Clinical Evidence of the Benefits of Phytonutrients in Human Healthcare
Abstract
1. Introduction
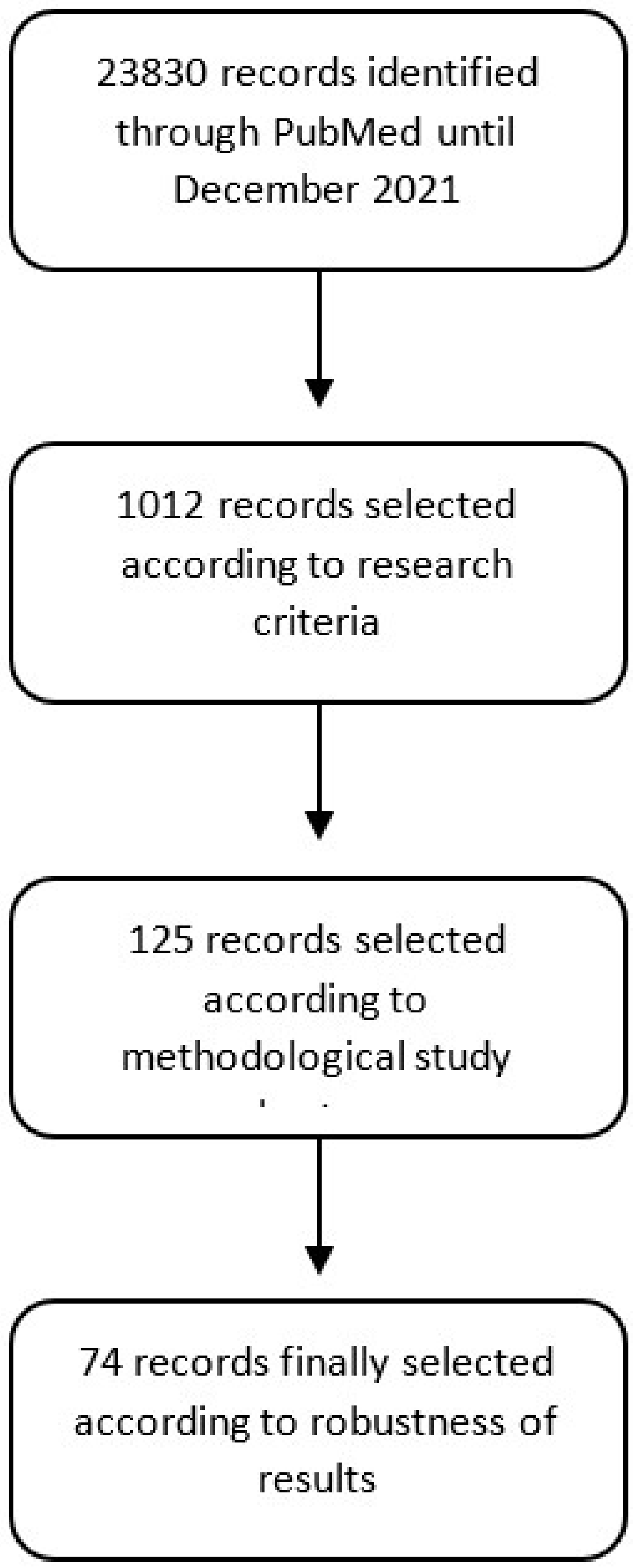
2. Materials and Methods
3. Results
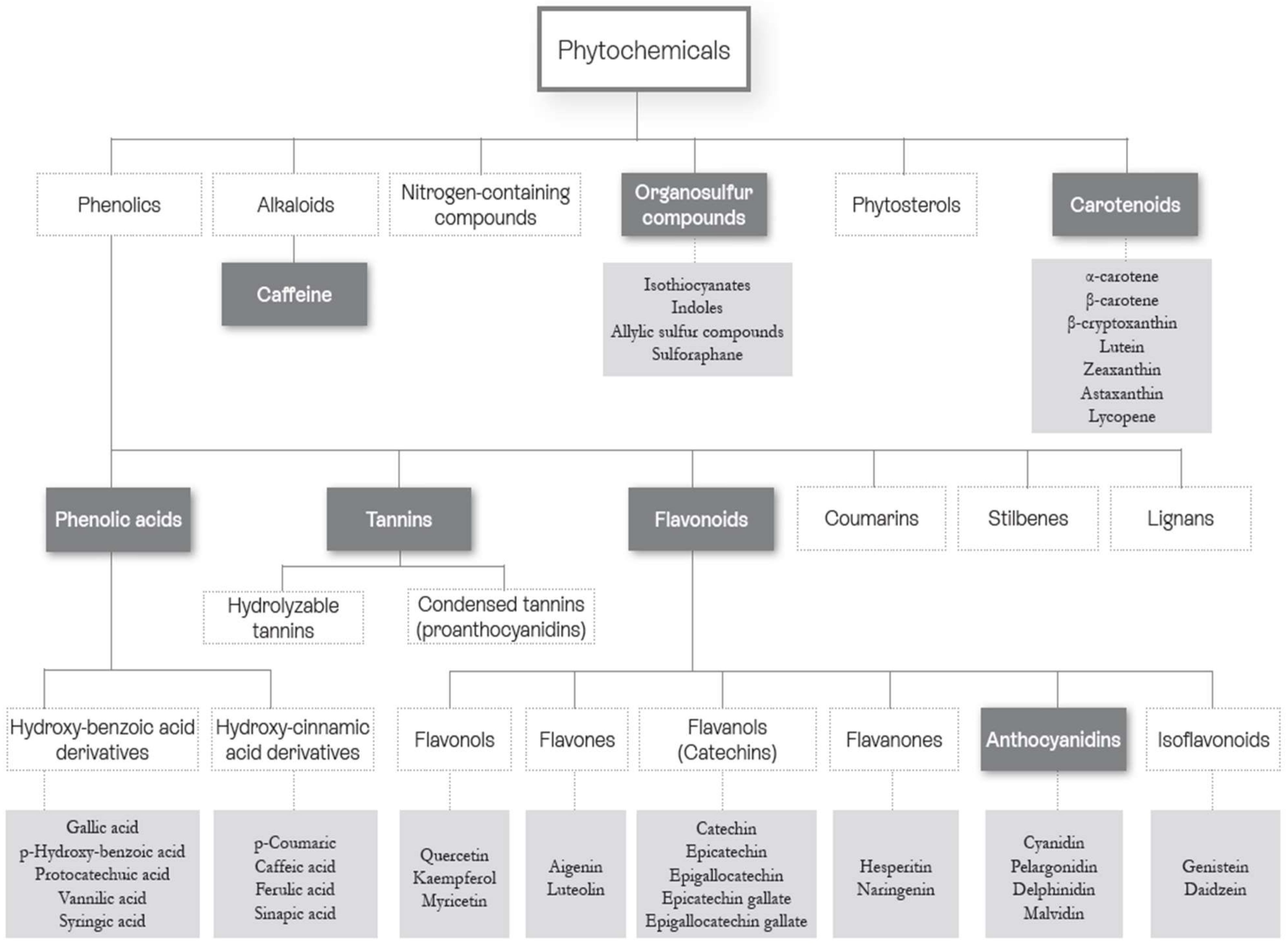
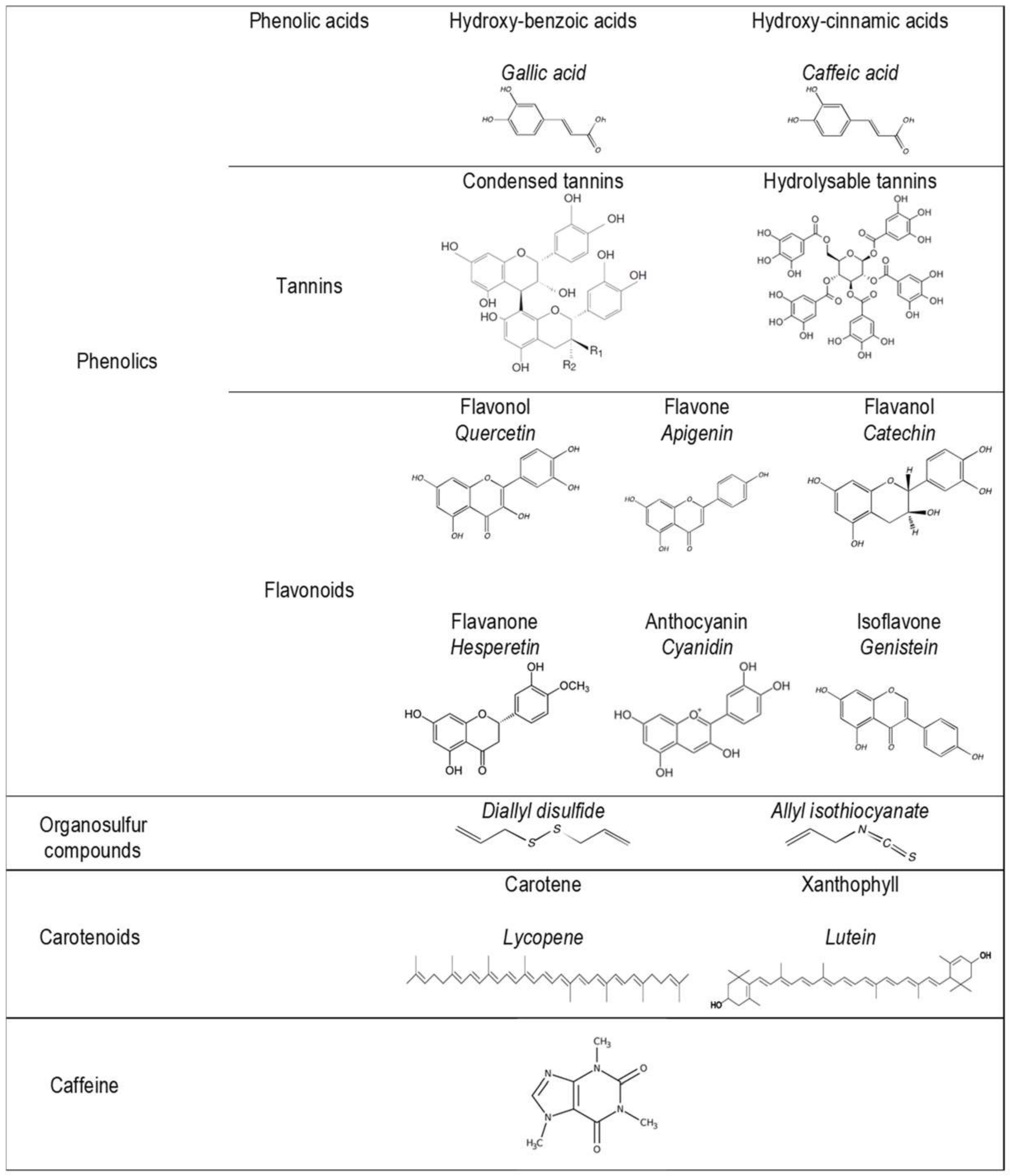
3.1. Phytonutrients
3.1.1. Phenolic Acids
3.1.2. Flavonoids
3.1.3. Anthocyanins
3.1.4. Tannins
3.1.5. Organosulfur Compounds
3.1.6. Carotenoids
3.1.7. Caffeine
3.2. Therapeutic Applications of the Value of Phytonutrients (Family Health)
3.2.1. Stress and Sleep
Stress
Sleep
3.2.2. Immunity and ENT
3.2.3. Digestive Health
3.2.4. Bones and Joints
3.2.5. Energy and Vitality
4. Conclusion and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zaynab, M.; Fatima, M.; Abbas, S.; Sharif, Y.; Umair, M.; Zafar, M.H.; Bahadar, K. Role of secondary metabolites in plant defense against pathogens. Microb. Pathog. 2018, 124, 198–202. [Google Scholar] [CrossRef] [PubMed]
- Slavin, J.L.; Lloyd, B. Health benefits of fruits and vegetables. Adv. Nutr. 2012, 3, 506–516. [Google Scholar] [CrossRef] [PubMed]
- Maggini, S.; Pierre, A.; Calder, P.C. Immune function and micronutrient requirements change over the life course. Nutrients 2018, 10, 1531. [Google Scholar] [CrossRef]
- Poiroux-Gonord, F.; Bidel, L.P.; Fanciullino, A.L.; Gautier, H.; Lauri-Lopez, F.; Urban, L. Health benefits of vitamins and secondary metabolites of fruits and vegetables and prospects to increase their concentrations by agronomic approaches. J. Agric. Food Chem. 2010, 58, 12065–12082. [Google Scholar] [CrossRef]
- Aune, D. Plant foods, antioxidant biomarkers, and the risk of cardiovascular disease, cancer, and mortality: A review of the evidence. Adv. Nutr. 2019, 10 (Suppl. 4), S404–S421. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.H. Health-promoting components of fruits and vegetables in the diet. Adv. Nutr. 2013, 4, 384S–392S. [Google Scholar] [CrossRef] [PubMed]
- Kris-Etherton, P.M.; Hecker, K.D.; Bonanome, A.; Coval, S.M.; Binkoski, A.E.; Hilpert, K.F.; Griel, A.E.; Etherton, T.D. Bioactive compounds in foods: Their role in the prevention of cardiovascular disease and cancer. Am. J. Med. 2002, 113 (Suppl. 9B), 71S–88S. [Google Scholar] [CrossRef]
- Tennant, D.R.; Davidson, J.; Day, A.J. Phytonutrient intakes in relation to European fruit and vegetable consumption patterns observed in different food surveys. Br. J. Nutr. 2014, 112, 1214–1225. [Google Scholar] [CrossRef]
- Amiot, M.J.; Latgé, C.; Plumey, L.; Raynal, S. Intake estimation of phytochemicals in a French well-balanced diet. Nutrients 2021, 13, 3628. [Google Scholar] [CrossRef]
- Jeffery, E.H.; Brown, A.F.; Kurilich, A.C.; Keck, A.S.; Matusheski, N.; Klein, B.P.; Juvik, J.A. Variation in content of bioactive components in broccoli. J. Food Compos. Anal. 2003, 16, 323–330. [Google Scholar] [CrossRef]
- King, A.; Young, G. Characteristics and occurrence of phenolic phytochemicals. J. Am. Diet Assoc. 1999, 99, 213–218. [Google Scholar] [CrossRef]
- Amiot, M.J.; Coxam, V.; Strigler, F. Les Phytomicronutriments, 1st ed.; Lavoisier: Paris, France, 2012; p. 386. [Google Scholar]
- Amiot, M.J.; Riva, C.; Vinet, A. Effects of dietary polyphenols on metabolic syndrome features in humans: A systematic review. Obes. Rev. 2016, 17, 573–586. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Tsao, R. Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. Curr. Opin. Food Sci. 2016, 8, 33–42. [Google Scholar] [CrossRef]
- Khameneh, B.; Iranshahy, M.; Soheili, V.; Fazly Bazzaz, B.S. Review on plant antimicrobials: A mechanistic viewpoint. AntiMicrob. Resist. Infect. Control 2019, 8, 118. [Google Scholar] [CrossRef] [PubMed]
- Sonawane, P.D.; Pollier, J.; Panda, S.; Szymanski, J.; Massalha, H.; Yona, M.; Unger, T.; Malitsky, S.; Arendt, P.; Pauwels, L.; et al. Plant cholesterol biosynthetic pathway overlaps with phytosterol metabolism. Nat. Plants 2016, 3, 16205. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J. Dietary flavonoid aglycones and their glycosides: Which show better biological significance? Crit. Rev. Food Sci. Nutr. 2017, 57, 1874–1905. [Google Scholar] [CrossRef] [PubMed]
- Kaulmann, A.; Bohn, T. Carotenoids, inflammation, and oxidative stress—Implications of cellular signaling pathways and relation to chronic disease prevention. Nutr. Res. 2014, 34, 907–929. [Google Scholar] [CrossRef]
- Dinkova-Kostova, A.T.; Kostov, R.V. Glucosinolates and isothiocyanates in health and disease. Trends Mol. Med. 2012, 18, 337–347. [Google Scholar] [CrossRef]
- Chandrasekara, A. Phenolic Acids. In Encyclopedia of Food Chemistry; Melton, L., Shahidi, F., Varelis, P., Eds.; Academic Press: Oxford, UK, 2019; pp. 535–545. [Google Scholar]
- Liu, R.H. Potential synergy of phytochemicals in cancer prevention: Mechanism of action. J. Nutr. 2004, 134 (Suppl. 12), 3479S–3485S. [Google Scholar] [CrossRef]
- Liu, R.H. Dietary bioactive compounds and their health implications. J. Food Sci. 2013, 78 (Suppl. 1), A18–A25. [Google Scholar] [CrossRef]
- Kumar, N.; Goel, N. Phenolic acids: Natural versatile molecules with promising therapeutic applications. Biotechnol. Rep. 2019, 24, e00370. [Google Scholar] [CrossRef] [PubMed]
- Bruneton, J. Pharmacognosie Phytochimie Plantes Médicinales, 5th ed.; Lavoisier Tec&Doc: Paris, France, 2016; pp. 287–1485. [Google Scholar]
- Banjarnahor, S.D.S.; Artanti, N. Antioxidant properties of flavonoids. Med. J. Indones. 2015, 23, 239–244. [Google Scholar] [CrossRef]
- Gupta, C.; Prakash, D. Phytonutrients as therapeutic agents. J. Complement. Integr. Med. 2014, 11, 151–169. [Google Scholar] [CrossRef] [PubMed]
- Parker, T.L.; Wang, X.H.; Pazmiño, J.; Engeseth, N.J. Antioxidant capacity and phenolic content of grapes, sun-dried raisins, and golden raisins and their effect on ex vivo serum antioxidant capacity. J. Agric. Food Chem. 2007, 55, 8472–8477. [Google Scholar] [CrossRef]
- Lin, B.W.; Gong, C.C.; Song, H.F.; Cui, Y.Y. Effects of anthocyanins on the prevention and treatment of cancer. Br. J. Pharmacol. 2017, 174, 1226–1243. [Google Scholar] [CrossRef]
- Derbel, S.; Ghedira, K. Les phytonutriments et leur impact sur la santé. Phytothérapie 2005, 3, 28–34. [Google Scholar] [CrossRef]
- Scalbert, A.; Williamson, G. Dietary intake and bioavailability of polyphenols. J. Nutr. 2000, 130 (Suppl. S8), 2073S–2085S. [Google Scholar] [CrossRef]
- Vlachojannis, C.; Zimmermann, B.F.; Chrubasik-Hausmann, S. Quantification of anthocyanins in elderberry and chokeberry dietary supplements. Phytother. Res. 2015, 29, 561–565. [Google Scholar] [CrossRef]
- Ghosh, D. Tannins from foods to combat diseases. Int. J. Pharma. Res. Rev. 2015, 4, 40–44. [Google Scholar]
- Chung, K.T.; Wong, T.Y.; Wei, C.I.; Huang, Y.W.; Lin, Y. Tannins and human health: A review. Crit. Rev. Food Sci. Nutr. 1998, 38, 421–464. [Google Scholar] [CrossRef]
- Sugiyama, R.; Hirai, M.Y. Atypical myrosinase as a mediator of glucosinolate functions in plants. Front. Plant Sci. 2019, 10, 1008. [Google Scholar] [CrossRef] [PubMed]
- Fiedor, J.; Burda, K. Potential role of carotenoids as antioxidants in human health and disease. Nutrients 2014, 6, 466–488. [Google Scholar] [CrossRef] [PubMed]
- Van der Sluis, A.A.; Dekker, M.; de Jager, A.; Jongen, W.M. Activity and concentration of polyphenolic antioxidants in apple: Effect of cultivar, harvest year, and storage conditions. J. Agric. Food Chem. 2001, 49, 3606–3613. [Google Scholar] [CrossRef] [PubMed]
- Bohn, T. Carotenoids and markers of oxidative stress in human observational studies and intervention trials: Implications for chronic diseases. Antioxidants 2019, 8, 179. [Google Scholar] [CrossRef] [PubMed]
- Niroula, A.; Khatri, S.; Timilsina, R.; Khadka, D.; Khadka, A.; Ojha, P. Profile of chlorophylls and carotenoids of wheat (Triticum aestivum L.) and barley (Hordeum vulgare L.) microgreens. J. Food Sci. Technol. 2019, 56, 2758–2763. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.L.; Norhaizan, M.E. Carotenoids: How effective are they to prevent age-related diseases? Molecules 2019, 24, 1801. [Google Scholar] [CrossRef]
- Ashihara, H.; Sano, H.; Crozier, A. Caffeine and related purine alkaloids: Biosynthesis, catabolism, function and genetic engineering. Phytochemistry 2008, 69, 841–856. [Google Scholar] [CrossRef]
- Actualisation des Repères du PNNS: ÉTude des Relations Entre Consommation de Groupes d’aliments et Risque de Maladies Chroniques Non Transmissibles. November 2016. Available online: https://www.researchgate.net/publication/312665902_Actualisation_des_reperes_du_PNNS_etude_des_relations_entre_consommation_de_groupes_d’aliments_et_risque_de_maladies_chroniques_non_transmissibles (accessed on 1 March 2022).
- Heckman, M.A.; Weil, J.; Gonzalez de Mejia, E. Caffeine (1,3,7-trimethylxanthine) in foods: A comprehensive review on consumption, functionality, safety, and regulatory matters. J. Food Sci. 2010, 75, R77–R87. [Google Scholar] [CrossRef]
- Stringham, N.T.; Holmes, P.V.; Stringham, J.M. Supplementation with macular carotenoids reduces psychological stress, serum cortisol, and sub-optimal symptoms of physical and emotional health in young adults. Nutr. Neurosci. 2018, 21, 286–296. [Google Scholar] [CrossRef]
- Scholey, A.; Downey, L.A.; Ciorciari, J.; Pipingas, A.; Nolidin, K.; Finn, M.; Wines, M.; Catchlove, S.; Terrens, A.; Barlow, E.; et al. Acute neurocognitive effects of epigallocatechin gallate (EGCG). Appetite 2012, 58, 767–770. [Google Scholar] [CrossRef]
- Kell, G.; Rao, A.; Beccaria, G.; Clayton, P.; Inarejos-García, A.M.; Prodanov, M. Affron® a novel saffron extract (Crocus sativus L.) improves mood in healthy adults over 4 weeks in a double-blind, parallel, randomized, placebo-controlled clinical trial. Complement. Ther. Med. 2017, 33, 58–64. [Google Scholar] [CrossRef] [PubMed]
- White, B.C.; Lincoln, C.A.; Pearce, N.W.; Reeb, R.; Vaida, C. Anxiety and muscle tension as consequences of caffeine withdrawal. Science 1980, 209, 1547–1548. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, G.A.; Carroll, M.E.; Crosby, R.D.; Perwien, A.R.; Go, F.S.; Benowitz, N.L. Caffeine effects on learning, performance, and anxiety in normal school-age children. J. Am. Acad. Child Adolesc. Psychiatry 1994, 33, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Micek, A.; Castellano, S.; Pajak, A.; Galvano, F. Coffee, tea, caffeine and risk of depression: A systematic review and dose-response meta-analysis of observational studies. Mol. Nutr. Food Res. 2016, 60, 223–234. [Google Scholar] [CrossRef]
- Terauchi, M.; Horiguchi, N.; Kajiyama, A.; Akiyoshi, M.; Owa, Y.; Kato, K.; Kubota, T. Effects of grape seed proanthocyanidin extract on menopausal symptoms, body composition, and cardiovascular parameters in middle-aged women: A randomized, double-blind, placebo-controlled pilot study. Menopause 2014, 21, 990–996. [Google Scholar] [CrossRef]
- Umigai, N.; Takeda, R.; Mori, A. Effect of crocetin on quality of sleep: A randomized, double-blind, placebo-controlled, crossover study. Complement. Ther. Med. 2018, 41, 47–51. [Google Scholar] [CrossRef]
- Hachul, H.; Brandão, L.C.; D’Almeida, V.; Bittencourt, L.R.; Baracat, E.C.; Tufik, S. Isoflavones decrease insomnia in postmenopause. Menopause 2011, 18, 178–184. [Google Scholar] [CrossRef]
- Losso, J.N.; Finley, J.W.; Karki, N.; Liu, A.G.; Prudente, A.; Tipton, R.; Yu, Y.; Greenway, F.L. Pilot study of the tart cherry juice for the treatment of insomnia and investigation of mechanisms. Am. J. Ther. 2018, 25, e194–e201. [Google Scholar] [CrossRef]
- Kuratsune, H.; Umigai, N.; Takeno, R.; Kajimoto, Y.; Nakano, T. Effect of crocetin from Gardenia jasminoides Ellis on sleep: A pilot study. Phytomedicine 2010, 17, 840–843. [Google Scholar] [CrossRef]
- Valko, M.; Izakovic, M.; Mazur, M.; Rhodes, C.J.; Telser, J. Role of oxygen radicals in DNA damage and cancer incidence. Mol. Cell. Biochem. 2004, 266, 37–56. [Google Scholar] [CrossRef]
- Stefanello, N.; Spanevello, R.M.; Passamonti, S.; Porciúncula, L.; Bonan, C.D.; Olabiyi, A.A.; Teixeira da Rocha, J.B.; Assmann, C.E.; Morsch, V.M.; Schetinger, M.R.C. Coffee, caffeine, chlorogenic acid, and the purinergic system. Food Chem. Toxicol. 2019, 123, 298–313. [Google Scholar] [CrossRef] [PubMed]
- Yahfoufi, N.; Alsadi, N.; Jambi, M.; Matar, C. The immunomodulatory and anti-inflammatory role of polyphenols. Nutrients 2018, 10, 1618. [Google Scholar] [CrossRef] [PubMed]
- Vrolijk, M.F.; Opperhuizen, A.; Jansen, E.H.; Godschalk, R.W.; Van Schooten, F.J.; Bast, A.; Haenen, G.R. The shifting perception on antioxidants: The case of vitamin E and β-carotene. Redox Biol. 2015, 4, 272–278. [Google Scholar] [CrossRef]
- Cos, P.; De Bruyne, T.; Hermans, N.; Apers, S.; Berghe, D.V.; Vlietinck, A.J. Proanthocyanidins in health care: Current and new trends. Curr. Med. Chem. 2004, 11, 1345–1359. [Google Scholar] [CrossRef] [PubMed]
- Van Acker, S.A.; van den Berg, D.J.; Tromp, M.N.; Griffioen, D.H.; van Bennekom, W.P.; van der Vijgh, W.J.; Bast, A. Structural aspects of antioxidant activity of flavonoids. Free Radic. Biol. Med. 1996, 20, 331–342. [Google Scholar] [CrossRef]
- Masuda, S.; Maeda-Yamamoto, M.; Usui, S.; Fujisawa, T. ‘Benifuuki’ green tea containing O-methylated catechin reduces symptoms of Japanese Cedar Pollinosis: A randomized, double-blind, placebo-controlled trial. Allergol. Int. 2014, 63, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Enomoto, T.; Nagasako-Akazome, Y.; Kanda, T.; Ikeda, M.; Dake, Y. Clinical effects of apple polyphenols on persistent allergic rhinitis: A randomized double-blind placebo-controlled parallel arm study. J. Investig. Allergol. Clin. Immunol. 2006, 16, 283–289. [Google Scholar] [PubMed]
- Matsumoto, K.; Yamada, H.; Takuma, N.; Niino, H.; Sagesaka, Y.M. Effects of green tea catechins and theanine on preventing influenza infection among healthcare workers: A randomized controlled trial. BMC Complement. Altern. Med. 2011, 11, 15. [Google Scholar] [CrossRef]
- Muller, L.; Meyer, M.; Bauer, R.N.; Zhou, H.; Zhang, H.; Jones, S.; Robinette, C.; Noah, T.L.; Jaspers, I. Effect of broccoli sprouts and live attenuated influenza virus on peripheral blood natural killer cells: A randomized, double-blind study. PLoS ONE 2016, 11, e0147742. [Google Scholar] [CrossRef]
- Nantz, M.P.; Rowe, C.A.; Muller, C.; Creasy, R.; Colee, J.; Khoo, C.; Percival, S.S. Consumption of cranberry polyphenols enhances human γδ-T cell proliferation and reduces the number of symptoms associated with colds and influenza: A randomized, placebo-controlled intervention study. Nutr. J. 2013, 12, 161. [Google Scholar] [CrossRef]
- Crişan, I.; Zaharia, C.N.; Popovici, F.; Jucu, V.; Belu, O.; Dascălu, C.; Mutiu, A.; Petrescu, A. Natural propolis extract NIVCRISOL in the treatment of acute and chronic rhinopharyngitis in children. Rom. J. Virol. 1995, 46, 115–133. [Google Scholar] [PubMed]
- Welsh, E.J.; Bara, A.; Barley, E.; Cates, C.J. Caffeine for asthma. Cochrane Database Syst. Rev. 2010, 2010, CD001112. [Google Scholar] [CrossRef] [PubMed]
- Nourollahian, M.; Rasoulian, B.; Gafari, A.; Anoushiravani, M.; Jabari, F.; Bakhshaee, M. Clinical comparison of the efficacy of spirulina platensis and cetirizine for treatment of allergic rhinitis. Acta Otorhinolaryngol. Ital. 2020, 40, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Cingi, C.; Conk-Dalay, M.; Cakli, H.; Bal, C. The effects of spirulina on allergic rhinitis. Eur. Arch. Otorhinolaryngol. 2008, 265, 1219–1223. [Google Scholar] [CrossRef]
- Cui, Y.; Oh, Y.J.; Lim, J.; Youn, M.; Lee, I.; Pak, H.K.; Park, W.; Jo, W.; Park, S. AFM study of the differential inhibitory effects of the green tea polyphenol (-)-epigallocatechin-3-gallate (EGCG) against Gram-positive and Gram-negative bacteria. Food Microbiol. 2012, 29, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Yoda, Y.; Hu, Z.Q.; Zhao, W.H.; Shimamura, T. Different susceptibilities of Staphylococcus and Gram-negative rods to epigallocatechin gallate. J. Infect. Chemother. 2004, 10, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Oneda, H.; Shiihara, M.; Inouye, K. Inhibitory effects of green tea catechins on the activity of human matrix metalloproteinase 7 (matrilysin). J. Biochem. 2003, 133, 571–576. [Google Scholar] [CrossRef]
- Yi, S.M.; Zhu, J.L.; Fu, L.L.; Li, J.R. Tea polyphenols inhibit Pseudomonas aeruginosa through damage to the cell membrane. Int. J. Food Microbiol. 2010, 144, 111–117. [Google Scholar] [CrossRef]
- Chen, C.C.; Huang, C.Y. Inhibition of Klebsiella pneumoniae DnaB helicase by the flavonol galangin. Protein J. 2011, 30, 59–65. [Google Scholar] [CrossRef]
- Huang, Y.H.; Huang, C.C.; Chen, C.C.; Yang, K.J.; Huang, C.Y. Inhibition of Staphylococcus aureus PriA helicase by flavonol kaempferol. Protein J. 2015, 34, 169–172. [Google Scholar] [CrossRef]
- Rauf, A.; Imran, M.; Abu-Izneid, T.; Iahtisham-Ul-Haq; Patel, S.; Pan, X.; Naz, S.; Sanches Silva, A.; Saeed, F.; Rasul Suleria, H.A. Proanthocyanidins: A comprehensive review. Biomed. Pharmacother. 2019, 116, 108999. [Google Scholar] [CrossRef]
- Anantharaju, P.G.; Gowda, P.C.; Vimalambike, M.G.; Madhunapantula, S.V. An overview on the role of dietary phenolics for the treatment of cancers. Nutr. J. 2016, 15, 99. [Google Scholar] [CrossRef] [PubMed]
- De Stefano, D.; Maiuri, M.C.; Simeon, V.; Grassia, G.; Soscia, A.; Cinelli, M.P.; Carnuccio, R. Lycopene, quercetin and tyrosol prevent macrophage activation induced by gliadin and IFN-gamma. Eur. J. Pharmacol. 2007, 566, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Patel, B.; Mann, G.E.; Chapple, S.J. Concerted redox modulation by sulforaphane alleviates diabetes and cardiometabolic syndrome. Free Radic. Biol. Med. 2018, 122, 150–160. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.S.; Fan, Y.E.; Lin, C.Y.; Hu, M.L. Lycopene inhibits matrix metalloproteinase-9 expression and down-regulates the binding activity of nuclear factor-kappa B and stimulatory protein-1. J. Nutr. Biochem. 2007, 18, 449–456. [Google Scholar] [CrossRef]
- Chen, C.C.; Chow, M.P.; Huang, W.C.; Lin, Y.C.; Chang, Y.J. Flavonoids inhibit tumor necrosis factor-alpha-induced up-regulation of intercellular adhesion molecule-1 (ICAM-1) in respiratory epithelial cells through activator protein-1 and nuclear factor-kappaB: Structure-activity relationships. Mol. Pharmacol. 2004, 66, 683–693. [Google Scholar] [CrossRef] [PubMed]
- Yamagishi, S.; Matsui, T. Protective role of sulphoraphane against vascular complications in diabetes. Pharm. Biol. 2016, 54, 2329–2339. [Google Scholar] [CrossRef] [PubMed]
- Sung, S.; Kwon, D.; Um, E.; Kim, B. Could polyphenols help in the control of rheumatoid arthritis? Molecules 2019, 24, 1589. [Google Scholar] [CrossRef]
- Zhu, R.; Chen, B.; Bai, Y.; Miao, T.; Rui, L.; Zhang, H.; Xia, B.; Li, Y.; Gao, S.; Wang, X.D.; et al. Lycopene in protection against obesity and diabetes: A mechanistic review. Pharmacol. Res. 2020, 159, 104966. [Google Scholar] [CrossRef]
- Martinez-Micaelo, N.; González-Abuín, N.; Ardèvol, A.; Pinent, M.; Blay, M.T. Procyanidins and inflammation: Molecular targets and health implications. Biofactors 2012, 38, 257–265. [Google Scholar] [CrossRef]
- Baek, H.I.; Ha, K.C.; Kim, H.M.; Choi, E.K.; Park, E.O.; Park, B.H.; Yang, H.J.; Kim, M.J.; Kang, H.J.; Chae, S.W. Randomized, double-blind, placebo-controlled trial of Ficus carica paste for the management of functional constipation. Asia Pac. J. Clin. Nutr. 2016, 25, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Ingersoll, G.L.; Wasilewski, A.; Haller, M.; Pandya, K.; Bennett, J.; He, H.; Hoffmire, C.; Berry, C. Effect of concord grape juice on chemotherapy-induced nausea and vomiting: Results of a pilot study. Oncol. Nurs. Forum. 2010, 37, 213–221. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dryden, G.W.; Lam, A.; Beatty, K.; Qazzaz, H.H.; McClain, C.J. A pilot study to evaluate the safety and efficacy of an oral dose of (-)-epigallocatechin-3-gallate-rich polyphenon E in patients with mild to moderate ulcerative colitis. Inflamm. Bowel Dis. 2013, 19, 1904–1912. [Google Scholar] [CrossRef] [PubMed]
- Barsalani, R.; Riesco, E.; Lavoie, J.M.; Dionne, I.J. Effect of exercise training and isoflavones on hepatic steatosis in overweight postmenopausal women. Climacteric 2013, 16, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Cheraghpour, M.; Imani, H.; Ommi, S.; Alavian, S.M.; Karimi-Shahrbabak, E.; Hedayati, M.; Yari, Z.; Hekmatdoost, A. Hesperidin improves hepatic steatosis, hepatic enzymes, and metabolic and inflammatory parameters in patients with nonalcoholic fatty liver disease: A randomized, placebo-controlled, double-blind clinical trial. Phytother. Res. 2019, 33, 2118–2125. [Google Scholar] [CrossRef]
- Venancio, V.P.; Kim, H.; Sirven, M.A.; Tekwe, C.D.; Honvoh, G.; Talcott, S.T.; Mertens-Talcott, S.U. Polyphenol-rich mango (Mangifera indica L.) ameliorate functional constipation symptoms in humans beyond equivalent amount of fiber. Mol. Nutr. Food Res. 2018, 62, e1701034. [Google Scholar] [CrossRef]
- Mangel, A.W.; Chaturvedi, P. Evaluation of crofelemer in the treatment of diarrhea-predominant irritable bowel syndrome patients. Digestion 2008, 78, 180–186. [Google Scholar] [CrossRef]
- Kikuchi, M.; Ushida, Y.; Shiozawa, H.; Umeda, R.; Tsuruya, K.; Aoki, Y.; Suganuma, H.; Nishizaki, Y. Sulforaphane-rich broccoli sprout extract improves hepatic abnormalities in male subjects. World J. Gastroenterol. 2015, 21, 12457–12467. [Google Scholar] [CrossRef]
- Yanaka, A. Daily intake of broccoli sprouts normalizes bowel habits in human healthy subjects. J. Clin. Biochem. Nutr. 2018, 62, 75–82. [Google Scholar] [CrossRef]
- Kaczmarek, J.L.; Liu, X.; Charron, C.S.; Novotny, J.A.; Jeffery, E.H.; Seifried, H.E.; Ross, S.A.; Miller, M.J.; Swanson, K.S.; Holscher, H.D. Broccoli consumption affects the human gastrointestinal microbiota. J. Nutr. Biochem. 2019, 63, 27–34. [Google Scholar] [CrossRef]
- Biedermann, L.; Mwinyi, J.; Scharl, M.; Frei, P.; Zeitz, J.; Kullak-Ublick, G.A.; Vavricka, S.R.; Fried, M.; Weber, A.; Humpf, H.U.; et al. Bilberry ingestion improves disease activity in mild to moderate ulcerative colitis—An open pilot study. J. Crohns. Colitis 2013, 7, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Ruhl, C.E.; Everhart, J.E. Coffee and tea consumption are associated with a lower incidence of chronic liver disease in the United States. Gastroenterology 2005, 129, 1928–1936. [Google Scholar] [CrossRef] [PubMed]
- Ambrosini, G.L.; Bremner, A.P.; Reid, A.; Mackerras, D.; Alfonso, H.; Olsen, N.J.; Musk, A.W.; de Klerk, N.H. No dose-dependent increase in fracture risk after long-term exposure to high doses of retinol or beta-carotene. Osteoporos Int. 2013, 24, 1285–1293. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.M.; Huang, C.Q.; Lin, Z.K.; Tian, N.F.; Ni, W.F.; Wang, X.Y.; Xu, H.Z.; Chi, Y.L. The relationship between vitamin A and risk of fracture: Meta-analysis of prospective studies. J. Bone Miner. Res. 2014, 29, 2032–2039. [Google Scholar] [CrossRef]
- Lee, D.R.; Lee, J.; Rota, M.; Lee, J.; Ahn, H.S.; Park, S.M.; Shin, D. Coffee consumption and risk of fractures: A systematic review and dose-response meta-analysis. Bone 2014, 63, 20–28. [Google Scholar] [CrossRef]
- Connelly, A.E.; Tucker, A.J.; Tulk, H.; Catapang, M.; Chapman, L.; Sheikh, N.; Yurchenko, S.; Fletcher, R.; Kott, L.S.; Duncan, A.M.; et al. High-rosmarinic acid spearmint tea in the management of knee osteoarthritis symptoms. J. Med. Food 2014, 17, 1361–1367. [Google Scholar] [CrossRef]
- Law, Y.Y.; Chiu, H.F.; Lee, H.H.; Shen, Y.C.; Venkatakrishnan, K.; Wang, C.K. Consumption of onion juice modulates oxidative stress and attenuates the risk of bone disorders in middle-aged and post-menopausal healthy subjects. Food Funct. 2016, 7, 902–912. [Google Scholar] [CrossRef]
- Hu, Y.; Cui, J.; Sparks, J.A.; Malspeis, S.; Costenbader, K.H.; Karlson, E.W.; Lu, B. Circulating carotenoids and subsequent risk of rheumatoid arthritis in women. Clin. Exp. Rheumatol. 2017, 35, 309–312. [Google Scholar]
- Javadi, F.; Ahmadzadeh, A.; Eghtesadi, S.; Aryaeian, N.; Zabihiyeganeh, M.; Rahimi Foroushani, A.; Jazayeri, S. The effect of quercetin on inflammatory factors and clinical symptoms in women with rheumatoid arthritis: A double-blind, randomized controlled trial. J. Am. Coll. Nutr. 2017, 36, 9–15. [Google Scholar] [CrossRef]
- Hosseinzadeh-Attar, M.J.; Alipoor, E.; Dehghani, S.; Salimzadeh, A. Increased efficacy of a garlic supplement on knee osteoarthritis symptoms in patients with obesity. J. Herb. Med. 2020, 24, 100392. [Google Scholar] [CrossRef]
- Kim, D.E.; Cho, S.H.; Park, H.M.; Chang, Y.K. Relationship between bone mineral density and dietary intake of β-carotene, vitamin C, zinc and vegetables in postmenopausal Korean women: A cross-sectional study. J. Int. Med. Res. 2016, 44, 1103–1114. [Google Scholar] [CrossRef] [PubMed]
- Lambert, M.N.T.; Hu, L.M.; Jeppesen, P.B. A systematic review and meta-analysis of the effects of isoflavone formulations against estrogen-deficient bone resorption in peri- and postmenopausal women. Am. J. Clin. Nutr. 2017, 106, 801–811. [Google Scholar] [CrossRef] [PubMed]
- Li, X.L.; Xu, J.H. Coffee consumption and hip fracture risk: A meta-analysis. J. Nutr. Sci. 2013, 2, e23. [Google Scholar] [CrossRef] [PubMed]
- Gambacciani, M.; Ciaponi, M.; Cappagli, B.; Piaggesi, L.; Genazzani, A.R. Effects of combined low dose of the isoflavone derivative ipriflavone and estrogen replacement on bone mineral density and metabolism in postmenopausal women. Maturitas 1997, 28, 75–81. [Google Scholar] [CrossRef]
- Ambrosini, G.L.; Alfonso, H.; Reid, A.; Mackerras, D.; Bremner, A.P.; Beilby, J.; Olsen, N.J.; Musk, A.W.; de Klerk, N.H. Plasma retinol and total carotenes and fracture risk after long-term supplementation with high doses of retinol. Nutrition 2014, 30, 551–556. [Google Scholar] [CrossRef] [PubMed]
- Wetmore, C.M.; Ichikawa, L.; LaCroix, A.Z.; Ott, S.M.; Scholes, D. Association between caffeine intake and bone mass among young women: Potential effect modification by depot medroxyprogesterone acetate use. Osteoporos. Int. 2008, 19, 519–527. [Google Scholar] [CrossRef]
- Pattison, D.J.; Symmons, D.P.; Lunt, M.; Welch, A.; Bingham, S.A.; Day, N.E.; Silman, A.J. Dietary beta-cryptoxanthin and inflammatory polyarthritis: Results from a population-based prospective study. Am. J. Clin. Nutr. 2005, 82, 451–455. [Google Scholar] [CrossRef]
- Rejnmark, L.; Vestergaard, P.; Charles, P.; Hermann, A.P.; Brot, C.; Eiken, P.; Mosekilde, L. No effect of vitamin A intake on bone mineral density and fracture risk in perimenopausal women. Osteoporos. Int. 2004, 15, 872–880. [Google Scholar] [CrossRef]
- Wattanathorn, J.; Somboonporn, W.; Sungkamanee, S.; Thukummee, W.; Muchimapura, S. A double-blind placebo-controlled randomized trial evaluating the effect of polyphenol-rich herbal congee on bone turnover markers of the perimenopausal and menopausal women. Oxid. Med. Cell Longev. 2018, 2018, 2091872. [Google Scholar] [CrossRef]
- Li, S.; Dai, Z.; Wu, Q. Effect of coffee intake on hip fracture: A meta-analysis of prospective cohort studies. Nutr. J. 2015, 14, 38. [Google Scholar] [CrossRef]
- Borota, D.; Murray, E.; Keceli, G.; Chang, A.; Watabe, J.M.; Ly, M.; Toscano, J.P.; Yassa, M.A. Post-study caffeine administration enhances memory consolidation in humans. Nat. Neurosci. 2014, 17, 201–203. [Google Scholar] [CrossRef] [PubMed]
- Carvalho-Peixoto, J.; Moura, M.R.; Cunha, F.A.; Lollo, P.C.; Monteiro, W.D.; Carvalho, L.M.; Farinatti Pde, T. Consumption of açai (Euterpe oleracea Mart.) functional beverage reduces muscle stress and improves effort tolerance in elite athletes: A randomized controlled intervention study. Appl. Physiol. Nutzr. Metab. 2015, 40, 725–733. [Google Scholar] [CrossRef] [PubMed]
- Alharbi, M.H.; Lamport, D.J.; Dodd, G.F.; Saunders, C.; Harkness, L.; Butler, L.T.; Spencer, J.P. Flavonoid-rich orange juice is associated with acute improvements in cognitive function in healthy middle-aged males. Eur. J. Nutr. 2016, 55, 2021–2029. [Google Scholar] [CrossRef] [PubMed]
- Bazzucchi, I.; Patrizio, F.; Ceci, R.; Duranti, G.; Sgrò, P.; Sabatini, S.; Di Luigi, L.; Sacchetti, M.; Felici, F. The effects of quercetin supplementation on eccentric exercise-induced muscle damage. Nutrients 2019, 11, 205. [Google Scholar] [CrossRef]
- Saitou, K.; Ochiai, R.; Kozuma, K.; Sato, H.; Koikeda, T.; Osaki, N.; Katsuragi, Y. Effect of chlorogenic acids on cognitive function: A randomized, double-blind, placebo-controlled trial. Nutrients 2018, 10, 1337. [Google Scholar] [CrossRef]
- Bowtell, J.L.; Aboo-Bakkar, Z.; Conway, M.E.; Adlam, A.R.; Fulford, J. Enhanced task-related brain activation and resting perfusion in healthy older adults after chronic blueberry supplementation. Appl. Physiol. Nutr. Metab. 2017, 42, 773–779. [Google Scholar] [CrossRef]
- Kesse-Guyot, E.; Andreeva, V.A.; Ducros, V.; Jeandel, C.; Julia, C.; Hercberg, S.; Galan, P. Carotenoid-rich dietary patterns during midlife and subsequent cognitive function. Br. J. Nutr. 2014, 111, 915–923. [Google Scholar] [CrossRef]
- Cook, M.D.; Myers, S.D.; Gault, M.L.; Willems, M.E.T. Blackcurrant alters physiological responses and femoral artery diameter during sustained isometric contraction. Nutrients 2017, 9, 556. [Google Scholar] [CrossRef]
- Falcone, P.H.; Tribby, A.C.; Vogel, R.M.; Joy, J.M.; Moon, J.R.; Slayton, C.A.; Henigman, M.M.; Lasrado, J.A.; Lewis, B.J.; Fonseca, B.A.; et al. Efficacy of a nootropic spearmint extract on reactive agility: A randomized, double-blind, placebo-controlled, parallel trial. J. Int. Soc. Sports Nutr. 2018, 15, 58. [Google Scholar] [CrossRef]
- Gratton, G.; Weaver, S.R.; Burley, C.V.; Low, K.A.; Maclin, E.L.; Johns, P.W.; Pham, Q.S.; Lucas, S.J.E.; Fabiani, M.; Rendeiro, C. Dietary flavanols improve cerebral cortical oxygenation and cognition in healthy adults. Sci. Rep. 2020, 10, 19409. [Google Scholar] [CrossRef]
- Grgic, J.; Trexler, E.T.; Lazinica, B.; Pedisic, Z. Effects of caffeine intake on muscle strength and power: A systematic review and meta-analysis. J. Int. Soc. Sports Nutr. 2018, 15, 11. [Google Scholar] [CrossRef] [PubMed]
- Haskell-Ramsay, C.F.; Jackson, P.A.; Forster, J.S.; Dodd, F.L.; Bowerbank, S.L.; Kennedy, D.O. The acute effects of caffeinated black coffee on cognition and mood in healthy young and older adults. Nutrients 2018, 10, 1386. [Google Scholar] [CrossRef] [PubMed]
- Imai, A.; Oda, Y.; Ito, N.; Seki, S.; Nakagawa, K.; Miyazawa, T.; Ueda, F. Effects of dietary supplementation of Astaxanthin and Sesamin on daily fatigue: A randomized, double-blind, placebo-controlled, two-way crossover study. Nutrients 2018, 10, 281. [Google Scholar] [CrossRef] [PubMed]
- Pilaczynska-Szczesniak, L.; Skarpanska-Steinborn, A.; Deskur, E.; Basta, P.; Horoszkiewicz-Hassan, M. The influence of chokeberry juice supplementation on the reduction of oxidative stress resulting from an incremental rowing ergometer exercise. Int. J. Sport Nutr. Exerc. Metab. 2005, 15, 48–58. [Google Scholar] [CrossRef] [PubMed]
- Lamport, D.J.; Lawton, C.L.; Merat, N.; Jamson, H.; Myrissa, K.; Hofman, D.; Chadwick, H.K.; Quadt, F.; Wightman, J.D.; Dye, L. Concord grape juice, cognitive function, and driving performance: A 12-wk, placebo-controlled, randomized crossover trial in mothers of preteen children. Am. J. Clin. Nutr. 2016, 103, 775–783. [Google Scholar] [CrossRef]
- Mastroiacovo, D.; Kwik-Uribe, C.; Grassi, D.; Necozione, S.; Raffaele, A.; Pistacchio, L.; Righetti, R.; Bocale, R.; Lechiara, M.C.; Marini, C.; et al. Cocoa flavanol consumption improves cognitive function, blood pressure control, and metabolic profile in elderly subjects: The Cocoa, Cognition, and Aging (CoCoA) Study—A randomized controlled trial. Am. J. Clin Nutr. 2015, 101, 538–548. [Google Scholar] [CrossRef]
- Duvnjak-Zaknich, D.M.; Dawson, B.T.; Wallman, K.E.; Henry, G. Effect of caffeine on reactive agility time when fresh and fatigued. Med. Sci. Sports Exerc. 2011, 43, 1523–1530. [Google Scholar] [CrossRef]
- Trombold, J.R.; Barnes, J.N.; Critchley, L.; Coyle, E.F. Ellagitannin consumption improves strength recovery 2-3 d after eccentric exercise. Med. Sci. Sports Exerc. 2010, 42, 493–498. [Google Scholar] [CrossRef]
- Whyte, A.R.; Williams, C.M. Effects of a single dose of a flavonoid-rich blueberry drink on memory in 8 to 10 y old children. Nutrition 2015, 31, 531–534. [Google Scholar] [CrossRef]
- Falcone, P.H.; Nieman, K.M.; Tribby, A.C.; Vogel, R.M.; Joy, J.M.; Moon, J.R.; Slayton, C.A.; Henigman, M.M.; Lasrado, J.A.; Lewis, B.J.; et al. The attention-enhancing effects of spearmint extract supplementation in healthy men and women: A randomized, double-blind, placebo-controlled, parallel trial. Nutr. Res. 2019, 64, 24–38. [Google Scholar] [CrossRef]
- Johnson, E.J.; McDonald, K.; Caldarella, S.M.; Chung, H.Y.; Troen, A.M.; Snodderly, D.M. Cognitive findings of an exploratory trial of docosahexaenoic acid and lutein supplementation in older women. Nutr. Neurosci. 2008, 11, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Ataka, S.; Tanaka, M.; Nozaki, S.; Mizuma, H.; Mizuno, K.; Tahara, T.; Sugino, T.; Shirai, T.; Kajimoto, Y.; Kuratsune, H.; et al. Effects of Applephenon and ascorbic acid on physical fatigue. Nutrition 2007, 23, 419–423. [Google Scholar] [CrossRef]
- Do Rosario, V.A.; Fitzgerald, Z.; Broyd, S.; Paterson, A.; Roodenrys, S.; Thomas, S.; Bliokas, V.; Potter, J.; Walton, K.; Weston-Green, K.; et al. Food anthocyanins decrease concentrations of TNF-α in older adults with mild cognitive impairment: A randomized, controlled, double blind clinical trial. Nutr. Metab. Cardiovasc. Dis. 2021, 31, 950–960. [Google Scholar] [CrossRef] [PubMed]
- Calapai, G.; Bonina, F.; Bonina, A.; Rizza, L.; Mannucci, C.; Arcoraci, V.; Laganà, G.; Alibrandi, A.; Pollicino, C.; Inferrera, S.; et al. A randomized, double-blinded, clinical trial on effects of a Vitis vinifera extract on cognitive function in healthy older adults. Front Pharmacol. 2017, 8, 776. [Google Scholar] [CrossRef] [PubMed]
- Caesar, L.K.; Cech, N.B. Synergy and antagonism in natural product extracts: When 1 + 1 does not equal 2. Nat. Prod Rep. 2019, 36, 869–888. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.I.; Apostolidis, E.; Kim, Y.C.; Shetty, K. Health benefits of traditional corn, beans, and pumpkin: In vitro studies for hyperglycemia and hypertension management. J. Med. Food. 2007, 10, 266–275. [Google Scholar] [CrossRef] [PubMed]
| Therapeutic Area | Key Words |
|---|---|
| Bones and joints | Joint, rheumatism, osteoarthritis, arthritis, arthrosis, tendonitis, tendinitis, bone mineral density, bone mineral turnover |
| Digestive health | Abdominal bloating, gut health, constipation, diarrhea, chronic diarrhea, nausea, hepatic crisis, hepatic steatosis, irritable bowel syndrome, inflammatory bowel, syndrome, irritable colon, colitis |
| Immunity and ENT diseases | Chronic rhinitis, allergic rhinitis, rhinopharyngitis, sinusitis antritis, chronic sinusitis, sinus infection, non-productive cough, dry cough, chronic cough, sore throat, pharyngitis, angina, aphonia, hoarseness, seasonal allergy, allergic and pollen, influenza, flu, immunodeficiency, immune deficiency, immunodepression, immunity, immune defense |
| Stress and sleep | Stress, nervosity, anxiety, sleep, sleep disorder, drowsiness, insomnia, mood |
| Vitality and cognition | Attention deficit, hyperactivity, cognition disorder, cognition, memory, memory disorder, memory deficit, semantic memory, short-term memory, long-term memory, reactive agility, cognitive performance, physical activity, performance, training, exercise, muscle strength, strength recovery, muscle recovery, recovery, muscular recovery, muscular recuperation |
| Phenolic Acids | Flavonoids | Anthocyanins | Tannins | Organosulfur Compound | Carotenoids | Caffeine | |
|---|---|---|---|---|---|---|---|
| Bones and joints | 2 | 3 | 0 | 0 | 2 | 7 | 3 |
| Digestive health | 0 | 5 | 1 | 2 | 3 | 0 | 1 |
| Immunity and ENT | 0 | 3 | 0 | 2 | 1 | 2 | 1 |
| Stress and sleep | 0 | 2 | 1 | 1 | 0 | 4 | 3 |
| Vitality and cognition | 4 | 6 | 6 | 2 | 0 | 3 | 4 |
| First Author (Year) | Target | Phytochemicals | Participants (Total Number, Female Number, Age Years) Country | Study Type (Duration) | Exposure, T (Number Treated) C (Number Control) Phytochemicals | Outcome Parameter | Results | Comments |
|---|---|---|---|---|---|---|---|---|
| Stringham (2018) [43] | Stress | Carotenoid (lutein, zeaxanthin) | 59 young adults with oxidative stress and inflammation-implicated in stress (32 female) Mean age: 21.5 years USA | Double-blind, placebo-controlled trial 12 months treatment, control 6 and 12 months | T: Macular carotenoids (lutein, zeaxanthin and meso-zeaxanthin) 13 mg/day (n = 24), 27 mg/day (n = 25) C: Placebo (n = 10) | Stress, serum cortisol, physical and emotional health (questionnaire) | After 6 months and 12 months, stress cortisol and symptoms of suboptimal emotional and physical health were reduced (p < 0.05) | Carotenoid reduced oxidative stress and inflammation implicated in stress |
| Scholey (2012) [44] | Mood Neurocognitive effect | Flavonoid Epigallocatechine gallate (EGCG) | Volunteers without pathology (n = 31; 19 female) Mean age: 27.7 years Australia | Double-blind, placebo-controlled, crossover trial (1 week treatment, 1 week washout 1 week treatment) | One testing session, with second testing session one week after T: Green tea 300 mg free of caffeine (n = 31) C: Placebo (n = 31) | Brain activity and self-reported mood, cardiovascular function, and electroencephalogram 120 min after intake | Increased self-rated calmness (p < 0.04) and reduced self-rated stress (p < 0.017) were reported | This pattern of results suggests that participants in the EGCC group may have been in a more relaxed and attentive state after consuming EGCC |
| Kell (2017) [45] | Mood | Carotenoid Crocins, safranal | 121 patients with low mood but no depression (75 females) Age: 18–77 years Australia | Double-blind, parallel, randomized, placebo- controlled trial (4 weeks) | T: Affron® (saffron extract) 28 mg/day (n = 41) or 22 mg/day (n = 42) C: Placebo: (n = 38) | Mood, stress, anxiety, sleep; sleep quality index (SQI) | Decreased negative mood and symptoms relative to stress at 28 mg and no effect with 22 mg (p < 0.001) SQI: No effect | The use of Affron® (saffron extract) increased mood and managed stress without side effects |
| White (1980) [46] | Anxiety and muscle tension | Caffeine | 36 student volunteers (Number of females and age not specified) USA | Double-blind, placebo-controlled trial | T: Caffeine citrate 300 mg 8 h intake and 11 h test (n = 19) C: Citric acid (Placebo) (n = 17) | Electromyography, reaction time and anxiety recorded 30 min after intake | Regular consumer of caffeine (376 mg) had higher muscle tension after 3 h then lower consumer (87 mg); brief abstinence of caffeine-induced anxiety in higher-consumption consumer | Brief abstinence from caffeine may produce anxiety |
| Bernstein (1994) [47] | Learning, performance, anxiety | Caffeine | 21 healthy prepubertal children (9 females) Age: 8–12 years USA | Double-blind, placebo-controlled, crossover trial (1 week of treatment) | T: Low-dose caffeine 2.5 mg/kg or high-dose caffeine 5 mg/kg C: Placebo (n = 21) | Learning, performance, anxiety, test of attention, manual dexterity, short-term memory, processing speed, anxiety rating, level of caffeine in saliva | Reduced sluggishness reported with caffeine 5 mg (p = 0.43); increased target stimulus with T group vs. placebo (p = 0.098) | Caffeine can enhance performance on test of attention and motor task, but can cause increased anxiety |
| Grosso (2016) [48] | Depression | Caffeine | 12 studies, 23 datasets, 346 913 individuals (8146 with depression; n umber of females and age not specified) Italy | Review and meta-analysis of observational study on depression | Dataset with coffee (n = 7) Dataset with tea (n = 6) Dataset with caffeine (n = 9) | Depression scale | J-shaped non-linear relation between coffee consumption and risk of depression; protective effect at 400 mL/day | Coffee has a protective effect against depression, which is only partially seen with tea and caffeine |
| Terauchi (2014) [49] | Menopausal symptoms, anxiety, sleep | Tannins Proanthocyanidin | Females with menopausal symptoms (n = 91) Age: 40–60 years Japan | Double-blind, randomized, placebo-controlled study (8 weeks) | T: Caffeine 100 mg (n = 32) and 200 mg (n = 30) C: Placebo (n = 29) | After 4 weeks and 8 weeks: menopausal health questionnaire, anxiety, depression, sleep, blood pressure (BP), muscle mass | Significant result after 8 weeks of treatment: Decreased physical symptom score in the T group (high dose) (p < 0.05); hot flash score improved in the T group (high dose) (p < 0.05); lower score in Athens insomnia scale with T group (high dose) (p < 0.01); decreased anxiety score (p < 0.01), BP decreased in the T group (high and low dose) (p < 0.01); increased muscle mass in the T group (high and low dose) (p < 0.05) | Caffeine improved the physical and psychological impact of menopause |
| Umigai (2018) [50] | Sleep | Carotenoid | 30 healthy men and postmenoposal women with mild sleep complaints (10 females) Age: 35–60 years Japan | Double-blind, randomized, placebo-controlled, crossover study (5 days baseline, 14 day intervention, 14 day washout period, 14 day intervention) | T: Crocetin 7.5 mg/day (n = 15) C: Placebo (n = 15) | Electroencephalography, subjective sleep parameters, Ogury Shirakawa Azumi sleep inventory—MA score | Sleep maintenance (p = 0.001); feeling refreshed (p = 0.007); subjective sleep quality; no effect in sleep latency, sleep efficiency, total sleep time, or sleep after sleep onset | Crocetin contributes to sleep maintainance and sleep quality |
| Hachul (2011) [51] | Insomnia | Flavonoids | Postmenoposal women with insomnia (n = 38) Age: 50–65 years Brazil | Double-blind, placebo-controlled study (4 months) | T: Isoflavones 80 mg/day (n = 19) C: Placebo (n = 19) | Subjective and objective sleep parameters; polysomnography | Decreased intensity and number of hot flashes and frequency of insomnia fversus placebo; increased sleep efficiency (p < 0.01) | Flavonoids can reduce the symptoms of insomnia |
| Losso (2018) [52] | Insomnia | Anthocyanin | Male (5) or female (3) individuals (n = 8; 3 excluded due to apnea) (5 females) Age: >50 years USA | Placebo-controlled, crossover study Two weeks of twice daily dosing Two weeks washout | T: Cherry juice 240 mL titrated with cynanidins twice daily (n = 4) C: Placebo (n = 4) | Five validated questionnaires: Pittsburgh sleep quality index, (insomnia severity index, Epworth sleepiness scale, Beck depression inventory, state-trait anxiety inventory; blood test: kynurenine:tryptophan ratio, serum indoleamine 2, 3-dioxygenase, and prostaglandine E2 | Increased sleep time for 84 min (p < 0.01) and sleep efficiency (p < 0.03) vs. placebo; increased tryptophan availability | Anthocyanins can increase sleep time and sleep efficiency |
| Kuratsune (2010) [53] | Insomnia | Carotenoid | Healthy adult men with a mild sleep complaint (n = 21) Age: 25–59 years Japan | Double-blind, placebo- controlled, crossover study (2 weeks of treatment, 2 week washout period, 2 weeks of treatment | T: Crocetin 7.5 mg (n = 10) C: Placebo (n = 10) | Actigraph; Mary’s Hospital sleep questionnaire | Reduced wakening episode with crocetin vs. placebo (p < 0.025); trend for improved quality of sleep with crocetin | Crocetin can improve quality of sleep |
| First Author (Year) | Target | Phytochemicals | Participants (Total Number, Female Number, Age Years) Country | Study Type (Duration) | Exposure, T (Number Treated) C (Number Control) Phytochemicals | Outcome Parameter | Results | Comments |
|---|---|---|---|---|---|---|---|---|
| Masuda (2014) [60] | Symptoms of Japanese Cedar Pollinosis (JCP) | Flavonoid (methylated catechin) | 51 adults with JCP (36 female) Age: 20–65 years Japan | Randomized, double-blind, placebo-controlled trial (12 weeks) | T: 700 mL of ‘Benifuuki’ green tea containing O-methylated epigallocatechine gallate (EGCG (n = 26) C: 700 mL of ‘Yabukita’ green tea not containing O-methylated EGCG (n = 25) | Area under the curve (AUC) of symptom scores of nasal symptoms (sneezing, runny nose and nasal congestion), ocular symptoms (itchy eyes and tearing), and sore throat during the peak pollen season; quality of life (QoL)-related questionnaire; peripheral eosinophil (PE) | AUC: Significantly smaller with T group (runny nose [p < 0.05], itchy eyes [p < 0.01], tearing [p < 0.01]); QoL score: Significantly better in the T group (p < 0.01); PE: Suppressed in the T group | ‘Benifuuki’ green tea containing O-methylated EGCG reduced symptoms of JCP and has potential as an alternative medicine for treating seasonal allergic rhinitis |
| Enomoto (2006) [61] | Allergic rhinitis | Tannins (procyanidins) | 33 adults with moderate or severe persistent allergic rhinitis (24 female) Age: 15–65 years Japan | Randomized, double-blind, placebo-controlled trial (4 weeks) | T1: High polyphenols (200 mg per bottle) (n = 11) T2: Low polyphenols (50 mg per bottle) (n = 11) C: Placebo (n = 11) | Sneezing attacks (SA); nasal discharge (ND); swelling nasal turbinate (SNT); color, or inferior turbinate | SA and ND: Significant improvement (p < 0.05 and p < 0.01, respectively) for the T1 group; SA: Significant improvement for the T2 group (p < 0.05); SNT: Significant improvement for T1 and T2 groups (both p < 0.05) | Tannins (procyanidins) are effective in alleviating symptoms of persistent allergic rhinitis |
| Matsumoto (2011) [62] | Influenza infection | Flavonoid catechins and theanine | 197 healthcare workers (152 female) Age: 21–69 years Japan | Randomized, double-blind, placebo-controlled trial (20 weeks) | T: Green tea catechins (378 mg/day) and theanine (210 mg/day (n = 98) C: Placebo (n = 99) | Incidence of clinically defined influenza infection (ICDII); incidence of laboratory-confirmed influenza infection (ILCII); time the patient was free from influenza infection (TFI) | ICDII: Significantly lower with T (p = 0.022); ILCII: Lower with T group but not significant (p = 0.112); TFI: Significantly different between the two groups (p = 0.023) | Taking green tea catechins and theanine may be effective prophylaxis for influenza infection |
| Müller (2016) [63] | Systemic effect in the context of live attenuated influenza virus (LAIV)-inoculation | Organosulfur compounds (sulforaphane) | 29 adults (19 female) Mean age: 26.7 ± 1.1 years USA | Randomized, double-blind, placebo-controlled (4 weeks) | T: Broccoli sprout Homogenate (BSH) shake of 200 g (111 g of fresh broccoli sprouts + water) (n = 13) C: Alfalfa sprout homogenate (ASH) (n = 16) | Blood sample (BS); neutrophils (N); monocytes (Mo); macrophages (Ma); T cells; NKT cells and NK cells; cytoxic potential; granzyme B (GB) | NKT, T, NK cells were significantly reduced; NKT: Day 2 and Day 21, p = 0.002 and p = 0.036, respectively; T cell: Day 2, p = 0.022; NK cells: CD56 and CD158b expression (p = 0.0084 and p = 0.0007, respectively); NK cells: Significantly increasing CD16 expression (p = 0.0095) and CP (Day 2); BSH increased LAIV-induced GB production vs. ASH (Day 2, p = 0.049); BSH GB negatively associated with influenza RNA levels in nasal lavage fluid cells (p = 0.088) | Nasal influenza infection may induce complex changes in peripheral blood NK cell activation, and BSH increases virus-induced peripheral blood NK cell granzyme B production, an effect that may be important for enhanced antiviral defense responses |
| Nantz (2013) [64] | Immunity (γδ-T cell proliferation), cold and influenza | Tannins | 45 adults (31 female) Age: 21–50 years USA | Randomized, double-blind, placebo-controlled, parallel intervention (10 weeks) | T: Powdered cranberry fraction (65–77% of proanthocyanins) (n = 22) C: Placebo (n = 23) | BS (γδ-T cells); peripheral blood mononuclear cell (PBMC) were cultured for 6 days with autologous serum and phytohemagglutinin stimulation; cold and influenza symptoms (CIS) | PBMC: γδ-T cells in culture were almost five times higher with T (p < 0.001); CIS: Significantly fewer symptoms of illness were reported (p = 0.031) | Consumption of the cranberry beverage modified the ex vivo proliferation of cells. As these cells are located in the epithelium and serve as a first line of defense, improving their function may be related to reducing the number of symptoms associated with a cold and flu |
| Crişan (1995) [65] | Acute and chronic rhinopharyngitis in children | Flavonoids | 94 children Preschool (P, n = 47) Mean age: 6 years–3 months and 24 days Schoolchildren (S, n = 47) 9 years, 1 month and 4 d. (mean) Mean age: 9 years, 1 month and 4 days Romania | Randomized, double-blind, placebo-controlled (20 weeks) | T: NIVCRISOL (aqueous propolis extract rich in flavonoids) 1 mL/day (n = 61; 26 P, 35 S) C: Physiological salt solution (n = 33; 21P, 12 S): | Clinical observation (CO) (NC: Nasal catarrh, pharyngeal congestion, conjunctival mucosa congestion [CMC]); fever or any other state alteration (F); nasopharyngeal exudate monthly (5-months): virus isolation (VI), bacteriological and fungal examinations | CO: Significant lowering of the number of cases; decrease in the number of respiratory infections with general state alteration (p < 0.01); VI: Decrease and sometimes suppression of symptom (F and CMC) and altered general state (p < 0.05) for P and S; persistent cough (p < 0.05) only for S; reduction in the number of days of disease in cases with respiratory illness (2.5 times lower in treated P and 1.2 lower in treated S) | NIVCRISOL predominantly acts on viruses frequently encountered in nasopharyngeal exudate of subjects with acute or chronic rhinopharyngitis or apparent good health |
| Welsh (2010) [66] | Lung function | Caffeine | 75 adults with mild to moserate asthma (16 female) Age: 16–73 years UK | Meta-analysis of randomized, controlled, crossover trials (seven studies: one study of exhaled nitric oxide concentration (FeNO) and six studies of pulmonary function test) | T: Oral caffeine or coffee (5–10 mg/kg of caffeine or 15 mg/kg of coffee) (n = 75) C: Placebo or decaffeinated coffee (n = 75) | Lung function (LF); forced expiratory volume in 1 s (FEV1); maximum mid-expiratory flow and specific airway conductance (FEF25–75); FeNO; maximal expiratory flow rates at 25 and 50% of viral capacity (Vmax50 and 25); symptoms, side effects and adverse effects SSEAE (heart rate [HR], blood pressure [BP]) | Six trials (n = 55): caffeine improved LF for up to 2 h after consumption; FeNO: No significant difference (p = 0.38); FEV1: Small improvement (up to 2 h, 5%), two studies have a 12% and 18% mean difference (p = 0.008); FEF25–75: Small improvement with caffeine (up to 4 h); SSEAE: One study had a significant result on HR (decrease up to 9%) and BP (increase up to 12%) | Caffeine appears to improve airways function modestly, for up to four hours, in people with asthma. When testing lung function, people may avoid caffeine consumption |
| Nourollahian (2020) [67] | Allergic rhinitis (AR) | Carotenoids | 53 patients with AR (30 female) Mean age: 26.8 ± 9.3 years Iran | Randomized, double-blind, placebo-controlled trial (2 months) | T: Spirulina (2 g/day) (n = 26) C: Cetirizine (10 mg/day) (n = 27) | Cardinal rhinitis symptoms; sneezing; nasal congestion; rhinorrhoea (R); smell disturbance (SD); nasal itching (NI); sleep condition (SC); social activity (SA); daily work (DW); interleukin (IL)-10, -4, -1; interferon-gamma (IFN-γ) | No difference between the groups before the clinical presentations (p > 0.05); R, NI, and SD: Significant improvement for the T group (p = 0.021, p = 0.039 and p = 0.030, respectively); SC, DW, and SA: Significantly improvement for the T group (p < 0.05); 1 month later: IL-1α, IL-1β, and IL-4 were significantly lower in the T group (p < 0.001, p < 0.001 and p = 0.008, respectively); IL-10: Significantly higher in the T group (p = 0.049) | Spirulina is more effective than cetirizine in improving cardinal symptoms of AR patients. Furthermore, spirulina can be considered as an alternative treatment in patients with AR |
| Cingi (2008) [68] | AR | Carotenoids | 129 patients 75 (female) Age: 19–49 years Turkey | Randomized, double-blind, placebo-controlled trial (6 months) | T: Spirulina (2000 mg/day) (n = 85) C: Placebo (n = 44) | Symptoms and physical findings: ND; sneezing; nasal congestion (NC); NI | ND, sneezing, NC, and NI): Significantly improved (p < 0.001) with T vs. C | Spirulina is clinically effective on allergic rhinitis when compared with placebo. Further studies should be performed in order to clarify the mechanism of this effect |
| First Author (Year) | Target | Phytochemicals | Participants (Total Number, Female Number, Age Years) Country | Study Type (Duration) | Exposure, T (Number Treated) C (Number Control) Phytochemicals | Outcome Parameter | Results | Comments |
|---|---|---|---|---|---|---|---|---|
| Stomach | ||||||||
| Ingersoll (2010) [86] | Chemotherapy-induced nausea and vomiting | Flavonoids | 77 adults with cancer (62 female) Mean age: 54.3 years USA | Double-blind, randomized clinical trial (1 week following each of four chemotherapy treatment cycles) | T: Grape juice (n = 40) C: No juice (n = 37) | Nausea and vomiting frequency, duration, and distress; quality of life; control over decision making; psychological state | No significant differences except for final anxiety and depression | Nausea and vomiting frequency, duration, and distress were lower for the treated group without any statistically significant difference over time |
| Intestine | ||||||||
| Biedermann (2013) [95] | Ulcerative colitis (UC) | Anthocyanins | 13 patients with mild-moderate UC (3 female) Age: 19–61 years Switzerland | Prospective, non-blinded, non-controlled pilot trial (6 weeks) | T: Bilberry 160 g (4 trays per day equivalent to an average dose of anthocyanin of 840 mg/day) | Clinical activity index (CAI) with remission defined as CAI<4; endoscopic Mayo score; short inflammatory bowel disease questionnaire (SIBDQ) | Remission was achieved in 63.4% of patients; significant reduction in the complete Mayo Score of at least two points in all patients; SIBDQ score was significantly higher at end of treatment for 81.8% of patients | Anthocyanins had a significant beneficial effect on inflammatory activity in UC |
| Dryden (2013) [87] | UC | Flavonoid (-)-epigallo-catechin-3-gallate (EGCG) | 17 patients with mild-to-moderate UC (11 female) Mean age: 44.9 years USA | Randomized, double-blind, placebo-controlled trial (56 days) | T: Polyphenn E green tea low dose (n = 5) and high dose (n = 8) C: Placebo (n = 3) | UC disease activity index (UCDAI); inflammatory bowel disease questionnaire (IBDQ) | Significant improvement of UCDAI for 66.7% patients with T vs. 0% with C (p = 0.03); no effect for IBDQ | Administration of Polyphenon E resulted in a therapeutic benefit for patients with UC who were refractory to 5-aminosalicylic and/or azathioprine. Polyphenon E treatment resulted in only minor side effects |
| Mangel (2008) [91] | Diarrhea-irritable bowel syndrome (D-IBS) | Tannins (oligomeric procyanidins) | 250 patients with D-IBS (185 female) Mean age: 50.2 years US | Double-blind, randomized trial: four groups comprising one placebo and three with different doses) (12 weeks) | T: Crofelemer (from Croton lechleri) 125 mg (n = 62), 250 mg (n = 59,) 500 mg (n = 62) C: Placebo (n = 61) | Stool consistency (ST); stool frequency (SF); pain score (PS); pain and discomfort-free days (PFD and DFD) | ST: No difference; SF: Significant with 500 mg/day dose (−0.43 vs. −0.98 for C; p 0.03); PS: No difference; PFD and DFD: Significant improvement with 500 mg/day dose; benefits increased after 3 months | No improvement of ST, except females with D-IBS treated with crofelemer 500 mg/day. Crofelemer displayed improvement for PFD and DFD |
| Baek (2016) [85] | Transit and bowel function | Flavonoids Polyphenols | 80 adults (71 female) Age: 19–39 years Korea | Randomized, double-blind, placebo-controlled trial (8 weeks) | T: Ficus carica paste (n = 40) Fiber: 1.7% Total phenolics compounds: 332 μg/g dry Total flavonoids: 44 μg/g dry C: Control paste (n = 40) | Colon transit time (CTT) | −38% with T vs. −24% with C, p < 0.0001 | After 8 weeks of supplementation Ficus carica paste, there was a significant reduction in CTT and an improvement in bowel function. No adverse effects were reported |
| Venancio (2018) [90] | Constipation symptoms | Tannins Gallotannins | 36 adults (28 female) Age: 18–65 years USA | Randomized trial (4 weeks) | Two T groups: Mango group (MG) (n = 19) Fibre group (FG) (n = 17) Intake of mango fruit (300 g) | Constipation symptoms; inflammatory biomarkers; hormones (gastrin); adipokines (interleukins [IL]); stool short-chain fatty acids (SCFA) | MG reported increased evacuation categorization; IL-6—23% in MG vs. FG (p = 0.01) IL-10—15.4% in MG vs. FG (p = 0.03); gastrin significantly increased in both MG and FG (+13% and +7%, respectively); valeric acid increased in both MG and FG (p = 0.336); endotoxin decreased in MG vs. FG (p = 0.025) | Mango consumption significantly improved constipation status, increased gastrin levels and fecal concentrations of SCFA (valeric acid), lowered plasma endotoxin and IL-6 |
| Yanaka (2018) [93] | Bowel habits | Organosulfur compound: sulforaphane glucosinolates (SGS) | 48 adults with constipation (44 female) Mean age: 35 years Japan | Randomized clinical trial (4 weeks) | Two T groups: Broccoli sprouts (BS) (n = 24) Alfalfa sprouts (AS) (n = 24) 20 g/day | Total constipation score: frequency of bowel movements, painful evacuation, incomplete evacuation, abdominal pain, duration of defecation attempt, assistance for evacuation, unsuccessful attempts of evacuation per 24 h; bacteria in stool samples | Significant reduction in constipation score for BS; significant effects on Bifidobacterium for BS and Lactobacillus for AS | A daily intake of 20 g/day of raw BS (4.4 mg/g SGS) for 4 weeks improves defaecation in healthy subjects. This effect was not demonstrated by an intake of the same amount of AS (no SGS). Beneficial effect of sulforaphane against chronic oxidative stress |
| Kaczmarek (2019) [94] | Gastrointestinal microbiota | Organosulfur compound: glucosinolates | 18 healthy adults) (10 female) Age: 21–70 years USA | Controlled feeding, randomized, crossover study (18 day treatment periods separated by a 24 day washout) | T: Diet + 200 g of cooked broccoli and 20 g of raw radish per day C: Diet excluding Brassica vegetables | Fecal samples/ beta diversity Urine and plasma: metabolites | Increase in Bacteroidetes for T vs. C (p = 0.03); decrease in Firmicutes for T vs. C (p = 0.05) | Broccoli increased Bacteroidetes and decreased Firmicutes. Broccoli consumption increased the abundance of Bacteroides. B. vulgatus and B. thetciiotciomicron showed no change by treatment type |
| Liver | ||||||||
| Ruhl (2005) [96] | Chronic liver disease (CLD) | Caffeine | 9849 adults (5995 female) Age: 25–74 years USA | Prospective (NHANES I) (mean 19 years) | Tea and coffee (>1, 1–2, >2 cups/day) | CLD | Multivariate-adjusted hazard ratio 0.36 (95% confidence interval: 0.17, 0.78) for >2 cups/day vs. <1 1 cup/day) | Coffee and tea drinking decreases the risk of CLD although the effect is limited to persons at increased risk of liver injury |
| Barsalani (2013) [88] | Hepatic steatosis | Flavonoïds Isoflavones | 54 overweight to obese post-menopausal women (body mass index: 240 kg/m2) Age: 50–70 years Canada | Randomized, double-blind, trial (6 months) | T: Exercise and soy isoflavones (70 mg/day) (n = 26) C: Exercise and placebo (n = 28) | Fatty liver index (FLI); plasma lipid profile; liver function enzymes: alanine aminotransferase(ALT), aspartate aminotransferase (AST), glutamyltransferase, alkaline phosphatase | All outcome parameters were improved in both T and C groups; significant improvements with isoflavones for glutamyltransferase and FLI after 6 months of treatment | In addition to exercise, isoflavones provided additional effects on FLI |
| Kikuchi (2015) [92] | Hepatic abnormalities | Organosulfur compound Glucoraphanin; sulforaphane precursor) | 55 men with fatty liver Age: 30– 69 years Japan | Randomized, placebo-controlled, double-blind trial (4 weeks) | T: Broccoli sprout extract 135 mg (approximately 310 μmol of glucoraphani per gram (n = 27) C: Placebo (n = 28) | Liver function markers: AST and ALT; γ-glutamyl transpeptidase (GTP); 8-hydroxydeoxyguanosine (8-OHdG) | Significant decrease in median (interquartile range) ALT before: 54.0 (34.5–79.0) vs. after T: 48.5 (33.3–65.3) international units (IU)/L; p < 0.05; significant decrease in GTP before: 51.5 (40.8–91.3) vs. after: 50.0 (37.8–85.3) IU/L; p < 0.05; 8-OHdG reduced in T not in C | Dietary supplementation with broccoli sprout extract containing the sulforaphane precursor is likely to be highly effective in improving liver function through reduction in oxidative stress |
| Cheraghpour (2019) [89] | Hepatic steatosis | Flavonoid Hesperidin (flavanone glycoside) | 49 adults with non-alcoholic fatty liver disease (NAFLD) (grades 2 and 3) (22 female) Iran | Randomized, placebo-controlled, double-blind clinical trial (12 weeks) | T: Hesperidin 1 g (n = 25) C: Placebo (n = 24) | ALT, GTP); total cholesterol (TC); triglyceride (TG); hepatic steatosis (HS); high-sensitivity C-reactive protein (hsCRP); tumor necrosis factor-α (TNF-α); nuclear factor-κB (NF-κB) | Significant reduction in ALT (p = 0.005), GTP (p = 0.004), TC (p = 0.016), TG (p = 0.049), HS (p = 0.041), hsCRP (p = 0.029), TNF-α, (p = 0.78) NF-κB (no significant reduction) | Hesperidin supplementation accompanied with lifestyle modification was superior to lifestyle modification alone in the management of NAFLD at least partially through inhibiting NF-κB activation and improving lipid profile |
| First Author (Year) | Target | Phytochemicals | Participants (Total Number, Female Number, Age Years) Country | Study Type (Duration) | Exposure, T (Number Treated) C (Number Control) Phytochemicals | Outcome Parameter | Results | Comments |
|---|---|---|---|---|---|---|---|---|
| Ambrosini (2013) [97] | Fracture risk | Carotenoids | 2322 individuals (664 female) Age: 40–62 years Australia | Observational studies (17 years) | T1: Synthetic all-trans β-carotene 30 mg/day T2: Synthetic all-trans β-carotene 0.75 mg/day T3: Retinol equivalents 7.5 mg/day as retinyl palmitate (25,000 international units) | Exploratory analysis of fracture (any fracture or osteoporotic fracture) risk as a secondary endpoint (the primary endpoint explored efficacy of retinol and β-carotene supplements for reducing the risk of mesothelioma and lung cancer in persons previously exposed to asbestos) | No increases in fracture risk after long-term supplementation with high doses of retinol and/or β-carotene (any fracture; p = 0.17; osteoporotic fracture, p = 0.79). NB: Previous cohort studies have reported positive associations between dietary retinol intake and fracture risk (p = 0.002) | This study observed no increases in fracture risk after long-term supplementation with high doses of retinol and/or β-carotene |
| Wu (2014) [98] | Fracture risk | Carotenoids | 283,930 individuals (≈252,835 female) Age: 15–90 years China and others | Meta-analysis of prospective studies (n = 12) | Assess the effects of vitamin A (n = 8 studies) or retinol or β-carotene (n = 4 studies) on fracture risk (mainly of the hip) | Adjusted relative risk (RR); risk of hip fracture; risk of total fracture; relation between serum retinol level and hip fracture risk | A high intake of vitamin A and retinol increased the risk of hip fracture (RR [95% confidence interval (CI)]: 1.87 [1.31, 2.65] and 1.56 [1.09, 2.22], respectively). Low concentration of retinol increased RR (dose–response meta-analysis showed a U-shaped relationship between serum retinol level and hip fracture risk), but not a high intake of β-carotene (RR [95% CI] 0.82 [0.59, 1.14]) | The meta-analysis suggested that blood retinol level is a double-edged sword for risk of hip fracture. To avoid the risk of hip fracture caused by too low or too high a level of retinol concentration, intake of β-carotene (provitamin A), which should be converted to retinol in blood, may be better than intake of retinol from meat, which is directly absorbed into blood after intake |
| Lee (2014) [99] | Fracture risk | Caffeine | 253,514 individuals (number of females not specified) Age: 25–103 years All countries, particularly Western countries (USA, Canada, Europe) | Systematic review and meta-analysis (n = 15) | Dose–response analysis to assess the risk of fractures according to the level of coffee consumption in the female population based on 12 939 fracture cases (cohort studies, n = 9; case–control studies, n = 6) | Urinary calcium excretion and expression of the protein receptor for vitamin D. RR of fracture | Daily coffee consumption is associated with an increased risk of fractures in women (RR of 1.02 per 2 cups to 1.54 per 8 cups per day) and a paradoxical decrease in risk in men | The meta-analysis suggested that daily consumption of coffee was associated with an increased risk of fractures in women and a contrasting decreased risk in men. However, future well-designed studies should be performed to confirm these findings |
| Connelly (2014) [100] | Knee osteoarthritis (OA) | Phenolic acid (rosmarinic acid) | 46 women with OA Age: 48–72 years Canada | Randomized, parallel-arm, double-blind study (16 weeks) | Effects of consuming spearmint infusion rich in rosmarinic acid, twice daily, on knee OA T: High-rosA spearmint plant (280 mg/day. rosA) (n = 22) C: Placebo (26 mg/day. rosA) (n = 24) | Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC); short-form 36-item health survey (SF-36); 6-min walk test (6MWT); stair climb test (SCT) | Daily consumption of spearmint tea significantly improved stiffness and physical disability scores in adults with knee OA, but only the high-rosA tea significantly decreased pain; WOMAC: Significant decrease with T (p = 0.002) at 16 weeks and significant decrease for C at 8 weeks (p = 0.04), but not at 16 weeks (p = 0.07). SF-36: Significant only for QoL score (p < 0.05); SCT and 6MWT: No significant difference (p = 0.43 for T, p = 0.44 for C, and p = 0.9 between the group) | Consumption of high-rosA tea warrants further consideration as a potential complementary therapy to reduce pain in OA |
| Law (2016) [101] | Bone mineral density (BMD), osteoporosis | Flavonoids | 30 healthy subjects (18 female) Age: 40–80 years China, Taiwan | Randomized, double-blind, placebo-controlled trial (8 weeks) | T: 100 mL of onion juice (n = 16) C: Placebo (n = 14) | BMD; alkaline phosphatase (ALP); free radicals; total antioxidant capacity | Onion juice consumption showed a positive modulatory effect on the bone loss and BMD (inhibitory effects on the differentiation of osteoclasts) and can be recommended for treating osteoporosis | Onion juice consumption showed a positive modulatory effect on the bone loss and BMD by improving antioxidant activities and thus can be recommended for treating various bone-related disorders, particularly osteoporosis |
| Hu (2017) [102] | Rheumatoid arthritis (RA) | Carotenoids | 227 incident RA patients and 671 matched controls (898 female) Age: 41–61 years USA | Prospective case–control study (10 years: Nurses’ Health Study and Nurses’ Health Study II) | To examine the associations between circulating carotenoids and future risk of RA | Measurement of plasma carotenoids (α-carotene, β-carotene, β- cryptoxanthin, lycopene and lutein/zeaxanthin) levels | No significant association was found between the level of circulating carotenoids and the risk of developing RA (p = 0.93) | Circulating carotenoids levels are not associated with reduced risk of RA. Further investigations using large prospective cohorts are warranted |
| Javadi (2017) [103] | RA | Flavonoids (Quercetin) | 50 women with RA Age: 35–56 years Iran | Double-blind, placebo-controlled clinical trial (8 weeks) | T: Quercetin (500 mg/day) (n = 25), C: Placebo (n = 25) | Plasma levels of TNF-α; sedimentation rate; clinical symptomatology including early morning stiffness, morning and after-activity pain, tender and swollen joint counts; disease activity score 28 (DAS-28); physician global assessment (PGA); health assessment questionnaire (HAQ) at the beginning and end of the study | Clinical symptomatology: Significantly reduced early morning stiffness, morning pain, and after-activity pain (p < 0.05); TNF-α: Significantly reduced in the T group compared with C (p < 0.05); DAS-28 and HAQ: DAS-28 significantly decreased in the T group (p = 0.04), DAS-28 and HAQ scores decreased in the T group compared with C (p = 0.001 for both); PGA: No significant change | Quercetin 500 mg/day supplementation for 8 weeks resulted in significant improvements in clinical symptoms, diseases activity, hs-TNF-α, and HAQ in women with RA |
| Hosseinzadeh-Attar (2020) [104] | Knee OA | Organosulfur compound (garlic) | 50 obese women (body mass index>30) with knee OA Age: 50–75 years Iran | Randomized, double-blind, placebo-controlled trial (12 weeks) | T: Daily odour-controlled garlic tablet 1000 mg (equivalent to 2500 mg of fresh garlic) containing 2.5 mg allicin (n = 23) C: Placebo (n = 25) | WOMAC questionnaire (including joint stiffness and physical function); visual analogue scale (VAS) for pain severity | WOMAC: Significant decrease in WOMAC total score (p = 0.013), joint stiffness (p = 0.019), and physical function (p = 0.018) in the T group compared with C; VAS: Marginal decrease (p = 0.073) | A 12 week garlic supplementation (1000 mg) exerted significant improvements in joint symptoms in obese women with knee OA. Future studies are required to address the potential better response of obese patients to interventions as well as relevant underlying mechanisms |
| Kim (2016) [105] | BMD, osteoporosis | Carotenoids (β-carotene) | 189 postmenopausal women Age: 50–75 years Korea | Cross-sectional study (6 months) | Relationship between nutritional intake (protein, carbohydrate, fat, micro, oligo elements and vitamins) and BMD | BMD T scores were measured at: lumbar spine, femoral neck, total hip; semiquantitative food-frequency questionnaire | Lumbar spine: Positively correlated with sodium, potassium, zinc, calcium, vitamin A, β-carotene and vitamin C (p < 0.05 for all); Femoral neck: Positive correlations with nutritional intake (protein, carbohydrate, fat, micro and oligo elements, and vitamins (p < 0.05 for all)); Total hip: Positive correlations with nutritional intake (protein, fat, vitamin, calcium, potassium, zinc, iron [p < 0.05 for all); β-carotene, Na and vitamin C had positive association with BDM-T scores (p < 0.001) | In postmenopausal Korean women, β-carotene, vitamin C, zinc, and sodium intakes were positively associated with bone mass Furthermore, frequency of vegetable consumption was positively associated with femoral neck and total hip T scores |
| Lambert (2017) [106] | BMD, osteoporosis | Flavonoids (isoflavones aglycones) | 2652 women analysis with postmenopausal or perimenopausal Age: 39–93 years Denmark | Systematic review of 26 randomized controlled trials | T: Isoflavone aglycones intakes C: Placebo | Weight mean difference (WMD) with the lumbar spine and femoral neck | WMD for lumbar spine: Isoflavone associated with a significant increase in BMD vs. placebo (p < 0.00001); WMD for femoral neck: Isoflavone associated with a significant increase in BMD vs. placebo (p < 0.01) | The effect appeared to be dependent on whether isoflavone treatments were in aglycone form. The beneficial effects against bone loss may be enhanced for isoflavone aglycones |
| Li (2013) [107] | Hip fracture risk | Caffeine | 4677 cases/159,307 controls Age: 50–70 years China, Europe, North America | Meta-analysis of prospective cohort studies and case–control studies (N = 6 cohort and N = 6 prospective studies) | Median coffee consumption | Establish the current evidence concerning the relationship between coffee consumption and hip fracture risk, according to study design and characteristics of study populations, and determine the potential dose–response pattern between coffee consumption and hip fracture risk | Pooled odds ratio: Increased hip fracture risk by 29.7% (p = 0.09) with high-dose caffeine result having no statistical significance. Subgroup analyses: Coffee consumption significantly increased hip fracture risk by 54.7% among women, by 40.1% for elderly participants aged >70 years, and by 68.3% for Northern Americans. Other subgroup analyses: Positive association between coffee and hip fracture risk. Follow-up duration also positively affected hip fracture risk (<13 years) | The meta-analysis provided insufficient evidence that coffee consumption significantly increases hip fracture risk. Coffee intake may increase hip fracture risk among women, elderly participants, and Northern Americans. No dose–response pattern was observed |
| Gambacciani (1997) [108] | BMD, osteoporosis | Flavonoids: isoflavones (ipriflavone) | 80 postmenopausal women Age: 40–49 years Italy | Longitudinal, comparative (2 years) | T1: Ipriflavone (IP) (600 mg/day) (n = 20) T2: Conjugated equine oestrogens (CE) (0.3 mg/day) (n = 20) T3: Low-dose IP (400 mg/day) and CE 0.3 mg/day (IP+CE) (n = 20) C: Placebo calcium supplementation (500 mg/day) (n = 20) | Bone mass measurement; bone metabolism marker measurements (urinary excretion [UE] of hydroxyproline, plasma osteocalcin level [POL], and measure of vertebral bone density [VBD]) | UE and POL in CE group: No modification of hydroxyproline; VBD: Significantly decreased with CE group and placebo (p < 0.0001); POL in IP and IP+CE: No modification;UE and VDB in IP and IP+CE: Significant decrease for UE (p < 0.05) and increase for VDB (p < 0.05) | Postmenopausal IP administration, at the standard dose of 600 mg/day, can prevent the increase in bone turnover and the decrease in BMD that follow ovarian failure. The same effect can be obtained with the combined administration of low-dose (400 mg/day) IP with low-dose (0.3 mg/day) CE |
| Ambrosini (2014) [109] | Fracture risk | Carotenoids (β-carotene) | 998 adults analyzed in cancer prevention program (335 female) Age: 15–80 years Australia | The Vitamin A Program, a cancer prevention program with supplementation of high-dose retinol and β-carotene (17 years) | From 1990 to 1996 randomly assigned to: T1: Retinol (7.5 mg /day) (n = 1006) T2: β-carotene (30 mg/day) (n = 1009) From 1996 to 2006, all assigned to: T1: Retinol (7.5 mg /day) (n = 1736) | Investigate plasma retinol and total carotene concentrations in relation to fracture risk | No convincing associations between plasma retinol concentration and fracture risk (hazard ratio [HR] 0.86 mmol/L; 95% CI: 0.65, 1.14) or osteoporotic fracture were observed (HR 0.97 mmol/L; 95% CI: 0.66, 1.43) | The possibility that higher plasma carotene concentrations may be associated with lower fracture risk is consistent with previous studies and warrants further study |
| Wetmore (2008) [110] | Bone mineral content (BMC) and BMD, osteoporosis | Caffeine | 625 women Age: 14–40 years USA | Prospective study | Associations between habitual caffeine intake and bone mass | BMC; BMD of total hip and lumbar spine | BMC and BMD: Intake >200 mg of caffeine per day had lower toal hip and lumbar spine (p < 0.01); caffeine intake not associated with either BMC or BMD (p > 0.5 for all models) | The data suggest that heavy habitual consumption of caffeinated beverages does not adversely impact bone mass among young women in general. Greater caffeine intake may be associated with lower BMC among depot medroxyprogesterone acetate users |
| Pattison (2005) [111] | RA | Carotenoids | >25,000 subjects who completed a baseline 7 d diet diary (European Prospective Investigation of Cancer Incidence (13,975 female) Age: 45–74 years UK | Prospective study | Carotenes: β-carotene, β-cryptoxanthin, zeaxanthin | Longitudinal follow-up of inflammatory polyarthritis (IP), ascertained via the Norfolk Arthritis Register | 88 cases of IP occurred: the mean of β-cryptoxanthin and zeaxanthin were 40% and 20% lower, respectively | These data are consistent with previous evidence showing that a modest increase in β-cryptoxanthin intake, equivalent to one glass of freshly squeezed orange juice per day, is associated with a reduced risk of developing inflammatory disorders, such as RA |
| Rejnmark (2004) [112] | BMD, osteoporosis | Carotenoids (vitamin A) | 2016 perimenopausal women Age: 48–52 years Denmark | Setting of the Danish Osteoporosis Prevention Study (5 year follow-up, case–control study) | Relationship between vitamin A and/or retinol at 0.53 mg/day. intake and BMD and fracture risk | BMD measurements: lumbar spine; femoral neck; cross-sectional analyses; longitudinal analyses | Cross-sectional and longitudinal analyses with femoral neck and lumbar spine: No associations between intakes (p = 0.93 with vitamin A and p = 0.92 with retinol) and BMD | During the 5 year study period, 163 subjects sustained a fracture (cases). Compared with 978 controls, logistic regression analyses revealed no difference in vitamin A intake. Thus, in a Danish population, average vitamin A intake was lower than in Sweden and the USA and not associated with detrimental effects on bone |
| Wattanathorn (2018) [113] | Risk factors of osteoporosis | Phenolic acids, gallic acid | 45 healthy perimenopausal and postmenopausal women Age: 45–60 years Thailand | Double-blind, placebo-controlled, randomized trial (8 weeks) | T1: Combined extract of M. Alba and P. odoratum (50 mg/day) (n = 15) T2: Combined extract of M. Alba and P. odoratum (1500 mg/day) (n = 15) C: Placebo (n = 15) | Osteocalcin (OC); ALP; carboxy-terminal collagen cross-links (β-CTX); total phenolic compounds (TPC); clinical chemistry changes (CCC) | ALP, OC, TPC, and CCC: Significantly increased in T2 group (p < 0.05, p < 0.01, p < 0.001, and p < 0.05, respectively); CTX: Significant decrease (p < 0.01) | Clinical safety assessment failed to show toxicity and adverse effects. Therefore, herbal congee containing the combined extract of M. alba and P. odoratum leaves is a potential functional food that can decrease the risk of osteoporosis |
| First Author (Year) | Target | Phytochemicals | Participants (Total Number, Female Number, Age Years) Country | Study Type (Duration) | Exposure, T (Number Treated) C (Number Control) Phytochemicals | Outcome Parameter | Results | Comments |
|---|---|---|---|---|---|---|---|---|
| Borota (2014) [115] | Recognition performance and memory consolidation | Caffeine | 160 individuals (80 female) Age: 18–30 years USA | Randomized, double-blind, placebo-controlled trial (48 h duration) | T (n = 122): 100 mg, n = 77) 200 mg, n = 35 300 mg, n = 10 C (Placebo), n = 38 | Salivary samples: caffeine metabolites; hippocampal memory (HM)–dependent task, particularly taxing pattern separation | Caffeine metabolites: Significant increase at the 1 h and 3 h time points, which then returned to baseline amounts over a 24 h washout period HM: More likely to call lure items ‘similar’ rather than ‘old’ vs. placebo in rates of target hits or foil rejection (p = 0.04) | Caffeine enhanced performance 24 h after administration according to an inverted U-shaped dose–response curve. Caffeine enhanced consolidation of the initial study session such that discrimination during retrieval was improved |
| Carvalho-Peixoto (2015) [116] | Physical performance enhancement | Anthocyanins | 14 athletes (all male) Mean age: 26 ± 6 years Brazil | Simple-blinded, randomized intervention study (four visits on four separate days) | T: Anthocyanins beverage of 300 mL containing 4% acaï (anthocyanins 27.6 mg) C: Placebo | Control of muscle (CM); cardiorespiratory responses (CR); time to exhaustion (TE); oxidative stress biomarkers (OSB) | CM: Reduction in perceived exertion (p < 0.05) CR: enhancement (p < 0.05) TE: Increased mean difference: 69 s (95% confidence interval [CI]: –296, 159; t = 2.2; p = 0.045) OSB: Attenuation of the metabolic stress induced by exercise (p < 0.05) | Anthocyanins beverages may be a useful and practical ergogenic aid to enhance performance during high-intensity training |
| Alharbi (2016) [117] | Cognitive function alertness and mood | Flavonoids | 24 men Age: 30–65 years UK | Randomized, double-blind, placebo-controlled, crossover (2 day exposure, 2 week washout) | T: Enriched orange juice (240 mL) containing flavonoids 272 mg C: Placebo | Immediate word recall (IWR); simple and complex finger tapping (SCFT); digit symbol substitution test (DSST); continuous performance test (CPT); serial sevens (SS); positives and negative affect scale (PANAS); contrast sensitivity (CS); delayed word recall (DWR) | No significant differences between drinks for any dependent variables. From a 2x2 ANOVA: no significant main effects or interactions for IWR, DSST, SS, PANAS, CS, DWR; SCFT: Significant mean change from baseline across time points, higher following flavonoid-rich drink (mean 1.4, SE 0.8) vs. placebo (mean -0.6, SE 0.5); CPT: Significant difference at 6 h (p < 0.05) but no significant difference at 2 h | Executive function and psychomotor speed significantly improved after the flavonoid-rich drink compared with control |
| Bazzucchi (2019) [118] | Neuromuscular function impairment caused by acute eccentric exercise-induced muscle damage | Flavonoids | 12 young men Mean age: 26.1 ± 3.1 years Italy | Randomized, double-blind, crossover (3 weeks of exposure, 14 days of washout) | T: Quercetin 1000 mg/day C: Placebo | Maximal voluntary isometric contraction (MVIC); force-velocity (FV); electromyography (EMG); isometric strength (IS); resting arm angle (RAA); arm circumference (AC); plasma creatine kinase (PCK); lactate dehydrogenase (LDH) | MVIC: Significant increase in IS recorded compared with baseline (+4.7%, p < 0.05); EMG, RAA, AC: Torque and muscle fibre conduction velocity (MFCV) decay significantly lower with T compared with C (p < 0.001); IS, FV, and MFCV significantly lower with C than T (p < 0.001); PCK, LDH: No significant findings | Quercetin supplementation appears to attenuate the severity of muscle weakness caused by eccentric-induced myofibrillar disruption and sarcolemma action potential propagation impairment |
| Saitou (2018) [119] | Cognitive function | Phenolic acids (chlorogenic acid [CGA]) | 38 healthy volunteers with subjective memory complaints (17 female) Age: 50–69 years Japan | Randomized, double-blind, placebo-controlled-parallel group (16 weeks) | T: CGA drink with dry green coffee extract without caffeine and rich in chlorogenic acids (caffeoylquinic acids [CQA] 67.5%, feruloylquinic acid [FQA] 13.8%, and dicaffeoylquinic acids 18.6%) CQA + FQA = 300 mg (n = 20) C: Placebo without CQA (n = 18) | Cognition vital signs (Cognitrax); verbal memory test (VBM); visual memory test (VIM); finger tapping test (FTT); symbol digit coding (SDC); stroop test (ST); shifting attention test (SAT); CPT; blood sample: levels of apolipoprotein (A1); transthyretin (TTR) | VBM: CGA scores lower vs. placebo group (p = 0.093); Cognition: CGA group significant increase (p < 0.05); SDC: scores for errors tended to be higher (p = 0.080) at 16 weeks; SDC, FTT/ ST, SAT, and CPT: significantly higher changes with CGA vs. placebo (p = 0.080, p = 0.071, p = 0.063, and p < 0.05, respectively); A1: higher score with CGA vs. placebo (p = 0.07); TTR: significantly higher in CGA group vs. placebo group (p < 0.05) | CGAs may improve some cognitive functions, including attention as well as motor speed, which would help in the efficient performance of complex tasks. Blood concentration of TTR and ApoA1 increased after the CGA treatment, which might reflect the improved cognitive functions observed in the neuropsychological tests |
| Bowtell (2017) [120] | Brain task-related activation, cognitive function, and resting perfusion | Anthocyanins | 26 healthy older adults (13 female) Mean age: 68.3 ± 1.7 years UK | Double-blind, randomized, controlled trial (12 weeks) | T: Blueberry concentrate (anthocyanidins 387 mg) (n = 12); C: Placebo (n = 14) | Magnetic resonance imaging (MRI); serum measurements; cognitive tests | MRI: Significant increase in brain activity for T vs. C group (p = 0.001); Serum measurements: Significant improvement in grey matter perfusion for T vs. C group in the parietal (p = 0.013) and occipital (p = 0.031) lobes; Cognitive tests: Improvement in working memory for T vs. C group (2-back test) (p = 0.05) | Supplementation with an anthocyanin-rich blueberry concentrate improved brain perfusion and activation in brain areas associated with cognitive function in healthy older adults |
| Kesse-Guyot (2014) [121] | Brain aging, particularly cognitive disorder | Carotenoids | 2983 middle-aged adults (1381 female) Age: 45–60 years France | Randomized, double-blind, placebo-controlled, primary prevention trial (5–7 years, continuation of an 8 year study) | T: Carotenoid-rich dietary pattern (CDP) C: No supplementation | Cognitive test performance (6 neuropsychological tests); recall tasks (RT); backward digit span task (BDST); trail-making test (TMT); semantic fluency task (SFT) | CDP associated with a higher composite cognitive test performance (p = 0.02); RT, BSDT, TMT, and SFT: Significant results (all p for trend <0.05) | Upon confirmation in other settings, these findings may argue that sufficient quantity and variety of colored fruits and vegetables in one’s diet may help to maintain brain health during ageing |
| Cook (2017) [122] | Physiological responses | Anthocyanins | 13 healthy men Mean age: 25 ± 4 years UK | Randomized, double-blind, crossover trial (7 day intake separated by a 14 day washout) | T: New Zealand blackcurrant extract 600 mg/day (CurraNZ) C: Placebo (anthocyanins 210 mg/day) | Isometric maximal voluntary contractions (iMVC) (from 0 and 120 s and from 30% to 100% iMVC) measured by: EMG; near-infrared spectroscopy (NIRS); hemodynamics; ultrasound | iMCV at 100%: No effect (p = 0.732); Hemodynamics: Significant result with 30% (p < 0.001); iMCV: Total peripheral resistance, systolic, diastolic, and mean arterial pressure were lower with increased cardiac output and stroke volume; EMG: Lower muscle oxygen saturation (p < 0.001) and root mean square (at 45 s, p = 0.05, at 60 s, p = 0.034, and at 75 s, p = 0.015); NIRS: No significant results; Ultrasound: Increase in femoral artery diameter at 30 s—30% iMVC (6.9%, p = 0.009), 60 s—30% iMVC (8.2%, p = 0.8), 90 s—30% iMVC (7.7%, p = 0.021), and 120 s—30% iMVC (6.0%, p = 0.022) | Seven-day intake of 600 mg of New Zealand blackcurrant extract containing 210 mg anthocyanins, with the final intake 2 to 3 h before testing, increased vasodilation during sustained submaximal isometric exercise in young adult healthy men |
| Falcone (2018) [123] | Nootropic effects | Phenolic acids | 142 healthy, recreationally active adults (44 female) Mean age: 27.5 ± 7.9 years USA | Randomized, double-blind, placebo-controlled, parallel trial (90 days) | T: Proprietary spearmint extract (PSE) (n = 73) 900 mg; C: Placebo (n = 69) | Number of hits and average reaction time (stationary and multi-directional test); complete blood count (CBC) | Average reaction time: Significant with PSE at Day 7 (p = 0.049) and Day 30 (p = 0.049); Stationary: Between group differences at Day 30 (p = 0.040) and Day 90 (p = 0.002); Multi-directional: Between group differences at Day 30 (p = 0.007) and Day 90 (p = 0.026); CBC: No significant difference | The findings of the current study demonstrate that consumption of PSE 900 mg improved specific measures of reactive agility in a young, active population |
| Gratton (2020) [124] | Cerebral cortical oxygenation and cognition improvement | Flavonoids | 18 healthy men Mean age: 23.9 ± 7.3 years USA | Randomized, double-blind, placebo-controlled, crossover trial (two visits with a 2 week washout) | T: High flavanol (HF) intake cocoa drink comprising epicatechin 150 mg and catechin 35.5 mg; C: Low flavanol (LF) intake placebo drink delivering <4 mg of both monomers | Cortical hemoglobin concentration; flow-mediated dilatation (FMD); functional near-infrared spectroscopy (fNIRS); cerebral CO2 reactivity (CCR); double-strop task (DST) | fNIRS: Blood oxygenation most evident in lateral frontal region; HF intake lead to earlier and larger response: significant interaction between intervention and latency of response reach 90% maximal oxygenation (p = 0.002); FMD: significant increase of 1% after HF intake (p < 0.001); DST: Significant difference between LF and HF conditions (p = 0.029); CCR: No significant improvement | Using dietary strategies containing plant-derived flavanols is useful for enhancement of blood oxygenation and cognitive performance in healthy populations, as well as for populations at higher risk of cognitive impairment or to help recovery from and treatment of brain injuries and disease |
| Grgic (2018) [125] | Muscle strength and power | Caffeine | 294 individuals (51 female) Age: 16−34 years Global | Systematic review and meta-analysis of randomized, double-blind studies (N = 20) | T: Caffeine 4.3–6.5 mg/kg (caps, liquid, or gel). C: Placebo | Upper and/ or lower body exercise (muscle strength); monitor unit recruitment; vertical jump magnitude | Upper and lower body exercise: Caffeine improved both strength and power (standardized mean difference [SMD] = 0.20, 95% CI: 0.03, 0.36; p = 0.023; and SMD = 0.17; 95% CI: 0.00, 0.34; p = 0.047, respectively). A subgroup indicated caffeine significantly improved upper body exercise (SMD = 0.21; 95% CI: 0.02, 0.39; p = 0.026) | Meta-analyses showed significant ergogenic effects of caffeine ingestion on maximal muscle strength of upper body and muscle power. Future studies should more rigorously control the effectiveness of blinding (female group and different type of caffeine) |
| Haskell-Ramsey (2018) [126] | Cognition and mood | Caffeine | 59 individuals (29 female) split into two age groups: older (61–80 years; n = 30, 16 female) and younger (20–34 years; n = 29, 13 female) UK | Randomized, placebo-controlled, double-blind, counter-balanced crossover trial (three visits, 7 days of washout between each visit) | T1: Regular coffee (caffeine 100 mg) T2: Decaffeinated coffee (caffeine ≈5 mg) C: Placebo | Saliva sample; immediate word recallIWR; delayed word recall (DWR) and recognition; delayed picture recognition (episodic memory); numeric working memory (working memory); simple reaction time (SRT); digit vigilance (DV); rapid visual (RV; attention); subjective state (SStt) | Saliva sample: Confirmed adherence to caffeine abstention instructions, SRT decreased with regular coffee; RV: increased alertness compared with placebo (p = 0.014) and faster accuracy (p = 0.009); decaffeinated coffee increased alertness compared with placebo (p = 0.0048); DV: increased with regular coffee compared with decaffeinated coffee (p = 0.01); SStt: decreased tiredness (p = 0.003) and headache ratings were observed (p = 0.0049) | These findings suggest behavioural activity of coffee beyond its caffeine content, raising issues with the use of decaffeinated coffee as a placebo and highlighting the need for further research into its psychoactive effects |
| Imai (2018) [127] | Oxidative stress severe fatigue | Carotenoids | 24 healthy volunteers (11 female) Age: 30–60 years Japan | Randomized, double-blind, placebo-controlled, two-way crossover trial (3-months with a 4 week washout between each period of supplementation) | T: 2 x capsules containing 3 mg of astaxanthin and 5 mg of sesamin (AS) (n = 12) C: Placebo (n = 12) | Visual analog scale (VAS); Calder fatigue questionnaire (daily subjective fatigue) [CFQ]; subjective feelings (SF); work efficiency (WE); autonomic nerve activity (ANA); OSB); safety; plasma AS concentration | CFQ: Significant improvement in time with AS vs. placebo from mental fatigue (p < 0.01); OSB: Significant decrease with AS vs. placebo (p < 0.05); VAS: Significant difference between AS vs. placebo group; Correlation coefficient between plasma AS and difference in VAS score: recovery 2 h-task 4 h was −0.451 (p < 0.05) and recovery 4 h-task 4 h was −0.502 (p < 0.05); SF and WE: No significant difference between AS and placebo; safety: no adverse effects | In conclusion, AS supplementation may be a candidate to promote recovery from mental fatigue which is experienced by many healthy people. Thus, antioxidative activity exhibited by AS could be a possible mechanism for its anti-fatigue effect |
| Pilaczynska-Szczesniak (2005) [128] | Oxidative stress from an incremental rowing ergometer exercise | Anthocyanins | 19 male athletes (rowing team members) Age: 20–24 years Poland | Randomized, double-blind, placebo-controlled trial (4 weeks) | T: Chokeberry juice (150 mL daily; 23 mg/ 100 mL of anthocyanins) (n = 9) C: Placebo (n = 10) | Redox; creatine kinase (CK); lactate levels (LA); thuibarbituric acid reactive substances (TBARS); super oxide dismutase (SOD); gluthanione peroxydase activity (GPx) | TBARS: Significantly lower with T group at 1 min (p < 0.05) and 24 h sample after exercise (p < 0.05); GPx: Significantly lower with T group at 1 min after exercise (p < 0.05); SOD: Significantly lower compared with placebo in the 24 h blood sample (p < 0.05); CK, LA, and redox: No significant result | These findings indicate that an increased intake of anthocyanins limits the exercise-induced oxidative damage to red blood cells, most probably by enhancing the endogenous antioxidant defense system |
| Lamport (2016) [129] | Cognitive function, driving performance, and blood pressure | Flavonoids and anthocyanins | 25 healthy mothers Mean age: 43 ± 0.6 years UK | Double-blind, randomized, crossover, placebo-controlled trial (12 weeks with 4 weeks washout between each trial) | T: Concord grape juice 355 mL containing 777 mg total polyphenolics as a gallic acid equivalent (167 mg anthocyanins as malvidin equivalent and 334 mg proanthocyanidins as catechin equivalent) C: Placebo | Visual verbal learning test (immediate and delayed recall) (VVLT); visual spatial learning test (immediate and delayed recall) (VSLT); rapid visual information processing (RVIP); attention (psychomotor skill); blood pressure (systolic and diastolic) (BP); mood; driving performance (match speed and direction of a lead vehicle) | Immediate spatial memory (VVLT, VS.LT, RVIP; p < 0.05): Significant improvements main effect of condition recall was higher after Concord grape juice (mean: 12.72 items; standard error [SE]: 0.39) vs. placebo (mean: 12.57 items; SE: 0.36; p < 0.05); Driving performance: More accurate with Concord grape juice vs. placebo (mean correlation: 0.96; SE: 0.01; p = 0.05); Attention: Significantly higher with Concord grape juice (p < 0.05); mood and BP: No significant result | Cognitive benefits associated with the long-term consumption of flavonoid-rich grape juice are not exclusive to adults with mild cognitive impairment. Moreover, these cognitive benefits are apparent in complex everyday tasks such as driving. Effects may persist beyond the cessation of flavonoid consumption |
| Mastroiacovo (2015) [130] | Cognitive performance | Flavonoids (flavanol) | 90 elderly individuals (53 female) Age: 61–85 years Italy | Randomized, double-blind, controlled, parallel-arm study (8 weeks) | T1: High cocoa flavanols 993 mg (n = 30) T2: Intermediate cocoa flavanols 520 mg (n = 30) C: Low cocoa flavanols 48 mg (n = 30) | Mini-mental state examination (MMSE); trail making test (TMT) A and B; verbal fluency test (VFT); insulin resistance (IR); BP; lipid peroxidation (LP) | MMSE: No significant difference; TMT A and B: significant with high flavanols (−8.6 ± 0.4 and −16.5 ± 0.8 s, respectively) and intermediate flavanols (−6.7 ± 0.5 and −14.2 ± 0.5 s, respectively) differed from low flavanols (−0.8 ± 1.6 and −1.1 ± 0.7 s, respectively); VFT: Significant improvement in all group, but greater with high flavanols (7.7 ± 1.1 words/60 s) vs. intermediate flavanols (3.6 ± 1.2 words/60 s) and low flavanols (1.3 ± 0.5 words/60 s); IR (p < 0.0001), BP (p < 0.0001), and LP (p = 0.001) better with high flavanols and intermediate flavanols vs. low flavanols | Regular cocoa flavanols consumption can reduce some measures of age-related cognitive dysfunction, possibly through an improvement in insulin sensitivity. These data suggest that the habitual intake of flavanols can support healthy cognitive function with age |
| Duvnjak-Zaknich (2011) [131] | Agility performance and decision-making accuracy after simulated team-sport exercise | Caffeine | 10 moderately trained male team-sport athletes Mean age: 21.6 ± 2 years Australia | Randomized, double-blind, counterbalanced trial (22 trials realized with a 1 week washout) | T: Caffeine (6 mg/kg) C: Placebo | Total time (TT); reactive agility time (RAT); decision time (DT); movement time (MT); decision-making accuracy | TT, RAT, MT, and DT: No interaction effect between trials (similar between time and conditions). Caffeine ingestion significantly increased TT (2.3%, p = 0.001), RAT (3.9%, p = 0.001), MT (2.7%, p = 0.043), and DT (9.3%, p = 0.045) vs. placebo | Caffeine ingestion may be beneficial to reactive agility performance when athletes are either fresh or fatigued |
| Trombold (2010) [132] | Recovery of skeletal muscle strength after eccentric exercise | Tannins | 16 recreationally active males Mean age: 24.2 ± 1.4 years USA | Double-blind, randomized, placebo-controlled crossover trial (two testing periods, each of 9 days, with a 14 day washout) | T: Pomegranate extract (POMx) 500 mL containing 650 mg of pomegranate (ellagitannins) C: Placebo (liquid) | IS (load cell); soreness (visual analog rank = subjective); BS: CK; myoglobin (Mb); interleukin 6 (IL-6); C-reactive protein (CRP) | IS: Significantly higher with POMx vs. placebo at 48 h (p = 0.01) and 72 h (p = 0.009) after exercise. BS (CK, Mb, IL-6, CRP): No significant changes | Supplementation with ellagitannins from POMx significantly improves recovery of IS 2–3 days after a damaging eccentric exercise |
| Whyte (2015) [133] | Cognitive performance | Anthocyanins | 14 children (4 female) Mean age: 9.17 ± 0.6 years UK | Controlled and crossover trial (≈1 week with a washout of 7 days [minimum]) | T: Flavonoid-rich blueberry (anthocyanins 143 mg) C: Placebo | Go-NoGo; ST; Rey’s auditory verbal learning task (RAVLT); object location task (OLT); visual N-back (VNB) | Go-NoGo, ST, VNB, and OLT: No significant results (RT and accuracy responses). RAVLT: Significant results vs. placebo (p < 0.001) | Although findings were mixed, the improvements in delayed recall found in this pilot study suggested that, following acute flavonoid-rich blueberry interventions, school-aged children encoded memory items more effectively (vs. without) |
| Falcone (2019) [134] | Cognitive performance and nootropic effects | Phenolic acids | 142 healthy, recreationally men and women (44 female) Age: 18–50 years USA | Randomized, double-blind, placebo-controlled, parallel design (90 days) | T: PSE 900 mg/day (n = 73) C: Placebo (n = 69) | FTT; SDC; complex attention (CA); ST; CPT; SAT; reasoning; sustained attention; four-part continuous performance test (FPCPT); Digi Span test; Leeds sleep evaluation questionnaire (LSEQ); quality of life (QoL); profile of mood states (POMS) | Sustained attention: Significant improvement with PSE at Day 30 (p = 0.001) and at Day 90 (p = 0.007); CA: Significant improvement with PSE at Day 7 (p = 0.016); SAT and FPCPT: Significant improvement in two individual tests vs. placebo (p < 0.05); QoL, POMS, and LSEQ: No significant difference with PSE vs. placebo | The current study demonstrates that chronic supplementation with PSE 900 mg improves cognitive performance in a young, active population, further supporting PSE as an efficacious nootropic |
| Johnson (2008) [135] | Cognitive performance | Carotenoids | 49 healthy, non-smoking women Age: 60–80 years USA | Randomized, double-blind intervention trial (4 months) | T1: Docosahexaenoic acid (DHA) 800 mg/d (n = 14) T2: Lutein 12 mg/day (n = 11) T3: Combination of DHA and lutein (n = 14) C: Placebo (n = 10) | Verbal fluency (VF); digit span forward and backward (DSFB); shopping list task (SLT); word list memory test (WLMT); memory in reality (MIR) apartment test; pattern comparison test (NES2 PC); ST; mood scale (NES2 MS) | VF: Significant improve of score for DHA, lutein, and combined treatment compared with placebo (p < 0.03); SLT: Learn significantly faster with the combined treatment (p = 0.03); trend for WLMT but not significant (p = 0.07); MIR (delayed recall): Significant improvement (p = 0.02); NES2 PC: Significant increase for the placebo group (p = 0.04); NES2 MS, ST, DSFB: No significant results | These exploratory findings suggest that DHA and lutein (carotenoid compounds) supplementation may provide cognitive benefits for older adults |
| Ataka (2007) [136] | Anti-fatigue effects | Tannins | 18 healthy volunteers (9 female) Mean age: 39.1 ± 9.1 years Japan | Double-blind, randomized, placebo-controlled, three-way crossover trial (8 days) | T1: Applephenon® (1200 mg/day)—a rich procyanidins product T2: Ascorbic acid (1000 mg/day) C: Placebo | Physical performance test (PPT); subjective rate of fatigue level (visual analog from 0 to 100) (SRFL); BP; heart rate (HR); BS | PPT: No difference at baseline. Significant change in maximum velocity between 30- and 120-min trials: higher in the T1 group than C (rpm: +2, p < 0.05). T2 had no effect. SRFL, BP, HR, and BS: No significant differences | These results suggest that Applephenon® attenuates physical fatigue, whereas ascorbic acid does not |
| Do Rosario (2021) [137] | Cognitive function | Anthocyanins | 31 participants (9 female) with mild cognitive impairment (MCI) Mean age: 75.3 ± 6.9 years Australia | Randomized, double-blind, placebo-controlled trial (8 weeks) | 250 mL fruit juice T1: High-dose anthocyanins (201 mg/day). T2: Low-dose anthocyanins (47 mg/day). C: Placebo | Microvascular function (MF), 24 h ambulatory blood pressure (ABP); serum inflammatory biomarkers (tumor necrosis factor-α (TNF-α), interleukin (IL)-6, IL-1β, c-reactive protein) | TNF-α: Significant reduction with T1 group (p = 0.002) vs. C and T2 (p < 0.05 for both). IL-6, IL-1β, c-reactive protein, MF, and 24 h ABP: Not altered by any treatment | A daily high dose of fruit-based anthocyanins for 8 weeks reduced concentrations of TNF-α in older adults with MCI. Anthocyanins did not alter other inflammatory biomarkers, microvascular function, or blood pressure parameters. Further studies with a larger sample size and longer period of follow-up are required to elucidate whether this change in the immune response will alter cardiovascular disease risk and progression of cognitive decline |
| Calapai (2017) [138] | Cognitive function | Anthocyanins | 111 healthy older adults (58 female) Age: 60–72 years Italy | Randomized, double-blind, placebo-controlled trial (12 weeks) | T: Cognigrape® (250 mg/day: 30/40% of V. vinifera extract) (n = 57) C: Placebo (n = 54) | MMSE: temporal orientation, spatial orientation, immediate memory (IM), attention and calculation (AC), recall memory (RM), language, praxia visuo-constructive; Beck depression inventory (BDI); Hamilton anxiety rating scale (HARS); repeatable battery for the assessment of neuropsychological status (RBANS) | MMSE: Significantly improved with T (p < 0.0001) at baseline and vs. C group (AC (p < 0.001), language (p < 0.05), IM (p < 0.0001)). BDI, HARS: Significant reduction of 15.8% (BDI), and 24.9% (HARS) (p < 0.0001) and vs. C (p < 0.0001 for BDI and p < 0.05 for HARS). RBANS: Significantly improved with T at baseline and vs. placebo (p < 0.0001) | The results show that 12 weeks of Cognigrape® supplementation is safe, can improve physiological cognitive profiles, and can concurrently ameliorate negative neuropsychological status in healthy older adults |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Monjotin, N.; Amiot, M.J.; Fleurentin, J.; Morel, J.M.; Raynal, S. Clinical Evidence of the Benefits of Phytonutrients in Human Healthcare. Nutrients 2022, 14, 1712. https://doi.org/10.3390/nu14091712
Monjotin N, Amiot MJ, Fleurentin J, Morel JM, Raynal S. Clinical Evidence of the Benefits of Phytonutrients in Human Healthcare. Nutrients. 2022; 14(9):1712. https://doi.org/10.3390/nu14091712
Chicago/Turabian StyleMonjotin, Nicolas, Marie Josèphe Amiot, Jacques Fleurentin, Jean Michel Morel, and Sylvie Raynal. 2022. "Clinical Evidence of the Benefits of Phytonutrients in Human Healthcare" Nutrients 14, no. 9: 1712. https://doi.org/10.3390/nu14091712
APA StyleMonjotin, N., Amiot, M. J., Fleurentin, J., Morel, J. M., & Raynal, S. (2022). Clinical Evidence of the Benefits of Phytonutrients in Human Healthcare. Nutrients, 14(9), 1712. https://doi.org/10.3390/nu14091712






