The Gliadin Hydrolysis Capacity of B. longum, L. acidophilus, and L. plantarum and Their Protective Effects on Caco-2 Cells against Gliadin-Induced Inflammatory Responses
Abstract
1. Introduction
2. Materials and Methods
2.1. Probiotic Preparations
2.2. Sourdough Fermentation
2.3. Gliadin Extract
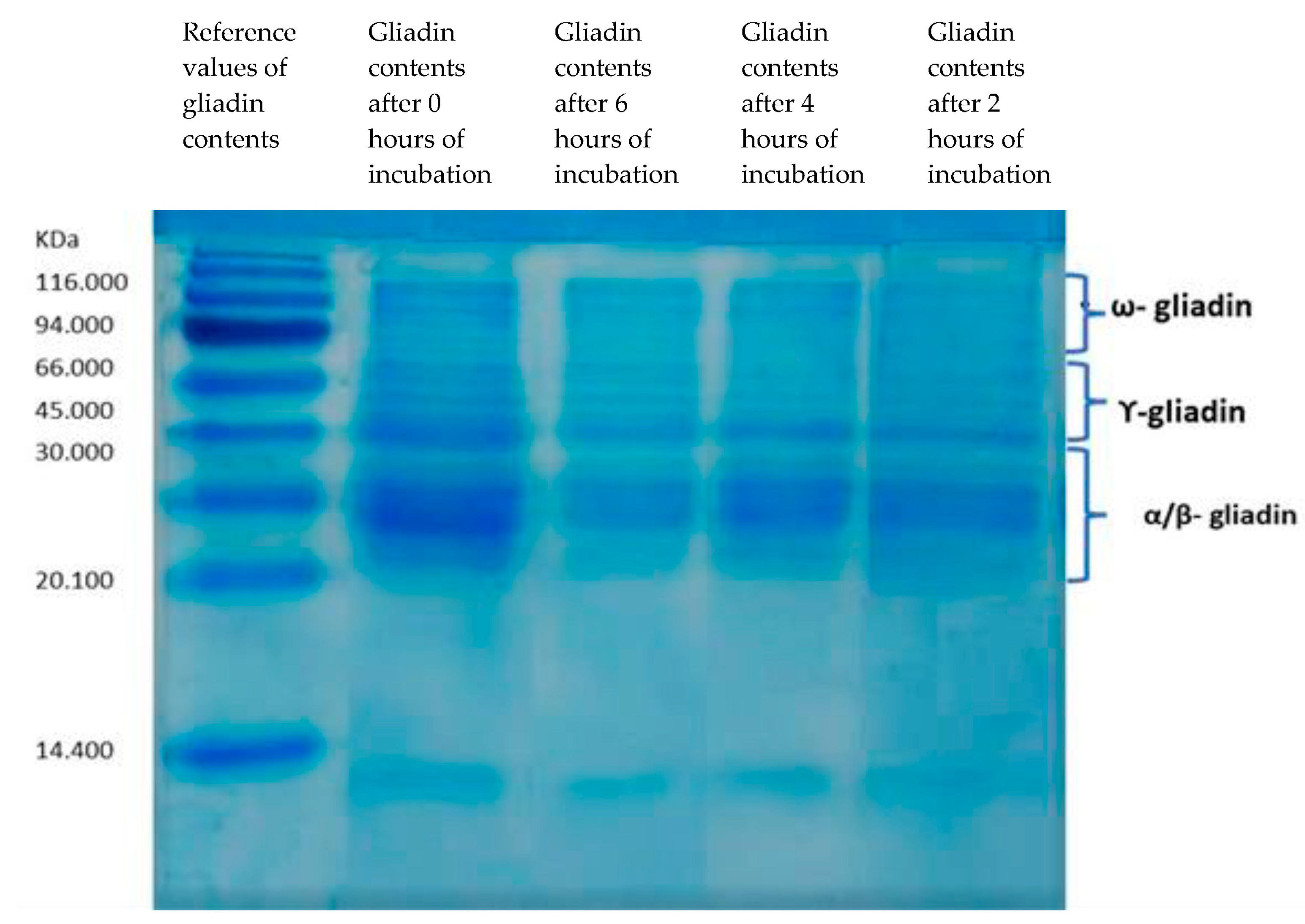
2.4. SDS-PAGE Electrophoresis of Gliadins
2.5. In Vitro Digestion of Gliadins
2.6. Cell Culture Conditions
2.7. Evaluating B. longum, L. acidophilous, and L. plantarum Effects on the Inflammatory Effects of Gliadin Peptides in Intestinal Epithelial (Caco-2) Cells
2.8. Sandwich Enzyme-Linked Immunosorbent Assay (ELISA)
2.9. RNA Extraction and Quantitative Real-Time PCR (qRT-PCR)
2.10. Statistical Analysis
2.11. Ethical Approval
3. Results
3.1. Gliadin Hydrolysis by Selected Probiotics
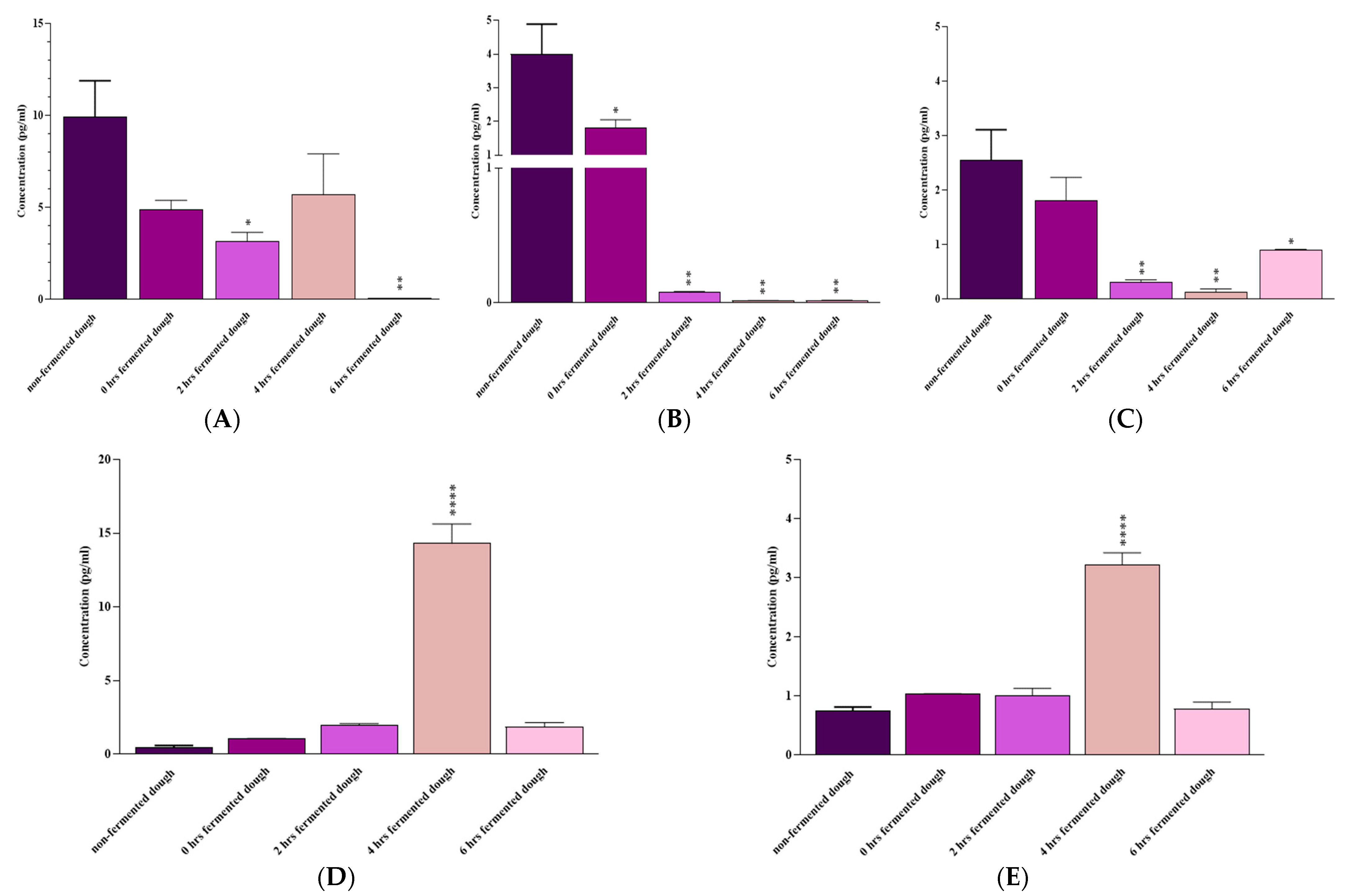
3.2. IL-6 and INF-γ Protein Expression
3.3. IL-6, IL-17A, IFN-γ, IL-10, and TGF-β mRNA Expression
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sharma, N.; Bhatia, S.; Chunduri, V.; Kaur, S.; Sharma, S.; Kapoor, P.; Kumari, A.; Garg, M. Pathogenesis of Celiac Disease and Other Gluten Related Disorders in Wheat and Strategies for Mitigating Them. Front. Nutr. 2020, 7, 6. [Google Scholar] [CrossRef]
- Tovoli, F.; Masi, C.; Guidetti, E.; Negrini, G.; Paterini, P.; Bolondi, L. Clinical and diagnostic aspects of gluten related disorders. World J. Clin. Cases 2015, 3, 275–284. [Google Scholar] [CrossRef]
- Roszkowska, A.; Pawlicka, M.; Mroczek, A.; Bałabuszek, K.; Nieradko-Iwanicka, B. Non-Celiac Gluten Sensitivity: A Review. Medicina 2019, 55, 222. [Google Scholar] [CrossRef] [PubMed]
- Rostami-Nejad, M.; Lahmi, F.; Zali, M. Non-celiac Gluten Sensitivity. J. Army Univ. Med. Sci. 2013, 11, 243–251. [Google Scholar]
- Taraghikhah, N.; Ashtari, S.; Asri, N.; Shahbazkhani, B.; Al-Dulaimi, D.; Rostami-Nejad, M.; Rezaei-Tavirani, M.; Razzaghi, M.R.; Zali, M.R. An updated overview of spectrum of gluten-related disorders: Clinical and diagnostic aspects. BMC Gastroenterol. 2020, 20, 258. [Google Scholar] [CrossRef]
- Volta, U.; Bardella, M.T.; Calabrò, A.; Troncone, R.; Corazza, G.R. An Italian prospective multicenter survey on patients suspected of having non-celiac gluten sensitivity. BMC Med. 2014, 12, 85. [Google Scholar] [CrossRef]
- Aziz, I.; Lewis, N.R.; Hadjivassiliou, M.; Winfield, S.N.; Rugg, N.; Kelsall, A.; Newrick, L.; Sanders, D.S. A UK study assessing the population prevalence of self-reported gluten sensitivity and referral characteristics to secondary care. Eur. J. Gastroenterol. Hepatol. 2014, 26, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Sapone, A.; Lammers, K.M.; Casolaro, V.; Cammarota, M.; Giuliano, M.T.; De Rosa, M.; Stefanile, R.; Mazzarella, G.; Tolone, C.; Russo, M.I.; et al. Divergence of gut permeability and mucosal immune gene expression in two gluten-associated conditions: Celiac disease and gluten sensitivity. BMC Med. 2011, 9, 23. [Google Scholar] [CrossRef] [PubMed]
- Uhde, M.; Ajamian, M.; Caio, G.; De Giorgio, R.; Indart, A.; Green, P.H.; Verna, E.C.; Volta, U.; Alaedini, A. Intestinal cell damage and systemic immune activation in individuals reporting sensitivity to wheat in the absence of coeliac disease. Gut 2016, 65, 1930–1937. [Google Scholar] [CrossRef]
- Hollon, J.; Leonard Puppa, E.; Greenwald, B.; Goldberg, E.; Guerrerio, A.; Fasano, A. Effect of Gliadin on Permeability of Intestinal Biopsy Explants from Celiac Disease Patients and Patients with Non-Celiac Gluten Sensitivity. Nutrients 2015, 7, 1565–1576. [Google Scholar] [CrossRef]
- Schuppan, D.; Pickert, G.; Ashfaq-Khan, M.; Zevallos, V. Non-celiac wheat sensitivity: Differential diagnosis, triggers and implications. Best Pr. Res. Clin. Gastroenterol. 2015, 29, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Escudero-Hernández, C.; Peña, A.S.; Bernardo, D. Immunogenetic Pathogenesis of Celiac Disease and Non-celiac Gluten Sensitivity. Curr. Gastroenterol. Rep. 2016, 18, 36. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.F.; Leffler, D.A.; Bai, J.C.; Biagi, F.; Fasano, A.; Green, P.H.R.; Hadjivassiliou, M.; Kaukinen, K.; Kelly, C.P.; Leonard, J.N.; et al. The Oslo definitions for coeliac disease and related terms. Gut 2013, 62, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Catassi, C.; Elli, L.; Bonaz, B.; Bouma, G.; Carroccio, A.; Castillejo, G.; Cellier, C.; Cristofori, F.; De Magistris, L.; Dolinsek, J.; et al. Diagnosis of Non-Celiac Gluten Sensitivity (NCGS): The Salerno Experts’ Criteria. Nutrients 2015, 7, 4966–4977. [Google Scholar] [CrossRef]
- Guandalini, S.; Polanco, I. Nonceliac gluten sensitivity or wheat intolerance syndrome? J. Pediatr. 2015, 166, 805–811. [Google Scholar] [CrossRef]
- Volta, U.; Caio, G.; Tovoli, F.; De Giorgio, R. Non-celiac gluten sensitivity: Questions still to be answered despite increasing awareness. Cell. Mol. Immunol. 2013, 10, 383–392. [Google Scholar] [CrossRef]
- Husby, S.; Murray, J.A. Gluten Sensitivity: Celiac Lite versus Celiac Like. J. Pediatr. 2014, 164, 436–438. [Google Scholar] [CrossRef]
- Bucci, C.; Zingone, F.; Russo, I.; Morra, I.; Tortora, R.; Pogna, N.; Scalia, G.; Iovino, P.; Ciacci, C. Gliadin Does Not Induce Mucosal Inflammation or Basophil Activation in Patients with Nonceliac Gluten Sensitivity. Clin. Gastroenterol. Hepatol. 2013, 11, 1294–1299.e1. [Google Scholar] [CrossRef]
- Aufiero, V.R.; Fasano, A.; Mazzarella, G. Non-Celiac Gluten Sensitivity: How Its Gut Immune Activation and Potential Dietary Management Differ from Celiac Disease. Mol. Nutr. Food Res. 2018, 62, e1700854. [Google Scholar] [CrossRef]
- Brottveit, M.; Beitnes, A.-C.R.; Tollefsen, S.; Bratlie, J.E.; Jahnsen, F.L.; Johansen, F.-E.; Sollid, L.M.; Lundin, K.E. Mucosal Cytokine Response after Short-Term Gluten Challenge in Celiac Disease and Non-Celiac Gluten Sensitivity. Am. J. Gastroenterol. 2013, 108, 842–850. [Google Scholar] [CrossRef] [PubMed]
- Losurdo, G.; Piscitelli, D.; Di Leo, A. Novel steps forward in the histopathology of non-celiac gluten sensitivity. Virchows Arch. 2018, 473, 523. [Google Scholar] [CrossRef]
- Zanini, B.; Villanacci, V.; Marullo, M.; Cadei, M.; Lanzarotto, F.; Bozzola, A.; Ricci, C. Duodenal histological features in suspected non-celiac gluten sensitivity: New insights into a still undefined condition. Virchows Arch. 2018, 473, 229–234. [Google Scholar] [CrossRef]
- Carroccio, A.; Giannone, G.; Mansueto, P.; Soresi, M.; La Blasca, F.; Fayer, F.; Iacobucci, R.; Porcasi, R.; Catalano, T.; Geraci, G.; et al. Duodenal and Rectal Mucosa Inflammation in Patients with Non-celiac Wheat Sensitivity. Clin. Gastroenterol. Hepatol. 2019, 17, 682–690.e3. [Google Scholar] [CrossRef]
- Losurdo, G.; Piscitelli, D.; Pezzuto, F.; Fortarezza, F.; Covelli, C.; Marra, A.; Iannone, A.; Amoruso, A.; Principi, M.; Ierardi, E.; et al. T Helper Lymphocyte and Mast Cell Immunohistochemical Pattern in Nonceliac Gluten Sensitivity. Gastroenterol. Res. Pr. 2017, 2017, 5023680. [Google Scholar] [CrossRef] [PubMed]
- Rostami, K.; Ensari, A.; Marsh, M.N.; Srivastava, A.; Villanacci, V.; Carroccio, A.; Aghdaei, H.A.; Bai, J.C.; Bassotti, G.; Becheanu, G.; et al. Gluten Induces Subtle Histological Changes in Duodenal Mucosa of Patients with Non-Coeliac Gluten Sensitivity: A Multicentre Study. Nutrients 2022, 14, 2487. [Google Scholar] [CrossRef]
- Volta, U.; Caio, G.; Ghirardi, C.; Lungaro, L.; Mansueto, P.; Carroccio, A.; de Giorgio, R. Minimal Lesions of the Small Intestinal Mucosa: More than Morphology. Dig. Dis. Sci. 2020, 65, 2761–2768. [Google Scholar] [CrossRef] [PubMed]
- Carroccio, A.; Mansueto, P.; Iacono, G.; Soresi, M.; D’Alcamo, A.; Cavataio, F.; Brusca, I.; Florena, A.M.; Ambrosiano, G.; Seidita, A.; et al. Non-Celiac Wheat Sensitivity Diagnosed by Double-Blind Placebo-Controlled Challenge: Exploring a New Clinical Entity. Am. J. Gastroenterol. ACG 2012, 107, 1898–1906. [Google Scholar] [CrossRef]
- Verdu, E.F.; Armstrong, D.; Murray, J.A. Between celiac disease and irritable bowel syndrome: The “no man’s land” of gluten sensitivity. Am. J. Gastroenterol. 2009, 104, 1587–1594. [Google Scholar] [CrossRef]
- Carroccio, A.; Soresi, M.; Chiavetta, M.; La Blasca, F.; Compagnoni, S.; Giuliano, A.; Fayer, F.; Mandreucci, F.; Castellucci, D.; Seidita, A.; et al. Frequency and Clinical Aspects of Neurological and Psychiatric Symptoms in Patients with Non-Celiac Wheat Sensitivity. Nutrients 2021, 13, 1971. [Google Scholar] [CrossRef] [PubMed]
- Mansueto, P.; Seidita, A.; Soresi, M.; Giuliano, A.; Riccio, G.; Volta, U.; Caio, G.; La Blasca, F.; Disclafani, R.; De Giorgio, R.; et al. Anemia in non-celiac wheat sensitivity: Prevalence and associated clinical and laboratory features. Dig. Liver Dis. 2022, 55, 735–742. [Google Scholar] [CrossRef] [PubMed]
- Catassi, C.; Alaedini, A.; Bojarski, C.; Bonaz, B.; Bouma, G.; Carroccio, A.; Castillejo, G.; De Magistris, L.; Dieterich, W.; Di Liberto, D.; et al. The Overlapping Area of Non-Celiac Gluten Sensitivity (NCGS) and Wheat-Sensitive Irritable Bowel Syndrome (IBS): An Update. Nutrients 2017, 9, 1268. [Google Scholar] [CrossRef] [PubMed]
- Biesiekierski, J.; Peters, S.L.; Newnham, E.D.; Rosella, O.; Muir, J.G.; Gibson, P.R. No Effects of Gluten in Patients with Self-Reported Non-Celiac Gluten Sensitivity after Dietary Reduction of Fermentable, Poorly Absorbed, Short-Chain Carbohydrates. Gastroenterology 2013, 145, 320–328.e3. [Google Scholar] [CrossRef] [PubMed]
- Skodje, G.I.; Sarna, V.K.; Minelle, I.H.; Rolfsen, K.L.; Muir, J.G.; Gibson, P.R.; Veierød, M.B.; Henriksen, C.; Lundin, K.E.A. Fructan, Rather Than Gluten, Induces Symptoms in Patients with Self-Reported Non-Celiac Gluten Sensitivity. Gastroenterol 2018, 154, 529–539.e2. [Google Scholar] [CrossRef] [PubMed]
- Graça, C.; Lima, A.; Raymundo, A.; Sousa, I. Sourdough Fermentation as a Tool to Improve the Nutritional and Health-Promoting Properties of Its Derived-Products. Fermentation 2021, 7, 246. [Google Scholar] [CrossRef]
- Xu, J.; Zhang, Y.; Wang, W.; Li, Y. Advanced properties of gluten-free cookies, cakes, and crackers: A review. Trends Food Sci. Technol. 2020, 103, 200–213. [Google Scholar] [CrossRef]
- Barbaro, M.; Cremon, C.; Stanghellini, V.; Barbara, G. Recent advances in understanding non-celiac gluten sensitivity. F1000Research 2018, 7, 1631. [Google Scholar] [CrossRef]
- Priyanka, P.; Gayam, S.; Kupec, J.T. The Role of a Low Fermentable Oligosaccharides, Disaccharides, Monosaccharides, and Polyol Diet in Nonceliac Gluten Sensitivity. Gastroenterol. Res. Pr. 2018, 2018, 1561476. [Google Scholar] [CrossRef]
- Sergi, C.; Villanacci, V.; Carroccio, A. Non-celiac wheat sensitivity: Rationality and irrationality of a gluten-free diet in individuals affected with non-celiac disease: A review. BMC Gastroenterol. 2021, 21, 5. [Google Scholar] [CrossRef]
- Carroccio, A.; D’alcamo, A.; Iacono, G.; Soresi, M.; Iacobucci, R.; Arini, A.; Geraci, G.; Fayer, F.; Cavataio, F.; La Blasca, F.; et al. Persistence of Nonceliac Wheat Sensitivity, Based on Long-term Follow-up. Gastroenterology 2017, 153, 56–58.e3. [Google Scholar] [CrossRef]
- Dennis, M.; Lee, A.R.; McCarthy, T. Nutritional Considerations of the Gluten-Free Diet. Gastroenterol. Clin. N. Am. 2019, 48, 53–72. [Google Scholar] [CrossRef]
- White, L.E.; Bannerman, E.; Gillett, P.M. Coeliac disease and the gluten-free diet: A review of the burdens; factors associated with adherence and impact on health-related quality of life, with specific focus on adolescence. J. Hum. Nutr. Diet. 2016, 29, 593–606. [Google Scholar] [CrossRef] [PubMed]
- Yano, H. Recent practical researches in the development of gluten-free breads. NPJ Sci. Food 2019, 3, 7. [Google Scholar] [CrossRef] [PubMed]
- Nionelli, L.; Rizzello, C.G. Sourdough-Based Biotechnologies for the Production of Gluten-Free Foods. Foods 2016, 5, 65. [Google Scholar] [CrossRef] [PubMed]
- Ghodsi, A.; Sarabi, M.; Ranjbar, G.; Afshari, A. The Gut Microbiota and Probiotics in Celiac Disease. J. Nutr. Fasting Health 2021, 9, 105–112. [Google Scholar]
- Kõiv, V.; Tenson, T. Gluten-degrading bacteria: Availability and applications. Appl. Microbiol. Biotechnol. 2021, 105, 3045–3059. [Google Scholar] [CrossRef] [PubMed]
- Wagh, S.K.; Lammers, K.M.; Padul, M.V.; Rodriguez-Herrera, A.; Dodero, V.I. Celiac Disease and Possible Dietary Interventions: From Enzymes and Probiotics to Postbiotics and Viruses. Int. J. Mol. Sci. 2022, 23, 11748. [Google Scholar] [CrossRef]
- Cristofori, F.; Francavilla, R.; Capobianco, D.; Dargenio, V.N.; Filardo, S.; Mastromarino, P. Bacterial-Based Strategies to Hydrolyze Gluten Peptides and Protect Intestinal Mucosa. Front. Immunol. 2020, 11, 567801. [Google Scholar] [CrossRef]
- Lindfors, K.; Rauhavirta, T.; Stenman, S.; Mäki, M.; Kaukinen, K. In vitro models for gluten toxicity: Relevance for celiac disease pathogenesis and development of novel treatment options. Exp. Biol. Med. 2012, 237, 119–125. [Google Scholar] [CrossRef]
- Stoven, S.; Murray, J.A.; Marietta, E.V. Latest in vitro and in vivo models of celiac disease. Expert Opin. Drug. Dis. 2013, 8, 445–457. [Google Scholar] [CrossRef]
- Herrera, M.G.; Nicoletti, F.; Gras, M.; Dörfler, P.W.; Tonali, N.; Hannappel, Y.; Ennen, I.; Hütten, A.; Hellweg, T.; Lammers, K.M.; et al. Pepsin Digest of Gliadin Forms Spontaneously Amyloid-Like Nanostructures Influencing the Expression of Selected Pro-Inflammatory, Chemoattractant, and Apoptotic Genes in Caco-2 Cells: Implications for Gluten-Related Disorders. Mol. Nutr. Food Res. 2021, 65, 2100200. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of Structural Proteins during the Assembly of the Head of Bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef] [PubMed]
- Silano, M.; Vincentini, O.; Iannuccelli, R.; Angelis, I.; Stammati, A.L.; Vincenzi, M. Mannan Prevents Gliadin Peptides Toxicity On Caco-2 Cells: OP2-02. J. Pediatr. Gastroenterol. Nutr. 2005, 40, 618–619. [Google Scholar] [CrossRef]
- Wei, C.-X.; Wu, J.-H.; Huang, Y.-H.; Wang, X.-Z.; Li, J.-Y. Lactobacillus plantarum improves LPS-induced Caco2 cell line intestinal barrier damage via cyclic AMP-PKA signaling. PLoS ONE 2022, 17, e0267831. [Google Scholar] [CrossRef]
- Tovar, L.E.R.; Gänzle, M.G. Degradation of Wheat Germ Agglutinin during Sourdough Fermentation. Foods 2021, 10, 340. [Google Scholar] [CrossRef] [PubMed]
- Ramedani, N.; Sharifan, A.; Gholam-Mostafaei, F.S.; Rostami-Nejad, M.; Yadegar, A.; Ehsani-Ardakani, M.J. The potentials of probiotics on gluten hydrolysis: A review study. Gastroenterol. Hepatol. Bed. Bench. 2020, 13 (Suppl. 1), S1–S7. [Google Scholar] [PubMed]
- Shewry, P.R. Do ancient types of wheat have health benefits compared with modern bread wheat? J. Cereal. Sci. 2018, 79, 469–476. [Google Scholar] [CrossRef]
- Seidita, A.; Mansueto, P.; Giuliano, A.; Chiavetta, M.; Mandreucci, F.; Soresi, M.; Pistone, M.; Compagnoni, S.; Castellucci, D.; Bisso, G.; et al. Potential tolerability of ancient grains in non-celiac wheat sensitivity patients: A preliminary evaluation. Front. Med. 2022, 9, 995019. [Google Scholar] [CrossRef]
- Giorgi, A.; Cerrone, R.; Capobianco, D.; Filardo, S.; Mancini, P.; Zanni, F.; Fanelli, S.; Mastromarino, P.; Mosca, L. A Probiotic Preparation Hydrolyzes Gliadin and Protects Intestinal Cells from the Toxicity of Pro-Inflammatory Peptides. Nutrients 2020, 12, 495. [Google Scholar] [CrossRef]
- Álvarez-Mercado, A.I.; Plaza-Díaz, J.; de Almagro, M.C.; Gil, Á.; Moreno-Muñoz, J.A.; Fontana, L. Bifidobacterium longum subsp. infantis CECT 7210 Reduces Inflammatory Cytokine Secretion in Caco-2 Cells Cultured in the Presence of Escherichia coli CECT 515. Int. J. Mol. Sci. 2022, 23, 10813. [Google Scholar] [CrossRef]
- Zhao, L.; Xie, Q.; Evivie, S.E.; Yue, Y.; Yang, H.; Lv, X.; Liu, F.; Li, B.; Huo, G. Bifidobacterium longum subsp. longum K5 alleviates inflammatory response and prevents intestinal barrier injury induced by LPS in vitro based on comparative genomics. J Funct. Foods 2022, 92, 105030. [Google Scholar] [CrossRef]
- Borthakur, A.; Bhattacharyya, S.; Kumar, A.; Anbazhagan, A.N.; Tobacman, J.K.; Dudeja, P.K. Lactobacillus acidophilus Alleviates Platelet-Activating Factor-Induced Inflammatory Responses in Human Intestinal Epithelial Cells. PLoS ONE 2013, 8, e75664. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.-J.; Chuang, C.-C.; Yang, H.-B.; Lu, C.-C.; Sheu, B.-S. Lactobacillus acidophilus ameliorates H. pylori-induced gastric inflammation by inactivating the Smad7 and NFκB pathways. BMC Microbiol. 2012, 12, 38. [Google Scholar] [CrossRef] [PubMed]
- Chiu, Y.-H.; Lu, Y.-C.; Ou, C.-C.; Lin, S.-L.; Tsai, C.-C.; Huang, C.-T.; Lin, M.-Y. Lactobacillus plantarum MYL26 induces endotoxin tolerance phenotype in Caco-2 cells. BMC Microbiol. 2013, 13, 190. [Google Scholar] [CrossRef] [PubMed]
- Scricciolo, A.; Lombardo, V.; Elli, L.; Bascuñán, K.A.; Doneda, L.; Rinaldi, F.; Pinto, D.; Araya, M.; Costantino, A.; Vecchi, M.; et al. Use of a proline-specific endopeptidase to reintroduce gluten in patients with non-coeliac gluten sensitivity: A randomized trial. Clin. Nutr. 2022, 41, 2025–2030. [Google Scholar] [CrossRef] [PubMed]
- Ido, H.; Matsubara, H.; Kuroda, M.; Takahashi, A.; Kojima, Y.; Koikeda, S.; Sasaki, M. Mix of Gluten-Digesting Enzymes Improved Symptoms of Non-Celiac Gluten Sensitivity: A Randomized Single-blind, Placebo-controlled Crossover Study. Clin. Transl. Gastroenterol. 2018, 9, 181. [Google Scholar] [CrossRef]
- Sakandar, H.A.; Kubow, S.; Azadi, B.; Faryal, R.; Ali, B.; Ghazanfar, S.; Quraishi, U.M.; Imran, M. Wheat Fermentation with Enterococcus mundtii QAUSD01 and Wickerhamomyces anomalus QAUWA03 Consortia Induces Concurrent Gliadin and Phytic Acid Degradation and Inhibits Gliadin Toxicity in Caco-2 Monolayers. Front. Microbiol. 2019, 9, 3312. [Google Scholar] [CrossRef]
- De Angelis, M.; Rizzello, C.G.; Fasano, A.; Clemente, M.G.; De Simone, C.; Silano, M.; De Vincenzi, M.; Losito, I.; Gobbetti, M. VSL#3 probiotic preparation has the capacity to hydrolyze gliadin polypeptides responsible for Celiac Sprue probiotics and gluten intolerance. Biochim. Et. Biophys. Acta (BBA)-Mol. Basis Dis. 2006, 1762, 80–93. [Google Scholar]
- Leszczyńska, J.; Diowksz, A.; Łącka, A.; Bryszewska, M.; Wolska, K.; Ambroziak, W. Decrease of wheat flour allergenicity via lactic acid fermentation. Food Agric. Immunol. 2009, 20, 139–145. [Google Scholar] [CrossRef]
- Dale, H.F.; Hatlebakk, J.G.; Hovdenak, N.; Ystad, S.O.; Lied, G.A. The effect of a controlled gluten challenge in a group of patients with suspected non-coeliac gluten sensitivity: A randomized, double-blind placebo-controlled challenge. Neurogastroenterol. Motil. 2018, 30, e13332. [Google Scholar] [CrossRef]
- Shahbazkhani, B.; Fanaeian, M.M.; Farahvash, M.J.; Aletaha, N.; Alborzi, F.; Elli, L.; Shahbazkhani, A.; Zebardast, J.; Rostami-Nejad, M. Prevalence of Non-Celiac Gluten Sensitivity in Patients with Refractory Functional Dyspepsia: A Randomized Double-blind Placebo Controlled Trial. Sci. Rep. 2020, 10, 2401. [Google Scholar] [CrossRef]
- Biesiekierski, J.R.; Newnham, E.D.; Irving, P.M.; Barrett, J.S.; Haines, M.; Doecke, J.D.; Shepherd, S.J.; Muir, J.G.; Gibson, P.R. Gluten Causes Gastrointestinal Symptoms in Subjects Without Celiac Disease: A Double-Blind Randomized Placebo-Controlled Trial. Am. J. Gastroenterol. 2011, 106, 508–514. [Google Scholar] [CrossRef] [PubMed]
- Capannolo, A.; Viscido, A.; Barkad, M.A.; Valerii, G.; Ciccone, F.; Melideo, D.; Frieri, G.; Latella, G. Non-Celiac Gluten Sensitivity among Patients Perceiving Gluten-Related Symptoms. Digestion 2015, 92, 8–13. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramedani, N.; Seidita, A.; Asri, N.; Azimirad, M.; Yadegar, A.; Jahani-Sherafat, S.; Sharifan, A.; Mansueto, P.; Carroccio, A.; Rostami-Nejad, M. The Gliadin Hydrolysis Capacity of B. longum, L. acidophilus, and L. plantarum and Their Protective Effects on Caco-2 Cells against Gliadin-Induced Inflammatory Responses. Nutrients 2023, 15, 2769. https://doi.org/10.3390/nu15122769
Ramedani N, Seidita A, Asri N, Azimirad M, Yadegar A, Jahani-Sherafat S, Sharifan A, Mansueto P, Carroccio A, Rostami-Nejad M. The Gliadin Hydrolysis Capacity of B. longum, L. acidophilus, and L. plantarum and Their Protective Effects on Caco-2 Cells against Gliadin-Induced Inflammatory Responses. Nutrients. 2023; 15(12):2769. https://doi.org/10.3390/nu15122769
Chicago/Turabian StyleRamedani, Najmeh, Aurelio Seidita, Nastaran Asri, Masoumeh Azimirad, Abbas Yadegar, Somayeh Jahani-Sherafat, Anousheh Sharifan, Pasquale Mansueto, Antonio Carroccio, and Mohammad Rostami-Nejad. 2023. "The Gliadin Hydrolysis Capacity of B. longum, L. acidophilus, and L. plantarum and Their Protective Effects on Caco-2 Cells against Gliadin-Induced Inflammatory Responses" Nutrients 15, no. 12: 2769. https://doi.org/10.3390/nu15122769
APA StyleRamedani, N., Seidita, A., Asri, N., Azimirad, M., Yadegar, A., Jahani-Sherafat, S., Sharifan, A., Mansueto, P., Carroccio, A., & Rostami-Nejad, M. (2023). The Gliadin Hydrolysis Capacity of B. longum, L. acidophilus, and L. plantarum and Their Protective Effects on Caco-2 Cells against Gliadin-Induced Inflammatory Responses. Nutrients, 15(12), 2769. https://doi.org/10.3390/nu15122769





