Vitamin B12—Multifaceted In Vivo Functions and In Vitro Applications
Abstract
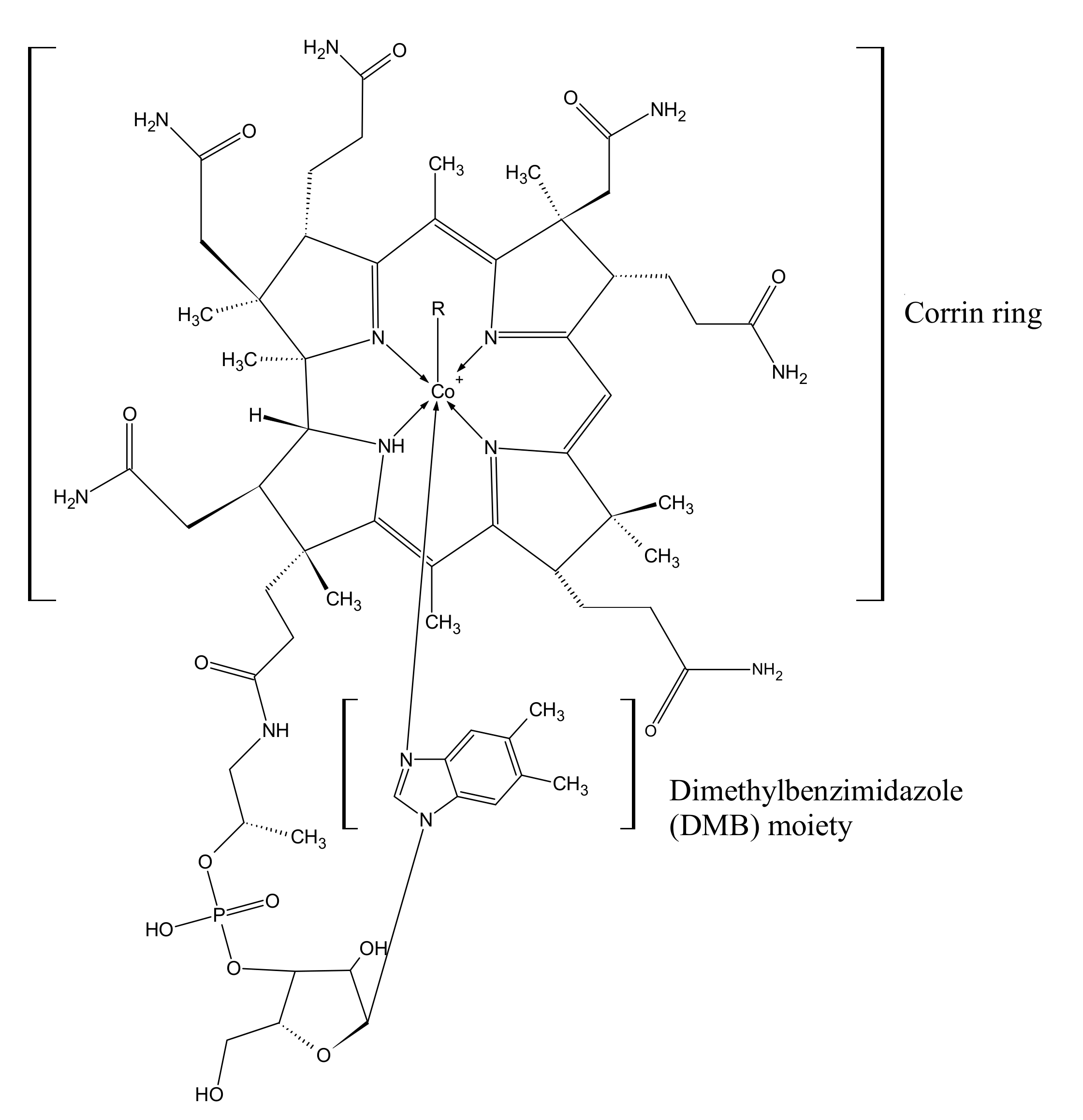
1. Chemical Structure and Biosynthesis of Cobalamin
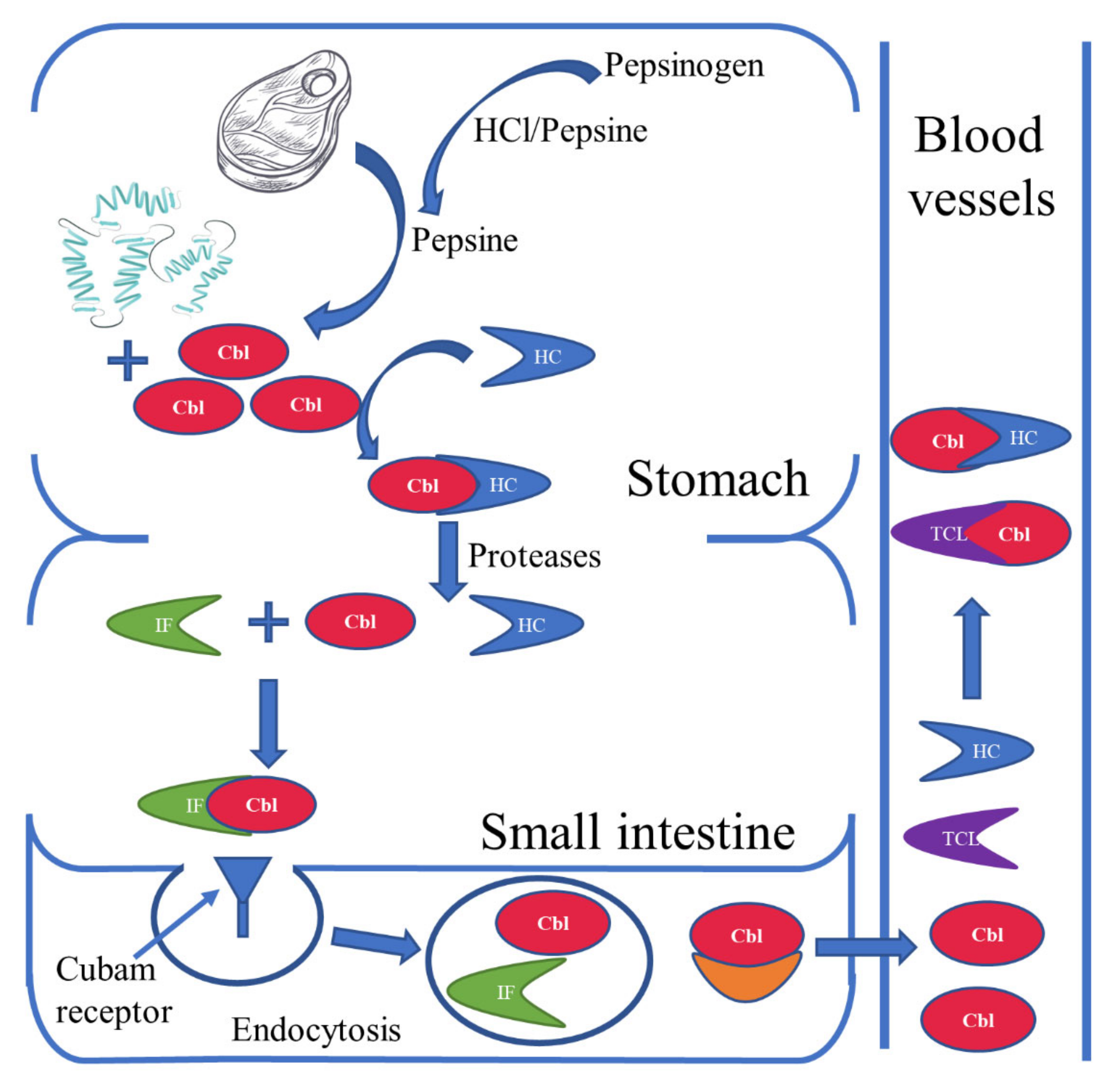
2. Sources and Absorption of Vitamin B12
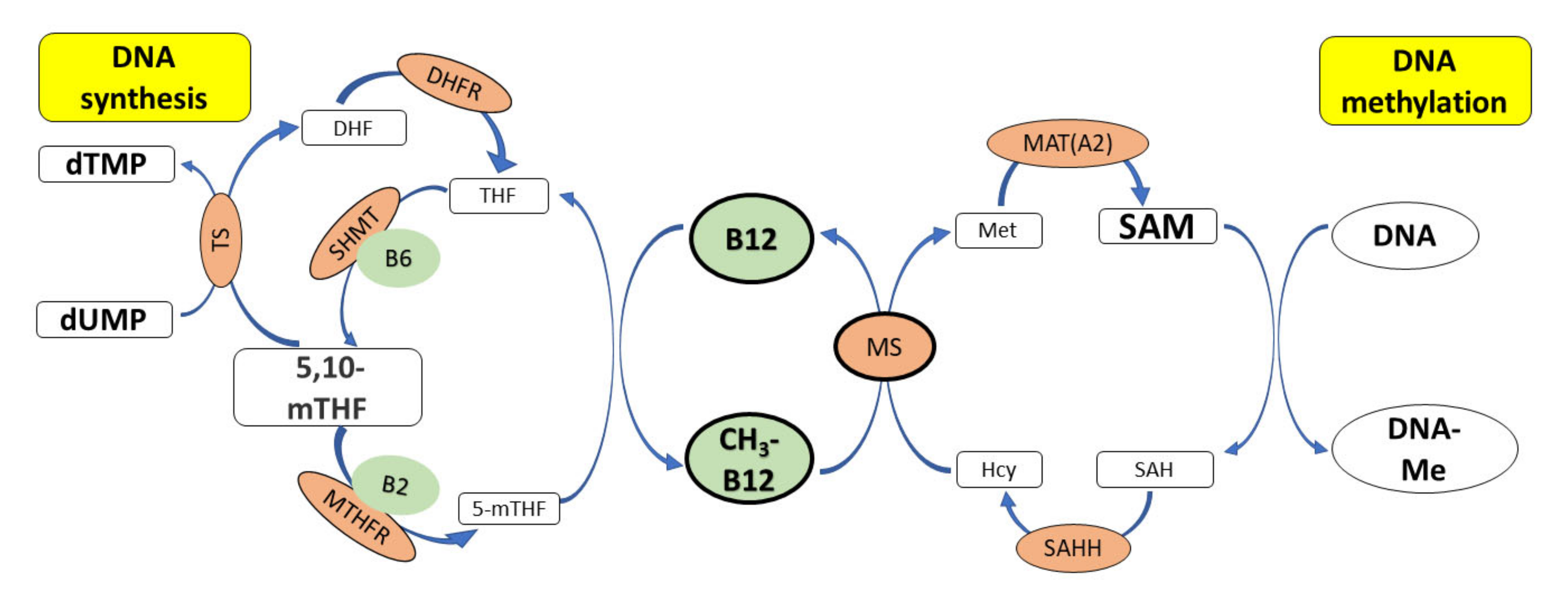
3. Cobalamin and Genome Stability
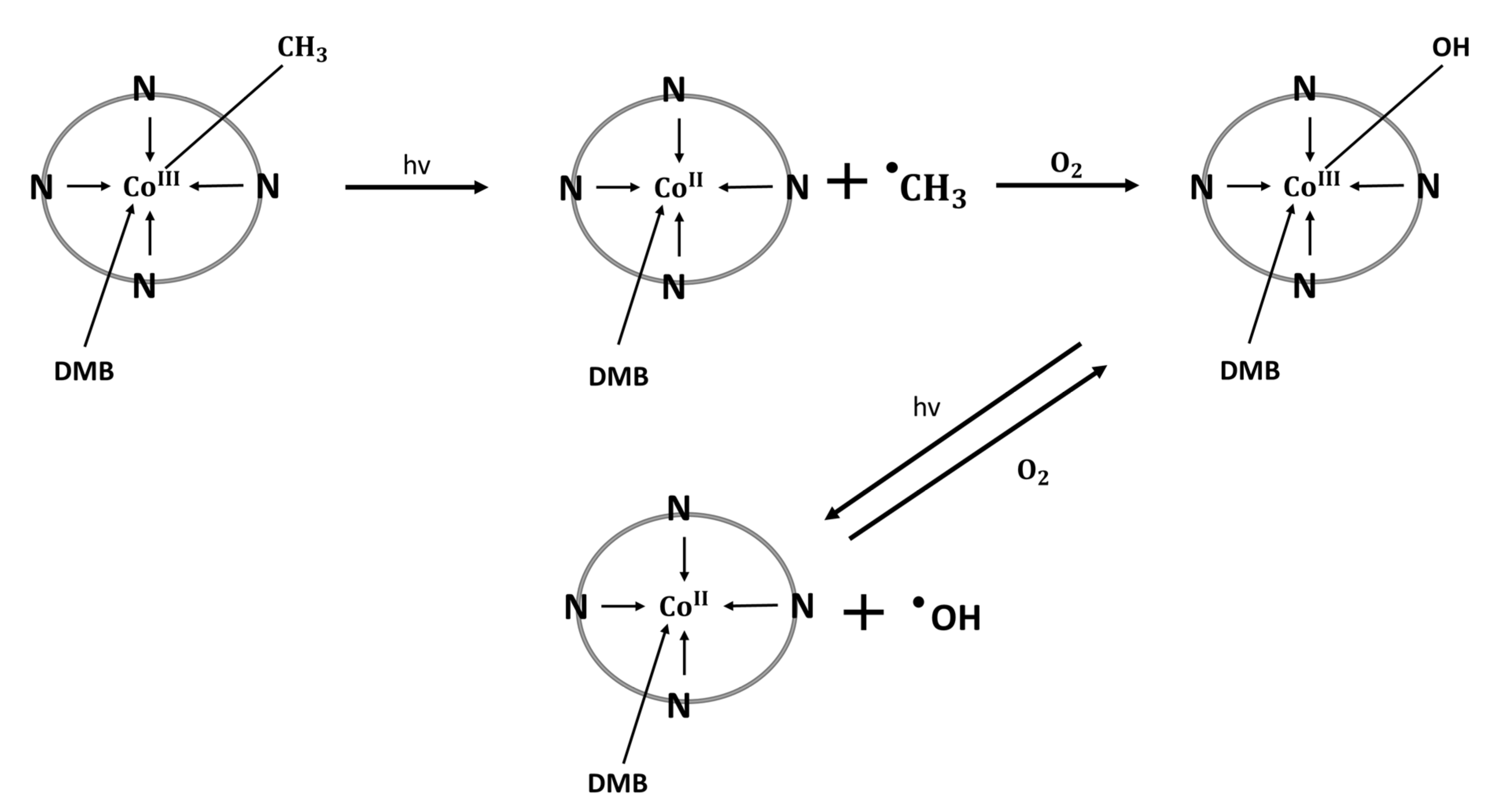
4. Cobalamin—Regulator of Inflammation and Oxidative Stress
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Froese, D.S.; Fowler, B.; Baumgartner, M.R. Vitamin B12, Folate, and the Methionine Remethylation Cycle-Biochemistry, Pathways, and Regulation. J. Inherit. Metab. Dis. 2019, 42, 673–685. [Google Scholar] [CrossRef] [PubMed]
- Gröber, U.; Kisters, K.; Schmidt, J. Neuroenhancement with Vitamin B12—Underestimated Neurological Significance. Nutrients 2013, 5, 5031–5045. [Google Scholar] [CrossRef] [PubMed]
- Okuda, K. Discovery of Vitamin B12 in the Liver and Its Absorption Factor in the Stomach: A Historical Review. J. Gastroenterol. Hepatol. 1999, 14, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.D.; Warren, M.J.; Refsum, H. Vitamin B12. Adv. Food Nutr. Res. 2018, 83, 215–279. [Google Scholar] [CrossRef] [PubMed]
- Moore, S.J.; Lawrence, A.D.; Biedendieck, R.; Deery, E.; Frank, S.; Howard, M.J.; Rigby, S.E.J.; Warren, M.J. Elucidation of the Anaerobic Pathway for the Corrin Component of Cobalamin (Vitamin B12). Proc. Natl. Acad. Sci. USA 2013, 110, 14906–14911. [Google Scholar] [CrossRef]
- Wan, Z.; Zheng, J.; Zhu, Z.; Sang, L.; Zhu, J.; Luo, S.; Zhao, Y.; Wang, R.; Zhang, Y.; Hao, K.; et al. Intermediate Role of Gut Microbiota in Vitamin B Nutrition and Its Influences on Human Health. Front. Nutr. 2022, 9, 1031502. [Google Scholar] [CrossRef]
- Lyon, P.; Strippoli, V.; Fang, B.; Cimmino, L. B Vitamins and One-Carbon Metabolism: Implications in Human Health and Disease. Nutrients 2020, 12, 2867. [Google Scholar] [CrossRef]
- Murray, R.K.; Granner, D.K.; Mayes, P.A.; Rodwell, V.W. Harper’s Illustrated Biochemistry, 31st ed.; Dalgleish, T., Williams, J.M.G., Golden, A.-M.J., Perkins, N., Barrett, L.F., Barnard, P.J., Au Yeung, C., Murphy, V., Elward, R., Tchanturia, K., et al., Eds.; Lange Medical Books/McGraw-Hill: Irvine, CA, USA, 2018; ISBN 978-1-25-983794-4. [Google Scholar]
- Temova Rakuša, Ž.; Roškar, R.; Hickey, N.; Geremia, S. Vitamin B12 in Foods, Food Supplements, and Medicines—A Review of Its Role and Properties with a Focus on Its Stability. Molecules 2022, 28, 240. [Google Scholar] [CrossRef]
- O’Leary, F.; Samman, S. Vitamin B12 in Health and Disease. Nutrients 2010, 2, 299–316. [Google Scholar] [CrossRef]
- Allen, L.H. Vitamin B-12. Adv. Nutr. 2012, 3, 54–55. [Google Scholar] [CrossRef]
- Gille, D.; Schmid, A. Vitamin B12 in Meat and Dairy Products. Nutr. Rev. 2015, 73, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.F.; Ross, S.K.; Mervyn, L.; Boddy, K.; King, P. Absorption of Cyanocobalamin, Coenzyme B12, Methylcobalamin, and Hydroxocobalamin at Different Dose Levels. Scand. J. Gastroenterol. 1971, 6, 249–252. [Google Scholar] [CrossRef] [PubMed]
- Allen, L.H.; Miller, J.W.; de Groot, L.; Rosenberg, I.H.; Smith, A.D.; Refsum, H.; Raiten, D.J. Biomarkers of Nutrition for Development (BOND): Vitamin B-12 Review. J. Nutr. 2018, 148, 1995S–2027S. [Google Scholar] [CrossRef]
- Allen, L.H. Bioavailability of Vitamin B12. Int. J. Vitam. Nutr. Res. 2010, 80, 330–335. [Google Scholar] [CrossRef] [PubMed]
- Doets, E.L.; In’t Veld, P.H.; Szczecińska, A.; Dhonukshe-Rutten, R.A.M.; Cavelaars, A.E.J.M.; Van’t Veer, P.; Brzozowska, A.; De Groot, L.C.P.G.M. Systematic Review on Daily Vitamin B12 Losses and Bioavailability for Deriving Recommendations on Vitamin B12 Intake with the Factorial Approach. Ann. Nutr. Metab. 2013, 62, 311–322. [Google Scholar] [CrossRef]
- Devi, S.; Pasanna, R.M.; Shamshuddin, Z.; Bhat, K.; Sivadas, A.; Mandal, A.K.; Kurpad, A.V. Measuring Vitamin B-12 Bioavailability with [13C]-Cyanocobalamin in Humans. Am. J. Clin. Nutr. 2020, 112, 1504–1515. [Google Scholar] [CrossRef]
- Doscherholmen, A.; McMahon, J.; Ripley, D. Inhibitory Effect of Eggs on Vitamin B12 Absorption: Description of a Simple Ovalbumin 57Co-Vitamin B12 Absorption Test. Br. J. Haematol. 1976, 33, 261–272. [Google Scholar] [CrossRef]
- Carmel, R. How I Treat Cobalamin (Vitamin B12) Deficiency. Blood 2008, 112, 2214–2221. [Google Scholar] [CrossRef]
- Shibata, K.; Hirose, J.; Fukuwatari, T. Relationship Between Urinary Concentrations of Nine Water-Soluble Vitamins and Their Vitamin Intakes in Japanese Adult Males. Nutr. Metab. Insights 2014, 7, 61. [Google Scholar] [CrossRef]
- Green, R.; Allen, L.H.; Bjørke-Monsen, A.L.; Brito, A.; Guéant, J.L.; Miller, J.W.; Molloy, A.M.; Nexo, E.; Stabler, S.; Toh, B.H.; et al. Vitamin B12 Deficiency. Nat. Rev. Dis. Prim. 2017, 3, 17040. [Google Scholar] [CrossRef]
- Obeid, R.; Heil, S.G.; Verhoeven, M.M.A.; van den Heuvel, E.G.H.M.; de Groot, L.C.P.G.M.; Eussen, S.J.P.M. Vitamin B12 Intake from Animal Foods, Biomarkers, and Health Aspects. Front. Nutr. 2019, 6, 93. [Google Scholar] [CrossRef]
- Rizzo, G.; Laganà, A.S.; Rapisarda, A.M.C.; La Ferrera, G.M.G.; Buscema, M.; Rossetti, P.; Nigro, A.; Muscia, V.; Valenti, G.; Sapia, F.; et al. Vitamin B12 among Vegetarians: Status, Assessment and Supplementation. Nutrients 2016, 8, 767. [Google Scholar] [CrossRef]
- Shipton, M.J.; Thachil, J. Vitamin B12 Deficiency—A 21st Century Perspective. Clin. Med. 2015, 15, 150. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on Nutrient Requirements and Dietary Intakes of Infants and Young Children in the European Union. EFSA J. 2013, 11, 3408. [Google Scholar] [CrossRef]
- Jarosz, M.; Wierzejska, R.; Rychlik, E.; Szponar, L.; Stoś, K.; Gielecińska, I.; Charzewska, J.; Pietraś, E.; Mojska, H.; Matczuk, E.; et al. Normy Żywienia Dla Populacji Polski i Ich Zastosowanie; Narodowy Instytut Zdrowia Publicznego—Państwowy Zakład Higieny: Warszawa, Poland, 2020; ISBN 978-83-65870-28-5.
- Golding, P.H. Holotranscobalamin (HoloTC, Active-B12) and Herbert’s Model for the Development of Vitamin B12 Deficiency: A Review and Alternative Hypothesis. Springerplus 2016, 5, 668. [Google Scholar] [CrossRef]
- Sobczyńska-Malefora, A.; Delvin, E.; McCaddon, A.; Ahmadi, K.R.; Harrington, D.J. Vitamin B12 Status in Health and Disease: A Critical Review. Diagnosis of Deficiency and Insufficiency—Clinical and Laboratory Pitfalls. Crit. Rev. Clin. Lab. Sci. 2021, 58, 399–429. [Google Scholar] [CrossRef] [PubMed]
- Thorpe, S.J.; Rigsby, P.; Roberts, G.; Lee, A.; Hamilton, M.; Craig, D. An International Standard for Holotranscobalamin (HoloTC): International Collaborative Study to Assign a HoloTC Value to the International Standard for Vitamin B12 and Serum Folate. Clin. Chem. Lab. Med. 2016, 54, 1467–1472. [Google Scholar] [CrossRef] [PubMed]
- Thorpe, S.J.; Heath, A.; Blackmore, S.; Lee, A.; Hamilton, M.; O’Broin, S.; Nelson, B.C.; Pfeiffer, C. International Standard for Serum Vitamin B(12) and Serum Folate: International Collaborative Study to Evaluate a Batch of Lyophilised Serum for B(12) and Folate Content. Clin. Chem. Lab. Med. 2007, 45, 380–386. [Google Scholar] [CrossRef]
- Carmel, R. Biomarkers of Cobalamin (Vitamin B-12) Status in the Epidemiologic Setting: A Critical Overview of Context, Applications, and Performance Characteristics of Cobalamin, Methylmalonic Acid, and Holotranscobalamin II. Am. J. Clin. Nutr. 2011, 94, 358S. [Google Scholar] [CrossRef]
- Devalia, V.; Hamilton, M.S.; Molloy, A.M. Guidelines for the Diagnosis and Treatment of Cobalamin and Folate Disorders. Br. J. Haematol. 2014, 166, 496–513. [Google Scholar] [CrossRef]
- Morkbak, A.L.; Hvas, A.M.; Milman, N.; Nexo, E. Holotranscobalamin Remains Unchanged during Pregnancy. Longitudinal Changes of Cobalamins and Their Binding Proteins during Pregnancy and Postpartum. Haematologica 2007, 92, 1711–1712. [Google Scholar] [CrossRef] [PubMed]
- Langan, R.C.; Goodbred, A.J. Vitamin B12 Deficiency: Recognition and Management. Am. Fam. Physician 2017, 96, 384–389. [Google Scholar] [PubMed]
- Hunt, A.; Harrington, D.; Robinson, S. Vitamin B12 Deficiency. BMJ 2014, 349, g5226. [Google Scholar] [CrossRef] [PubMed]
- Andrès, E.; Zulfiqar, A.A.; Vogel, T. State of the Art Review: Oral and Nasal Vitamin B12 Therapy in the Elderly. QJM 2020, 113, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Lacombe, V.; Lenaers, G.; Urbanski, G. Diagnostic and Therapeutic Perspectives Associated to Cobalamin-Dependent Metabolism and Transcobalamins’ Synthesis in Solid Cancers. Nutrients 2022, 14, 2058. [Google Scholar] [CrossRef] [PubMed]
- Huemer, M.; Diodato, D.; Schwahn, B.; Schiff, M.; Bandeira, A.; Benoist, J.F.; Burlina, A.; Cerone, R.; Couce, M.L.; Garcia-Cazorla, A.; et al. Guidelines for Diagnosis and Management of the Cobalamin-Related Remethylation Disorders CblC, CblD, CblE, CblF, CblG, CblJ and MTHFR Deficiency. J. Inherit. Metab. Dis. 2017, 40, 21–48. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Aryee, M.J.; Padyukov, L.; Daniele Fallin, M.; Hesselberg, E.; Runarsson, A.; Reinius, L.; Acevedo, N.; Taub, M.; Ronninger, M.; et al. Epigenome-Wide Association Data Implicate DNA Methylation as an Intermediary of Genetic Risk in Rheumatoid Arthritis HHS Public Access Author Manuscript. Nat. Biotechnol. 2013, 31, 142–147. [Google Scholar] [CrossRef]
- Rakyan, V.K.; Beyan, H.; Down, T.A.; Hawa, M.I.; Maslau, S.; Aden, D.; Daunay, A.; Busato, F.; Mein, C.A.; Manfras, B.; et al. Identification of Type 1 Diabetes–Associated DNA Methylation Variable Positions That Precede Disease Diagnosis. PLoS Genet. 2011, 7, 1002300. [Google Scholar] [CrossRef]
- Feinberg, A.P.; Irizarry, R.A.; Fradin, D.; Aryee, M.J.; Murakami, P.; Aspelund, T.; Eiriksdottir, G.; Harris, T.B.; Launer, L.; Gudnason, V.; et al. Personalized Epigenomic Signatures That Are Stable Over Time and Covary with Body Mass Index. Sci. Transl. Med. 2010, 2, 49ra67. [Google Scholar] [CrossRef]
- Li, Z.; Gueant-Rodriguez, R.M.; Quilliot, D.; Sirveaux, M.A.; Meyre, D.; Gueant, J.L.; Brunaud, L. Folate and Vitamin B12 Status Is Associated with Insulin Resistance and Metabolic Syndrome in Morbid Obesity. Clin. Nutr. 2018, 37, 1700–1706. [Google Scholar] [CrossRef]
- Igarai, T.; Nishimtjra, Y.; Obara, K.; Ono, S. Serum Vitamin B12 Levels of Patients with Rheumatoid Arthritis. Tohoku J. Exp. Med. 1978, 125, 287–301. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.F.; Morabia, A.; Carroll, J.; Gonzalez, K.; Fulda, K.; Kaur, M.; Vishwanatha, J.K.; Santella, R.M.; Cardarelli, R. Dietary Patterns Are Associated with Levels of Global Genomic DNA Methylation in a Cancer-Free Population. J. Nutr. 2011, 141, 1165. [Google Scholar] [CrossRef]
- Mandaviya, P.R.; Joehanes, R.; Brody, J.; Castillo-Fernandez, J.E.; Dekkers, K.F.; Do, A.N.; Graff, M.; Hänninen, I.K.; Tanaka, T.; De Jonge, E.A.L.; et al. Association of Dietary Folate and Vitamin B-12 Intake with Genome-Wide DNA Methylation in Blood: A Large-Scale Epigenome-Wide Association Analysis in 5841 Individuals. Am. J. Clin. Nutr. 2019, 110, 437–450. [Google Scholar] [CrossRef] [PubMed]
- Zafon, C.; Gil, J.; Pérez-González, B.; Jordà, M. DNA Methylation in Thyroid Cancer. Endocr. Relat. Cancer 2019, 26, R415–R439. [Google Scholar] [CrossRef] [PubMed]
- Halczuk, K.M.; Boguszewska, K.; Urbaniak, S.K.; Szewczuk, M.; Karwowski, B.T. 8-Oxo-7,8-Dihydro-2′-Deoxyguanosine (8-OxodG) And8-Hydroxy-2′-Deoxyguanosine (8-OHdG) as a Cause of Autoimmune Thyroid Diseases(AITD) during Pregnancy? Yale J. Biol. Med. 2020, 93, 501. [Google Scholar] [PubMed]
- Offringa, A.K.; Bourgonje, A.R.; Schrier, M.S.; Deth, R.C.; van Goor, H. Clinical Implications of Vitamin B12 as Redox-Active Cofactor. Trends Mol. Med. 2021, 27, 931–934. [Google Scholar] [CrossRef]
- Moreira, E.S.; Brasch, N.E.; Yun, J. Vitamin B12 Protects against Superoxide-Induced Cell Injury in Human Aortic Endothelial Cells. Free Radic. Biol. Med. 2011, 51, 876. [Google Scholar] [CrossRef]
- Birch, C.S.; Brasch, N.E.; McCaddon, A.; Williams, J.H.H. A Novel Role for Vitamin B12: Cobalamins Are Intracellular Antioxidants in Vitro. Free Radic. Biol. Med. 2009, 47, 184–188. [Google Scholar] [CrossRef]
- Van de Lagemaat, E.E.; de Groot, L.C.P.G.M.; van den Heuvel, E.G.H.M. Vitamin B12 in Relation to Oxidative Stress: A Systematic Review. Nutrients 2019, 11, 482. [Google Scholar] [CrossRef]
- Waly, M.I.; Ali, A.; Al-Nassri, A.; Al-Mukhaini, M.; Valliatte, J.; Al-Farsi, Y. Low Nourishment of B-Vitamins Is Associated with Hyperhomocysteinemia and Oxidative Stress in Newly Diagnosed Cardiac Patients. Exp. Biol. Med. 2016, 241, 46. [Google Scholar] [CrossRef]
- McCue, A.C.; Moreau, W.M.; Shell, T.A. Visible Light-Induced Radical Mediated DNA Damage. Photochem. Photobiol. 2018, 94, 551. [Google Scholar] [CrossRef] [PubMed]
- Shell, T.A.; Lawrence, D.S. A New Trick (Hydroxyl Radical Generation) for an Old Vitamin (B12). J. Am. Chem. Soc. 2011, 133, 2150. [Google Scholar] [CrossRef]
- Marvin, C.M.; Ding, S.; White, R.E.; Orlova, N.; Wang, Q.; Zywot, E.M.; Vickerman, B.M.; Harr, L.; Tarrant, T.K.; Dayton, P.A.; et al. On Command Drug Delivery via Cell-Conveyed Phototherapeutics. Small 2019, 15, e1901442. [Google Scholar] [CrossRef] [PubMed]
- Ikotun, O.F.; Marquez, B.V.; Fazen, C.H.; Kahkoska, A.R.; Doyle, R.P.; Lapi, S.E. Investigation of a Vitamin B12 Conjugate as a PET Imaging Probe. ChemMedChem 2014, 9, 1244–1251. [Google Scholar] [CrossRef]
- Kräutler, B. Biochemistry of B12-Cofactors in Human Metabolism. In Water Soluble Vitamins. Subcellular Biochemistry; Springer: New York, NY, USA, 2012; Volume 56, pp. 323–346. [Google Scholar]
- Hogenkamp, H.P.C. A Cyclic Nucleoside Derived from Coenzyme B12. J. Biol. Chem. 1963, 238, 477–480. [Google Scholar] [CrossRef]
- Hogenkamp, H.P.C. The Photolysis of Methylcobalamin. Biochemistry 1966, 5, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Brooks, P.J. The Case for 8,5′-Cyclopurine-2′-Deoxynucleosides as Endogenous DNA Lesions That Cause Neurodegeneration in Xeroderma Pigmentosum. Neuroscience 2007, 145, 1407–1417. [Google Scholar] [CrossRef]
- Mori, T.; Nakane, H.; Iwamoto, T.; Krokidis, M.G.; Chatgilialoglu, C.; Tanaka, K.; Kaidoh, T.; Hasegawa, M.; Sugiura, S. High Levels of Oxidatively Generated DNA Damage 8,5′-Cyclo-2′-Deoxyadenosine Accumulate in the Brain Tissues of Xeroderma Pigmentosum Group A Gene-Knockout Mice. DNA Repair 2019, 80, 52–58. [Google Scholar] [CrossRef]
- Fenech, M. Folate (Vitamin B9) and Vitamin B12 and Their Function in the Maintenance of Nuclear and Mitochondrial Genome Integrity. Mutat. Res. Fundam. Mol. Mech. Mutagen. 2012, 733, 21–33. [Google Scholar] [CrossRef]
- Blount, B.C.; Ames, B.N. DNA Damage in Folate Deficiency. Baillieres Clin. Haematol. 1995, 8, 461–478. [Google Scholar] [CrossRef]
- Blount, B.C.; Mack, M.M.; Wehr, C.M.; Macgregor, J.T.; Hiatt, R.A.; Wang, G.; Wickramasinghe, S.N.; Everson, R.B.; Ames, B.N. Folate Deficiency Causes Uracil Misincorporation into Human DNA and Chromosome Breakage: Implications for Cancer and Neuronal Damage. Proc. Natl. Acad. Sci. USA 1997, 94, 3290–3295. [Google Scholar] [CrossRef]
- Pieroth, R.; Paver, S.; Day, S.; Lammersfeld, C. Folate and Its Impact on Cancer Risk. Curr. Nutr. Rep. 2018, 7, 70. [Google Scholar] [CrossRef] [PubMed]
- Vollset, S.E.; Igland, J.; Jenab, M.; Fredriksen, Å.; Meyer, K.; Eussen, S.; Gjessing, H.K.; Ueland, P.M.; Pera, G.; Sala, N.; et al. The Association of Gastric Cancer Risk with Plasma Folate, Cobalamin, and Methylenetetrahydrofolate Reductase Polymorphisms in the European Prospective Investigation into Cancer and Nutrition. Cancer Epidemiol. Biomark. Prev. 2007, 16, 2416–2424. [Google Scholar] [CrossRef]
- Fenech, M.; Aitken, C.; Rinaldi, J. Folate, Vitamin B12, Homocysteine Status and DNA Damage in Young Australian Adults. Carcinogenesis 1998, 19, 1163–1171. [Google Scholar] [CrossRef]
- Fenech, M.F.; Dreosti, I.E.; Rinaldi, J.R. Folate, Vitamin B12, Homocysteine Status and Chromosome Damage Rate in Lymphocytes of Older Men. Carcinogenesis 1997, 18, 1329–1336. [Google Scholar] [CrossRef] [PubMed]
- Frnech, M.; Rinaldi, J. The Relationship between Micronuclei in Human Lymphocytes and Plasma Levels of Vitamin C, Vitamin E, Vitamin B12 and Folic Acid. Carcinogenesis 1994, 15, 1405–1411. [Google Scholar] [CrossRef]
- Milić, M.; Rozgaj, R.; Kašuba, V.; Oreščanin, V.; Balija, M.; Jukić, I. Correlation between Folate and Vitamin B12 and Markers of DNA Stability in Healthy Men: Preliminary Results. Acta Biochem. Pol. 2010, 57, 339–345. [Google Scholar] [CrossRef]
- Minnet, C.; Koc, A.; Aycicek, A.; Kocyigit, A. Vitamin B12 Treatment Reduces Mononuclear DNA Damage. Pediatr. Int. 2011, 53, 1023–1027. [Google Scholar] [CrossRef]
- Firat, C.K.; Ozkan, B.N.; Guler, E.M. Beneficial Effects of Vitamin B12 Treatment in Pediatric Patients Diagnosed with Vitamin B12 Deficiency Regarding Total-Native Thiol, Oxidative Stress, and Mononuclear Leukocyte DNA Damage. Free Radic. Res. 2022, 56, 631–639. [Google Scholar] [CrossRef]
- Choi, S.W.; Friso, S.; Ghandour, H.; Bagley, P.J.; Selhub, J.; Mason, J.B. Vitamin B-12 Deficiency Induces Anomalies of Base Substitution and Methylation in the DNA of Rat Colonic Epithelium. J. Nutr. 2004, 134, 750–755. [Google Scholar] [CrossRef]
- Fernàndez-Roig, S.; Lai, S.C.; Murphy, M.M.; Fernandez-Ballart, J.; Quadros, E.V. Vitamin B12 Deficiency in the Brain Leads to DNA Hypomethylation in the TCblR/CD320 Knockout Mouse. Nutr. Metab. 2012, 9, 41. [Google Scholar] [CrossRef] [PubMed]
- Padmanabhan, S.; Waly, M.I.; Taranikanti, V.; Guizani, N.; Ali, A.; Rahman, M.S.; Al-Attabi, Z.; Al-Malky, R.N.; Al-Maskari, S.N.M.; Al-Ruqaishi, B.R.S.; et al. Folate/Vitamin B12 Supplementation Combats Oxidative Stress-Associated Carcinogenesis in a Rat Model of Colon Cancer. Nutr. Cancer 2019, 71, 100–110. [Google Scholar] [CrossRef] [PubMed]
- Acharyya, N.; Deb, B.; Chattopadhyay, S.; Maiti, S. Arsenic-Induced Antioxidant Depletion, Oxidative DNA Breakage, and Tissue Damages Are Prevented by the Combined Action of Folate and Vitamin B12. Biol. Trace Elem. Res. 2015, 168, 122–132. [Google Scholar] [CrossRef]
- Alzoubi, K.; Khabour, O.; Khader, M.; Mhaidat, N.; Al-Azzam, S. Evaluation of Vitamin B12 Effects on DNA Damage Induced by Paclitaxel. Drug Chem. Toxicol. 2014, 37, 276–280. [Google Scholar] [CrossRef]
- Alzoubi, K.; Khabour, O.; Hussain, N.; Al-azzam, S.; Mhaidat, N. Evaluation of Vitamin B12 Effects on DNA Damage Induced by Pioglitazone. Mutat. Res. 2012, 748, 48–51. [Google Scholar] [CrossRef]
- Palmer, A.M.; Kamynina, E.; Field, M.S.; Stover, P.J. Folate Rescues Vitamin B12 Depletion-Induced Inhibition of Nuclear Thymidylate Biosynthesis and Genome Instability. Proc. Natl. Acad. Sci. USA 2017, 114, E4095–E4102. [Google Scholar] [CrossRef]
- Jeon, Y.M.; Kwon, Y.; Lee, S.; Kim, S.; Jo, M.; Lee, S.; Kim, S.R.; Kim, K.; Kim, H.J. Vitamin B12 Reduces TDP-43 Toxicity by Alleviating Oxidative Stress and Mitochondrial Dysfunction. Antioxidants 2021, 11, 82. [Google Scholar] [CrossRef]
- Chan, W.; Almasieh, M.; Catrinescu, M.M.; Levin, L.A. Cobalamin-Associated Superoxide Scavenging in Neuronal Cells Is a Potential Mechanism for Vitamin B12-Deprivation Optic Neuropathy. Am. J. Pathol. 2018, 188, 160–172. [Google Scholar] [CrossRef]
- Suarez-Moreira, E.; Yun, J.; Birch, C.S.; Williams, J.H.H.; McCaddon, A.; Brasch, N.E. Vitamin B(12) and Redox Homeostasis: Cob(II)Alamin Reacts with Superoxide at Rates Approaching Superoxide Dismutase (SOD). J. Am. Chem. Soc. 2009, 131, 15078–15079. [Google Scholar] [CrossRef]
- Karamshetty, V.; Acharya, J.D.; Ghaskadbi, S.; Goel, P. Mathematical Modeling of Glutathione Status in Type 2 Diabetics with Vitamin B12 Deficiency. Front. Cell Dev. Biol. 2016, 4, 16. [Google Scholar] [CrossRef] [PubMed]
- Ouma, P.A.; Mwaeni, V.K.; Amwayi, P.W.; Isaac, A.O.; Nyariki, J.N. Calcium Carbide-Induced Derangement of Hematopoiesis and Organ Toxicity Ameliorated by Cyanocobalamin in a Mouse Model. Lab. Anim. Res. 2022, 38, 26. [Google Scholar] [CrossRef]
- Hoffman, M. Hypothesis: Hyperhomocysteinemia Is an Indicator of Oxidant Stress. Med. Hypotheses 2011, 77, 1088–1093. [Google Scholar] [CrossRef]
- Gospodarczyk, A.; Marczewski, K.; Gospodarczyk, N.; Widuch, M.; Tkocz, M.; Zalejska-Fiolka, J. Homocysteine and Cardiovascular Disease—A Current Review. Wiadomosci. Lek. 2022, 75, 2862–2866. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Yang, H.; Li, N.; Li, C.; Liang, W.; Zhang, X. Increased Serum Homocysteine in First Episode and Drug-Naïve Individuals with Schizophrenia: Sex Differences and Correlations with Clinical Symptoms. BMC Psychiatry 2022, 22, 759. [Google Scholar] [CrossRef] [PubMed]
- Shen, Z.; Zhang, Z.; Zhao, W. Relationship between Plasma Homocysteine and Chronic Kidney Disease in US Patients with Type 2 Diabetes Mellitus: A Cross-Sectional Study. BMC Nephrol. 2022, 23, 419. [Google Scholar] [CrossRef] [PubMed]
- Bouguerra, K.; Tazir, M.; Melouli, H.; Khelil, M. The Methylenetetrahydrofolate Reductase C677T and A1298C Genetic Polymorphisms and Plasma Homocysteine in Alzheimer’s Disease in an Algerian Population. Int. J. Neurosci. 2022, 29, 1–6. [Google Scholar] [CrossRef]
- Tyagi, N.; Sedoris, K.C.; Steed, M.; Ovechkin, A.V.; Moshal, K.S.; Tyagi, S.C. Mechanisms of Homocysteine-Induced Oxidative Stress. Am. J. Physiol. Heart Circ. Physiol. 2005, 289, 2649–2656. [Google Scholar] [CrossRef]
- Politis, A.; Olgiati, P.; Malitas, P.; Albani, D.; Signorini, A.; Polito, L.; de Mauro, S.; Zisaki, A.; Piperi, C.; Stamouli, E.; et al. Vitamin B12 Levels in Alzheimer’s Disease: Association with Clinical Features and Cytokine Production. J. Alzheimers Dis. 2010, 19, 481–488. [Google Scholar] [CrossRef]
- Zuo, L.; Prather, E.R.; Stetskiv, M.; Garrison, D.E.; Meade, J.R.; Peace, T.I.; Zhou, T. Inflammaging and Oxidative Stress in Human Diseases: From Molecular Mechanisms to Novel Treatments. Int. J. Mol. Sci. 2019, 20, 4472. [Google Scholar] [CrossRef] [PubMed]
- Matsuzawa, A.; Ichijo, H. Redox Control of Cell Fate by MAP Kinase: Physiological Roles of ASK1-MAP Kinase Pathway in Stress Signaling. Biochim. Biophys. Acta 2008, 1780, 1325–1336. [Google Scholar] [CrossRef]
- Gupta, N.; Verma, K.; Nalla, S.; Kulshreshtha, A.; Lall, R.; Prasad, S. Free Radicals as a Double-Edged Sword: The Cancer Preventive and Therapeutic Roles of Curcumin. Molecules 2020, 25, 5390. [Google Scholar] [CrossRef]
- Sainz, R.M.; Lombo, F.; Mayo, J.C. Radical Decisions in Cancer: Redox Control of Cell Growth and Death. Cancers 2012, 4, 442–474. [Google Scholar] [CrossRef] [PubMed]
- Schieber, M.; Chandel, N.S. ROS Function in Redox Signaling and Oxidative Stress. Curr. Biol. 2014, 24, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Guéant, J.L.; Caillerez-Fofou, M.; Battaglia-Hsu, S.; Alberto, J.M.; Freund, J.N.; Dulluc, I.; Adjalla, C.; Maury, F.; Merle, C.; Nicolas, J.P.; et al. Molecular and Cellular Effects of Vitamin B12 in Brain, Myocardium and Liver through Its Role as Co-Factor of Methionine Synthase. Biochimie 2013, 95, 1033–1040. [Google Scholar] [CrossRef] [PubMed]
- Brasky, T.M.; Ray, R.M.; Navarro, S.L.; Schenk, J.M.; Newton, A.M.; Neuhouser, M.L. Supplemental One-Carbon Metabolism Related B Vitamins and Lung Cancer Risk in the Women’s Health Initiative. Int. J. Cancer 2020, 147, 1374–1384. [Google Scholar] [CrossRef] [PubMed]
- Arendt, J.F.H.; Sørensen, H.T.; Horsfall, L.J.; Petersen, I. Elevated Vitamin B12 Levels and Cancer Risk in UK Primary Care: A THIN Database Cohort Study. Cancer Epidemiol. Biomark. Prev. 2019, 28, 814–821. [Google Scholar] [CrossRef]
- Cheng, W.W.; Wang, Z.K.; Shangguan, H.F.; Zhu, Q.; Zhang, H.Y. Are Vitamins Relevant to Cancer Risks? A Mendelian Randomization Investigation. Nutrition 2020, 78, 110870. [Google Scholar] [CrossRef]
- Flodh, H.; Ullberg, S. Accumulation of Labelled Vitamin B12 in Some Transplanted Tumours. Int. J. Cancer 1968, 3, 694–699. [Google Scholar] [CrossRef]
- Pettenuzzo, A.; Pigot, R.; Ronconi, L. Vitamin B12–Metal Conjugates for Targeted Chemotherapy and Diagnosis: Current Status and Future Prospects. Eur. J. Inorg. Chem. 2017, 2017, 1625–1638. [Google Scholar] [CrossRef]
- Abdulwahhab, R.Q.; Ali Alabdali, S.M. Study of the Protective Effects of Cyanocobalamin on Methotrexate Induced Nephrotoxicity in Rats. F1000Research 2022, 11, 1012. [Google Scholar] [CrossRef]
- Solomon, B.; Bunn, P.A. Clinical Activity of Pemetrexed: A Multitargeted Antifolate Anticancer Agent. Future Oncol. 2005, 1, 733–746. [Google Scholar] [CrossRef]
- Atoum, M.F.; Alzoughool, F.E.; Al-Mazaydeh, Z.A.; Rammaha, M.S.; Tahtamouni, L.H. Vitamin B12 Enhances the Antitumor Activity of 1,25-Dihydroxyvitamin D3 via Activation of Caspases and Targeting Actin Cytoskeleton. Tumour Biol. 2022, 44, 17–35. [Google Scholar] [CrossRef] [PubMed]
- Misra, U.K.; Kalita, J.; Singh, S.K.; Rahi, S.K. Oxidative Stress Markers in Vitamin B12 Deficiency. Mol. Neurobiol. 2017, 54, 1278–1284. [Google Scholar] [CrossRef] [PubMed]
- Demirtas, M.S.; Erdal, H. Evaluation of Thiol Disulfide Balance in Adolescents with Vitamin B12 Deficiency. Ital. J. Pediatr. 2023, 49, 3. [Google Scholar] [CrossRef] [PubMed]
- Collin, S.M.; Metcalfe, C.; Refsum, H.; Lewis, S.J.; Zuccolo, L.; Smith, G.D.; Chen, L.; Harris, R.; Davis, M.; Marsden, G.; et al. Circulating Folate, Vitamin B12, Homocysteine, Vitamin B12 Transport Proteins, and Risk of Prostate Cancer: A Case-Control Study, Systematic Review, and Meta-Analysis. Cancer Epidemiol. Biomark. Prev. 2010, 19, 1632–1642. [Google Scholar] [CrossRef] [PubMed]
- Fanidi, A.; Carreras-Torres, R.; Larose, T.L.; Yuan, J.M.; Stevens, V.L.; Weinstein, S.J.; Albanes, D.; Prentice, R.; Pettinger, M.; Cai, Q.; et al. Is High Vitamin B12 Status a Cause of Lung Cancer? Int. J. Cancer 2019, 145, 1499–1503. [Google Scholar] [CrossRef]
- Ermens, A.A.M.; Vlasveld, L.T.; Lindemans, J. Significance of Elevated Cobalamin (Vitamin B12) Levels in Blood. Clin. Biochem. 2003, 36, 585–590. [Google Scholar] [CrossRef]
- Lin, C.Y.; Kuo, C.S.; Lu, C.L.; Wu, M.Y.; Huang, R.F.S. Elevated Serum Vitamin B(12) Levels in Association with Tumor Markers as the Prognostic Factors Predictive for Poor Survival in Patients with Hepatocellular Carcinoma. Nutr. Cancer 2010, 62, 190–197. [Google Scholar] [CrossRef]
- World Cancer Research Fund; American Institute for Cancer Research. Continuous Update Project Expert Report 2018. Recommendations and Public Health and Policy Implications; World Cancer Research Fund: London, UK; American Institute for Cancer Research: Washington, DC, USA, 2018. [Google Scholar]
| EFSA (2015) | WHO/FAO (2004) | SCF (1993) | Poland | |
|---|---|---|---|---|
| Age (years) | >18 | ≥19 | ≥19 | ≥19 |
| Men + Women | ||||
| Reference value (μg/day) | 4.0 | 2.4 | 1.4 | 2.4 |
| Pregnant Women | ||||
| Reference value (μg/day) | 4.5 | 2.6 | 1.6 | 2.6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Halczuk, K.; Kaźmierczak-Barańska, J.; Karwowski, B.T.; Karmańska, A.; Cieślak, M. Vitamin B12—Multifaceted In Vivo Functions and In Vitro Applications. Nutrients 2023, 15, 2734. https://doi.org/10.3390/nu15122734
Halczuk K, Kaźmierczak-Barańska J, Karwowski BT, Karmańska A, Cieślak M. Vitamin B12—Multifaceted In Vivo Functions and In Vitro Applications. Nutrients. 2023; 15(12):2734. https://doi.org/10.3390/nu15122734
Chicago/Turabian StyleHalczuk, Krzysztof, Julia Kaźmierczak-Barańska, Bolesław T. Karwowski, Aleksandra Karmańska, and Marcin Cieślak. 2023. "Vitamin B12—Multifaceted In Vivo Functions and In Vitro Applications" Nutrients 15, no. 12: 2734. https://doi.org/10.3390/nu15122734
APA StyleHalczuk, K., Kaźmierczak-Barańska, J., Karwowski, B. T., Karmańska, A., & Cieślak, M. (2023). Vitamin B12—Multifaceted In Vivo Functions and In Vitro Applications. Nutrients, 15(12), 2734. https://doi.org/10.3390/nu15122734







