Translational Research on Bee Pollen as a Source of Nutrients: A Scoping Review from Bench to Real World
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
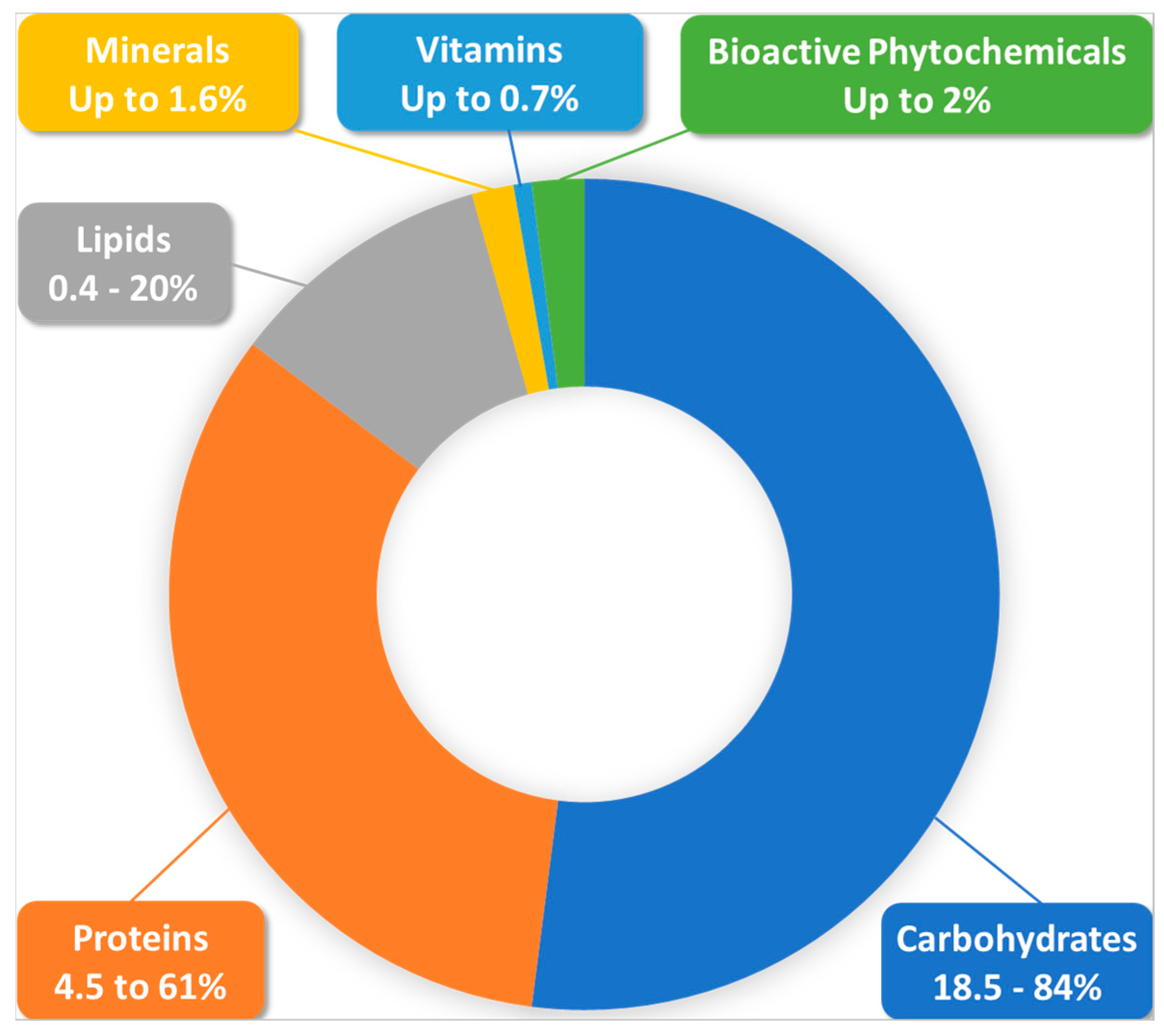
3.1. Nutritional Value of BP
3.2. BP as a Functional Nutrient Source
3.2.1. Nutritional Deficiencies
3.2.2. Antioxidant Potential
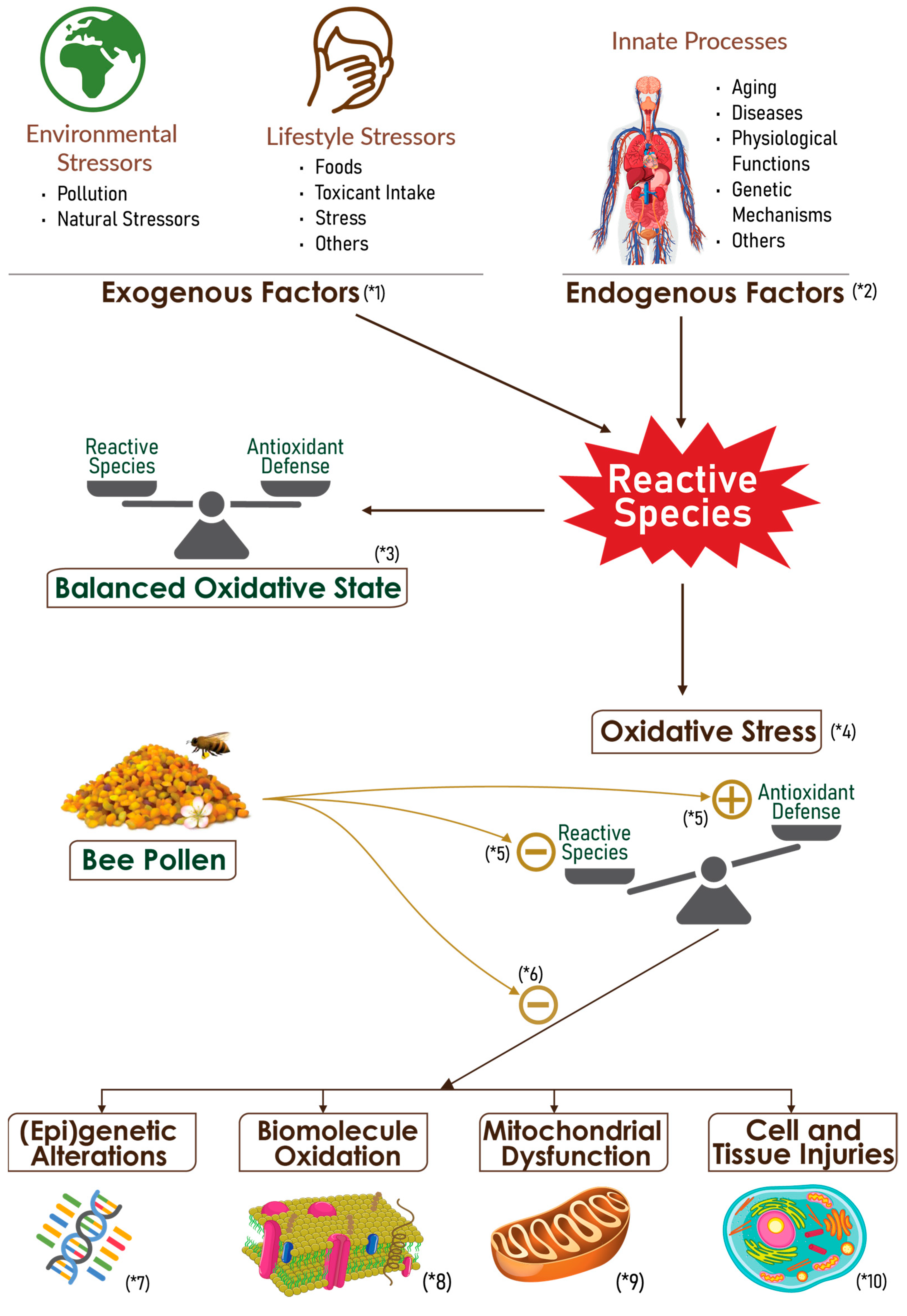
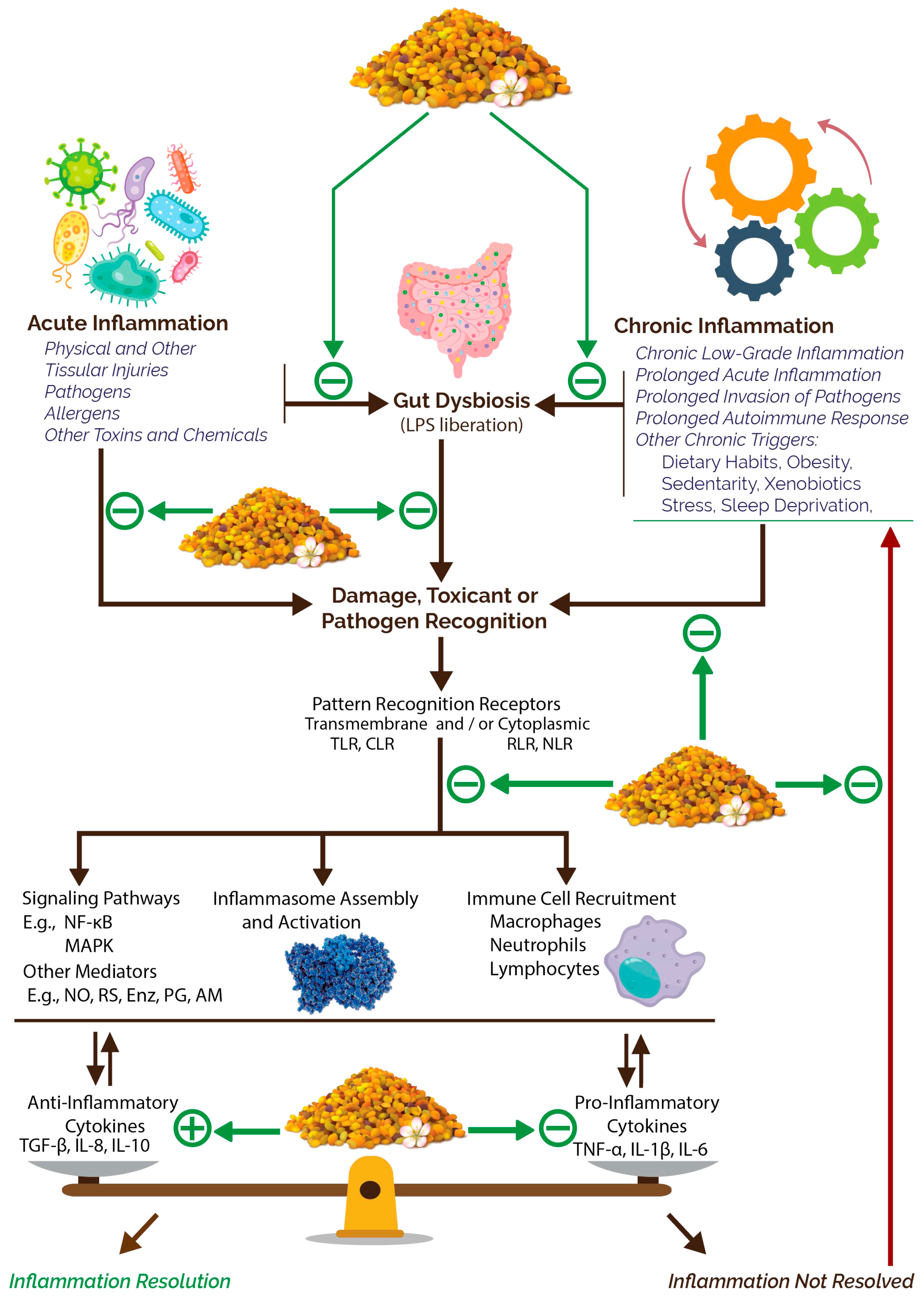
3.2.3. Anti-Inflammatory Potential
3.2.4. Digestive Health
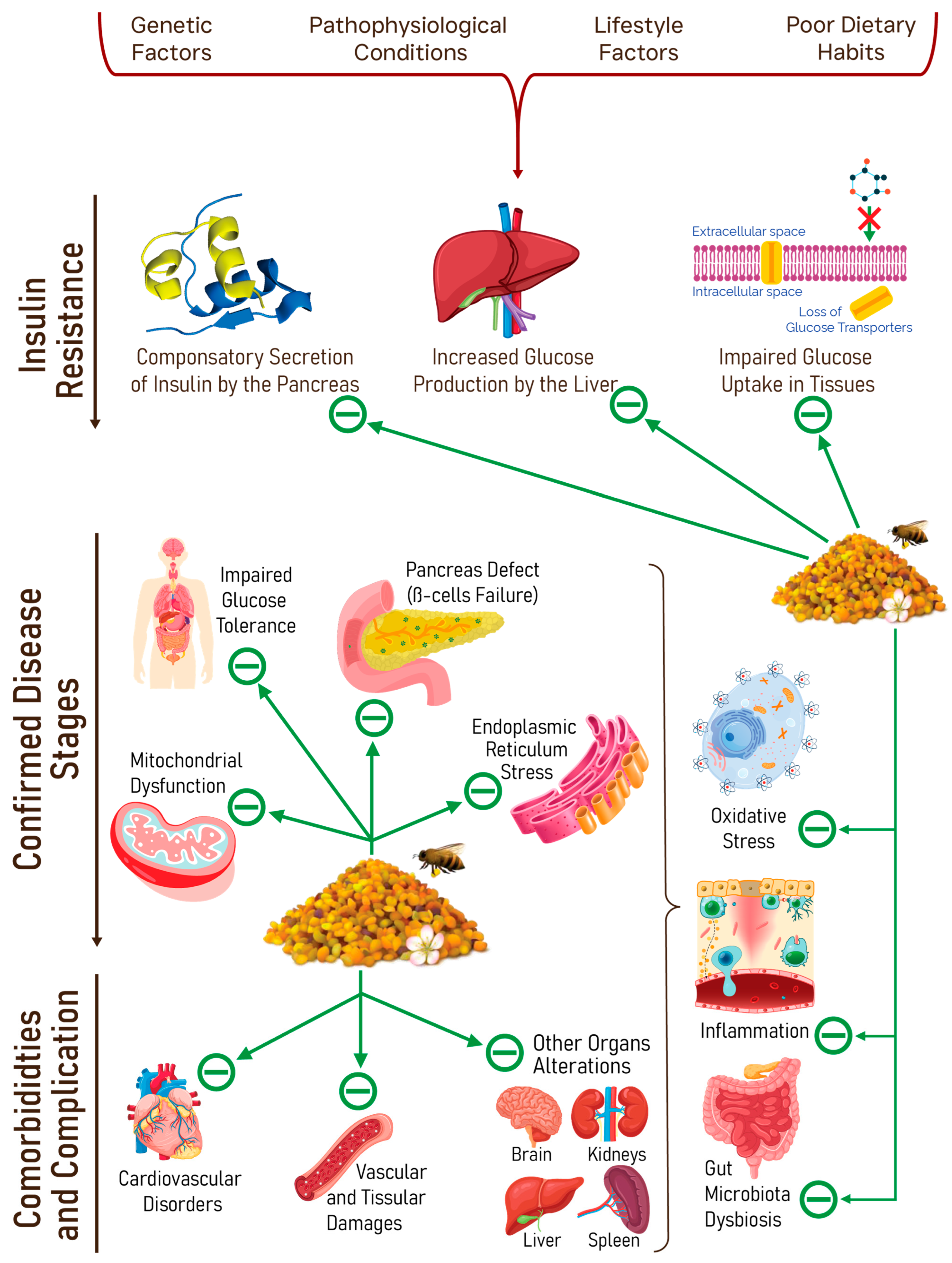
3.2.5. Metabolic Disorders
Obesity
Diabetes Mellitus
Dyslipidemia
Hyperuricemia
3.2.6. Cardiovascular Diseases
3.3. BP as a Food Adjuvant
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Campos, M.G.; Frigerio, C.; Bobiş, O.; Urcan, A.C.; Gomes, N.G.M. Infrared irradiation drying impact on bee pollen: Case study on the phenolic composition of Eucalyptus globulus labill and Salix atrocinerea Brot. pollens. Processes 2021, 9, 890. [Google Scholar] [CrossRef]
- Campos, M.G.; Anjos, O.; Chica, M.; Campoy, P.; Nozkova, J.; Almaraz-Abarca, N.; Barreto, L.M.R.C.; Nordi, J.C.; Estevinho, L.M.; Pascoal, A.; et al. Standard methods for pollen research. J. Apic. Res. 2021, 60, 1–109. [Google Scholar] [CrossRef]
- Matuszewska, E.; Plewa, S.; Pietkiewicz, D.; Kossakowski, K.; Matysiak, J.; Rosiński, G.; Matysiak, J. Mass Spectrometry-Based Identification of Bioactive Bee Pollen Proteins: Evaluation of Allergy Risk after Bee Pollen Supplementation. Molecules 2022, 27, 7733. [Google Scholar] [CrossRef] [PubMed]
- Kostić, A.Ž.; Milinčić, D.D.; Barać, M.B.; Shariati, M.A.; Tešić, L.; Pešić, M.B. The Application of Pollen as a Functional Food and Feed Ingredient—The Present and Perspectives. Biomolecules 2020, 10, 84. [Google Scholar] [CrossRef]
- Baky, M.H.; Abouelela, M.B.; Wang, K.; Farag, M.A. Bee Pollen and Bread as a Super-Food: A Comparative Review of Their Metabolome Composition and Quality Assessment in the Context of Best Recovery Conditions. Molecules 2023, 28, 715. [Google Scholar] [CrossRef]
- Xue, F.; Li, C. Effects of ultrasound assisted cell wall disruption on physicochemical properties of camellia bee pollen protein isolates. Ultrason. Sonochem. 2023, 92, 106249. [Google Scholar] [CrossRef]
- Alshallash, K.S.; Abolaban, G.; Elhamamsy, S.M.; Zaghlool, A.; Nasr, A.; Nagib, A.; El-Hakim, A.F.A.; Zahra, A.A.; Hamdy, A.E.; Taha, I. Bee Pollen as a Functional Product–Chemical Constituents and Nutritional Properties. J. Ecol. Eng. 2023, 24, 173–183. [Google Scholar] [CrossRef]
- Végh, R.; Csóka, M.; Stefanovits-Bányai, É.; Juhász, R.; Sipos, L. Biscuits Enriched with Monofloral Bee Pollens: Nutritional Properties, Techno-Functional Parameters, Sensory Profile, and Consumer Preference. Foods 2022, 12, 18. [Google Scholar] [CrossRef]
- Rojo, S.; Escuredo, O.; Rodríguez-Flores, M.S.; Seijo, M.C. Botanical Origin of Galician Bee Pollen (Northwest Spain) for the Characterization of Phenolic Content and Antioxidant Activity. Foods 2023, 12, 294. [Google Scholar] [CrossRef]
- Hanafy, N.A.N.; Salim, E.I.; Mahfouz, M.E.; Eltonouby, E.A.; Hamed, I.H. Fabrication and characterization of bee pollen extract nanoparticles: Their potential in combination therapy against human A549 lung cancer cells. Food Hydrocoll. Heal. 2023, 3, 100110. [Google Scholar] [CrossRef]
- Chelucci, E.; Chiellini, C.; Cavallero, A.; Gabriele, M. Bio-Functional Activities of Tuscan Bee Pollen. Antioxidants 2023, 12, 115. [Google Scholar] [CrossRef] [PubMed]
- Zhou, E.; Xue, X.; Xu, H.; Zhao, L.; Wu, L.; Li, Q. Effects of covalent conjugation with quercetin and its glycosides on the structure and allergenicity of Bra c p from bee pollen. Food Chem. 2023, 406, 135075. [Google Scholar] [CrossRef] [PubMed]
- Qiao, J.; Feng, Z.; Zhang, Y.; Xiao, X.; Dong, J.; Haubruge, E.; Zhang, H. Phenolamide and flavonoid glycoside profiles of 20 types of monofloral bee pollen. Food Chem. 2023, 405, 134800. [Google Scholar] [CrossRef] [PubMed]
- Oroian, M.; Dranca, F.; Ursachi, F. Characterization of Romanian Bee Pollen—An Important Nutritional Source. Foods 2022, 11, 2633. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Chen, L.; Zhang, Y.; Xie, S.; Yang, J.; Su, S.; Yao, H.; Shi, P. Lotus Bee Pollen Extract Inhibits Isoproterenol-Induced Hypertrophy via JAK2/STAT3 Signaling Pathway in Rat H9c2 Cells. Antioxidants 2022, 12, 88. [Google Scholar] [CrossRef] [PubMed]
- Algethami, J.S.; El-Wahed, A.A.A.; Elashal, M.H.; Ahmed, H.R.; Elshafiey, E.H.; Omar, E.M.; Al Naggar, Y.; Algethami, A.F.; Shou, Q.; Alsharif, S.M.; et al. Bee Pollen: Clinical Trials and Patent Applications. Nutrients 2022, 14, 2858. [Google Scholar] [CrossRef]
- El-Seedi, H.R.; Khalifa, S.A.M.; El-Wahed, A.A.; Gao, R.; Guo, Z.; Tahir, H.E.; Zhao, C.; Du, M.; Farag, M.A.; Musharraf, S.G.; et al. Honeybee products: An updated review of neurological actions. Trends Food Sci. Technol. 2020, 101, 17–27. [Google Scholar] [CrossRef]
- Camacho-Bernal, G.I.; Cruz-Cansino, N.d.S.; Ramírez-Moreno, E.; Delgado-Olivares, L.; Zafra-Rojas, Q.Y.; Castañeda-Ovando, A.; Suárez-Jacobo, Á. Addition of Bee Products in Diverse Food Sources: Functional and Physicochemical Properties. Appl. Sci. 2021, 11, 8156. [Google Scholar] [CrossRef]
- Xi, X.; Li, J.; Guo, S.; Li, Y.; Xu, F.; Zheng, M.; Cao, H.; Cui, X.; Guo, H.; Han, C. The Potential of Using Bee Pollen in Cosmetics: A Review. J. Oleo Sci. 2018, 67, 1071–1082. [Google Scholar] [CrossRef]
- Lu, P.; Takiguchi, S.; Honda, Y.; Lu, Y.; Mitsui, T.; Kato, S.; Kodera, R.; Furihata, K.; Zhang, M.; Okamoto, K.; et al. NMR and HPLC profiling of bee pollen products from different countries. Food Chem. Mol. Sci. 2022, 5, 100119. [Google Scholar] [CrossRef]
- Tao, Y.; Zhou, E.; Li, F.; Meng, L.; Li, Q.; Wu, L. Allergenicity Alleviation of Bee Pollen by Enzymatic Hydrolysis: Regulation in Mice Allergic Mediators, Metabolism, and Gut Microbiota. Foods 2022, 11, 3454. [Google Scholar] [CrossRef] [PubMed]
- Munn, Z.; Pollock, D.; Khalil, H.; Alexander, L.; Mclnerney, P.; Godfrey, C.M.; Peters, M.; Tricco, A.C. What are scoping reviews? Providing a formal definition of scoping reviews as a type of evidence synthesis. JBI Évid. Synth. 2022, 20, 950–952. [Google Scholar] [CrossRef] [PubMed]
- Bradbury-Jones, C.; Aveyard, H.; Herber, O.R.; Isham, L.; Taylor, J.; O’malley, L. Scoping reviews: The PAGER framework for improving the quality of reporting. Int. J. Soc. Res. Methodol. 2022, 25, 457–470. [Google Scholar] [CrossRef]
- Maggio, L.A.; Larsen, K.; Thomas, A.; Costello, J.A.; Artino, A.R. Scoping reviews in medical education: A scoping review. Med. Educ. 2021, 55, 689–700. [Google Scholar] [CrossRef] [PubMed]
- Peters, M.D.; Marnie, C.; Tricco, A.C.; Pollock, D.; Munn, Z.; Alexander, L.; McInerney, P.; Godfrey, C.M.; Khalil, H. Updated methodological guidance for the conduct of scoping reviews. JBI Évid. Synth. 2020, 18, 2119–2126. [Google Scholar] [CrossRef]
- Khalil, H.; Tricco, A.C. Differentiating between mapping reviews and scoping reviews in the evidence synthesis ecosystem. J. Clin. Epidemiol. 2022, 149, 175–182. [Google Scholar] [CrossRef]
- Pollock, D.; Tricco, A.C.; Peters, M.D.; Mclnerney, P.A.; Khalil, H.; Godfrey, C.M.; Alexander, L.A.; Munn, Z. Methodological quality, guidance, and tools in scoping reviews: A scoping review protocol. JBI Évid. Synth. 2022, 20, 1098–1105. [Google Scholar] [CrossRef]
- Buus, N.; Nygaard, L.; Berring, L.L.; Hybholt, L.; Kamionka, S.L.; Rossen, C.B.; Søndergaard, R.; Juel, A. Arksey and O′Malley’s consultation exercise in scoping reviews: A critical review. J. Adv. Nurs. 2022, 78, 2304–2312. [Google Scholar] [CrossRef]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Thakur, M.; Nanda, V. Composition and functionality of bee pollen: A review. Trends Food Sci. Technol. 2020, 98, 82–106. [Google Scholar] [CrossRef]
- Khalifa, S.A.M.; Elashal, M.H.; Yosri, N.; Du, M.; Musharraf, S.G.; Nahar, L.; Sarker, S.D.; Guo, Z.; Cao, W.; Zou, X.; et al. Bee Pollen: Current Status and Therapeutic Potential. Nutrients 2021, 13, 1876. [Google Scholar] [CrossRef] [PubMed]
- Sevin, S.; Tutun, H.; Yipel, M.; Aluç, Y.; Ekici, H. Concentration of essential and non-essential elements and carcinogenic/non-carcinogenic health risk assessment of commercial bee pollens from Turkey. J. Trace Elem. Med. Biol. 2023, 75, 127104. [Google Scholar] [CrossRef] [PubMed]
- Ben Bacha, A.; Norah, A.-O.; Al-Osaimi, M.; Harrath, A.H.; Mansour, L.; El-Ansary, A. The therapeutic and protective effects of bee pollen against prenatal methylmercury induced neurotoxicity in rat pups. Metab. Brain Dis. 2020, 35, 215–224. [Google Scholar] [CrossRef]
- Al-Osaimi, M.; El-Ansary, A.; Al-Daihan, S.; Bhat, R.S.; Ben Bacha, A. Therapeutic and Protective Potency of Bee Pollen Against Neurotoxic Effects Induced by Prenatal Exposure of Rats to Methyl Mercury. J. Mol. Neurosci. 2018, 65, 327–335. [Google Scholar] [CrossRef]
- Crone, M.K.; Grozinger, C.M. Pollen protein and lipid content influence resilience to insecticides in honey bees (Apis mellifera). J. Exp. Biol. 2021, 224, jeb242040. [Google Scholar] [CrossRef] [PubMed]
- Prđun, S.; Svečnjak, L.; Valentić, M.; Marijanović, Z.; Jerković, I. Characterization of Bee Pollen: Physico-Chemical Properties, Headspace Composition and FTIR Spectral Profiles. Foods 2021, 10, 2103. [Google Scholar] [CrossRef]
- Lau, P.; Lesne, P.; Grebenok, R.J.; Rangel, J.; Behmer, T.S. Assessing pollen nutrient content: A unifying approach for the study of bee nutritional ecology. Philos. Trans. R. Soc. B Biol. Sci. 2022, 377, 20210510. [Google Scholar] [CrossRef]
- Aylanc, V.; Falcão, S.I.; Vilas-Boas, M. Bee pollen and bee bread nutritional potential: Chemical composition and macronutrient digestibility under in vitro gastrointestinal system. Food Chem. 2023, 413, 135597. [Google Scholar] [CrossRef]
- Didaras, N.A.; Karatasou, K.; Dimitriou, T.G.; Amoutzias, G.D.; Mossialos, D. Antimicrobial Activity of Bee-Collected Pollen and Beebread: State of the Art and Future Perspectives. Antibiotics 2020, 9, 811. [Google Scholar] [CrossRef]
- dos Santos, T.R.; Melo, J.d.S.; dos Santos, A.V.; Severino, P.; Lima, S.; Souto, E.B.; Zielińska, A.; Cardoso, J.C. Development of a Protein-Rich By-Product by 23 Factorial Design: Characterization of Its Nutritional Value and Sensory Analysis. Molecules 2022, 27, 8918. [Google Scholar] [CrossRef] [PubMed]
- Hsu, P.-S.; Wu, T.-H.; Huang, M.-Y.; Wang, D.-Y.; Wu, M.-C. Nutritive Value of 11 Bee Pollen Samples from Major Floral Sources in Taiwan. Foods 2021, 10, 2229. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Campos, M.G.; Fratini, F.; Altaye, S.Z.; Li, J. New Insights into the Biological and Pharmaceutical Properties of Royal Jelly. Int. J. Mol. Sci. 2020, 21, 382. [Google Scholar] [CrossRef] [PubMed]
- Darwish, A.M.G.; El-Wahed, A.A.A.; Shehata, M.G.; El-Seedi, H.R.; Masry, S.H.D.; Khalifa, S.A.M.; Mahfouz, H.M.; El-Sohaimy, S.A. Chemical Profiling and Nutritional Evaluation of Bee Pollen, Bee Bread, and Royal Jelly and Their Role in Functional Fermented Dairy Products. Molecules 2022, 28, 227. [Google Scholar] [CrossRef] [PubMed]
- Jarosz, P.M.; Jasielski, P.P.; Zarobkiewicz, M.K.; Sławiński, M.A.; Wawryk-Gawda, E.; Jodłowska-Jędrych, B. Changes in Histological Structure, Interleukin 12, Smooth Muscle Actin and Nitric Oxide Synthase 1. and 3. Expression in the Liver of Running and Non-Running Wistar Rats Supplemented with Bee Pollen or Whey Protein. Foods 2022, 11, 1131. [Google Scholar] [CrossRef]
- Čeksterytė, V.; Treigytė, G.; Matuzevičius, D.; Jaškūnė, K.; Navakauskas, D.; Kurtinaitienė, B.; Navakauskienė, R. Comparative proteomic profile in bee- and manually-collected Taraxacum officinale pollen. J. Apic. Res. 2022, 61, 543–556. [Google Scholar] [CrossRef]
- Ares, A.M.; Martín, M.T.; Toribio, L.; Bernal, J. Determination of Free Amino Acids in Bee Pollen by Liquid Chromatography with Fluorescence Detection. Food Anal. Methods 2022, 15, 2172–2180. [Google Scholar] [CrossRef]
- Taha, E.-K.A.; Al-Kahtani, S.; Taha, R. Protein content and amino acids composition of bee-pollens from major floral sources in Al-Ahsa, eastern Saudi Arabia. Saudi J. Biol. Sci. 2019, 26, 232–237. [Google Scholar] [CrossRef]
- Jeannerod, L.; Carlier, A.; Schatz, B.; Daise, C.; Richel, A.; Agnan, Y.; Baude, M.; Jacquemart, A.-L. Some bee-pollinated plants provide nutritionally incomplete pollen amino acid resources to their pollinators. PLoS ONE 2022, 17, e0269992. [Google Scholar] [CrossRef] [PubMed]
- Sommano, S.R.; Bhat, F.M.; Wongkeaw, M.; Sriwichai, T.; Sunanta, P.; Chuttong, B.; Burgett, M. Amino Acid Profiling and Chemometric Relations of Black Dwarf Honey and Bee Pollen. Front. Nutr. 2020, 7, 558579. [Google Scholar] [CrossRef]
- Habryka, C.; Socha, R.; Juszczak, L. Effect of Bee Pollen Addition on the Polyphenol Content, Antioxidant Activity, and Quality Parameters of Honey. Antioxidants 2021, 10, 810. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarti, P.; Lucas, H.M.; Sagili, R.R. Evaluating Effects of a Critical Micronutrient (24-Methylenecholesterol) on Honey Bee Physiology. Ann. EÈntomol. Soc. Am. 2020, 113, 176–182. [Google Scholar] [CrossRef]
- Vaudo, A.D.; Tooker, J.F.; Patch, H.M.; Biddinger, D.J.; Coccia, M.; Crone, M.K.; Fiely, M.; Francis, J.S.; Hines, H.M.; Hodges, M.; et al. Pollen Protein: Lipid Macronutrient Ratios May Guide Broad Patterns of Bee Species Floral Preferences. Insects 2020, 11, 132. [Google Scholar] [CrossRef] [PubMed]
- Grund-Mueller, N.; Ruedenauer, F.A.; Spaethe, J.; Leonhardt, S.D. Adding Amino Acids to a Sucrose Diet Is Not Sufficient to Support Longevity of Adult Bumble Bees. Insects 2020, 11, 247. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Liu, J.; Gao, F.; Chen, M.; Jiang, Y.; Zhao, H.; Ma, W. Electrophysiological and Behavioral Responses of Apis mellifera and Bombusterrestris to Melon Flower Volatiles. Insects 2022, 13, 973. [Google Scholar] [CrossRef]
- Dimins, F.; Cinkmanis, I.; Radenkovs, V.; Augspole, I.; Valdovska, A. Analysis of 18 Free Amino Acids in Honeybee and Bumblebee Honey from Eastern and Northern Europe and Central Asia Using HPLC-ESI-TQ-MS/MS Approach Bypassing Derivatization Step. Foods 2022, 11, 2744. [Google Scholar] [CrossRef]
- Hao, K.; Xu, Q.; Huang, S. Pollen-feeding behavior of diverse insects on Geranium delavayi, a flower with large, accessible pollen grains. Am. J. Bot. 2023, 110, e16113. [Google Scholar] [CrossRef]
- Li, Q.; Liang, X.; Xue, X.; Wang, K.; Wu, L. Lipidomics Provides Novel Insights into Understanding the Bee Pollen Lipids Transepithelial Transport and Metabolism in Human Intestinal Cells. J. Agric. Food Chem. 2020, 68, 907–917. [Google Scholar] [CrossRef]
- Wang, J.; Chen, Y.; Zhao, L.; Zhang, Y.; Fang, X. Lipidomics reveals the molecular mechanisms underlying the changes in lipid profiles and lipid oxidation in rape bee pollen dried by different methods. Food Res. Int. 2022, 162, 112104. [Google Scholar] [CrossRef]
- Giampieri, F.; Quiles, J.L.; Cianciosi, D.; Forbes-Hernández, T.Y.; Orantes-Bermejo, F.J.; Alvarez-Suarez, J.M.; Battino, M. Bee Products: An Emblematic Example of Underutilized Sources of Bioactive Compounds. J. Agric. Food Chem. 2022, 70, 6833–6848. [Google Scholar] [CrossRef]
- Chakrabarti, P.; Lucas, H.M.; Sagili, R.R. Novel Insights into Dietary Phytosterol Utilization and Its Fate in Honey Bees (Apis mellifera L.). Molecules 2020, 25, 571. [Google Scholar] [CrossRef] [PubMed]
- Zu, P.; Koch, H.; Schwery, O.; Pironon, S.; Phillips, C.; Ondo, I.; Farrell, I.W.; Nes, W.D.; Moore, E.; Wright, G.A.; et al. Pollen sterols are associated with phylogeny and environment but not with pollinator guilds. New Phytol. 2021, 230, 1169–1184. [Google Scholar] [CrossRef] [PubMed]
- Nattagh-Eshtivani, E.; Barghchi, H.; Pahlavani, N.; Barati, M.; Amiri, Y.; Fadel, A.; Khosravi, M.; Talebi, S.; Arzhang, P.; Ziaei, R.; et al. Biological and pharmacological effects and nutritional impact of phytosterols: A comprehensive review. Phytother. Res. 2022, 36, 299–322. [Google Scholar] [CrossRef] [PubMed]
- Nainu, F.; Masyita, A.; Bahar, M.A.; Raihan, M.; Prova, S.R.; Mitra, S.; Bin Emran, T.; Simal-Gandara, J. Pharmaceutical Prospects of Bee Products: Special Focus on Anticancer, Antibacterial, Antiviral, and Antiparasitic Properties. Antibiotics 2021, 10, 822. [Google Scholar] [CrossRef] [PubMed]
- Alfawaz, H.A.; El-Ansary, A.; Al-Ayadhi, L.; Bhat, R.S.; Hassan, W.M. Protective Effects of Bee Pollen on Multiple Propionic Acid-Induced Biochemical Autistic Features in a Rat Model. Metabolites 2022, 12, 571. [Google Scholar] [CrossRef] [PubMed]
- Al-Kahtani, S.N.; Taha, E.-K.A.; Farag, S.A.; Taha, R.A.; Abdou, E.A.; Mahfouz, H.M. Harvest Season Significantly Influences the Fatty Acid Composition of Bee Pollen. Biology 2021, 10, 495. [Google Scholar] [CrossRef]
- Zhou, E.; Wang, Q.; Li, X.; Zhu, D.; Niu, Q.; Li, Q.; Wu, L. Effects of Bee Pollen Derived from Acer mono Maxim. or Phellodendron amurense Rupr. on the Lipid Composition of Royal Jelly Secreted by Honeybees. Foods 2023, 12, 625. [Google Scholar] [CrossRef]
- Bennett, M.M.; Welchert, A.C.; Carroll, M.; Shafir, S.; Smith, B.H.; Corby-Harris, V. Unbalanced fatty acid diets impair discrimination ability of honey bee workers to damaged and healthy brood odors. J. Exp. Biol. 2022, 225, jeb244103. [Google Scholar] [CrossRef]
- Barta, D.G.; Cornea-Cipcigan, M.; Margaoan, R.; Vodnar, D.C. Biotechnological Processes Simulating the Natural Fermentation Process of Bee Bread and Therapeutic Properties—An Overview. Front. Nutr. 2022, 9, 871896. [Google Scholar] [CrossRef]
- Zhang, H.; Zhu, X.; Huang, Q.; Zhang, L.; Liu, X.; Liu, R.; Lu, Q. Antioxidant and anti-inflammatory activities of rape bee pollen after fermentation and their correlation with chemical components by ultra-performance liquid chromatography-quadrupole time of flight mass spectrometry-based untargeted metabolomics. Food Chem. 2023, 409, 135342. [Google Scholar] [CrossRef]
- Miyata, R.; Hoshino, S.; Ahn, M.-R.; Kumazawa, S. Chemical Profiles of Korean Bee Pollens and Their Catechol-O-methyltransferase Inhibitory Activities. J. Agric. Food Chem. 2022, 70, 1174–1181. [Google Scholar] [CrossRef] [PubMed]
- Ares, A.M.; Martín, M.T.; Tapia, J.A.; González-Porto, A.V.; Higes, M.; Martín-Hernández, R.; Bernal, J. Differentiation of bee pollen samples according to the betaines and other quaternary ammonium related compounds content by using a canonical discriminant analysis. Food Res. Int. 2022, 160, 111698. [Google Scholar] [CrossRef] [PubMed]
- Arumugam, M.K.; Paal, M.C.; Donohue, T.M.; Ganesan, M.; Osna, N.A.; Kharbanda, K.K. Beneficial Effects of Betaine: A Comprehensive Review. Biology 2021, 10, 456. [Google Scholar] [CrossRef] [PubMed]
- Ilie, C.-I.; Oprea, E.; Geana, E.-I.; Spoiala, A.; Buleandra, M.; Pircalabioru, G.G.; Badea, I.A.; Ficai, D.; Andronescu, E.; Ficai, A.; et al. Bee Pollen Extracts: Chemical Composition, Antioxidant Properties, and Effect on the Growth of Selected Probiotic and Pathogenic Bacteria. Antioxidants 2022, 11, 959. [Google Scholar] [CrossRef]
- Pełka, K.; Otłowska, O.; Worobo, R.W.; Szweda, P. Bee Bread Exhibits Higher Antimicrobial Potential Compared to Bee Pollen. Antibiotics 2021, 10, 125. [Google Scholar] [CrossRef]
- Kieliszek, M.; Piwowarek, K.; Kot, A.M.; Błażejak, S.; Chlebowska-Śmigiel, A.; Wolska, I. Pollen and bee bread as new health-oriented products: A review. Trends Food Sci. Technol. 2018, 71, 170–180. [Google Scholar] [CrossRef]
- Zhang, H.; Lu, Q.; Liu, R. Widely targeted metabolomics analysis reveals the effect of fermentation on the chemical composition of bee pollen. Food Chem. 2021, 375, 131908. [Google Scholar] [CrossRef]
- Adaškevičiūtė, V.; Kaškonienė, V.; Barčauskaitė, K.; Kaškonas, P.; Maruška, A. The Impact of Fermentation on Bee Pollen Polyphenolic Compounds Composition. Antioxidants 2022, 11, 645. [Google Scholar] [CrossRef]
- Martinello, M.; Mutinelli, F. Antioxidant activity in bee products: A review. Antioxidants 2021, 10, 71. [Google Scholar] [CrossRef]
- Filannino, P.; Di Cagno, R.; Vincentini, O.; Pinto, D.; Polo, A.; Maialetti, F.; Porrelli, A.; Gobbetti, M. Nutrients Bioaccessibility and Anti-inflammatory Features of Fermented Bee Pollen: A Comprehensive Investigation. Front. Microbiol. 2021, 12, 622091. [Google Scholar] [CrossRef]
- Yan, S.; Wang, K.; Wang, X.; Ou, A.; Wang, F.; Wu, L.; Xue, X. Effect of fermented bee pollen on metabolic syndrome in high-fat diet-induced mice. Food Sci. Hum. Wellness 2021, 10, 345–355. [Google Scholar] [CrossRef]
- Kaškonienė, V.; Adaškevičiūtė, V.; Kaškonas, P.; Mickienė, R.; Maruška, A. Antimicrobial and antioxidant activities of natural and fermented bee pollen. Food Biosci. 2020, 34, 100532. [Google Scholar] [CrossRef]
- Yan, S.; Li, Q.; Xue, X.; Wang, K.; Zhao, L.; Wu, L. Analysis of improved nutritional composition of bee pollen (Brassica campestris L.) after different fermentation treatments. Int. J. Food Sci. Technol. 2019, 54, 2169–2181. [Google Scholar] [CrossRef]
- Yin, S.; Tao, Y.; Jiang, Y.; Meng, L.; Zhao, L.; Xue, X.; Li, Q.; Wu, L. A Combined Proteomic and Metabolomic Strategy for Allergens Characterization in Natural and Fermented Brassica napus Bee Pollen. Front. Nutr. 2022, 9, 822033. [Google Scholar] [CrossRef] [PubMed]
- Barkhidarian, B.; Roldos, L.; Iskandar, M.M.; Saedisomeolia, A.; Kubow, S. Probiotic Supplementation and Micronutrient Status in Healthy Subjects: A Systematic Review of Clinical Trials. Nutrients 2021, 13, 3001. [Google Scholar] [CrossRef]
- Kobylińska, M.; Antosik, K.; Decyk, A.; Kurowska, K. Malnutrition in Obesity: Is It Possible? Obes. Facts 2021, 15, 19–25. [Google Scholar] [CrossRef]
- Cashman, K.D. 100 Years of Vitamin D: Global differences in vitamin D status and dietary intake: A review of the data. Endocr. Connect. 2022, 11, e210282. [Google Scholar] [CrossRef]
- Mehl, S.; Sun, Q.; Görlich, C.L.; Hackler, J.; Kopp, J.F.; Renko, K.; Mittag, J.; Schwerdtle, T.; Schomburg, L. Cross-sectional analysis of trace element status in thyroid disease. J. Trace Elem. Med. Biol. 2020, 58, 126430. [Google Scholar] [CrossRef]
- Giacconi, R.; Chiodi, L.; Boccoli, G.; Costarelli, L.; Piacenza, F.; Provinciali, M.; Malavolta, M. Reduced levels of plasma selenium are associated with increased inflammation and cardiovascular disease in an Italian elderly population. Exp. Gerontol. 2021, 145, 111219. [Google Scholar] [CrossRef]
- Vural, Z.; Avery, A.; Kalogiros, D.I.; Coneyworth, L.J.; Welham, S.J.M. Trace Mineral Intake and Deficiencies in Older Adults Living in the Community and Institutions: A Systematic Review. Nutrients 2020, 12, 1072. [Google Scholar] [CrossRef]
- Richardson, D.P.; Lovegrove, J.A. Nutritional status of micronutrients as a possible and modifiable risk factor for COVID-19: A UK perspective. Br. J. Nutr. 2021, 125, 678–684. [Google Scholar] [CrossRef] [PubMed]
- Marley, A.; Smith, S.C.; Ahmed, R.; Nightingale, P.; Cooper, S.C. Vitamin A deficiency: Experience from a tertiary referral UK hospital; not just a low- and middle-income country issue. Public Health Nutr. 2021, 24, 6466–6471. [Google Scholar] [CrossRef] [PubMed]
- Forman, H.J.; Zhang, H. Targeting oxidative stress in disease: Promise and limitations of antioxidant therapy. Nat. Rev. Drug Discov. 2021, 20, 689–709. [Google Scholar] [CrossRef] [PubMed]
- Feelisch, M.; Cortese-Krott, M.M.; Santolini, J.; Wootton, S.A.; Jackson, A.A. Systems Redox Biology in Health and Disease. EXCLI J. 2022, 21, 623–646. [Google Scholar] [CrossRef] [PubMed]
- Manzoor, M.F.; Arif, Z.; Kabir, A.; Mehmood, I.; Munir, D.; Razzaq, A.; Ali, A.; Goksen, G.; Coşier, V.; Ahmad, N.; et al. Oxidative stress and metabolic diseases: Relevance and therapeutic strategies. Front. Nutr. 2022, 9, 994309. [Google Scholar] [CrossRef] [PubMed]
- Raut, S.K.; Khullar, M. Oxidative stress in metabolic diseases: Current scenario and therapeutic relevance. Mol. Cell. Biochem. 2023, 478, 185–196. [Google Scholar] [CrossRef]
- Panda, P.; Verma, H.K.; Lakkakula, S.; Merchant, N.; Kadir, F.; Rahman, S.; Jeffree, M.S.; Lakkakula, B.V.K.S.; Rao, P.V. Biomarkers of Oxidative Stress Tethered to Cardiovascular Diseases. Oxidative Med. Cell. Longev. 2022, 2022, 9154295. [Google Scholar] [CrossRef]
- Batty, M.; Bennett, M.R.; Yu, E. The Role of Oxidative Stress in Atherosclerosis. Cells 2022, 11, 3843. [Google Scholar] [CrossRef]
- Aylanc, V.; Tomás, A.; Russo-Almeida, P.; Falcão, S.I.; Vilas-Boas, M. Assessment of bioactive compounds under simulated gastrointestinal digestion of bee pollen and bee bread: Bioaccessibility and antioxidant activity. Antioxidants 2021, 10, 651. [Google Scholar] [CrossRef]
- El-Seedi, H.R.; Eid, N.; Abd El-Wahed, A.A.; Rateb, M.E.; Afifi, H.S.; Algethami, A.F.; Zhao, C.; Al Naggar, Y.; Alsharif, S.M.; Tahir, H.E. Honey Bee Products: Preclinical and Clinical Studies of Their Anti-inflammatory and Immunomodulatory Properties. Front. Nutr. 2022, 8, 1109. [Google Scholar] [CrossRef]
- Lawag, I.L.; Yoo, O.; Lim, L.Y.; Hammer, K.; Locher, C. Optimisation of Bee Pollen Extraction to Maximise Extractable Antioxidant Constituents. Antioxidants 2021, 10, 1113. [Google Scholar] [CrossRef] [PubMed]
- Thakur, M.; Nanda, V. Screening of Indian bee pollen based on antioxidant properties and polyphenolic composition using UHPLC-DAD-MS/MS: A multivariate analysis and ANN based approach. Food Res. Int. 2021, 140, 110041. [Google Scholar] [CrossRef] [PubMed]
- Barbieri, D.; Gabriele, M.; Summa, M.; Colosimo, R.; Leonardi, D.; Domenici, V.; Pucci, L. Antioxidant, Nutraceutical Properties, and Fluorescence Spectral Profiles of Bee Pollen Samples from Different Botanical Origins. Antioxidants 2020, 9, 1001. [Google Scholar] [CrossRef] [PubMed]
- Mazurek, S.; Szostak, R.; Kondratowicz, M.; Węglińska, M.; Kita, A.; Nemś, A. Modeling of Antioxidant Activity, Polyphenols and Macronutrients Content of Bee Pollen Applying Solid-State 13C NMR Spectra. Antioxidants 2021, 10, 1123. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Hu, J.; Huang, W.; Zhu, L.; Shao, M.; Dordoe, C.; Ahn, Y.-J.; Wang, D.; Zhao, Y.; Xiong, Y.; et al. The botanical origin and antioxidant, anti-BACE1 and antiproliferative properties of bee pollen from different regions of South Korea. BMC Complement. Med. Ther. 2020, 20, 236. [Google Scholar] [CrossRef]
- Węglińska, M.; Szostak, R.; Kita, A.; Nemś, A.; Mazurek, S. Determination of nutritional parameters of bee pollen by Raman and infrared spectroscopy. Talanta 2020, 212, 120790. [Google Scholar] [CrossRef]
- Castiglioni, S.; Stefano, M.; Astolfi, P.; Pisani, M.; Carloni, P. Characterisation of Bee Pollen from the Marche Region (Italy) According to the Botanical and Geographical Origin with Analysis of Antioxidant Activity and Colour, Using a Chemometric Approach. Molecules 2022, 27, 7996. [Google Scholar] [CrossRef]
- Alimoglu, G.; Guzelmeric, E.; Yuksel, P.I.; Celik, C.; Deniz, I.; Yesilada, E. Monofloral and polyfloral bee pollens: Comparative evaluation of their phenolics and bioactivity profiles. LWT 2021, 142, 110973. [Google Scholar] [CrossRef]
- Sawicki, T.; Starowicz, M.; Kłębukowska, L.; Hanus, P. The Profile of Polyphenolic Compounds, Contents of Total Phenolics and Flavonoids, and Antioxidant and Antimicrobial Properties of Bee Products. Molecules 2022, 27, 1301. [Google Scholar] [CrossRef]
- Rana, A.; Samtiya, M.; Dhewa, T.; Mishra, V.; Aluko, R.E. Health benefits of polyphenols: A concise review. J. Food Biochem. 2022, 46, e14264. [Google Scholar] [CrossRef]
- Wang, S.; Du, Q.; Meng, X.; Zhang, Y. Natural polyphenols: A potential prevention and treatment strategy for metabolic syndrome. Food Funct. 2022, 13, 9734–9753. [Google Scholar] [CrossRef] [PubMed]
- Gasmi, A.; Mujawdiya, P.K.; Noor, S.; Lysiuk, R.; Darmohray, R.; Piscopo, S.; Lenchyk, L.; Antonyak, H.; Dehtiarova, K.; Shanaida, M.; et al. Polyphenols in Metabolic Diseases. Molecules 2022, 27, 6280. [Google Scholar] [CrossRef] [PubMed]
- Aravind, S.M.; Wichienchot, S.; Tsao, R.; Ramakrishnan, S.; Chakkaravarthi, S. Role of dietary polyphenols on gut microbiota, their metabolites and health benefits. Food Res. Int. 2021, 142, 110189. [Google Scholar] [CrossRef]
- Zhang, Y.; Yu, W.; Zhang, L.; Wang, M.; Chang, W. The Interaction of Polyphenols and the Gut Microbiota in Neurodegenerative Diseases. Nutrients 2022, 14, 5373. [Google Scholar] [CrossRef]
- Cherubim, D.J.D.L.; Martins, C.V.B.; Fariña, L.O.; Lucca, R.A.D.S.D. Polyphenols as natural antioxidants in cosmetics applications. J. Cosmet. Dermatol. 2020, 19, 33–37. [Google Scholar] [CrossRef]
- de Araújo, F.F.; de Paulo Farias, D.; Neri-Numa, I.A.; Pastore, G.M. Polyphenols and their applications: An approach in food chemistry and innovation potential. Food Chem. 2021, 338, 127535. [Google Scholar] [CrossRef]
- Winiarska-Mieczan, A.; Kwiecień, M.; Jachimowicz-Rogowska, K.; Donaldson, J.; Tomaszewska, E.; Baranowska-Wójcik, E. Anti-Inflammatory, Antioxidant, and Neuroprotective Effects of Polyphenols—Polyphenols as an Element of Diet Therapy in Depressive Disorders. Int. J. Mol. Sci. 2023, 24, 2258. [Google Scholar] [CrossRef]
- Morén, C.; Desouza, R.M.; Giraldo, D.M.; Uff, C. Antioxidant Therapeutic Strategies in Neurodegenerative Diseases. Int. J. Mol. Sci. 2022, 23, 9328. [Google Scholar] [CrossRef]
- Bjørklund, G.; Shanaida, M.; Lysiuk, R.; Butnariu, M.; Peana, M.; Sarac, I.; Strus, O.; Smetanina, K.; Chirumbolo, S. Natural Compounds and Products from an Anti-Aging Perspective. Molecules 2022, 27, 7084. [Google Scholar] [CrossRef]
- Maiuolo, J.; Gliozzi, M.; Carresi, C.; Musolino, V.; Oppedisano, F.; Scarano, F.; Nucera, S.; Scicchitano, M.; Bosco, F.; Macri, R.; et al. Nutraceuticals and cancer: Potential for natural polyphenols. Nutrients 2021, 13, 3834. [Google Scholar] [CrossRef]
- Di Lorenzo, C.; Colombo, F.; Biella, S.; Stockley, C.; Restani, P. Polyphenols and human health: The role of bioavailability. Nutrients 2021, 13, 273. [Google Scholar] [CrossRef]
- Shen, N.; Wang, T.; Gan, Q.; Liu, S.; Wang, L.; Jin, B. Plant flavonoids: Classification, distribution, biosynthesis, and antioxidant activity. Food Chem. 2022, 383, 132531. [Google Scholar] [CrossRef]
- Rajendran, P.; Abdelsalam, S.A.; Renu, K.; Veeraraghavan, V.; Ben Ammar, R.; Ahmed, E.A. Polyphenols as Potent Epigenetics Agents for Cancer. Int. J. Mol. Sci. 2022, 23, 11712. [Google Scholar] [CrossRef]
- Doseděl, M.; Jirkovský, E.; Macáková, K.; Krčmová, L.K.; Javorská, L.; Pourová, J.; Mercolini, L.; Remião, F.; Nováková, L.; Mladěnka, P.; et al. Vitamin C-sources, physiological role, kinetics, deficiency, use, toxicity, and determination. Nutrients 2021, 13, 615. [Google Scholar] [CrossRef]
- Caritá, A.C.; Fonseca-Santos, B.; Shultz, J.D.; Michniak-Kohn, B.; Chorilli, M.; Leonardi, G.R. Vitamin C: One compound, several uses. Advances for delivery, efficiency and stability. Nanomed. Nanotechnol. Biol. Med. 2020, 24, 102117. [Google Scholar] [CrossRef]
- Moslemi, E.; Musazadeh, V.; Kavyani, Z.; Naghsh, N.; Shoura, S.M.S.; Dehghan, P. Efficacy of vitamin D supplementation as an adjunct therapy for improving inflammatory and oxidative stress biomarkers: An umbrella meta-analysis. Pharmacol. Res. 2022, 186, 106484. [Google Scholar] [CrossRef]
- Albergamo, A.; Apprato, G.; Silvagno, F. The Role of Vitamin D in Supporting Health in the COVID-19 Era. Int. J. Mol. Sci. 2022, 23, 3621. [Google Scholar] [CrossRef]
- Ciarcià, G.; Bianchi, S.; Tomasello, B.; Acquaviva, R.; Malfa, G.A.; Naletova, I.; La Mantia, A.; Di Giacomo, C. Vitamin E and Non-Communicable Diseases: A Review. Biomedicines 2022, 10, 2473. [Google Scholar] [CrossRef]
- Liao, S.; Omage, S.O.; Börmel, L.; Kluge, S.; Schubert, M.; Wallert, M.; Lorkowski, S. Vitamin E and Metabolic Health: Relevance of Interactions with Other Micronutrients. Antioxidants 2022, 11, 1785. [Google Scholar] [CrossRef]
- Wu, X.; Cai, X.; Ai, J.; Zhang, C.; Liu, N.; Gao, W. Extraction, Structures, Bioactivities and Structure-Function Analysis of the Polysaccharides From Safflower (Carthamus tinctorius L.). Front. Pharmacol. 2021, 12, 767947. [Google Scholar] [CrossRef]
- Adaškevičiūtė, V.; Kaškonienė, V.; Kaškonas, P.; Barčauskaitė, K.; Maruška, A. Comparison of Physicochemical Properties of Bee Pollen with Other Bee Products. Biomolecules 2019, 9, 819. [Google Scholar] [CrossRef] [PubMed]
- Naseri, L.; Khazaei, M.R.; Khazaei, M. Potential Therapeutic Effect of Bee Pollen and Metformin Combination on Testosterone and Estradiol Levels, Apoptotic Markers and Total Antioxidant Capacity in A Rat Model of Polycystic Ovary Syndrome. Int. J. Fertil. Steril. 2021, 15, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Saisavoey, T.; Sangtanoo, P.; Srimongkol, P.; Reamtong, O.; Karnchanatat, A. Hydrolysates from bee pollen could induced apoptosis in human bronchogenic carcinoma cells (ChaGo-K-1). J. Food Sci. Technol. 2021, 58, 752–763. [Google Scholar] [CrossRef] [PubMed]
- Bakour, M.; Laaroussi, H.; Ferreira-Santos, P.; Genisheva, Z.; Ousaaid, D.; Teixeira, J.A.; Lyoussi, B. Exploring the Palynological, Chemical, and Bioactive Properties of Non-Studied Bee Pollen and Honey from Morocco. Molecules 2022, 27, 5777. [Google Scholar] [CrossRef] [PubMed]
- Bridi, R.; Atala, E.; Pizarro, P.N.; Montenegro, G. Honeybee Pollen Load: Phenolic Composition and Antimicrobial Activity and Antioxidant Capacity. J. Nat. Prod. 2019, 82, 559–565. [Google Scholar] [CrossRef]
- Aylanc, V.; Larbi, S.; Calhelha, R.; Barros, L.; Rezouga, F.; Rodríguez-Flores, M.S.; Seijo, M.C.; El Ghouizi, A.; Lyoussi, B.; Falcão, S.I.; et al. Evaluation of Antioxidant and Anticancer Activity of Mono- and Polyfloral Moroccan Bee Pollen by Characterizing Phenolic and Volatile Compounds. Molecules 2023, 28, 835. [Google Scholar] [CrossRef]
- Kędzierski, W.; Janczarek, I.; Kowalik, S.; Jamioł, M.; Wawak, T.; Borsuk, G.; Przetacznik, M. Bee Pollen Supplementation to Aged Horses Influences Several Blood Parameters. J. Equine Vet. Sci. 2020, 90, 103024. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Zhao, Y.; Yan, Y.; Mi, J.; Lu, L.; Luo, Q.; Li, X.; Zeng, X.; Cao, Y. Antioxidant and immunomodulatory activities in vitro of polysaccharides from bee collected pollen of Chinese wolfberry. Int. J. Biol. Macromol. 2020, 163, 190–199. [Google Scholar] [CrossRef] [PubMed]
- Saral, Ö.; Şahin, H.; Saral, S.; Alkanat, M.; Akyıldız, K.; Topçu, A.; Yılmaz, A. Bee pollen increases hippocampal brain-derived neurotrophic factor and suppresses neuroinflammation in adult rats with chronic immobilization stress. Neurosci. Lett. 2021, 766, 136342. [Google Scholar] [CrossRef]
- Novaković, S.; Djekic, I.; Pešić, M.; Kostić, A.; Milinčić, D.; Stanisavljević, N.; Radojević, A.; Tomasevic, I. Bee pollen powder as a functional ingredient in frankfurters. Meat Sci. 2021, 182, 108621. [Google Scholar] [CrossRef]
- Di Chiacchio, I.M.; Gómez-Abenza, E.; Paiva, I.M.; de Abreu, D.J.M.; Rodríguez-Vidal, J.F.; Carvalho, E.E.N.; Carvalho, S.M.; Solis-Murgas, L.D.; Mulero, V. Bee pollen in zebrafish diet affects intestinal microbiota composition and skin cutaneous melanoma development. Sci. Rep. 2022, 12, 9998. [Google Scholar] [CrossRef]
- Al-Kahtani, S.N.; Alaqil, A.A.; Abbas, A.O. Modulation of Antioxidant Defense, Immune Response, and Growth Performance by Inclusion of Propolis and Bee Pollen into Broiler Diets. Animals 2022, 12, 1658. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, J.; Zhou, Q.; Wang, L.; Huang, W.; Wang, R. Effect of ultrasonic and ball-milling treatment on cell wall, nutrients, and antioxidant capacity of rose (Rosa rugosa) bee pollen, and identification of bioactive components. J. Sci. Food Agric. 2019, 99, 5350–5357. [Google Scholar] [CrossRef]
- Al-Salem, H.S.; Al-Yousef, H.M.; Ashour, A.E.; Ahmed, A.F.; Amina, M.; Issa, I.S.; Bhat, R.S. Antioxidant and hepatorenal protective effects of bee pollen fractions against propionic acid-induced autistic feature in rats. Food Sci. Nutr. 2020, 8, 5114–5127. [Google Scholar] [CrossRef]
- El Ghouizi, A.; El Menyiy, N.; Falcão, S.I.; Vilas-Boas, M.; Lyoussi, B. Chemical composition, antioxidant activity, and diuretic effect of Moroccan fresh bee pollen in rats. Vet. World 2020, 13, 1251–1261. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, R.; Lu, Q. Separation and Characterization of Phenolamines and Flavonoids from Rape Bee Pollen, and Comparison of Their Antioxidant Activities and Protective Effects Against Oxidative Stress. Molecules 2020, 25, 1264. [Google Scholar] [CrossRef]
- Gercek, Y.C.; Celik, S.; Bayram, S. Screening of Plant Pollen Sources, Polyphenolic Compounds, Fatty Acids and Antioxidant/Antimicrobial Activity from Bee Pollen. Molecules 2021, 27, 117. [Google Scholar] [CrossRef]
- Şahin, S.; Karkar, B. The antioxidant properties of the chestnut bee pollen extract and its preventive action against oxidatively induced damage in DNA bases. J. Food Biochem. 2019, 43, e12888. [Google Scholar] [CrossRef]
- Laaroussi, H.; Bakour, M.; Ousaaid, D.; Aboulghazi, A.; Ferreira-Santos, P.; Genisheva, Z.; Teixeira, J.A.; Lyoussi, B. Effect of antioxidant-rich propolis and bee pollen extracts against D-glucose induced type 2 diabetes in rats. Food Res. Int. 2020, 138, 109802. [Google Scholar] [CrossRef]
- Kostić, A.; Milinčić, D.D.; Nedić, N.; Gašić, U.M.; Trifunović, B.; Vojt, D.; Tešić, L.; Pešić, M.B. Phytochemical Profile and Antioxidant Properties of Bee-Collected Artichoke (Cynara scolymus) Pollen. Antioxidants 2021, 10, 1091. [Google Scholar] [CrossRef]
- Xu, Y.; Cao, X.; Zhao, H.; Yang, E.; Wang, Y.; Cheng, N.; Cao, W. Impact of Camellia japonica Bee Pollen Polyphenols on Hyperuricemia and Gut Microbiota in Potassium Oxonate-Induced Mice. Nutrients 2021, 13, 2665. [Google Scholar] [CrossRef]
- Gulhan, M.F. Therapeutic potentials of propolis and pollen on biochemical changes in reproductive function of L-NAME induced hypertensive male rats. Clin. Exp. Hypertens. 2019, 41, 292–298. [Google Scholar] [CrossRef]
- Belina-Aldemita, M.D.; Schreiner, M.; D’Amico, S. Characterization of phenolic compounds and antioxidative potential of pot-pollen produced by stingless bees (Tetragonula biroi Friese) from the Philippines. J. Food Biochem. 2020, 44, e13102. [Google Scholar] [CrossRef]
- Gonçalves, A.C.; Lahlou, R.A.; Alves, G.; Garcia-Viguera, C.; Moreno, D.A.; Silva, L.R. Potential activity of abrantes pollen extract: Biochemical and cellular model studies. Foods 2021, 10, 2804. [Google Scholar] [CrossRef]
- Bujang, J.S.; Zakaria, M.H.; Ramaiya, S.D. Chemical constituents and phytochemical properties of floral maize pollen. PLoS ONE 2021, 16, e0247327. [Google Scholar] [CrossRef]
- Gabriele, M.; Frassinetti, S.; Pucci, L. Antimicrobial activity and protective effect of Tuscan bee pollens on oxidative and endoplasmic reticulum stress in different cell-based models. Foods 2021, 10, 1422. [Google Scholar] [CrossRef]
- Shen, Z.; Geng, Q.; Huang, H.; Yao, H.; Du, T.; Chen, L.; Wu, Z.; Miao, X.; Shi, P. Antioxidative and cardioprotective effects of Schisandra chinensis bee pollen extract on isoprenaline-induced myocardial infarction in rats. Molecules 2019, 24, 1090. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, A.E.; El-Magd, M.A.; El-Said, K.S.; El-Sharnouby, M.; Tousson, E.M.; Salama, A.F. Potential therapeutic effect of thymoquinone and/or bee pollen on fluvastatin-induced hepatitis in rats. Sci. Rep. 2021, 11, 15688. [Google Scholar] [CrossRef]
- Oyarzún, J.E.; Andia, M.E.; Uribe, S.; Núñez Pizarro, P.; Núñez, G.; Montenegro, G.; Bridi, R. Honeybee Pollen Extracts Reduce Oxidative Stress and Steatosis in Hepatic Cells. Molecules 2021, 26, 6. [Google Scholar] [CrossRef]
- Prado, A.; Brunet, J.-L.; Peruzzi, M.; Bonnet, M.; Bordier, C.; Crauser, D.; Le Conte, Y.; Alaux, C. Warmer winters are associated with lower levels of the cryoprotectant glycerol, a slower decrease in vitellogenin expression and reduced virus infections in winter honeybees. J. Insect Physiol. 2022, 136, 104348. [Google Scholar] [CrossRef]
- Ricigliano, V.A.; Cank, K.B.; Todd, D.A.; Knowles, S.L.; Oberlies, N.H. Metabolomics-Guided Comparison of Pollen and Microalgae-Based Artificial Diets in Honey Bees. J. Agric. Food Chem. 2022, 70, 9790–9801. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Liang, X.; Guo, N.; Hu, L.; Maruthi, P.E.; Wu, Y.; Xue, X.; Wu, L.; Wang, K. Protective effects of Bee pollen extract on the Caco-2 intestinal barrier dysfunctions induced by dextran sulfate sodium. Biomed. Pharmacother. 2019, 117, 109200. [Google Scholar] [CrossRef] [PubMed]
- Laaroussi, H.; Ferreira-Santos, P.; Genisheva, Z.; Bakour, M.; Ousaaid, D.; El Ghouizi, A.; Teixeira, J.A.; Lyoussi, B. Unveiling the techno-functional and bioactive properties of bee pollen as an added-value food ingredient. Food Chem. 2023, 405, 134958. [Google Scholar] [CrossRef] [PubMed]
- Khongkarat, P.; Ramadhan, R.; Phuwapraisirisan, P.; Chanchao, C. Safflospermidines from the bee pollen of Helianthus annuus L. exhibit a higher in vitro antityrosinase activity than kojic acid. Heliyon 2020, 6, e03638. [Google Scholar] [CrossRef]
- Lopes, A.J.O.; Vasconcelos, C.C.; Pereira, F.A.N.; Silva, R.H.M.; Queiroz, P.F.d.S.; Fernandes, C.V.; Garcia, J.B.S.; Ramos, R.M.; da Rocha, C.Q.; Lima, S.T.d.J.R.M.; et al. Anti-Inflammatory and Antinociceptive Activity of Pollen Extract Collected by Stingless Bee Melipona fasciculata. Int. J. Mol. Sci. 2019, 20, 4512. [Google Scholar] [CrossRef]
- Chen, S.; Zhao, H.; Cheng, N.; Cao, W. Rape bee pollen alleviates dextran sulfate sodium (DSS)-induced colitis by neutralizing IL-1β and regulating the gut microbiota in mice. Food Res. Int. 2019, 122, 241–251. [Google Scholar] [CrossRef]
- Chen, S.; Wang, X.; Cheng, N. Ultrasound-assisted ethanol extraction of Actinidia arguta pollen possesses antioxidant activity and protects DNA from oxidative damage. J. Food Biochem. 2021, 45, e13603. [Google Scholar] [CrossRef]
- Shi, P.; Geng, Q.; Chen, L.; Du, T.; Lin, Y.; Lai, R.; Meng, F.; Wu, Z.; Miao, X.; Yao, H. Schisandra chinensis bee pollen’s chemical profiles and protective effect against H2O2-induced apoptosis in H9c2 cardiomyocytes. BMC Complement. Med. Ther. 2020, 20, 274. [Google Scholar] [CrossRef]
- Messina, C.M.; Panettieri, V.; Arena, R.; Renda, G.; Ruiz, C.E.; Morghese, M.; Piccolo, G.; Santulli, A.; Bovera, F. The Inclusion of a Supercritical Fluid Extract, Obtained From Honey Bee Pollen, in the Diet of Gilthead Sea Bream (Sparus aurata), Improves Fish Immune Response by Enhancing Anti-oxidant, and Anti-bacterial Activities. Front. Vet. Sci. 2020, 7, 95. [Google Scholar] [CrossRef]
- Kostić, A.; Milinčić, D.D.; Stanisavljević, N.S.; Gašić, U.M.; Lević, S.; Kojić, M.O.; Tešić, L.; Nedović, V.; Barać, M.B.; Pešić, M.B. Polyphenol bioaccessibility and antioxidant properties of in vitro digested spray-dried thermally-treated skimmed goat milk enriched with pollen. Food Chem. 2021, 351, 129310. [Google Scholar] [CrossRef]
- Lopes, A.J.O.; Vasconcelos, C.C.; Garcia, J.B.S.; Pinheiro, M.S.D.; Pereira, F.A.N.; Camelo, D.d.S.; de Morais, S.V.; Freitas, J.R.B.; da Rocha, C.Q.; Ribeiro, M.N.d.S.; et al. Anti-Inflammatory and Antioxidant Activity of Pollen Extract Collected by Scaptotrigona affinis postica: In silico, in vitro, and in vivo Studies. Antioxidants 2020, 9, 103. [Google Scholar] [CrossRef]
- Saral, O.; Dokumacioglu, E.; Saral, S.; Sumer, A.; Bulmus, O.; Kaya, S.O.; Canpolat, S. The effect of bee pollen on reproductive and biochemical parameters in methotrexate-induced testicular damage in adult rats. J. Basic Clin. Physiol. Pharmacol. 2020, 32, 1001–1006. [Google Scholar] [CrossRef] [PubMed]
- Amalia, E.; Diantini, A.; Subarnas, A. Water-soluble propolis and bee pollen of Trigona spp. from South Sulawesi Indonesia induce apoptosis in the human breast cancer MCF-7 cell line. Oncol. Lett. 2020, 20, 274. [Google Scholar] [CrossRef]
- Elghouizi, A.; Al-Waili, N.; Elmenyiy, N.; Elfetri, S.; Aboulghazi, A.; Al-Waili, A.; Lyoussi, B. Protective effect of bee pollen in acute kidney injury, proteinuria, and crystalluria induced by ethylene glycol ingestion in rats. Sci. Rep. 2022, 12, 8351. [Google Scholar] [CrossRef] [PubMed]
- Khongkarat, P.; Phuwapraisirisan, P.; Chanchao, C. Phytochemical content, especially spermidine derivatives, presenting antioxidant and antilipoxygenase activities in Thai bee pollens. PeerJ 2022, 10, e13506. [Google Scholar] [CrossRef] [PubMed]
- Gulcin, İ. Antioxidants and antioxidant methods: An updated overview. Arch. Toxicol. 2020, 94, 651–715. [Google Scholar] [CrossRef]
- Ivanova, A.; Gerasimova, E.; Gazizullina, E. Study of Antioxidant Properties of Agents from the Perspective of Their Action Mechanisms. Molecules 2020, 25, 4251. [Google Scholar] [CrossRef]
- Munteanu, I.G.; Apetrei, C. Analytical Methods Used in Determining Antioxidant Activity: A Review. Int. J. Mol. Sci. 2021, 22, 3380. [Google Scholar] [CrossRef]
- Acquaviva, A.; Di Simone, S.C.; Canini, A.; Braglia, R.; Di Marco, G.; Campana, C.; Angelini, P.; Flores, G.A.; Venanzoni, R.; Libero, M.L.; et al. Phytochemical and biological investigations on the pollen from industrial hemp male inflorescences. Food Res. Int. 2022, 161, 111883. [Google Scholar] [CrossRef]
- Altiner, D.D.; Altunatmaz, S.S.; Sabuncu, M.; Aksu, F.; Sahan, Y. In-vitro bioaccessibility of antioxidant properties of bee pollen in Turkey. Food Sci. Technol. 2021, 41 (Suppl. S1), 133–141. [Google Scholar] [CrossRef]
- Saisavoey, T.; Sangtanoo, P.; Chanchao, C.; Reamtong, O.; Karnchanatat, A. Identification of novel anti-inflammatory peptides from bee pollen (Apis mellifera) hydrolysate in lipopolysaccharide-stimulated RAW264.7 macrophages. J. Apic. Res. 2021, 60, 280–289. [Google Scholar] [CrossRef]
- Nwachukwu, I.D.; Sarteshnizi, R.A.; Udenigwe, C.C.; Aluko, R.E. A Concise Review of Current In Vitro Chemical and Cell-Based Antioxidant Assay Methods. Molecules 2021, 26, 4865. [Google Scholar] [CrossRef] [PubMed]
- Gong, E.S.; Liu, C.; Li, B.; Zhou, W.; Chen, H.; Li, T.; Wu, J.; Zeng, Z.; Wang, Y.; Si, X.; et al. Phytochemical profiles of rice and their cellular antioxidant activity against ABAP induced oxidative stress in human hepatocellular carcinoma HepG2 cells. Food Chem. 2020, 318, 126484. [Google Scholar] [CrossRef] [PubMed]
- Nassar, A.M.K.; Salim, Y.M.; Eid, K.S.; Shaheen, H.M.; Saati, A.A.; Hetta, H.F.; Elmistekawy, A.; Batiha, G.E.-S. Ameliorative Effects of Honey, Propolis, Pollen, and Royal Jelly Mixture against Chronic Toxicity of Sumithion Insecticide in White Albino Rats. Molecules 2020, 25, 2633. [Google Scholar] [CrossRef] [PubMed]
- Suleiman, J.B.; Abu Bakar, A.B.; Mohamed, M. Review on Bee Products as Potential Protective and Therapeutic Agents in Male Reproductive Impairment. Molecules 2021, 26, 3421. [Google Scholar] [CrossRef]
- Khanna, D.; Khanna, S.; Khanna, P.; Kahar, P.; Patel, B.M. Obesity: A Chronic Low-Grade Inflammation and Its Markers. Cureus 2022, 14, e22711. [Google Scholar] [CrossRef]
- Fabozzi, G.; Verdone, G.; Allori, M.; Cimadomo, D.; Tatone, C.; Stuppia, L.; Franzago, M.; Ubaldi, N.; Vaiarelli, A.; Ubaldi, F.M.; et al. Personalized Nutrition in the Management of Female Infertility: New Insights on Chronic Low-Grade Inflammation. Nutrients 2022, 14, 1918. [Google Scholar] [CrossRef]
- Herrero-Cervera, A.; Soehnlein, O.; Kenne, E. Neutrophils in chronic inflammatory diseases. Cell. Mol. Immunol. 2022, 19, 177–191. [Google Scholar] [CrossRef]
- Livshits, G.; Kalinkovich, A. A cross-talk between sestrins, chronic inflammation and cellular senescence governs the development of age-associated sarcopenia and obesity. Ageing Res. Rev. 2023, 86, 101852. [Google Scholar] [CrossRef]
- Wang, X.-L.; Feng, S.-T.; Wang, Y.-T.; Yuan, Y.-H.; Li, Z.-P.; Chen, N.-H.; Wang, Z.-Z.; Zhang, Y. Mitophagy, a Form of Selective Autophagy, Plays an Essential Role in Mitochondrial Dynamics of Parkinson’s Disease. Cell. Mol. Neurobiol. 2022, 42, 1321–1339. [Google Scholar] [CrossRef]
- Teissier, T.; Boulanger, E.; Cox, L.S. Interconnections between Inflammageing and Immunosenescence during Ageing. Cells 2022, 11, 359. [Google Scholar] [CrossRef] [PubMed]
- Di Giosia, P.; Stamerra, C.A.; Giorgini, P.; Jamialahamdi, T.; Butler, A.E.; Sahebkar, A. The role of nutrition in inflammaging. Ageing Res. Rev. 2022, 77, 101596. [Google Scholar] [CrossRef] [PubMed]
- Henein, M.Y.; Vancheri, S.; Longo, G.; Vancheri, F. The Role of Inflammation in Cardiovascular Disease. Int. J. Mol. Sci. 2022, 23, 12906. [Google Scholar] [CrossRef] [PubMed]
- Walker, K.A.; Basisty, N.; Wilson, D.M.; Ferrucci, L. Connecting aging biology and inflammation in the omics era. J. Clin. Investig. 2022, 132, e158448. [Google Scholar] [CrossRef]
- Kosyreva, A.M.; Sentyabreva, A.V.; Tsvetkov, I.S.; Makarova, O.V. Alzheimer’s Disease and Inflammaging. Brain Sci. 2022, 12, 1237. [Google Scholar] [CrossRef]
- Zhu, L.; Li, J.; Wei, C.; Luo, T.; Deng, Z.; Fan, Y.; Zheng, L. A polysaccharide from Fagopyrum esculentum Moench bee pollen alleviates microbiota dysbiosis to improve intestinal barrier function in antibiotic-treated mice. Food Funct. 2020, 11, 10519–10533. [Google Scholar] [CrossRef]
- Kotipalli, R.S.S.; Tirunavalli, S.K.; Pote, A.B.; Sahu, B.D.; Kuncha, M.; Jerald, M.K.; Sistla, R.; Andugulapati, S.B. Sinigrin Attenuates the Dextran Sulfate Sodium-induced Colitis in Mice by Modulating the MAPK Pathway. Inflammation 2023, 46, 787–807. [Google Scholar] [CrossRef]
- Khongkarat, P.; Traiyasut, P.; Phuwapraisirisan, P.; Chanchao, C. First report of fatty acids in Mimosadiplotricha bee pollen with in vitro lipase inhibitory activity. PeerJ 2022, 10, e12722. [Google Scholar] [CrossRef]
- Kosedag, M.; Gulaboglu, M. Pollen and bee bread expressed highest anti-inflammatory activities among bee products in chronic inflammation: An experimental study with cotton pellet granuloma in rats. Inflammopharmacology 2023, 1–9. [Google Scholar] [CrossRef]
- Chen, S.; Xu, Y.; Cheng, N.; Li, F.; Zhao, H.; Bai, N.; El-Seedi, H.R.; Cao, W. Mitigation of DSS-Induced Colitis Potentially via Th1/Th2 Cytokine and Immunological Function Balance Induced by Phenolic-Enriched Buckwheat (Fagopyrum esculentum Moench) Bee Pollen Extract. Foods 2022, 11, 1293. [Google Scholar] [CrossRef]
- Zhang, J.; Cao, W.; Zhao, H.; Guo, S.; Wang, Q.; Cheng, N.; Bai, N. Protective Mechanism of Fagopyrum esculentum Moench. Bee Pollen EtOH Extract Against Type II Diabetes in a High-Fat Diet/Streptozocin-Induced C57BL/6J Mice. Front. Nutr. 2022, 9, 925351. [Google Scholar] [CrossRef] [PubMed]
- Shaldoum, F.; El-Kott, A.F.; Ouda, M.M.A.; Abd-Ella, E.M. Immunomodulatory effects of bee pollen on doxorubicin-induced bone marrow/spleen immunosuppression in rat. J. Food Biochem. 2021, 45, e13747. [Google Scholar] [CrossRef] [PubMed]
- Eteraf-Oskouei, T.; Shafiee-Khamneh, A.; Heshmati-Afshar, F.; Delazar, A. Anti-inflammatory and anti-angiogenesis effect of bee pollen methanolic extract using air pouch model of inflammation. Res. Pharm. Sci. 2020, 15, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Sun, M.; Wan, Z.; Liang, J.; Betti, M.; Hrynets, Y.; Xue, X.; Wu, L.; Wang, K. Bee Pollen Extracts Modulate Serum Metabolism in Lipopolysaccharide-Induced Acute Lung Injury Mice with Anti-Inflammatory Effects. J. Agric. Food Chem. 2019, 67, 7855–7868. [Google Scholar] [CrossRef] [PubMed]
- Cheng, N.; Chen, S.; Liu, X.; Zhao, H.; Cao, W. Impact of SchisandraChinensis Bee Pollen on Nonalcoholic Fatty Liver Disease and Gut Microbiota in HighFat Diet Induced Obese Mice. Nutrients 2019, 11, 346. [Google Scholar] [CrossRef]
- Aabed, K.; Bhat, R.S.; Al-Dbass, A.; Moubayed, N.; Algahtani, N.; Merghani, N.M.; Alanazi, A.; Zayed, N.; El-Ansary, A. Bee pollen and propolis improve neuroinflammation and dysbiosis induced by propionic acid, a short chain fatty acid in a rodent model of autism. Lipids Health Dis. 2019, 18, 200. [Google Scholar] [CrossRef]
- Rivera-Jiménez, J.; Berraquero-García, C.; Gálvez, A.R.P.; García-Moreno, P.J.; Espejo-Carpio, F.J.; Guadix, A.; Guadix, E.M. Peptides and protein hydrolysates exhibiting anti-inflammatory activity: Sources, structural features and modulation mechanisms. Food Funct. 2022, 13, 12510–12540. [Google Scholar] [CrossRef]
- Mansinhbhai, C.H.; Sakure, A.; Maurya, R.; Bishnoi, M.; Kondepudi, K.K.; Das, S.; Hati, S. Significance of whey protein hydrolysate on anti-oxidative, ACE-inhibitory and anti-inflammatory activities and release of peptides with biofunctionality: An in vitro and in silico approach. J. Food Sci. Technol. 2021, 59, 2629–2642. [Google Scholar] [CrossRef]
- Pisoschi, A.M.; Pop, A.; Iordache, F.; Stanca, L.; Geicu, O.I.; Bilteanu, L.; Serban, A.I. Antioxidant, anti-inflammatory and immunomodulatory roles of vitamins in COVID-19 therapy. Eur. J. Med. Chem. 2022, 232, 114175. [Google Scholar] [CrossRef]
- Garbicz, J.; Całyniuk, B.; Górski, M.; Buczkowska, M.; Piecuch, M.; Kulik, A.; Rozentryt, P. Nutritional Therapy in Persons Suffering from Psoriasis. Nutrients 2021, 14, 119. [Google Scholar] [CrossRef]
- Coniglio, S.; Shumskaya, M.; Vassiliou, E. Unsaturated Fatty Acids and Their Immunomodulatory Properties. Biology 2023, 12, 279. [Google Scholar] [CrossRef] [PubMed]
- Gocher, A.M.; Workman, C.J.; Vignali, D.A.A. Interferon-γ: Teammate or opponent in the tumour microenvironment? Nat. Rev. Immunol. 2022, 22, 158–172. [Google Scholar] [CrossRef]
- Benci, J.L.; Johnson, L.R.; Choa, R.; Xu, Y.; Qiu, J.; Zhou, Z.; Xu, B.; Ye, D.; Nathanson, K.L.; June, C.H.; et al. Opposing Functions of Interferon Coordinate Adaptive and Innate Immune Responses to Cancer Immune Checkpoint Blockade. Cell 2019, 178, 933–948.e14. [Google Scholar] [CrossRef] [PubMed]
- Mertowska, P.; Mertowski, S.; Smarz-Widelska, I.; Grywalska, E. Biological Role, Mechanism of Action and the Importance of Interleukins in Kidney Diseases. Int. J. Mol. Sci. 2022, 23, 647. [Google Scholar] [CrossRef] [PubMed]
- Djuric, N.; Lafeber, G.C.M.; Vleggeert-Lankamp, C.L.A. The contradictory effect of macrophage-related cytokine expression in lumbar disc herniations: A systematic review. Eur. Spine J. 2020, 29, 1649–1659. [Google Scholar] [CrossRef] [PubMed]
- Stumpf, F.; Keller, B.; Gressies, C.; Schuetz, P. Inflammation and Nutrition: Friend or Foe? Nutrients 2023, 15, 1159. [Google Scholar] [CrossRef]
- Sierra-Galicia, M.I.; Lara, R.R.-D.; Orzuna-Orzuna, J.F.; Lara-Bueno, A.; Ramírez-Valverde, R.; Fallas-López, M. Effects of Supplementation with Bee Pollen and Propolis on Growth Performance and Serum Metabolites of Rabbits: A Meta-Analysis. Animals 2023, 13, 439. [Google Scholar] [CrossRef]
- Khurelchuluun, A.; Uehara, O.; Paudel, D.; Morikawa, T.; Kawano, Y.; Sakata, M.; Shibata, H.; Yoshida, K.; Sato, J.; Miura, H.; et al. Bee Pollen Diet Alters the Bacterial Flora and Antimicrobial Peptides in the Oral Cavities of Mice. Foods 2021, 10, 1282. [Google Scholar] [CrossRef]
- Aaseth, J.; Ellefsen, S.; Alehagen, U.; Sundfør, T.M.; Alexander, J. Diets and drugs for weight loss and health in obesity—An update. Biomed. Pharmacother. 2021, 140, 111789. [Google Scholar] [CrossRef]
- Lin, X.; Li, H. Obesity: Epidemiology, Pathophysiology, and Therapeutics. Front. Endocrinol. 2021, 12, 706978. [Google Scholar] [CrossRef]
- Zhang, Q.; Bai, Y.; Wang, W.; Li, J.; Zhang, L.; Tang, Y.; Yue, S. Role of herbal medicine and gut microbiota in the prevention and treatment of obesity. J. Ethnopharmacol. 2023, 305, 116127. [Google Scholar] [CrossRef] [PubMed]
- Endalifer, M.L.; Diress, G. Epidemiology, Predisposing Factors, Biomarkers, and Prevention Mechanism of Obesity: A Systematic Review. J. Obes. 2020, 2020, 6134362. Available online: https://pubmed.ncbi.nlm.nih.gov/32566274/ (accessed on 26 March 2023). [CrossRef] [PubMed]
- Savova, M.S.; Mihaylova, L.V.; Tews, D.; Wabitsch, M.; Georgiev, M.I. Targeting PI3K/AKT signaling pathway in obesity. Biomed. Pharmacother. 2023, 159, 114244. [Google Scholar] [CrossRef]
- Sandoval, V.; Sanz-Lamora, H.; Arias, G.; Marrero, P.F.; Haro, D.; Relat, J. Metabolic impact of flavonoids consumption in obesity: From central to peripheral. Nutrients 2020, 12, 2393. [Google Scholar] [CrossRef]
- Oliveira, A.K.D.S.; Silva, A.M.D.O.E.; Pereira, R.O.; Santos, A.S.; Junior, E.V.B.; Bezerra, M.T.; Barreto, R.S.S.; Quintans-Junior, L.J.; Quintans, J.S.S. Anti-obesity properties and mechanism of action of flavonoids: A review. Crit. Rev. Food Sci. Nutr. 2022, 62, 7827–7848. [Google Scholar] [CrossRef] [PubMed]
- Ngamsamer, C.; Sirivarasai, J.; Sutjarit, N. The Benefits of Anthocyanins against Obesity-Induced Inflammation. Biomolecules 2022, 12, 852. [Google Scholar] [CrossRef] [PubMed]
- Martín, M.Á.; Ramos, S. Dietary Flavonoids and Insulin Signaling in Diabetes and Obesity. Cells 2021, 10, 1474. [Google Scholar] [CrossRef]
- Rufino, A.T.; Costa, V.M.; Carvalho, F.; Fernandes, E. Flavonoids as antiobesity agents: A review. Med. Res. Rev. 2020, 41, 556–585. [Google Scholar] [CrossRef]
- Kadar, N.N.M.A.; Ahmad, F.; Teoh, S.L.; Yahaya, M.F. Caffeic Acid on Metabolic Syndrome: A Review. Molecules 2021, 26, 5490. [Google Scholar] [CrossRef]
- Pérez-Torres, I.; Castrejón-Téllez, V.; Soto, M.E.; Rubio-Ruiz, M.E.; Manzano-Pech, L.; Guarner-Lans, V. Oxidative Stress, Plant Natural Antioxidants, and Obesity. Int. J. Mol. Sci. 2021, 22, 1786. [Google Scholar] [CrossRef]
- Yang, S.; Qu, Y.; Chen, J.; Chen, S.; Sun, L.; Zhou, Y.; Fan, Y. Bee Pollen Polysaccharide From Rosa rugosa Thunb. (Rosaceae) Promotes Pancreatic β-Cell Proliferation and Insulin Secretion. Front. Pharmacol. 2021, 12, 688073. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Ren, C.; Yan, S.; Wang, K.; Hrynets, Y.; Xiang, L.; Xue, X.; Betti, M.; Wu, L. Extract of Unifloral Camellia sinensis L. Pollen Collected by Apis mellifera L. Honeybees Exerted Inhibitory Effects on Glucose Uptake and Transport by Interacting with Glucose Transporters in Human Intestinal Cells. J. Agric. Food Chem. 2021, 69, 1877–1887. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.; Li, C.; Wang, S.; Song, X. Green Tea (Camellia sinensis): A Review of Its Phytochemistry, Pharmacology, and Toxicology. Molecules 2022, 27, 3909. [Google Scholar] [CrossRef]
- Brimson, J.M.; Prasanth, M.I.; Kumaree, K.K.; Thitilertdecha, P.; Malar, D.S.; Tencomnao, T.; Prasansuklab, A. Tea Plant (Camellia sinensis): A Current Update on Use in Diabetes, Obesity, and Cardiovascular Disease. Nutrients 2023, 15, 37. [Google Scholar] [CrossRef]
- Noreen, S.; Rizwan, B.; Khan, M.; Farooq, S. Health Benefits of Buckwheat (Fagopyrum Esculentum), Potential Remedy for Diseases, Rare to Cancer: A Mini Review. Infect. Disord. Drug Targets 2021, 21, e170721189478. [Google Scholar] [CrossRef] [PubMed]
- Llanaj, E.; Ahanchi, N.S.; Dizdari, H.; Taneri, P.E.; Niehot, C.D.; Wehrli, F.; Khatami, F.; Raeisi-Dehkordi, H.; Kastrati, L.; Bano, A.; et al. Buckwheat and Cardiometabolic Health: A Systematic Review and Meta-Analysis. J. Pers. Med. 2022, 12, 1940. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, G.; Baroni, L.; Lombardo, M. Promising Sources of Plant-Derived Polyunsaturated Fatty Acids: A Narrative Review. Int. J. Environ. Res. Public Health 2023, 20, 1683. [Google Scholar] [CrossRef]
- Sasani, N.; Kazemi, A.; Babajafari, S.; Amiri-Ardekani, E.; Rezaiyan, M.; Barati-Boldaji, R.; Mazloomi, S.M.; Clark, C.C.T.; Ashrafi-Dehkordi, E. The effect of acorn muffin consumption on glycemic indices and lipid profile in type 2 diabetic patients: A randomized double-blind placebo-controlled clinical trial. Food Sci. Nutr. 2023, 11, 883–891. [Google Scholar] [CrossRef]
- Choi, D.-H.; Han, J.-H.; Hong, M.; Lee, S.-Y.; Lee, S.-U.; Kwon, T.-H. Antioxidant and lipid-reducing effects of Rosa rugosa root extract in 3T3-L1 cell. Food Sci. Biotechnol. 2022, 31, 121–129. [Google Scholar] [CrossRef]
- Baiyisaiti, A.; Wang, Y.; Zhang, X.; Chen, W.; Qi, R. Rosa rugosa flavonoids exhibited PPARα agonist-like effects on genetic severe hypertriglyceridemia of mice. J. Ethnopharmacol. 2019, 240, 111952. [Google Scholar] [CrossRef]
- Farah, S.; Yammine, K. A systematic review on the efficacy of vitamin B supplementation on diabetic peripheral neuropathy. Nutr. Rev. 2022, 80, 1340–1355. [Google Scholar] [CrossRef] [PubMed]
- Mascolo, E.; Vernì, F. Vitamin B6 and Diabetes: Relationship and Molecular Mechanisms. Int. J. Mol. Sci. 2020, 21, 3669. [Google Scholar] [CrossRef] [PubMed]
- Prabhakar, B.; Kulkarni, Y.A. Water Soluble Vitamins and their Role in Diabetes and its Complications. Curr. Diabetes Rev. 2020, 16, 649–656. [Google Scholar] [CrossRef]
- Mason, S.A.; Parker, L.; van der Pligt, P.; Wadley, G.D. Vitamin C supplementation for diabetes management: A comprehensive narrative review. Free. Radic. Biol. Med. 2023, 194, 255–283. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Park, S. A Causal Relationship between Vitamin C Intake with Hyperglycemia and Metabolic Syndrome Risk: A Two-Sample Mendelian Randomization Study. Antioxidants 2022, 11, 857. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Karp, J.; Sun, K.M.; Weaver, C.M. Decreasing Vitamin C Intake, Low Serum Vitamin C Level and Risk for US Adults with Diabetes. Nutrients 2022, 14, 3902. [Google Scholar] [CrossRef]
- Pittas, A.G.; Kawahara, T.; Jorde, R.; Dawson-Hughes, B.; Vickery, E.M.; Angellotti, E.; Nelson, J.; Trikalinos, T.A.; Balk, E.M. Vitamin D and Risk for Type 2 Diabetes in People With Prediabetes: A Systematic Review and Meta-analysis of Individual Participant Data From 3 Randomized Clinical Trials. Ann. Intern. Med. 2023, 176, 355–363. [Google Scholar] [CrossRef]
- Abugoukh, T.M.; Al Sharaby, A.; Elshaikh, A.O.; Joda, M.; Madni, A.; Ahmed, I.; Abdalla, R.S.; Ahmed, K.; Elazrag, S.E.; Abdelrahman, N. Does Vitamin D Have a Role in Diabetes? Cureus 2022, 14, e30432. [Google Scholar] [CrossRef]
- Michael, W.; Couture, A.D.; Swedlund, M.; Hampton, A.; Eglash, A.; Schrager, S. An Evidence-Based Review of Vitamin D for Common and High-Mortality Conditions. J. Am. Board Fam. Med. 2022, 35, 1217–1229. [Google Scholar] [CrossRef]
- Asbaghi, O.; Nazarian, B.; Yousefi, M.; Anjom-Shoae, J.; Rasekhi, H.; Sadeghi, O. Effect of vitamin E intake on glycemic control and insulin resistance in diabetic patients: An updated systematic review and meta-analysis of randomized controlled trials. Nutr. J. 2023, 22, 10. [Google Scholar] [CrossRef]
- Ziegler, M.; Wallert, M.; Lorkowski, S.; Peter, K. Cardiovascular and metabolic protection by Vitamin E: A matter of treatment strategy? Antioxidants 2020, 9, 935. [Google Scholar] [CrossRef] [PubMed]
- Dubey, P.; Thakur, V.; Chattopadhyay, M. Role of minerals and trace elements in diabetes and insulin resistance. Nutrients 2020, 12, 1864. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Yu, J.; Xu, H.; Zhou, Y.; Li, H.; Yin, S.; Xu, D.; Wang, Y.; Xia, H.; Liao, W.; et al. Comparative effects of vitamin and mineral supplements in the management of type 2 diabetes in primary care: A systematic review and network meta-analysis of randomized controlled trials. Pharmacol. Res. 2023, 188, 106647. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.B.; Sorenson, J.C.; Pollard, E.L.; Kirby, J.K.; Audhya, T. Evidence-based recommendations for an optimal prenatal supplement for women in the U.S., part two: Minerals. Nutrients 2021, 13, 1849. [Google Scholar] [CrossRef]
- Le, Y.; Wang, B.; Xue, M. Nutraceuticals use and type 2 diabetes mellitus. Curr. Opin. Pharmacol. 2022, 62, 168–176. [Google Scholar] [CrossRef]
- Xiao, Y.; Zhang, Q.; Liao, X.; Elbelt, U.; Weylandt, K.H. The effects of omega-3 fatty acids in type 2 diabetes: A systematic review and meta-analysis. Prostaglandins Leukot. Essent. Fat. Acids 2022, 182, 102456. [Google Scholar] [CrossRef]
- Bhat, S.; Sarkar, S.; Zaffar, D.; Dandona, P.; Kalyani, R.R. Omega-3 Fatty Acids in Cardiovascular Disease and Diabetes: A Review of Recent Evidence. Curr. Cardiol. Rep. 2023, 25, 51–65. [Google Scholar] [CrossRef]
- Prasad, M.; Jayaraman, S.; Eladl, M.A.; El-Sherbiny, M.; Abdelrahman, M.A.E.; Veeraraghavan, V.P.; Vengadassalapathy, S.; Umapathy, V.R.; Hussain, S.F.J.; Krishnamoorthy, K.; et al. A Comprehensive Review on Therapeutic Perspectives of Phytosterols in Insulin Resistance: A Mechanistic Approach. Molecules 2022, 27, 1595. [Google Scholar] [CrossRef]
- Vezza, T.; Canet, F.; de Marañón, A.M.; Bañuls, C.; Rocha, M.; Víctor, V.M. Phytosterols: Nutritional health players in the management of obesity and its related disorders. Antioxidants 2020, 9, 1266. [Google Scholar] [CrossRef]
- Menezes, R.; Matafome, P.; Freitas, M.; García-Conesa, M.-T. Updated Information of the Effects of (Poly)phenols against Type-2 Diabetes Mellitus in Humans: Reinforcing the Recommendations for Future Research. Nutrients 2022, 14, 3563. [Google Scholar] [CrossRef]
- Shamsudin, N.F.; Ahmed, Q.U.; Mahmood, S.; Shah, S.A.A.; Sarian, M.N.; Khattak, M.M.A.K.; Khatib, A.; Sabere, A.S.M.; Yusoff, Y.M.; Latip, J. Flavonoids as Antidiabetic and Anti-Inflammatory Agents: A Review on Structural Activity Relationship-Based Studies and Meta-Analysis. Int. J. Mol. Sci. 2022, 23, 12605. [Google Scholar] [CrossRef] [PubMed]
- Akpoveso, O.-O.P.; Ubah, E.E.; Obasanmi, G. Antioxidant Phytochemicals as Potential Therapy for Diabetic Complications. Antioxidants 2023, 12, 123. [Google Scholar] [CrossRef] [PubMed]
- Mahboob, A.; Senevirathne, D.K.L.; Paul, P.; Nabi, F.; Khan, R.H.; Chaari, A. An investigation into the potential action of polyphenols against human Islet Amyloid Polypeptide aggregation in type 2 diabetes. Int. J. Biol. Macromol. 2023, 225, 318–350. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Arroo, R. The protective effects of flavonoids and carotenoids against diabetic complications—A review of in vivo evidence. Front. Nutr. 2023, 10, 1020950. [Google Scholar] [CrossRef]
- Islam, F.; Khadija, J.F.; Islam, R.; Shohag, S.; Mitra, S.; Alghamdi, S.; Babalghith, A.O.; Theyab, A.; Rahman, M.T.; Akter, A.; et al. Investigating Polyphenol Nanoformulations for Therapeutic Targets against Diabetes Mellitus. Evid.-Based Complement. Altern. Med. 2022, 2022, 5649156. [Google Scholar] [CrossRef]
- Chu, A.J. Quarter-Century Explorations of Bioactive Polyphenols: Diverse Health Benefits. Front. Biosci. 2022, 27, 134. [Google Scholar] [CrossRef]
- Olas, B. Bee Products as Interesting Natural Agents for the Prevention and Treatment of Common Cardiovascular Diseases. Nutrients 2022, 14, 2267. [Google Scholar] [CrossRef]
- Copur, S.; Demiray, A.; Kanbay, M. Uric acid in metabolic syndrome: Does uric acid have a definitive role? Eur. J. Intern. Med. 2022, 103, 4–12. [Google Scholar] [CrossRef]
- Ozdemir, B.; Gulhan, M.F.; Sahna, E.; Selamoglu, Z. The investigation of antioxidant and anti-inflammatory potentials of apitherapeutic agents on heart tissues in nitric oxide synthase inhibited rats via Nω-nitro-L-arginine methyl ester. Clin. Exp. Hypertens. 2021, 43, 69–76. [Google Scholar] [CrossRef]
- Brainin, M.; Grisold, W.; Hankey, G.J.; Norrving, B.; Feigin, V.L. Time to revise primary prevention guidelines for stroke and cardiovascular disease. Lancet Neurol. 2022, 21, 686–687. [Google Scholar] [CrossRef]
- Kong, A.S.-Y.; Lai, K.S.; Hee, C.-W.; Loh, J.Y.; Lim, S.H.E.; Sathiya, M. Oxidative Stress Parameters as Biomarkers of Cardiovascular Disease towards the Development and Progression. Antioxidants 2022, 11, 1175. [Google Scholar] [CrossRef] [PubMed]
- Adorni, M.P.; Ferri, N. Nutrition Intervention and Cardiovascular Disease. Nutrients 2022, 14, 1435. [Google Scholar] [CrossRef] [PubMed]
- Cainzos-Achirica, M.; Patel, K.V.; Nasir, K. The Evolving Landscape of Cardiovascular Disease Prevention. Methodist DeBakey Cardiovasc. J. 2021, 17, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Hinnen, D.; Kruger, D.; Magwire, M. Type 2 diabetes and cardiovascular disease: Risk reduction and early intervention. Postgrad. Med. 2023, 135, 2–12. [Google Scholar] [CrossRef] [PubMed]
- Sadeq, O.; Mechchate, H.; Es-Safi, I.; Bouhrim, M.; Jawhari, F.Z.; Ouassou, H.; Kharchoufa, L.; AlZain, M.N.; Alzamel, N.M.; Al Kamaly, O.M.; et al. Phytochemical Screening, Antioxidant and Antibacterial Activities of Pollen Extracts from Micromeria fruticosa, Achillea fragrantissima, and Phoenix dactylifera. Plants 2021, 10, 676. [Google Scholar] [CrossRef] [PubMed]
- Weis, W.A.; Ripari, N.; Conte, F.L.; Honorio, M.D.S.; Sartori, A.A.; Matucci, R.H.; Sforcin, J.M. An overview about apitherapy and its clinical applications. Phytomedicine Plus 2022, 2, 100239. [Google Scholar] [CrossRef]
- Durazzo, A.; Lucarini, M.; Plutino, M.; Lucini, L.; Aromolo, R.; Martinelli, E.; Souto, E.B.; Santini, A.; Pignatti, G. Bee Products: A Representation of Biodiversity, Sustainability, and Health. Life 2021, 11, 970. [Google Scholar] [CrossRef]
- Koksal, A.R.; Verne, G.N.; Zhou, Q. Endoplasmic Reticulum Stress in Biological Processing and Disease. J. Investig. Med. 2021, 69, 309–315. [Google Scholar] [CrossRef]
- Ye, J.; Liu, X. Interactions between endoplasmic reticulum stress and extracellular vesicles in multiple diseases. Front. Immunol. 2022, 13, 955419. [Google Scholar] [CrossRef]
- Fernandes-Da-Silva, A.; Miranda, C.S.; Santana-Oliveira, D.A.; Oliveira-Cordeiro, B.; Rangel-Azevedo, C.; Silva-Veiga, F.M.; Martins, F.F.; Souza-Mello, V. Endoplasmic reticulum stress as the basis of obesity and metabolic diseases: Focus on adipose tissue, liver, and pancreas. Eur. J. Nutr. 2021, 60, 2949–2960. [Google Scholar] [CrossRef]
- Ajoolabady, A.; Liu, S.; Klionsky, D.J.; Lip, G.Y.; Tuomilehto, J.; Kavalakatt, S.; Pereira, D.M.; Samali, A.; Ren, J. ER stress in obesity pathogenesis and management. Trends Pharmacol. Sci. 2021, 43, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Lan, J.; Bao, B.; Yao, W.; Cao, Y.; Shan, M.; Cheng, F.; Chen, P.; Zhang, L. Effects of carbonized process on quality control, chemical composition and pharmacology of Typhae Pollen: A review. J. Ethnopharmacol. 2020, 270, 113774. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.M.; Kunugi, H. Apitherapy for Age-Related Skeletal Muscle Dysfunction (Sarcopenia): A Review on the Effects of Royal Jelly, Propolis, and Bee Pollen. Foods 2020, 9, 1362. [Google Scholar] [CrossRef] [PubMed]
- El-Hakam, F.E.-Z.A.; Laban, G.A.; El-Din, S.B.; El-Hamid, H.A.; Farouk, M.H. Apitherapy combination improvement of blood pressure, cardiovascular protection, and antioxidant and anti-inflammatory responses in dexamethasone model hypertensive rats. Sci. Rep. 2022, 12, 20765. [Google Scholar] [CrossRef]
- Al Rifai, M.; Virani, S.S.; Nambi, V. Identifying Differences: A Key Step in Precision Cardiovascular Disease Prevention. J. Am. Hear. Assoc. 2022, 11, e026600. [Google Scholar] [CrossRef]
- Wu, W.; Qiao, J.; Xiao, X.; Kong, L.; Dong, J.; Zhang, H. In vitro and In vivo digestion comparison of bee pollen with or without wall-disruption. J. Sci. Food Agric. 2021, 101, 2744–2755. [Google Scholar] [CrossRef]
- Pohl, P.; Dzimitrowicz, A.; Lesniewicz, A.; Welna, M.; Szymczycha-Madeja, A.; Cyganowski, P.; Jamroz, P. Room temperature solvent extraction for simple and fast determination of total concentration of Ca, Cu, Fe, Mg, Mn, and Zn in bee pollen by FAAS along with assessment of the bioaccessible fraction of these elements using in vitro gastrointestinal digestion. J. Trace Elem. Med. Biol. 2020, 60, 126479. [Google Scholar] [CrossRef]
- Luo, X.; Dong, Y.; Gu, C.; Zhang, X.; Ma, H. Processing Technologies for Bee Products: An Overview of Recent Developments and Perspectives. Front. Nutr. 2021, 8, 727181. [Google Scholar] [CrossRef]
- Aylanc, V.; Falcão, S.I.; Ertosun, S.; Vilas-Boas, M. From the hive to the table: Nutrition value, digestibility and bioavailability of the dietary phytochemicals present in the bee pollen and bee bread. Trends Food Sci. Technol. 2021, 109, 464–481. [Google Scholar] [CrossRef]
| Section | Item | Prisma-ScR Checklist Item | Reported on Page # |
|---|---|---|---|
| Title | |||
| Title | 1 | Identify the report as a scoping review. | 1 |
| Abstract | |||
| Structured summary | 2 | Provide a structured summary that includes (as applicable): background, objectives, eligibility criteria, sources of evidence, charting methods, results, and conclusions that relate to the review questions and objectives. | 1 |
| Introduction | |||
| Rationale | 3 | Describe the rationale for the review in the context of what is already known. Explain why the review questions/objectives lend themselves to a scoping review approach. | 1, 2 |
| Objectives | 4 | Provide an explicit statement of the questions and objectives being addressed with reference to their key elements (e.g., population or participants, concepts, and context) or other relevant key elements used to conceptualize the review questions and/or objectives. | 1, 2 |
| Methods | |||
| Protocol and registration | 5 | Indicate whether a review protocol exists; state if and where it can be accessed (e.g., a web address); and if available, provide registration information, including the registration number. | None |
| Eligibility criteria | 6 | Specify characteristics of the sources of evidence used as eligibility criteria (e.g., years considered, language, and publication status), and provide a rationale. | 4, 7 |
| Information sources | 7 | Describe all information sources in the search (e.g., databases with dates of coverage and contact with authors to identify additional sources), as well as the date the most recent search was executed. | 7 |
| Search | 8 | Present the full electronic search strategy for at least 1 database, including any limits used, such that it could be repeated. | 7 |
| Selection of sources of evidence | 9 | State the process for selecting sources of evidence (i.e., screening and eligibility) included in the scoping review. | 4, 7 |
| Data charting process | 10 | Describe the methods of charting data from the included sources of evidence (e.g., calibrated forms or forms that have been tested by the team before their use, and whether data charting was performed independently or in duplicate) and any processes for obtaining and confirming data from investigators. | 3, 4, 7 |
| Data items | 11 | List and define all variables for which data were sought and any assumptions and simplifications made. | 2, 7–46 |
| Critical appraisal of individual sources of evidence | 12 | If carried out, provide a rationale for conducting a critical appraisal of included sources of evidence; describe the methods used and how this information was used in any data synthesis (if appropriate). | None |
| Synthesis of results | 13 | Describe the methods of handling and summarizing the data that were charted. | 4–7 |
| Results | |||
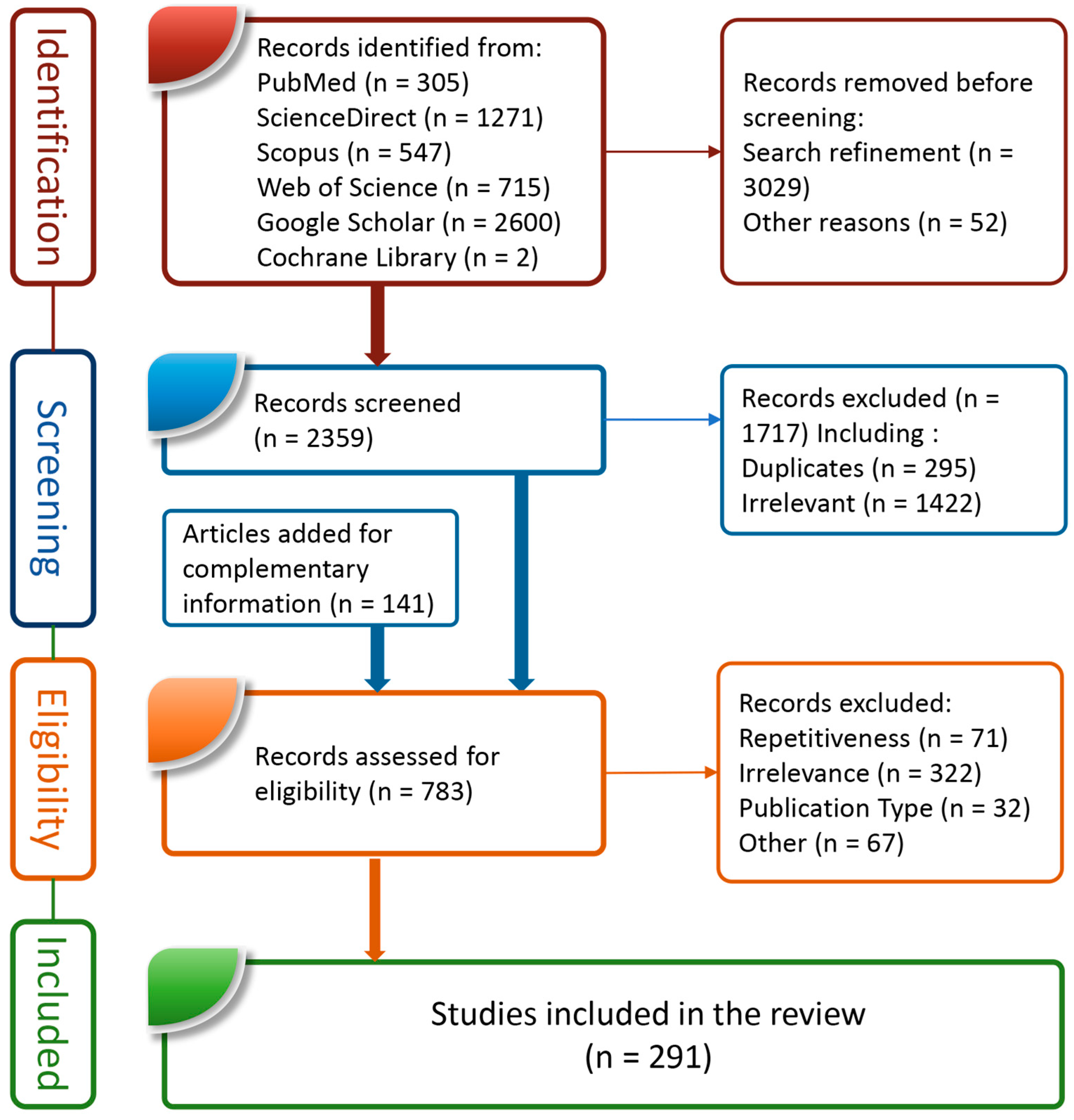
| Selection of sources of evidence | 14 | Give numbers of sources of evidence screened, assessed for eligibility, and included in the review, with reasons for exclusions at each stage, ideally using a flow diagram. | 4,7 |
| Characteristics of sources of evidence | 15 | For each source of evidence, present characteristics for which data were charted and provide the citations. | 4, 7 |
| Critical appraisal within sources of evidence | 16 | If carried out, present data on critical appraisal of included sources of evidence (see item 12). | 35, 39, 42, 47, 48 |
| Results of individual sources of evidence | 17 | For each included source of evidence, present the relevant data that were charted that relate to the review questions and objectives. | 7–46 |
| Synthesis of results | 18 | Summarize and/or present the charting results as they relate to the review questions and objectives. | 8, 14–25, 28–33, 36, 39, 41, |
| Discussion | |||
| Summary of evidence | 19 | Summarize the main results (including an overview of concepts, themes, and types of evidence available), link to the review questions and objectives, and consider the relevance to key groups. | 28, 35, 38, 42, 45–48 |
| Limitations | 20 | Discuss the limitations of the scoping review process. | 48 |
| Conclusions | 21 | Provide a general interpretation of the results with respect to the review questions and objectives, as well as potential implications and/or next steps. | 46–48 |
| Funding | |||
| Funding | 22 | Describe sources of funding for the included sources of evidence, as well as sources of funding for the scoping review. Describe the role of the funders of the scoping review. | 49 |
| Form of BP Use | Botanical Origin | Geographical Origin | Experiment Protocol | Reported Results | Unveiled Mechanisms | Ref. | Correlation with TPC (*) |
|---|---|---|---|---|---|---|---|
| 80% ME | NA (*) | Poland | ABTS (*), DPPH (*), FRAP (*), CUPRAC (*), PCL (*) | Marked AO (*) activity (much higher than bee bread, wax and honey). | Radical scavenging, ion reduction. | [109] | Strong |
| Whole dried BP in water | NA | European countries | Oxidation-reduction potential (ORP) | The first study of ORP in BP. Marked improvement in AO status. | Radical scavenging. Overall decrease in oxidant abundance. | [131] | Moderate |
| BP fed to animal models of POS (*) | NA | NA | TAC (*) of serum measured by FRAP | TAC increased. | Radical scavenging. Apoptosis increase in ovarian cysts. | [132] | NA |
| BP protein hydrolysates (enzymatic) | NA | Thailand | ABTS and DPPH | AO activity, and apoptosis activation in a human lung cancer cell line. | Radical scavenging. Promoting cancer cells apoptosis. | [133] | NA |
| Ethanol 95% and water extracts | Multifloral: Castanea sativa, Hedera helix, Rubus ulmifolius. | Italy | In vitro: ABTS, DPPH, ORAC (*), FRAP, Fe2+. In erythrocytes: CAA (*), oxidative lysis | Good AO activities in all assays. Erythrocytes hemolysis prevention. BP microbiota yeasts also showed AO activity. | Radical scavenging, ion reduction, inhibition of free radical production, cell lysis prevention, metal-chelating ability. | [11] | NA. |
| Water extract | Multifloral: Citrus aurantium, Apiaceae | Morocco | DPPH, ABTS, and FRAP. | AO activity of BP was much more pronounced than honey in all assays. | Radical scavenging, ion reduction. | [134] | Strong |
| Absolute EE | Mainly Brassica rapa, Eschscholzia californica. Others. | Chile | FRAP, ORAC-fluorescein. | The AO activity varied significantly between samples, independently of TPC. | Radical scavenging, ion reduction. | [135] | No correlation |
| 70% EE | Many monoflorals, Other Multiflorals | Morocco | DPPH, ABTS, Reducing Power Assay (ferric ion reduction). | Authors curiously reported that an Ononis Monofloral BP had the lowest activity in DPPH and ABTS test, and another botanically similar BP had the highest activity in all assays. | Radical scavenging, ion reduction. | [136] | High for DPPH and ABTS, but nil for FRAP. |
| BP feeding to aged horses during autumn | NA | NA | In the blood: FRAP, GSH-Px (*), [SH] (*) as indicator of OS (*). | FRAP declined significantly, and SH increase was prevented, while GSH-Px activity was not changed | Radical scavenging, protein oxidation abolition, but AO defense (GSH-Px activity) not enhanced | [137] | NA |
| 80% ME of fermented and non-fermented BP | NA | Different European regions | In vitro: DPPH (fermentation either spontaneous or by lactic bacteria) | Increased radical scavenging activity and total phenolic and flavonoid contents in fermented BP | Radical scavenging. | [78] | High |
| Polysaccharide fractions of BP | Lycium chinense | China | In vitro: DPPH, O2− radical scavenging, ABTS, Fe2+ chelating | Fraction with lower molecular weight and higher uronic acid content had the highest AO activity. | Radical scavenging, low ferrous ion chelating ability. | [138] | NA |
| EE | NA | Turkey | In vitro: FRAPIn vivo: Rat hippocampus | FRAP reduction in vitro and BDNF (*) increase in vivo. | Radical scavenging, AO defense enhancement. | [139] | High |
| 80% methanol for assays, BP powder to frankfurters | Predominantly Brassica napus and Fraxinus spp. | Serbia | TAC, FRP, ABTS, DPPH, TBARS (*) | Strong AO activity in all tests, good stabilization of sausages. | Radical scavenging, ion reduction, prevention of free radial production, food stabilization | [140] | NA |
| Pollen feeding to zebrafish | NA | Brazil | ABTS | BP had radical scavenging activity but induced higher growth of melanoma without weight gain. | Such results have never been reported in other studies. | [141] | NA |
| BP feeding to broilers | NA | Saudi Arabia | DPPH, TAC, total SOD (*) (T-SOD), and CAT (*) in blood samples. | In vitro: DPPH decrease, In vivo: Increased TAC, T-SOD, and CAT activities. | Radical scavenging, AO defense enhancement. | [142] | NA |
| 80% ethanol extract | Miscellaneous multifloral BPs | Spain | DPPH, ABTS | Botanical origin and climatic conditions determine AO activity of BP | Radical scavenging. | [9] | High |
| In vitro: 70% EE of wall-broken BP Animals: raw and wall-broken BP | Rosa rugosa | China | ORAC, DPPH, ABTS | In vitro: AO activity improved by BP wall-breaking. In vivo: Elevated SOD and CAT. MDA (*) decrease. Protection of kidney, liver, spleen and thymus. | Radical scavenging, reduction in free radical production, enhancement of AO defense, cell and tissue protection. | [143] | NA |
| 95% EE fractionated by organic solvents | NA | Saudi Arabia | MDA levels and AO status (CAT, Vit. C, GSH), GST (*) in rat brains. | Inhibition of lipid peroxidation and increase in AO defense biomarkers depending on BP fraction. Protective effects in other organs than the brain. | Radical scavenging, AO defense enhancement, prevention of free radical generation. | [144] | Correlation present but extent not assessed |
| Water extract | NA | Morocco | DPPH, FRAP, Phosphomolybdate assay. | Marked DPPH decrease and ferric ion reduction. | Radical scavenging, ion reduction. | [145] | NA |
| 70% ME | Diverse botanical species | Italy | ORAC, DPPH, ABTS | AO capacity varied with botanical species and geographical area even for the same botanical species. It was higher for multi-floral pollens. | Radical scavenging. | [107] | Significant with some exceptions |
| 80% EE after petroleum treatment to remove lipids | Rape (Brassica sp.) | China | DPPH, ABTS, FRAP | AO activity decreased as follows: phenolamines fraction > crude extract > flavonoid fraction. | Radical scavenging, ion reduction. | [146] | NA |
| 70% EE | 4 Mono-floral, One bi-floral, and 13 Multifloral BPs. | South Korea | DPPH | 16/18 samples had potent AO activity depended on botanical and geographical origins and climate | Radical scavenging | [105] | Absent for both TPC and TFC (*) |
| 70% ME | NA | Poland | ABTS | AO activity varied largely between samples although always present. | Radical scavenging. | [104] | High |
| 95% EE | Species from diverse botanical families | Turkey | TAC assessed by CERAC and CUPRAC (*). | Strong AO activity in all samples with high TPC and unsaturated fatty acid contents | Radical scavenging, ion reduction. | [147] | NA |
| EE | Chestnut (Castanea sp.) | Turkey | FRAP, CHROMAC (*), and ABTS. Fenton reaction for DNA (*) oxidative damage | Marked AO activity, protection against DNA oxidative damages. | Radical scavenging, ion reduction, Inhibition of free radical production, especially DNA oxidation byproducts | [148] | No correlation |
| 80% EE | Multifloral (17 pollen types present at >3%) | Portugal |
| Marked decrease in radical scavenging and reducing power after digestion (only one BP has the highest AO capacity at the end of the digestion). | Radical scavenging, ion reducing effect | [99] | Strong correlation |
| 70% EE | Rape (Brassica sp.) BP: fermented and unfermented | China | In vitro: DPPH, ABTS, FRAP. In human hepatocytes: CAA | Both pollen types were active, but the fermented one was more potent in all assays. | Radical scavenging and reducing effect. High increase in TPC and TFC after fermentation | [70] | NA |
| 95% EE | Mono-floral BPs. | Italy | FRAP in vitro, Cellular AO Activity in Red Blood Cells (ex vivo) | BP composition and activity varied even for similar botanical origin. Marked AO activity in both assays | Ion reduction, and cell protection against oxidative injuries. | [103] | Positive for TPC and TFC |
| 70% EE | BP from a region with limited botanical species | Morocco | TAC evaluated by the phosphor-molybdenum method; DPPH | High AO and radical scavenging activities. TPC, TFC and TAA of BP were markedly lower than propolis | Radical scavenging and reducing effect. | [149] | NA |
| 85% ME |
| India | DPPH, FRAP, ABTS, Metal chelating activity (MCA) on Fe2+ | All samples were active with variations according to the sample and used test | Radical scavenging, ion reduction and metal chelation. Similar botanical origins resulted in equivalent AO activities. | [102] | Very strong except for very few molecules |
| Multistep extraction by different organic solvents | Cynara scolymus | Serbia | DPPH, ABTS, TAC, FRAP, and FCC. | Extractable phenolic fraction largely more potent in ABTS, FRP and TAC. Lipid fraction had high TAC. Hydrolysable fraction: highest DPPH. | Radical scavenging, ion reduction, metal chelating, and inhibition of free radical production. | [150] | NA |
| 75% EE dissolved in methanol/water, then extracted in 2 fractions: ethyl acetate and n-hexane | Camellia japonica | China | DPPH, FCC, FRAP, Effect on DNA Oxidation induced by Hydroxyl Radicals |
| Radical scavenging, AO defense enhancement, prevention of free radical generation, Increase in the gut microbiota diversity and beneficial strain abundance. | [151] | Significant in the two studied fractions |
| 70% EE | Multifloral samples | Romania | ABTS | Studied AO activity correlated fully with both TPC and TFC. | Radical scavenging, ion reduction. | [74] | Strong correlation |
| Micronized BP added to multi-flower honey | NA | Poland | TAC, DPPH, ABTS, FRAP, CUPRAC | Increase in TPC, TFC, phenolic acids, anthocyanin, and carotenoid contents of honey. | Excellent augmentation of total AO, antiradical and reducing activity of honey. | [51] | NA |
| Ethanol, methanol, water, and 70% ethanol extracts | Eucalyptus marginata and Corymbia calophylla | Australia | FRAP, DPPH | An extensive analysis of used extraction protocols in the literature and multiple fractions in this study. | Non-pulverized BP extracted with 70% EE coupled with agitation is the best method to maximize phenolics and AOs. | [101] | NA |
| Ethanol extract | NA | NA | In hypertensive rat testes: TAS (*), TOS (*), NF-κB (*), MDA, NO (*), PON1 (*) and CAT. | BP markedly restored the activity of PON1 and CAT, enhanced TAS, reduced TOS, NF-κB, and MDA levels, and relatively restored NO levels. | Reducing total oxidant status, increasing AO defense, and thus abolishing reproductive function damages. | [152] | NA |
| Hexane and then methanol or acidified methanol extraction | NA. Stingless bee (Tetragonula biroi) pot pollen. | Philippines | FRAP, DPPH, and ABTS, and lipid peroxidation inhibition (BCB (*) activity). | Significant differences between different extracts (all were active). DPPH and FRAP lower than those of A. mellifera BP in other studies. | Radical scavenging, ion reduction, inhibiting of free radical production (from lipid peroxidation). | [153] | High for TPC and TFC, except on lipid peroxidation |
| Enrichment of biscuits with dried BP | Mono-floral BP: Brassica napus, Phacelia tanacetifolia and Helianthus annuus | Hungary | DPPH, FRAP, TEAC (*) | Marked increases in AO activities and TPC in all samples with a ratio of 10% BP/Biscuit (w/w). TPC and AO activity decreased as follows: phacelia > rapeseed > sunflower | Radical scavenging, ion reduction, food stabilization. | [8] | High |
| 70% EE | Multi-floral: Echium plantagineum, Cistus ladanifer and Quercus sp., etc. | Portugal | In vitro: DPPH, NO and O2- radical assaysIn erythrocytes: HB, lipid and others; hemolysis | Radical scavenging was much lower than that of ascorbic acid. | Radical scavenging, inhibition of free radical production and intracellular reactions, cell protection against oxidation and lysis. | [154] | High, especially for selected compounds |
| 70% EE | Zea mays | Malaysia | DPPH | Strong AO activities and high TPC. | Radical scavenging. | [155] | strong |
| BP Lipid fraction evaluated by lipidomics | Camellia sinensis | China | Antioxidative gene expression after DSS(*) OS, and gut barrier function | Activating the Nrf2-ARE-dependent pathway (*) and activating NF-κB, TOR (*), and Nrf2 signaling factors. | Enhancing the AO defense and the general barrier function and structure in the gut epithelium | [58] | NA |
| 95% EE | Multi-floral: Castanea, Rubus, and Cistus. | Italy | Ex vivo (erythrocytes): CAA, hemolysis, cellular ROS (*) production | All samples were similarly efficient in preventing AAPH (*)-induced hemolysis and OS. | Boost of cellular AO status, prevention of membrane alteration and cell injuries and lysis. | [156] | Judged probable but not confirmed |
| 70% EE | Schisandra chinensis | China | In vitro: ABTS, FRAP Infarcted rat myocardium: SOD, GSH-Px, CAT | AO activity in vitro and significant in vivo increase in AO enzyme activities at high doses of BP extract. | Radical scavenging, ion reduction, AO defense activation and myocardial tissue protection. | [157] | NA |
| Water extract | NA | Egypt | In Fluvastatin-induced hepatitis rat liver: MDA, GSH, GSH-Px and CAT | Lower MDA and higher GSH levels, and higher GSH-Px, and CAT activities. Synergism with thymoquinone. | Inhibition of free radical production, activation of AO defense, and liver tissue protection. | [158] | NA |
| Absolute EE | Diverse botanical origins | Chile | In vitro: FRAP, ORAC In a hepatic cell line: Cytotoxicity and hepatoprotection AAPH | AO activity in all samples but varied largely. Hepatic cell death prevented. Significant reduction in lipid accumulation in steatosis. | Radical scavenging, ion reduction. Cell protection against lipid loads and AAPH. | [159] | Present for TPC and TFC |
| BP diet feeding to Apis mellifera isolated in cages | Predominantly Solidago spp. | United States | In bee abdomens: SOD, CAT, vitellogenin (VG), HSP70 and HSP90 (*). | VG expression very high in BP-fed compared to sugar-fed bees. Other parameters did not differ. | Improvement in a vital protein status. In honeybees, VG is a vital protein with AO activity [160]. | [161] | NA |
| Hexane, 70% ethanol and ethyl acetate successively | Camellia sinensis | China | NQO1 (*), Txnrd1 (*), and Nrf2 genes in colon adenocarcinoma cells | Potent increase in NQO1, Txnrd1 and Nrf2 gene expressions following the DSS-induced oxidative stress. | Enhancing cellular AO defense. | [162] | NA |
| 70% EE | Mono-floral and multi-floral BPs | Morocco | DPPH, ABTS, FRAP | Potential AO activity with great difference in all samples, and Thymus vulgaris BP being the most active. | Radical scavenging, ion reduction. | [163] | No correlation |
| ME fractioned with hexane and dichloro-methane | Helianthus annuus | Thailand | DPPH | AO potential of different fractions varied from nil to low, while it was absent for crude extracts. | No AO activity. Botanical origin and extraction method may determine the AO activity. | [164] | NA |
| 70% EE of BP from Melipona fasciculata | NA | Brazil | DPPH, FRAP and ABTS | AO activity present in all samples, varied largely and is higher than that of Apis mellifera BP in other studies. | Radical scavenging, ion reduction. | [165] | No correlation |
| 75% EE | Rape (Brassica sp.) | China | In mice models of DSS-induced colitis: Oxidative and microbiota markers | Increase in SOD and GSH-Px activities decrease in NO and MPO (*) levels. Increase in microbiota diversity and beneficial strains abundance. | Radical scavenging, AO defense enhancement, prevention of free radical generation, microbiota modulation. | [166] | NA |
| Water or 75% EE assisted with ultrasounds or heat reflux | Actinidia arguta | China | In vitro: FRAP, FCC, and DPPH. In a plasmid DNA and in lymphocytes: OS-induced damages | Strong AO activities. EEs were more potent (more with ultrasound in FRAP and FCC). Potent inhibition of DNA alterations | Radical scavenging, reduction Abolishing oxidative damages to circulating DNA and cytoprotective effect on lymphocytes. | [167] | Evident (but only few samples were studied) |
| 70% EE from pulverized BP powder | Schisandra chinensis | China | In H2O2-injured cardiomyocyte: Cell survival, morphology, MDA, SOD, and GSH. | Increase in cell viability, SOD and GSH, marked prevention of morphological alterations, and decrease in MDA levels. | AO defense enhancement, prevention of free radical generation, cell protection against oxidative injuries and death. | [168] | NA |
| Water, 80% EE and supercritical fluid extracts (SFE) | Chestnut (Castanea sp.) | Italy | DPPH, ferrous ion reduction. | EE by far the most active in both assays followed by SFE and water extracts. SFE was the richest in TPC. | Radical scavenging, ion reduction. | [169] | No correlation |
| BP powder added to skimmed goat milk | Helianthus annuus | Serbia | Polyphenol bio-accessibility in a digestion model. TAC, ABTS, FCC | BP reduced milk TAC and TPC, and reduced polyphenol bioaccessibility. Digestion increased RSA. | Enhancing AO potential and functionality. The TAC decrease is probably due to polyphenol capture by casein micelles. | [170] | NA |
| Water extracts | Different botanical origins | Egypt | DPPH | BP was the most active compared to bee bread, royal jelly, and different mixes of bee products. | Radical scavenging. | [44] | No correlation |
| 70% EE | NA. BP of stingless bee Scaptotrigona affinis postica | Brazil | DPPH, ABTS, FRAP | DPPH AO activity and TPC much higher than Apis mellifera BP in other studies. FRAP and ABTS AO activity similar to other bee species. | Radical scavenging, ion reduction. | [171] | NA |
| 70% ME | Multifloral BPs | Poland | ABTS | ABTS AO activity varied with TPC | Radical scavenging. | [106] | High |
| Water | NA | NA. | MDA, CAT and SOD in rat testes damaged by methotrexate | Decrease in MDA levels and SOD activity. No change in CAT activity. BP restored rat fertility parameters. | Reducing lipid peroxidation, enhancing AO state, and reproductive organ structure and function | [172] | NA |
| Water | NA. BP of stingless bee Trigona spp. | Indonesia | DPPH. | Mild activity: the 1/4th of vitamin C, better than propolis and honey. | Radical scavenging. | [173] | NA |
| 70% EE | NA | Morocco | DPPH, ABTS and FRAP | AO activity in all assays. Remained however lower than vitamin C. | Radical scavenging, ion reduction, protection against induced cell and tissue injury. | [174] | NA |
| ME partitioned by hexane, DCM and methanol | Monofloral BPs | Thailand | DPPH, ABTS, FRAP | Only DCM fraction from M. diplotricha showed strong AO potential (however less than ascorbic acid). | Radical scavenging, ion reduction. | [175] | No correlation |
| 70% EE | Monofloral Lotus BP | China | In vitro: GSH, SOD and MDA levels. In isoproterenol-injured cardiomyocytes: cell protection | Significant increase in GSH and SOD activity, and significant decrease in MDA levels in the studied cell line. | AO defense enhancement, prevention of free radical generation, cell protection against oxidative injuries and apoptosis. | [15] | NA |
| BP Preparation | Experiment Details | Reported Results | Ref. |
|---|---|---|---|
| 70% EE of unfermented and S. cerevisiae-fermented rape BP from China | In a lipopolysaccharide-treated macrophage cell line: NO, COX-2, TNF-α (*), IL-1β and IL-6 levels. | NO, COX-2, TNF-α, IL-1β, and IL-6 levels markedly decreased dose-dependently (fermented BP more potent in all tests. | [70] |
| 95% EE of Italian BP composed mainly from Castanea sativa (88.8%) and Hedera helix sp. (4.2%). | Cell viability and gene expression of IL-8, COX-2, and ICAM-1 (*) were assessed in a TNFα-inflamed human colorectal adenocarcinoma cell line. | Pretreatment with BP extract reduced IL-8, COX-2, and ICAM-1 levels and did not alter cell viability even at very high doses. | [11] |
| Animals fed with BP from Turkey. Other products assessed (bee bread, honey, royal jelly and propolis). | Bee products were administered separately to rat models of chronic inflammation. Pro- and anti-inflammatory cytokines were measured in the blood. | BP increased anti-inflammatory cytokines IL-4, IL-10, and IL-1RA, and decreased pro-inflammatory cytokines TNF-α, IL-1ß, and IL-6. BP was more potent than all other bee products. | [199] |
| Extraction with 90% ethanol of Fagopyrum esculentum BP from China. | Colitis was induced by DSS in male mice. DAI and other inflammation markers (ICAM-1 expression and colon tissue imaging) were assessed. | BP extract markedly reduced DAI; corrected DSS-induced loss of weight and colon weight and length, significantly reduced spleen swelling and lymphocytes and iCAM-1 expression; corrected damages in epithelial barrier and villi, and restored AO defense and activated/regulatory T-cell balance. | [200] |
| Pulverized Brazilian BP was fed to zebrafish (botanical origin was not specified). | In transgenic zebrafish experimentally-induced melanoma: Metagenomic analysis of gut microbiome, and assessment of pro-inflammatory mediators in gut. | Significant increase in beneficial microbiota strains and a decrease in some known pathogenic strains. Unexpectedly, BP did not change tested pro-inflammatory mediators and induced a higher tumor growth than in control animals. | [141] |
| Mice models fed with 90% EE of Fagopyrum esculentum BP from China. | In high-fat diet- and streptozocin-induced diabetes mice: Pro- (TNF-α, IL-2 and IL-6) and anti-inflammatory (TGF-ß (*)) cytokines, and PI3K/AKT (*) in rat pancreas. Genomic study of colon microbiota. | BP reduced liver infiltration of inflammatory cells, decreased gene expression of TNF-α and TGF-ß, and increased that of PI3K and AKT, and enhanced microbiota richness, diversity, and beneficial strain abundance. Other inflammation triggers markedly reduced (e.g., OS markers, liver injuries, steatosis) | [201] |
| An extract of Lotus BP with 70% ethanol was added to a myocardial cell culture. | Cardiomyocyte hypertrophy was induced with isoproterenol. | BP inhibited the increase in pro-inflammatory cytokines (IL-6 and TNF-α) and the JAK2/STAT3 (*) signaling pathway, and increased anti-apoptosis related factors (lowered Bax and Caspases levels, and increased Bcl-2 and Bcl-2/Bax ratio (*)). | [15] |
| Commercial BP was fed to rats. Botanical origin not identified. | Previously PB-fed rats were treated by propionic acid to induce autistic state. Cytokines, OS markers, and gut microbiota were analyzed. | In the rat brains, BP induced marked decrease in IFN-γ (*), IL-1α, and IL-6, and had no effect on IL-12, TNF-α, and VEGF (*). BP induced a significant decrease in fecal pathogen strains and protected animals against other inflammation comorbidities such as OS and glutamate excitotoxicity. | [65] |
| BP, raw or fermented either spontaneously or by a selected inoculum, was used for in vitro experiments. | BP samples added either to an in vitro model of gastrointestinal batch digestion, or to a human adenocarcinoma or keratinocyte cell line cultures. Pro-inflammatory mediators, ROS, intestinal barrier function and cell viability were assessed. | Serum availability of phenolics increased by 22% with selectively fermented BP and not changed by raw and spontaneously fermented BPs. Inhibitory potential on Il-6, IL-8, MCP-1 and TNF-α was significant by all samples only in inflamed cells (selectively fermented > spontaneously fermented > raw). | [80] |
| Extract from Chinese Camellia japonica BP with 75% ethanol, dissolved in methanol and water, and then fractionated with ethyl acetate and n-hexane. | Hyperuricemic mice fed with the ethyl acetate fraction. Pro-inflammatory cytokines in serum and kidney, kidney histopathology, gut microbiota analysis and fecal SCFA (*) were assessed. | All assessed cytokines (TNF-α, IL-6, IL-1β and IL-18) in animal serum and kidneys were decreased, TLR4/MyD88/NF-κB (*) and NLRP3/ASC/Caspase-1 (*) inflammatory pathways were both inhibited, microbiota diversity was enhanced, and levels of SCFA increased. BP reduced renal histological damages and interstitial infiltration of inflammatory cells. Persistent tissular alterations in allopurinol-treated mice surprisingly disappeared in BP-treated ones. | [151] |
| BP water extract was administered orally to rats. Botanical and geographical origins were not indicated. | A bone marrow/spleen suppression in rats was induced by doxorubicin. BP was administered at 100 and 200 mg/kg body weight. Inflammatory cytokines, oxidative and other hematological parameters, and apoptosis-related genes were measured. | BP induced a significant decrease in PF4 (*), IL-6 and, IL-1β and a significant increase in serum GM-CSF (*), G-CSF (*), and IL-10. BP increased hematopoietic factors, and blood cells and platelet numbers, reduced OS and apoptotic mechanisms, and repaired histological injuries. | [202] |
| A 70% EE of BP collected by the stingless bee Scaptotrigona affinis postica from Brazil. Botanical Origin not indicated. | Paw edema was induced in mice either by Carrageenan (with indomethacin as standard) or Dextran (cyproheptadine as a standard). Antinociceptive effect was assessed using acetic acid or formalin. | In the acute inflammation phase, BP reduced edema volume in a more potent manner than the two drug references. It also reduced nociceptive response more markedly than indomethacin. | [171] |
| Methanolic extract of BP. Botanical and geographical origins not indicated. | Air pouch was induced in rats and then carrageenan was injected in it with or without BP. Morphological and molecular inflammation markers were assessed. | BP extract significantly reduced inflammatory exudate accumulation, leukocyte infiltration, granulation tissue formation, and angiogenesis. TNF-α and VEGF significantly lowered. | [203] |
| Purified polysaccharide fraction (BPPF) of Fagopyrum esculentum BP from China. | A microbiota dysbiosis was induced with ceftriaxone in mice. Effect of the BPPF on inflammatory mediators, intestinal structure and function, microbiota, and immune response were assessed. | BPPF restored blood LPS (*) and thymus and spleen atrophies to the normal values. In colonic mucosa, inflammatory cell infiltration disappeared, goblet cells and mucus area were fully restored, structural alterations were corrected, TNF-α levels were not affected, and IL-6 and IL-1β levels were fully restored. Despite some differences with control group, BPPF restored microbiota diversity, richness and corrected its balance. | [196] |
| Multistep and multi-solvent extracts of fresh monofloral BPs from Camellia sinensis, Nelumbo nucifera, and Brassica campestris. | Molecular methods, metabolomics and a macrophage cell line with LPS-induced inflammation were used to study the anti-inflammatory potential of the extracts. C. sinensis BP was further studied in an LPS-induced acute lung injury mouse model. | In macrophages, all extracts reduced NO release and IL-1β, IL-6, IL-10, iNOS (*), and COX-2 gene expressions, and increased HO-1 gene expressions (except N. nucifera BP on IL-1β, IL-6, and COX-2). MAPK (*) and NF-κB pathways were markedly suppressed by C. sinensis and B. campestris BP extracts. In all cited effects, C. sinensis BP was the most active and the richest in TPC. In the lungs, this extract reduced inflammatory cell infiltration, COX-2 and iNOS expression and NLRP3 inflammasome activation. All effects were more marked than dexamethasone used as standard. | [204] |
| Commercial BP bought in Saudi Arabia was fed to rats (botanical and geographical origin not indicated). | Neurotoxicity was induced in rats by prenatal exposure to methylmercury. Markers of neuroinflammation, along with other neurological parameters, were examined in rat brains after a BP diet. | Brain IFN-γ significantly reduced by BP with a greater effect obtained when mothers were fed with BP before the offspring birth. Levels of many neurotransmitters, which may interfere in an interplay with neuroinflammation, were also significantly corrected. | [34] |
| Chinese Camellia sinensis BP was treated with hexane. The residue was then treated with 70% ethanol and later with ethyl acetate. | BP extract was applied on a DSS-challenged adenocarcinoma colonic monolayer cell line. inflammatory cytokine genes expression, gut barrier function, MAPK signaling pathway were studied. | Remarkable increase in TGF-β1 and decrease in TNF-α and IL-6 gene expression. Alterations of monolayer integrity, tight junction disruption and gut permeability significantly corrected. MAPK signaling pathway was markedly suppressed. | [162] |
| Extract of Chinses Schisandra chinensis BP with 75% ethanol. | Extract was fed to obese mice models of high-fat-induced nonalcoholic fatty liver disease. | IL-1β, TNF-α, NF-κB and iNOS markedly reduced in the liver and increases in serum IL-6 and TNF-α levels were completely inhibited. Shifts in microbiota diversity and abundance were corrected. | [205] |
| Purified polysaccharide fractions from Chinese Lycium chinense BP. | DSS-challenged macrophage cell line was treated by the polysaccharide fractions. Inflammatory mediators were then assessed. | A significant increase in NO and TNF-α levels were obtained. IL-1β and IL-6 were also increased. The effects depended more tightly on the fraction type than the used dose. | [138] |
| Brazilian BP collected by the stingless bee Melipona fasciculata was extracted with 70% ethanol. Botanical origin not identified. | COX inhibition was performed in vitro. Paw edema was induced in mice either by Carrageenan (with indomethacin as standard) or Dextran (cyproheptadine as a standard). Antinociceptive effect was assessed using acetic acid or formalin. | The extract inhibited COX-1 and, in a largely more potent manner (100% inhibition with 500mg BP/kg body weight), COX-2 activity. BP extract reduced edema volume in a more potent manner than the two drug references. It also reduced nociceptive response more markedly than indomethacin. | [165] |
| 75% EE from Chinese Brassica sp. BP. | Mice models of DSS-induced colitis were fed with BP extract. Histomorphometry, inflammatory mediators measurement, and microbiota analysis were performed. | Downregulation was very potent on IL-1β, and marked on IL-6, NF-κB, and IκB (*). BP extract abolished weight loss, colon shortening, spleen swelling, and villi, crypts, glandular, and epithelial alterations. The overall colitis DAI was markedly reduced. BP extract ameliorated gut microbiota diversity and the relative abundance of many beneficial genera, and completely reversed the abundance of some strains which was raised by DSS. | [166] |
| Hamsters were fed with BP. Botanical and geographical origin were not indicated. | BP was fed to animals after autism induction with propionic acid. Markers of neuroinflammation were evaluated in animal brain tissues. Fecal Clostridium difficile was searched. | BP increased IL-10, completely abolished the propionic acid-induced increase in IL-6, and significantly decreased IFN-γ, IL-1α, and VEGF. TNF-α was slightly decreased. Effect on IL-12 was insignificant. C. difficile toxins not detected in BP-treated animals. | [206] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kacemi, R.; Campos, M.G. Translational Research on Bee Pollen as a Source of Nutrients: A Scoping Review from Bench to Real World. Nutrients 2023, 15, 2413. https://doi.org/10.3390/nu15102413
Kacemi R, Campos MG. Translational Research on Bee Pollen as a Source of Nutrients: A Scoping Review from Bench to Real World. Nutrients. 2023; 15(10):2413. https://doi.org/10.3390/nu15102413
Chicago/Turabian StyleKacemi, Rachid, and Maria G. Campos. 2023. "Translational Research on Bee Pollen as a Source of Nutrients: A Scoping Review from Bench to Real World" Nutrients 15, no. 10: 2413. https://doi.org/10.3390/nu15102413
APA StyleKacemi, R., & Campos, M. G. (2023). Translational Research on Bee Pollen as a Source of Nutrients: A Scoping Review from Bench to Real World. Nutrients, 15(10), 2413. https://doi.org/10.3390/nu15102413







