The Impact of Anorexia Nervosa and the Basis for Non-Pharmacological Interventions
Abstract
1. Introduction
- Family therapy (changing and solving family problems to cure AN)
- Family based treatment (parents’ involvement in adolescents’ food consumption)
- Joint family therapy (collaborative work among adolescents, the entire family and a therapist with monitoring of the family’s emotional issues)
- Behavioral family system therapy (three-step behavioral weight gain program with family involvement)
- Cognitive-behavioral therapy (modifications of irrational beliefs and problematic eating behavior)
- Specialist supportive clinical management (Education with supportive therapy)
2. Materials and Methods
3. The Impact of Anorexia Nervosa on an Individual’s Life
3.1. Psychological Profile
3.2. Family and Anorexia
3.3. Social Context and Anorexia
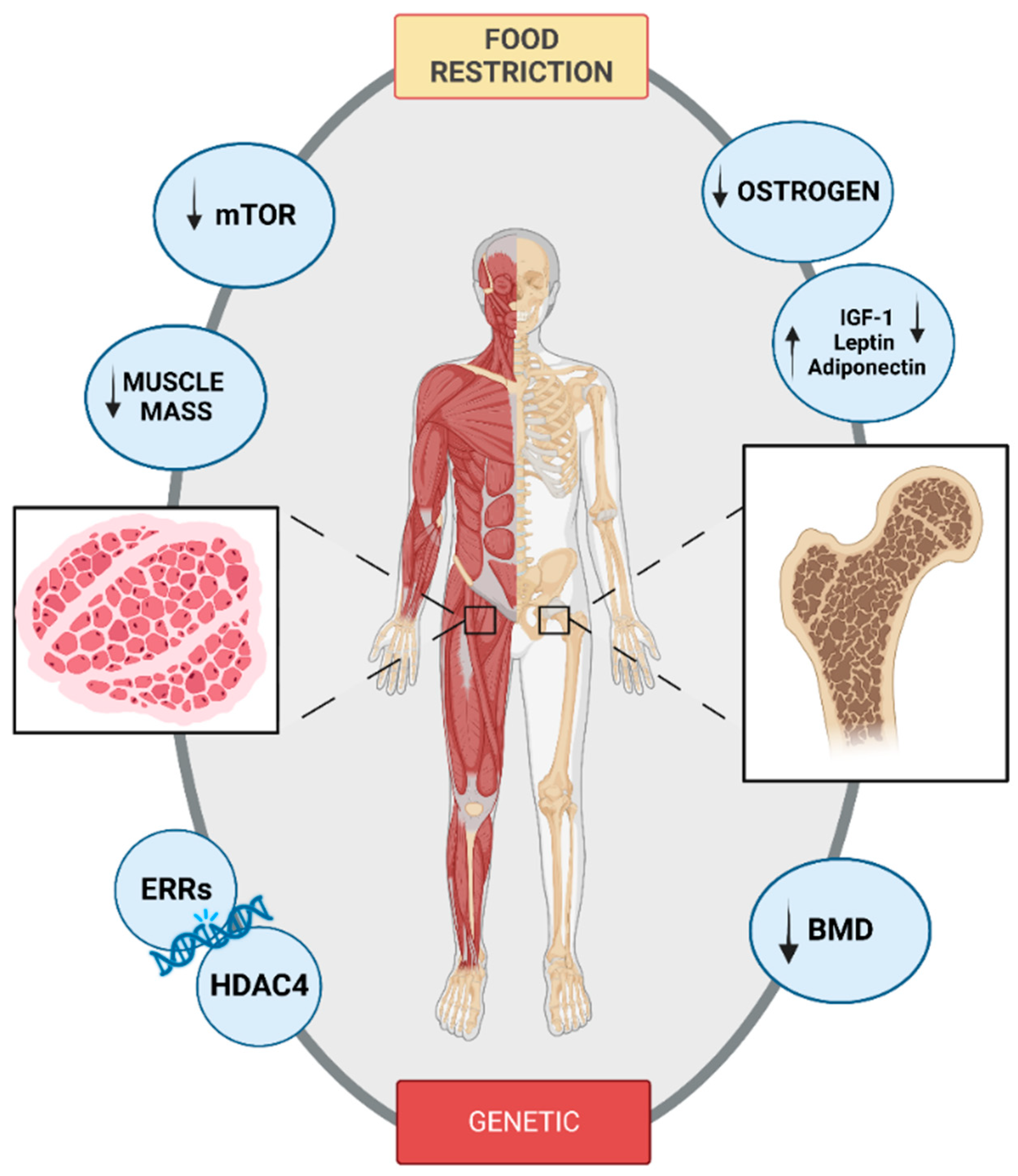
3.4. Bone and Muscular Implications of Anorexia
3.5. Microbiota and Anorexia
3.5.1. Diversity and Microbial Metabolites in Anorexia Nervosa
3.5.2. Psychopathology in Anorexia Nervosa
3.5.3. Gut Microbiome Rehabilitation
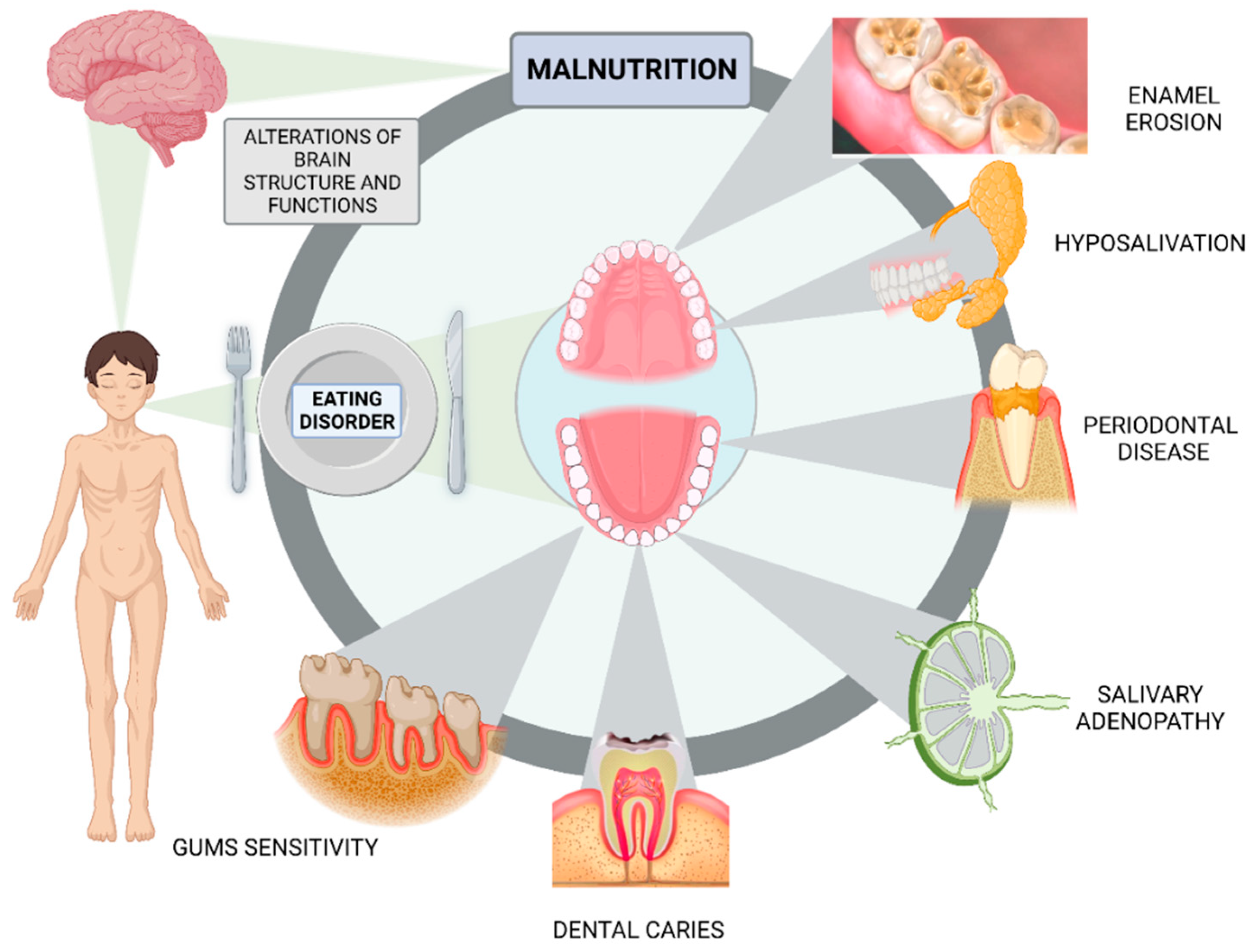
3.6. Dental Health in Anorexia
4. Non-Pharmacological Interventions in Anorexia Nervosa
4.1. Nutritional Interventions
4.2. Physical Activity Interventions
4.3. Psychological Interventions
4.4. Psychosocial Interventions
4.5. Physical Therapy Interventions
5. Practical Applications
- -
- Understanding the individual profile that characterizes each anorexia patient may help in providing a more effective treatment and preventive strategies.
- -
- Family support and involvement during the treatment of anorexia patients can have a positive impact on the outcome.
- -
- Managing the relationship between the patient and social media will help avoid setbacks in the treatment and avoid any body image influence during and after the treatment.
- -
- Eating disorders might be linked to mutations in the transcription factor ESRR α and the transcriptional repressor histone deacetylase 4 (HDAC4). Even so, studies have not proven the relationship between a specific gene and the development of eating disorders.
- -
- A diet rich in fiber can improve the diversity of the gut microbiome which is linked to weight gain.
- -
- Controlled and supervised physical exercise could help improve the composition of the intestinal microbiota and improve eating disorder symptomatology. It should be included in the patient’s treatment plan.
- -
- An analysis of the intestinal microbiota in patients with AN could be useful for nutritional rehabilitation.
- -
- Oral care should be included in the treatment of patients with AN, since they have a higher risk of developing gingival and dental disease.
- -
- A healthy mouth and smile could help improve the low self-esteem that patients with AN usually present with.
- -
- A good clinic history and physical examination are crucial in treating patients with AN.
- -
- The rate of weight gain and energy requirements should be calculated individually for each patient.
- -
- Nutritional education is a key aspect in treating patients with this disorder.
- -
- Each patient needs an individualized nutritional plan.
- -
- AN can be accompanied by other psychological disorders, depression being the most common one.
- -
- The most used psychological therapy for patients with AN is cognitive behavioral therapy.
- -
- Physical therapy can help ease back pain or increase the tone of hypotonic muscles.
- -
- Massages can help decrease anxiety and stress, promoting relaxation.
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Knekt, P.; Lindfors, O.; Laaksonen, M.A.; Raitasalo, R.; Haaramo, P.; Järvikoski, A.; Group, H.P.S. Effectiveness of Short-Term and Long-Term Psychotherapy on Work Ability and Functional Capacity—A Randomized Clinical Trial on Depressive and Anxiety Disorders. J. Affect. Disord. 2008, 107, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Van Eeden, A.E.; van Hoeken, D.; Hoek, H.W. Incidence, Prevalence and Mortality of Anorexia Nervosa and Bulimia Nervosa. Curr. Opin. Psychiatry 2021, 34, 515. [Google Scholar] [CrossRef] [PubMed]
- Staudt, M.A.; Rojo, N.M.; Ojeda, G.A. Trastornos de La Conducta Alimentaria: Anorexia Nerviosa. Revisión Bibliográfica. Rev. Posgrado VI Cátedra Med. Rev. En Internet 2006, 156, 24–30. [Google Scholar]
- Zipfel, S.; Wild, B.; Groß, G.; Friederich, H.-C.; Teufel, M.; Schellberg, D.; Giel, K.E.; de Zwaan, M.; Dinkel, A.; Herpertz, S. Focal Psychodynamic Therapy, Cognitive Behaviour Therapy, and Optimised Treatment as Usual in Outpatients with Anorexia Nervosa (ANTOP Study): Randomised Controlled Trial. Lancet 2014, 383, 127–137. [Google Scholar] [CrossRef]
- Griffiths, S.; Murray, S.B.; Bentley, C.; Gratwick-Sarll, K.; Harrison, C.; Mond, J.M. Sex Differences in Quality of Life Impairment Associated with Body Dissatisfaction in Adolescents. J. Adolesc. Health 2017, 61, 77–82. [Google Scholar] [CrossRef]
- Gan, J.K.E.; Wu, V.X.; Chow, G.; Chan, J.K.Y.; Klainin-Yobas, P. Effectiveness of Non-Pharmacological Interventions on Individuals with Anorexia Nervosa: A Systematic Review and Meta-Analysis. Patient Educ. Couns. 2022, 105, 44–55. [Google Scholar] [CrossRef]
- Mikhaylova, O.; Dokuka, S. Anorexia and Young Womens’ Personal Networks: Size, Structure, and Kinship. Front. Psychol. 2022, 13, 848774. [Google Scholar] [CrossRef]
- Breton, J.; Déchelotte, P.; Ribet, D. Intestinal Microbiota and Anorexia Nervosa. Clin. Nutr. Exp. 2019, 28, 11–21. [Google Scholar] [CrossRef]
- Cucarella, J.O.; Tortajada, R.E.; Moreno, L.R. Neuropsicología y Anorexia Nerviosa. Hallazgos Cognitivos y Radiológicos. Neurología 2012, 27, 504–510. [Google Scholar] [CrossRef]
- Reville, M.-C.; O’Connor, L.; Frampton, I. Literature Review of Cognitive Neuroscience and Anorexia Nervosa. Curr. Psychiatry Rep. 2016, 18, 18. [Google Scholar] [CrossRef]
- Kaye, W. Neurobiology of Anorexia and Bulimia Nervosa. Physiol. Behav. 2008, 94, 121–135. [Google Scholar] [CrossRef] [PubMed]
- Casper, R.C. Personality Features of Women with Good Outcome from Restricting Anorexia Nervosa. Psychosom. Med. 1990, 52, 156–170. [Google Scholar] [CrossRef] [PubMed]
- Srinivasagam, N.M.; Kaye, W.H.; Plotnicov, K.H.; Greeno, C.; Weltzin, T.E.; Rao, R. Persistent Perfectionism, Symmetry, and Exactness after Long-Term Recovery from Anorexia Nervosa. Am. J. Psychiatry 1995, 152, 1630–1634. [Google Scholar] [PubMed]
- Strober, M. Personality and Symptomatological Features in Young, Nonchronic Anorexia Nervosa Patients. J. Psychosom. Res. 1980, 24, 353–359. [Google Scholar] [CrossRef]
- Miles, S.; Nedeljkovic, M.; Phillipou, A. Can Cognitive Flexibility and Clinical Perfectionism Be Used to Identify People with Anorexia Nervosa? J. Clin. Med. 2022, 11, 1954. [Google Scholar] [CrossRef]
- Shafran, R.; Lee, M.; Fairburn, C.G. Clinical Perfectionism: A Case Report. Behav. Cogn. Psychother. 2004, 32, 353–357. [Google Scholar] [CrossRef]
- Cederlöf, M.; Thornton, L.M.; Baker, J.; Lichtenstein, P.; Larsson, H.; Rück, C.; Bulik, C.M.; Mataix-Cols, D. Etiological Overlap between Obsessive-compulsive Disorder and Anorexia Nervosa: A Longitudinal Cohort, Multigenerational Family and Twin Study. World Psychiatry 2015, 14, 333–338. [Google Scholar] [CrossRef]
- Swinbourne, J.; Hunt, C.; Abbott, M.; Russell, J.; St Clare, T.; Touyz, S. The Comorbidity between Eating Disorders and Anxiety Disorders: Prevalence in an Eating Disorder Sample and Anxiety Disorder Sample. Aust. N. Z. J. Psychiatry 2012, 46, 118–131. [Google Scholar] [CrossRef]
- Pinto, A.; Mancebo, M.C.; Eisen, J.L.; Pagano, M.E.; Rasmussen, S.A. The Brown Longitudinal Obsessive Compulsive Study: Clinical Features and Symptoms of the Sample at Intake. J. Clin. Psychiatry 2006, 67, 703–711. [Google Scholar] [CrossRef]
- Adamson, J.; Ozenc, C.; Baillie, C.; Tchanturia, K. Self-Esteem Group: Useful Intervention for Inpatients with Anorexia Nervosa? Brain Sci. 2019, 9, 12. [Google Scholar] [CrossRef]
- Yellowlees, A.; Forbes, C. Working with Eating Disorders and Self-Esteem; Folens: Dublin, Ireland, 1997; ISBN 1854672800. [Google Scholar]
- Surgenor, L.J.; Maguire, S.; Russell, J.; Touyz, S. Self-liking and Self-competence: Relationship to Symptoms of Anorexia Nervosa. Eur. Eat. Disord. Rev. Prof. J. Eat. Disord. Assoc. 2007, 15, 139–145. [Google Scholar] [CrossRef]
- Halvorsen, I.; Heyerdahl, S. Girls with Anorexia Nervosa as Young Adults: Personality, Self-esteem, and Life Satisfaction. Int. J. Eat. Disord. 2006, 39, 285–293. [Google Scholar] [CrossRef]
- Cervera, S.; Lahortiga, F.; Angel Martínez-González, M.; Gual, P.; Irala-Estévez, J.D.; Alonso, Y. Neuroticism and Low Self-esteem as Risk Factors for Incident Eating Disorders in a Prospective Cohort Study. Int. J. Eat. Disord. 2003, 33, 271–280. [Google Scholar] [CrossRef]
- Fairburn, C.G.; Cooper, Z.; Shafran, R. Cognitive Behaviour Therapy for Eating Disorders: A “Transdiagnostic” Theory and Treatment. Behav. Res. Ther. 2003, 41, 509–528. [Google Scholar] [CrossRef]
- Jacobi, F.; Wittchen, H.-U.; Hölting, C.; Höfler, M.; Pfister, H.; Müller, N.; Lieb, R. Prevalence, Co-Morbidity and Correlates of Mental Disorders in the General Population: Results from the German Health Interview and Examination Survey (GHS). Psychol. Med. 2004, 34, 597–611. [Google Scholar] [CrossRef]
- Silverstone, P.H. Low Self-Esteem in Eating Disordered Patients in the Absence of Depression. Psychol. Rep. 1990, 67, 276–278. [Google Scholar] [CrossRef]
- Wilksch, S.; Wade, T.D. Differences between Women with Anorexia Nervosa and Restrained Eaters on Shape and Weight Concerns, Self-Esteem, and Depression. Int. J. Eat. Disord. 2004, 35, 571–578. [Google Scholar] [CrossRef]
- Costa, P.T., Jr.; McCrae, R.R. Revised NEO Personality Inventory (NEO-PI-R) and NEO Five-Factor (NEO-FFI) Inventory Professional Manual; PAR: Odessa, FL, USA, 1992. [Google Scholar]
- Strus, W.; Cieciuch, J. Beyond the Big Five–Review of New Models of Personality Structure. In Polskie Forum Psychologiczne; Wydawnictwo Uniwersytetu Kazimierza Wielkiego w Bydgoszczy: Bydgoszcz, Poland, 2014; Volume 1, no. XIX; pp. 17–49. [Google Scholar]
- MacNeill, L.P.; Best, L.A.; Davis, L.L. The Role of Personality in Body Image Dissatisfaction and Disordered Eating: Discrepancies between Men and Women. J. Eat. Disord. 2017, 5, 44. [Google Scholar] [CrossRef]
- Swami, V.; Taylor, R.; Carvalho, C. Body Dissatisfaction Assessed by the Photographic Figure Rating Scale Is Associated with Sociocultural, Personality, and Media Influences. Scand. J. Psychol. 2011, 52, 57–63. [Google Scholar] [CrossRef]
- Miller, J.L.; Schmidt, L.A.; Vaillancourt, T.; McDougall, P.; Laliberte, M. Neuroticism and Introversion: A Risky Combination for Disordered Eating among a Non-Clinical Sample of Undergraduate Women. Eat. Behav. 2006, 7, 69–78. [Google Scholar] [CrossRef]
- Sternheim, L.C.; van Passel, B.; Dingemans, A.; Cath, D.; Danner, U.N. Cognitive and Experienced Flexibility in Patients with Anorexia Nervosa and Obsessive Compulsive Disorder. Front. Psychiatry 2022, 13, 868921. [Google Scholar] [CrossRef] [PubMed]
- Murray, D.; Lesser, M.; Lawson, W. Attention, Monotropism and the Diagnostic Criteria for Autism. Autism 2005, 9, 139–156. [Google Scholar] [CrossRef] [PubMed]
- Abbate-Daga, G.; Buzzichelli, S.; Amianto, F.; Rocca, G.; Marzola, E.; McClintock, S.M.; Fassino, S. Cognitive Flexibility in Verbal and Nonverbal Domains and Decision Making in Anorexia Nervosa Patients: A Pilot Study. BMC Psychiatry 2011, 11, 162. [Google Scholar] [CrossRef]
- Jáuregui-Lobera, I. Neuropsychology of Eating Disorders: 1995–2012. Neuropsychiatr. Dis. Treat. 2013, 9, 415–430. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, S.R.; Fineberg, N.A.; Menzies, L.A.; Blackwell, A.D.; Bullmore, E.T.; Robbins, T.W.; Sahakian, B.J. Impaired Cognitive Flexibility and Motor Inhibition in Unaffected First-Degree Relatives of Patients with Obsessive-Compulsive Disorder. Am. J. Psychiatry 2007, 164, 335–338. [Google Scholar] [CrossRef] [PubMed]
- Meiran, N.; Diamond, G.M.; Toder, D.; Nemets, B. Cognitive Rigidity in Unipolar Depression and Obsessive Compulsive Disorder: Examination of Task Switching, Stroop, Working Memory Updating and Post-Conflict Adaptation. Psychiatry Res. 2011, 185, 149–156. [Google Scholar] [CrossRef]
- Fineberg, N.A.; Day, G.A.; de Koenigswarter, N.; Reghunandanan, S.; Kolli, S.; Jefferies-Sewell, K.; Hranov, G.; Laws, K.R. The Neuropsychology of Obsessive-Compulsive Personality Disorder: A New Analysis. CNS Spectr. 2015, 20, 490–499. [Google Scholar] [CrossRef]
- Paast, N.; Khosravi, Z.; Memari, A.H.; Shayestehfar, M.; Arbabi, M. Comparison of Cognitive Flexibility and Planning Ability in Patients with Obsessive Compulsive Disorder, Patients with Obsessive Compulsive Personality Disorder, and Healthy Controls. Shanghai Arch. Psychiatry 2016, 28, 28. [Google Scholar]
- Buzzichelli, S.; Marzola, E.; Amianto, F.; Fassino, S.; Abbate-Daga, G. Perfectionism and Cognitive Rigidity in Anorexia Nervosa: Is There an Association? Eur. Eat. Disord. Rev. 2018, 26, 360–366. [Google Scholar] [CrossRef]
- Hosseini, S.A.; Padhy, R.K. Body Image Distortion. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- King, I.C.C. Body Image in Paediatric Burns: A Review. Burns Trauma 2018, 6. [Google Scholar] [CrossRef]
- Damiano, S.R.; Hart, L.M.; Paxton, S.J. Development and Validation of Parenting Measures for Body Image and Eating Patterns in Childhood. J. Eat. Disord. 2015, 3, 5. [Google Scholar] [CrossRef]
- Shoraka, H.; Amirkafi, A.; Garrusi, B. Review of Body Image and Some of Contributing Factors in Iranian Population. Int. J. Prev. Med. 2019, 10. [Google Scholar] [CrossRef]
- Bearman, S.K.; Presnell, K.; Martinez, E.; Stice, E. The Skinny on Body Dissatisfaction: A Longitudinal Study of Adolescent Girls and Boys. J. Youth Adolesc. 2006, 35, 217–229. [Google Scholar] [CrossRef]
- Ciwoniuk, N.; Wayda-Zalewska, M.; Kucharska, K. Distorted Body Image and Mental Pain in Anorexia Nervosa. Int. J. Environ. Res. Public. Health 2023, 20, 718. [Google Scholar] [CrossRef]
- Díaz-Marsá, M.; Alberdi-Páramo, Í.; Niell-Galmés, L. Nutritional Supplements in Eating Disorders. Actas Esp. Psiquiatr. 2017, 45, 26–36. [Google Scholar]
- Nitsch, K.; Prajs, E.; Kurpisz, J.; Tyburski, E. Obraz Ciała i Jego Zaburzenia. Aspekty Teoretyczne w Kontekście Wybranych Jednostek Psychopatologicznych. Psychiatr. Psychol. Klin. 2012, 3, 176–182. [Google Scholar]
- Guillaume, S.; Gorwood, P.; Jollant, F.; Van den Eynde, F.; Courtet, P.; Richard-Devantoy, S. Impaired Decision-Making in Symptomatic Anorexia and Bulimia Nervosa Patients: A Meta-Analysis. Psychol. Med. 2015, 45, 3377–3391. [Google Scholar] [CrossRef]
- Couturier, J.; Kimber, M.; Szatmari, P. Efficacy of Family-based Treatment for Adolescents with Eating Disorders: A Systematic Review and Meta-analysis. Int. J. Eat. Disord. 2013, 46, 3–11. [Google Scholar] [CrossRef]
- le Grange, D.; Eisler, I. Family Interventions in Adolescent Anorexia Nervosa. Child Adolesc. Psychiatr. Clin. N. Am. 2009, 18, 159–173. [Google Scholar] [CrossRef]
- Lock, J. Evaluation of Family Treatment Models for Eating Disorders. Curr. Opin. Psychiatry 2011, 24, 274–279. [Google Scholar] [CrossRef]
- Eisler, I.; Simic, M.; Blessitt, E.; Dodge, L.; MCCAED Team. Maudsley Service Manual for Child and Adolescent Eating Disorders. 2016. Available online: https://mccaed.slam.nhs.uk/wp-content/uploads/2019/11/Maudsley-Service-Manual-for-Child-and-Adolescent-Eating-Disorders-July-2016.pdf (accessed on 9 May 2023).
- Pilecki, M.W.; Sałapa, K.; Józefik, B. Socio-Cultural Context of Eating Disorders in Poland. J. Eat. Disord. 2016, 4, 11. [Google Scholar] [CrossRef] [PubMed]
- Tchanturia, K.; Davies, H.; Harrison, A.; Fox, J.R.E.; Treasure, J.; Schmidt, U. Altered Social Hedonic Processing in Eating Disorders. Int. J. Eat. Disord. 2012, 45, 962–969. [Google Scholar] [CrossRef]
- Momeñe, J.; Estévez, A.; Griffiths, M.D.; Macía, P.; Herrero, M.; Olave, L.; Iruarrizaga, I. Eating Disorders and Intimate Partner Violence: The Influence of Fear of Loneliness and Social Withdrawal. Nutrients 2022, 14, 2611. [Google Scholar] [CrossRef] [PubMed]
- Harney, M.B.; Fitzsimmons-Craft, E.E.; Maldonado, C.R.; Bardone-Cone, A.M. Negative Affective Experiences in Relation to Stages of Eating Disorder Recovery. Eat. Behav. 2014, 15, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Elwyn, R. A Lived Experience Response to the Proposed Diagnosis of Terminal Anorexia Nervosa: Learning from Iatrogenic Harm, Ambivalence and Enduring Hope. J. Eat. Disord. 2023, 11, 2. [Google Scholar] [CrossRef]
- Rymarczyk, K. The Role of Personality Traits, Sociocultural Factors, and Body Dissatisfaction in Anorexia Readiness Syndrome in Women. J. Eat. Disord. 2021, 9, 51. [Google Scholar] [CrossRef]
- Levine, M.P.; Smolak, L.; Hayden, H. The Relation of Sociocultural Factors to Eating Attitudes and Behaviors among Middle School Girls. J. Early Adolesc. 1994, 14, 471–490. [Google Scholar] [CrossRef]
- Jones, D.C.; Crawford, J.K. The Peer Appearance Culture During Adolescence: Gender and Body Mass Variations. J. Youth Adolesc. 2006, 35, 243–255. [Google Scholar] [CrossRef]
- Bozzola, E.; Spina, G.; Agostiniani, R.; Barni, S.; Russo, R.; Scarpato, E.; Di Mauro, A.; Di Stefano, A.V.; Caruso, C.; Corsello, G.; et al. The Use of Social Media in Children and Adolescents: Scoping Review on the Potential Risks. Int. J. Environ. Res. Public. Health 2022, 19, 9960. [Google Scholar] [CrossRef]
- Dufour, R.; Novack, K.; Picard, L.; Chadi, N.; Booij, L. The Use of Technology in the Treatment of Youth with Eating Disorders: A Scoping Review. J. Eat. Disord. 2022, 10, 182. [Google Scholar] [CrossRef]
- Wiberg, A.-C.; Ghaderi, A.; Danielsson, H.B.; Safarzadeh, K.; Parling, T.; Carlbring, P.; Jansson, M.; Welch, E. Internet-Based Cognitive Behavior Therapy for Eating Disorders—Development and Feasibility Evaluation. Internet Interv. 2022, 30, 100570. [Google Scholar] [CrossRef]
- Vaccaro, C.M.; Guarino, G.; Conte, D.; Ferrara, E.; Ragione, L.D.; Bracale, R. Social Networks and Eating Disorders during the COVID-19 Pandemic. Open Med. 2021, 16, 1170–1174. [Google Scholar] [CrossRef]
- Frieiro, P.; González-Rodríguez, R.; Domínguez-Alonso, J. Self-esteem and Socialisation in Social Networks as Determinants in Adolescents’ Eating Disorders. Health Soc. Care Community 2022, 30, e4416–e4424. [Google Scholar] [CrossRef]
- Bonfanti, R.C.; Lo Coco, G.; Salerno, L.; Di Blasi, M. The Thin Ideal and Attitudes towards Appearance as Correlates of Exercise Addiction among Sporty People during the COVID-19 Pandemic. Behav. Sci. 2022, 12, 187. [Google Scholar] [CrossRef]
- Clarke, J.; Peyre, H.; Alison, M.; Bargiacchi, A.; Stordeur, C.; Boizeau, P.; Mamou, G.; Crépon, S.G.; Alberti, C.; Léger, J. Abnormal Bone Mineral Density and Content in Girls with Early-Onset Anorexia Nervosa. J. Eat. Disord. 2021, 9, 9. [Google Scholar] [CrossRef]
- Misra, M.; Klibanski, A. Bone Health in Anorexia Nervosa. Curr. Opin. Endocrinol. Diabetes Obes. 2011, 18, 376. [Google Scholar] [CrossRef]
- Robinson, L.; Micali, N.; Misra, M. Eating Disorders and Bone Metabolism in Women. Curr. Opin. Pediatr. 2017, 29, 488. [Google Scholar] [CrossRef]
- Miller, K.K.; Lee, E.E.; Lawson, E.A.; Misra, M.; Minihan, J.; Grinspoon, S.K.; Gleysteen, S.; Mickley, D.; Herzog, D.; Klibanski, A. Determinants of Skeletal Loss and Recovery in Anorexia Nervosa. J. Clin. Endocrinol. Metab. 2006, 91, 2931–2937. [Google Scholar] [CrossRef]
- Vestergaard, P.; Emborg, C.; Støving, R.K.; Hagen, C.; Mosekilde, L.; Brixen, K. Fractures in Patients with Anorexia Nervosa, Bulimia Nervosa, and Other Eating Disorders—A Nationwide Register Study. Int. J. Eat. Disord. 2002, 32, 301–308. [Google Scholar] [CrossRef]
- Jada, K.; Djossi, S.K.; Khedr, A.; Neupane, B.; Proskuriakova, E.; Mostafa, J.A. The Pathophysiology of Anorexia Nervosa in Hypothalamic Endocrine Function and Bone Metabolism. Cureus 2021, 13, e20548. [Google Scholar] [CrossRef]
- Legroux-Gerot, I.; Vignau, J.; Biver, E.; Pigny, P.; Collier, F.; Marchandise, X.; Duquesnoy, B.; Cortet, B. Anorexia Nervosa, Osteoporosis and Circulating Leptin: The Missing Link. Osteoporos. Int. 2010, 21, 1715–1722. [Google Scholar] [CrossRef] [PubMed]
- Legroux-Gérot, I.; Vignau, J.; D’Herbomez, M.; Collier, F.; Marchandise, X.; Duquesnoy, B.; Cortet, B. Evaluation of Bone Loss and Its Mechanisms in Anorexia Nervosa. Calcif. Tissue Int. 2007, 81, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Legroux, I.; Cortet, B. Factors Influencing Bone Loss in Anorexia Nervosa: Assessment and Therapeutic Options. RMD Open 2019, 5, e001009. [Google Scholar] [CrossRef] [PubMed]
- Zipfel, S.; Löwe, B.; Reas, D.L.; Deter, H.-C.; Herzog, W. Long-Term Prognosis in Anorexia Nervosa: Lessons from a 21-Year Follow-up Study. Lancet 2000, 355, 721–722. [Google Scholar] [CrossRef]
- Misra, M.; Miller, K.K.; Cord, J.; Prabhakaran, R.; Herzog, D.B.; Goldstein, M.; Katzman, D.K.; Klibanski, A. Relationships between Serum Adipokines, Insulin Levels, and Bone Density in Girls with Anorexia Nervosa. J. Clin. Endocrinol. Metab. 2007, 92, 2046–2052. [Google Scholar] [CrossRef]
- Legroux-Gerot, I.; Vignau, J.; Collier, F.; Cortet, B. Factors Influencing Changes in Bone Mineral Density in Patients with Anorexia Nervosa-Related Osteoporosis: The Effect of Hormone Replacement Therapy. Calcif. Tissue Int. 2008, 83, 315–323. [Google Scholar] [CrossRef]
- Rosa-Caldwell, M.E.; Eddy, K.T.; Rutkove, S.B.; Breithaupt, L. Anorexia Nervosa and Muscle Health: A Systematic Review of Our Current Understanding and Future Recommendations for Study. Int. J. Eat. Disord. 2023, 56, 483–500. [Google Scholar] [CrossRef]
- Solenberger, S.E. Exercise and Eating Disorders: A 3-Year Inpatient Hospital Record Analysis. Eat. Behav. 2001, 2, 151–168. [Google Scholar] [CrossRef]
- McLoughlin, D.M.; Spargo, E.; Wassif, W.S.; Newham, D.J.; Peters, T.J.; Lantos, P.L.; Russell, G.F.M. Structural and Functional Changes in Skeletal Muscle in Anorexia Nervosa. Acta Neuropathol. 1998, 95, 540–632. [Google Scholar] [CrossRef]
- Fernández-del-Valle, M.; Larumbe-Zabala, E.; Morande-Lavin, G.; Perez Ruiz, M. Muscle Function and Body Composition Profile in Adolescents with Restrictive Anorexia Nervosa: Does Resistance Training Help? Disabil. Rehabil. 2016, 38, 346–353. [Google Scholar] [CrossRef]
- Leitner, L.M.; Wilson, R.J.; Yan, Z.; Gödecke, A. Reactive Oxygen Species/Nitric Oxide Mediated Inter-Organ Communication in Skeletal Muscle Wasting Diseases. Antioxid. Redox Signal. 2017, 26, 700–717. [Google Scholar] [CrossRef]
- Hodson, N.; Philp, A. The Importance of MTOR Trafficking for Human Skeletal Muscle Translational Control. Exerc. Sport Sci. Rev. 2019, 47, 46. [Google Scholar] [CrossRef]
- Shimkus, K.L.; Jefferson, L.S.; Gordon, B.S.; Kimball, S.R. Repressors of MTORC1 Act to Blunt the Anabolic Response to Feeding in the Soleus Muscle of a Cast-immobilized Mouse Hindlimb. Physiol. Rep. 2018, 6, e13891. [Google Scholar] [CrossRef]
- Alaaraj, N.; Soliman, A.; Hamed, N.; Alyafei, F.; De Sanctis, V. Understanding the Complex Role of Mtorc as an Intracellular Critical Mediator of Whole-Body Metabolism in Anorexia Nervosa: A Mini Review. Acta Bio Medica Atenei Parm. 2021, 92, e2021170. [Google Scholar]
- Tripathi, D.; Kant, S.; Pandey, S.; Ehtesham, N.Z. Resistin in Metabolism, Inflammation, and Disease. FEBS J. 2020, 287, 3141–3149. [Google Scholar] [CrossRef]
- Cui, H.; Moore, J.; Ashimi, S.S.; Mason, B.L.; Drawbridge, J.N.; Han, S.; Hing, B.; Matthews, A.; McAdams, C.J.; Darbro, B.W. Eating Disorder Predisposition Is Associated with ESRRA and HDAC4 Mutations. J. Clin. Investig. 2013, 123, 4706–4713. [Google Scholar] [CrossRef]
- Horard, B.; Vanacker, J.M. Estrogen Receptor-Related Receptors: Orphan Receptors Desperately Seeking a Ligand. J. Mol. Endocrinol. 2003, 31, 349–357. [Google Scholar] [CrossRef]
- Bookout, A.L.; Jeong, Y.; Downes, M.; Ruth, T.Y.; Evans, R.M.; Mangelsdorf, D.J. Anatomical Profiling of Nuclear Receptor Expression Reveals a Hierarchical Transcriptional Network. Cell 2006, 126, 789–799. [Google Scholar] [CrossRef]
- Singh, B.K.; Sinha, R.A.; Ohba, K.; Yen, P.M. Role of Thyroid Hormone in Hepatic Gene Regulation, Chromatin Remodeling, and Autophagy. Mol. Cell. Endocrinol. 2017, 458, 160–168. [Google Scholar] [CrossRef]
- Rangwala, S.M.; Li, X.; Lindsley, L.; Wang, X.; Shaughnessy, S.; Daniels, T.G.; Szustakowski, J.; Nirmala, N.R.; Wu, Z.; Stevenson, S.C. Estrogen-Related Receptor α Is Essential for the Expression of Antioxidant Protection Genes and Mitochondrial Function. Biochem. Biophys. Res. Commun. 2007, 357, 231–236. [Google Scholar] [CrossRef]
- Wallace, M.A.; Hock, M.B.; Hazen, B.C.; Kralli, A.; Snow, R.J.; Russell, A.P. Striated Muscle Activator of Rho Signalling (STARS) Is a PGC-1α/Oestrogen-related Receptor-α Target Gene and Is Upregulated in Human Skeletal Muscle after Endurance Exercise. J. Physiol. 2011, 589, 2027–2039. [Google Scholar] [CrossRef] [PubMed]
- Ranhotra, H.S. Up-Regulation of Orphan Nuclear Estrogen-Related Receptor Alpha Expression during Long-Term Caloric Restriction in Mice. Mol. Cell. Biochem. 2009, 332, 59–65. [Google Scholar] [CrossRef]
- Giguére, V. To ERR in the Estrogen Pathway. Trends Endocrinol. Metab. 2002, 13, 220–225. [Google Scholar] [CrossRef] [PubMed]
- Sender, R.; Fuchs, S.; Milo, R. Revised Estimates for the Number of Human and Bacteria Cells in the Body. PLoS Biol. 2016, 14, e1002533. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, M.; Knight, R.; Leibel, R.L. The Gut Microbiota in Human Energy Homeostasis and Obesity. Trends Endocrinol. Metab. 2015, 26, 493–501. [Google Scholar] [CrossRef]
- Lozupone, C.A.; Stombaugh, J.I.; Gordon, J.I.; Jansson, J.K.; Knight, R. Diversity, Stability and Resilience of the Human Gut Microbiota. Nature 2012, 489, 220–230. [Google Scholar] [CrossRef]
- Borgo, F.; Riva, A.; Benetti, A.; Casiraghi, M.C.; Bertelli, S.; Garbossa, S.; Anselmetti, S.; Scarone, S.; Pontiroli, A.E.; Morace, G. Microbiota in Anorexia Nervosa: The Triangle between Bacterial Species, Metabolites and Psychological Tests. PLoS ONE 2017, 12, e0179739. [Google Scholar] [CrossRef]
- Mack, I.; Cuntz, U.; Grämer, C.; Niedermaier, S.; Pohl, C.; Schwiertz, A.; Zimmermann, K.; Zipfel, S.; Enck, P.; Penders, J. Weight Gain in Anorexia Nervosa Does Not Ameliorate the Faecal Microbiota, Branched Chain Fatty Acid Profiles and Gastrointestinal Complaints. Sci. Rep. 2016, 6, 26752. [Google Scholar] [CrossRef]
- Mörkl, S.; Lackner, S.; Müller, W.; Gorkiewicz, G.; Kashofer, K.; Oberascher, A.; Painold, A.; Holl, A.; Holzer, P.; Meinitzer, A. Gut Microbiota and Body Composition in Anorexia Nervosa Inpatients in Comparison to Athletes, Overweight, Obese, and Normal Weight Controls. Int. J. Eat. Disord. 2017, 50, 1421–1431. [Google Scholar] [CrossRef]
- Morita, C.; Tsuji, H.; Hata, T.; Gondo, M.; Takakura, S.; Kawai, K.; Yoshihara, K.; Ogata, K.; Nomoto, K.; Miyazaki, K. Gut Dysbiosis in Patients with Anorexia Nervosa. PLoS ONE 2015, 10, e0145274. [Google Scholar] [CrossRef]
- Ruusunen, A.; Rocks, T.; Jacka, F.; Loughman, A. The Gut Microbiome in Anorexia Nervosa: Relevance for Nutritional Rehabilitation. Psychopharmacology 2019, 236, 1545–1558. [Google Scholar] [CrossRef]
- Armougom, F.; Henry, M.; Vialettes, B.; Raccah, D.; Raoult, D. Monitoring Bacterial Community of Human Gut Microbiota Reveals an Increase in Lactobacillus in Obese Patients and Methanogens in Anorexic Patients. PLoS ONE 2009, 4, e7125. [Google Scholar] [CrossRef]
- Ghoshal, U.; Shukla, R.; Srivastava, D.; Ghoshal, U.C. Irritable Bowel Syndrome, Particularly the Constipation-Predominant Form, Involves an Increase in Methanobrevibacter Smithii, Which Is Associated with Higher Methane Production. Gut Liver 2016, 10, 932. [Google Scholar] [CrossRef]
- Breton, J.; Legrand, R.; Akkermann, K.; Järv, A.; Harro, J.; Déchelotte, P.; Fetissov, S.O. Elevated Plasma Concentrations of Bacterial ClpB Protein in Patients with Eating Disorders. Int. J. Eat. Disord. 2016, 49, 805–808. [Google Scholar] [CrossRef]
- Kleiman, S.C.; Watson, H.J.; Bulik-Sullivan, E.C.; Huh, E.Y.; Tarantino, L.M.; Bulik, C.M.; Carroll, I.M. The Intestinal Microbiota in Acute Anorexia Nervosa and during Renourishment: Relationship to Depression, Anxiety, and Eating Disorder Psychopathology. Psychosom. Med. 2015, 77, 969. [Google Scholar] [CrossRef]
- Prochazkova, P.; Roubalova, R.; Dvorak, J.; Tlaskalova-Hogenova, H.; Cermakova, M.; Tomasova, P.; Sediva, B.; Kuzma, M.; Bulant, J.; Bilej, M.; et al. Microbiota, Microbial Metabolites, and Barrier Function in A Patient with Anorexia Nervosa after Fecal Microbiota Transplantation. Microorganisms 2019, 7, 338. [Google Scholar] [CrossRef]
- Gouba, N.; Raoult, D.; Drancourt, M. Gut Microeukaryotes during Anorexia Nervosa: A Case Report. BMC Res. Notes 2014, 7, 33. [Google Scholar] [CrossRef]
- Mörkl, S.; Lackner, S.; Meinitzer, A.; Mangge, H.; Lehofer, M.; Halwachs, B.; Gorkiewicz, G.; Kashofer, K.; Painold, A.; Holl, A.K. Gut Microbiota, Dietary Intakes and Intestinal Permeability Reflected by Serum Zonulin in Women. Eur. J. Nutr. 2018, 57, 2985–2997. [Google Scholar] [CrossRef]
- Speranza, E.; Cioffi, I.; Santarpia, L.; Del Piano, C.; De Caprio, C.; Naccarato, M.; Marra, M.; De Filippo, E.; Contaldo, F.; Pasanisi, F. Fecal Short Chain Fatty Acids and Dietary Intake in Italian Women with Restrictive Anorexia Nervosa: A Pilot Study. Front. Nutr. 2018, 5, 119. [Google Scholar] [CrossRef]
- Tang, W.K.; Sun, F.C.S.; Ungvari, G.S.; O’donnell, D. Oral Health of Psychiatric In-Patients in Hong Kong. Int. J. Soc. Psychiatry 2004, 50, 186–191. [Google Scholar] [CrossRef]
- Kisely, S.; Baghaie, H.; Lalloo, R.; Johnson, N.W. Association between Poor Oral Health and Eating Disorders: Systematic Review and Meta-Analysis. Br. J. Psychiatry 2015, 207, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Ramon, T.; Grinshpoon, A.; Zusman, S.P.; Weizman, A. Oral Health and Treatment Needs of Institutionalized Chronic Psychiatric Patients in Israel. Eur. Psychiatry 2003, 18, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Hasan, S.; Ahmed, S.; Panigrahi, R.; Chaudhary, P.; Vyas, V.; Saeed, S. Oral Cavity and Eating Disorders: An Insight to Holistic Health. J. Fam. Med. Prim. Care 2020, 9, 3890. [Google Scholar]
- Scharner, S.; Stengel, A. Alterations of Brain Structure and Functions in Anorexia Nervosa. Clin. Nutr. Exp. 2019, 28, 22–32. [Google Scholar] [CrossRef]
- DeBate, R.D.; Tedesco, L.A.; Kerschbaum, W.E. Knowledge of Oral and Physical Manifestations of Anorexia and Bulimia Nervosa among Dentists and Dental Hygienists. J. Dent. Educ. 2005, 69, 346–354. [Google Scholar] [CrossRef]
- Lourenço, M.; Azevedo, Á.; Brandão, I.; Gomes, P.S. Orofacial Manifestations in Outpatients with Anorexia Nervosa and Bulimia Nervosa Focusing on the Vomiting Behavior. Clin. Oral Investig. 2018, 22, 1915–1922. [Google Scholar] [CrossRef]
- Johansson, A.-K.; Mjanger Øvretvedt, T.; Reinholtsen, K.K.; Johansson, A. Eating Disorders: An Analysis of Self-Induced Vomiting, Binge Eating, and Oral Hygiene Behavior. Int. J. Clin. Pract. 2022, 2022, 6210372. [Google Scholar] [CrossRef]
- Jagielska, G.; Kacperska, I. Outcome, Comorbidity and Prognosis in Anorexia Nervosa. Psychiatr. Pol. 2017, 51, 205–218. [Google Scholar] [CrossRef]
- Roberts, M.W.; Li, S.-H. Oral Findings in Anorexia Nervosa and Bulimia Nervosa: A Study of 47 Cases. J. Am. Dent. Assoc. 1987, 115, 407–410. [Google Scholar] [CrossRef]
- Favaro, A.; Ferrara, S.; Santonastaso, P. The Spectrum of Eating Disorders in Young Women: A Prevalence Study in a General Population Sample. Psychosom. Med. 2003, 65, 701–708. [Google Scholar] [CrossRef]
- Paszynska, E.; Hernik, A.; Slopien, A.; Roszak, M.; Jowik, K.; Dmitrzak-Weglarz, M.; Tyszkiewicz-Nwafor, M. Risk of Dental Caries and Erosive Tooth Wear in 117 Children and Adolescents’ Anorexia Nervosa Population—A Case-Control Study. Front. Psychiatry 2022, 13, 874263. [Google Scholar] [CrossRef]
- Panico, R.; Piemonte, E.; Lazos, J.; Gilligan, G.; Zampini, A.; Lanfranchi, H. Oral Mucosal Lesions in Anorexia Nervosa, Bulimia Nervosa and EDNOS. J. Psychiatr. Res. 2018, 96, 178–182. [Google Scholar] [CrossRef]
- Mascitti, M.; Coccia, E.; Vignini, A.; Aquilanti, L.; Santarelli, A.; Salvolini, E.; Sabbatinelli, J.; Mazzanti, L.; Procaccini, M.; Rappelli, G. Anorexia, Oral Health and Antioxidant Salivary System: A Clinical Study on Adult Female Subjects. Dent. J. 2019, 7, 60. [Google Scholar] [CrossRef]
- Vergata, R.T. Oral Manifestations of Eating Disorders in Adolescent Patients. A Review. Eur. J. Paediatr. Dent. 2021, 22, 155. [Google Scholar]
- Aranha, A.C.; Eduardo, C.d.P.; Cordás, T.A. Eating Disorders. Part I: Psychiatric Diagnosis and Dental Implications. J. Contemp. Dent. Pract. 2008, 9, 73–81. [Google Scholar] [CrossRef]
- Hermont, A.P.; Oliveira, P.A.D.; Martins, C.C.; Paiva, S.M.; Pordeus, I.A.; Auad, S.M. Tooth Erosion and Eating Disorders: A Systematic Review and Meta-Analysis. PLoS ONE 2014, 9, e111123. [Google Scholar] [CrossRef]
- Rangé, H.; Pallier, A.; Ali, A.; Huas, C.; Colon, P.; Godart, N. Risk Factors for Oral Health in Anorexia Nervosa: Comparison of a Self-Report Questionnaire and a Face-to-Face Interview. Int. J. Environ. Res. Public. Health 2021, 18, 4212. [Google Scholar] [CrossRef]
- Nijakowski, K.; Walerczyk-Sas, A.; Surdacka, A. Regular Physical Activity as a Potential Risk Factor for Erosive Lesions in Adolescents. Int. J. Environ. Res. Public. Health 2020, 17, 3002. [Google Scholar] [CrossRef]
- Pleple, A.; Lalanne, C.; Huas, C.; Mattar, L.; Hanachi, M.; Flament, M.F.; Carchon, I.; Jouen, F.; Berthoz, S.; Godart, N. Nutritional Status and Anxious and Depressive Symptoms in Anorexia Nervosa: A Prospective Study. Sci. Rep. 2021, 11, 771. [Google Scholar] [CrossRef]
- Lobera, I.J.; Ríos, P.B. Choice of Diet in Patients with Anorexia Nervosa. Nutr. Hosp. 2009, 24, 682–687. [Google Scholar]
- Garrido-Martínez, P.; Domínguez-Gordillo, A.; Cerero-Lapiedra, R.; Burgueño-García, M.; Martínez-Ramírez, M.-J.; Gómez-Candela, C.; Cebrián-Carretero, J.-L.; Esparza-Gómez, G. Oral and Dental Health Status in Patients with Eating Disorders in Madrid, Spain. Med. Oral Patol. Oral Cirugia Bucal 2019, 24, e595. [Google Scholar] [CrossRef] [PubMed]
- Silverstein, L.S.; Haggerty, C.; Sams, L.; Phillips, C.; Roberts, M.W. Impact of an Oral Health Education Intervention among a Group of Patients with Eating Disorders (Anorexia Nervosa and Bulimia Nervosa). J. Eat. Disord. 2019, 7, 29. [Google Scholar] [CrossRef] [PubMed]
- Marzola, E.; Nasser, J.A.; Hashim, S.A.; Shih, P.-A.B.; Kaye, W.H. Nutritional Rehabilitation in Anorexia Nervosa: Review of the Literature and Implications for Treatment. BMC Psychiatry 2013, 13, 290. [Google Scholar] [CrossRef] [PubMed]
- Andrewes, D.G.; O’Connor, P.; Mulder, C.; McLennan, J.; Derham, H.; Weigall, S.; Say, S. Computerised Psychoeducation for Patients with Eating Disorders. Aust. N. Z. J. Psychiatry 1996, 30, 492–497. [Google Scholar] [CrossRef]
- Ng, L.W.C.; Ng, D.P.; Wong, W.P. Is Supervised Exercise Training Safe in Patients with Anorexia Nervosa? A Meta-Analysis. Physiotherapy 2013, 99, 1–11. [Google Scholar] [CrossRef]
- Rizk, M.; Kern, L.; Lalanne, C.; Hanachi, M.; Melchior, J.; Pichard, C.; Mattar, L.; Group, E.; Berthoz, S.; Godart, N. High-intensity Exercise Is Associated with a Better Nutritional Status in Anorexia Nervosa. Eur. Eat. Disord. Rev. 2019, 27, 391–400. [Google Scholar] [CrossRef]
- Dalle Grave, R.; Calugi, S.; El Ghoch, M.; Conti, M.; Fairburn, C.G. Inpatient Cognitive Behavior Therapy for Adolescents with Anorexia Nervosa: Immediate and Longer-Term Effects. Front. Psychiatry 2014, 5, 14. [Google Scholar] [CrossRef]
- Steinglass, J.; Albano, A.M.; Simpson, H.B.; Carpenter, K.; Schebendach, J.; Attia, E. Fear of Food as a Treatment Target: Exposure and Response Prevention for Anorexia Nervosa in an Open Series. Int. J. Eat. Disord. 2012, 45, 615–621. [Google Scholar] [CrossRef]
- Fisher, C.A.; Skocic, S.; Rutherford, K.A.; Hetrick, S.E. Family Therapy Approaches for Anorexia Nervosa. Cochrane Database Syst. Rev. 2018, 10, CD004780. [Google Scholar] [CrossRef]
- Barber, S.; Thornicroft, G. Reducing the Mortality Gap in People with Severe Mental Disorders: The Role of Lifestyle Psychosocial Interventions. Front. Psychiatry 2018, 9, 463. [Google Scholar] [CrossRef]
- Hart, S.; Field, T.; Hernandez-Reif, M.; Nearing, G.; Shaw, S.; Schanberg, S.; Kuhn, C. Anorexia Nervosa Symptoms Are Reduced by Massage Therapy. Eat. Disord. 2001, 9, 289–299. [Google Scholar] [CrossRef]
- Fogarty, S.; Smith, C.A.; Touyz, S.; Madden, S.; Buckett, G.; Hay, P. Patients with Anorexia Nervosa Receiving Acupuncture or Acupressure; Their View of the Therapeutic Encounter. Complement. Ther. Med. 2013, 21, 675–681. [Google Scholar] [CrossRef]
- American Dietetic Association. Position of the American Dietetic Association: Nutrition Intervention in the Treatment of Anorexia Nervosa, Bulimia Nervosa, and Eating Disorders Not Otherwise Specified (EDNOS). J. Am. Diet. Assoc. 2001, 101, 810–819. [Google Scholar] [CrossRef]
- Rock, C.L. Nutritional Rehabilitation for Anorexia Nervosa. In The Treatment of Eating Disorders: A Clinical Handbook; Guilford Press: New York, NY, USA, 2010; pp. 187–197. [Google Scholar]
- Wilson, G.T.; Grilo, C.M.; Vitousek, K.M. Psychological Treatment of Eating Disorders. Am. Psychol. 2007, 62, 199. [Google Scholar] [CrossRef]
- Gómez Candela, C.; Palma Milla, S.; Miján de la Torre, A.; Rodríguez Ortega, P.; Matía Martín, P.; Loria Kohen, V.; Campos del Portillo, R.; Virgili Casas, M.; Martínez Olmos, M.Á.; Mories Álvarez, M.T. Consenso Sobre La Evaluación y El Tratamiento Nutricional de Los Trastornos de La Conducta Alimentaria: Anorexia Nerviosa. Nutr. Hosp. 2018, 35, 11–48. [Google Scholar] [CrossRef]
- De La Guía, G.D.T. Guía de Práctica Clínica Sobre Trastornos de La Conducta Alimentaria; Hospital Psiquiátrico “Román Alberca”: Murcia, Spain, 2009. [Google Scholar]
- Association, A.P. Practice Guideline for the Treatment of Patients with Eating Disorders. Am. J. Psychiatry 2006, 163, 4–54. [Google Scholar]
- Reiter, C.S.; Graves, L. Nutrition Therapy for Eating Disorders. Nutr. Clin. Pract. 2010, 25, 122–136. [Google Scholar] [CrossRef]
- Wakefield, A.; Williams, H. Practice Recommendations for the Nutritional Management of Anorexia Nervosa in Adults; Dietitians Association of Australia: Deakin, Australia, 2009. [Google Scholar]
- National Collaborating Centre for Mental Health (UK). Eating Disorders: Core Interventions in the Treatment and Management of Anorexia Nervosa, Bulimia Nervosa and Related Eating Disorders; National Collaborating Centre for Mental Health: Leicester, UK, 2004. [Google Scholar]
- McMaster, C.M.; Wade, T.; Franklin, J.; Hart, S. Development of Consensus-based Guidelines for Outpatient Dietetic Treatment of Eating Disorders: A Delphi Study. Int. J. Eat. Disord. 2020, 53, 1480–1495. [Google Scholar] [CrossRef]
- Trocki, O.; Shepherd, R.W. Change in Body Mass Index Does Not Predict Change in Body Composition in Adolescent Girls with Anorexia Nervosa. J. Acad. Nutr. Diet. 2000, 100, 457. [Google Scholar] [CrossRef]
- Bartrina, J.A.; Majem, L.S. Objetivos Nutricionales Para La Población Española: Consenso de La Sociedad Española de Nutrición Comunitaria 2011. Rev. Esp. Nutr. Comunitaria Span. J. Community Nutr. 2011, 17, 178–199. [Google Scholar]
- Capo-chichi, C.D.; Guéant, J.-L.; Lefebvre, E.; Bennani, N.; Lorentz, E.; Vidailhet, C.; Vidailhet, M. Riboflavin and Riboflavin-Derived Cofactors in Adolescent Girls with Anorexia Nervosa. Am. J. Clin. Nutr. 1999, 69, 672–678. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Grinspoon, S.; Herzog, D.; Klibanski, A. Mechanisms and Treatment Options for Bone Loss in Anorexia Nervosa. Psychopharmacol. Bull. 1997, 33, 399–404. [Google Scholar] [PubMed]
- Ortega Anta, M.R.; Aparicio Vizuete, A.; Lopez Sobaler, A.M. Educación Nutricional; Gil Hernández, Á., Ed.; Tratado de Nutrición; Editorial Medica Panamericana: Madrid, Spain, 2005; pp. 514–554. [Google Scholar]
- Dwyer, J.T. Role of the Nutritionist in Eating Disorders. New Dir. Ment. Health Serv. 1986, 1986, 21–28. [Google Scholar] [CrossRef]
- Ozier, A.D.; Henry, B.W. Position of the American Dietetic Association: Nutrition Intervention in the Treatment of Eating Disorders. J. Am. Diet. Assoc. 2011, 111, 1236–1241. [Google Scholar] [CrossRef]
- Mayer, L.E.S.; Schebendach, J.; Bodell, L.P.; Shingleton, R.M.; Walsh, B.T. Eating Behavior in Anorexia Nervosa: Before and after Treatment. Int. J. Eat. Disord. 2012, 45, 290–293. [Google Scholar] [CrossRef]
- Nomdedeu, C.L. Educación Nutricional de La Población General y de Riesgo. In Nutrición y Metabolismo en Trastornos de la Conducta Alimentaria; Fundación Dialnet: La Rioja, Spain, 2004; pp. 459–476. [Google Scholar]
- Quiles Marcos, Y.; Leon Zarceno, E.; Lopez Lopez, J.A. Effectiveness of Exercise-based Interventions in Patients with Anorexia Nervosa: A Systematic Review. Eur. Eat. Disord. Rev. 2021, 29, 3–19. [Google Scholar] [CrossRef]
- Melissa, R.; Lama, M.; Laurence, K.; Sylvie, B.; Jeanne, D.; Odile, V.; Nathalie, G. Physical Activity in Eating Disorders: A Systematic Review. Nutrients 2020, 12, 183. [Google Scholar] [CrossRef]
- Vancampfort, D.; Vanderlinden, J.; De Hert, M.; Soundy, A.; Adamkova, M.; Skjaerven, L.H.; Catalan-Matamoros, D.; Lundvik Gyllensten, A.; Gomez-Conesa, A.; Probst, M. A Systematic Review of Physical Therapy Interventions for Patients with Anorexia and Bulemia Nervosa. Disabil. Rehabil. 2014, 36, 628–634. [Google Scholar] [CrossRef]
- Le Grange, D.; Eisler, I. The Link between Anorexia Nervosa and Excessive Exercise: A Review. Eur. Eat. Disord. Rev. 1993, 1, 100–119. [Google Scholar] [CrossRef]
- Stice, E. Risk and Maintenance Factors for Eating Pathology: A Meta-Analytic Review. Psychol. Bull. 2002, 128, 825. [Google Scholar] [CrossRef]
- Leuenberger, A. Endorphins, Exercise, and Addictions: A Review of Exercise Dependence. 2006. Available online: https://impulse.pubpub.org/pub/ff9iap4s (accessed on 9 May 2023).
- Cook, B.; Engel, S.; Crosby, R.; Hausenblas, H.; Wonderlich, S.; Mitchell, J. Pathological Motivations for Exercise and Eating Disorder Specific Health-related Quality of Life. Int. J. Eat. Disord. 2014, 47, 268–272. [Google Scholar] [CrossRef]
- Strober, M.; Freeman, R.; Morrell, W. The Long-term Course of Severe Anorexia Nervosa in Adolescents: Survival Analysis of Recovery, Relapse, and Outcome Predictors over 10–15 Years in a Prospective Study. Int. J. Eat. Disord. 1997, 22, 339–360. [Google Scholar] [CrossRef]
- Ruiz, J.R.; Sui, X.; Lobelo, F.; Morrow, J.R.; Jackson, A.W.; Sjöström, M.; Blair, S.N. Association between Muscular Strength and Mortality in Men: Prospective Cohort Study. BMJ 2008, 337, a439. [Google Scholar] [CrossRef]
- Chen, L.; Nelson, D.R.; Zhao, Y.; Cui, Z.; Johnston, J.A. Relationship between Muscle Mass and Muscle Strength, and the Impact of Comorbidities: A Population-Based, Cross-Sectional Study of Older Adults in the United States. BMC Geriatr. 2013, 13, 74. [Google Scholar] [CrossRef]
- Trombetti, A.; Reid, K.F.; Hars, M.; Herrmann, F.R.; Pasha, E.; Phillips, E.M.; Fielding, R.A. Age-Associated Declines in Muscle Mass, Strength, Power, and Physical Performance: Impact on Fear of Falling and Quality of Life. Osteoporos. Int. 2016, 27, 463–471. [Google Scholar] [CrossRef]
- Martin, S.P.K.; Bachrach, L.K.; Golden, N.H. Controlled Pilot Study of High-Impact Low-Frequency Exercise on Bone Loss and Vital-Sign Stabilization in Adolescents with Eating Disorders. J. Adolesc. Health 2017, 60, 33–37. [Google Scholar] [CrossRef]
- Calogero, R.M.; Pedrotty, K.N. The Practice and Process of Healthy Exercise: An Investigation of the Treatment of Exercise Abuse in Women with Eating Disorders. Eat. Disord. 2004, 12, 273–291. [Google Scholar] [CrossRef]
- Carei, T.R.; Fyfe-Johnson, A.L.; Breuner, C.C.; Brown, M.A. Randomized Controlled Clinical Trial of Yoga in the Treatment of Eating Disorders. J. Adolesc. Health 2010, 46, 346–351. [Google Scholar] [CrossRef]
- Borden, A.; Cook-Cottone, C. Yoga and Eating Disorder Prevention and Treatment: A Comprehensive Review and Meta-Analysis. Eat. Disord. 2020, 28, 400–437. [Google Scholar] [CrossRef]
- Rizzuto, L.; Hay, P.; Noetel, M.; Touyz, S. Yoga as Adjunctive Therapy in the Treatment of People with Anorexia Nervosa: A Delphi Study. J. Eat. Disord. 2021, 9, 111. [Google Scholar] [CrossRef] [PubMed]
- del Valle, M.F.; Pérez, M.; Santana-Sosa, E.; Fiuza-Luces, C.; Bustamante-Ara, N.; Gallardo, C.; Villasenor, A.; Graell, M.; Morandé, G.; Romo, G.R. Does Resistance Training Improve the Functional Capacity and Well Being of Very Young Anorexic Patients? A Randomized Controlled Trial. J. Adolesc. Health 2010, 46, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-del-Valle, M.; Larumbe-Zabala, E.; Villaseñor-Montarroso, A.; Cardona Gonzalez, C.; Diez-Vega, I.; Lopez Mojares, L.M.; Perez Ruiz, M. Resistance Training Enhances Muscular Performance in Patients with Anorexia Nervosa: A Randomized Controlled Trial. Int. J. Eat. Disord. 2014, 47, 601–609. [Google Scholar] [CrossRef] [PubMed]
- Trott, M.; Jackson, S.E.; Firth, J.; Jacob, L.; Grabovac, I.; Mistry, A.; Stubbs, B.; Smith, L. A Comparative Meta-Analysis of the Prevalence of Exercise Addiction in Adults with and without Indicated Eating Disorders. Eat. Weight Disord.-Stud. Anorex. Bulim. Obes. 2021, 26, 37–46. [Google Scholar] [CrossRef]
- Galmiche, M.; Déchelotte, P.; Lambert, G.; Tavolacci, M.P. Prevalence of Eating Disorders over the 2000–2018 Period: A Systematic Literature Review. Am. J. Clin. Nutr. 2019, 109, 1402–1413. [Google Scholar] [CrossRef]
- Ward, Z.J.; Rodriguez, P.; Wright, D.R.; Austin, S.B.; Long, M.W. Estimation of Eating Disorders Prevalence by Age and Associations with Mortality in a Simulated Nationally Representative US Cohort. JAMA Netw. Open 2019, 2, e1912925. [Google Scholar] [CrossRef]
- Keski-Rahkonen, A. Epidemiology of Binge Eating Disorder: Prevalence, Course, Comorbidity, and Risk Factors. Curr. Opin. Psychiatry 2021, 34, 525–531. [Google Scholar] [CrossRef]
- Grilo, C.M.; Ivezaj, V.; Lydecker, J.A.; White, M.A. Toward an Understanding of the Distinctiveness of Body-Image Constructs in Persons Categorized with Overweight/Obesity, Bulimia Nervosa, and Binge-Eating Disorder. J. Psychosom. Res. 2019, 126, 109757. [Google Scholar] [CrossRef]
- Perrotta, G. Neural Correlates in Eating Disorders: Definition, Contexts and Clinical Strategies. J. Pub. Heath Cat. 2019, 2, 137–148. [Google Scholar]
- Hay, P. Current Approach to Eating Disorders: A Clinical Update. Intern. Med. J. 2020, 50, 24–29. [Google Scholar] [CrossRef]
- Solmi, M.; Radua, J.; Stubbs, B.; Ricca, V.; Moretti, D.; Busatta, D.; Carvalho, A.F.; Dragioti, E.; Favaro, A.; Monteleone, A.M. Risk Factors for Eating Disorders: An Umbrella Review of Published Meta-Analyses. Braz. J. Psychiatry 2020, 43, 314–323. [Google Scholar] [CrossRef]
- McCabe, M.; Tatangelo, G.; Watson, B.; Fuller-Tyszkiewicz, M.; Rodgers, R.F.; Aimé, A.; Mellor, D.; Granero-Gallegos, A.; Strodl, E.; Caltabiano, M. Development and Testing of a Model for Risk and Protective Factors for Eating Disorders and Higher Weight among Emerging Adults: A Study Protocol. Body Image 2019, 31, 139–149. [Google Scholar] [CrossRef]
- Herle, M.; De Stavola, B.; Hübel, C.; Abdulkadir, M.; Ferreira, D.S.; Loos, R.J.F.; Bryant-Waugh, R.; Bulik, C.M.; Micali, N. A Longitudinal Study of Eating Behaviours in Childhood and Later Eating Disorder Behaviours and Diagnoses. Br. J. Psychiatry 2020, 216, 113–119. [Google Scholar] [CrossRef]
- Graell, M.; Morón-Nozaleda, M.G.; Camarneiro, R.; Villaseñor, Á.; Yáñez, S.; Muñoz, R.; Martínez-Núñez, B.; Miguélez-Fernández, C.; Muñoz, M.; Faya, M. Children and Adolescents with Eating Disorders during COVID-19 Confinement: Difficulties and Future Challenges. Eur. Eat. Disord. Rev. 2020, 28, 864–870. [Google Scholar] [CrossRef]
- Cooper, M.; Reilly, E.E.; Siegel, J.A.; Coniglio, K.; Sadeh-Sharvit, S.; Pisetsky, E.M.; Anderson, L.M. Eating Disorders during the COVID-19 Pandemic and Quarantine: An Overview of Risks and Recommendations for Treatment and Early Intervention. Eat. Disord. 2022, 30, 54–76. [Google Scholar] [CrossRef]
- Fernández-Aranda, F.; Munguía, L.; Mestre-Bach, G.; Steward, T.; Etxandi, M.; Baenas, I.; Granero, R.; Sánchez, I.; Ortega, E.; Andreu, A. COVID Isolation Eating Scale (CIES): Analysis of the Impact of Confinement in Eating Disorders and Obesity—A Collaborative International Study. Eur. Eat. Disord. Rev. 2020, 28, 871–883. [Google Scholar] [CrossRef]
- Murray, S.B. Updates in the Treatment of Eating Disorders in 2018: A Year in Review in Eating Disorders: The Journal of Treatment & Prevention. Eat. Disord. 2019, 27, 6–17. [Google Scholar]
- Hale, M.D.; Logomarsino, J.V. The Use of Enteral Nutrition in the Treatment of Eating Disorders: A Systematic Review. Eat. Weight Disord.-Stud. Anorex. Bulim. Obes. 2019, 24, 179–198. [Google Scholar] [CrossRef]
- Agras, W.S. Cognitive Behavior Therapy for the Eating Disorders. Psychiatr. Clin. 2019, 42, 169–179. [Google Scholar] [CrossRef]
- Bohon, C. Binge Eating Disorder in Children and Adolescents. Child Adolesc. Psychiatr. Clin. 2019, 28, 549–555. [Google Scholar] [CrossRef]
- Puls, H.; Schmidt, R.; Hilbert, A. Therapist Adherence and Therapeutic Alliance in Individual Cognitive-behavioural Therapy for Adolescent Binge-eating Disorder. Eur. Eat. Disord. Rev. 2019, 27, 182–194. [Google Scholar] [CrossRef] [PubMed]
- Flynn, M.; Austin, A.; Lang, K.; Allen, K.; Bassi, R.; Brady, G.; Brown, A.; Connan, F.; Franklin-Smith, M.; Glennon, D. Assessing the Impact of First Episode Rapid Early Intervention for Eating Disorders on Duration of Untreated Eating Disorder: A Multi-centre Quasi-experimental Study. Eur. Eat. Disord. Rev. 2021, 29, 458–471. [Google Scholar] [CrossRef] [PubMed]
- Le Grange, D.; Eckhardt, S.; Dalle Grave, R.; Crosby, R.D.; Peterson, C.B.; Keery, H.; Lesser, J.; Martell, C. Enhanced Cognitive-Behavior Therapy and Family-Based Treatment for Adolescents with an Eating Disorder: A Non-Randomized Effectiveness Trial. Psychol. Med. 2022, 52, 2520–2530. [Google Scholar] [CrossRef] [PubMed]
- Davis, L.E.; Attia, E. Recent Advances in Therapies for Eating Disorders. F1000Research 2019, 8. [Google Scholar] [CrossRef]
- Linardon, J.; Shatte, A.; Messer, M.; Firth, J.; Fuller-Tyszkiewicz, M. E-Mental Health Interventions for the Treatment and Prevention of Eating Disorders: An Updated Systematic Review and Meta-Analysis. J. Consult. Clin. Psychol. 2020, 88, 994. [Google Scholar] [CrossRef]
- Kakhramonovich, T.P. Epidemiology of Pysichiatric Disorders. Tex. J. Med. Sci. 2022, 12, 102–105. [Google Scholar]
- Todisco, P.; Meneguzzo, P.; Garolla, A.; Antoniades, A.; Vogazianos, P.; Tozzi, F. Impulsive Behaviors and Clinical Outcomes Following a Flexible Intensive Inpatient Treatment for Eating Disorders: Findings from an Observational Study. Eat. Weight Disord.-Stud. Anorex. Bulim. Obes. 2021, 26, 869–877. [Google Scholar] [CrossRef]
- Dahlenburg, S.C.; Gleaves, D.H.; Hutchinson, A.D. Treatment Outcome Research of Enhanced Cognitive Behaviour Therapy for Eating Disorders: A Systematic Review with Narrative and Meta-Analytic Synthesis. Eat. Disord. 2019, 27, 482–502. [Google Scholar] [CrossRef]
- Joshua, P.R.; Lewis, V.; Kelty, S.F.; Boer, D.P. Is Schema Therapy Effective for Adults with Eating Disorders? A Systematic Review into the Evidence. Cogn. Behav. Ther. 2023, 52, 1–19. [Google Scholar] [CrossRef]
- Smith, E.; Simpson, S.; McIntosh, V.V.W. Introduction to Schema Therapy for Eating Disorders. In Schema Therapy for Eating Disorders; Routledge: Abingdon, UK, 2019; pp. 3–11. ISBN 0429295715. [Google Scholar]
- Thomas, J.J.; Becker, K.R.; Kuhnle, M.C.; Jo, J.H.; Harshman, S.G.; Wons, O.B.; Keshishian, A.C.; Hauser, K.; Breithaupt, L.; Liebman, R.E. Cognitive-behavioral Therapy for Avoidant/Restrictive Food Intake Disorder: Feasibility, Acceptability, and Proof-of-concept for Children and Adolescents. Int. J. Eat. Disord. 2020, 53, 1636–1646. [Google Scholar] [CrossRef]
- Linardon, J.; Kothe, E.J.; Fuller-Tyszkiewicz, M. Efficacy of Psychotherapy for Bulimia Nervosa and Binge-eating Disorder on Self-esteem Improvement: Meta-analysis. Eur. Eat. Disord. Rev. 2019, 27, 109–123. [Google Scholar] [CrossRef]
- Butler, R.M.; Heimberg, R.G. Exposure Therapy for Eating Disorders: A Systematic Review. Clin. Psychol. Rev. 2020, 78, 101851. [Google Scholar] [CrossRef]
- Citrome, L. Binge Eating Disorder Revisited: What’s New, What’s Different, What’s next. CNS Spectr. 2019, 24, 4–13. [Google Scholar] [CrossRef]
- Coelho, J.S.; Suen, J.; Clark, B.A.; Marshall, S.K.; Geller, J.; Lam, P.-Y. Eating Disorder Diagnoses and Symptom Presentation in Transgender Youth: A Scoping Review. Curr. Psychiatry Rep. 2019, 21, 107. [Google Scholar] [CrossRef]
- Zickgraf, H.F.; Ellis, J.M.; Essayli, J.H. Disentangling Orthorexia Nervosa from Healthy Eating and Other Eating Disorder Symptoms: Relationships with Clinical Impairment, Comorbidity, and Self-Reported Food Choices. Appetite 2019, 134, 40–49. [Google Scholar] [CrossRef]
- Van Zyl, N.; Andrews, L.; Williamson, H.; Meyrick, J. The Effectiveness of Psychosocial Interventions to Support Psychological Well-Being in Post-Operative Bariatric Patients: A Systematic Review of Evidence. Obes. Res. Clin. Pract. 2020, 14, 404–420. [Google Scholar] [CrossRef]
- Fiorillo, A.; Luciano, M.; Pompili, M.; Sartorius, N. Reducing the Mortality Gap in People with Severe Mental Disorders: The Role of Lifestyle Psychosocial Interventions. Front. Psychiatry 2019, 10, 434. [Google Scholar] [CrossRef]
- Barbui, C.; Purgato, M.; Abdulmalik, J.; Acarturk, C.; Eaton, J.; Gastaldon, C.; Gureje, O.; Hanlon, C.; Jordans, M.; Lund, C. Efficacy of Psychosocial Interventions for Mental Health Outcomes in Low-Income and Middle-Income Countries: An Umbrella Review. Lancet Psychiatry 2020, 7, 162–172. [Google Scholar] [CrossRef]
- Unikel-Santoncini, C.; de León-Vázquez, C.D.; Rivera-Márquez, J.A.; Bojorquez-Chapela, I.; Méndez-Ríos, E. Dissonance-Based Program for Eating Disorders Prevention in Mexican University Students. Psychosoc. Interv. 2019, 28, 29–35. [Google Scholar] [CrossRef]
- Pina, A.A.; Polo, A.J.; Huey, S.J. Evidence-Based Psychosocial Interventions for Ethnic Minority Youth: The 10-Year Update. J. Clin. Child Adolesc. Psychol. 2019, 48, 179–202. [Google Scholar] [CrossRef]
- Derenne, J.L.; Beresin, E. V Body Image, Media, and Eating Disorders. Acad. Psychiatry 2006, 30, 257–261. [Google Scholar] [CrossRef] [PubMed]
- Ríos, P.B. Nutritional Treatment in Eating Disorders. In Relevant Topics in Eating Disorders; IntechOpen: London, UK, 2012; ISBN 9535100017. [Google Scholar]
- Napolitano, F.; Bencivenga, F.; Pompili, E.; Angelillo, I.F. Assessment of Knowledge, Attitudes, and Behaviors toward Eating Disorders among Adolescents in Italy. Int. J. Environ. Res. Public. Health 2019, 16, 1448. [Google Scholar] [CrossRef] [PubMed]
- Nagata, J.M.; Ganson, K.T.; Austin, S.B. Emerging Trends in Eating Disorders among Sexual and Gender Minorities. Curr. Opin. Psychiatry 2020, 33, 562. [Google Scholar] [CrossRef]
- Favaro, A.; Busetto, P.; Collantoni, E.; Santonastaso, P. The Age of Onset of Eating Disorders. In Age of Onset of Mental Disorders; Springer: Cham, Switzerland; Midtown Manhattan: New York, NY, USA, 2019; pp. 203–216. [Google Scholar]
- Demmler, J.C.; Brophy, S.T.; Marchant, A.; John, A.; Tan, J.O.A. Shining the Light on Eating Disorders, Incidence, Prognosis and Profiling of Patients in Primary and Secondary Care: National Data Linkage Study. Br. J. Psychiatry 2020, 216, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Qian, J.; Wu, Y.; Liu, F.; Zhu, Y.; Jin, H.; Zhang, H.; Wan, Y.; Li, C.; Yu, D. An Update on the Prevalence of Eating Disorders in the General Population: A Systematic Review and Meta-Analysis. Eat. Weight Disord.-Stud. Anorex. Bulim. Obes. 2022, 27, 415–428. [Google Scholar] [CrossRef]
- Linardon, J.; Gleeson, J.; Yap, K.; Murphy, K.; Brennan, L. Meta-Analysis of the Effects of Third-Wave Behavioural Interventions on Disordered Eating and Body Image Concerns: Implications for Eating Disorder Prevention. Cogn. Behav. Ther. 2019, 48, 15–38. [Google Scholar] [CrossRef]
- Nightingale, B.A.; Cassin, S.E. Disordered Eating among Individuals with Excess Weight: A Review of Recent Research. Curr. Obes. Rep. 2019, 8, 112–127. [Google Scholar] [CrossRef]
- Cameron, K.; Ogrodniczuk, J.; Hadjipavlou, G. Changes in Alexithymia Following Psychological Intervention: A Review. Harv. Rev. Psychiatry 2014, 22, 162–178. [Google Scholar] [CrossRef]
- Vandereycken, W.; Depreitere, L.; Probst, M. Body-Oriented Therapy for Anorexia Nervosa Patients. Am. J. Psychother. 1987, 41, 252–259. [Google Scholar] [CrossRef]
- Nozaki, T.; Takao, M.; Takakura, S.; Koreeda-Arimura, C.; Ishido, K.; Yamada, Y.; Kawai, K.; Takii, M.; Kubo, C. Psychopathological Features of Patients with Prolonged Anorexia Nervosa as Assessed by the Minnesota Multiphasic Personality Inventory. Eat. Weight Disord.-Stud. Anorex. Bulim. Obes. 2006, 11, 59–65. [Google Scholar] [CrossRef]
- Probst, M.; Majeweski, M.L.; Albertsen, M.N.; Catalan-Matamoros, D.; Danielsen, M.; De Herdt, A.; Duskova Zakova, H.; Fabricius, S.; Joern, C.; Kjölstad, G. Physiotherapy for Patients with Anorexia Nervosa. Adv. Eat. Disord. Theory Res. Pract. 2013, 1, 224–238. [Google Scholar] [CrossRef]
- Thien, V.; Thomas, A.; Markin, D.; Birmingham, C.L. Pilot Study of a Graded Exercise Program for the Treatment of Anorexia Nervosa. Int. J. Eat. Disord. 2000, 28, 101–106. [Google Scholar] [CrossRef]
- Agne, A.; Quesnel, D.A.; Larumbe-Zabala, E.; Olmedillas, H.; Graell-Berna, M.; Perez-Ruiz, M.; Fernandez-del-Valle, M. Progressive Resistance Exercise as Complementary Therapy Improves Quality of Life and Body Composition in Anorexia Nervosa: A Randomized Controlled Trial. Complement. Ther. Clin. Pract. 2022, 48, 101576. [Google Scholar] [CrossRef]
- Field, T.; Schanberg, S.; Kuhn, C.; Field, T.; Fierro, K.; Henteleff, T.; Mueller, C.; Yando, R.; Shaw, S.; Burman, I. Bulimic Adolescents Benefit from Massage Therapy. Adolescence 1998, 33, 555–564. [Google Scholar]
- Field, T.M. Massage Therapy Effects. Am. Psychol. 1998, 53, 1270. [Google Scholar] [CrossRef] [PubMed]
- Pawlow, L.A.; O’neil, P.M.; Malcolm, R.J. Night Eating Syndrome: Effects of Brief Relaxation Training on Stress, Mood, Hunger, and Eating Patterns. Int. J. Obes. 2003, 27, 970–978. [Google Scholar] [CrossRef] [PubMed]
- Wanden-Berghe, R.G.; Sanz-Valero, J.; Wanden-Berghe, C. The Application of Mindfulness to Eating Disorders Treatment: A Systematic Review. Eat. Disord. 2010, 19, 34–48. [Google Scholar] [CrossRef] [PubMed]
- Probst, M.; Van Coppenolle, H.; Vandereycken, W. Body Experience in Anorexia Nervosa Patients: An Overview of Therapeutic Approaches. Eat. Disord. 1995, 3, 145–157. [Google Scholar] [CrossRef]
- Zipfel, S.; Giel, K.E.; Bulik, C.M.; Hay, P.; Schmidt, U. Anorexia Nervosa: Aetiology, Assessment, and Treatment. Lancet Psychiatry 2015, 2, 1099–1111. [Google Scholar] [CrossRef]
| Author and Year | Study Title | Aim of Study | Main Outcomes and Effectiveness | Duration | Type of Intervention |
|---|---|---|---|---|---|
| Marzola et al. (2013) [138] | Nutritional Rehabilitation in Anorexia nervosa: Review of the Literature and Implications for Treatment | To describe issues related to the caloric requirements needed to gain and maintain weight for short and long-term recovery for AN inpatients and outpatients. | The restoration of both nutrient status and weight starts slowly and gradually accelerate as tolerated. | Several weeks | Nutritional |
| Andrewes et al. (1996) [139] | Computerised psychoeducation for patients with eating disorders | To assess a new computer-based method of health education for patients with bulimia and AN. | The DIET group members were significantly improved when compared to the placebo group in terms of both their knowledge and attitudes towards their disorder. | Not specified | Nutritional |
| Ng et al. (2013) [140] | Is supervised exercise training safe in patients with anorexia nervosa? A meta-analysis | To examine the effects of supervised exercise training in patients with AN. | Significant improvement in weight and body fat; strength and cardiovascular fitness were also shown to improve. | >2 h/week | Physical Activity |
| Rizk et al. (2018) [141] | High-intensity exercise is associated with a better nutritional status in anorexia nervosa. | To investigate the links between duration and intensity of exercise and the nutritional status in terms of body composition in acute AN patients. | Exercising at higher intensity in AN is associated with a better nutritional status. | >9 h/week | Physical Activity |
| Grave et al. (2014) [142] | Inpatient cognitive behavior therapy for adolescents with anorexia nervosa: immediate and longer-term effects. | To establish the immediate and longer-term effects of a novel inpatient program for adolescents that was designed to produce enduring change. | Enhanced cognitive behavior therapy is a promising approach to the treatment of adolescents with severe anorexia nervosa. | 20 weeks | Psychological |
| Steinglass et al. (2012) [143] | Fear of Food as a Treatment Target: Exposure and Response Prevention for Anorexia Nervosa in an Open Series. | To evaluate the potential utility of addressing eating-related fear in the treatment of AN using psychotherapy techniques known to be effective in the treatment of anxiety disorders | Change in anxiety with AN was associated with greater caloric intake | 4 weeks | Psychological |
| Fisher et al. (2018) [144] | Family therapy approaches for anorexia nervosa | To evaluate the efficacy of family therapy approaches compared with standard treatment and other treatments for AN | There was some evidence of a small effect favoring family based therapy compared with other psychosocial interventions in terms of weight gain post-intervention. | No specified | Psychosocial |
| Barber et al. (2018) [145] | Reducing the Mortality in People with Severe Mental Disorders: The Role of Lifestyle Psychosocial Interventions | To explore the causes of death in high income and low and middle-income countries and review the multi-level risk factor model for mortality in severe mental disorders | Nurse-led services and the utilization of peer support are showing promise outcomes. | >6 months | Psychosocial |
| Hart et al. (2001) [146] | Anorexia nervosa Symptoms are Reduced by Massage Therapy | To Evaluate massage therapy for women with AN for (1) reducing stress and stress hormone levels, (2) decreasing depression, (3) improving mood, (4) reducing eating disorder symptoms, and (5) increasing dopamine values | Reduced anxiety following their first and last treatment; decreases in body dissatisfaction on the Eating Disorder Inventory and increased dopamine and norepinephrine levels. | 5 weeks | Physical Therapy |
| Fogarty et al. (2013) [147] | Patients with anorexia nervosa receiving acupuncture or acupressure their view of the therapeutic encounter | To investigate the views of patients with AN receiving an acupuncture or acupressure intervention. | Patients perceive the therapeutic relationship and empathy as important qualities of the acupuncture or acupressure intervention as an adjunct therapy for the treatment of AN. | 3 weeks | Physical Therapy |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clemente-Suárez, V.J.; Ramírez-Goerke, M.I.; Redondo-Flórez, L.; Beltrán-Velasco, A.I.; Martín-Rodríguez, A.; Ramos-Campo, D.J.; Navarro-Jiménez, E.; Yáñez-Sepúlveda, R.; Tornero-Aguilera, J.F. The Impact of Anorexia Nervosa and the Basis for Non-Pharmacological Interventions. Nutrients 2023, 15, 2594. https://doi.org/10.3390/nu15112594
Clemente-Suárez VJ, Ramírez-Goerke MI, Redondo-Flórez L, Beltrán-Velasco AI, Martín-Rodríguez A, Ramos-Campo DJ, Navarro-Jiménez E, Yáñez-Sepúlveda R, Tornero-Aguilera JF. The Impact of Anorexia Nervosa and the Basis for Non-Pharmacological Interventions. Nutrients. 2023; 15(11):2594. https://doi.org/10.3390/nu15112594
Chicago/Turabian StyleClemente-Suárez, Vicente Javier, Maria Isabel Ramírez-Goerke, Laura Redondo-Flórez, Ana Isabel Beltrán-Velasco, Alexandra Martín-Rodríguez, Domingo Jesús Ramos-Campo, Eduardo Navarro-Jiménez, Rodrigo Yáñez-Sepúlveda, and José Francisco Tornero-Aguilera. 2023. "The Impact of Anorexia Nervosa and the Basis for Non-Pharmacological Interventions" Nutrients 15, no. 11: 2594. https://doi.org/10.3390/nu15112594
APA StyleClemente-Suárez, V. J., Ramírez-Goerke, M. I., Redondo-Flórez, L., Beltrán-Velasco, A. I., Martín-Rodríguez, A., Ramos-Campo, D. J., Navarro-Jiménez, E., Yáñez-Sepúlveda, R., & Tornero-Aguilera, J. F. (2023). The Impact of Anorexia Nervosa and the Basis for Non-Pharmacological Interventions. Nutrients, 15(11), 2594. https://doi.org/10.3390/nu15112594










