Linoleic Acid Induced Changes in SZ95 Sebocytes—Comparison with Palmitic Acid and Arachidonic Acid
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Culture and Treatments
2.2. Determination of mRNA Levels
2.3. RNA-Seq Data Analysis
2.4. Lipid Analyses
2.5. Statistical Analyses
3. Results
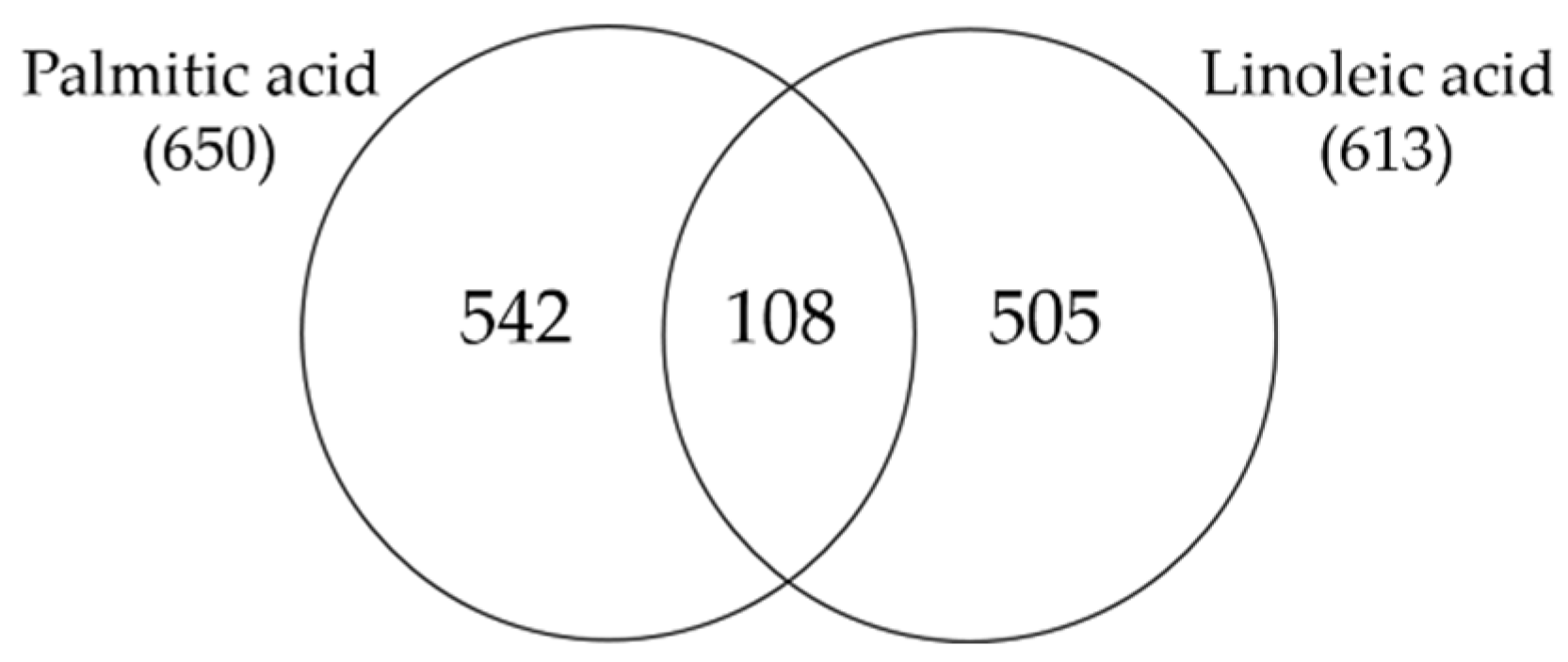
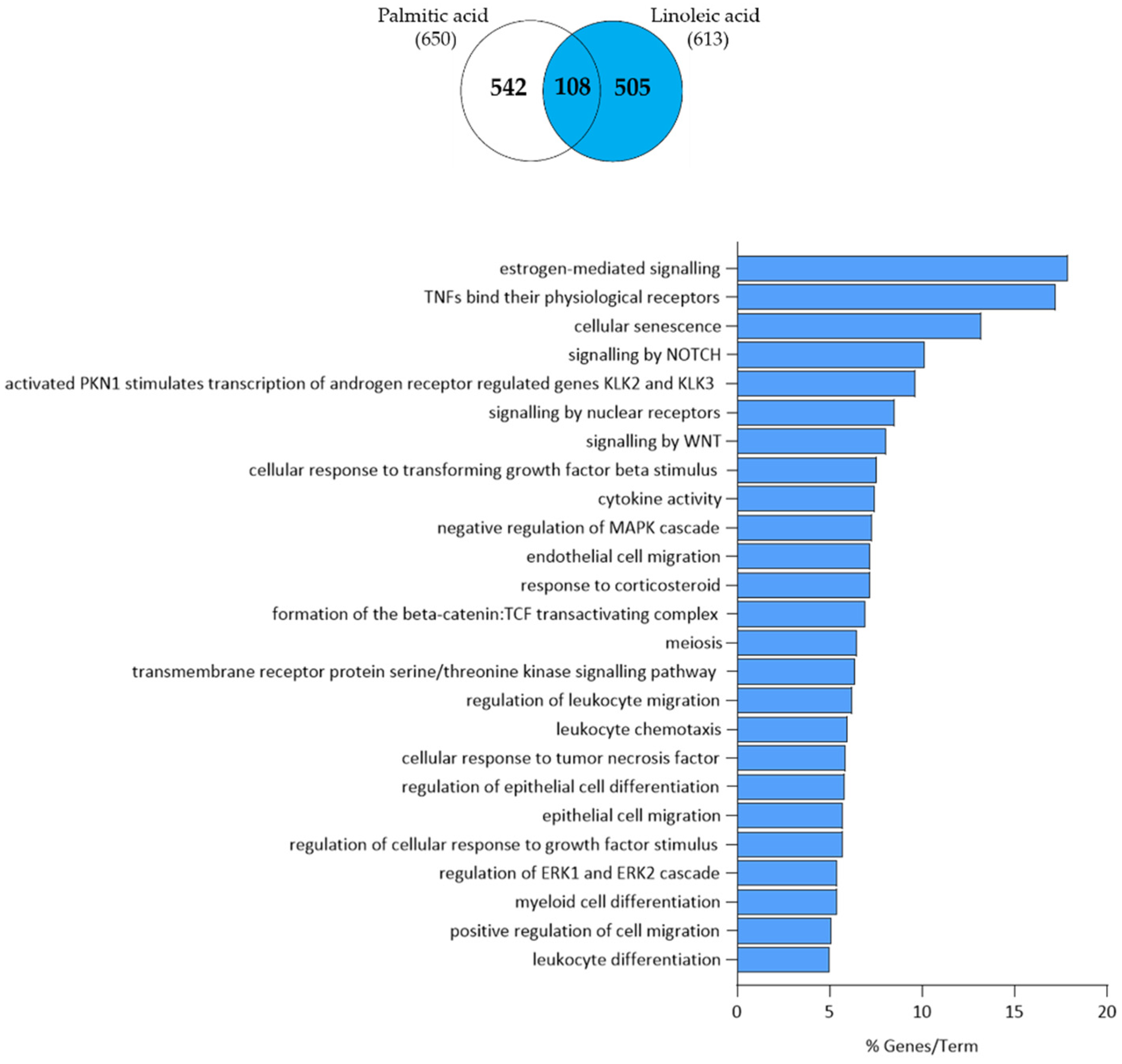
3.1. Linoleic Acid Regulates a Specific Set of Genes in SZ95 Sebocytes
3.2. Linoleic Acid-Induced Gene Expression Changes in SZ95 Sebocytes
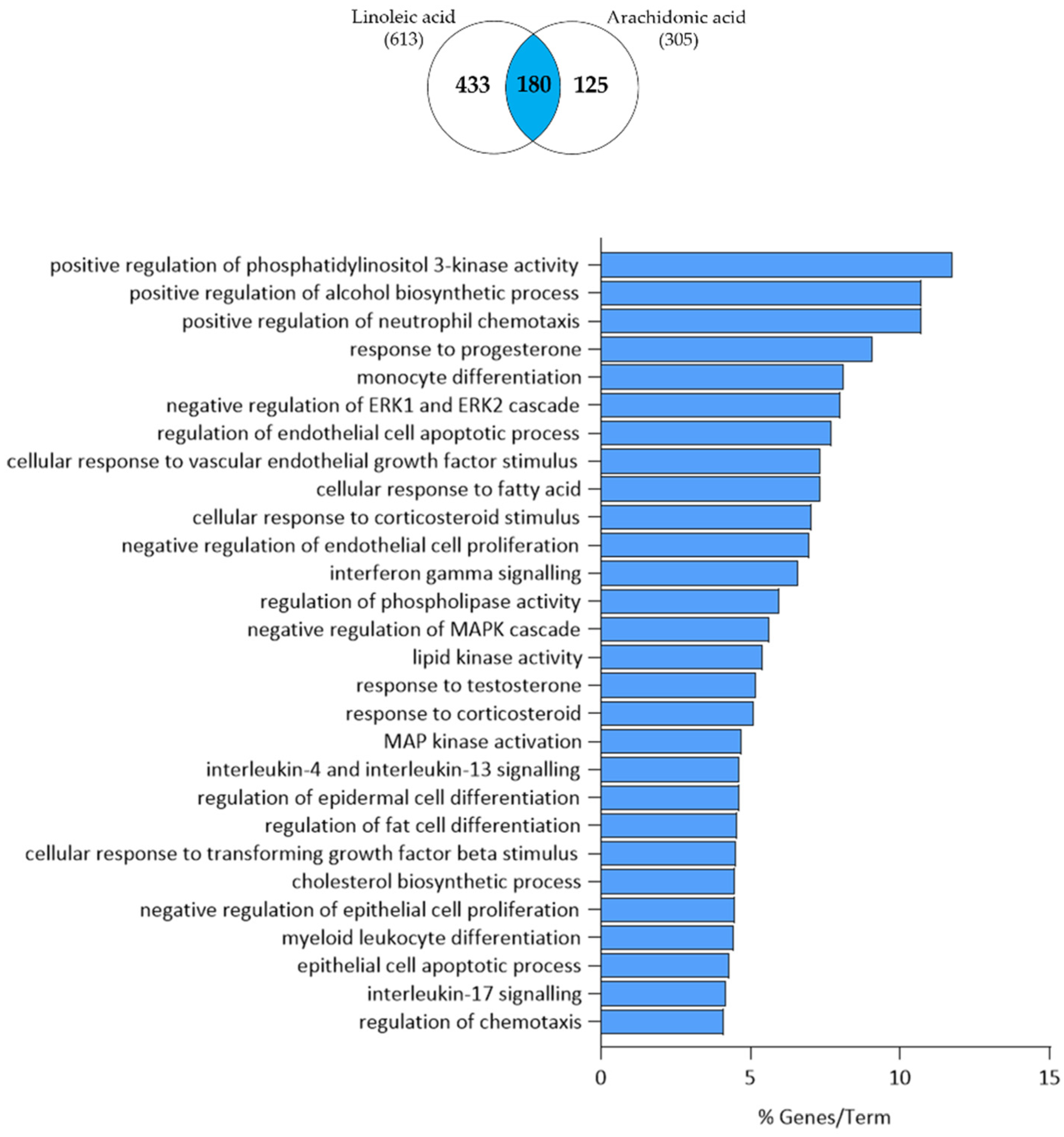
3.3. Comparison of Linoleic Acid and Arachidonic Acid-Induced Gene Expression Changes in SZ95 Sebocytes
3.4. Arachidonic Acid-Independent Effects of Linoleic Acid
3.5. Cholesterol Biosynthesis Is Only Affected by Linoleic Acid and Not by Arachidonic Acid at the Gene Expression Level
3.6. Linoleic and Arachidonic Acid-Induced Changes in the Lipid Profile of SZ95 Sebocytes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zouboulis, C.C.; Baron, J.M.; Böhm, M.; Kippenberger, S.; Kurzen, H.; Reichrath, J.; Thielitz, A. Frontiers in sebaceous gland biology and pathology. Exp. Dermatol. 2008, 17, 542–551. [Google Scholar] [CrossRef] [PubMed]
- Fluhr, J.W.; Mao-Qiang, M.; Brown, B.E.; Wertz, P.W.; Crumrine, D.; Sundberg, J.P.; Feingold, K.R.; Elias, P.M. Glycerol regulates stratum corneum hydration in sebaceous gland deficient (asebia) mice. J. Investig. Dermatol. 2003, 120, 728–737. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Picardo, M.; Zompetta, C.; De Luca, C.; Cirone, M.; Faggioni, A.; Nazzaro-Porro, M.; Passi, S.; Prota, G. Role of skin surface lipids in UV-induced epidermal cell changes. Arch. Dermatol. Res. 1991, 283, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Lovászi, M.; Szegedi, A.; Zouboulis, C.C.; Törőcsik, D. Sebaceous-immunobiology is orchestrated by sebum lipids. Derm. Endocrinol. 2017, 9, e1375636. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lovaszi, M.; Mattii, M.; Eyerich, K.; Gacsi, A.; Csanyi, E.; Kovacs, D.; Ruhl, R.; Szegedi, A.; Kemeny, L.; Stahle, M.; et al. Sebum lipids influence macrophage polarization and activation. Br. J. Dermatol. 2017, 177, 1671–1682. [Google Scholar] [CrossRef] [Green Version]
- Törőcsik, D.; Fazekas, F.; Póliska, S.; Gregus, A.; Janka, E.A.; Dull, K.; Szegedi, A.; Zouboulis, C.C.; Kovács, D. Epidermal Growth Factor Modulates Palmitic Acid-Induced Inflammatory and Lipid Signaling Pathways in SZ95 Sebocytes. Front. Immunol. 2021, 12, 600017. [Google Scholar] [CrossRef]
- Nakatsuji, T.; Kao, M.C.; Zhang, L.; Zouboulis, C.C.; Gallo, R.L.; Huang, C.M. Sebum free fatty acids enhance the innate immune defense of human sebocytes by upregulating beta-defensin-2 expression. J. Investig. Dermatol. 2010, 130, 985–994. [Google Scholar] [CrossRef] [Green Version]
- Wille, J.J.; Kydonieus, A. Palmitoleic acid isomer (C16:1delta6) in human skin sebum is effective against gram-positive bacteria. Ski. Pharmacol. Appl. Ski. Physiol. 2003, 16, 176–187. [Google Scholar] [CrossRef]
- Elias, P.M.; Brown, B.E.; Ziboh, V.A. The permeability barrier in essential fatty acid deficiency: Evidence for a direct role for linoleic acid in barrier function. J. Investig. Dermatol. 1980, 74, 230–233. [Google Scholar] [CrossRef] [Green Version]
- Dahlhoff, M.; Camera, E.; Picardo, M.; Zouboulis, C.C.; Chan, L.; Chang, B.H.; Schneider, M.R. PLIN2, the major perilipin regulated during sebocyte differentiation, controls sebaceous lipid accumulation in vitro and sebaceous gland size in vivo. Biochim. Biophys. Acta 2013, 1830, 4642–4649. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.; Yang, C.C.; Sheu, H.M.; Seltmann, H.; Zouboulis, C.C. Expression of peroxisome proliferator-activated receptor and CCAAT/enhancer binding protein transcription factors in cultured human sebocytes. J. Investig. Dermatol. 2003, 121, 441–447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zouboulis, C.C.; Hossini, A.M.; Hou, X.; Wang, C.; Weylandt, K.H.; Pietzner, A. Effects of Moringa oleifera Seed Oil on Cultured Human Sebocytes In Vitro and Comparison with Other Oil Types. Int. J. Mol. Sci. 2023, 24, 10332. [Google Scholar] [CrossRef]
- Ottaviani, M.; Camera, E.; Picardo, M. Lipid mediators in acne. Mediat. Inflamm. 2010, 2010, 858176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Downing, D.T.; Stewart, M.E.; Wertz, P.W.; Strauss, J.S. Essential fatty acids and acne. J. Am. Acad. Dermatol. 1986, 14, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Letawe, C.; Boone, M.; Piérard, G.E. Digital image analysis of the effect of topically applied linoleic acid on acne microcomedones. Clin. Exp. Dermatol. 1998, 23, 56–58. [Google Scholar] [CrossRef]
- Valacchi, G.; De Luca, C.; Wertz, P.W. Lipid mediators in skin inflammation: Updates and current views. Mediat. Inflamm. 2010, 2010, 398926. [Google Scholar] [CrossRef] [Green Version]
- Choi, C.W.; Kim, Y.; Kim, J.E.; Seo, E.Y.; Zouboulis, C.C.; Kang, J.S.; Youn, S.W.; Chung, J.H. Enhancement of lipid content and inflammatory cytokine secretion in SZ95 sebocytes by palmitic acid suggests a potential link between free fatty acids and acne aggravation. Exp. Dermatol. 2019, 28, 207–210. [Google Scholar] [CrossRef]
- Zouboulis, C.C.; Angres, S.; Seltmann, H. Regulation of stearoyl-coenzyme A desaturase and fatty acid delta-6 desaturase-2 expression by linoleic acid and arachidonic acid in human sebocytes leads to enhancement of proinflammatory activity but does not affect lipogenesis. Br. J. Dermatol. 2011, 165, 269–276. [Google Scholar] [CrossRef]
- Dozsa, A.; Dezso, B.; Toth, B.I.; Bacsi, A.; Poliska, S.; Camera, E.; Picardo, M.; Zouboulis, C.C.; Bíró, T.; Schmitz, G.; et al. PPARγ-mediated and arachidonic acid-dependent signaling is involved in differentiation and lipid production of human sebocytes. J. Investig. Dermatol. 2014, 134, 910–920. [Google Scholar] [CrossRef] [Green Version]
- Zouboulis, C.C.; Coenye, T.; He, L.; Kabashima, K.; Kobayashi, T.; Niemann, C.; Nomura, T.; Oláh, A.; Picardo, M.; Quist, S.R.; et al. Sebaceous immunobiology—Skin homeostasis, pathophysiology, coordination of innate immunity and inflammatory response and disease associations. Front. Immunol. 2022, 13, 1029818. [Google Scholar] [CrossRef]
- Alestas, T.; Ganceviciene, R.; Fimmel, S.; Muller-Decker, K.; Zouboulis, C.C. Enzymes involved in the biosynthesis of leukotriene B4 and prostaglandin E2 are active in sebaceous glands. J. Mol. Med. 2006, 84, 75–87. [Google Scholar] [CrossRef]
- Oh, S.Y.; Lee, S.J.; Jung, Y.H.; Lee, H.J.; Han, H.J. Arachidonic acid promotes skin wound healing through induction of human MSC migration by MT3-MMP-mediated fibronectin degradation. Cell Death Dis. 2015, 6, e1750. [Google Scholar] [CrossRef] [Green Version]
- Ge, L.; Gordon, J.S.; Hsuan, C.; Stenn, K.; Prouty, S.M. Identification of the delta-6 desaturase of human sebaceous glands: Expression and enzyme activity. J. Investig. Dermatol. 2003, 120, 707–714. [Google Scholar] [CrossRef]
- Zouboulis, C.C.; Seltmann, H.; Neitzel, H.; Orfanos, C.E. Establishment and characterization of an immortalized human sebaceous gland cell line (SZ95). J. Investig. Dermatol. 1999, 113, 1011–1020. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bindea, G.; Mlecnik, B.; Hackl, H.; Charoentong, P.; Tosolini, M.; Kirilovsky, A.; Fridman, W.H.; Pages, F.; Trajanoski, Z.; Galon, J. ClueGO: A Cytoscape plug-in to decipher functionally grouped gene ontology and pathway annotation networks. Bioinformatics 2009, 25, 1091–1093. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Törőcsik, D.; Kovács, D.; Camera, E.; Lovászi, M.; Cseri, K.; Nagy, G.G.; Molinaro, R.; Rühl, R.; Tax, G.; Szabó, K.; et al. Leptin promotes a proinflammatory lipid profile and induces inflammatory pathways in human SZ95 sebocytes. Br. J. Dermatol. 2014, 171, 1326–1335. [Google Scholar] [CrossRef] [Green Version]
- Ludovici, M.; Kozul, N.; Materazzi, S.; Risoluti, R.; Picardo, M.; Camera, E. Influence of the sebaceous gland density on the stratum corneum lipidome. Sci. Rep. 2018, 8, 11500. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Isseroff, R.R.; Ziboh, V.A.; Chapkin, R.S.; Martinez, D.T. Conversion of linoleic acid into arachidonic acid by cultured murine and human keratinocytes. J. Lipid Res. 1987, 28, 1342–1349. [Google Scholar] [CrossRef] [PubMed]
- Chapkin, R.S.; Ziboh, V.A.; Marcelo, C.L.; Voorhees, J.J. Metabolism of essential fatty acids by human epidermal enzyme preparations: Evidence of chain elongation. J. Lipid Res. 1986, 27, 945–954. [Google Scholar] [CrossRef] [PubMed]
- Mathay, C.; Pierre, M.; Pittelkow, M.R.; Depiereux, E.; Nikkels, A.F.; Colige, A.; Poumay, Y. Transcriptional profiling after lipid raft disruption in keratinocytes identifies critical mediators of atopic dermatitis pathways. J. Investig. Dermatol. 2011, 131, 46–58. [Google Scholar] [CrossRef] [Green Version]
- Saini, R.K.; Keum, Y.S. Omega-3 and omega-6 polyunsaturated fatty acids: Dietary sources, metabolism, and significance—A review. Life Sci. 2018, 203, 255–267. [Google Scholar] [CrossRef]
- Innes, J.K.; Calder, P.C. Omega-6 fatty acids and inflammation. Prostaglandins Leukot. Essent. Fat. Acids 2018, 132, 41–48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Whelan, J.; Fritsche, K. Linoleic acid. Adv. Nutr. 2013, 4, 311–312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cho, Y.; Ziboh, V.A. Incorporation of 13-hydroxyoctadecadienoic acid (13-HODE) into epidermal ceramides and phospholipids: Phospholipase C-catalyzed release of novel 13-HODE-containing diacylglycerol. J. Lipid Res. 1994, 35, 255–262. [Google Scholar] [CrossRef]
- Hansen, H.S.; Jensen, B. Essential function of linoleic acid esterified in acylglucosylceramide and acylceramide in maintaining the epidermal water permeability barrier. Evidence from feeding studies with oleate, linoleate, arachidonate, columbinate and alpha-linolenate. Biochim. Biophys. Acta 1985, 834, 357–363. [Google Scholar] [CrossRef]
- Danby, S.G.; AlEnezi, T.; Sultan, A.; Lavender, T.; Chittock, J.; Brown, K.; Cork, M.J. Effect of olive and sunflower seed oil on the adult skin barrier: Implications for neonatal skin care. Pediatr. Dermatol. 2013, 30, 42–50. [Google Scholar] [CrossRef]
- Hanley, K.; Jiang, Y.; He, S.S.; Friedman, M.; Elias, P.M.; Bikle, D.D.; Williams, M.L.; Feingold, K.R. Keratinocyte differentiation is stimulated by activators of the nuclear hormone receptor PPARalpha. J. Investig. Dermatol. 1998, 110, 368–375. [Google Scholar] [CrossRef] [Green Version]
- Zouboulis Ch, C.; Saborowski, A.; Boschnakow, A. Zileuton, an oral 5-lipoxygenase inhibitor, directly reduces sebum production. Dermatology 2005, 210, 36–38. [Google Scholar] [CrossRef]
- Pappas, A.; Johnsen, S.; Liu, J.C.; Eisinger, M. Sebum analysis of individuals with and without acne. Derm. Endocrinol. 2009, 1, 157–161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hong, I.; Lee, M.H.; Na, T.Y.; Zouboulis, C.C.; Lee, M.O. LXRalpha enhances lipid synthesis in SZ95 sebocytes. J. Investig. Dermatol. 2008, 128, 1266–1272. [Google Scholar] [CrossRef] [Green Version]
- Makrantonaki, E.; Zouboulis, C.C. Testosterone metabolism to 5alpha-dihydrotestosterone and synthesis of sebaceous lipids is regulated by the peroxisome proliferator-activated receptor ligand linoleic acid in human sebocytes. Br. J. Dermatol. 2007, 156, 428–432. [Google Scholar] [CrossRef]
- Farvid, M.S.; Ding, M.; Pan, A.; Sun, Q.; Chiuve, S.E.; Steffen, L.M.; Willett, W.C.; Hu, F.B. Dietary linoleic acid and risk of coronary heart disease: A systematic review and meta-analysis of prospective cohort studies. Circulation 2014, 130, 1568–1578. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chowdhury, R.; Warnakula, S.; Kunutsor, S.; Crowe, F.; Ward, H.A.; Johnson, L.; Franco, O.H.; Butterworth, A.S.; Forouhi, N.G.; Thompson, S.G.; et al. Association of dietary, circulating, and supplement fatty acids with coronary risk: A systematic review and meta-analysis. Ann. Intern. Med. 2014, 160, 398–406. [Google Scholar] [CrossRef]
- Ramsden, C.E.; Zamora, D.; Majchrzak-Hong, S.; Faurot, K.R.; Broste, S.K.; Frantz, R.P.; Davis, J.M.; Ringel, A.; Suchindran, C.M.; Hibbeln, J.R. Re-evaluation of the traditional diet-heart hypothesis: Analysis of recovered data from Minnesota Coronary Experiment (1968–1973). BMJ Clin. Res. Ed. 2016, 353, i1246. [Google Scholar] [CrossRef] [Green Version]
- Pappas, A.; Anthonavage, M.; Gordon, J.S. Metabolic fate and selective utilization of major fatty acids in human sebaceous gland. J. Investig. Dermatol. 2002, 118, 164–171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zouboulis, C.C.; Picardo, M.; Ju, Q.; Kurokawa, I.; Torocsik, D.; Biro, T.; Schneider, M.R. Beyond acne: Current aspects of sebaceous gland biology and function. Rev. Endocr. Metab. Disord. 2016, 17, 319–334. [Google Scholar] [CrossRef] [PubMed]
- Fritsch, M.; Orfanos, C.E.; Zouboulis, C.C. Sebocytes are the key regulators of androgen homeostasis in human skin. J. Investig. Dermatol. 2001, 116, 793–800. [Google Scholar] [CrossRef]
- Zouboulis, C.C. Acne and sebaceous gland function. Clin. Dermatol. 2004, 22, 360–366. [Google Scholar] [CrossRef]
- Schneider, M.R.; Paus, R. Sebocytes, multifaceted epithelial cells: Lipid production and holocrine secretion. Int. J. Biochem. Cell Biol. 2010, 42, 181–185. [Google Scholar] [CrossRef]
- Yanes, D.A.; Mosser-Goldfarb, J.L. Emerging therapies for atopic dermatitis: The prostaglandin/leukotriene pathway. J. Am. Acad. Dermatol. 2018, 78, S71–S75. [Google Scholar] [CrossRef]
- Huang, X.; Yi, S.; Hu, J.; Du, Z.; Wang, Q.; Ye, Z.; Su, G.; Kijlstra, A.; Yang, P. Linoleic acid inhibits in vitro function of human and murine dendritic cells, CD4(+)T cells and retinal pigment epithelial cells. Graefe’s Arch. Clin. Exp. Ophthalmol. 2021, 259, 987–998. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Kesarwala, A.H.; Eggert, T.; Medina-Echeverz, J.; Kleiner, D.E.; Jin, P.; Stroncek, D.F.; Terabe, M.; Kapoor, V.; ElGindi, M.; et al. NAFLD causes selective CD4(+) T lymphocyte loss and promotes hepatocarcinogenesis. Nature 2016, 531, 253–257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vaughan, J.E.; Walsh, S.W. Neutrophils from pregnant women produce thromboxane and tumor necrosis factor-alpha in response to linoleic acid and oxidative stress. Am. J. Obstet. Gynecol. 2005, 193, 830–835. [Google Scholar] [CrossRef] [PubMed]
- Hatanaka, E.; Levada-Pires, A.C.; Pithon-Curi, T.C.; Curi, R. Systematic study on ROS production induced by oleic, linoleic, and gamma-linolenic acids in human and rat neutrophils. Free. Radic. Biol. Med. 2006, 41, 1124–1132. [Google Scholar] [CrossRef]
- Mena, S.J.; Manosalva, C.; Carretta, M.D.; Teuber, S.; Olmo, I.; Burgos, R.A.; Hidalgo, M.A. Differential free fatty acid receptor-1 (FFAR1/GPR40) signalling is associated with gene expression or gelatinase granule release in bovine neutrophils. Innate Immun. 2016, 22, 479–489. [Google Scholar] [CrossRef] [Green Version]
- Kain, V.; Halade, G.V. Immune responsive resolvin D1 programs peritoneal macrophages and cardiac fibroblast phenotypes in diversified metabolic microenvironment. J. Cell. Physiol. 2019, 234, 3910–3920. [Google Scholar] [CrossRef]
- Navarini, A.A.; Simpson, M.A.; Weale, M.; Knight, J.; Carlavan, I.; Reiniche, P.; Burden, D.A.; Layton, A.; Bataille, V.; Allen, M.; et al. Genome-wide association study identifies three novel susceptibility loci for severe Acne vulgaris. Nat. Commun. 2014, 5, 4020. [Google Scholar] [CrossRef] [Green Version]
- Kelhala, H.L.; Palatsi, R.; Fyhrquist, N.; Lehtimaki, S.; Vayrynen, J.P.; Kallioinen, M.; Kubin, M.E.; Greco, D.; Tasanen, K.; Alenius, H.; et al. IL-17/Th17 pathway is activated in acne lesions. PLoS ONE 2014, 9, e105238. [Google Scholar] [CrossRef]
- Mattii, M.; Lovászi, M.; Garzorz, N.; Atenhan, A.; Quaranta, M.; Lauffer, F.; Konstantinow, A.; Küpper, M.; Zouboulis, C.C.; Kemeny, L.; et al. Sebocytes contribute to skin inflammation by promoting the differentiation of T helper 17 cells. Br. J. Dermatol. 2018, 178, 722–730. [Google Scholar] [CrossRef] [Green Version]
- Zhang, C.; Chinnappan, M.; Prestwood, C.A.; Edwards, M.; Artami, M.; Thompson, B.M.; Eckert, K.M.; Vale, G.; Zouboulis, C.; McDonald, J.G.; et al. Interleukins 4 and 13 drive lipid abnormalities in skin cells through regulation of sex steroid hormone synthesis. Proc. Natl. Acad. Sci. USA 2021, 118, e2100749118. [Google Scholar] [CrossRef]
- Balić, A.; Vlašić, D.; Žužul, K.; Marinović, B.; Bukvić Mokos, Z. Omega-3 Versus Omega-6 Polyunsaturated Fatty Acids in the Prevention and Treatment of Inflammatory Skin Diseases. Int. J. Mol. Sci. 2020, 21, 741. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jung, J.Y.; Kwon, H.H.; Hong, J.S.; Yoon, J.Y.; Park, M.S.; Jang, M.Y.; Suh, D.H. Effect of dietary supplementation with omega-3 fatty acid and gamma-linolenic acid on acne vulgaris: A randomised, double-blind, controlled trial. Acta Derm. Venereol. 2014, 94, 521–525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dahlhoff, M.; Camera, E.; Ludovici, M.; Picardo, M.; Muller, U.; Leonhardt, H.; Zouboulis, C.C.; Schneider, M.R. EGFR/ERBB receptors differentially modulate sebaceous lipogenesis. FEBS Lett. 2015, 589, 1376–1382. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holleran, W.M.; Williams, M.L.; Gao, W.N.; Elias, P.M. Serine-palmitoyl transferase activity in cultured human keratinocytes. J. Lipid Res. 1990, 31, 1655–1661. [Google Scholar] [CrossRef]









Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kovács, D.; Camera, E.; Póliska, S.; Cavallo, A.; Maiellaro, M.; Dull, K.; Gruber, F.; Zouboulis, C.C.; Szegedi, A.; Törőcsik, D. Linoleic Acid Induced Changes in SZ95 Sebocytes—Comparison with Palmitic Acid and Arachidonic Acid. Nutrients 2023, 15, 3315. https://doi.org/10.3390/nu15153315
Kovács D, Camera E, Póliska S, Cavallo A, Maiellaro M, Dull K, Gruber F, Zouboulis CC, Szegedi A, Törőcsik D. Linoleic Acid Induced Changes in SZ95 Sebocytes—Comparison with Palmitic Acid and Arachidonic Acid. Nutrients. 2023; 15(15):3315. https://doi.org/10.3390/nu15153315
Chicago/Turabian StyleKovács, Dóra, Emanuela Camera, Szilárd Póliska, Alessia Cavallo, Miriam Maiellaro, Katalin Dull, Florian Gruber, Christos C. Zouboulis, Andrea Szegedi, and Dániel Törőcsik. 2023. "Linoleic Acid Induced Changes in SZ95 Sebocytes—Comparison with Palmitic Acid and Arachidonic Acid" Nutrients 15, no. 15: 3315. https://doi.org/10.3390/nu15153315
APA StyleKovács, D., Camera, E., Póliska, S., Cavallo, A., Maiellaro, M., Dull, K., Gruber, F., Zouboulis, C. C., Szegedi, A., & Törőcsik, D. (2023). Linoleic Acid Induced Changes in SZ95 Sebocytes—Comparison with Palmitic Acid and Arachidonic Acid. Nutrients, 15(15), 3315. https://doi.org/10.3390/nu15153315







