The Effect of Vitamin D Supplementation on the Length of Hospitalisation, Intensive Care Unit Admission, and Mortality in COVID-19—A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Eligibility Criteria
- Population: adult and paediatric population with COVID-19;
- Intervention: high dose of vitamin D supplementation after a SARS-CoV-2 positive test;
- Comparison: control group without vitamin D supplementation or low dose of vitamin D supplementation vs. high dose;
- Outcome: the outcomes are the effects on mortality related to COVID-19, the length of hospital stay (LOS), and the admission to the ICU;
- Study design: randomized controlled trials were included.
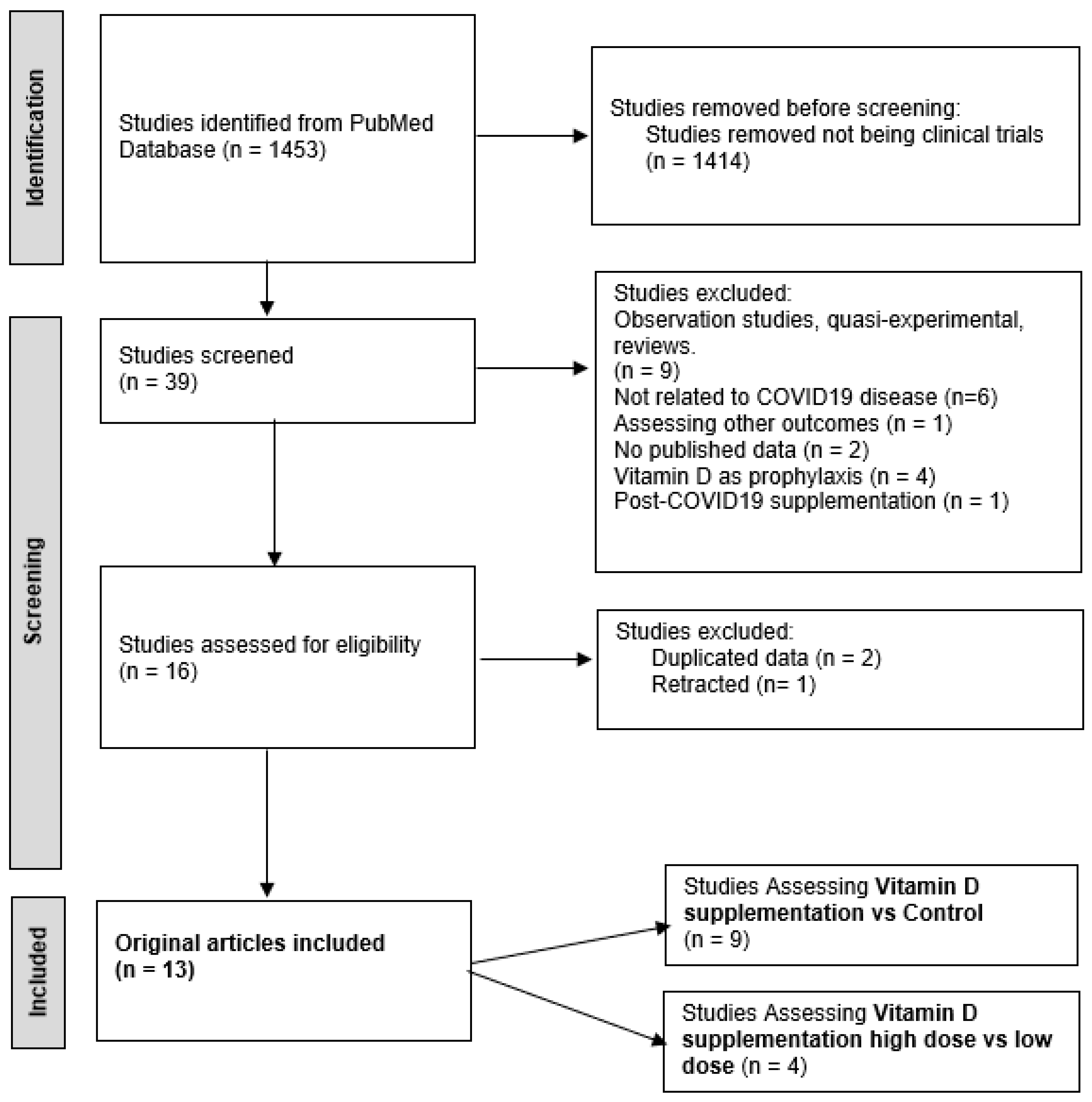
2.3. Study Screening and Selection
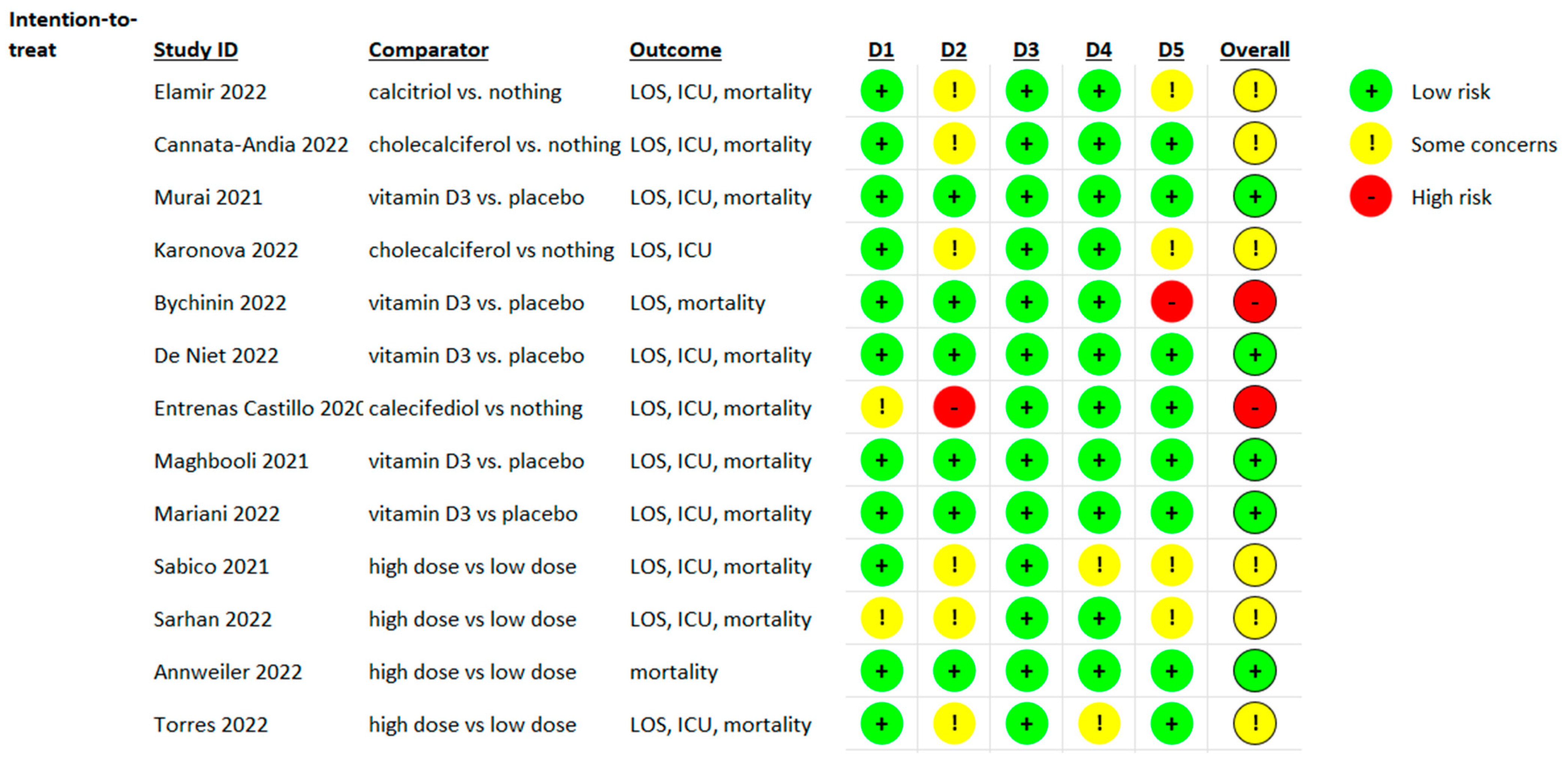
2.4. Data Extraction and Quality Assessment
2.5. Statistical Analysis
3. Results
3.1. Vitamin D Supplementation vs. Control
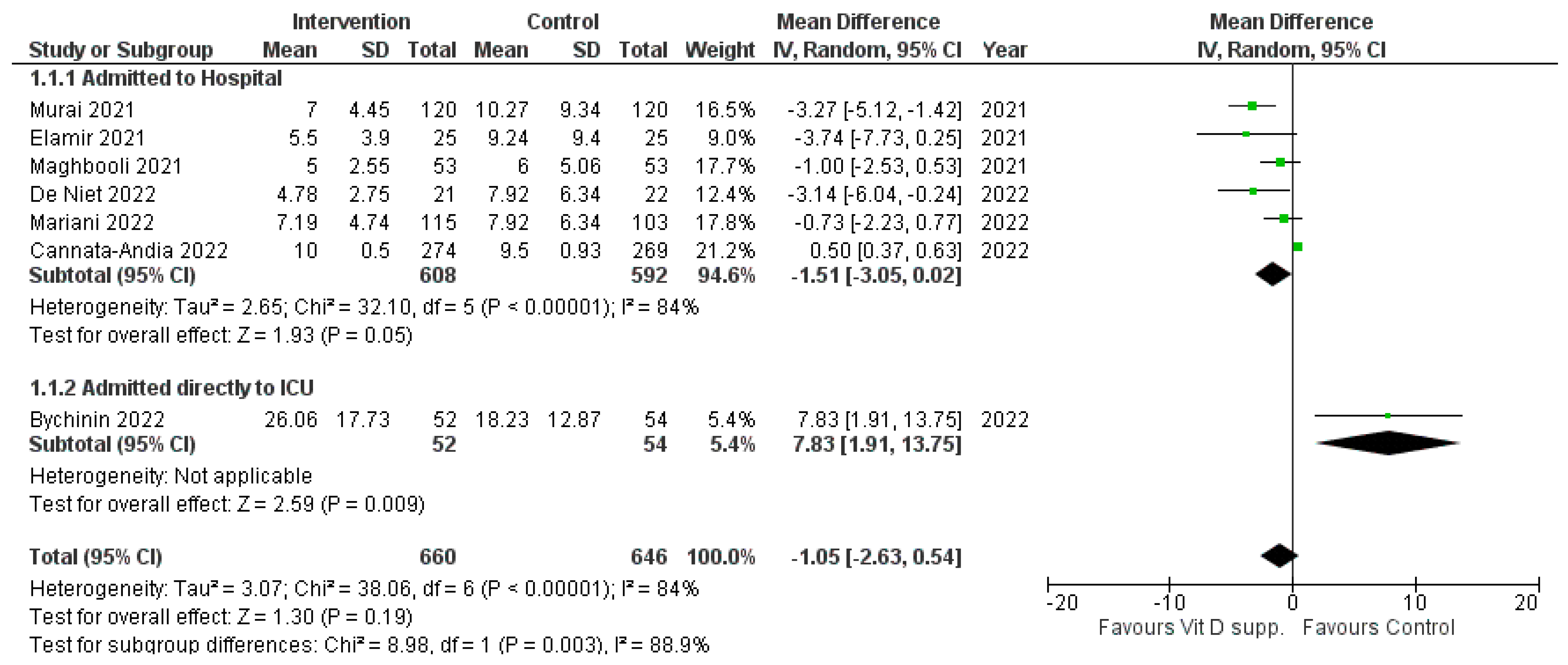
3.1.1. Length of Hospital Stay
3.1.2. Admission to the ICU
3.1.3. Mortality
3.2. Vitamin D Supplementation: High Dose vs. Low Dose
3.3. Risk of Bias Evaluation
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Coronavirus Disease (COVID-19) Pandemic; World Health Organization: Geneva, Switzerland, 2020; Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019 (accessed on 27 March 2022).
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D Deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.B.; Lahore, H.; McDonnell, S.L.; Baggerly, C.A.; French, C.B.; Aliano, J.L.; Bhattoa, H.P. Evidence that Vitamin D Supplementation Could Reduce Risk of Influenza and COVID-19 Infections and Deaths. Nutrients 2020, 12, 988. [Google Scholar] [CrossRef] [PubMed]
- Cashman, K.D.; Dowling, K.G.; Škrabáková, Z.; Gonzalez-Gross, M.; Valtueña, J.; De Henauw, S.; Moreno, L.; Damsgaard, C.T.; Michaelsen, K.F.; Mølgaard, C.; et al. Vitamin D deficiency in Europe: Pandemic? Am. J. Clin. Nutr. 2016, 103, 1033–1044. [Google Scholar] [CrossRef]
- Amrein, K.; Scherkl, M.; Hoffmann, M.; Neuwersch-Sommeregger, S.; Köstenberger, M.; Berisha, A.T.; Martucci, G.; Pilz, S.; Malle, O. Vitamin D deficiency 2.0: An update on the current status worldwide. Eur. J. Clin. Nutr. 2020, 74, 1498–1513. [Google Scholar] [CrossRef]
- Sutherland, J.P.; Zhou, A.; Hyppönen, E. Vitamin D Deficiency Increases Mortality Risk in the UK Biobank: A Nonlinear Mendelian Randomization Study. Ann. Intern. Med. 2022, 175, 1552–1559. [Google Scholar] [CrossRef]
- Gunville, C.F.; Mourani, P.M.; Ginde, A.A. The Role of Vitamin D in Prevention and Treatment of Infection. Inflamm. Allergy-Drug Targets 2013, 12, 239–245. [Google Scholar] [CrossRef]
- Feketea, G.; Vlacha, V.; Bocsan, I.C.; Vassilopoulou, E.; Stanciu, L.A.; Zdrenghea, M. Vitamin D in Corona Virus Disease 2019 (COVID-19) Related Multisystem Inflammatory Syndrome in Children (MIS-C). Front. Immunol. 2021, 12, 648546. [Google Scholar] [CrossRef]
- Dror, A.A.; Morozov, N.; Daoud, A.; Namir, Y.; Yakir, O.; Shachar, Y.; Lifshitz, M.; Segal, E.; Fisher, L.; Mizrachi, M.; et al. Pre-infection 25-hydroxyvitamin D3 levels and association with severity of COVID-19 illness. PLoS ONE 2022, 17, e0263069. [Google Scholar] [CrossRef]
- Chetty, V.V.; Chetty, M. Potential benefit of vitamin D supplementation in people with respiratory illnesses, during the COVID-19 pandemic. Clin. Transl. Sci. 2021, 14, 2111–2116. [Google Scholar] [CrossRef]
- Li, B.; Yang, S.; Hou, N. Could vitamin D supplementation play a role against COVID-19? Front. Immunol. 2022, 13, 967215. [Google Scholar] [CrossRef] [PubMed]
- BPetrelli, F.; Luciani, A.; Perego, G.; Dognini, G.; Colombelli, P.L.; Ghidini, A. Therapeutic and prognostic role of vitamin D for COVID-19 infection: A systematic review and meta-analysis of 43 observational studies. J. Steroid Biochem. Mol. Biol. 2021, 211, 105883. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef] [PubMed]
- McGrath, S.; Zhao, X.; Steele, R.; Thombs, B.D.; Benedetti, A.; Levis, B.; Riehm, K.E.; Saadat, N.; Levis, A.W.; Azar, M.; et al. Estimating the sample mean and standard deviation from commonly reported quantiles in meta-analysis. Stat. Methods Med. Res. 2020, 29, 2520–2537. [Google Scholar] [CrossRef] [PubMed]
- Bychinin, M.V.; Klypa, T.V.; Mandel, I.A.; Yusubalieva, G.M.; Baklaushev, V.P.; Kolyshkina, N.A.; Troitsky, A.V. Effect of vitamin D3 supplementation on cellular immunity and inflammatory markers in COVID-19 patients admitted to the ICU. Sci. Rep. 2022, 12, 18604. [Google Scholar] [CrossRef] [PubMed]
- Cannata-Andía, J.B.; Díaz-Sottolano, A.; Fernández, P.; Palomo-Antequera, C.; Herrero-Puente, P.; Mouzo, R.; Carrillo-López, N.; Panizo, S.; Ibañez, G.H.; Cusumano, C.A.; et al. A single-oral bolus of 100,000 IU of cholecalciferol at hospital admission did not improve outcomes in the COVID-19 disease: The COVID-VIT-D—A randomised multicentre international clinical trial. BMC Med. 2022, 20, 83. [Google Scholar] [CrossRef]
- De Niet, S.; Trémège, M.; Coffiner, M.; Rousseau, A.-F.; Calmes, D.; Frix, A.-N.; Gester, F.; Delvaux, M.; Dive, A.-F.; Guglielmi, E.; et al. Positive Effects of Vitamin D Supplementation in Patients Hospitalized for COVID-19: A Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients 2022, 14, 3048. [Google Scholar] [CrossRef]
- Elamir, Y.M.; Amir, H.; Lim, S.; Rana, Y.P.; Lopez, C.G.; Feliciano, N.V.; Omar, A.; Grist, W.P.; Via, M.A. A randomized pilot study using calcitriol in hospitalized COVID-19 patients. Bone 2022, 154, 116175. [Google Scholar] [CrossRef]
- Entrenas Castillo, M.E.; Entrenas Costa, L.M.E.; Vaquero Barrios, J.M.V.; Alcalá Díaz, J.F.A.; López Miranda, J.L.; Bouillon, R.; Quesada Gomez, J.M.Q. Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: A pilot randomized clinical study. J. Steroid Biochem. Mol. Biol. 2020, 203, 105751. [Google Scholar] [CrossRef]
- Karonova, T.L.; Golovatyuk, K.A.; Kudryavtsev, I.V.; Chernikova, A.T.; Mikhaylova, A.A.; Aquino, A.D.; Lagutina, D.I.; Zaikova, E.K.; Kalinina, O.V.; Golovkin, A.S.; et al. Effect of Cholecalciferol Supplementation on the Clinical Features and Inflammatory Markers in Hospitalized COVID-19 Patients: A Randomized, Open-Label, Single-Center Study. Nutrients 2022, 14, 2602. [Google Scholar] [CrossRef]
- Maghbooli, Z.; Sahraian, M.A.; Jamalimoghadamsiahkali, S.; Asadi, A.; Zarei, A.; Zendehdel, A.; Varzandi, T.; Mohammadnabi, S.; Alijani, N.; Karimi, M.; et al. Treatment With 25-Hydroxyvitamin D3 (Calcifediol) Is Associated with a Reduction in the Blood Neutrophil-to-Lymphocyte Ratio Marker of Disease Severity in Hospitalized Patients With COVID-19: A Pilot Multicenter, Randomized, Placebo-Controlled, Double-Blinded Clinical Trial. Endocr. Pract. 2021, 27, 1242–1251. [Google Scholar] [CrossRef]
- Mariani, J.; Antonietti, L.; Tajer, C.; Ferder, L.; Inserra, F.; Cunto, M.S.; Brosio, D.; Ross, F.; Zylberman, M.; López, D.E.; et al. High-dose vitamin D versus placebo to prevent complications in COVID-19 patients: Multicentre randomized controlled clinical trial. PLoS ONE 2022, 17, e0267918. [Google Scholar] [CrossRef] [PubMed]
- Murai, I.H.; Fernandes, A.L.; Sales, L.P.; Pinto, A.J.; Goessler, K.F.; Duran, C.S.C.; Silva, C.B.R.; Franco, A.S.; Macedo, M.B.; Dalmolin, H.H.H.; et al. Effect of a Single High Dose of Vitamin D3 on Hospital Length of Stay in Patients with Moderate to Severe COVID-19: A Randomized Clinical Trial. JAMA 2021, 325, 1053–1060. [Google Scholar] [CrossRef] [PubMed]
- Annweiler, C.; Beaudenon, M.; Gautier, J.; Gonsard, J.; Boucher, S.; Chapelet, G.; Darsonval, A.; Fougère, B.; Guérin, O.; Houvet, M.; et al. High-dose versus standard-dose vitamin D supplementation in older adults with COVID-19 (COVIT-TRIAL): A multicenter, open-label, randomized controlled superiority trial. PLoS Med. 2022, 19, e1003999. [Google Scholar] [CrossRef] [PubMed]
- Sabico, S.; Enani, M.A.; Sheshah, E.; Aljohani, N.J.; Aldisi, D.A.; Alotaibi, N.H.; Alshingetti, N.; Alomar, S.Y.; Alnaami, A.M.; Amer, O.E.; et al. Effects of a 2-Week 5000 IU versus 1000 IU Vitamin D3 Supplementation on Recovery of Symptoms in Patients with Mild to Moderate Covid-19: A Randomized Clinical Trial. Nutrients 2021, 13, 2170. [Google Scholar] [CrossRef] [PubMed]
- Sarhan, N.; Warda, A.E.A.; Sarhan, R.M.; Boshra, M.S.; Mostafa-Hedeab, G.; Alruwaili, B.F.; Ibrahim, H.S.G.; Schaalan, M.F.; Fathy, S. Evidence for the Efficacy of a High Dose of Vitamin D on the Hyperinflammation State in Moderate-to-Severe COVID-19 Patients: A Randomized Clinical Trial. Medicina 2022, 58, 1358. [Google Scholar] [CrossRef]
- Torres, M.; Casado, G.; Vigón, L.; Rodríguez-Mora, S.; Mateos, E.; Ramos-Martín, F.; López-Wolf, D.; Sanz-Moreno, J.; Ryan-Murua, P.; Taboada-Martínez, M.L.; et al. Changes in the immune response against SARS-CoV-2 in individuals with severe COVID-19 treated with high dose of vitamin D. Biomed. Pharmacother. 2022, 150, 112965. [Google Scholar] [CrossRef]
- Yang, H.; Sun, D.; Wu, F.; Xu, X.; Liu, X.; Wang, Z.; Zhou, L. Effects of Vitamin D on Respiratory Function and Immune Status for Patients with Chronic Obstructive Pulmonary Disease (COPD): A Systematic Review and Meta-Analysis. Comput. Math. Methods Med. 2022, 2022, 2910782. [Google Scholar] [CrossRef]
- Zhang, Y.; Fang, F.; Tang, J.; Jia, L.; Feng, Y.; Xu, P.; Faramand, A. Association between vitamin D supplementation and mortality: Systematic review and meta-analysis. BMJ 2019, 366, l4673, Erratum in BMJ 2020, 370, m2329. [Google Scholar]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef]
- Jolliffe, D.; Camargo, C.A., Jr.; Sluyter, J.D.; Aglipay, M.; Aloia, J.F.; Ganmaa, D.; Bergman, P.; A Bischoff-Ferrari, H.; Borzutzky, A.; Damsgaard, C.T.; et al. Vitamin D supplementation to prevent acute respiratory infections: A systematic review and meta-analysis of aggregate data from randomised controlled trials. Lancet Diabetes Endocrinol. 2021, 9, 276–292. [Google Scholar] [CrossRef] [PubMed]
- Menshawey, E.; Menshawey, R.; Nabeh, O.A. Shedding light on vitamin D: The shared mechanistic and pathophysiological role between hypovitaminosis D and COVID-19 risk factors and complications. Inflammopharmacology 2021, 29, 1017–1031. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, B.; El Abd, A.; Ducharme, F.M. Effects of Vitamin D Supplementation on COVID-19 Related Outcomes: A Systematic Review and Meta-Analysis. Nutrients 2022, 14, 2134. [Google Scholar] [CrossRef] [PubMed]
- Bassatne, A.; Basbous, M.; Chakhtoura, M.; El Zein, O.; Rahme, M.; Fuleihan, G.E.-H. The link between COVID-19 and VItamin D (VIVID): A systematic review and meta-analysis. Metabolism 2021, 119, 154753. [Google Scholar] [CrossRef]
- D’ecclesiis, O.; Gavioli, C.; Martinoli, C.; Raimondi, S.; Chiocca, S.; Miccolo, C.; Bossi, P.; Cortinovis, D.; Chiaradonna, F.; Palorini, R.; et al. Vitamin D and SARS-CoV2 infection, severity and mortality: A systematic review and meta-analysis. PLoS ONE 2022, 17, e0268396. [Google Scholar] [CrossRef]
- Varikasuvu, S.R.; Thangappazham, B.; Vykunta, A.; Duggina, P.; Manne, M.; Raj, H.; Aloori, S. COVID-19 and vitamin D (Co-VIVID study): A systematic review and meta-analysis of randomized controlled trials. Expert Rev. Anti-Infective Ther. 2022, 20, 907–913. [Google Scholar] [CrossRef]


| Author | Year | Study Design | Population | Intervention | Vitamin D Deficiency Prior to Evaluation | Primary and Secondary Outcomes |
|---|---|---|---|---|---|---|
| Bychinin et al. [16] | 2022 | Placebo-controlled RCT | Patients admitted to the ICU for symptomatic SARS-CoV-2 infection | 60,000 IU of vitamin D3 followed by daily maintenance doses of 5000 IU (p.o) | Yes, median points to patients with severe deficiency | Lymphocyte counts, natural killer (NK) and natural killer T (NKT) cell counts, neutrophil-to-lymphocyte ratio (NLR), and serum levels of inflammatory markers on 7th day of treatment; LOS, ICU admission, and mortality |
| Cannata-Andía et al. [17] | 2022 | Open-label RCT | Patients hospitalized for mild to moderate-severe SARS-CoV-2 infection | 100,000 IU of vitamin D3 (p.o, single dose) | Yes, mild to severe deficiency | LOS, ICU, admission, mortality |
| De Niet et al. [18] | 2022 | Placebo-controlled RCT | Vitamin D-deficient patients hospitalized for SARS-CoV-2 infection | 25,000 IU/d over 4 consecutive days, followed by 25,000 IU/week up to 6 weeks (p.o) | Yes, moderate to severe | WHO Ordinal Scale and Inflammation Risk Categories in COVID-19, LOS, ICU admission, time until absence of fever, oxygen support, mechanical ventilation or additional organ support, mortality |
| Elamir et al. [19] | 2021 | Open-label RCT | Patients hospitalized for symptomatic SARS-CoV-2 infection | 0.5 μg of Calcitriol daily for 14 days or hospital discharge | Not evaluated | Oxygen support, LOS, ICU admission, mortality, and readmission |
| Entrenas et al. [20] | 2020 | Open-label RCT | Patients hospitalized for symptomatic SARS-CoV-2 infection | 0.532 mg of Calcifediol, then 0.266 mg on days 3 and 7, and then weekly until discharge or ICU admission(p.o) | Not evaluated | ICU admission and mortality |
| Karonova et al. [21] | 2022 | Open-label RCT | Patients hospitalized for symptomatic SARS-CoV-2 infection | 50,000 IU on the 1st and the 8th day of hospitalization (p.o) | Yes, mild to severe deficiency | Complete blood count, CRP level, and B cell subsets on the 9th day of hospitalization compared to the 1st day, severity of COVID-19, oxygen support, mechanical ventilation, LOS, and ICU admission |
| Maghbooli et al. [22] | 2021 | Placebo-controlled RCT | Vitamin D-deficient patients with symptomatic SARS-CoV-2 infection | 3000 to 6000 IU (p.o, daily for 60 d) | Yes, mild and moderate deficiency | Severity of COVID-19, LOS, oxygen support, mortality, lymphocyte count and percentage |
| Mariani et al. [23] | 2022 | Placebo-controlled RCT | Patients hospitalized for mild to moderate SARS-CoV-2 infection | 500,000 IU of vitamin D3 (p.o, single dose) | Sample points to patients without deficiency | Change in the respiratory SOFA parameters between baseline up to day 7, change in SpO2, oxygen support, mechanical ventilation, the change in the quick SOFA, LOS, ICU admission, acute kidney injury, and mortality |
| Murai et al. [24] | 2021 | Placebo-controlled RCT | Patients with moderate to severe SARS-CoV-2 infection | 200,000 IU of vitamin D3 (p.o, single dose) | Yes, mild to severe deficiency | LOS, mortality, ICU admission, mechanical ventilation, total calcium, creatinine, and C-reactive protein |
| Author | Intervention/Control | LOS | p-Value LOS | Mortality | p-Value Mortality | Admission to ICU | p-Value Admission to ICU |
|---|---|---|---|---|---|---|---|
| Bychinin et al. [16] | n = 52/54 | 20.5 [15–33] vs. 14.5 [10–23] | 0.007 | 37% vs. 50% | 0.16 | 100% | - |
| Cannata-Andía et al. [17] | n = 274/269 | 10.0 [9.0–10.5] vs. 9.5 [9.0–10.5] | 0.19 | 8.0% vs. 5.6% | 0.69 | 17.2% vs. 16.4% | 0.65 |
| De Niet et al. [18] | n = 21/22 | 4.0 [3.0–6.0] vs. 8.0 [6.0–12.0] | 0.003 | 4.8% vs. 12% | 0.129 | 9.5% vs. 23% | 0.412 |
| Elamir et al. [19] | n = 25/25 | 5.5 (± 3.9) vs. 9.24 (± 9.4) | 0.14 | 0% vs. 12% | 0.23 | 20% vs. 32% | 0.33 |
| Entrenas Castillo et al. [20] | n = 50/26 | - | - | 0% vs. 7.7% | - | 2% vs. 50% | <0.001 |
| Karonova et al. [21] | n = 56/54 | - | - | - | - | 0% vs. 6% | - |
| Maghbooli et al. [22] | n = 53/53 | 5 [3] vs. 6 [5.5] | 0.1 | 6% vs. 9% | 0.7 | 11% vs. 19% | 0.3 |
| Mariani et al. [23] | n = 115/103 | 6.0 [4.0–9.0] vs. 6.0 [4.0–10.0] | 0.632 | 4.3% vs. 1.9% | 0.451 | 7.8% vs. 10.7% | 0.622 |
| Murai et al. [24] | n = 119/118 | 7.0 [4.0–10.0] vs. 7.0 [5.0–13.0] | 0.59 | 7.6% vs. 5.1% | 0.43 | 16% vs. 21.2% | 0.3 |
| Author | Year | Study Design | Population | Intervention | Vitamin D Deficiency Prior to Evaluation | Primary and Secondary Outcomes |
|---|---|---|---|---|---|---|
| Annweiler et al. [25] | 2022 | Open-label trial | Elderly patients with symptomatic SARS-CoV-2 infection not requiring admission to the ICU | Vitamin D 400,000 IU vs. 50,000 IU (orally, single dose) | Not evaluated | Mortality within 14 days, mortality within 28 days, and between-group comparison of safety |
| Sabico et al. [26] | 2021 | Open-label trial | Vitamin D-deficient patients with SARS-CoV-2 infection with mild to moderate SARS-CoV-2 infection | Vitamin D 5000 IU vs. 1000 IU (orally for 2 weeks) | Yes, mild deficiency | Number of days to resolve symptoms, changes in the metabolic profile, LOS, ICU admission, and mortality |
| Sarhan et al. [27] | 2022 | Open-label trial | Patients hospitalized for symptomatic SARS-CoV-2 infection | Single high-dose vitamin D cholecalciferol (200,000 IU) IM vs. vitamin D alfacalcidol (1 microgram/day) | Not evaluated | Improvement in oxygenation parameters, LOS, mortality, inflammatory profile, and occurrence of secondary infections |
| Torres et al. [28] | 2022 | Open-label trial | Vitamin D deficient patients hospitalized for symptomatic SARS-CoV-2 infection | Vitamin D 10,000 IU vs. 2000 IU(orally, for 2 weeks) | Yes, mild to moderate deficiency | 25(OH)D serum level, LOS, inflammatory profile, and the cytotoxic immune response |
| Author | High Dose/Low Dose | LOS | p-Value LOS | Mortality | p-Value Mortality | Admission to ICU | p-Value Admission to ICU |
|---|---|---|---|---|---|---|---|
| Annweiler et al. [25] | n = 127/127 | - | - | 6% vs. 14% 15% vs. 17% * | 0.39 0.70 | - | - |
| Sabico et al. [26] | n = 36/33 | 6 (5–8) vs. 7 (0–10) | 0.14 | 2.77% vs. 0% | - | 5.5% vs. 9% | 1.0 |
| Sarhan et al. [27] | n = 58/58 | 6.1 (±3.4) vs. 8.9 (±5.1) | 0.04 | 45% vs. 51% | 0.49 | 42% vs. 65% | 0.016 |
| Torres et al. [28] | n = 41/44 | 6.44 vs. 9.36 | >0.05 | 2.4% vs. 2.2% | - | 4.9% vs. 11.3% | >0.05 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sîrbu, A.C.; Sabin, O.; Bocșan, I.C.; Vesa, Ș.C.; Buzoianu, A.D. The Effect of Vitamin D Supplementation on the Length of Hospitalisation, Intensive Care Unit Admission, and Mortality in COVID-19—A Systematic Review and Meta-Analysis. Nutrients 2023, 15, 3470. https://doi.org/10.3390/nu15153470
Sîrbu AC, Sabin O, Bocșan IC, Vesa ȘC, Buzoianu AD. The Effect of Vitamin D Supplementation on the Length of Hospitalisation, Intensive Care Unit Admission, and Mortality in COVID-19—A Systematic Review and Meta-Analysis. Nutrients. 2023; 15(15):3470. https://doi.org/10.3390/nu15153470
Chicago/Turabian StyleSîrbu, Alexandru Constantin, Octavia Sabin, Ioana Corina Bocșan, Ștefan Cristian Vesa, and Anca Dana Buzoianu. 2023. "The Effect of Vitamin D Supplementation on the Length of Hospitalisation, Intensive Care Unit Admission, and Mortality in COVID-19—A Systematic Review and Meta-Analysis" Nutrients 15, no. 15: 3470. https://doi.org/10.3390/nu15153470
APA StyleSîrbu, A. C., Sabin, O., Bocșan, I. C., Vesa, Ș. C., & Buzoianu, A. D. (2023). The Effect of Vitamin D Supplementation on the Length of Hospitalisation, Intensive Care Unit Admission, and Mortality in COVID-19—A Systematic Review and Meta-Analysis. Nutrients, 15(15), 3470. https://doi.org/10.3390/nu15153470








