong, A.W.; Harskamp, C.T.; Armstrong, E.J. Psoriasis and metabolic syndrome: A systematic review and meta-analysis of observational studies. J. Am. Acad. Dermatol. 2013, 68, 654–662. [Google Scholar] [CrossRef]
- Armstrong, A.W. Psoriasis. JAMA Dermatol. 2017, 153, 956. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Constantin, C.; Surcel, M.; Munteanu, A.; Neagu, M. Insights into Nutritional Strategies in Psoriasis. Nutrients 2023, 15, 3528. https://doi.org/10.3390/nu15163528
Constantin C, Surcel M, Munteanu A, Neagu M. Insights into Nutritional Strategies in Psoriasis. Nutrients. 2023; 15(16):3528. https://doi.org/10.3390/nu15163528
Chicago/Turabian StyleConstantin, Carolina, Mihaela Surcel, Adriana Munteanu, and Monica Neagu. 2023. "Insights into Nutritional Strategies in Psoriasis" Nutrients 15, no. 16: 3528. https://doi.org/10.3390/nu15163528
APA StyleConstantin, C., Surcel, M., Munteanu, A., & Neagu, M. (2023). Insights into Nutritional Strategies in Psoriasis. Nutrients, 15(16), 3528. https://doi.org/10.3390/nu15163528







